
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Oncol. , 01 April 2025
Sec. Neuro-Oncology and Neurosurgical Oncology
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1525427
 Sanghyeok Park1
Sanghyeok Park1 Jung Han Seo1
Jung Han Seo1 Sang Woo Song1
Sang Woo Song1 Young Hyun Cho1
Young Hyun Cho1 Chang-Ki Hong1
Chang-Ki Hong1 Jeong Hoon Kim1
Jeong Hoon Kim1 Ho Sung Kim2
Ho Sung Kim2 Ji Eun Park2
Ji Eun Park2 Soo Jung Nam3
Soo Jung Nam3 Young-Hoon Kim1*
Young-Hoon Kim1*Purpose: Pilocytic astrocytomas (PA) in adult patients are rare and the efficacy of postoperative adjuvant treatments remains unclear. This study aims to investigate the survival outcome and prognostic factors in surgically treated adult PA.
Methods: A total of 90 consecutive adult patients with newly diagnosed PA were enrolled. Among the patients, 47 (52%) were male, with a median age of 28 years (18–70 years). Preoperative neurological deficits were observed in 43 (48%) patients. The most common tumor locations were cerebellar and cerebral hemispheres (28% and 27%, respectively), while 23% of tumors were located in deeper brain structures. The median follow-up duration was 88months (12–304 months).
Results: Gross total removal (GTR) was achieved in 55 (61%) patients. At the final follow-up, 12 (13%) patients had died, and 23 (26%) experienced disease progression. The 1, 2, and 5-year overall survival (OS) rates were 93%, 91%, and 87%, respectively, while the progression-free survival (PFS) rates were 88%, 80%, and 77%, respectively. The recurrence rate in patients who underwent GTR was 11%, compared with 53% and 45% in those without GTR, with or without adjuvant treatments, respectively. The tumors in the deeper brain locations had significantly lower GTR rates (14%) compared with other locations (75%; p < 0.001). Multivariate analysis identified the absence of preoperative neurological deficits (p = 0.048; HR = 2.878), not deeper tumor location (p = 0.017; HR = 3.471) and GTR (p = 0.007; HR = 3.884) as significant factors for improved PFS.
Conclusion: Adult PA exhibited more aggressive behavior compared with pediatric PA. These aggressive behaviors including preoperative neurological deficits, deeper tumor location, and lower GTR rates were significantly associated with poor prognosis.
Pilocytic astrocytomas (PA) are primary brain tumors classified as World Health Organization (WHO) grade 1 (1–3). They are the most common pediatric brain tumors in individuals aged 0–14 years and the second most common in those aged 15–19 years, accounting for 15.2% of all primary brain and central nervous system (CNS) tumors in individuals under 19 years. The incidence of PA in this age group is 0.95 per 100,000 person-years (4). In the pediatric population, PA typically occur in the cerebellum, whereas in adults, they are more frequently located in the cerebral hemisphere, followed by the cerebellum (5).
Despite their generally benign characteristics, some studies suggest that PA in adult patients may exhibit more aggressive behavior than in pediatric patients (5–8). However, Brown et al. suggested that PA in adults are as benign as those in children (9). The behavior of PA in adults remains poorly understood due to the rarity of the condition in this population, with an incidence of less than 0.1 per 100,000 person-years in individuals over 45 years old (4). The standard treatment for PA is surgical resection, with gross total removal (GTR) offering the most favorable prognosis. However, achieving GTR can be challenging, particularly for tumors located in deep regions such as the thalamus or brainstem. In such cases where GTR is not feasible, adjuvant radiation therapy (RTx) or stereotactic radiosurgery may be considered in some centers (10, 11). The efficacy of RTx in these situations, however, remains controversial (12, 13).
The aim of this study was to evaluate the survival outcomes of surgically treated adult PA and to identify prognostic factors in this patient population.
A total of 97 consecutive adult patients were diagnosed with primary PA between 1999 and 2022 at our institution. Inclusion criteria required patients to be aged 18 years or older, have histopathologically confirmed PA, a newly diagnosed primary PA, and a minimum clinical and radiological follow-up of 12 months. Two (2%) patients were excluded due to insufficient radiological data, and five (5%) were excluded due to loss of follow-up. Ultimately, 90 (93%) patients were enrolled in the study. All clinical information and neuroimaging data were collected with the approval of our institutional review board. This study was conducted in accordance with the Declaration of Helsinki, and informed consent was obtained from all participants.
