
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Oncol. , 03 April 2025
Sec. Genitourinary Oncology
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1534739
Background: Cryotherapy plays a crucial role in managing radio-recurrent prostate cancer (PCa) after initial treatment. This study aims to provide a comprehensive review of its effectiveness and associated complications.
Methods: A systematic review was conducted using PubMed and EMBASE databases up to June 2024, focusing on recurrence-free survival (RFS) with salvage cryotherapy across various subgroups. Severe complications were also assessed. Survival curves were reconstructed using WebPlotDigitizer and a newly developed Shiny application. The incidence of complications was summarized with a 95% confidence interval (CI) using a random-effects model. Complications were evaluated using the Clavien-Dindo Scale (CDS).
Results: Thirty-six studies were included, with 15 papers (3174 patients) contributing to survival curve reconstruction. Among 1593 patients treated with salvage cryotherapy, the median RFS was 56.7 months, with 2-, 3-, and 5-year rates of 67.6%, 59.5%, and 47.3%, respectively. Factors associated with better RFS included a longer time from primary treatment to salvage therapy (TRS) [> 70 months vs. < 70 months, hazard ratio (HR) (95% CI):0.75(0.58-0.97), p=0.031], lower pre-salvage prostate-specific antigen (PSA) levels [< 5 ng/mL vs. > 5 ng/mL, HR (95% CI):0.78 (0.65-0.93), p=0.005], salvage whole-gland cryotherapy (SWC) [whole vs. focal, HR (95% CI):0.45 (0.37-0.56), p < 0.001], neoadjuvant androgen deprivation therapy (ADT) [Yes vs. No, HR (95% CI):0.79 (0.69-0.89), p < 0.001], and higher adjuvant ADT usage [16.5-34.2% vs. 0-10.5%, HR (95% CI):0.47(0.39-0.56), p < 0.001]. Concerning severe complications, 78 out of 876 patients (8.9%, 95% CI: 7-11) experienced genitourinary (GU) events, 53 out of 633 patients (8.5%, 95% CI: 6-11) suffered from urinary incontinence, 15 out of 493 patients (3.0%, 95% CI: 2-5) had urethral sloughing/stenosis, and 6 out of 522 patients (1.1%, 95% CI: 0-2) developed recto-urethral/vesical fistula. No cases of severe haematuria, urinary tract infection, or urinary retention were reported.
Conclusions: Cryotherapy demonstrates a favorable safety profile and significant RFS benefits for salvage treatment of radio-recurrent PCa. Longer TRS, lower pre-salvage PSA, SWC, and peri-salvage ADT usage appear to be promising prognostic factors for RFS. However, confirmation of these findings requires randomized controlled trials (RCTs) due to the low evidence levels and study heterogeneity.
Approximately 30–40% of individuals diagnosed with localized prostate cancer (PCa) choose non-extirpative treatments, such as external beam radiation therapy (EBRT), brachytherapy (BT), or cryotherapy, as their primary management options (1, 2). Within this cohort, 20 to 50% are expected to experience prostate recurrence, influenced by various risk factors. A subset of these patients may benefit from salvage therapies (3–5). Salvage interventions following non-extirpative treatments include salvage radical prostatectomy (SRP), stereotactic body radiation therapy (SBRT), BT, high-intensity focused ultrasound (HIFU), and salvage cryotherapy, among others. However, due to the limited number of high-quality clinical trials and the prevalence of low-quality evidence, recommendations for their use remain inconclusive (6).
A prior meta-analysis revealed that patients undergoing salvage cryotherapy had 2-year and 5-year recurrence-free survival (RFS) rates of 68% (95% confidence interval [CI], 62–73) and 50% (95% CI, 44–56), respectively. Genitourinary (GU) complications occurred in 15% (95% CI, 10–22) of cases (7). However, the literature search for this meta-analysis was conducted up until 2019, which is relatively early. Due to space limitations, the descriptions of each study in the analysis were insufficiently detailed. Moreover, the presence of duplicate cases among the studies may have affected the accuracy of the reported results.
In our recent meta-analysis (8), we reconstructed and summarized the RFS curves and evaluated toxicities in patients with radio-recurrent PCa undergoing salvage high-dose-rate brachytherapy (HDR-BT). This analysis yielded significant subgroup findings and a summary of toxicities. However, no comparable meta-analysis has assessed salvage cryotherapy. Thus, the aim of this systematic review and meta-analysis is to evaluate the efficacy and complications of cryotherapy for radio-recurrent PCa.
This meta-analysis adhered to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA). The evaluation protocol was prospectively registered in the International Prospective Register of Systematic Reviews (PROSPERO) and is publicly available with the registration number CRD42024552270.
A comprehensive and systematic literature search was conducted across two reputable electronic databases, Embase and PubMed, covering articles from their inception through June 15, 2024. Full-text eligibility screening was independently performed by two investigators. The search strategy included the following terms: (cryotherapy OR cryosurgery OR cryoablation OR cryosurgical OR cold therapy) AND (prostate OR prostatic) AND (recurrence OR recurrent OR relapse OR salvage OR Recrudescence OR local failure OR radio-recurrent) (\Supplementary Table 1). Additionally, reference lists of eligible studies were manually reviewed for potential additional inclusions.
Inclusion criteria: 1) Patients with a confirmed diagnosis of radio-recurrent PCa; 2) Availability of quantitative data on either RFS or severe complications treated with cryotherapy, with RFS curves demonstrating rates exceeding two years.
Exclusion criteria: 1) Duplicate publications; 2) Articles lacking full-text access; 3) Non-English language publications; 4) Studies that did not employ the Clavien-Dindo Scale (CDS) to assess severe urinary complications.
Inclusion criteria for RFS curve reconstruction: 1) Fulfillment of the inclusion and exclusion criteria outlined above; 2) Availability of risk tables within the RFS curves.
Exclusion criteria for RFS curve reconstruction:1) Duplicate data.
Survival curve reconstruction was performed independently by two investigators, with any discrepancies resolved through consensus.
