
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Oncol. , 23 May 2022
Sec. Pharmacology of Anti-Cancer Drugs
Volume 12 - 2022 | https://doi.org/10.3389/fonc.2022.908031
This article is part of the Research Topic Pharmacological Targeting of Tumor Microenvironment: From Bench to Bedside, Volume II View all 21 articles
 Minghui Peng1†
Minghui Peng1† Zhuifeng Zheng1,2†
Zhuifeng Zheng1,2† Shaoyang Chen1†
Shaoyang Chen1† Le Fang3
Le Fang3 Rongxiu Feng4
Rongxiu Feng4 Lijun Zhang5
Lijun Zhang5 Qingnan Tang1
Qingnan Tang1 Xuewen Liu1*
Xuewen Liu1*Objective: To explore the impacts of AE (aloe-emodin) in gefitinib-resistant NSCLC (non-small cell lung cancer) cells and the corresponding mechanism.
Methods: PC9 and PC9-GR cells were cultured and treated by gefitinib, AE, or the combination of the two drugs. Then, viability, apoptosis, migration and invasion of cells were investigated using CCK-8, TUNEL, wound healing assay, and transwell assay, respectively. Female BALB/c nude mice were employed for the establishment of xenograft tumor models to examine the role of AE in tumor growth.
Results: PC9-GR cells showed reduced apoptosis and enhanced cell viability, migration and invasion upon treatment by gefitinib, compared with PC9 cells. E-cahherin in PC9-GR cells was down-regulated, while Vimentin, Snail2 (or Slug) and Twist1 in PC9-GR cells were up-regulated, compared with PC9 cells. Meanwhile, treatment by a combination of gefitinib and AE significantly strengthened apoptosis of PC9-GR cells, while attenuated their migration and invasion, compared with the control group or treatment by gefitinib or AE alone. WB results showed that AE could reverse EMT and activation of PI3K/AKT signalling pathway in PC9-GR cells. In vivo experiments showed that tumor growth and EMT of PC9-GR cells were dramatically repressed after treatment by a combination of AE and gefitinib. Additionally, the use of SC97 (a PI3K/Akt pathway activator) could counteract the effects of AE in gefitinib-resistant PC9 cells.
Conclusions: AE could enhance the gefitinib sensitivity of PC9-GR cells and reverse EMT by blocking PI3K/Akt/TWIS1 signal pathway.
Although great advances in cancer treatment have been achieved in recent decades, the prognosis of patients remains unsatisfactory due to the development of drug resistance during the treatment (1, 2). Lung cancer, which consists of about 85% NSCLC and 15% SCLC, is the top cancer globally and a leading cause of cancer death (3, 4). The therapeutic techniques for NSCLC, which depend on pathological types, clinical stages as well as other medical conditions, include surgery, adjuvant chemotherapy, immunotherapy, and targeted therapy (5).
Epidermal growth factor tyrosine inhibitors (EGFR-TKIs) are considered typical paradigms of targeted therapies in NSCLC, but acquired drug resistance unavoidably limits the clinical efficacy of EGFR-TKIs, which therefore makes fighting against EGFR-TKI resistance an absorbing topic for NSCLC treatment (6, 7). Gefitinib, one of the widely used EGFR-TKIs, has been approved for the first-line treatment of NSCLC with sensitizing EGFR mutations (exon 19 deletion or L858R point mutation) (8), while drug resistance in cancer cells contributes to the limited clinical efficacy of the targeted drug. Therefore, safe yet effective antitumor drugs or treatments are urgently needed for the management of LC.
The intimate connection between epithelial-mesenchymal transition (EMT) activation and drug resistance are founded in many diseases, including NSCLC (9). In addition, several mechanisms including abnormal activation of PI3K/Akt pathway have been proved to generate drug resistance in cancer (10). One study reported that miR-30a-5p overexpression can overcome EGFR-TKI resistance in NSCLC by regulating PI3K/Akt pathway (11). Fang et al. revealed the distribution of common mutations involving PI3K activation in EGFR-TKI resistant patients, which were difficult to overcome (12). Zhou et al. found that PI3K/Akt signaling pathway was suppressed by miR-200c to strengthen the sensitivity of drug-resistant NSCLC cells to gefitinib (13). Hao et al. demonstrated that IRS4 conferred therapy resistance in lung cancer cells through activating PI3K/Akt signaling (14). These studies collectively indicate PI3K/Akt signaling pathway may be a promising target of overcoming the gefitinib resistance in NSCLC.
As a natural anthraquinone derivative and an active ingredient of Chinese herbs, Aloe-emodin (AE, Figure 2A) exhibits pharmacological effects on various diseases, including liver fibrosis, LC, and leukemia (15). Ma et al. found that AE could trigger EMT in SkBr3 cells (breast cancer cells) to repress cancer metastasis (16). Dou et al. reported that AE could alleviate renal fibrosis by repressing PI3K/Akt/mTOR signaling pathway both in vivo and in vitro (17). Additionally, it has been demonstrated that AE could mitigate drug resistance of breast cancer cells and melanoma cells, indicating a regulatory role of AE in drug resistance (18, 19). Nonetheless, effects of AE on drug resistance of NSCLCs have not been fully explored.
In this study, the role of AE in the development of drug resistance of NSCLCs and its underlying mechanism were investigated to provide insights for NSCLC treatment.
The PC9 cell line was obtained from the Chinese Academy of Science (Shanghai, China) and cultured in RPMI 1640 with 10% FBS under 5% CO2 at 37°C. Then, gefitinib was added with elevating concentrations to develop and maintain the resistant cell line for half a year (20).
PC9-GR and PC9 cells were then subjected to gefitinib of different concentrations (0, 0.01, 0.1, 1, 10, 100 μM), while PC9-GR cells were further treated by DMSO (the control group), 10 μM gefitinib (the gefitinib group), 10 μM AE (the AE group), the combination of gefitinib and AE (the gefitinib+AE group), and the combination of gefitinib and AE plus SC97 (the gefitinib+AE+SC97 group).
Cell viability was determined using CCK8. Briefly, cell seeding was executed in 96-well plates and a confluence was reached in 24 h. Then, cells were treated with gefitinib of different concentrations and/or AE. After 24 h, the cells were further incubated with CCK‐8 for 120 min, and the viable cells were subsequently counted according to the absorbance at 450 nm.
