- 1Neonatologie, Universitätsklinikum Leipzig, Leipzig, Germany
- 2Kinder- und Jugendklinik, Universitätsmedizin Rostock, Rostock, Germany
Purpose: This study assesses whether peripherally inserted central venous catheters (PICC), impregnated with anti-infective drugs, reduce the rate of infections in neonates compared with unimpregnated catheters.
Methods: A retrospective analysis was conducted on electronic patient records of neonates born between August 2014 and May 2020, who had PICCs inserted, either standard (S-PICC) or with anti-infective drugs (A-PICC). Catheter-related bloodstream infections (CRBSI) were diagnosed based on clinical symptoms, laboratory results, and mentioning of infection in the patient record. Data on dwell time, mechanical ventilation, insertion site, maximum C-reactive protein (CRP) concentration, and anti-infective drug use were analyzed.
Results: A total of 223 PICCs were included. The infection rates were A-PICC (18.9%) and S-PICC (12.5%), which were not significantly different (p = 0.257). A-PICCs had significantly longer dwell times than S-PICCs (median 372 vs. 219 h, p = 0.004). The time to infection was not different between the groups (p = 0.3). There were also no significant differences in maximum CRP, insertion site abnormalities, or anti-infective drug use between the groups.
Conclusion: This retrospective study did not find a significant reduction in infection rates by using PICCs containing anti-infective drugs in neonates. Current antibiotic impregnations do not seem to be effective in preventing blood stream infections.
Introduction
Peripherally inserted central venous catheters (PICC) remain an important tool to ensure adequate parenteral nutrition and application of intravenous medications in small preterm infants for bridging the time until full enteral nutrition has been established (1–5). PICCs are introduced into peripheral veins—most commonly Vena saphena, Vena basilica, Vena cephalica, and the veins of the dorsal hand (1, 2, 6). They are advanced until the tip is located in one of the caval veins (2, 3, 7). Catheter-related blood stream infections (CRBSI) are a common and serious complication (5, 8).
PICCs impregnated with the anti-infective drugs rifampicin and miconazole were designed to reduce CRBSI (9). No recommendation has been made on their routine use in neonates due to a lack of evidence (10). Only the Infusion Nurse Society of the USA suggests using them in high-dependency patients (11). Flemmer et al. spoke at the 42nd conference of the German Society for Neonatology and Pediatric Intensive Care about a reduction in bacteriological complications (12). The multicenter randomized PREVAIL trial did not find a significant difference regarding infection rates (13). Similarly, a monocentric retrospective study at Doha (Qatar) including several catheter types did not find a benefit of anti-infective impregnated catheters regarding culture proven bloodstream infections (14). The following study made use of a large database of patient records accumulated at our university hospital to assess infection rates with PICCs impregnated or non-impregnated with anti-infective drugs in the clinical routine.
Methods
Setting
This study was conducted at the University of Leipzig Medical Center, a large tertiary care perinatal center in the German state of Saxony. Due to the retrospective nature of this study, neither treatment nor outcomes were influenced by this investigation. Anti-infective drug-incorporated PICCs (A-PICC, Premistar®, Vygon) were automatically identified from electronic patient records of all patients born between August 2014 and May 2020. This interval was chosen because both types of catheters were inserted during this time. An approximated number of the standard, i.e., drug-free, standard PICCs (S-PICC, Premicath®, Vygon) were randomly selected. PICC that were started during infection by other endovascular devices were excluded as were ambiguously documented PICC. We analyzed all microbiology studies taken during dwell time of the studied PICCs from catheter tips and blood cultures drawn peripherally.
Definition of CRBSI
Based on the National Health Safety Networks (NHSN) (7) our definition of CRBSI considered three criteria: (i) clinical symptoms, (ii) laboratory results, and (iii) mention of an infection in the patient record (15).
Any PICC was considered infected if two of these three criteria were positive within 7 days (7):
(I) Clinical symptoms were considered positive if at least five of the following eight parameters were present (7, 15, 16): (a) central body temperature >38.5°C, (b) difference between central and peripheral body temperature >2.5°C, (c) tachycardia >180 bpm, (d) bradycardia >6 alarms within 2 h, (e) FiO2 increase >20% from baseline, (d) SpO2 > 6 alarms within 2 h, (e) pathological alteration of skin color, and (f) prolonged capillary refill time >3 s. Of note, the values for subcriteria (a), (b), (c), and (e) are recorded in our electronic patient records as 15 min averages; they are considered to be positive if the threshold was passed at least once.
(II) Laboratory results were positive if either CRP > 10 mg/L or IL-6 > 200 ng/L.
