
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pharmacol. , 22 March 2021
Sec. Drugs Outcomes Research and Policies
Volume 12 - 2021 | https://doi.org/10.3389/fphar.2021.586893
 Yan Wang1,2,3
Yan Wang1,2,3 Ying Zhang2
Ying Zhang2 Treasure M. McGuire3,4,5
Treasure M. McGuire3,4,5 Samantha A. Hollingworth3
Samantha A. Hollingworth3 Mieke L. Van Driel6
Mieke L. Van Driel6 Lu Cao7
Lu Cao7 Xue Wang8
Xue Wang8 Yalin Dong2*
Yalin Dong2*Background: The relationship between antibiotic use and the incidence of triazole-resistant phenotypes of invasive candidiasis (IC) in critically ill patients is unclear. Different methodologies on determining this relationship may yield different results.
Methods: A retrospective multicenter observational analysis was conducted to investigate exposure to antibiotics and the incidence of non-duplicate clinical isolates of Candida spp. resistant to fluconazole, voriconazole, or both during November 2013 to April 2018, using two different methodologies: group-level (time-series analysis) and individual-patient-level (regression analysis and propensity-score adjusting).
Results: Of 393 identified Candida spp. from 388 critically ill patients, there were three phenotypes of IC identified: fluconazole-resistance (FR, 63, 16.0%); voriconazole-resistance (VR, 46, 11.7%); and cross-resistance between fluconazole and voriconazole (CR, 32, 8.1%). Exposure to several antibacterial agents with activity against the anaerobic gastrointestinal flora, especially third-generation cefalosporins (mainly cefoperazone/sulbactam and ceftriaxone), but not triazoles, have an immediate effect (time lag = 0) on subsequent ICU-acquired triazole-resistant IC in the group-level (p < 0.05). When the same patient database was analyzed at the individual-patient-level, we found that exposure to many antifungal agents was significantly associated with triazole-resistance (fluconazole [adjusted odds ratio (aOR) = 2.73] or caspofungin [aOR = 11.32] on FR, voriconazole [aOR = 2.87] on CR). Compared to the mono-triazole-resistant phenotype, CR IC has worse clinical outcomes (14-days mortality) and a higher level of resistance.
Conclusion: Group-level and individual-patient-level analyses of antibiotic-use-versus-resistance relations yielded distinct but valuable results. Antibacterials with antianaerobic activity and antifungals might have “indirect” and “direct” effect on triazole-resistant IC, respectively.
Invasive candidiasis includes both bloodstream and deep-seated invasive infections caused by Candida species (Martin-Loeches et al., 2019). Candida spp. account for up to 17% of all intensive-care unit (ICU)-acquired infections globally (Vincent et al., 2009). Half of candidemia episodes occur in the ICU setting reflecting the complexity of illness usually associated with this infection (Pappas et al., 2018). The incidence and mortality rates for IC in ICU patients have progressively increased over time (Lortholary et al., 2014); a mortality rate approaching 40% has been reported for ICU-acquired IC in most series (Calandra et al., 2016).
Echinocandins and triazoles (mainly fluconazole and voriconazole) are backbones for the treatment of IC and are recommended by several guidelines (Wang et al., 2019). Although echinocandins are increasingly regarded as first-line treatment superior to fluconazole, the echinocandin-resistant rate remains relatively low (Perlin, 2015). Triazoles are still the most wildly used antifungal agents worldwide (Guo et al., 2013; Vallabhaneni et al., 2018). Given that antifungal resistance is an emerging threat to the initiation of appropriate antifungal treatment, early recognition of risk factors for patients who may experience triazole-resistant IC can improve patient survival (Perlin et al., 2017).
Previous studies demonstrated that exposure to antibiotics, (i.e. antifungal or antibacterial agents) is a prominent factor for increasing the risk of emergence and resistance of Candida species (Ben-Ami et al., 2012; Martin-Loeches et al., 2014; Jensen et al., 2015). However, most of these studies were based on data from individual-patients and it is known that antimicrobial treatment may exert effects on individual-patients that are substantially different in magnitude to the effects at the population-level, (i.e. group-level) (Lipsitch and Samore, 2002). For many types of pathogens, the relationship between antibiotic usage and resistance is mediated by selection at the population-level due to patient-to-patient transmission; it also applies to Candida spp. in ICU (Hammarskjold et al., 2013). Therefore, adopting the perspective of both the group and individual-patient level to identify the relationship between antibiotic usage and triazole-resistant IC will inform therapeutic decision making.
The objectives of this study were to: 1) model the time-varying effect of antibiotic use, (i.e. antifungals and antibacterials; aggregated data) on the incidence of three triazole-resistant phenotypes (fluconazole-resistance [FR], voriconazole-resistance [VR], and cross-resistance between voriconazole and fluconazole [CR]) in ICU patients using autoregressive integrated moving average (ARIMA) models and a transfer function, (i.e. group-level); 2) perform individual-patient-level analyses to identify potential antibiotic-risk factors associated with the three resistant phenotypes by using the same dataset, comparing the similarities and differences of the results from two different methodologies.
