
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Oncol. , 12 April 2022
Sec. Breast Cancer
Volume 12 - 2022 | https://doi.org/10.3389/fonc.2022.861439
 Jian Wan1,2†
Jian Wan1,2† Shizhen Chen3†
Shizhen Chen3† Anqin Zhang2
Anqin Zhang2 Yiting Liu2
Yiting Liu2 Yangyang Zhang2
Yangyang Zhang2 Qinghua Li2
Qinghua Li2 Ziqi Yu3
Ziqi Yu3 Yuwei Wan3
Yuwei Wan3 Lei Yang3*
Lei Yang3* Qi Wang1,2*
Qi Wang1,2*Background: Adenosine-to-inosine RNA editing (ATIRE) is increasingly being used to characterize cancer. However, no studies have been conducted to identify an ATIRE signature for predicting cancer survival.
Methods: Breast cancer (BRCA) samples with ATIRE profiles from The Cancer Genome Atlas were divided into training (n = 452) and internal validation cohorts (n = 311), and 197 additional BRCA patients were recruited as an external validation cohort. The ATIRE signature for BRCA overall survival (OS) and disease-free survival (DFS) were identified using forest algorithm analysis and experimentally verified by direct sequencing. An ATIRE-based risk score (AIRS) was established with these selected ATIRE sites. Significantly prognostic factors were incorporated to generate a nomogram that was evaluated using Harrell’s C-index and calibration plot for all cohorts.
Results: Seven ATIRE sites were revealed to be associated with both BRCA OS and DFS, of which four sites were experimentally confirmed. Patients with high AIRS displayed a higher risk of death than those with low AIRS in the training (hazard ratio (HR) = 3.142, 95%CI = 1.932–5.111), internal validation (HR = 2.097, 95%CI = 1.123–3.914), and external validation cohorts (HR = 2.680, 95%CI = 1.000–7.194). A similar hazard effect of high AIRS on DFS was also observed. The nomogram yielded Harrell’s C-indexes of 0.816 (95%CI = 0.784–0.847), 0.742 (95%CI = 0.684–0.799), and 0.869 (95%CI = 0.835–0.902) for predicting OS and 0.767 (95%CI = 0.708–0.826), 0.684 (95%CI = 0.605–0.763), and 0.635 (95%CI = 0.566–0.705) for predicting DFS in the three cohorts.
Conclusion: AIRS nomogram could help to predict OS and DFS of patients with BRCA.
Female breast cancer (BRCA) has now been the most commonly diagnosed cancer with an estimated 2.3 million new cases in 2020 worldwide (1). Over the past decades, the long-term survival rate of female BRCA has dramatically increased due to advances in early diagnosis and prompt treatment. The data from Surveillance, Epidemiology, and End Results (SEER) shows the 5-year survival rate of female BRCA approached 90.3% in the United States (https://seer.cancer.gov/statfacts/html/breast.html), while in China, the number was about 82.0% (2). However, the high incidence of female BRCA still caused an estimated 684,996 deaths in 2020 (1). Besides, improved treatments such as adjuvant systemic therapy result in inappropriate treatment in many patients, including both unnecessary treatment and insufficient curing. Therefore, it is of great value to develop a reliable prognostic model to identify patients at high risk of death or disease recurrence, which may suggest changes in life health management.
Accompanied by the revelation of transcriptomics in BRCA, various prognostic models have been developed to help physicians assess the survival of BRCA patients (3–10). These prediction models were mostly established by gene expression data recorded by The Cancer Genome Atlas (TCGA) database (https://portal.gdc.cancer.gov/) or Gene Expression Omnibus (GEO) database (https://www.ncbi.nlm.nih.gov/geo/). Nevertheless, all these models have limitations in clinical practice in terms of their veracity, reliability, and reproducibility, which are always at the mercy of mobility and detection instability in gene expression. A novel type of molecular markers may contribute to overcoming these challenges. RNA editing is a molecular process through which some cells can make discrete changes to specific nucleotide sequences within an RNA molecule (11). Compared to gene expression, RNA editing is more tumor-specific (12, 13) and irrespective of inter-individual variability at the amount of isolated RNA and reference gene selection. Therefore, as a biomarker, RNA editing is superior to RNA expression in terms of test stability and reliability.
More than 70% of RNA editing in humans is adenosine-to-inosine RNA editing (ATIRE) (14), which results in adenosine-to-inosine transitions at particular sites of pre-mRNA. In the process of post-transcription and translation, inosine is recognized as guanosine (G). Recent bioinformatics analyses have revealed transcriptome-wide ATIRE profiling and identified survival-related ATIRE sites in cancer by analyzing TCGA sequencing data (15, 16). However, no studies have validated both the reality and prognostic correlation of these sites in cancer. In this study, we aimed to develop and validate an ATIRE-based risk score (AIRS) for assessing the probability of overall survival (OS) and disease-free survival (DFS) in patients affected by BRCA, and for the first time, we constructed a novel prognostic model using ATIRE with considerable accuracy on predicting BRCA OS and DFS.
ATIRE profiling of TCGA-BRCA patients in cancer tissues was downloaded from the synapse (https://www.synapse.org/#!Synapse:syn4382382) (15). The corresponding clinicopathological parameters of indicated patients were downloaded from the cBioPortal (https://www.cbioportal.org/). Patients were excluded if they lacked data about ATIRE level, OS, and TNM stages. The remaining patients were randomly allocated to a training cohort (n = 452) and an internal validation cohort (n = 311). Besides, an external validation cohort (n = 197) was randomly recruited in Guangdong Women and Children’s Hospital and Health Institute from January 2014 to September 2019 in Guangzhou city. All cases were histopathologically confirmed and followed up by phone by one of us (JW). After a median follow-up of 68 months, 89.3% of patients (176/197) were followed up successfully and provided complete OS information. Meanwhile, 83.8% of patients provided DFS data. OS was defined as the length of time from the date of diagnosis for BRCA that patients diagnosed with the disease are still alive. DFS was defined as the time after surgery to relapse, second cancer, or all-cause death, whichever came first. There were no significant differences in frequency distributions of clinicopathological characteristics between the cases lost to follow-up and those to be analyzed (Supplementary Table S1). All individuals have signed written informed consent. The study was approved by the Institutional Review Board of Guangzhou medical university.
Figure 1 presents the flowchart of AIRS model construction for BRCA survival. After elimination of sites with low authenticity whose editing levels were not reported in more than half of the samples, 30,001 ATIRE sites were included in the univariate Cox proportional hazards (Cox-PH) model in the training cohort. Then the sites with p < 0.001 were submitted to the forest algorithm analysis, a non-parametric machine learning strategy that was recently used for building a risk prediction model in survival analysis (17). Due to the limitation of forest algorithm analysis, these sites with undetermined editing levels in more than 5% of samples and samples that had undetermined editing levels at any of the inclusive sites were removed. After the median split of samples into high and low editing groups according to the editing level, the common sites for OS and DFS with relative importance > 0.1 were preselected for the construction of the AIRS model.