All patients underwent preoperative and postoperative magnetic resonance imaging (MRI) within 48 hours of surgery. Patients were followed for more than 6 months after surgery, with additional MRI evaluations to assess the extent of resection. The extent of resection was determined by comparing preoperative and postoperative MRIs. GTR was defined as no visible residual tumor on the immediate postoperative MRI. Subtotal removal (STR) was defined as less than 10% of the tumor remaining, while partial removal (PR) was defined as more than 10% of the tumor remaining on postoperative MRI. Biopsy was defined as obtaining tissue for histopathological examination (14).
At our institution, GTR was the primary surgical objective for suspected adult PA. If GTR was achieved, patients were monitored with follow-up imaging without additional treatment. For residual or recurrent tumors, reoperation was considered if feasible. If reoperation was not possible, follow-up observation was prioritized.
For deep-seated tumors with a high risk of progression or challenging reoperations, adjuvant therapy such as RTx, chemotherapy (CTx), or stereotactic radiosurgery (SRS) were considered. In cases of recurrence, salvage treatments, including reoperation, RTx, CTx or SRS, were performed.
Data were collected retrospectively by reviewing medical records and radiological findings. Collected variables included sex, age at diagnosis, presenting symptoms, presence of neurological deficits, tumor location, tumor size, infiltration, cyst components, gadolinium enhancement on brain MRI, perilesional edema, hydrocephalus, pathologic reports, extent of tumor removal, residual tumor size, and use of adjuvant or salvage treatments. The associations between clinical and radiological factors and progression-free survival (PFS) were analyzed.
OS and PFS were the primary endpoints. OS was defined as the time from initial diagnosis to death, while PFS was defined as the time from initial treatment to tumor recurrence or progression, as determined by radiological findings. Cumulative rates of OS and PFS were evaluated using Kaplan–Meier survival methods. Prognostic factors for PFS were analyzed using logistic regression and Cox proportional hazard models, employing a backward stepwise method. To reduce the risk of type II errors due to the modest sample size, variables were included in the multivariate analysis only if they had a p-value of < 0.05 in univariate analyses. A p-value of < 0.05 was considered statistically significant. All statistical analyses were conducted using SPSS ver. 21.0 (SPSS Inc, Chicago, IL).
Among the 90 patients, 47 (52%) were male and 43 (48%) were female. The median age at diagnosis was 28 years old (range: 18–71 years), with 50 (56%) patients diagnosed before the age of 30. The most common preoperative symptoms were headache (54%), dizziness (31%), nausea and vomiting (23%), seizure (20%), visual disturbances such as diplopia (19%), gait disturbance (14%), and motor weakness (10%). Notably, three patients (3%) were asymptomatic. Forty-three (48%) patients presented with preoperative neurological deficits.
The most common tumor location was the cerebellar hemisphere (25 patients, 28%) followed by the cerebral hemisphere (24 patients, 27%), cerebellar vermis (15 patients, 17%), and lateral ventricle (5 patients, 5%). Twenty-one patients (23%) had tumors in deeper locations, such as the brainstem, third or fourth ventricle, basal ganglia, thalamus, pineal gland, or suprasellar area. The median tumor size was 4.0 cm (range: 1.2–8.0 cm), with 11 tumors (12%) showing infiltrative features. Thirty-seven patients (41%) presented with preoperative hydrocephalus, and 11 (12%) underwent external ventricular drainage or endoscopic third ventriculostomy prior to surgery. The median follow-up period was 88 months (range, 12–304 months). A summary of preoperative clinical and radiological characteristics is provided in Table 1.
