
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Oncol. , 28 March 2025
Sec. Thoracic Oncology
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1496752
This article is part of the Research Topic Case Reports in Pulmonary Medicine 2024 View all 30 articles
Lung carcinomas usually spread to the liver, lungs, pleura, pericardium, adrenal glands, brain, and bones. Anaplastic lymphoma kinase gene (ALK) fusion occurs in approximately 5% of non-small cell lung cancer (NSCLC) cases and most frequently in adenocarcinoma. Here, we report a rare case of intestinal metastasis originating from pulmonary pleomorphic carcinoma in a 49-year-old male heavy smoker. At the local hospital, the patient was initially considered to have an ALK-positive intestinal tumor, leading to a differential diagnosis of inflammatory myofibroblastic tumor (IMT). Due to the tumor’s peculiar morphology (including epithelioid and spindle cell components), pathologists of the local hospital sent slides of the case to our hospital for further consultation. Immunohistochemical analysis revealed that the epithelioid and spindle neoplastic cells were positive for CK7, TTF1, and ALK-V. Fluorescence in situ hybridization (FISH) confirmed the presence of the echinoderm microtubule-associated protein-like 4 (EML4):: ALK fusion. Based on these findings, we established the final diagnosis as intestinal metastasis of ALK-positive pulmonary pleomorphic carcinoma. A subsequent enhanced CT scan of the chest revealed a 3.0 cm solid mass in the right upper lung, further supporting the diagnosis of intestinal metastasis originating from pulmonary pleomorphic carcinoma. In conclusion, this case exhibited highly unusual clinicopathological features that could easily lead to misdiagnosis as primary intestinal tumors with ALK rearrangement. Pathologists must know this possibility to ensure accurate diagnosis and appropriate management.
Lung cancer is mainly divided into small-cell lung carcinoma (SCLC) and non-small cell carcinoma (NSCLC). The NSCLC primarily includes squamous cell carcinoma (SqCC), adenocarcinoma (ADC), large cell carcinoma, adenosquamous carcinoma, and other types of NSCLC (1). Pulmonary pleomorphic carcinoma (PLC) is a pretty rare type of NSCLC, which is composed of a mesenchymal component of spindle cells, giant cells, or both, with a sarcomatoid tumor component of more than 10% according to the 2021 World Health Organization’s (WHO) histological classification of lung tumors. Moreover, reports indicate that it accounts for no more than 0.5% of lung carcinomas (1).
There are many genetic abnormalities that have been described, the most common genes including tumor protein p53 (TP53), KRAS proto-oncogene (KRAS), and epidermal growth factor receptor (EGFR) (1). Notably, anaplastic lymphoma kinase gene (ALK) rearrangement occurs in approximately 5% of pulmonary PLCs (2), which is similar to that in cases with non-small cell lung cancer (NSCLC) and is most often seen in ADC (3).
The most frequent metastatic sites of pulmonary PLC are the liver, adrenal glands, brain, and bones. Metastases to the intestine from lung cancer are uncommon and only account for about 8% of all lung cancer cases in autopsy (4) and even less in clinical incidence (5, 6), let alone the first clinical indication.
Researchers have only reported a small number of lung cancers with intestinal metastasis, including few ones with ALK rearrangement, but most of them are ADC subtypes instead of PLCs (7–9). Here, we described an extremely rare case of EML4::ALK positive pulmonary PLC with intestinal metastasis, which histologically mimics inflammatory myofibroblastic tumor (IMT).
A 49-year-old heavy-smoker (37 pack per year) male patient was admitted to the local hospital with acute abdomen pain. The patient has no history of chronic diseases. Neither the patient nor their family members have a history of cancer. An enhanced computed tomography (CT) scan revealed a 4.0 cm soft tissue mass within the lumen of the small intestine and mid-lower intestine intussusception (Figure 1).
