- 1Department of Hematology, Institute of Hematology, West China Hospital of Sichuan University, Chengdu, China
- 2Department of Neurology, West China Hospital of Sichuan University, Chengdu, China
- 3Department of Respiratory and Critical Care Medicine, West China Hospital, Sichuan University, Chengdu, China
- 4Department of Thoracic Surgery, West China Hospital, Sichuan University, Chengdu, China
Lung cancer remains the leading cause of mortality worldwide. Non-small cell lung cancer (NSCLC) is the most common subtype of lung cancer with a generally poor prognosis. In recent years, advances in targeted therapy and sequencing technology have brought significant improvement in the therapeutic outcomes of patients with advanced NSCLC. Targeted inhibitors directed against specific mutated or rearranged oncogenes, such as epidermal growth factor receptor (EGFR), anaplastic lymphoma kinase (ALK), and receptor tyrosine kinase ROS proto-oncogene 1(ROS1) among others, exhibit promising anti-tumor activity. Unfortunately, some patients develop acquired resistance and disease progression soon after initial remission. Despite the continuous development of new drugs and strategies to overcome drug resistance, it is still a major challenge in the treatment of NSCLC. The landscape of targeted therapy for NSCLC is evolving rapidly in response to the pace of scientific research. This study aimed to provide a comprehensive review of tumor target antigens and agents related to targeted therapy in NSCLC.
1 Introduction
In recent years, targeted therapy has achieved significant success in advanced non-small cell lung cancer (NSCLC). Patients with metastatic lung cancer who qualify for targeted therapies now experience prolonged survival, with 5-year survival rates ranging from 15% to 60%, contingent on the specific biomarker identified (1–4). Consequently, molecular and immune biomarker testing of lung cancer specimens is crucial to identifying potentially effective targeted treatments, especially in patients with metastatic NSCLC (3–7). It aims to alleviate symptoms, decrease tumor burden, and improve overall survival (OS).
Classic actionable biomarkers included various genetic alterations that are the targets of several tyrosine kinase inhibitors (TKI) such as anaplastic lymphoma kinase (ALK) rearrangement, V-RAF mouse sarcoma virus oncogene homolog B1 (BRAF) p.V600E mutation, epidermal growth factor receptor (EGFR) mutation, erb-b2 receptor tyrosine kinase 2 (ERBB2, also known as human epidermal growth factor receptor 2, HER2) mutation, Kirsten rat sarcoma virus (KRAS) mutation, mesenchymal-epithelial transition factor (MET) exon 14 (METex14) skipping mutation, neurotrophic tyrosine receptor kinase 1/2/3 (NTRK1/2/3) gene fusion, rearranged in transfection (RET) rearrangement, receptor tyrosine kinase ROS proto-oncogene 1(ROS1) rearrangement, and high-level MET amplification. These gene alterations typically occur in a non-overlapping manner. However, 1%–3% of patients may have coexistence of more than one of these biomarkers (8).
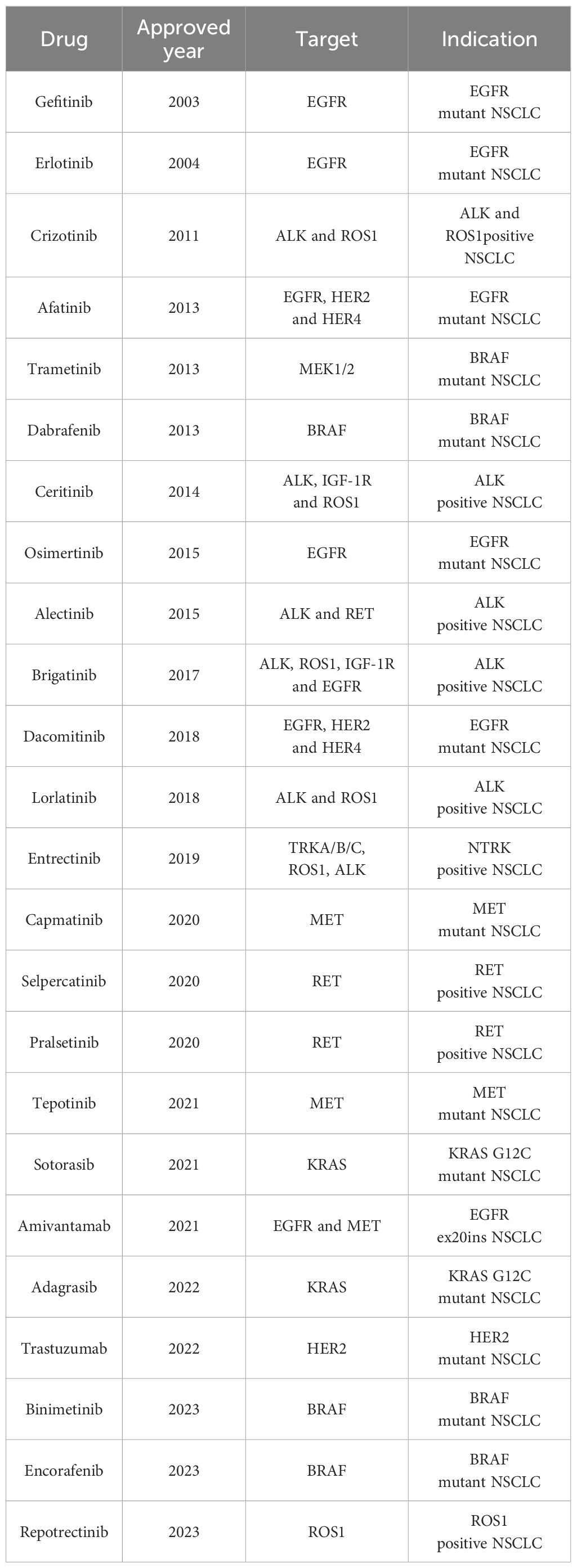
Here we summarize the essential therapeutic targets and targeted drugs for NSCLC (Table 1) and provide insights into the treatment response and resistance mechanisms associated with targeted therapies.
2 Biomarkers and target therapies
2.1 EGFR inhibitors
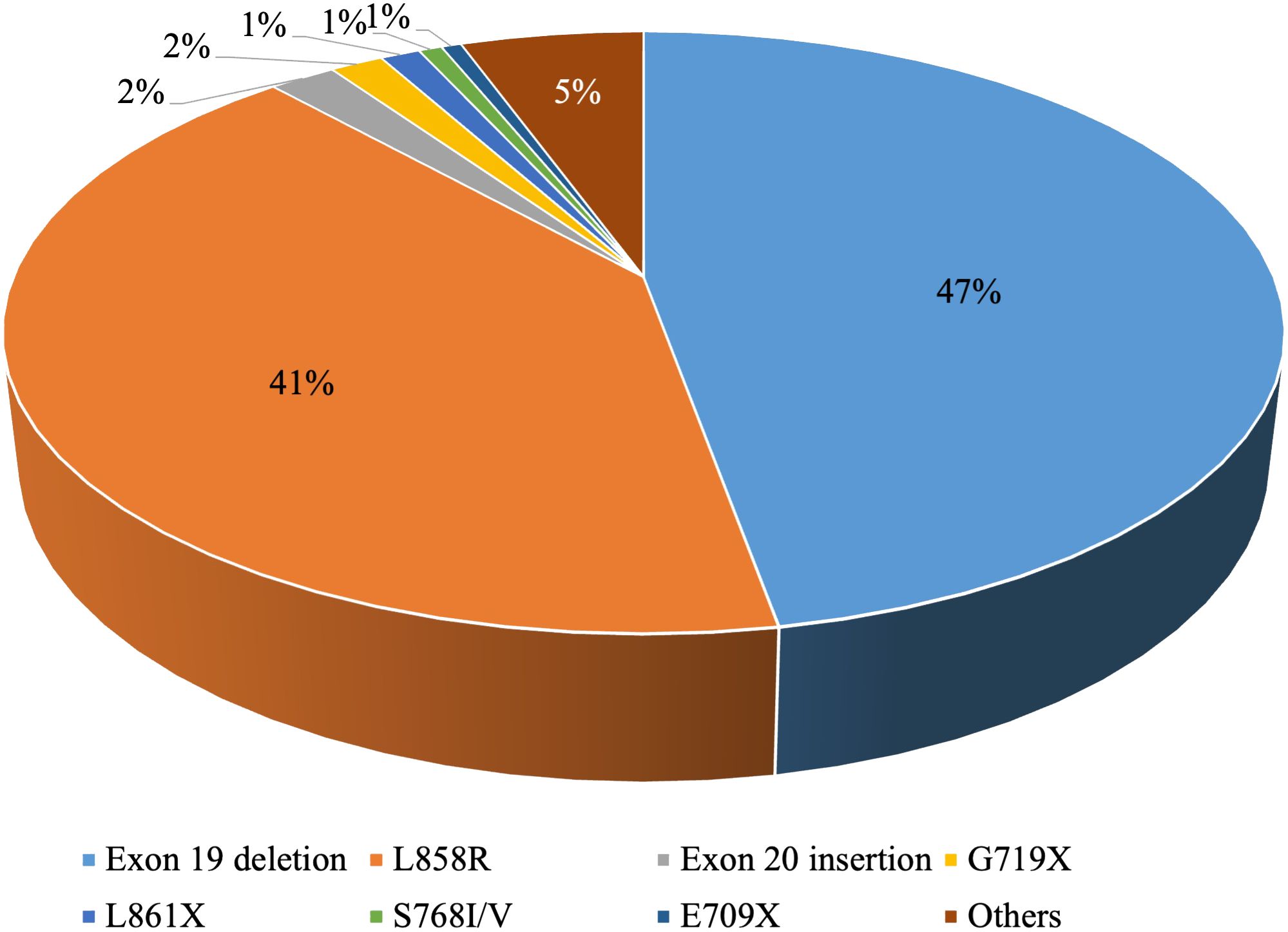
EGFR is the most common driver gene in NSCLC. The mutation frequency is approximately 10-15% in Western Europe and North America and can be as high as 30%-50% in East Asia (9, 10). Common EGFR mutations involve exon 19 deletions and the exon 21 mutation p.L858R, while less frequent mutations include p.S768I/V, p.L861X, and p.G719X (11) (Figure 1).
2.1.1 First-generation medications
Gefitinib and erlotinib were both reversible inhibitors of the first-generation EGFR TKIs. They can selectively and reversibly prevent ATP binding, thereby inhibiting EGFR autophosphorylation (12). An analysis of five clinical studies in which erlotinib or gefitinib was used as first-line treatment in NSCLC (stage IIIB or IV) revealed that the response rate was 67% in patients with sensitizing EGFR mutations (13).
2.1.1.1 Erlotinib
Erlotinib has shown better efficacy than conventional chemotherapy in advanced NSCLC patients with EGFR mutations in multiple randomized phase III trials. In the EURTAC trial, patients receiving erlotinib demonstrated a response rate of 58% with a median PFS of 9.7 months, whereas those receiving conventional chemotherapy exhibited a response rate of 15% with a median PFS of 5.2 months (14). In the trial CALGB30406, erlotinib monotherapy achieved an impressive response rate of 70% (15). Another phase III trial reported a higher objective response rate in the gefitinib group compared to the chemotherapy group (73.7% vs. 30.7%) (16).
2.1.1.2 Gefitinib
The phase III randomized trial IPASS evaluated the efficacy of gefitinib in previously untreated NSCLC patients in East Asia, patients treated with gefitinib exhibited a significantly high objective response rate of 71.2% compared to those treated with carboplatin–paclitaxel (17). The OPTIMAL trial also reported a superior response rate in the gefitinib group compared to the chemotherapy group (83% vs. 36%) (18). The phase III randomized trial WJOG5108L reported similar response rates for gefitinib and erlotinib at 55.0% and 58.9%, respectively (19).
2.1.2 Second-generation medications
2.1.2.1 Afatinib
Afatinib, a second-generation oral TKI, exerts irreversible inhibition targeting the ErbB/HER receptor family including EGFR and HER2 (20). In a phase IIB trial comparing afatinib and gefitinib for first-line treatment in common EGFR mutation metastatic adenocarcinoma patients, afatinib demonstrated a significantly higher objective tumor response rate compared to gefitinib (70% vs. 56%) (21). Updated results revealed no significant difference in OS between the two groups (22). A subgroup analysis of several LUX-LUNG trials (LUX-LUNG 2, 3, and 6) evaluated the efficacy of afatinib in patients with mutation-positive metastatic NSCLC. The response rate was 77.8% in patients with EGFR p.G719X mutation, 100% in p.S768I, and 56.3% in p.L861Q (23). Notably, these findings should be interpreted cautiously as treatment crossover occurred in most patients (72% in LUX-LUNG 3 and 80% in LUX-LUNG 6).
2.1.2.2 Dacomitinib
Dacomitinib is a second-generation oral TKI, that exerts irreversible inhibition on ErbB/HER receptors, including EGFR, HER1, HER2, and HER4.In the phase III randomized trial ARCHER1050, patients receiving dacomitinib as first-line treatment exhibited an objective response rate of 75% (24). Subsequent updated data indicated that dacomitinib-treated patients experienced longer progression-free survival (PFS) (14.7 months vs. 9.2 months) and OS (34.1 months vs. 27 months) compared to the gefitinib group (25, 26).
