
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Oncol. , 31 March 2025
Sec. Neuro-Oncology and Neurosurgical Oncology
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1493542
Objectives: Primary central nervous system lymphoma is an extremely aggressive type of non-Hodgkin lymphoma, and there is no consensus regarding the optimal management strategy for this disease. This study aimed to analyze the impact of consolidation therapy among young patients with intracranial primary diffuse large B-cell lymphoma (DLBCL).
Methods: This retrospective study analyzed the clinical data of 55 young patients (age < 60 years) with intracranial primary DLBCL who achieved complete remission (CR) after high-dose methotrexate (HD-MTX)-based chemotherapy from March 2001 to October 2021. Among these patients, 33 patients received consolidation therapy, and 22 patients did not. Overall survival (OS) and disease-free survival (DFS) were compared between the two groups via Kaplan–Meier analysis, the multivariate Cox proportional hazards method, and propensity score matching (PSM).
Results: The median follow-up time was 60.1 months. A total of 13 patients (23.6%) died, and 20 patients (36.4%) experienced recurrence. Patients who received consolidation therapy had higher 2-year OS (96.8% vs. 71.1%, P = 0.036) and DFS (90.9% vs. 56.4%, P = 0.006) rates than those without consolidation therapy. Multivariate analysis after PSM revealed that consolidation therapy was an independent predictor of DFS (HR = 0.282, 95% CI = 0.084–0.942, P = 0.040). Furthermore, rituximab was an independent predictor of favorable OS, and performance status was an independent predictor of OS and DFS. Subgroup analysis showed rituximab significantly improved OS in patients without consolidation therapy (88.9% vs. 45.0%, P = 0.006), but not in those with consolidation therapy (95.0% vs. 100%, P = 0.528).
Conclusions: Consolidation therapy improved DFS in young intracranial primary DLBCL patients achieving CR after HD-MTX-based chemotherapy. Autologous stem cell transplantation and radiotherapy showed comparable consolidation benefits. Good performance status correlated with favorable outcomes. Adding rituximab to induction chemotherapy may improve OS in patients without consolidation therapy, but it might be unnecessary for those eligible for consolidation. Further research involving a larger patient cohort is warranted to ascertain rituximab’s efficacy.
Primary central nervous system lymphoma (PCNSL) represents a highly aggressive subtype of non-Hodgkin lymphoma, predominantly manifesting as intracranial tumors, with a minority presenting as primarily intraocular or spinal cord lesions. PCNSL constitutes approximately 2% of all primary central nervous system tumors (1, 2). The most common type of PCNSL is diffuse large B-cell lymphoma (DLBCL), which has distinct biological properties and clinical behavior, resulting in a more aggressive nature than systemic DLBCL (3, 4). The introduction of high-dose methotrexate (HD-MTX) (dose ranging from 3.5 to 8.0 g/m²) as the standard systemic therapy has significantly enhanced treatment responses in PCNSL patients (5). However, a substantial subset of patients continues to exhibit resistance to upfront treatment, resulting in poor prognostic outcomes (6).
Following induction chemotherapy, the prevailing treatment strategies encompass consolidation therapy through whole-brain radiotherapy (WBRT) and high-dose chemotherapy in conjunction with autologous stem cell transplantation (ASCT) for patients who achieve complete remission (CR). In contrast, patients who do not attain CR are managed with salvage or palliative radiotherapy (7). Nonetheless, the effectiveness and safety of implementing consolidation therapy subsequent to HD-MTX induction therapy remain uncertain. Consequently, treatment regimens may vary considerably across institutions and regions.
This study aims to assess the impact of consolidation therapy on young patients with intracranial primary DLBCL by employing propensity score matching (PSM).
This retrospective study analyzed the clinical, treatment, and follow-up data of newly diagnosed PCNSL patients treated at the Sun Yat-sen University Cancer Center between March 2001 and October 2021. The patients were pathologically diagnosed in accordance with the fifth edition of the WHO Guidelines for the Classification of Tumors of the CNS (8).
The inclusion criteria were as follows: 1) age < 60 years, 2) primary lesion located in the brain, 3) pathologically confirmed DLBCL, and 4) CR was achieved after HD-MTX-based (dose ranging from 3.5 to 8.0 g/m2) chemotherapy according to the International PCNSL Collaborative Group 2005 criteria (9).
The exclusion criteria were as follows: 1) patients with solitary intraocular lymphoma, 2) patients with secondary PCNSL, 3) immunocompromised patients, 4) patients who underwent prior radiotherapy, 5) patients who were followed up for less than one month, 6) patients with other central nervous system diseases, and 7) patients with other malignant tumors.
Data from physical examinations, medical history, bone marrow biopsy, blood tests, and imaging were collected. The baseline characteristics included age, sex, lesion site, Eastern Cooperative Oncology Group performance status (ECOG PS), surgical approach, and immunohistochemistry.
The recurrence and survival data of patients were obtained via regular outpatient re-examination or telephone follow-up, and census and surveillance data collected by government agencies were used for patients who were lost to follow-up.
A database was established via SPSS 26.0 (IBM Corp., Armonk, NY, USA). Figures were drawn via GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA). Overall survival (OS) and disease-free survival (DFS) were the primary outcome measures and were examined via the Kaplan–Meier method with the log-rank test. The chi-square test was used to compare clinicopathological characteristics between the non-consolidation and consolidation therapy groups.
To balance the covariates between the non-consolidation and consolidation therapy groups, PSM analysis was conducted on confounding factors. Based on prior literature (10, 11), the inclusion criteria for the matching variables were as follows: 1) variables identified by univariate analysis as significantly associated (P < 0.10) with survival outcomes (ECOG PS, molecular subtype and targeted therapy); 2) variables that may potentially influence treatment selection and survival outcomes (sex, deep structure involvement and surgical procedure). A 1:1 ratio nearest neighbor match with a caliper value of 0.2 was used. A Cox proportional hazards model was used for multivariate analysis. All tests were two-tailed, and P < 0.05 was considered statistically significant.
