
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Oncol. , 23 December 2021
Sec. Neuro-Oncology and Neurosurgical Oncology
Volume 11 - 2021 | https://doi.org/10.3389/fonc.2021.788819
 Harry Subramanian1
Harry Subramanian1 Rahul Dey1
Rahul Dey1 Waverly Rose Brim1
Waverly Rose Brim1 Niklas Tillmanns1
Niklas Tillmanns1 Gabriel Cassinelli Petersen1
Gabriel Cassinelli Petersen1 Alexandria Brackett2
Alexandria Brackett2 Amit Mahajan1
Amit Mahajan1 Michele Johnson1
Michele Johnson1 Ajay Malhotra1
Ajay Malhotra1 Mariam Aboian1*
Mariam Aboian1*Purpose: Machine learning has been applied to the diagnostic imaging of gliomas to augment classification, prognostication, segmentation, and treatment planning. A systematic literature review was performed to identify how machine learning has been applied to identify gliomas in datasets which include non-glioma images thereby simulating normal clinical practice.
Materials and Methods: Four databases were searched by a medical librarian and confirmed by a second librarian for all articles published prior to February 1, 2021: Ovid Embase, Ovid MEDLINE, Cochrane trials (CENTRAL), and Web of Science-Core Collection. The search strategy included both keywords and controlled vocabulary combining the terms for: artificial intelligence, machine learning, deep learning, radiomics, magnetic resonance imaging, glioma, as well as related terms. The review was conducted in stepwise fashion with abstract screening, full text screening, and data extraction. Quality of reporting was assessed using TRIPOD criteria.
Results: A total of 11,727 candidate articles were identified, of which 12 articles were included in the final analysis. Studies investigated the differentiation of normal from abnormal images in datasets which include gliomas (7 articles) and the differentiation of glioma images from non-glioma or normal images (5 articles). Single institution datasets were most common (5 articles) followed by BRATS (3 articles). The median sample size was 280 patients. Algorithm testing strategies consisted of five-fold cross validation (5 articles), and the use of exclusive sets of images within the same dataset for training and for testing (7 articles). Neural networks were the most common type of algorithm (10 articles). The accuracy of algorithms ranged from 0.75 to 1.00 (median 0.96, 10 articles). Quality of reporting assessment utilizing TRIPOD criteria yielded a mean individual TRIPOD ratio of 0.50 (standard deviation 0.14, range 0.37 to 0.85).
Conclusion: Systematic review investigating the identification of gliomas in datasets which include non-glioma images demonstrated multiple limitations hindering the application of these algorithms to clinical practice. These included limited datasets, a lack of generalizable algorithm training and testing strategies, and poor quality of reporting. The development of more robust and heterogeneous datasets is needed for algorithm development. Future studies would benefit from using external datasets for algorithm testing as well as placing increased attention on quality of reporting standards.
Systematic Review Registration: www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42020209938, International Prospective Register of Systematic Reviews (PROSPERO 2020 CRD42020209938).
As the healthcare needs of the population increase and the volume of imaging grows, there is a critical need for computer assisted models to provide support to radiologists in routine clinical practice. Brain tumors, and specifically gliomas, are of particular interest to neuro-oncologists and radiologists. Machine learning research in neuro-oncology has become increasingly popular as sufficient computing power and large datasets have come to be more available to researchers. Machine learning refers to a subset of artificial intelligence consisting of algorithms that analyze data without explicit programming (1, 2). Deep learning is a subtype of machine learning that utilizes neural networks, which refer to algorithm models composed of neurons represented by nodes and interconnections between nodes (3). Machine learning has been applied to the diagnostic imaging of gliomas to augment classification, prognostication, segmentation, and treatment planning (4). Algorithms which can differentiate gliomas from other entities such as normal examinations, stroke, or demyelinating disease remain in the early stages of development. Until now, most studies have focused on brain tumor segmentation accuracy, and provide segmentation algorithms which are developed on datasets containing only glioma images. The identification of gliomas in a heterogeneous group of images is a critical function but less well studied. In clinical practice, most studies contain normal images or other non-oncologic pathology. Algorithms developed on datasets containing only glioma images are unlikely to be generalizable to clinical practice. Therefore, in this study we investigate how machine learning has been applied to the identification of gliomas in datasets which contain non-glioma images. A systematic review was performed to assess the existing body of literature and identify the most optimal targets for future research.
