
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Oncol., 28 June 2022
Sec. Thoracic Oncology
Volume 12 - 2022 | https://doi.org/10.3389/fonc.2022.869390
This article is part of the Research TopicPrimary and Acquired Resistance in Lung CancerView all 10 articles
 Po-Chun Hsieh1
Po-Chun Hsieh1 Yao-Kuang Wu2,3
Yao-Kuang Wu2,3 Chun-Yao Huang2,3
Chun-Yao Huang2,3 Mei-Chen Yang2,3
Mei-Chen Yang2,3 Chan-Yen Kuo4
Chan-Yen Kuo4 I-Shiang Tzeng4
I-Shiang Tzeng4 Chou-Chin Lan2,3*
Chou-Chin Lan2,3*Background: Lung adenocarcinoma is a common disease with a high mortality rate. Epidermal growth factor receptor (EGFR) mutations are found in adenocarcinomas, and oral EGFR-tyrosine kinase inhibitors (EGFR-TKIs) show good responses. EGFR-TKI therapy eventually results in resistance, with the most common being T790M. T790M is also a biomarker for predicting resistance to first- and second-generation EGFR-TKIs and is sensitive to osimertinib. The prognosis was better for patients with acquired T790M who were treated with osimertinib than for those treated with chemotherapy. Therefore, T790M mutation is important for deciding further treatment and prognosis. Previous studies based on small sample sizes have reported very different T790 mutation rates. We conducted a meta-analysis to evaluate the T790M mutation rate after EGFR-TKI treatment.
Methods: We systematic reviewed the electronic databases to evaluate the T790M mutation rate after treatment with first-generation (gefitinib, erlotinib, and icotinib) and second-generation (afatinib and dacomitinib) EGFR-TKIs. Random-effects network meta-analysis and single-arm meta-analysis were conducted to estimate the T790M mutation rate of the target EGFR-TKIs.
Results: A total of 518 studies were identified, of which 29 were included. Compared with afatinib, a higher odds ratio (OR) of the T790M mutation rate was observed after erlotinib [OR = 1.48; 95% confidence interval (CI):1.09–2.00] and gefitinib (OR = 1.45; 95% CI: 1.11–1.90) treatments. An even OR of the T790M mutation rate was noted after icotinib treatment (OR = 0.91, 95% CI: 0.46–1.79) compared with that after afatinib. The T790M mutation rate was significantly lower with afatinib (33%) than that with gefitinib (49%) and erlotinib treatments (47%) (p < 0.001). The acquired T790M mutation rate in all participants was slightly lower in Asians (43%) than that in Caucasians (47%).
Conclusions: Erlotinib and gefitinib had a higher OR for the T790M mutation than afatinib. The T790M mutation rate was significantly lower in afatinib than in gefitinib and erlotinib. T790M is of great significance because osimertinib shows a good prognosis in patients with T790M mutation.
Systematic Review Registration: PROSPERO, identifier CRD42021257824.
Lung cancer is associated with significant mortality rates worldwide. Non-small-cell lung cancer (NSCLC) accounts for approximately 80% of all lung cancer cases, and its treatment depends on the stage and gene profiles of the tumors (1). Most patients with NSCLC are at an advanced stage at the time of diagnosis, have unresectable tumors, and usually present with a poor prognosis (1). Therefore, targeted therapy and chemotherapy are major treatments for these patients (1).
Traditionally, chemotherapy has been the standard treatment for patients with NSCLC. However, chemotherapy often causes serious adverse reactions and complications that can render patients unable to receive a complete course of treatment. Adenocarcinomas account for 80% of all NSCLC cases (1). Epidermal growth factor receptor (EGFR) mutations occur in approximately 50% of Asian and 20% of Caucasian patients with lung adenocarcinoma (2). Oral EGFR-tyrosine kinase inhibitors (EGFR-TKIs) have become promising treatments for patients with adenocarcinoma because of their good curative effects and few adverse reactions.
EGFR is a tyrosine kinase receptor that plays a key role in tumor cell proliferation and vascularization. Hence, it is an important molecular target in cancer treatment. Previous studies have shown that EGFR-TKIs are superior to paclitaxel/carboplatin in NSCLC patients with EGFR-sensitizing gene mutations. This finding implies that the effective treatment of NSCLC consists of EGFR-TKIs. Currently, the available EGFR-TKIs for NSCLC are first- (gefitinib, erlotinib, and icotinib), second- (afatinib and dacomitinib), and third-generation TKIs (osimertinib) (3).
Gefitinib was approved for patients with advanced NSCLC and sensitive EGFR mutations in July 2015 (4). Gefitinib as the first line of treatment for NSCLC patients with sensitive EGFR mutations showed an objective response rate (ORR) of 62–71%, progression-free survival (PFS) of 8–13 months, and overall survival (OS) of 21–30 months (4). Erlotinib was approved in 2004 for patients harboring EGFR exon 21 L858R mutations and exon 19 deletions (5). Erlotinib as the first line of treatment for NSCLC patients with sensitive EGFR gene mutations revealed an ORR of 58–83%, PFS of 9.7–13 months, and OS of 23–33 months (5). Afatinib is an irreversible covalent inhibitor of the ErbB receptor family, which includes EGFR, ErbB2/HER2, and ErbB4/HER4 (6). It was approved by the FDA for treating NSCLC patients with exon 21 L858R substitutions and exon 19 deletions in 2013 and for uncommon EGFR mutations such as L861Q in exon 21 and G719X in exon 18 in 2018 (6). Afatinib, as the first line of treatment for NSCLC patients with sensitive EGFR gene mutations, showed an ORR of 70%–81.8%, PFS of 13.4–15.2 months, and OS of 27.9–49 months (6).
Predictive biomarkers are important for the treatment of NSCLC. In previous studies, PDL-1 expression was found to be a predictive biomarker for the therapeutic response to immunotherapy (7). The clinical outcomes of patients with higher PDL-1 expression were better PFS and OS associated with immunotherapy (7). However, evidence shows that patients with metastatic squamous cell lung cancer tend to benefit from immunotherapy, regardless of PD-L1 status (7). Tumor mutational burden (TMB) also serves as a predictive biomarker for immunotherapy, and OS was in favor of chemotherapy for patients with low TMB and immunotherapy for patients with high TMB (7). Previous evidence suggests that micro RNAs may serve as biomarkers of response to cancer treatment and enable better management decisions (8). Furthermore, micro RNAs can be used as biomarkers for lung cancer screening and are associated with OS (8). EGFR mutations are the most important biomarkers for predicting treatment response to EGRF-TKIs. EGFR gene mutations mainly occur in the 18–21 exon and classical mutations refer to deletions in exon 19 and point mutation L858R in exon 21, which account for approximately 85% of all EGFR mutations (9). These mutations are associated with sensitivity to EGFR-TKIs such as gefitinib, erlotinib, afatinib, and icotinib (9, 10). EGFR-TKIs have better outcomes than chemotherapy as the first line of treatment in patients with EGFR-mutant NSCLC (10).