2.1.3 Third-generation medications
Approximately 60% of patients who progressed on first- and second-generation EGFR TKI treatment harbor EGFR p.T790M mutation. The third-generation EGFR TKIs were originally designed to overcome the resistance caused by acquired EGFR p.T790M mutation. Osimertinib, an oral and irreversible TKI, exhibits selectivity for both common EGFR mutations and p.T790M mutation, with activity within the central nervous system (CNS) (27–29). Osimertinib is the first third-generation EGFR-TKI approved by the Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for metastatic NSCLC patients with EGFR p.T790M mutation (30).
In a phase III randomized trial (AURA3), involving patients with EGFR p.T790M-positive metastatic NSCLC progressing after first-line treatment, the objective response rate was significantly superior with osimertinib (71%) compared to chemotherapy (31%). Osimertinib also exhibited a longer PFS (10.1 vs. 4.4 months). Notably, in the subgroup of patients with CNS metastases, osimertinib presented a prolonged PFS compared to those treated with platinum–pemetrexed (8.5 vs. 4.2 months) (31). The BLOOM study which increased the standard dose of osimertinib from 80 mg once daily to 160 mg once daily, have proved beneficial of the higher dose of osimertinib for patients with leptomeningeal disease progression with EGFR mutations, irrespective of p.T790M status, with an objective response rate of 62% (32, 33).
In addition to the second- or third-line use to overcome resistance of first- and second-generation EGFR TKI treatment, osimertinib has been used as first-line to treat EGFR mutant NSCLC patients. A Multicenter, Phase II Trial (KCSG-LU15-09) demonstrated an objective response rate of 50% for osimertinib as first-line treatment in 37 patients with EGFR rare mutations, including p.S768I, p.L861Q, and p.G719X (34). The phase III randomized trial (FLAURA) also proved a longer median OS with osimertinib as first-line treatment than with erlotinib or gefitinib (38.6 months vs. 31.8 months), though the objective response rate was comparable (80% vs 76%) (35, 36).
2.1.4 Other medications
2.1.4.1 Amivantamab
Amivantamab is a bispecific human antibody to both EGFR and MET receptors that bypasses resistance to EGFR TKIs (37). CHRYSALIS study, a phase I study, evaluated the efficacy of Amivantamab-vmjw as a subsequent treatment in 81 metastatic NSCLC patients with EGFR exon 20 insertion. The overall response rate reported in this cohort was 40% (37). In a phase III study (PAPILLON), amivantamab-chemotherapy significantly improved PFS of patients with EGFR exon 20 insertions who had not received previous systemic therapy when compared to chemotherapy alone (median, 11.4 months and 6.7 months, respectively) (38). And MARIPOSA evaluated the therapeutic efficacy of Amivantamab plus carboplatin-pemetrexed (chemotherapy) with and without Lazertinib in patients with EGFR-mutated (exon 19 deletions or L858R) locally advanced or metastatic NSCLC after disease progression on Osimertinib. The median PFS was significantly longer for amivantamab-chemotherapy and amivantamab-lazertinib-chemotherapy versus chemotherapy (6.3 and 8.3 versus 4.2 months, respectively) (39).
2.1.4.2 Mobocertinib
Mobocertinib is an oral TKI selectively inhibiting EGFR and HER2 exon 20 insertion mutations (40, 41). A phase I/II study evaluated the efficacy of mobocertinib as a subsequent treatment in patients with EGFR exon 20 insertion mutation. The objective response rate was 28%, with a median duration of response of 17.5 months and a median PFS of 7.3 months (40). Subsequently, mobocertinib received FDA accelerated approval for advanced or metastatic NSCLC in adults with EGFR exon 20 insertion mutations who progressed during or after platinum-based chemotherapy.
However, results from the phase III trial, EXCLAIM-2, indicated that the objective response rates and disease control rates between the mobocertinib and chemotherapy groups are similar (response rate: 32% vs. 30%, control rate: 87% vs. 80%) (42). As a result, the FDA and Takeda withdrew mobocertinib in America in October 2023, as it did not meet the primary endpoint of the study.
2.1.4.3 Cetuximab
Cetuximab is a monoclonal antibody to EGFR. In a large phase III randomized trial, FLEX, the combination of chemotherapy and cetuximab proved higher overall response rates than chemotherapy alone (36% vs. 29%) and comparable median OS (11.3 vs. 10.1 months) (43). However, this combination exhibited poorer tolerability considering the nearly 40% incidence of grade 4 neutropenia. Therefore, the use of cetuximab is not yet recommended in NSCLC.
2.2 ALK inhibitors
ALK gene rearrangements occurred in approximately 3-5% of NSCLC patients (44). So far, more than 19 distinct ALK fusion partners have been identified in NSCLC, including EML4, KIF5B, KLC1, and TPR (45). The most common fusion was EML4::ALK, existing in about 85% of ALK-rearrangement NSCLC.
2.2.1 First-generation medications
2.2.1.1 Crizotinib
Crizotinib is a first-generation oral TKI and the first TKI approved for treating ALK-positive NSCLC, effectively inhibits ALK rearrangements, ROS1 rearrangements, high-level MET amplification, and METex14 skipping mutations. In phase I and II studies, crizotinib demonstrated objective tumor responses in approximately 60% of ALK-positive NSCLC patients, with a median PFS ranging from 7 to 10 months (46–48). A phase III randomized study, PROFILE 1014, assessing the efficacy of crizotinib as first-line targeted therapy, yielded promising results with an objective response rate of 74% (49). For ALK-positive patients progressing after first-line chemotherapy, crizotinib has shown efficacy in improving PFS (7.7 months) and enhancing response rates (65%) (50).
2.2.2 Second-generation medications
2.2.2.1 Alectinib
Alectinib is a selective second-generation oral ALK inhibitor with high CNS penetration. It has demonstrated activity against several secondary mutations associated with acquired resistance to crizotinib, such as p.T1151L, p.1152insT, p.L1196M, p.C1156Y, p.F1174L, and p.G1269A (51, 52).
The ALEX trial, a phase III randomized study, compared the efficacy of alectinib and crizotinib as first-line treatments in 303 ALK-positive advanced NSCLC patients, including those with asymptomatic brain metastases. The response rate in the alectinib group was 82.9% and 75.5% in the crizotinib group (53). Another phase III trial, J-ALEX, enrolled 207 ALK inhibitor-naive Japanese patients with ALK-positive NSCLC, also proved that alectinib as a first-line treatment achieved a higher objective response rate compared to crizotinib (92% vs. 79%) (54).
Efficacy of alectinib as subsequent treatments was reported by phase II trials with a total response rate of 48% to 50% in metastatic NSCLC patients with ALK rearrangement progressing after crizotinib treatment (55, 56).
2.2.2.2 Brigatinib
Brigatinib is a second-generation TKI that inhibits a broad spectrum of ALK rearrangements. As first-line treatment, brigatinib was reported a higher systemic objective response rate of 71% than crizotinib (60%) in the ALTA-1L trial. The intracranial response rate was also notably higher with brigatinib (78%) compared to crizotinib (29%) (57). Updated data further confirmed that the 3-year PFS in the brigatinib group was superior to crizotinib (43% vs. 19%) (58).
A phase II study, ALTA, evaluated the efficacy of two different doses of brigatinib in ALK-positive metastatic NSCLC patients who had experienced disease progression on or intolerance to crizotinib. The overall response rate ranged from 45% to 54%. In patients with measurable brain metastases, the intracranial overall response rate was observed to be between 42% and 67% (59, 60).
2.2.2.3 Ceritinib
Ceritinib is a second-generation oral TKI designed for ALK and ROS1 rearrangements (61), showing promising results in various clinical trials. In the ASCEND-4 trial, the overall response to ceritinib as first-line therapy was 72·5% with a median PFS of 16.6 months, as compared with 26·7% with a median PFS of 8.1 months in the chemotherapy group (62).
As subsequent treatment in patients with prior exposure to at least two treatments, ceritinib was reported an overall response rate of 38.6%, with a concurrent intracranial response rate of 45.0% in a phase II study (ASCEND-2) (63), and a higher overall response rate of 45% than pemetrexed or docetaxel chemotherapy (8%) (64).
2.2.3 Third-generation medications
Lorlatinib, a third-generation oral TKI with excellent CNS penetration, selectively inhibits ALK and ROS1. It exhibits the ability to inhibit ALK resistance mutations that emerge following treatment with first and second-generation ALK inhibitors (65–68). In the phase III randomized trial, CROWN, lorlatinib demonstrated complete CNS responses in 61% of patients with baseline brain metastases, compared to only 15% with crizotinib (69). Updated data reveals a lower cumulative CNS progression rate with lorlatinib (7%) than crizotinib (72%) over 12 months, and higher 1-year PFS rates of 78% than 22% for crizotinib in patients with brain metastases (70).
Lorlatinib also remains effective for patients experiencing progression after treatment of other ALK inhibitors, especially those with CNS involvement. Among patients with measurable baseline CNS lesions, 47% achieved objective responses, and 63% achieved an objective intracranial response (66, 67).
2.3 BRAF inhibitors
BRAF mutations manifest in 1%–5% of NSCLC patients (71–74). The most common mutation is p.V600E, accounting for approximately 50% of BRAF-mutated cases (75). Other BRAF mutations include p.D594G and p.G469A/V, observed in 35% and 6% of BRAF-mutated NSCLC patients, respectively (74). For NSCLC patients with p.V600E mutation, the FDA has currently approved two combinations of RAF and MEK inhibitors: dabrafenib/trametinib and encorafenib/binimetinib.
2.3.1 Dabrafenib/trametinib
In a phase II trial, dabrafenib/trametinib as first-line treatment demonstrated a robust overall response rate of 64% in 36 patients with BRAF p.V600E mutation (76). An updated analysis of this trial revealed a 5-year OS rate of 22% (1). Another dual-cohort phase II study conducted a comparative analysis between patients receiving dabrafenib monotherapy and combination therapy with dabrafenib and trametinib. The results indicated a distinct overall response rate of 33% and 67%, and median PFS durations of 5.5 months and 10.2 months, respectively (77).
2.3.2 Encorafenib/binimetinib
In the PHAROS trial, an open-label, multicenter, single-arm study, an impressive overall response rate of 75% was observed among the 59 treatment-naive patients with BRAF p.V600E mutation, with a median duration of response not achieved. In the cohort of 39 previously treated patients, the overall response rate was 46%, and the median duration of response was 16.7 months (78).
2.4 ERBB2 (HER2) inhibitors
2.4.1 Ado-trastuzumab emtansine
Ado-Trastuzumab Emtansine, also known as T-DM1, is a humanized antibody-drug conjugate comprising the HER2-targeting antibody trastuzumab and the microtubule inhibitor emtansine (79). In a phase II basket trial, the efficacy of ado-trastuzumab emtansine was assessed in patients with metastatic NSCLC and HER2 mutations, revealing a partial response rate of 44% (79, 80). Another study focused on patients with HER2 exon20 insertion mutations, indicating an objective response rate of 38% with ado-trastuzumab emtansine (81). These findings underscore the potential of ado-trastuzumab emtansine as a targeted therapeutic option for patients with HER2-mutated NSCLC.
2.4.2 Fam-trastuzumab deruxtecan-nxki
Fam-trastuzumab deruxtecan-nxki, a humanized monoclonal antibody-drug conjugate comprising trastuzumab linked to deruxtecan, is a topoisomerase I inhibitor (82). A phase I trial investigated the efficacy of fam-trastuzumab deruxtecan-nxki in HER2-mutant NSCLC patients, representing an objective response rate of 72.7% (83). The DESTINY-Lung01, a phase II study, revealed an objective response rate of 55% in 91 patients treated with fam-trastuzumab deruxtecan-nxki (82).
2.5 KRAS inhibitors
KRAS mutations are identified in approximately 30% of NSCLC patients (84). These mutations are predominantly (>95%) located at codons 12 and 13. The p.G12C variant was the most prevalent, constituting 39% of all KRAS mutations, followed by p.G12V (21%) and p.G12D (17%) variants (85). Sotorasib and adagrasib are both an oral inhibitor to the RAS GTPase family, demonstrating efficacy in inhibiting the KRAS p.G12C mutation in patients with metastatic NSCLC who have previously undergone chemotherapy (± immunotherapy).
2.5.1 Sotorasib
Sotorasib, as a small-molecule inhibitor, irreversibly binds to the non-active GDP pocket of KRAS, forming an irreversible covalent bond with the cysteine residue in KRAS p.G12C. This covalent interaction locks the protein in an inactive state. By disrupting the KRAS signaling pathway, sotorasib inhibits cell growth as well as tumor progression both in vitro and in vivo and induces apoptosis in KRAS p.G12C tumor cell lines (86, 87).
In a phase II study involving 126 patients with KRAS p.G12C-positive advanced NSCLC who had prior platinum-based chemotherapy (with or without immunotherapy), subsequent treatment with sotorasib showed a partial response rate of 33.9% and complete response rate of 4.2% (88). The phase III randomized study, CodeBreaK200 trial, has also reported the efficacy of sotorasib in patients in a similar situation (89). Sotorasib demonstrated a significantly higher overall response rate of 28.1% than docetaxel (13.2%). Moreover, the disease-control rate in the sotorasib group was 82.5%, compared to 60.3% in the docetaxel group.
2.5.2 Adagrasib
In a phase II study involving 116 patients who had previously undergone platinum-based chemotherapy with or without immunotherapy, adagrasib demonstrated an objective response rate of 42.9%. The efficacy of adagrasib in cases with KRAS mutations beyond p.G12C remains to be systematically evaluated (90).
