- 1Pathology Unit, Centro di Riferimento Oncologico di Aviano (CRO) Istituto di ricovero e cura a carattere scientifico (IRCCS), Aviano, Italy
- 2Department of Molecular Science and Nanosystems, Ca’ Foscary University, Venice, Italy
Small extracellular vesicles (EVs) in the last 20 years are demonstrated to possess promising properties as potential new drug delivery systems, biomarkers, and therapeutic targets. Moreover, EVs are described to be involved in the most important steps of tumor development and progression including drug resistance. The acquired or intrinsic capacity of cancer cells to resist chemotherapies is one of the greatest obstacles to overcome to improve the prognosis of many patients. EVs are involved in this mechanism by exporting the drugs outside the cells and transferring the drug efflux pumps and miRNAs in recipient cells, in turn inducing drug resistance. In this mini-review, the main mechanisms by which EVs are involved in drug resistance are described, giving a rapid and clear overview of the field to the readers.
Introduction
Extracellular vesicles (EVs) are small cell-released particles with a diameter ranging from 30 to 1,000 nm (1). EVs are a heterogeneous population that can differ in size, properties, and biological function and classified according to their biogenesis pathway (2). In addition, from the first attributing role, consisting in managing cellular waste, nowadays it is well recognized that EVs play a central role in cell–cell communication (3), both in physiological and in pathological conditions, and their cargoes have been distinguished in different components from proteins to miRNA, going through mRNA and lncRNA, among others (4). EVs were also employed as drug delivery systems (DDS) displaying very suitable properties for this purpose and obtaining interesting results in preclinical and clinical trials (2, 5).
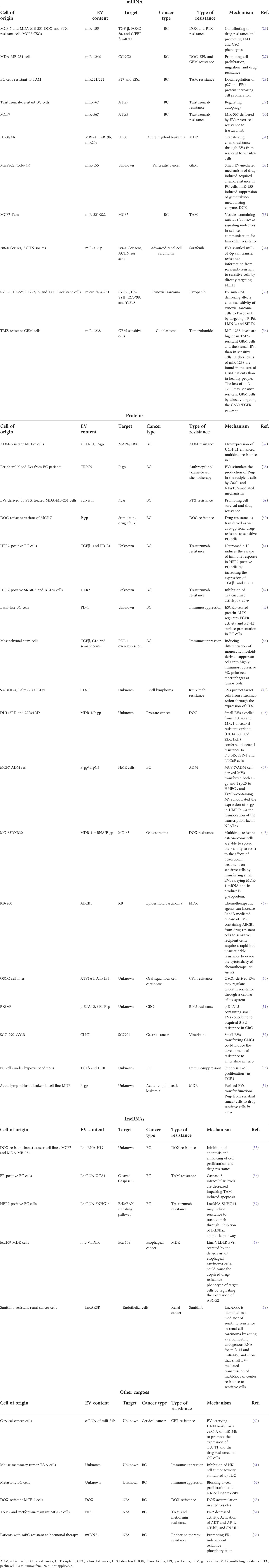
The history of EVs started in the second half of the 1940s in the previous century, when in 1945 Chargaff working on blood coagulation observed small “membrane debris” sedimented at high-speed centrifugation of plasma supernatant (6). The following year, his observation was reported as “a variety of minute breakdown products of blood corpuscules” (7). Twenty-one years later, Peter Wolf described in more detail Chargaff’s remarks, saying that it could be a material “originated from platelets but it is distinguishable from the intact ones”. This claim was confirmed by electron microscopy images that Wolf himself described as “platelet dust” (8). For almost 20 years, other electron microscopy images showed structures with a size under 1,000 nm. In particular, in 1974, Nunez et al. reported, for the first time, structures later called multivesicular bodies (MVB) (9), opening up the path in the identification of a subtype of EVs that originated from MVB, later called exosomes or small EVs (30–150 nm). The biogenesis of these structures was demonstrated to start from late endosomes, which are formed by the inward budding of MVB membranes forming intraluminal vesicles (ILVs), which fuse back with the plasmatic membrane and released by cells as small EVs [later called exosomes (10)] as described by Cliff Harding in 1983 (11). Starting from the early 1980s, many studies on EVs have increased the knowledge in this field and scientists began to deeply understand the multiple biological functions in which EVs are involved. For almost a decade, small EVs were identified as a vehicle to remove unnecessary molecules from cells, like a cellular garbage disposal (12). In the 1990s, small EVs were identified to have an immunological function (13), followed by a large number of studies highlighting that EVs were involved in intercellular communication mechanisms playing a role in physiological or biologically important processes, such as lactation, inflammation, cell proliferation, and neuronal function (14–16). Moreover, other studies showed that EVs are implicated not only in pathological processes, namely, thrombosis (17), diabetes, and atherosclerosis (18), but also in the development and progression of diseases such as liver (19) and neurodegenerative diseases (20) and, recently, in cancer (21, 22). In cancer, many processes like cell proliferation, migration, invasion, epithelial-to-mesenchymal transition, angiogenesis, lymphogenesis, immune suppression, and metastasis (23) are regulated by EVs. In the late 1990s, important studies were published about EVs. Starting with the work of Raposo et al. (13) that demonstrates that EVs derived from immune cells are capable of presenting antigens, other groups started new projects about a new vaccine approach based on EVs. The first approach on vaccines using EVs was explored by Zitvogel et al. in 1998 (24). In their work, the authors described how EVs secreted by dendritic cells loaded with tumor antigens are able to eradicate cancer cells. Based on advances in the next decades, Escudier et al. conducted a clinical trial (25). This work has been a starting point for many studies on the physiological role of EVs and their possible applications as biomarkers, and an opportunity to new therapeutic approaches. In the last few years, lines of evidence for the implication of EVs in the development of anticancer drug resistance have increased and have been extensively studied. This mini-review will focus on the role of EVs in cancer drug resistance exploring and describing the main mechanisms of action through a synthetic description of the major scientific works in the field. Also, a brief description of the most important research papers is provided in Table 1, which aims to give an impression of this field and, overall, to give the readers a rapid and clear overview of the involvement of EVs in drug resistance mechanisms.
The term EVs used in this review, independent of the term used in the article referred to, refers to a mixed population of small EVs ranging from 30 to 200 nm since the available isolation methods are not able to discriminate vesicles originated from different pathways.
EVs mediated drug resistance
The hallmarks of drug resistance are basically summarized in six points: (1) alteration of drug targets (2), activation of drug pumps, (3) detoxification mechanisms, (4) reduced susceptibility to apoptosis, (5) increased ability to repair DNA damage, and (6) altered proliferation. Also, local modifications of stroma, tumor microenvironment (TME), and local immunity could contribute to the development of resistance (66). Keeping in mind these notions, EVs are involved in cell–cell communication and cargo sharing/delivery, and these characteristics have been associated with chemo- and targeted therapies’ resistance as detailed here. In the next paragraphs, the most important mechanisms by which EVs regulate drug resistance will be described.
Activation of drug-efflux pumps
Efflux pump mechanisms are physiologically important in many processes such as toxin clearance from the gastrointestinal tract, elimination of bile from the hepatocytes, effective functioning of the blood–brain and placental barrier, and the renal excretion of drugs. In drug-resistant tumors, the overexpression of these proteins (67) allow the cells to reduce the intracellular drug concentration to a sublethal dose. Many research papers described the role of EVs in the transferring of drug efflux pumps from resistant to sensitive cancer cells. Among the delivered proteins are frequently described ATP-binding cassette (ABC) family, like P-glycoprotein (P-gp, MDR1, and ABCB1), breast cancer (BC)-resistant proteins (ABCG2, BRCP, and ABCA3), and multidrug-resistant protein 1 (MRP-1) (45, 46, 68–72). The mechanism by which EVs transfer proteins among cells is commonly called EVs-mediated horizontal transfer of drug efflux pumps. BC cells were able to export doxorubicin in the extracellular medium by EVs shedding, thus reducing intracellular accumulation of the drug. Moreover, EVs mediate the transfer of functional proteins or RNAs (miRNA and mRNA) that modulate the expression and function of P-gp. The P-gp is found to be overproduced in cancer cells to remove cytotoxic drugs from cells and is demonstrated to cover a pivotal role in drug resistance together with TRPC (transient receptor potential channel) proteins (73).
