
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Oncol. , 27 March 2025
Sec. Gastrointestinal Cancers: Hepato Pancreatic Biliary Cancers
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1551038
 Koushiro Ohtsubo1*
Koushiro Ohtsubo1* Shigeki Sato1
Shigeki Sato1 Hiroyuki Sakaguchi1
Hiroyuki Sakaguchi1 Hiroshi Kotani1
Hiroshi Kotani1 Akihiro Nishiyama1
Akihiro Nishiyama1 Kaname Yamashita1
Kaname Yamashita1 Seiji Yano2
Seiji Yano2 Fumihito Toshima3
Fumihito Toshima3 Dai Inoue3
Dai Inoue3 Toshifumi Gabata3
Toshifumi Gabata3 Hiroko Ikeda4
Hiroko Ikeda4 Atsushi Watanabe5
Atsushi Watanabe5 Kenji Notohara6
Kenji Notohara6 Takao Fujisawa7,8,9
Takao Fujisawa7,8,9 Yoshiaki Nakamura8,9,10
Yoshiaki Nakamura8,9,10 Takayuki Yoshino8,9,10
Takayuki Yoshino8,9,10 Kunio Miyake11
Kunio Miyake11 Kazuhiro Miwa12
Kazuhiro Miwa12 Shinji Takeuchi1
Shinji Takeuchi1We report the case of a 75-year-old woman with a pancreatic body mass. Pathological findings from endoscopic ultrasonography-guided fine-needle aspiration revealed medullary carcinoma of the pancreas (MCP). Deficient mismatch repair (dMMR) and high microsatellite instability (MSI-H) were identified through immunohistochemistry and next generation sequencing, respectively. While immunohistochemistry suggested MLH1 abnormality, no MLH1 mutation was; hypermethylation of the MLH1 promoter was later confirmed via bisulfite sequencing. The patient initially received nab-paclitaxel plus gemcitabine, achieving tumor shrinkage. Upon tumor regrowth, she was treated with the anti-programmed cell death-1 immune checkpoint inhibitor (ICI) pembrolizumab, which resulted in significant tumor reduction. This is the first case report of MCP with dMMR/MSI-H due to MLH1 promoter hypermethylation, effectively treated with an ICI.
Pancreatic cancer (PC) is a highly fatal disease with a 5-year survival rate of approximately 10% and is becoming an increasingly common cause of cancer-related mortality (1, 2). Approximately 95% of PCs are exocrine cell tumors, with ductal adenocarcinomas being the most prevalent subtype (3).
Medullary carcinoma of the pancreas (MCP) is a rare subtype of pancreatic ductal adenocarcinoma recognized in the current World Health Organization classification (4). Pathologically, MCP is characterized as a poorly differentiated carcinoma with limited gland formation, sheets and nests with pushing borders, syncytial growth patterns, and often abundant tumor-infiltrating lymphocytes (4). Although MCP has frequently been associated with deficient mismatch repair (dMMR) and high microsatellite instability (MSI-H) (5–12), most cases are linked to Lynch syndrome, with MLH1 promoter hypermethylation being reported only anechdotally (9). Furthermore, the effectiveness of immune checkpoint inhibitors (ICI) in MCP remains unclear.
We report a rare case of MCP with dMMR/MSI-H due to MLH1 promoter hypermethylation, successfully treated with immunotherapy.
A 75-year-old woman presented to a local hospital complaining of lumbago. Abdominal computed tomography (CT) revealed a large pancreatic body mass, and she was referred to our hospital for further evaluation and treatment.
Her medical history included ischemic heart disease and uterine myoma. Her family history was significant for gastric cancer in an elder brother and breast cancer in two elder sisters. She neither smoked, nor drank alcohol. Pancreatic enzyme levels were elevated, including lipase (54 IU/L; reference range: 11–53 IU/L), elastase I (329 ng/dL; reference range: <300 ng/dL), and trypsin (716 ng/mL; reference range: 110-550 ng/mL), while amylase remained normal. Among tumor markers, carcinoembryonic antigen (CEA), carbohydrate antigen 19-9 (CA 19-9), and Duke pancreatic monoclonal antigen type 2 (DUPAN-2) were within normal ranges, but cytokeratin-19 fragment (CYFRA 21-1) was elevated to 4.1 ng/mL (reference range: <3.5 ng/mL). Contrast-enhanced abdominal CT revealed a 77-mm hypovascular mass in the pancreatic body with celiac artery invasion (Figure 1A), but no definitive distant metastases were identified. The pancreatic tumor was classified as T4N0M0, stage III according to the Union for International Cancer Control (UICC) 8th edition.

Figure 1. (A) Contrast-enhanced abdominal computed tomography showing a 77-mm hypovascular mass with celiac artery invasion in the pancreatic body (white arrows). (B) Significant reduction in pancreatic tumor size, indicating partial response, after two months of treatment with nab-paclitaxel plus gemcitabine (white arrows). (C) Enlargement of the pancreatic tumor with obstructive jaundice after six months of treatment (white arrows). (D) Marked reduction in pancreatic tumor size, nearly achieving partial response, four months after initiating pembrolizumab treatment (white arrows).
