
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Oncol. , 02 April 2025
Sec. Genitourinary Oncology
Volume 15 - 2025 | https://doi.org/10.3389/fonc.2025.1448079
Objective: Kidney-sparing surgery (KSS) has been increasingly performed in patients with upper tract urothelial carcinoma (UTUC) in recent years. We aim to conduct a systematic review and meta-analysis comparing the long-term oncologic and renal function outcomes of KSS with those of radical nephroureterectomy (RNU) for UTUC.
Materials and methods: A literature search was conducted on PubMed, Embase, and Web of Science in January 2024. A meta-analysis was performed to analyze overall survival (OS), cancer-specific survival (CSS), recurrence-free survival (RFS), intravesical recurrence-free survival (IVRFS), metastasis-free survival (MFS), and surgery-related estimated glomerular filtration rate (eGFR) variations.
Results: A total of 32 studies with 21615 patients were included in this meta-analysis. Patients treated with KSS were less likely to have hydronephrosis, more often had low-grade tumors, and were more frequently at a low-stage compared to those undergoing RNU. There were no significant differences between the KSS and RNU groups in terms of 5-year OS, 5-year CSS, 5-year RFS, 5-year IVRFS, 5-year MFS, and hazard ratios (HRs) for OS and CSS based on univariate or multivariable Cox regression analysis. Similar results were found in subgroup analyses comparing segmental urethrectomy (SU) with RNU. In the comparison between the endoscopic management (EM) and RNU groups, EM was associated with worse overall survival outcomes (HR,1.40; 95%CI,1.08-1.82; P=0.01) based on multivariable Cox regression analysis, and the upper tract recurrence rate (OR,39.06; 95%CI, 14.55-104.85; P<0.00001) was significantly higher in the EM group. On the other hand, in patients treated with KSS, postoperative renal function as measured by eGFR increased by 0.4ml/min/1.73 m2, while it decreased by 11.4ml/min/1.73 m2 in the RNU group (WMD, 11.81 ml/min/1.73 m2; 95%CI,9.06-14.56; P<0.0001).
Conclusion: Our meta-analysis supports similar oncological outcomes between KSS and RNU, although tumor characteristics were not equally balanced. KSS can be considered the best choice option for patients with low-risk UTUC, as it offers better preservation of renal function. In terms of kidney-sparing options, SU could be a better alternative for the treatment of ureteral tumors compared to ureteroscopy, due to the higher local recurrence rate associated with EM.
Systematic review registration: https://inplasy.com/inplasy-2024-5-0051/, identifier (INPLASY202450051).
Upper tract urothelial carcinoma (UTUC) is a relatively rare cancer that accounts for 5-10% of all urothelial carcinomas affecting the renal pelvis and ureter (1). Regardless of tumor location, radical nephroureterectomy (RNU) with bladder cuff excision is still considered the standard treatment for UTUC. However, RNU has a significant and enduring detrimental impact on renal function, which puts the patient at risk of chronic kidney disease (CKD) and related sequelae, particularly in patients with impaired renal function (2). Kidney-sparing surgery (KSS) has been proposed as an alternative to RNU for the treatment of selected cases of UTUC, including ureteroscopy, percutaneous access, and segmental urethrectomy (SU). The European Association of Urology (EAU) guidelines recommend KSS as a treatment option for UTUC patients with low-grade, low-stage, unifocal, or small-volume tumors (≤ 2 cm) (3). Patients with compelling indications such as solitary kidney, renal insufficiency, or bilateral UTUC can also be considered KSS as a treatment for UTUC.
However, the safety and efficacy of KSS are still controversial in some literature, the aim of this study is to perform a systematic review and meta-analysis on the oncologic and renal function outcomes between KSS and RNU for patients with UTUC.
Two investigators (LQZ and SS) performed a computerized bibliographic search using a combined text and MeSH heading search strategy in Pubmed, Embase, and Web of Science up to January 2024. The following search strategy was used for this study: (Ureteroscopic OR Percutaneous Nephroscopy OR Endoscopy OR Segmental Ureterectomy OR Partial Ureterectomy OR Distal Ureterectomy OR Nephron Sparing OR Kidney Sparing) AND (Upper Tract Urothelial Carcinoma OR Urothelial Carcinoma OR Transitional Cell Carcinoma) AND (Radical OR Nephroureterectomy). In addition, the reference lists in recent reviews, meta-analyses, and included articles were manually searched to identify related articles. The research was focused on English-language studies and did not include conference abstracts, conference papers, notes, letters, editorials, and short surveys.
According to the Preferred Reporting Items for Systematic Reviews and Meta-analysis (PRISMA) guidelines, we used the Population, Intervention, Comparator, Outcome (PICO) approach to define study eligibility: patients with UTUC (P) undergoing KSS (I) or RNU (C) to compare oncological and renal function outcomes (O). We defined the inclusion and exclusion criteria for study selection at the initiation of the search. The inclusion criteria were: (1) Patients diagnosed with non-metastatic upper tract urothelial carcinoma; (2) The intervention group included only patients treated with ureteroscopic or percutaneous surgery, SU, or distal urethrectomy; (3) The control group included only patients treated with RNU; (4) The study reported at least one of the following outcomes: overall survival (OS), cancer-specific survival (CSS), recurrence-free survival (RFS), intravesical RFS (IVRFS), metastasis-free survival (MFS), and changes in estimated glomerular filtration rate (eGFR) related to surgery. The exclusion criteria were: (1) Single-arm studies; (2) Metastatic UTUC; (3) Adolescents (under 18 years of age); (4) The study period in the literature before 1990.
Two investigators (KPN and CYH) independently extracted the data from all eligible publications. Any differences among evaluators were resolved through discussion with a third investigator (ST). A standardized pre-piloted data extraction sheet was used. The extracted information included: (1) Baseline features: First author's name, year of publication, country, recruitment period, sample size, patient's age, gender (male), hydronephrosis status, follow-up time, and details regarding intervention and control; (2) Pathologic outcomes: tumor grade, tumor stage; (3) Survival outcomes: OS, CSS, RFS, IVRFS, MFS, and hazard ratios (HRs) with 95% confidence intervals (CIs) of OS and CSS in univariate or multivariate Cox analyses for comparing surgical techniques: KSS vs. RNU; (4) Functional outcomes: preoperative eGFR, postoperative eGFR, and changes in eGFR.
The quality of included studies was independently evaluated by two authors (LQZ and SS) using the Newcastle-Ottawa Scale (NOS) (4), which includes three domains: selection of the study population, comparability of the groups, and ascertainment of the outcome. High-quality articles were identified as those with NOS scores ranging from 6 to 9, whereas scores ranging from 0 to 5 were considered to indicate poor quality.
Oncological outcomes were primarily assessed using available HRs from univariable or multivariate analyses and their corresponding 95% CIs and 5-year survival endpoints. When 5-year survival endpoints were not available, we applied the formula described by Parmar et al. (5). We used the software Engauge Digitizer version 4.1 to calculate values derived from published Kaplan-Meier curves.
Mean changes in eGFR were used to assess the surgery-related alterations in renal function. Mean preoperative and postoperative eGFR values were recorded, along with their standard deviations (SDs), to evaluate the weighted mean difference.
Continuous and dichotomous variables were considered: inverse variance weighted mean difference (WMD) was used to summarize continuous variables, while the Mantel-Haenszel test was used to calculate odds ratios (ORs) with 95% CIs for binary values. Meta-analyses of pooled data were conducted using a fixed-effects or random-effects model. Statistical heterogeneity was assessed using the Chi-square test and I2 test. The I2 values ranges from 0% to 100%, with higher values indicating a greater degree of heterogeneity. If I2 > 50%, significant between-study heterogeneity was present, and the random-effect model was used. Two-tailed p < 0.05 was considered statistically significant. The data were analyzed with Review Manager (RevMan) 5.3, which was developed by the Cochrane Collaboration and Stata 17 software (Stat Corp, College Station, TX, USA).