This retrospective study was performed at two urban, tertiary hospitals (2,700-bed and 2,200-bed, respectively), affiliated with the same university. These are the two largest tertiary hospitals in the area of central and south of Xi’an, and adopt similar medical models and practice guidelines. The ICUs of these two hospitals have 100 and 50 beds, respectively. The data collection procedures were approved by each hospital’s institutional review board. All data in this study were collected retrospectively, all patient data were anonymous and all laboratory information were part of routine testing, so informed consent was waived (No. 2015-XJTUFAH-002 and No. XJTUSAH-2020071).
Adult patients (age ≥18 years) were enrolled if diagnosed with proven IC in ICU and ICU stay ≥3 days. Proven IC was defined as a clinical illness consistent with an IC diagnosis combined with a Candida spp. isolate being detected by positive specimen culture taken from a sterile site (tissue or blood) or through histopathological evidence.
The four exclusion criteria were: 1) patients with neutropenia (absolute neutrophil count <500 cells/μl), hematologic malignancy, or after hematopoietic stem cell transplant; 2) microbiological isolates collected within 48 h of ICU admission; 3) being infected with rare Candida species whose clinical breakpoints or epidemiological cutoff-values for fluconazole and voriconazole have not been determined; and 4) incomplete data on susceptibility testing.
The details of antibiotic use, Candida spp. identification and susceptibility testing, and individual-patient data collection are provided in the (Supplementary Appendix SA1).
We determined if there were significant changes in monthly antibiotic use, the incidence of the most commonly isolated Candida spp., and the triazole-resistant Candida spp., (i.e. FR, VR and CR) using linear regression over the study period. Application of simple regression analyses would be inappropriate as the temporally sequenced observations on antibiotic use, the incidence of Candida spp., and the resistance isolates are not independent (Lopez-Lozano et al., 2000). Therefore, we assessed the relationship between monthly antibiotic use and monthly incidence of triazole-resistant Candida spp.; as well as monthly incidence of Candida spp. using a dynamic regression model. In the current study, this method comprises modeling the monthly incidence of Candida spp. or triazole-resistant Candida spp (explained variable) using ARIMA models with the Box–Jenkins method, integrating the stochastic dependence of consecutive data over time (Helfenstein, 1996). Then, the transfer function (TF) modeling method was employed to quantify the dynamic relationship by adding antibiotic use as an explanatory variable, and the possible time delays (lags) of up to six months was considered (Lopez-Lozano et al., 2000; Vernaz et al., 2011). This methodology has been proposed by Lopez-Lozano et al. as a suitable method to examine the relationship between antibiotic exposure and the emergence of drug-resistance (Lopez-Lozano et al., 2000) (See details of the statistical analysis of ARIMA and TF model in the Supplementary Appendix SA2).
A multivariate multinomial regression analysis was performed to identify risk factors for IC due to non-albicans Candida species, using C. albicans as reference.
A multivariate stepwise logistic regression model with backward selection was used to determine the relevant factors of FR, VR and CR. A p < 0.05 was considered statistically significant in the final multivariate model. To limit confounding by nonantibiotic-risk factors, we calculated the conditional probability of recent exposure to specific antibiotics based on nonantibiotic-risk factors using propensity-score adjusting analysis. We estimated the cumulative incidence of 14 days all-cause mortality by using the Kaplan-Meier product-limit method. The difference in cumulative incidence was compared by the log-rank test for trend and Cox regression analysis (see details of the statistical analysis of the regression model in the Supplementary Appendix SA3). All statistical analyses were performed using SAS 9.4 (SAS Inc.).
We identified 393 episodes of IC in 388 patients during the 54 months study period. Of these episodes, the median age was 63 years (IQR 46, 74 years) and, 221 (56%) patients were male. There were a median of 119 (IQR 109, 130) ICU discharges per month and the median duration of ICU stay was 11.38 days (IQR 9.74, 13.42).
Detailed antibiotic use and Candida spp. results are presented in the Supplementary Material (Supplementary Appendix SA4). Trends in the use of each class of antibiotics are summarized in the Supplementary Table S1 (Supplementary Appendix SA5). The distribution and the incidence of triazole-resistance of Candida spp. are shown in Table 1.