Endoscopic ultrasonography (EUS) demonstrated a low-echoic, lobulated mass, and EUS-guided fine-needle aspiration (EUS-FNA) was performed using a 19-gauge needle. Pathological examination identified medullary carcinoma, characterized by solid sheets of carcinoma cells with minimal stroma (Figure 2A) and abundant tumor-infiltrating lymphocytes (Figure 2B). Staining for mismatch repair (MMR) proteins revealed loss of MLH1 and PMS2 expression, with retained MSH2 and MSH6, indicating deficient mismatch repair (dMMR) due to MLH1 dysfunction (Figures 2C–F). Although solid medullary neoplasms of the pancreas were raised as differential diagnosis, neuroendocrine neoplasms were excluded based on the negative immunohistochemical staining for synaptophysin, chromogranin A, and insulinoma-associated protein 1 (INSM1). Additionally, Bcl-10 immunostaining for acinar cell carcinoma and Epstein-Barr virus (EBV)-encoded RNA (EBER) in situ hybridization for EBV-related poorly differentiated carcinoma were also negative, further supporting the diagnosis of pancreatic medullary carcinoma.
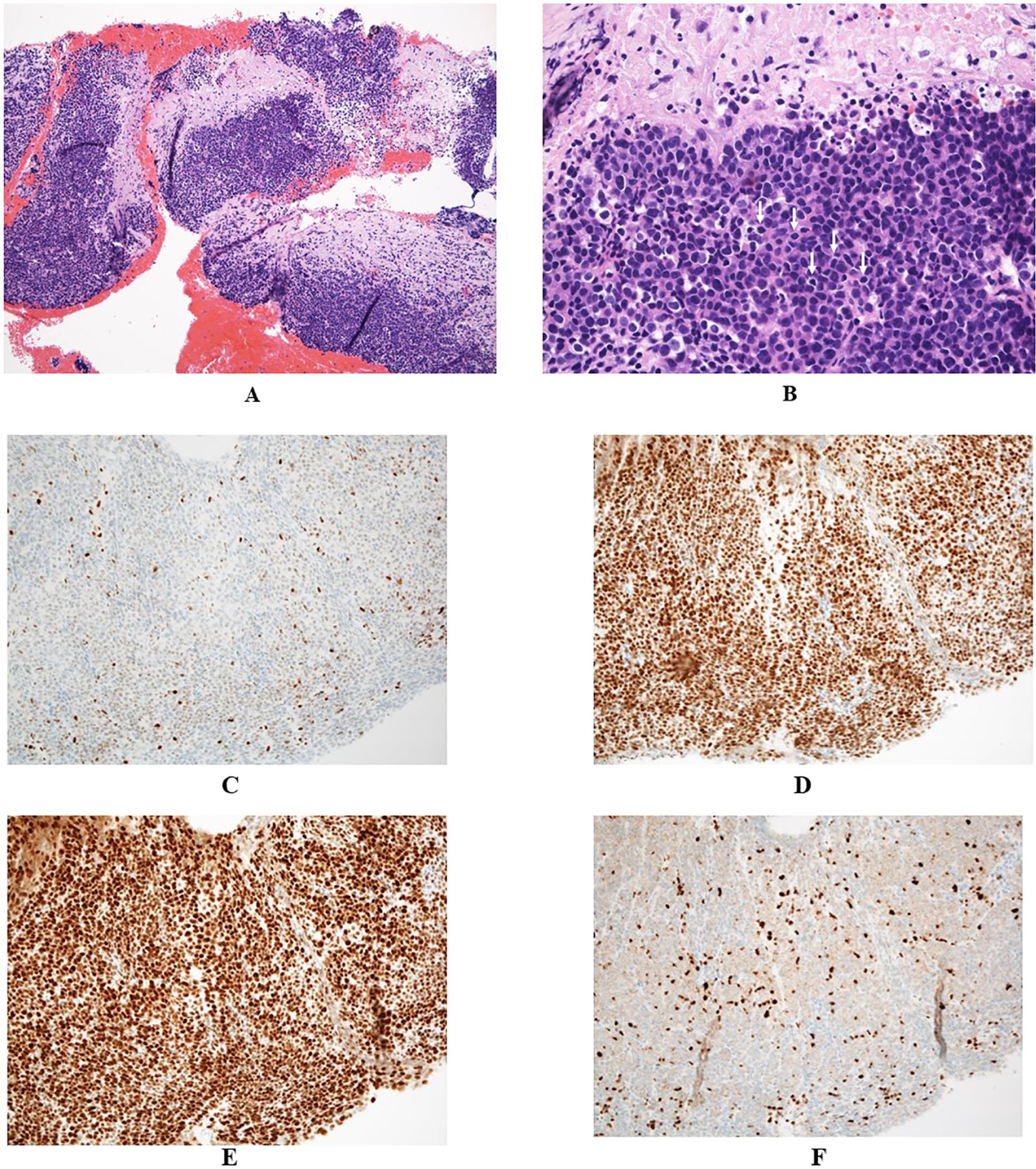
Figure 2. (A, B) Pathological findings revealed poorly differentiated carcinoma with syncytial growth, tumor-infiltrating lymphocytes (examples marked with white arrows), formation of sheets and nests, and pushing borders, suggesting medullary carcinoma (A:×10, B:×40). (C–F) Immunohistochemical findings demonstrated deficient mismatch repair proteins: MLH1 (C) and PMS2 (F), and proficient MSH2 (D) and MSH6 (E), suggesting an MLH1 gene abnormality (C–F:×20).