A summary of surgical and survival outcomes is presented in Table 2. Of the 90 patients who underwent surgery for PA, GTR was achieved in 55 (61%) patients. STR, PR, and biopsy were performed in 10 (11%), 12 (13%), and 13 (14%) patients, respectively. The median residual tumor was 2.8 cm (range: 1.0–5.6 cm). GTR rates highest in tumors located in the cerebral (88%) and cerebellar hemisphere (76%), whereas tumors in the deeper brain locations had significantly lower GTR rates (14%) compared with other locations (75%; p < 0.001). Infiltrative tumors (18%) and predominantly solid (51%) tumors had significantly lower GTR rates compared with circumscribed (67%) and cystic (79%) tumors (p = 0.003 and 0.013, respectively). Tumor size, enhancement, peri-tumoral edema, and hydrocephalus were not significantly associated with GTR rates (p = 0.384, 1.000, 0.270, and 0.278, respectively).
The outcomes associated with adjuvant or salvage treatments are depicted in Figure 1; Table 2. None of the 55 patients who underwent GTR received adjuvant treatments. Of the 35 patients who did not receive GTR, 20 (57%) received adjuvant therapy. Tumor recurrence occurred in 6 (11%) patients who had undergone GTR, compared with 17 (49%) of the 35 patients without GTR, regardless of whether they received adjuvant therapy (45% or 53%, respectively).

Figure 1. Treatment protocols and surgical outcomes of adult patients with pilocytic astrocytomas (n = 90). Among the 55 patients who underwent GTR, none received adjuvant treatments. Of the 35 patients who did not achieve GTR, 20 patients (57%) underwent adjuvant treatments. Tumor recurrence occurred in 6 patients (11%) after GTR, whereas 17 patients (49%) without GTR showed disease progression, regardless of adjuvant treatments (45% or 53%, respectively). GTR, gross total removal; Tx, treatments; Reop, reoperation; RTx, radiation therapy; SRS, stereotactic radiosurgery; CTx, chemotherapy; CR, complete remission; SD, stable disease; PD, progressive disease.
Among the 35 patients without GTR, four underwent reoperation, with only 1 (25%) showing disease progression. Of the remaining 31 patients without reoperation, 16 (52%) had tumor recurrence. After initial disease progression, 12 patients (52%) died due to disease progression, irrespective of the type of salvage. The treatment failure rates for recurrent tumors were 50% (5/10) for salvage surgery, 80% (4/5) for RTx, 40% (2/5) for SRS, and 67% (4/6) for CTx.
During the follow-up period, 23 patients (26%) showed disease progression, and 12 (13%) died due to disease progression. The mean PFS was 223.0 months (95% confidence interval [CI], 194.2–251.7 months), with cumulative 1-, 2-, and 5-year PFS rates of 87.8%, 79.6%, and 77.1%, respectively. Mean OS was 257.5 months (95% CI, 232.7–282.2 months), with cumulative 1-, 2-, and 5-year OS rates of 93.2%, 90.8%, and 86.6%, respectively. The survival outcomes are summarized in Table 2.
Prognostic factors for PFS in adult patients with PA are analyzed in Table 3, and Kaplan–Meier PFS curves for various clinical and radiological factors are presented in Figure 2. Patient sex (Figure 2A), tumor size (Figure 2E), cystic morphology (Figure 2G), enhancement (Figure 2H), peri-tumoral edema (Figure 2I), preoperative hydrocephalus (Figure 2J), and pathology (Figure 2K) were not significantly associated with PFS (p = 0.704, 0.255, 0.200, 0.151, 0.614, 0.156, and 0.308, respectively). Patient age (Figure 2B) was marginally associated with PFS, but did not reach statistical significance (p = 0.055).
