
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Oncol. , 18 March 2025
Sec. Hematologic Malignancies
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1517223
This article is part of the Research Topic Metabolism Dysregulation in Hematologic Malignancies View all articles
 Yin Jin
Yin Jin Wenfei Gu*
Wenfei Gu*Background: The effect of the controlling nutritional status (CONUT) score on forecasting multiple myeloma (MM) prognosis is previously analyzed, whereas the results remained inconsistent. The present meta-analysis focused on identifying the exact function of CONUT in forecasting MM prognosis.
Methods: Web of Science, PubMed, Embase, CNKI, and Cochrane Library were comprehensively searched between inception and 1 February 2025. The effect of CONUT on forecasting MM overall survival (OS) and progression-free survival (PFS) was determined by computing pooled hazard ratios (HRs) together with 95% confidence intervals (CIs).
Results: There were nine studies with 1,176 patients being recruited into the present work. As indicated by our pooled data, elevated CONUT was related to the dismal OS (HR = 1.87, 95% CI = 1.37–2.54, p < 0.001) of patients with MM. Nonetheless, CONUT was not significantly related to PFS (HR = 1.33, 95% CI = 0.81–2.19, p = 0.254) of MM. Furthermore, higher CONUT score showed a significant relationship to bone marrow plasma cells >30% (OR = 2.30, 95% CI = 1.32–3.99, p = 0.003). On the other hand, CONUT was not markedly correlated with gender (OR = 2.68, 95% CI = 0.81–8.82, p = 0.105), ISS stage (OR = 1.28, 95% CI = 0.94–1.75, p = 0.119), or ECOG PS (OR = 1.30, 95% CI = 0.84–2.01, p = 0.234) of MM.
Conclusion: Collectively, according to our results in this meta-analysis, higher CONUT score is markedly related to dismal OS, but not PFS in patients with MM. CONUT score can be used as a candidate marker used to predict MM prognosis in the clinic in the future.
Multiple myeloma (MM), the plasma cell-malignant cancer accumulating within the bone marrow, can result in bone damage and marrow failure (1). Generally, malignant plasma cells can be detected from the bone marrow, which produce abnormal antibodies (M-protein) (2). MM ranks second among hematological malignancies, occurring in 6.5 people per 100,000 worldwide (3). According to GLOBOCAN, 176,404 newly diagnosed MM cases along with 117,077 death cases were reported in 2020 globally (4). There have been considerable advancements in treatment options for MM in recent decades, including proteasome inhibitors, immunomodulatory drugs, and anti-CD38 antibody immunotherapies (5). Despite these advances, most MM patients eventually relapse and cannot be cured, with 5- and 10-year survival rates of 55.6% and 17%, respectively (6, 7). Prognostic markers are important to improve the survival outcomes from MM (8). Consequently, identifying new and reliable markers for predicting MM prognosis is urgently needed.
Current lines of evidence show that inflammation and nutrition play pivotal roles in cancer progression and development (9). In recent years, many nutrition-related indexes, including prognostic nutritional index (PNI) (10), geriatric nutritional risk index (GNRI) (11), albumin-to-globulin ratio (12), and C-reactive protein-to-albumin ratio (CAR) (13), represent key markers for predicting cancer prognosis. First proposed by Ignacio et al. in 2005, the controlling nutritional status (CONUT) score has been used to detect hospital undernutrition (14). In peripheral blood, cholesterol, albumin, and lymphocyte levels can be easily calculated to determine the CONUT score, which ranges from 0 to 12 (Table 1). Many studies have shown the prominent significance of CONUT on predicting cancer prognosis such as pancreatic ductal carcinoma (15), cervical cancer (16), esophageal squamous cell carcinoma (17), colon cancer (18), and non-small cell lung cancer (NSCLC) (19). The impact of CONUT on predicting MM prognosis is analyzed previously, whereas conflicting results are reported (20–28). Consequently, this meta-analysis focused on examining the exact impact of CONUT on forecasting MM prognosis. Additionally, we also investigated the relationship of CONUT with clinicopathological factors of MM through this meta-analysis.
This meta-analysis was carried out following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines (29).
PubMed, Web of Science, Embase, Cochrane Library, and CNKI were thoroughly searched between inception and 1 February 2025 using the search items below: (controlling nutritional status score or CONUT or controlling nutritional status) and (multiple myeloma or myeloma). There was no restriction on the language of publications. Reference lists of electronically selected articles were checked manually to obtain more relevant articles.
Articles were included based on the following criteria: 1) pathological diagnosis of MM was made; 2) the CONUT was determined based on pretreatment peripheral blood test; 3) those that mentioned the association between CONUT and MM survival outcomes; (4) those that provided available or computed hazard ratios (HRs) and 95% confidence intervals (CIs); (5) threshold CONUT was available to stratify the patients; and (6) studies published in any language. The following articles were eliminated: 1) reviews, comments, letters, meeting abstracts, and case reports; 2) publications with duplicated or overlapped data; and 3) animal studies.
Two authors (YJ and WG) independently collected data from the qualified articles. Any dispute between them was solved through discussion until reaching a consensus. The following data were extracted: first author, year, country, sample size, gender, age, study design, study period, International Staging System (ISS) stage, threshold, threshold determination approach, follow-up, survival endpoints, and survival analysis types, as well as HRs with 95% CIs. We selected overall survival (OS) and progression-free survival (PFS) as our primary and secondary survival outcomes, respectively. Newcastle-Ottawa Scale (NOS) scores were used to evaluate study quality (30), with studies scoring 6 or higher considered high quality.