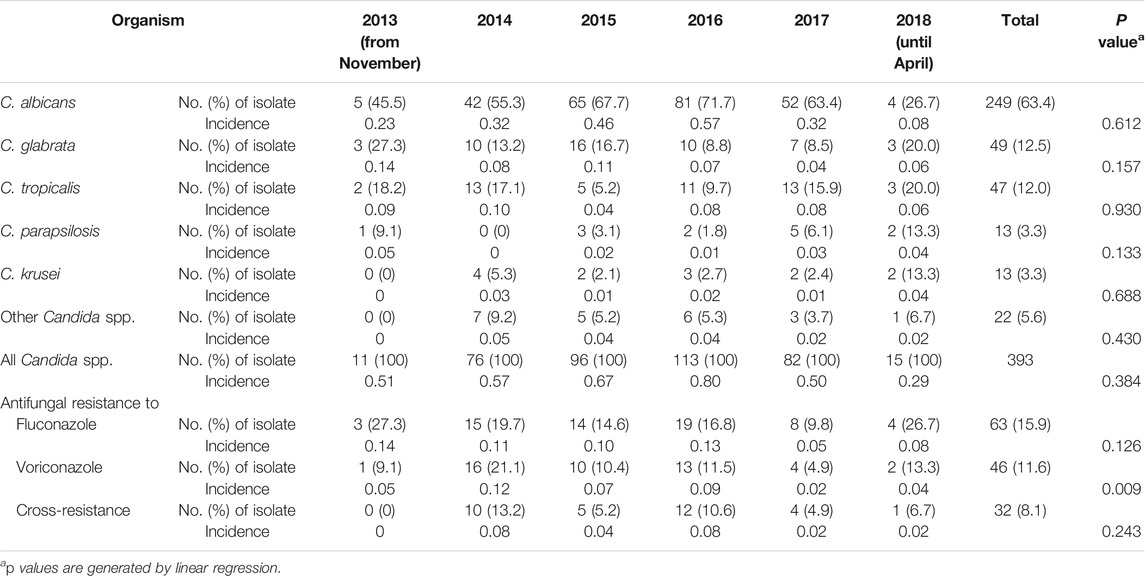
TABLE 1. Episodes of invasive candidiasis in number and incidence (per 100 patient-days) of non-duplicate Candida spp. distribution and resistance to fluconazole and voriconazole (November 2013 to April 2018).
The number of species associated with a high fluconazole MIC (≥64 μg/ml) related to CR Candida infections (16/32, 50.0%) was higher than that of mono-resistance Candida infections (5/31, 16.1%, p = 0.004), and a non-significant trend for high MIC (≥2 μg/ml) of voriconazole (16/32 [50.0%] vs. 3/14 [21.4%], respectively, p = 0.070).
The results of correlations between antibiotic use and Candida spp. distribution were presented in the Supplementary Material (Supplementary Appendixes SA6, SA7; Supplementary Table S2).
There were positive relationships between the incidence of FR IC and several potential explanatory variables in the dynamic regression model; for third-generation cefalosporins (mainly cefoperazone/sulbactam and ceftriaxone), first-generation cefalosporins, and linezolid. Temporal variations in third-generation cefalosporins use were followed by variations in FR IC incidence immediately, (i.e. no delay). After the third-generation cefalosporins usage increase (or decrease) by one DDD/100PD, the incidence of FR IC immediately increase (or decrease) by 0.0016/100PD. The variation of FR IC incidence was 0.083/100PD for first-generation cefalosporin (3 months delay) and 0.011/100PD for linezolid (2 months delay) (Table 2 and Figure 1A). Over the study period, half (52%) of the variations of the monthly incidence of FR IC were explained by the factors included in the model (R2 0.52).
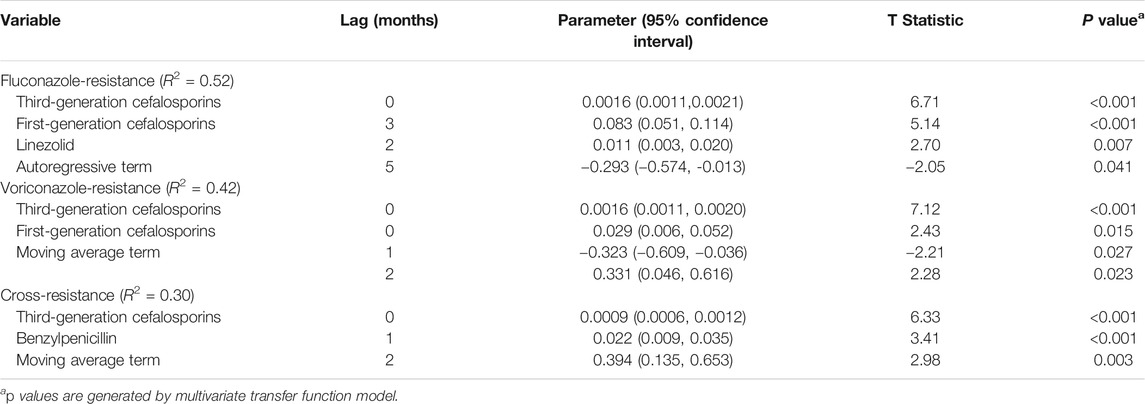
TABLE 2. Antibiotic use for the incidence (per 100 patient-days) of non-duplicate Candida spp. resistant to azoles (November 2013 to April 2018) using a multivariate transfer function model.