Cell migration was detected using the wound healing assay. First, cell seeding was executed in 6‐well plates and cultured for 24 h. Thereafter, a single scratch was created using a 100 μL pipette tip, and the cells were incubated with FBS‐free culture medium containing DMSO or gefitinib and/or AE after being rinsed with PBS. Finally, the scratches at 0 and 24 hours were captured using an Olympus IX71 inverted microscope.
Cell invasion was assessed using the transwell assay. 60 μL of Matrigel was added into each chamber, and the cells were seeded in the upper chamber of the insert with or without drugs. Then, the upper chamber was cleaned and cells on the membrane bottom were fixed and stained after a 24-hour incubation. A light microscope was used to count the cells on the membrane bottom.
Cell apoptosis was assessed using the TUNEL assay. First, cells were rinsed with PBS twice, incubated with formaldehyde for 20 min, and permeabilized with 0.1% TritonX‐100 for 10 min. Then, cells were incubated in PBS for 5 min, TdT reaction mixture was added and cells were incubated in a dark humidified chamber at 37°C for 60 min. Thereafter, 2 × SSC buffer was used to stop the reaction, and DAPI (Abcam, MA, USA) was used to counterstain the cell nuclei after being rinsed with PBS. Finally, a fluorescence microscope was used to capture images.
Total RNA extraction from PC9-GR cells was executed using Trizol Reagent. Then, cDNA of the mRNA was obtained, and the mixture of cDNA and SYBR Green was subjected to qPCR by a real-time PCR System. The results were quantified using the 2-ΔΔCT method. GAPDH was employed as an internal reference for mRNA standardization in each sample. Specific primers used for qPCR are listed in Table S1.
PC9-GR or PC9 cells were lysed in RIPA buffer to extract total proteins, which were subsequently segregated on SDS‐PAGE gels and transferred to PVDF membranes. Then, primary antibodies were added and cells were incubated at 4°C overnight upon blockage of 5% skim milk. After three rinsing cycles, appropriate secondary antibodies were added and cells were incubated at 37°C for 2 h. Finally, samples were characterized by the ECL system and quantified by Image J. The detailed information of antibodies used is listed in Table S2.
Female BALB/c nude mice (age = 4-5 weeks and weight = 16-20 g) were provided by the Guangdong Medical Laboratory Center (China). The animals were raised in a SPF environment, which were given ab libtum to water and standard chow. After acclimated for one week, subcutaneous inoculation of 1 × 107 PC9-GR cells was executed into the right flank of the animals (n = 4 in each group) to develop xenograft models. Once the tumor volume approached 50 mm3 by average, gefitinib (30 mg/kg), AE (30 mg/kg) and their combination were administered, respectively, while the same volume of saline was administered as the control group. The tumor size was evaluated every week using a digital caliper, while tumor volumes were calculated by: Volume = (Length × Width2)/2. The mice were euthanized on Day 21 and tumors were weighed immediately.
