
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Pediatr. , 18 March 2024
Sec. Pediatric Urology
Volume 12 - 2024 | https://doi.org/10.3389/fped.2024.1334544
Fetal Wilms tumor (WT) is extremely rare, but with advances in fetal imaging, more cases are being reported. The management of these cases remains challenging. Herein, we present the case of a full-term female infant diagnosed antenatally at 32 weeks of gestation with a right solid renal mass detected on routine prenatal ultrasound without polyhydramnios. At birth, the infant was healthy, with no evidence of dysmorphic features or abnormal laboratory tests to suggest a predisposition syndrome. Her family history was also unremarkable. A successful radical right nephrectomy was performed on day 2 of life revealing a classic WT. She received vincristine as adjuvant chemotherapy without any complications. At the age of 1 month, the infant developed isolated lateralized overgrowth of the right lower limb suspicious of Beckwith–Wiedemann syndrome. At the latest follow-up of 4 years, the child is healthy and disease-free with conserved asymmetry of lower limbs. The case provides insights into the challenging diagnosis and treatment of fetal WT. A review of the literature suggests that the presence of polyhydramnios is a worse prognostic factor while the combination of best supportive care and surgery remains the best management. Fetal WT can be associated with predisposition syndromes; however, their first manifestations can develop after the diagnosis of cancer has been made, as in our patient. We propose starting active surveillance programs and genetic testing for any case of fetal WT.
Fetal tumors are rare entities with an estimated prevalence of 7–10 per 100,000 live births. Only 5% of these tumors arise from the kidneys (1, 2). Wilms tumor (WT) is the second most common neonatal renal tumor after congenital mesoblastic nephroma (CMN), accounting for approximately 20% of all cases, with 16% of them identified prenatally as an abdominal mass (3). Some cases are associated with prenatal polyhydramnios and fetal hydrops (4). Less than 10% of neonatal WT cases are associated with congenital syndromes such as Wilms tumor, aniridia, genitourinary abnormalities, range of developmental delays (WAGR) syndrome, Beckwith–Wiedemann syndrome (BWS)/isolated lateralized overgrowth (ILO), Denys–Drash syndrome, and Simpson–Golabi–Behmel syndrome (2, 5). In the present study, we report the case of a fetal WT detected in the third trimester by routine prenatal ultrasound in a female infant before she developed isolated hemihypertrophy of her lower limb postnatally.
A PubMed search was performed for articles indexed through the Medline database. Keywords used included “fetal renal tumors” and “Wilms Tumor” or “Wilms’ Tumour” and “prenatal diagnosis”. In total, 44 articles were identified, of which 13 were case reports of interest. A total of 13 different patients were described within these 13 case reports published between 1984 and 2021 (Table 1). A total of 31 articles were excluded: 16 articles were not about WT, 13 were not related, and 2 were not case reports. Furthermore, when we added the term “hemihypertrophy,” we retrieved only one article that was not related to our topic. A qualitative analysis was performed on the included cases, and the findings are presented.
A 27-year-old previously healthy primigravida mother with an uneventful pregnancy presented at 32 weeks of gestation (WG), with a routine prenatal ultrasound showing an isolated heterogeneous fetal mass of 2 cm occupying the right renal fossa. Family history was negative for any malignancies or renal diseases. At 34 WG, a fetal magnetic resonance imaging (MRI) scan was performed and confirmed the presence of a solid multinodular right renal mass of 6 cm with a normal contralateral kidney and no other associated congenital malformations (Figures 1A,B). The pregnancy went smoothly until term, with weekly follow-up fetal ultrasound examinations showing a gradual increase in the size of the mass but with normal amniotic fluid and fetal growth. There were no signs of fetal distress. The delivery was performed at 38 WG by elective cesarean section (CS) to avoid mass rupture.