2.6 MET inhibitors
The oncogenic driver genomic alterations associated with MET comprise METex14 skipping mutations and high-level MET amplification. High-level MET amplification was recently identified as an emerging biomarker. Its definition may vary depending on the reagent kits. When employing Next-Generation Sequencing (NGS), high-level MET amplification is defined as the copy number greater than 10 (91). The FDA has not yet approved recommended drugs for NSCLC patients carrying these mutations, despite their approval in other tumor types.
2.6.1 Capmatinib
Capmatinib is an oral TKI selectively targeting MET alterations. The GEOMETRY trial revealed that capmatinib achieved an overall response rate of 68% as a first-line treatment, and 41% as subsequent treatment in patients with METex14 skipping mutations. While in patients with high-level MET amplification, the response rate was 40% as the first-line therapy, and 29% as subsequent therapy (91). Notably, the updated data of GEOMETRY indicate that capmatinib exhibits anti-tumor efficacy within the brain (92). Another study revealed an overall response rate of 50% in a cohort of 10 patients with high-level MET amplification (93).
2.6.2 Crizotinib
Crizotinib is an oral TKI that inhibits METex14 skipping mutation and high-level MET amplification. A phase II study evaluated the efficacy of crizotinib in 69 patients with METex14 skipping mutations. The objective response rate was 32%, with a median PFS of 7.3 months (94). The PROFILE1001 study investigated the efficacy of crizotinib in advanced NSCLC patients with varying levels of MET amplification. Patients with MET genomic copy number over 10 demonstrated an overall response rate of 29% (95).
2.6.3 Tepotinib
Tepotinib is a selective oral TKI that inhibits METex14 skipping mutation and high-level MET amplification. A phase II study (VISION) assessed the efficacy of tepotinib in patients with MET mutations. The response rate in patients with METex14 skipping mutations was 46%. Another cohort comprising 24 patients with MET amplification but lacking METex14 skipping mutations exhibited an overall response rate of 41.7% (96, 97).
2.7 NTRK1/2/3 inhibitors
NTRK1/2/3 gene fusions encode TRK fusion proteins, serving as oncogenic drivers in multiple solid tumors, including lung, thyroid, salivary gland, and sarcoma (98). Entrectinib and larotrectinib are both inhibitors of TRK fusion proteins in unresectable or metastatic solid tumors.
2.7.1 Entrectinib
The efficacy of entrectinib was evaluated in three phase I or II trials (STARTRK-2, STARTRK-1, ALKA-372-001). A pooled analysis revealed an overall response rate of 70% in 10 NTRK gene fusion-positive NSCLC patients treated with entrectinib (99–101).
2.7.2 Larotrectinib
A study comprising 55 patients with various solid tumors and positive NTRK gene fusions revealed an overall response rate of 75% with larotrectinib (98). The updated data demonstrated that 90% of patients still remained alive one year after treatment. Furthermore, among 35 NTRK fusion cancer patients, the overall response rate reached 74% (102).
2.8 RET inhibitors
The RET gene is observed in 1-2% of all NSCLC patients with chromosomal rearrangements and is involved in various fusion partners such as KIF5B, TRIM33, CCDC6, and NCOA4 (103, 104).
2.8.1 Pralsetinib
In a phase I/II study (ARROW), pralsetinib was assessed in metastatic NSCLC patients with RET rearrangements. The overall response rate of pralsetinib was 70% as a first-line treatment, and 61% as a subsequent treatment reached 61% (105). The FDA approved pralsetinib in 2020 for the treatment of metastatic RET fusion-positive NSCLC patients. It is the first oral TKI targeting RET fusions (106).
2.8.2 Selpercatinib
A phase I/II study, Libretto-001, along with its updated results, reveals that selpercatinib exhibits remarkable efficacy in NSCLC patients with RET rearrangements. The overall response rate for first-line treatment was 85%, while 64% for subsequent treatment. Notably, in patients with brain metastases, selpercatinib demonstrated effectiveness in 91% of cases (107, 108).
2.8.3 Cabozantinib
In a prospective phase II trial involving 26 RET fusion-positive patients treated with cabozantinib, the overall response rate was 28% (109, 110).
2.9 ROS1 inhibitors
2.9.1 Crizotinib
Crizotinib is a multitargeted inhibitor targeting MET, ALK, and ROS1. In an early-phase study, crizotinib demonstrated considerable efficacy in ROS1-rearranged NSCLC (111). The objective response rate in the expansion cohort treated with crizotinib reached 72%. The overall response duration was 17.6 months, with a median PFS of 19.2 months (112).
Three phase II studies confirmed an overall response rate of more than 70% with crizotinib in patients with ROS1 rearrangement. A phase II trial evaluating the efficacy of crizotinib in 127 East Asian patients reported an overall response rate of 72% (113). The PROFILE 1001 study and updated data reported an objective response rate of 72% in 53 ROS1-positive advanced NSCLC patients, including 3 complete responses and 33 partial responses (4, 114). The multicenter trial, EUCROSS study, reported a total response rate of 70% in 30 patients treated with crizotinib (115). Additionally, a retrospective study assessing crizotinib in stage IV ROS1-rearranged NSCLC patients (n=30) reported an overall response rate of 80%, with a median PFS of 9.1 months (116).
2.9.2 Lorlatinib
Lorlatinib is an oral third-generation TKI targeting both ALK and ROS1 with significant CNS penetration. It was evaluated in a phase I/II trial for its efficacy in ROS1-positive metastatic NSCLC patients. The objective response rate in patients previously treated with crizotinib reached 35%, while treatment-naive patients demonstrated a 62% objective response rate. Notably, intracranial responses were observed in 50% of patients with prior crizotinib treatment and 64% of treatment-naive patients (65).
2.9.3 Entrectinib
Entrectinib is an oral TKI inhibiting multiple tyrosine kinases, including ROS1 and TRK. A pooled analysis of 53 patients with ROS1 rearrangement across several phase I and II trials (STARTRK-2 trial, STARTRK-1 trial, ALKA-372-001 trial) who received entrectinib as first-line treatment demonstrated an overall response rate of 77%, with a 55% intracranial response rate (100, 101, 117). Although entrectinib exhibits superior CNS penetration compared to crizotinib, it comes with higher toxicity, with an incidence of grade 3 or 4 adverse events of 34% (117).
2.9.4 Ceritinib
Ceritinib is a second-generation oral TKI inhibiting ALK and ROS1 rearrangements. In a phase II trial assessing ceritinib as first-line treatment in ROS1-rearranged NSCLC patients (28 evaluable patients), the reported overall response rate was 62%, with 1 case of complete response and 19 cases of partial responses (61).
2.9.5 Repotrectinib
A phase I/II trial assessed the efficacy and safety of repotrectinib in patients with advanced ROS1 fusion-positive NSCLC. The confirmed overall response rate was 79% among ROS1 TKI-naive patients and 38% among patients previously treated with other ROS1 inhibitors. Notably, responses were observed in intracranial lesions in patients with measurable CNS metastases, as well as in those with resistance mutations following TKI therapy (118).
2.10 VEGF or VEGF receptors inhibitors
2.10.1 Bevacizumab
Bevacizumab is a recombinant monoclonal antibody targeting VEGF. In a phase III randomized trial, ECOG4599, involving relapsed or advanced non-squamous NSCLC, the corresponding response rates were 35% in patients treated with a combination of bevacizumab chemotherapy and 15% in those treated with chemotherapy alone (119). Another phase III trial, NEJ026, compared the efficacy of erlotinib combined with bevacizumab to erlotinib monotherapy as first-line treatments in EGFR-positive advanced non-squamous NSCLC patients. The objective response rates were similar (erlotinib/Ramucirumab: 72% vs. erlotinib monotherapy: 67%) (120).
2.10.2 Ramucirumab
Ramucirumab is a recombinant monoclonal antibody targeting VEGF receptors. In the phase III randomized trial RELAY, first-line treatment with erlotinib/ramucirumab was compared to erlotinib monotherapy in EGFR-mutated advanced NSCLC patients. The overall response rates were similar (erlotinib/ramucirumab: 76% vs. erlotinib monotherapy: 75%) (121). The REVEL trial, a phase III randomized study in metastatic NSCLC patients who experienced disease progression, evaluated the efficacy of ramucirumab/docetaxel compared to docetaxel alone as subsequent therapy. The ramucirumab/docetaxel group exhibited higher overall response rates (23% vs. 14%) and disease control rates (64% vs. 53%) (122).
2.10.3 Nintedanib
Nintedanib is a potent, oral angiokinase inhibitor that targets the pro-angiogenic pathways mediated by VEGFR1-3 (123). In the phase III randomized controlled trial LUME-Lung 1, 1314 stage IIIB/IV patients progressing after first-line chemotherapy were randomly assigned to receive docetaxel plus nintedanib (n=655) or docetaxel plus placebo therapy (n=659). PFS was significantly improved in the nintedanib plus docetaxel group when compared to the docetaxel plus placebo group (median 3.4 months vs. 2.7 months) (PMID: (124)).
3 Resistance to targeted therapy
3.1 Overview of the mechanisms of resistance to targeted therapies
Resistance to targeted therapies is categorized as either primary (intrinsic) or secondary (acquired) (125). Primary resistance describes a de novo lack of therapeutic response, while secondary resistance indicates disease progression after the initial response. Despite distinct resistance mechanisms identified in patients with different gene alterations, there are common mechanisms shared among these cohorts (126). The acquired resistance mechanisms can be broadly classified into two categories.
The first category involves the development of additional genetic alterations in the primary oncogenes, activating continued downstream signaling. This is often attributed to secondary mutations in kinase targets or gene amplifications of the kinase itself (127). The second category of resistance development can occur independently of changes in the target gene. This scenario includes upregulation of bypass signaling pathways, histological changes of tumor tissue, or alterations in drug metabolism (128, 129). Moreover, about 14% of small-cell lung cancer can histologically transform into NSCLC, often accompanied by resistance to the original TKI (130, 131).
In 2010, Jackman et al. proposed the criteria of acquired resistance in EGFR-mutant NSCLC (132): 1) Patients must have previously received EGFR inhibitor treatment. 2) Patients harbor either tumor-genotyping confirmed typical EGFR mutations associated with drug sensitivity, or objective clinical benefit from treatment with an EGFR inhibitor. 3) Patients develop systemic progression while on continuous treatment with gefitinib or erlotinib within the last 30 days. 4) No additional systemic treatment between cessation of EGFR inhibitor and initiation of new therapy.
3.2 Resistance to EGFR inhibitors
3.2.1 Primary resistance
Primary resistance to EGFR inhibitors may be partially attributed to differential TKI sensitivity for different EGFR mutations. Typical EGFR mutations, including exon 19 deletions and p.L858R, are associated with significant sensitivity to TKIs (128). Conversely, exon 20 insertions or duplications, accounting for about 4% of patients with EGFR mutations, appear to have resistance to EGFR inhibitors (133).
3.2.2 Acquired resistance
The earliest report of TKI resistance in EGFR-mutant NSCLC identified a substitution of threonine for methionine at residue 790 (p.T790M) (134). Subsequent reports confirmed that p.T790M is the most common mutation responsible for TKI resistance, which is identified in approximately 60% of patients who experience disease progression after initial response to first-line EGFR TKIs treatment (125, 134–140).
Threonine 790 serves as the “gatekeeper” residue, crucial for inhibitor specificity in the ATP binding pocket. The p.T790M mutation activates wild-type (WT) EGFR, introducing an increase in the ATP affinity of the p.L858R mutant by more than an order of magnitude. This is the main mechanism by which the p.T790M mutation confers TKI resistance, reducing the efficacy of any ATP-competitive kinase inhibitor. Irreversible inhibitors can simply overcome this resistance through covalent binding rather than alternative binding (141). Therefore, in patients with EGFR p.T790M-positive metastatic NSCLC experiencing progression after first-line treatment, osimertinib as an irreversible EGFR-TKI can achieve an objective response rate of over 70% (31).
Other secondary mutations include p.D761Y, p.L747S, and p.T854A. They reduce the sensitivity to EGFR inhibitors, but the resistance mechanism remains unknown (142). In the AURA trial, the acquired p.C797S mutation was observed in 14% of the samples (31). The p.C797S mutation frequency was 7% when osimertinib was used as first-line therapy (35). The EGFR p.C797S mutation, in which cysteine at codon 797 is replaced by serine in the ATP-binding site, results in the loss of the covalent bond between osimertinib and mutated EGFR. Predictably, the p.C797S mutation also leads to cross-resistance by preventing other irreversible third-generation TKIs from binding to the EGFR active site (143–145).
TKI resistance may also activate bypass signaling pathways, such as MET amplification (15-19%), PIK3CA mutations (6-7%), KRAS mutations (3%), and HER2 amplification (2-5%) (146, 147). Bypass pathway activation leads to TKI resistance by sustaining activation of EGFR downstream signaling pathways.
3.3 Resistance to ALK inhibitors
The primary resistance to ALK inhibitors may be due to the different sensitivity of EML4::ALK variants and other ALK fusion genes to ALK inhibitors (148). Acquired resistance to ALK inhibitors typically occurs within the first year of treatment (125). Secondary mutations in the enzyme are the common mechanism of TKI resistance. It is noteworthy that multiple secondary mutations can occur in ALK-positive patients upon TKI resistance. The first “gatekeeper” mutation identified in the EML4::ALK kinase domain is p.L1196M (149). The substitution of leucine for methionine at position 1196 in the ATP binding pocket generates a mutated large amino acid side chain, which hinders crizotinib from binding to its receptor. Other identified acquired resistance point mutations include p.G1128A, p.1151Tins, p.L1152P/R, p.C1156Y, p.I1171T/N/S, p.F1174V, p.V1180L, p.G1202R, p.S1206Y/C, p.E1210K, and p.G1269A (150–155).