The effect of CONUT on forecasting MM OS and PFS was analyzed through calculating pooled HRs and 95% CIs. Among-study heterogeneities were assessed through Cochran’s Q-test and I2 statistics. I2 >50% and p <0.10 (Q-test) indicate obvious heterogeneity, so the random-effects model is chosen; otherwise, the fixed-effects model is used. Meanwhile, the prognostic value of CONUT in various patient subgroups was investigated through subgroup analysis. Also, the heterogeneity source was identified, and the pooled result stability was evaluated by conducting sensitivity analysis. The relationship of CONUT with MM clinicopathological characteristics was assessed based on pooled odds ratios (ORs) and 95% CIs. Begg’s test and Egger’s test were adopted in evaluating publication bias. Stata version 12 (StataCorp, College Station, TX, USA) was applied in the statistical analyses. A p-value <0.05 indicated statistical significance.
Through primary search, a total of 61 studies were obtained, with 47 being maintained when removing duplicates (Figure 1). Through title and abstract checking, 36 studies were excluded because of irrelevance and animal studies. Subsequently, the full texts of 11 studies were assessed, with two being eliminated due to not focusing on MM (n = 1) and no survival data provided (n = 1). Ultimately, nine studies with 1,176 patients (20–28) were enrolled for the present meta-analysis (Figure 1).
Table 2 displays the identified study features. The publication year was 2020–2024 (20–28). Two studies were conducted in Japan (20, 21) and seven in China (22–28). Three studies were published in English (20, 21, 24) and six were in Chinese (22, 23, 25–28). Their sample sizes were 57–245 (median, 119). The enrolled studies were of retrospective design. Eight studies recruited ISS stage I–III patients (20–26, 28) and one enrolled patient with recurrent disease (27). In terms of cutoff value, three studies adopted ≥3.5 (22, 23, 27), three used ≥5 (20, 21, 28), two selected ≥6.5 (25, 26), and one used ≥3 (24). Six studies determined the cutoff value by using the receiver operating characteristic (ROC) curve (20, 22, 23, 25–27), two studies referred to the literature (21, 28), and one adopted the X-tile software (24). All nine studies mentioned the relationship of CONUT with OS (20–28) and three studies showed the significance of CONUT in predicting PFS (22, 26, 28) in MM. Five articles obtained the HRs and 95% CIs from univariate regression (22, 25–28) and four studies adopted multivariate analysis (20, 21, 23, 24). The NOS scores were 7–9, suggesting high quality (Table 2).
All nine studies (20–28) provided the information regarding the impact of CONUT on forecasting OS of MM patients. Due to significant heterogeneity (I2 = 70.4%, p = 0.001), this study adopted the random-effects model. As indicated by our combined data, higher CONUT was connected with unfavorable OS of MM patients (HR = 1.87, 95% CI = 1.37–2.54, p < 0.001; Table 3, Figure 2). As shown by subgroup analysis, high CONUT remained the significant prognostic indicator of dismal OS, despite country, sample size, stage, threshold determination, or survival analysis (all p < 0.05; Table 3).
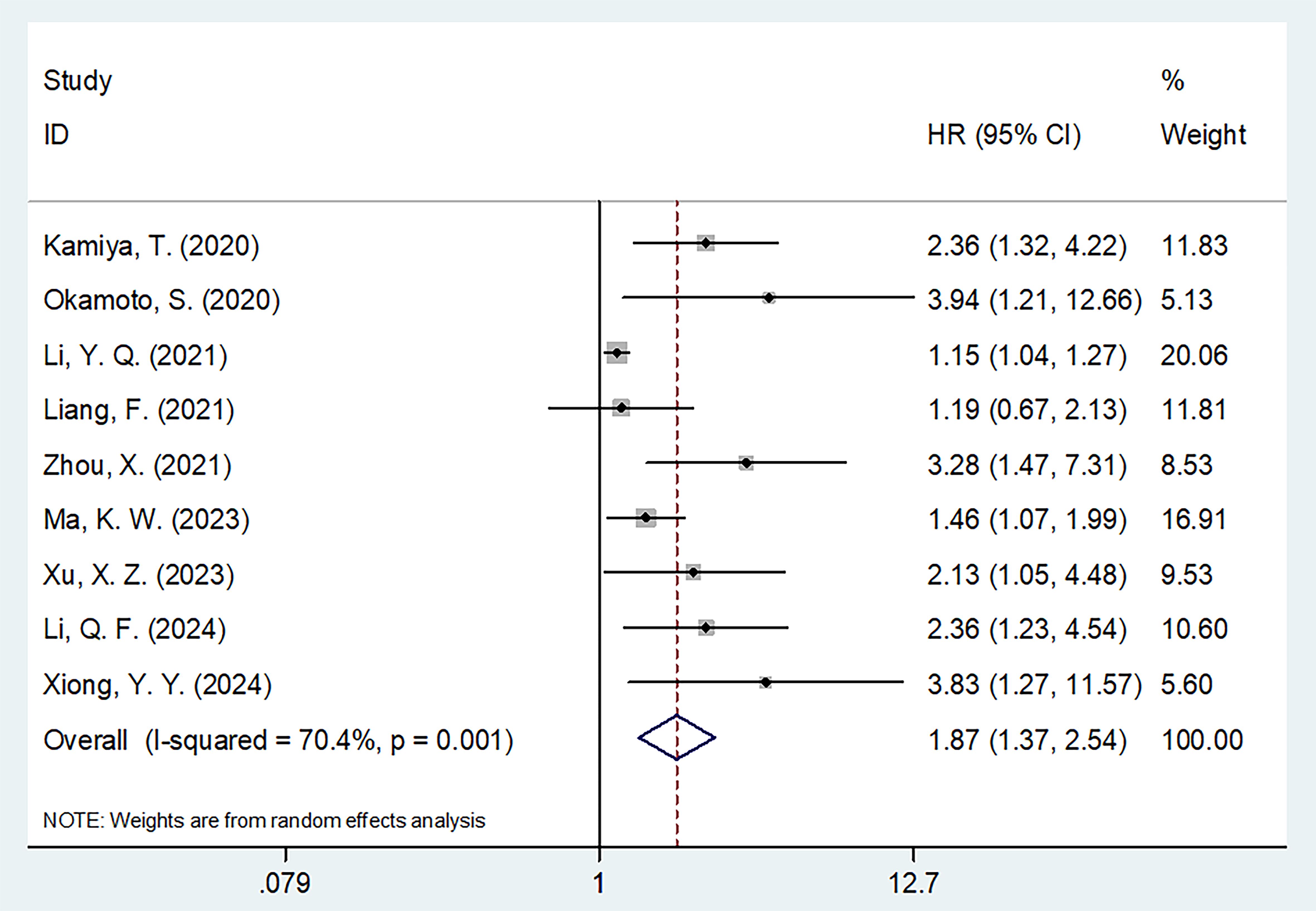
Figure 2. Forest plots of the association between CONUT score and OS in patients with multiple myeloma.
