
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Pediatr., 09 August 2021
Sec. Pediatric Urology
Volume 9 - 2021 | https://doi.org/10.3389/fped.2021.718647
 Tariq O. Abbas1,2,3,4*
Tariq O. Abbas1,2,3,4* Abubakr Elawad2
Abubakr Elawad2 Aamir Kareem2
Aamir Kareem2 Abdul Kareem Pullattayil S5
Abdul Kareem Pullattayil S5 Mansour Ali2
Mansour Ali2 Abdulla Alnaimi6
Abdulla Alnaimi6Background: There is a steadily growing number of different reconstructive surgical procedures for hypospadias that were tested on animal models prior to their human application. However, the clinical translatability and reproducibility of the results encountered in preclinical urethral reconstruction experiments is considered poor, with significant factors contributing to the poor design and reporting of animal experiments. Our objective was to evaluate the quality of the design and reporting in published articles of urethral reconstructive preclinical studies.
Methods: Both PubMed and EMBASE databases were searched for animal urethral repair experiments between January 2014 and September 2019. Internal quality (bias) was evaluated through several signaling questions arising from the Systematic Review Centre for Laboratory animal Experimentation (SYRCLE), while the quality of reporting was assessed by the Animal Research: Reporting of In vivo Experiments (ARRIVE) guidelines by scoring of a 20-item checklist.
Results: A total of 638 articles were initially screened after the literature search. Employing the inclusion and exclusion criteria, 30 studies were chosen for full-text screening and 21 studies were considered eligible for the quality assessment. The mean score of the checklist was 66%. The elements that accomplished the highest grades included the number of animals utilized, the number in each investigational and control group, and the delineation of investigational conclusions. The items that were least commonly stated comprised information about the experimental method, housing and husbandry, rationalization of the number of animals, and reporting of adverse events. No paper stated the sample size estimation.
Conclusion: We found that several critical experiment design principles were poorly reported, which hinders a rigorous appraisal of the scientific quality and reproducibility of the experiments. A comprehensive implementation of the ARRIVE guidelines in animal studies exploring urethral repair is necessary to facilitate the effective translation of preclinical research findings into clinical therapies.
Hypospadias is considered a common birth defect with an incidence of about 1 in 300 live births and has significant clinical and social impacts (1). Furthermore, the reconstructive urethroplasty operations are technically demanding and associated with significant complication rates (1–5). It is considered vitally important that preclinical experiments evaluating the different surgical procedures utilized are well-designed and appropriately reported in order to achieve sound translation to human and generalizability scores (6–9).
Several animal models have been utilized to evaluate several hypospadias repair techniques, with rabbits being the most frequently used (8, 10, 11). This might be because the rabbit's urethra is easily accessed and displays significant functional and structural similarities to human urethra, where a robust envelop of well-vascularized spongiosa encircles a thin epithelial layer underneath (12–16). Furthermore, transurethral endoscopes can be readily used as the size of an adult rabbit's urethra is comparable to that of an infant boy, where most of the hypospadias surgery are mostly conducted.
Several reports have raised concerns that the translation of preclinical experiments to humans has several challenges, including the variations of species and strains with subsequent physiological impact (17), absence of blinding (18), insufficient reporting of technical details, and under-reporting of complications or uncertain results, which could prime false conclusions (19). As a result, in 2010, the Animal Research: Reporting In vivo Experiments (ARRIVE) guidelines (20–22) were introduced by the National Centre for Replacement, Reduction, and Refinement (NC3Rs). Despite the increasing utilization of these guidelines, several research territories still struggle to capture the targeted levels of adoption and compliance (23–25). On the other hand, poor study design and incomplete reporting of outcomes might partly explain the hindrance of the clinical translation of urethroplasty procedures (26).
The utilization of systematic reviews to reflect and summarize the findings of animal experimental studies is less common than in clinical studies. Some systematic review features of animal studies need to be tailored accordingly and are mainly affected by bias. Therefore, the degree of translatability of such systematic reviews to clinical practice depends on a sound methodology and the design quality of the included experiments (27). The main goal of this systematic review was to explore these research demands by executing a quality evaluation using the ARRIVE and the Systematic Review Centre for Laboratory Animal Experimentation (SYRCLE) (28) recommendations as checklists. The range of the review has not been restricted to experiments using a certain animal model.
A search in MEDLINE of the PubMed database and EMBASE of the OVID SP database was conducted in September 2020. The search terms nominated were: urethra, urethroplasty, urethral reconstruction, urethral graft, and animal experimentation reconstructive surgery. The search arenas were organized by database grounds like MeSH term, Text Word, and All Fields suitable to the databases. “Publication date: 01/01/2014 to present” and “English language” filters have been used. Details of the search are represented in the PRISMA flow diagram (Figure 1).
All retrieved publications were screened in the abstract level initially by two authors (TA and AKPS). Articles have been excluded, including 12 duplicates (see exclusion criteria in the flowchart in Figure 1). Group discussions resolved disputes regarding the appropriateness of an article. Eligible articles were included for full-text analysis. The reasons for the further exclusion of articles are mentioned in the flowchart.
Extraction into a standardized data framework derived from the ARRIVE guidelines (22) (Supplementary Table 1) was conducted separately by three reviewers. In certain ARRIVE questions that were considered to be vital for urethroplasty experiments, the option (NA) was removed and the two options (yes) and (no) were kept for the reviewers to select from. For the possibility of discrepancies between the reviewers, a training phase through detailed descriptions and examples of scoring was conducted with the three reviewers before the commencement of the data collection.
Information on the ARRIVE guidelines consists of 38 items (Supplementary Table 1). Every item was evaluated as “yes” if the item was reported in the study, “no” if the item was not reported, and “not applicable” if the item was not relevant. All authors read the selected full-text articles independently and extracted the data blinded for the analysis from the other reviewers. Inconsistent data were consequently settled by decision of the third reviewer.
