
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pain Res. , 14 March 2025
Sec. Musculoskeletal Pain
Volume 6 - 2025 | https://doi.org/10.3389/fpain.2025.1568227
This article is part of the Research Topic Advancing Intervertebral Disc Degeneration Research: Focus on Annulus Fibrosus and Pain Management View all articles
 Leyu Zhao1,†
Leyu Zhao1,† Qi Yan1,†
Qi Yan1,† Lijie Yuan2,†
Lijie Yuan2,† Tianyi Wu1
Tianyi Wu1 Yun Teng1
Yun Teng1 Junjie Niu1
Junjie Niu1 Dawei Song1
Dawei Song1 Jinning Wang1
Jinning Wang1 Xiao Sun1
Xiao Sun1 Rui Chen1
Rui Chen1 Xianggu Zhong1
Xianggu Zhong1 Jiarong Li3*
Jiarong Li3* Xiaolan Gu2*
Xiaolan Gu2* Jun Zou1*
Jun Zou1*
Background: Lumbar disc herniation (LDH) is a prevalent degenerative disc disorder frequently resulting in lumbar and leg pain. Obese patients with LDH often encounter the scenario where the disc herniation is not completely resolved in the short term following percutaneous transforaminal endoscopic discectomy (PTED), necessitating subsequent surgical intervention, or where long-term reherniation occurs post-procedure. Currently, the literature provides little information regarding the application of annulus fibrosus suture (AFS) as a supplementary measure to PTED for diminishing the recurrence of disc herniation. Our aim was to evaluate the short-term and long-term therapeutic outcomes of combining PTED with AFS, with a particular focus on the impact of AFS on the recurrence rate of disc herniation following PTED.
Methods: We recruited 23 obese patients with single-level LDH diagnosed between December 2021 and December 2023. All patients successfully underwent PTED in conjunction with AFS and the postoperative follow-up. We collected and analyzed data related to baseline parameters, disc degeneration grading, clinical effectiveness, surgery-related factors, lumbar spine function, pain severity, quality of life, and adverse prognosis events.
Results: Compared with preoperative assessments, all patients exhibited significant improvements in Visual Analog Scale for leg pain (VAS-LP), Oswestry Disability Index (ODI), and Japanese Orthopaedic Association (JOA) scores (P < 0.05). During the short-term follow-up period, no patient required a secondary conventional microdiscectomy due to severe complications. At the one-year follow-up, no patient experienced significant recurrent radicular leg pain that would raise suspicion of LDH recurrence. However, when PTED was combined with AFS, the improvement in Visual Analog Scale for back pain (VAS-BP) was relatively less pronounced.
Conclusions: The synergy of PTED and AFS seems to be a comparatively safe and efficacious approach for treating LDH in obese patients. AFS reduces the incidence of long-term recurrent leg pain, which may in turn reduce the probability of LDH recurrence after PTED. Consequently, AFS should be regarded as an efficacious supplementary procedure to PTED, adept at efficiently reducing the recurrence rate in obese individuals with LDH.
The severity of lumbar radiculopathy due to lumbar disc heriation (LDH), primarily characterized by leg pain, significantly impacts patients' daily activities and overall quality of life, often serving as a critical factor in determining the need for surgical intervention (1–4). In patients suffering from persistent sciatica, early discectomy demonstrates superior effectiveness in relieving radicular leg pain compared to conservative treatment (5–7). In comparison to nonoperative treatment modalities, lumbar discectomy has been demonstrated to elicit more robust therapeutic effects with regard to the amelioration of lumbar radiculopathy (8). However, given an average postoperative follow-up period exceeding one year, the incidence of recurrent lumbar disc herniation (rLDH) subsequent to conventional lumbar discectomy remains between 5% and 10%, and can still reach up to 10% in recent years, resulting in enduring postoperative recurrent radicular leg pain and substantially augmenting healthcare expenditures (4, 9–13). percutaneous transforaminal endoscopic discectomy (PTED) is an emerging minimally invasive technique in spinal surgery that demonstrates comparable therapeutic efficacy and safety to conventional lumbar discectomy for treating LDH, offering evident minimally invasive advantages and promoting rapid postoperative recovery (14, 15). However, it fails to demonstrate a statistical advantage in relation to recurrence rate of rLDH and the incidence of postoperative recurrent radicular pain (16, 17). Thus, there is an urgent need in clinical practice for a new approach to diminish the recurrence rate following intervertebral disc surgery. Obesity is frequently considered a risk factor for LDH due to its detrimental impact on spinal biomechanics (18, 19). Moreover, the recurrence rate after discectomy in obese patients can even reach 10% to 15% (20, 21). Therefore, in comparison to non-obese patients, the long-term radicular leg pain linked to postoperative recurrence is a more critical concern for obese patients who have undergone discectomy.
It is currently asserted that annular defects correlate with symptom recurrence and the risk of reoperation, whereas annular competence can predict the recurrence rate following lumbar discectomy for leg pain (22–24). Annular defects not only furnish an anatomical aperture for rLDH but also engender conditions conducive to inflammation-induced neuropathic pain (24–27). Against this backdrop, the urgent development of a suture technique for the torn annulus fibrosus post-lumbar discectomy is essential. In the initial phases of study, the extensive surgical incisions and disruption of the neural environment associated with conventional lumbar discectomy led to considerable postoperative adhesions. Additionally, the lack of devices designed to simplify the suturing process increased the cost of repairing the annulus fibrosus (28, 29). These issues cumulatively led to unsatisfactory therapy outcomes and engendered pessimism about the achievement of the expected benefits of annulus fibrosus repair. Hence, the reparation of annular defects persists in presenting challenges. PTED, characterized by its minimal invasiveness and negligible harm to surrounding anatomical structures, is a novel approach for annular repair. Concurrently, the evolution of the Disposable Annulus Stapler has kindled a glimmer of hope for the therapeutic use of Annulus Fibrosus Suture (AFS). preliminary trials are commencing to assess the therapeutic effectiveness of employing a disposable annulus stapler for the treatment of annular defects subsequent to PTED (30). However, there is seldom literature reporting the effects of AFS on reducing the recurrence rate and alleviating residual pain after PTED. Moreover, a study gap persists about the therapeutic outcomes in obese patients following the synergy of PTED and AFS. To verify our hypothesis that the combination of AFS and PTED may effectively reduce the recurrence rate and alleviate residual pain after PTED in obese patients, we conducted a retrospective study, aiming to evaluate the potential for large-scale clinical application of this combined approach in obese patients with LDH.