A total of 55 individuals were included in this study. All patients were < 60 years old at diagnosis, and the median age was 48 years (range, 15–60 years). Histopathological diagnoses were made in 15 patients via stereotactic biopsy, 18 patients via open biopsy and 22 patients via surgical resection. Following histological confirmation, all patients received HD-MTX-based (dose ranging from 3.5 to 8.0 g/m2) regimens with a median of 6 cycles (ranging from 2 to 10 cycles). Rituximab was administered to 36 patients. Specifically, the treatment regimens comprised HD-MTX–temozolomide–rituximab for 29 patients, HD-MTX–rituximab for 4 patients, and HD-MTX–cytarabine–rituximab for 3 patients.
All patients achieved CR after chemotherapy. Among them, 12 patients who had sufficient autologous peripheral blood stem cell collection proceeded to undergo ASCT as previously described (3, 12). The conditioning regimens consisted of carmustine and thiotepa for 7 patients, thiotepa, busulfan, and cyclophosphamide for 3 patients, and etoposide and thiotepa for 2 patients. A total of 21 patients received consolidation WBRT as previously defined (2). The median total dose was 40.0 Gy (ranging from 23.4 to 56.0 Gy), and the median whole-brain dose was 30.0 Gy (ranging from 20.0 to 40.0 Gy). In addition, 22 patients were monitored via a watchful waiting strategy. Among patients receiving rituximab during induction therapy, 10/36 (27.8%) underwent ASCT, 12/36 (33.3%) received WBRT, and 14/36 (38.9%) were managed with watchful waiting. For those not receiving rituximab, 2/19 (10.5%) underwent ASCT, 9/19 (47.4%) received WBRT, and 8/19 (42.1%) were managed with watchful waiting. Table 1 summarizes the primary baseline characteristics of the participants.
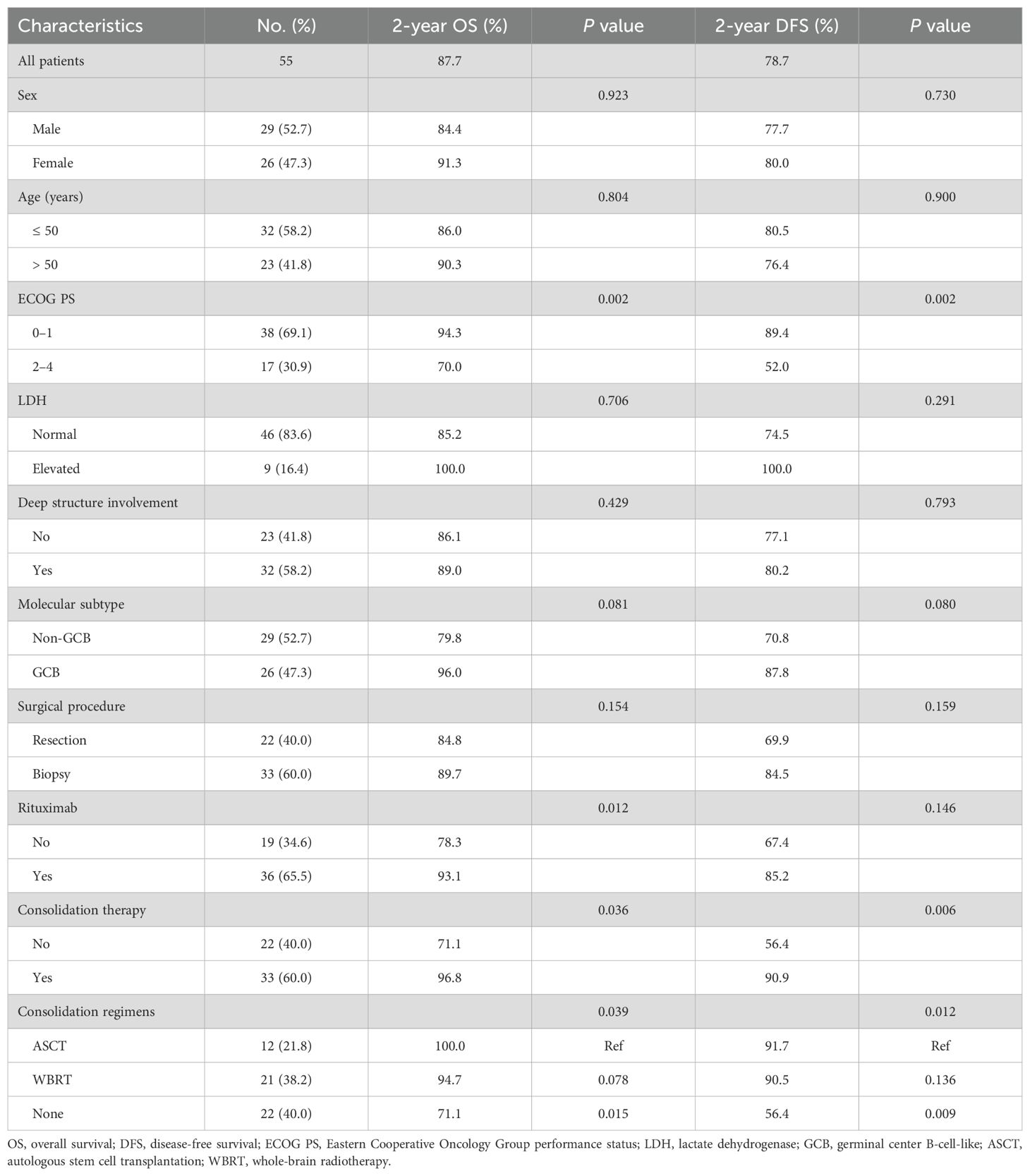
Table 1. Univariate survival analysis based on baseline clinicopathologic characteristics for all patients (n = 55).