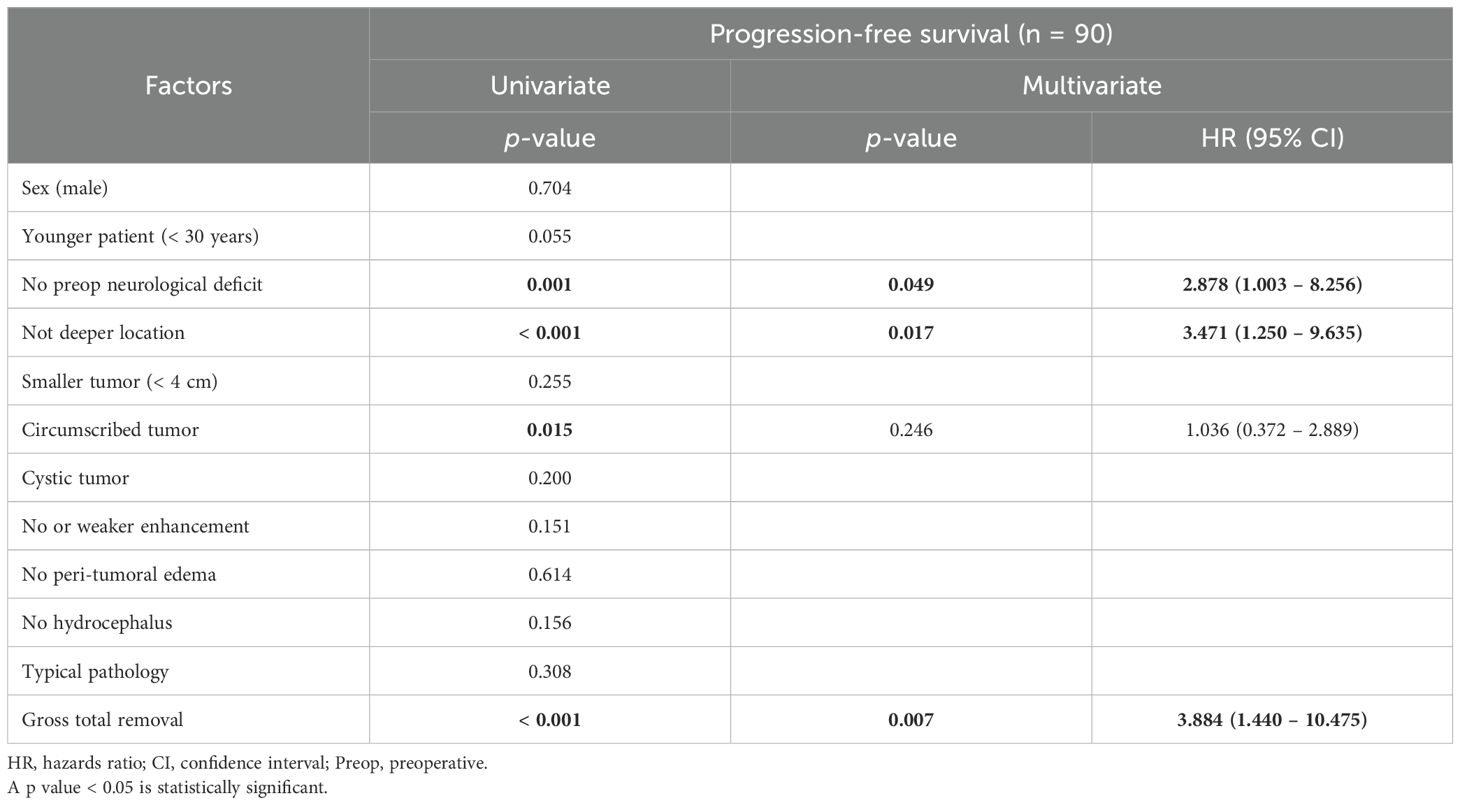
Table 3. Favorable prognostic factors for progression-free survival in adult patients who underwent surgical treatments for pilocytic astrocytomas (n = 90).