Although these EGFR-TKIs show good responses in NSCLC patients with EGFR-sensitizing genes (3), all treated patients eventually develop acquired resistance. The mechanisms of acquired resistance to first- or second-generation EGFR-TKIs include the T790M mutation, ERBB2 amplification, MET amplification, and transformation to small-cell lung cancer, of which T790M mutations are the most common resistance mechanism (11). The main process of developing T790M is a single nucleotide transition mutation in EGFR, a cytosine to thymine (C>T) mutation at position 2369, causing a threonine to methionine amino acid change at codon 790 (12). The T790M mutation leads to steric hindrance, increased binding affinity for ATP, and downstream signal transduction. When encountering patients with T790M, physicians can choose osimertinib as further therapy. As second-line therapy for NSCLC patients with acquired T790M mutations, osimertinib has better outcomes than platinum-based chemotherapy (13). Therefore, research on T790M mutation rate is of great significance because it may be related to further treatment strategies and prognosis. Therefore, T790M also serves as a biomarker for resistance to first- and second-generation EGFR-TKIs and sensitivity to osimertinib (14). The prognosis was better for patients with acquired T790M who were treated with osimertinib than for those treated with chemotherapy (13, 15–17). In patients without acquired T790M, PFS with chemotherapy is worse (13, 17). Therefore, T790M mutation is a prognostic factor.
It is important to understand the T790M mutation rate in patients with NSCLC after treatment with EGFR-TKIs. Many studies have been conducted on the T790M mutation rate after treatment with EGFR-TKIs. These studies suggest that the acquired T790M mutation rate is approximately 50%–60% (11), and the acquired T790M mutation rate with afatinib is lower than that with gefitinib or erlotinib (11). However, the range of positive rates for acquired T790M was considerably wide in these studies. In addition, many of these data come from studies with very small sample sizes, some even fewer than ten patients. Therefore, such statistics produce significant errors and no definite conclusions can be obtained.
Due to the very small number of subjects in these studies and the wide range of T790M mutation rates, it is difficult to determine whether the T790M mutation rate after afatinib treatment is lower than that after first-generation EGFR-TKIs. To solve this problem, we conducted a meta-analysis to analyze the T790M mutation rate after treatment with first- and second-generation EGFR-TKIs using direct and indirect comparisons.
This study was performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) extension guidelines for network meta-analysis (18). A prospective protocol was created in advance and registered on the International Prospective Register of Systematic Reviews PROSPERO website (registration number: CRD42021257824).
We performed a comprehensive literature search of electronic databases, including Embase, Cochrane Library, PubMed, and ClinicalTrials.gov, from their inception until May 31, 2021, without language restrictions. We aimed to compare the acquired T790M acquisition rates after treatment with different first-generation (gefitinib, erlotinib, and icotinib) and second-generation (afatinib and dacomitinib) EGFR-TKIs in patients with NSCLC. The detailed definitions of PICOS are listed in Table S1. The full details of the search strategy are listed in Table S2.
Studies were included under the following conditions: (1) observational studies, including prospective and retrospective cohort studies; (2) patients with NSCLC treated with only one EGFR-TKI during the study; (3) reported acquired T790M acquisition rates in separate EGFR-TKI groups; and (4) published as full-length articles. The exclusion criteria were (1) case-control studies or case reports; (2) T790M acquisition detected before EGFR-TKI treatment; (3) patients administered more than one EGFR-TKI; (4) EGFR-TKIs combined with chemotherapy or anti-vascular endothelial growth factor therapy; and (5) published articles, posters, or abstracts with limited information that could not be used for analysis. Bibliographies of the included studies and related systematic review articles were manually reviewed for relevant references. Two reviewers (PCH and YKW) independently reviewed the titles and abstracts of identified articles. Discrepancies or issues between reviewers were resolved by consulting a third reviewer (CCL) as an arbiter.
A predetermined form was used by two reviewers (PCH and YKW) independently for data extraction of the following information: (1) publication year, (2) authors, (3) countries where the research was conducted, (4) NSCLC stages, (5) EGFR-TKIs, (6) number of patients with acquired resistance, (7) baseline characteristics and outcomes (sex, age, L858R mutation, exon 19 deletion, PFS, and OS); (8) biopsy sample types for examination; and (9) detection methods of T790M acquisition.
The outcome was the acquired T790M acquisition rate in the research cohort after first- or second-generation EGFR-TKI treatment.
We summarized the odds ratio (OR) with a 95% confidence interval (CI) as the effective size for measuring the acquired T790M acquisition rate. All graph generation and statistical analyses were performed using the statistical software RStudio (version 1.4.1106) (19). To compare the acquired T790M acquisition rate between the target EGFR-TKIs, network meta-analyses were conducted using “netmeta”, “ggplot2”, and “reshape2” packages. A random-effects network meta-analysis was performed using a consistency model. Single-arm meta-analyses with random-effects models were conducted using “meta” and “metafor” packages to estimate the specific acquired T790M acquisition rate of the target EGFR-TKIs. Subgroup analyses with Asian or Caucasian populations were conducted because of the varying characteristics of different races. Q and I2 statistics were used to quantify heterogeneity among the included studies.
If more than 10 studies were included in the analysis, a funnel plot was used to examine publication bias. We performed Egger’s test to assess the existence of bias in small-sample studies. Within the network meta-analysis results, a plot of direct evidence proportions was constructed to quantify the percentage of direct and indirect evidence proportions for each network estimate (20). Inconsistent assumptions were assessed using a node‐splitting model and design‐by‐treatment interaction model. Within the single-arm meta-analysis, a meta-regression analysis was performed to explore the potential associations between the effect size and target EGFR-TKIs. If more than two studies were included in the single-arm meta-analysis, an influence analysis was performed using the leave-one-out method.
Two reviewers (PCH and YKW) independently assessed the methodological quality of the retrieved multi-cohort studies using the ROBINS-I tool (21), and discrepancies were resolved by a third reviewer (CCL).
The review process is illustrated in Figure 1. A total of 518 studies were identified using the search terms in the electronic databases, with 200 studies on PubMed, 265 on Embase, 31 on Cochrane Library, and 22 on ClinicalTrials.gov (Table S2). After removing duplicate studies and excluding titles and abstracts, 56 studies were considered for full-text evaluation, and 27 studies were excluded for different reasons (Table S3). Finally, 29 studies [including 23 multi-cohort studies (5, 11, 22–42) and six single-cohort studies (43–48)] were included in the risk of bias assessment and single-arm meta-analysis, and 20 multi-cohort studies were included in the network meta-analysis. Among the studies identified in the search results, acquired T790M acquisition rates after treatment with gefitinib, erlotinib, icotinib, or afatinib were noted. To our knowledge, no study has reported the acquired T790M acquisition rate after dacomitinib treatment. A summary of the retrieved studies is shown in Table 1.
The characteristics of the participants are presented in Tables 1 and S4. The final quantitative analysis included 3385 participants (age: 27–93-years-old), with stages I–IV and advanced, recurrent, or metastatic NSCLC. Twenty-four studies were conducted in Asia (12 in Japan, 5 in Korea, 4 in China, and 3 in Taiwan; with 2883 Asian participants), and 5 studies were conducted in Europe and North America (2 in Italy, 1 in Germany, and 2 in the USA; with 502 Caucasian participants).