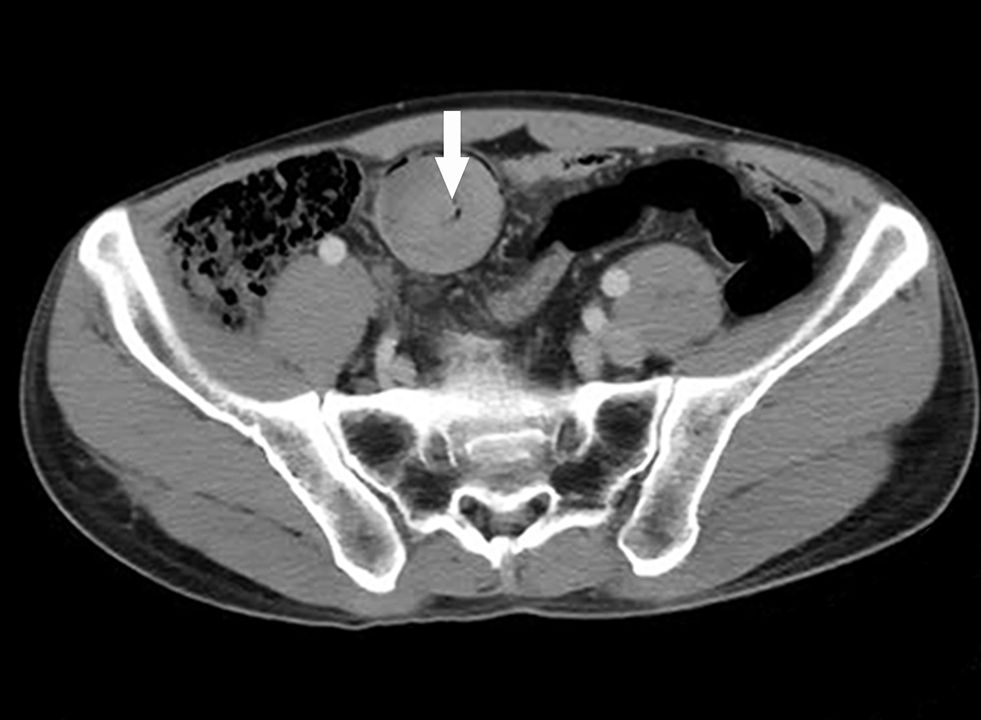
Figure 1. Enhanced computed tomography (CT) scan showing a well-defined soft tissue mass (arrow) in the lumen of the ileum.
The patient was admitted to the general surgery ward, and an intussusception surgery was performed on April 8, 2019. An irregular, hard, solid white mass involving the intestine was identified during surgery. The patient underwent complete resection of the mass with a partial small intestine resection. The resected specimen comprised a multinodular mass with a tan-white cut surface and firm consistency, whereas a thin envelope was observed in the periphery of the mass. ALK-positive IMT was diagnosed and was highly suspicious for epithelioid inflammatory myofibroblastic sarcoma (EIMS) for showing ALK(5A4) positive in neoplastic cells immunochemically. Our department received the consultation slides from the local hospital.
Histologically, in low-power fields, the tumor was located within the submucosa and lamina propria of the intestinal wall with mucosal erosion (Figure 2A), and the neoplastic cells were embedded within the inflammatory background. In high power fields, it showed that neoplastic components of fat spindle-shaped cells and large epithelioid cells with ovoid nuclei interwoven with each other distributed in the background of many neutrophil cells (Figure 2B, C). Mitotic activity was frequent (up to 10 per 10 high-power fields), and atypical forms were observed. The neoplastic cells showed moderate cytoplasmic positivity for ALK (OTI1H7) and were negative for CD30, CD45, desmin, smooth muscle actin, DOG1, CD117, CD34, and S-100 protein. After reviewing those consultation slides, additional immunostainings were performed and results showed that the neoplastic cells were positive for ALK-V (D5F3) (cytoplasmic pattern) (Figure 2D), ALK (5A4), thyroid transcription factor 1 (TTF-1, 8G7G3) (Figure 2E), TTF-1(SPT2), epithelial membrane antigen (EMA), cytokeratin AE1/AE3, and cytokeratin seven (CK7) (Figure 2F). The tumor cells were negative for p63, cytokeratin 5/6, and Napsin-A. Fluorescence in situ hybridization analysis revealed ALK gene rearrangement in spindle, oval, and epithelial-like neoplastic cells (Figure 3A). Subsequently, next-generation sequencing involving 68 lung cancer-related genes identified an EML4 (exon 6):: ALK (exon 20) gene fusion(V3) (Figure 3B). No genetic abnormality of EGFR, KRAS, TPM3, or other genes was detected.