To evaluate the risk of bias of the studies, we used the SYRCLE (28) via its 10 signaling questions (Supplementary Table 2). These entries are related to six types of bias: selection bias, performance bias, detection bias, attrition bias, reporting bias, and other biases. If the criteria recommended fitted, we indicated the answer YES (i.e., the risk of bias was low). In contrast, if the guidelines were not met, we assigned the answer NO, symbolizing a considerable risk of bias. If there were inadequate data or the study did not address the expected criteria, either YES or NO could be selected. Alternatively, UNCLEAR was granted (i.e., the risk of bias was unknown). Two independent reviewers did the evaluations, and discrepancies were settled by consensus-oriented discussion.
The data were compiled employing a Microsoft Excel spreadsheet and analyzed using the IBM SPSS Statistics, version 21 (IBM Corp., Armonk, NY, USA). For each of the selected studies, a score was calculated, which represents the percentage of positively reported items. The score was calculated using the following formula:
where Nyes is the number of “yes” entries, Nna is the number of “not applicable” entries, and 38 is the total number of items in the ARRIVE guideline. To compare the different scores of the included articles over the years they were published, the Mann–Kendall test was utilized. Statistical significance was set at P < 0.05.
As shown in Figure 1, a total of 638 articles were initially screened after the literature search. Following the inclusion and exclusion criteria, 30 studies were chosen for full-text screening and 21 studies were considered eligible for quality assessment. All studies used male rabbits, except two studies that included Wistar and Sprague–Dawley rats. The most commonly studied approach was evaluating the tabularized incised posterior urethral plate (TIP). These studies comprise a range of approaches for urethral repair, which are summarized in Table 1. The table provides details about the strain, sex, age, weight, number of animals, procedural approach, the complications encountered, and duration of follow-up. The average number of animals in each experiment was 22 and varied between 8 and 38. The average of the post-surgical follow-up duration was 12 weeks and ranged between 2 and 52 weeks (Table 1).
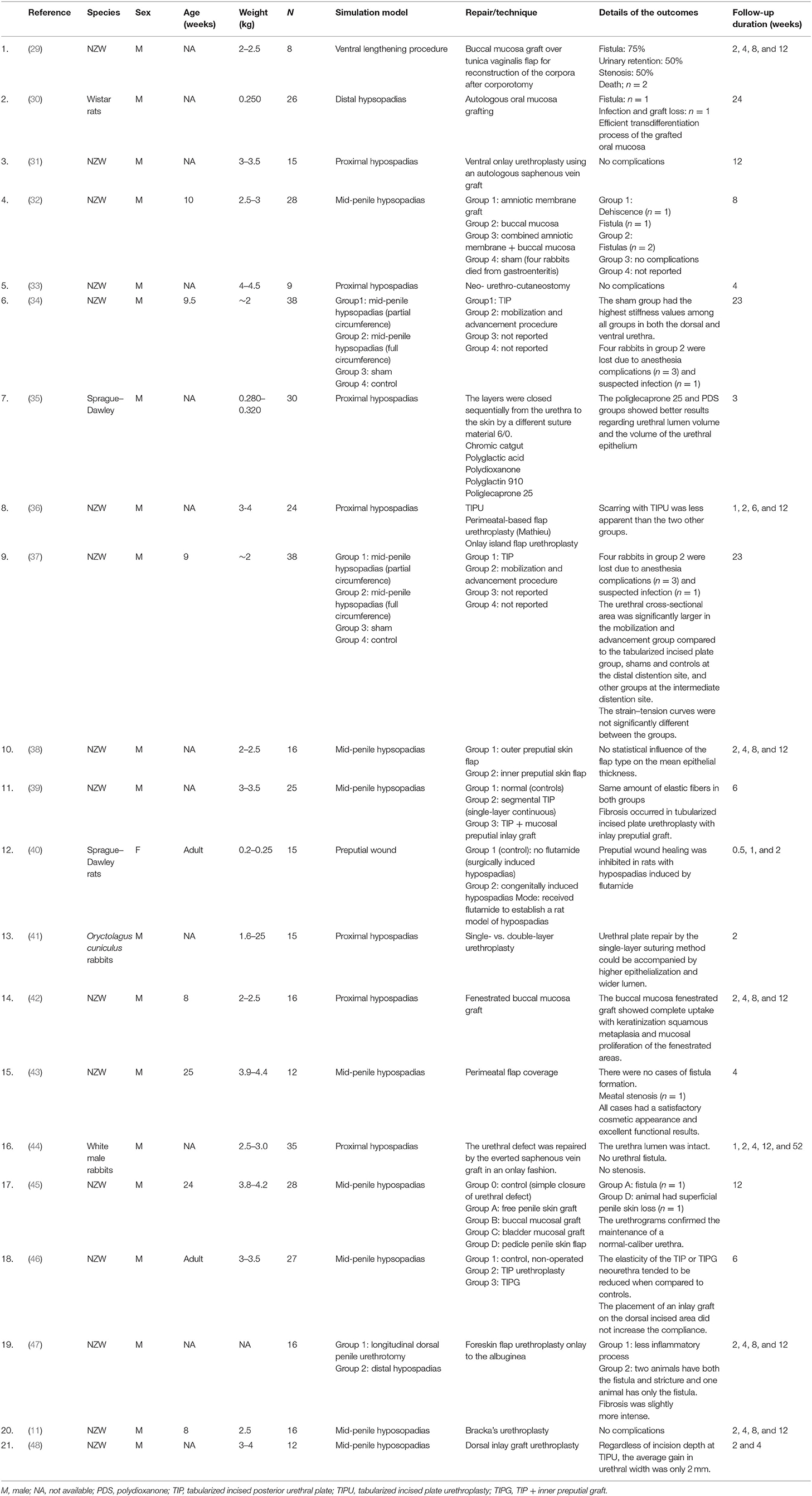
Table 1. Overview of the studies included with a summary of the age, sex, and weight of the animal models utilized and evaluation of the hypospadias simulation model and repair.
