
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Oncol. , 20 March 2025
Sec. Neuro-Oncology and Neurosurgical Oncology
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1477978
This article is part of the Research Topic Advances in Brain Tumor Therapy View all 11 articles
H3 K27-Altered Diffuse Midline Gliomas are commonly found in children and adolescents in midline locations such as the thalamus, brain stem, and spinal cord. It is rare for these tumors to affect adults and to occur in locations like the lateral ventricles. Despite aggressive treatment methodologies, there is no cure for this disease. The median survival is between 8-12 months. A 24-year-old white male presented to the emergency department due to severe headache refractory to pain medications with a 2-month history of progressive headaches and eventual memory problems. Computed tomography (CT) and magnetic resonance imaging (MRI) showed an intraventricular enhancing mass and hydrocephalus. The final diagnosis was an intraventricular H3 K27-Altered Diffuse Midline Glioma. The patient underwent two craniotomies, one laser interstitial thermal ablation (LITT), chemoradiotherapy, and bevacizumab and ONC206, through compassionate use. Despite a reduction in the tumor size, it continued to spread to other brain areas, leading to further complications and, eventually, his death, 10 months after initial diagnosis. From review of the literature, 21 cases were identified, and the median age was 24. Their median survival is 10.5 months (ranges 1 - 24 months). This case report presents the clinical, radiological, pathological, and molecular characteristics of a 24-year-old white man diagnosed with a ventricular H3 K27-Altered diffuse midline glioma, highlighting the rare presentation, management, and outcomes.
Diffuse midline gliomas (DMGs) characterized by the histone H3 K27M mutation are rare and aggressive high-grade tumors predominantly affecting children ages 5-10 years (1). The midline location defines this tumor type, diffuse growth pattern/infiltrating, and lysine-to-methionine substitution at position 27 on the H3 histone genes (2). This tumor type was first recognized in the 2016 World Health Organization (WHO) classification of central nervous system (CNS) tumors as DMG H3 K27M mutant. In 2021, WHO CNS tumor terminology changed to DMG H3 K27-altered to include subtypes of DMG with alternative mechanisms for the loss of H3K27 methylation, such as EGFR mutant DMG or EZH inhibitory protein overexpression DMG. This classification is categorized as “pediatric-type diffuse midline glioma” and is subdivided into 4 subtypes (DMG H3 K27-altered; diffuse hemispheric glioma, H3 G34-mutant; diffuse pediatric-type high-grade glioma H3-wildtype and IDH-wildtype; and infant-type hemispheric glioma), each of which possess characteristic molecular profiles (3). DMGs commonly arise in the thalamus, brainstem, and spinal cord—regions critical for vital functions—making these tumors particularly challenging to treat. These tumors are typically diagnosed in children and are associated with a very poor prognosis. The 5-year survival rate for patients with DMGs is less than 1%, and the median overall survival ranges from eight to twelve months (4).
Ventricular tumors are also rare and represent 0.8-1.6% of intracranial tumors, but tend to be benign, such as central neurocytomas, choroid plexus papillomas or carcinomas, astrocytomas, meningiomas, ependymomas, colloid cysts, or craniopharyngiomas. This case report describes the clinical, radiological, pathological, and molecular characteristics of a 24-year-old white male diagnosed with a ventricular H3 K27-altered diffuse midline glioma, highlighting the challenges and complexities of management.
A 24-year-old white male firefighter with a history of asthma and attention deficit hyperactivity disorder (ADHD) presented with a 2-month history of nonspecific memory problems. He was described by family members as exhibiting forgetfulness of events and tasks. Developed progressive headaches that were alleviated by lying down and taking NASID medications with limited benefit. These headaches were subsequently accompanied by photophobia, phonophobia, nausea, and vomiting.
He presented to the emergency department due to a severe prolonged headache refractory to usual treatment, which led to a CT scan showing an intraventricular mass. A magnetic resonance image (MRI) of the brain with and without contrast revealed an irregular 6.4x7x4.5 cm (AP x Lat x CC) heterogeneous mass in the lateral ventricles appearing to be coming from the pineal region. This image did not identify involvement of midline structures like the thalamus. It occupied both lateral ventricles causing an 8 mm right midline shift (Figures 1A–D). The mass exhibited aggressive features, including restricted diffusion, necrosis, and heterogeneous contrast enhancement (Figures 1B, C) (5).