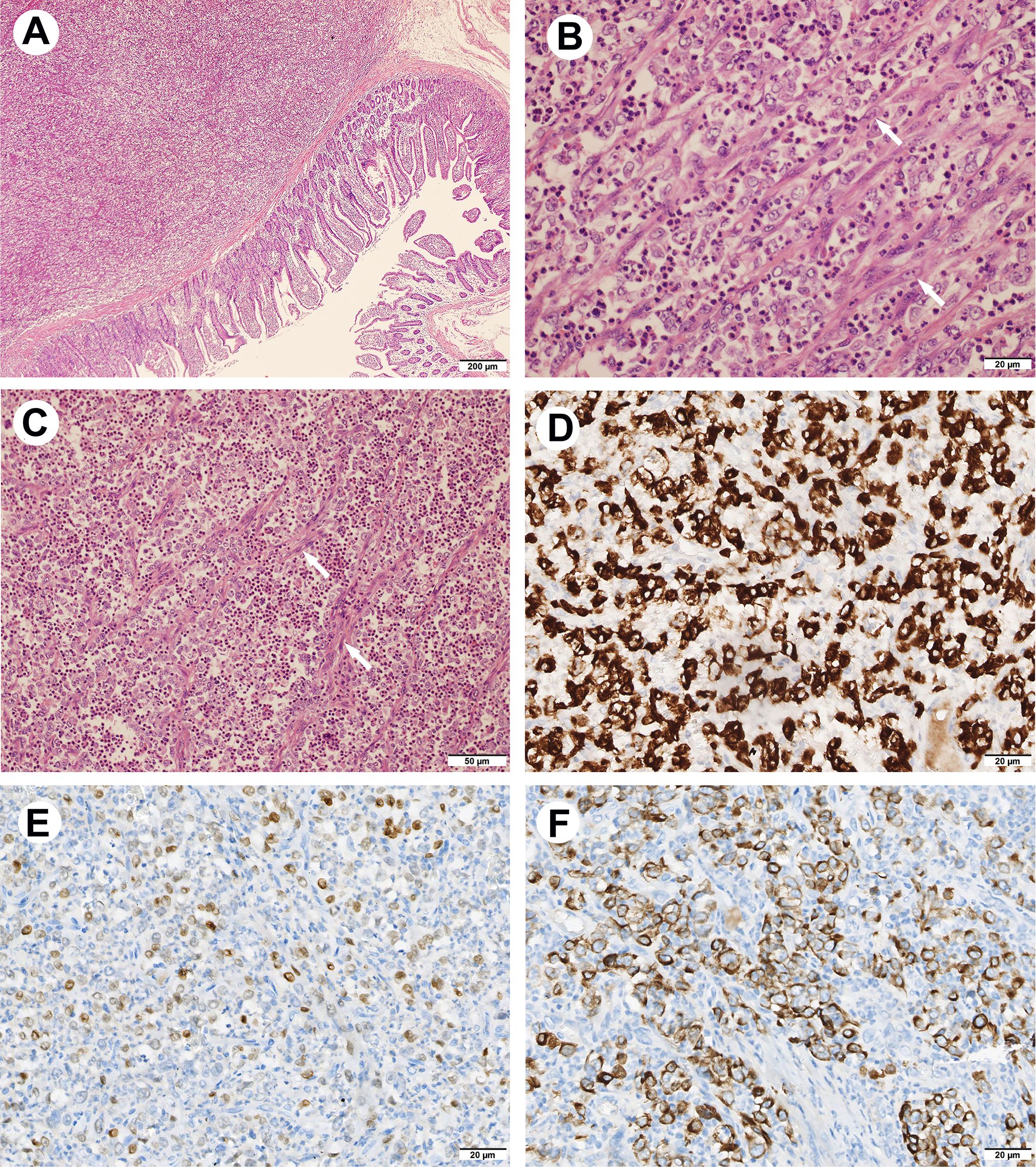
Figure 2. Hematoxylin & eosin and immunohistochemical findings of the tumor. (A) Low-power view showing the tumor located within the intestinal wall (HE, 40×). (B, C) The neoplastic components of fat spindle-shaped cells (arrow) and large epithelioid cells with ovoid nuclei interwoven with each other distributed in the background of large number of neutrophil cells ((HE, 200x&400×). (D) The neoplastic cells show cytoplasmic positivity for ALK (D5F3) (IHC, 400×). (E) The nuclear of neoplastic cells are positivity for TTF-1(8G7G3) (IHC, 400×). (F) The neoplastic cells show cytoplasmic reactivity for CK7 (IHC, 400×).