EVs could also transfer drug metabolizing enzymes to inactivate drugs. Yang et al. described that the expression of GSTP1 (glutathione S-transferase P1), an enzyme belonging to phase II drug-metabolizing proteins, was higher in doxorubicin-resistant cells and in their EVs, which are capable of transferring the GSTP1 enzyme to sensitive cells (74). Accordingly, a high level of GSTP1 in circulating EVs may be an indication of a drug-resistant profile and could be used as a drug resistance predictive marker (74) as already demonstrated for the expression of GSTP1 on tumor cells (75, 76).
Cell viability could even be enhanced by EVs’ transferring of pro-survival factors like cell surface receptors, miRNAs, and cellular proteins. These cargoes could improve cell viability by decreasing apoptosis and activating proliferative signals (77–81).
Intercellular communication between the microenvironment and tumor cells
As described in the Introduction, EVs are involved in cell–cell communication. This mechanism could play a role in the bidirectional crosstalk between tumoral and stromal cells also regarding drug resistance mechanisms. EVs derived from cancer-associated fibroblasts (CAFs) are described to be involved in drug resistance in different types of tumor. In colorectal cancer, EVs derived from CAFs are able to induce chemoresistance to 5-FU and oxaliplatin both in vitro and on patient-derived mouse xenografts (82). CAF-EVs are able to promote stemness and resistance in CRC cells in vitro and in vivo also by transferring the lncRNA H19 (83), H19 is an activator of β-catenin pathway. Previous studies demonstrated that the β-catenin pathway is involved in tumor progression and drug resistance (84–86). Interestingly, CAFs are naturally resistant to gemcitabine and their EVs transfer the gemcitabine chemoresistance phenotype in pancreatic ductal adenocarcinoma (PDAC) by delivering the SNAIL mRNA that increase SNAIL protein expression promoting proliferation and drug resistance (87). A recent work highlighted that CAF-EVs are involved in oxaliplatin resistance in CRC by transferring the CCAL (colorectal cancer-associated lncRNA) and activating the β-catenin pathway (88). CCAL interacts with mRNA-stabilizing protein HuR (human antigen R) increasing β-catenin mRNA and protein levels. Another work described the effect of stromal EVs in multiple myeloma cells inducing resistance to bortezomib, which could be linked to the activation of JNK, p38, p53, and Akt pathways (89). The release of EVs from mesenchymal stem cells carrying miR-222/miR-223 is linked to drug resistance in BC cells (90). ZEB1 mRNA encapsulated in EVs derived from mesenchymal transformed lung cells can transfer gemcitabine and cisplatin chemoresistance and the mesenchymal phenotypes to epithelial NSCLC cell line (91).
RNA (miRNA, lncRNA, and mRNA)-mediated drug resistance
Micro RNAs are small noncoding RNAs of 13–29 nucleotides involved in gene regulation and different biological and pathological processes, including the formation and development of tumors and drug resistance. In the last years, miRNAs are one of the most studied cargoes of EVs. As described, drug resistance mechanisms are heterogeneous and complex, and most of them are also regulated by miRNAs (92). miRNAs could promote drug resistance through the activation of metabolizing enzymes, in turn favoring drug inactivation or the expression of drug efflux pumps. The transfer of miRNA-365 by tumor-associated macrophage (TAM)-derived EVs to pancreatic ductal cells is described to induce resistance to gemcitabine in pancreatic adenocarcinoma by upregulating the triphosphate-nucleotide pool in cancer cells and inducing the cytidine deaminase enzyme that is able to inactivate gemcitabine (93). As mentioned, EV miRNAs could regulate the expression of ABC transporters that are involved in the efflux of intracellular drugs. It is described that, in ovarian cancer (OC) cells, there is an inverse correlation between the expression of Caveolin 1 (Cav1) and ABCB1, and this proportion is supposed to be driven by Cav1 (94, 95). Kanlikilicer et al. demonstrated that Cav1 levels in macrophages when co-cultured with OC cells are selectively dysregulated by the release of miR-1246 via EVs by OC cells. miR-1246 secreted in EVs inhibits the expression of Cav1 and upregulates ABCB1 expression to induce tumor-promoting phenotype and drug resistance in vitro and in vivo (96). As described for the transport of drug efflux pumps, even miRNAs could display a double-action behavior in the occurrence and development of drug resistance. Some miRNAs could have a positive effect on drug resistance, enhancing drug sensitivity in cancer cells. An analysis conducted by Liu et al. showed that EVs containing miR-128-3p were able to downregulate the expression of the MDR5 protein thus enhancing oxaliplatin sensitivity in resistant colorectal cancer cells (97). Another way to inhibit drug resistance is the regulation of glycolysis. Cancer metastasis, invasion, and drug resistance are also dependent on the anabolic profile of tumor cells that promotes the decrease in extracellular pH leading to the reduction of cytotoxic T-cell function in the TME acquiring strong survival advantages (98, 99). The GLUT protein family is involved in the intracellular uptake of glucose (100) and the regulation of glycolysis could be a strategy to contrast drug resistance (101). GLUT1 is demonstrated to be overexpressed in several tumors (102, 103), and its activation is associated with the regulation of mTOR. A decreased expression of mir-100-5p is described to be involved in drug resistance in many tumors. mTOR is the target gene of miR-100-5p that decreases its expression, enhancing drug sensitivity in cancer cells (104).
mRNA-mediated EVs are another player in the resistance process. Cao et al. demonstrated that EVs containing the DNMT1 mRNA (DNA methyltranferase 1) induce the overexpression of this enzyme in the recipient cells, playing an important role in the cisplatin resistance mediated by EVs in the xenograft model (105). In this research work, the underlying mechanism is not investigated, but it could be speculated that the dysregulation of Wnt and PI3K/AKT/mTOR signaling pathways, caused by an altered methylation status in a variety of genes, was described to be associated with resistance to standard treatments in many types of cancer (106). It was also demonstrated that BC cells resistant to doxorubicin possess an increased level of mRNA coding for a detoxifying enzyme (GSTP1) and EVs derived from those cells are capable of transferring the mRNA to sensitive cells and inducing resistance (74). In vitro and in vivo experiments demonstrated that normal astrocytes can protect glioma cells from apoptosis induced by Temozolomide (TMZ) through the transfer of the mRNA of O-6-methylguaninene-DNA methyltransferase (MGMT) by EVs (107). EVs transfer of Zinc finger E-box homeobox 1 (ZEB1), a transcription factor involved in the epithelial-to-mesenchymal transition (EMT) process, induces the mesenchymal phenotype and drug resistance in recipient lung cancer cells (91, 108). In particular, this work described how EVs derived from mesenchymal oncogenically transformed lung cells can transfer chemoresistance and the mesenchymal phenotype to recipient cells.