The frequencies of the options “yes,” “no,” and “n/a” of the selected studies according to the ARRIVE checklist are shown in Figure 2. Calculation of the scores of each individual study is presented in Figure 3. The mean checklist score of the studies published from 2014 to 2019 was 66%.
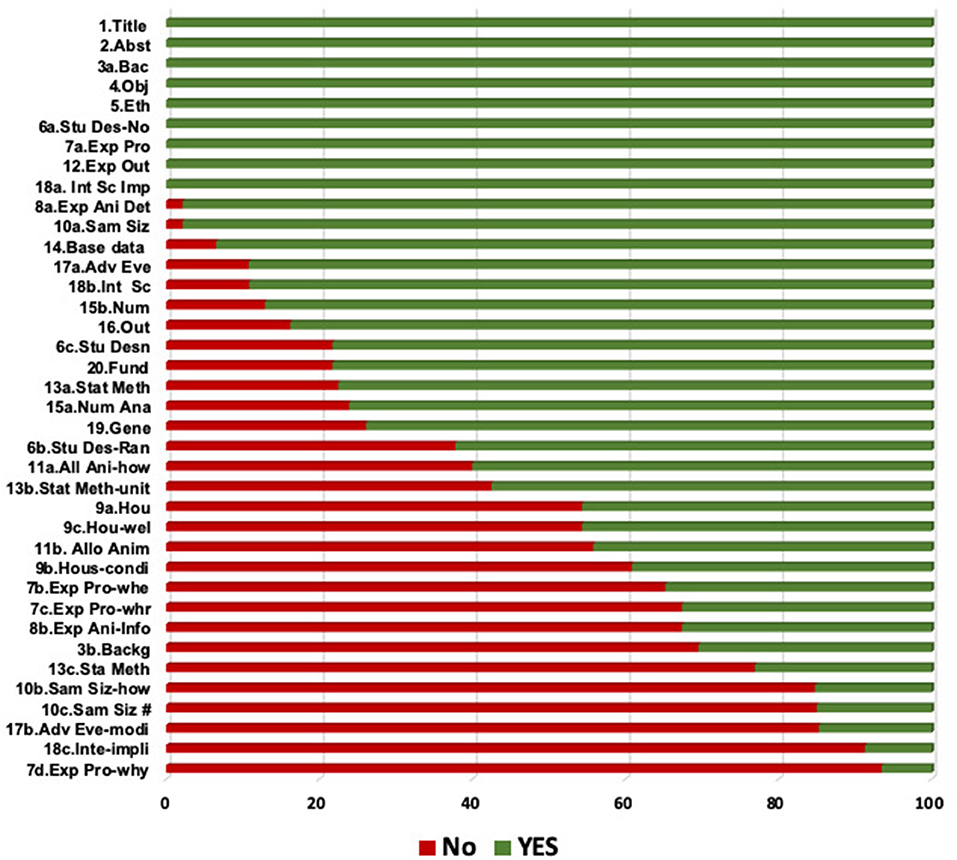
Figure 2. Bar chart displaying the frequencies of the options “Yes” and “No” among the 22 articles selected for the analysis. Items are indicated by their corresponding numbers and a label associated with their content. Alloc Ani, allocation of animals; Exp ani, experimental animals; Basel, baseline data; Fund, funding; Generali, generalizability; Stu desig, study design; Sample, sample size.
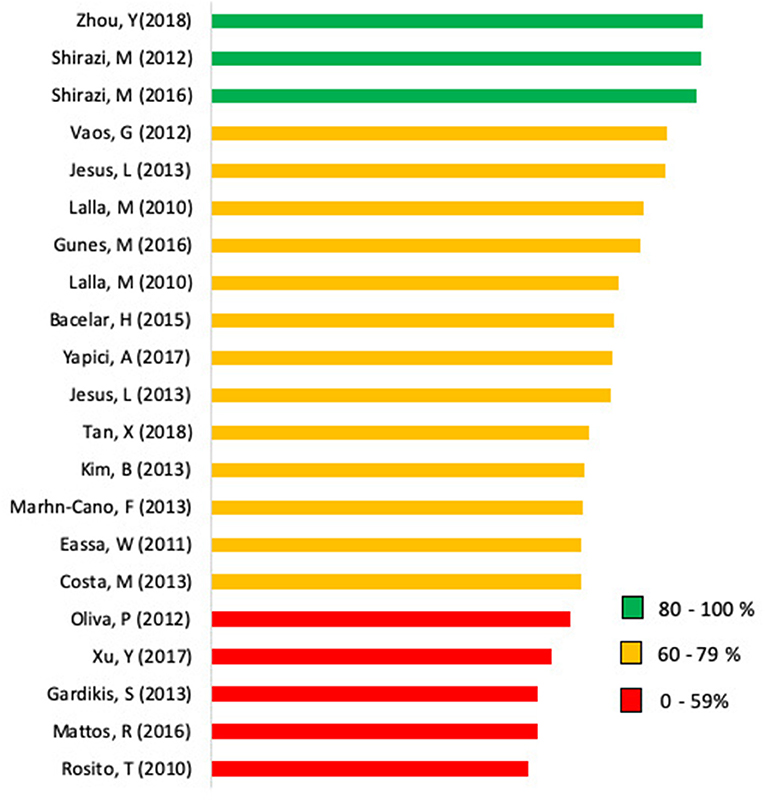
Figure 3. Bar chart displaying the scores of the analyzed articles. Scores represent the percentage adherence to the ARRIVE (Animal Research: Reporting of In vivo Experiments) guidelines on a scale from 0 to 100.