The date of the last follow-up was January 31, 2023, resulting in a median patient follow-up of 60.1 months (ranging from 3.6 to 118.1 months). Overall, 13 patients (23.6%) died, and 20 patients (36.4%) experienced disease recurrence. In the non-consolidation group, 7 patients (31.8%) died, and 11 patients (50.0%) experienced recurrence, while in the consolidation group, 6 patients (18.2%) died, and 9 patients (27.3%) experienced recurrence.
As shown in Table 1; Figure 1, survival outcomes were significantly better in patients receiving consolidation therapy than in the non-consolidation group, with no significant difference between WBRT and ASCT. Additionally, an ECOG PS of 0–1 was a predictor of favorable OS and DFS, and rituximab was a predictor of favorable OS. In addition, to further validate the effect of rituximab, we performed a stratified survival analysis of rituximab based on whether patients received consolidation therapy. The results indicated that rituximab significantly improved OS in patients who did not receive consolidation therapy, whereas no significant differences in DFS and OS were observed among patients who received consolidation therapy (Table 2).
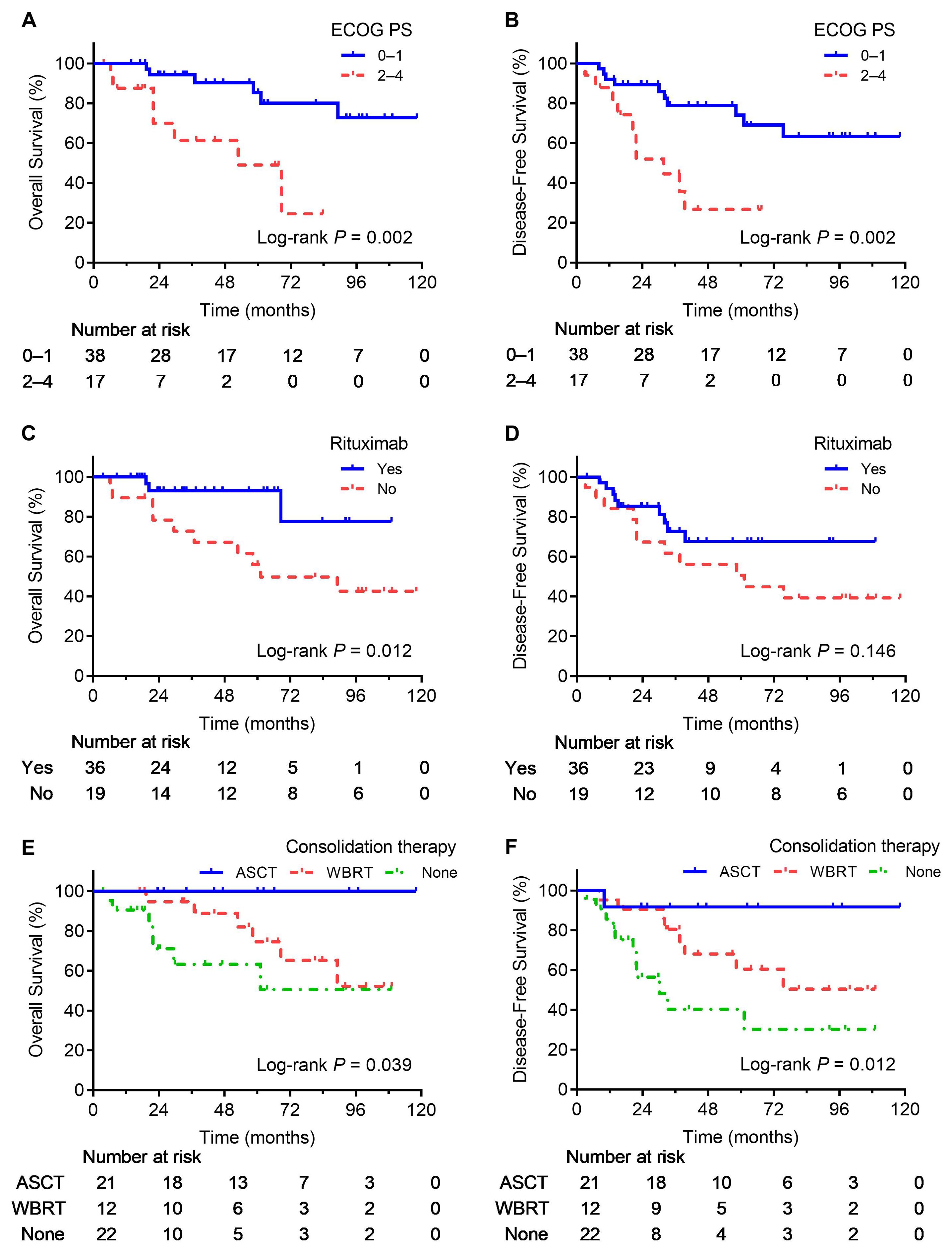
Figure 1. Survival curves for significant prognostic factors. OS (A) and DFS (B) curves for patients with different ECOG PS. OS (C) and DFS (D) curves for patients treated with or without rituximab. OS (E) and DFS (F) curves for patients treated with different consolidation regimens.