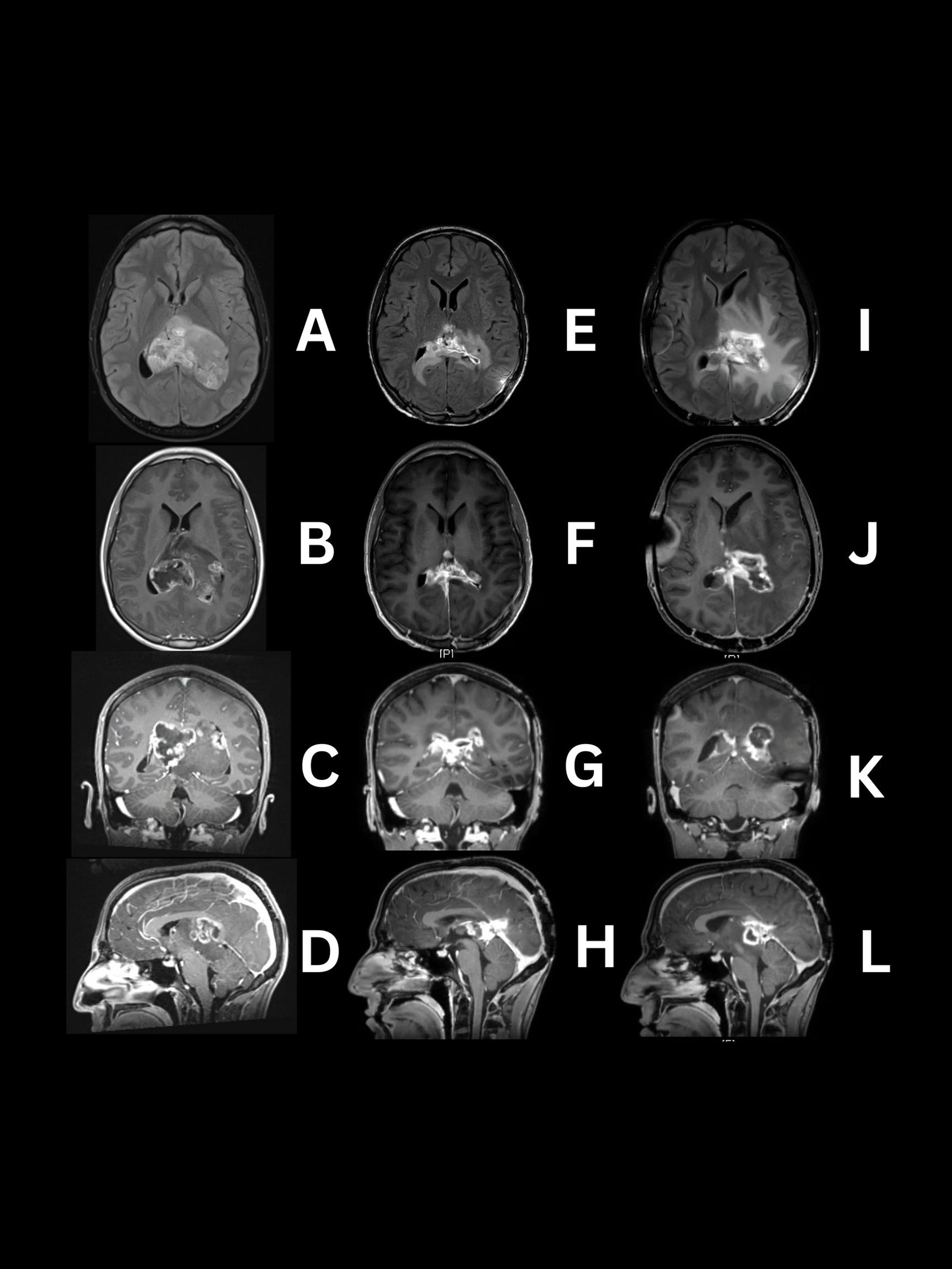
Figure 1. MRI images demonstrating serial progression of H3 K27-altered diffuse midline glioma. Preoperative (A–D) tumor located in lateral ventricles predominantly solid with patchy areas showing high intensity and central necrosis on the right side on T2/FLAIR and T1W post contrast. Day 104 post operation and chemoradiotherapy (E–H) reduced intraventricular tumor with periventricular FLAIR signal abnormality and contrast enhancement extending to the surrounding structures in T1W post-contrast images. Day 199 post-final treatment, (I–L) Progression of FLAIR signal (which is most likely a mixture of tumor infiltration with edema and radiation change) extending to basal ganglia and parietal lobes, with enhancement and necrosis of the intraventricular tumor and adjacent structures.