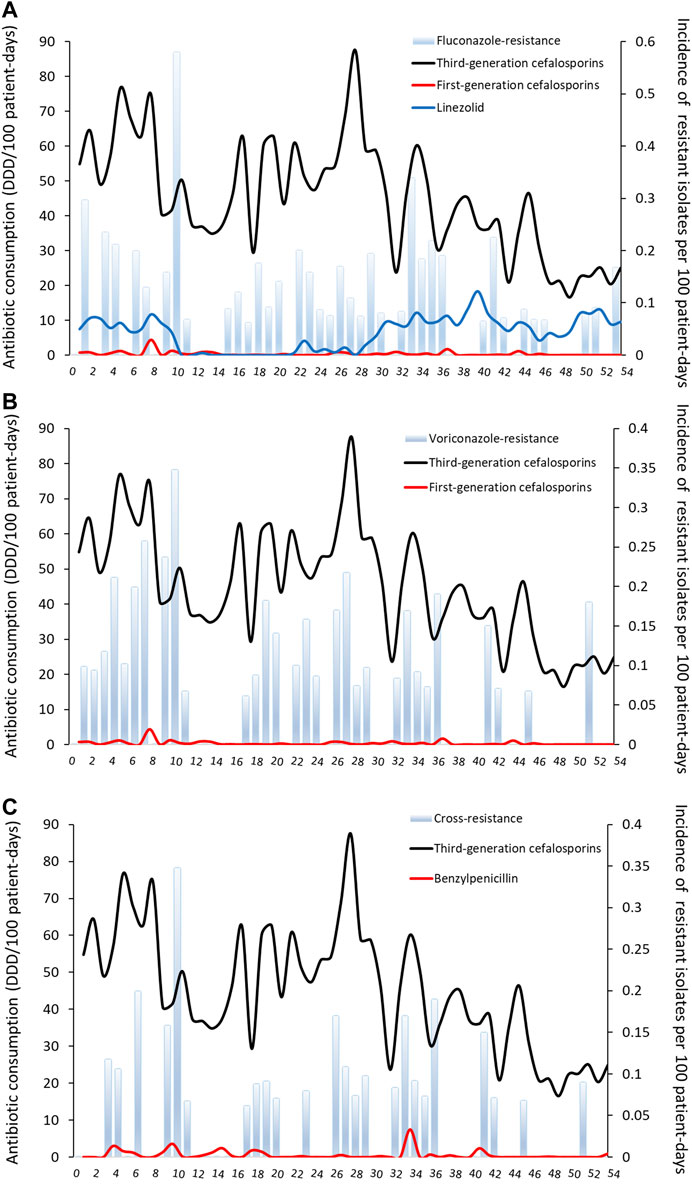
FIGURE 1. Monthly incidence of non-duplicate triazole-resistant isolates and antibiotic consumption per 100 patient-days (A) Fluconazole-resistant invasive candidiasis (B) Voriconazole-resistant invasive candidiasis (C) Cross-resistant invasive candidiasis. The number on the x-axis are months (from November 2013 to April 2018).
The incidence of VR IC was positively correlated with the previous use of third-generation cefalosporins and first-generation cefalosporins. The variation of VR was 0.0016/100PD for third-generation cefalosporins and 0.029/100PD for first-generation cefalosporins (both for no delay) (R2 0.42, Table 2 and Figure 1B). For CR IC, the incidence of CR IC was positively correlated with previous use of third-generation cefalosporins (0.0009/100PD, no delay) and benzylpenicillin (0.022/100PD, average delay of one month; R2 0.30, Table 2 and Figure 1C).
The results of multinomial regression analysis for different Candida species were presented in the Supplementary Material (Supplementary Appendixes SA6, SA8; Supplementary Table S3).
The demographic and clinical characteristics of the study patients (n = 393) and patients with different triazole-resistant phenotypes, as well as some associations for FR, VR, and CR IC identified in the bivariate analysis are presented in the Supplementary Table S4; Supplementary Appendix SA9).
The independent antibiotic-risk factors for FR IC included previous exposure to fluconazole (odds ratio [OR] 2.73, 95% confidence interval [CI] 1.38, 5.39, p = 0.004), caspofungin (OR 11.32, 95% CI 2.48, 51.74, p = 0.002), or glycopeptides (mainly vancomycin) (OR 2.32, 95% CI 1.22, 4.42, p = 0.010; Table 3). We found no associations with VR IC after adjusted for propensity-scores (Table 3). CR IC was positively associated with previous exposure to voriconazole only (OR 2.87, 95%CI 1.03, 8.01, p = 0.045) (Table 3). After adjusting for propensity-scores and SOFA score, exposure to fluconazole was associated with a non-statistically significant trend for CR IC (OR 2.26, 95%CI 0.96, 5.30, p = 0.062).
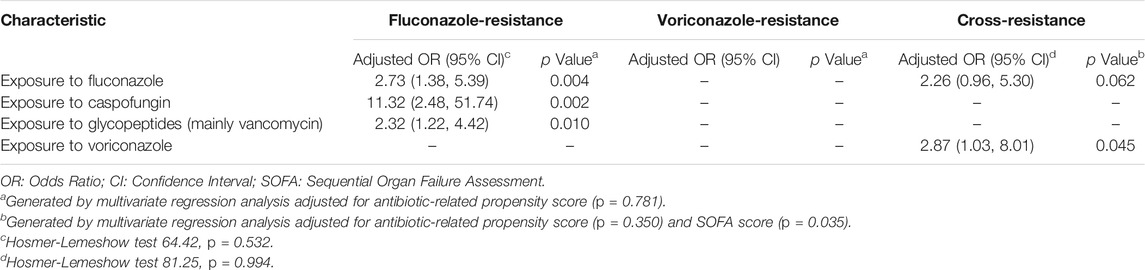
TABLE 3. Adjusted odds of antibiotics associated with fluconazole-resistant, voriconazole-resistant and cross-resistant invasive candidiasis using multivariate analyses.