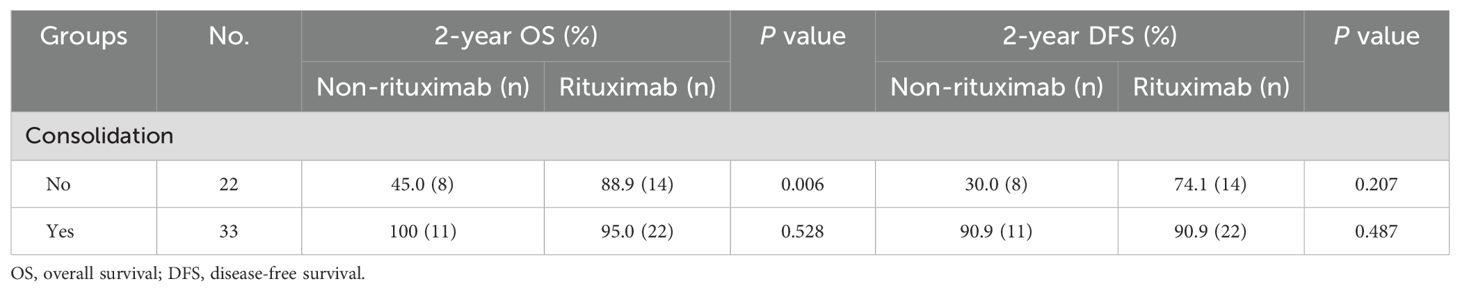
Table 2. Stratified survival analysis of rituximab by consolidation therapy for all patients (n = 55).
Before PSM, the proportion of patients who underwent biopsy was greater in the consolidation group. PSM analysis was performed in the non-consolidation and consolidation groups based on sex, ECOG PS, deep structure involvement, molecular subtype, surgical procedure, and targeted therapy. A total of 20 pairs of patients were included, and the patient characteristics of the two groups were balanced after PSM (Table 3).
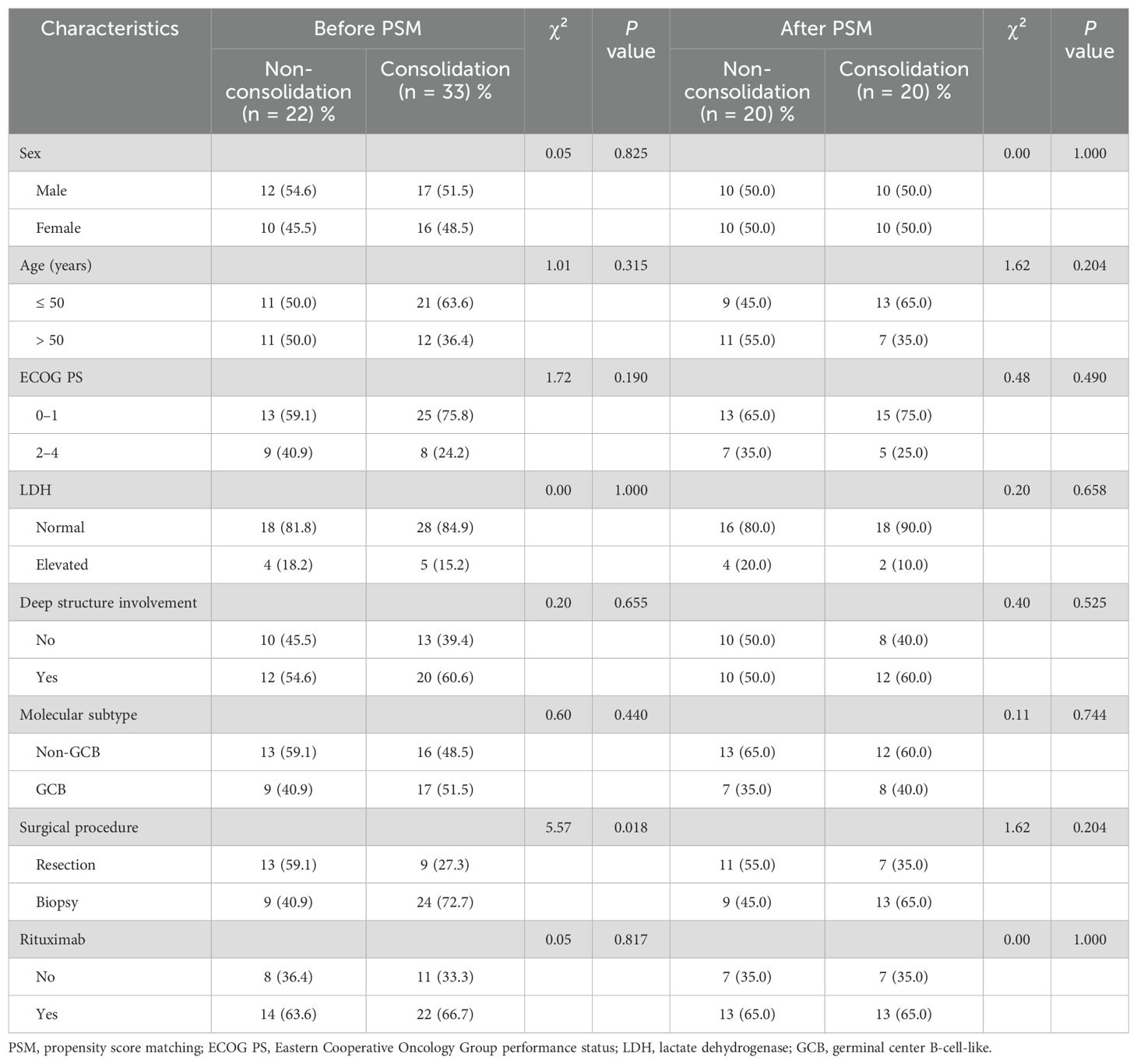
Table 3. Distribution of characteristics in the non-consolidation and consolidation groups before and after PSM.