Student’s t-tests were employed to compare the two groups. One- or two-way ANOVA was performed on multiple-group comparison. All data were in the format of mean ± standard deviation (SD) and the three experiments were independent from each other. P<0.05 indicated statistically significant differences.
First, gefitinib‐resistant PC9 cells were constructed and their viability in gefitinib was enhanced compared with their parent PC9 cells (p < 0.001, Figure 1A). Then, the effects of 1 μM gefitinib on apoptosis, migration, and invasion of the cell samples were examined. Investigation by TUNEL assay showed that the apoptosis rate of PC9-GR cells was prominently lower than that of PC9 cells (p = 0.005, Figure 1B). Enhanced migration (p = 0.016, Figure 1C) and reduced apoptosis (p < 0.001, Figure 1D) were founded in PC9-GR cells, as shown in the results of wound healing assay and cell invasion assay. WB results indicated that the E-cadherin level was significantly higher in PC9 cells than in PC9-GR cells, while levels of Vimentin, Slug, Twist1 in PC9 cells were significantly lower in PC9 cells than in PC9-GR cells (p < 0.001, Figure 1E). In summary, EMT was observed in gefitinib‐resistant cells and it might be associated with the development of gefitinib resistance.
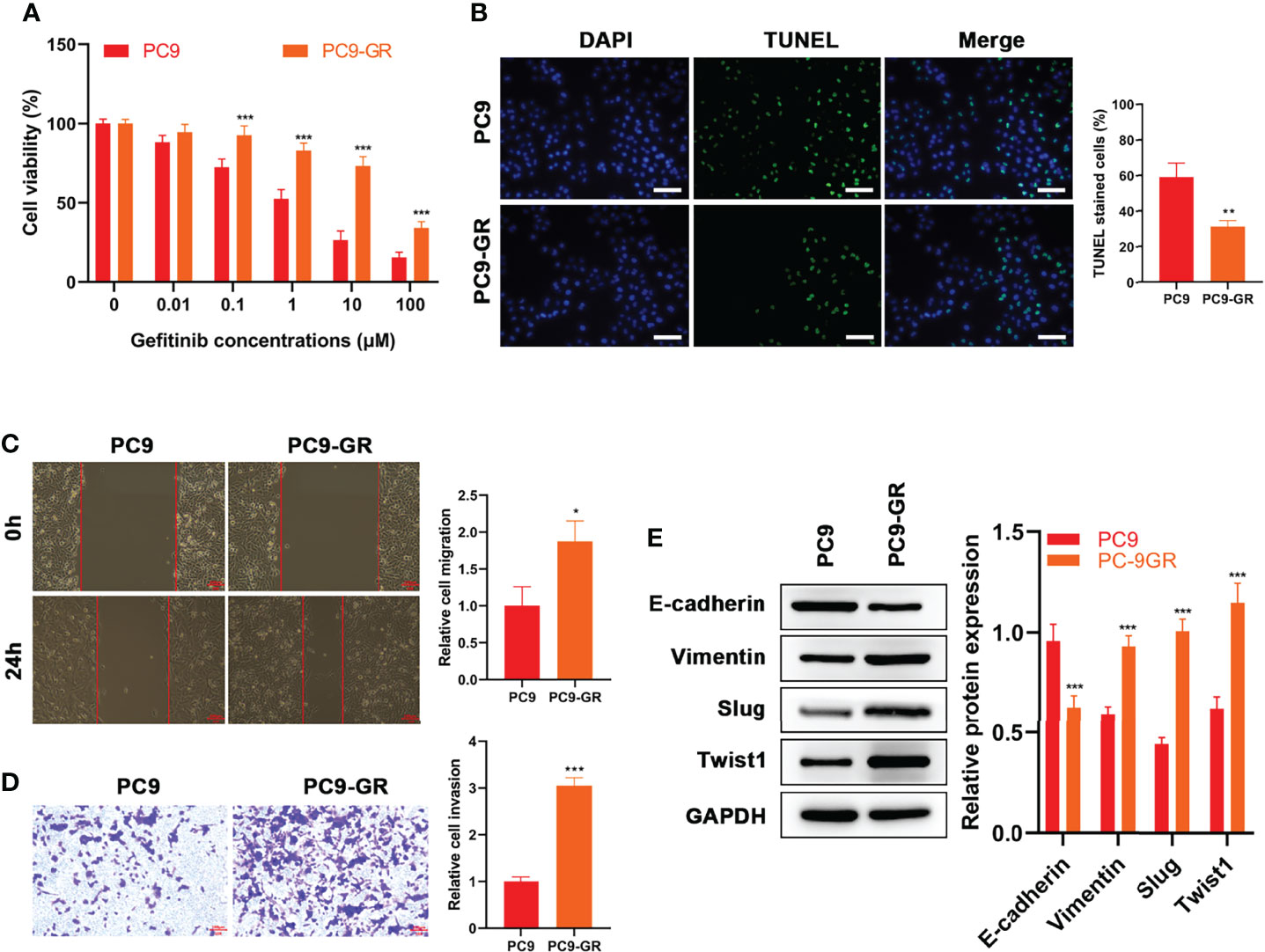
Figure 1 EMT Occurred in Gefitinib‐Resistant Cells. (A) CCK8 results of PC9 and PC9-GR cells at different concentrations of gefitinib treatment (B) TUNEL results, (C) wound healing assay results and (D) Transwell assay of PC9 and PC9-GR cells at 1 μM of gefitinib (E) Expressions of EMT-related proteins (E-cadherin, Vimentin, Slug and Twist1) in the two cell lines at 1 μM of gefitinib detected by WB results. Scale bar = 100 μm. *p<0.05, **p<0.01, ***p<0.001.
The effects of AE (Figure 2A) on PC9-GR cells were explored. The results showed that the presence of AE led to a significant decrease in cell viability (p = 0.005, Figure 2B), migration (p < 0.001, Figure 2D), and invasion (p = 0.001, Figure 2E), and an increase in apoptosis (p = 0.003, Figure 2C). Meanwhile, the combination of AE and gefitinib achieved a notable suppression on the proliferation, migratory and invasive abilities of PC9-GR cells, which was refractory to gefitinib treatment alone (p < 0.001, Figures 2B–E). These results demonstrated that the combination was more effective than either one alone, and AE could reduce the resistance of PC9-GR cells to gefitinib.
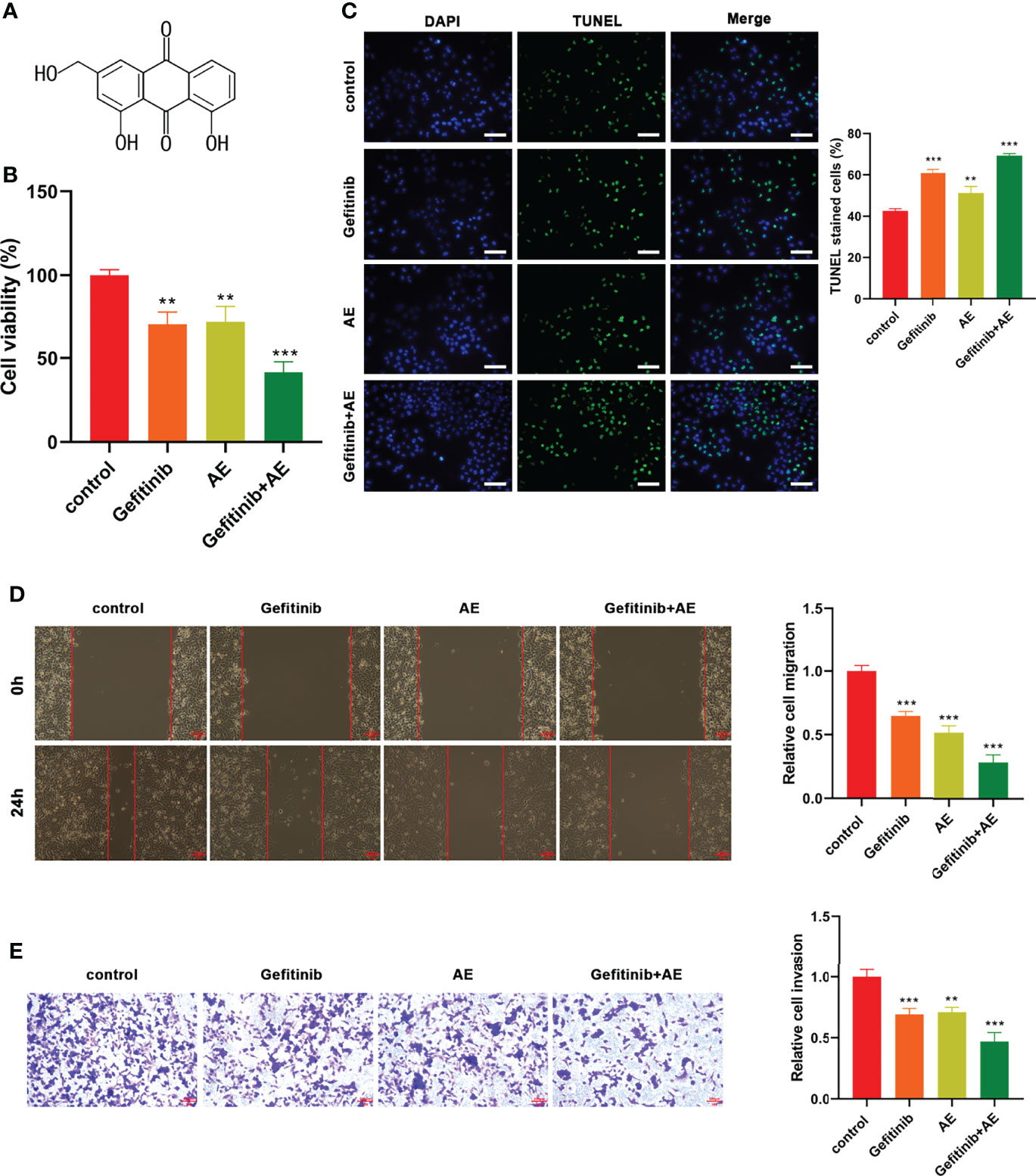
Figure 2 AE Could Enhance the Gefitinib Sensitivity of PC9-GR Cells. (A) Chemical structure of AE. (B) CCK8 results, (C) TUNEL assay results, (D) wound healing assay results, and (E) Transwell assay results of PC9-GR cells after different treatments. Scale bar = 100 μm. **p<0.01, ***p<0.001.