Genetic analysis was performed using both pancreatic tumor tissue and blood samples under MONSTAR-SCREEN-2, a component of the Cancer Genome Screening Project for Individualized Medicine in Japan (SCRUM-Japan) (13). Tumor tissue next-generation sequencing (NGS) was performed using MI Profile® (Caris Life Sciences, Phoenix, AZ), while plasma and buffy coat NGS was performed using Caris Assure® (Caris Life Sciences, Phoenix, AZ). No abnormalities were detected in the four major driver genes of pancreatic cancer (KRAS, TP53, CDKN2A, SMAD4). However, MSI-H and high tumor mutational burden (TMB-H) were identified in both tumor tissue (31 Mut/Mb) and plasma (19 Mut/Mb) (Tables 1A, B). Germline analysis detected no pathogenic variants in the MMR genes. The absence of somatic or germline mutations in the MMR genes suggested an epigenetic alteration of MLH1. Bisulfite sequencing confirmed hypermethylation of the MLH1 promoter in tumor tissue (Figure 3). Additionaly, an E-cadherin (CDH1) mutation with high variant allele frequency was identified in both pancreatic tumor tissue and plasma (Tables 1A, B).
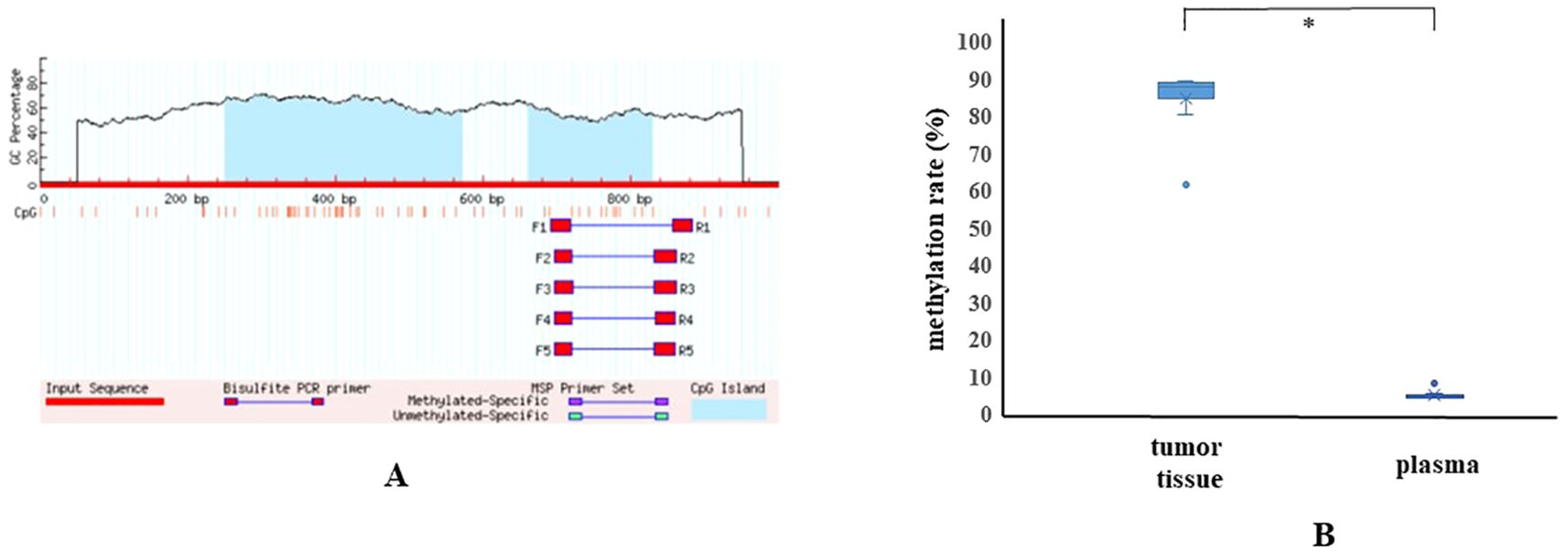
Figure 3. (A) CpG sites in the MLH1 gene are shown. Methylation analysis of MLH1 was performed at 11 CpG sites using 5 primers by bisulfite sequencing. (B) The methylation rate of the MLH1 promoter region in pancreatic tumor tissue (61.4–89.5%, average: 84.5%) was significantly higher than that in plasma (4.2–10.4%, average: 5.4%) (p=0.0001; Mann-Whitney U test). The methylation rate in normal pancreatic tissue could not be analyzed due to insufficient material.