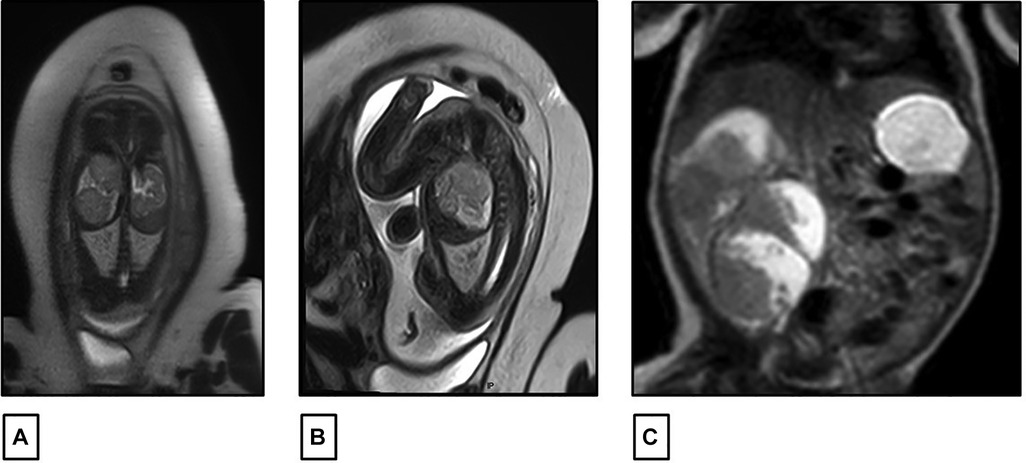
Figure 1. (A,B) Fetal MRI performed at 34 weeks of gestation showing a right renal fossa occupied by a 60 mm × 30 mm multinodular solid mass with intermediate signal intensity on T2. (C) Postnatal abdominal MRI showing a lobulated heterogeneous mass in the right renal fossa 81 mm × 58 mm.
At birth, the female infant was healthy with a good Apgar score. The examination of the placenta was normal and did not show placental mesenchymal dysplasia. The infant’s physical exam was unremarkable except for a palpable right upper quadrant mass. No macroglossia, omphalocele, macrosomia, or other dysmorphic features were detected. Laboratory workups were carried out and were normal, except for a mild elevation of serum lactate dehydrogenase (LDH) at 534 U/L. There were no recorded episodes of hypoglycemia or hypercalcemia. An abdominal ultrasound was also controlled postnatally, revealing a significant increase in tumor size to 8 cm. The mass had multiple lobules surrounded by vessels without a clear intra-tumoral Doppler flow. The left kidney appeared normal in size and echogenicity with no dilatation of the excretory system. The liver and spleen were normal; there was no evidence of nephrogenic rest, nor adrenal cytomegaly or pancreatic adenomatosis. There was no evidence of renal vein or inferior vena cava thrombus on Doppler study. A complementary abdominal MRI scan was also performed on the same day, confirming the same radiological findings with a lobulated heterogeneous mass at the right renal fossa, measuring 81 mm × 58 mm, with hyper- and hypo-intense signals respectively on T1 and T2. It was well limited to the kidney with no evidence of enlarged lymph nodes (Figure 1C).
On the second day of life, the patient underwent a right radical nephrectomy with excision of the supra-hilar and para-aortic lymph nodes. A pathology study revealed a well-circumscribed and encapsulated tumoral proliferation that was limited to the kidney (Figure 2). It contained three types of cells, composed of epithelial cells that formed immature tubules and primitive-looking glomeruli, mesenchymatous cells made up of spindle cells, and blastemal cells that were round and blue with scanty cytoplasm. Foci of necrosis were present. The adjacent renal parenchyma showed cystic dilations in the cortex and medulla. The cysts were lined by a flattened cuboid epithelium. No anaplastic features were identified. There was no vascular invasion, and the excised lymph nodes had no signs of malignancy. Thus, the diagnosis of triphasic WT or nephroblastoma was confirmed. Staging was completed with a computed tomography scan of the chest, which came back normal, and the tumor was classified as stage I according to the International Society of Pediatric Oncology (SIOP) and the National Wilms Tumor Study (NWTS) Group Staging System (19). Three weeks after surgery, adjuvant chemotherapy was started with weekly vincristine for a total of 10 weeks, without major adverse events.