Previous studies have demonstrated the presence of EMT during the development of drug resistance (21–23). Hence, effects of AE on EMT in PC9-GR cells were investigated. qPCR analysis revealed that the E-cadherin levels increased significantly when treated with AE alone (p < 0.001), whereas the levels of Vimentin (p = 0.009) and Twist1 (p = 0.002) decreased under the administration of AE alone (Figure 3A). The combination of AE and gefitinib exhibited a stronger effect on reversing EMT, as evidenced by a significant increase in E-cadherin levels (p < 0.001) and a decrease in levels of Vimentin (p = 0.004), Slug (p = 0.022) and Twist1 (p < 0.001) (Figure 3A). These results were further corroborated by WB analysis (Figure 3B). Meanwhile, the MAPK/ERK and PI3K/Akt signaling pathways were detected in PC9-GR cells with the treatment of AE. There is no significant difference in the phosphorylation of ERK1/2 among all groups (all p > 0.05), while a remarkable decline in the phosphorylation of PI3K (p =0.035) and Akt (p < 0.001) was conformed upon AE treatment (Figure 3C). Notably, the combination of AE and gefitinib exhibited a stronger suppression effect than AE alone on PI3K and Akt phosphorylation (all p< 0.001, Figure 3C). These results indicated that AE could reverse the EMT in PC9-GR cells, and inactivate the PI3K/Akt pathway.
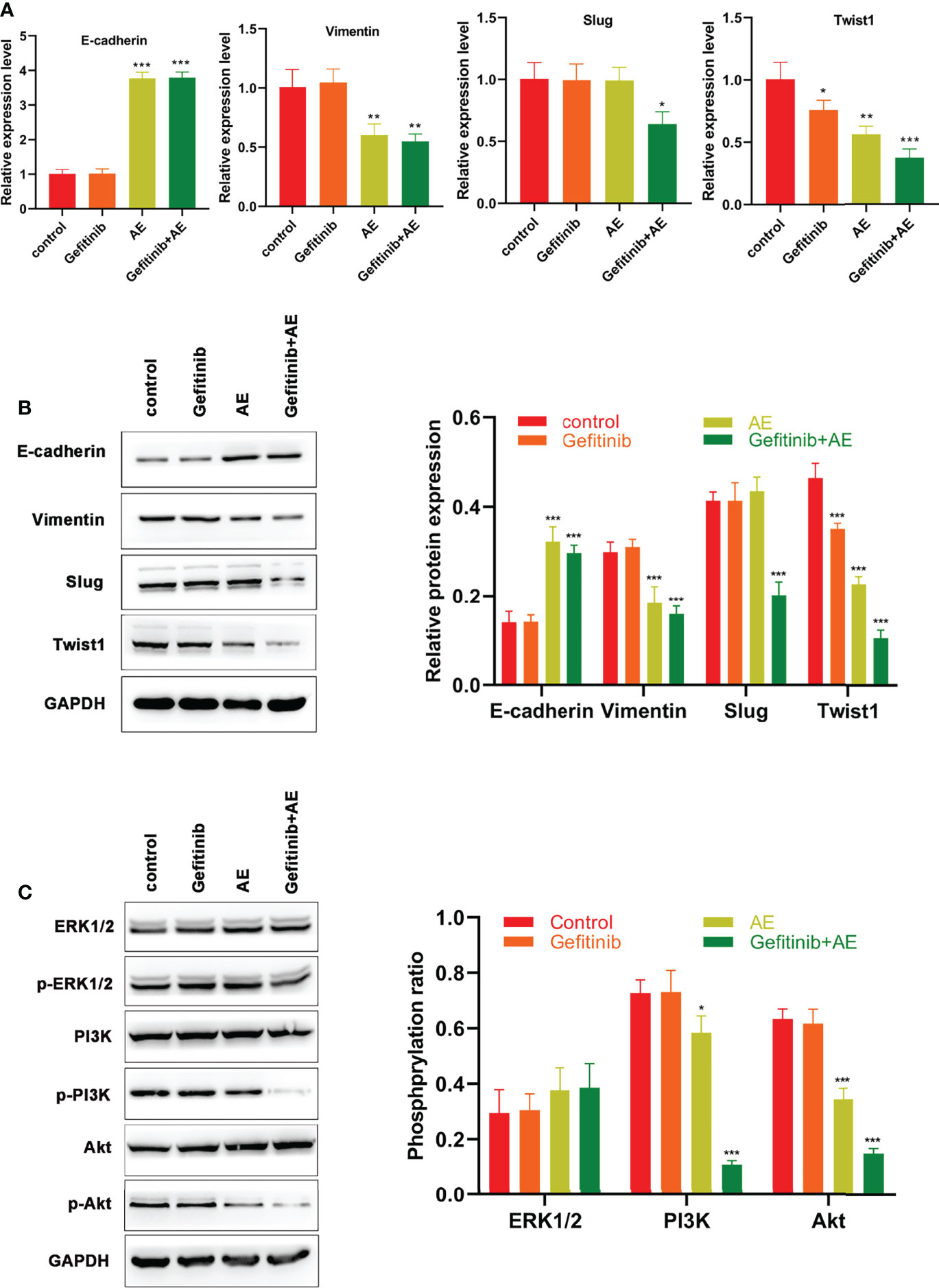
Figure 3 AE Could Reverse EMT Induced by Drug Resistance in PC9-GR Cells. (A) qPCR results of E-cadherin, Vimentin, Slug and Twist1 for PC9-GR cells. (B) WB results of E-cadherin, Vimentin, Slug, and Twist 1 for PC9-GR cells (C) WB results of the PI3K/Akt pathway related proteins for PC9-GR cells. *p<0.05, **p<0.01, ***p<0.001.