Before PSM, the ECOG PS was an independent predictor for OS and DFS, and consolidation therapy was an independent predictor for DFS. After PSM, rituximab-based therapy was also found to be an independent predictor of OS in addition to the above findings (Figure 2).
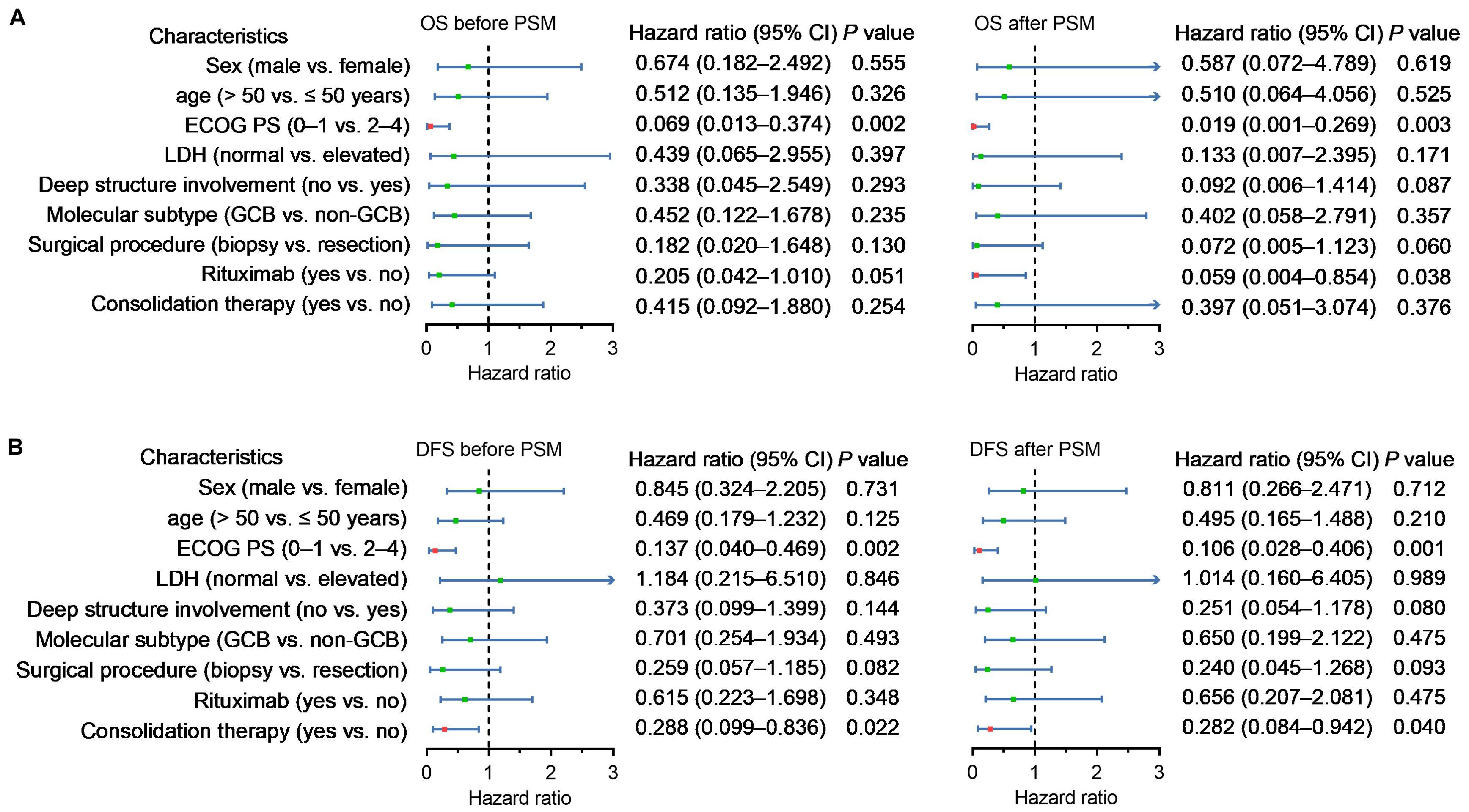
Figure 2. Forest plots representing multivariate analysis of risk factors associated with OS (A) and DFS (B) for all patients and matched patients. The bars represent the 95% confidence intervals of the hazard ratios.
Treatment-related neurotoxicity was defined as progressive neurological or cognitive impairment confirmed by serial clinical examinations in the absence of recurrent lymphoma. A total of 5 patients (23.8%) who received consolidation WBRT showed clinical evidence of treatment-related neurotoxicity, which included leukoencephalopathy in 2 patients, memory impairment in 2 patients, and cognitive disturbance in 1 patient. Among patients receiving ASCT as consolidation therapy, no patients developed treatment-related neurotoxicity.
HD-MTX-based chemotherapy is currently endorsed as the induction therapy for patients newly diagnosed with PCNSL (3, 7). A meta-analysis of clinical trials has demonstrated that HD-MTX-based regimens achieve an overall CR rate of 41% in this patient population (13). Consequently, we included patients undergoing HD-MTX-based induction chemotherapy in the present study.