LncRNA delivered by EVs often serves as competing endogenous RNA (ceRNA) to help miRNA in their drug resistance regulatory mechanisms (109). LncRNAs have been identified to be involved in cancer drug resistance by affecting the expression of drug metabolizing enzymes (110). Two studies described that EVs transferring lncRNA linc-ROR (111) and linc-VLDRLR (112) induced sorafenib and doxorubicin resistance in HepG2 cells (hepatocellular carcinoma) by activating the TGF-β pathway (111) and increasing the expression of ABCG2 (112). LncRNA urothelial carcinoma-associated 1 (UCA1) in NSCLC is associated with the modulation of a gefitinib-resistant phenotype by decreasing the expression of miR-143 and consequently increasing the expression of its target FOS-like 2 (113). LncRNA SBF2-AS1 is identified to be ceRNA of miR-151 and is involved in the mechanism of DNA repair that is one of the leading mechanisms of resistance to TMZ in neurological cancers (114). In glioblastoma patients, the presence of EVs lncRNA SBF-AS1 in the serum was found to be associated with TMZ resistance (115). LncRNA could also act by regulating some RNA-binding proteins as demonstrated for AFAP1-AS1 associated with shorter time survival of HER-2-positive BC patients linked to trastuzumab resistance. AFAP1-AS1 is responsible for trastuzumab resistance by upregulating HER-2 expression through the binding of the RNA binding protein AU-binding factor 1 (116).
EVs and possible applications as biomarkers of tumor therapy resistance
EVs can be isolated from various types of body fluids including blood, urine, and saliva. It is demonstrated that in the cancer patient population, the amount of EVs present in the blood is more than double compared to healthy individuals (117), suggesting that they could be new biomarker candidates (118). A correlation between serum EVs containing miR-146-5p could predict the efficacy of cisplatin in NSCLC patients in advanced stages and utilized for the real-time monitoring of drug resistance manifestation (119). Xiao et al. described that EVs derived from the serum of drug-resistant CRC patients (5-FU resistance) are enriched in TAG72 (tumor-associated glycoprotein 72) (120, 121). In BC preclinical models, it is demonstrated that the cargoes of EVs are influenced by the stress induced from drugs and could be correlated to the transfer of resistance in metastatic sites mediated by the Pg-P protein (40) or by miR-423-5p (122). Leukemia-derived EVs are described to induce IL-8 release in bone marrow stromal cells, thus protecting the cells from the effects induced by chemotherapy (123). A high level of IL8 promotes the expression of Pg-P and is required for the expression of the MDR profile in BC (124) and, in renal cancer, is described to be associated with sunitinib resistance (125). According to the described implication of EVs in drug resistance, it could be useful to set up methods for rapid isolation and characterization of tumor-derived EVs to improve the personalization of the therapies and to predict the drug response of the patient. Moreover, targeted drugs against tumor-derived EVs should be studied to reduce their non-beneficial effects as described in the next paragraph.
Targeting EVs to reduce cancer chemoresistance
Considering the importance of EVs in the regulation of chemoresistance, a few drugs were utilized to inhibit their production, release, or action.
It is described that drug-resistant cancer cells could produce an increased number of EVs than their drug-sensitive counterpart, thus contributing to the spread of resistance (45, 126–128). Some studies reported that, in drug-resistant cells, there is a direct association between the presence of drug resistance mediators and molecules involved in the production of EVs. For instance, Annexin A3 is a protein involved in OC platinum resistance and is also demonstrated to have a role in the EVs’ production (129, 130). In the last years, an increasing number of studies investigated the possibility to inhibit the release of EVs from cancer cells. GW4869 is an inhibitor of the neutral sphingomyelinase (131) and is able to sensitize cisplatin-resistant OC cells by reducing EVs trafficking (105). Moreover, rhamnose-emodin is a molecule that is described to reduce the secretion of EVs from doxorubicin-resistant BC cells, thus reducing the expression of EVs miRNAs involved in chemoresistance (132). Therapeutic targeted antibodies against cell surface receptors may be neutralized by EVs interaction. Aung et al.’s research group described how Rituximab (anti-CD20) is quenched by EVs expressing the target protein. The authors also demonstrate that by blocking EVs biogenesis with indomethacin, the therapeutic benefits of the therapy were restored (45). In another work, indomethacin is used to block EVs secretion in order to increase the amount of cytoplasmatic doxorubicin, its accumulation in the nucleus, and cytotoxicity (133). In a CRC model, it was demonstrated that the interaction mediated by EVs between cancer stem cells and fibroblasts promoted the resistance to 5-FU and oxaliplatin and can be reverted by blocking the release of EVs (82). Xie et al. developed functionalized silica mesoporous nanoparticles (NPs) able to selectively bind EGFR+-EVs derived from NSCLC through aptamer recognition. NPs, after binding to EVs in the bloodstream, are delivered to the liver and excreted in the intestinal tract to be removed from the organism. It was demonstrated that by employing this system, the in vivo cancer metastatic overgrowth could be reduced (134).
Conclusions
The discovery of new cancer therapies is a stimulating topic that is investigated by a lot of researchers all over the world. The need for new therapeutic approaches is required because of the interpatient variability in terms of drug response, and also the development of drug resistance represents a very hard hurdle to overcome. Drug resistance appears in almost all types of cancer, and the underlying mechanisms are not yet clearly understood. In the last few years, the wide implication of EVs in drug resistance has been investigated, and in this manuscript, the major implications in this process are described and summarized. Although many described experiments are limited to preclinical and often to an in vitro stage, it is necessary to deeply investigate the roles of EVs in cancer drug resistance for many important aspects.
First of all, the involvement of EVs in drug resistance and their profiling could be exploited in the clinical approach to define new hallmarks of prognosis of drug response avoiding invasive procedures. On the other hand, as already explained, clarifying the role of EVs on drug resistance could stimulate the development of new anti-cancer strategies based on EVs targeting to revert drug resistance. Most importantly, cancer-released EVs should be deeply characterized, and their peculiar properties should be investigated. In this way, the development of new targeted strategies able to discriminate tumor-derived EVs could be set up. Moreover, the employment of artificial EVs could be considered in order to revert drug sensitivity (135, 136). EVs have already been described to possess very suitable properties as DDS to be loaded with different cargoes (drugs, miRNA, and proteins) displaying high biocompatibility, and the capacity to target cells is 10 times higher compared to liposomes of the same size (137–140). EVs could also represent a new DDS against neurological malignancies due to their ability to cross the blood–brain barrier (141, 142). Due to the possibility to produce engineered EVs in vitro, they could be developed to target different types of malignancies. There is also the possibility of studying artificial EVs for therapeutic employment with the advantage of producing standardized EVs with a defined content to facilitate the transition into a clinical application.
Author contributions
SP wrote and revised the text. FR and VC conceptualized the work and edit the final version. All authors contributed to the article and approved the submitted version.
Funding
This research was funded by Italian Ministry of Health – Ricerca Corrente.