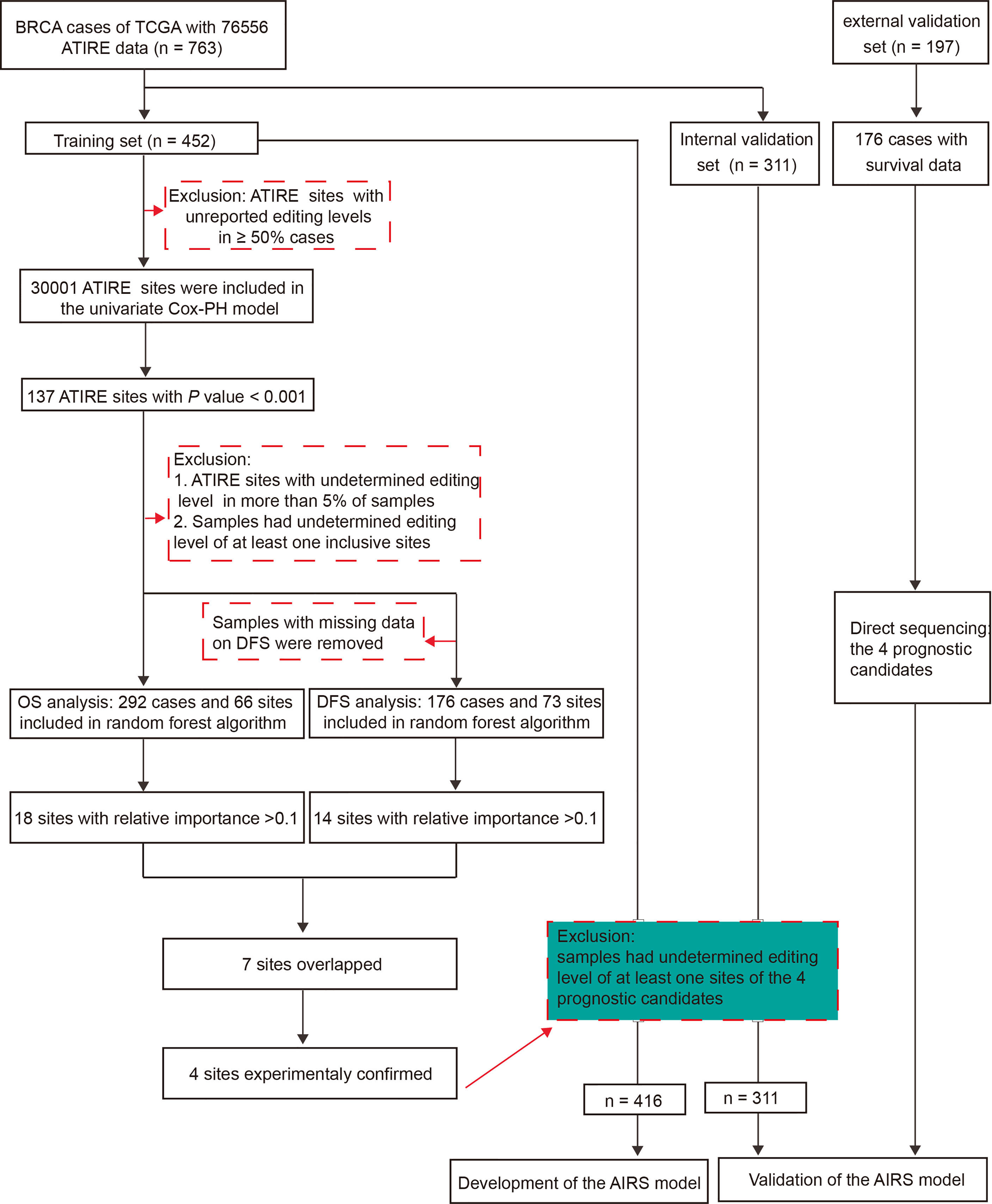
Figure 1 The workflow of generation of an AIRS model for BRCA OS and DFS. AIRS, adenosine-to-inosine RNA editing risk score; BRCA, breast cancer; OS, overall survival; DFS, disease-free survival.
To verify the reality of these preselected ATIRE sites, complementary DNA (cDNA) and genomic DNA (gDNA) were isolated from five randomly selected BRCA tissues and were used as templates for PCR. The PCR products were purified and then directly sequenced. The primers used are presented in Supplementary Table S2.
Empirically supported ATIRE sites were finally selected for AIRS model construction. To maximize the efficiency and robustness of the model, the editing level of each ATIRE was classified into low, medium, and high through the optimal grouping method using the X-Tile Software (https://medicine.yale.edu/lab/rimm/research/software/) (18). Cox-PH was used to measure the hazard ratio (HR) and 95% CI for the association between each ATIRE and BRCA survival. Then, an AIRS model was established by summing the effect of each site by its respective weight (lnHR).
Associations of the AIRS model with BRCA OS and DFS were validated in the internal and external validation cohorts. The PCR followed by sequencing was performed to determine the editing level of each selected ATIRE site in the external validation cohort. The editing level was calculated by the ratio between the height of peak corresponding to G and the sum of the height of peaks corresponding to G and A.
A nomogram incorporating AIRS and clinicopathological features including age at diagnosis, TNM stages, estrogen receptor (ER), and progesterone receptor (PR) status was constructed by means of the “rms” package in R (version 4.0.1). To validate the nomogram, the total point of each patient in the two validation cohorts was calculated according to the established nomogram. The predictive performance of the nomogram was measured by Harrell’s C-index and calibration with 100 bootstrap samples (19).
Since the four AIRS sites are all located at the 3′-untranslated region (3′-UTR) of host genes, correlations between the editing level of each site and expression of host gene were analyzed by integrating TCGA-BRCA RNA-seq data with ATIRE profiling.
To determine the effect of AIRS sites on whole transcriptome expression, RNA-seq data were downloaded from TCGA. The difference in an expressional file between low and medium/high AIRS patients was assessed using the “limma” package in R software (version 4.0.1). Differences with adjusted p-value <0.05 and |log2(Fold change)| ≥ 1.5 were considered to be significant. Then, Gene Set Enrichment Analysis (GSEA) was performed to determine the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways (20).
In addition to the aforementioned statistical analysis, correlations between the editing level of ATIRE sites and the expression of host genes were tested by Spearman’s correlation test. All tests were two-sided and evaluated by GraphPad Prism 9.0. p < 0.05 was considered to be statistically significant.
The clinicopathological characteristics of all patients to be analyzed were presented in Table 1. There were no obvious differences in distributions of clinicopathological characteristics between the training and internal validation cohorts, but more cases in the external cohort were young and diagnosed at early stages with negative PR or positive HER2.