Three studies consisting of 255 patients (22, 26, 28) reported the correlation between CONUT and PFS in MM. The combined results suggested HR = 1.33, 95% CI = 0.81–2.19, p = 0.254, indicating that CONUT was not a significant prognostic marker for PFS in MM (Table 3; Figure 3). Subgroup analysis demonstrated that elevated CONUT exhibited obvious relationship to PFS in MM using the threshold determined according to the literature (p = 0.012; Table 3).
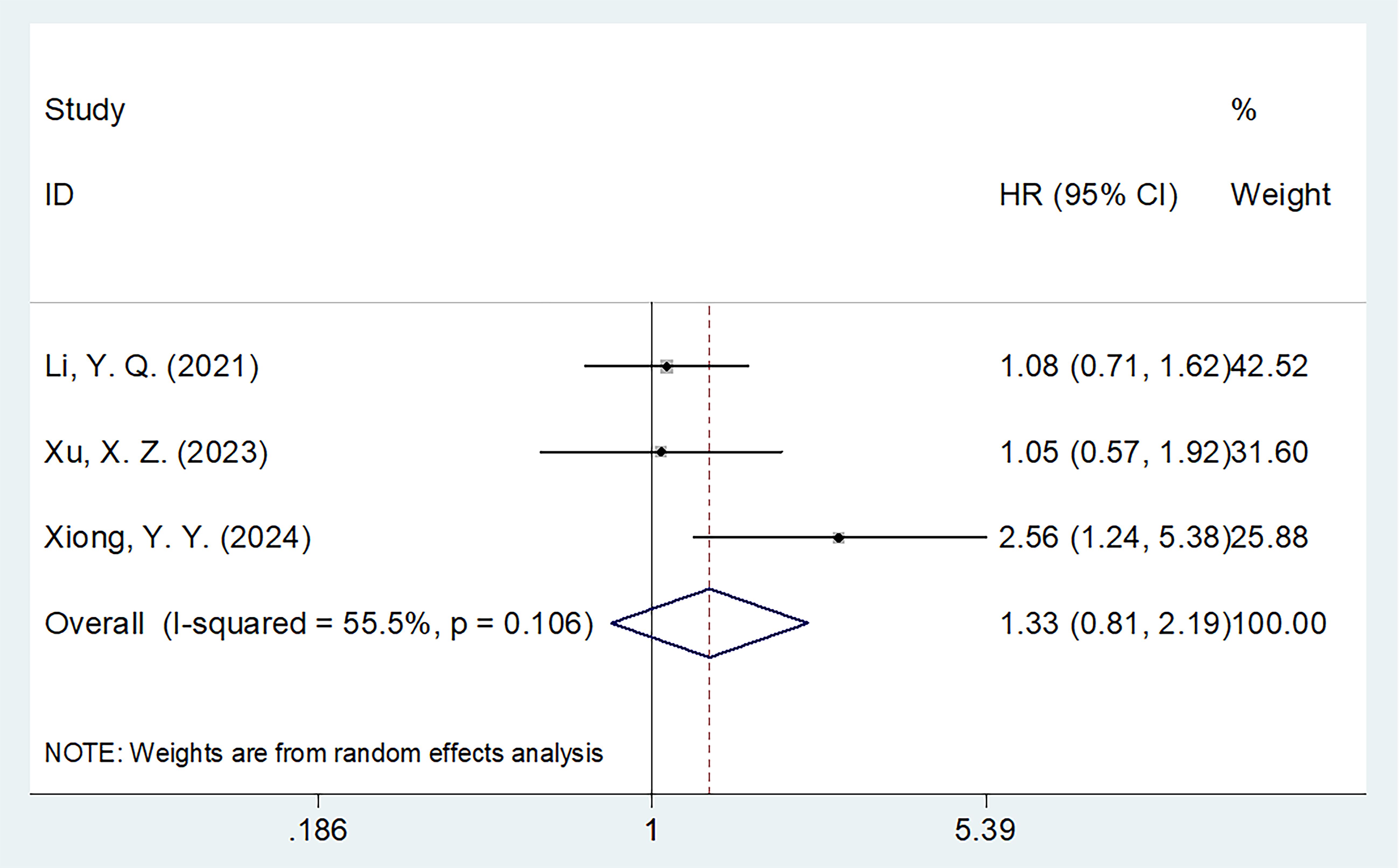
Figure 3. Forest plots of the association between CONUT score and PFS in patients with multiple myeloma.