Acknowledgments
We thank all the colleagues from “Aviano Centro di Riferimento Oncologico” and from Ca’ Foscari University for the constructive discussions about the topic of the review helping us to develop the final work.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2022.948843/full#supplementary-material
References
1. Doyle L, Wang M. Overview of extracellular vesicles, their origin, composition, purpose, and methods for exosome isolation and analysis. Cells (2019) 8:727. doi: 10.3390/CELLS8070727
2. Palazzolo S, Memeo L, Hadla M, Duzagac F, Steffan A, Perin T, et al. Cancer extracellular vesicles: Next-generation diagnostic and drug delivery nanotools. Cancers (Basel) (2020) 12:1–36. doi: 10.3390/CANCERS12113165
3. Tkach M, Théry C. Communication by extracellular vesicles: Where we are and where we need to go. Cell (2016) 164:1226–32. doi: 10.1016/j.cell.2016.01.043
4. Abels ER, Breakefield XO. Introduction to extracellular vesicles: Biogenesis, RNA cargo selection, content, release, and uptake. Cell Mol Neurobiol (2016) 36:301–12. doi: 10.1007/S10571-016-0366-Z
5. Herrmann IK, Wood MJA, Fuhrmann G. Extracellular vesicles as a next-generation drug delivery platform. Nat Nanotechnol (2021) 16:748–59. doi: 10.1038/S41565-021-00931-2
6. Chargaff E. Cell structure and the problem of blood coagulation. J Biol Chem (1945) 160:351–9. doi: 10.1016/S0021-9258(18)43131-6
7. Chargaff E, West R. The biological significance of the thromboplastic protein of blood. J Biol Chem (1946) 166:189–97. doi: 10.1016/s0021-9258(17)34997-9
8. Wolf P. The nature and significance of platelet products in human plasma. Br J Haematol (1967) 13:269–88. doi: 10.1111/j.1365-2141.1967.tb08741.x
9. Nunez EA, Wallis J, Gershon MD. Secretory processes in follicular cells of the bat thyroid. III. the occurrence of extracellular vesicles and colloid droplets during arousal from hibernation. Am J Anat (1974) 141:179–201. doi: 10.1002/aja.1001410203
10. Pan BT, Johnstone RM. Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: Selective externalization of the receptor. Cell (1983) 33:967–78. doi: 10.1016/0092-8674(83)90040-5
11. Harding C, Heuser J, Stahl P. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol (1983) 97:329–39. doi: 10.1083/jcb.97.2.329
12. Johnstone RM. The Jeanne manery-Fisher memorial lecture 1991. maturation of reticulocytes: formation of exosomes as a mechanism for shedding membrane proteins. Biochem Cell Biol (1992) 70:179–90. doi: 10.1139/o92-028
13. Raposo G, Nijman HW, Stoorvogel W, Liejendekker R, Harding CV, Melief CJ, et al. B lymphocytes secrete antigen-presenting vesicles. J Exp Med (1996) 183:1161–72. doi: 10.1084/jem.183.3.1161
14. Gu Y, Li M, Wang T, Liang Y, Zhong Z, Wang X, et al. Lactation-related microRNA expression profiles of porcine breast milk exosomes. PloS One (2012) 7. doi: 10.1371/journal.pone.0043691
15. Azevedo LCP, Janiszewski M, Pontieri V, Pedro M de A, Bassi E, Tucci PJF, et al. Platelet-derived exosomes from septic shock patients induce myocardial dysfunction. Crit Care (2007) 11. doi: 10.1186/cc6176
16. Liu W, Bai X, Zhang A, Huang J, Xu S, Zhang J. Role of exosomes in central nervous system diseases. Front Mol Neurosci (2019) 12:240. doi: 10.3389/fnmol.2019.00240
17. Zifkos K, Dubois C, Schäfer K. Extracellular vesicles and thrombosis: Update on the clinical and experimental evidence. Int J Mol Sci (2021) 22. doi: 10.3390/IJMS22179317
18. Yanan W, Yingyu X, Ao Z, Mingyang W, Zihan F, Junping Z. Exosomes: An emerging factor in atherosclerosis. BioMed Pharmacother (2019) 115. doi: 10.1016/j.biopha.2019.108951
19. Masyuk AI, Masyuk TV, Larusso NF. Exosomes in the pathogenesis, diagnostics and therapeutics of liver diseases. J Hepatol (2013) 59:621–5. doi: 10.1016/j.jhep.2013.03.028
20. Rastogi S, Sharma V, Bharti PS, Rani K, Modi GP, Nikolajeff F, et al. The evolving landscape of exosomes in neurodegenerative diseases: Exosomes characteristics and a promising role in early diagnosis. Int J Mol Sci (2021) 22:1–31. doi: 10.3390/ijms22010440
21. Tai YL, Chen KC, Hsieh JT, Shen TL. Exosomes in cancer development and clinical applications. Cancer Sci (2018) 109:2364–74. doi: 10.1111/cas.13697
22. Dai J, Su Y, Zhong S, Cong L, Liu B, Yang J, et al. Exosomes: key players in cancer and potential therapeutic strategy. Signal Transduct Target Ther (2020) 5. doi: 10.1038/s41392-020-00261-0
23. Xu R, Rai A, Chen M, Suwakulsiri W, Greening DW, Simpson RJ. Extracellular vesicles in cancer — implications for future improvements in cancer care. Nat Rev Clin Oncol (2018) 15:617–38. doi: 10.1038/S41571-018-0036-9
24. Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D, et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med (1998) 4:594–600. doi: 10.1038/nm0598-594
25. Escudier B, Dorval T, Chaput N, André F, Caby M-P, Novault S, et al. Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: results of thefirst phase I clinical trial. J Transl Med (2005) 3:10. doi: 10.1186/1479-5876-3-10
26. Santos JC, Lima NDS, Sarian LO, Matheu A, Ribeir ML, Derchain SFM. Exosome-mediated breast cancer chemoresistance via miR-155 transfer. Sci Rep (2018 ) 8(1):829. doi:10.1038/s41598-018-19339-5
27. Li XJ, Ren ZJ, Tang JH, Yu Q Exosomal MicroRNA MiR-1246 promotes cell proliferation, invasion and drug resistance by targeting CCNG2 in breast cancer Cell Physiol Biochem (2017) 44:1741–8. doi: 10.1159/000485780
28. Semina SE, Scherbakov AM, Vnukova AA, Bagrov DV, Evtushenko EG, Safronova VM, et al. Exosome-mediated transfer of cancer cell resistance to antiestrogen drugs. Molecules (2018) 23. doi: 10.3390/MOLECULES23040829
29. Martinez VG, O’Neill S, Salimu J, Breslin S, Clayton A, Crown J, et al. Resistance to HER2-targeted anti-cancer drugs is associated with immune evasion in cancer cells and their derived extracellular vesicles. Oncoimmunology (2017) 6. doi: 10.1080/2162402X.2017.1362530
30. Han M, Hu J, Lu P, Cao H, Yu C, Li X, et al. Exosome-transmitted miR-567 reverses trastuzumab resistance by inhibiting ATG5 in breast cancer. Cell Death Dis (2020) 11. doi: 10.1038/S41419-020-2250-5
31. Bouvy C, Wannez A, Laloy J, Chatelain C, Dogné JM. Transfer of multidrug resistance among acute myeloid leukemia cells via extracellular vesicles and their microRNA cargo. Leuk Res (2017) 62:70–6. doi: 10.1016/J.LEUKRES.2017.09.014
32. Patel GK, Khan MA, Bhardwaj A, Srivastava SK, Zubair H, Patton MC, et al. Exosomes confer chemoresistance to pancreatic cancer cells by promoting ROS detoxification and miR-155-mediated suppression of key gemcitabine-metabolising enzyme, DCK. Br J Cancer (2017) 116:609–19. doi: 10.1038/BJC.2017.18
33. Wei Y, Lai X, Yu S, Chen S, Ma Y, Zhang Y, et al. Exosomal miR-221/222 enhances tamoxifen resistance in recipient ER-positive breast cancer cells. Breast Cancer Res Treat (2014) 147:423–31. doi: 10.1007/S10549-014-3037-0