The full process of the systematic literature review is shown in Figure 1. According to the PRISMA search strategy, no randomized controlled trials were available. Thirty-one retrospective studies and one prospective study, comprising a total of 21615 patients (7048 in the KSS group and 14567 in the RNU group), were ultimately included in the meta-analysis (Table 1) (6–37). Among these 32 articles, 10 focused on endoscopic management (EM), which included ureteroscopy surgery and percutaneous nephroscopy surgery, compared to RNU (6, 7, 10, 13, 14, 16, 28, 29, 34, 35), 19 studies compared SU to RNU (9, 11, 12, 15, 17, 18, 20–27, 30, 32, 33, 36, 37), and 3 involved comparisons among EM, SU, and RNU (8, 19, 31). The oncological characteristics and eGFR changes associated with surgery of the included studies are shown in Supplementary Table 1. The assessments of the NOS are presented in Supplementary Table 2, and the results showed that all the studies were of high quality with scores ranging from 6 to 8.
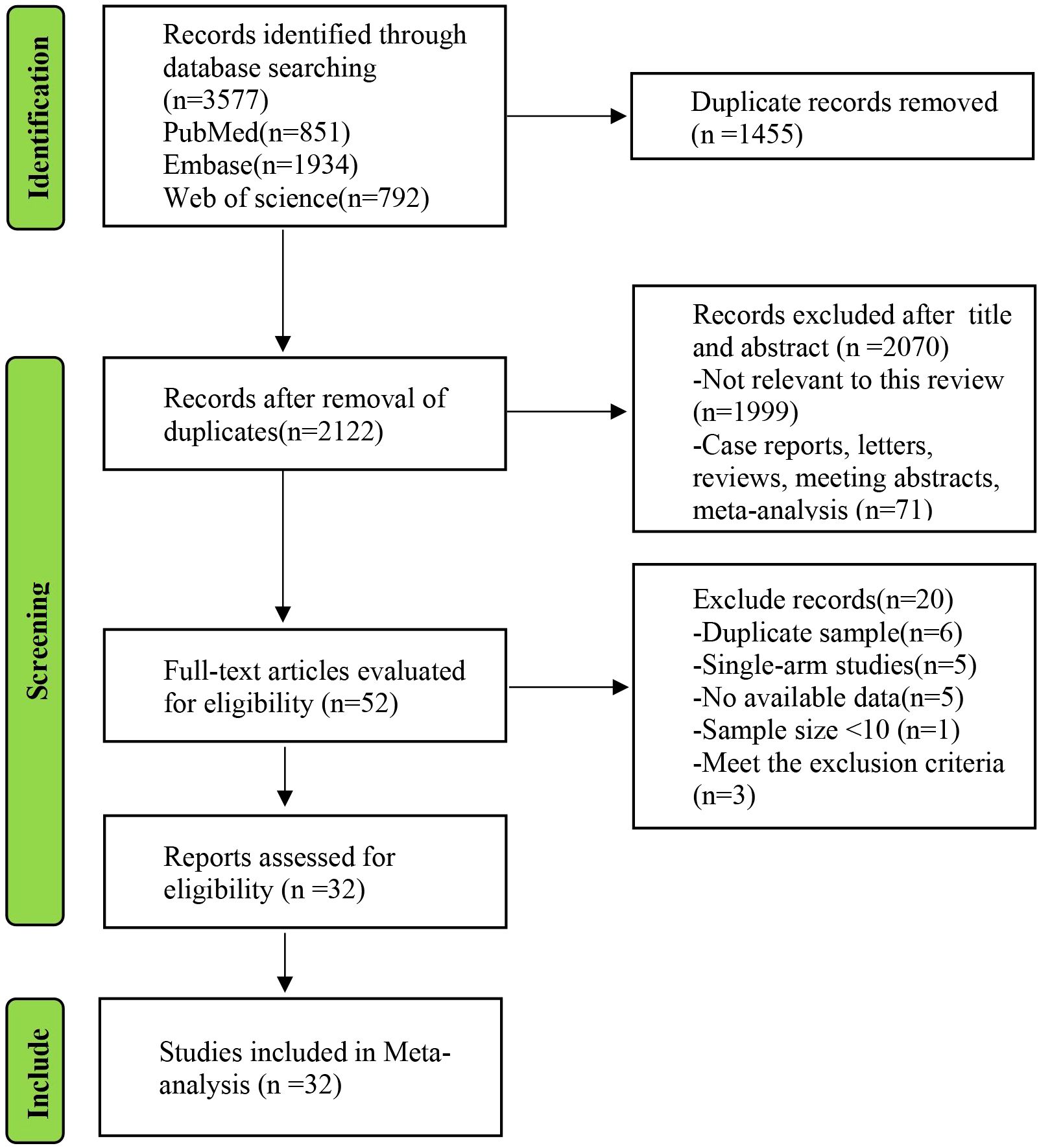
Figure 1. PRISMA flow diagram of the study process. PRISMA, Preferred Reporting Items for Systematic review and Meta-analysis.
The survival outcomes in the included studies are described in Table 2. No statistically significant differences were found in terms of age or gender (Supplementary Figure 1A, B). However, some significant differences were observed between the KSS and RNU groups. Patients treated with KSS were less likely to have hydronephrosis (OR,0.45; 95%CI, 0.27-0.76; P=0.003) (Supplementary Figure 1C) and were more likely to have low-grade tumors (OR,1.80; 95%CI,1.39-2.33; P<0.0001) compared with those treated with RNU (Supplementary Figure 1D). Additionally, patients in the KSS group also presented with a lower tumor stage of ≤pT1 (OR, 1.69; 95% CI, 1.33-2.16; P < 0.0001) (Supplementary Figure 1E).
Our investigation revealed no significant differences in terms of 5-year OS, 5-year CSS, 5-year RFS, 5-year IVRFS, and 5-year MFS between the KSS and RNU groups, as shown in Figures 2A–E. This finding was confirmed by both univariate and multivariate Cox regression analysis. The surgical treatment-related hazard ratios (HRs) for KSS versus RNU from univariate analyses for OS were HR, 0.99; 95% CI, 0.94-1.03; P=0.54 (Figure 2F). From multivariate analyses for OS, the HR was 1.34; 95% CI, 0.95-1.89; P=0.09 (Figure 2G). Univariate analyses for CSS showed an HR of 1.06 (95% CI, 0.94-1.19; P=0.37) (Figure 2H), while multivariate analyses for CSS showed an HR of 1.34 (95% CI, 0.92-1.96; P=0.13) (Figure 2I). Similarly, the observed differences were not statistically significant.