(III) Documentation was positive if either a nurse or doctor logged an infection in the patient record at that time.
The time of the first positive criterion was regarded as the start of the infection.
Any documented site of infection other than the PICC including other CRBSI led to the exclusion of the infection according to this definition.
Data acquisition and statistics
Our hospital's Data Integration Center extracted the data from our electronic patient data management system. Additional data were extracted from the hospital information and quality assurance systems. We used the R software environment version 3.6 (17) with the packages survival (18), survminer (19), and ggpubr (20) for time-to-event analyses. If not stated otherwise, discrete variables were compared with Welch's two sample t-test or Wilcoxon's rank sum test if normality could not be assumed, categorical variables, with either χ² test, or, if the former was inappropriate, Fisher's exact test against the canonical α = 0.05. The results are reported as median [IQR] unless noted otherwise. Data collection and analysis were approved by the institutional review board of the medical faculty of the University of Leipzig (017/20-ek).
Results
Sample description
Patient data are listed in Table 1; of note, patients receiving A-PICCs on average had a lower weight (0.90 [0.68, 1.47]) than patients receiving S-PICCs (1.35 [0.87, 2.26], W = 2834.5, p < 0.001), and a lower gestational age (A-PICC 27 4/7 wks [25 3/7, 31 2/7] vs. S-PICC 30 3/7 wks [27 2/7, 34 6/7], W = 2878, p < 0.001). One patient died in the S-PICC group of fulminant sepsis, 16 days after the last PICC had been removed.

Table 1. Patient characteristics by catheter type used (comparisons are χ²-test for categorical and t-test for discrete variables).
Primary outcome
Catheter-related infections were found in 15.7% of the PICCs, but not less often in A-PICCs (18.9%) than in S-PICCs (12.5%) (χ2(1) = 1.285, p = 0.257). Furthermore, A-PICCs had a significantly longer dwell time (Figure 1A) (A-PICC median 372 h [95%CI: 291,399] vs. S-PICC 219 h [192,260], χ2(1) = 8.1, p = 0.004). The time to infection was not different between the two groups (Figure 1B) (p = 0.3).
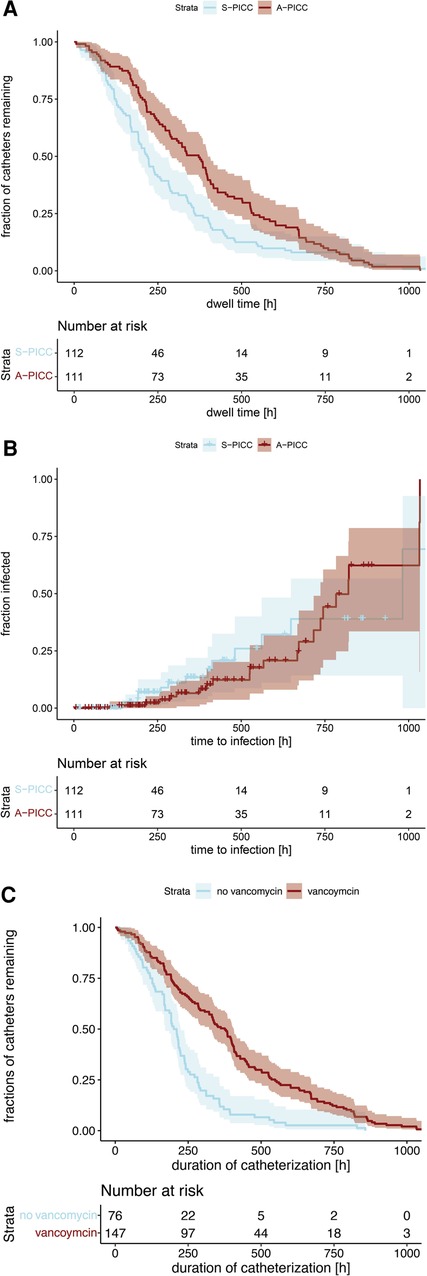
Figure 1. (A) Kaplan–Meier curves of catheter dwell time grouped by anti-infective incorporation. (A-PICC median 372 h [95%CI: 291,399] vs. S-PICC 219 h [192,260], χ2(1) = 8.1, p = 0.004). (B) Kaplan–Meier curves of time to infection. No difference was demonstrable regarding anti-infective incorporation (p = 0.3). (C) Kaplan–Meier curves of catheter dwell time grouped by use of vancomycin during dwell time. If vancomycin was used, PICCs had a significantly longer dwell time (with vancomycin 376 h [95%CI: 322,407], without 206 h [171,224], χ2(1) = 35.1, p < 0.001).