The Manhattan plot (Figure 2A) shows 137 ATIRE sites with p < 0.001 in the Cox-PH model for testing associations between 30,001 ATIRE sites and BRCA OS in the training cohort. Then the forest algorithm analysis featured 18 (Figure 2B) and 14 (Figure 2C) sites as foremost predictive factors for BRCA OS and DFS, of which seven sites, that are, ARSD A2874>I, ZNF791 A2280>I, H6PD A8760>I, MED18 A1552>I, MEGF8 A9749>I, SSU27 A1727>I, and RAD1 A1415>I, were overlapped. The number residing in A>I indicates the distance between the transcription start site and the ATIRE site.
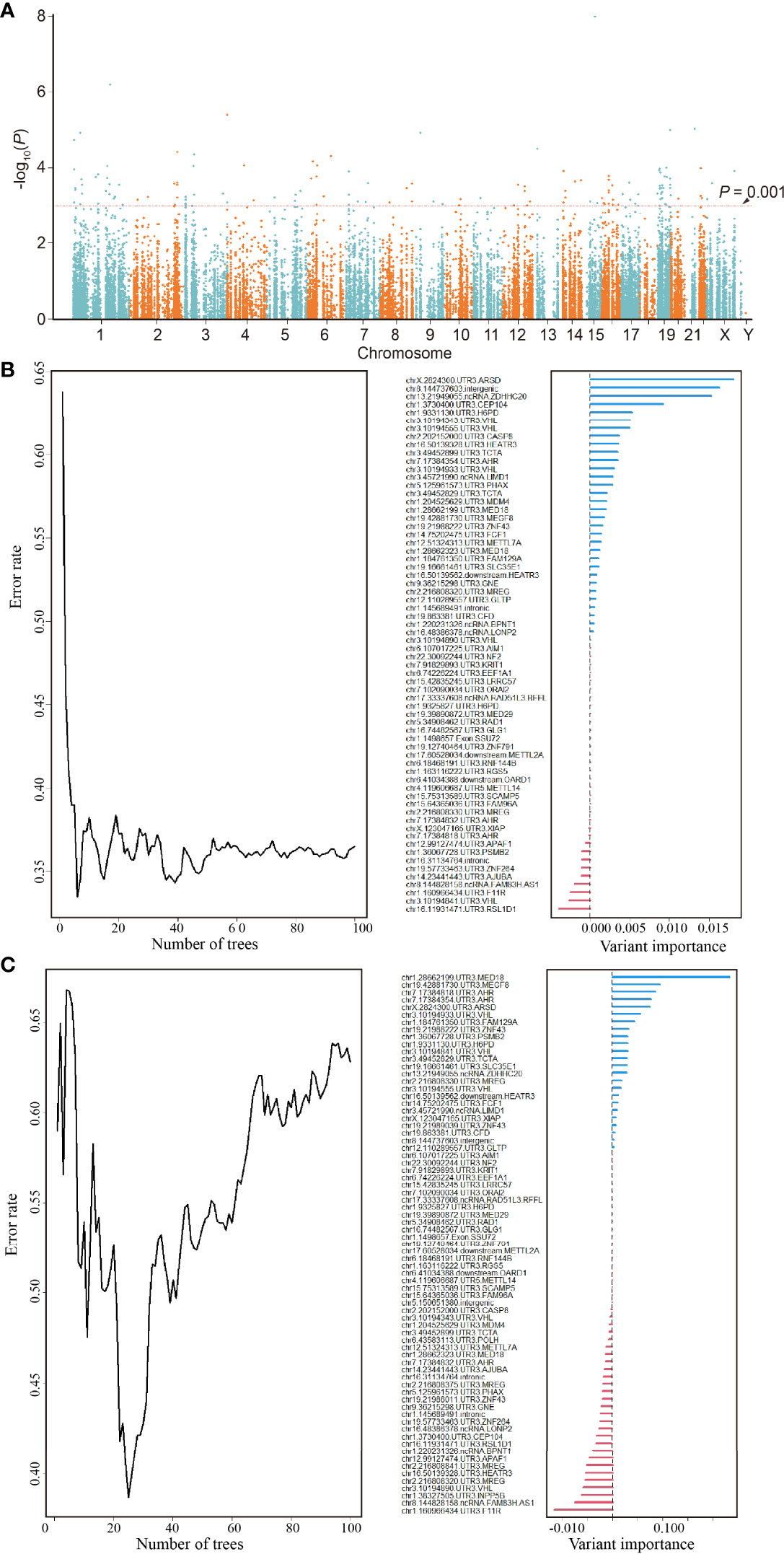
Figure 2 Identification of an ATIRE prognostic signature for OS and DFS of BRCA patients. (A) Scatter plot of p-values in −log10 scale from the univariate Cox-PH model on associations between all ATIRE sites and OS of TCGA-BRCA patients. y-Axis refers to p-values in –log10 scale, and x-axis refers to chromosomal location of these ATIRE sites. (B) Random forest modeling (left) and analysis of variable importance of ATIRE sites (right) to OS of BRCA. Left: survival forest of 100 trees was created, using the log-rank splitting rule with ATIRE predictors randomly selected at each split. Right: survival random forest analysis furnishes a ranking of ATIRE predictors’ importance in determining the accuracy of prediction. (C) Random forest modeling (left) and analysis of variable importance of ATIRE sites (right) to DFS of BRCA. Left: survival forest of 100 trees was created, using the log-rank splitting rule with ATIRE predictors randomly selected at each split. Right: survival random forest analysis furnishes a ranking of ATIRE predictors’ importance in determining the accuracy of prediction. ATIRE, adenosine-to-inosine RNA editing; OS, overall survival; DFS, disease-free survival; BRCA, breast cancer; TCGA, The Cancer Genome Atlas.
Direct sequencing (Figure 3A) shows ARSD A2874>I, ZNF791 A2280>I, MED18 A1552>I, and RAD1 A1415>I with distinct G peak in the cDNA but not in the corresponding gDNA, suggesting that these sites were edited. However, no edit of MEGF8 A9749>I and SSU27 A1727>I were observed. Besides, the trace peaks for H6PD were misshapen, causing a failed determination of H6PD A8760>I. Thus, the four experimentally confirmed ATIRE sites were selected to construct the AIRS model for BRCA OS and DFS.