The patient was initially treated with nab-paclitaxel plus gemcitabine, resulting in dramatic tumor shrinkage and a partial response (PR) after two months (Figure 1B). However, six months after initiating treatment, tumor regrowth occurred, accompanied by obstructive jaundice (Figure 1C). Endoscopic placement of a biliary plastic stent was performed, followed by treatment with the anti-programmed cell death-1 (PD-1) ICI pembrolizumab. The tumor significantly reduced in size again, achieving near-PR after four months (Figure 1D). Although tumor shrinkage was maintained for two months, tumor regrowth occurred seven months after the initiation of pembrolizumab, leading to treatment discontinuation (Figure 4).
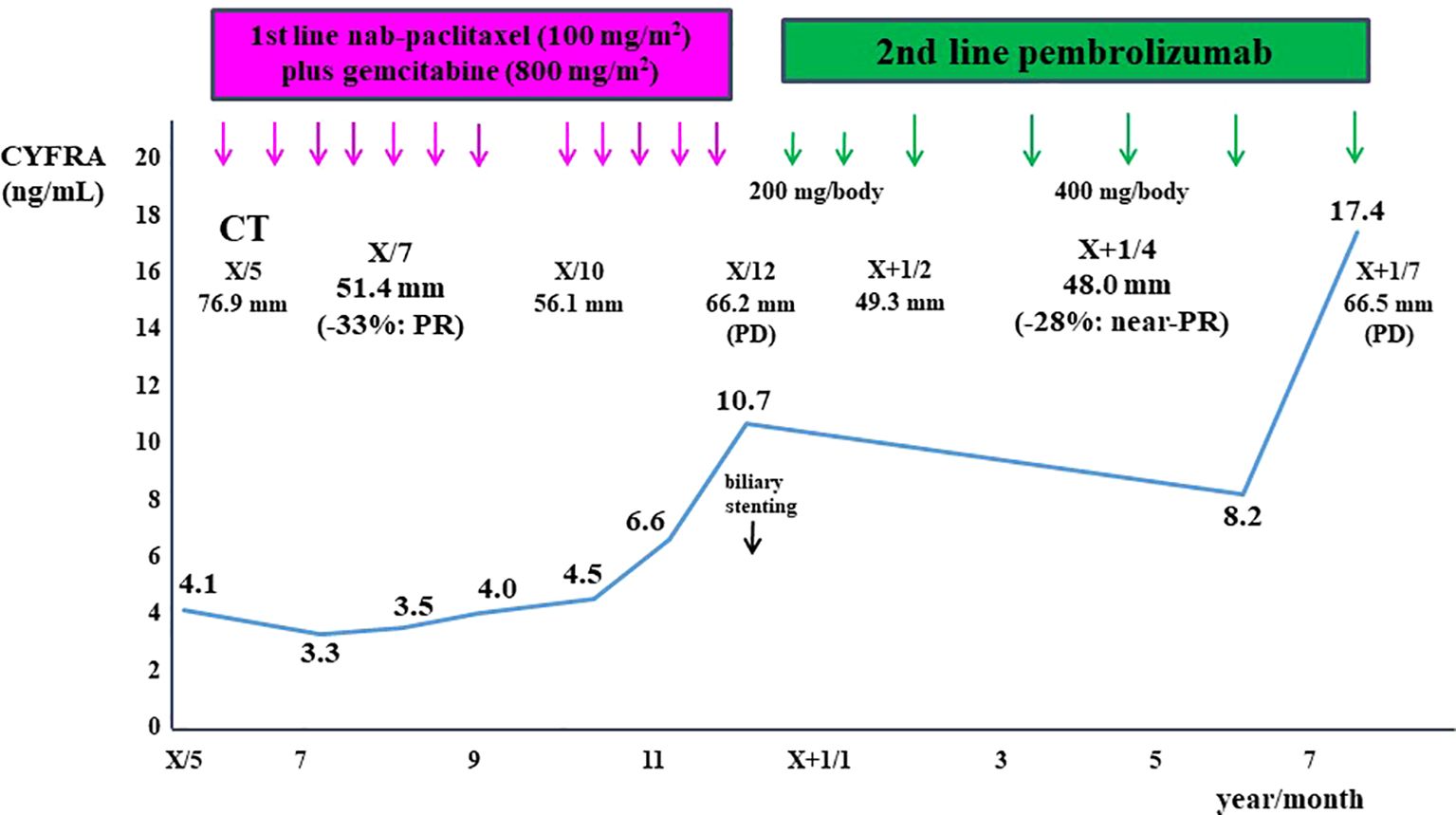
Figure 4. Clinical course of this patient is shown. The patient was initially treated with nab-paclitaxel plus gemcitabine, resulting in a partial response after two months. However, tumor regrowth occurred six months after treatment initiation. The patient was subsequently treated with pembrolizumab, achieving near- partial response after four months. However, tumor regrowth occurred seven months after the initiation of pembrolizumab. The cytokeratin-19 fragment (CYFRA 21-1) levels closely correlated with tumor size.