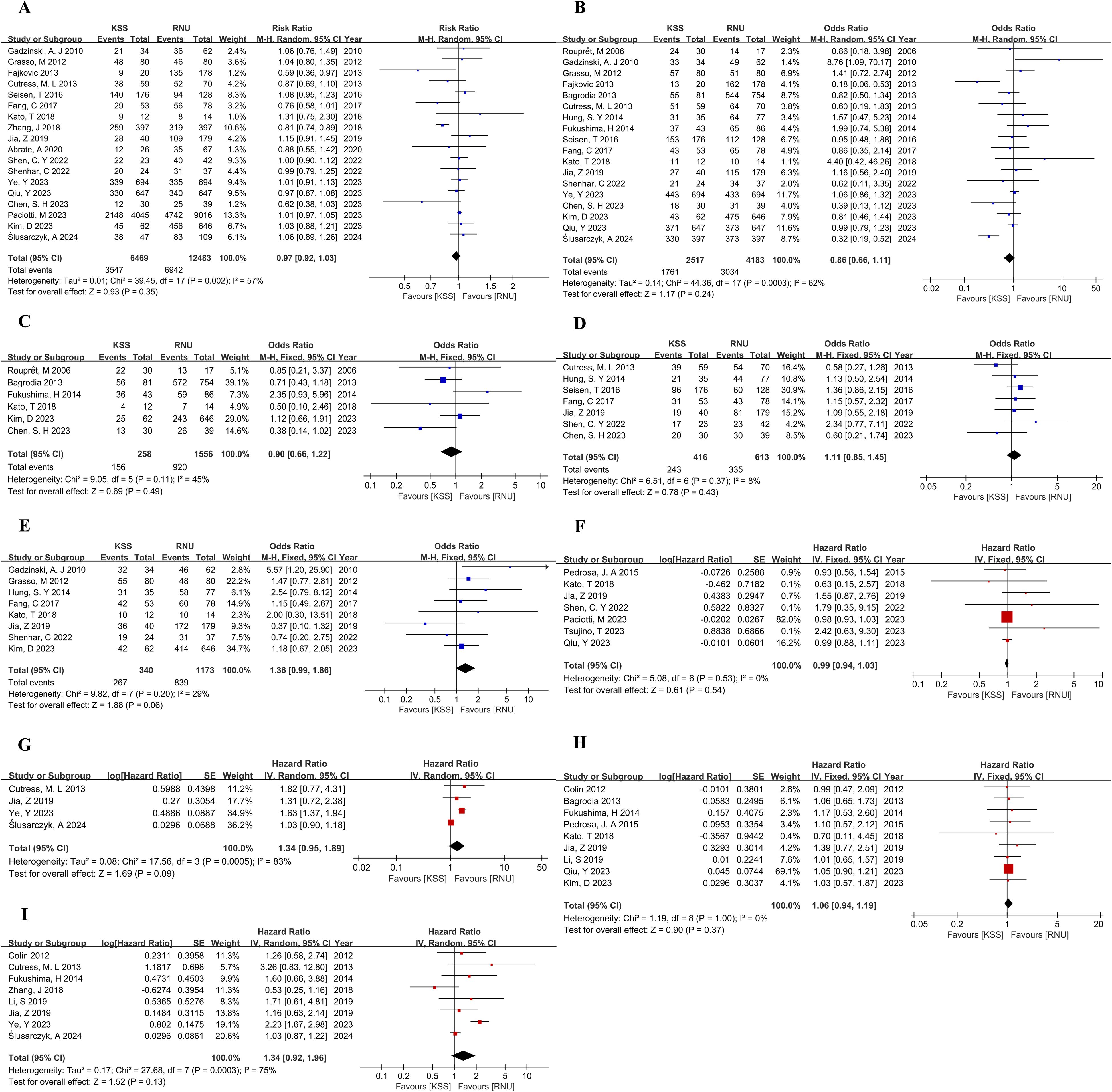
Figure 2. Forest plot of meta-analyses comparing oncologic outcomes between the KSS and RNU groups: (A) 5-year OS; (B) 5-year CSS; (C) 5-year RFS; (D) 5-year IVRFS; (E) 5-year MFS; (F) HR for OS from univariate analyses; (G) HR for OS from multivariate analyses; (H) HR for CSS from univariate analyses; (I) HR for CSS from multivariate analyses.
To reduce heterogeneity, we conducted a comprehensive comparison of survival outcomes between different treatment groups. Subgroup analysis was performed on SU versus RNU and EM versus RNU. In terms of pathologic outcomes between the SU and RNU groups, no statistically significant differences were found in tumor grade (OR,1.24; 95%CI, 0.99-1.55; P=0.06)(Supplementary Figure 2A). However, the SU group had a lower tumor stage of ≤pT1 (OR, 1.47; 95% CI, 1.19-1.93; P =0.0004)(Supplementary Figure 2B), which was statistically significant. The 5-year OS, 5-year CSS, 5-year RFS,5-year IVRFS, and 5-year MFS showed no significant differences between SU and RNU groups. The same results for OS and CSS were observed in both univariate and multivariate Cox regression analyses between SU and RNU groups (Figure 3).
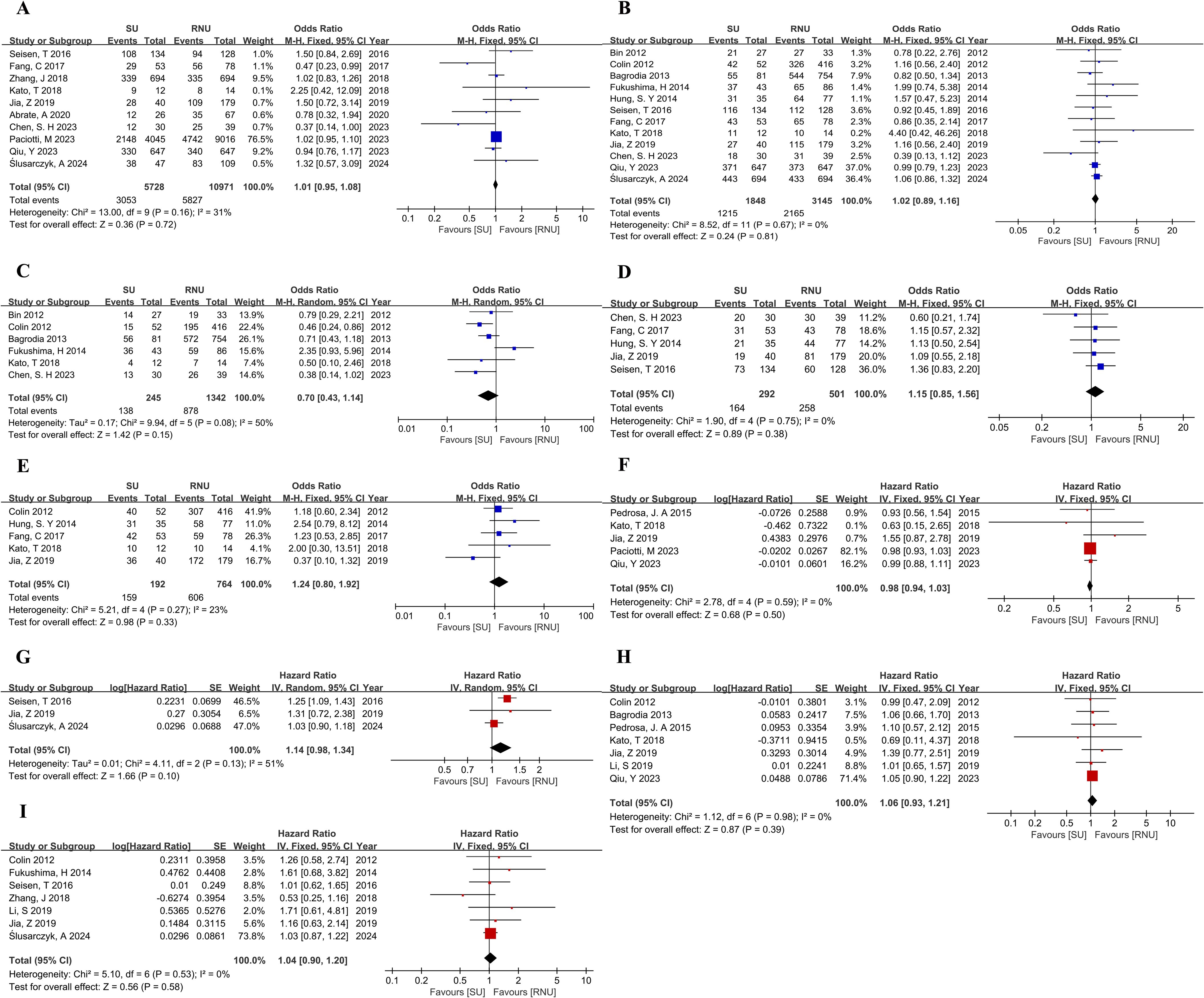
Figure 3. Forest plot of meta-analyses comparing oncologic outcomes between the SU and RNU groups: (A) 5-year OS; (B) 5-year CSS; (C) 5-year RFS; (D) 5-year IVRFS; (E) 5-year MFS; (F) HR for OS from univariate analyses; (G) HR for OS from multivariate analyses; (H) HR for CSS from univariate analyses; (I) HR for CSS from multivariate analyses.
