- 1Key Laboratory of Medical Science and Laboratory Medicine of Jiangsu Province, School of Medicine, Jiangsu University, Zhenjiang, China
- 2Department of Laboratory Medicine, Affiliated Tumor Hospital of Nantong University, Nantong, China
- 3Department of Laboratory Medicine, The Affiliated People’s Hospital, Jiangsu University, Zhenjiang, China
- 4Department of Surgery, The Affiliated Hospital of Jiangsu University, Jiangsu University, Zhenjiang, China
- 5Department of Oncology, Jiangsu Cancer Hospital, Jiangsu Institute of Cancer Research, Nanjing Medical University Affiliated Cancer Hospital, Nanjing, China
- 6Department of Clinical Laboratory, Affiliated Kunshan Hospital of Jiangsu University, Suzhou, China
- 7Department of Clinical Laboratory, Maternal and Child Health Care Hospital of Kunshan, Suzhou, China
A Corrigendum on
Exosomal CD44 transmits lymph node metastatic capacity between gastric cancer cells via YAP-CPT1A-mediated FAO reprogramming
By Wang M, Yu W, Cao X, Gu H, Huang J, Wu C, Wang L, Sha X, Shen B, Wang T, Yao Y, Zhu W and Huang F (2022). Front. Oncol. 12:860175. doi: 10.3389/fonc.2022.860175
In the published article, there were errors in Figure 1E, Figure 3C and Figures 4L–M as published. Due to carelessness during the creation of the figures, images were pasted incorrectly. We found that Figures 1E (invasion, HGC-27) and 4L (migration, HGC-27-L ex si-CD44) do not correspond to the original data. Besides, the cell counts in Figure 4M do not match the observations in Figure 4L. For Figure 3C, the original data were lost, so independent repeat experiments were conducted, and the results were consistent with the initial findings. Thus, Figures 3C, D were updated.
The corrected Figure 1 and its caption appear below.
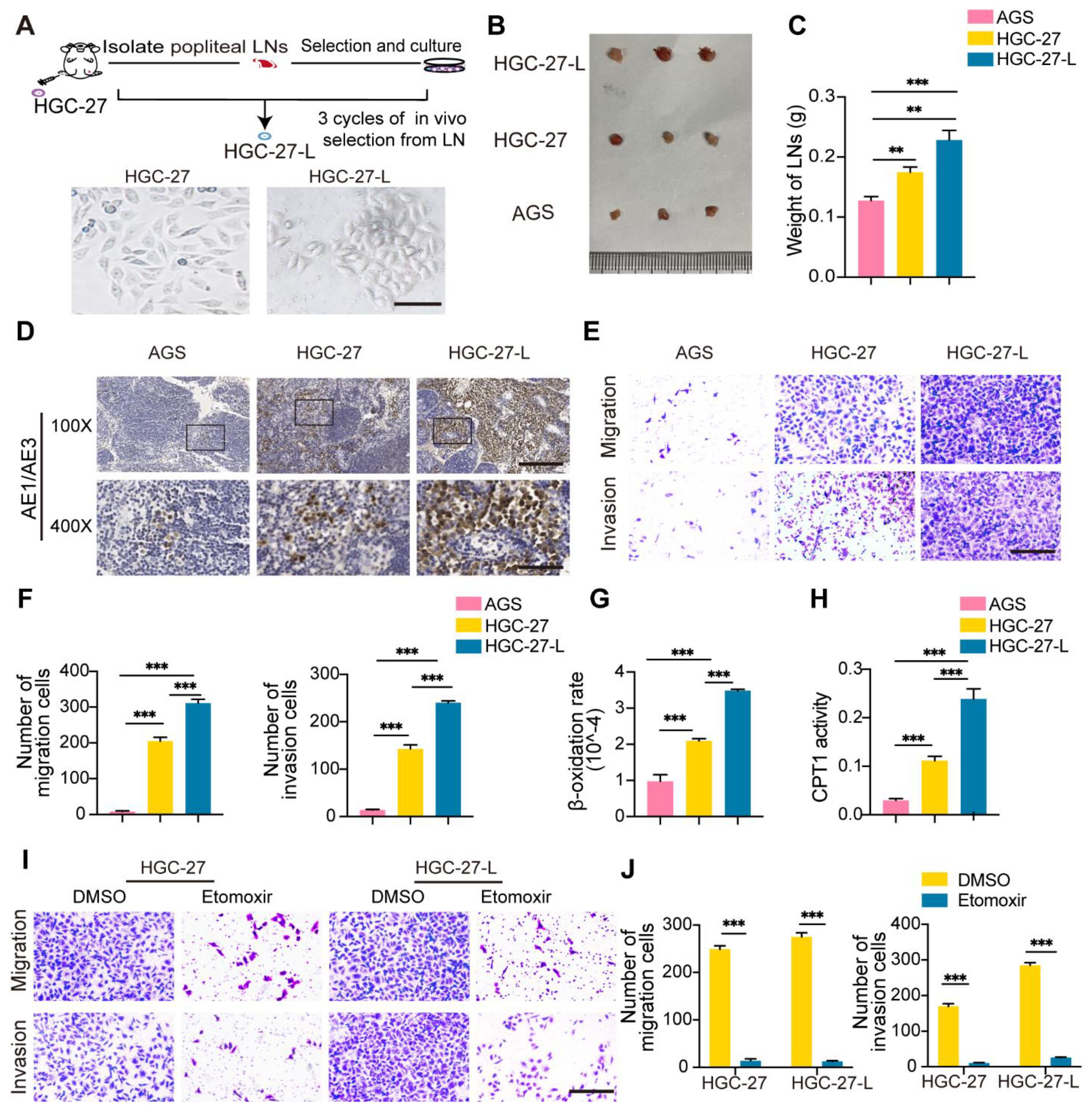