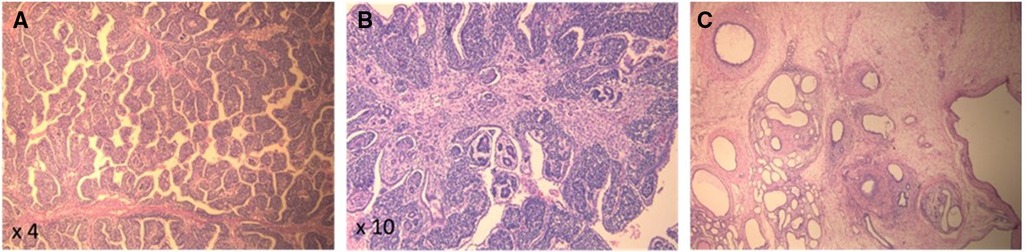
Figure 2. (A,B) Pathology (H&E) of the kidney tumor showing pupillary structures supported by triphasic neoplastic proliferation comprising immature glomerular and tubular structures and immature small round blue cells. (C) Background renal parenchyma showing microcystic dilated tubules, strangled by concentric layers of spindled mesenchymal cells.
At 1 month of age, the infant developed an isolated right lower limb hemihypertrophy without skin staining (Figure 3). No vascular or capillary limb malformation was noted on Doppler study. The cardiac ultrasound and retinal examination were unremarkable. With the addition of this new clinical feature, Beckwith–Wiedemann syndrome was highly suspected and genetic testing was recommended. However, the parents did not give their consent to proceed with molecular studies. The child had since then regular clinical, biological, and radiological surveillance every 3 months. At the last follow-up at 4 years of age, the girl had normal growth (75th percentile for weight and height) and developmental milestones for her age, with no tumor recurrence and stable lower limb asymmetry with a difference of 1 cm.
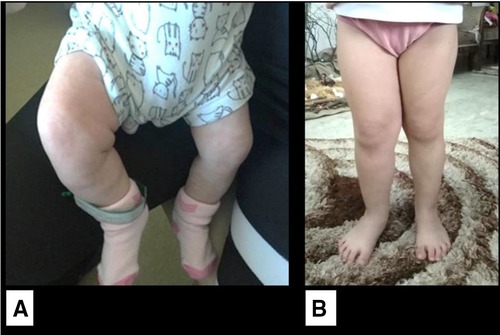
Figure 3. Isolated lateralized hemihypertrophy of the right lower limb. (A) At 1 month of age. (B) At 4 years of age.
Fetal renal tumors are defined as tumors involving the kidney discovered before birth. They are extremely rare, occurring in 7 out of 100,000 births (20). The majority are benign, with CMN being the most common tumor. WT, on the other hand, is the most common malignant renal tumor, with a prevalence of 0.12% (21). Reports of fetal WT are scarce and are limited to case reports (Table 1).
In their review, Berger and von Schweinitz (1) proposed an approach to the management of fetal kidney tumors. Prenatal ultrasound remains the most preferred modality in the diagnosis and follow-up of antenatal renal tumors. They are usually detected in the third trimester and have a predilection to the right side. Although prenatal fetal MRI does not differentiate between CMN and WT (22), it is highly valuable in evaluating large renal masses (>10 cm), in assessing the contralateral kidney and adjacent structures and in planning resection. Since the majority of fetal tumors diagnosed early are either benign or low-stage malignant tumors, Berger and von Schweinitz recommended that initial management should focus on immediate counseling and perinatal care. When it comes to delivery, vaginal deliveries can be safely performed in non-complicated cases, while a cesarean section is indicated for complicated pregnancies and large renal tumors (23). Postnatal management of prenatally detected renal tumors remains surgical (1). There is no indication for percutaneous needle biopsy unless there is evidence of metastatic disease or unresectable tumor (24). Once the newborn has achieved adequate postnatal adaptation and hemodynamic stability, a radical nephrectomy should be attempted. Complete surgical resection with negative margins and without tumoral spillage is recommended to prevent local recurrence. Although nephron-sparing surgery is gaining in popularity (25), it is still reserved for cases of either bilateral WT or whenever there is concern for contralateral kidney involvement or function (26).