In the comparison between the EM and RNU groups, significant differences were also observed in pathologic outcomes. The EM group had more low-grade tumors (OR,4.67; 95%CI,1.77-12.32; P=0.002) and low-stage tumors (≤pT1) (OR, 2.65; 95% CI, 1.31-5.37; P=0.007) (Supplementary Figure 3). There were no significant differences in 5-year OS, 5-year CSS, 5-year IVRFS, and 5-year MFS between the EM and RNU groups, as depicted in Figure 4A–D. However, in multivariate Cox regression analysis, the HR for OS was 1.42 (95%CI,1.07-1.88; P=0.01) between the EM and RNU groups, indicating a statistically significant difference and suggesting had worse overall survival outcomes for endoscopic excision compared to RNU (Figure 4E). There was no statistically significant difference in CSS (HR,1.71; 95%CI,0.84-3.48; P=0.14) in multivariate analysis (Figure 4F). Moreover, the upper tract recurrence rate was calculated and found to be significantly higher in the EM group compared to the RNU group (OR,39.06; 95%CI,14.55-104.85; P<0.00001) (Figure 4G).
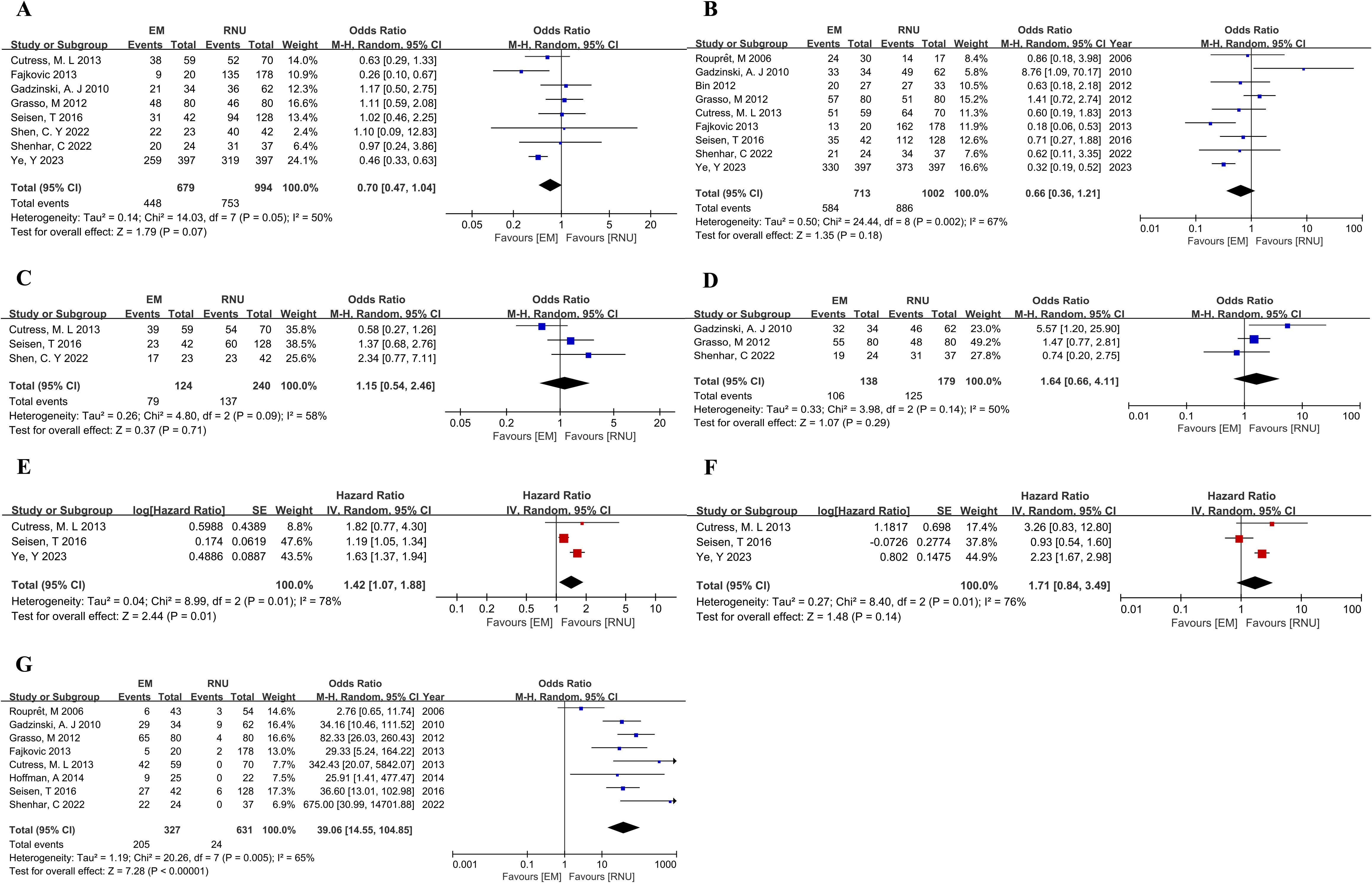
Figure 4. Forest plot of meta-analyses comparing oncologic outcomes between the EM and RNU groups: (A) 5-year OS; (B) 5-year CSS; (C) 5-year IVRFS; (D) 5-year MFS; (E) HR for OS from multivariate analyses; (F) HR for CSS from multivariate analyses; (G) Upper tract recurrence rate.
No statistically significant difference was found in preoperative eGFR between the KSS and RNU groups (WMD, 2.81 ml/min/1.73 m2; 95%CI,-1.43-7.05;P=0.19) (Figure 5A). Howere, significant differences were observed in the weighted mean changes in eGFR between the two groups. In patients treated with KSS, postoperative eGFR increased by 0.4 ml/min/1.73m2, whereas it decreased by 11.4ml/min/1.73 m2 in the RNU group (WMD, 11.81 ml/min/1.73m2; 95%CI, 9.06-14.56; P<0.0001) (Figure 5B). That indicates that patients treated with KSS experienced significantly better preservation of renal function compared to those treated with RNU.
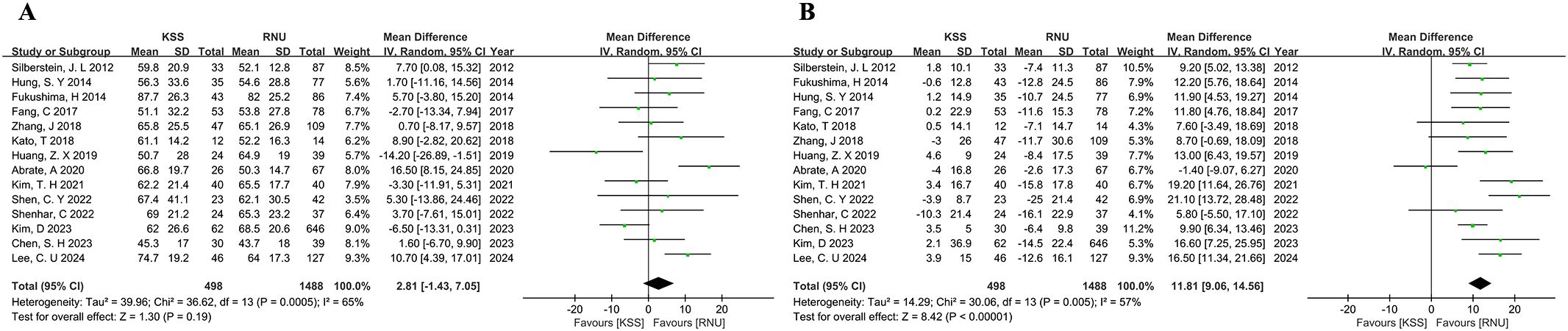
Figure 5. Forest plot of meta-analyses comparing renal function between the KSS and RNU groups: (A) Preoperative eGFR; (B) Weighted mean changes in eGFR.
A sensitivity analysis was performed on the 5-year OS, 5-year CSS, HR for OS from univariate analyses, HR for CSS from univariate analyses, preoperative eGFR, and weighted mean changes in eGFR (as depicted in Figure 6) between the KSS and RNU groups. The significance of the pooled comparison between the two groups was not influenced by the removal of any single study, indicating that the results of our meta-analysis were stable.