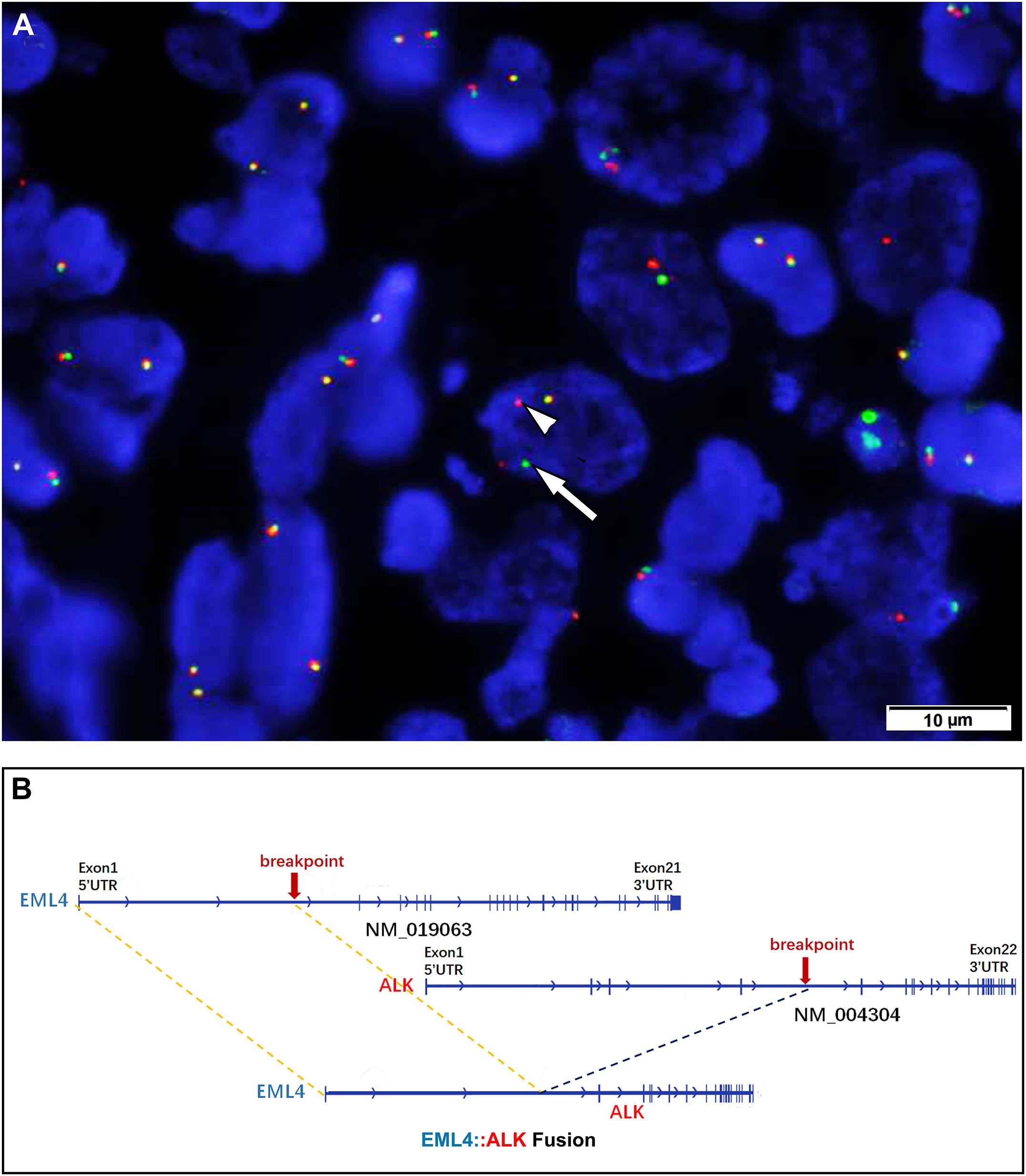
Figure 3. (A) Fluorescence in situ hybridization demonstrating a rearrangement of the ALK locus in the neoplastic cells (separation of the red [arrow] and green [arrowhead] signals). (B) Next-generation sequencing involving 68 lung cancer-related genes identified an EML4 (exion 6)::ALK (exon 20)(V3) gene fusion.
Given that the neoplastic cells were positive for CK7 and TTF1, this strongly suggests that the tumor likely originated from the pulmonary epithelium rather than the soft tissue of the intestine. Additionally, this case revealed the presence of an EML4 (exon 6):: ALK (exon 20) (V3) fusion, which was one of the most common genetic alterations observed in lung cancer. In contrast, EIMS typically harbored RANBP2::ALK or RRBP1::ALK fusions (10). Based on these findings, we considered this case to represent intestinal metastasis of an ALK-rearranged pulmonary adenocarcinoma. Therefore, we recommend further clinical investigations to confirm the diagnosis.
A subsequent enhanced CT scan of the chest revealed a 3.0 cm solid mass in the right upper lung, further confirming the diagnosis. Therefore, the oncologists recommended the ALK-tyrosine kinase inhibitors (TKI) therapy to the patient. Unfortunately, the patient refused to apply therapeutic approaches despite the doctor firmly suggesting them. Enhanced CT was performed seven months later; it identified a mass of 9.3cm×8.5cm invading the abdominal ln the lower abdomen, which is considered a metastasis or recurrence. The patient showed rapid disease progression and died in December 2019, 6 months after surgery.