Numerous studies suggest that second-generation drugs such as alectinib, ceritinib, brigatinib, and ensatinib may be more effective than chemotherapy when treating NSCLC patients with no response to first-generation ALK inhibitors (64, 156–158). In patients treated with second-generation ALK inhibitors, the p.G1202R mutation is the most common secondary ALK mutation, appearing in 21% of ceritinib-treated patients, 29% of alectinib-treated patients, and 43% of brigatinib-treated patients (159).
A gain in ALK gene fusion copy number (more than two-fold increase) has recently been proposed as a mechanism of resistance to crizotinib in both in vitro and in patients (150, 155). Based on single circulating tumor cell sequencing, another study reported repeated mutations in the RTK-KRAS (EGFR, KRAS, BRAF genes), TP53, and other genes in the ALK-independent pathway in crizotinib-resistant patients (160).
Resistance to ALK inhibitors can also occur through the activation of bypass signaling pathways, including YAP transcription co-regulator, EGFR signaling, KIT amplification, the IGF-1R pathway, MAPK amplification, the BRAF p.V600E mutation, and MET amplification (155, 161–165). MET amplification was observed in 15% of tumor samples from patients progressing after second-generation ALK inhibitors, and in 12% and 22% of tumor biopsy samples from patients progressing on second-generation inhibitors or lorlatinib, respectively (166).
3.4 Resistance to ROS1 inhibitors
Single nucleotide mutations in the ROS1 kinase domain, such as p.D2033N, p.G2032R/K, p.L2026M, p.L2155S, and p.S1986F/Y, have been reported leading to acquired resistance to ROS1 TKIs in ROS1 fusion-positive NSCLC through preclinical and clinical studies (167–171). These mutations diminish the efficacy of kinase inhibitors (112, 168, 172).
A study evaluating biopsies from 55 patients progressing after TKI treatment found that ROS1 mutations were identified in 38% of post-crizotinib biopsies and 46% of post-lorlatinib biopsies. Approximately one-third of patients harbored the most common mutation ROS1 p.G2032R. Additional ROS1 mutations emerged following crizotinib treatment, including p.D2033N (2.4%), p.S1986F (2.4%), p.L2086F (3.6%), p.G2032R/p.L2086F (3.6%), and p.G2032R/p.S1986F/p.L2086F (3.6%). p.S1986F/p.L2000V (3.6%) was detected in 3.6% of patients receiving lorlatinib treatment (170).
The p.D2033N mutation causes the substitution of aspartate for asparagine at position 2033 in the ROS1 kinase hinge region, thus leading to significant resistance to ROS1 inhibitors in vitro (173, 174). The p.L2026M and p.G2032R mutations in the ROS1 kinase domain confer crizotinib resistance by altering the “gatekeeper” position of ROS1 inhibitor binding (168, 175). Additionally, p.S1986F/Y in the kinase domain disrupts crucial activation sites, thereby increasing kinase activity. p.L2155S is anticipated to confer crizotinib resistance through protein dysfunction (176).
The mutations and/or copy number increases of genes in other RTKs or downstream MAPK pathway are also involved in the mechanism of resistance to ROS1 inhibitor (177). Mediators involved in this pathway include KRAS, NRAS, EGFR, HER2, MET, KIT, BRAF, and MEK, either as downstream or bypass mediators (167, 168, 172, 174). KRAS p.G12D and BRAF p.V600E mutations are associated with crizotinib treatment, while NRAS p.Q61K is associated with entrectinib treatment (178).
4 Strategies for overcoming resistance to TKIs
Targeted therapies have significantly improved the prognosis of NSCLC patients with relevant genetic alterations, which is a major progress in the history of NSCLC treatment. However, part of the patients acquires TKI resistance and disease progression shortly after initial remission. Strategies have been investigated to overcoming resistance to TKIs, which include the continuation of TKI therapy beyond disease progression, combination with other TKIs, and the use of immune checkpoint inhibitors.
4.1 Continuation of TKI therapy beyond disease progression
A phase II open-label single-arm trial named ASPIRATION reported that the post-progression erlotinib patients exhibited deeper responses, longer PFS, prolonged time from overall response to progression, and fewer new lung lesions (179). A retrospective analysis of 414 ALK-positive NSCLC patients enrolled in PROFILE 1001 and PROFILE 1005 showed that continuation of crizotinib (>3 weeks) after progression conferred extended progression time and longer OS (180). However, more evidence supports the timely detection of potential resistance mutations and prompt switching to sensitive targeted therapies after disease progression.
4.2 Combination with other TKIs
In a phase Ib/II single-arm trial, 47% of EGFR TKI-resistant NSCLC patients with MET gene amplification and 32% of EGFR TKI-resistant patients with MET overexpression responded to the MET inhibitor capmatinib in combination with EGFR TKI (181). In another phase 1b study of the combination of the MET inhibitors savolitinib and gefitinib, up to 52% of patients with EGFR TKI-resistant NSCLC with MET gene amplification had an objective response to the combination treatment regimen (182). In the subsequent INSIGHT study, 67% of EGFR TKI-resistant NSCLC patients with MET gene amplification had an objective therapeutic response to treatment with the MET inhibitor tepotinib combined with gefitinib (183). And in the phase Ib trial of the TATTON study, 64% of NSCLC patients who were resistant to first- or second-generation EGFR TKIs and had MET gene amplification showed improved response to savolitinib combined with osimertinib. However, only 30% of patients who were resistant to third-generation EGFR TKIs and had MET gene amplification showed an objective response to this combination therapy (184).
4.3 Immune checkpoint inhibitors
In recent years, checkpoint inhibitor antibodies, including programmed cell death protein 1 (PD-1) inhibitors and programmed death ligand 1 (PD-L1) inhibitors, have demonstrated favorable outcomes in NSCLC treatment by blocking the PD-1 and PD-L1 interaction and enhancing the antitumor effects of endogenous T cells. Pembrolizumab, atezolizumab, and cemiplimab have all been reported to prolong PFS and OS in eligible patients (185–189). However, the efficacy of checkpoint inhibitor antibodies depends on the expression level of PD-L1, and for certain mutations such as EGFR exon 19 deletions, EGFR p.L858R mutations, or ALK rearrangements, they appeared to be less effective (190–194).
In conclusion, targeted therapy has brought significant benefits to NSCLC patients, but the emergence of TKI resistance poses a formidable obstacle. The treatment of NSCLC still has a long way to go.
Author contributions
HZ: Writing – original draft, Writing – review & editing. YZhang: Writing – original draft, Writing – review & editing. YZhu: Visualization, Writing – review & editing. TD: Writing – review & editing. ZL: Funding acquisition, Supervision, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported by the Science and Technology Support Program of Sichuan Province (2023NSFSC0732).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Planchard D, Besse B, Groen HJM, Hashemi SMS, Mazieres J, Kim TM, et al. Phase 2 study of dabrafenib plus trametinib in patients with BRAF V600E-mutant metastatic NSCLC: updated 5-year survival rates and genomic analysis. J Thorac Oncol. (2022) 17:103–15. doi: 10.1016/j.jtho.2021.08.011
2. Mok T, Camidge DR, Gadgeel SM, Rosell R, Dziadziuszko R, Kim DW, et al. Updated overall survival and final progression-free survival data for patients with treatment-naive advanced ALK-positive non-small-cell lung cancer in the ALEX study. Ann Oncol. (2020) 31:1056–64. doi: 10.1016/j.annonc.2020.04.478
3. Lin JJ, Cardarella S, Lydon CA, Dahlberg SE, Jackman DM, Jänne PA, et al. Five-year survival in EGFR-mutant metastatic lung adenocarcinoma treated with EGFR-TKIs. J Thorac Oncol. (2016) 11:556–65. doi: 10.1016/j.jtho.2015.12.103
4. Shaw AT, Riely GJ, Bang YJ, Kim DW, Camidge DR, Solomon BJ, et al. Crizotinib in ROS1-rearranged advanced non-small-cell lung cancer (NSCLC): updated results, including overall survival, from PROFILE 1001. Ann Oncol. (2019) 30:1121–6. doi: 10.1093/annonc/mdz131
5. Garon EB, Hellmann MD, Rizvi NA, Carcereny E, Leighl NB, Ahn MJ, et al. Five-year overall survival for patients with advanced non−Small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 study. J Clin Oncol. (2019) 37:2518–27. doi: 10.1200/JCO.19.00934
6. Singhi EK, Horn L, Sequist LV, Heymach J, Langer CJ. Advanced non-small cell lung cancer: sequencing agents in the EGFR-mutated/ALK-rearranged populations. Am Soc Clin Oncol Educ Book. (2019) 39:e187–97. doi: 10.1200/EDBK_237821
7. Antonia SJ, Borghaei H, Ramalingam SS, Horn L, De Castro Carpeño J, Pluzanski A, et al. Four-year survival with nivolumab in patients with previously treated advanced non-small-cell lung cancer: a pooled analysis. Lancet Oncol. (2019) 20:1395–408. doi: 10.1016/S1470-2045(19)30407-3
8. Zhao Y, Wang S, Yang Z, Dong Y, Wang Y, Zhang L, et al. Co-occurring potentially actionable oncogenic drivers in non-small cell lung cancer. Front Oncol. (2021) 11:665484. doi: 10.3389/fonc.2021.665484
9. Graham RP, Treece AL, Lindeman NI, Vasalos P, Shan M, Jennings LJ, et al. Worldwide frequency of commonly detected EGFR mutations. Arch Pathol Lab Med. (2018) 142:163–7. doi: 10.5858/arpa.2016-0579-CP
10. Han B, Tjulandin S, Hagiwara K, Normanno N, Wulandari L, Laktionov K, et al. EGFR mutation prevalence in Asia-Pacific and Russian patients with advanced NSCLC of adenocarcinoma and non-adenocarcinoma histology: The IGNITE study. Lung Cancer. (2017) 113:37–44. doi: 10.1016/j.lungcan.2017.08.021
11. Harrison PT, Vyse S, Huang PH. Rare epidermal growth factor receptor (EGFR) mutations in non-small cell lung cancer. Semin Cancer Biol. (2020) 61:167–79. doi: 10.1016/j.semcancer.2019.09.015
12. Martinez-Marti A, Navarro A, Felip E. Epidermal growth factor receptor first generation tyrosine-kinase inhibitors. Transl Lung Cancer Res. (2019) 8:S235–s246. doi: 10.21037/tlcr.2019.04.20
13. Jackman DM, Miller VA, Cioffredi LA, Yeap BY, Jänne PA, Riely GJ, et al. Impact of epidermal growth factor receptor and KRAS mutations on clinical outcomes in previously untreated non-small cell lung cancer patients: results of an online tumor registry of clinical trials. Clin Cancer Res. (2009) 15:5267–73. doi: 10.1158/1078-0432.CCR-09-0888
14. Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. (2012) 13:239–46. doi: 10.1016/S1470-2045(11)70393-X
15. Jänne PA, Wang X, Socinski MA, Crawford J, Stinchcombe TE, Gu L, et al. Randomized phase II trial of erlotinib alone or with carboplatin and paclitaxel in patients who were never or light former smokers with advanced lung adenocarcinoma: CALGB 30406 trial. J Clin Oncol. (2012) 30:2063–9. doi: 10.1200/JCO.2011.40.1315
16. Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. (2010) 362:2380–8. doi: 10.1056/NEJMoa0909530
17. Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med. (2009) 361:947–57. doi: 10.1056/NEJMoa0810699
18. Zhou C, Wu YL, Chen G, Feng J, Liu XQ, Wang C, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol. (2011) 12:735–42. doi: 10.1016/S1470-2045(11)70184-X