A systematic literature review was performed to identify how machine learning has been applied to identify gliomas in datasets which include non-glioma images, thereby simulating normal clinical practice. The study was registered with the International Prospective Register of Systematic Reviews (PROSPERO, CRD42020209938) and conducted in concordance with preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) guidelines (5). The primary literature search is summarized in the PRISMA flow diagram in Figure 1, and involved a query of four databases to identify all published articles investigating machine learning and gliomas. The queried databases were Ovid Embase, Ovid MEDLINE, Cochrane trials (CENTRAL), and Web of Science-Core Collection. The initial search included articles published prior to September 1, 2020, and a second search was performed to identify articles published between September 1, 2020 and February 1, 2021. The search strategy included both keywords and controlled vocabulary combining the terms for: artificial intelligence, machine learning, deep learning, radiomics, magnetic resonance imaging, glioma, as well as related terms. The search strategy and syntax are demonstrated in Supplementary Figure S1. The search was executed by a medical librarian and reviewed by a second institutional librarian.
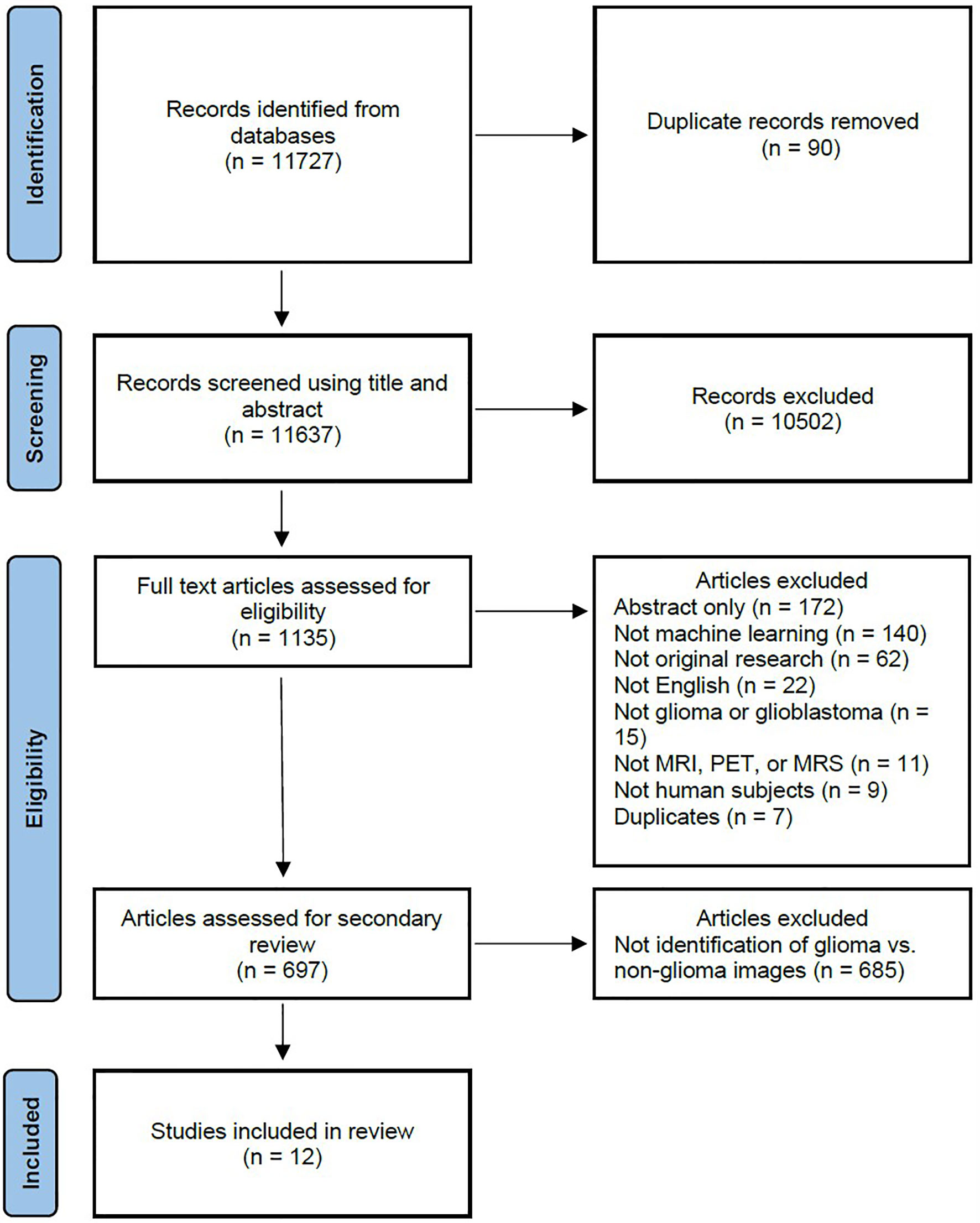
Figure 1 PRISMA flow diagram depicting the systematic review search strategy. (MRI, magnetic resonance imaging; MRS, magnetic resonance spectroscopy; PET, positron emission tomography.).