A total of six studies including 877 cases (22–25, 27, 28) presented the relationship of CONUT with MM clinicopathological factors. Based on our combined findings, higher CONUT score showed a significant relationship to bone marrow plasma cells >30% (OR = 2.30, 95% CI = 1.32–3.99, p = 0.003; Table 4, Figure 4). Nonetheless, CONUT was not apparently connected with gender (OR = 2.68, 95% CI = 0.81–8.82, p = 0.105), ISS stage (OR = 1.28, 95% CI = 0.94–1.75, p = 0.119), or Eastern Cooperative Oncology Group Performance Status (ECOG PS) (OR = 1.30, 95% CI = 0.84–2.01, p = 0.234) of MM (Table 4, Figure 4).
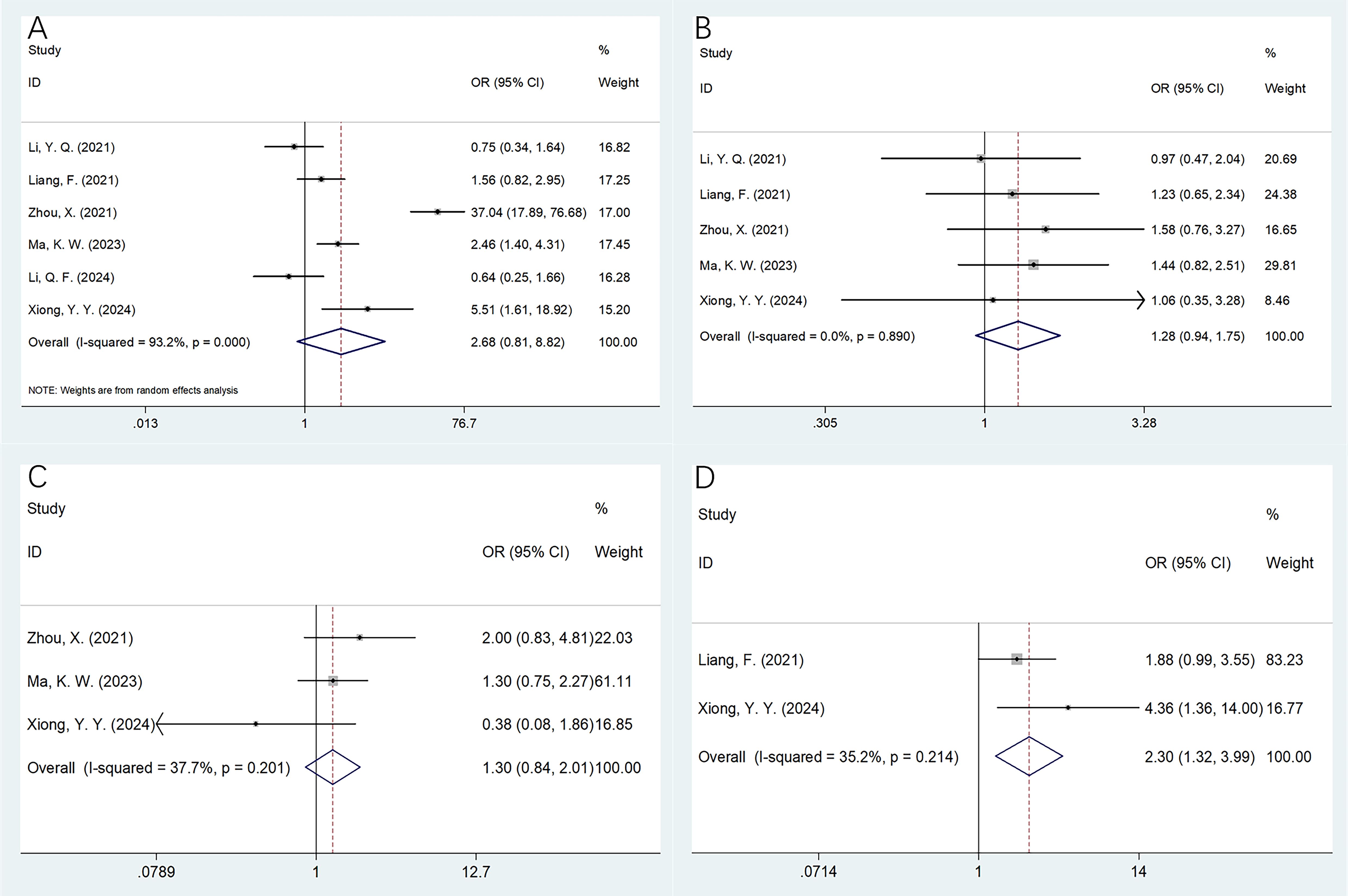
Figure 4. Forest plots of the correlation between CONUT and clinicopathological features of multiple myeloma. (A) Gender (male vs. female); (B) ISS stage (III vs. I–II); (C) ECOG PS (≥2 vs. <2); and (D) bone marrow plasma cells (>30% vs. ≤30%).
Sensitivity analysis indicated that no single study significantly impacted the effect size for the relationship between CONUT and OS or PFS of MM (Figure 5), which suggested that our results were reliable.