Metastases to the intestine from lung cancer are rare. Historically, clinical incidence rates of intestinal metastasis of lung cancer have been reported to be no more than 1% (4). However, a review study showed that small bowel obstruction caused by lung carcinoma was up to 11.1% in all secondary tumors (5), implying that some cases may be overlooked in clinical diagnoses.
A SNOMED search of the West China Hospital surgical pathology files from January 2009 to May 2023 identified 73132 lung carcinomas, whereas only 24 cases (0.03%) of lung carcinomas metastasized to the intestine have been identified. Among the 24 lung carcinomas (including the present one), ADC (11/24, 45.8%) was the most common subtype, followed by SqCC (5/24,20.8%), PLC (4/24, 8.7%), small cell lung carcinoma (3/24, 12.5%) and large cell carcinoma (1/24, 4.2%). Four patients have performed ALK protein detection (one ADC, one SCC, and one PLC), and the present case is the only positive for ALK. In our hospital, the current case is the only ALK-positive PLC confirmed by FISH. Additionally, the data we summarized demonstrated that most cases of intestinal metastasis involved the small intestine (17/24, 70.8%), and only a minimal number of cases involved the large intestine (3/24, 12.5%), the ileocecal junction (2/24, 8.3%) and appendix (2/24, 8.3%). As far as we know, carcinoma originating from the small bowel is sporadic, which would remind clinicians to preferentially explore the primary sites for carcinomas in the small intestine.
A search of the English literature indicated that only 21 cases of ALK-rearranged pulmonary pleomorphic carcinomas had been reported so far (including the present one) (Supplementary Table) (2, 11–23). All patients with available age data (13 cases) are adults aged from 38 to 87 years old (median, 58 years old). Among those 14 cases with gender information, males comprised far more than females, with a ratio of 9 to 5. Of those patients (13 cases) with smoking information, 3 have a smoking history, while 10 denied it. The tumor size was 2.4 cm to 7 cm (median, 3cm). For the morphology, most cases (9/11, 81.8%) were composed of sarcomatoid spindle cell components and ADC in varying proportions, while a majority of cases (2/11, 18.2%) comprised giant cells and other components (spindle cells, SqCC or ADC). All those patients with ALK rearrangement with ALK immunostaining results (17 cases) showed positive for ALK (17/17,100%). EML4 is the only fusion partner of the ALK gene in those patients with identified fusion partners (8/8, 100%). Among those five patients with follow-up information, local or distant metastases occurred in four patients, and the present case is the only one that metastasized to the small intestine. There were two patients who died in 3 and 23 months after surgery, despite they adopt aggressive treatment. Two patients have survived for 11 months after admission. The present case died 6 months ago after refusing any treatments after diagnosis, considering his poor physical condition.
Small intestine metastasis in ALK-rearrangement lung cancer is infrequent. Four cases (including the present one) have been reported in the literature (7–9), and details was shown in Table 1. They shared some clinical and pathological features but also showed differences. In two of these cases, abdominal pain was the initial symptom, and they were admitted to the hospital with intestinal lesions as the primary concern. Intestinal metastasis presents with more pronounced symptoms than lung symptoms, which might be one of the reasons for misdiagnosis. Notably, all three cases with detailed genetic findings involved the EML4::ALK (V3) fusion type, which has been reported to be associated with poor prognosis of ALK-rearranged non-small cell lung cancer (24). All these four patients were already showed distant metastasis at the initial consultation and two of the passed away in 14 months after diagnosis; however, whether this is related to the V3 fusion type requires further investigation. Among the four pathological cases, three contained only ADC components. In contrast, the present case comprised epithelioid and spindle cell components, and it was the only case of pleomorphic carcinoma among these four cases. Currently, there is no definitive experimental evidence regarding the mechanisms of lung cancer metastasis. Still, previous studies suggest it might be related to the intestine’s abundant blood supply and lymphatic tissue (8, 9).