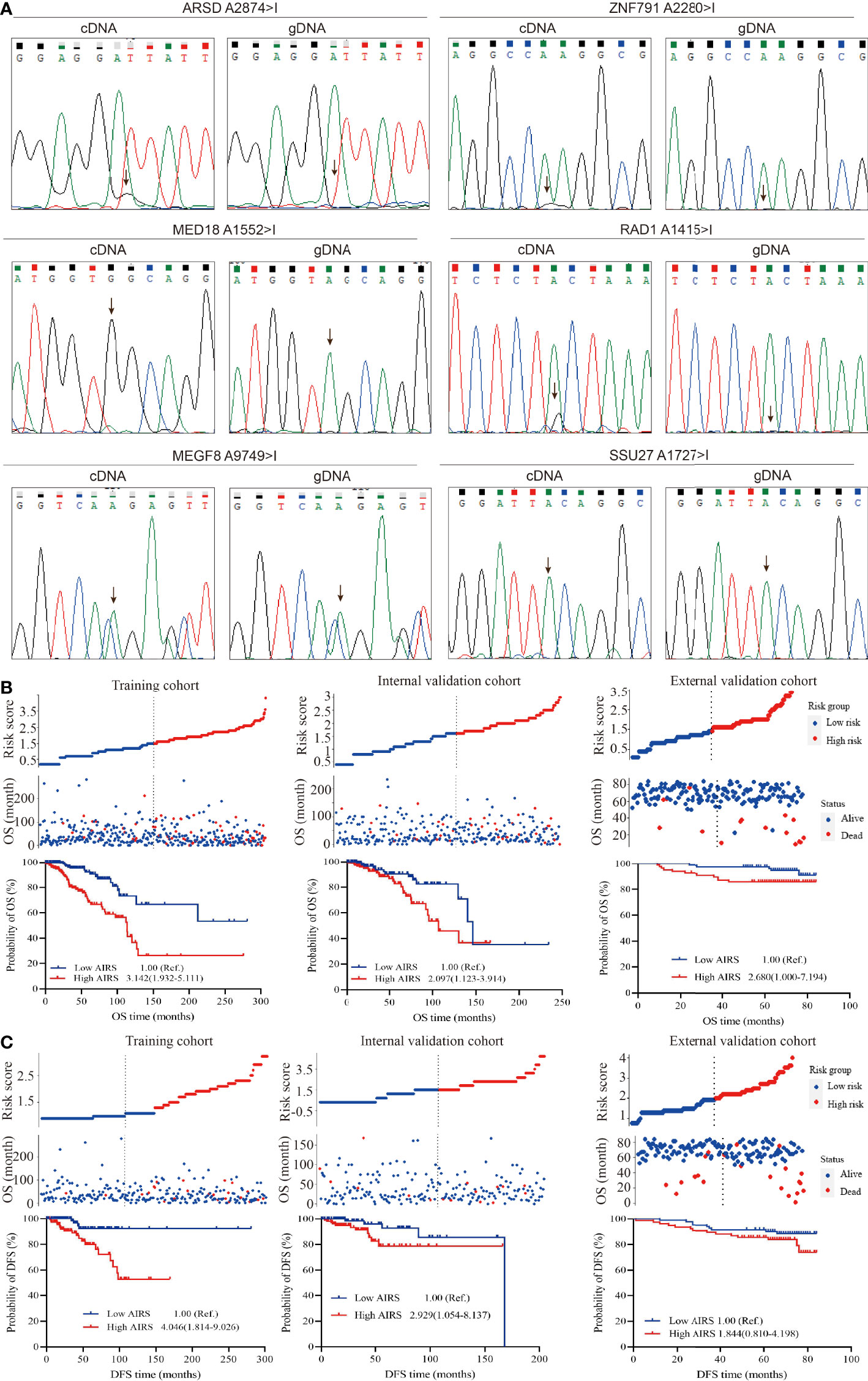
Figure 3 Correlation between AIRS, OS, and DFS of BRCA patients. (A) Validation of the selected ATIRE sites by direct sequencing. cDNA, complementary DNA; gDNA, genomic DNA. (B) Distribution of the risk score (top), survival status (middle), and Kaplan–Meier plot (bottom) to visualize the OS probabilities for AIRS in the training cohort (left), internal validation cohort (middle), and external validation cohort (right). (C) Distribution of the risk score (top), survival status (middle), and Kaplan–Meier plot (bottom) to visualize the DFS probabilities for AIRS in the training cohort (left), internal validation cohort (middle), and external validation cohort (right). Hazard ratio (HR) values were calculated by the Cox regression model, as shown in the Kaplan–Meier plot. AIRS, adenosine-to-inosine RNA editing-based risk score; OS, overall survival; DFS, disease-free survival; BRCA, breast cancer; ATIRE, adenosine-to-inosine RNA editing.
After grouping the editing level of aforesaid screened ATIRE sites into low, medium, and high using the X-Tile Software, the HRs (Supplementary Table S3) for associations between these sites and BRCA OS were used to calculate AIRS. We generated a simple applet in Excel to easily calculate AIRS (Supplementary File 1). As shown in Figure 3B, after grouping AIRS by the median, we found that high AIRS trebled (HR = 3.142, 95%CI = 1.932–5.111) and doubled (HR = 2.097, 95%CI = 1.123–3.914) a BRCA patient’s risk of death in the training and internal validation cohorts. Being consistent, after removing 3 cases with undetermined editing levels in any of the AIRS sites, the high AIRS group displayed poorer OS than the low AIRS group (HR = 2.680, 95%CI = 1.000–7.194).
Similarly, the HRs for associations (Supplementary Table S4) between the aforesaid four sites and BRCA DFS were used to calculate AIRS. We also generated a simple applet in Excel to easily calculate AIRS for DFS (Supplementary File 1). As shown in Figure 3C, compared to the patients with low AIRS, those with high AIRS had a 4.046-fold (HR = 4.046, 95%CI = 1.814–9.026) and 2.929-fold (HR = 2.929, 95%CI = 1.054–8.137) risk of progression, recurrence, or death in the training and internal validation cohorts. In the external cohort, the high AIRS was associated with an increased risk (HR = 1.844, 95%CI = 0.810–4.198), but the association did not reach statistical significance, which may be due to the limited sample size. Taken together, these data suggest that AIRS is a prognostic indicator of BRCA OS and DFS.
The multivariate Cox-PH analysis showed that AIRS, ER/PR status, T stage, N stage, and M stage were independent predictors of mortality in TCGA patients affected by BRCA (Figure 4A). Thus, these factors were incorporated to develop a nomogram for predicting BRCA OS (Figure 4B). As shown in Figure 4C, the nomogram-predicted survival rate displayed a superior agreement with the observed survival rate in all the three cohorts, with Harrell’s C-indexes as 0.816 (95%CI = 0.784–0.847), 0.742 (95%CI = 0.684–0.799), and 0.869 (95%CI = 0.835–0.902).