34. He J, He J, Min L, He Y, Guan H, Wang J, et al. Extracellular vesicles transmitted miR-31-5p promotes sorafenib resistance by targeting MLH1 in renal cell carcinoma. Int J Cancer (2020) 146:1052–63. doi: 10.1002/IJC.32543
35. Shiozawa K, Shuting J, Yoshioka Y, Ochiya T, Kondo T. Extracellular vesicle-encapsulated microRNA-761 enhances pazopanib resistance in synovial sarcoma. Biochem Biophys Res Commun (2018) 495:1322–7. doi: 10.1016/J.BBRC.2017.11.164
36. Yin J, Zeng A, Zhang Z, Shi Z, Yan W, You Y. Exosomal transfer of miR-1238 contributes to temozolomide-resistance in glioblastoma. EBioMedicine (2019) 42:238–51. doi: 10.1016/J.EBIOM.2019.03.016
37. Ning K, Wang T, Sun X, Zhang P, Chen Y, Jin J, et al. UCH-L1-containing exosomes mediate chemotherapeutic resistance transfer in breast cancer. J Surg Oncol (2017) 115:932–40. doi: 10.1002/JSO.24614
38. Wang T, Ning K, Lu TX, Sun X, Jin L, Qi X, et al. Increasing circulating exosomes-carrying TRPC5 predicts chemoresistance in metastatic breast cancer patients. Cancer Sci (2017) 108:448–54. doi: 10.1111/CAS.13150
39. Kreger BT, Johansen ER, Cerione RA, Antonyak MA. The enrichment of survivin in exosomes from breast cancer cells treated with paclitaxel promotes cell survival and chemoresistance. Cancers (Basel) (2016) 8. doi: 10.3390/CANCERS8120111
40. Lv M, Zhu X, Chen W, Zhong S, Hu Q, Ma T, et al. Exosomes mediate drug resistance transfer in MCF-7 breast cancer cells and a probable mechanism is delivery of p-glycoprotein. Tumour Biol (2014) 35:10773–9. doi: 10.1007/s13277-014-2377-z
41. Martinez VG, O'Neill S, Salimu J, Breslin S, Clayton A, Crown J, et al. Resistance to HER2-targeted anti-cancer drugs is associated with immune evasion in cancer cells and their derived extracellular vesicles. Oncoimmunology (2017) 6(1):e1362530. doi: 10.1080/2162402X.2017.1362530
42. Ciravolo V, Huber V, Ghedini GC, Venturelli E, Bianchi F, Campiglio M, et al. Potential role of HER2-overexpressing exosomes in countering trastuzumab-based therapy. J Cell Physiol (2012) 227(2):658–67. doi: 10.1002/jcp.22773
43. Monypenny J, Milewicz H, Flores-Borja F, Weitsman G, Cheung A, Chowdhury R, et al. ALIX regulates tumor-mediated immunosuppression by controlling EGFR activity and PD-L1 presentation. Cell Rep (2018) 24:630–41. doi: 10.1016/J.CELREP.2018.06.066
44. Biswas S, Mandal G, Roy Chowdhury S, Purohit S, Payne KK, Anadon C, et al. Exosomes produced by mesenchymal stem cells drive differentiation of myeloid cells into immunosuppressive M2-polarized macrophages in breast cancer. J Immunol (2019) 203:3447–60. doi: 10.4049/JIMMUNOL.1900692
45. Aung T, Chapuy B, Vogel D, Wenzel D, Oppermann M, Lahmann M, et al. Exosomal evasion of humoral immunotherapy in aggressive b-cell lymphoma modulated by ATP-binding cassette transporter A3. Proc Natl Acad Sci U.S.A. (2011) 108:15336–41. doi: 10.1073/pnas.1102855108
46. Corcoran C, Rani S, O’Brien K, O’Neill A, Prencipe M, Sheikh R, et al. Docetaxel-resistance in prostate cancer: evaluating associated phenotypic changes and potential for resistance transfer via exosomes. PloS One (2012) 7:e50999. doi: 10.1371/journal.pone.0050999
47. Dong Y, Pan Q, Jiang L, Chen Z, Zhang F, Liu Y, et al. Tumor endothelial expression of p-glycoprotein upon microvesicular transfer of TrpC5 derived from adriamycin-resistant breast cancer cells. Biochem Biophys Res Commun (2014) 446:85–90. doi: 10.1016/J.BBRC.2014.02.076
48. Torreggiani E, Roncuzzi L, Perut F, Zini N, Baldini N. Multimodal transfer of MDR by exosomes in human osteosarcoma. Int J Oncol (2016) 49:189–96. doi: 10.3892/IJO.2016.3509
49. Wang X, Qiao D, Chen L, Xu M, Chen S, Huang L, et al. Chemotherapeutic drugs stimulate the release and recycling of extracellular vesicles to assist cancer cells in developing an urgent chemoresistance. Mol Cancer (2019) 18. doi: 10.1186/S12943-019-1114-Z
50. Khoo XH, Paterson IC, Goh BH, Lee WL. Cisplatin-resistance in oral squamous cell carcinoma: Regulation by tumor cell-derived extracellular vesicles. Cancers (Basel) (2019) 11. doi: 10.3390/CANCERS11081166
51. Zhang Q, Liu RX, Chan KW, Hu J, Zhang J, Wei L, et al. Exosomal transfer of p-STAT3 promotes acquired 5-FU resistance in colorectal cancer cells. J Exp Clin Cancer Res (2019) 38. doi: 10.1186/S13046-019-1314-9
52. Zhao K, Wang Z, Li X, lu LJ, Tian L, Chen Jq. Exosome-mediated transfer of CLIC1 contributes to the vincristine-resistance in gastric cancer. Mol Cell Biochem (2019) 462:97–105. doi: 10.1007/S11010-019-03613-9
53. Rong L, Li R, Li S, Luo R. Immunosuppression of breast cancer cells mediated by transforming growth factor-β in exosomes from cancer cells. Oncol Lett (2016) 11:500–4. doi: 10.3892/OL.2015.3841
54. Bebawy M, Combes V, Lee E, Jaiswal R, Gong J, Bonhoure A, et al. Membrane microparticles mediate transfer of p-glycoprotein to drug sensitive cancer cells. Leukemia (2009) 23:1643–9. doi: 10.1038/LEU.2009.76
55. Wang X, Pei X, Guo G, Qian X, Dou D, Zhang Z, et al. Exosome-mediated transfer of long noncoding RNA H19 induces doxorubicin resistance in breast cancer. J Cell Physiol (2020) 235:6896–904. doi: 10.1002/JCP.29585
56. Xu CG, Yang MF, Ren YQ, Wu CH, Wang LQ. Exosomes mediated transfer of lncRNA UCA1 results in increased tamoxifen resistance in breast cancer cells. Eur Rev Med Pharmacol Sci (2016) 20:4362–8.
57. Dong H, Wang W, Chen R, Zhang Y, Zou K, Ye M, et al. Exosome-mediated transfer of lncRNA SNHG14 promotes trastuzumab chemoresistance in breast cancer. Int J Oncol (2018) 53(3):1013–26. doi: 10.3892/ijo.2018.4467
58. Chen YT, Liu L, Li J, Du Y, Wang J, Liu JH. Effects of long noncoding RNA (linc-VLDLR) existing in extracellular vesicles on the occurrence and multidrug resistance of esophageal cancer cells. Pathol Res Pract (2019) 215:470–7. doi: 10.1016/J.PRP.2018.12.033
59. Qu L, Ding J, Chen C, Wu ZJ, Liu B, Gao Y, et al. Exosome-transmitted lncARSR promotes sunitinib resistance in renal cancer by acting as a competing endogenous RNA. Cancer Cell (2016) 29:653–68. doi: 10.1016/J.CCELL.2016.03.004
60. Luo X, Wei J, Yang FL, Pang XX, Shi F, Wei YX, et al. Exosomal lncRNA HNF1A-AS1 affects cisplatin resistance in cervical cancer cells through regulating microRNA-34b/TUFT1 axis. Cancer Cell Int (2019) 19. doi: 10.1186/S12935-019-1042-4
61. Liu C, Yu S, Zinn K, Wang J, Zhang L, Jia Y, et al. Murine mammary carcinoma exosomes promote tumor growth by suppression of NK cell function. J Immunol (2006) 176:1375–85. doi: 10.4049/JIMMUNOL.176.3.1375
63. Shedden K, Xie XT, Chandaroy P, Chang YT, Rosania GR. Expulsion of small molecules in vesicles shed by cancer cells: Association with gene expression and chemosensitivity profiles. Cancer Res (2003) 63:4331–7.