While the addition of rituximab has been shown to enhance the prognosis of patients with systemic DLBCL, clinical investigations into its efficacy in PCNSL patients have produced inconsistent findings. For instance, a randomized phase III clinical trial (HOVON 105/ALLG NHL 24) indicated that the incorporation of rituximab into HD-MTX-based induction chemotherapy did not lead to improved disease outcomes (14, 15). In contrast, a phase II trial conducted by the International Extranodal Lymphoma Study Group-32 (IELSG 32) reported that the combination of HD-MTX–cytarabine with rituximab significantly increased the CR rate (30% vs. 23%), 7-year PFS rate (29% vs. 20%), and OS rate (37% vs. 21%) (5, 16). The divergence in outcomes between these two trials may be attributed to variations in protocol design and treatment intensity. A meta-analysis of two randomized trials indicated that the combination of rituximab with HD-MTX-based induction chemotherapy prolonged PFS, although its effects on OS remain uncertain (17). Another meta-analysis examining the impact of rituximab in patients with PCNSL found that rituximab use was significantly associated with higher CR rates and improved OS and PFS at both 3 and 5 years. This suggests that rituximab may positively influence the prognosis of PCNSL patients (18). Our findings revealed that, while DFS did not exhibit a statistically significant enhancement, patients undergoing rituximab combination therapy experienced an improvement in OS. This observation may be attributed to the proposed interaction between WBRT and rituximab, wherein WBRT-induced disruption of the blood-brain barrier potentially facilitates increased brain penetration of residual rituximab, thereby contributing to the observed OS improvement (15). Additionally, the effect of consolidation therapy on DFS may have also impacted the DFS survival analysis in the context of rituximab treatment. Notably, subgroup analysis stratified by consolidation therapy revealed that the OS benefit of rituximab was noticed in patients without consolidation therapy (88.9% vs. 45.0%, P = 0.006), but not in those with consolidation therapy (95.0% vs. 100%, P = 0.528). This finding, along with the PSM-adjusted multivariate analysis, suggested that rituximab contributes to OS improvement independently of consolidation therapy, which implied that the inclusion of rituximab in induction regimens may not provide additional survival benefits for patients undergoing consolidation therapy. Therefore, rituximab may be considered unnecessary in the induction therapy for young PCNSL patients eligible for consolidation strategies. Considering the limited sample size of the present study, further validation in a larger patient cohort is warranted to ascertain the efficacy of rituximab in young patients with PCNSL. A post-hoc analysis of the Phase III HOVON 105/ALLG NHL 24 trial demonstrated that rituximab significantly enhanced event-free survival in patients aged 60 years or younger who received WBRT. Nevertheless, the authors urge caution in interpreting these findings and recommend a randomized Phase III study specifically targeting OS in patients under 60 with PCNSL to validate rituximab’s efficacy (15). Our results provide retrospective support for this recommendation.
As a consolidation to HD-MTX chemotherapy, WBRT has been shown to extend survival in patients with PCNSL (19, 20). However, the effectiveness of consolidation WBRT remains contentious. A Phase III randomized trial conducted by the German PCNSL Study Group indicated that the addition of WBRT to the HD-MTX regimen prolonged PFS but did not enhance OS and increased neurotoxicity compared to HD-MTX-based monotherapy (21). Gavrilovic et al. reported that WBRT following the HD-MTX regimen significantly extended OS in patients under 60 years, with 74% surviving until the final follow-up. They recommended deferring WBRT in patients over 60 to mitigate treatment-related neurotoxicity, as the median OS was 29 months, irrespective of WBRT administration (22). Furthermore, existing literature suggests that consolidation WBRT does not enhance survival rates in patients who achieve CR following HD-MTX-based induction chemotherapy, indicating that the omission of WBRT could be considered for these patients (23). Contrarily, research by Omuro et al. indicates that for PCNSL patients under 60 years of age who achieve CR after HD-MTX induction chemotherapy, deferring WBRT may not be optimal due to suboptimal PFS (24). In our study, multivariate analysis following PSM demonstrated a significant improvement in DFS but not OS in patients who achieved CR after HD-MTX induction chemotherapy and received consolidation therapy, including WBRT and ASCT, compared to those who did not receive consolidation therapy.
The primary concern associated with WBRT in patients with PCNSL is the risk of delayed neurotoxicity, which undermines the long-term survival advantages, particularly among elderly individuals (20, 23). ASCT has emerged as an effective and promising alternative consolidation strategy to WBRT following HD-MTX-based induction therapy. Evidence from two randomized phase II trials, IELSG 32 and PRECIS, indicates that ASCT as a consolidation therapy is noninferior to WBRT in terms of PFS and OS for patients newly diagnosed with PCNSL (16, 25). Furthermore, both studies revealed that ASCT is significantly associated with the preservation of neurocognitive function. However, it is important to note that treatment-related mortality and hematologic toxicity rates were significantly higher with ASCT compared to WBRT (16, 25). A meta-analysis of PCNSL consolidation therapies found no significant differences between ASCT and WBRT in terms of OS and PFS. Nonetheless, concerning neurocognitive function, patients receiving WBRT exhibited a significant decline in attention and executive function (26). Our study produced comparable results, indicating no significant difference in survival outcomes between patients undergoing WBRT and those undergoing ASCT following CR with HD-MTX-based chemotherapy. Additionally, treatment-related neurotoxicity was observed in patients who received WBRT, whereas those who underwent ASCT did not experience significant neurocognitive decline. This finding suggests that ASCT may serve as a viable alternative to reduce WBRT-associated neurotoxicity in younger patients who prioritize long-term cognitive preservation. Therefore, when selecting a consolidation therapy regimen, it is crucial to thoroughly consider patients’ age, performance status, and the potential impact on their quality of life (26). In this context, ASCT may be more suitable for younger patients who respond favorably to induction chemotherapy (12).
Among the predictors of PCNSL, the ECOG PS has been identified as a key baseline predictor (27). In this study, patients with a favorable ECOG PS demonstrated significantly better survival outcomes.
This study is subject to several potential limitations. As a retrospective analysis, our assessment of the neurotoxicity profile was constrained to clinical records, rather than formal neuropsychological testing. Additionally, the findings may be affected by selection bias and variability in management practices. Nevertheless, the study’s focus on data from patients treated at a single hospital allowed for the standardization and consistency of diagnostic methods, treatment protocols, and follow-up procedures. Moreover, we utilized PSM to mitigate the influence of confounding variables, thereby enhancing the robustness of the evidence supporting the efficacy of consolidation therapy.