Figure 1. Establishment of a highly lymphatic metastatic GC cell line HGC-27-L and lymphatic metastatic GC cells sustained LNM capacity depending on FAO (A) A flow chart illustrates the establishment of HGC-27-L by serial transplantation of parental cell HGC-27 in vivo. Images of cell morphology are presented. (magnification, 400×; scale bars, 50 μm); (B–D) In vivo comparison of LNM capacity among AGS, HGC-27 and HGC-27-L by popliteal LNs analysis. (B) Pictures of LNs. Ruler unit, mm; (C) Weight of LNs; (D) Pancytokeratin AE1/AE3 staining (magnification, 100×; scale bars, 200 μm; magnification, 400×; scale bars, 50 μm); (E, F) In vitro comparison of migration and invasion capacity among the three cell lines. (E) Morphology of migrated and invaded cells (magnification, 200×; scale bars, 100 μm); (F) Count of migrated and invaded cells; (G) β-oxidation rate detection; (H) CPT1 activity analysis; (I, J) Effect of etomoxir treatment on migration and invasion capacity of HGC-27 and HGC-27-L cells. (I) Morphology of migrated and invaded cells (magnification, 200×; scale bars, 100 μm); (J) Number of migrated and invaded cells. **P < 0.01; ***P < 0.001.
The corrected Figure 3 and its caption appear below.

Figure 3. Lymphatic metastatic GC cell-exosomes conferred LNM capacity on primary GC cells depending on FAO (A, B) FAO detection in AGS after treatment with different GC cell exosomes. (A) β-oxidation rate measurement; (B) CPT1 activity analysis; (C–G) Effect of etomoxir pretreatment on lymphatic metastatic GC cell-exosome increasing AGS LNM capacity. (C, D) In vitro migration and invasion assay. (C) Representative images of migrated and invaded cells (magnification, 200×; scale bars, 100 μm); (D) Number of migrated and invaded cells; (E–G) In vivo LNM capacity detection. (E) Images of popliteal LNs. Ruler unit, mm; (F) Weight of LNs; (G) Pancytokeratin AE1/AE3 staining in LNs (magnification, 100×; scale bars, 200 μm; magnification, 400×; scale bars, 50 μm); ex, exosomes. **P < 0.01; ***P < 0.001.
The corrected Figure 4 and its caption appear below.
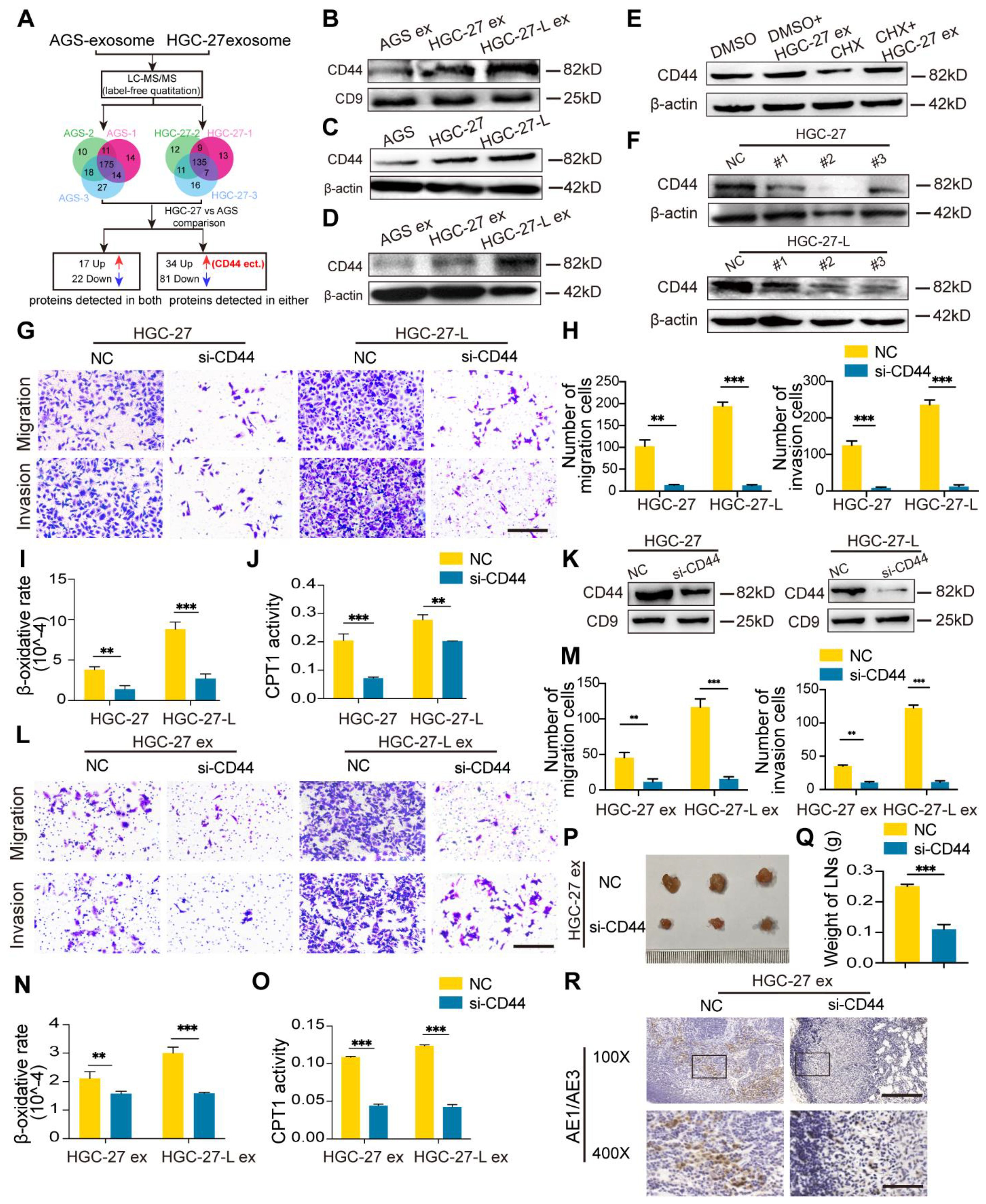
