- 1Department of Paediatric Medicine, KK Women's and Children's Hospital, Singapore, Singapore
- 2Duke-NUS Medical School, Singapore, Singapore
- 3Department of Emergency Medicine, KK Women's and Children's Hospital, Singapore, Singapore
- 4Centre for Quantitative Medicine, Duke-NUS Medical School, Singapore, Singapore
- 5Department of Paediatric Subspecialties, Children's Intensive Care, KK Women's and Children's Hospital, Singapore, Singapore
Introduction: Fear of missed serious bacterial infections (SBIs) results in many febrile young infants receiving antibiotics. We aimed to compare the time to antibiotics between infants with SBIs and those without.
Materials and Methods: We recruited febrile infants ≤ 90 days old seen in the emergency department (ED) between December 2017 and April 2021. SBI was defined as (1) urinary tract infection, (2) bacteremia or (3) bacterial meningitis. We compared the total time (median with interquartile range, IQR) from ED arrival to infusion of antibiotics, divided into (i) time from triage to decision for antibiotics and (ii) time from decision for antibiotics to administration of antibiotics.
Results: We analyzed 81 and 266 infants with and without SBIs. Median age of those with and without SBIs were 44 (IQR 19–72) and 29 (IQR 7–56) days, respectively (p = 0.002). All infants with SBIs and 168/266 (63.2%) infants without SBIs received antibiotics. Among 249 infants who received antibiotics, the median total time from ED arrival to infusion of antibiotics was 277.0 (IQR 236.0–385.0) mins for infants with SBIs and 304.5 (IQR 238.5–404.0) mins for those without (p = 0.561). The median time to decision for antibiotics was 156.0 (IQR 115.0–255.0) mins and 144.0 (IQR 105.5–211.0) mins, respectively (p = 0.175). Following decision for antibiotics, infants with SBIs received antibiotics much faster compared to those without [107.0 (IQR 83.0–168.0) vs. 141.0 (94.0–209.5) mins, p = 0.017].
Conclusion: There was no difference in total time taken to antibiotics between infants with SBIs and without SBIs. Both recognition and administration delays were observed. While all infants with SBIs were adequately treated, more than half of the infants without SBIs received unnecessary antibiotics. This highlights the challenge in managing young febrile infants at initial presentation, and demonstrates the need to examine various aspects of care to improve the overall timeliness to antibiotics.
Introduction
Serious bacterial infections (SBIs) are a preventable source of infant mortality, with the majority of these deaths attributed to sepsis, meningitis and pneumonia (1–3). Although most of these deaths are in low- to middle-income countries (3), sepsis alone still accounts for an estimated 11% of young infant mortality in high-income countries (4). Survivors develop disabilities such as cerebral palsy, impaired growth, and suffer from cognitive deficits (5–8).
Recognizing that SBIs have potentially devastating consequences, clinicians have developed guidelines for the optimal management of sepsis, pneumonia and urinary tract infection (UTI), where judicious and timely administration of antimicrobial therapy is critical (9–11). The Surviving Sepsis Campaign (SSC) International Guidelines recommend starting antibiotics as soon as possible, within 1 h of recognizing a child with septic shock, or within 3 h of recognizing sepsis-associated organ dysfunction without shock (9). Early administration of antibiotics, has been demonstrated to reduce subsequent resource utilization, including progression to organ dysfunction, hospital length of stay (LOS), and mortality (12, 13).
Young infants <90 days old with SBIs pose a diagnostic challenge to emergency department (ED) physicians and pediatricians. Algorithms such as the Rochester criteria, Boston criteria and the Philadelphia criteria were developed to guide physicians to differentiate between high and low-risk infants (14–16). Newer protocols like the “Step-by-Step” approach and the Pediatric Emergency Care Applied Research Network (PECARN) rule were derived to define low-risk febrile infants and were found to have a higher sensitivity and negative predictive value than the RC (17, 18). Although the newer algorithms focus on defining a low-risk group, these protocols are not used systematically in clinical practice (19–21).
Due to fear of missed SBIs, there is a low threshold to perform invasive investigations and administer empirical antibiotics to most febrile infants. While this ensures that clinicians err on the side of caution, the suboptimal specificities (46.9–60.0%) of the most recent risk stratification tools mean that febrile infants without SBIs are also extensively investigated and treated (17, 18). This may result in delays in antibiotic administration, especially considering the busy nature of the emergency department, where overcrowding and resource management can challenge the provision of timely optimal care.
Delays in time-to-antibiotics are divided into 2 categories: First, time from arrival in the ED to time of decision to administer antibiotics, which requires prompt recognition of a child with SBI and measures “recognition delay”. Second, time from decision to administer antibiotics to time of actual administration of first dose antibiotics, which measures “administration delay” in the delivery of antibiotics (22). Both delays are associated with increased mortality (22–24). Studying these 2 key time points would allow targeted interventions to improve clinical outcomes.