Catheter ingrowth
To prevent ingrowth, a slight retraction was performed in 31 PICCs during dwell time; there was no difference between the groups (p = 0.257) or presence of catheter infection (p = 0.287); but PICCs were described as difficult to remove in four cases, all of them had a catheter-related infection (p = 0.013).
Mechanical ventilation
Infants not requiring any ventilator support never met the catheter infection definition (p = 0.018). If infants without catheter infection needed mechanical ventilation, it was more often non-invasive than invasive (χ2(1) = 16.037, p < 0.001). Invasive ventilation was needed more often in A-PICC (χ2(1) = 5.874, p = 0.015) and in catheter infections (χ2(1) = 32.9, p < 0.001). The patients with S-PICCs had significantly more often no mechanical ventilation (χ2(1) = 7.227, p = 0.007).
Insertion site
Abnormalities, such as redness, at the insertion sites were not different between A-PICC and S-PICC (p = 0.773). Catheter locations were not different (see Table 2), but infections were more common in catheters inserted at the lower extremities (χ2(1) = 4.104, p = 0.043).
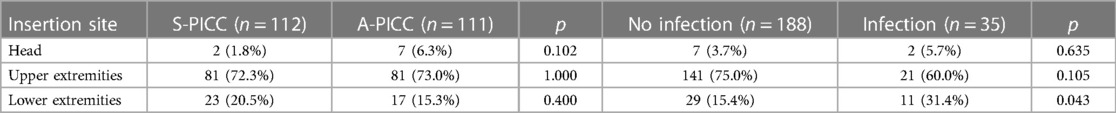
Table 2. Insertion site of catheters grouped by infection and anti-infective incorporation. Infection was more common in catheters in the lower extremities (χ2(1) = 4.104, p = 0.043).
Maximum CRP
Maximum CRP during dwell time was not different between the two groups (p = 0.410), but differed, as expected, between infected and uninfected PICCs (infected 74.36 mg/l [25.73, 104.00] vs. uninfected 5.99 mg/l [1.52, 33.48], W = 3766, p < 0.001).
Post-removal
Records were analyzed for a further 48 h after PICC removal. In infected catheters, SpO2 alarms were more common (χ2(1) = 12.757, p < 0.001), although notably no abnormalities in body temperature or skin color occurred. The maximum CRP after removal was above reference values, but not different between the two groups (A-PICC 13.59 mg/l [2.74, 35.43] vs. S-PICC 5.63 mg/l [1.85, 13.70], W = 885.5, p = 0.089). A higher post-removal CRP was seen in those meeting our infection definition (infected 20.18 mg/l [5.23, 33.10], uninfected 5.95 mg/l [1.57, 17.12], W = 797, p = 0.005).
Microbiology
Peripheral blood cultures were collected more often in cases with infected PICC (infection 48.6% vs. no infection 22.5%, χ2(1) = 9.007, p = 0.003); 7 of 23 cultures in patients with S-PICCs vs. 11 of 36 A-PICCs returned positive, so did 12 of 42 of those with no infection vs. 6 of 17 of the infected; Klebsiella oxytoca, Lactobacillus fermentum, Enterobacter cloacae, Klebsiella pneumoniae, Bacillus subtilis, Bacillus cereus, and Serratia marcescens were found. Skin flora, namely, Staphylococcus epidermidis, Staphylococcus haemolyticus, and Staphylococcus hominis, were significantly more often detected on infected catheters (p = 0.005). Candida antigen studies were not different between the groups.
Anti-infective drugs and resistance: Table 3 sums up aniinfective drug use in relation to the outcomes. In S-PICC, there was significantly more often no anti-infective treatment (A-PICC 5.4% untreated vs. S-PICC 17.9%, χ2(1) = 7.227, p = 0.007). All PICCs meeting the infection definition received anti-infective drugs (infection 100% vs. no infection 86.2%, p = 0.018). Five or more anti-infective drugs were used more often in A-PICC (A-PICC 27.0% vs. S-PICC 11.6%, χ2(1) = 7.555, p = 0.006) and in infected PICCs (infection 57.1% vs. no infection: 12.2%, χ2(1) = 35.403, p < 0.001).