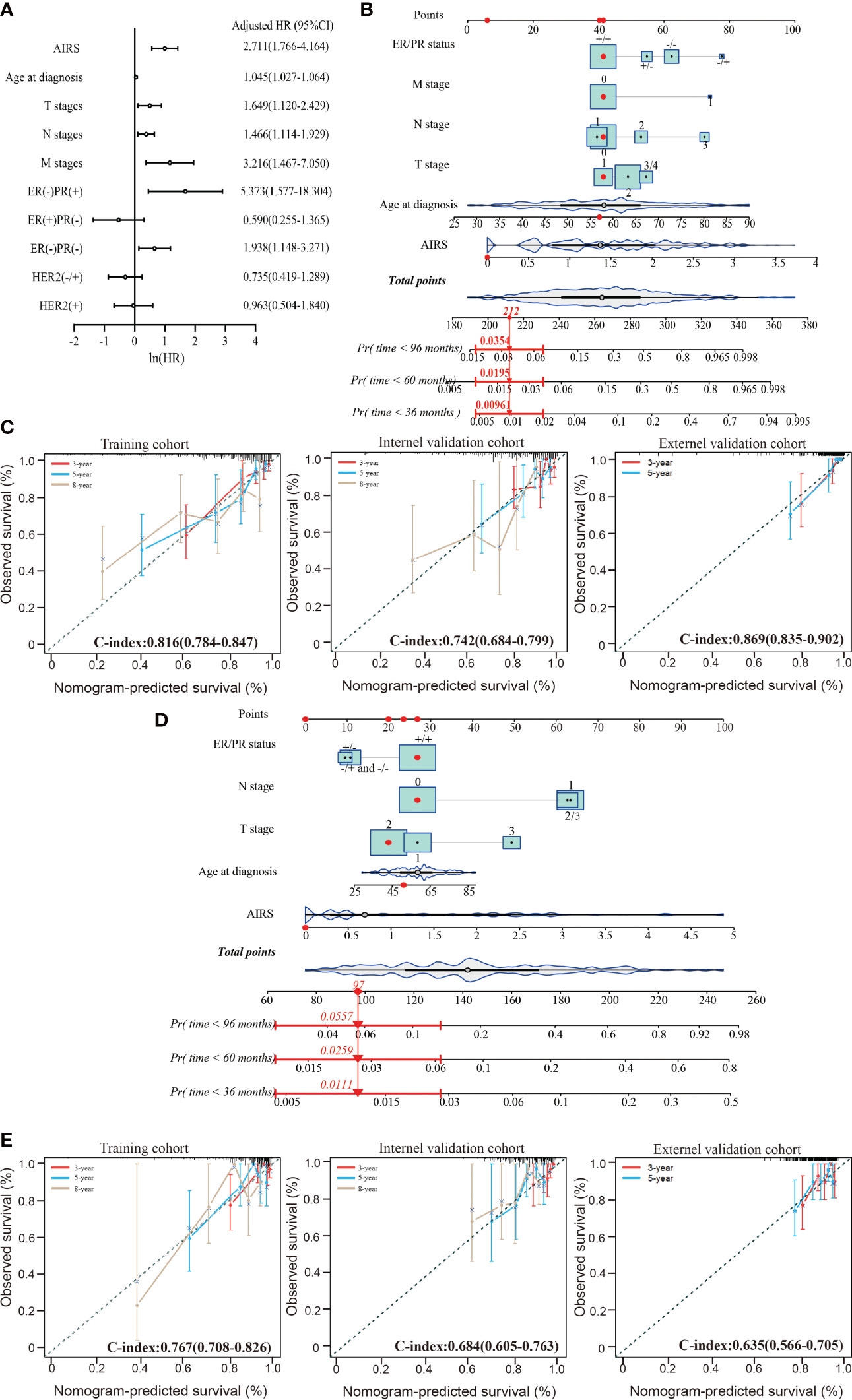
Figure 4 Performance of nomogram based on the AIRS and clinicopathological features. (A) Forest plot to visualize the HRs and 95%CIs for associations between the AIRS, clinicopathological features, and OS, after the multivariate Cox-PH model. (B) AIRS nomogram predicting 36-, 60-, and 96-month OS probability of BRCA patients. Each clinicopathological variable or AIRS has a certain number of the score (top row) ranging from 0 to 100. The sum of points of each variable was related to the probability of OS at specific time points (36, 60, and 96 months). The red dots and straight line with arrow show an example illustrating the use of the nomogram. This patient was one of TCGA-BRCA patients. The patient was ER+/PR+ (point = 41), no lymphatic metastasis (point = 41), no distant metastasis (point = 41), T1-stage (point = 41), 57-year-old (point = 42), and 0 ATRS (point = 6); thus, the total points of this patient is 212, which corresponds to a 99.9% probability of 36-month OS, a 98.1% probability of 60-month OS, and a 96.5% probability of 96-month OS. (C) The calibration curves for predicting patients’ OS at 3-year (red line), 5-year (violet-red line), and 8-year (tan line) in the training cohort (left), internal validation cohort (middle), and external validation cohort (right). (D) AIRS nomogram predicting 36-, 60-, and 96-month DFS probability of BRCA patient and an example illustrating the use of the nomogram. (E) The calibration curves for predicting patients’ DFS at 3-year (red line), 5-year (violet-red line), and 8-year (tan line) in the training cohort (left), internal validation cohort (middle), and external validation cohort (right). ER, estrogen receptor; PR, progesterone receptor; AIRS, adenosine-to-inosine RNA editing-based risk score; HRs, hazard ratios; OS, overall survival; TCGA, The Cancer Genome Atlas; BRCA, breast cancer.
The aforementioned prognostic factors were also used to develop a nomogram for predicting BRCA DFS based on the training cohort (Figure 4D). In this nomogram, the M stage was removed, because there was only one case with distant metastasis. As shown in Figure 4E, the nomogram-predicted survival rate displayed a noteworthy agreement with the observed survival rate in the training cohort and validation cohorts, with Harrell’s C-indexes as 0.767 (95%CI = 0.708–0.826), 0.684 (95%CI = 0.605–0.763), and 0.635 (95%CI = 0.566–0.705). These data suggest that AIRS-based nomograms could be used to predict BRCA OS and DFS.
Integrating TCGA-BRCA RNA-seq datasets with ATIRE profiling showed that a high editing level of A2280>I was significantly associated with a decreased level of ZNF791 when compared to a low or medium editing level (p = 0.002). Meanwhile, there was an inverse dose–effect relationship between the editing level of A1552>I and MED18 expression (p = 0.020). The editing level of A2874>I exerted a correlation with ARSD expression with a trend towards statistical significance (p = 0.067). However, no such effect was observed for A1415>I editing and RAD1 expression (Figures 5A–D). Moreover, a comparison of transcriptomic data between the high AIRS and low AIRS groups of TCGA-BRCA samples demonstrated that AIRS was correlated with expressions of 20 genes, of which 16 were lower and 4 were higher in the high AIRS group (Figure 5E; Supplementary File 2). KEGG pathways enrichment analysis showed that high AIRS actives several pathways such as oxidative phosphorylation and proteasome and suppresses pathways such as the calcium signaling pathway (Figure 5F). Consistently, GSEA with the 50 hallmarks found that signaling pathways of target genes are involved in oxidative phosphorylation, proteasome, and calcium signaling pathway (Figure 5G).