19. Urata Y, Katakami N, Morita S, Kaji R, Yoshioka H, Seto T, et al. Randomized phase III study comparing gefitinib with erlotinib in patients with previously treated advanced lung adenocarcinoma: WJOG 5108L. J Clin Oncol. (2016) 34:3248–57. doi: 10.1200/JCO.2015.63.4154
20. Nelson V, Ziehr J, Agulnik M, Johnson M. Afatinib: emerging next-generation tyrosine kinase inhibitor for NSCLC. Onco Targets Ther. (2013) 6:135–43. doi: 10.2147/OTT
21. Park K, Tan EH, O’Byrne K, Zhang L, Boyer M, Mok T, et al. Afatinib versus gefitinib as first-line treatment of patients with EGFR mutation-positive non-small-cell lung cancer (LUX-Lung 7): a phase 2B, open-label, randomised controlled trial. Lancet Oncol. (2016) 17:577–89. doi: 10.1016/S1470-2045(16)30033-X
22. Paz-Ares L, Tan EH, O’Byrne K, Zhang L, Hirsh V, Boyer M, et al. Afatinib versus gefitinib in patients with EGFR mutation-positive advanced non-small-cell lung cancer: overall survival data from the phase IIb LUX-Lung 7 trial. Ann Oncol. (2017) 28:270–7. doi: 10.1093/annonc/mdw611
23. Yang JC, Sequist LV, Geater SL, Tsai CM, Mok TS, Schuler M, et al. Clinical activity of afatinib in patients with advanced non-small-cell lung cancer harbouring uncommon EGFR mutations: a combined post-hoc analysis of LUX-Lung 2, LUX-Lung 3, and LUX-Lung 6. Lancet Oncol. (2015) 16:830–8. doi: 10.1016/S1470-2045(15)00026-1
24. Wu YL, Cheng Y, Zhou X, Lee KH, Nakagawa K, Niho S, et al. Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): a randomised, open-label, phase 3 trial. Lancet Oncol. (2017) 18:1454–66. doi: 10.1016/S1470-2045(17)30608-3
25. Mok TS, Cheng Y, Zhou X, Lee KH, Nakagawa K, Niho S, et al. Improvement in overall survival in a randomized study that compared dacomitinib with gefitinib in patients with advanced non-small-cell lung cancer and EGFR-activating mutations. J Clin Oncol. (2018) 36:2244–50. doi: 10.1200/JCO.2018.78.7994
26. Mok TS, Cheng Y, Zhou X, Lee KH, Nakagawa K, Niho S, et al. Updated overall survival in a randomized study comparing dacomitinib with gefitinib as first-line treatment in patients with advanced non-small-cell lung cancer and EGFR-activating mutations. Drugs. (2021) 81:257–66. doi: 10.1007/s40265-020-01441-6
27. Cross DA, Ashton SE, Ghiorghiu S, Eberlein C, Nebhan CA, Spitzler PJ, et al. overcomes T790M-mediated resistance to EGFR inhibitors in lung cancer. Cancer Discovery. (2014) 4:1046–61. doi: 10.1158/2159-8290.CD-14-0337
28. Ballard P, Yates JW, Yang Z, Kim DW, Yang JC, Cantarini M, et al. Preclinical comparison of osimertinib with other EGFR-TKIs in EGFR-mutant NSCLC brain metastases models, and early evidence of clinical brain metastases activity. Clin Cancer Res. (2016) 22:5130–40. doi: 10.1158/1078-0432.CCR-16-0399
29. Yang JC-H, Kim D-W, Kim S-W, Cho BC, Lee J-S, Ye X, et al. Osimertinib activity in patients (pts) with leptomeningeal (LM) disease from non-small cell lung cancer (NSCLC): Updated results from BLOOM, a phase I study. J Clin Oncol. (2016) 34. doi: 10.1200/JCO.2016.34.15_suppl.9002
30. Remon J, Steuer CE, Ramalingam SS, Felip E. Osimertinib and other third-generation EGFR TKI in EGFR-mutant NSCLC patients. Ann Oncol. (2018) 29:i20–7. doi: 10.1093/annonc/mdx704
31. Mok TS, Wu YL, Ahn MJ, Garassino MC, Kim HR, Ramalingam SS, et al. Osimertinib or platinum-pemetrexed in EGFR T790M-positive lung cancer. N Engl J Med. (2017) 376:629–40. doi: 10.1056/NEJMoa1612674
32. Yang JCH, Kim SW, Kim DW, Lee JS, Cho BC, Ahn JS, et al. Osimertinib in patients with epidermal growth factor receptor mutation-positive non-small-cell lung cancer and leptomeningeal metastases: the BLOOM study. J Clin Oncol. (2020) 38:538–47. doi: 10.1200/JCO.19.00457
33. Yang JC-H, Cho BC, Kim D-W, Kim S-W, Lee J-S, Su W-C, et al. Osimertinib for patients (pts) with leptomeningeal metastases (LM) from EGFR-mutant non-small cell lung cancer (NSCLC): Updated results from the BLOOM study. J Clin Oncol. (2017). doi: 10.1200/JCO.2017.35.15_suppl.2020
34. Cho JH, Lim SH, An HJ, Kim KH, Park KU, Kang EJ, et al. Osimertinib for patients with non-small-cell lung cancer harboring uncommon EGFR mutations: A multicenter, open-label, phase II trial (KCSG-LU15-09). J Clin Oncol. (2020) 38:488–95. doi: 10.1200/JCO.19.00931
35. Ramalingam SS, Vansteenkiste J, Planchard D, Cho BC, Gray JE, Ohe Y, et al. Overall survival with osimertinib in untreated, EGFR-mutated advanced NSCLC. N Engl J Med. (2020) 382:41–50. doi: 10.1056/NEJMoa1913662
36. Soria JC, Ohe Y, Vansteenkiste J, Reungwetwattana T, Chewaskulyong B, Lee KH, et al. Osimertinib in untreated EGFR-mutated advanced non-small-cell lung cancer. N Engl J Med. (2018) 378:113–25. doi: 10.1056/NEJMoa1713137
37. Park K, Haura EB, Leighl NB, Mitchell P, Shu CA, Girard N, et al. Amivantamab in EGFR exon 20 insertion-mutated non-small-cell lung cancer progressing on platinum chemotherapy: initial results from the CHRYSALIS phase I study. J Clin Oncol. (2021) 39:3391–402. doi: 10.1200/JCO.21.00662
38. Zhou C, Tang KJ, Cho BC, Liu B, Paz-Ares L, Cheng S, et al. Amivantamab plus chemotherapy in NSCLC with EGFR exon 20 insertions. N Engl J Med. (2023) 389:2039–51. doi: 10.1056/NEJMoa2306441
39. Passaro A, Wang J, Wang Y, Lee SH, Melosky B, Shih JY, et al. Amivantamab plus chemotherapy with and without lazertinib in EGFR-mutant advanced NSCLC after disease progression on osimertinib: primary results from the phase III MARIPOSA-2 study. Ann Oncol. (2024) 35:77–90. doi: 10.1016/j.annonc.2023.10.117
40. Zhou C, Ramalingam SS, Kim TM, Kim SW, Yang JC, Riely GJ, et al. Treatment outcomes and safety of mobocertinib in platinum-pretreated patients with EGFR exon 20 insertion-positive metastatic non-small cell lung cancer: A phase 1/2 open-label nonrandomized clinical trial. JAMA Oncol. (2021) 7:e214761. doi: 10.1001/jamaoncol.2021.4761
41. Riely GJ, Neal JW, Camidge DR, Spira AI, Piotrowska Z, Costa DB, et al. Activity and safety of mobocertinib (TAK-788) in previously treated non-small cell lung cancer with EGFR exon 20 insertion mutations from a phase I/II trial. Cancer Discovery. (2021) 11:1688–99. doi: 10.1158/2159-8290.CD-20-1598
42. Janne P, Wang B, Cho B, Zhao J, Li J, Hochmair M, et al. EXCLAIM-2: Phase III trial of first-line (1L) mobocertinib versus platinum-based chemotherapy in patients (pts) with epidermal growth factor receptor (EGFR) exon 20 insertion (ex20ins)+ locally advanced/metastatic NSCLC. Ann OF Oncol. (2023) 34:S1663–4. doi: 10.1016/j.annonc.2023.10.586
43. Pirker R, Pereira JR, Szczesna A, von Pawel J, Krzakowski M, Ramlau R, et al. Cetuximab plus chemotherapy in patients with advanced non-small-cell lung cancer (FLEX): an open-label randomised phase III trial. Lancet. (2009) 373:1525–31. doi: 10.1016/S0140-6736(09)60569-9
44. Kohno T, Nakaoku T, Tsuta K, Tsuchihara K, Matsumoto S, Yoh K, et al. Beyond ALK-RET, ROS1 and other oncogene fusions in lung cancer. Transl Lung Cancer Res. (2015) 4:156–64. doi: 10.3978/j.issn.2218-6751.2014.11.11
45. Zhang SS, Nagasaka M, Zhu VW, Ou SI. Going beneath the tip of the iceberg. Identifying and understanding EML4-ALK variants and TP53 mutations to optimize treatment of ALK fusion positive (ALK+) NSCLC. Lung Cancer. (2021) 158:126–36. doi: 10.1016/j.lungcan.2021.06.012
46. Kim D-W, Ahn M-J, Shi Y, De Pas TM, Yang P-C, Riely GJ, et al. Results of a global phase II study with crizotinib in advanced ALK-positive non-small-cell lung cancer (NSCLC). Ann Oncol. (2012) 23:xi32–3. doi: 10.1016/S0923-7534(20)32006-8
47. Kwak EL, Bang YJ, Camidge DR, Shaw AT, Solomon B, Maki RG, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med. (2010) 363:1693–703. doi: 10.1056/NEJMoa1006448
48. Camidge DR, Bang YJ, Kwak EL, Iafrate AJ, Varella-Garcia M, Fox SB, et al. Activity and safety of crizotinib in patients with ALK-positive non-small-cell lung cancer: updated results from a phase 1 study. Lancet Oncol. (2012) 13:1011–9. doi: 10.1016/S1470-2045(12)70344-3
49. Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. (2014) 371:2167–77. doi: 10.1056/NEJMoa1408440
50. Shaw AT, Kim DW, Nakagawa K, Seto T, Crinó L, Ahn MJ, et al. Crizotinib versus chemotherapy in advanced ALK-positive lung cancer. N Engl J Med. (2013) 368:2385–94. doi: 10.1056/NEJMoa1214886
51. Kodama T, Tsukaguchi T, Yoshida M, Kondoh O, Sakamoto H. Selective ALK inhibitor alectinib with potent antitumor activity in models of crizotinib resistance. Cancer Lett. (2014) 351:215–21. doi: 10.1016/j.canlet.2014.05.020
52. Gadgeel SM, Gandhi L, Riely GJ, Chiappori AA, West HL, Azada MC, et al. Safety and activity of alectinib against systemic disease and brain metastases in patients with crizotinib-resistant ALK-rearranged non-small-cell lung cancer (AF-002JG): results from the dose-finding portion of a phase 1/2 study. Lancet Oncol. (2014) 15:1119–28. doi: 10.1016/S1470-2045(14)70362-6
53. Peters S, Camidge DR, Shaw AT, Gadgeel S, Ahn JS, Kim DW, et al. Alectinib versus crizotinib in untreated ALK-positive non-small-cell lung cancer. N Engl J Med. (2017) 377:829–38. doi: 10.1056/NEJMoa1704795
54. Hida T, Nokihara H, Kondo M, Kim YH, Azuma K, Seto T, et al. Alectinib versus crizotinib in patients with ALK-positive non-small-cell lung cancer (J-ALEX): an open-label, randomised phase 3 trial. Lancet. (2017) 390:29–39. doi: 10.1016/S0140-6736(17)30565-2
55. Ou SH, Ahn JS, De Petris L, Govindan R, Yang JC, Hughes B, et al. Alectinib in crizotinib-refractory ALK-rearranged non-small-cell lung cancer: A phase II global study. J Clin Oncol. (2016) 34:661–8. doi: 10.1200/JCO.2015.63.9443
56. Shaw AT, Gandhi L, Gadgeel S, Riely GJ, Cetnar J, West H, et al. Alectinib in ALK-positive, crizotinib-resistant, non-small-cell lung cancer: a single-group, multicentre, phase 2 trial. Lancet Oncol. (2016) 17:234–42. doi: 10.1016/S1470-2045(15)00488-X
57. Camidge DR, Kim HR, Ahn MJ, Yang JC, Han JY, Lee JS, et al. Brigatinib versus crizotinib in ALK-positive non-small-cell lung cancer. N Engl J Med. (2018) 379:2027–39. doi: 10.1056/NEJMoa1810171
58. Camidge DR, Kim HR, Ahn MJ, Yang JCH, Han JY, Hochmair MJ, et al. Brigatinib versus crizotinib in ALK inhibitor-naive advanced ALK-positive NSCLC: final results of phase 3 ALTA-1L trial. J Thorac Oncol. (2021) 16:2091–108. doi: 10.1016/j.jtho.2021.07.035
59. Kim DW, Tiseo M, Ahn MJ, Reckamp KL, Hansen KH, Kim SW, et al. Brigatinib in patients with crizotinib-refractory anaplastic lymphoma kinase-positive non-small-cell lung cancer: A randomized, multicenter phase II trial. J Clin Oncol. (2017) 35:2490–8. doi: 10.1200/JCO.2016.71.5904
60. Camidge DR, Tiseo M, Ahn M-J, Reckamp K, Hansen K, Kim S-W, et al. P3. 02a-013 brigatinib in crizotinib-refractory ALK+ NSCLC: central assessment and updates from ALTA, a pivotal randomized phase 2 trial: topic: ALK clinical. J Thorac Oncol. (2017) 12:S1167–9. doi: 10.1016/j.jtho.2016.11.1643
61. Lim SM, Kim HR, Lee JS, Lee KH, Lee YG, Min YJ, et al. Open-label, multicenter, phase II study of ceritinib in patients with non-small-cell lung cancer harboring ROS1 rearrangement. J Clin Oncol. (2017) 35:2613–8. doi: 10.1200/JCO.2016.71.3701