The background of the studies was described adequately, including the rationale and the context in all the experiments. The elements that accomplished the highest grades comprised the number of animals utilized, the number in each investigational and control group, and the delineation of investigational conclusions. The items that were least commonly stated comprised information about the experimental method, housing and husbandry, rationalization of the number of animals, and reporting of adverse events.
No tendency or steady pattern in the grade of the scores could be recognized over the studied duration (2014–2019) as the Kendall's rank correlation coefficient (tau) was unfolded to be very low at 0.055 (P = 0.701).
Regarding the study design, numerous vital elements were poorly communicated. Only one study answered the item (7d) about study processes. Recording of randomization scored 62%. None of the 21 studies reported sample size estimation. The least frequently reported items (reported in ≤ 20% of the studies) were items 18c (interpretation), 10b and 10c (sample size), 7d (experimental procedures), and 17b (adverse events). As is evident from Figures 3, 4, none of the analyzed studies fully complied with the ARRIVE guidelines.
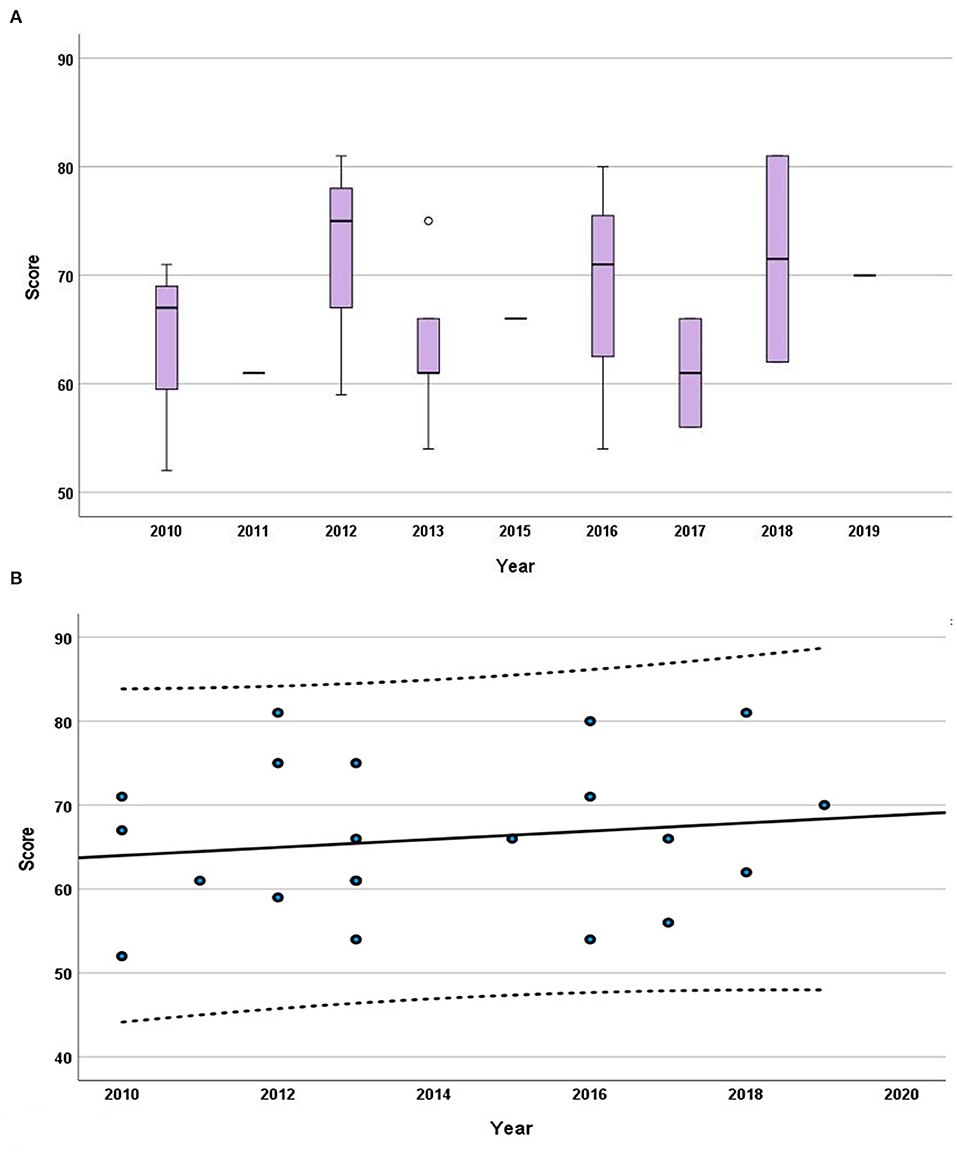
Figure 4. Progression of the grades throughout the years. (A) Box plot displaying the median, interquartile range, minimum, and maximum of the scores for each year from 2014 to 2019. (B) The trendline presented is from the Mann–Kendall trend analysis on the median of the scores. The interrupted lines indicate the 95% confidence interval.
Figure 5 displays the global grades of the bias risk appraisal of the 21 studies involved in this systematic review. Of the studies, 92% stated that the experimental groups were similar at baseline or were adjusted for confounders. None of the papers described whether the allocation to the different groups during the randomization process was concealed. Sixteen percent of the studies reported that the outcome assessment has been blinded.