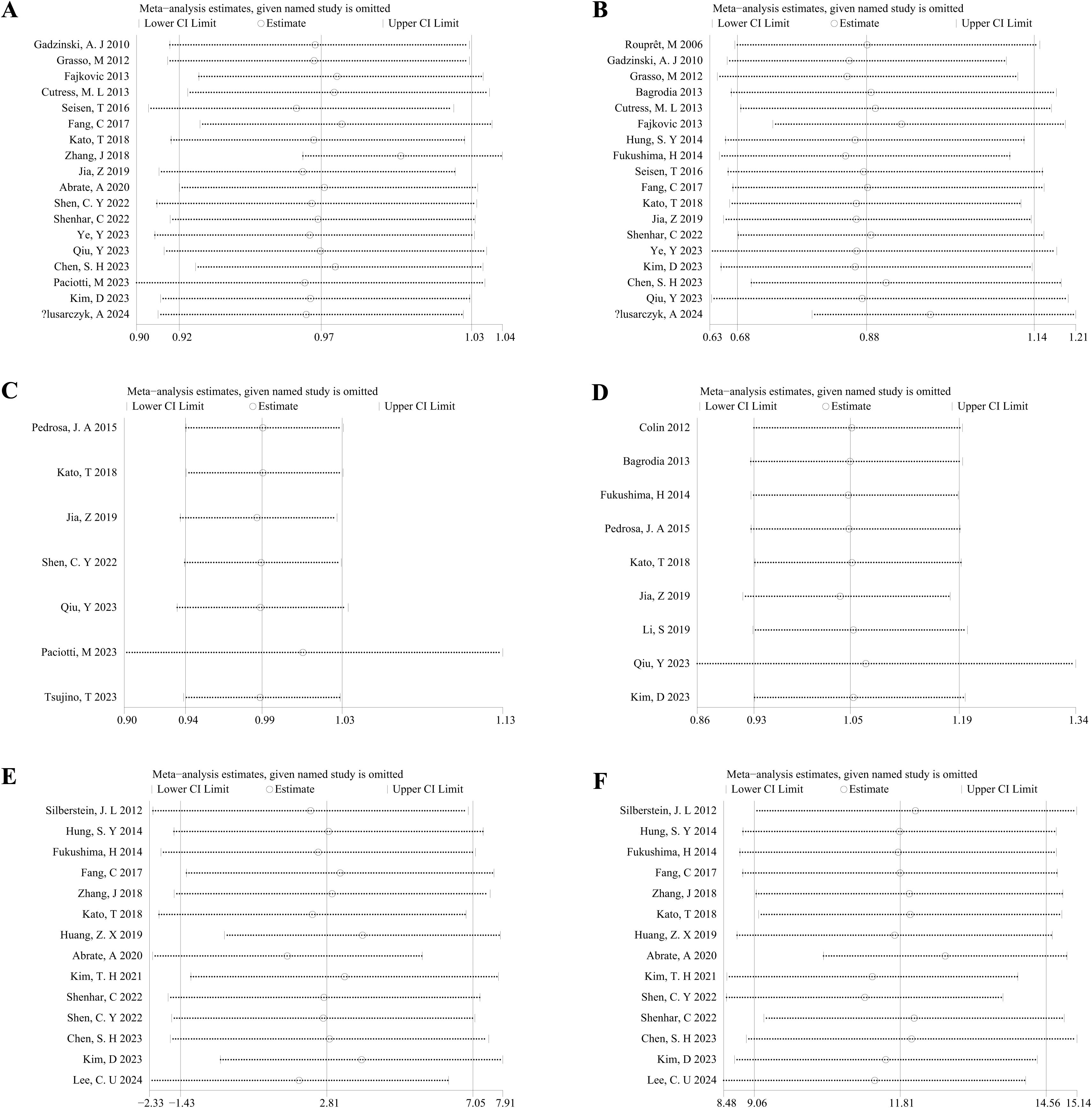
Figure 6. Sensitivity analysis of the association between the KSS and RNU groups: (A) 5-year OS; (B) 5-year CSS; (C) HR for OS from univariate analyses; (D) HR for CSS from univariate analyses; (E) Preoperative eGFR; (F) Weighted mean changes in eGFR.
The funnel plots were used to assess the publication bias of the included studies, and no significant publication bias was detected between KSS and RNU groups regarding 5-year OS, 5-year CSS, HR for OS from univariate analyses, HR for CSS from univariate analyses, preoperative eGFR, weighted mean changes in eGFR (Figure 7).
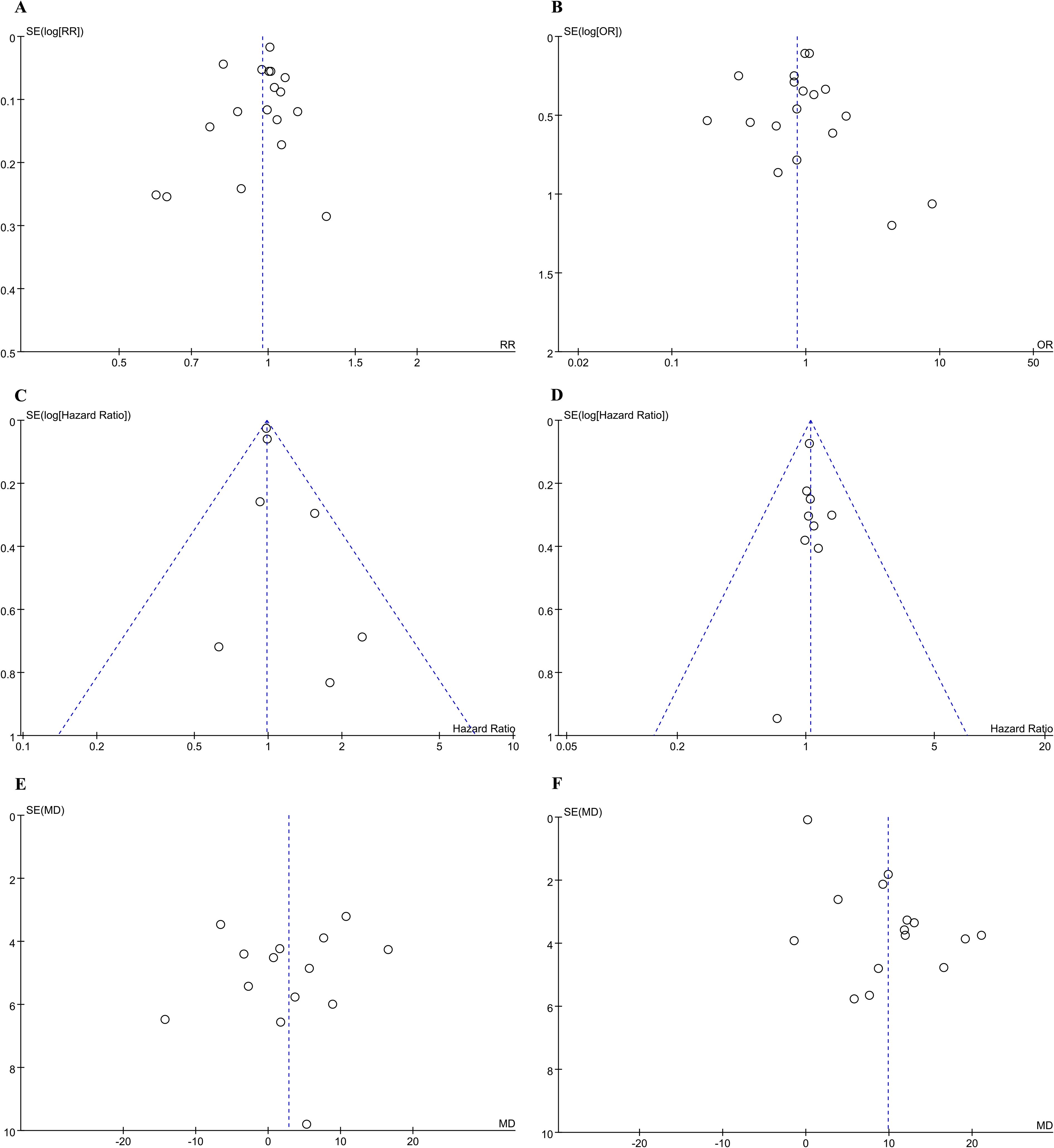
Figure 7. Funnel plot for the evaluation of potential publication bias between the KSS and RNU groups: (A) 5-year OS; (B) 5-year CSS; (C) HR for OS from univariate analyses; (D) HR for CSS from univariate analyses; (E) Preoperative eGFR; (F) Weighted mean changes in eGFR.