64. Muluhngwi P, Klinge CM. Identification of miRNAs as biomarkers for acquired endocrine resistance in breast cancer. Mol Cell Endocrinol (2017) 456:76–86. doi: 10.1016/J.MCE.2017.02.004
65. Sansone P, Savini C, Kurelac I, Chang Q, Amato LB, Strillacci A, et al. Packaging and transfer of mitochondrial DNA via exosomes regulate escape from dormancy in hormonal therapy-resistant breast cancer. Proc Natl Acad Sci USA (2017) 114(43):E9066–75. doi: 10.1073/pnas.1704862114
66. Kurywchak P, Tavormina J, Kalluri R. The emerging roles of exosomes in the modulation of immune responses in cancer. Genome Med (2018) 10. doi: 10.1186/S13073-018-0535-4
67. Ughachukwu P, Unekwe P. Efflux pump-mediated resistance in chemotherapy. Ann Med Health Sci Res (2012) 2:191–8. doi: 10.4103/2141-9248.105671
68. Li J, Li ZN, Yu LC, Bao QL, Wu JR, Shi SB, et al. Association of expression of MRP1, BCRP, LRP and ERCC1 with outcome of patients with locally advanced non-small cell lung cancer who received neoadjuvant chemotherapy. Lung Cancer (2010) 69:116–22. doi: 10.1016/j.lungcan.2009.09.013
69. Bagnoli M, Beretta GL, Gatti L, Pilotti S, Alberti P, Tarantino E, et al. Clinicopathological impact of ABCC1/MRP1 and ABCC4/MRP4 in epithelial ovarian carcinoma. BioMed Res Int (2013) 2013. doi: 10.1155/2013/143202
70. Cole SPC. Multidrug resistance protein 1 (mrp1, abcc1), a ‘multitasking’ atp-binding cassette (abc,) transporter. J Biol Chem (2014) 289:30880–8. doi: 10.1074/jbc.R114.609248
71. Munoz M, Henderson M, Haber M, Norris M. Role of the MRP1/ABCC1 multidrug transporter protein in cancer. IUBMB Life (2007) 59:752–7. doi: 10.1080/15216540701736285
72. Lu JF, Luk F, Gong J, Jaiswal R, Grau GER, Bebawy M. Microparticles mediate MRP1 intercellular transfer and the re-templating of intrinsic resistance pathways. Pharmacol Res (2013) 76:77–83. doi: 10.1016/j.phrs.2013.07.009
73. Ma X, Cai Y, He D, Zou C, Zhang P, Lo CY, et al. Transient receptor potential channel TRPC5 is essential for p-glycoprotein induction in drug-resistant cancer cells. Proc Natl Acad Sci U.S.A. (2012) 109:16282–7. doi: 10.1073/PNAS.1202989109
74. Yang Sj, Wang Dd, Li J, Xu H, Shen H, Chen X, et al. Predictive role of GSTP1-containing exosomes in chemotherapy-resistant breast cancer. Gene (2017) 623:5–14. doi: 10.1016/j.gene.2017.04.031
75. Arai T, Miyoshi Y, Kim SJ, Akazawa K, Maruyama N, Taguchi T, et al. Association of GSTP1 expression with resistance to docetaxel and paclitaxel in human breast cancers. Eur J Surg Oncol (2008) 34:734–8. doi: 10.1016/j.ejso.2007.07.008
76. Miyake T, Nakayama T, Naoi Y, Yamamoto N, Otani Y, Kim SJ, et al. GSTP1 expression predicts poor pathological complete response to neoadjuvant chemotherapy in ER-negative breast cancer. Cancer Sci (2012) 103:913–20. doi: 10.1111/J.1349-7006.2012.02231.X
77. Khan S, Jutzy JMS, Aspe JR, McGregor DW, Neidigh JW, Wall NR. Survivin is released from cancer cells via exosomes. Apoptosis (2011) 16:1–12. doi: 10.1007/s10495-010-0534-4
78. Al-Nedawi K, Meehan B, Micallef J, Lhotak V, May L, Guha A, et al. Intercellular transfer of the oncogenic receptor EGFRvIII by microvesicles derived from tumour cells. Nat Cell Biol (2008) 10:619–24. doi: 10.1038/ncb1725
79. Song X, Ding Y, Liu G, Yang X, Zhao R, Zhang Y, et al. Cancer cell-derived exosomes induce mitogen-activated protein kinase-dependent monocyte survival by transport of functional receptor tyrosine kinases. J Biol Chem (2016) 291:8453–64. doi: 10.1074/jbc.M116.716316
80. Valenzuela MMA, Ferguson Bennit HR, Gonda A, Diaz Osterman CJ, Hibma A, Khan S, et al. Exosomes secreted from human cancer cell lines contain inhibitors of apoptosis (IAP). Cancer Microenviron (2015) 8:65–73. doi: 10.1007/s12307-015-0167-9
81. Khan S, Aspe JR, Asumen MG, Almaguel F, Odumosu O, Acevedo-Martinez S, et al. Extracellular, cell-permeable survivin inhibits apoptosis while promoting proliferative and metastatic potential. Br J Cancer (2009) 100:1073–86. doi: 10.1038/sj.bjc.6604978
82. Hu Y, Yan C, Mu L, Huang K, Li X, Tao D, et al. Fibroblast-derived exosomes contribute to chemoresistance through priming cancer stem cells in colorectal cancer. PloS One (2015) 10. doi: 10.1371/journal.pone.0125625
83. Ren J, Ding L, Zhang D, Shi G, Xu Q, Shen S, et al. Carcinoma-associated fibroblasts promote the stemness and chemoresistance of colorectal cancer by transferring exosomal lncRNA H19. Theranostics (2018) 8:3932–48. doi: 10.7150/thno.25541
84. Teng Y, Wang X, Wang Y, Ma D. Wnt/β-catenin signaling regulates cancer stem cells in lung cancer A549 cells. Biochem Biophys Res Commun (2010) 392:373–9. doi: 10.1016/j.bbrc.2010.01.028
85. Cui J, Jiang W, Wang S, Wang L, Xie K. Role of wnt/β-catenin signaling in drug resistance of pancreatic cancer. Curr Pharm Des (2012) 18:2464–71. doi: 10.2174/13816128112092464
86. Fang X, Gu P, Zhou C, Liang A, Ren S, Liu F, et al. β-catenin overexpression is associated with gefitinib resistance in non-small cell lung cancer cells. Pulm Pharmacol Ther (2014) 28:41–8. doi: 10.1016/j.pupt.2013.05.005
87. Richards KE, Zeleniak AE, Fishel ML, Wu J, Littlepage LE, Hill R. Cancer-associated fibroblast exosomes regulate survival and proliferation of pancreatic cancer cells. Oncogene (2017) 36:1770–8. doi: 10.1038/onc.2016.353
88. Deng X, Ruan H, Zhang X, Xu X, Zhu Y, Peng H, et al. Long noncoding RNA CCAL transferred from fibroblasts by exosomes promotes chemoresistance of colorectal cancer cells. Int J Cancer (2020) 146:1700–16. doi: 10.1002/IJC.32608
89. Wang J, Hendrix A, Hernot S, Lemaire M, De Bruyne E, Van Valckenborgh E, et al. Bone marrow stromal cell-derived exosomes as communicators in drug resistance in multiple myeloma cells. Blood (2014) 124:555–66. doi: 10.1182/blood-2014-03-562439
90. Bliss SA, Sinha G, Sandiford OA, Williams LM, Engelberth DJ, Guiro K, et al. Mesenchymal stem cell-derived exosomes stimulate cycling quiescence and early breast cancer dormancy in bone marrow. Cancer Res (2016) 76:5832–44. doi: 10.1158/0008-5472.CAN-16-1092