This study concentrated on patients under 60 years of age with intracranial primary DLBCL who attained CR following HD-MTX induction chemotherapy. Our results indicate that consolidation therapy significantly enhances DFS in this cohort, thereby informing clinical decision-making regarding consolidation strategies. Furthermore, our research demonstrated comparable efficacy between WBRT and ASCT as consolidation therapies concerning survival outcomes. This finding offers clinicians alternative treatment options, particularly considering the potential neurotoxic effects associated with WBRT. ASCT may be recommended for younger patients who prioritize the preservation of long-term cognitive function. Lastly, the observed potential benefit of rituximab in improving OS further supports its inclusion in induction chemotherapy regimens.
In young patients diagnosed with intracranial primary DLBCL who achieved CR following HD-MTX-based chemotherapy, those who underwent consolidation therapy exhibited superior DFS outcomes. The efficacy of consolidation therapy was comparable between ASCT and WBRT. Patients exhibiting a favorable ECOG PS also experienced improved prognoses. Additionally, the incorporation of rituximab into the HD-MTX-based chemotherapy regimen was associated with improved OS in patients who did not receive consolidation therapy. Conversely, rituximab may be considered unnecessary for induction therapy in patients eligible for consolidation strategies. Further investigation is warranted to ascertain the efficacy of rituximab within a larger patient cohort.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by Research Ethics Committee of the Sun Yat-sen University Cancer Center. The studies were conducted in accordance with the local legislation and institutional requirements. The ethics committee/institutional review board waived the requirement of written informed consent for participation from the participants or the participants’ legal guardians/next of kin because Informed consent was waived due to the retrospective design of the study.
GW: Data curation, Formal analysis, Investigation, Methodology, Writing – original draft. MX: Data curation, Formal analysis, Investigation, Writing – original draft. HW: Data curation, Investigation, Writing – original draft. JW: Project administration, Resources, Writing – original draft. HS: Investigation, Validation, Writing – original draft. JZ: Project administration, Resources, Supervision, Writing – review & editing. YZ: Conceptualization, Supervision, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Ostrom QT, Price M, Neff C, Cioffi G, Waite KA, Kruchko C, et al. CBTRUS statistical report: primary brain and other central nervous system tumors diagnosed in the United States in 2015-2019. Neuro Oncol. (2022) 24:v1–v95. doi: 10.1093/neuonc/noac202
2. National Health Commission of the People’s Republic of China. National guidelines for diagnosis and treatment of Malignant lymphoma 2022 in China. Chin J Cancer Res. (2022) 34:425–46. doi: 10.21147/j.issn.1000-9604.2022.05.01
3. Ferreri AJM, Illerhaus G, Doorduijn JK, Auer DP, Bromberg JEC, Calimeri T, et al. Primary central nervous system lymphomas: EHA–ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. HemaSphere. (2024) 8:e89. doi: 10.1002/hem3.89
4. Lauw MIS, Lucas CG, Ohgami RS, Wen KW. Primary central nervous system lymphomas: a diagnostic overview of key histomorphologic, immunophenotypic, and genetic features. Diagnostics (Basel). (2020) 10:22. doi: 10.3390/diagnostics10121076
5. Ferreri AJ, Cwynarski K, Pulczynski E, Ponzoni M, Deckert M, Politi LS, et al. Chemoimmunotherapy with methotrexate, cytarabine, thiotepa, and rituximab (MATRix regimen) in patients with primary CNS lymphoma: results of the first randomisation of the International Extranodal Lymphoma Study Group-32 (IELSG32) phase 2 trial. Lancet Haematol. (2016) 3:e217–e27. doi: 10.1016/S2352-3026(16)00036-3
6. Houillier C, Soussain C, Ghesquieres H, Soubeyran P, Chinot O, Taillandier L, et al. Management and outcome of primary CNS lymphoma in the modern era: An LOC network study. Neurology. (2020) 94:e1027–e39. doi: 10.1212/WNL.0000000000008900
7. Hoang-Xuan K, Deckert M, Ferreri AJM, Furtner J, Gallego-Perez-Larraya J, Henriksson R, et al. European Association of Neuro-Oncology (EANO) guidelines for treatment of primary central nervous system lymphoma (PCNSL). Neuro Oncol. (2023) 25:37–53. doi: 10.1093/neuonc/noac196
8. Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, et al. The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol. (2021) 23:1231–51. doi: 10.1093/neuonc/noab106
9. Abrey LE, Batchelor TT, Ferreri AJ, Gospodarowicz M, Pulczynski EJ, Zucca E, et al. Report of an international workshop to standardize baseline evaluation and response criteria for primary CNS lymphoma. J Clin Oncol. (2005) 23:5034–43. doi: 10.1200/JCO.2005.13.524
10. Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. (2011) 46:399–424. doi: 10.1080/00273171.2011.568786
11. Brookhart MA, Schneeweiss S, Rothman KJ, Glynn RJ, Avorn J, Stürmer T. Variable selection for propensity score models. Am J Epidemiol. (2006) 163:1149–56. doi: 10.1093/aje/kwj149