This study utilized Begg’s and Egger’s tests in examining potential publication bias, which existed in the present work (p = 0.118 and 0.102 for OS; p = 0.296 and 0.462 for PFS through Begg’s and Egger’s tests, respectively) (Figure 6).
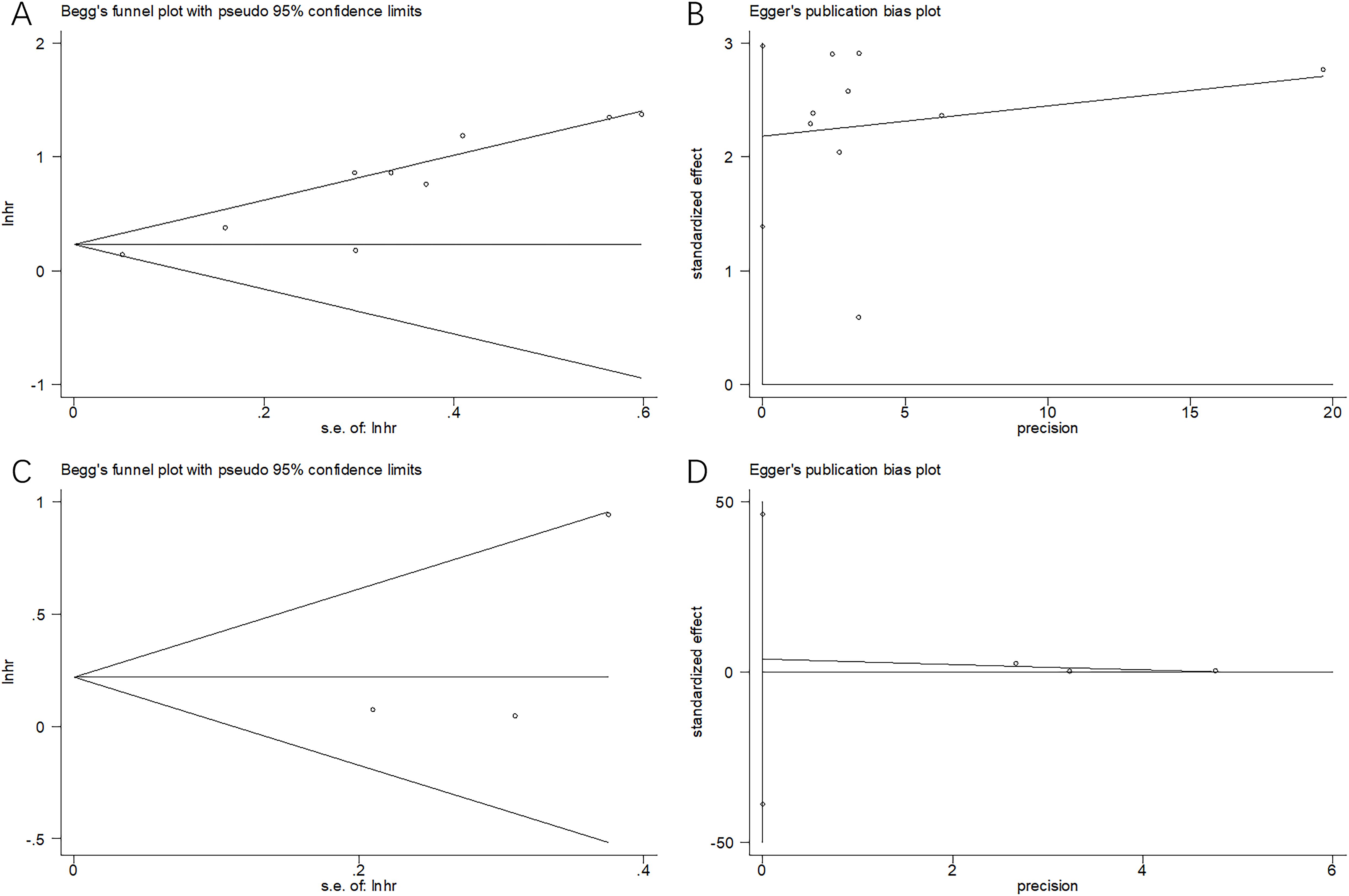
Figure 6. Publication bias test. (A) Begg’s test for OS, p = 0.118; (B) Egger’s test for OS, p = 0.102; (C) Begg’s test for PFS, p = 0.296; and (D) Egger’s test for PFS, p = 0.462.
The value of CONUT in forecasting MM prognosis is widely explored, with controversial results presented. This work aggregated data in nine articles with 1,176 patients to shed light on this issue. As discovered, higher CONUT significantly forecasted the dismal OS of MM. Nonetheless, CONUT was not significantly related to PFS in patients with MM. Furthermore, according to our meta-analysis, higher CONUT showed an obvious relationship to bone marrow plasma cells >30% in MM. Our results were robust, as validated by sensitivity and publication bias analyses. Higher CONUT was a significant prognostic factor for OS, but not PFS in MM. For the first time, this meta-analysis investigated the effect of CONUT on predicting MM prognosis.