62. Soria JC, Tan DSW, Chiari R, Wu YL, Paz-Ares L, Wolf J, et al. First-line ceritinib versus platinum-based chemotherapy in advanced ALK-rearranged non-small-cell lung cancer (ASCEND-4): a randomised, open-label, phase 3 study. Lancet. (2017) 389:917–29. doi: 10.1016/S0140-6736(17)30123-X
63. Crinò L, Ahn MJ, De Marinis F, Groen HJ, Wakelee H, Hida T, et al. Multicenter phase II study of whole-body and intracranial activity with ceritinib in patients with ALK-rearranged non-small-cell lung cancer previously treated with chemotherapy and crizotinib: results from ASCEND-2. J Clin Oncol. (2016) 34:2866–73. doi: 10.1200/jco.2015.65.5936
64. Shaw AT, Kim TM, Crinò L, Gridelli C, Kiura K, Liu G, et al. Ceritinib versus chemotherapy in patients with ALK-rearranged non-small-cell lung cancer previously given chemotherapy and crizotinib (ASCEND-5): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. (2017) 18:874–86. doi: 10.1016/S1470-2045(17)30339-X
65. Shaw AT, Solomon BJ, Chiari R, Riely GJ, Besse B, Soo RA, et al. Lorlatinib in advanced ROS1-positive non-small-cell lung cancer: a multicentre, open-label, single-arm, phase 1-2 trial. Lancet Oncol. (2019) 20:1691–701. doi: 10.1016/S1470-2045(19)30655-2
66. Solomon BJ, Besse B, Bauer TM, Felip E, Soo RA, Camidge DR, et al. Lorlatinib in patients with ALK-positive non-small-cell lung cancer: results from a global phase 2 study. Lancet Oncol. (2018) 19:1654–67. doi: 10.1016/S1470-2045(18)30649-1
67. Besse B, Solomon BJ, Felip E, Bauer TM, Ou S-HI, Soo RA, et al. Lorlatinib in patients (Pts) with previously treated ALK+ advanced non-small cell lung cancer (NSCLC): Updated efficacy and safety. J Clin Oncol. (2018). doi: 10.1200/JCO.2018.36.15_suppl.9032
68. Shaw AT, Solomon BJ, Besse B, Bauer TM, Lin CC, Soo RA, et al. ALK resistance mutations and efficacy of lorlatinib in advanced anaplastic lymphoma kinase-positive non-small-cell lung cancer. J Clin Oncol. (2019) 37:1370–9. doi: 10.1200/JCO.18.02236
69. Shaw AT, Bauer TM, de Marinis F, Felip E, Goto Y, Liu G, et al. First-line lorlatinib or crizotinib in advanced ALK-positive lung cancer. N Engl J Med. (2020) 383:2018–29. doi: 10.1056/NEJMoa2027187
70. Solomon BJ, Bauer TM, Ignatius Ou SH, Liu G, Hayashi H, Bearz A, et al. Post hoc analysis of lorlatinib intracranial efficacy and safety in patients with ALK-positive advanced non-small-cell lung cancer from the phase III CROWN study. J Clin Oncol. (2022) 40:3593–602. doi: 10.1200/JCO.21.02278
71. Baik CS, Myall NJ, Wakelee HA. Targeting BRAF-mutant non-small cell lung cancer: from molecular profiling to rationally designed therapy. Oncologist. (2017) 22:786–96. doi: 10.1634/theoncologist.2016-0458
72. Nguyen-Ngoc T, Bouchaab H, Adjei AA, Peters S. BRAF alterations as therapeutic targets in non-small-cell lung cancer. J Thorac Oncol. (2015) 10:1396–403. doi: 10.1097/JTO.0000000000000644
73. Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lung cancer. Nature. (2018) 553:446–54. doi: 10.1038/nature25183
74. Litvak AM, Paik PK, Woo KM, Sima CS, Hellmann MD, Arcila ME, et al. Clinical characteristics and course of 63 patients with BRAF mutant lung cancers. J Thorac Oncol. (2014) 9:1669–74. doi: 10.1097/JTO.0000000000000344
75. Marchetti A, Felicioni L, Malatesta S, Grazia Sciarrotta M, Guetti L, Chella A, et al. Clinical features and outcome of patients with non-small-cell lung cancer harboring BRAF mutations. J Clin Oncol. (2011) 29:3574–9. doi: 10.1200/JCO.2011.35.9638
76. Planchard D, Smit EF, Groen HJM, Mazieres J, Besse B, Helland Å, et al. Dabrafenib plus trametinib in patients with previously untreated BRAF(V600E)-mutant metastatic non-small-cell lung cancer: an open-label, phase 2 trial. Lancet Oncol. (2017) 18:1307–16. doi: 10.1016/S1470-2045(17)30679-4
77. O’Leary CG, Andelkovic V, Ladwa R, Pavlakis N, Zhou C, Hirsch F, et al. Targeting BRAF mutations in non-small cell lung cancer. Transl Lung Cancer Res. (2019) 8:1119–24. doi: 10.21037/tlcr.2019.10.22
78. Riely GJ, Smit EF, Ahn MJ, Felip E, Ramalingam SS, Tsao A, et al. Open-label study of encorafenib plus binimetinib in patients with BRAF(V600)-mutant metastatic non-small-cell lung cancer. J Clin Oncol. (2023) 41:3700–11. doi: 10.1200/JCO.23.00774
79. Li BT, Shen R, Buonocore D, Olah ZT, Ni A, Ginsberg MS, et al. Ado-trastuzumab emtansine for patients with HER2-mutant lung cancers: results from a phase II basket trial. J Clin Oncol. (2018) 36:2532–7. doi: 10.1200/JCO.2018.77.9777
80. Li BT, Shen R, Buonocore D, Olah ZT, Ni A, Ginsberg MS, et al. Ado-trastuzumab emtansine in patients with HER2 mutant lung cancers: Results from a phase II basket trial. J Clin Oncol. (2017). doi: 10.1200/JCO.2017.35.15_suppl.8510
81. Iwama E, Zenke Y, Sugawara S, Daga H, Morise M, Yanagitani N, et al. Trastuzumab emtansine for patients with non-small cell lung cancer positive for human epidermal growth factor receptor 2 exon-20 insertion mutations. Eur J Cancer. (2022) 162:99–106. doi: 10.1016/j.ejca.2021.11.021
82. Li BT, Smit EF, Goto Y, Nakagawa K, Udagawa H, Mazières J, et al. Trastuzumab deruxtecan in HER2-mutant non-small-cell lung cancer. N Engl J Med. (2022) 386:241–51. doi: 10.1056/NEJMoa2112431
83. Tsurutani J, Iwata H, Krop I, Jänne PA, Doi T, Takahashi S, et al. Targeting HER2 with trastuzumab deruxtecan: A dose-expansion, phase I study in multiple advanced solid tumors. Cancer Discovery. (2020) 10:688–701. doi: 10.1158/2159-8290.CD-19-1014
84. Skoulidis F, Heymach JV. Co-occurring genomic alterations in non-small-cell lung cancer biology and therapy. Nat Rev Cancer. (2019) 19:495–509. doi: 10.1038/s41568-019-0179-8
85. Dogan S, Shen R, Ang DC, Johnson ML, D’Angelo SP, Paik PK, et al. Molecular epidemiology of EGFR and KRAS mutations in 3,026 lung adenocarcinomas: higher susceptibility of women to smoking-related KRAS-mutant cancers. Clin Cancer Res. (2012) 18:6169–77. doi: 10.1158/1078-0432.CCR-11-3265
86. Canon J, Rex K, Saiki AY, Mohr C, Cooke K, Bagal D, et al. The clinical KRAS(G12C) inhibitor AMG 510 drives anti-tumour immunity. Nature. (2019) 575:217–23. doi: 10.1038/s41586-019-1694-1
87. Nakajima EC, Drezner N, Li X, Mishra-Kalyani PS, Liu Y, Zhao H, et al. FDA approval summary: sotorasib for KRAS G12C-mutated metastatic NSCLC. Clin Cancer Res. (2022) 28:1482–6. doi: 10.1158/1078-0432.CCR-21-3074
88. Skoulidis F, Li BT, Dy GK, Price TJ, Falchook GS, Wolf J, et al. Sotorasib for lung cancers with KRAS p.G12C mutation. N Engl J Med. (2021) 384:2371–81. doi: 10.1056/NEJMoa2103695
89. de Langen AJ, Johnson ML, Mazieres J, Dingemans AC, Mountzios G, Pless M, et al. Sotorasib versus docetaxel for previously treated non-small-cell lung cancer with KRAS(G12C) mutation: a randomised, open-label, phase 3 trial. Lancet. (2023) 401:733–46. doi: 10.1016/S0140-6736(23)00221-0
90. Jänne PA, Riely GJ, Gadgeel SM, Heist RS, Ou SI, Pacheco JM, et al. Adagrasib in non-small-cell lung cancer harboring a KRAS(G12C) mutation. N Engl J Med. (2022) 387:120–31. doi: 10.1056/NEJMoa2204619
91. Wolf J, Seto T, Han JY, Reguart N, Garon EB, Groen HJM, et al. Capmatinib in MET exon 14-mutated or MET-amplified non-small-cell lung cancer. N Engl J Med. (2020) 383:944–57. doi: 10.1056/NEJMoa2002787
92. Garon EB, Heist RS, Seto T, Han J-Y, Reguart N, Groen HJ, et al. Abstract CT082: Capmatinib in MET ex14-mutated (mut) advanced non-small cell lung cancer (NSCLC): Results from the phase II GEOMETRY mono-1 study, including efficacy in patients (pts) with brain metastases (BM). Cancer Res. (2020) 80:CT082–2. doi: 10.1158/1538-7445.AM2020-CT082
93. Choi W, Park SY, Lee Y, Lim KY, Park M, Lee GK, et al. The clinical impact of capmatinib in the treatment of advanced non-small cell lung cancer with MET exon 14 skipping mutation or gene amplification. Cancer Res Treat. (2021) 53:1024–32. doi: 10.4143/crt.2020.1331
94. Drilon A, Clark JW, Weiss J, Ou SI, Camidge DR, Solomon BJ, et al. Antitumor activity of crizotinib in lung cancers harboring a MET exon 14 alteration. Nat Med. (2020) 26:47–51. doi: 10.1038/s41591-019-0716-8
95. Camidge DR, Otterson GA, Clark JW, Ignatius Ou SH, Weiss J, Ades S, et al. Crizotinib in patients with MET-amplified NSCLC. J Thorac Oncol. (2021) 16:1017–29. doi: 10.1016/j.jtho.2021.02.010
96. Paik PK, Felip E, Veillon R, Sakai H, Cortot AB, Garassino MC, et al. Tepotinib in non-small-cell lung cancer with MET exon 14 skipping mutations. N Engl J Med. (2020) 383:931–43. doi: 10.1056/NEJMoa2004407
97. Le X, Paz-Ares LG, Van Meerbeeck J, Viteri S, Galvez CC, Smit EF, et al. Tepotinib in patients with non-small cell lung cancer with high-level MET amplification detected by liquid biopsy: VISION Cohort B. Cell Rep Med. (2023) 4:101280. doi: 10.1016/j.xcrm.2023.101280
98. Drilon A, Laetsch TW, Kummar S, DuBois SG, Lassen UN, Demetri GD, et al. Efficacy of larotrectinib in TRK fusion-positive cancers in adults and children. N Engl J Med. (2018) 378:731–9. doi: 10.1056/NEJMoa1714448
99. Doebele RC, Drilon A, Paz-Ares L, Siena S, Shaw AT, Farago AF, et al. Entrectinib in patients with advanced or metastatic NTRK fusion-positive solid tumours: integrated analysis of three phase 1-2 trials. Lancet Oncol. (2020) 21:271–82. doi: 10.1016/S1470-2045(19)30691-6
100. Drilon A, Barlesi F, Braud FD, Cho BC, Ahn M-J, Siena S, et al. Abstract CT192: Entrectinib in locally advanced or metastatic ROS1 fusion-positive non-small cell lung cancer (NSCLC): Integrated analysis of ALKA-372-001, STARTRK-1 and STARTRK-2. Cancer Res. (2019) 79:CT192–2. doi: 10.1158/1538-7445.AM2019-CT192
101. Doebele R, Ahn M, Siena S, Drilon A, Krebs M, Lin C, et al. OA02. 01 Efficacy and safety of entrectinib in locally advanced or metastatic ROS1 fusion-positive non-small cell lung cancer (NSCLC). J Thorac Oncol. (2018) 13:S321–2. doi: 10.1016/j.jtho.2018.08.239
102. Lassen U, Albert CM, Kummar S, Van Tilburg C, DuBois SG, Geoerger B, et al. Larotrectinib efficacy and safety in TRK fusion cancer: an expanded clinical dataset showing consistency in an age and tumor agnostic approach. Ann Oncol. (2018) 29:viii133. doi: 10.1093/annonc/mdy279.397
103. Collisson EA, Campbell JD, Brooks AN, Berger AH, Lee W, Chmielecki J, et al. Comprehensive molecular profiling of lung adenocarcinoma. Nature. (2014) 511:543–50. doi: 10.1038/nature13385
104. Bronte G, Ulivi P, Verlicchi A, Cravero P, Delmonte A, Crinò L. Targeting RET-rearranged non-small-cell lung cancer: future prospects. Lung Cancer (Auckl). (2019) 10:27–36. doi: 10.2147/LCTT
105. Gainor JF, Curigliano G, Kim DW, Lee DH, Besse B, Baik CS, et al. Pralsetinib for RET fusion-positive non-small-cell lung cancer (ARROW): a multi-cohort, open-label, phase 1/2 study. Lancet Oncol. (2021) 22:959–69. doi: 10.1016/S1470-2045(21)00247-3
106. Wright KM. FDA approves pralsetinib for treatment of adults with metastatic RET fusion-positive NSCLC. Oncol (Williston Park). (2020) 34:406–6. doi: 10.46883/ONCOLOGY
107. Drilon A, Oxnard GR, Tan DSW, Loong HHF, Johnson M, Gainor J, et al. Efficacy of selpercatinib in RET fusion-positive non-small-cell lung cancer. N Engl J Med. (2020) 383:813–24. doi: 10.1056/NEJMoa2005653
108. Drilon A, Oxnard G, Wirth L, Besse B, Gautschi O, Tan S, et al. PL02. 08 registrational results of LIBRETTO-001: a phase 1/2 trial of LOXO-292 in patients with RET fusion-positive lung cancers. J Thorac Oncol. (2019) 14:S6–7.