This retrospective study included 23 obese patients diagnosed with single-level LDH from December 2021 to December 2023. All patients successfully underwent PTED + AFS surgery. All participants received a comprehensive explanation of the benefits and risks associated with AFS and made informed decisions regarding their surgical intervention. All procedures involving human participants were conducted in accordance with the Declaration of Helsinki. The Institutional Review Board waived the necessity for informed consent because of the retrospective study design.
The criteria for inclusion were as follows: (1) BMI ≥ 30 kg/m2; (2) Clinically diagnosed with single-level lumbar disc herniation; (3) Symptoms including chronic low back pain with radiating pain in the extremities, limited lumbar mobility, and a positive straight leg raise test; (4) Clinical diagnosis of lumbar disc herniation with consistent clinical symptoms, signs, and imaging findings; (5) Poor response or recurrent episodes after 12 weeks of conservative treatment; (6) Complete clinical data before surgery and follow-up records for more than 1 years post-surgery; (7) Patient consent for annular suturing, performed after discectomy with a relatively intact and defect-free annulus fibrosus.
The criteria for exclusion were as follows: (1) Patients with multilevel LDH; (2) Presence of lumbar spondylolisthesis, spinal stenosis, spinal infection, bone tuberculosis, lumbar tumors, or other lumbar diseases; (3) Clinical symptoms and signs inconsistent with imaging findings; (4) History of previous lumbar spine surgery; (5) Severe osteoporosis and significant calcification of the affected segment; (6) Annulus fibrosus rupture unsuitable for suturing; (7) Cognitive or mental disorders.
Patients were positioned prone under local anesthesia. The surgeon performed routine disinfection and draping. A guide needle was put in with the help of C-arm fluoroscopy, and a working cannula was connected to the foraminoscopic system. Under direct visualization with the foraminoscope, the protruding nucleus pulposus was identified. The annulus fibrosus was either incised or the protruding portion was excised from an existing rupture in the annulus fibrosus, with strict protection of the annular fissure during the procedure. An EFit Disposable Annulus Stapler was required for this study. Subsequently, the Disposable Annulus Stapler was inserted into the annulus fibrosus on one side of the defect, taking care to sense the toughness of the annulus and the depth of penetration. Once a sufficient depth was reached, the slider on the tail end of the gun-shaped suturing device was pushed forward until it could no longer be moved. At this point, the transverse anchor device at the head end of the gun-shaped suturing device would gradually emerge from the head end to the deep side of the annulus (Figure 1a). The transverse anchor device would rotate 90 degrees on its own and lock under the annulus fibrosus once it was fully out of the head end of the gun-shaped suturing device (Figure 1b). The gun-shaped annular suturing device was then removed, and the suture was pulled upward to confirm the security of the first stitch (Figure 1c). Another annular suturing device was used to puncture the other side of the defect. The head end of the second annular suturing device would have a suture loop. Before performing the second puncture, the suture of the first stitch was threaded through this loop (Figure 1d). The procedure for the second puncture mirrored that of the initial one, except that the deployment of the transverse anchor device was facilitated by a push rod (Figures 1e,f). Since the suture from the first stitch had been previously passed through the suture loop of the second stitch, tightening the suture was sufficient to secure the first knot (Figure 1g). Subsequently, a second knot was formed outside the body, and an endoscopic knot pusher, specifically designed for minimally invasive procedures, was employed to advance the second knot toward the first, ensuring a robust ligation (Figures 1h,i). After irrigating the incision with 0.9% sodium chloride solution and achieving hemostasis with radiofrequency, the working channel was removed, followed by routine suturing and dressing of the incision. Postoperatively, patients routinely received mecobalamin, mannitol, and other anti-inflammatory, dehydrating, and analgesic treatments. On the first postoperative day, all patients were guided to perform in-bed functional exercises, including straight-leg raising exercises and five-point or three-point support exercises. Medical staff also assisted patients in attempting ambulation. Patients were informed that they could begin swallowtail (fly-like) functional exercises on the third postoperative day. To ensure the scientific rigor of postoperative rehabilitation training after discharge, all patients received educational instructions from our team prior to discharge. Surgeons with similar experience and skill recorded the surgical time, postoperative hospital stay, incision length, working channel placement time, and intraoperative blood loss for both groups.