Two investigators independently utilized a standardized data extraction form to collect relevant information, with any discrepancies addressed through discussion. Patient characteristics were categorized into two main areas: 1) Primary disease and treatment characteristics; 2) Disease and treatment details during the peri-salvage cryotherapy period. Moreover, we extracted raw data points and numbers at risk from the original studies to reconstruct individual patient data (IPD) for RFS analysis. To mitigate the impact of duplicate reports, we meticulously excluded redundant data by considering the enrollment institutions and times, ensuring the accuracy of the data related to RFS and severe complications.
The primary objective of this study is to evaluate the RFS of PCa patients treated with salvage cryotherapy across various subgroups, with a secondary focus on assessing the occurrence of severe complications.
The definition of RFS varies among studies, with the Phoenix criteria (9) being the most widely adopted standard. While some studies follow the criteria set by the American Society for Radiation Oncology (ASTRO) (10), others regard local failure, metastatic progression, or the initiation of hormone suppression therapy as indicators of recurrence. In this study, we define biochemical RFS, failure-free survival, and disease-free survival as equivalent to RFS. Urinary complications will be evaluated using the CDS, where Grade ≥ 3a is considered indicative of severe events (11).
To reconstruct survival data, screenshots of necessary survival curves and risk tables will be obtained from each publication. Raw data coordinates will then be extracted using the semi-automated tool WebPlotDigitizer. IPD will be reconstructed utilizing a novel application developed by Liu et al. (12), followed by survival curve plotting using R (version 4.0.3). Summary outcomes of complications will be presented as incidence rates with 95% CIs. These rates and CIs will be calculated using a random effects model with logit transformation, as delineated by Nyaga et al. (13), and implemented in STATA 14.0. A two-sided test will be performed, with statistical significance set at α = 0.05, and results will be considered significant if the p-value is below this threshold.
After removing duplicate records, a total of 2084 entries were retrieved from two databases. A preliminary review of titles and abstracts led to the exclusion of records that did not meet the inclusion criteria, resulting in 119 records being retained (Figure 1). Following a comprehensive review of the full texts, 36 studies were included (14–49). Among these, curve data extraction software was used to derive 2-year or 5-year RFS rates from 34 papers (14–47) (Supplementary Table 2). RFS curves were reconstructed for 15 studies (16–19, 21, 26, 29–31, 37, 38, 40, 41, 43, 49), with near-complete duplication observed in 5 studies (18, 19, 26, 29, 41) (Table 1). A summary of severe urinary complications was compiled from 11 studies (14–17, 20, 21, 23, 27, 28, 48, 49), noting partial duplication in 2 studies (27, 49) (Supplementary Table 3).
Table 2 outlines the patient characteristics regarding primary disease and treatment across 15 studies used for the reconstruction of RFS curves. These studies, published between 2007 and 2023, included three prospective and the remainder retrospective designs. Patient enrollment occurred from 1992 to 2021, with the majority of studies conducted in North America. The number of patients enrolled in each study ranged from 19 to 486, with median pre-treatment prostate-specific antigen (PSA) levels ranging from 12 to 36 ng/mL. The distribution of Gleason score (GS) was summarized across the studies, although missing data were noted in several of them. Most patients received definitive treatment based on external beam radiation therapy (EBRT), while other treatments included BT and unspecified radiotherapy.
Table 3 summarizes the disease and treatment characteristics during the peri-salvage cryotherapy period. In the studies reviewed, the median age at recurrence ranged from 66 to 72 years, and the median time from primary treatment to salvage therapy (TRS) ranged from < 60 to 84 months. Median pre-treatment PSA levels ranged from < 4 to 7.8 ng/mL. Imaging methods for diagnosing pelvic recurrence primarily included magnetic resonance imaging (MRI), computed tomography (CT), bone scans, and positron emission tomography-computed tomography (PET-CT). Pathological biopsies of recurrent lesions were performed for all enrolled patients, and recurrence was primarily defined according to the Phoenix criteria. The majority of studies implemented salvage whole-gland cryotherapy (SWC), though salvage focal-gland cryotherapy (SFC) was used in several cases. Additionally, the proportion of patients receiving neoadjuvant/adjuvant androgen deprivation therapy (ADT) and the follow-up time after salvage therapy were also summarized.
The RFS curves for the total cohort were reconstructed from data of 1,593 patients across 8 studies (16, 17, 21, 30, 31, 37, 40, 43). The median RFS duration was 56.7 months (range: 51.2–64.4 months). The pooled 2-, 3-, and 5-year RFS rates were 67.6% (95% CI: 64.7–69.8%), 59.5% (95% CI: 56.7–62.5%), and 47.3% (95% CI: 44.0–50.6%), respectively (Figure 2).