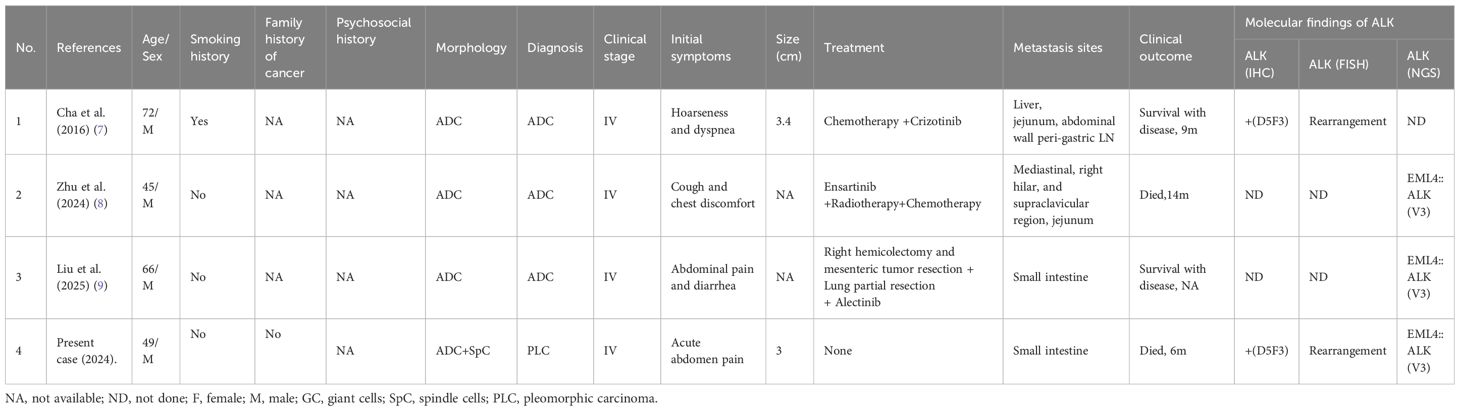
Table 1. Clinicopathologic and molecular features of ALK-rearrangement lung carcinomas with small intestinal metastases.
Compared with other reported ALK-rearranged PLC cases, the present case exhibited peculiar clinicopathological and molecular features. Epidemiologically, the initial clinical indications of intestinal metastasis of lung cancer and PLC are sporadic. Histologically, this tumor is rich in inflammatory cells and diffusely scattered in epithelioid and spindle cell areas. Molecularly, this tumor harbors ALK gene rearrangement. All of the above-mentioned characteristics make the diagnosis of the present case more challenging. For the present case, inflammatory spindle cell lesions of the cavum abdominals should be dominantly considered as differential diagnosis, where IMT and anaplastic large cell lymphoma (ALCL) would be much more likely to be identified as ALK-positive tumors, especially IMT (including its subtypes). IMTs are the much more common types of ALK-positive gastrointestinal neoplasm, far outnumbering metastatic NSCLCs. Besides, some classic IMTs can express focal positivity for cytokeratin, and a minority of tumors can harbor EML4::ALK fusion, making the diagnosis more confusing (6). In the present case, the morphology is more prone to be a high-grade tumor, and epithelioid inflammatory fibroblastic sarcoma (IMS), an aggressive subtype of IMTs, should be excluded. IMS has a marked predilection for the abdominal cavity and comprises large epithelioid cells. However, we should emphasize that most EIMSs show a nuclear membrane or a perinuclear accentuated cytoplasmic pattern of ALK staining and usually have RANBP2:: ALK or RRBP1:: ALK fusions (10). The current case showed a cytoplasmic pattern and harbored EML4::ALK fusion. ALCL is a common type of tumor that harbors ALK gene fusion; furthermore, about 10% of ALCLs present large round neoplastic cells admixed with a large number of reactive histiocytes (25), which is similar to the morphology of the present case. Although the gastrointestinal tract is not the common site for ALCL, it does occur in the gastrointestinal tract (26). However, the hallmark cells of anaplastic large cell lymphoma (ALCL) are absent in the present case. As reported, these cells usually have a variable proportion, with eccentric, horseshoe- or kidney-shaped nuclei and often an adjacent eosinophilic region (25). In addition, ALCL is generally positive for CD30. All the above-mentioned characteristics assist us in ruling out the possibility of ALCL. Notably, positive TTF1 staining combined with detailed clinical findings can be valuable in confirming the diagnosis.
The present case exhibited extremely peculiar clinicopathological features and can easily be misdiagnosed as primary intestinal tumors with ALK rearrangement. A comprehensive analysis from clinical, histological, immunohistochemical, and molecular aspects offers a full view of these case. It reminded that pathologists should be aware of the possibility of other ALK-positive neoplasms, including metastatic lesions, in the diagnosis-making process. However, being a single case study, its findings lacked generalizability and there was no experimental evidence on metastasis mechanisms, and more cases are needed to clarify it.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author/s.