The 14 days all-cause mortality rate was 26.0% (101/388) among the ICU patients. The rate varied: CR IC was 43.8% (14/32), mono-triazole-resistant IC was 26.7% (12/45) while those with non-resistant IC was 24.1% (75/311, p = 0.006; Figure 2). There was an association between 14 days all-cause mortality and resistant phenotypes (adjusted hazards ratio [aHR] 1.55, 95% CI 1.17, 2.05, p = 0.002) after adjusting for APACHE II scores at IC diagnosis (aHR 1.08, 95%CI 1.05, 1.11, p < 0.001), and invasive mechanical ventilation (aHR 1.80, 95%CI 1.10, 2.95, p = 0.021). There was no difference in the length of stay following infection between patients with FR IC and patients with non-FR IC, between patients with VR IC and patients with non-VR IC, nor between patients with CR IC and patients with non-CR IC (Supplementary Table S4; Supplementary Appendix SA9).

FIGURE 2. Cumulative survival in patients with cross-resistant, mono-triazole-resistant, and non-resistant invasive candidiasis using Kaplan-Meier curves. The dashed line indicates the 95% confidence interval: yellow for non-resistant; green for mono-triazole-resistant; red for cross-resistant invasive candidiasis.
Exposure to antibiotic remains one of the most important risk factors for acquiring antibiotic-resistant Candida spp. (Ben-Ami et al., 2012). We applied two different methodologies (group-level and individual-patient-level) to analyze antibiotic-use-versus-resistance relations for Candida spp. isolates in critically ill patients, yielding distinct results. Exposure to several antibacterial agents, specifically third-generation cefalosporins, but not triazoles, have an immediate effect (time lag = 0) on subsequent ICU-acquired triazole-resistant IC in the group-level, indicating an "indirect" effect on resistance. When the same patient database was analyzed at the individual-patient-level, we found that exposure to many antifungal agents was significantly associated with triazole-resistance.
In group-level analyses, previous fluconazole use (for 3-months delay) was associated with the emergence of FR, VR and CR IC when using univariate TF models. However, when we introduced third-generation cefalosporins exposure (mainly cefoperazone/sulbactam and ceftriaxone) into the multivariate TF model, exposure to fluconazole was no longer a significant risk factor for developing triazole-resistant IC and the model performance became better to predict the real incidence of resistant isolates. The antibiotic-associated collateral damage which implies certain antibacterials with an antianaerobic spectrum of activity promote subsequent infection with C. glabrata infection (potentially resistant) (Lin et al., 2005) or FR infection (Ben-Ami et al., 2012) has been noted. Candida species coexists with a vast number of bacterial species in the human gastrointestinal tract, most of which are facultative or obligatory anaerobes. Most IC are caused by translocation from endogenous reservoir to normally sterile body sites. Cefoperazone/sulbactam, ceftriaxone, linezolid and benzylpenicillin, which were identified by group-level analyses for triazole-resistant IC, all have certain anti-anaerobic activity. The probable mechanism is that these antibiotics significantly alter the balance of different components of the indigenous microbial flora, which may persist for months after a relatively short course of treatment (Dethlefsen and Relman, 2011). Simply by killing off competing flora of different species, treatment with one agent may increase the burden of a pathogen resistant to another agent. For example, this has been observed with anti-anaerobic treatments that increase the load of vancomycin-resistant Enterococci (Donskey et al., 2000), as well as ceftriaxone, piperacillin-tazobactam, and metronidazole treatments that promote Candida glabrata colonization in the feces of mice (Pultz et al., 2005). Other proposed mechanisms include direct antifungal activities or modulating azole resistance by inducing the expression of efflux-pump-encoding genes of some antibacterials (de Micheli et al., 2002; Zhai et al., 2010).
Regarding the patient-level analyses, previous exposure to fluconazole or caspofungin was positively correlated with the emergence of FR IC. Given that previous exposure to echinocandins is a significant risk factor for echinocandin-resistance and multidrug resistance, our results might suggest echinocandin-resistance was strongly associated with FR, which is in agreement with previous reports (Farmakiotis et al., 2014; Pham et al., 2014). We only found that exposure to fluconazole can affect FR, but not VR and CR, while exposure to voriconazole would affect CR, but not VR. These findings were in accord with another report (Bailly et al., 2016), suggesting that we should use voriconazole carefully to avoid causing more serious drug-resistance. No triazole affected VR IC: this was unexpected and needs to be confirmed by further studies.