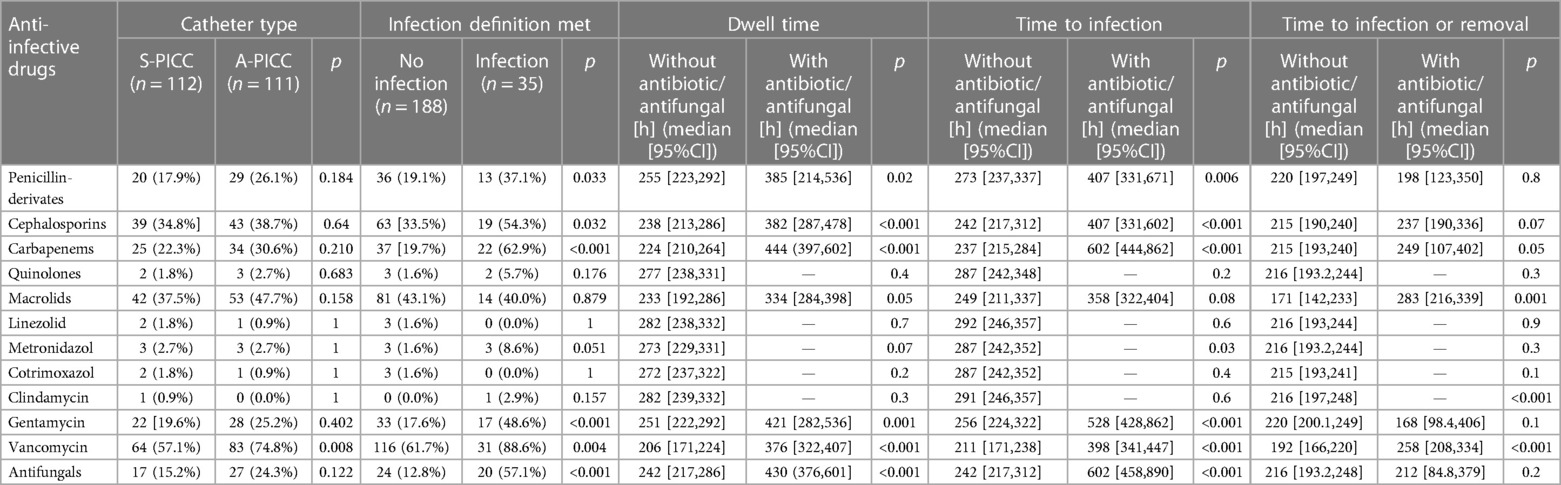
Table 3. Use of antibiotics by either catheter type and infection and comparisons of dwell times, times to infection, and time to infection or removal with and without anti-infectives.
Penicillin-derivatives (Ampicillin, Ampicillin + Sulbactam, Piperacillin + Tazobactam) were used significantly more often in infections (infection 37.1% vs. no infection 19.1%, χ2(1) = 4.572, p = 0.032) as were cephalosporins (54.3% vs. 33.5%, χ2(1) = 4.620, p = 0.032), gentamicin (48.6% vs. 17.6%, χ2(1) = 14.587, p < 0.001), and antimycotics (57.1% vs. 12.8%, χ2(1) = 33.941, p < 0.001).
Vancomycin was used more often in A-PICC (A-PICC 74.8% vs. S-PICC 57.1%, χ2(1) = 6.950, p = 0.008). It was used in all but four cases of infection. If vancomycin was used, PICCs had a significantly longer dwell time (Figure 1C) (with vancomycin median 376 h [95%CI: 322,407], without 206 h [171,224], χ2(1) = 35.1, p < 0.001).
Only in three A-PICCs bacteria resistant to the incorporated rifampin were found (p = 0.049). The resistance to miconazole was not studied.
Discussion
In this retrospective analysis, infection rates in A-PICC and S-PICC were similar, with a slight trend toward A-PICC. This is in line with the PREVAIL (13) trial and studies by Klemme et al. (21) and Bayoumi et al. (14). None of them detected differences between A-PICC and S-PICC. If the slight trend toward more infections with A-PICC was indeed an effect, the number needed to harm in our population would result in a high value of 15.58. In contrast, catheters incorporating minocycline and rifampicin had been found to have less infections in older children (22).
Flemmer et al. (12) were the only ones to report a reduction in bacteriological complications by A-PICCs in neonates and preterm infants. Unlike their study, bacterial colonization was not tested in every PICC included in our analysis.
The dwell times of PICCs were considerably longer than reported in the literature (our study 12.3 days, other studies 8.2 and approximately 6.5 days) (13, 21). There were no rules prescribing maximum dwell times in our unit. While the removal of infected PICCs was more often complicated, no new methods—as described by Van Mechelen and Mahieu (23)—or surgery were needed.