109. Drilon A, Rekhtman N, Arcila M, Wang L, Ni A, Albano M, et al. Cabozantinib in patients with advanced RET-rearranged non-small-cell lung cancer: an open-label, single-centre, phase 2, single-arm trial. Lancet Oncol. (2016) 17:1653–60. doi: 10.1016/S1470-2045(16)30562-9
110. Drilon A, Wang L, Hasanovic A, Suehara Y, Lipson D, Stephens P, et al. Response to Cabozantinib in patients with RET fusion-positive lung adenocarcinomas. Cancer Discovery. (2013) 3:630–5. doi: 10.1158/2159-8290.CD-13-0035
111. Lin JJ, Shaw AT. Recent advances in targeting ROS1 in lung cancer. J Thorac Oncol. (2017) 12:1611–25. doi: 10.1016/j.jtho.2017.08.002
112. Dziadziuszko R, Le AT, Wrona A, Jassem J, Camidge DR, Varella-Garcia M, et al. An activating KIT mutation induces crizotinib resistance in ROS1-positive lung cancer. J Thorac Oncol. (2016) 11:1273–81. doi: 10.1016/j.jtho.2016.04.001
113. Wu YL, Yang JC, Kim DW, Lu S, Zhou J, Seto T, et al. Phase II study of crizotinib in east asian patients with ROS1-positive advanced non-small-cell lung cancer. J Clin Oncol. (2018) 36:1405–11. doi: 10.1200/JCO.2017.75.5587
114. Shaw AT, Ou SH, Bang YJ, Camidge DR, Solomon BJ, Salgia R, et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N Engl J Med. (2014) 371:1963–71. doi: 10.1056/NEJMoa1406766
115. Michels S, Massutí B, Schildhaus H-U, Franklin J, Sebastian M, Felip E, et al. Safety and efficacy of crizotinib in patients with advanced or metastatic ROS1-rearranged lung cancer (EUCROSS): a European phase II clinical trial. J Thorac Oncol. (2019) 14:1266–76. doi: 10.1016/j.jtho.2019.03.020
116. Mazières J, Zalcman G, Crinò L, Biondani P, Barlesi F, Filleron T, et al. Crizotinib therapy for advanced lung adenocarcinoma and a ROS1 rearrangement: results from the EUROS1 cohort. J Clin Oncol. (2015) 33:992–9. doi: 10.1200/JCO.2014.58.3302
117. Drilon A, Siena S, Dziadziuszko R, Barlesi F, Krebs MG, Shaw AT, et al. Entrectinib in ROS1 fusion-positive non-small-cell lung cancer: integrated analysis of three phase 1-2 trials. Lancet Oncol. (2020) 21:261–70. doi: 10.1016/S1470-2045(19)30690-4
118. Drilon A, Camidge DR, Lin JJ, Kim SW, Solomon BJ, Dziadziuszko R, et al. Repotrectinib in ROS1 fusion-positive non-small-cell lung cancer. N Engl J Med. (2024) 390:118–31. doi: 10.1056/NEJMoa2302299
119. Sandler A, Gray R, Perry MC, Brahmer J, Schiller JH, Dowlati A, et al. Paclitaxel-carboplatin alone or with bevacizumab for non-small-cell lung cancer. N Engl J Med. (2006) 355:2542–50. doi: 10.1056/NEJMoa061884
120. Saito H, Fukuhara T, Furuya N, Watanabe K, Sugawara S, Iwasawa S, et al. Erlotinib plus bevacizumab versus erlotinib alone in patients with EGFR-positive advanced non-squamous non-small-cell lung cancer (NEJ026): interim analysis of an open-label, randomised, multicentre, phase 3 trial. Lancet Oncol. (2019) 20:625–35. doi: 10.1016/S1470-2045(19)30035-X
121. Nakagawa K, Garon EB, Seto T, Nishio M, Ponce Aix S, Paz-Ares L, et al. Ramucirumab plus erlotinib in patients with untreated, EGFR-mutated, advanced non-small-cell lung cancer (RELAY): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet Oncol. (2019) 20:1655–69. doi: 10.1016/S1470-2045(19)30634-5
122. Garon EB, Ciuleanu TE, Arrieta O, Prabhash K, Syrigos KN, Goksel T, et al. Ramucirumab plus docetaxel versus placebo plus docetaxel for second-line treatment of stage IV non-small-cell lung cancer after disease progression on platinum-based therapy (REVEL): a multicentre, double-blind, randomised phase 3 trial. Lancet. (2014) 384:665–73. doi: 10.1016/S0140-6736(14)60845-X
123. Hilberg F, Roth GJ, Krssak M, Kautschitsch S, Sommergruber W, Tontsch-Grunt U, et al. BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res. (2008) 68:4774–82. doi: 10.1158/0008-5472.CAN-07-6307
124. Reck M, Kaiser R, Mellemgaard A, Douillard JY, Orlov S, Krzakowski M, et al. Docetaxel plus nintedanib versus docetaxel plus placebo in patients with previously treated non-small-cell lung cancer (LUME-Lung 1): a phase 3, double-blind, randomised controlled trial. Lancet Oncol. (2014) 15:143–55. doi: 10.1016/S1470-2045(13)70586-2
125. Gainor JF, Shaw AT. Emerging paradigms in the development of resistance to tyrosine kinase inhibitors in lung cancer. J Clin Oncol. (2013) 31:3987–96. doi: 10.1200/JCO.2012.45.2029
126. Zhang W, Lei P, Dong X, Xu C. The new concepts on overcoming drug resistance in lung cancer. Drug Des Devel Ther. (2014) 8:735–44. doi: 10.2147/DDDT
127. Engelman JA, Jänne PA. Mechanisms of acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small cell lung cancer. Clin Cancer Res. (2008) 14:2895–9. doi: 10.1158/1078-0432.CCR-07-2248
128. Pao W, Chmielecki J. Rational, biologically based treatment of EGFR-mutant non-small-cell lung cancer. Nat Rev Cancer. (2010) 10:760–74. doi: 10.1038/nrc2947
129. Ellis LM, Hicklin DJ. Resistance to targeted therapies: refining anticancer therapy in the era of molecular oncology. Clin Cancer Res. (2009) 15:7471–8. doi: 10.1158/1078-0432.CCR-09-1070
130. Oser MG, Niederst MJ, Sequist LV, Engelman JA. Transformation from non-small-cell lung cancer to small-cell lung cancer: molecular drivers and cells of origin. Lancet Oncol. (2015) 16:e165–72. doi: 10.1016/S1470-2045(14)71180-5
131. Rath B, Plangger A, Hamilton G. Non-small cell lung cancer-small cell lung cancer transformation as mechanism of resistance to tyrosine kinase inhibitors in lung cancer. Cancer Drug Resist. (2020) 3:171–8. doi: 10.20517/cdr.2019.85
132. Jackman D, Pao W, Riely GJ, Engelman JA, Kris MG, Jänne PA, et al. Clinical definition of acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small-cell lung cancer. J Clin Oncol. (2010) 28:357–60. doi: 10.1200/JCO.2009.24.7049
133. Yasuda H, Kobayashi S, Costa DB. EGFR exon 20 insertion mutations in non-small-cell lung cancer: preclinical data and clinical implications. Lancet Oncol. (2012) 13:e23–31. doi: 10.1016/S1470-2045(11)70129-2
134. Pao W, Miller VA, Politi KA, Riely GJ, Somwar R, Zakowski MF, et al. Acquired resistance of lung adenocarcinomas to gefitinib or erlotinib is associated with a second mutation in the EGFR kinase domain. PloS Med. (2005) 2:e73. doi: 10.1371/journal.pmed.0020073
135. Yu PP, Vose JM, Hayes DF. Genetic cancer susceptibility testing: increased technology, increased complexity. J Clin Oncol. (2015) 33:3533–4. doi: 10.1200/JCO.2015.63.3628
136. Yu HA, Arcila ME, Rekhtman N, Sima CS, Zakowski MF, Pao W, et al. Analysis of tumor specimens at the time of acquired resistance to EGFR-TKI therapy in 155 patients with EGFR-mutant lung cancers. Clin Cancer Res. (2013) 19:2240–7. doi: 10.1158/1078-0432.CCR-12-2246
137. Riely GJ, Yu HA. EGFR: the paradigm of an oncogene-driven lung cancer. Clin Cancer Res. (2015) 21:2221–6. doi: 10.1158/1078-0432.CCR-14-3154
138. Finlay MR, Anderton M, Ashton S, Ballard P, Bethel PA, Box MR, et al. Discovery of a potent and selective EGFR inhibitor (AZD9291) of both sensitizing and T790M resistance mutations that spares the wild type form of the receptor. J Med Chem. (2014) 57:8249–67. doi: 10.1021/jm500973a
139. Kosaka T, Yatabe Y, Endoh H, Yoshida K, Hida T, Tsuboi M, et al. Analysis of epidermal growth factor receptor gene mutation in patients with non-small cell lung cancer and acquired resistance to gefitinib. Clin Cancer Res. (2006) 12:5764–9. doi: 10.1158/1078-0432.CCR-06-0714
140. Onitsuka T, Uramoto H, Nose N, Takenoyama M, Hanagiri T, Sugio K, et al. Acquired resistance to gefitinib: the contribution of mechanisms other than the T790M, MET, and HGF status. Lung Cancer. (2010) 68:198–203. doi: 10.1016/j.lungcan.2009.05.022
141. Yun CH, Mengwasser KE, Toms AV, Woo MS, Greulich H, Wong KK, et al. The T790M mutation in EGFR kinase causes drug resistance by increasing the affinity for ATP. Proc Natl Acad Sci U.S.A. (2008) 105:2070–5. doi: 10.1073/pnas.0709662105
142. Huang L, Fu L. Mechanisms of resistance to EGFR tyrosine kinase inhibitors. Acta Pharm Sin B. (2015) 5:390–401. doi: 10.1016/j.apsb.2015.07.001
143. Bean J, Riely GJ, Balak M, Marks JL, Ladanyi M, Miller VA, et al. Acquired resistance to epidermal growth factor receptor kinase inhibitors associated with a novel T854A mutation in a patient with EGFR-mutant lung adenocarcinoma. Clin Cancer Res. (2008) 14:7519–25. doi: 10.1158/1078-0432.CCR-08-0151
144. Costa DB, Halmos B, Kumar A, Schumer ST, Huberman MS, Boggon TJ, et al. BIM mediates EGFR tyrosine kinase inhibitor-induced apoptosis in lung cancers with oncogenic EGFR mutations. PloS Med. (2007) 4:1669–79. doi: 10.1371/journal.pmed.0040315
145. Toyooka S, Date H, Uchida A, Kiura K, Takata M. The epidermal growth factor receptor D761Y mutation and effect of tyrosine kinase inhibitor. Clin Cancer Res. (2007) 13:3431. doi: 10.1158/1078-0432.CCR-07-0070
146. Leonetti A, Sharma S, Minari R, Perego P, Giovannetti E, Tiseo M. Resistance mechanisms to osimertinib in EGFR-mutated non-small cell lung cancer. Br J Cancer. (2019) 121:725–37. doi: 10.1038/s41416-019-0573-8
147. Takeda M, Nakagawa K. First- and second-generation EGFR-TKIs are all replaced to osimertinib in chemo-naive EGFR mutation-positive non-small cell lung cancer? Int J Mol Sci. (2019) 20. doi: 10.3390/ijms20010146
148. Heuckmann JM, Balke-Want H, Malchers F, Peifer M, Sos ML, Koker M, et al. Differential protein stability and ALK inhibitor sensitivity of EML4-ALK fusion variants. Clin Cancer Res. (2012) 18:4682–90. doi: 10.1158/1078-0432.CCR-11-3260
149. Choi YL, Soda M, Yamashita Y, Ueno T, Takashima J, Nakajima T, et al. EML4-ALK mutations in lung cancer that confer resistance to ALK inhibitors. N Engl J Med. (2010) 363:1734–9. doi: 10.1056/NEJMoa1007478
150. Doebele RC, Pilling AB, Aisner DL, Kutateladze TG, Le AT, Weickhardt AJ, et al. Mechanisms of resistance to crizotinib in patients with ALK gene rearranged non-small cell lung cancer. Clin Cancer Res. (2012) 18:1472–82. doi: 10.1158/1078-0432.CCR-11-2906
151. Kim S, Kim TM, Kim DW, Go H, Keam B, Lee SH, et al. Heterogeneity of genetic changes associated with acquired crizotinib resistance in ALK-rearranged lung cancer. J Thorac Oncol. (2013) 8:415–22. doi: 10.1097/JTO.0b013e318283dcc0
152. Ai X, Niu X, Chang L, Chen R, Ou SI, Lu S. Next generation sequencing reveals a novel ALK G1128A mutation resistant to crizotinib in an ALK-Rearranged NSCLC patient. Lung Cancer. (2018) 123:83–6. doi: 10.1016/j.lungcan.2018.07.004
153. Yanagitani N, Uchibori K, Koike S, Tsukahara M, Kitazono S, Yoshizawa T, et al. Drug resistance mechanisms in Japanese anaplastic lymphoma kinase-positive non-small cell lung cancer and the clinical responses based on the resistant mechanisms. Cancer Sci. (2020) 111:932–9. doi: 10.1111/cas.14314
154. Dehghanian F, Kay M, Vallian S. F1174V mutation alters the ALK active conformation in response to Crizotinib in NSCLC: Insight from molecular simulations. J Mol Graph Model. (2017) 75:287–93. doi: 10.1016/j.jmgm.2017.06.010