In recent years, KSS has been increasingly performed to avoid complications associated with RNU and to theoretically preserve postoperative renal function. However, the assumptions regarding the comparative outcomes between KSS and RNU are predominantly based on heterogeneous data derived from small retrospective cohort studies, which are considered to provide a low level of evidence in the current literature (38, 39). To address this gap, we conducted a systematic review and meta-analysis to compare oncological and renal function outcomes between KSS and RNU. Our analysis was based on data obtained from all available comparative studies, with additional subgroup analyses conducted to examine differences between SU and RNU, as well as between EM and RNU.
In our meta-analysis, KSS demonstrated feasible efficacy regarding oncological outcomes. Specifically, there were no significant differences in the 5-year OS, 5-year CSS, 5-year RFS, 5-year IVRFS, and 5-year MFS between the KSS and RNU groups. This finding was consistent across both univariate and multivariate Cox regression analyses for OS and CSS. In the survival outcomes mentioned above, it is generally believed that the IVRFS of the KSS group is often inferior to that of the RNU group due to the complete bladder cuff excision performed in the RNU group. However, our study found no significant difference in 5-year IVRFS between the two groups, which may be attributed to the lower grade (18, 19, 31, 32, 34) or lower stage (11, 18, 19, 26, 31, 34, 40) of disease in the KSS group. Additionally, patients undergoing RNU were more likely to present with preoperative hydronephrosis (11, 41) and more advanced disease at the outset, which may have contributed to a reduced survival rate. This patient selection bias may have potentially skewed the results. Despite these considerations, based on the outcomes of our study, KSS can be a viable treatment option for patients with lower-grade and lower-stage UTUC.
Since KSS encompasses both the EM and SU approaches, we conducted a subgroup analysis between SU and RNU to reduce selection bias. Patients treated with ureteroureterostomy or ureteral reimplantation for ureteral tumors are collectively referred to as undergoing SU. Our study found no significant differences in the 5-year OS, 5-year CSS, 5-year RFS,5-year IVRFS, and 5-year MFS between the SU and RNU groups. This was further supported by the results of univariate and multivariate Cox regression analyses for OS and CSS, which showed no significant difference between the two groups. According to the EAU guidelines, SU is recommended only for imperative cases or highly selected patients with low-risk UTUC, due to the potential increased risk of ipsilateral upper urinary tract recurrence and CSS compared to radical extirpation surgery (3). However, our study demonstrated no statistically significant difference in RFS, IVRFS, and CSS between the SU and RNU groups. Even though all included trials in our meta-analysis involved high-grade tumors, the survival outcomes showed no significant differences between SU and RNU. This suggests that SU can be a valid option for patients with high-grade ureteric urothelial cancer, particularly for those with distally located tumors in the ureter. SU could provide complete removal of tumors, lymph nodes, and possibly invaded tissues. Based on our findings, SU can be safely performed regardless of tumor grade for patients with distal ureteral tumors, without increasing patient mortality.
Over the past decade, advancements in material and laser technology have led to an increased adoption of endoscopic management for UTUC by many surgical teams (42). EM, which includes percutaneous and ureteroscopic resection, fulguration, or ablation, has been widely applied (19). Numerous case-control studies have suggested that endoscopic management can be recommended as an alternative to nephroureterectomy for low-risk or superficial UTUC, with no observed impact on survival (16, 28, 29, 34). However, our meta-analysis revealed that the local recurrence rate (OR, 39.06) was substantially higher in the EM group compared to the RNU group. Upper tract recurrence is an inevitable consequence of endoscopic treatment, regardless of tumor grade. The high local recurrence rate necessitates repeated endoscopic treatments or delayed nephrectomy, imposing an increased mental, physical, and financial burden on patients (35). Furthermore, in multivariate Cox regression analysis, the HR for OS was found to be higher in the EM group, indicating a shorter OS for this group and multiple studies have reached the same conclusion (19, 35). Another limitation of the ureteroscopic treatment for UTUC includes the potential for under grading on initial biopsy and the inability to fully assess the extent of UTUC (35). Despite this, no significant differences were observed in the 5-year OS, 5-year CSS, 5-year IVRFS, 5-year MFS, and CSS in multivariate analysis.
The primary advantage of KSS is the preservation of kidney function, which can lead to better outcomes in renal function and increased eligibility for adjuvant chemotherapy (27). In our meta-analysis, we observed that postoperative eGFR increased by 0.4ml/min/1.73m2 in patients treated with KSS, while it decreased by 10.4ml/min/1.73 m2 in the RNU group (WMD,10.78ml/min/1.73m2). This indicates that preserving the ipsilateral renal unit results in significantly less reduction in renal function compared to RNU. The improved renal function following KSS is likely due to the resection of a partially or completely obstructing ureteral tumor, thereby allowing for enhanced ipsilateral renal function (36). Another study found that the greatest difference in renal function was observed during follow-up at 3 months after intervention, but no significant difference was seen at 2and 5 years after intervention. This also indicates that, at least within the first 2 years, the renal function in the KSS group is superior to that of the RNU group (43). Moreover, patients who underwent KSS were more likely to be candidates for adjuvant chemotherapy because of the preservation of eGFR (44). More importantly, the preservation of the renal unit is crucial for enhancing life expectancy (29). Thus, our results suggest that patients may experience better postoperative renal function after KSS compared to RNU, thereby avoiding the unnecessary risk of postoperative dialysis, which can significantly impact the quality of life.
To our knowledge, this is the only meta-analysis comparing KSS with RNU, and it is also the most up-to-date systematic review and meta-analysis comparing SU with RNU, as well as EM with RNU. However, our study is limited by its retrospective design and the heterogeneity in definitions, inclusion criteria, therapies, follow-up periods, reporting methods, and surgeon expertise. These factors make the study susceptible to selection and reporting biases. Nevertheless, this limitation is inherent to the field of UTUC. In the future, there is a need for more multicenter, randomized controlled trials with large sample sizes and high quality. Ideally, these studies would provide more accurate data, including detailed information on tumor size, location, stage, and grade.
Our systematic review and meta-analysis support the notion that KSS yields similar oncological outcomes compared to RNU, albeit with the caveat that tumor characteristics were not equally balanced between the two groups. KSS can be considered a viable treatment option for patients with low-grade and low-stage UTUC. Furthermore, we observed that renal function preservation is significantly better after KSS compared to RNU. Although EM is part of KSS and is associated with a higher local recurrence rate and a shorter OS in multivariate analysis, it may necessitate repeated surgical interventions or eventual acceptance of delayed RNU. On the other hand, the distinct benefits of SU include the en-bloc resection of the ureteral tumor with surrounding soft tissue and lymph node dissection. Thus, we believe that SU could be a better alternative for the treatment of ureteral tumors compared with ureteroscopic approaches. Additionally, renal function preservation is significantly better after SU when compared to RNU. These findings suggest that SU should be considered a first-line treatment for low-grade UTUC of the ureter and may also be appropriate for selected cases of high-grade UTUC.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
LZ: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Software, Writing – original draft. CH: Conceptualization, Investigation, Project administration, Supervision, Writing – review & editing. SS: Data curation, Investigation, Methodology, Software, Writing – original draft. KN: Project administration, Software, Supervision, Validation, Writing – review & editing. ST: Conceptualization, Methodology, Project administration, Resources, Supervision, Validation, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2025.1448079/full#supplementary-material
Supplementary Figure 1 | Forest plot of meta-analyses comparing baseline characteristics between the KSS and RNU groups: (A) Age; (B) Gender; (C) Hydronephrosis; (D) Low-grade tumor; (E) Tumor Stage≤pT1.