Our primary aim was to compare the timing of antibiotic administration between young infants with SBIs and those without, divided into: (i) total time from ED arrival to infusion of antibiotics, (ii) time from arrival in the ED to time of decision to administer antibiotics (“recognition delay”), and (iii) time from decision to administer antibiotics to time of first dose antibiotics administered (“administration delay”). We hypothesized that young infants with SBIs would have an overall shorter time to antibiotics and have both shorter recognition and administration delays, compared to infants without SBIs.
Article Types
Tier 1 article: Original Research.
Materials and Methods
Study Design, Setting and Population
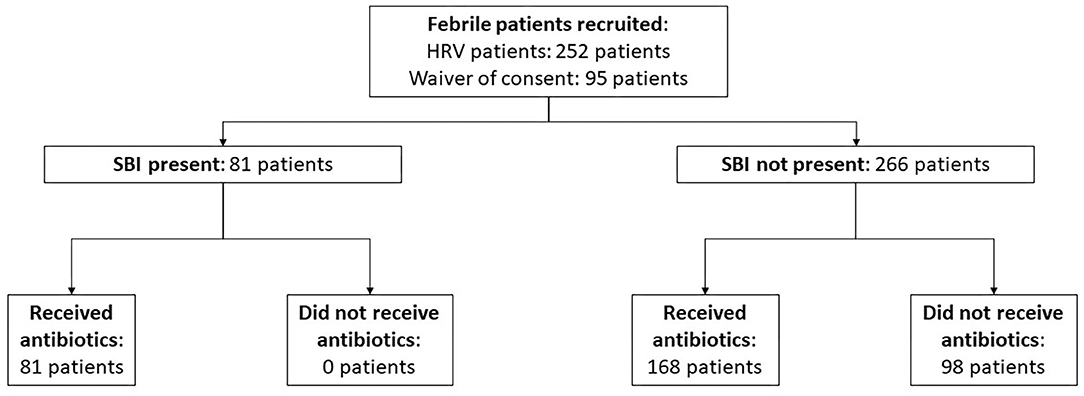
We performed a prospective cohort study among infants ≤ 90 days old, with fever, who attended the ED of KK Women's and Children's Hospital (KKH), Singapore between December 2017 and April 2021. The initial cohort was a secondary analysis of infants recruited for a heart rate variability study, from December 2017 to December 2020 (25). Subsequently, we obtained ethics approval to study all febrile infants from January to April 2021 (Figure 1). KKH is one of the two major tertiary centers for pediatric care in Singapore.
We excluded premature infants with a gestation of <35weeks, infants with major congenital malformations, chronic respiratory conditions requiring home non-invasive support or perinatal conditions requiring at least 7 days of stay in the neonatal intensive care unit prior to this hospitalization. We chose to exclude these because they formed a high-risk population that would automatically receive urgent attention in the ED and inpatient services and the approach is different from other febrile infants. We also excluded infants who received antibiotics in the last 48 h before presenting to the ED, and infants who were discharged against medical advice from the ED.
In our institution, infants ≤ 90 days old who are clinically unwell or remain febrile (temperature ≥38.5°C or ≥38.0°C on 2 occasions) will receive a full septic work up including full blood count, C-reactive protein (CRP) and procalcitonin, blood, urine and cerebrospinal fluid (CSF) cultures. They will also be started on empirical intravenous antibiotics while awaiting culture growth. For infants who remain afebrile or do not reach the fever criteria during the hospital stay, a urinalysis is ordered. Subsequent investigations and decision for antibiotics are physician-dependent. The decision for antibiotics in these cases is based on the clinical status as well as initial biochemical results including CRP and procalcitonin. All infants hospitalized for fever are monitored until they are well and afebrile for 24 h before discharge.
Study Definitions
Fever was defined as an axillary or rectal temperature of ≥38.0°C. SBI was defined as (1) UTI, (2) bacteremia or (3) bacterial meningitis (26). UTI was defined as growth of a single pathogen of (1) >100,000 colony-forming unit (CFU)/ml in a clean catch specimen, or (2) ≥50,000 CFU/ml in a catheterized specimen, or (3) 10,000–50,000 CFU/ml in a catheterized specimen with an abnormal urinalysis (positive for leucocyte esterase, nitrite or >5 white blood cell/per high-powered-field). Bacteremia was defined as growth of a single pathogen in blood culture. When the bacteria was considered to be a likely contaminant, for example coagulase-negative staphylococcus, it was excluded. Bacterial meningitis was defined as (1) growth of a single pathogen in the CSF, or (2) sterile CSF pleocytosis with biochemical changes consistent with bacterial meningitis where CSF protein is more than 100 mg/dL and CSF to plasma glucose ratio <0.6 (27, 28). Invasive bacterial infection (IBI) is defined as bacterial meningitis and/or bacteremia (29).
Data Collection
Data on baseline characteristics was collected prospectively from medical records and entered into a structured data entry form. We collected data on age, sex, presence of co-morbidities, prematurity status and vital signs at the ED.