Positive cultures were not mandatory for defining an infection, because otherwise many infections would be missed owing to the low yield of blood cultures (24). To maintain specificity, we mandated more clinical and laboratory signs to be present during a fixed time interval as suggested by our national German guideline for counting an episode as an infection (15). We feel this is justified as the decision to start anti-infective treatment is in clinical routine often based on clinical criteria and laboratory results like CRP and interleukin 6. Their combination has been ascribed a high positive predictive value (15). Waiting for microbiology results would delay treatment unacceptably. We relinquished from assessing procalcitonin (PCT) since IL-6 and CRP have been shown to be superior (25, 26).
Preterms with lower birth weights are at an increased risk of late-onset sepsis (LOS) (27) and CRBSI (28); longer dwell times have been associated with a rise in sepsis (27) and CRBSI (28, 29) risks. Interestingly, changing central lines after a fixed time is not recommended, because the replacement line has the same infection risk (29).
Catheter tips were not routinely sent for cultures and documentation of catheter removal is often scant. Any discussion on the microbiology from catheter tips must be interpreted after taking into account their unclear role in the diagnosis of CRBSI (30) and the scarcity of studies included in this analysis. Catheter tip cultures were not assigned a role in the definition of infection by their results or in clinical practice. Arguments have been put forward to forgo them altogether (31). Only with a synchronously drawn blood culture might their results be interpretable (30). Our data are hence not suitable for deriving clear statements on colonization. Staphylococcus epidermidis was the most common bacterial species identified at the catheter tip with no difference between PICCs considered infected vs. not infected. Such coagulase-negative staphylococci (CNS) have been described as most abundant there (32) and as a relevant cause of LOS (27) and CRBSI (33) in the literature.
Vancomycin is the drug of choice for CNS infection in the preterm; Rodriguez-Guerineau et al. found it especially effective if combined with rifampicin (16). In our department, vancomycin is being used frequently as an anti-infective prophylaxis if catheters are used beyond a certain dwell time. We were unable to distinguish such prophylaxis from actual treatments in our data; it is thus unclear whether prophylaxis actually prevented infection.
We identified multiple limitations in our data. First, the retrospective design: since the decision between A-PICC and S-PICC was made by the physician inserting it, a sum of subjective judgements may have caused A-PICCs being used in children weighing less and having a lower gestational age, possibly because A-PICCs might have been attributed increased safety for longer dwell times, which might have been considered especially advantageous in smaller infants. Since these differences may be confounding variables, some findings in our data may be difficult to interpret.
A-PICCs in our study were inserted in patients weighing significantly less, but were also used longer. The trend toward slightly higher infection rates in the A-PICC group may hence be rather due to the infection risk being generally higher in more immature infants.
Due to low numbers of infection in both groups, estimated power is 25.04%, which is too low to accept the Null Hypothesis, i.e., to demonstrate practical equivalence. A-PICCs and S-PICCs were inserted in patients different in weight and gestational age to which the literature ascribes different CRBSI risks. This lowers the informative value significantly, especially with regard to comparability with other studies. Our infection definition hinges on infection symptoms and laboratory studies, which we feel is representative of clinical practice. The number of infections in our study would have been severely reduced if we had kept to the definition by NHSN demanding a positive blood culture, because our department orders few microbiological studies. It seems unlikely that in our case more microbiological studies would have helped increase the number of infections since only 4%–12% return positive (34–37). Missing a large number of infections, however, would also not improve the conclusions. Of note, there is a high variability in peripheral venous blood culture positive rates: Klemme et al. had no positive blood cultures, whereas the PREVAIL trial reported 61.3%. Limitation of the analysis to catheter dwell time (plus 48 h) and exclusion if any other site of infection had been documented was further reducing the number of infections. Despite all of these shortcomings, we feel that our definition is very close to what is happening in the clinical routine, where catheter infection is most often diagnosed by ruling out other explanations, and antibiotic treatment is started long before microbiology results are available.
If our observations were used to estimate the sample size of a trial with an ɑ = 0.05 and a power of 90%, about 700 PICCs would be needed in both arms.
Finally, because of the few infections in our study, a clear benefit from repeatedly retracting longer-dwelling PICCs was not discernible.
In spite of these limitations, this manuscript is a description of the current use and complications of PICCs in our department with an infection definition that resembles routine rather than an artificial study environment.
Conclusion
In line with other studies on this topic, we could find no clear advantage regarding infections by incorporating anti-infective drugs into peripherally inserted central catheters.