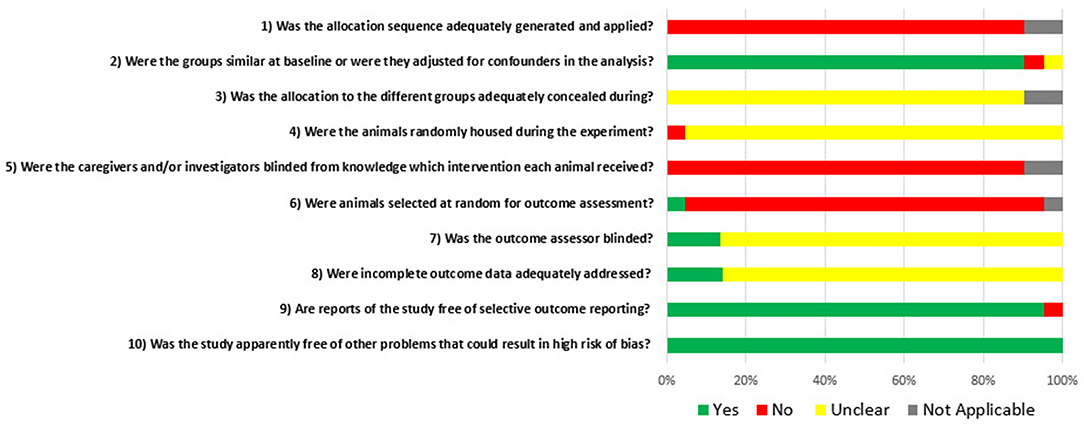
Figure 5. Risk of bias, averaged per each signal question. Yes = low risk of bias; No = high risk of bias; Unclear = unclear risk of bias; and Not applicable.
This systematic review unfolded that the reporting quality of some crucial pieces was commonly poor in preclinical studies of hypospadias repair, which did not seem to progress over the years. Preclinical studies play an important role in scientific progress and the discovery of new and potentially successful surgical procedures, provided that they are designed, conducted, assessed, and sufficiently reported according to internationally agreed guidelines. Several principal parts of the study design are frequently missed, which significantly adds to failure of reproducibility following these experiments, and urethral reconstructive studies seem not to be an exception. Fundamental experimental design components are often overlooked in scientific papers, which contributes to the irreproducibility of the experiments (49), and preclinical studies investigating urethral repair do not appear to be an exemption.
This study revealed that there is still room for methodological improvements of experiments on animals in the urethral reconstruction and hypospadiology fields. Most domains were judged to have an unclear risk of bias, and therefore, it is not possible to determine the degree of bias of the described treatment effects. Note that the risk of bias and the quality of reporting should be considered distinct from each other. Although, the former relates to the internal efficacy of a trial, the latter points to how researchers report their conclusions. Although, personal communication with the authors of the study might be an attempt at clarifying dubious or lack of information, this does not warrant the correctness of the information rendered (50).
Strikingly, particulars to safeguard the reproducibility of such experiments, like animal housing, husbandry, and anesthetics, were seldom communicated, which might meaningfully disturb the study conclusions (51). Prager et al. (52) revealed that different animal husbandry conditions could affect several research outcomes. Furthermore, caring for the study animals in tiny cages or in big groups following urethroplasties likely brings hazard of infections, probabilities of dislodgements of stents, and surgical site trauma.
None of the included experiments reported sample size calculation similar to the previous signal that displays scarcity of reporting concerning sample size calculation in animal studies (53). Additionally, not defining the study design before the commencement of experiments could result in inappropriate analysis of the null hypothesis and insufficient sample size, potentially leading to debatable conclusions. The required sample size of equivalence trials is usually larger than that for superiority trials (54). Experiments with low power may create false-negative results, i.e., so-called type II errors (55). The low average number of animals in most studies could be partly rationalized by the high cost and the difficulty of testing, handling, and monitoring animals throughout the experiment. Nonetheless, approaches to verify the number of animals used exist, such as performing previous pilot investigations or utilizing Mead's resource equation in situations where there is no information on the standard deviation and it is challenging to define an effect size (56).
Only a few of the included studies have deliberated the “why and how” regarding the type of animal model employed and its applicability to human pathology. We consider that arguments are of noteworthy significance in all surgical reconstructive experiments because of the substantial variations between the genital anatomy of humans and animals. We found that blinding was not steadily described. However, it might be impractical to blind the investigator performing different urethroplasty procedures. Therefore, we did not attempt to analyze blinding within the included studies.
An implementation approach to increasing compliance of reporting quality would be stringent polices by the editorial committees of the journals (57). However, a recent randomized controlled trial exposed that instructing the accomplishment of an ARRIVE checklist throughout submission of the paper, with no additional pressure on reporting by the editorial team, did not encourage compliance (58). The recently published PREPARE (Planning Research and Experimental Procedures on Animals: Recommendations for Excellence) guidelines (59) can likewise aid in implementing more consideration to investigational precision at an earlier stage of the planning course.
The limitations of this review include the potential subjectivity of the assessment by the evaluators. Yet, the pronounced inter-observer consensus established that the assessors had a rather similar approach to applying these guidelines. Additionally, the rather small number of studies involved restricted a broad appraisal of the conditions persuading reporting. Calculating a summary score for each study using the SYRCLE was not done as a summary score inevitably involves assigning “weights” to specific domains in the tool, and it is difficult to justify the weights assigned. Also, these weights might differ per outcome and review. Using the existing animal experimental literature is also challenging because the current reporting quality of animal studies is low; several details regarding the housing conditions or timing outcome assessment are often unreported. Users also indicated that they had to judge many entries as having an “unclear risk of bias.”
This review exposed that the experiments testing urethral reconstructive procedures suffer from significant internal (design/bias) limitations and significant reporting shortages. We encourage the usage of the ARRIVE procedures in all animal experiments to benefit the production of manuscripts that deliver defined conclusions of scientific value.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
TA conceived the article, planned, and prepared its structure. TA performed the bibliographical search along with AP, AE, AK, and AA. AE, TA, AK, and AP analyzed the results. TA wrote the manuscript draft. TA, AK, AP, MA, AA, and AE edited sections of the manuscript and contributed to the critical revision of the final draft.