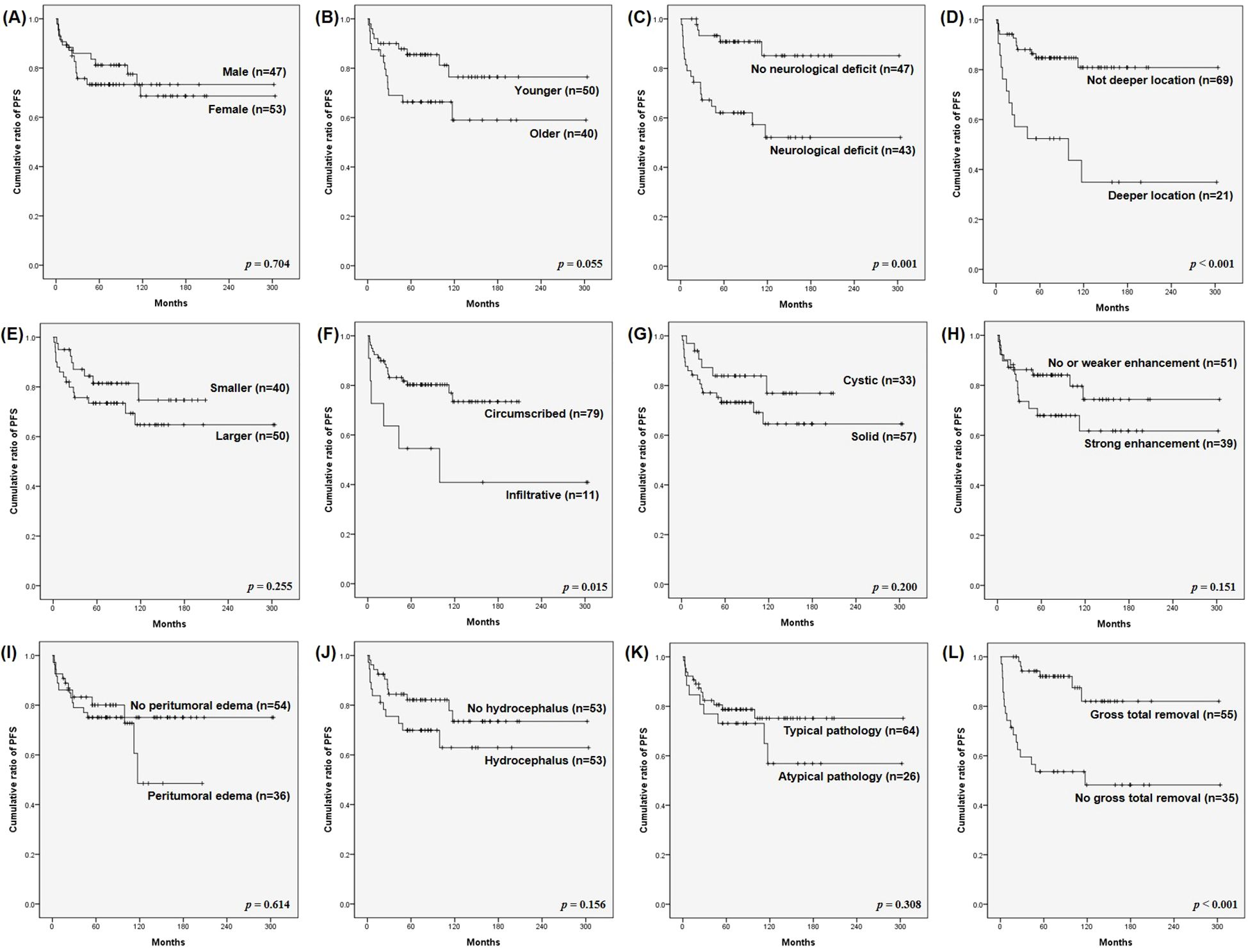
Figure 2. Kaplan–Meier survival curves for progression-free survival (PFS) of surgically treated adult pilocytic astrocytomas (PA). PFS were analyzed based on: (A) patient sex, (B) patient age, (C) preoperative neurological deficit, (D) tumor location, (E) tumor size, (F) infiltrative vs. circumscribed tumor pattern, (G) cystic vs. solid tumor, (H) enhancement pattern, (I) peritumoral edema, (J) hydrocephalus, (K) typical pathology, and (L) extent of resection. The (C) absence of preoperative neurological deficit, (D) non-deep tumor location, (F) circumscribed tumor pattern, and (L) gross total removal (GTR) were significant favorable prognostic factors for PFS of adult PA (p = 0.001, < 0.001, 0.015, and < 0.001, respectively).
In contrast, the absence of preoperative neurological deficits, non-deeper tumor location, circumscribed tumors, and GTR were significant favorable prognostic factors for PFS (p = 0.001, < 0.001, 0.015, and < 0.001, respectively). The 5-year PFS rates were significantly higher for patients without preoperative neurological deficits (Figure 2C), non-deep tumors (Figure 2D), circumscribed tumors (Figure 2F), and GTR (Figure 2L) (85.1%, 80.9%, 73.4%, and 82.0%, respectively) compared with those with preoperative neurological deficits, deep tumors, infiltrative tumors, and without GTR (52.1%, 34.9%, 40.9%, and 48.2%, respectively) (p = 0.001, < 0.001, 0.015, and < 0.001, respectively).