To further verify the conclusions, in vivo experiments were conducted. Compared with the control group, gefitinib alone showed a slight repression in tumor growth, while the combination of AE and gefitinib achieved a notable growth suppression on PC9-GR xenografts (all p < 0.001, Figures 4A–C). Similar to the in vitro results, WB results suggested that E-cadherin was significantly up-regulated when treated with AE and gefitinib, whereas Vimentin, Slug and Twist1 were down-regulated when treated with the combination of these two drugs (all p < 0.001, Figure 4D). In summary, AE could enhance the sensitivity of xenograft tumors to gefitinib and reverse the EMT in vivo.
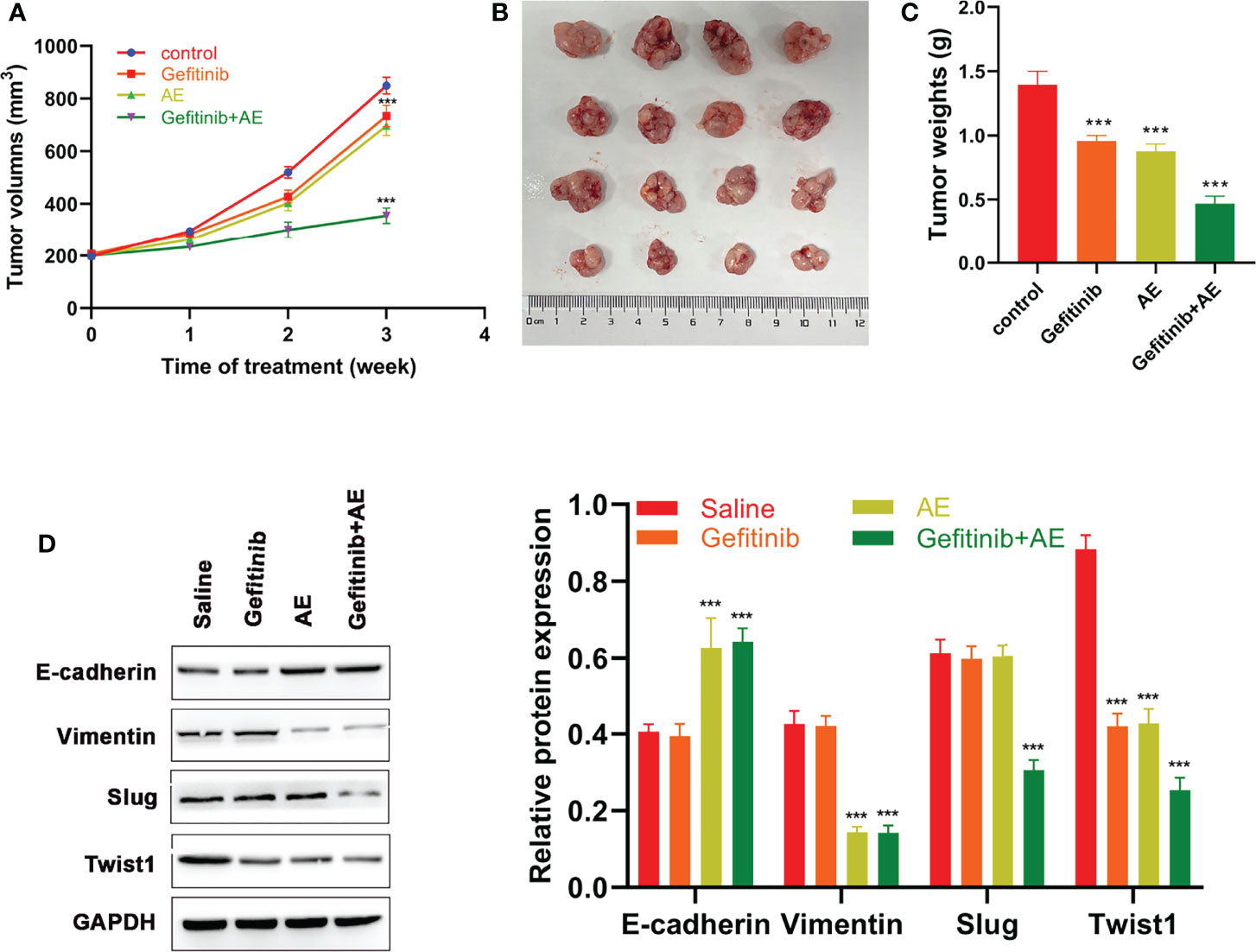
Figure 4 AE Could Enhance the Sensitivity of Xenograft Tumors to Gefitinib. (A) Volumes of xenograft tumors, (B) tumor growth, (C) weights of xenograft tumors and (D) WB results of EMT-related proteins of different groups after different treatments. ***p<0.001.
Based on the results mentioned above, AE was supposed to reverse EMT and block PI3K/Akt pathway. Therefore, a PI3K activator SC97 was used to verify this hypothesis. PC9-GR cells were subjected to gefitinib, SC97, or the combination of gefitinib and AE plus SC97. Then, viability, apoptosis, migration, and invasion of the cells were determined. Consistent with the foregoing results, administration of SC97 led to attenuated apoptosis (p = 0.003, Figure 5B), and enhanced viability (p < 0.001, Figure 5A), migration (p = 0.002, Figure 5C) and invasion (p = 0.012, Figure 5D) of PC9-GR cells, while AE significantly alleviated this effect (all p > 0.05, Figures 5A–D).