12. Gritsch D, Mrugala MM, Marks LA, Mangipudi K, Neal M, Wingerchuk DM, et al. Is autologous stem cell transplantation a safe and effective alternative to whole brain radiation as consolidation therapy in patients with primary central nervous system lymphoma?: a critically appraised topic. Neurologist. (2021) 26:137–42. doi: 10.1097/NRL.0000000000000328
13. Yu J, Du H, Ye X, Zhang L, Xiao H. High-dose methotrexate-based regimens and post-remission consolidation for treatment of newly diagnosed primary CNS lymphoma: meta-analysis of clinical trials. Sci Rep. (2021) 11:2125. doi: 10.1038/s41598-020-80724-0
14. Bromberg JEC, Issa S, van der Holt B, van der Meulen M, Dirven L, Minnema MC, et al. Survival, neurocognitive function, and health-related quality of life outcomes after rituximab-methotrexate, BCNU, teniposide, and prednisolone for primary CNS lymphoma: Final results of the HOVON 105/ALLG NHL 24 study. Neuro Oncol. (2024) 26:724–34. doi: 10.1093/neuonc/noad224
15. Bromberg JEC, Issa S, Bakunina K, Minnema MC, Seute T, Durian M, et al. Rituximab in patients with primary CNS lymphoma (HOVON 105/ALLG NHL 24): a randomised, open-label, phase 3 intergroup study. Lancet Oncol. (2019) 20:216–28. doi: 10.1016/S1470-2045(18)30747-2
16. Ferreri AJM, Cwynarski K, Pulczynski E, Fox CP, Schorb E, Celico C, et al. Long-term efficacy, safety and neurotolerability of MATRix regimen followed by autologous transplant in primary CNS lymphoma: 7-year results of the IELSG32 randomized trial. Leukemia. (2022) 36:1870–8. doi: 10.1038/s41375-022-01582-5
17. Schmitt AM, Herbrand AK, Fox CP, Bakunina K, Bromberg JEC, Cwynarski K, et al. Rituximab in primary central nervous system lymphoma-A systematic review and meta-analysis. Hematol Oncol. (2019) 37:548–57. doi: 10.1002/hon.2666
18. Zhang YH, Liu ZH, Gao CW, Bian HY, Ma YS, Jing FJ, et al. Role of rituximab in treatment of patients with primary central nervous system lymphoma (PCNSL): A systematic review and meta. Clin Lymphoma Myeloma Leukemia. (2023) 23:733–41. doi: 10.1016/j.clml.2023.06.008
19. Shibamoto Y, Sumi M, Takemoto M, Tsuchida E, Onodera S, Matsushita H, et al. Analysis of radiotherapy in 1054 patients with primary central nervous system lymphoma treated from 1985 to 2009. Clin Oncol (R Coll Radiol). (2014) 26:653–60. doi: 10.1016/j.clon.2014.06.011
20. O’Brien PC, Roos DE, Pratt G, Liew KH, Barton MB, Poulsen MG, et al. Combined-modality therapy for primary central nervous system lymphoma: long-term data from a Phase II multicenter study (Trans-Tasman Radiation Oncology Group). Int J Radiat Oncol Biol Phys. (2006) 64:408–13. doi: 10.1016/j.ijrobp.2005.07.958
21. Korfel A, Thiel E, Martus P, Mohle R, Griesinger F, Rauch M, et al. Randomized phase III study of whole-brain radiotherapy for primary CNS lymphoma. Neurology. (2015) 84:1242–8. doi: 10.1212/WNL.0000000000001395
22. Gavrilovic IT, Hormigo A, Yahalom J, DeAngelis LM, Abrey LE. Long-term follow-up of high-dose methotrexate-based therapy with and without whole brain irradiation for newly diagnosed primary CNS lymphoma. J Clin Oncol. (2006) 24:4570–4. doi: 10.1200/JCO.2006.06.6910
23. Ekenel M, Iwamoto FM, Ben-Porat LS, Panageas KS, Yahalom J, DeAngelis LM, et al. Primary central nervous system lymphoma: the role of consolidation treatment after a complete response to high-dose methotrexate-based chemotherapy. Cancer. (2008) 113:1025–31. doi: 10.1002/cncr.23670
24. Omuro A, Taillandier L, Chinot O, Sierra Del Rio M, Carnin C, Barrie M, et al. Primary CNS lymphoma in patients younger than 60: can whole-brain radiotherapy be deferred? J Neurooncol. (2011) 104:323–30. doi: 10.1007/s11060-010-0497-x
25. Houillier C, Dureau S, Taillandier L, Houot R, Chinot O, Molucon-Chabrot C, et al. Radiotherapy or autologous stem-cell transplantation for primary CNS lymphoma in patients age 60 years and younger: long-term results of the randomized phase II PRECIS study. J Clin Oncol. (2022) 40:3692–8. doi: 10.1200/JCO.22.00491
26. Epperla N, Reljic T, Chowdhury SM, Ferreri AJM, Kumar A, Hamadani M. Autologous hematopoietic cell transplantation versus whole-brain radiotherapy consolidation in primary central nervous system lymphoma: A systematic review and meta-analysis. Hematological Oncol. (2023) 41:88–96. doi: 10.1002/hon.3083
Keywords: diffuse large B-cell lymphoma, central nervous system neoplasm, radiotherapy, methotrexate, stem cell transplantation
Citation: Wen G, Xiang M, Wang H, Wang J, Shao H, Zhang J and Zhang Y (2025) Consolidation therapy impact on survival outcomes in young patients with intracranial primary diffuse large B-cell lymphoma achieving complete remission: a propensity score matching analysis. Front. Oncol. 15:1493542. doi: 10.3389/fonc.2025.1493542
Received: 09 September 2024; Accepted: 17 March 2025;
Published: 31 March 2025.
Edited by:
Xiao-Tang Kong, University of California, Irvine, United StatesReviewed by:
Jun-Ping Zhang, Capital Medical University, ChinaCopyright © 2025 Wen, Xiang, Wang, Wang, Shao, Zhang and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yujing Zhang, eXVqaW5nX3poYW5nMTk2OUAxNjMuY29t; Jinshan Zhang, emhhbmdqc2dkMTM3QDE2My5jb20=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.