Laboratory investigations included full blood count, CRP, procalcitonin and blood, urine and CSF cultures.
Outcome Measures
We computed the time taken to antibiotics, defined as total time from ED triage to the administration of the first dose of antibiotics. The timing was further divided into (i) time taken from arrival in the ED to decision to administer antibiotics (“recognition delay”), and (ii) time between the decision to administer antibiotics to time which the first dose of antibiotics was administered (“administration delay”). Time stamps were collected from the electronic health record by a study team member who was blinded to the SBI status of the study subjects. The recognition delay time stamp was taken as the time the attending physician reviewed the infant and made the decision for antibiotics, as captured by the physician's consult note in the electronic health record. In cases where the attending physician made a note to monitor for a fever spike before starting antibiotics, the time of the fever spike was used as the recognition delay time stamp. We also computed the need for fluid resuscitation, use of inotropes, admission to the high dependency unit (HDU) or intensive care unit (ICU), total hospital LOS, and mortality.
Statistical Analyses
Categorical data for baseline demographics, clinical characteristics and laboratory results were summarized as frequency counts and percentages and compared between study groups using a chi-square test; continuous data was summarized as median (interquartile range, IQR) and compared between study groups using a Wilcoxon rank-sum test. The Wilcoxon rank sum test was used to compare time to antibiotics between study groups with differences reported using the Hodges-Lehmann shift estimate (median of all possible differences) with 95% confidence interval. Statistical significance was set at p ≤ 0.05. We performed a sensitivity analysis defining SBIs as those with culture positive growth, thereby excluding those who were diagnosed with meningitis based on CSF pleocytosis with biochemical changes. We also performed a subgroup analysis on neonates (<28 days old). Analysis was performed using SPSS v26 (Chicago) (30) and SAS v9.4 (SAS Inc. Cary NC) (31).
Results
Patient Demographics and Clinical Characteristics
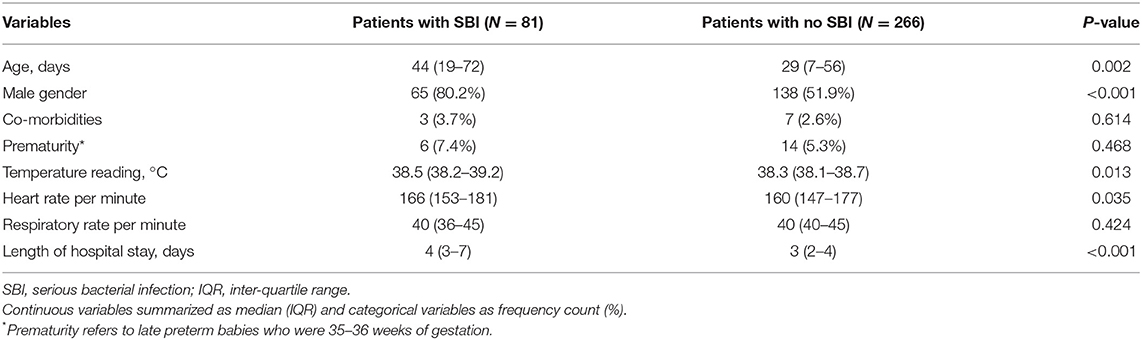
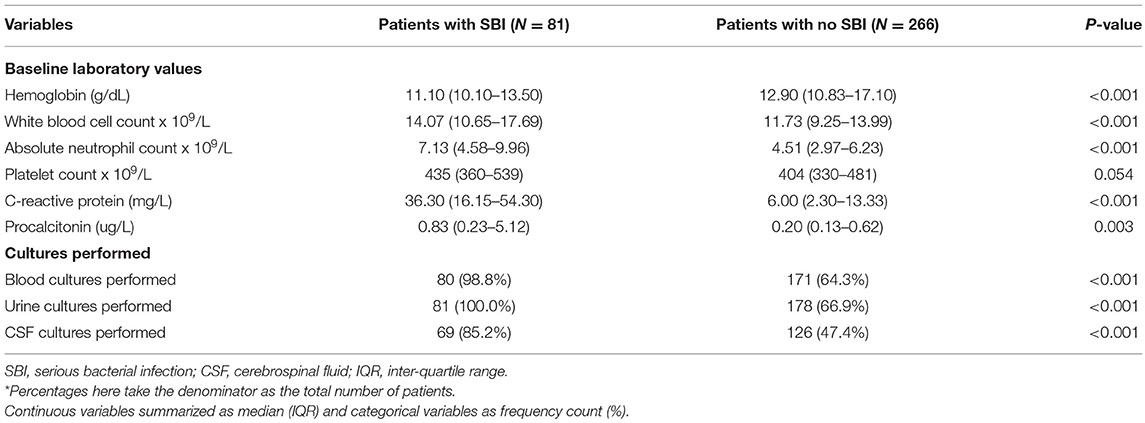
Among 347 patients analyzed, 81 infants (23.3%) had SBIs and 266 infants (76.7%) did not have SBIs. There were 98 neonates, 28 (28.6%) had SBIs and 70 (71.4%) did not have SBIs. Infants with SBIs were older (median 44 days, IQR 19–72 vs. 29 days, IQR 7–56, p = 0.002), more likely to be males (80.2% vs. 51.9%, p <0.001), had a higher temperature (median 38.5°C, IQR 38.2–39.2 vs. 38.3°C, IQR 38.1–38.7, p = 0.013), and had a higher heart rate (median 166/min, IQR 153–181 vs. 160/min, IQR 147–177, p = 0.035) than infants without SBIs (Table 1). Among the infants who received blood investigations, infants in the SBI group had higher median inflammatory markers like white blood cell count, absolute neutrophil count, CRP and procalcitonin (Table 2).