In the multivariate analysis, the absence of preoperative neurological deficits (p = 0.049; hazard ratio [HR] = 2.878; 95% CI, 1.003–8.256), not deeper tumor location (p = 0.017; HR = 3.471; 95% CI, 1.250–9.635) and GTR (p = 0.007; HR = 3.884; 95% CI, 1.440–10.475) were the only significant favorable prognostic factors.
PA are WHO grade I primary brain tumors with a characteristic biphasic architecture, comprising densely packed fibrillary tissue and looser microcystic compartments. They typically display Rosenthal fibers and eosinophilic granular bodies (1, 3). PA are the most common primary brain tumor in the pediatric population (0–18 years), representing 15.2% of all primary CNS tumors in this age group (4). In the pediatric population, PA generally have an indolent nature and favorable prognosis, with 5-year OS rates nearing 100% and 10-year survival rates of 95.8% (12). However, adult PA are less well-characterized and can exhibit more aggressive behavior, with an incidence of less than 0.1 per 100,000 person-years in a population over 45 years (4). Stüer et al. reported a 30% tumor recurrence rate in adult PA, with 18% of patients succumbing to the disease over a 10-year follow-up period (15). Similarly, Theeler et al. reported a recurrence rate of 42% and reported 13 tumor-related deaths over a 22-year study of 127 adult patients with PA (6).
Our study aligns with these findings, with more than half of patients (50 of 90, 56%) diagnosed before the age of 30 years. Tumor locations were similar to those reported in other studies, with cerebellar hemisphere (28%), cerebral hemisphere (27%), and cerebellar vermis (17%) being the most common. According to the National Cancer Institute Surveillance data from 1973 to 2008, 27% and 30% of adult PA were located in the cerebellum and cerebrum, respectively (5). Our study reported a tumor progression rate of 26% (23 of 90) and a mortality rate of 13% (12 of 90), both significantly higher than in pediatric populations (5, 7, 16).
In our study, younger patients exhibited relatively favorable PFS compared with older patients, although this difference was not statistically significant (p = 0.055). Previous studies have consistently shown that pediatric PA has a more favorable prognosis than adult PA (5, 6), with increasing age of diagnosis associated with more aggressive tumor behavior. Johnson et al. concluded that the varying prognosis across age groups is attributed to differences in PA aggressiveness (5). While age at diagnosis was a significant prognostic factor in their multivariate analysis, this was not the case in our study (5). This discrepancy may be attributed to differences in how patient age groups were categorized. Johnson et al. divided patients into several age brackets (under 5 years, under 20 years, under 40 years, under 60 years, and over 60 years) (5), which allowed them to demonstrate a prognostic impact of age. In contrast, we divided our cohort into just two groups: those under 30 and those over 30.
Tumor location and characteristics significantly impacted progression in our study, though these effects were largely due to the strong correlation between tumor location, features, and extent of resection. The three factors identified as significant factors for prognosis in this study, such as the presence or absence of preoperative neurological deficits, the location of the tumor, and the GTR, seem to be all related. In the case of tumors located deeper in the brain, the possibility of preoperative neurological deficits is high and the probability of GTR is significantly reduced. It can be seen that this aggressive behaviors of adult PA affects a poor prognosis.
GTR remains the most important factor influencing survival and prognosis in adult PA. Stüer et al. found that the recurrence rate for partially resected tumors was four times higher than for completely resected tumors (15). Similarly, Johnson et al. reported a significantly lower hazard ratio (0.3) for death in patients who underwent GTR compared with those who had STR or biopsy (5). Our results further confirm the positive impact of GTR on PFS. Even if GTR is not achieved during the initial surgery, reoperation with complete tumor resection can still yield favorable outcomes.
In cases where GTR is not feasible, reoperation to achieve complete resection offers the best prognosis. However, when reoperation is not possible, adjuvant therapy, such as RTx, should be considered for patients with residual or recurrent tumors. In our study, the effects of RTx on residual or recurrent tumors did not show significantly better outcomes compared with other treatments or no treatment. The role of postoperative adjuvant RTx remains controversial, with conflicting reports regarding its efficacy. Ishkanian et al. reported superior PFS in patients who received adjuvant RTx in a retrospective analysis, supporting its use in adults (17). In contrast, Theeler et al. found significantly reduced PFS with adjuvant RTx (6).