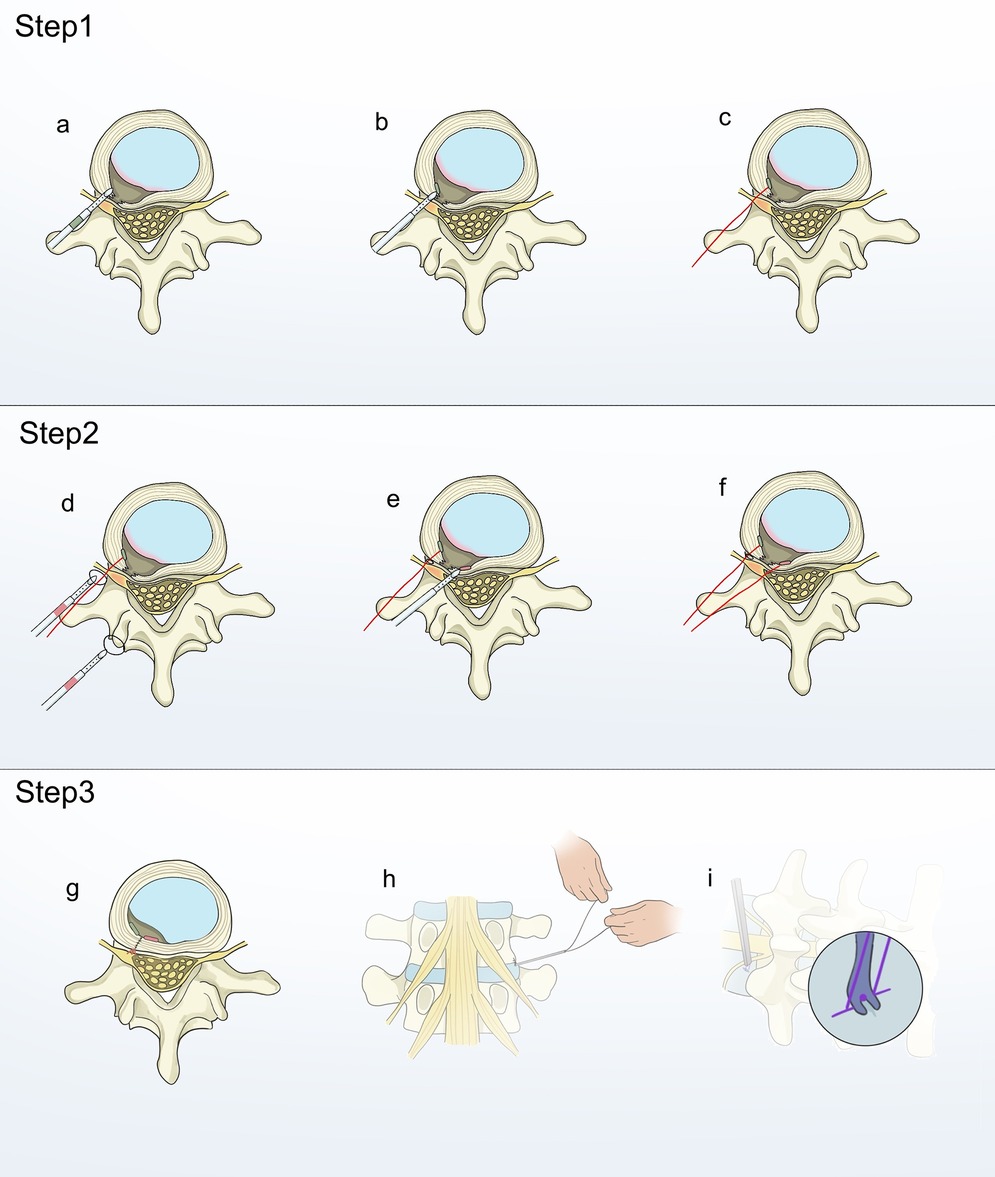
Figure 1. Step 1: Schematic diagram of annulus defect closure using a double-stitch suture technique. The disposable annulus stapler was inserted into the annulus on one side of the defect (a) The slider at the tail end of the gun-type suturing device was pushed forward to perform transverse anchoring (b) The gun-type annulus fibrosus suturing device was withdrawn, and the suture was pulled upward to confirm the security of the first stitch (c) Step 2: The suture loop of the second stitch was passed over the suture of the first stitch (d) The second stitch performed transverse anchoring (e,f). Step 3:The suture was tightened to secure the first knot (g) Form the second knot externally and use a minimally invasive endoscopic knot pusher to advance it towards the first knot (h,i).
This study used the Pfirrmann grading system to assess the degree of intervertebral disc degeneration in patients prior to surgery. We used the Modified MacNab Criteria to statistically assess the clinical outcomes at one year postoperatively. The ODI and JOA scores at one month, three months, six months, and one year after surgery served as indicators to evaluate the recovery of lumbar spine function. The VAS was utilized to assess postoperative low back pain (VAS-BP) and leg pain (VAS-LP). Special attention was paid to the incidence of severe short-term complications and the recurrence rate at six months postoperatively. A recurrence of lumbar disc herniation was suspected to have occurred if, compared to the condition at one month postoperatively, patients experienced aggravated low back or leg pain, or a decline in lumbar spine function.
The software utilized for statistical analysis was IBM SPSS Statistics 25.0 (IBM Corp, Armonk, NY, USA). Repeated measures analysis of variance (ANOVA) was conducted to compare the mean scores of VAS-LP, VAS-BP, JOA score, and ODI in patients from preoperatively to postoperatively over time. The significance threshold was rigorously set at a p-value of 0.05. Statistical graphs were created using GraphPad Prism version 10.0.
23 obese patients underwent PTED combined with AFS for the treatment of LDH. Clinical data were collected before and after the procedure. The study included 11 male patients (47.83%) and 12 female patients (52.17%), with ages ranging from 21 to 61 years. All patients were diagnosed with single-level LDH. Preoperatively, 9 patients (39.13%) were graded as Pfirrmann Grade III, and 14 patients (60.87%) were graded as Pfirrmann Grade IV (Tables 1,2).
All patients successfully underwent PTED + AFS surgical intervention, with no significant surgical accidents occurring during the surgery (Table 3). Preoperative MRI (L5-S1) and intraoperative data of the representative case are shown in the figures (Figure 2).
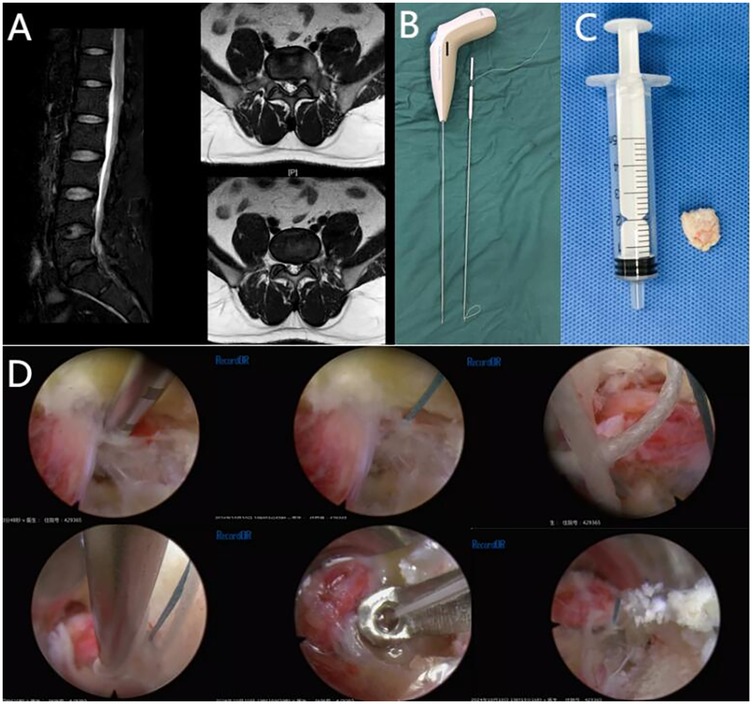
Figure 2. Preoperative MRI (L5-S1) and intraoperative data of the representative case are shown in the figures. (A) Preoperative MRI images of a 43-year-old obese patient with lumbar disc herniation (L5-S1). (B) The Disposable Annulus Stapler. (C) The nucleus pulposus removed from the patient during PTED. (D) Post-PTED double-stitch suture technique for annulus defect closure.
Repeated-measures ANOVA was conducted on VAS-LP, VAS-BP, JOA, and ODI scores, and it showed that there were significant differences between time points (P < 0.05). Further LSD multiple comparisons indicated that scores at postoperative month 1 were lower than preoperative levels, scores at postoperative month 3 were lower than both preoperative and postoperative month 1 levels. There were no differences between postoperative months 6 and 12 in VAS-LP scores, although both were lower than preoperative, postoperative month 1, and postoperative month 3 levels (Figure 3A). B. For VAS-BP, there were no differences among postoperative months 1, 3, 6, and 12, but all were lower than preoperative levels (Figure 3B). Regarding ODI scores, there were no differences between postoperative months 6 and 12, but both were higher than preoperative, postoperative month 1, and postoperative month 3 levels. Additionally, scores at postoperative month 3 were higher than both preoperative and postoperative month 1 levels, with no difference between postoperative months 6 and 12, although both were higher than preoperative, postoperative month 1, and postoperative month 3 levels (Figure 3C). JOA scores were higher than preoperative levels at postoperative month 1, higher than both preoperative and postoperative month 1 levels at postoperative month 3, and higher than preoperative, postoperative month 1, and postoperative month 12 levels at postoperative month 6 (Figure 3D).