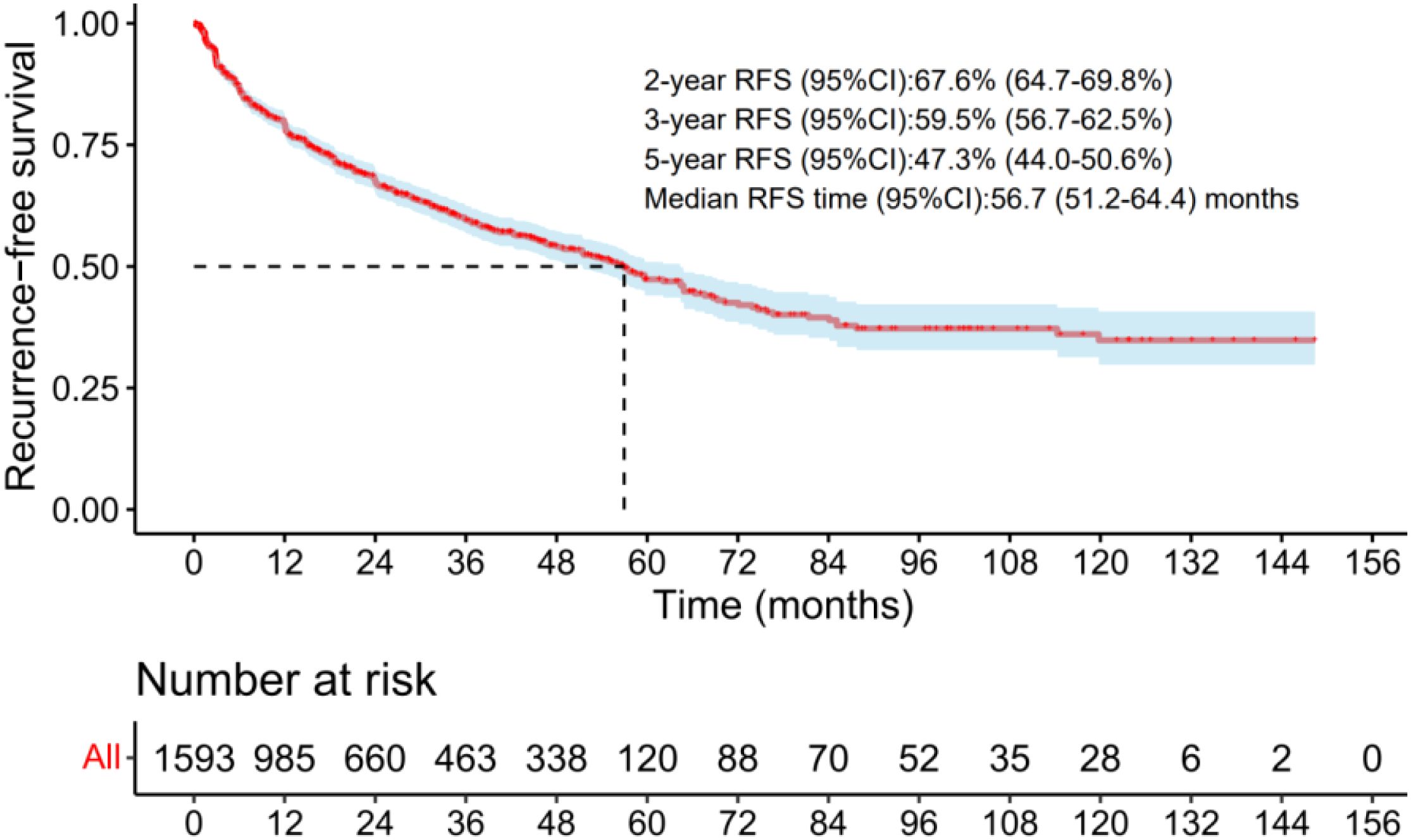
Figure 2. The RFS curves of the radio-recurrent patients treated with salvage cryotherapy in the total group. .
Various factors were assessed to determine their impact on RFS following salvage cryotherapy. Patients from the studies by Campbell SP et al. (15), Tan WP et al. (17), and Kovac E et al. (29) exhibited significantly higher RFS rates compared to those in studies by Overduin CG et al. (26), Spiess PE et al. (40), and Ismail M et al. (43) (Figure 3A). Similarly, individuals with TRS >70 months exhibited notably higher RFS rates than those < 70 months (hazard ratio, HR: 0.75, 95% CI: 0.58-0.97, p=0.031) (Figure 3B). Patients with pre-salvage PSA <5 ng/mL exhibited significantly higher RFS rates compared to >5 ng/mL (HR: 0.78, 95% CI: 0.65-0.93, p = 0.005) (Figure 3C). Moreover, patients treated with SWC showed significantly higher RFS rates than those treated with SFC (HR: 0.45, 95% CI: 0.37-0.56, p<0.001) (Figure 3D). Patients who received neoadjuvant ADT exhibited significantly better RFS rates compared to those who did not (HR: 0.79, 95% CI: 0.69-0.89, p < 0.001) (Figure 3E). Furthermore, patients with an adjuvant ADT proportion ranging from 16.5% to 34.2% demonstrated significantly higher RFS rates than those with a proportion of 0 to 10.5% (HR: 0.47, 95% CI: 0.39-0.56, p < 0.001) (Figure 3F). However, no significant differences in RFS rates were observed based on median age (≤ 70 years vs. >70 years) or median Gleason score (GS ≤7 vs. GS ≥8) (Figures 3G–H).
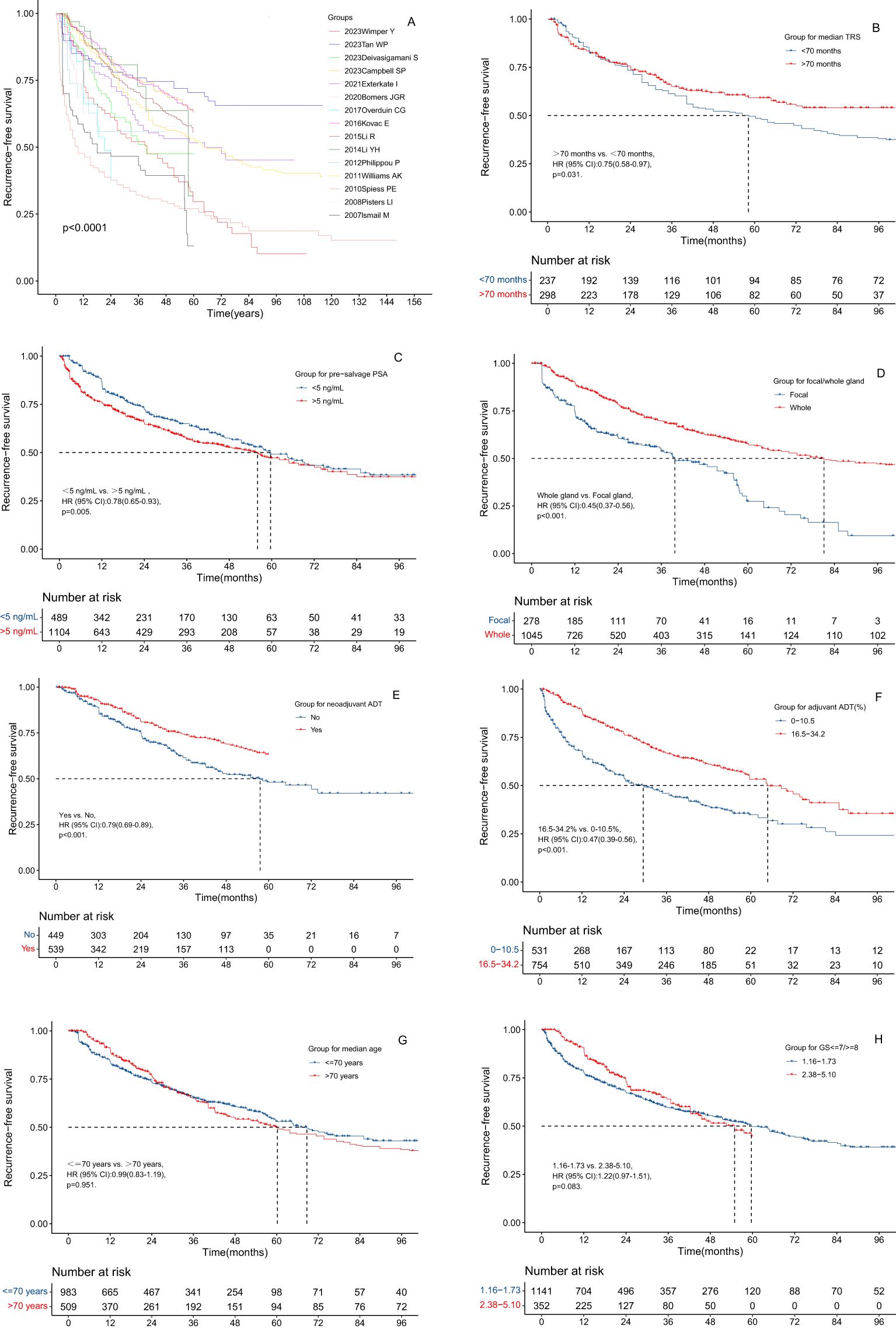
Figure 3. The RFS curves of the radio-recurrent patients treated with salvage cryotherapy in different subgroups. (A) Grouping of different papers. (B) Grouping of different median age at time of recurrence. (C) Grouping of different median TRS. (D) Grouping of different median pre-salvage PSA level. (E) Grouping of different pre-salvage values of GS ≤7/≥8. (F) Grouping of SFC vs. SWC. (G) Grouping of different median proportion of neoadjuvant ADT. (H) Grouping of different median proportion of adjuvant ADT.
As shown in Supplementary Table 2, the 2-year RFS rates reported in 34 studies ranged from 15.4% to 92%, with a median rate of 72.0%. The 5-year RFS rates, available from 26 studies, ranged from 0% to 86.5%, with a median of 46.5% (13–46).