In terms of the acquired T790M acquisition rate following treatment with gefitinib, erlotinib, icotinib, and afatinib, 20 multi-cohort studies (5, 22–37, 39–41) were included in the network meta-analysis. The structure of the network is shown in Figure 2A. A forest plot of the network meta-analysis is shown in Figure 2B. There was no statistical heterogeneity among the included studies, with an I2 of 0% (95% CI: 0–32.6), and the Q statistic was 25.04% (p = 0.57) for within-design and 1.56 (p = 0.66) for between-designs, indicating no heterogeneity and consistency in the model used. Compared with afatinib, a higher OR of acquired T790M acquisition rate was observed after erlotinib (OR = 1.48; 95% CI: 1.09–2.00) and gefitinib (OR = 1.45; 95% CI: 1.11–1.90) treatments. The results also indicated an even OR of acquired T790M acquisition rate after treatment with icotinib (OR = 0.91, 95% CI: 0.46–1.79) compared with that after afatinib treatment in NSCLC patients. According to the league table (Table 2) and P-scores (Table S5), erlotinib was associated with the highest risk of acquired T790M acquisition rate, followed by gefitinib.
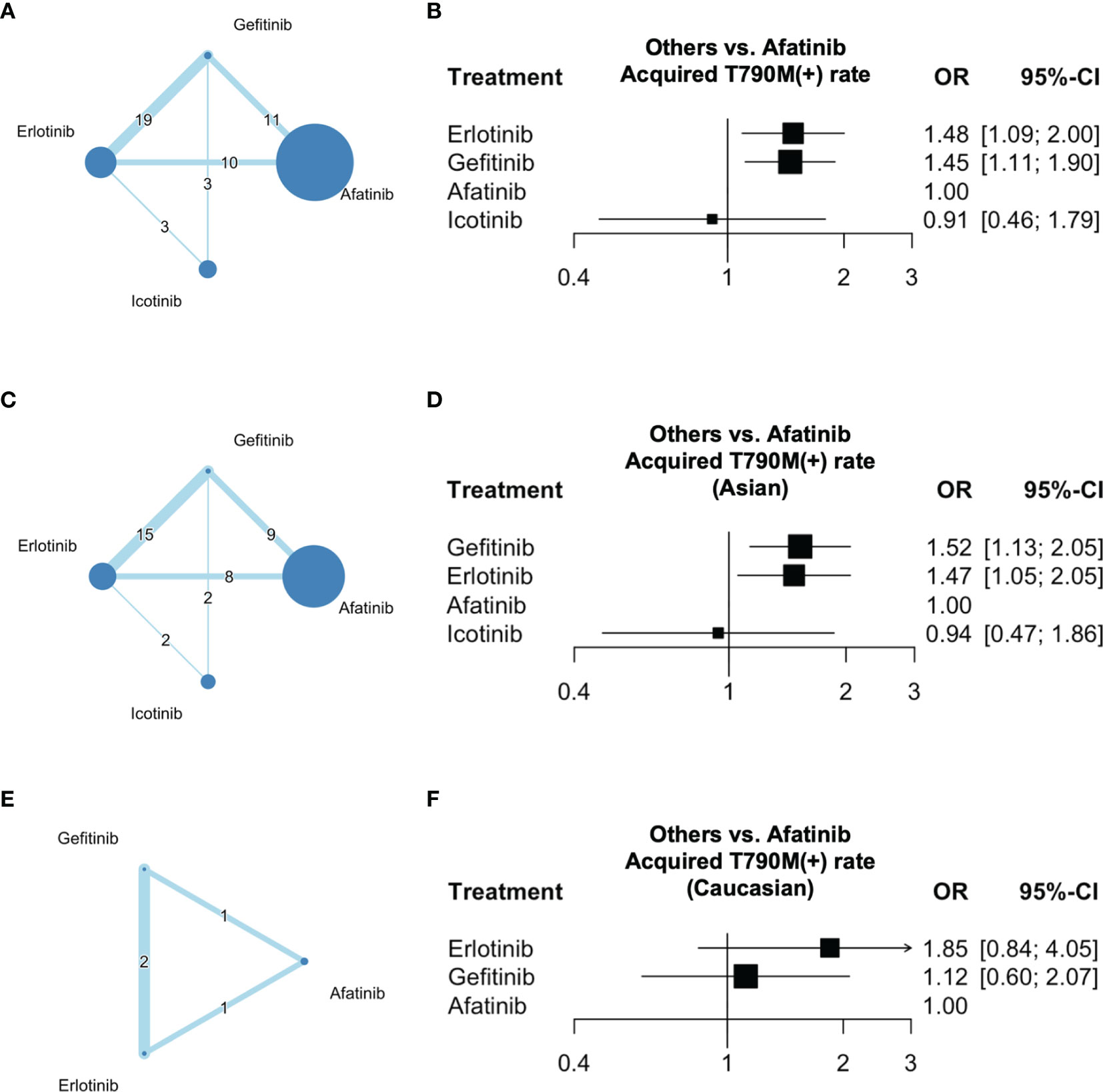
Figure 2 Network of the comparisons and forest plot for the network meta-analysis. Networks of eligible EGFR-TKIs comparisons for outcomes of acquired T790M mutation rate for (A) all participants, (C) Asians, and (E) Caucasians. Forest plots of eligible EGFR-TKI comparisons for outcomes of acquired T790M mutation rate for (B) all participants, (D) Asians, and (F) Caucasians. Network: The size of the nodes corresponds to the number of studies for each treatment. The lines between nodes represent a direct comparison of the trials and the thickness of the lines linking nodes corresponds to the number of trials included.

Table 2 League table with network meta-analysis estimates of acquired T790M mutation rate in all participants.
In terms of the acquired T790M acquisition rates in Asian patients following treatment with gefitinib, erlotinib, icotinib, and afatinib, 18 multi-cohort studies (5, 23–37, 39, 41) were included in the subgroup network meta-analysis. The structure of the network is shown in Figure 2C. A forest plot of the network meta-analysis is shown in Figure 2D. There was no statistical heterogeneity among the included studies, with I2 0% (95% CI: 0–34.8), and the Q statistic was 22.35 (p = 0.55) for within-design and 1.70 (p = 0.63) for between-designs, indicating no heterogeneity and consistency in the model used. The results indicated a higher OR of acquired T790M acquisition rate after treatment with gefitinib (OR = 1.52; 95% CI: 1.13–2.05) and erlotinib (OR = 1.47; 95% CI: 1.05–2.05) than that after afatinib treatment in patients with NSCLC. Furthermore, an even OR of acquired T790M acquisition rate was observed after icotinib treatment (OR = 0.94; 95% CI: 0.47–1.86) compared with that in afatinib-treated patients with NSCLC. According to the league table (Table 3) and P-scores (Table S5), gefitinib was associated with the highest risk of acquired T790M acquisition rate, followed by erlotinib.

Table 3 League table with network meta-analysis estimates of acquired T790M mutation rate in Asian patients.
In terms of the acquired T790M acquisition rate in Caucasian patients following treatment with gefitinib, erlotinib, and afatinib, two multi-cohort studies (22, 40) were included in the subgroup network meta-analysis. The structure of the network is shown in Figure 2E. No statistical heterogeneity was observed among the included studies, with an I2 value of 0%. The Q statistic was 0.11 (p = 0.91) for between-designs, indicating no heterogeneity and consistency in the model used. A forest plot of the network meta-analysis is shown in Figure 2F. The results indicated an even OR of acquired T790M acquisition rate after treatment with erlotinib (OR = 1.85; 95% CI: 0.84–4.05) and gefitinib (OR = 1.12, 95% CI: 0.60–2.07) compared with that in afatinib-treated patients with NSCLC. According to the league table (Table 4) and P-scores (Table S5), gefitinib, erlotinib, and afatinib were associated with an even risk of an acquired T790M acquisition rate.