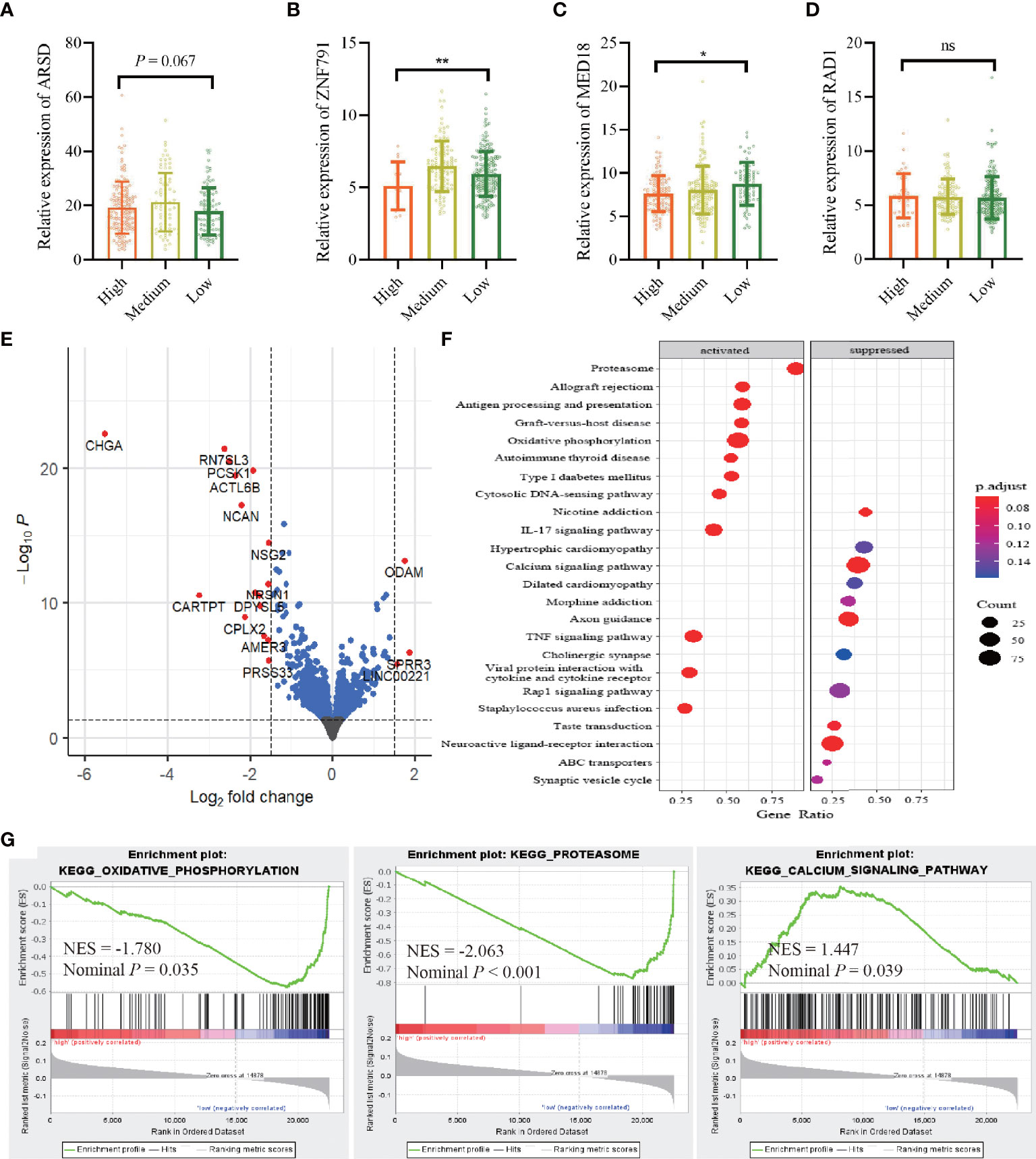
Figure 5 Correlation between the AIRS sites and gene expressions. (A–D) Correlations between editing levels of the four AIRS sites and expression levels of host genes. Data are presented as mean ± SD, and p-values are calculated by the one-way ANOVA test. *p < 0.05,**p < 0.01, ns, not significant. (E) Visualization of differentially expressed genes with volcano plot in high AIRS patients versus low AIRS patients. The expression difference of a log2 fold change of 1.5 (outer light gray broken vertical line) and for an adjusted p-value of 0.05 (dark broken horizontal line). y-Axis refers to p-values in–log10 scale, and x-axis refers to the fold change in log 2 scale. (F) Plot of the KEGG pathway enrichment analysis for AIRS-related genes based on TCGA data. y-Axis represents pathways; x-axis represents the amount of AIRS-related genes enriched in KEGG pathways. The color and size of each bubble represent enrichment significance and the number of AIRS-related genes enriched in KEGG, respectively. (G) GSEA enrichment plot of KEGG pathway genes in high AIRS versus low AIRS. KEGG, Kyoto Encyclopedia of Genes and Genomes; GSEA, Gene Set Enrichment Analysis; NES, normalized enrichment score; AIRS, adenosine-to-inosine RNA editing-based risk score; TCGA, The Cancer Genome Atlas.
Although an overwhelming majority of studies have established many prognostic prediction models for BRCA via analyzing the transcriptomics data, most of these studies around this topic redundantly disaggregated transcriptomics data into gene sets by functional annotation to create new nomograms such as N6-methyladenosine regulator-related or autophagy gene-related nomogram (21, 22). This excessive mining of transcriptomics actually contributes little to improve predictive precision of cancer prognosis, even if we combined these models considering the collinearity of gene expression. Besides, a lot of similar nomograms confuse the selection of such models for application. To overcome these problems, new developments in discovering novel biomarkers are needed. In this study, we for the first time constructed an AIRS model that exerts high associations with both OS and DFS of BRCA. We then developed a nomogram incorporating the AIRS and clinicopathological features, which performed precisely in predicting the survival of patients affected by BRCA.
Emerging evidence has demonstrated that dysregulated ATIRE was implicated in cancer development by causing amino acid changes, mRNA abundance and splicing anomaly, and re-direction of mRNA–microRNA binding (23, 24). The bioinformatics analyses on high-throughput RNA-seq data have widely revealed ATIRE sites and reported cancer survival-related ATIRE profiles, although these sites have not been actually verified (15, 16). Indeed, in this study, only four of the seven preselected sites were successfully verified by direct sequencing, demonstrating that identification of ATIRE sites via bioinformatics had some disadvantages of the uncertainties, with quite a deviation. Furthermore, multiple experiment studies targeting ATIRE in indicated genes or microRNAs have revealed several functional ATIRE sites to be associated with various cancer survival (25–32). These findings make it possible to use ATIRE sites as a potential tool in the assessment of cancer prognosis.