CONUT is calculated by serum albumin, cholesterol, and total lymphocyte count, and reductions of these three elements lead to high CONUT. The mechanisms related to the effect of CONUT on predicting OS in MM remain to be fully illustrated, yet they may be explained below. Firstly, a patient’s serum albumin, the most common plasma protein, can serve as an objective measure of their nutritional status (31). Albumin directly indicates a host’s nutritional status, and it performs an essential function, such as damage repair and material transport, during pathophysiological processes (32). Lower serum albumin contents are indicative of chronic inflammation activation and low nutritional status. Secondly, as a key component in the human immune system, lymphocytes suppress carcinogenesis and recurrence by producing cytokines and inducing cytotoxic death, thereby regulating immune function (33). The lymphocytes are also critical to maintaining the adaptive immune system, and a decreased lymphocyte count is associated with dismal prognostic outcome of many cancers (34). It is common for lymphocytopenia to be accompanied by an increase in leukocytes, which may allow tumor cells to hide from the immune system (35). Thirdly, in healthy cells, cholesterol is a key structural component of lipid metabolism, and it impacts the membrane properties like function and fluidity of membrane proteins (36). The presence of cholesterol on the cellular membrane is closely related to tumor proliferation. Moreover, patients having lower cholesterol levels usually do not show the best prognostic outcome (37). Cancer development and tumorigenesis are linked to cholesterol metabolism disorders. Consequently, a reduction of serum cholesterol suggests the reduction of cell membrane cholesterol, which affects the cancer-fighting capacity of immune-active cells.
Notably, this meta-analysis revealed that CONUT was a significant prognostic marker for patients with MM, which could be considered an effective biomarker in clinical practice. Current lines of evidence have shown that there are many types of prognostic markers for MM, such as serum free light chain (FLC) levels (38), generation sequencing (NGS) panels (39), circulating tumor cells (CTCs) (40), proteomics panels (41), and genomic biomarkers (8, 42). Compared with these prognostic markers, CONUT score has the following strengths. First, CONUT is easily available. CONUT is derived from blood test results, being commonly examined in clinical management. Second, the prognostic efficiency of CONUT is reliable. The current meta-analysis indicated that CONUT score is a significant prognostic biomarker for long-term survival outcomes in MM. Third, the CONUT test is cost-effective and did not increase medical expenses because CONUT can be calculated from blood tests and no specific examination for CONUT is needed. Therefore, CONUT could serve as a promising and cost-effective prognostic marker for MM in clinical settings.
Recently, many articles also reported the effect of CONUT on predicting cancer prognosis through meta-analysis (43–47). Wang et al. showed that higher CONUT score showed a close relationship with OS and disease-free survival (DFS) in head and neck cancer through their meta-analysis including 1,478 cases (43). As reported by Li et al., increased CONUT scores showed a close relationship to the dismal OS and PFS of lymphoma in their meta-analysis involving seven studies (44). As demonstrated by one latest meta-analysis comprising 17 articles, a high CONUT score increased the risk of tumor progression, advanced tumor stage, microvascular invasion, and postoperative complications in gastric cancer patients (45). Niu et al. discovered from a meta-analysis involving 2,569 cases that increased CONUT scores had a close relationship to the reduced OS and PFS of gynecological cancer (46). According to Peng et al. in a meta-analysis including 3,029 cases, a high CONUT score was positively related to dismal prognoses in NSCLC (47). Our results conform to the prognostic value of CONUT in additional cancers.
This work has some limitations. Firstly, our studies were from Asia, mainly China. Therefore, the results were more applicable to Asian MM patients. Secondly, our sample was limited in size. Although nine studies were included, the total sample size was 1,176, which was relatively small. Thirdly, all included studies were retrospective, which might introduce selection bias. Consequently, large-scale multicenter prospective studies should be conducted for further validation.
In summary, a higher CONUT score is apparently related to shortened OS but not PFS in patients with MM. CONUT score can be a candidate marker to predict MM prognosis clinically.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
YJ: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Resources, Software, Supervision, Validation, Visualization, Writing – original draft. WG: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Validation, Visualization, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. van de Donk N, Pawlyn C, Yong KL. Multiple myeloma. Lancet. (2021) 397:410–27. doi: 10.1016/s0140-6736(21)00135-5
2. Swerdlow SH, Campo E, Pileri SA, Harris NL, Stein H, Siebert R, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms. Blood. (2016) 127:2375–90. doi: 10.1182/blood-2016-01-643569
3. Manier S, Ingegnere T, Escure G, Prodhomme C, Nudel M, Mitra S, et al. Current state and next-generation CAR-T cells in multiple myeloma. Blood Rev. (2022) 54:100929. doi: 10.1016/j.blre.2022.100929
4. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA: Cancer J Clin. (2021) 71:209–49. doi: 10.3322/caac.21660
5. Pinto V, Bergantim R, Caires HR, Seca H, Guimarães JE, Vasconcelos MH. Multiple myeloma: available therapies and causes of drug resistance. Cancers (Basel). (2020) 12. doi: 10.3390/cancers12020407
6. Podar K, Leleu X. Relapsed/refractory multiple myeloma in 2020/2021 and beyond. Cancers (Basel). (2021) 13. doi: 10.3390/cancers13205154
7. Kumar SK, Dispenzieri A, Lacy MQ, Gertz MA, Buadi FK, Pandey S, et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia. (2014) 28:1122–8. doi: 10.1038/leu.2013.313
8. Wallington-Beddoe CT, Mynott RL. Prognostic and predictive biomarker developments in multiple myeloma. J Hematol Oncol. (2021) 14:151. doi: 10.1186/s13045-021-01162-7
9. Mantovani A, Allavena P, Sica A, Balkwill F. Cancer-related inflammation. Nature. (2008) 454:436–44. doi: 10.1038/nature07205
10. Li B, Lu Z, Wang S, Hou J, Xia G, Li H, et al. Pretreatment elevated prognostic nutritional index predicts a favorable prognosis in patients with prostate cancer. BMC Cancer. (2020) 20:361. doi: 10.1186/s12885-020-06879-1
11. Sakamoto T, Makinoya M, Sunaguchi T, Goto K, Morimoto M, Murakami Y, et al. Geriatric nutritional risk index as a prognostic factor in patients with recurrent pancreatic cancer. PloS One. (2022) 17:e0271073. doi: 10.1371/journal.pone.0271073
12. Taguchi S, Kawai T, Nakagawa T, Nakamura Y, Kamei J, Obinata D, et al. Prognostic significance of the albumin-to-globulin ratio for advanced urothelial carcinoma treated with pembrolizumab: a multicenter retrospective study. Sci Rep. (2021) 11:15623. doi: 10.1038/s41598-021-95061-z
13. Alkurt EG, Durak D, Turhan VB, Sahiner IT. Effect of C-reactive protein-to-albumin ratio on prognosis in gastric cancer patients. Cureus. (2022) 14:e23972. doi: 10.7759/cureus.23972
14. Ignacio de Ulíbarri J, González-Madroño A, de Villar NG, González P, González B, Mancha A, et al. CONUT: a tool for controlling nutritional status. First validation in a hospital population. Nutricion hospital. (2005) 20:38–45.