Table 4 League table with network meta-analysis estimates of acquired T790M mutation rate in Caucasian patients.
In terms of specific acquired T790M acquisition rates with EGFR-TKIs following treatment with gefitinib, erlotinib, icotinib, and afatinib, 29 studies (5, 11, 22–48) were included in the single-arm meta-analysis. A forest plot of the analysis is shown in Figure 3. The overall rate of acquired T790M acquisition was 44% (95% CI: 40–47; I2 = 71%). The specific acquired T790M acquisition rates were 49% for gefitinib (95% CI: 44–54; I2 = 74%), 47% for erlotinib (95% CI: 43–52; I2 = 16%), 37% for icotinib (95% CI: 0–46; I2 = 0%), and 33% for afatinib (95% CI, 24–41; I2 = 76%). The meta-regression results indicated potential associations between the acquired T790M acquisition rate and different EGFR-TKIs, with statistical significance (p < 0.0001) (Table S6).
In terms of specific acquired T790M acquisition rates in Asian patients following treatment with gefitinib, erlotinib, icotinib, and afatinib, 24 studies (5, 23–39, 42–45, 47, 48) were included in the single-arm meta-analysis. A forest plot of the analysis is shown in Figure 4. The overall acquired T790M acquisition rate was 43% (95% CI: 39–47; I2 = 73%). The specific acquired T790M acquisition rates were 49% for gefitinib (95% CI: 43–52; I2 = 76%), 46% for erlotinib (95% CI: 41–50; I2 = 5%), 37% for icotinib (95% CI: 0–46; I2 = 0%), and 30% for afatinib (95% CI: 0–41; I2 = 80%). Meta-regression results indicated potential associations between the acquired T790M acquisition rate and different EGFR-TKIs in Asians, with statistical significance (p < 0.0001) (Table S6).
In terms of the acquired T790M acquisition rate of EGFR-TKIs in Caucasian patients following treatment with gefitinib, erlotinib, and afatinib, five studies (11, 22, 40, 41, 46) were included in the single-arm meta-analysis. A forest plot of the analysis is shown in Figure 5. The overall acquired T790M acquisition rate was 47% (95% CI: 42–53; I2 = 12%). The specific acquired T790M acquisition rates were 49% for gefitinib (95% CI: 40–57; I2 = 0%), 57% for erlotinib (95% CI: 45–68; I2 = 0%), and 42% for afatinib (95% CI: 35–50; I2 = 0%). Meta-regression results indicated no potential association between the acquired T790M acquisition rate and different EGFR-TKIs in Caucasians (p = 0.6621) (Table S6).
We also conducted a meta-regression analysis to investigate the potential associations between gefitinib, erlotinib, and afatinib treatments and Asians/Caucasians. In terms of the acquired T790M acquisition rate of gefitinib, the results indicated potential associations between the acquired T790M acquisition rate and Asian/Caucasian populations (p < 0.0001). In terms of the acquired T790M acquisition rate of erlotinib, the results indicated no potential association between the acquired T790M acquisition rate and Asians/Caucasians (p = 0.3941). In terms of the acquired T790M acquisition rate of afatinib, the results indicated potential associations between the acquired T790M acquisition rate and Asian/Caucasian populations (p < 0.0001). Gefitinib and afatinib treatments had different effects on the acquired T790M acquisition rates in Asians and Caucasians (Table S7).
We conducted single-arm meta-analyses and meta-regression analyses to evaluate the rate of acquired T790M acquisition in tissue or plasma biopsy samples. Forest plots of the analyses are shown in Figure S1. The results showed that after treatment with gefitinib, the acquired T790M acquisition rate was 52% (95% CI: 48–56; I2 = 30%) in tissue biopsy samples, whereas the acquired T790M acquisition rate was 27% (95% CI: 21–33) in plasma biopsy samples. After treatment with erlotinib, the acquired T790M acquisition rate was 46% (95% CI: 40–52; I2 = 22%) in tissue biopsy samples, whereas the acquired T790M acquisition rate was 40% (95% CI: 26–54) in plasma biopsy samples. Furthermore, after treatment with afatinib, the acquired T790M acquisition rate was 39% (95% CI: 34–45; I2 = 0%) in tissue biopsy samples, whereas the acquired T790M mutation rate was 33% (95% CI: 23–42; I2 = 65%) in plasma biopsy samples. Meta-regression analysis results showed potential associations between the acquired T790M mutation rate and tissue/plasma biopsy after treatment with gefitinib, erlotinib, or afatinib (Table S8). After treatment with gefitinib, erlotinib, or afatinib, the detection rate of the acquired T790M mutation was significantly lower in plasma biopsy samples than that in tissue biopsy samples.
Among the network meta-analyses of all Asian patients, funnel plots of publication bias showed general symmetry. Egger’s test showed no significant publication bias among the included studies (Figures S2A, B). Because only two studies were included in the network meta-analysis for Caucasians, no further assessment of publication bias was performed.
In the single-arm meta-analysis of all participants, funnel plots of publication bias showed general symmetry (Figure S3A). Because the intercept was close to zero, the small study bias was not significant (Figure S3B). In the single-arm meta-analysis for Asians, funnel plots of publication bias showed a general symmetry (Figure S3C). Because the intercept was significantly close to zero, the small study bias was not significant (Figure S3D). As only three studies were included in the single-arm meta-analysis for Caucasians, no further assessment of publication bias was performed.
In the network meta-analyses, direct evidence plots for all patients, Asian and Caucasian are presented in Figure S4. We found no evidence of inconsistencies using either node-splitting (Figure S5) or design-by-treatment interaction model approaches (Figure S6).
In the single-arm meta-analysis for gefitinib, erlotinib, icotinib, and afatinib treatment in Asian patients, the results indicated no significant changes in the integrated or after eliminating each study individually (Figure S7). For the single-arm meta-analysis of afatinib in Caucasians, the results indicated no significant changes in the integrated OR while eliminating each study individually (Figure S8).
The ROBINS-I results are presented in Table S9. Most of the studies had a moderate risk of overall bias. There were three main reasons. (1) For baseline EGFR mutation types, the number of patients with L858R mutations or exon 19 deletions was not balanced or adjusted for different EGFR-TKI-treated groups. Hence, we propose a moderate risk of bias owing to confounding factors. (2) For the detection of T790M mutation, five studies used multiple methods (Table 1) (35–37, 41, 42). As the detection accuracy differed according to the detection method (49, 50), we proposed a moderate risk of bias in the measurement of outcomes. (3) For the re-biopsy samples, six studies used multiple types of samples, including tissue, plasma, and fluid (Table 1) (27, 28, 32, 35, 37, 42). As the detection accuracy differed according to the type of examination sample (50), we proposed a moderate risk of bias in the measurement of outcomes.
This is the first meta-analysis of the acquired T790M mutation rate associated with treatment using different EGFR-TKIs, and it has revealed that the acquired T790M mutation rate was significantly lower with afatinib (33%) than that with gefitinib (49%) and erlotinib (47%) (p < 0.05) treatments in the overall population. The first- and second-generation EGFR-TKIs are different. The mechanisms underlying the lower T790M mutation rate after afatinib treatment are unclear; several hypotheses can explain this result.