By mining TCGA-BRCA ATIRE profiling with a machine learning strategy, forest algorithm analysis, and by proving experimentation with direct sequencing, we for the first time identified the four ATIRE-relevant prognostic signatures and established the AIRS model for both OS and DFS of BRCA. It was found that the AIRS was associated with OS and DFS of BRCA in the internal and external validation cohorts, which agrees with the result from the training cohort. This indicates the validity of the model. The AIRS model was applied together with age at diagnosis, TNM stages, and ER and PR status to build a nomogram for predicting OS and DFS probability of patients affected by BRCA. Overall, our nomogram presents a good performance, robustness, and stability on predicting BRCA OS with Harrell’s C-indexes as 0.816, 0.742, and 0.869 in the three cohorts. The nomogram also exerts a considerable accuracy on predicting BRCA DFS with the C-indexes as 0.767, 0.684, and 0.635 in the three cohorts. The predictive efficiencies were also confirmed by the calibration plots. Thus, the nomogram may help the clinics for better health administration of patients with BRCA.
Since the four AIRS sites are all located in the 3′-UTR of host genes, it is plausible to observe the correlations between A2280>I editing level and ZNF791 expression, A1552>I editing level, and MED18 expression. However, the biological functions of ZNF791 and MED18 in BRCA are largely unknown, making it impossible to infer the plausibility of associations between the two sites and BRCA survival. Moreover, the cellular pathway clearly demonstrated that AIRS was associated with several signaling pathways, such as oxidative phosphorylation (33), proteasome (34), and calcium signaling pathway (35). These pathways are all promising targets for the treatment of BRCA, which suggests the AIRS as new targets for BRCA treatment.
One of the strengths of this study is that it represents a comprehensive examination of the whole transcriptomic ATIRE sites. The other one is that we used both internal and external validation cohorts to verify the AIRS model and nomogram. Nevertheless, there still remain some limitations in our research. First, functional analysis is lacking to support the biological plausibility of the association between the ATIRE sites and BRCA survival. Second, the omission of detailed treatment options in TCGA data prevents us to analyze the effect of the ATIES model on the effectiveness of different treatment strategies, which is essential in the future application of prognostic nomogram for treatment selection. Finally, bias, especially selection bias, may affect the authenticity of the association.
The novel four-ATIRE signature serves as a promising model to predict the survival of patients with BRCA, and these ATIRE sites might be novel therapeutic targets for BRCA treatment, which is warranted to be further studied.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
The studies involving human participants were reviewed and approved by the Institutional Review Board of Guangzhou Medical University. The patients/participants provided their written informed consent to participate in this study.
Conceptualization: LY and QW. Data analysis: JW and SC. Sample resources: JW, AZ, YZ, QL, and YL. Data curation, LY. Experimental operation: ZY and YW. Writing—original draft preparation: JW. Writing—review and editing: LY and QW. Funding acquisition: LY. All authors have read and agreed to the published version of the manuscript.
This research was funded by the National Natural Science Foundation of China grants, grant numbers 81871876, 82073628, and 81672303 (LY). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2022.861439/full#supplementary-material
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin (2021) 71(3):209–49. doi: 10.3322/caac.21660
2. Zeng H, Chen W, Zheng R, Zhang S, Ji JS, Zou X, et al. Changing Cancer Survival in China During 2003-15: A Pooled Analysis of 17 Population-Based Cancer Registries. Lancet Glob Health (2018) 6(5):e555–67. doi: 10.1016/S2214-109X(18)30127-X
3. Yap WK, Shih MC, Kuo C, Pai PC, Chou WC, Chang KP, et al. Development and Validation of a Nomogram for Assessing Survival in Patients With Metastatic Lung Cancer Referred for Radiotherapy for Bone Metastases. JAMA Netw Open (2018) 1(6):e183242. doi: 10.1001/jamanetworkopen.2018.3242
4. Zheng K, Liu X, Ji W, Lu J, Cui J, Li W. The Efficacy of Different Inflammatory Markers for the Prognosis of Patients With Malignant Tumors. J Inflammation Res (2021) 14:5769–85. doi: 10.2147/JIR.S334941
5. Xi G, Guo W, Kang D, Ma J, Fu F, Qiu L, et al. Large-Scale Tumor-Associated Collagen Signatures Identify High-Risk Breast Cancer Patients. Theranostics (2021) 11(7):3229–43. doi: 10.7150/thno.55921
6. Peng Y, Yu H, Jin Y, Qu F, Ren H, Tang Z, et al. Construction and Validation of an Immune Infiltration-Related Gene Signature for the Prediction of Prognosis and Therapeutic Response in Breast Cancer. Front Immunol (2021) 12:666137. doi: 10.3389/fimmu.2021.666137
7. Sun X, Zhou ZR, Fang Y, Ding S, Lu S, Wang Z, et al. A Novel Metabolic Gene Signature-Based Nomogram to Predict Overall Survival in Breast Cancer. Ann Transl Med (2021) 9(5):367. doi: 10.21037/atm-20-4813
8. van Zonneveld AJ, Kolling M, Bijkerk R, Lorenzen JM. Circular RNAs in Kidney Disease and Cancer. Nat Rev Nephrol (2021) 17(12):814–26. doi: 10.1038/s41581-021-00465-9
9. Zhang D, Yang S, Li Y, Yao J, Ruan J, Zheng Y, et al. Prediction of Overall Survival Among Female Patients With Breast Cancer Using a Prognostic Signature Based on 8 DNA Repair-Related Genes. JAMA Netw Open (2020) 3(10):e2014622. doi: 10.1001/jamanetworkopen.2020.14622