Particularly important findings of this study were the all-cause mortality were closely related to different resistant phenotypes (CR vs. mono-triazole-resistance vs. non-resistance) after adjusting for severity of illness, and the number of species associated with high fluconazole or voriconazole MIC was greater for CR IC than for mono-triazole-resistant IC, indicating a higher level of resistance for CR species. Altogether, CR IC may have a worrisome propensity compared to mono-triazole-resistant IC. Nevertheless, the new Chinese medical insurance policy 2020 may still force Chinese clinicians to prioritize triazoles (mainly fluconazole and voriconazole), despite investigations showing that initial echinocandin treatment reduces mortality in critically ill patients with candidemia (Garnacho-Montero et al., 2018). This paradoxical dilemma might lead to therapeutic failure and decrease the survival of patients with IC.
Different types of epidemiological studies have been performed to quantify the association between antibiotic exposure and Candida resistance (Ben-Ami et al., 2012; Farmakiotis et al., 2014; Bailly et al., 2016). Our results indicated the different methodological approaches are not mutually transposable. Some experts indicated that group-level analytic methods alone cannot reliably clarify causal relationships between exposure to antibiotic and resistance due to some ecological biases, (e.g. the key information was lost in the data aggregation process-the sequence of antibiotic exposure and the emergence of a resistant organism has not been reflected). However, ecological studies (aggregated data) are potentially helpful for the researches of infectious diseases, since they can estimate the overall effect of antibiotic exposure, (i.e. not just the direct effects on the individual receiving the antibiotic but also the indirect effects mediated through effects on transmission capacity). (Harbarth et al., 2001). Therefore, considering the different ways in which antibiotics affect antimicrobial resistance, antibacterials with antianaerobic activity seem to have an “indirect” effect, while antifungal agents have a “direct” effect on triazole-resistant IC.
We acknowledge five limitations of our study. Firstly, this is a retrospective analysis and our sample size was relatively small for differentiating individual resistant Candida species. Inherent risk factors may differ between single species and the combined ones we used but several studies examining potential factors for resistance also treated multiple species as a single group. Secondly, the antifungal agents tested in the present study were limited to only two triazoles so multidrug resistance among different antifungal classes could not be assessed. Multidrug-resistant Candida species are not uncommon—a third (36%) of echinocandin-resistant isolates were also resistant to fluconazole (Pham et al., 2014). Thirdly, both the ARIMA or regression models could have missed several confounding factors, which might bias the results and lead to the observed relationships. Additionally, the stepwise regression analysis does not always choose the best possible combination of risk factors. When there is a small sample size compared to the number of variables being studied, the selection of variables using a stepwise regression will be unstable. Nevertheless, this instability is reduced when there is a sample size (or number of events) greater than 50 per candidate variable (Steyerberg et al., 2001). Moreover, by reducing the number of variables, stepwise selection will yield a simple and easily interpretable model. Fourthly, we adopted incidence as our measure for the burden of IC. It is noted that measuring the proportion of resistant isolates among all isolates is a common method, especially for situation where the overall number of infections does not change dramatically (Burton et al., 2009; Schechner et al., 2013). Nevertheless, the use of antibiotics will reduce the number of susceptible isolates, so even if the number of resistant isolates does not increase, the proportion of resistant organisms would increase. Schechner and colleagues, therefore instead recommend the “incidence” as an indicator of the burden of resistance (Schechner et al., 2013). Finally, the resistance mechanism of triazoles and defining genotype relationships between the species by molecular typing have not been clarified in our study. The clarification of these mechanisms can better help understand the level of resistance to triazoles and confirm the patient-to-patient transmission.
Despite these limitations, we believe that this study is valuable since it combines two different statistical methods (group-level and individual-patient-level) to provide further evidence on how antibiotic use influences the incidence of varying triazole-resistance in ICU patients, which allows us to interpret the results in two divergent perspectives. Moreover, we were able to investigate clinical outcomes for different resistant phenotypes to confirm the worrying status of antifungal resistance.
In conclusion, antibacterials with antianaerobic activity and antifungals might have “indirect” and “direct” effect on triazole-resistant IC, respectively, suggesting that preventing the misuse of antifungals and antibacterials with activity against the anaerobic gastrointestinal flora in ICU is necessary to avoid triazole-resistance phenomena without compromising the efficacy of antifungal therapy. Inappropriate initial treatment might lead to the more-complicated and worse cross-resistant phenotype, which should be considered when formulating healthcare policies.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving human participants were reviewed and approved by the Ethics Committee of the First Affiliated Hospital and the Second Affiliated Hospital of Xi’an Jiaotong University. Written informed consent for participation was not required for this study in accordance with the national legislation and the institutional requirements.
Concept and design: YW and YD. Acquisition, analysis or interpretation of data: YW, YZ, and LC. Drafting of the manuscript: YW, TM, SH, MD, LC, and XW. Critical revision of the manuscript for important intellectual content: all authors. All authors read and approved the final manuscript.
This work was supported by internal funding, (i.e. the National Natural Science Foundation of China (No. 71904155)).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors acknowledge the Junhui Liu for isolation and identification of isolates.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2021.586893/full#supplementary-material.