Due to the low incidence of fetal WT, management has not been standardized, but it is in line with what has been described for fetal kidney tumors in general. In our review, all 13 cases were diagnosed in the third trimester using routine ultrasound. There were four bilateral WTs, seven tumors originating from the left kidney, and two from the right kidney. Five cases were associated with polyhydramnios. There were five preterm deliveries secondary to fetal distress. Delivery was by CS in six patients. Radical nephrectomy was performed in eight patients while three patients with bilateral WT had nephron-sparing surgery. Tumor staging was low: six tumors were stage 1; and one tumor was stage 2. Only two tumors were stage 3. Five patients received adjuvant chemotherapy. None of the reviewed cases were associated with BWS or ILO; however, there was one case of nephroblastomatosis (6), which is a condition generally associated with WT.
In general, the outcome of WT is excellent, with overall survival (OS) of 90% at 5 years (24, 27). In their report, van den Heuvel-Eibrink et al. showed that patients with WT aged less than 7 months had an OS of 93%; however, they did not specify the outcome of the patients diagnosed antenatally (28). In fact, the prognosis of fetal WT depends on several factors, including the presence of polyhydramnios, staging, histology type, and the presence of metastasis. Reports from Leclair et al. showed that 39% of fetal renal tumors had polyhydramnios as the main perinatal complication (23). Other reported complications include hypercalcemia, intra-tumoral hemorrhage, and non-immune hydrops fetalis leading to prematurity, fetal distress, and fetal demise (29). Our review of the data showed that the survival rate is excellent for infants who had isolated fetal WT, except for one patient who had disease progression secondary to therapeutic delays and suboptimal management leading to death (12) and another fetus that had several malformations that could be related to a syndrome and can explain the early mortality (14). On the other hand, all five patients who had fetal WT associated with polyhydramnios had a poor outcome and died in the early neonatal period. Moreover, polyhydramnios was responsible for fetal distress, hydrops, prematurity, and hemodynamic instability. Two patients died within 24 h of birth before the attempt of surgery. As for the remaining three patients, they were hemodynamically unstable, and when surgery was attempted to relieve the pressure from the tumor, it resulted in high morbidity, major postoperative complications, and death. In addition to kidney tumors, polyhydramnios has also been associated with other conditions, such as fetal gastrointestinal obstruction, neuromuscular disorders, fetal malformation syndromes, severe fetal anemia, maternal diabetes mellitus, macrosomia, and fetal hydrops and infection (TORCH), resulting in a poor neonatal outcome (30). Whether polyhydramnios was caused solely by WT or secondary to any other associated conditions could not be verified in this review and needs a larger study to investigate this hypothesis. Therefore, polyhydramnios is a worse prognostic factor associated with fetal WT. Special consideration should be directed to infants who are diagnosed with WT and polyhydramnios, as they need to be followed in a tertiary care center by an experienced multidisciplinary team.