15. Kuwabara S, Takeuchi Y, Sato O, Mizota T, Ichinokawa M, Murakawa K, et al. Prognostic value of combined psoas muscle mass and controlling nutritional status in patients with pancreatic ductal adenocarcinoma: a retrospective cohort study. BMC Surg. (2024) 24:116. doi: 10.1186/s12893-024-02395-2
16. Fu J, Xu X, Tian M, Wang H, Jin X. The controlling nutritional status score as a new prognostic predictor in patients with cervical cancer receiving radiotherapy: a propensity score matching analysis. BMC Cancer. (2024) 24:1093. doi: 10.1186/s12885-024-12872-9
17. Fang P, Zhou J, Liang Z, Yang Y, Luan S, Xiao X, et al. The prognostic value of controlling nutritional status score on esophageal squamous cell carcinoma patients with neoadjuvant therapy followed by esophagectomy-a retrospective research. J Thorac Dis. (2024) 16:4460–73. doi: 10.21037/jtd-24-187
18. Cozzani F, Ricchiuto M, Virgilio E, Viani L, Rossini M, Pedrazzi G, et al. The controlling nutritional status (CONUT) score predicts post-operatory risks and prognosis in patients with surgically treated colon cancer: A retrospective study. Anticancer Res. (2024) 44:3955–64. doi: 10.21873/anticanres.17224
19. Jiang S, Wang X, Xing Y, Wu J, Yuan X. Value of prognostic nutritional index and controlling nutritional status score for advanced non-small cell lung cancer patients receiving PD-1 inhibitors. Am J Cancer Res. (2024) 14:2894–904. doi: 10.62347/xqhl4852
20. Kamiya T, Ito C, Fujita Y, Ogura S, Mizuno K, Sakurai A, et al. The prognostic value of the controlling nutritional status score in patients with multiple myeloma. Leuk Lymph. (2020) 61:1894–900. doi: 10.1080/10428194.2020.1749608
21. Okamoto S, Ureshino H, Kidoguchi K, Kusaba K, Kizuka-Sano H, Sano H, et al. Clinical impact of the CONUT score in patients with multiple myeloma. Ann Hematol. (2020) 99:113–9. doi: 10.1007/s00277-019-03844-2
22. Li YQ, Wang Y, Song Y, Han Y, Li JJ, Yue LL, et al. The influence of CONUT score on the prognosis of patients with multiple myeloma. J Exp Hematol. (2021) 29:781–6. doi: 10.19746/j.cnki.issn.1009-2137.2021.03.020
23. Liang F, Dong XY, Tang GF, Qi KM, Chen W, Sang W, et al. Influence of prognostic nutritional index and controlling nutritional status on the prognosis of patients with multiple myeloma. Chin J Hematol. (2021) 42:332–7. doi: 10.3760/cma.j.issn.0253-2727.2021.04.011
24. Zhou X, Lu Y, Xia J, Mao J, Wang J, Guo H. Association between baseline Controlling Nutritional Status score and clinical outcomes of patients with multiple myeloma. Cancer biomark: sect A Dis Markers. (2021) 32:65–71. doi: 10.3233/cbm-210073
25. Ma KW, Ye JN, Mao JJ, Zhou X, Sun C, Li JY. Analysis of long-term prognostic factors based on nutritional status in patients with multiple myeloma. J Exp Hematol. (2023) 31:455–61. doi: 10.19746/j.cnki.issn.1009-2137.2023.02.021
26. Xu XZ, Liu R, Zhao WH, Yang Y, Liu J, Zhang WG, et al. Alteration and significance of serum lipid levels and nutritional status during BCMA-CAR-T-cell therapy in patients with refractory or relapsed multiple myeloma: a retrospective study based on LEGEND-2. Chin J Hematol. (2023) 44:838–44. doi: 10.3760/cma.j.issn.0253-2727.2023.10.008
27. Li QF, Zhang QK, Wei XF, Feng YF, Fu Y, Zhao YY, et al. Effects of different nutritional scoring systems on prognosis of elderly patients with multiple myeloma. J Exp Hematol. (2024) 32:499–504. doi: 10.19746/j.cnki.issn.1009-2137.2024.02.027
28. Xiong YY, Zhou Q, Chen L, Yu W, Zhang HB, Chen JB. Effects of pre-transplant CONUT and post-transplant MRD on prognosis of patients with multiple myeloma after auto-HSCT. J Exp Hematol. (2024) 32:146–54. doi: 10.19746/j.cnki.issn.1009-2137.2024.01.023
29. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA Statement. Open Med. (2009) 3:e123–30.
30. Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. (2010) 25:603–5. doi: 10.1007/s10654-010-9491-z
31. Gupta D, Lis CG. Pretreatment serum albumin as a predictor of cancer survival: a systematic review of the epidemiological literature. Nutr J. (2010) 9:69. doi: 10.1186/1475-2891-9-69
32. Zeng X, Liu G, Pan Y, Li Y. Prognostic value of clinical biochemistry-based indexes in nasopharyngeal carcinoma. Front Oncol. (2020) 10:146. doi: 10.3389/fonc.2020.00146
33. Dunn GP, Old LJ, Schreiber RD. The immunobiology of cancer immunosurveillance and immunoediting. Immunity. (2004) 21:137–48. doi: 10.1016/j.immuni.2004.07.017
34. Fumagalli LA, Vinke J, Hoff W, Ypma E, Brivio F, Nespoli A. Lymphocyte counts independently predict overall survival in advanced cancer patients: a biomarker for IL-2 immunotherapy. J immunother (Hagerstown Md: 1997). (2003) 26:394–402. doi: 10.1097/00002371-200309000-00002
35. Quigley DA, Kristensen V. Predicting prognosis and therapeutic response from interactions between lymphocytes and tumor cells. Mol Oncol. (2015) 9:2054–62. doi: 10.1016/j.molonc.2015.10.003
36. Resnik N, Sepcic K, Plemenitas A, Windoffer R, Leube R, Veranic P. Desmosome assembly and cell-cell adhesion are membrane raft-dependent processes. J Biol Chem. (2011) 286:1499–507. doi: 10.1074/jbc.M110.189464
37. Zhou P, Li B, Liu B, Chen T, Xiao J. Prognostic role of serum total cholesterol and high-density lipoprotein cholesterol in cancer survivors: A systematic review and meta-analysis. Clinica chimica acta; Int J Clin Chem. (2018) 477:94–104. doi: 10.1016/j.cca.2017.11.039
38. Tacchetti P, Cavo M, Rocchi S, Pezzi A, Pantani L, Brioli A, et al. Prognostic impact of serial measurements of serum-free light chain assay throughout the course of newly diagnosed multiple myeloma treated with bortezomib-based regimens. Leuk Lymph. (2016) 57:2058–64. doi: 10.3109/10428194.2015.1124994
39. Walker BA, Boyle EM, Wardell CP, Murison A, Begum DB, Dahir NM, et al. Mutational spectrum, copy number changes, and outcome: results of a sequencing study of patients with newly diagnosed myeloma. J Clin Oncol. (2015) 33:3911–20. doi: 10.1200/jco.2014.59.1503
40. Chakraborty R, Muchtar E, Kumar SK, Jevremovic D, Buadi FK, Dingli D, et al. Risk stratification in myeloma by detection of circulating plasma cells prior to autologous stem cell transplantation in the novel agent era. Blood Cancer J. (2016) 6:e512. doi: 10.1038/bcj.2016.117
41. Dytfeld D, Rosebeck S, Kandarpa M, Mayampurath A, Mellacheruvu D, Alonge MM, et al. Proteomic profiling of naïve multiple myeloma patient plasma cells identifies pathways associated with favourable response to bortezomib-based treatment regimens. Br J Haematol. (2015) 170:66–79. doi: 10.1111/bjh.13394
42. Chretien ML, Corre J, Lauwers-Cances V, Magrangeas F, Cleynen A, Yon E, et al. Understanding the role of hyperdiploidy in myeloma prognosis: which trisomies really matter? Blood. (2015) 126:2713–9. doi: 10.1182/blood-2015-06-650242
43. Wang Y, Qian C. Prognostic and clinicopathological value of the controlling nutritional status (CONUT) score in patients with head and neck cancer: a meta-analysis. World J Surg Oncol. (2024) 22:223. doi: 10.1186/s12957-024-03505-3
44. Li L, Shou L. Prognostic and clinicopathological significance of the Controlling Nutritional Status (CONUT) score in patients with lymphoma: a meta-analysis. BMJ Open. (2024) 14:e078320. doi: 10.1136/bmjopen-2023-078320
45. Yin J, Qu J, Liang X, Wang M. Prognostic significance of controlling nutritional status score for patients with gastric cancer: A systematic review and meta−analysis. Exp Ther Med. (2023) 25:202. doi: 10.3892/etm.2023.11901
46. Niu Z, Yan B. Prognostic and clinicopathological impacts of Controlling Nutritional Status (CONUT) score on patients with gynecological cancer: a meta-analysis. Nutr J. (2023) 22:33. doi: 10.1186/s12937-023-00863-8
Keywords: CONUT score, multiple myeloma, meta-analysis, evidence-based medicine, prognosis
Citation: Jin Y and Gu W (2025) Prognostic and clinicopathological value of the controlling nutritional status score in patients with multiple myeloma: a meta-analysis. Front. Oncol. 15:1517223. doi: 10.3389/fonc.2025.1517223
Received: 25 October 2024; Accepted: 28 February 2025;
Published: 18 March 2025.
Edited by:
Jiao Ma, Shanghai Jiao Tong University, ChinaCopyright © 2025 Jin and Gu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wenfei Gu, d2YxMTM4NTg1QDE2My5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.