MCP is a rare histological subtype of PC (4, 14), first described by Goggins et al. in 1998 (5). Pathologically, MCP is characterized by poorly differentiated carcinoma with pushing borders, limited gland formation, and syncytial growth of tumor cells, often accompanied by abundant tumor-infiltrating lymphocytes (4). MCP is associated with a relatively favorable prognosis (6, 7, 15, 16). While MCP has been reported in EBV-associated carcinomas with lymphoepithelioma-like features (6), EBV involvement was excluded in this case due to a negative EBER in situ hybridization.
Carcinomas with dMMR/MSI-H are frequently observed in endometrial, gastric, small bowel, and colorectal cancers (CRCs) (17, 18). In CRCs, Alexander et al. identified medullary carcinoma, intraepithelial lymphocytosis, and poor differentiation as key features distinguishing MSI-H from microsatellite-stable (MSS) cancers with high specificity (19). Although the overall frequency of dMMR/MSI-H in PCs is extremely low (around 1–2%) (10, 17, 18, 20), it is relatively high in MCPs (5, 6, 10, 12). A systematic review of 34 studies involving 8,323 patients by Luchini et al. (10) reported that dMMR/MSI-H PCs are rare but are strongly associated with medullary or mucinous/colloid histology and a KRAS/TP53 wild-type molecular background.
Lynch syndrome, also known as hereditary nonpolyposis colorectal cancer syndrome, is an autosomal dominant disorder caused by germline mutations in any of the four genes involved in the MMR process: MLH1, MSH2, MSH6, and PMS2 (21). The most common malignancies in Lynch syndrome patients are CRCs and endometrial cancers, with penetrance rates of 50–80% and 25–60%, respectively (22). PC associated with Lynch syndrome was firstly reported in 1985 (23). Lynch syndrome is associated with an approx. 8.6-fold increased risk of developing PC (24) and has been identified in patients with MCP (6, 8). Given that MCP generally exhibits the histological features of a poorly differentiated carcinoma, we propose that poorly differentiated carcinomas reported in Lynch syndrome patients (8, 10) are highly likely to represent MCP (25, 26).
In this case, hypermethylation of MLH1 is estimated to be main cause of tumorigenesis, as bisulfite sequencing validated MLH1 hypermethylation while no mutations in MMR genes were detected. Hypermethylation serves as an alternative mechanism for the genetic silencing of MLH1. In CRC, it has been documented that dMMR/MSI-H is induced by hypermethylation of the MLH1 promoter region in over 75% of dMMR carcinomas, while germline mutations associated with Lynch syndrome account for less than 25% (27). Although the reported prevalence of MLH1 hypermethylation ranges from 0% to 54% (28–30), its precise role in MLH1 gene silencing remains unclear. Only one case of MCP harboring MLH1 promoter hypermethylation has been reported, identified using methylation-specific PCR (9).
In most PC patients, dMMR/MSI-H is routinely investigated using PCR and/or MMR immunohistochemistry rather than NGS (10). However, germline and methylation analyses have been scarcely performed. Consequently, it remains unclear whether genetic (somatic or germline mutations) or epigenetic mechanisms predominate in the development of MCP.
The typical genetic pattern of MCP is believed to involve dMMR/MSI-H and wild-type KRAS (5, 6). However, dMMR/MSI-H has been detected in only 22% (4/18) of MCP cases, with the majority being MSS (5, 6, 31). Mutations in the proofreading domain of polymerase epsilon (POLE) result in DNA repair deficiency characterized by MSS and an ultramutated phenotype (32). A case of MCP with a somatic POLE mutation and MSS has been reported, where the POLE mutation and resulting hypermutation were proposed as an altenative genetic backgound for MCP distinct from dMMR/MSI-H (33).
In a recent report, none of the six patients with MSI-H PC who received chemotherapy achieved a clinical response, including four patients treated with nab-paclitaxel plus gemcitabine (20). However, the patient was initially treated with nab-paclitaxel plus gemcitabine, resulting in a PR. Conversely, although the objective response rate of the anti-PD-1 ICI, pembrolizumab, in patients with previously treated advanced noncolorectal dMMR/MSI-H cancers has been reported as 34.3% (80 of 233 patients), the response rate in PC was only 18.2% (4 of 22 patients) in the Phase II KEYNOTE-158 study (34). Additionally, objective responses were observed in 29% (30 of 102 patients) of the tissue TMB-H group treated with pembrolizumab (35). In the present case, both dMMR/MSI-H and TMB-H (31 Mut/Mb) were identified in pancreatic tumor tissue, and the tumor size was further reduced following secondary treatment with pembrolizumab, resulting in nearly a PR. To the best of our knowledge, this is the first case report demonstrating the efficacy of immunotherapy in a patient with MCP. However, tumor regrowth occurred seven months after the initiation of pembrolizumab. Acquired resistance to anti-PD-1 ICI in patients with melanoma has been reported to be associated with defects in the pathways involving interferon-receptor–associated Janus kinase 1 (JAK1) or Janus kinase 2 (JAK2), as well as in the antigen-presenting protein beta-2-microglobulin (B2M) (36). Further investigation is necessary to clarify acquired resistance to anti-PD-1 ICI in patients with MCP.