Our case is illustrative of the management of a fetal WT. The fetal ultrasound detected a right renal mass at 32 WG without polyhydramnios nor fetal distress, and fetal MRI confirmed the presence of a solitary kidney mass without local dissemination. After the initial diagnosis, the parents were immediately referred to a tertiary care center, and the pregnancy was followed by a multidisciplinary team, including a specialized fetal radiologist, a pediatric nephrologist, a pediatric surgeon, and a neonatologist. Although the delivery could be done by the vaginal route, it was agreed with the parents to proceed with a CS because of the large size of the mass, thus decreasing the risk of tumor rupture. At birth, there were no documented episodes of hypercalcemia nor hypoglycemia. In addition, there was no family history, no dysmorphic features, or anomalies to suggest a predisposition syndrome. The infant underwent radical nephrectomy on the second day of life since she was hemodynamically stable. The pathologic examination of the mass showed a favorable histology WT, and confined to the renal capsule with negative lymph nodes, thus making it stage 1. These findings are similar to those in international studies where the majority of neonatal WT have favorable histology and low staging (28) and will not receive adjuvant chemotherapy. However, this infant had several concerning risk factors including a large tumor (approximately 500 g), unknown status of loss of heterozygosity (LOH) of 1p/16q, and the absence of a genetic study. After discussing the case in the multidisciplinary meeting, we upgraded this tumor to intermediate risk (19). Therefore, the female infant received dose-adjusted weekly vincristine over a period of 10 weeks according to the SIOP Umbrella RTSG 2016 protocol (24) without any complications.
What makes our case unique is the development after birth of isolated hemihypertrophy of the right lower limb, which can be associated with a predisposition syndrome (31). Hemihypertrophy is defined as an increase in length or circumference of one side of the body or limb compared to the contralateral part. It can be part of a syndrome, such as in BWS, or isolated, hence the new term ILO (32). On the other hand, BWS is a growth disorder commonly associated with WT. Manifestations of BWS can be detected during the fetal or neonatal period and include fetal macrosomia, macroglossia, polyhydramnios, placental mesenchymal dysplasia, and neonatal hypoglycemia (33). Our patient had no positive family history, no dysmorphic features, or other perinatal abnormal findings to suggest a diagnosis of BWS, making the diagnosis of ILO more likely. However, given the high index of suspicion, we continued screening for a potential predisposing syndrome until the development of hemihypertrophy. In a large review, MacFarland et al. reported that children with “syndromic” WT tend to be diagnosed earlier than “sporadic” WT, probably due to successful cancer screening programs. Furthermore, approximately 6% of children developed WT before the diagnosis of BWS/ILO by several months, with the youngest patient being 2 days old (34). In a prospective multicenter study of ILO, the prevalence of WT is approximately 3.5% (35), and in all reported cases, hemihypertrophy was present before the diagnosis of WT by at least 3 months (36). In our case report, the diagnosis of WT was made during the fetal period long before the development of the lower limb hemihypertrophy, an order of presentation that has not been reported before. The child benefitted, and continues to benefit, from a close screening program for early cancer detection since early infancy.
Early recognition of a predisposition syndrome is important not only to detect and promptly treat possible manifestations, but also to properly initiate surveillance programs. The benefits of screening for associated cancers (WT, hepatoblastomas, neuroblastomas) include early-stage detection of small and localized tumors, improved prognosis, less intensive treatment, and the omission of radiation therapy. Screening for non-cancerous conditions is equally essential for the early detection and management of potential morbidities, namely, hypoglycemia/hyperinsulinism and kidney and musculoskeletal disorders. Genetic testing remains essential in confirming the diagnosis and for genetic counseling.
This case illustrates the challenging management of fetal WT. Prenatal ultrasound remains an essential tool for diagnosis. Immediate care should be offered in a tertiary care center by an experienced multidisciplinary team. Surgical resection is the standard of care after stabilization of the newborn. The prognosis is generally excellent but depends on the tumor's staging, histology, and on the presence of polyhydramnios, which is a worse prognostic factor. Fetal WT can be associated with predisposition syndromes, but their manifestations may develop after the diagnosis of cancer has been made. Physicians should be aware of this possibility and not be satisfied with treating only the tumor. Therefore, they are encouraged to continue screening for predisposition syndromes and to offer genetic testing to start surveillance programs and plan early interventions.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Written informed consent was obtained from the minor(s)’ legal guardian/next of kin for the publication of any potentially identifiable images or data included in this article.