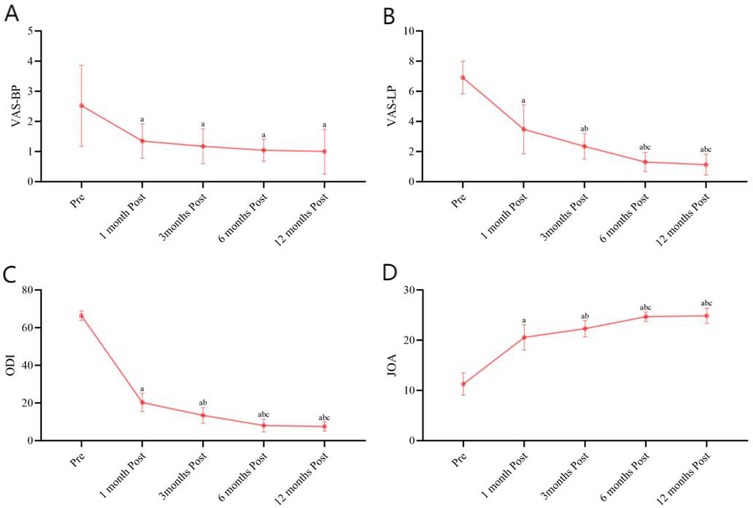
Figure 3. Preoperative and postoperative (1, 3, 6, and 12 months) VAS-LP, VAS-BP, ODI, and JOA scores. (A) Preoperative and postoperative (1, 3, 6, and 12 months) VAS-BP. (B) Preoperative and postoperative (1, 3, 6, and 12 months) VAS-LP. (C) Preoperative and postoperative (1, 3, 6, and 12 months) ODI. (D) Preoperative and postoperative (1, 3, 6, and 12 months) JOA scores.
After one year of follow-up, the excellent and good rate of PTED combined with AFS can reach 91.30% (Table 4). It is worth mentioning that we observed no patients experiencing severe short-term complications requiring secondary surgical intervention, and no patients presented with severe recurrent radicular pain at the one-year follow-up.
Irrespective of the surgical approach—be it conventional lumbar discectomy, microsurgical lumbar discectomy, or the minimally invasive PTED—patients may still be at risk for recurrent LDH (rLDH) and the sequelae of persistent postoperative disability and pain, which can substantially impair their quality of life (31–33).The competence of the annulus is considered an important factor influencing the risk of rLDH after discectomy, and larger annular defects may be associated with accelerated disc height loss postoperatively (22, 34, 35). In fact, annular defects left after discectomy often heal poorly. Peripheral healing in injured discs may not occur, or if it does, it may only result in a thin layer of tissue (36). In the presence of a highly hydrated nucleus pulposus, the increased tensile stress on the inner annulus fibrosus associated with the damaged outer layer following discectomy initially leads to deformation and bulging of the collagen bundles, and ultimately results in the inner extension and persistent opening of the tear (37). This limited healing allows for the re-diffusion of nuclear irritants to surrounding neural structures under even slight increases in intradiscal pressure. When the annular defect fails to close effectively, the swelling pressure of the annulus fibrosus decreases with degeneration and injury. The loss of swelling pressure leads to increased matrix deformation, weakened strength and elasticity of the fiber structure, and the inability of the intervertebral disc (IVD) to effectively absorb and distribute compressive loads, thereby increasing the risk of rLDH (38). The integrity of the lamellar structure of the annulus fibrosus ensures its anisotropic and inhomogeneous characteristics. Annular defects may lead to weakened or disrupted interactions between fiber populations in the multilamellae annulus fibrosus, resulting in decreased tensile strength and stiffness, while increasing its deformability. Consequently, the annulus becomes more susceptible to deformation under mechanical overload, potentially further preventing the closure of the annular defect (39–41). Radial annular defects or posterior disc protrusions can lead to a decrease in the pressure of the adjacent disc nucleus, while simultaneously doubling the stress peaks within the annulus fibrosus. This accentuates the transfer of load from nucleus to annulus, further compromising the structural integrity of the annulus fibrosus (42). Thus, this creates a vicious cycle in which the presence of annular defects keeps making the loss of annular competence worse, which is bad for patients' long-term outcomes after discectomy.
Obesity, defined by a Body Mass Index (BMI) ≥ 30 kg/m² (43), is also widely recognized as a risk factor for recurrent lumbar disc herniation (rLDH) following various surgical interventions, including percutaneous transforaminal endoscopic discectomy (PTED) (18, 44, 45). Existing literature has confirmed that obesity is associated with reduced lumbar disc height (46, 47). The increased body weight in obese individuals exerts greater forces on spinal motion segments, particularly the lumbar spine, thereby delaying the healing of the annulus fibrosus through mechanical overload. In obese patients, increased flexion of the sacroiliac joints leads to degeneration of the joint surfaces. The altered biomechanical stability generates high torque, which can tear the annular structure (48–52). The disordered internal environment further affects the biosynthesis of extracellular matrix components in the outer region of the intervertebral disc (53–56). Therefore, after lumbar discectomy, the incision site of the annulus fibrosus in obese patients may become a weak point due to scar formation or non-healing, leading to reherniation of the nucleus pulposus. Based on these findings, we propose that the incidence of rLDH is higher in obese patients than in the general population (Figure 4). Thus, we selected obese patients as the subjects of our study, which is more in line with clinical needs.