Open access fees is sponsored by Sidra Medicine, Doha, Qatar.
AA is employed by Hamad Medical Corporation.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fped.2021.718647/full#supplementary-material
1. Keays MA, Dave S. Current hypospadias management: diagnosis, surgical management, and long-term patient-centred outcomes. Can Urol Assoc J. (2017) 11(Suppl. 1):S48–53. doi: 10.5489/cuaj.4386
2. Chapple C, Andrich D, Atala A, Barbagli G, Cavalcanti A, Kulkarni S, et al. SIU/ICUD consultation on urethral strictures: the management of anterior urethral stricture disease using substitution urethroplasty. Urology. (2014) 83:S31–47. doi: 10.1016/j.urology.2013.09.012
3. Snodgrass W, Bush N. Primary hypospadias repair techniques: a review of the evidence. Urol Ann. (2016) 8:403–8. doi: 10.4103/0974-7796.192097
4. Barbagli G, Sansalone S, Djinovic R, Romano G, Lazzeri M. Current controversies in reconstructive surgery of the anterior urethra: a clinical overview. Int Braz J Urol. (2012) 38:307–16; discussion 316. doi: 10.1590/S1677-55382012000300003
5. Mundy AR, Andrich DE. Urethral strictures. BJU Int. (2011) 107:6–26. doi: 10.1111/j.1464-410X.2010.09800.x
6. Abbas TO, Mahdi E, Hasan A, AlAnsari A, Pennisi CP. Current status of tissue engineering in the management of severe hypospadias. Front Pediatr. (2018) 5:283. doi: 10.3389/fped.2017.00283
7. Abbas TO, Yalcin HC, Pennisi CP. From acellular matrices to smart polymers: degradable scaffolds that are transforming the shape of urethral tissue engineering. Int J Mol Sci. (2019) 20:1763. doi: 10.3390/ijms20071763
8. Versteegden LRM, de Jonge PKJD, IntHout J, van Kuppevelt TH, Oosterwijk E, Feitz WFJ, et al. Tissue engineering of the urethra: a systematic review and meta-analysis of preclinical and clinical studies. Eur Urol. (2017) 72:1–13. doi: 10.1016/j.eururo.2017.03.026
9. Sievert K-D. Tissue engineering of the urethra: solid basic research and farsighted planning are required for clinical application. Eur Urol. (2017) 72:607–9. doi: 10.1016/j.eururo.2017.04.025
10. Qi N, Li W, Tian H. A systematic review of animal and clinical studies on the use of scaffolds for urethral repair. J Huazhong Univ Sci Technolog Med Sci. (2016) 36:111–7. doi: 10.1007/s11596-016-1551-5
11. Rosito TE, Pires JAS, Delcelo R, Ortiz V, Macedo A Jr. Macroscopic and histological evaluation of tunica vaginalis dorsal grafting in the first stage of Bracka's urethroplasty: an experimental study in rabbits. BJU Int. (2011) 108:E17–22. doi: 10.1111/j.1464-410X.2010.09708.x
12. Faydaci G, Tarhan F, Tuncer M, Eryildirim B, Celik O, Keser SH, et al. Comparison of two experimental models for urethral stricture in the anterior urethra of the male rabbit. Urology. (2012) 80:225.e7–e11. doi: 10.1016/j.urology.2012.04.025
13. Zhang K, Fu Q, Yoo J, Chen X, Chandra P, Mo X, et al. 3D bioprinting of urethra with PCL/PLCL blend and dual autologous cells in fibrin hydrogel: an in vitro evaluation of biomimetic mechanical property and cell growth environment. Acta Biomater. (2017). 50:154–64. doi: 10.1016/j.actbio.2016.12.008
14. Theodorescu D, Balcom A, Smith CR, McLorie GA, Churchill BM, Khoury AE. Urethral replacement with vascularized tunica vaginalis: defining the optimal form of use. J Urol. (1998) 159:1708–11. doi: 10.1097/00005392-199805000-00098
15. Kemppainen E, Talja M, Riihelä M, Pohjonen T, Törmälä P, Alfthan O. A bioresorbable urethral stent. An experimental study. Urol Res. (1993) 21:235–8. doi: 10.1007/BF00590042
16. Bradley JE, Anderson UA, Woolsey SM, Thornbury KD, McHale NG, Hollywood MA. Characterization of T-type calcium current and its contribution to electrical activity in rabbit urethra. Am J Physiol Physiol. (2004) 286:C1078–88. doi: 10.1152/ajpcell.00463.2003
17. Festing MFW. The choice of animal model and reduction. Altern Lab Anim. (2004) 32(Suppl. 2):59–64. doi: 10.1177/026119290403202s12
18. Mignini LE, Khan KS. Methodological quality of systematic reviews of animal studies: a survey of reviews of basic research. BMC Med Res Methodol. (2006) 6:10. doi: 10.1186/1471-2288-6-10
19. Korevaar DA, Hooft L, Ter Riet G. Systematic reviews and meta-analyses of preclinical studies: publication bias in laboratory animal experiments. Lab Anim. (2011) 45:225–30. doi: 10.1258/la.2011.010121
20. Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG NC3Rs Reporting Guidelines Working Group. Animal research: reporting in vivo experiments: The ARRIVE guidelines. Br J Pharmacol. (2010) 160:1577–9. doi: 10.1111/j.1476-5381.2010.00872.x
21. van der Worp HB, Howells DW, Sena ES, Porritt MJ, Rewell S, O'Collins V, et al. Can animal models of disease reliably inform human studies? PLoS Med. (2010) 7:e1000245. doi: 10.1371/journal.pmed.1000245