10. Duan F, Li J, Huang J, Hua X, Song C, Wang L, et al. Establishment and Validation of Prognostic Nomograms Based on Serum Copper Level for Patients With Early-Stage Triple-Negative Breast Cancer. Front Cell Dev Biol (2021) 9:770115. doi: 10.3389/fcell.2021.770115
11. Christofi T, Zaravinos A. RNA Editing in the Forefront of Epitranscriptomics and Human Health. J Transl Med (2019) 17(1):319. doi: 10.1186/s12967-019-2071-4
12. Nishikura K. A-To-I Editing of Coding and Non-Coding RNAs by ADARs. Nat Rev Mol Cell Biol (2016) 17(2):83–96. doi: 10.1038/nrm.2015.4
13. Ben-Aroya S, Levanon EY. A-To-I RNA Editing: An Overlooked Source of Cancer Mutations. Cancer Cell (2018) 33(5):789–90. doi: 10.1016/j.ccell.2018.04.006
14. Bazak L, Haviv A, Barak M, Jacob-Hirsch J, Deng P, Zhang R, et al. A-To-I RNA Editing Occurs at Over a Hundred Million Genomic Sites, Located in a Majority of Human Genes. Genome Res (2014) 24(3):365–76. doi: 10.1101/gr.164749.113
15. Han L, Diao L, Yu S, Xu X, Li J, Zhang R, et al. The Genomic Landscape and Clinical Relevance of A-To-I RNA Editing in Human Cancers. Cancer Cell (2015) 28(4):515–28. doi: 10.1016/j.ccell.2015.08.013
16. Wang Y, Xu X, Yu S, Jeong KJ, Zhou Z, Han L, et al. Systematic Characterization of A-To-I RNA Editing Hotspots in microRNAs Across Human Cancers. Genome Res (2017) 27(7):1112–25. doi: 10.1101/gr.219741.116
17. Mogensen UB, Ishwaran H, Gerds TA. Evaluating Random Forests for Survival Analysis Using Prediction Error Curves. J Stat Softw (2012) 50(11):1–23. doi: 10.18637/jss.v050.i11
18. Camp RL, Dolled-Filhart M, Rimm DL. X-Tile: A New Bio-Informatics Tool for Biomarker Assessment and Outcome-Based Cut-Point Optimization. Clin Cancer Res (2004) 10(21):7252–9. doi: 10.1158/1078-0432.CCR-04-0713
19. Steyerberg EW, Vergouwe Y. Towards Better Clinical Prediction Models: Seven Steps for Development and an ABCD for Validation. Eur Heart J (2014) 35(29):1925–31. doi: 10.1093/eurheartj/ehu207
20. Yu G, Wang LG, Han Y, He QY. Clusterprofiler: An R Package for Comparing Biological Themes Among Gene Clusters. OMICS (2012) 16(5):284–7. doi: 10.1089/omi.2011.0118
21. Wu Q, Li Q, Zhu W, Zhang X, Li H. Identification of Autophagy-Related Long Non-Coding RNA Prognostic Signature for Breast Cancer. J Cell Mol Med (2021) 25(8):4088–98. doi: 10.1111/jcmm.16378
22. Wang S, Zou X, Chen Y, Cho WC, Zhou X. Effect of N6-Methyladenosine Regulators on Progression and Prognosis of Triple-Negative Breast Cancer. Front Genet (2020) 11:580036. doi: 10.3389/fgene.2020.580036
23. Xu X, Wang Y, Liang H. The Role of A-To-I RNA Editing in Cancer Development. Curr Opin Genet Dev (2018) 48:51–6. doi: 10.1016/j.gde.2017.10.009
24. Chan TW, Fu T, Bahn JH, Jun HI, Lee JH, Quinones-Valdez G, et al. RNA Editing in Cancer Impacts mRNA Abundance in Immune Response Pathways. Genome Biol (2020) 21(1):268. doi: 10.1186/s13059-020-02171-4
25. Fu L, Qin YR, Ming XY, Zuo XB, Diao YW, Zhang LY, et al. RNA Editing of SLC22A3 Drives Early Tumor Invasion and Metastasis in Familial Esophageal Cancer. Proc Natl Acad Sci USA (2017) 114(23):E4631–40. doi: 10.1073/pnas.1703178114
26. Chen W, He W, Cai H, Hu B, Zheng C, Ke X, et al. A-To-I RNA Editing of BLCAP Lost the Inhibition to STAT3 Activation in Cervical Cancer. Oncotarget (2017) 8(24):39417–29. doi: 10.18632/oncotarget.17034
27. Nakano M, Fukami T, Gotoh S, Nakajima M. A-To-I RNA Editing Up-Regulates Human Dihydrofolate Reductase in Breast Cancer. J Biol Chem (2017) 292(12):4873–84. doi: 10.1074/jbc.M117.775684
28. Gumireddy K, Li A, Kossenkov AV, Sakurai M, Yan J, Li Y, et al. The mRNA-Edited Form of GABRA3 Suppresses GABRA3-Mediated Akt Activation and Breast Cancer Metastasis. Nat Commun (2016) 7:10715. doi: 10.1038/ncomms10715
29. Shoshan E, Mobley AK, Braeuer RR, Kamiya T, Huang L, Vasquez ME, et al. Reduced Adenosine-to-Inosine miR-455-5p Editing Promotes Melanoma Growth and Metastasis. Nat Cell Biol (2015) 17(3):311–21. doi: 10.1038/ncb3110
30. Han SW, Kim HP, Shin JY, Jeong EG, Lee WC, Kim KY, et al. RNA Editing in RHOQ Promotes Invasion Potential in Colorectal Cancer. J Exp Med (2014) 211(4):613–21. doi: 10.1084/jem.20132209
31. Chan TH, Lin CH, Qi L, Fei J, Li Y, Yong KJ, et al. A Disrupted RNA Editing Balance Mediated by ADARs (Adenosine DeAminases That Act on RNA) in Human Hepatocellular Carcinoma. Gut (2014) 63(5):832–43. doi: 10.1136/gutjnl-2012-304037
32. Hochberg M, Gilead L, Markel G, Nemlich Y, Feiler Y, Enk CD, et al. Insulin-Like Growth Factor-Binding Protein-7 (IGFBP7) Transcript: A-To-I Editing Events in Normal and Cancerous Human Keratinocytes. Arch Dermatol Res (2013) 305(6):519–28. doi: 10.1007/s00403-013-1338-5
33. Bueno MJ, Ruiz-Sepulveda JL, Quintela-Fandino M. Mitochondrial Inhibition: A Treatment Strategy in Cancer? Curr Oncol Rep (2021) 23(4):49. doi: 10.1007/s11912-021-01033-x
34. Xu J, Brosseau JP, Shi H. Targeted Degradation of Immune Checkpoint Proteins: Emerging Strategies for Cancer Immunotherapy. Oncogene (2020) 39(48):7106–13. doi: 10.1038/s41388-020-01491-w
Keywords: A-to-I RNA editing, breast cancer, nomogram, overall survival, disease-free survival
Citation: Wan J, Chen S, Zhang A, Liu Y, Zhang Y, Li Q, Yu Z, Wan Y, Yang L and Wang Q (2022) Development and Validation of a Four Adenosine-to-Inosine RNA Editing Site-Relevant Prognostic Signature for Assessing Survival in Breast Cancer Patients. Front. Oncol. 12:861439. doi: 10.3389/fonc.2022.861439
Received: 24 January 2022; Accepted: 14 March 2022;
Published: 12 April 2022.
Edited by:
Mohammad Taheri, University Hospital Jena, GermanyCopyright © 2022 Wan, Chen, Zhang, Liu, Zhang, Li, Yu, Wan, Yang and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lei Yang, bGVpeWFuZ0BnemhtdS5lZHUuY24=; Qi Wang, d2FuZ3FpZ3pAMjFjbi5jb20=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.