Initially, gefitinib, erlotinib, and afatinib were considered similar EGFR-TKIs. However, gefitinib and erlotinib are reversible EGRF-TKIs with similar activities in in vitro and xenograft assays. In our analysis, the incidence of acquired T790M mutations was similar following treatment with gefitinib and erlotinib. In contrast, afatinib is an irreversible EGFR-TKI that inhibits the ErbB receptor family and causes rare EGFR mutations, including exon 18 p.G719X and exon 21 p.L861Q point mutations (6). Afatinib has pharmacological characteristics that differ from those of gefitinib and erlotinib.
El Kadi et al. found that acquired EGFR T790M occurs mainly through activation-induced cytidine deaminase (AICDA)-mediated deamination of 5-methylcytosine following TKI treatment (12). They reported that EGFR-TKI treatment leads to activation of the nuclear factor-kappa B pathway, which in turn induces the expression of AICDA, further causing deamination of 5-methylcytosine to thymine at position c.2369 to generate the T790M mutation. The different pharmacological characteristics of these EGFR-TKIs may lead to different rates of acquired T790M mutation. Gefitinib and erlotinib showed higher AICDA expression than afatinib (12). Therefore, it is rational to understand the higher frequencies of the T790M mutation rates following treatment with gefitinib and erlotinib.
Another hypothesis is that clonal selection during different EGFR-TKI treatments may lead to different clonality of the acquired resistance. Previous studies have shown that afatinib suppresses the growth of lung cancer cells harboring T790M cells (51, 52). Furthermore, afatinib exerts a 100-fold potent activity against T790M cell lines than first-generation EGFR-TKIs (51). Afatinib was initially considered a potential salvage therapy after first-generation TKIs. However, the clinical use of afatinib as salvage therapy after first-generation TKIs has been disappointing (52) because of the difficulty in increasing the clinical dose of afatinib to reach the afatinib concentration in the human body in an in vitro study (52). Although afatinib cannot effectively overcome T790M at a clinically achievable dose, it may reduce the occurrence of T790M colonies. Therefore, T790M subclonies are likely to be enriched under the different effects of gefitinib, erlotinib, and afatinib (39). Previous studies have shown that prolonged exposure to EGFR-TKIs promotes selective survival of T790M-positive cells (5, 53, 54). These results support the hypothesis of clonal selection for EGFR-TKI therapy.
Interestingly, we found that the rate of acquired T790M mutation was slightly lower in Asians (43%) than that in Caucasians (47%). Asian and Caucasian patients with lung cancer have different genetic susceptibilities (2). For example, common EGFR mutations in adenocarcinoma occur in approximately 50% of Asian patients and 20% of Caucasian patients (2). In contrast, the incidence of KRAS mutations in European populations (30%) is higher than that in Asian populations (<10%) (2). The exact mechanisms for the different EGFR or KRAS mutations in Asian and Caucasian populations remain unclear. In this meta-analysis, we found that the rate of acquired T790M mutation was higher in Caucasian patients than that in Asian patients. However, the reason for this difference remains unclear.
The clinical outcomes of EGFR-TKIs are controversial. It has been shown that for NSCLC patients with sensitive EGFR mutations, first-line treatment with gefitinib yielded PFS of 8–13 months (55), erlotinib yielded a PFS of 9.7–13 months (5), and afatinib yielded a PFS of 13.4–15.2 months (6). The LUX-Lung 7 trial suggested that afatinib achieved superior clinical outcomes compared with gefitinib-treated patients bearing EGFR L858R or exon 19 deletions (56). However, other studies have shown that PFS and OS are similar in gefitinib- and afatinib-treated patients (39, 57). In a real-world study, the clinical outcomes of PFS or OS were similar among patients treated with gefitinib, erlotinib, or afatinib for NSCLC patients bearing sensitizing EGFR mutations (5). Therefore, the clinical outcomes were similar among the three EGFR-TKIs. However, these EGFR-TKIs have different incidences of T790M mutation.
Patients with acquired T790M can choose osimertinib treatment, whereas those without acquired T790M can only receive chemotherapy. As shown in Figure 6, the clinical prognosis was better for patients with acquired T790M mutations who were treated with osimertinib. PFS of NSCLC patients with acquired T790M after treatment with gefitinib, erlotinib, and afatinib was 10.4-15.6 months (13, 15, 16). The PFS of NSCLC patients with acquired T790M mutation treated with platinum-based chemotherapy was only 6 months (17). In patients without acquired T790M, PFS after platinum-based chemotherapy was worse, only 4.4–5.1 months (13, 17). Joo et al. also suggested that osimertinib treatment was independently associated with better outcomes, such as longer OS and PFS (54). A preclinical model also suggested that T790M-positive cells grow more slowly than T790M-negative cells (53). Thus, T790M appears to be a prognostic marker. As the presence of T790M is an important factor in choosing treatment and determining prognosis, assessing which population will develop T790M is vital. This should be considered when selecting EGFR-TKIs, and patients must be screened for acquired EGFR T790M mutations at the time of tumor progression.
The occurrence of T790M mutation has important implications for further treatment and prognosis after first- or second-generation EGFR-TKI therapy. The PFS after afatinib treatment was similar to or slightly higher than that after gefitinib and erlotinib treatments. However, the T790M mutation rate of afatinib was significantly lower than those of gefitinib and erlotinib. Patients with acquired T790M mutations during EGFR-TKI treatment showed better PFS and OS with osimertinib treatment. Accordingly, we suggest gefitinib and erlotinib as the first-line treatments for patients with advanced NSCLC. However, in precision medicine, selecting patients with a high probability of receiving first-generation EGFR-TKIs is a better strategy. Ouyang et al. showed that a lower body mass index (≤ 25 kg/m2), higher levels of neuron-specific enolase (> 17.9 ng/ml), and retroperitoneal lymph node metastasis before treatment are independent risk factors for the acquired T790M mutation (58). Lin et al. revealed that the independent factors for T790M mutation were first-generation EGFR-TKIs, initial liver metastasis, male sex, and uncommon EGFR mutations (5).
This meta-analysis had several limitations. First, the literature cited in this meta-analysis was retrospective but not a randomized control trial. The clinical conditions of the subjects who received the three EGFR-TKIs were unequal. Second, the study was not designed to determine the incidence of T790M mutations. Therefore, the timing of biopsy, sample collection (tissues or blood), and methods for detecting T790M were not well designed. Our analysis showed a significantly lower detection rate of acquired T790M mutations in plasma biopsy samples than that in tissue biopsy samples. There are many methods for detecting the T790M mutation. Droplet digital PCR (ddPCR) and matrix-assisted laser desorption/ionization-time of flight mass spectrometry (MALDI-TOF MS) are highly sensitive approaches capable of detecting mutations, and studies using ddPCR and MALDI-TOF MS have shown a higher incidence of detecting T790M (40, 47). The duration of exposure to EGFR-TKIs is an independent factor for the occurrence of T790M mutations. However, the time required for rebiopsy has not been standardized. Third, we included all the available first- and second-generation EGFR-TKIs. However, studies on acquired T790M in patients treated with icotinib or dacomitinib are limited. Therefore, we did not discuss the effects of icotinib or dacomitinib. Further meta-analysis should be performed with more studies on icotinib and dacomitinib.