The following day, the patient underwent a left parietal craniotomy with a transparietal approach for mass resection and ventriculoperitoneal shunt (VPS) placement. The tumor location, firm and rubbery consistency, and similarity to adjacent brain tissue necessitated an initial partial resection. A postoperative MRI four days later showed expected surgical changes with a mass reduction to approximately 4.4x7x3.7 cm (AP x Lat x CC). Five days after the initial surgery, he underwent a second craniotomy with an interhemispheric approach focusing on the right lateral ventricle for further mass resection. MRI performed one day after the second surgery showed expected surgical changes and residual left intraventricular tumor of approximately 1.8x1.8x0.7cm with post-surgical periventricular enhancement of the thalamus.
The pathology report diagnosed a DMG H3 K27-Altered CNS WHO grade 4 with positive immunohistochemistry for H3K27M mutant nuclear expression, loss of H3K27me3 nuclear expression, (Figures 2A–C), weak to moderate nuclear expression of p53, strong expression of EGFR, and a Ki67 labeling index of 30-40%. The tumor was negative for IDH1 mutant protein expression and loss of ATRX expression. The Next Generation Sequencing (NGS) showed gene H3F3A K28M mutation, PTEN G132V – subclonal, RAD51B loss on exon 8, TSC2 E1344del, ATRX splice site 6849 + 2T>C.

Figure 2. H&E and immunohistochemistry results. (A) Hematoxylin and Eosin (H&E) staining showed monomorphic to pleomorphic glial cells with high mitotic activity. (B) H3 K27M showed positive nuclear staining. (C) H3K27me3 showed loss of nuclear stain. The final diagnosis was DMG, H3 K27-altered, CNS WHO grade-4. Images were captured with the Leica Thunder imaging system (Danaher, Washington, DC).
Following discharge, the patient continued to experience memory difficulties and newly developed right side homonymous hemianopsia. On day 30, he began chemoradiotherapy, receiving fractionated external beam radiotherapy (54-60 Gy in 30 fractions) with concurrent temozolomide at 75 mg/m2 x 42 days. Throughout therapy, he experienced ongoing attention and memory difficulties, further visual field reduction at his right side, and seizures due to missed doses of anti-seizure medication. He completed chemoradiotherapy on Day 69.
On day 105, MRI evaluation revealed new enhancement within the splenium, left thalamus, and 3rd ventricle with dimensions of 2.6x3.2x2.2 cm, raising questions about regrowth versus radiation changes (Figures 1E–H). The treatment approach included laser interstitial thermal ablation (LITT) through the parietal lobes with intraoperative fluoroscopy for precise targeting and ventriculoperitoneal shunt (VPS) placement. The patient was subsequently treated under a compassionate use regimen with ONC206, a more potent analogue of ONC201, a selective dopamine receptor D2 (DRD2) antagonist and mitochondrial protease ClpP agonist, at 120mg once per week, oral, for approximately 3 months.
On Day 189, his VPS malfunctioned due to blockage causing gait instability and right hemiparesis, requiring the placement of new bilateral ventriculoperitoneal shunts. He began bevacizumab therapy at a dose of 10 mg/kg IV, (without 600mg) IV, every 14 days as a salvage therapy, experiencing side effects, such as nausea, vomiting, and asthenia. The last MRI, Day 199 showed a progression in FLAIR signal in basal ganglia and parietal lobes, most likely due to tumor infiltration, radiation changes with edema or likely a mixture all three components (Figures 1I–L).
On Day 314, the patient’s condition deteriorated, leading to hospitalization. Despite intensive treatment efforts, he ultimately succumbed to his illness on the same day (Figure 3).