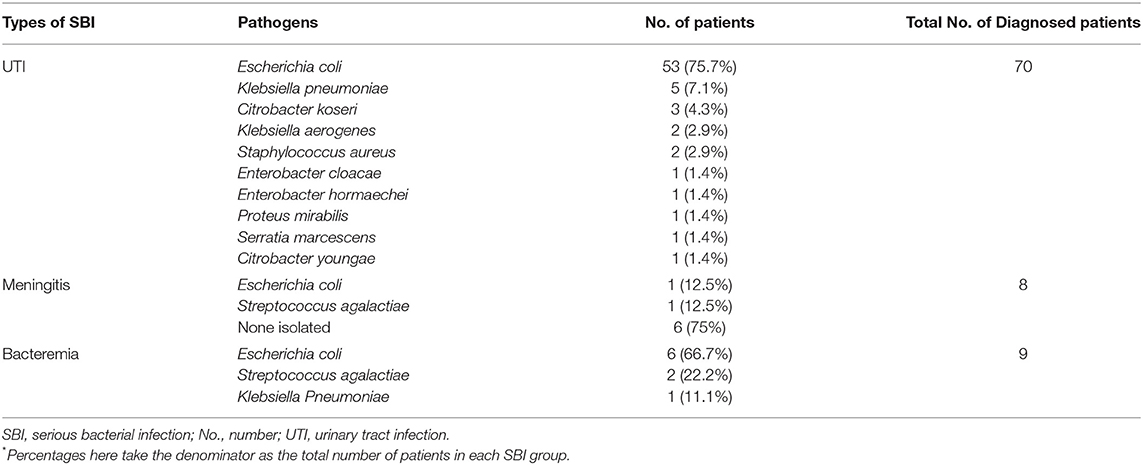
Among 81 infants with SBIs, 70 (86.4%) infants were diagnosed with a UTI. Among 15 (4.3% of all febrile infants) infants with IBI, 8 (53.3%) had the diagnosis of meningitis and 9 (60%) had bacteremia (Table 3). Six out of eight (75%) of the infants with CSF culture negative bacterial meningitis were diagnosed by CSF biochemical changes. All of them had their lumbar puncture done prior to receiving antibiotics. All the infants had their CSF sent for culture and filmarray for various viruses and bacteria including E. coli, Haemophilus influenzae and Streptococcus agalactiae. Six patients had multiple-source infections (2 had bacteremia and meningitis while 4 had UTI and bacteremia).
Antibiotics Timings
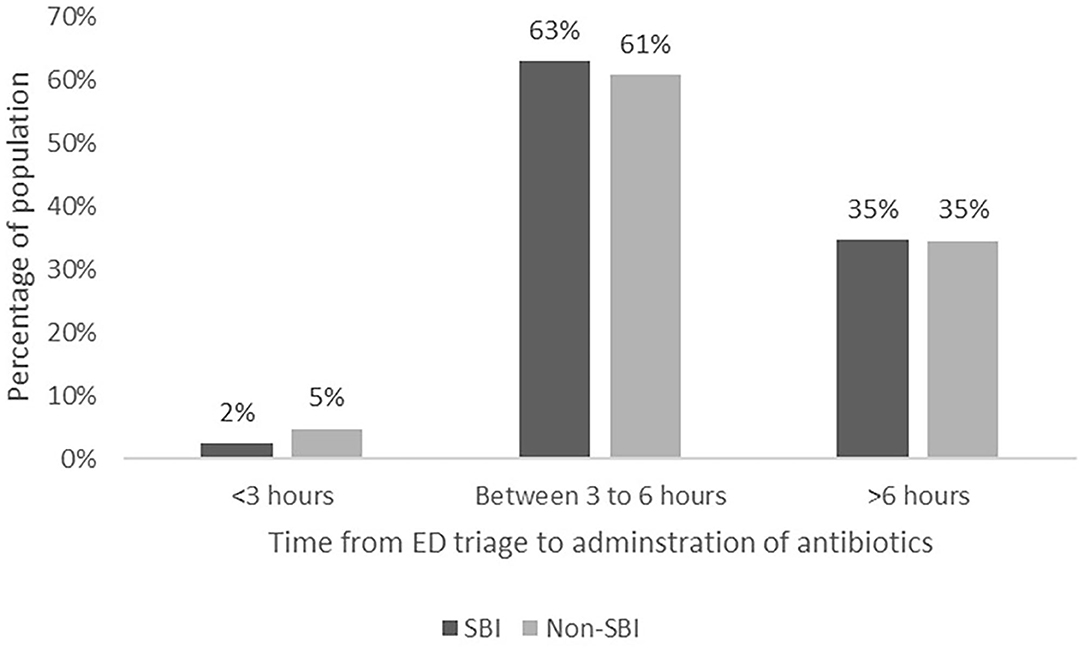
All infants with SBIs and 168/266 (63.2%) infants without SBIs received antibiotics. As shown in Table 4, the median time from triage at ED to first dose of antibiotics was 277.0 min (IQR 236.0–385.0) in the SBI group, and 304.5 min (IQR 238.5–404.0) in the non-SBI group (difference−8.0 mins, 95% confidence interval (CI)−38.0–21.0, p = 0.561). We found no difference in time from arrival in the ED to decision to administer antibiotics in the SBI group (median 156.0 min; IQR, 115.0–255.0) compared to the non-SBI group (144.0 min; IQR, 105.5–211.0) (difference 15.0 mins, 95% CI−7.0–38.0, p = 0.175). The median time between the decision for antibiotics and administration of first dose of antibiotics was shorter for the SBI group (107.0 min; IQR 83.0–168.0), compared to the non-SBI group (141.0 min; IQR 94.0–209.5) (difference−24.0 mins, 95% CI−44.0–4.0, p = 0.017). Subgroup analysis for neonates (<28 days) yielded consistent results. Figure 2 shows the proportion of infants in each group who received early (within 3 h of presentation) versus late antibiotics administration, as per the SSC guidelines (9).
We performed a sensitivity analysis by excluding cases of CSF pleocytosis without culture positive growth, and found consistent results (Supplementary Table 1). For the outcome of total time taken from ED arrival to antibiotics, there remained no difference between the groups (difference−12.0 mins, 95%CI−60.2–36.2, p = 0.625). For time from ED triage to decision for antibiotics, there remained no difference between the groups (difference 13.0 mins, 95%CI−14.0–40.0, p = 0.343). For time taken from decision for antibiotics to first infusion of antibiotics, the difference remained significant between the groups (difference−30.0 mins, 95% CI−54.8 to −5.2, p = 0.018).
Secondary Outcomes