Supplementary Figure 2 | Forest plot of meta-analyses comparing pathologic outcomes between the SU and RNU groups: (A) Low-grade tumor; (B) Tumor Stage≤pT1.
Supplementary Figure 3 | Forest plot of meta-analyses comparing pathologic outcomes between the EM and RNU groups: (A) Low-grade tumor; (B) Tumor Stage≤pT1.
1. Collà Ruvolo C, Nocera L, Stolzenbach LF, Wenzel M, Cucchiara V, Tian Z, et al. Incidence and survival rates of contemporary patients with invasive upper tract urothelial carcinoma. Eur Urol Oncol. (2021) 4:792–801. doi: 10.1016/j.euo.2020.11.005
2. Lane BR, Smith AK, Larson BT, Gong MC, Campbell SC, Raghavan D, et al. Chronic kidney disease after nephroureterectomy for upper tract urothelial carcinoma and implications for the administration of perioperative chemotherapy. Cancer. (2010) 116:2967–73. doi: 10.1002/cncr.25043
3. Rouprêt M, Babjuk M, Burger M, Capoun O, Cohen D, Compérat EM, et al. European association of urology guidelines on upper urinary tract urothelial carcinoma: 2020 update. Eur Urol. (2021) 79:62–79. doi: 10.1016/j.eururo.2020.05.042
4. Stang A. Critical evaluation of the newcastle-ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur J Epidemiol. (2010) 25:603–5. doi: 10.1007/s10654-010-9491-z
5. Parmar MK, Torri V, Stewart L. Extracting summary statistics to perform meta-analyses of the published literature for survival endpoints. Stat Med. (1998) 17:2815–34. doi: 10.1002/(sici)1097-0258(19981230)17:24<2815::aid-sim110>3.0.co;2-8
6. Rouprêt M, Hupertan V, Traxer O, Loison G, Chartier-Kastler E, Conort P, et al. Comparison of open nephroureterectomy and ureteroscopic and percutaneous management of upper urinary tract transitional cell carcinoma. Urology. (2006) 67:1181–7. doi: 10.1016/j.urology.2005.12.034
7. Gadzinski AJ, Roberts WW, Faerber GJ, Wolf JS Jr. Long-term outcomes of nephroureterectomy versus endoscopic management for upper tract urothelial carcinoma. J Urol. (2010) 183:2148–53. doi: 10.1016/j.juro.2010.02.005
8. Bin X, Roy OP, Ghiraldi E, Manglik N, Liang T, Vira M, et al. Impact of tumour location and surgical approach on recurrence-free and cancer-specific survival analysis in patients with ureteric tumours. BJU Int. (2012) 110:E514–9. doi: 10.1111/j.1464-410X.2012.11199.x
9. Colin P, Ouzzane A, Pignot G, Ravier E, Crouzet S, Ariane MM, et al. Comparison of oncological outcomes after segmental ureterectomy or radical nephroureterectomy in urothelial carcinomas of the upper urinary tract: results from a large french multicentre study. BJU Int. (2012) 110:1134–41. doi: 10.1111/j.1464-410X.2012.10960.x
10. Grasso M, Fishman AI, Cohen J, Alexander B. Ureteroscopic and extirpative treatment of upper urinary tract urothelial carcinoma: A 15-year comprehensive review of 160 consecutive patients. BJU Int. (2012) 110:1618–26. doi: 10.1111/j.1464-410X.2012.11066.x
11. Silberstein JL, Power NE, Savage C, Tarin TV, Favaretto RL, Su D, et al. Renal function and oncologic outcomes of parenchymal sparing ureteral resection versus radical nephroureterectomy for upper tract urothelial carcinoma. J Urol. (2012) 187:429–34. doi: 10.1016/j.juro.2011.09.150
12. Bagrodia A, Kuehhas FE, Gayed BA, Wood CG, Raman JD, Kapur P, et al. Comparative analysis of oncologic outcomes of partial ureterectomy vs radical nephroureterectomy in upper tract urothelial carcinoma. Urology. (2013) 81:972–7. doi: 10.1016/j.urology.2012.12.059
13. Cutress ML, Stewart GD, Tudor EC, Egong EA, Wells-Cole S, Phipps S, et al. Endoscopic versus laparoscopic management of noninvasive upper tract urothelial carcinoma: 20-year single center experience. J Urol. (2013) 189:2054–60. doi: 10.1016/j.juro.2012.12.006
14. Fajkovic H, Klatte T, Nagele U, Dunzinger M, Zigeuner R, Hübner W, et al. Results and outcomes after endoscopic treatment of upper urinary tract carcinoma: the Austrian experience. World J Urol. (2013) 31:37–44. doi: 10.1007/s00345-012-0948-4
15. Fukushima H, Saito K, Ishioka J, Matsuoka Y, Numao N, Koga F, et al. Equivalent survival and improved preservation of renal function after distal ureterectomy compared with nephroureterectomy in patients with urothelial carcinoma of the distal ureter: A propensity score-matched multicenter study. Int J urology: Off J Japanese Urological Assoc. (2014) 21:1098–104. doi: 10.1111/iju.12554
16. Hoffman A, Yossepowitch O, Erlich Y, Holland R, Lifshitz D. Oncologic results of nephron sparing endoscopic approach for upper tract low grade transitional cell carcinoma in comparison to nephroureterectomy - a case control study. BMC Urol. (2014) 14:97. doi: 10.1186/1471-2490-14-97
17. Hung SY, Yang WC, Luo HL, Hsu CC, Chen YT, Chuang YC. Segmental ureterectomy does not compromise the oncologic outcome compared with nephroureterectomy for pure ureter cancer. Int Urol Nephrol. (2014) 46:921–6. doi: 10.1007/s11255-013-0514-z
18. Pedrosa JA, Masterson TA, Rice KR, Kaimakliotis HZ, Monn MF, Bihrle R, et al. Oncologic outcomes and prognostic impact of urothelial recurrences in patients undergoing segmental and total ureterectomy for upper tract urothelial carcinoma. Can Urological Assoc J = J l’Association Des urologues du Canada. (2015) 9:E187–92. doi: 10.5489/cuaj.2408
19. Seisen T, Nison L, Remzi M, Klatte T, Mathieu R, Lucca I, et al. Oncologic outcomes of kidney sparing surgery versus radical nephroureterectomy for the elective treatment of clinically organ confined upper tract urothelial carcinoma of the distal ureter. J Urol. (2016) 195:1354–60. doi: 10.1016/j.juro.2015.11.036
20. Fang C, Xie X, Xu T, He W, He H, Wang X, et al. Segmental Ureterectomy Is Not Inferior to Radical Nephroureterectomy for Either Middle or Distal Ureter Urothelial Cell Carcinomas within 3.5 cm. Int Urol Nephrol. (2017) 49:1177–82. doi: 10.1007/s11255-017-1576-0
21. Kato T, Nakayama R, Haba T, Kawaguchi M, Komiya A, Koike H. Oncological and renal outcomes of segmental ureterectomy vs. Radical nephroureterectomy for upper tract urothelial carcinoma. Oncol Lett. (2018) 16:6861–7. doi: 10.3892/ol.2018.9463
22. Zhang J, Yang F, Wang M, Niu Y, Chen W, Xing N. Comparison of radical nephroureterectomy and partial ureterectomy for the treatment of upper tract urothelial carcinoma. BioMed Res Int. (2018) 2018:2793172. doi: 10.1155/2018/2793172
23. Huang ZX, Zhang XW, Zhang XP, Li Q, Liu SJ, Yu LP, et al. Segmental ureterectomy is acceptable for high-risk ureteral carcinoma comparing to radical nephroureterectomy. J Invest Surg. (2019) 32:746–53. doi: 10.1080/08941939.2018.1457192