The studies involving humans were approved by West China Hospital Institutional Review Board. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the participant/patient(s) for the publication of this case report.
CS: Formal Analysis, Resources, Writing – original draft. YQ: Writing – review & editing, Writing – original draft. KH: Resources, Writing – review & editing. YL: Data curation, Writing – review & editing. QW: Resources, Writing – review & editing. JY: Resources, Writing – review & editing. HZ: Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the National Natural Science Foundation of China (No. 81972520, 82203180); Natural Science Foundation Project of Science & Technology Department of Sichuan Province (No.2024NSFSC0763, 2024NSFSC7843) and the Cadre Health Research Project of Sichuan Province (No.2024-113).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2025.1496752/full#supplementary-material
1. WHO Classification of tumors Editorial Board Thoracic Tumors. WHO Classification of Tumor. Lyon: International Agency for Research on Cancer (2021).
2. Huang Y, Guo J, Li S, Liu J, Xu J, Ye W, et al. The correlation between histologic, immunophenotypic, and molecular characteristics of pulmonary sarcomatoid carcinoma reveals that sarcomatoid change is potentially derived from epithelial carcinoma cells undergoing epithelial-mesenchymal transition. Appl Immunohistochem Mol Morphol. (2023) 31:17–25. doi: 10.1097/PAI.0000000000001060
3. Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature. (2007) 448:561–6. doi: 10.1038/nature05945
4. Rossi G, Marchioni A, Romagnani E, Bertolini F, Longo L, Cavazza A, et al. Primary lung cancer presenting with gastrointestinal tract involvement: clinicopathologic and immunohistochemical features in a series of 18 consecutive cases. J Thorac Oncol. (2007) 2:115–20. doi: 10.1016/S1556-0864(15)30037-X
5. Idelevich E, Kashtan H, Mavor E, Brenner B. Small bowel obstruction caused by secondary tumors. Surg Oncol. (2006) 15:29–32. doi: 10.1016/j.suronc.2006.05.004
6. Coffin CM, Hornick JL, Fletcher CD. Inflammatory myofibroblastic tumor: comparison of clinicopathologic, histologic, and immunohistochemical features including ALK expression in atypical and aggressive cases. Am J Surg Pathol. (2007) 31:509–20. doi: 10.1097/01.pas.0000213393.57322.c7
7. Cha YJ, Cho BC, Kim HR, Lee HJ, Shim HS. A case of ALK-rearranged adenocarcinoma with small cell carcinoma-like transformation and resistance to crizotinib. J Thorac Oncol. (2016) 11:e55–8. doi: 10.1016/j.jtho.2015.12.097
8. Zhu L, Zhao Y, Zhang Y, Liu Z, Ma W, Guo Y, et al. Small intestinal metastasis in a lung adenocarcinoma patient with concurrent EML4-ALK V3 and TP53 mutations after distinct responses to tyrosine kinase inhibitors: A case report. Heliyon. (2024) 10:e38839. doi: 10.1016/j.heliyon.2024.e38839
9. Liu TH, Chen YZ, Xu ZQ, Dong M. Oncogenic ALK fusion in rare subtype of small intestine metastasis from occult lung cancer. Lung Cancer Manag. (2025) 14:2364582. doi: 10.1080/17581966.2024.2364582
10. Lee JC, Li CF, Huang HY, Zhu MJ, Mariño-Enríquez A, Lee CT, et al. ALK oncoproteins in atypical inflammatory myofibroblastic tumours: novel RRBP1-ALK fusions in epithelioid inflammatory myofibroblastic sarcoma. J Patho. (2017) 241:316–23. doi: 10.1002/path.4836
11. Yoshida A, Tsuta K, Nakamura H, Kohno T, Takahashi F, Asamura H, et al. Comprehensive histologic analysis of ALK-rearranged lung carcinomas. Am J Surg Pathol. (2011) 35:1226–34. doi: 10.1097/PAS.0b013e3182233e06
12. Lee HY, Ahn HK, Jeong JY, Kwon MJ, Han JH, Sun JM, et al. Favorable clinical outcomes of pemetrexed treatment in anaplastic lymphoma kinase positive non-small-cell lung cancer. Lung Cancer. (2013) 79:40–5. doi: 10.1016/j.lungcan.2012.10.002