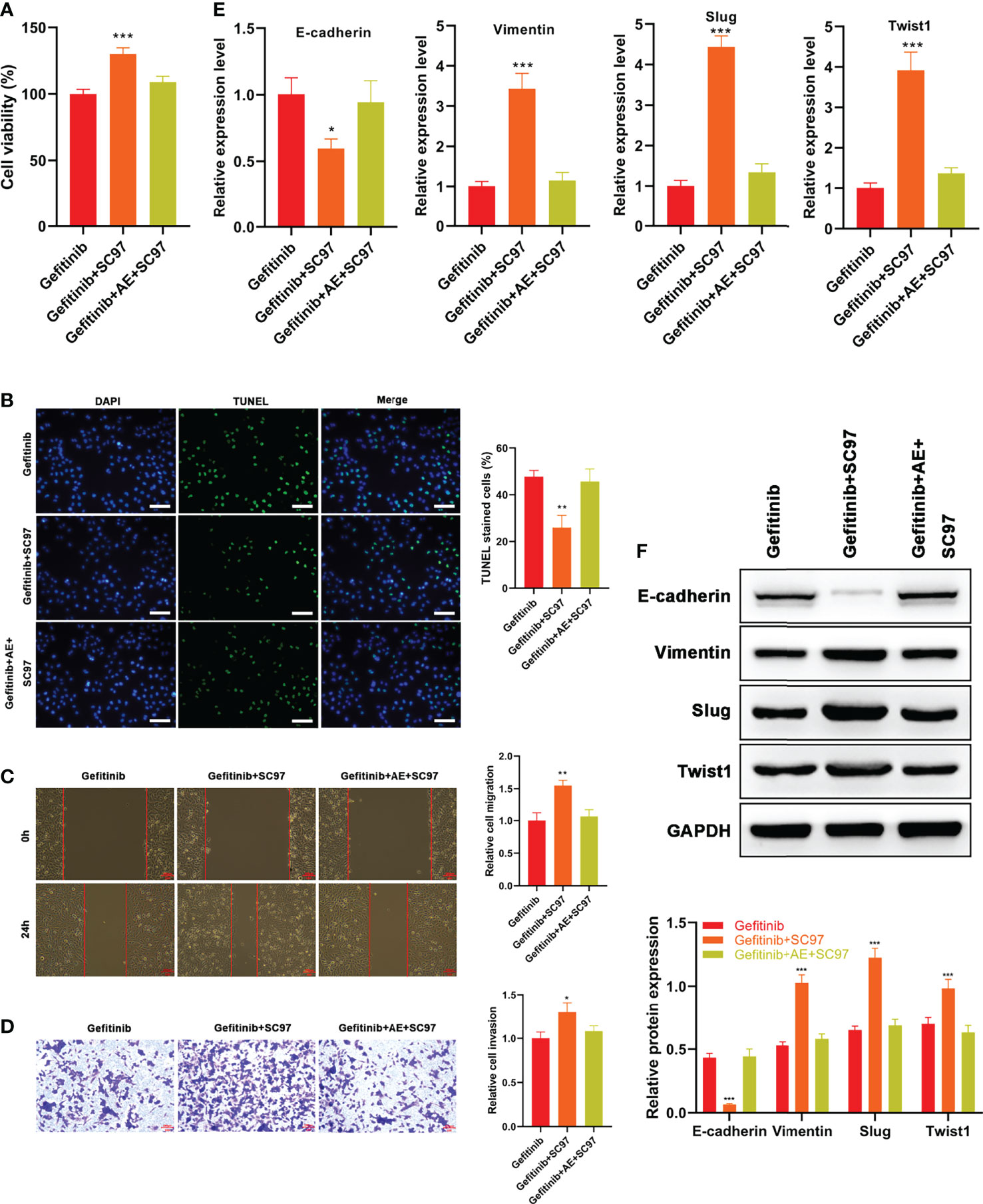
Figure 5 AE Strengthened the Gefitinib Sensitivity of PC9-GR Cells by Reversing EMT Via PI3K/Akt/TWIS1 Pathway. (A) Relative cell viability of PC9-GR cells treated with different treatments (B) TUNEL assay results, (C) wound healing assay results, (D) Transwell assay results, (E) qPCR results of EMT-related genes, and (F) WB results of EMT-related genes in PC9-GR cells after different treatments. Scale bar = 100 μm. *p<0.05, **p<0.01, ***p<0.001.
qPCR and WB results suggested AE reversed the reduced expression of E-cadherin and enhanced expressions of Vimentin, Slug and Twist1 by SC97 (all p > 0.05, Figures 5E, F). Overall, the results revealed that AE could strengthen the gefitinib sensitivity of PC9-GR cells and reverse EMT by blocking PI3K/Akt/TWIS1 pathway.
Many advances in therapeutic strategies for cancer have been made in recent years, but the occurrence of drug resistance cuts down the curative effects of therapies (24, 25). Among the therapeutic methods for NSCLC, targeted therapy is characterized by high efficiency, reduced side effects, and wide adaptability (26). Gefitinib is a commonly used targeted drug for the treatment of NSCLC with favorable efficacy, but the drug resistance gradually weakens the clinical therapeutic effect of the targeted drug, resulting in an ineffective prognosis. Owing to a long period for development and clinical applications of new drugs, the treatment of many patients might not be timely. Therefore, exploration of a potential adjuvant to gefitinib in NSCLC treatment to overcome drug resistance is promising in improving the treatment efficacy for patients.
The anticancer properties of AE on several types of cancers, including bladder cancer, cervical cancer, and colon cancer, have been explored (27). The results of this study confirmed the anticancer properties of AE on NSCLC by treating PC9-GR cells or xenograft models with a combination of gefitinib and AE or AE alone. Cheng et al. presented that AE sensitized tumor cells to chemotherapeutic agents, indicating the potential of AE for improving drug resistance (18). In this study, AE could improve the drug resistance of PC9-GR cells and xenograft models.
It has been demonstrated that EMT plays an important role in drug resistance in various cases, such as lung cancer and breast cancer, etc. (22, 28, 29). Likewise, the results of this study showed that E-cadherin was significantly down-regulated in PC9-GR cells, whereas vimentin, slug, and twist1 were up-regulated in PC9-GR cells, indicating the presence of EMT in drug-resistant cells. Additionally, the combination of AE and gefitinib or AE alone can reverse the EMT both in vitro and in vivo.
It has been found that FAT4 (FAT tumor suppressor homolog 4) could regulate the autophagy and the EMT process in colorectal cancer cells by blocking the PI3K-Akt signaling pathway (30). Meanwhile, GPER1 regulates the EMT process by blocking the PI3K-Akt signaling pathway in gastric cancer (31). In addition, the ERK1/2 signal pathway was involved in EMT (32–36). These studies suggested that the ERK1/2 or PI3K/Akt signaling pathway might be involved in drug resistance.
The determination of phosphorylation of relevant indices of ERK1/2 and PI3K-Akt pathways in this study showed no significant difference in the activation of ERK signaling pathway, while the activation of PI3K/Akt pathway exhibited significant upregulation in the AE group and the gefitinib+AE group, suggesting that the PI3K/Akt signaling pathway participated the AE regulation in drug resistance. Additionally, SC97, a PI3K/Akt pathway activator, was employed to verify that AE strengthened the gefitinib sensitivity of PC9-GR cells and reversed EMT via the PI3K/Akt axis.