EB: Writing – original draft. CS: Writing – review & editing. CG: Writing – review & editing. DC: Writing – review & editing. PA: Writing – review & editing.
The authors declare that no financial support was received for the research, authorship, and/or publication of this article.
We would like to thank Dr Zarouhie Meguerian, MD, for providing the pathology description and images for this case and Dr Sandra Hasbani, MD, for providing the MRI images.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Berger M, von Schweinitz D. Current management of fetal and neonatal renal tumors. Curr Pediatr Rev. (2015) 11(3):188–94. doi: 10.2174/1573396311666150714105403
2. Powis M. Neonatal renal tumours. Early Hum Dev. (2010) 86(10):607–12. doi: 10.1016/j.earlhumdev.2010.08.018
3. Sze SK. Neonatal renal tumors. Clin Perinatol. (2021) 48(1):71–81. doi: 10.1016/j.clp.2020.11.004
4. Isaacs H J. Fetal and neonatal renal tumors. J Pediatr Surg. (2008) 43(9):1587–95. doi: 10.1016/j.jpedsurg.2008.03.052
5. Shapiro E. Upper urinary tract anomalies and perinatal renal tumors. Clin Perinatol. (2014) 41(3):679–94. doi: 10.1016/j.clp.2014.05.014
6. Roth A. An unusual renal tumor, associating nephroblastoma, nephroblastomatosis, teratoma, and cystic dysplasia with arterial hypertension and secretion of alpha-fetoprotein. J Urol (Paris). (1984) 90(1):7–10.6202802
7. Tomá P, Lucigrai G, Dodero P, Lituania M. Prenatal detection of an abdominal mass by MR imaging performed while the fetus is immobilized with pancuronium bromide. AJR Am J Roentgenol. (1990) 154(5):1049–50. doi: 10.2214/ajr.154.5.2157328
8. Suresh I, Suresh S, Arumugam R, Govindarajan M, Reddy MP, Sulochana NV. Antenatal diagnosis of Wilms tumor. J Ultrasound Med. (1997) 16(1):69–72. doi: 10.7863/jum.1997.16.1.69
9. Applegate KE, Ghei M, Perez-Atayde AR. Prenatal detection of a Wilms’ tumor. Pediatr Radiol. (1999) 29(1):65–7. doi: 10.1007/s002470050538
10. Vadeyar S, Ramsay M, James D, O'Neill D. Prenatal diagnosis of congenital Wilms’ tumor (nephroblastoma) presenting as fetal hydrops. Ultrasound Obstet Gynecol. (2000) 16(1):80–3. doi: 10.1046/j.1469-0705.2000.00169.x
11. Linam LE, Yu X, Calvo-Garcia MA, Rubio EI, Crombleholme TM, Bove K, et al. Contribution of magnetic resonance imaging to prenatal differential diagnosis of renal tumors: report of two cases and review of the literature. Fetal Diagn Ther. (2010) 28(2):100–8. doi: 10.1159/000313655
12. Jain V, Mohta A, Sengar M, Khurana N. Is antenatal detection of Wilms’ tumor a bad prognostic marker? Indian J Med Paediatr Oncol. (2011) 32(4):214–6. doi: 10.4103/0971-5851.95144
13. Sarin YK, Rahul SK, Sinha S, Khurana N, Ramji S. Antenatally diagnosed Wilms’ tumour. J Neonatal Surg. (2014) 3(1):8. doi: 10.47338/jns.v3.72
14. Mitra S, Chatterjee D, Gowda K, Das A. A rare coexistence of bilateral congenital Wilms tumor with ductal plate malformation at autopsy. Fetal Pediatr Pathol. (2016) 35(3):186–91. doi: 10.3109/15513815.2016.1153176