13. Kodama T, Motoi N, Ninomiya H, Sakamoto H, Kitada K, Tsukaguchi T, et al. A novel mechanism of EML4-ALK rearrangement mediated by chromothripsis in a patient-derived cell line. J Thorac Oncol. (2014) 9:1638–46. doi: 10.1097/JTO.0000000000000311
14. Shiroyama T, Tanaka A, Tamiya M, Hamaguchi M, Osa A, Takeoka S, et al. A rare case of pleomorphic carcinoma of the lung harboring an anaplastic lymphoma kinase (ALK) rearrangement. Intern Med. (2015) 54:2741–3. doi: 10.2169/internalmedicine.54.4474
15. Murakami Y, Saka H, Oki M. Response to crizotinib and clinical outcome in ALK-rearranged pulmonary pleomorphic carcinoma. J Thorac Oncol. (2015) 10:e28–9. doi: 10.1097/JTO.0000000000000450
16. Maruyama R, Matsumura F, Shibata Y, Takahashi H, Okabayashi H, Kosai S, et al. Detection of ALK rearrangement in an octogenarian patient with pleomorphic carcinoma of the lung. Gen Thorac Cardiovasc Surg. (2016) 64:167–9. doi: 10.1007/s11748-014-0428-4
17. Jang JS, Bi L, Kipp BR, Jen J, Yi ES, Boland JM. Molecular characterization of pulmonary sarcomatoid carcinoma: analysis of 33 cases. Mod Pathol. (2016) 29:824–31. doi: 10.1038/modpathol.2016.89
18. Li X, Wang D, Zhao Q, Ren D, Ren F, Chen G, et al. Clinical significance and next-generation sequencing of Chinese pulmonary sarcomatoid carcinoma. Sci Rep. (2017) 7:3947. doi: 10.1038/s41598-017-04296-2
19. Chen X, Zhang Y, Lu J, Xu C, Liang J, Wang F, et al. Pulmonary sarcomatoid carcinoma with ALK rearrangement: frequency, clinical-pathologic characteristics, and response to ALK inhibitor. Transl Oncol. (2017) 10:115–20. doi: 10.1016/j.tranon.2016.11.009
20. Lin L, Huang F, Chen F, He Y, Hu J, Cao X. Anaplastic lymphoma kinase (ALK)-rearranged pulmonary pleomorphic carcinoma successfully treated with crizotinib. J Int Med Res. (2018) 46:3491–7. doi: 10.1177/0300060517748262
21. Alì G, Bruno R, Poma AM, Affinito O, Monticelli A, Piaggi P, et al. Whole transcriptome targeted gene quantification provides new insights on pulmonary sarcomatoid carcinomas. Sci Rep. (2019) 9:3536. doi: 10.1038/s41598-019-40016-8
22. Manabe S, Kasajima R, Murakami S, Miyagi Y, Yokose T, Kondo T, et al. Analysis of targeted somatic mutations in pleomorphic carcinoma of the lung using next-generation sequencing technique. Thorac Cancer. (2020) 11:2262–9. doi: 10.1111/1759-7714.13536
23. Hashimoto H, Komori K, Kameda K, Taguchi S, Ozeki Y. Successful salvage surgery followed by second ALK-TKI after alectinib failure in a patient with ALK-positive NSCLC. Surg Case Rep. (2022) 8:59. doi: 10.1186/s40792-022-01408-7
24. Noh KW, Lee MS, Lee SE, Song JY, Shin HT, Kim YJ, et al. Molecular breakdown: a comprehensive view of anaplastic lymphoma kinase (ALK)-rearranged non-small cell lung cancer. J Pathol. (2017) 243:307–19. doi: 10.1002/path.4950
25. Tsuyama N, Sakamoto K, Sakata S, Dobashi A, Takeuchi K. Anaplastic large cell lymphoma: pathology, genetics, and clinical aspects. J Clin Exp Hematop. (2017) 57:120–42. doi: 10.3960/jslrt.17023
Keywords: ALK, inflammatory myofibroblastic tumor, epithelioid inflammatory myofibroblastic sarcoma, lung carcinoma, intestinal metastasis
Citation: Shi C, Qiu Y, He K, Li Y, Wan Q, Yao J and Zhang H (2025) Case Report: Intestinal metastasis from ALK-rearranged pulmonary pleomorphic carcinomas mimicking inflammatory myofibroblastic tumors. Front. Oncol. 15:1496752. doi: 10.3389/fonc.2025.1496752
Received: 15 September 2024; Accepted: 04 March 2025;
Published: 28 March 2025.
Edited by:
Karolina Henryka Czarnecka-Chrebelska, Medical University of Lodz, PolandReviewed by:
Swarna Kanchan, Marshall University, United StatesCopyright © 2025 Shi, Qiu, He, Li, Wan, Yao and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hongying Zhang, aHlfemhhbmdAc2N1LmVkdS5jbg==
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.