To sum up, AE can induce the apoptosis and reduce the viability, migration and invasion of gefitinib-resistant NSCLC cells. More importantly AE enhance the gefitinib sensitivity of PC9-GR cells in NSCLC cells and xenograft models and reverse EMT by blocking PI3K/Akt signaling pathway. These findings highlight the anti-resistance property of AE, indicating that AE is a promising sensitizer of gefitinib in NSCLC treatment. Also, this study provides insights for other researchers to combat drug resistance, and offers a novel question that whether AE could be used as an adjuvant in chemotherapy and targeted therapy for other diseases.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Conceptualization, MP, ZZ, SC and XL. Validation and formal analysis, LF and RF. Investigation anddata curation, LZ and QT. Writing—original draft preparation, MP, ZZ and SC. Writing—review and editing, XL. All authors have read and agreed to the published version of the manuscript.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
This study was supported by Wisdom Gathering and Talent Cultivating Program from the Third Xiangya Hospital.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2022.908031/full#supplementary-material
1. Sharifi E, Bigham A, Yousefiasl S, Trovato M, Ghomi M, Esmaeili Y, et al. Mesoporous Bioactive Glasses in Cancer Diagnosis and Therapy: Stimuli-Responsive, Toxicity, Immunogenicity, and Clinical Translation. Adv Sci (Weinh) (2022) 9:e2102678. doi: 10.1002/advs.202102678
2. Makvandi P, Chen M, Sartorius R, Zarrabi A, Ashrafizadeh M, Dabbagh Moghaddam F, et al. Endocytosis of Abiotic Nanomaterials and Nanobiovectors: Inhibition of Membrane Trafficking. Nano Today (2021) 40:101279. doi: 10.1016/j.nantod.2021.101279
4. Bade BC, Dela Cruz CS. Lung Cancer 2020: Epidemiology, Etiology, and Prevention. Clin Chest Med (2020) 41:1–24. doi: 10.1016/j.ccm.2019.10.001
5. Duma N, Santana-Davila R, Molina JR. Non-Small Cell Lung Cancer: Epidemiology, Screening, Diagnosis, and Treatment Mayo. Clin Proc (2019) 94:1623–40. doi: 10.1016/j.mayocp.2019.01.013
6. Zhu X, Chen L, Liu L, Niu X. EMT-Mediated Acquired EGFR-TKI Resistance in NSCLC: Mechanisms and Strategies. Front Oncol (2019) 9:1044. doi: 10.3389/fonc.2019.01044
7. Terlizzi M, Colarusso C, Pinto A, Sorrentino R. Drug Resistance in non-Small Cell Lung Cancer (NSCLC): Impact of Genetic and Non-Genetic Alterations on Therapeutic Regimen and Responsiveness. Pharmacol Ther (2019) 202:140–8. doi: 10.1016/j.pharmthera.2019.06.005
8. Rawluk J, Waller CF. Gefitinib Recent Results. Cancer Res (2018) 211:235–46. doi: 10.1007/978-3-319-91442-8_16
9. Shibue T, Weinberg RA EMT. CSCs, and Drug Resistance: The Mechanistic Link and Clinical Implications. Nat Rev Clin Oncol (2017) 14:611–29. doi: 10.1038/nrclinonc.2017.44
10. Liu R, Chen Y, Liu G, Li C, Song Y, Cao Z, et al. PI3K/AKT Pathway as a Key Link Modulates the Multidrug Resistance of Cancers. Cell Death Dis (2020) 11:797. doi: 10.1038/s41419-020-02998-6
11. Meng F, Wang F, Wang L, Wong SC, Cho WC, Chan LW. MiR-30a-5p Overexpression May Overcome EGFR-Inhibitor Resistance Through Regulating PI3K/AKT Signaling Pathway in Non-Small Cell Lung Cancer Cell Lines. Front Genet (2016) 7:197. doi: 10.3389/fgene.2016.00197
12. Fang W, Huang Y, Gu W, Gan J, Wang W, Zhang S, et al. PI3K-AKT-mTOR Pathway Alterations in Advanced NSCLC Patients After Progression on EGFR-TKI and Clinical Response to EGFR-TKI Plus Everolimus Combination Therapy. Trans Lung Cancer Res (2020) 9:1258–67. doi: 10.21037/tlcr-20-141
13. Zhou G, Zhang F, Guo Y, Huang J, Xie Y, Yue S, et al. miR-200c Enhances Sensitivity of Drug-Resistant non-Small Cell Lung Cancer to Gefitinib by Suppression of PI3K/Akt Signaling Pathway and Inhibites Cell Migration via Targeting ZEB1. BioMed Pharmacother (2017) 85:113–9. doi: 10.1016/j.biopha.2016.11.100
14. Hao P, Huang Y, Peng J, Yu J, Guo X, Bao F, et al. IRS4 Promotes the Progression of Non-Small Cell Lung Cancer and Confers Resistance to EGFR-TKI Through the Activation of PI3K/Akt and Ras-MAPK Pathways. Exp Cell Res (2021) 403:112615. doi: 10.1016/j.yexcr.2021.112615
15. Dong X, Zeng Y, Liu Y, You L, Yin X, Fu J, et al. Aloe-Emodin: A Review of Its Pharmacology, Toxicity, and Pharmacokinetics. Phytother Res (2020) 34:270–81. doi: 10.1002/ptr.6532
16. Ma JW, Hung CM, Lin YC, Ho CT, Kao JY, Way TD. Aloe-Emodin Inhibits HER-2 Expression Through the Downregulation of Y-Box Binding Protein-1 in HER-2-Overexpressing Human Breast Cancer Cells. Oncotarget (2016) 7:58915–30. doi: 10.18632/oncotarget.10410
17. Dou F, Liu Y, Liu L, Wang J, Sun T, Mu F, et al. Aloe-Emodin Ameliorates Renal Fibrosis Via Inhibiting PI3K/Akt/mTOR Signaling Pathway In Vivo and In Vitro. Rejuvenation Res (2019) 22:218–29. doi: 10.1089/rej.2018.2104