15. Toussi A, Granberg CF, Gargollo PC. A case of prenatally diagnosed Wilms tumor managed with laparoscopic nephrectomy. Urology. (2018) 113:197–9. doi: 10.1016/j.urology.2017.10.045
16. Ogawa S, Schlaepfer CH, Weaver J, Meenakshi-Sundaram B, Coplen D, Rove KO, et al. Antenatal presentation of Wilms’ tumor. Urology. (2019) 134:225–7. doi: 10.1016/j.urology.2019.08.011
17. Rampersad F, Diljohn J, Goetz C. The clinical presentation, imaging features and differential diagnoses of congenital Wilms tumour. BMJ Case Rep. (2019) 12(5):e228651. doi: 10.1136/bcr-2018-228651
18. Meng D, Chang X, Ren Q, Xu J, Wang H. Congenital bilateral Wilms tumor: a case report. Urology. (2021) 157:242–5. doi: 10.1016/j.urology.2021.08.011
19. Theilen T-M, Braun Y, Bochennek K, Rolle U, Fiegel HC, Friedmacher F. Multidisciplinary treatment strategies for Wilms tumor: recent advances, technical innovations and future directions. Front Pediatr. (2022) 10:852185. doi: 10.3389/fped.2022.852185
20. Parkes S, Muir K, Southern L, Cameron A, Darbyshire P, Stevens M. Neonatal tumours: a thirty-year population-based study. Med Pediatr Oncol. (1994) 22(5):309–17. doi: 10.1002/mpo.2950220503
21. Hrabovsky EE, Othersen HB Jr., deLorimier A, Kelalis P, Beckwith JB, Takashima J. Wilms’ tumor in the neonate: a report from the national Wilms’ tumor study. J Pediatr Surg. (1986) 21(5):385–7. doi: 10.1016/S0022-3468(86)80502-4
22. Che M, Yang F, Huang H, Zhang H, Han C, Sun N. Prenatal diagnosis of fetal congenital mesoblastic nephroma by ultrasonography combined with MR imaging: a case report and literature review. Medicine (Baltimore). (2021) 100(3):e24034. doi: 10.1097/MD.0000000000024034
23. Leclair MD, El-Ghoneimi A, Audry G, Ravasse P, Moscovici J, Heloury Y, et al. The outcome of prenatally diagnosed renal tumors. J Urol. (2005) 173(1):186–9. doi: 10.1097/01.ju.0000147300.53837.8f
24. van den Heuvel-Eibrink MM, Hol JA, Pritchard-Jones K, van Tinteren H, Furtwängler R, Verschuur AC, et al. Position paper: rationale for the treatment of Wilms tumour in the UMBRELLA SIOP-RTSG 2016 protocol. Nat Rev Urol. (2017) 14(12):743–52. doi: 10.1038/nrurol.2017.163
25. Wilde JCH, Aronson DC, Sznajder B, Van Tinteren H, Powis M, Okoye B, et al. Nephron sparing surgery (NSS) for unilateral Wilms tumor (UWT): the SIOP 2001 experience. Pediatr Blood Cancer. (2014) 61(12):2175–9. doi: 10.1002/pbc.25185
26. Ehrlich PF, Chi YY, Chintagumpala MM, Hoffer FA, Perlman EJ, Kalapurakal JA, et al. Results of treatment for patients with multicentric or bilaterally predisposed unilateral Wilms tumor (AREN0534): a report from the children’s oncology group. Cancer. (2020) 126(15):3516–25. doi: 10.1002/cncr.32958
27. Dome JS, Mullen EA, Dix DB, Gratias EJ, Ehrlich PF, Daw NC, et al. Impact of the first generation of children’s oncology group clinical trials on clinical practice for Wilms tumor. J Natl Compr Cancer Netw. (2021) 19(8):978–85. doi: 10.6004/jnccn.2021.7070