24. Jia Z, Gong YQ, Zhang CJ, Bao ZQ, Li XS, Hao H, et al. Segmental ureterectomy can be performed safely in patients with urothelial carcinoma of distal ureter. Can Urological Assoc J = J l’Association Des urologues du Canada. (2019) 13:E202–e9. doi: 10.5489/cuaj.5555
25. Li S, Pan Y, Hu J. Oncologic outcomes comparison of partial ureterectomy and radical nephroureterectomy for urothelial carcinoma. BMC Urol. (2019) 19:120. doi: 10.1186/s12894-019-0557-2
26. Abrate A, Sessa F, Campi R, Preto M, Olivero A, Varca V, et al. Segmental ureterectomy vs. Radical nephroureterectomy for ureteral carcinoma in patients with a preoperative glomerular filtration rate less than 90 ml/min/1.73 M(2): A multicenter study. Urologic Oncol. (2020) 38:601. doi: 10.1016/j.urolonc.2020.03.005
27. Kim TH, Lee CU, Kang M, Jeon HG, Jeong BC, Seo SI, et al. Comparison of oncologic and functional outcomes between radical nephroureterectomy and segmental ureterectomy for upper urinary tract urothelial carcinoma. Sci Rep. (2021) 11:7828. doi: 10.1038/s41598-021-87573-5
28. Shen CY, Jou YC, Kan WC, Tzai TS, Tsai YS. Outcome of non-muscle invasive upper tract urothelial carcinoma receiving endoscopic ablation: an inverse probability of treatment weighting analysis. J Clin Med. (2022) 11:1307. doi: 10.3390/jcm11051307
29. Shenhar C, Veredgorn Y, Bulis S, Aviv T, Darawsha AE, Gilad R, et al. Endoscopic management of low-grade upper tract urothelial carcinoma: characterizing the long-term burden of care in comparison to radical nephroureterectomy. Urology. (2022) 159:152–9. doi: 10.1016/j.urology.2021.06.053
30. Chen SH, Wu CH, Wu R, Mai HC, Lin V. Oncologic and renal function outcomes of segmental ureterectomy versus radical nephroureterectomy for urothelial carcinoma of the ureter. Urological Sci. (2023) 34:80–5. doi: 10.4103/UROS.UROS_54_22
31. Kim D, You D, Jeong IG, Hong JH, Ahn H, Hong B. Kidney sparing surgery in upper tract urothelial carcinoma: paradigm change in surgical treatment for ureter cancer. J Cancer Res Clin Oncol. (2023) 149:13717–25. doi: 10.1007/s00432-023-05207-x
32. Paciotti M, Alkhatib KY, Nguyen DD, Yim K, Lipsitz SR, Mossanen M, et al. Is segmental ureterectomy associated with inferior survival for localized upper-tract urothelial carcinoma of the ureter compared to radical nephroureterectomy? Cancers. (2023) 15:1373. doi: 10.3390/cancers15051373
33. Qiu Y, Zhang XH, Dong ZY, Zhang YF, Zhai QL, Lv SD, et al. Comparable survival benefits of partial ureterectomy to radical nephroureterectomy in non-metastatic ureter carcinoma: A population-matched study. Int Urol Nephrol. (2023) 55:579–88. doi: 10.1007/s11255-022-03429-5
34. Tsujino T, Komura K, Inamoto T, Maenosono R, Hashimoto T, Adachi T, et al. Nephron-sparing ureteroscopic surgery vs. Radical nephroureterectomy: comparable survival-outcomes in upper tract urothelial carcinoma. World J Urol. (2023) 41:3585–91. doi: 10.1007/s00345-023-04687-3
35. Ye Y, Zheng Y, Li J, Miao Q, Lin M, Chen J, et al. Endoscopic excision versus radical nephroureterectomy for non-muscle invasive upper tract urothelial carcinoma: A population-based large cohort study. Heliyon. (2023) 9:e22408. doi: 10.1016/j.heliyon.2023.e22408
36. Lee CU, Lee JH, Lee HW, Chung JH, Song W, Kang M, et al. Analysis of progression after elective distal ureterectomy and effects of salvage radical nephroureterectomy in patients with distal ureteral urothelial carcinoma. Sci Rep. (2024) 14:3497. doi: 10.1038/s41598-024-54232-4
37. Ślusarczyk A, Zapała P, Zapała Ł, Rajwa P, Moschini M, Laukhtina E, et al. Oncologic outcomes of patients treated with kidney-sparing surgery or radical nephroureterectomy for upper urinary tract urothelial cancer: A population-based study. Urologic Oncol. (2024) 42:22.e1–.e11. doi: 10.1016/j.urolonc.2023.09.019
38. Veccia A, Antonelli A, Checcucci E, Falagario U, Carrieri G, Guruli G, et al. Segmental ureterectomy for upper tract urothelial carcinoma: A systematic review and meta-analysis of comparative studies. Clin genitourinary Cancer. (2020) 18:e10–20. doi: 10.1016/j.clgc.2019.10.015
39. Kawada T, Laukhtina E, Quhal F, Yanagisawa T, Rajwa P, Pallauf M, et al. Oncologic and safety outcomes for endoscopic surgery versus radical nephroureterectomy for upper tract urothelial carcinoma: an updated systematic review and meta-analysis. Eur Urol Focus. (2023) 9:236–40. doi: 10.1016/j.euf.2022.11.016
40. Chen YT, Yu CC, Yeh HC, Lee HY, Jiang YH, Lee YK, et al. Endoscopic management versus radical nephroureterectomy for localized upper tract urothelial carcinoma in a high endemic region. Sci Rep. (2021) 11:4040. doi: 10.1038/s41598-021-83495-4
41. Giannarini G, Schumacher MC, Thalmann GN, Bitton A, Fleischmann A, Studer UE. Elective management of transitional cell carcinoma of the distal ureter: can kidney-sparing surgery be advised? BJU Int. (2007) 100:264–8. doi: 10.1111/j.1464-410X.2007.06993.x
42. Netsch C, Gross AJ, Herrmann TRW, Becker B. Current use of thulium lasers in endourology and future perspectives. Archivos espanoles urologia. (2020) 73:682–8.
43. Hendriks N, Baard J, Beerlage HP, Schout BMA, Doherty KSG, Pelger RCM, et al. Survival and long-term effects of kidney-sparing surgery versus radical nephroureterectomy on kidney function in patients with upper urinary tract urothelial carcinoma. Eur Urol Open Sci. (2022) 40:104–11. doi: 10.1016/j.euros.2022.04.007
44. Singla N, Gayed BA, Bagrodia A, Krabbe LM, Palazzi KL, Mirheydar H, et al. Multi-institutional analysis of renal function outcomes following radical nephroureterectomy and partial ureterectomy for upper tract urothelial carcinoma. Urologic Oncol. (2015) 33:268.e1–7. doi: 10.1016/j.urolonc.2015.03.006
Keywords: upper tract urothelial carcinoma, kidney-sparing surgery, segmental urethrectomy, endoscopic management, survival outcomes, renal function, meta-analysis
Citation: Zhou L, Huang C, Sun S, Ning K and Tang S (2025) Kidney sparing surgery versus radical nephroureterectomy in upper tract urothelial carcinoma: a meta-analysis and systematic review. Front. Oncol. 15:1448079. doi: 10.3389/fonc.2025.1448079
Received: 02 August 2024; Accepted: 17 March 2025;
Published: 02 April 2025.
Edited by:
Riccardo Tellini, Careggi University Hospital, ItalyReviewed by:
Arcangelo Sebastianelli, University of Florence, ItalyCopyright © 2025 Zhou, Huang, Sun, Ning and Tang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Leqing Zhou, emhvdWxlcWluZzE2OEBzaW5hLmNvbQ==; Shan Tang, dGFuZ3NoYW4yMDA0MDY4QDE2My5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.