Eleven (13.6%) infants with SBIs, and 15 (5.6%) without SBIs received a fluid bolus (p = 0.027). Only 1 infant was admitted to the HDU, and 1 other infant to ICU. Both infants had SBIs. There was neither use of inotropes, nor deaths in the study cohort.
Discussion
In our study of 249 febrile infants who received antibiotics, there was no significant difference in either the overall time taken from triage at ED to first dose of antibiotics, or in the time taken from arrival in the ED to time of decision to administer antibiotics (“recognition delay”) between the SBI and non-SBI cohort. We did find that the SBI group received antibiotics faster than the non-SBI group, once the decision for antibiotics had been made (“administration delay”).
We report a high prevalence of SBIs (23.3%) and IBIs (4.3%) in our cohort. This is higher than that reported by other studies, with a SBI prevalence of 5–15% (32) and an IBI prevalence of 2.3–3.3% (12, 33). We recognize important health service differences in our study population. We are the larger of 2 pediatric tertiary centers in the country and receive referrals from primary care centers. Infants who examine well with a clear source of fever may not be referred to our center for further evaluation. This could have accounted for the higher prevalence of SBIs and IBIs compared to the published literature.
ED providers have difficulty discerning which young febrile infants are at high risk of SBIs. This is likely because infants present with a non-specific complaint of fever and no other reliable signs and symptoms (32). Previously, we demonstrated that current triage tools including the National Institute for Health and Care Excellence (NICE) Traffic Light System and the Severity Index Score (SIS) were insufficient for young febrile infants (34). In our earlier studies, high temperature, tachycardia and a low SIS were associated with serious infections (25, 35). In this study, although there were differences in the height of fever and median heart rate at triage, these were clinically small differences that cannot provide discrimination between infants with SBIs and those without. Many febrile infants continue to be hospitalized and undergo invasive testing and empiric antibiotics treatment (36). Our institution is working to implement a modified algorithm based on the “Step-by-Step” approach to streamline the diagnosis and management of these febrile infants. Using point of care tests with faster turnaround time will provide earlier stratification of febrile infants so that those at high risk of SBIs receive antibiotics more promptly, and infants who are low risk will not be subjected to unnecessary invasive tests and antibiotics.