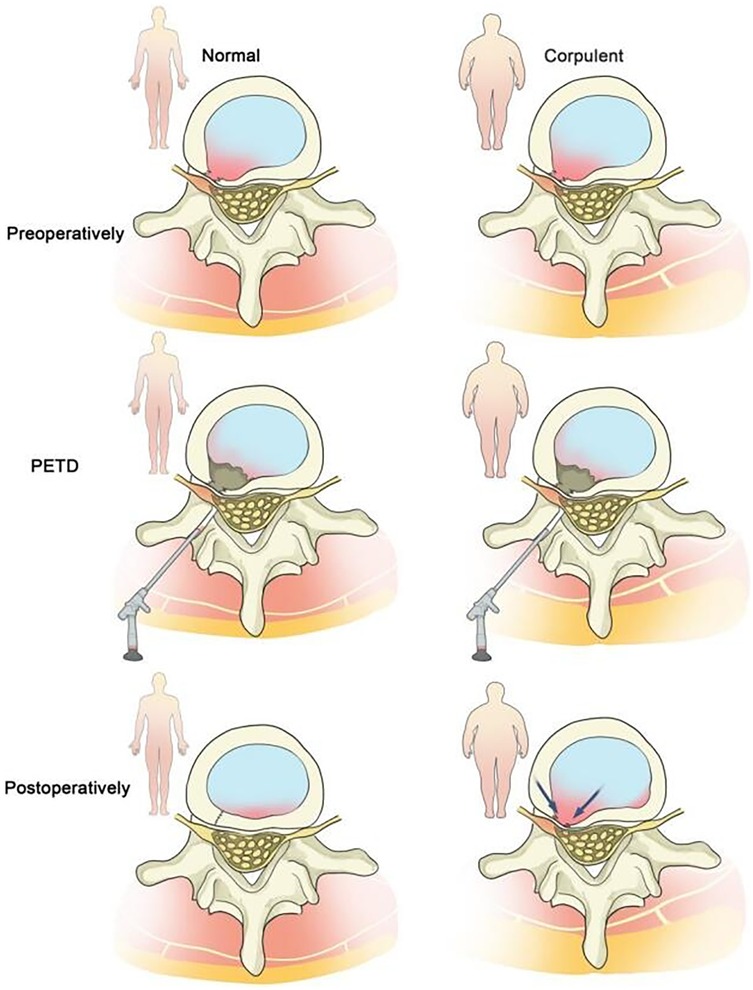
Figure 4. Obese patients are more prone to rLDH after PTED compared to patients with normal body weight.
The suture technique for a ruptured annulus was initially referenced in a sheep model study, where it was hypothesized that direct repair of annular incisions through tight suturing in sheep undergoing lumbar discectomy would not significantly affect the healing strength of the IVD (29). The advent of a simplified anular repair device heralds a new dawn for the practical clinical application of suturing the annular defects. Regrettably, in the study conducted in 2013, although suturig the annular defects does not adversely affect the positive patient outcomes inherent after a lumbar discectomy, the data are insufficient to conclusively demonstrate the effectiveness of suturing the annular defects in reducing the need for subsequent reoperations due to rLDH (28). At that juncture, the suturing technique is not yet fully developed, and the lack of a more simplified suturing device greatly increases the cost of repairing the annulus defects. This situation, where the benefits gained are not proportional to the costs paid, makes it uncertain whether the suturing of annular defects has achieved its anticipated benefits. In fact, due to the large surgical wound and extensive exposure range associated with conventional lumbar discectomy, there is significant destruction to the surrounding neural environment, often leading to extensive adhesions postoperatively. Under such conditions, the suturing technique is akin to a drop in the ocean. It is essential to emphasize that suturing the annular defects may merely enhance resistance to pressure through mechanical blocking, rather than by promoting healing. The healing of the annulus is a gradual process, not driven by direct repair. Significant damage to the normal anatomy largely overshadows the protective role of suturing in the gradual healing process. PTED is a way to remove protruding or trapped disc fragments without doing a full discectomy. It helps avoid the risks of spinal instability that come with doing a full discectomy in traditional LDH surgeries. The safety and efficacy of PTED have been continuously improved with the effective prevention of various complications arising from the ongoing refinement of PTED techniques (57–59). PTED exhibits comparability to traditional lumbar discectomy with respect to therapeutic efficacy and safety in the treatment of LDH, possessing evident minimally invasive benefits and facilitating rapid postoperative recovery. However, it doesn't show a statistically significant advantage over lumbar discectomy when it comes to the rate of recurrence and the number of cases of recurrent radicular pain after surgery. To address the issue of rLDH after PTED, there are currently only a few reports on the use of the Disposable Annulus Stapler to address annular defects following PTED. It remains uncertain whether the AFS can be widely and effectively applied in clinical practice to significantly reduce the incidence of rLDH. In a network meta-analysis conducted in 2021, researchers found that in the SUCRA plot for pain scores, percutaneous endoscopic discectomy and annulus fibrosus repair ranked second and third, respectively, just after open discectomy (60). This not only highlights the significant potential of PTED combined with AFS in reducing the recurrence of disc herniation after discectomy, but also provides inspiration for our research.
Previous studies have suggested that obesity is a significant factor for recurrence after PTED and can also affect the incidence of postoperative disc reherniation. Data from these research suggest that the long-term recurrence rate after PTED in obese patients can reach −5–10 percent within a one-year follow-up period (61–63). The proportion of obese patients who experience severe pain immediately postoperatively due to incomplete decompression of lumbar disc herniation on the nerve root, necessitating intervention with conventional microdiscectomy on the same postoperative day, can even reach up to 10% (63). The data from these references have provided prospective conditions for our research. In this retrospective study, our suturing technique differs from the conventional single-stitch method by incorporating the Disposable Annulus Stapler. Specifically, the loop of the second stitch is pre-threaded over the suture of the first stitch, enabling the first knot to be secured simply by tightening the suture. This ensures the stability of the suture knot. This approach reduces the prognostic variability associated with technical errors in traditional single-stitch suturing and significantly decreases the time required for physicians to learn the suturing technique. As a result, it ensures the reliability of annular fibrosus suturing within a shorter timeframe, thereby enhancing the reliability of the study outcomes. In our investigation, we observed that obese patients who underwent PTED combined with AFS did not require secondary traditional discectomy interventions within the short term, as previously reported in the literature. Additionally, no instances of recurrence were documented throughout the one-year follow-up duration. Individuals who received PTED in conjunction with AFS exhibited marked alleviation of postoperative pain and substantial enhancements in quality of life, with no instances of reherination. Our findings further substantiate the reliability of PTED combined with AFS in treating LDH and reducing the incidence of recurrent radicular leg pain following PTED. The synergy of AFS and PTED is fundamentally attributed to its ability to address the underlying cause of LDH. The procedure facilitates early closure of the annular defects, which prevents residual nucleus pulposus in obese patients from re-herniating through the annular defect under mechanical stress and reduces the release of inflammatory mediators within the disc that could irritate nerve roots. Therefore, closing the annular defects not only reduces the short-term recurrence of disc herniation after surgery but also effectively decreases the mechanical irritation of the ruptured and bulging annulus on the nerves, thereby alleviating the postoperative residual symptoms of low back pain and leg pain associated with recurrent disc herniation.