Supplementary Table 3 summarizes the severe complications reported in 11 studies (14–17, 20, 21, 23, 27, 28, 48, 49). After excluding duplicates, we compiled a summary of the most common severe complications (Supplementary Tables 4–10). Among 876 patients, 78 (8.9%, 95% CI: 7-11) experienced GU events. Of 633 patients, 53 (8.5%, 95% CI: 6-11) suffered from urinary incontinence, 15 out of 493 patients (3.0%, 95% CI: 2-5) developed urethral sloughing/stenosis, and 6 out of 522 patients (1.1%, 95% CI: 0-2) had recto-urethral or vesical fistulae (Figure 4). No cases of severe hematuria, urinary tract infection, or urinary retention were reported (Figure 4).
To the best of our knowledge, this meta-analysis represents the first comprehensive attempt to assess the RFS rates and the incidence of severe complications associated with salvage cryotherapy in patients with radio-recurrent PCa, utilizing a survival curve reconstruction methodology.
Our findings revealed that the estimated 2-year and 5-year RFS rates following salvage cryotherapy were 67.6% (95% CI: 64.7–69.8%) and 47.3% (95% CI: 44.0–50.6%), respectively. These rates are notably higher than those reported for salvage stereotactic body radiation therapy (SBRT), which demonstrated 2-year and 5-year RFS rates of 64.8% (95% CI: 60.3–67.9%) and 40.6% (95% CI: 34.4–46.7%) (50), but lower than those for salvage low-dose-rate brachytherapy (LDR-BT), which showed 2-year and 5-year RFS rates of 84.6% (95% CI: 81.5–87.5%) and 63.5% (95% CI: 59.0–68.7%) (51), as well as for salvage high-dose-rate brachytherapy (HDR-BT), which reported 2-year and 5-year RFS rates of 75.9% (95% CI: 72.8–79.2%) and 52.3% (95% CI: 47.5–57.4%) (8). All pooled analyses in this study utilized survival curve reconstruction techniques to ensure comparability and precision across the data.
Additionally, the aggregated incidence of severe GU complications in our cohort of 876 patients from 9 studies was 8.9% (95% CI: 7–11%). This figure was higher than the rates reported for salvage SBRT (5.8% [95% CI: 4.5–7.4%]) (50) and salvage HDR-BT (5.8% [95% CI: 4–7%]) (8), but lower than that for salvage LDR-BT (12.7% [95% CI: 10–15%]) (51). It is important to note that the CDS, employed in the current study, does not classify urinary incontinence as a graded complication. However, the incidence of severe urinary incontinence following salvage cryotherapy was non-negligible, with our pooled analysis revealing a rate of 8.5% (95% CI: 6–11%). In contrast, studies using the Common Terminology Criteria for Adverse Events (CTCAE) for toxicity assessment reported a very low incidence of severe urinary incontinence with other treatments such as SBRT, LDR-BT, and HDR-BT. Consequently, it is not possible to conclusively state that salvage cryotherapy is safer than salvage LDR-BT in terms of GU toxicity.