In our study, we observed that all infants with SBIs (100%) received antibiotics while 168/266 (63.2%) of the infants without SBIs received antibiotics. This underscores our findings that the current approach is highly sensitive in the final diagnosis of SBIs. While we did not miss SBIs in our population (all SBIs received antibiotics), our low threshold for intervention resulted in more than half the infants without SBIs receiving antibiotics. As such, merely treating all infants with SBIs with antibiotics is not sufficient, we seek to identify them early so that we can prioritize empirical antibiotics to those who really need them. This will result in more judicious use of invasive investigations and empirical antibiotics. Once identified, the SBI group received antibiotics faster than the non-SBI group (shorter “administration delay”). This is reassuring that once identified, the hospital has the resources and processes in place to administer antibiotics to infants with likely SBIs rapidly. These include a mechanism for early review in the wards if the young infant is deemed to look unwell or have potential for deterioration, in the ED. Additionally, given that the majority of the infants with SBIs had a UTI, once the urinalysis returned as positive, it may have prompted more timely administration of antibiotics.
The median total time taken from presentation at ED to administration of antibiotics exceeded the SSC recommendation of 3 h for both groups of infants (9). It would be important to examine the various clinical, administrative and logistic processes. The current workflow for febrile infants includes an initial assessment at the ED by the ED physician. Unstable infants receive investigations and urgent antibiotics in the ED. Stable infants are transferred to receive care from the inpatient team, before a decision is made for initiation of antibiotics. The various points of assessment could contribute to a delay in time to antibiotics. Besides focusing on early sepsis recognition, systemic changes to reduce delays could include effective communication between the medical, nursing and bed management teams, robust monitoring systems to detect sudden deterioration in infants, and institution protocols that mandate minimum standards for time to antibiotics.
Limitations and Strengths
We acknowledge the limitations of our study. Patients recruited in the first part of the study were part of another study and therefore could inadvertently have resulted in selection bias. However, once the team received ethics approval, we were able to track and follow outcomes for all febrile infants from the start of 2021. Not all infants without SBIs had a complete workup performed. However, all febrile infants were monitored until they were well and afebrile for 24 h before discharge, to ensure that no SBIs were missed. Our cohort was largely stable with only 2 infants requiring a higher level of care. We recognize that we may have been underpowered to study difference in time to antibiotics. We used the sample standard deviation (study σ = 350) as an estimate of the population standard (σ). Taking a difference between groups of 60 min as clinically relevant, a sample size of 488 patients per arm would be needed in a similar endeavor to ensure adequate power. Our study provides useful information for sample size computations in future related investigations. Being a single center study, we recognize that our findings may not be generalizable to other healthcare settings. Finally, we recognize that given the rapid changes in clinical status for these young infants, any risk stratification tool, even if methodically derived and validated, may not successfully differentiate infants with SBIs from those without, early in their sickness.
We recognize the strengths of this study, which include prospective data collection, independent verification of time stamps and therefore improved veracity of the timings reported in this study, and none of the subjects were lost to follow-up.
Conclusion
Given that all our infants with SBIs received antibiotics, our practice is overall safe. However, the goal is an improved level of care where early recognition of infants with SBIs translates to shorter time to antibiotics. We found that when all febrile infants compete for the same resources, early antibiotics was not prioritized for infants with SBIs. Future research should investigate how early accurate risk stratification and robust monitoring systems improve early recognition of SBIs and ensure prompt delivery of antibiotics.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available on reasonable request to the Corresponding Author.
Ethics Statement
The studies involving human participants were reviewed and approved by Study title: Rapid Triage for Serious Infections in Infants Younger than 3 months using a Novel Heart Rate Variability Tool IRB No: 2017/2680 SingHealth Centralised Institutional Review Board (CIRB). Written informed consent from the participants' legal guardian/next of kin was not required to participate in this study in accordance with the national legislation and the institutional requirements.
Author Contributions
JY, WO, JL, and S-LC contributed to the conception and design of the study. JY, WO, and RP collected and organized the database. JA and S-LC performed the statistical analysis. JY and WO wrote the first draft of the manuscript. JA, JL, and S-LC made critical revisions to the manuscript. All authors contributed to manuscript revision, read and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fped.2022.873043/full#supplementary-material
References
1. Liu L, Johnson HL, Cousens S, Perin J, Scott S, Lawn JE, et al. Child Health Epidemiology Reference Group of WHO and UNICEF. Global, regional, and national causes of child mortality: an updated systematic analysis for 2010 with time trends since 2000. Lancet. (2012) 379:2151-61. doi: 10.1016/S0140-6736(12)60560-1
2. World Health Organization and UNICEF. WHO/UNICEF Joint Statement. Managing Possible Serious Bacterial Infection in Young Infants 0 – 59 Days Old When Referral is Not Feasible. World Health Organization (2017). 6 p. Licence: CC BY-NC-SA 3.0 IGO