The limitations of this study include the lack of a control group and insufficient sample size, which means that the accuracy and reliability of the results should be viewed with caution. Another limitation stems from the heterogeneity between this study and previous studies. Although the heterogeneity of the participant population itself is considered relatively low, such indirect comparisons may lead to further imprecision.
PTED combined with AFS alleviated patients' pain, improved their functional outcomes, and significantly reduced the likelihood of severe short-term complications and long-term recurrent leg pain during a 12-month observation period.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding authors.
The studies involving humans were approved by the Medical Research Ethics Committee of the First Affiliated Hospital of Soochow University. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
LZ: Writing – original draft, Data curation, Formal analysis, Investigation. QY: Data curation, Formal analysis, Investigation, Writing – original draft. LY: Data curation, Formal analysis, Investigation, Writing – original draft. TW: Conceptualization, Methodology, Writing – review & editing. YT: Conceptualization, Methodology, Writing – review & editing. JN: Conceptualization, Methodology, Writing – review & editing. DS: Conceptualization, Methodology, Writing – review & editing. JW: Conceptualization, Methodology, Writing – review & editing. XS: Conceptualization, Methodology, Writing – review & editing. RC: Conceptualization, Methodology, Writing – review & editing. XZ: Conceptualization, Methodology, Writing – review & editing. JL: Conceptualization, Data curation, Methodology, Project administration, Supervision, Writing – review & editing. XG: Conceptualization, Data curation, Methodology, Project administration, Supervision, Writing – review & editing. JZ: Conceptualization, Data curation, Methodology, Project administration, Supervision, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the National Key Research and Development Program of China (Grant Nos: 2022YFC2009700).
We thank all the staff and patients who participated in this study.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpain.2025.1568227/full#supplementary-material
1. Hasvik E, Haugen AJ, Grøvle L. Symptom descriptors and patterns in lumbar radicular pain caused by disc herniation: a 1-year longitudinal cohort study. BMJ Open. (2022) 12(12):e065500. doi: 10.1136/bmjopen-2022-065500
2. Liu C, Ferreira GE, Abdel Shaheed C, Chen Q, Harris IA, Bailey CS, et al. Surgical versus non-surgical treatment for sciatica: systematic review and meta-analysis of randomised controlled trials. Br Med J. (2023) 381:e070730. doi: 10.1136/bmj-2022-070730
3. Porchet F, Wietlisbach V, Burnand B, Daeppen K, Villemure JG, Vader JP. Relationship between severity of lumbar disc disease and disability scores in sciatica patients. Neurosurgery. (2002) 50(6):1253–9. discussion 9-60. doi: 10.1097/00006123-200206000-00014
4. Rogerson A, Aidlen J, Jenis LG. Persistent radiculopathy after surgical treatment for lumbar disc herniation: causes and treatment options. Int Orthop. (2019) 43(4):969–73. doi: 10.1007/s00264-018-4246-7
5. Bailey CS, Rasoulinejad P, Taylor D, Sequeira K, Miller T, Watson J, et al. Surgery versus conservative care for persistent sciatica lasting 4 to 12 months. N Engl J Med. (2020) 382(12):1093–102. doi: 10.1056/NEJMoa1912658
6. Carr JC. In persistent sciatica and lumbar disc herniation, surgery vs. conservative care reduced leg pain at 6 mo. Ann Intern Med. (2020) 173(4):Jc20. doi: 10.7326/ACPJ202008180-020
7. Atlas SJ, Keller RB, Wu YA, Deyo RA, Singer DE. Long-term outcomes of surgical and nonsurgical management of sciatica secondary to a lumbar disc herniation: 10 year results from the maine lumbar spine study. Spine. (2005) 30(8):927–35. doi: 10.1097/01.brs.0000158954.68522.2a
8. Weinstein JN, Lurie JD, Tosteson TD, Skinner JS, Hanscom B, Tosteson AN, et al. Surgical vs nonoperative treatment for lumbar disc herniation: the spine patient outcomes research trial (SPORT) observational cohort. Jama. (2006) 296(20):2451–9. doi: 10.1001/jama.296.20.2451
9. Benzakour A, Benzakour T. Lumbar disc herniation: long-term outcomes after mini-open discectomy. Int Orthop. (2020) 44(2):409. doi: 10.1007/s00264-019-04457-0
10. Ambrossi GL, McGirt MJ, Sciubba DM, Witham TF, Wolinsky JP, Gokaslan ZL, et al. Recurrent lumbar disc herniation after single-level lumbar discectomy: incidence and health care cost analysis. Neurosurgery. (2009) 65(3):574–8. discussion 8. doi: 10.1227/01.NEU.0000350224.36213.F9
11. Jönsson B, Strömqvist B. Clinical characteristics of recurrent sciatica after lumbar discectomy. Spine. (1996) 21(4):500–5. doi: 10.1097/00007632-199602150-00017
12. McGirt MJ, Ambrossi GL, Datoo G, Sciubba DM, Witham TF, Wolinsky JP, et al. Recurrent disc herniation and long-term back pain after primary lumbar discectomy: review of outcomes reported for limited versus aggressive disc removal. Neurosurgery. (2009) 64(2):338–44. discussion 44-5. doi: 10.1227/01.NEU.0000337574.58662.E2
13. Davis RA. A long-term outcome analysis of 984 surgically treated herniated lumbar discs. J Neurosurg. (1994) 80(3):415–21. doi: 10.3171/jns.1994.80.3.0415
14. Gadjradj PS, Rubinstein SM, Peul WC, Depauw PR, Vleggeert-Lankamp CL, Seiger A, et al. Full endoscopic versus open discectomy for sciatica: randomised controlled non-inferiority trial. Br Med J. (2022) 376:e065846. doi: 10.1136/bmj-2021-065846
15. Chen Q, Zhang Z, Liu B, Liu S. Evaluation of percutaneous transforaminal endoscopic discectomy in the treatment of lumbar disc herniation: a retrospective study. Orthop Surg. (2021) 13(2):599–607. doi: 10.1111/os.12839