Lung cancer is associated with significant mortality rates worldwide. Lung adenocarcinoma is the most common type of NSCLC. EGFR mutations occur frequently in adenocarcinoma, and oral EGFR-TKIs with good responses are promising treatments for patients with advanced NSCLC. T790M mutation is the most common mechanism of acquired resistance. Our meta-analysis of 29 studies showed that erlotinib and gefitinib had a higher OR for the T790M mutation than afatinib. The acquired T790M mutation rate was significantly lower with afatinib treatment than that with gefitinib or erlotinib in the overall population. The T790M mutation rate is of great significance because osimertinib treatment in patients with the T790M mutation shows a good prognosis.
The original contributions of this study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
The authors confirm their contribution to the paper as follows: study conception and design: Y-KW and C-CL. Data collection: P-CH and Y-KW. Statistical analysis: P-CH, C-YK, and I-ST. Interpretation of results: Y-KW, C-YH, M-CY, and C-CL. Drafting manuscript: P-CH and C-CL. And project administration: C-CL. All authors have reviewed the results and approved the final version of the manuscript.
This study was supported by grants from Taipei Tzu Chi Hospital and Buddhist Tzu Chi Medical Foundation [TCRD-TPE-109-24(3/3) and TCRD-TPE-111-11].
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2022.869390/full#supplementary-material
1. Chang YS TS, Chen YC, Liu TY, Lee YT, Yen JC, Fang HY, et al. Mutation Profile of Non-Small Cell Lung Cancer Revealed by Next Generation Sequencing. Respir Res (2021) 22(1):3. doi: 10.1186/s12931-020-01608-5
2. Lui NS, Benson J, He H, Imielski BR, Kunder CA, Liou DZ, et al. Sub-Solid Lung Adenocarcinoma in Asian Versus Caucasian Patients: Different Biology But Similar Outcomes. J Thorac Dis (2020) 12(5):2161–71. doi: 10.21037/jtd.2020.04.37
3. You JHS, Cho WCS, Ming WK, Li YC, Kwan CK, Au KH, et al. Egfr Mutation-Guided Use of Afatinib, Erlotinib and Gefitinib for Advanced Non-Small-Cell Lung Cancer in Hong Kong - a Cost-Effectiveness Analysis. PLoS One (2021) 16(3):e0247860. doi: 10.1371/journal.pone.0247860
4. Solassol I, Pinguet F, Quantin X. Fda- and Ema-Approved Tyrosine Kinase Inhibitors in Advanced Egfr-Mutated Non-Small Cell Lung Cancer: Safety, Tolerability, Plasma Concentration Monitoring, and Management. Biomolecules (2019) 9(11)1–19. doi: 10.3390/biom9110668
5. Lin YT, Chen JS, Liao WY, Ho CC, Hsu CL, Yang CY, et al. Clinical Outcomes and Secondary Epidermal Growth Factor Receptor (Egfr) T790m Mutation Among First-Line Gefitinib, Erlotinib and Afatinib-Treated Non-Small Cell Lung Cancer Patients With Activating Egfr Mutations. Int J Cancer (2019) 144(11):2887–96. doi: 10.1002/ijc.32025
6. de Marinis F LK, Poltoratskiy A, Egorova I, Hochmair M, Passaro A, Migliorino MR, et al. Afatinib in Egfr Tki-Naïve Patients With Locally Advanced or Metastatic Egfr Mutation-Positive Non-Small Cell Lung Cancer: Interim Analysis of a Phase 3b Study. Lung Cancer (Amsterdam Netherlands) (2021) 152:127–34. doi: 10.1016/j.lungcan.2020.12.011
7. Khwaja RM, Chu QS. Present and Emerging Biomarkers in Immunotherapy for Metastatic Non-Small Cell Lung Cancer: A Review. Curr Oncol (Toronto Ont) (2022) 29(2):479–89. doi: 10.3390/curroncol29020043
8. Herath S, Sadeghi Rad H, Radfar P, Ladwa R, Warkiani M, O'Byrne K, et al. The Role of Circulating Biomarkers in Lung Cancer. Front Oncol (2021) 11:801269. doi: 10.3389/fonc.2021.801269
9. Xu CW, Lei L, Wang WX, Lin L, Zhu YC, Wang H, et al. Molecular Characteristics and Clinical Outcomes of Egfr Exon 19 C-Helix Deletion in Non-Small Cell Lung Cancer and Response to Egfr Tkis. Trans Oncol (2020) 13(9):100791. doi: 10.1016/j.tranon.2020.100791
10. Jiang H, Zhu M, Li Y, Li Q. Association Between Egfr Exon 19 or Exon 21 Mutations and Survival Rates After First-Line Egfr-Tki Treatment in Patients With Non-Small Cell Lung Cancer. Mol Clin Oncol (2019) 11(3):301–8. doi: 10.3892/mco.2019.1881
11. Wagener-Ryczek S, Heydt C, Süptitz J, Michels S, Falk M, Alidousty C, et al. Mutational Spectrum of Acquired Resistance to Reversible Versus Irreversible Egfr Tyrosine Kinase Inhibitors. BMC Cancer (2020) 20(1):408. doi: 10.1186/s12885-020-06920-3
12. El Kadi N, Wang L, Davis A, Korkaya H, Cooke A, Vadnala V, et al. The Egfr T790m Mutation Is Acquired Through Aicda-Mediated Deamination of 5- Methylcytosine Following Tki Treatment in Lung Cancer. Cancer Res (2018) 78(24):6728–35. doi: 10.1158/0008-5472.Can-17-3370
13. Mok TS, Wu YL, Ahn MJ, Garassino MC, Kim HR, Ramalingam SS, et al. Osimertinib or Platinum-Pemetrexed in Egfr T790m-Positive Lung Cancer. New Engl J Med (2017) 376(7):629–40. doi: 10.1056/NEJMoa1612674
14. Hayashi H, Nadal E, Gray JE, Ardizzoni A, Caria N, Puri T, et al. Overall Treatment Strategy for Patients With Metastatic Nsclc With Activating Egfr Mutations. Clin Lung Cancer (2022) 23(1):e69–82. doi: 10.1016/j.cllc.2021.10.009
15. Chiang CL, Huang HC, Shen CI, Luo YH, Chen YM, Chiu CH. Post-Progression Survival in Secondary Egfr T790m-Mutated Non-Small-Cell Lung Cancer Patients With and Without Osimertinib After Failure of a Previous Egfr Tki. Targeted Oncol (2020) 15(4):503–12. doi: 10.1007/s11523-020-00737-7
16. Hochmair MJ, Morabito A, Hao D, Yang CT, Soo RA, Yang JC, et al. Sequential Afatinib and Osimertinib in Patients With Egfr Mutation-Positive Non-Small-Cell Lung Cancer: Updated Analysis of the Observational Giotag Study. Future Oncol (London England) (2019) 15(25):2905–14. doi: 10.2217/fon-2019-0346
17. Yoshida T, Kuroda H, Oya Y, Shimizu J, Horio Y, Sakao Y, et al. Clinical Outcomes of Platinum-Based Chemotherapy According to T790m Mutation Status in Egfr-Positive Non-Small Cell Lung Cancer Patients After Initial Egfr-Tki Failure. Lung Cancer (Amsterdam Netherlands) (2017) 109:89–91. doi: 10.1016/j.lungcan.2017.05.001
18. Hutton B, Salanti G, Caldwell DM, Chaimani A, Schmid CH, Cameron C, et al. The Prisma Extension Statement for Reporting of Systematic Reviews Incorporating Network Meta-Analyses of Health Care Interventions: Checklist and Explanations. Ann Intern Med (2015) 162(11):777–84. doi: 10.7326/m14-2385
20. König J, Krahn U, Binder H. Visualizing the Flow of Evidence in Network Meta-Analysis and Characterizing Mixed Treatment Comparisons. Stat Med (2013) 32(30):5414–29. doi: 10.1002/sim.6001
21. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. Robins-I: A Tool for Assessing Risk of Bias in Non-Randomised Studies of Interventions. BMJ (2016) 355:i4919. doi: 10.1136/bmj.i4919
22. Sequist LV, Waltman BA, Dias-Santagata D, Digumarthy S, Turke AB, Fidias P, et al. Genotypic and Histological Evolution of Lung Cancers Acquiring Resistance to Egfr Inhibitors. Sci Trans Med (2011) 3(75):75ra26. doi: 10.1126/scitranslmed.3002003
23. Yano S, Yamada T, Takeuchi S, Tachibana K, Minami Y, Yatabe Y, et al. Hepatocyte Growth Factor Expression in Egfr Mutant Lung Cancer With Intrinsic and Acquired Resistance to Tyrosine Kinase Inhibitors in a Japanese Cohort. J Thorac Oncol (2011) 6(12):2011–7. doi: 10.1097/JTO.0b013e31823ab0dd
24. Hata A, Katakami N, Yoshioka H, Takeshita J, Tanaka K, Nanjo S, et al. Rebiopsy of Non-Small Cell Lung Cancer Patients With Acquired Resistance to Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitor: Comparison Between T790m Mutation-Positive and Mutation-Negative Populations. Cancer (2013) 119(24):4325–32. doi: 10.1002/cncr.28364
25. Sun JM, Ahn MJ, Choi YL, Ahn JS, Park K. Clinical Implications of T790m Mutation in Patients With Acquired Resistance to Egfr Tyrosine Kinase Inhibitors. Lung Cancer (Amsterdam Netherlands) (2013) 82(2):294–8. doi: 10.1016/j.lungcan.2013.08.023
26. Li W, Ren S, Li J, Li A, Fan L, Li X, et al. T790m Mutation Is Associated With Better Efficacy of Treatment Beyond Progression With Egfr-Tki in Advanced Nsclc Patients. Lung Cancer (2014) 84(3):295–300. doi: 10.1016/j.lungcan.2014.03.011
27. Jin Y, Shao Y, Shi X, Lou G, Zhang Y, Wu X, et al. Mutational Profiling of Non-Small-Cell Lung Cancer Patients Resistant to First-Generation Egfr Tyrosine Kinase Inhibitors Using Next Generation Sequencing. Oncotarget (2016) 7(38):61755–63. doi: 10.18632/oncotarget.11237
28. Ko R, Kenmotsu H, Serizawa M, Koh Y, Wakuda K, Ono A, et al. Frequency of Egfr T790m Mutation and Multimutational Profiles of Rebiopsy Samples From Non-Small Cell Lung Cancer Developing Acquired Resistance to Egfr Tyrosine Kinase Inhibitors in Japanese Patients. BMC Cancer (2016) 16(1):864. doi: 10.1186/s12885-016-2902-0
29. Matsuo N, Azuma K, Sakai K, Hattori S, Kawahara A, Ishii H, et al. Association of Egfr Exon 19 Deletion and Egfr-Tki Treatment Duration With Frequency of T790m Mutation in Egfr-Mutant Lung Cancer Patients. Sci Rep (2016) 6:36458. doi: 10.1038/srep36458
30. Nosaki K, Satouchi M, Kurata T, Yoshida T, Okamoto I, Katakami N, et al. Re-Biopsy Status Among Non-Small Cell Lung Cancer Patients in Japan: A Retrospective Study. Lung Cancer (Amsterdam Netherlands) (2016) 101:1–8. doi: 10.1016/j.lungcan.2016.07.007
31. Takahama T, Sakai K, Takeda M, Azuma K, Hida T, Hirabayashi M, et al. Detection of the T790m Mutation of Egfr in Plasma of Advanced Non-Small Cell Lung Cancer Patients With Acquired Resistance to Tyrosine Kinase Inhibitors (West Japan Oncology Group 8014ltr Study). Oncotarget (2016) 7(36):58492–9. doi: 10.18632/oncotarget.11303
32. Tseng JS, Su KY, Yang TY, Chen KC, Hsu KH, Chen HY, et al. The Emergence of T790m Mutation in Egfr-Mutant Lung Adenocarcinoma Patients Having a History of Acquired Resistance to Egfr-Tki: Focus on Rebiopsy Timing and Long-Term Existence of T790m. Oncotarget (2016) 7(30):48059–69. doi: 10.18632/oncotarget.10351
33. Lee CK, Kim S, Lee JS, Lee JE, Kim SM, Yang IS, et al. Next-Generation Sequencing Reveals Novel Resistance Mechanisms and Molecular Heterogeneity in Egfr-Mutant Non-Small Cell Lung Cancer With Acquired Resistance to Egfr-Tkis. Lung Cancer (2017) 113:106–14. doi: 10.1016/j.lungcan.2017.09.005
34. Oya Y, Yoshida T, Kuroda H, Shimizu J, Horio Y, Sakao Y, et al. Association Between Egfr T790m Status and Progression Patterns During Initial Egfr-Tki Treatment in Patients Harboring Egfr Mutation. Clin Lung Cancer (2017) 18(6):698–705.e2. doi: 10.1016/j.cllc.2017.05.004
35. Wang W, Song Z, Zhang Y. A Comparison of Ddpcr and Arms for Detecting Egfr T790m Status in Ctdna From Advanced Nsclc Patients With Acquired Egfr-Tki Resistance. Cancer Med (2017) 6(1):154–62. doi: 10.1002/cam4.978
36. Zhang Q, Ke E, Niu F, Deng W, Chen Z, Xu C, et al. The Role of T790m Mutation in Egfr-Tki Re-Challenge for Patients With Egfr-Mutant Advanced Lung Adenocarcinoma. Oncotarget (2017) 8(3):4994–5002. doi: 10.18632/oncotarget.14007
37. Kaburagi T, Kiyoshima M, Nawa T, Ichimura H, Saito T, Hayashihara K, et al. Acquired Egfr T790m Mutation After Relapse Following Egfr-Tki Therapy: A Population-Based Multi-Institutional Study. Anticancer Res (2018) 38(5):3145–50. doi: 10.21873/anticanres.12577
38. Lee K, Kim Y, Jung HA, Lee SH, Ahn JS, Ahn MJ, et al. Repeat Biopsy Procedures and T790m Rates After Afatinib, Gefitinib, or Erlotinib Therapy in Patients With Lung Cancer. Lung Cancer (Amsterdam Netherlands) (2019) 130:87–92. doi: 10.1016/j.lungcan.2019.01.012
39. Yoon BW, Kim JH, Lee SH, Choi CM, Rho JK, Yoon S, et al. Comparison of T790m Acquisition Between Patients Treated With Afatinib and Fefitinib as First-Line Therapy: Retrospective Propensity Score Matching Analysis. Trans Oncol (2019) 12(6):852–8. doi: 10.1016/j.tranon.2019.04.004
40. Del Re M, Petrini I, Mazzoni F, Valleggi S, Gianfilippo G, Pozzessere D, et al. Incidence of T790m in Patients With Nsclc Progressed to Gefitinib, Erlotinib, and Afatinib: A Study on Circulating Cell-Free DNA. Clin Lung Cancer (2020) 21(3):232–7. doi: 10.1016/j.cllc.2019.10.003
41. Dal Maso A, Lorenzi M, Roca E, Pilotto S, Macerelli M, Polo V, et al. Clinical Features and Progression Pattern of Acquired T790m-Positive Compared With T790m-Negative Egfr Mutant Non–Small-Cell Lung Cancer: Catching Tumor and Clinical Heterogeneity Over Time Through Liquid Biopsy. Clin Lung Cancer (2020) 21(1):1–14.e3. doi: 10.1016/j.cllc.2019.07.009
42. Oya Y, Yoshida T, Asada K, Oguri T, Inui N, Morikawa S, et al. Clinical Utility of Liquid Biopsy for Egfr Driver, T790m Mutation and Egfr Amplification in Plasma in Patients With Acquired Resistance to Afatinib. BMC Cancer (2021) 21(1)1–9. doi: 10.1186/s12885-020-07777-2
43. Onitsuka T, Uramoto H, Nose N, Takenoyama M, Hanagiri T, Sugio K, et al. Acquired Resistance to Gefitinib: The Contribution of Mechanisms Other Than the T790m, Met, and Hgf Status. Lung Cancer (Amsterdam Netherlands) (2010) 68(2):198–203. doi: 10.1016/j.lungcan.2009.05.022
44. Uramoto H, Yamada T, Yano S, Kondo N, Hasegawa S, Tanaka F. Prognostic Value of Acquired Resistance-Related Molecules in Japanese Patients With Nsclc Treated With an Egfr-Tki. Anticancer Res (2012) 32(9):3785–90.