If one were still to stipulate more evidence, for deciding whether to use A-PICCs, a larger trial would be required.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
This retrospective analysis was carried out in accordance with state law (Sächsisches Krankenhausgesetz §29). In addition, the institutional review board of the University of Leipzig waived informed consent (internal reference number 017/20-ek).
Author contributions
JK: Data curation, Formal analysis, Investigation, Visualization, Writing – original draft, review & editing. RGA: Conceptualization, Formal analysis, Methodology, Software, Supervision, Validation, Visualization, Writing – review & editing. UHT: Conceptualization, Funding acquisition, Project administration, Resources, Supervision, Writing – review & editing. FP: Conceptualization, Funding acquisition, Methodology, Supervision, Writing – review & editing.
Acknowledgments
For preparing patient data, we used services of our Hospital’s Data Integration Center, funded by the German Federal Ministry of Education and Research (Grant No 01ZZ1803D).
Conflict of interest
UHT and FP received an unrestricted grant of 4,500€ by Vygon GmbH und Co. KG.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
A-PICC, anti-infective drugs-incorporated PICC (rifampicin/miconazole); BPD, bronchopulmonary dysplasia; CNS, coagulase-negative staphylococci; CRBSI, catheter-related blood stream infection; CRIB, Clinical Risk Index for Babies; CRP, C-reactive protein; FiO2, fraction of inspired oxygen; IQR, interquartile range; IVH, intraventricular hemorrhage; NEC, necrotizing enterocolitis; PICC, peripherally inserted central catheter; RDS, respiratory distress syndrome; SD, standard deviation; SIP, spontaneous intestinal perforation; S-PICC, standard PICC, drug-free; SpO2, pulseoxymetric oxygen saturation.
References
1. Westergaard B, Classen V, Walther-Larsen S. Peripherally inserted central catheters in infants and children—indications, techniques, complications and clinical recommendations: PICCs in children. Acta Anaesthesiol Scand. (2013) 57(3):278–87. doi: 10.1111/aas.12024
2. Stocker M, Berger TM. Arterielle und zentralvenöse Katheter bei Neugeborenen und Säuglingen. Anaesthesist. (2006) 55(8):873–82. doi: 10.1007/s00101-006-1052-1
3. Colacchio K, Deng Y, Northrup V, Bizzarro MJ. Complications associated with central and non-central venous catheters in a neonatal intensive care unit. J Perinatol. (2012) 32(12):941–6. doi: 10.1038/jp.2012.7
4. Vachharajani AJ, Vachharajani NA, Morris H, Niesen A, Elward A, Linck DA, et al. Reducing peripherally inserted central catheters in the neonatal intensive care unit. J Perinatol. (2017) 37(4):409–13. doi: 10.1038/jp.2016.243
5. Balain M, Oddie SJ, McGuire W. Antimicrobial-impregnated central venous catheters for prevention of catheter-related bloodstream infection in newborn infants. Cochrane Database Syst Rev. (2015) 2015(9):CD011078. doi: 10.1002/14651858.CD011078.pub2
6. Erhard DM, Nguyen S, Guy KJ, Casalaz DM, König K. Dwell times and risk of non-elective removal of 1-French peripherally inserted central catheters according to catheter tip position in very preterm infants. Eur J Pediatr. (2017) 176(3):407–11. doi: 10.1007/s00431-017-2854-7
7. National Healthcare Safety Network (CDC). 4. Bloodstream infection event (central line-associated bloodstream infection and non-central line associated bloodstream infection). In: NHSN Patient Safety Component Manual. Washington, D. C., USA (2022). p. 4-1-4-48. Available from: https://www.cdc.gov/nhsn/pdfs/pscmanual/pcsmanual_current.pdf (accessed December 11, 2022).
8. McMullan RL, Gordon A. Antibiotics at the time of removal of central venous catheter to reduce morbidity and mortality in newborn infants. Cochrane Database Syst Rev. (2018) 3(3):CD012181. doi: 10.1002/14651858.CD012181.pub2
9. VYGON. Pädiatrie und Neonatologie. Pädiatrische Venenverweilkatheter. Premistar®. Größter Schutz für kleine Leben (2016). Available at: https://www.vygon.de/download/689/PDF-Paediatrische_Venenkatheter.pdf (Accessed January 8, 2022).
10. Kommission für Krankenhaushygiene und Infektionsprävention (KRINKO) beim Robert Koch-Institut. Prävention von Gefäßkatheter-assoziierten Infektionen bei Früh- und Neugeborenen: Empfehlung der Kommission für Krankenhaushygiene und Infektionsprävention (KRINKO) beim Robert Koch-Institut. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz. (2018) 61(5):608–26. doi: 10.1007/s00103-018-2718-y
11. Infusion Nurse Society. Infusion therapy standards of practice updates (2021). Available at: https://journals.lww.com/10.1097/NAN.0000000000000436 (Accessed September 29, 2023).