16. Zhao XM, Chen AF, Lou XX, Zhang YG. Comparison of three common intervertebral disc discectomies in the treatment of lumbar disc herniation: a systematic review and meta-analysis based on multiple data. J Clin Med. (2022) 11(22):6604. doi: 10.3390/jcm11226604
17. Gu YT, Cui Z, Shao HW, Ye Y, Gu AQ. Percutaneous transforaminal endoscopic surgery (PTES) for symptomatic lumbar disc herniation: a surgical technique, outcome, and complications in 209 consecutive cases. J Orthop Surg Res. (2017) 12(1):25. doi: 10.1186/s13018-017-0524-0
18. Zileli M, Oertel J, Sharif S, Zygourakis C. Lumbar disc herniation: prevention and treatment of recurrence: WFNS spine committee recommendations. World Neurosurg X. (2024) 22:100275. doi: 10.1016/j.wnsx.2024.100275
19. Takatalo J, Karppinen J, Taimela S, Niinimäki J, Laitinen J, Sequeiros RB, et al. Association of abdominal obesity with lumbar disc degeneration–a magnetic resonance imaging study. PLoS One. (2013) 8(2):e56244. doi: 10.1371/journal.pone.0056244
20. Bae JS, Lee SH. Transforaminal full-endoscopic lumbar discectomy in obese patients. Int J Spine Surg. (2016) 10:18. doi: 10.14444/3018
21. Meredith DS, Huang RC, Nguyen J, Lyman S. Obesity increases the risk of recurrent herniated nucleus pulposus after lumbar microdiscectomy. Spine J. (2010) 10(7):575–80. doi: 10.1016/j.spinee.2010.02.021
22. Carragee EJ, Han MY, Suen PW, Kim D. Clinical outcomes after lumbar discectomy for sciatica: the effects of fragment type and anular competence. J Bone Joint Surg Am. (2003) 85(1):102–8. doi: 10.2106/00004623-200301000-00016
23. Carragee EJ, Spinnickie AO, Alamin TF, Paragioudakis S. A prospective controlled study of limited versus subtotal posterior discectomy: short-term outcomes in patients with herniated lumbar intervertebral discs and large posterior anular defect. Spine. (2006) 31(6):653–7. doi: 10.1097/01.brs.0000203714.76250.68
24. Buttermann GR. Treatment of lumbar disc herniation: epidural steroid injection compared with discectomy. A prospective, randomized study. J Bone Joint Surg Am. (2004) 86(4):670–9. doi: 10.2106/00004623-200404000-00002
25. Freemont AJ, Peacock TE, Goupille P, Hoyland JA, O'Brien J, Jayson MI. Nerve ingrowth into diseased intervertebral disc in chronic back pain. Lancet. (1997) 350(9072):178–81. doi: 10.1016/S0140-6736(97)02135-1
26. Peng Y, Chen X, Rao Z, Wu W, Zuo H, Chen K, et al. Multifunctional annulus fibrosus matrix prevents disc-related pain via inhibiting neuroinflammation and sensitization. Acta Biomater. (2023) 170:288–302. doi: 10.1016/j.actbio.2023.08.028
27. Kirnaz S, Capadona C, Lintz M, Kim B, Yerden R, Goldberg JL, et al. Pathomechanism and biomechanics of degenerative disc disease: features of healthy and degenerated discs. Int J Spine Surg. (2021) 15(s1):10–25. doi: 10.14444/8052
28. Bailey A, Araghi A, Blumenthal S, Huffmon GV. Prospective, multicenter, randomized, controlled study of anular repair in lumbar discectomy: two-year follow-up. Spine. (2013) 38(14):1161–9. doi: 10.1097/BRS.0b013e31828b2e2f
29. Ahlgren BD, Lui W, Herkowitz HN, Panjabi MM, Guiboux JP. Effect of anular repair on the healing strength of the intervertebral disc: a sheep model. Spine. (2000) 25(17):2165–70. doi: 10.1097/00007632-200009010-00004
30. Li ZZ, Cao Z, Zhao HL, Shang WL, Hou SX. A pilot study of full-endoscopic annulus fibrosus suture following lumbar discectomy: technique notes and one-year follow-up. Pain Physician. (2020) 23(5):E497–e506.32967400
31. Ebeling U, Reichenberg W, Reulen HJ. Results of microsurgical lumbar discectomy. Review on 485 patients. Acta Neurochir. (1986) 81(1-2):45–52. doi: 10.1007/BF01456264
32. den Boer JJ, Oostendorp RA, Beems T, Munneke M, Evers AW. Continued disability and pain after lumbar disc surgery: the role of cognitive-behavioral factors. Pain. (2006) 123(1-2):45–52. doi: 10.1016/j.pain.2006.02.008
33. Tacconi L, Baldo S, Merci G, Serra G. Transforaminal percutaneous endoscopic lumbar discectomy: outcome and complications in 270 cases. J Neurosurg Sci. (2020) 64(6):531–6. doi: 10.23736/S0390-5616.18.04395-3
34. McGirt MJ, Eustacchio S, Varga P, Vilendecic M, Trummer M, Gorensek M, et al. A prospective cohort study of close interval computed tomography and magnetic resonance imaging after primary lumbar discectomy: factors associated with recurrent disc herniation and disc height loss. Spine. (2009) 34(19):2044–51. doi: 10.1097/BRS.0b013e3181b34a9a
35. Bono CM, Leonard DA, Cha TD, Schwab JH, Wood KB, Harris MB, et al. The effect of short (2-weeks) versus long (6-weeks) post-operative restrictions following lumbar discectomy: a prospective randomized control trial. Eur Spine J. (2017) 26(3):905–12. doi: 10.1007/s00586-016-4821-9
36. Hampton D, Laros G, McCarron R, Franks D. Healing potential of the anulus fibrosus. Spine. (1989) 14(4):398–401. doi: 10.1097/00007632-198904000-00009
37. Osti OL, Vernon-Roberts B, Fraser RD. Volvo award in experimental studies. Anulus tears and intervertebral disc degeneration. An experimental study using an animal model. Spine. (1990) 15(8):762–7. doi: 10.1097/00007632-199008010-00005
38. Iatridis JC, Setton LA, Foster RJ, Rawlins BA, Weidenbaum M, Mow VC. Degeneration affects the anisotropic and nonlinear behaviors of human anulus fibrosus in compression. J Biomech. (1998) 31(6):535–44. doi: 10.1016/S0021-9290(98)00046-3
39. Elliott DM, Setton LA. Anisotropic and inhomogeneous tensile behavior of the human anulus fibrosus: experimental measurement and material model predictions. J Biomech Eng. (2001) 123(3):256–63. doi: 10.1115/1.1374202
40. Iatridis JC, Setton LA, Weidenbaum M, Mow VC. Alterations in the mechanical behavior of the human lumbar nucleus pulposus with degeneration and aging. J Orthop Res. (1997) 15(2):318–22. doi: 10.1002/jor.1100150224