22. Kilkenny C, Browne WJ, Cuthill IC, Emerson M, Altman DG. Improving bioscience research reporting: the ARRIVE guidelines for reporting animal research. PLoS Biol. (2010) 8:e1000412. doi: 10.1371/journal.pbio.1000412
23. Leung V, Rousseau-Blass F, Beauchamp G, Pang DSJ. ARRIVE has not ARRIVEd: Support for the ARRIVE (Animal Research: Reporting of in vivo Experiments) guidelines does not improve the reporting quality of papers in animal welfare, analgesia or anesthesia. PLoS ONE. (2018) 13:e0197882. doi: 10.1371/journal.pone.0197882
24. Bezdjian A, Klis SFL, Peters JPM, Grolman W, Stegeman I. Quality of reporting of otorhinolaryngology articles using animal models with the ARRIVE statement. Lab Anim. (2018) 52:79–87. doi: 10.1177/0023677217718862
25. Ting KHJ, Hill CL, Whittle SL. Quality of reporting of interventional animal studies in rheumatology: a systematic review using the ARRIVE guidelines. Int J Rheum Dis. (2015) 18:488–94. doi: 10.1111/1756-185X.12699
26. Collins FS, Tabak LA. Policy: NIH plans to enhance reproducibility. Nature. (2014) 505:612–3. doi: 10.1038/505612a
27. Macleod MR, Fisher M, O'Collins V, Sena ES, Dirnagl U, Bath PMW, et al. Good laboratory practice: preventing introduction of bias at the bench. Stroke. (2009) 40:e50–2. doi: 10.1161/STROKEAHA.108.525386
28. Hooijmans CR, Rovers MM, de Vries RBM, Leenaars M, Ritskes-Hoitinga M, Langendam MW. SYRCLE's risk of bias tool for animal studies. BMC Med Res Methodol. (2014) 14:43. doi: 10.1186/1471-2288-14-43
29. Mattos RM de, Araújo SRR de, Quitzan JG, Leslie B, Bacelar H, Parizi JLG, et al. Can a graft be placed over a flap in complex hypospadias surgery? An experimental study in rabbits. Int Braz J Urol. (2016) 42:1228–36. doi: 10.1590/s1677-5538.ibju.2016.0168
30. Martín-Cano F, Garzón I, Marañés C, Liceras E, Martín-Piedra MA, Ruiz-Montes AM, et al. Histological and immunohistochemical changes in the rat oral mucosa used as an autologous urethral graft. J Pediatr Surg. (2013) 48:1557–64. doi: 10.1016/j.jpedsurg.2013.01.056
31. Kim BS, Kim HT, Kwon SY, Chun SY, Choi KH, Park M, et al. Non-transected ventral onlay-augmented urethroplasty using autologous saphenous vein graft in a rabbit model of urethral stricture. Urology. (2014) 83:225–31. doi: 10.1016/j.urology.2013.08.030
32. Güneş M, Altok M, Özmen Ö, Umul M, Güneş A, Baş E, et al. A novel experimental method for penile augmentation urethroplasty with a combination of buccal mucosa and amniotic membrane in a rabbit model. Urology. (2017) 102:240–6. doi: 10.1016/j.urology.2016.10.061
33. Yapici AK, Uguz S, Bayram Y, Sari S, Karslioglu Y, Guven A, et al. Use of a fibrovascular tube in creation of neo-urethra during penile reconstruction. J Pediatr Urol. (2017) 13:273.e1–e8. doi: 10.1016/j.jpurol.2016.12.026
34. Lalla M, Riis C, Jørgensen CS, Danielsen CC, Jørgensen TM. A biomechanical, histological and biochemical study in an experimental rabbit hypospadias repair model using scanning acoustic microscopy. J Pediatr Urol. (2011) 7:404–11. doi: 10.1016/j.jpurol.2010.07.011
35. Shirazi M, Noorafshan A, Serhan A, Serhan A. Effects of different suture materials used for the repair of hypospadias: a stereological study in a rat model. Urol Int. (2012) 89:395–401. doi: 10.1159/000343423
36. Tan X-H, Liu X, Long C-L, Zhang D-Y, Lin T, He D-W, et al. Histological and biochemical evaluation of urethral scar following three different hypospadias repairs: an experimental study in rabbits. Eur J Pediatr Surg. (2018) 28:420–5. doi: 10.1055/s-0037-1605347
37. Lalla M, Gregersen H, Olsen LH, Jørgensen TM. In vivo biomechanical assessment of anterior rabbit urethra after repair of surgically created hypospadias. J Urol. (2010) 184:675–82. doi: 10.1016/j.juro.2010.03.055
38. Costa M, Leslie B, Rondon A, Bacelar H, Mattos R, Barbosa B, et al. Keratinized versus non-keratinized preputial flap onlay urethroplasty: does it make any difference in the histological analysis? An experimental study in rabbits. Int Braz J Urol. (2013) 39:884–92. doi: 10.1590/S1677-5538.IBJU.2013.06.16
39. Jesus LE, Schanaider A, Kirwan T, Aitken KJ, Caldas MLR, Fonseca E, et al. Reduced flow after tubularized incised plate urethroplasty—increased fibrogenesis, elastin fiber loss or neither? J Urol. (2014) 191:1856–62. doi: 10.1016/j.juro.2013.11.098
40. Zhou Y, Peng J, Cao X, Yan C, Huang F, Shen L, et al. Retardation of preputial wound healing in rats with hypospadias induced by flutamide. J Investig Surg. (2020) 33:164–71. doi: 10.1080/08941939.2018.1483448
41. Shirazi M, Rahimi M, Noorafshan A. Single vs. double layer suturing method repair of the urethral plate in the rabbit model of hypospadias. Cent Eur J Urol. (2016) 69:425–30. doi: 10.5173/ceju.2016.875