In the present case, a CDH1 mutation with a high variant allele frequency was detected, and a germline pathogenic variant was suspected based on NGS analysis. Additionally, the presence of gastric cancer in the patient’s elder brother and breast cancers in two elder sisters aligns with the phenotype associated with CDH1 mutation. It has been reported that CDH1 germline mutations are linked to hereditary diffuse gastric cancer and lobular breast cancer (37–39). A recent report also described a relationship between CDH1 germline mutations and colorectal signet-ring cell cancer; however, the development of pancreatic cancer has been rarely documented (38). Furthermore, since no germline variants were detected in buffy coat analysis, we determined that the CDH1 mutation was somatic. Therefore, in the current case, the CDH1 mutation is unlikely to be significantly associated with the development of MCP.
In conclusion, we present a rare case of MCP with dMMR/MSI-H attributed to MLH1 promoter hypermethylation, successfully treated with immunotherapy. This case highlights the importance of testing for dMMR/MSI-H in MCP cases to assess their potential responsiveness to immunotherapy. Further research is needed to elucidate the pathogesesis of MCP.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
KO: Conceptualization, Writing – original draft, Writing – review & editing. SS: Writing – review & editing. HS: Writing – review & editing. HK: Writing – review & editing. AN: Writing – review & editing. KY: Writing – review & editing. SY: Writing – review & editing. FT: Formal Analysis, Writing – review & editing. DI: Formal Analysis, Writing – review & editing. TG: Formal Analysis, Writing – review & editing. HI: Formal Analysis, Writing – review & editing. AW: Formal Analysis, Writing – review & editing. KN: Formal Analysis, Writing – review & editing. TF: Formal Analysis, Writing – review & editing. YN: Formal Analysis, Writing – review & editing. TY: Formal Analysis, Writing – review & editing. KuM: Formal Analysis, Writing – review & editing. KaM: Writing – review & editing. ST: Validation, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors thank the patient and her family for providing informed consent for the use of her data in this publication. The authors also thank David Price of English Services for Scientists, based in Hiroshima for proofreading.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Egawa S, Toma H, Ohigashi H, Okusaka T, Nakao A, Hatori T, et al. Japan Pancreatic Cancer Registry; 30th year anniversary: Japan Pancreas Society. Pancreas. (2012) 41:985–92. doi: 10.1097/MPA.0b013e318258055c
2. Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. (2023) 73:17–48. doi: 10.3322/caac.21763
3. Grossberg AJ, Chu LC, Deig CR, Fishman EK, Hwang WL, Maitra A, et al. Multidisciplinary standards of care and recent progress in pancreatic ductal adenocarcinoma. CA Cancer J Clin. (2020) 70:375–403. doi: 10.3322/caac.21626
4. WHO Classification of Tumours Editorial Board. WHO classification of tumours. In: Digestive System Tumours, 10 Tumours of the Pancreas, 5th ed. Lyon: World Health Organization (2019). p. 295–372.
5. Goggins M, Offerhaus GJ, Hilgers W, Griffin CA, Shekher M, Tang D, et al. Pancreatic adenocarcinomas with DNA replication errors (RER+) are associated with wild-type K-ras and characteristic histopathology. Poor differentiation, a syncytial growth pattern, and pushing borders suggest RER+. Am J Pathol. (1998) 152:1501–7.