18. Cheng G, Pi Z, Zhuang X, Zheng Z, Liu S, Liu Z, et al. The Effects and Mechanisms of Aloe-Emodin on Reversing Adriamycin-Induced Resistance of MCF-7/ADR Cells. Phytother Res (2021) 35:3886–97. doi: 10.1002/ptr.7096
19. Tabolacci C, Cordella M, Turcano L, Rossi S, Lentini A, Mariotti S, et al. Aloe-Emodin Exerts a Potent Anticancer and Immunomodulatory Activity on BRAF-Mutated Human Melanoma Cells. Eur J Pharmacol (2015) 762:283–92. doi: 10.1016/j.ejphar.2015.05.057
20. Xiao F, Liu Y, Zhang Z, Wang L, Wang T, Wang X. Tobacco Extracts Promote PD-L1 Expression and Enhance Malignant Biological Differences via mTOR in Gefitinib-Resistant Cell Lines. Thorac Cancer (2020) 11:2237–51. doi: 10.1111/1759-7714.13533
21. Singh A, Settleman J EMT. Cancer Stem Cells and Drug Resistance: An Emerging Axis of Evil in the War on Cancer. Oncogene (2010) 29:4741–51. doi: 10.1038/onc.2010.215
22. Erin N, Grahovac J, Brozovic A, Efferth T. Tumor Microenvironment and Epithelial Mesenchymal Transition as Targets to Overcome Tumor Multidrug Resistance. Drug Resist Updates (2020) 53:100715. doi: 10.1016/j.drup.2020.100715
23. Aleksakhina SN, Kashyap A, Imyanitov EN. Mechanisms of Acquired Tumor Drug Resistance Biochim Biophys. Acta Rev Cancer (2019) 1872:188310. doi: 10.1016/j.bbcan.2019.188310
24. Ramezani Farani M, Azarian M, Heydari Sheikh Hossein H, Abdolvahabi Z, Mohammadi Abgarmi Z, Moradi A, et al. Folic Acid-Adorned Curcumin-Loaded Iron Oxide Nanoparticles for Cervical Cancer. ACS Appl Bio Mater (2022) 5:1305–18. doi: 10.1021/acsabm.1c01311
25. Moammeri A, Abbaspour K, Zafarian A, Jamshidifar E, Motasadizadeh H, Dabbagh Moghaddam F, et al. pH-Responsive, Adorned Nanoniosomes for Codelivery of Cisplatin and Epirubicin: Synergistic Treatment of Breast Cancer. ACS Appl Bio Mater (2022) 5:675–90. doi: 10.1021/acsabm.1c01107
26. Herbst RS, Morgensztern D, Boshoff C. The Biology and Management of non-Small Cell Lung Cancer. Nature (2018) 553:446–54. doi: 10.1038/nature25183
27. Sanders B, Ray AM, Goldberg S, Clark T, McDaniel HR, Atlas SE, et al. Anti-Cancer Effects of Aloe-Emodin: A Systematic Review. J Clin Transl Res (2018) 3:283–96. doi: 10.1016/j.bbcan.2019.188310
28. Raoof S, Mulford IJ, Frisco-Cabanos H, Nangia V, Timonina D, Labrot E, et al. Targeting FGFR Overcomes EMT-Mediated Resistance in EGFR Mutant Non-Small Cell Lung Cancer. Oncogene (2019) 38:6399–413. doi: 10.1038/s41388-019-0887-2
29. Gooding AJ, Schiemann WP. Epithelial–mesenchymal Transition Programs and Cancer Stem Cell Phenotypes: Mediators of Breast Cancer Therapy Resistance. Mol Cancer Res (2020) 18:1257–70. doi: 10.1158/1541-7786.MCR-20-0067
30. Wei R, Xiao Y, Song Y, Yuan H, Luo J, Xu W. FAT4 Regulates the EMT and Autophagy in Colorectal Cancer Cells in Part via the PI3K-AKT Signaling Axis. J Exp Clin Cancer Res (2019) 38:112. doi: 10.1186/s13046-019-1043-0
31. Xu E, Xia X, Jiang C, Li Z, Yang Z, Zheng C, et al. GPER1 Silencing Suppresses the Proliferation, Migration, and Invasion of Gastric Cancer Cells by Inhibiting PI3K/AKT-Mediated EMT. Front Cell Dev Biol (2020) 8:591239. doi: 10.3389/fcell.2020.591239
32. Chen Y, Liu P, Shen D, Liu H, Xu L, Wang J, et al. FAM172A Inhibits EMT in Pancreatic Cancer via ERK-MAPK Signalling. Biol Open (2020) 9:bio048462. doi: 10.1242/bio.048462
33. Wang J, Zhang Z, Li R, Mao F, Sun W, Chen J, et al. ADAM12 Induces EMT and Promotes Cell Migration, Invasion and Proliferation in Pituitary Adenomas via EGFR/ERK Signaling Pathway. BioMed Pharmacother (2018) 97:1066–77. doi: 10.1016/j.biopha.2017.11.034
34. Wen L, Zhang X, Zhang J, Chen S, Ma Y, Hu J, et al. Paxillin Knockdown Suppresses Metastasis and Epithelial−Mesenchymal Transition in Colorectal Cancer via the ERK Signalling Pathway. Oncol Rep (2020) 44:1105–15. doi: 10.3892/or.2020.7687
35. Wang K, Ji W, Yu Y, Li Z, Niu X, Xia W, et al. FGFR1-ERK1/2-SOX2 Axis Promotes Cell Proliferation, Epithelial-Mesenchymal Transition, and Metastasis in FGFR1-Amplified Lung Cancer. Oncogene (2018) 37:5340–54. doi: 10.1038/s41388-018-0311-3
Keywords: aloe-emodin, gefitinib, NSCLC, drug resistance, EMT
Citation: Peng M, Zheng Z, Chen S, Fang L, Feng R, Zhang L, Tang Q and Liu X (2022) Sensitization of Non-Small Cell Lung Cancer Cells to Gefitinib and Reversal of Epithelial–Mesenchymal Transition by Aloe-Emodin Via PI3K/Akt/TWIS1 Signal Blockage. Front. Oncol. 12:908031. doi: 10.3389/fonc.2022.908031
Received: 30 March 2022; Accepted: 19 April 2022;
Published: 23 May 2022.
Edited by:
Gautam Sethi, National University of Singapore, SingaporeReviewed by:
Peramaiyan Rajendran, King Faisal University, Saudi ArabiaCopyright © 2022 Peng, Zheng, Chen, Fang, Feng, Zhang, Tang and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xuewen Liu, YXZlaW5saXVAMTYzLmNvbQ==
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.