45. Ji W, Choi CM, Rho JK, Jang SJ, Park YS, Chun SM, et al. Mechanisms of Acquired Resistance to Egfr-Tyrosine Kinase Inhibitor in Korean Patients With Lung Cancer. BMC Cancer (2013) 13:606. doi: 10.1186/1471-2407-13-606
46. Campo M, Gerber D, Gainor JF, Heist RS, Temel JS, Shaw AT, et al. Acquired Resistance to First-Line Afatinib and the Challenges of Prearranged Progression Biopsies. J Thorac Oncol (2016) 11(11):2022–6. doi: 10.1016/j.jtho.2016.06.032
47. Liang SK, Hsieh MS, Lee MR, Keng LT, Ko JC, Shih JY. Real-World Experience of Afatinib as a First-Line Therapy for Advanced Egfr Mutation-Positive Lung Adenocarcinoma. Oncotarget (2017) 8(52):90430–43. doi: 10.18632/oncotarget.19563
48. Tanaka K, Nosaki K, Otsubo K, Azuma K, Sakata S, Ouchi H, et al. Acquisition of the T790m Resistance Mutation During Afatinib Treatment in Egfr Tyrosine Kinase Inhibitor-Naïve Patients With Non-Small Cell Lung Cancer Harboring Egfr Mutations. Oncotarget (2017) 8(40):68123–30. doi: 10.18632/oncotarget.19243
49. Passiglia F, Rizzo S, Di Maio M, Galvano A, Badalamenti G, Listì A, et al. The Diagnostic Accuracy of Circulating Tumor DNA for the Detection of Egfr-T790m Mutation in Nsclc: A Systematic Review and Meta-Analysis. Sci Rep (2018) 8(1):13379. doi: 10.1038/s41598-018-30780-4
50. Li X, Zhou C. Comparison of Cross-Platform Technologies for Egfr T790m Testing in Patients With Non-Small Cell Lung Cancer. Oncotarget (2017) 8(59):100801–18. doi: 10.18632/oncotarget.19007
51. Li D, Ambrogio L, Shimamura T, Kubo S, Takahashi M, Chirieac LR, et al. Bibw2992, an Irreversible Egfr/Her2 Inhibitor Highly Effective in Preclinical Lung Cancer Models. Oncogene (2008) 27(34):4702–11. doi: 10.1038/onc.2008.109
52. Miller VA, Hirsh V, Cadranel J, Chen YM, Park K, Kim SW, et al. Afatinib Versus Placebo for Patients With Advanced, Metastatic Non-Small-Cell Lung Cancer After Failure of Erlotinib, Gefitinib, or Both, and One or Two Lines of Chemotherapy (Lux-Lung 1): A Phase 2b/3 Randomised Trial. Lancet Oncol (2012) 13(5):528–38. doi: 10.1016/s1470-2045(12)70087-6
53. Chmielecki J, Foo J, Oxnard GR, Hutchinson K, Ohashi K, Somwar R, et al. Optimization of Dosing for Egfr-Mutant Non-Small Cell Lung Cancer With Evolutionary Cancer Modeling. Sci Trans Med (2011) 3(90):90ra59. doi: 10.1126/scitranslmed.3002356
54. Joo JW, Hong MH, Shim HS. Clinical Characteristics of T790m-Positive Lung Adenocarcinoma After Resistance to Epidermal Growth Factor Receptor-Tyrosine Kinase Inhibitors With an Emphasis on Brain Metastasis and Survival. Lung Cancer (Amsterdam Netherlands) (2018) 121:12–7. doi: 10.1016/j.lungcan.2018.04.013
55. Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, et al. Gefitinib or Chemotherapy for Non-Small-Cell Lung Cancer With Mutated Egfr. New Engl J Med (2010) 362(25):2380–8. doi: 10.1056/NEJMoa0909530
56. Park K, Tan EH, O'Byrne K, Zhang L, Boyer M, Mok T, et al. Afatinib Versus Gefitinib as First-Line Treatment of Patients With Egfr Mutation-Positive Non-Small-Cell Lung Cancer (Lux-Lung 7): A Phase 2b, Open-Label, Randomised Controlled Trial. Lancet Oncol (2016) 17(5):577–89. doi: 10.1016/s1470-2045(16)30033-x
57. Paz-Ares L, Tan EH, O'Byrne K, Zhang L, Hirsh V, Boyer M, et al. Afatinib Versus Gefitinib in Patients With Egfr Mutation-Positive Advanced Non-Small-Cell Lung Cancer: Overall Survival Data From the Phase Iib Lux-Lung 7 Trial. Ann Oncol (2017) 28(2):270–7. doi: 10.1093/annonc/mdw611
Keywords: non-small cell lung cancer, adenocarcinoma, epidermal growth factor receptor, tyrosine kinase inhibitors, T790M acquisition
Citation: Hsieh P-C, Wu Y-K, Huang C-Y, Yang M-C, Kuo C-Y, Tzeng I-S and Lan C-C (2022) Comparison of T790M Acquisition After Treatment With First- and Second-Generation Tyrosine-Kinase Inhibitors: A Systematic Review and Network Meta-Analysis. Front. Oncol. 12:869390. doi: 10.3389/fonc.2022.869390
Received: 04 February 2022; Accepted: 30 May 2022;
Published: 28 June 2022.
Edited by:
Iacopo Petrini, University of Pisa, ItalyReviewed by:
Francesco Pepe, University of Naples Federico II, ItalyCopyright © 2022 Hsieh, Wu, Huang, Yang, Kuo, Tzeng and Lan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chou-Chin Lan, Ymx1ZXNjb3B5QHlhaG9vLmNvbS50dw==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.