In the subgroup analysis of salvage cryotherapy, we identified several prognostic factors influencing RFS. Specifically, SWC and a higher proportion of ADT were associated with improved RFS rates. Given that solitary lesions are more common in localized recurrent PCa than multifocal lesions, salvage focal-gland therapy (SFC) is theoretically a viable option (52). Observational data suggest that localized ablative treatments, such as high-intensity focused ultrasound (HIFU) and cryotherapy, yield oncological outcomes comparable to those of whole-gland treatments but with reduced toxicity (53–57). In a study by Tan WP et al. (22), which included 385 patients with radio-recurrent PCa (72 of whom underwent SFC), no significant difference in 2-year progression-free survival was found between SWC and SFC (79.8% vs. 77.0%, P = 0.11) after propensity score matching. However, SFC was associated with a significantly lower rate of transient urinary retention compared to SWC (5.6% vs. 22.4%, P < 0.001) (22). Similarly, de Castro Abreu AL et al. (34) conducted a study involving 50 patients, 25 of whom were treated with SFC, and found that SWC resulted in significantly higher 5-year RFS rates compared to SFC (86% vs. 54%). Another study by Wenske S et al. (58), which included 328 patients with radio-recurrent PCa (55 of whom underwent SFC), also reported higher 5-year RFS rates for SWC compared to SFC (63% vs. 47%). Our analysis further supports these findings, showing that SWC significantly improves RFS rates relative to SFC (HR: 0.45, 95% CI: 0.37-0.56, p < 0.001). However, due to variability in baseline characteristics among the studies included in our meta-analysis, the relative effectiveness of SFC versus SWC in terms of RFS or long-term survival remains inconclusive.
In addition, the present study identified a longer duration of treatment-free survival (TRS) as a favorable prognostic factor for RFS, with patients exhibiting TRS > 70 months showing significantly better outcomes compared to those with TRS < 70 months (HR: 0.75, 95% CI: 0.58–0.97, p = 0.031). While several recent studies have investigated the effect of TRS on RFS (21, 38, 43, 59), only Exterkate L et al. (21) observed a significant association between TRS duration and RFS (HR: 0.87, 95% CI: 0.78–0.99, p = 0.03). Therefore, caution should be exercised when interpreting these results. Furthermore, our study demonstrated that the pre-salvage PSA level is a significant prognostic factor for RFS. Specifically, patients with pre-salvage PSA levels <5 ng/mL had superior RFS rates compared to those with PSA >5 ng/mL (HR: 0.78, 95% CI: 0.65–0.93, p = 0.005), although similar studies (8, 51) did not find PSA level to be predictive of RFS.
Furthermore, we found that neoadjuvant ADT prior to salvage therapy was associated with significantly higher RFS rates compared to those who did not receive neoadjuvant ADT (HR: 0.79, 95% CI: 0.69-0.89, p < 0.001). Despite extensive exploration of this issue in many studies (17, 18, 21, 26, 30, 31, 42, 48), only a few have suggested the benefits of neoadjuvant ADT for RFS (26, 30). Consequently, our findings are particularly valuable for patients with high-risk recurrent PCa, recommending peri-salvage ADT for this patient subset.
It is noteworthy that our study is the first meta-analysis to comprehensively examine the prevalence of severe GU complications, such as recto-urethral/vesical fistula, urinary incontinence, and urethral sloughing/stenosis, among others. The incidence of these severe complications was consistently low, with rates not exceeding 10%, and many instances were reported as zero. Given the findings from previous studies on salvage prostatectomy (7), our results suggest that salvage cryotherapy may offer a safer profile than salvage prostatectomy in terms of severe GU complications.
While our study provides valuable insights, it is not without its limitations. Firstly, although survival curve reconstruction for indirect comparison of survival outcomes across different treatment groups is a robust method, the homogeneity of the included studies plays a critical role in ensuring the reliability of the results. Most studies in this meta-analysis were single-arm or retrospective, with relatively low levels of evidence. Additionally, there were considerable variations in baseline patient characteristics, such as primary treatment type, pre-salvage age, median TRS, pre-salvage PSA level, pre-salvage GS, and peri-salvage ADT usage, which may have influenced the observed differences in RFS across subgroups. Furthermore, despite our efforts to avoid duplication, some studies in our analysis, particularly those from the Cryo Online Data (COLD) registry, may have overlapping patient populations. Moreover, discrepancies in data extraction tools, survival reconstruction methodologies, curve resolution techniques, and variations in researchers’ approaches could affect the accuracy of data restoration during survival reconstruction. Consequently, the reliability of our findings may be compromised, necessitating confirmation through relevant randomized controlled trials (RCTs).
Cryotherapy demonstrates strong safety and offers significant benefits in RFS as salvage therapy for radio-recurrent PCa. Particularly, patients with longer TRS, lower pre-salvage PSA, SWC, and peri-salvage ADT usage experience superior RFS outcomes with minimal severe urinary complications. However, these findings require validation through RCTs due to the low evidence quality and variability across studies.
SZ: Data curation, Writing – original draft. JL: Formal Analysis, Writing – original draft. BS: Investigation, Writing – original draft. HX: Methodology, Supervision, Writing – review & editing. WZ: Investigation, Writing – original draft. SJ: Conceptualization, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by grants from Shaoxing People’s Hospital Youth Fund (2023YB30) and Health Science and Technology Plan Project of Zhejiang (2024KY1714;2023KY366).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2025.1534739/full#supplementary-material
PCa, prostate cancer; RFS, recurrence-free survival; CI, confidence interval; CDS, Clavien-Dindo Scale; TRS, time from primary treatment to salvage therapy; HR, hazard ratio; PSA, prostate-specific antigen; SWC, salvage whole-gland cryotherapy; ADT, androgen deprivation therapy; RCTs, randomized controlled trials; EBRT, external beam radiation therapy; BT, brachytherapy; SRP, salvage radical prostatectomy; SBRT, stereotactic body radiation therapy; HIFU, high-intensity focused ultrasound; GU, genitourinary; HDR-BT, high-dose-rate brachytherapy; PRISMA, Systematic Reviews and Meta-Analyses; PROSPERO, the International Prospective Register of Systematic Reviews; IPD, individual patient data; ASTRO, American Society for Radiation Oncology; GS, Gleason score; MRI, magnetic resonance imaging; CT, computed tomography; PET-CT, positron emission tomography-computed tomography; SFC, salvage focal-gland cryotherapy; COLD, Cryo Online Data; CTCAE, Common Terminology Criteria for Adverse Events.