3. World Health Organization. Newborns: Improving Survival and Well-Being. World Health Organisation (2019).
4. Watson RS, Carcillo JA, Linde-Zwirble WT, Clermont G, Lidicker J, Angus DC. The epidemiology of severe sepsis in children in the United States. Am J Respir Crit Care Med. (2003) 167:695–701. doi: 10.1164/rccm.200207-682OC
5. Stephens BE, Vohr BR. Neurodevelopmental outcome of the premature infant. Pediatr Clin North Am. (2009) 56:631–46. doi: 10.1016/j.pcl.2009.03.005
6. Stoll BJ, Hansen NI, Adams-Chapman I, Fanaroff AA, Hintz SR, Vohr B, et al. National Institute of Child Health and Human Development Neonatal Research Network. Neurodevelopmental and growth impairment among extremely low-birth-weight infants with neonatal infection. JAMA. (2004) 292:2357–65. doi: 10.1001/jama.292.19.2357
7. Pek JH, Yap BJ, Gan MY, Seethor STT, Greenberg R, Hornik CPV, et al. Neurocognitive impairment after neonatal sepsis: protocol for a systematic review and meta-analysis. BMJ Open. (2020) 10:e038816. doi: 10.1136/bmjopen-2020-038816
8. Pek JH, Gan MY, Yap BJ, Seethor STT, Greenberg RG, Hornik CPV, et al. Contemporary trends in global mortality of sepsis among young infants less than 90 days old: protocol for a systematic review and meta-analysis. BMJ Open. (2020) 10:e038815. doi: 10.1136/bmjopen-2020-038815
9. Weiss SL, Peters MJ, Alhazzani W, Agus MSD, Flori HR, Inwald DP, et al. Surviving sepsis campaign international guidelines for the management of septic shock and sepsis-associated organ dysfunction in children. Pediatr Crit Care Med. (2020) 21:e52–e106. doi: 10.1097/PCC.0000000000002197
10. Roberts KB, Downs SM, Finnell SME, Hellerstein S, Shortliffe LD, Wald ER, et al. Subcommittee on Urinary Tract Infection, Steering Committee on Quality Improvement and Management. Urinary tract infection: clinical practice guideline for the diagnosis and management of the initial UTI in febrile infants and children 2 to 24 months. Pediatrics. (2011) 128:595–610. doi: 10.1542/peds.2011-1330
11. Bradley JS, Byington CL, Shah SS, Alverson B, Carter ER, Harrison C, et al. Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. The management of community-acquired pneumonia in infants and children older than 3 months of age: clinical practice guidelines by the Pediatric Infectious Diseases Society and the Infectious Diseases Society of America. Clin Infect Dis. (2011) 53:e25–76. doi: 10.1093/cid/cir531
12. Huang YH, Yan JH, Kuo KC, Wu WT, Su CM, Chiu IM. Early antibiotics use in young infants with invasive bacterial infection visiting emergency department, a single medical center's experience. Pediatr Neonatol. (2020) 61:155–9. doi: 10.1016/j.pedneo.2019.08.003
13. Weiss SL, Fitzgerald JC, Balamuth F, Alpern ER, Lavelle J, Chilutti M, et al. Delayed antimicrobial therapy increases mortality and organ dysfunction duration in pediatric sepsis. Crit Care Med. (2014) 42:2409–17. doi: 10.1097/CCM.0000000000000509
14. Dagan R, Powell KR, Hall CB, Menegus MA. Identification of infants unlikely to have serious bacterial infection although hospitalized for suspected sepsis. J Pediatr. (1985) 107:855–60. doi: 10.1016/S0022-3476(85)80175-X
15. Baker MD, Bell LM, Avner JR. Outpatient management without antibiotics of fever in selected infants. N Engl J Med. (1993) 329:1437-41. doi: 10.1056/NEJM199311113292001
16. Baskin MN, O'Rourke EJ, Fleisher GR. Outpatient treatment of febrile infants 28 to 89 days of age with intramuscular administration of ceftriaxone. J Pediatr. (1992) 120:22–7. doi: 10.1016/S0022-3476(05)80591-8
17. Gomez B, Mintegi S, Bressan S, Da Dalt L, Gervaix A, Lacroix L. European Group for Validation of the Step-by-Step Approach. Validation of the “step-by-step” approach in the management of young febrile infants. Pediatrics. (2016) 138:e20154381. doi: 10.1542/peds.2015-4381
18. Kuppermann N, Dayan PS, Levine DA, Vitale M, Tzimenatos L, Tunik MG, et al. Febrile infant working group of the Pediatric Emergency Care Applied Research Network (PECARN). A clinical prediction rule to identify febrile infants 60 days and younger at low risk for serious bacterial infections. JAMA Pediatr. (2019) 173:342–51. doi: 10.1001/jamapediatrics.2018.5501
19. Aronson PL, Thurm C, Alpern ER, Alessandrini EA, Williams DJ, Shah SS, et al. Febrile young infant research collaborative. Variation in care of the febrile young infant <90 days in US pediatric emergency departments. Pediatrics. (2014) 134:667–77. doi: 10.1542/peds.2014-1382