91. Lobb RJ, van Amerongen R, Wiegmans A, Ham S, Larsen JE, Möller A. Exosomes derived from mesenchymal non-small cell lung cancer cells promote chemoresistance. Int J Cancer (2017) 141:614–20. doi: 10.1002/ijc.30752
92. Ma J, Dong C, Ji C. MicroRNA and drug resistance. Cancer Gene Ther (2010) 17:523–31. doi: 10.1038/cgt.2010.18
93. Binenbaum Y, Fridman E, Yaari Z, Milman N, Schroeder A, Ben David G, et al. Transfer of miRNA in macrophage-derived exosomes induces drug resistance in pancreatic adenocarcinoma. Cancer Res (2018) 78:5287–99. doi: 10.1158/0008-5472.CAN-18-0124
94. Cai C, Chen J. Overexpression of caveolin-1 induces alteration of multidrug resistance in Hs578T breast adenocarcinoma cells. Int J Cancer (2004) 111:522–9. doi: 10.1002/ijc.20300
95. Wiechen K, Diatchenko L, Agoulnik A, Scharff KM, Schober H, Arlt K, et al. Caveolin-1 is down-regulated in human ovarian carcinoma and acts as a candidate tumor suppressor gene. Am J Pathol (2001) 159:1635–43. doi: 10.1016/S0002-9440(10)63010-6
96. Kanlikilicer P, Bayraktar R, Denizli M, Rashed MH, Ivan C, Aslan B, et al. Exosomal miRNA confers chemo resistance via targeting Cav1/p-gp/M2-type macrophage axis in ovarian cancer. EBioMedicine (2018) 38:100–12. doi: 10.1016/j.ebiom.2018.11.004
97. Liu T, Zhang X, Du L, Wang Y, Liu X, Tian H, et al. Exosome-transmitted miR-128-3p increase chemosensitivity of oxaliplatin-resistant colorectal cancer. Mol Cancer (2019) 18. doi: 10.1186/s12943-019-0981-7
98. Bhattacharya B, Low SHH, Soh C, Kamal Mustapa N, Beloueche-Babari M, Koh KX, et al. Increased drug resistance is associated with reduced glucose levels and an enhanced glycolysis phenotype. Br J Pharmacol (2014) 171:3255–67. doi: 10.1111/bph.12668
99. Fischer K, Hoffmann P, Voelkl S, Meidenbauer N, Ammer J, Edinger M, et al. Inhibitory effect of tumor cell-derived lactic acid on human T cells. Blood (2007) 109:3812–9. doi: 10.1182/blood-2006-07-035972
100. Navale AM, Paranjape AN. Glucose transporters: physiological and pathological roles. Biophys Rev (2016) 8:5–9. doi: 10.1007/s12551-015-0186-2
101. Morandi A, Indraccolo S. Linking metabolic reprogramming to therapy resistance in cancer. Biochim Biophys Acta - Rev Cancer (2017) 1868:1–6. doi: 10.1016/j.bbcan.2016.12.004
102. Carvalho KC, Cunha IW, Rocha RM, Ayala FR, Cajaíba MM, Begnami MD, et al. GLUT1 expression in malignant tumors and its use as an immunodiagnostic marker. Clinics (2011) 66:965–72. doi: 10.1590/S1807-59322011000600008
103. Kunkel M, Reichert TE, Benz P, Lehr HA, Jeong JH, Wieand S, et al. Overexpression of glut-1 and increased glucose metabolism in tumors are associated with a poor prognosis in patients with oral squamous cell carcinoma. Cancer (2003) 97:1015–24. doi: 10.1002/cncr.11159
104. Ye Y, Li S-L, Wang J-J. miR-100-5p downregulates mTOR to suppress the proliferation, migration, and invasion of prostate cancer cells. Front Oncol (2020) 10:578948. doi: 10.3389/fonc.2020.578948
105. Cao YL, Zhuang T, Xing BH, Li N, Li Q. Exosomal DNMT1 mediates cisplatin resistance in ovarian cancer. Cell Biochem Funct (2017) 35:296–303. doi: 10.1002/cbf.3276
106. Romero-Garcia S, Prado-Garcia H, Carlos-Reyes A. Role of DNA methylation in the resistance to therapy in solid tumors. Front Oncol (2020) 10:1152. doi: 10.3389/fonc.2020.01152
107. Yu T, Wang XF, Zhi T, Zhang J, Wang Y, Nie E, et al. Delivery of MGMT mRNA to glioma cells by reactive astrocyte-derived exosomes confers a temozolomide resistance phenotype. Cancer Lett (2018) 433:210–20. doi: 10.1016/j.canlet.2018.06.041
108. Franke FC, Slusarenko BO, Engleitner T, Johannes W, Laschinger M, Rad R, et al. Novel role for CRK adaptor proteins as essential components of SRC/FAK signaling for epithelial–mesenchymal transition and colorectal cancer aggressiveness. Int J Cancer (2020) 147:1715–31. doi: 10.1002/ijc.32955
109. Wang L, Cho KB, Li Y, Tao G, Xie Z, Guo B. Long noncoding RNA (lncRNA)-mediated competing endogenous RNA networks provide novel potential biomarkers and therapeutic targets for colorectal cancer. Int J Mol Sci (2019) 20. doi: 10.3390/IJMS20225758
110. Liu K, Gao L, Ma X, Huang JJ, Chen J, Zeng L, et al. Long non-coding RNAs regulate drug resistance in cancer. Mol Cancer (2020) 19. doi: 10.1186/s12943-020-01162-0
111. Takahashi K, Yan IK, Kogure T, Haga H, Patel T. Extracellular vesicle-mediated transfer of long non-coding RNA ROR modulates chemosensitivity in human hepatocellular cancer. FEBS Open Bio (2014) 4:458–67. doi: 10.1016/j.fob.2014.04.007
112. Takahashi K, Yan IK, Wood J, Haga H, Patel T. Involvement of extracellular vesicle long noncoding RNA (linc-VLDLR) in tumor cell responses to chemotherapy. Mol Cancer Res (2014) 12:1377–87. doi: 10.1158/1541-7786.MCR-13-0636
113. Chen X, Wang Z, Tong F, Dong X, Wu G, Zhang R. lncRNA UCA1 promotes gefitinib resistance as a ceRNA to target FOSL2 by sponging miR-143 in non-small cell lung cancer. Mol Ther Nucleic Acids (2020) 19:643–53. doi: 10.1016/J.OMTN.2019.10.047
114. Mahinfar P, Baradaran B, Davoudian S, Vahidian F, Cho WCS, Mansoori B. Long non-coding RNAs in multidrug resistance of glioblastoma. Genes (Basel) (2021) 12. doi: 10.3390/genes12030455
115. Zhang Z, Yin J, Lu C, Wei Y, Zeng A, You Y. Exosomal transfer of long non-coding RNA SBF2-AS1 enhances chemoresistance to temozolomide in glioblastoma. J Exp Clin Cancer Res (2019) 38. doi: 10.1186/s13046-019-1139-6
116. Han M, Gu Y, Lu P, Li J, Cao H, Li X, et al. Exosome-mediated lncRNA AFAP1-AS1 promotes trastuzumab resistance through binding with AUF1 and activating ERBB2 translation. Mol Cancer (2020) 19. doi: 10.1186/S12943-020-1145-5
117. Kalluri R. The biology and function of exosomes in cancer. J Clin Invest (2016) 126:1208–15. doi: 10.1172/JCI81135
118. Melo SA, Luecke LB, Kahlert C, Fernandez AF, Gammon ST, Kaye J, et al. Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature (2015) 523:177–82. doi: 10.1038/nature14581
119. Yuwen DL, Sheng BB, Liu J, Wenyu W, Shu YQ. MiR-146a-5p level in serum exosomes predicts therapeutic effect of cisplatin in non-small cell lung cancer. Eur Rev Med Pharmacol Sci (2017) 21:2650–8.