1. Scherzer ND, DiBiase ZS, Srivastav SK, Thomas R, DiBiase SJ. Regional differences in the treatment of localized prostate cancer: an analysis of surgery and radiation utilization in the United States. Adv Radiat Oncol. (2019) 4:331–6. doi: 10.1016/j.adro.2019.01.004
2. Mahal BA, Butler S, Franco I, Spratt DE, Rebbeck TR, D’Amico AV, et al. Use of active surveillance or watchful waiting for low-risk prostate cancer and management trends across risk groups in the United States, 2010-2015. JAMA. (2019) 321:704–6. doi: 10.1001/jama.2018.19941
3. Autran-Gomez AM, Scarpa RM, Chin J. High-intensity focused ultrasound and cryotherapy as salvage treatment in local radio-recurrent prostate cancer. Urol Int. (2012) 89:373–9. doi: 10.1159/000339616
4. Duijzentkunst DA, Peters M, van der Voort van Zyp JR, Moerland MA, van Vulpen M. Focal salvage therapy for local prostate cancer recurrences after primary radiotherapy: a comprehensive review. World J Urol. (2016) 34:1521–31. doi: 10.1007/s00345-016-1811-9
5. Golbari NM, Katz AE. Salvage therapy options for local prostate cancer recurrence after primary radiotherapy: a literature review. Curr Urol Rep. (2017) 18:63. doi: 10.1007/s11934-017-0709-4
6. Mottet N, van den Bergh RCN, Briers E, Van den Broeck T, Cumberbatch MG, De Santis M, et al. EAU-EANM-ESTRO-ESUR-SIOG guidelines on prostate cancer-2020 update. Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. (2021) 79:243–62. doi: 10.1016/j.eururo.2020.09.042
7. Valle LF, Lehrer EJ, Markovic D, Elashoff D, Levin-Epstein R, Karnes RJ, et al. A systematic review and meta-analysis of local salvage therapies after radiotherapy for prostate cancer (MASTER). Eur Urol. (2021) 80:280–92. doi: 10.1016/j.eururo.2020.11.010
8. Shen B, Liu J, Wu D, Guo J. Evaluation of the safety and efficacy of high-dose rate brachytherapy for radiorecurrent prostate cancer: a systematic review and meta-analysis. Strahlenther Onkol. (2024) 200:655–70. doi: 10.1007/s00066-024-02205-x
9. Roach M 3rd, Hanks G, Thames H Jr, Schellhammer P, Shipley WU, Sokol GH, et al. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int J Radiat Oncol Biol Phys. (2006) 65:965–74. doi: 10.1016/j.ijrobp.2006.04.029
10. Cox JD, Grignon DJ, Kaplan RS, Parsons JT, Schellhammer PF, Zagars GK. Consensus statement: guidelines for PSA following radiation therapy. American Society for Therapeutic Radiology and Oncology Consensus Panel. Int J Radiat Oncol Biol Phys. (1997) 37:1035–41.
11. Clavien PA, Barkun J, de Oliveira ML, Vauthey JN, Dindo D, Schulick RD, et al. The Clavien-Dindo classification of surgical complications: Five-year experience. Ann Surg. (2009) 250:187–96. doi: 10.1097/SLA.0b013e3181b13ca2
12. Liu N, Zhou Y, Lee JJ. IPDfromKM: reconstruct individual patient data from published Kaplan-Meier survival curves. BMC Med Res Methodol. (2021) 21:111. doi: 10.1186/s12874-021-01308-8
13. Nyaga VN, Arbyn M, Aerts M. Metaprop: a Stata command to perform meta-analysis of binomial data. Arch Public Health. (2014) 72:39. doi: 10.1186/2049-3258-72-39
14. Brito DV E, Pereira JA, Ferreira AM, Lourenço M, Godinho R, Pereira B, et al. Salvage cryotherapy for prostate cancer. Arch Ital Urol Androl. (2024) 95:11897. doi: 10.4081/aiua.2023.11897
15. Carbonell E, Mercader C, Sureda J, Gutiérrez A, Muñoz J, Gallardo E, et al. Nadir prostate-specific antigen after salvage cryotherapy as a potential prognostic factor for oncologic outcomes. World J Urol. (2024) 42:133. doi: 10.1007/s00345-024-04806-8
16. Wimper Y, Overduin CG, Sedelaar JPM, Veltman J, Jenniskens SFM, Bomers JGR, et al. MRI-guided salvage focal cryoablation: A 10-year single-center experience in 114 patients with localized recurrent prostate cancer. Cancers (Basel). (2023) 15:4093. doi: 10.3390/cancers15164093
17. Tan WP, Kotamarti S, Ayala A, Mahle R, Chen E, Arcot R, et al. Oncological and functional outcomes for men undergoing salvage whole-gland cryoablation for radiation-resistant prostate cancer. Eur Urol Oncol. (2023) 6:289–94. doi: 10.1016/j.euo.2023.02.007
18. Deivasigamani S, Orabi H, El-Shafei A, Adams ES, Kotamarti S, Aminsharifi A, et al. Intermediate-term oncological and functional outcomes of salvage cryotherapy for the management of prostate cancer recurrence after primary brachytherapy versus primary cryotherapy: A propensity score-matched analysis. Prostate. (2023) 83:1373–86. doi: 10.1002/pros.24600
19. Campbell SP, Deivasigamani S, Arcot R, Adams ES, Orabi H, Elshafei A, et al. Salvage cryoablation for recurrent prostate cancer following primary external beam radiotherapy or primary cryotherapy: A propensity score matched analysis of mid-term oncologic and functional outcomes. Clin Genitourin Cancer. (2023) 21:555–62. doi: 10.1016/j.clgc.2023.06.015
20. Vestris PG, Giwerc A, Hennequin C, Goujon A, Meria P, Verine J, et al. Operative and midterm oncological outcome of focal salvage cryotherapy for localized prostate cancer. Urol Int. (2022) 106:897–902. doi: 10.1159/000518980
21. Exterkate L, Peters M, Somford DM, Vergunst H. Functional and oncological outcomes of salvage cryosurgery for radiorecurrent prostate cancer. BJU Int. (2021) 128:46–56. doi: 10.1111/bju.15269