Figure 4. Identification of CD44 was a critical protein mediating exosome- transmission of LNM phenotype (A) A flow chart shows the identification of differential protein profile between HGC-27-exosomes and AGS-exosomes by label-free quantitation; (B) CD44 protein detection in different GC cell-exosomes; (C) Comparison of CD44 protein in GC cells; (D) Change of CD44 protein levels in AGS after treatment with exosomes; (E) Effect of CHX pretreatment on CD44 protein in AGS incubation with HGC-27-exosomes; (F) Screening for the most efficient si-CD44 in HGC-27 and HGC-27-L; (G–J) Effect of CD44 knockdown on the migration and invasion capacity (G, H) and FAO (I, J) of HGC-27 and HGC-27-L; (K) CD44 protein detection in exosomes derived from HGC-27 and HGC-27-L with CD44 knockdown; (L–O) Effect of CD44-less exosomes on migration and invasion capacity (L, M) and FAO (N, O) of AGS; (P–R) Effect of CD44-less exosomes on LNM capacity of AGS in vivo. Ruler unit, mm. CHX, cycloheximide; ex, exosomes. *P < 0.05; **P < 0.01; ***P < 0.001.
The authors apologize for these errors and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Keywords: exosomes, lymph node metastasis, gastric cancer, CD44, fatty acid oxidation, yes-associated protein (YAP), carnitine palmitoyltransferase 1A (CPT1A)
Citation: Wang M, Yu W, Cao X, Gu H, Huang J, Wu C, Wang L, Sha X, Shen B, Wang T, Yao Y, Zhu W and Huang F (2024) Corrigendum: Exosomal CD44 transmits lymph node metastatic capacity between gastric cancer cells via YAP-CPT1A-mediated FAO reprogramming. Front. Oncol. 14:1495349. doi: 10.3389/fonc.2024.1495349
Received: 12 September 2024; Accepted: 17 September 2024;
Published: 11 October 2024.
Edited and Reviewed by:
João Pessoa, University of Aveiro, PortugalCopyright © 2024 Wang, Yu, Cao, Gu, Huang, Wu, Wang, Sha, Shen, Wang, Yao, Zhu and Huang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mei Wang, d2FuZ21laTg0MTdAMTYzLmNvbQ==; Feng Huang, aHVhbmdmZW5na3NybXl5QDE2My5jb20=
†ORCID: Mei Wang, orcid.org/0000-0003-4483-7286
Feng Huang, orcid.org/0000-0002-0348-4296
 Mei Wang
Mei Wang Wanjun Yu
Wanjun Yu Xiaoli Cao2
Xiaoli Cao2 Yongliang Yao
Yongliang Yao Wei Zhu
Wei Zhu