Bailly, S., Maubon, D., Fournier, P., Pelloux, H., Schwebel, C., Chapuis, C., et al. (2016). Impact of antifungal prescription on relative distribution and susceptibility of candida spp.—trends over 10 years. J. Infect. 72, 103–111. doi:10.1016/j.jinf.2015.09.041
Ben-Ami, R., Olshtain-Pops, K., Krieger, M., Oren, I., Bishara, J., Dan, M., et al. (2012). Antibiotic exposure as a risk factor for fluconazole-resistant candida bloodstream infection. Antimicrob. Agents Chemother. 56, 2518–2523. doi:10.1128/AAC.05947-11
Burton, D. C., Edwards, J. R., Horan, T. C., Jernigan, J. A., and Fridkin, S. K. (2009). Methicillin-resistant staphylococcus aureus central line-associated bloodstream infections in us intensive care units, 1997-2007. JAMA 301, 727–736. doi:10.1001/jama.2009.153
Calandra, T., Roberts, J. A., Antonelli, M., Bassetti, M., and Vincent, J. L. (2016). Diagnosis and management of invasive candidiasis in the icu: an updated approach to an old enemy. Crit. Care 20, 125. doi:10.1186/s13054-016-1313-6
de Micheli, M., Bille, J., Schueller, C., and Sanglard, D. (2002). A common drug-responsive element mediates the upregulation of the candida albicans abc transporters cdr1 and cdr2, two genes involved in antifungal drug resistance. Mol. Microbiol. 43, 1197–1214. doi:10.1046/j.1365-2958.2002.02814.x
Dethlefsen, L., and Relman, D. A. (2011). Incomplete recovery and individualized responses of the human distal gut microbiota to repeated antibiotic perturbation. Proc. Natl. Acad. Sci. USA 108 (Suppl. 1), 4554–4561. doi:10.1073/pnas.1000087107
Donskey, C. J., Chowdhry, T. K., Hecker, M. T., Hoyen, C. K., Hanrahan, J. A., Hujer, A. M., et al. (2000). Effect of antibiotic therapy on the density of vancomycin-resistant enterococci in the stool of colonized patients. N. Engl. J. Med. 343, 1925–1932. doi:10.1056/NEJM200012283432604
Farmakiotis, D., Tarrand, J. J., and Kontoyiannis, D. P. (2014). Drug-resistant candida glabrata infection in cancer patients. Emerging Infect. Dis. 20, 1833–1840. doi:10.3201/eid2011.140685
Garnacho-Montero, J., Díaz-Martín, A., Cantón-Bulnes, L., Ramírez, P., Sierra, R., Arias-Verdú, D., et al. (2018). Initial antifungal strategy reduces mortality in critically ill patients with candidemia: a propensity score-adjusted analysis of a multicenter study. Crit. Care Med. 46, 384–393. doi:10.1097/CCM.0000000000002867
Guo, F., Yang, Y., Kang, Y., Zang, B., Cui, W., Qin, B., et al. (2013). Invasive candidiasis in intensive care units in China: a multicentre prospective observational study. J. Antimicrob. Chemother. 68, 1660–1668. doi:10.1093/jac/dkt083
Hammarskjöld, F., Mernelius, S., Andersson, R. E., Berg, S., Hanberger, H., Löfgren, S., et al. (2013). Possible transmission of candida albicans on an intensive care unit: genotype and temporal cluster analyses. J. Hosp. Infect. 85, 60–65. doi:10.1016/j.jhin.2013.06.002
Harbarth, S., Harris, A. D., Carmeli, Y., and Samore, M. H. (2001). Parallel analysis of individual and aggregated data on antibiotic exposure and resistance in gram-negative bacilli. Clin. Infect. Dis. 33, 1462–1468. doi:10.1086/322677
Helfenstein, U. (1996). Box-jenkins modelling in medical research. Stat. Methods Med. Res. 5, 3–22. doi:10.1177/096228029600500102
Jensen, J. U., Hein, L., Lundgren, B., Bestle, M. H., Mohr, T., Andersen, M. H., et al. (2015). Invasive candida infections and the harm from antibacterial drugs in critically ill patients: data from a randomized, controlled trial to determine the role of ciprofloxacin, piperacillin-tazobactam, meropenem, and cefuroxime. Crit. Care Med. 43, 594–602. doi:10.1097/CCM.0000000000000746
Lin, M. Y., Carmeli, Y., Zumsteg, J., Flores, E. L., Tolentino, J., Sreeramoju, P., et al. (2005). Prior antimicrobial therapy and risk for hospital-acquired candida glabrata and candida krusei fungemia: a case-case-control study. Antimicrob. Agents Chemother. 49, 4555–4560. doi:10.1128/AAC.49.11.4555-4560.2005
Lipsitch, M., and Samore, M. H. (2002). Antimicrobial use and antimicrobial resistance: a population perspective. Emerging Infect. Dis. 8, 347–354. doi:10.3201/eid0804.010312
López-Lozano, J. M., Monnet, D. L., Yagüe, A., Burgos, A., Gonzalo, N., Campillos, P., et al. (2000). Modelling and forecasting antimicrobial resistance and its dynamic relationship to antimicrobial use: a time series analysis. Int. J. Antimicrob. Agents 14, 21–31. doi:10.1016/s0924-8579(99)00135-1