22. Tan WP, ElShafei A, Aminsharifi A, Khalifa AO, Polascik TJ. Salvage focal cryotherapy offers similar short-term oncologic control and improved urinary function compared with salvage whole gland cryotherapy for radiation-resistant or recurrent prostate cancer. Clin Genitourin Cancer. (2020) 18:e260–5. doi: 10.1016/j.clgc.2019.11.009
23. Bain A, Kinnaird A, McLarty R, Senthilselvan A, Todd G, Chetner MP. Oncological outcomes of salvage cryotherapy after primary radiation therapy vs. primary cryotherapy: 10-year experience at a large Canadian referral center. Can Urol Assoc J. (2020) 14:E604–6. doi: 10.5489/cuaj.6244
24. Safavy S, Jabaji RB, Lu SM, Slezak JM, Cosmatos HA, Williams SG, et al. Salvage cryoablation for radiorecurrent prostate cancer: initial experience at a regional health care system. Perm J. (2019) 23:18–153. doi: 10.7812/TPP/18-153
25. Barat M, Colleter L, Mongiat-Artus P, Jolibois Z, Quero L, Hennequin C, et al. Salvage cryoablation for local recurrence of prostatic cancer after curative therapy. Diagn Interv Imaging. (2019) 100:679–87. doi: 10.1016/j.diii.2019.07.001
26. Overduin CG, Jenniskens SFM, Sedelaar JPM, Bomers JGR, Fütterer JJ. Percutaneous MR-guided focal cryoablation for recurrent prostate cancer following radiation therapy: retrospective analysis of iceball margins and outcomes. Eur Radiol. (2017) 27:4828–36. doi: 10.1007/s00330-017-4833-9
27. Siddiqui KM, Billia M, Al-Zahrani A, Williams A, Goodman C, Arifin A, et al. Long-term oncologic outcomes of salvage cryoablation for radio-recurrent prostate cancer. J Urol. (2016) 196:1105–11. doi: 10.1016/j.juro.2016.04.080
28. Lian H, Yang R, Lin T, Wang W, Zhang G, Guo H. Salvage cryotherapy with third-generation technology for locally recurrent prostate cancer after radiation therapy. Int Urol Nephrol. (2016) 48:1461–6. doi: 10.1007/s11255-016-1339-3
29. Kovac E, ElShafei A, Tay KJ, Mendez M, Polascik TJ, Jones JS. Five-year biochemical progression-free survival following salvage whole-gland prostate cryoablation: defining success with nadir prostate-specific antigen. J Endourol. (2016) 30:624–31. doi: 10.1089/end.2015.0719
30. Li R, Ruckle HC, Schlaifer AE, El-Shafei A, Yu C, Jones JS. The effect of androgen deprivation therapy before salvage whole-gland cryoablation after primary radiation failure in prostate cancer treatment. Urology. (2015) 85:1137–42. doi: 10.1016/j.urology.2014.12.025
31. Li YH, Elshafei A, Agarwal G, Ruckle H, Powsang J, Jones JS. Salvage focal prostate cryoablation for locally recurrent prostate cancer after radiotherapy: initial results from the cryo on-line data registry. Prostate. (2015) 75:1–7. doi: 10.1002/pros.22881
32. Spiess PE, Levy DA, Mouraviev V, Pisters LL, Jones JS. Predictors of biochemical failure in patients undergoing prostate whole-gland salvage cryotherapy: a novel risk stratification model. BJU Int. (2013) 112:E256–61. doi: 10.1111/j.1464-410X.2012.11695.x
33. Elkjær MC, Borre M. Only carefully selected patients may have a beneficial effect of salvage cryoablation in recurrent prostate cancer after radiotherapy. Dan Med J. (2013) 60:A4756.
34. de Castro Abreu AL, Bahn D, Leslie S, Shoji S, Silverman P, Desai MM, et al. Salvage focal and salvage total cryoablation for locally recurrent prostate cancer after primary radiation therapy. BJU Int. (2013) 112:298–307. doi: 10.1111/bju.12151
35. Ahmad I, Kalna G, Ismail M, Birrell F, Asterling S, McCartney E, et al. Prostate gland lengths and iceball dimensions predict micturition functional outcome following salvage prostate cryotherapy in men with radiation recurrent prostate cancer. PloS One. (2013) 8:e69243. doi: 10.1371/journal.pone.0069243
36. Spiess PE, Levy DA, Pisters LL, Mouraviev V, Jones JS. Outcomes of salvage prostate cryotherapy stratified by pre-treatment PSA: update from the COLD registry. World J Urol. (2013) 31:1321–5. doi: 10.1007/s00345-012-0982-2
37. Philippou P, Yap T, Chinegwundoh F. Third-generation salvage cryotherapy for radiorecurrent prostate cancer: a centre’s experience. Urol Int. (2012) 88:137–44. doi: 10.1159/000332220
38. Williams AK, Martínez CH, Lu C, Ng CK, Pautler SE, Chin JL. Disease-free survival following salvage cryotherapy for biopsy-proven radio-recurrent prostate cancer. Eur Urol. (2011) 60:405–10. doi: 10.1016/j.eururo.2010.12.012
39. Ng CK, Touma NJ, Chalasani V, Moussa M, Downey DB, Chin JL. The pattern of prostate cancer local recurrence after radiation and salvage cryoablation. Can Urol Assoc J. (2011) 5:E125–8. doi: 10.5489/cuaj.09116
40. Spiess PE, Katz AE, Chin JL, Bahn D, Cohen JK, Shinohara K, et al. A pretreatment nomogram predicting biochemical failure after salvage cryotherapy for locally recurrent prostate cancer. BJU Int. (2010) 106:194–8. doi: 10.1111/j.1464-410X.2009.09078.x
41. Pisters LL, Rewcastle JC, Donnelly BJ, Lugnani FM, Katz AE, Jones JS. Salvage prostate cryoablation: initial results from the cryo on-line data registry. J Urol. (2008) 180:559–63; discussion 563-4. doi: 10.1016/j.juro.2008.04.005
42. Ng CK, Moussa M, Downey DB, Chin JL. Salvage cryoablation of the prostate: followup and analysis of predictive factors for outcome. J Urol. (2007) 178:1253–7;discussion 1257. doi: 10.1016/j.juro.2007.05.137