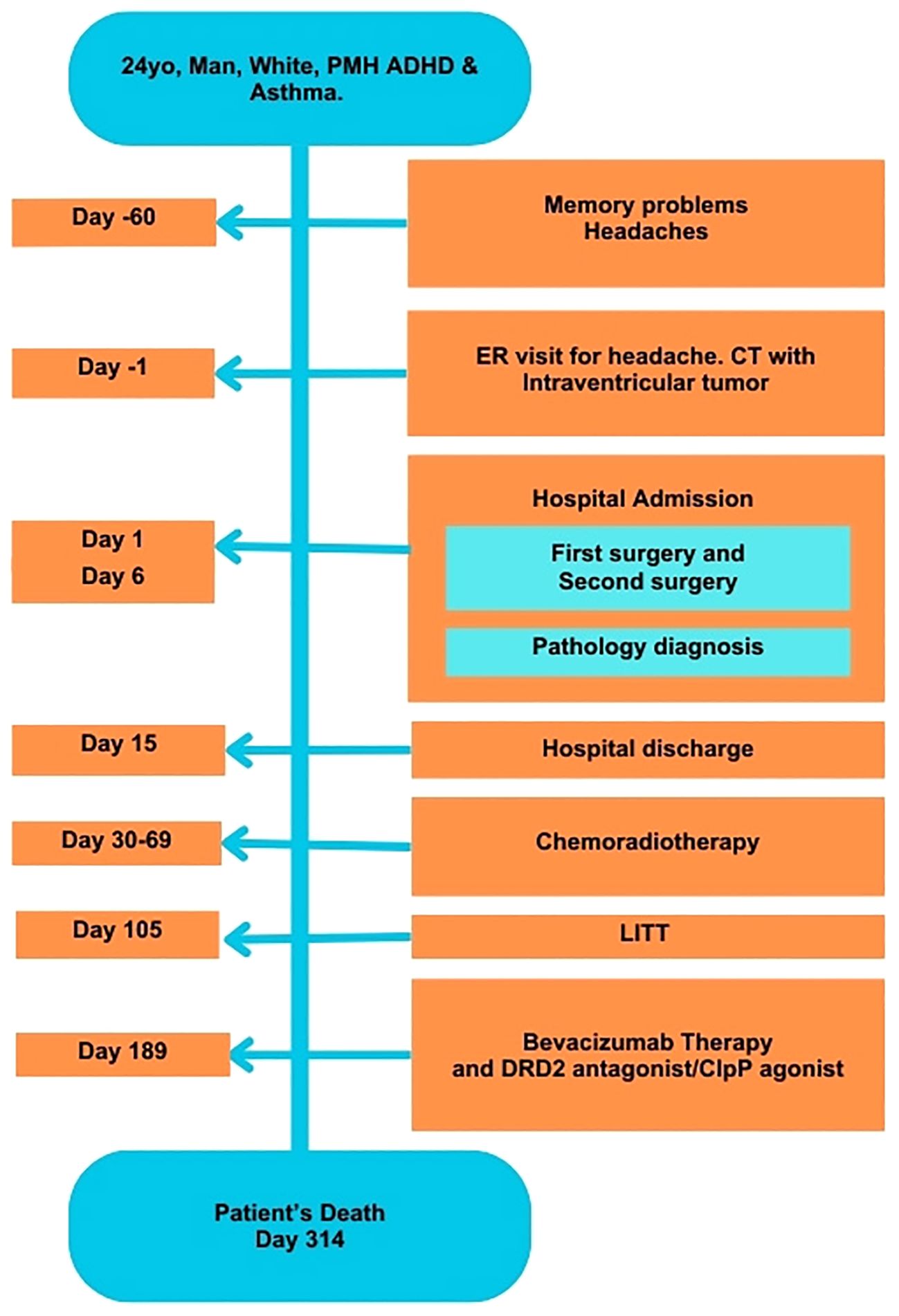
Figure 3. Timeline of development of symptoms and tumor treatments. ADHD, Attention Deficit Hyperactivity Disorder; ER, Emergency Room; CT, Computed tomography; LITT, Laser Interstitial Thermal ablation; PMH, Past Medical History.
Diffuse Midline Gliomas (DMGs) with the H3K27M mutation predominantly affect midline structures, such as the thalamus, brainstem, and spinal cord in children and adolescents. In adults, these tumors more commonly involve the diencephalic region, particularly the thalamus, with a mean age of onset around 42 years (1). This case report highlights the aggressive nature and complex management of an H3 K27-altered DMG in an adult with an unusual ventricular location, presenting significant clinical challenges.
Intraventricular tumors are rare and represent 0.8-1.6% of intracranial tumors. Intraventricular tumors are benign and are more common in childhood than adulthood. Some examples of these tumors are neoplasm of choroid plexus, ventricular wall and septum pellucid, and secondary malignant intraventricular tumors like glioblastoma multiforme. The most common clinical presentation is secondary to high ventricular pressure.
Table 1 presents a comparative analysis of 22 reported DMG cases with ventricular involvement. Most of these cases involve patients of Asian descent, suggesting a possible racial predisposition for ventricular involvement. The median age was 24, with a median survival of 10.5 months (1-24 months).