12. Flemmer AW, DeMaio N, Schubert S, Wurster T, Klemme M. FV56. Effekt einer antiinfektösen Katheterbeschichtung von Einschwemmkathetern auf Katheterkolonisierung und Sekundärsepsis bei Frühgeborenen. In: Abstracts der 42. Jahrestagung der Gesellschaft für Neonatologie und Pädiatrische Intensivmedizin. Monatsschr Kinderheilkd. (2016) 164(Suppl 2):S145. doi: 10.1007/s00112-016-0091-1
13. Gilbert R, Brown M, Rainford N, Donohue C, Fraser C, Sinha A, et al. Antimicrobial-impregnated central venous catheters for prevention of neonatal bloodstream infection (PREVAIL): an open-label, parallel-group, pragmatic, randomised controlled trial. Lancet Child Adolesc Health. (2019) 3(6):381–90. doi: 10.1016/S2352-4642(19)30114-2
14. Bayoumi MAA, Van Rens MFPT, Chandra P, Masry A, D’Souza S, Khalil AM, et al. Does the antimicrobial-impregnated peripherally inserted central catheter decrease the CLABSI rate in neonates? Results from a retrospective cohort study. Front Pediatr. (2022) 10:1012800. doi: 10.3389/fped.2022.1012800
15. Zemlin M, Gille C, Härtel C, Küster H, Müller A, Pohlandt F, et al. AWMF Leitlinie 024-008—Bakterielle Infektionen bei Neugeborenen. Available at: https://register.awmf.org/assets/guidelines/024-008l_S2k_Bakterielle_Infektionen_Neugeborene_2021-03.pdf (Accessed January 8, 2022).
16. Rodriguez-Guerineau L, Salvia-Roigés MD, León-Lozano M, Rodríguez-Miguélez JM, Figueras-Aloy J. Combination of vancomycin and rifampicin for the treatment of persistent coagulase-negative staphylococcal bacteremia in preterm neonates. Eur J Pediatr. (2013) 172(5):693–7. doi: 10.1007/s00431-012-1927-x
17. R Core Team. R: A Language and Environment for Statistical Computing. Vienna, Austria (2022). Available at: https://www.R-project.org/ (Accessed January 02, 2023).
18. Therneau T. A package for survival analysis in R. R package version 3.2-11 (2021). Available at: https://CRAN.R-project.org/package=survival (Accessed January 2, 2023).
19. Kassambara A, Kosinski M, and Biecek P. survminer: drawing survival Curves using “ggplot2”. R package version 0.4.9 (2021). Available at: https://CRAN.R-project.org/package=survminer (Accessed January 2, 2023).
20. Kassambara A. ggpubr: “ggplot2” based publication ready plots. R package version 0.4.0. 2020. Available at: https://CRAN.R-project.org/package=ggpubr (Accessed January 2, 2023)
21. Klemme M, Staffler A, De Maio N, Lauseker M, Schubert S, Innocenti P, et al. Use of impregnated catheters to decrease colonization rates in neonates—a randomized controlled pilot trial. J Neonatal Perinat Med. (2020) 13(2):231–7. doi: 10.3233/NPM-190273
22. Gilbert RE, Mok Q, Dwan K, Harron K, Moitt T, Millar M, et al. Impregnated central venous catheters for prevention of bloodstream infection in children (the CATCH trial): a randomised controlled trial. Lancet. (2016) 387(10029):1732–42. doi: 10.1016/S0140-6736(16)00340-8
23. Van Mechelen K, Mahieu L. A new technique for difficult removal of a peripherally inserted central venous catheter (PICC) in a neonate. Eur J Pediatr. (2021) 180(3):973–6. doi: 10.1007/s00431-020-03797-z
24. Guang CX, Sheng ZS, Yan WC, Jin K, Dong MH. The diagnostic value of next-generation sequencing technology in sepsis. Front Cell Infect Microbiol. (2022) 12:899508. doi: 10.3389/fcimb.2022.899508
25. Franz AR, Kron M, Pohlandt F, Steinbach G. Comparison of procalcitonin with interleukin 8, C-reactive protein and differential white blood cell count for the early diagnosis of bacterial infections in newborn infants. Pediatr Infect Dis J. (1999) 18(8):666. doi: 10.1097/00006454-199908000-00003