43. Ismail M, Ahmed S, Kastner C, Davies J. Salvage cryotherapy for recurrent prostate cancer after radiation failure: a prospective case series of the first 100 patients. BJU Int. (2007) 100:760–4. doi: 10.1111/j.1464-410X.2007.07045.x
44. Donnelly BJ, Saliken JC, Ernst DS, Weber B, Robinson JW, Brasher PM, et al. Role of transrectal ultrasound guided salvage cryosurgery for recurrent prostate carcinoma after radiotherapy. Prostate Cancer Prostatic Dis. (2005) 8:235–42. doi: 10.1038/sj.pcan.4500811
45. Bahn DK, Lee F, Silverman P, Bahn E, Badalament R, Kumar A, et al. Salvage cryosurgery for recurrent prostate cancer after radiation therapy: a seven-year follow-up. Clin Prostate Cancer. (2003) 2:111–4. doi: 10.3816/cgc.2003.n.018
46. Chin JL, Pautler SE, Mouraviev V, Touma N, Moore K, Downey DB. Results of salvage cryoablation of the prostate after radiation: identifying predictors of treatment failure and complications. J Urol. (2001) 165:1937–41;discussion 1941-2. doi: 10.1097/00005392-200106000-00022
47. Ghafar MA, Johnson CW, de la Taille A, Benson MC, Bagiella E, Fatal M, et al. Salvage cryotherapy using an argon based system for locally recurrent prostate cancer after radiation therapy: the Columbia experience. J Urol. (2001) 166:1333–7;discussion 1337-8. doi: 10.1016/S0022-5347(05)65763-1
48. Chin JL, Lavi A, Metcalfe MJ, Siddiqui K, Dewar M, Petros FG, et al. Long-term outcomes of whole gland salvage cryotherapy for locally recurrent prostate cancer following radiation therapy: A combined analysis of two centers. J Urol. (2021) 206:646–54. doi: 10.1097/JU.0000000000001831
49. Bomers JGR, Overduin CG, Jenniskens SFM, Cornel EB, van Lin ENJT, Sedelaar JPM, et al. Focal salvage MR imaging-guided cryoablation for localized prostate cancer recurrence after radiotherapy: 12-month follow-up. J Vasc Interv Radiol. (2020) 31:35–41. doi: 10.1016/j.jvir.2019.07.001
50. Meng Y, Liu J, Shen B, Xu H, Wu D, Ying Y. Evaluation of the safety and efficacy of stereotactic body radiotherapy for radio-recurrent prostate cancer: a systematic review and meta-analysis. Prostate Cancer Prostatic Dis. (2024). doi: 10.1038/s41391-024-00927-8. [Epub ahead of print].
51. Xie S, Liu J, Shen B, Xu H, Ni J. Salvage low-dose vs high-dose brachytherapy for radio-recurrent prostate cancer. BJU Int. 2025. doi: 10.1111/bju.16639. [Epub ahead of print].
52. Anttinen M, Mäkelä P, Viitala A, Nurminen P, Suomi V, Sainio T, et al. Salvage magnetic resonance imaging-guided transurethral ultrasound ablation for localized radiorecurrent prostate cancer: 12-month functional and oncological results. Eur Urol Open Sci. (2020) 22:79–87. doi: 10.1016/j.euros.2020.10.007
53. Hopstaken JS, Bomers JGR, Sedelaar MJP, Valerio M, Fütterer JJ, Rovers MM. An updated systematic review on focal therapy in localized prostate cancer: what has changed over the past 5 years? Eur Urol. (2022) 81:5–33. doi: 10.1016/j.eururo.2021.08.005
54. Shah TT, Reddy D, Peters M, Ball D, Kim NH, Gomez EG, et al. Focal therapy compared to radical prostatectomy for non-metastatic prostate cancer: a propensity score-matched study. Prostate Cancer Prostatic Dis. (2021) 24:567–74. doi: 10.1038/s41391-020-00315-y
55. Abreu AL, Peretsman S, Iwata A, Shakir A, Iwata T, Brooks J, et al. High intensity focused ultrasound hemigland ablation for prostate cancer: initial outcomes of a United States series. J Urol. (2020) 204:741–7. doi: 10.1097/JU.0000000000001126
56. Oishi M, Gill IS, Tafuri A, Shakir A, Cacciamani GE, Iwata T, et al. Hemigland cryoablation of localized low, intermediate and high risk prostate cancer: oncologic and functional outcomes at 5 years. J Urol. (2019) 202:1188–98. doi: 10.1097/JU.0000000000000456
57. Lane JA, Donovan JL, Young GJ, Davis M, Walsh EI, Avery KNL, et al. Prostate Testing for Cancer and Treatment (ProtecT) Study Group. Functional and quality of life outcomes of localised prostate cancer treatments (Prostate Testing for Cancer and Treatment [ProtecT] study). BJU Int. (2022) 130:370–80. doi: 10.1111/bju.15739
58. Wenske S, Quarrier S, Katz AE. Salvage cryosurgery of the prostate for failure after primary radiotherapy or cryosurgery: long-term clinical, functional, and oncologic outcomes in a large cohort at a tertiary referral centre. Eur Urol. (2013) 64:1–7. doi: 10.1016/j.eururo.2012.07.008
Keywords: prostate cancer, recurrence, radio-recurrent, salvage, cryotherapy
Citation: Zhu S, Liu J, Shen B, Xu H, Zhong W and Jin S (2025) Oncological effects and complications of salvage cryotherapy for radio-recurrent prostate cancer: a systematic review and meta-analysis. Front. Oncol. 15:1534739. doi: 10.3389/fonc.2025.1534739
Received: 26 November 2024; Accepted: 27 February 2025;
Published: 03 April 2025.
Edited by:
Maurizio Brausi, Hesperia Hospital, ItalyReviewed by:
Konstantinos Ferentinos, German Oncology Center, CyprusCopyright © 2025 Zhu, Liu, Shen, Xu, Zhong and Jin. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sheng Jin, ajEzNjc2ODY2MDYyQDE2My5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.