Clinical presentation varies depending on the anatomical area affected. In this case, the intraventricular mass caused headaches, attention, and memory problems for at least two months. These nonspecific symptoms, likely indicative of increased intracranial pressure, may have delayed diagnosis. However, given the aggressive nature of these tumors and the lack of highly effective treatments, it is unclear if an earlier diagnosis would have significantly altered the outcome.
The patient underwent two intraventricular surgeries, one-time LITT, fractionated external beam radiation (30 fractions) in combination with daily temozolomide, 3 sessions of monoclonal antibody therapy with bevacizumab at 10mg/kg every 14 days, and ONC206 drug through compassionate use. These therapies were based on the ASCO-SNO guidelines for diffuse astrocytic and oligodendroglial tumors in adults, encouraging radiotherapy and enrollment in clinical trials with this alteration (10).
The H3K27M mutation in DMG tumors is currently the primary negative prognostic factor in both adults and children. However, differentiation based on other genetic alterations could provide valuable targets for therapy, prognostication, and risk assessment. DMG tumors with H3K27M mutations in the midline region can also exhibit alterations, such as IDH negativity, ATRX loss, CDK2A deletion, TP53 overexpression, EGFR expression, and MGMT promoter methylation (11, 12).
In this case, the tumor exhibited the classic H3K27M protein expression alongside ATRX loss, moderate TP53 expression, EGFR overexpression by IHC, and H3F3A K28M mutation along with PTEN G132V – subclonal, RAD51B loss on exon 8, TSC2 E1344del, ATRX splice site 6849 + 2T>C by next generation sequencing (Foundation Medicine, Cambridge, MA). Immunohistochemical markers commonly observed in intraventricular DMG tumor case reports, were GFAP, Olig2, Ki67, and S100, were also expressed (Table 1).
Despite aggressive treatment, the primary ventricular tumor reduced in size by nearly 70%, yet continued to proliferate into adjacent areas like the thalamus and splenium. This progression might be linked to the tumor’s immunophenotype, characterized by EGFR overexpression, loss of H3 K27 trimethylation, a high Ki67 proliferation index (30-40%), and weak P53 expression (13).
EGFR overexpression and loss of H3 K27 trimethylation are associated with increased migratory potential and greater propensity for thalamic invasion. Furthermore, TP53 loss, a common alteration in DMGs, is known to promote tumor self-renewal, induce epigenetic dysregulation, and confer resistance to radiotherapy (12, 13).
The molecular profile of this tumor likely played a critical role in its development, migration, and response or lack of response to treatment. Interestingly, the immunophenotype in this case differed from previously reported intraventricular DMG cases, aligning more closely with diffuse midline gliomas originating in the thalamus. This unique molecular profile may have facilitated the tumor’s expansion from the ventricles into adjacent structures, including the thalamus, splenium, and third ventricle, contributing to its aggressive progression and resistance to conventional therapies.
The classification of DMG H3 K27-altered tumors was first designated in 2016 and was updated in 2021 to incorporate alternative mechanisms of H3K27 methylation loss. As such, documenting and analyzing atypical cases is vital to improving our understanding of this complex disease. These tumors are more frequently seen in children and primarily affect deep midline structures. Intracranial intraventricular tumors are rare, comprising less than 1.6% of all tumors, and typically have favorable outcomes when treated. However, this case presents a difficult-to-treat tumor with an unusual growth pattern from the ventricle to diencephalic structures.
Cases like the one presented here offer an opportunity to explore whether delayed diagnoses in adults are due to different growth rates, whether the tumor phenotype varies across age groups, or if molecular markers can predict tumor progression or indicate epigenetic alterations. Current treatment protocols are not specifically designed to target this mutation, leading to highly variable prognoses. Unfortunately, no single factor has yet been definitively identified as having a significant impact on outcomes, highlighting the ongoing need for research and the development of more effective, mutation based targeted therapies.
The original contributions presented in the study are included in the article. Further inquiries can be directed to the corresponding author.
The studies involving humans were approved by the Committee for the Protection of Human Subjects (CPHS). The studies were conducted in accordance with the local legislation and institutional requirements. However, the consent states that the patient will not be personally identified in any reports or publications that may result from this study. The content of this case report does not identify the patient. The patient is no longer alive to obtain consent for this specific publication.