120. Xiao Y, Li Y, Yuan Y, Liu B, Pan S, Liu Q, et al. The potential of exosomes derived from colorectal cancer as a biomarker. Clin Chim Acta (2019) 490:186–93. doi: 10.1016/J.CCA.2018.09.007
121. Hitchcock CL, Povoski SP, Mojzisik CM, Martin EW. Survival advantage following TAG-72 antigen-directed cancer surgery in patients with colorectal carcinoma: Proposed mechanisms of action. Front Oncol (2021) 11:731350. doi: 10.3389/FONC.2021.731350
122. Wang B, Zhang Y, Ye M, Wu J, Ma L, Chen H. Cisplatin-resistant MDA-MB-231 cell-derived exosomes increase the resistance of recipient cells in an exosomal miR-423-5p-dependent manner. Curr Drug Metab (2019) 20:804–14. doi: 10.2174/1389200220666190819151946
123. Chen T, Zhang G, Kong L, Xu S, Wang Y, Dong M. Leukemia-derived exosomes induced IL-8 production in bone marrow stromal cells to protect the leukemia cells against chemotherapy. Life Sci (2019) 221:187–95. doi: 10.1016/J.LFS.2019.02.003
124. Shi Z, Yang WM, Chen LP, Yang DH, Zhou Q, Zhu J, et al. Enhanced chemosensitization in multidrug-resistant human breast cancer cells by inhibition of IL-6 and IL-8 production. Breast Cancer Res Treat (2012) 135:737–47. doi: 10.1007/S10549-012-2196-0
125. Huang D, Ding Y, Zhou M, Rini BI, Petillo D, Qian CN, et al. Interleukin-8 mediates resistance to antiangiogenic agent sunitinib in renal cell carcinoma. Cancer Res (2010) 70:1063–71. doi: 10.1158/0008-5472.CAN-09-3965
126. Kharaziha P, Chioureas D, Rutishauser D, Baltatzis G, Lennartsson L, Fonseca P, et al. Molecular profiling of prostate cancer derived exosomes may reveal a predictive signature for response to docetaxel. Oncotarget (2015) 6:21740–54. doi: 10.18632/ONCOTARGET.3226
127. Gong J, Luk F, Jaiswal R, George AM, Grau GER, Bebawy M. Microparticle drug sequestration provides a parallel pathway in the acquisition of cancer drug resistance. Eur J Pharmacol (2013) 721:116–25. doi: 10.1016/j.ejphar.2013.09.044
128. Lopes-Rodrigues V, Di Luca A, Sousa D, Seca H, Meleady P, Henry M, et al. Multidrug resistant tumour cells shed more microvesicle-like EVs and less exosomes than their drug-sensitive counterpart cells. Biochim Biophys Acta (2016) 1860:618–27. doi: 10.1016/J.BBAGEN.2015.12.011
129. Yin J, Yan X, Yao X, Zhang Y, Shan Y, Mao N, et al. Secretion of annexin A3 from ovarian cancer cells and its association with platinum resistance in ovarian cancer patients. J Cell Mol Med (2012) 16:337–48. doi: 10.1111/J.1582-4934.2011.01316.X
130. Yan X, Yin J, Yao H, Mao N, Yang Y, Pan L. Increased expression of annexin A3 is a mechanism of platinum resistance in ovarian cancer. Cancer Res (2010) 70:1616–24. doi: 10.1158/0008-5472.CAN-09-3215
131. Menck K, Sönmezer C, Worst TS, Schulz M, Dihazi GH, Streit F, et al. Neutral sphingomyelinases control extracellular vesicles budding from the plasma membrane. J Extracell Vesicles (2017) 6:1378056. doi: 10.1080/20013078.2017.1378056
132. Chen WX, Xu LY, Qian Q, He X, Peng WT, Fan WQ, et al. D rhamnose β-hederin reverses chemoresistance of breast cancer cells by regulating exosome-mediated resistance transmission. Biosci Rep (2018) 38. doi: 10.1042/BSR20180110
133. Koch R, Aung T, Vogel D, Chapuy B, Wenzel D, Becker S, et al. Nuclear trapping through inhibition of exosomal export by indomethacin increases cytostatic efficacy of doxorubicin and pixantrone. Clin Cancer Res (2016) 22:395–404. doi: 10.1158/1078-0432.CCR-15-0577
134. Xie X, Nie H, Zhou Y, Lian S, Mei H, Lu Y, et al. Publisher correction: Eliminating blood oncogenic exosomes into the small intestine with aptamer-functionalized nanoparticles. Nat Commun (2020) 11. doi: 10.1038/S41467-019-13863-2
135. Li S, Yi M, Dong B, Jiao Y, Luo S, Wu K. The roles of exosomes in cancer drug resistance and its therapeutic application. Clin Transl Med (2020) 10. doi: 10.1002/CTM2.257
136. Li YJ, Wu JY, Liu J, Xu W, Qiu X, Huang S, et al. Artificial exosomes for translational nanomedicine. J Nanobiotechnol (2021) 19. doi: 10.1186/S12951-021-00986-2
137. Bastos N, Ruivo CF, da Silva S, Melo SA. Exosomes in cancer: Use them or target them? Semin Cell Dev Biol (2018) 78:13–21. doi: 10.1016/J.SEMCDB.2017.08.009
138. Hadla M, Palazzolo S, Corona G, Caligiuri I, Canzonieri V, Toffoli G, et al. Exosomes increase the therapeutic index of doxorubicin in breast and ovarian cancer mouse models. Nanomedicine (2016) 11:2431–41. doi: 10.2217/nnm-2016-0154
139. Toffoli G, Hadla M, Corona G, Caligiuri I, Palazzolo S, Semeraro S, et al. Exosomal doxorubicin reduces the cardiac toxicity of doxorubicin. Nanomed (Lond) (2015). doi: 10.2217/nnm.15.118
140. Liang Y, Duan L, Lu J, Xia J. Engineering exosomes for targeted drug delivery. Theranostics (2021) 11:3183–95. doi: 10.7150/THNO.52570
141. Li X, Tsibouklis J, Weng T, Zhang B, Yin G, Feng G, et al. Nano carriers for drug transport across the blood-brain barrier. J Drug Target (2017) 25:17–28. doi: 10.1080/1061186X.2016.1184272
Keywords: extra cellular vesicles, cancer, drug resistance, therapy, oncology
Citation: Palazzolo S, Canzonieri V and Rizzolio F (2022) The history of small extracellular vesicles and their implication in cancer drug resistance. Front. Oncol. 12:948843. doi: 10.3389/fonc.2022.948843
Received: 20 May 2022; Accepted: 06 July 2022;
Published: 24 August 2022.
Edited by:
Nadia Rucci, University of L’Aquila, ItalyReviewed by:
Natasha Kyprianou, Icahn School of Medicine at Mount Sinai, United StatesKrisztina V. Vukman, Semmelweis University, Hungary
Copyright © 2022 Palazzolo, Canzonieri and Rizzolio. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Flavio Rizzolio, ZmxhdmlvLnJpenpvbGlvQHVuaXZlLml0; Stefano Palazzolo, c3BhbGF6em9sb0Bjcm8uaXQ=
 Stefano Palazzolo
Stefano Palazzolo Vincenzo Canzonieri
Vincenzo Canzonieri Flavio Rizzolio
Flavio Rizzolio