41. Iatridis JC, MacLean JJ, Roughley PJ, Alini M. Effects of mechanical loading on intervertebral disc metabolism in vivo. J Bone Joint Surg Am. (2006) 88(0 2):41–6. doi: 10.2106/JBJS.E.01407
42. Adams MA, McNally DS, Dolan P. ’Stress' distributions inside intervertebral discs. The effects of age and degeneration. J Bone Joint Surg Br. (1996) 78(6):965–72. doi: 10.1302/0301-620X.78B6.0780965
43. NHLBI Obesity Education Initiative Expert Panel on the Identification, Evaluation, and Treatment of Obesity in Adults (US). Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults–the evidence report. National institutes of health. Obes Res. (1998) 6(Suppl 2):51s–209.9813653
44. Luo M, Wang Z, Zhou B, Yang G, Shi Y, Chen J, et al. Risk factors for lumbar disc herniation recurrence after percutaneous endoscopic lumbar discectomy: a meta-analysis of 58 cohort studies. Neurosurg Rev. (2023) 46(1):159. doi: 10.1007/s10143-023-02041-0
45. Siccoli A, Staartjes VE, Klukowska AM, Muizelaar JP, Schröder ML. Overweight and smoking promote recurrent lumbar disc herniation after discectomy. Eur Spine J. (2022) 31(3):604–13. doi: 10.1007/s00586-022-07116-y
46. Urquhart DM, Kurniadi I, Triangto K, Wang Y, Wluka AE, O'Sullivan R, et al. Obesity is associated with reduced disc height in the lumbar spine but not at the lumbosacral junction. Spine. (2014) 39(16):E962–6. doi: 10.1097/BRS.0000000000000411
47. Samartzis D, Karppinen J, Chan D, Luk KD, Cheung KM. The association of lumbar intervertebral disc degeneration on magnetic resonance imaging with body mass index in overweight and obese adults: a population-based study. Arthritis Rheum. (2012) 64(5):1488–96. doi: 10.1002/art.33462
48. Onyemaechi NO, Anyanwu GE, Obikili EN, Onwuasoigwe O, Nwankwo OE. Impact of overweight and obesity on the musculoskeletal system using lumbosacral angles. Patient Prefer Adherence. (2016) 10:291–6. doi: 10.2147/PPA.S90967
49. Evcik D, Yücel A. Lumbar lordosis in acute and chronic low back pain patients. Rheumatol Int. (2003) 23(4):163–5. doi: 10.1007/s00296-002-0268-x
50. Sheng B, Feng C, Zhang D, Spitler H, Shi L. Associations between obesity and spinal diseases: a medical expenditure panel study analysis. Int J Environ Res Public Health. (2017) 14(2):183. doi: 10.3390/ijerph14020183
51. Videman T, Levälahti E, Battié MC. The effects of anthropometrics, lifting strength, and physical activities in disc degeneration. Spine. (2007) 32(13):1406–13. doi: 10.1097/BRS.0b013e31806011fa
52. Wang D, Lai A, Gansau J, Nasser P, Lee Y, Laudier DM, et al. Ex vivo biomechanical evaluation of acute lumbar endplate injury and comparison to annulus fibrosus injury in a rat model. J Mech Behav Biomed Mater. (2022) 131:105234. doi: 10.1016/j.jmbbm.2022.105234
53. Elmasry S, Asfour S, de Rivero Vaccari JP, Travascio F. A computational model for investigating the effects of changes in bioavailability of insulin-like growth factor-1 on the homeostasis of the intervertebral disc. Comput Biol Med. (2016) 78:126–37. doi: 10.1016/j.compbiomed.2016.09.016
54. Cannata F, Vadalà G, Ambrosio L, Fallucca S, Napoli N, Papalia R, et al. Intervertebral disc degeneration: a focus on obesity and type 2 diabetes. Diabetes Metab Res Rev. (2020) 36(1):e3224. doi: 10.1002/dmrr.3224
55. Tilg H, Moschen AR. Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nat Rev Immunol. (2006) 6(10):772–83. doi: 10.1038/nri1937
56. Ruiz-Fernández C, Francisco V, Pino J, Mera A, González-Gay MA, Gómez R, et al. Molecular relationships among obesity, inflammation and intervertebral disc degeneration: are adipokines the common link? Int J Mol Sci. (2019) 20(8):2030. doi: 10.3390/ijms20082030
57. Abdullah AF, Ditto EW 3rd, Byrd EB, Williams R. Extreme-lateral lumbar disc herniations. Clinical syndrome and special problems of diagnosis. J Neurosurg. (1974) 41(2):229–34. doi: 10.3171/jns.1974.41.2.0229
58. Lew SM, Mehalic TF, Fagone KL. Transforaminal percutaneous endoscopic discectomy in the treatment of far-lateral and foraminal lumbar disc herniations. J Neurosurg. (2001) 94(2 Suppl):216–20. doi: 10.3171/spi.2001.94.2.0216
59. Ahn Y. Transforaminal percutaneous endoscopic lumbar discectomy: technical tips to prevent complications. Expert Rev Med Devices. (2012) 9(4):361–6. doi: 10.1586/erd.12.23
60. Rickers KW, Pedersen PH, Tvedebrink T, Eiskjær SP. Comparison of interventions for lumbar disc herniation: a systematic review with network meta-analysis. Spine J. (2021) 21(10):1750–62. doi: 10.1016/j.spinee.2021.02.022
61. Yin S, Du H, Yang W, Duan C, Feng C, Tao H. Prevalence of recurrent herniation following percutaneous endoscopic lumbar discectomy: a meta-analysis. Pain Physician. (2018) 21(4):337–50.30045591
62. Kim HS, You JD, Ju CI. Predictive scoring and risk factors of early recurrence after percutaneous endoscopic lumbar discectomy. Biomed Res Int. (2019) 2019:6492675. doi: 10.1155/2019/6492675
Keywords: annulus fibrosus suture, percutaneous transforaminal endoscopic discectomy, obesity, lumbar radiculopathy, lumbar disc herniation
Citation: Zhao L, Yan Q, Yuan L, Wu T, Teng Y, Niu J, Song D, Wang J, Sun X, Chen R, Zhong X, Li J, Gu X and Zou J (2025) The effcet of annulus fibrosus suture combined with percutaneous transforaminal endoscopic discectomy on obese patients with lumbar disc herniation. Front. Pain Res. 6:1568227. doi: 10.3389/fpain.2025.1568227
Received: 29 January 2025; Accepted: 3 March 2025;
Published: 14 March 2025.
Edited by:
Jiawei Lu, Tongji University, ChinaCopyright: © 2025 Zhao, Yan, Yuan, Wu, Teng, Niu, Song, Wang, Sun, Chen, Zhong, Li, Gu and Zou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jun Zou, anpvdUBzdWRhLmVkdS5jbg==; Xiaolan Gu, MTg2NjIxMDAwMDdAc29odS5jb20=; Jiarong Li, bGVlanI4MEAxNjMuY29t
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.