26. Chiesa C, Pellegrini G, Panero A, Osborn JF, Signore F, Assumma M, et al. C-reactive protein, interleukin-6, and procalcitonin in the immediate postnatal period: influence of illness severity, risk status, antenatal and perinatal complications, and infection. Clin Chem. (2003) 49(1):60–8. doi: 10.1373/49.1.60
27. Stoll BJ, Hansen N, Fanaroff AA, Wright LL, Carlo WA, Ehrenkranz RA, et al. Late-onset sepsis in very low birth weight neonates: the experience of the NICHD neonatal research network. Pediatrics. (2002) 110(2):285–91. doi: 10.1542/peds.110.2.285
28. Hu Y, Ling Y, Ye Y, Zhang L, Xia X, Jiang Q, et al. Analysis of risk factors of PICC-related bloodstream infection in newborns: implications for nursing care. Eur J Med Res. (2021) 26(1):80. doi: 10.1186/s40001-021-00546-2
29. Buetti N, Marschall J, Drees M, Fakih MG, Hadaway L, Maragakis LL, et al. Strategies to prevent central line-associated bloodstream infections in acute-care hospitals: 2022 update. Infect Control Hosp Epidemiol. (2022) 43(5):553–69. doi: 10.1017/ice.2022.87
30. Miller JM, Binnicker MJ, Campbell S, Carroll KC, Chapin KC, Gilligan PH, et al. A guide to utilization of the microbiology laboratory for diagnosis of infectious diseases: 2018 update by the Infectious Diseases Society of America and the American Society for Microbiology. Clin Infect Dis. (2018) 67(6):e1–94. doi: 10.1093/cid/ciy381
31. Peterson LR, Smith BA. Nonutility of catheter tip cultures for the diagnosis of central line-associated bloodstream infection. Clin Infect Dis. (2015) 60(3):492–3. doi: 10.1093/cid/ciu845
32. Bouza E, San Juan R, Muñoz P, Pascau J, Voss A, Desco M. A European perspective on intravascular catheter-related infections: report on the microbiology workload, aetiology and antimicrobial susceptibility (ESGNI-005 study). Clin Microbiol Infect. (2004) 10(9):838–42. doi: 10.1111/j.1469-0691.2004.00936.x
33. Graham PL, Begg MD, Larson E, Della-Latta P, Allen A, Saiman L. Risk factors for late onset gram-negative sepsis in low birth weight infants hospitalized in the neonatal intensive care unit. Pediatr Infect Dis J. (2006) 25(2):113–7. doi: 10.1097/01.inf.0000199310.52875.10
34. Marks L, Waal K, Ferguson JK. Time to positive blood culture in early onset neonatal sepsis: a retrospective clinical study and review of the literature. J Paediatr Child Health. (2020) 56(9):1371–5. doi: 10.1111/jpc.14934
35. Mintz A, Mor M, Klinger G, Scheuerman O, Pirogovsky A, Sokolover N, et al. Changing epidemiology and resistance patterns of pathogens causing neonatal bacteremia. Eur J Clin Microbiol Infect Dis. (2020) 39(10):1879–84. doi: 10.1007/s10096-020-03921-9
36. Bromiker R, Elron E, Klinger G. Do neonatal infections require a positive blood culture? Am J Perinatol. (2020) 37(S 02):S18–21. doi: 10.1055/s-0040-1714079
Keywords: neonate, preterm, central line, catheter-related infections, medicated catheters
Citation: Koppitz J, Ascherl RG, Thome UH and Pulzer F (2024) Incorporating anti-infective drugs into peripherally inserted catheters does not reduce infection rates in neonates. Front. Pediatr. 11:1255492. doi: 10.3389/fped.2023.1255492
Received: 8 July 2023; Accepted: 29 November 2023;
Published: 5 January 2024.
Edited by:
Suksham Jain, Government Medical College and Hospital, IndiaReviewed by:
Tuuli Metsvaht, University of Tartu, EstoniaMohammad A. A. Bayoumi, Hamad Medical Corporation, Qatar
Matheus (Roland) Van Rens, Hamad Medical Corporation, Qatar
© 2024 Koppitz, Ascherl, Thome and Pulzer. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Rudolf Georg Ascherl YXNjaGVybEBtZWRpemluLnVuaS1sZWlwemlnLmRl
†These authors have contributed equally to this work and share first authorship
 Julia Koppitz
Julia Koppitz Rudolf Georg Ascherl
Rudolf Georg Ascherl Ulrich Herbert Thome
Ulrich Herbert Thome Ferdinand Pulzer
Ferdinand Pulzer