6. Wilentz RE, Goggins M, Redston M, Marcus VA, Adsay NV, Sohn TA, et al. Genetic, immunohistochemical, and clinical features of medullary carcinoma of the pancreas: A newly described and characterized entity. Am J Pathol. (2000) 156:1641–51. doi: 10.1016/S0002-9440(10)65035-3
7. Maple JT, Smyrk TC, Boardman LA, Johnson RA, Thibodeau SN, Chari ST. Defective DNA mismatch repair in long-term (> or =3 years) survivors with pancreatic cancer. Pancreatology. (2005) 5:220–7; discussion 227-228. doi: 10.1159/000085275
8. Banville N, Geraghty R, Fox E, Leahy DT, Green A, Keegan D, et al. Medullary carcinoma of the pancreas in a man with hereditary nonpolyposis colorectal cancer due to a mutation of the MSH2 mismatch repair gene. Hum Pathol. (2006) 37:1498–502. doi: 10.1016/j.humpath.2006.06.024
9. Laghi L, Beghelli S, Spinelli A, Bianchi P, Basso G, Di Caro G, et al. Irrelevance of microsatellite instability in the epidemiology of sporadic pancreatic ductal adenocarcinoma. PLoS One. (2012) 7:e46002. doi: 10.1371/journal.pone.0046002
10. Luchini C, Brosens LAA, Wood LD, Chatterjee D, Shin JI, Sciammarella C, et al. Comprehensive characterisation of pancreatic ductal adenocarcinoma with microsatellite instability: histology, molecular pathology and clinical implications. Gut. (2021) 70:148–56. doi: 10.1136/gutjnl-2020-320726
11. Verocq C, Racu ML, Bafort D, Butorano G, Perez-Casanova-Garcia L, Navez J, et al. Pancreatic medullary carcinoma developed on a pancreatic intraductal papillary mucinous neoplasm with loss of MSH2 and MSH6 expression: a case report. Diagn Pathol. (2021) 16:117. doi: 10.1186/s13000-021-01178-0
12. Luchini C, Scarpa A. Microsatellite instability in pancreatic and ampullary carcinomas: histology, molecular pathology, and clinical implications. Hum Pathol. (2023) 132:176–82. doi: 10.1016/j.humpath.2022.06.009
13. Nakamura Y, Yamashita R, Okamoto W, Komatsu Y, Yuki S, Ueno M, et al. Efficacy of targeted trials and signaling pathway landscape in advanced gastrointestinal cancers from SCRUM-Japan GI-SCREEN: A nationwide genomic profiling program. JCO Precis Oncol. (2023) 7:e2200653. doi: 10.1200/PO.22.00653
14. Satake T, Morizane C, Rikitake R, Higashi T, Okusaka T, Kawai A. The epidemiology of rare types of hepatobiliary and pancreatic cancer from national cancer registry. J Gastroenterol. (2022) 57:890–901. doi: 10.1007/s00535-022-01920-5
15. Lüttges J, Klöppel G. Update on the pathology and genetics of exocrine pancreatic tumors with ductal phenotype: precursor lesions and new tumor entities. Dig Dis. (2001) 19:15–23. doi: 10.1159/000050649
16. Schlitter AM, Segler A, Steiger K, Michalski CW, Jäger C, Konukiewitz B, et al. Molecular, morphological and survival analysis of 177 resected pancreatic ductal adenocarcinomas (PDACs): Identification of prognostic subtypes. Sci Rep. (2017) 7:41064. doi: 10.1038/srep41064
17. Le DT, Durham JN, Smith KN, Wang H, Bartlett BR, Aulakh LK, et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science. (2017) 357:409–13. doi: 10.1126/science.aan6733
18. Akagi K, Oki E, Taniguchi H, Nakatani K, Aoki D, Kuwata T, et al. Real-world data on microsatellite instability status in various unresectable or metastatic solid tumors. Cancer Sci. (2021) 112:1105–13. doi: 10.1111/cas.14798
19. Alexander J, Watanabe T, Wu TT, Rashid A, Li S, Hamilton SR. Histopathological identification of colon cancer with microsatellite instability. Am J Pathol. (2001) 158:527–35. doi: 10.1016/S0002-9440(10)63994-6
20. Sakakida T, Ishikawa T, Doi T, Morita R, Kataoka S, Miyake H, et al. Genomic profile and clinical features of MSI-H and TMB-high pancreatic cancers: real-world data from C-CAT database. J Gastroenterol. (2024) 59:145–56. doi: 10.1007/s00535-023-02058-8
21. Lynch HT, Snyder CL, Shaw TG, Heinen CD, Hitchins MP. Milestones of lynch syndrome: 1895-2015. Nat Rev Cancer. (2015) 15:181–94. doi: 10.1038/nrc3878
22. Morani AC, Hanafy AK, Ramani NS, Katabathina VS, Yedururi S, Dasyam AK, et al. Hereditary and sporadic pancreatic ductal adenocarcinoma: current update on genetics and imaging. Radiol Imaging Cancer. (2020) 2:e190020. doi: 10.1148/rycan.2020190020
23. Lynch HT, Voorhees GJ, Lanspa SJ, McGreevy PS, Lynch JF. Pancreatic carcinoma and hereditary nonpolyposis colorectal cancer: a family study. Br J Cancer. (1985) 52:271–3. doi: 10.1038/bjc.1985.187
24. Kastrinos F, Mukherjee B, Tayob N, Wang F, Sparr J, Raymond VM, et al. Risk of pancreatic cancer in families with Lynch syndrome. JAMA. (2009) 302:1790–5. doi: 10.1001/jama.2009.1529
25. Yamamoto H, Itoh F, Nakamura H, Fukushima H, Sasaki S, Perucho M, et al. Genetic and clinical features of human pancreatic ductal adenocarcinomas with widespread microsatellite instability. Cancer Res. (2001) 61:3139–44.