The observed association between adjuvant RTx and higher progression rates is likely a result of patient selection bias, as RTx was primarily recommended for high-risk patients based on clinical, radiological, or pathologic factors. Further randomized prospective studies with larger populations are needed to elucidate the efficacy of adjuvant RTx for adult PA.
The primary limitation of this study is its retrospective nature, conducted at a single institution. To account for the rarity of adult PA, we collected data over a 20-year period. During this time, surgical techniques and equipment, as well as diagnostic criteria, evolved. Consequently, there is a potential for misdiagnosis, particularly in earlier cases. In the current era, molecular diagnosis of brain tumors has gained prominence, and future studies will require detailed pathological analysis, including immunohistochemistry, to better establish prognostic factors for adult PA. For example, in the case of KIAA1549-BRAF fusion, which is expressed in 70-80% of pilocytic astocytoma, this mutation increases the possibility of gross total removal and makes it possible to predict a better prognosis. Therefore, molecular biological diagnosis of tumor tissue can be said to be an essential process for future glioma research (18).
Adult PA tend to exhibit more aggressive behavior compared with pediatric PA. In our study, 26% of patients experienced tumor recurrence, and more than half of these patients died due to disease progression. The absence of preoperative neurological deficits, not deeper tumor location and GTR were the only significant prognostic factors associated with favorable PFS in adult PA. Further research is needed to better understand the efficacy and optimal selection of adjuvant or salvage treatments for adult PA.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
The studies involving humans were approved by the Institutional Review Board of Asan Medical Center (Approval number: S2024-2253). The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants' legal guardians/next of kin in accordance with the national legislation and institutional requirements.
SP: Conceptualization, Data curation, Formal analysis, Methodology, Validation, Writing – original draft, Writing – review & editing. JS: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing – original draft. SS: Conceptualization, Investigation, Resources, Supervision, Validation, Visualization, Writing – review & editing. YC: Conceptualization, Formal analysis, Investigation, Resources, Supervision, Validation, Visualization, Writing – review & editing. C-KH: Conceptualization, Investigation, Resources, Supervision, Validation, Visualization, Writing – review & editing. JK: Conceptualization, Resources, Writing – review & editing. HK: Conceptualization, Resources, Supervision, Validation, Visualization, Writing – review & editing. JP: Investigation, Resources, Supervision, Validation, Visualization, Writing – review & editing. SN: Investigation, Resources, Validation, Visualization, Writing – review & editing. Y-HK: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Louis DN, Perry A, Reifenberger G, Deimling Av, Figarella−Branger D, Cavenee WK, et al. The 2016 world health organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. (2016) 131:803–20. doi: 10.1007/s00401-016-1545-1
2. Chen Y-H, Gutmann D. The molecular and cell biology of pediatric low-grade gliomas. Oncogene. (2014) 33:2019–26. doi: 10.1038/onc.2013.148
3. Sadighi Z, Slopis J. Pilocytic astrocytoma: A disease with evolving molecular heterogeneity. J Child Neurol. (2013) 28:625–32. doi: 10.1177/0883073813476141
4. Ostrom QT, Cioffi G, Waite K, Kruchko C, Barnholtz-Sloan JS. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2014–2018. Neuro-Oncology. (2021) 23:iii1–iii105. doi: 10.1093/neuonc/noab200