155. Katayama R, Shaw AT, Khan TM, Mino-Kenudson M, Solomon BJ, Halmos B, et al. Mechanisms of acquired crizotinib resistance in ALK-rearranged lung Cancers. Sci Transl Med. (2012) 4:120ra17. doi: 10.1126/scitranslmed.3003316
156. Novello S, Mazières J, Oh IJ, de Castro J, Migliorino MR, Helland Å, et al. Alectinib versus chemotherapy in crizotinib-pretreated anaplastic lymphoma kinase (ALK)-positive non-small-cell lung cancer: results from the phase III ALUR study. Ann Oncol. (2018) 29:1409–16. doi: 10.1093/annonc/mdy121
157. Yang Y, Zhou J, Zhou J, Feng J, Zhuang W, Chen J, et al. Efficacy, safety, and biomarker analysis of ensartinib in crizotinib-resistant, ALK-positive non-small-cell lung cancer: a multicentre, phase 2 trial. Lancet Respir Med. (2020) 8:45–53. doi: 10.1016/S2213-2600(19)30252-8
158. Camidge DR, Kim DW, Tiseo M, Langer CJ, Ahn MJ, Shaw AT, et al. Exploratory analysis of brigatinib activity in patients with anaplastic lymphoma kinase-positive non-small-cell lung cancer and brain metastases in two clinical trials. J Clin Oncol. (2018) 36:2693–701. doi: 10.1200/JCO.2017.77.5841
159. Gainor JF, Dardaei L, Yoda S, Friboulet L, Leshchiner I, Katayama R, et al. Molecular mechanisms of resistance to first- and second-generation ALK inhibitors in ALK-rearranged lung cancer. Cancer Discovery. (2016) 6:1118–33. doi: 10.1158/2159-8290.CD-16-0596
160. Pailler E, Faugeroux V, Oulhen M, Mezquita L, Laporte M, Honoré A, et al. Acquired resistance mutations to ALK inhibitors identified by single circulating tumor cell sequencing in ALK-rearranged non-small-cell lung cancer. Clin Cancer Res. (2019) 25:6671–82. doi: 10.1158/1078-0432.CCR-19-1176
161. Sasaki T, Koivunen J, Ogino A, Yanagita M, Nikiforow S, Zheng W, et al. A novel ALK secondary mutation and EGFR signaling cause resistance to ALK kinase inhibitors. Cancer Res. (2011) 71:6051–60. doi: 10.1158/0008-5472.CAN-11-1340
162. Lovly CM, McDonald NT, Chen H, Ortiz-Cuaran S, Heukamp LC, Yan Y, et al. Rationale for co-targeting IGF-1R and ALK in ALK fusion-positive lung cancer. Nat Med. (2014) 20:1027–34. doi: 10.1038/nm.3667
163. Crystal AS, Shaw AT, Sequist LV, Friboulet L, Niederst MJ, Lockerman EL, et al. Patient-derived models of acquired resistance can identify effective drug combinations for cancer. Science. (2014) 346:1480–6. doi: 10.1126/science.1254721
164. Tsuji T, Ozasa H, Aoki W, Aburaya S, Funazo T, Furugaki K, et al. Alectinib resistance in ALK-rearranged lung cancer by dual salvage signaling in a clinically paired resistance model. Mol Cancer Res. (2019) 17:212–24. doi: 10.1158/1541-7786.MCR-18-0325
165. Shi R, Filho SNM, Li M, Fares A, Weiss J, Pham NA, et al. BRAF V600E mutation and MET amplification as resistance pathways of the second-generation anaplastic lymphoma kinase (ALK) inhibitor alectinib in lung cancer. Lung Cancer. (2020) 146:78–85. doi: 10.1016/j.lungcan.2020.05.018
166. Dagogo-Jack I, Yoda S, Lennerz JK, Langenbucher A, Lin JJ, Rooney MM, et al. MET alterations are a recurring and actionable resistance mechanism in ALK-positive lung cancer. Clin Cancer Res. (2020) 26:2535–45. doi: 10.1158/1078-0432.CCR-19-3906
167. Gainor JF, Tseng D, Yoda S, Dagogo-Jack I, Friboulet L, Lin JJ, et al. Patterns of metastatic spread and mechanisms of resistance to crizotinib in ROS1-positive non-small-cell lung cancer. JCO Precis Oncol. (2017) 2017. doi: 10.1200/PO.17.00063
168. McCoach CE, Le AT, Gowan K, Jones K, Schubert L, Doak A, et al. Resistance mechanisms to targeted therapies in ROS1(+) and ALK(+) non-small cell lung cancer. Clin Cancer Res. (2018) 24:3334–47. doi: 10.1158/1078-0432.CCR-17-2452
169. D’Angelo A, Sobhani N, Chapman R, Bagby S, Bortoletti C, Traversini M, et al. Focus on ROS1-positive non-small cell lung cancer (NSCLC): crizotinib, resistance mechanisms and the newer generation of targeted therapies. Cancers (Basel). (2020) 12. doi: 10.3390/cancers12113293
170. Lin JJ, Choudhury NJ, Yoda S, Zhu VW, Johnson TW, Sakhtemani R, et al. Spectrum of mechanisms of resistance to crizotinib and lorlatinib in ROS1 fusion-positive lung cancer. Clin Cancer Res. (2021) 27:2899–909. doi: 10.1158/1078-0432.CCR-21-0032
171. Zhou Y, Jiang W, Zeng L, Mi J, Song L, Lizaso A, et al. A novel ROS1 G2032 K missense mutation mediates lorlatinib resistance in a patient with ROS1-rearranged lung adenocarcinoma but responds to nab-paclitaxel plus pembrolizumab. Lung Cancer. (2020) 143:55–9. doi: 10.1016/j.lungcan.2020.03.019
172. Facchinetti F, Loriot Y, Kuo MS, Mahjoubi L, Lacroix L, Planchard D, et al. Crizotinib-resistant ROS1 mutations reveal a predictive kinase inhibitor sensitivity model for ROS1- and ALK-rearranged lung cancers. Clin Cancer Res. (2016) 22:5983–91. doi: 10.1158/1078-0432.CCR-16-0917
173. Facchinetti F, Rossi G, Bria E, Soria JC, Besse B, Minari R, et al. Oncogene addiction in non-small cell lung cancer: Focus on ROS1 inhibition. Cancer Treat Rev. (2017) 55:83–95. doi: 10.1016/j.ctrv.2017.02.010
174. Drilon A, Somwar R, Wagner JP, Vellore NA, Eide CA, Zabriskie MS, et al. A novel crizotinib-resistant solvent-front mutation responsive to cabozantinib therapy in a patient with ROS1-rearranged lung cancer. Clin Cancer Res. (2016) 22:2351–8. doi: 10.1158/1078-0432.CCR-15-2013
175. Zou HY, Li Q, Engstrom LD, West M, Appleman V, Wong KA, et al. PF-06463922 is a potent and selective next-generation ROS1/ALK inhibitor capable of blocking crizotinib-resistant ROS1 mutations. Proc Natl Acad Sci U.S.A. (2015) 112:3493–8. doi: 10.1073/pnas.1420785112
176. Song A, Kim TM, Kim DW, Kim S, Keam B, Lee SH, et al. Molecular changes associated with acquired resistance to crizotinib in ROS1-rearranged non-small cell lung cancer. Clin Cancer Res. (2015) 21:2379–87. doi: 10.1158/1078-0432.CCR-14-1350
177. Drilon A, Jenkins C, Iyer S, Schoenfeld A, Keddy C, Davare MA. ROS1-dependent cancers - biology, diagnostics and therapeutics. Nat Rev Clin Oncol. (2021) 18:35–55. doi: 10.1038/s41571-020-0408-9
178. Zhu YC, Lin XP, Li XF, Wu LX, Chen HF, Wang WX, et al. Concurrent ROS1 gene rearrangement and KRAS mutation in lung adenocarcinoma: A case report and literature review. Thorac Cancer. (2018) 9:159–63. doi: 10.1111/1759-7714.12518
179. Park K, Ahn M, Yu C, Kim S, Lin M, Sriuranpong V, et al. ASPIRATION: first-line erlotinib (E) until and beyond RECIST progression (PD) in Asian patients (pts) with EGFR mutation-positive (mut+) NSCLC. Ann Oncol. (2014) 25:iv426. doi: 10.1093/annonc/mdu349.2
180. Ou SH, Jänne PA, Bartlett CH, Tang Y, Kim DW, Otterson GA, et al. Clinical benefit of continuing ALK inhibition with crizotinib beyond initial disease progression in patients with advanced ALK-positive NSCLC. Ann Oncol. (2014) 25:415–22. doi: 10.1093/annonc/mdt572
181. Wu YL, Zhang L, Kim DW, Liu X, Lee DH, Yang JC, et al. Phase ib/II study of capmatinib (INC280) plus gefitinib after failure of epidermal growth factor receptor (EGFR) inhibitor therapy in patients with EGFR-mutated, MET factor-dysregulated non-small-cell lung cancer. J Clin Oncol. (2018) 36:3101–9. doi: 10.1200/JCO.2018.77.7326
182. Yang JJ, Fang J, Shu YQ, Chang JH, Chen GY, He JX, et al. A phase Ib study of the highly selective MET-TKI savolitinib plus gefitinib in patients with EGFR-mutated, MET-amplified advanced non-small-cell lung cancer. Invest New Drugs. (2021) 39:477–87. doi: 10.1007/s10637-020-01010-4
183. Wu YL, Cheng Y, Zhou J, Lu S, Zhang Y, Zhao J, et al. Tepotinib plus gefitinib in patients with EGFR-mutant non-small-cell lung cancer with MET overexpression or MET amplification and acquired resistance to previous EGFR inhibitor (INSIGHT study): an open-label, phase 1b/2, multicentre, randomised trial. Lancet Respir Med. (2020) 8:1132–43. doi: 10.1016/S2213-2600(20)30154-5
184. Sequist LV, Han JY, Ahn MJ, Cho BC, Yu H, Kim SW, et al. Osimertinib plus savolitinib in patients with EGFR mutation-positive, MET-amplified, non-small-cell lung cancer after progression on EGFR tyrosine kinase inhibitors: interim results from a multicentre, open-label, phase 1b study. Lancet Oncol. (2020) 21:373–86. doi: 10.1016/S1470-2045(19)30785-5
185. Hanna NH, Schneider BJ, Temin S, Baker S Jr., Brahmer J, Ellis PM, et al. Therapy for stage IV non-small-cell lung cancer without driver alterations: ASCO and OH (CCO) joint guideline update. J Clin Oncol. (2020) 38:1608–32. doi: 10.1200/JCO.19.03022
186. Paz-Ares L, Luft A, Vicente D, Tafreshi A, Gümüş M, Mazières J, et al. Pembrolizumab plus chemotherapy for squamous non-small-cell lung cancer. N Engl J Med. (2018) 379:2040–51. doi: 10.1056/NEJMoa1810865
187. Socinski MA, Jotte RM, Cappuzzo F, Orlandi F, Stroyakovskiy D, Nogami N, et al. Atezolizumab for first-line treatment of metastatic nonsquamous NSCLC. N Engl J Med. (2018) 378:2288–301. doi: 10.1056/NEJMoa1716948
188. Gogishvili M, Melkadze T, Makharadze T, Giorgadze D, Dvorkin M, Penkov K, et al. Cemiplimab plus chemotherapy versus chemotherapy alone in non-small cell lung cancer: a randomized, controlled, double-blind phase 3 trial. Nat Med. (2022) 28:2374–80. doi: 10.1038/s41591-022-01977-y
189. Sezer A, Kilickap S, Gümüş M, Bondarenko I, Özgüroğlu M, Gogishvili M, et al. Cemiplimab monotherapy for first-line treatment of advanced non-small-cell lung cancer with PD-L1 of at least 50%: a multicentre, open-label, global, phase 3, randomised, controlled trial. Lancet. (2021) 397:592–604. doi: 10.1016/s0140-6736(21)00228-2
190. Borghaei H, Paz-Ares L, Horn L, Spigel DR, Steins M, Ready NE, et al. Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med. (2015) 373:1627–39. doi: 10.1056/NEJMoa1507643
191. Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WE, Poddubskaya E, et al. Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med. (2015) 373:123–35. doi: 10.1056/NEJMoa1504627
192. Gainor JF, Shaw AT, Sequist LV, Fu X, Azzoli CG, Piotrowska Z, et al. EGFR mutations and ALK rearrangements are associated with low response rates to PD-1 pathway blockade in non-small cell lung cancer: A retrospective analysis. Clin Cancer Res. (2016) 22:4585–93. doi: 10.1158/1078-0432.CCR-15-3101
193. Rittmeyer A, Barlesi F, Waterkamp D, Park K, Ciardiello F, von Pawel J, et al. Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. (2017) 389:255–65. doi: 10.1016/S0140-6736(16)32517-X
Keywords: non-small cell lung cancer, targeted therapy, treatment response, drug resistance, tyrosine kinase inhibitor
Citation: Zhang H, Zhang Y, Zhu Y, Dong T and Liu Z (2024) Understanding the treatment response and resistance to targeted therapies in non-small cell lung cancer: clinical insights and perspectives. Front. Oncol. 14:1387345. doi: 10.3389/fonc.2024.1387345
Received: 17 February 2024; Accepted: 25 June 2024;
Published: 11 July 2024.
Edited by:
Yong Zhang, Central South University, ChinaCopyright © 2024 Zhang, Zhang, Zhu, Dong and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zheng Liu, bGl1emhlbmc1MTZAc2luYS5jbg==
†These authors have contributed equally to this work and share first authorship
 Hang Zhang
Hang Zhang Yingying Zhang2†
Yingying Zhang2†