20. Aronson PL, Thurm C, Williams DJ, Nigrovic LE, Alpern ER, Tieder JS, et al. Febrile Young Infant Research Collaborative. Association of clinical practice guidelines with emergency department management of febrile infants ≤ 56 days of age. J Hosp Med. (2015) 10:358–65. doi: 10.1002/jhm.2329
21. Jain S, Cheng J, Alpern ER, Thurm C, Schroeder L, Black K, et al. Management of febrile neonates in US pediatric emergency departments. Pediatrics. (2014) 133:187–95. doi: 10.1542/peds.2013-1820
22. Taylor SP, Anderson WE, Beam K, Taylor B, Ellerman J, Kowalkowski MA. The association between antibiotic delay intervals and hospital mortality among patients treated in the emergency department for suspected sepsis. Crit Care Med. (2021) 49:741–7. doi: 10.1097/CCM.0000000000004863
23. Curtiss J, Karvetski C, Taylor B, Taylor S. 1476: Intervals of delay in antibiotic administration for emergency department patients with sepsis. Crit Care Med. (2018) 46:722. doi: 10.1097/01.ccm.0000529478.57644.99
24. Kashiouris MG, Zemore Z, Kimball Z, Stefanou C, Fowler AA. 3rd, Fisher B, et al. Supply Chain Delays in Antimicrobial Administration After the Initial Clinician Order and Mortality in Patients With Sepsis. Crit Care Med. (2019) 47:1388–95. doi: 10.1097/CCM.0000000000003921
25. Chong SL, Ong GY, Allen JC, Lee JH, Piragasam R, Koh GZX, et al. Early prediction of serious infections in febrile infants incorporating heart rate variability in an emergency department: a pilot study. Emerg Med J. (2021) 38:607–12. doi: 10.1136/emermed-2020-210675
26. Mahajan P, Kuppermann N, Mejias A, Suarez N, Chaussabel D, Casper TC, et al. Pediatric Emergency Care Applied Research Network (PECARN). Association of RNA biosignatures with bacterial infections in febrile infants aged 60 days or younger. JAMA. (2016) 316:846–57. doi: 10.1001/jama.2016.9207
27. Tunkel AR, Hartman BJ, Kaplan SL, Kaufman BA, Roos KL, Scheld WM, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. (2004) 39:1267–84. doi: 10.1086/425368
28. Tacon CL, Flower O. Diagnosis and management of bacterial meningitis in the paediatric population: a review. Emerg Med Int. (2012) 2012:320309. doi: 10.1155/2012/320309
29. Mintegi S, Gomez B, Carro A, Diaz H, Benito J. Invasive bacterial infections in young afebrile infants with a history of fever. Arch Dis Child. (2018) 103:665–9. doi: 10.1136/archdischild-2017-313578
32. Esposito S, Rinaldi VE, Argentiero A, Farinelli E, Cofini M, D'Alonzo R, et al. Approach to neonates and young infants with fever without a source who are at risk for severe bacterial infection. Mediators Inflamm. (2018) 2018:4869329. doi: 10.1155/2018/4869329
33. DePorre AG, Aronson PL, McCulloh RJ. Facing the ongoing challenge of the febrile young infant. Crit Care. (2017) 21:68. doi: 10.1186/s13054-017-1646-9
34. Yao SHW, Ong GY, Maconochie IK, Lee KP, Chong SL. Analysis of emergency department prediction tools in evaluating febrile young infants at risk for serious infections. Emerg Med J. (2019) 36:729–35. doi: 10.1136/emermed-2018-208210
35. Chong SL, Ong GY, Chin WYW, Chua JM, Nair P, Ong ASZ, et al. A retrospective review of vital signs and clinical outcomes of febrile infants younger than 3 months old presenting to the emergency department. PLoS ONE. (2018) 13:e0190649. doi: 10.1371/journal.pone.0190649
Keywords: serious bacterial infections, infants, recognition delay, sepsis, antimicrobial therapy
Citation: Yang J, Ong WJ, Piragasam R, Allen JC, Lee JH and Chong S-L (2022) Delays in Time-To-Antibiotics for Young Febrile Infants With Serious Bacterial Infections: A Prospective Single-Center Study. Front. Pediatr. 10:873043. doi: 10.3389/fped.2022.873043
Received: 10 February 2022; Accepted: 22 March 2022;
Published: 29 April 2022.
Edited by:
Silvia Bressan, University of Padua, ItalyReviewed by:
Todd William Lyons, Boston Children's Hospital, United StatesElliot Long, Royal Children's Hospital, Australia
Damian Roland, University of Leicester, United Kingdom
Copyright © 2022 Yang, Ong, Piragasam, Allen, Lee and Chong. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jinghui Yang, eWFuZy5qaW5naHVpQHNpbmdoZWFsdGguY29tLnNn
 Jinghui Yang
Jinghui Yang Wei Jie Ong2
Wei Jie Ong2 Rupini Piragasam
Rupini Piragasam John Carson Allen
John Carson Allen Jan Hau Lee
Jan Hau Lee Shu-Ling Chong
Shu-Ling Chong