28. van den Heuvel-Eibrink MM, Grundy P, Graf N, Pritchard-Jones K, Bergeron C, Patte C, et al. Characteristics and survival of 750 children diagnosed with a renal tumor in the first seven months of life: a collaborative study by the SIOP/GPOH/SFOP, NWTSG, and UKCCSG Wilms tumor study groups. Pediatr Blood Cancer. (2008) 50(6):1130–4. doi: 10.1002/pbc.21389
29. Panda SS, Mandelia A, Gupta DK, Singh A. Antenatally detected solid tumour of kidney. BMJ Case Rep. (2014) 2014:bcr2013202084. doi: 10.1136/bcr-2013-202084
30. Hamza A, Herr D, Solomayer EF, Meyberg-Solomayer G. Polyhydramnios: causes, diagnosis and therapy. Geburtshilfe Frauenheilkd. (2013) 73(12):1241–46. doi: 10.1055/s-0033-1360163
31. Turner JT, Brzezinski J, Dome JS. Wilms tumor predisposition. In: Adam MP, Feldman J, Mirzaa GM, et al., editors. Genereviews®. Seattle, WA: University of Washington, Seattle (2003). p. 1–14. [Updated 2022 Mar 24]; 1993–2023.
32. Mussa A, Carli D, Cardaropoli S, Ferrero GB, Resta N. Lateralized and segmental overgrowth in children. Cancers (Basel). (2021) 13(24):6166. doi: 10.3390/cancers13246166
33. Brioude F, Netchine I, Praz F, Le Jule M, Calmel C, Lacombe D, et al. Mutations of the imprinted CDKN1C gene as a cause of the overgrowth Beckwith–Wiedemann syndrome: clinical spectrum and functional characterization. Hum Mutat. (2015) 36(9):894–902. doi: 10.1002/humu.22824
34. MacFarland SP, Duffy KA, Bhatti TR, Bagatell R, Balamuth NJ, Brodeur GM, et al. Diagnosis of Beckwith–Wiedemann syndrome in children presenting with Wilms tumor. Pediatr Blood Cancer. (2018) 65(10):e27296. doi: 10.1002/pbc.27296
35. Hoyme HE, Seaver LH, Jones KL, Procopio F, Crooks W, Feingold M. Isolated hemihyperplasia (hemihypertrophy): report of a prospective multicenter study of the incidence of neoplasia and review. Am J Med Genet. (1998) 79(4):274–8. doi: 10.1002/(SICI)1096-8628(19981002)79:4%3C274::AID-AJMG8%3E3.0.CO;2-M
Keywords: fetal Wilms tumor, prenatal diagnosis, isolated lateralized overgrowth, hemihypertrophy, Beckwith–Wiedemann syndrome, cancer, polyhydramnios
Citation: Bechara E, Saadé C, Geagea C, Charouf D and Abou Jaoude P (2024) Fetal Wilm's tumor detection preceding the development of isolated lateralized overgrowth of the limb: a case report and review of literature. Front. Pediatr. 12:1334544. doi: 10.3389/fped.2024.1334544
Received: 13 November 2023; Accepted: 16 February 2024;
Published: 18 March 2024.
Edited by:
Pierluigi Marzuillo, University of Campania Luigi Vanvitelli, Italy© 2024 Bechara, Saadé, Geagea, Charouf and Abou Jaoude. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Pauline Abou Jaoude cGF1bGluZS5hYm91amFvdWRlQGhkZi51c2ouZWR1Lmxi
Abbreviations WT: Wilms tumor; MRI, magnetic resonance imaging; CMN, congenital mesoblastic nephroma; WAGR, Wilms tumor, aniridia, genitourinary abnormalities, range of developmental delays; BWS, Beckwith–Wiedemann syndrome; ILO, isolated lateralized overgrowth; WG, weeks of gestation; CS, cesarean section; SIOP, international society of pediatric oncology; NWTS, national wilms tumor study; OS, overall survival.
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.