26. Hu ZI, Shia J, Stadler ZK, Varghese AM, Capanu M, Salo-Mullen E, et al. Evaluating mismatch repair deficiency in pancreatic adenocarcinoma: Challenges and recommendations. Clin Cancer Res. (2018) 24:1326–36. doi: 10.1158/1078-0432.CCR-17-3099
27. Taieb J, Svrcek M, Cohen R, Basile D, Tougeron D, Phelip JM. Deficient mismatch repair/microsatellite unstable colorectal cancer: Diagnosis, prognosis and treatment. Eur J Cancer. (2022) 175:136–57. doi: 10.1016/j.ejca.2022.07.020
28. Ueki T, Toyota M, Sohn T, Yeo CJ, Issa JP, Hruban RH, et al. Hypermethylation of multiple genes in pancreatic adenocarcinoma. Cancer Res. (2000) 60:1835–9.
29. Kondo E, Furukawa T, Yoshinaga K, Kijima H, Semba S, Yatsuoka T, et al. Not hMSH2 but hMLH1 is frequently silenced by hypermethylation in endometrial cancer but rarely silenced in pancreatic cancer with microsatellite instability. Int J Oncol. (2000) 17:535–41. doi: 10.3892/ijo.17.3.535
30. Li M, Zhao ZW. Clinical implications of mismatched repair gene promoter methylation in pancreatic cancer. Med Oncol. (2012) 29:970–6. doi: 10.1007/s12032-011-9968-y
31. Yago A, Furuya M, Mori R, Yabushita Y, Sawada Y, Kumamoto T, et al. Medullary carcinoma of the pancreas radiologically followed up as a cystic lesion for 9 years: a case report and review of the literature. Surg Case Rep. (2018) 4:80. doi: 10.1186/s40792-018-0487-3
32. Palles C, Cazier JB, Howarth KM, Domingo E, Jones AM, Broderick P, et al. Germline mutations affecting the proofreading domains of POLE and POLD1 predispose to colorectal adenomas and carcinomas. Nat Genet. (2013) 45:136–44. doi: 10.1038/ng.2503
33. Kryklyva V, Ter Linden E, Kroeze LI, de Voer RM, van der Kolk BM, Stommel MWJ, et al. Medullary pancreatic carcinoma due to somatic POLE mutation: A distinctive pancreatic carcinoma with marked long-term survival. Pancreas. (2020) 49:999–1003. doi: 10.1097/MPA.0000000000001588
34. Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De-Jesus-Acosta A, Delord JP, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair-deficient cancer: Results from the Phase II KEYNOTE-158 Study. J Clin Oncol. (2020) 38:1–10. doi: 10.1200/JCO.19.02105
35. Marabelle A, Fakih M, Lopez J, Shah M, Shapira-Frommer R, Nakagawa K, et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol. (2020) 21:1353–65. doi: 10.1016/S1470-2045(20)30445-9
36. Zaretsky JM, Garcia-Diaz A, Shin DS, Escuin-Ordinas H, Hugo W, Hu-Lieskovan S, et al. Mutations associated with acquired resistance to PD-1 blockade in melanoma. N Engl J Med. (2016) 375:819–29. doi: 10.1056/NEJMoa1604958
37. Hansford S, Kaurah P, Li-Chang H, Woo M, Senz J, Pinheiro H, et al. Hereditary diffuse gastric cancer syndrome: CDH1 mutations and beyond. JAMA Oncol. (2015) 1:23–32. doi: 10.1001/jamaoncol.2014.168
38. Girardi A, Magnoni F, Vicini E, Kouloura A, La Vecchia C, Veronesi P, et al. CDH1 germline mutations in families with hereditary lobular breast cancer. Eur J Cancer Prev. (2022) 31:274–8. doi: 10.1097/CEJ.0000000000000688
Keywords: medullary carcinoma of the pancreas, deficient mismatch repair, microsatellite instability, MLH1 hypermethylation, immune checkpoint inhibitor, pembrolizumab
Citation: Ohtsubo K, Sato S, Sakaguchi H, Kotani H, Nishiyama A, Yamashita K, Yano S, Toshima F, Inoue D, Gabata T, Ikeda H, Watanabe A, Notohara K, Fujisawa T, Nakamura Y, Yoshino T, Miyake K, Miwa K and Takeuchi S (2025) Case Report: Medullary carcinoma of the pancreas with MLH1 promoter hypermethylation, induced deficient mismatch repair, successfully treated with an immune checkpoint inhibitor. Front. Oncol. 15:1551038. doi: 10.3389/fonc.2025.1551038
Received: 24 December 2024; Accepted: 10 March 2025;
Published: 27 March 2025.
Edited by:
Massimo Broggini, Mario Negri Institute for Pharmacological Research (IRCCS), ItalyReviewed by:
Shuai Wang, New York University, United StatesCopyright © 2025 Ohtsubo, Sato, Sakaguchi, Kotani, Nishiyama, Yamashita, Yano, Toshima, Inoue, Gabata, Ikeda, Watanabe, Notohara, Fujisawa, Nakamura, Yoshino, Miyake, Miwa and Takeuchi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Koushiro Ohtsubo, b2h0c3Vib0BzdGFmZi5rYW5hemF3YS11LmFjLmpw
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.