5. Johnson DR, Brown PD, Galanis E, Hammack JE. Pilocytic astrocytoma survival in adults: analysis of the Surveillance, Epidemiology, and End Results Program of the National Cancer Institute. J Neuro-oncol. (2012) 108:187–93. doi: 10.1007/s11060-012-0829-0
6. Theeler BJ, Ellezam B, Sadighi ZS, Mehta V, Tran MD, Adesina AM, et al. Adult pilocytic astrocytomas: clinical features and molecular analysis. Neuro-oncology. (2014) 16:841–7. doi: 10.1093/neuonc/not246
7. Yang W, Porras JL, Khalafallah AM, Sun Y, Bettegowda A, Mukherjee D. Comparison of adult and pediatric pilocytic astrocytomas using competing risk analysis: A population-based study. Clin Neurol Neurosurg. (2022) 212:107084. doi: 10.1016/j.clineuro.2021.107084
8. Muhsen BA, Aljariri AI, Elayyan M, Hirbawi H, Masri MA. Insight about the characteristics and surgical resectability of adult pilocytic astrocytoma: tertiary center experience. CNS Oncol. (2022) 11:CNS81. doi: 10.2217/cns-2021-0014
9. Brown PD, Anderson SK, Carrero XW, O’Neill BP, Giannini C, Galanis E, et al. Adult patients with supratentorial pilocytic astrocytoma: long-term follow-up of prospective multicenter clinical trial NCCTG-867251 (Alliance). Neuro-oncol Pract. (2015) 2:199–204. doi: 10.1093/nop/npv031
10. Trifiletti DM, Peach MS, Xu Z, Kersh R, Showalter TN, Sheehan JP. Evaluation of outcomes after stereotactic radiosurgery for pilocytic astrocytoma. J Neurooncol. (2017) 134:297–302. doi: 10.1007/s11060-017-2521-x
11. Hafez RFA, Fahmy OM, Hassan HT, Ganz JC. Gamma Knife Radiosurgery for symptomatic eloquently deep-seated cystic pilocytic astrocytoma mural nodules: Retrospective case series of effective outcomes. Acta Neurochir (Wien). (2024) 166:466. doi: 10.1007/s00701-024-06366-7
12. Burkhard C, Patre P-LD, Schüler D, Schüler G, Yas¸Argil MG, Yonekawa Y, et al. A population-based study of the incidence and survival rates in patients with pilocytic astrocytoma. J Neurosurg. (2003) 98:1170–4. doi: 10.3171/jns.2003.98.6.1170
13. Khalafallah AM, Jimenez AE, Shah PP, Brem H, Mukherjee D. Effect of radiation therapy on overall survival following subtotal resection of adult pilocytic astrocytoma. J Clin Neurosci. (2020) 81:340–5. doi: 10.1016/j.jocn.2020.10.020
14. Byun J, Kim YH, Nam SJ, Park JE, Cho YH, Kim HS, et al. Comparison of survival outcomes between partial resection and biopsy for primary glioblastoma: A propensity score-matched study. World Neurosurg. (2019) 121:e858–66. doi: 10.1016/j.wneu.2018.09.237
15. Stüer C, Vilz B, Majores M, Becker A, Schramm J, Simon M. Frequent recurrence and progression in pilocytic astrocytoma in adults. Cancer. (2007) 110:2799–808. doi: 10.1002/cncr.v110:12
16. Kim JW, Phi JH, Kim SK, Lee JH, Park SH, Won JK, et al. Comparison of the clinical features and treatment outcomes of pilocytic astrocytoma in pediatric and adult patients. Childs Nerv Syst. (2023) 39:583–91. doi: 10.1007/s00381-023-05839-x
17. Ishkanian A, Laperriere NJ, Xu W, Millar B-A, Payne D, Mason W, et al. Upfront observation versus radiation forAdult pilocytic astrocytoma. Cancer. (2011) 117:4070–9. doi: 10.1002/cncr.v117.17
Keywords: pilocytic astrocytoma, adult, prognosis, gross total removal, neurological deficit, deep location
Citation: Park S, Seo JH, Song SW, Cho YH, Hong C-K, Kim JH, Kim HS, Park JE, Nam SJ and Kim Y-H (2025) Survival outcomes and prognostic factors in surgically treated adult pilocytic astrocytomas. Front. Oncol. 15:1525427. doi: 10.3389/fonc.2025.1525427
Received: 09 November 2024; Accepted: 18 March 2025;
Published: 01 April 2025.
Edited by:
Rony Benson, Mar Sleeva Medicity, IndiaReviewed by:
Marcello D’Andrea, U.O. Neurochirurgia Ospedale “M. Bufalini”, ItalyCopyright © 2025 Park, Seo, Song, Cho, Hong, Kim, Kim, Park, Nam and Kim. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Young-Hoon Kim, anVzdGluNTI3QGhhbm1haWwubmV0
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.