Lortholary, O., Renaudat, C., Sitbon, K., Madec, Y., Denoeud-Ndam, L., Wolff, M., et al. (2014). Worrisome trends in incidence and mortality of candidemia in intensive care units (paris area, 2002-2010). Intensive Care Med. 40, 1303–1312. doi:10.1007/s00134-014-3408-3
Martin-Loeches, I., Antonelli, M., Cuenca-Estrella, M., Dimopoulos, G., Einav, S., De Waele, J. J., et al. (2019). Esicm/escmid task force on practical management of invasive candidiasis in critically ill patients. Intensive Care Med. 45, 789–805. doi:10.1007/s00134-019-05599-w
Martín-Loeches, I., Diaz, E., and Vallés, J. (2014). Risks for multidrug-resistant pathogens in the icu. Curr. Opin. Crit. Care 20, 516–524. doi:10.1097/MCC.0000000000000124
Pappas, P. G., Lionakis, M. S., Arendrup, M. C., Ostrosky-Zeichner, L., and Kullberg, B. J. (2018). Invasive candidiasis. Nat. Rev. Dis. Primers 4, 18026. doi:10.1038/nrdp.2018.26
Perlin, D. S. (2015). Echinocandin resistance in candida. Clin. Infect. Dis. 61 (Suppl. 6), S612–S617. doi:10.1093/cid/civ791
Perlin, D. S., Rautemaa-Richardson, R., and Alastruey-Izquierdo, A. (2017). The global problem of antifungal resistance: prevalence, mechanisms, and management. Lancet Infect. Dis. 17, e383–e392. doi:10.1016/S1473-3099(17)30316-X
Pham, C. D., Iqbal, N., Bolden, C. B., Kuykendall, R. J., Harrison, L. H., Farley, M. M., et al. (2014). Role of fks mutations in candida glabrata: mic values, echinocandin resistance, and multidrug resistance. Antimicrob. Agents Chemother. 58, 4690–4696. doi:10.1128/AAC.03255-14
Pultz, N. J., Stiefel, U., Ghannoum, M., Helfand, M. S., and Donskey, C. J. (2005). Effect of parenteral antibiotic administration on establishment of intestinal colonization by candida glabrata in adult mice. Antimicrob. Agents Chemother. 49, 438–440. doi:10.1128/AAC.49.1.438-440.2005
Schechner, V., Temkin, E., Harbarth, S., Carmeli, Y., and Schwaber, M. J. (2013). Epidemiological interpretation of studies examining the effect of antibiotic usage on resistance. Clin. Microbiol. Rev. 26, 289–307. doi:10.1128/CMR.00001-13
Steyerberg, E. W., Eijkemans, M. J., Harrell, F. E., and Habbema, J. D. (2001). Prognostic modeling with logistic regression analysis: in search of a sensible strategy in small data sets. Med. Decis. Making 21, 45–56. doi:10.1177/0272989X0102100106
Vallabhaneni, S., Baggs, J., Tsay, S., Srinivasan, A. R., Jernigan, J. A., and Jackson, B. R. (2018). Trends in antifungal use in us hospitals, 2006-12. J. Antimicrob. Chemother. 73, 2867–2875. doi:10.1093/jac/dky270
Vernaz, N., Huttner, B., Muscionico, D., Salomon, J. L., Bonnabry, P., López-Lozano, J. M., et al. (2011). Modelling the impact of antibiotic use on antibiotic-resistant escherichia coli using population-based data from a large hospital and its surrounding community. J. Antimicrob. Chemother. 66, 928–935. doi:10.1093/jac/dkq525
Vincent, J. L., Rello, J., Marshall, J., Silva, E., Anzueto, A., Martin, C. D., et al. (2009). International study of the prevalence and outcomes of infection in intensive care units. JAMA 302, 2323–2329. doi:10.1001/jama.2009.1754
Wang, Y., McGuire, T. M., Hollingworth, S. A., Dong, Y., and Van Driel, M. L. (2019). Antifungal agents for invasive candidiasis in non-neutropenic critically ill adults: what do the guidelines recommend?. Int. J. Infect. Dis. 89, 137–145. doi:10.1016/j.ijid.2019.10.016
Keywords: invasive candidiasis, critical illness, drug resistance, fungal, time series analysis, regression analysis
Citation: Wang Y, Zhang Y, McGuire TM, Hollingworth SA, Van Driel ML, Cao L, Wang X and Dong Y (2021) ICU Patients’ Antibiotic Exposure and Triazole-Resistance in Invasive Candidiasis: Parallel Analysis of Aggregated and Individual Data. Front. Pharmacol. 12:586893. doi: 10.3389/fphar.2021.586893
Received: 11 August 2020; Accepted: 10 February 2021;
Published: 22 March 2021.
Edited by:
Jean Paul Deslypere, Aesculape CRO, BelgiumReviewed by:
Scott Olesen, OpenBiome, Cambridge, United StatesCopyright © 2021 Wang, Zhang, McGuire, Hollingworth, Van Driel, Cao, Wang and Dong. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yalin Dong, ZG9uZ3lhbGluQG1haWwueGp0dS5lZHUuY24=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.