MJ: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing. JZ: Formal analysis, Investigation, Supervision, Validation, Writing – review & editing. MBB: Writing – review & editing, Data curation, Methodology, Supervision, Formal analysis, Validation, Investigation, Resources, Visualization. GH: Conceptualization, Formal analysis, Methodology, Resources, Supervision, Validation, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Bin-Alamer O, Ae J, Td A, Bettegowda C, Mukherjee D. H3K27-altered diffuse midline gliomas among adult patients: A systematic review of clinical features and survival analysis. World Neurosurg. (2022) 165:e251–64. doi: 10.1016/j.wneu.2022.06.020
2. Wang L, Li Z, Zhang M, Piao Y, Chen L, Liang H, et al. H3 K27M–mutant diffuse midline gliomas in different anatomical locations. Hum Pathol. (2018) 78:89–96. doi: 10.1016/j.humpath.2018.04.015
3. Louis D, Perry A, Wesseling P. The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro-Oncol. (2021) 23:1231–51. doi: 10.1093/neuonc/noab106
4. van den Bent M, Saratsis AM, Geurts M, Franceschi E. H3 K27M-altered glioma and diffuse intrinsic pontine glioma: semi-systematic review of treatment landscape and future directions. Neuro-oncol. (2023) 26(Supplement_2):S110–S124. doi: 10.1093/neuonc/noad220
5. Daoud EV, Rajaram V, Cai C, Oberle R, Matin G, Raisanen J, et al. Adult brainstem gliomas with H3K27M mutation: radiology, pathology and prognosis. J Neuropathol Exp Neurol. (2018) 77:302–11. doi: 10.1093/jnen/nly006
6. Wang Y, Feng L, Ji P, Liu J, Guo S, Zhai Y, et al. Clinical features and molecular markers on diffuse midline gliomas with H3K27M mutations: A 43 cases retrospective cohort study. Front Oncol. (2021) 10:602553. doi: 10.3389/fonc.2020.602553
7. Luo Y, Zeng L, Xie XQ, Wang F, Li YZ, Wu DB, et al. H3K27M mutant diffuse midline glioma: a case report. Eur Rev Med Pharmacol Sci. (2020) 24:2579–84. doi: 10.26355/eurrev_202003_20527
8. Zhao B, Sun K, Zhang Z, Xu T, Zhao L, Liu C, et al. A rare presentation of primary lateral ventricle H3 K27-altered diffuse midline glioma in a 14-year-old girl: a case description. Quantitative Imaging Med Surg. (2022) 12:5288–95. doi: 10.21037/qims-22-339
9. Zheng L, Gong J, Yu T, Zou Y, Zhang M, Nie L, et al. Diffuse midline gliomas with histone H3 K27M mutation in adults and children: A retrospective series of 164 cases. Am J Surg Pathol. (2022) 46:863–71. doi: 10.1097/PAS.0000000000001897
10. Mohile NA, Messersmith H, Gatson NT, Hottinger A, Lassman A, Morton J, et al. Therapy for diffuse astrocytic and oligodendroglial tumors in adults: ASCO-SNO guideline. J Clin Oncol. (2022) 40:403–26. doi: 10.1200/JCO.21.02036
11. Schulte J, Buerki R, Lapointe S, Molinaro A, Zhang Y, Villanueva-Meyer J, et al. Clinical, radiologic and genetic characteristics of histone H3 K27M-mutant diffuse midline gliomas in adults. Neuro-Oncol Adv. (2020) 2:1–10. doi: 10.1093/noajnl/vdaa142
12. Gong X, Kuang S, Deng D, Wu J, Zhang L, Liu C. Differences in survival prognosticators between children and adults with H3K27M-mutant diffuse midline glioma. CNS Neurosci Ther. (2023) 29:3863–75. doi: 10.1111/cns.14307
Keywords: diffuse midline glioma (DMG), diffuse midline glioma H3 K27-altered, adult DMG, H3 K27, H3K27M mutation
Citation: Jasso M, Zhu JJ, Bhattacharjee MB and Hergenroeder GW (2025) Case Report: Rare intraventricular H3 K27-altered diffuse midline glioma in an adult. Front. Oncol. 15:1477978. doi: 10.3389/fonc.2025.1477978
Received: 08 August 2024; Accepted: 18 February 2025;
Published: 20 March 2025.
Edited by:
Ramcharan Singh Angom, Mayo Clinic Florida, United StatesCopyright © 2025 Jasso, Zhu, Bhattacharjee and Hergenroeder. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Merari Jasso, bWVyYXJpamphc3NvQGhvdG1haWwuY29t
†ORCID: Georgene W. Hergenroeder, orcid.org/0000-0002-6170-2191
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.