42. Oliva P, Delcelo R, Bacelar H, Rondon A, Barroso U Jr, Ortiz V, et al. The buccal mucosa fenestrated graft for Bracka first stage urethroplasty: experimental study in rabbits. Int Braz J Urol. (2012) 38:825–32. doi: 10.1590/1677-553820133806825
43. Gardikis S, Kambouri K, Giatromanolaki A, Agelidou M, Kalaitzis C, Giannakopoulos S, et al. The use of a perimeatal-based penile skin flap to cover the repair of a tubularized incised plate urethroplasty: from experimental surgery to clinical practice. J Pediatr Urol. (2014) 10:469–73. doi: 10.1016/j.jpurol.2013.12.010
44. Xu Y, Shen Z, Liu G, Liu B, Hua X, Xiang Y, et al. Urethral reconstruction using everted saphenous vein graft in a rabbit model: one-year outcomes. Urol Int. (2017) 99:110–7. doi: 10.1159/000464473
45. Vaos G, Gardikis S, Giatromanolaki A, Kambouri K, Trιpsianis G, Ypsilantis P, et al. Long-term angiogenic activity of free grafts and pedicle flap in a rabbit urethroplasty model. World J Urol. (2013) 31:919–24. doi: 10.1007/s00345-012-0875-4
46. Jesus LE, Schanaider A, Patterson G, Marchenko A, Aitken KJ, Leslie B, et al. Urethral compliance in hypospadias operated by tubularized incised urethral plate (TIP) with and without a dorsal inlay graft: an experimental controlled study. World J Urol. (2013) 31:971–5. doi: 10.1007/s00345-012-1004-0
47. Bacelar H, Rondon AV, Mattos R, Quitzan JG, Leslie B, Delcelo R, et al. Onlay foreskin flap anastomosed directly to the tunica albuginea: a short-term experimental study in rabbits. J Pediatr Urol. (2015) 11:274.e1–e6. doi: 10.1016/j.jpurol.2015.04.027
48. Eassa W, He X, El-Sherbiny M. How much does the midline incision add to urethral diameter after tubularized incised plate urethroplasty? An Experimental Animal Study. J Urol. (2011) 186:1625–30. doi: 10.1016/j.juro.2011.03.073
49. Landis SC, Amara SG, Asadullah K, Austin CP, Blumenstein R, Bradley EW, et al. A call for transparent reporting to optimize the predictive value of preclinical research. Nature. (2012) 490:187–91. doi: 10.1038/nature11556
50. Haahr MT, Hróbjartsson A. Who is blinded in randomized clinical trials? A study of 200 trials and a survey of authors. Clin Trials. (2006) 3:360–5. doi: 10.1177/1740774506069153
51. von der Behrens W. Animal models of subjective tinnitus. Neural Plast. (2014) 2014:1–13. doi: 10.1155/2014/741452
52. Prager EM, Bergstrom HC, Grunberg NE, Johnson LR. The importance of reporting housing and husbandry in rat research. Front Behav Neurosci. (2011) 5:38. doi: 10.3389/fnbeh.2011.00038
53. Faggion CM, Schmitter M, Tu Y-K. Assessment of replication of research evidence from animals to humans in studies on peri-implantitis therapy. J Dent. (2009) 37:737–47. doi: 10.1016/j.jdent.2009.06.010
54. Piaggio G, Elbourne DR, Altman DG, Pocock SJ, Evans SJW, CONSORT Group. Reporting of non-inferiority and equivalence randomized trials: an extension of the CONSORT statement. JAMA. (2006) 295:1152–60. doi: 10.1001/jama.295.10.1152
55. Tsang R, Colley L, Lynd LD. Inadequate statistical power to detect clinically significant differences in adverse event rates in randomized controlled trials. J Clin Epidemiol. (2009) 62:609–16. doi: 10.1016/j.jclinepi.2008.08.005
56. Festing MFW, Altman DG. Guidelines for the design and statistical analysis of experiments using laboratory animals. ILAR J. (2002) 43:244–58. doi: 10.1093/ilar.43.4.244
57. Macleod MR, group TNC. Findings of a retrospective, controlled cohort study of the impact of a change in nature journals' editorial policy for life sciences research on the completeness of reporting study design and execution. bioRxiv. (2017) 187245. doi: 10.1101/187245
58. Percie du Sert N, Hurst V, Ahluwalia A, Alam S, Altman DG, Avey MT, et al. Revision of the ARRIVE guidelines: rationale and scope. BMJ Open Sci. (2018) 2:e000002. doi: 10.1136/bmjos-2018-000002
Keywords: hypospadias, animal experiments, quality assessment, clinical translation challenge, translational research
Citation: Abbas TO, Elawad A, Kareem A, Pullattayil S AK, Ali M and Alnaimi A (2021) Preclinical Experiments for Hypospadias Surgery: Systematic Review and Quality Assessment. Front. Pediatr. 9:718647. doi: 10.3389/fped.2021.718647
Received: 01 June 2021; Accepted: 05 July 2021;
Published: 09 August 2021.
Edited by:
Alexander Springer, Medical University of Vienna, AustriaReviewed by:
Einar Olafur Arnbjornsson, Skåne University Hospital, SwedenCopyright © 2021 Abbas, Elawad, Kareem, Pullattayil S, Ali and Alnaimi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Tariq O. Abbas, YWJiYXNAaHN0LmFhdS5kaw==; dGFyaXEyY0Bob3RtYWlsLmNvbQ==; orcid.org/0000-0002-8425-4029
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.