Screening of the articles was performed by two independent reviewers (H.S. and M.A.), which includes one board certified neuroradiologist (M.A.), utilizing Covidence (Covidence systematic review software, Veritas Health Innovation, Melbourne, Australia. Available at www.covidence.org). Articles were initially screened by title and abstract, after which the remaining articles were screened by full text. To meet inclusion criteria, the articles were required to be original research, investigate machine learning, investigate gliomas in human subjects, be published in the English language, and utilize imaging with either MRI, MRS, or PET. Further screening was then performed to identify articles which investigated the identification of gliomas in datasets including non-glioma images. Each reviewer screened each article independently and disagreement was resolved by discussion.
Data extraction was performed by two independent reviewers (H.S. and R.D.). Each reviewer extracted the whole data independently and disagreement was resolved by discussion. Major data points included the study objective, dataset, number of patients and images, machine learning algorithm training and testing strategy, and magnetic resonance imaging (MRI) sequences. Quantitative data was also collected where available, including accuracy, sensitivity, specificity, area under the receiver operating characteristic curve (AUC) and Dice coefficient. When multiple algorithms were evaluated in a study, the best performing algorithm was reported.
Risk of bias assessment was performed using Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD) guidelines (6). The TRIOPD checklist contains 22 primary features as well as multiple subitems, resulting in a total of 37 features. A TRIPOD score was created using 1 possible point for each subitem. Adherence to a subitem was given 1 point, while non-adherence was scored as 0 points. Features not assessed in an article due to the nature of the study were deemed as not applicable and excluded from analysis. The primary features are title (1 point), abstract (1 point), introduction - background and objectives (2 points), methods - source of data (2 points), methods - participants (3 points), methods - outcome (2 points), methods - predictors (2 points), methods - sample data (1 point), methods - missing data (1 point), methods - statistical analysis (5 points), methods - risk groups (1 point), methods - development and validation (1 point), results - participants (3 points), results - model development (2 points), results - model specification (2 points), results - model performance (1 point), results - model updating (1 point), discussion - limitations (1 point), discussion - interpretation (2 points), discussion - implications (1 point), supplementary information (1 point) and funding (1 point). The individual TRIPOD ratio was calculated for each article as the ratio of the TRIPOD score to the maximum possible points calculated from the included features. The TRIPOD adherence ratio for each feature was calculated as the ratio of the total points for a specific feature to the total possible points from all of the articles assessing that feature.
Descriptive statistics were calculated and visualized using GraphPad Prism version 9.1.2 for Windows, GraphPad Software, San Diego, California USA, www.graphpad.com.
The primary literature search returned 11,727 candidate articles, of which 90 duplicates were removed. The remaining 11,637 articles were screened using title and abstract, of which 10,502 articles that did not involve neuro-oncology were excluded. The full text of the remaining 1,135 articles was reviewed, of which 438 articles were excluded. The 438 excluded articles consisted of 172 conference abstracts, 140 articles not utilizing machine learning, 62 not representing original research, 22 not published in the English language, 15 not investigating gliomas, 11 not utilizing MRI, magnetic resonance spectroscopy (MRS), or positron emission tomography (PET) imaging, 9 not utilizing human subjects, and 7 duplicate articles. The remaining 697 articles underwent further review, of which 685 articles were excluded and 12 articles (7–18) investigating the use of machine learning to identify gliomas in datasets which include non-glioma images were identified for inclusion in the final analysis.
The main data points extracted from the 12 articles are summarized in Table 1. The distribution of the objective of the articles is depicted in Figure 2, the distribution of datasets utilized is depicted in Figure 3, and algorithm testing strategies are depicted in Figure 4. Seven articles investigated the differentiation of normal from abnormal images in datasets which include gliomas, and five articles investigated the differentiation of glioma images from non-glioma or normal images. The most frequent dataset used was a single institution dataset (5 articles, of which 4 used the Harvard Medical School dataset), followed by the Multimodal Brain Tumor Image Segmentation Benchmark (BRATS; 3 articles), multicenter datasets (2 articles), and The Cancer Imaging Archive (TCIA; 2 articles). BRATS (19–21) and TCIA (22) are publicly available databases of annotated MR images of gliomas. The ground truth in the BRATS and TCIA datasets is defined by pathology. Additionally, there was pathologic ground truth in the single institution dataset used by Dube et al. In the Harvard Medical School (23) dataset used by four studies, the method of ground truth establishment is unknown. Additionally, in the two studies using other multicenter datasets, the method to establish ground truth is unknown for at least part of the data.
Algorithm training and testing strategies consisted of five-fold cross validation (5 articles), and use of exclusive sets of images within the same dataset for training and for testing (7 articles). The range of sample sizes is shown in Figure 5. The median sample size was 280 patients (reported in 9 articles, range 42 to 717). The three articles not reporting the number of patients did report the number of images, with a median of 298 images (range 90 to 400). The sequences of magnetic resonance images used in each study was variable, consisting of some combination of T1-weighted, T2-weighted, contrast enhanced T1-weigthed, T2 fluid attenuated inversion recovery (FLAIR), and diffusion weighted (DWI) images.
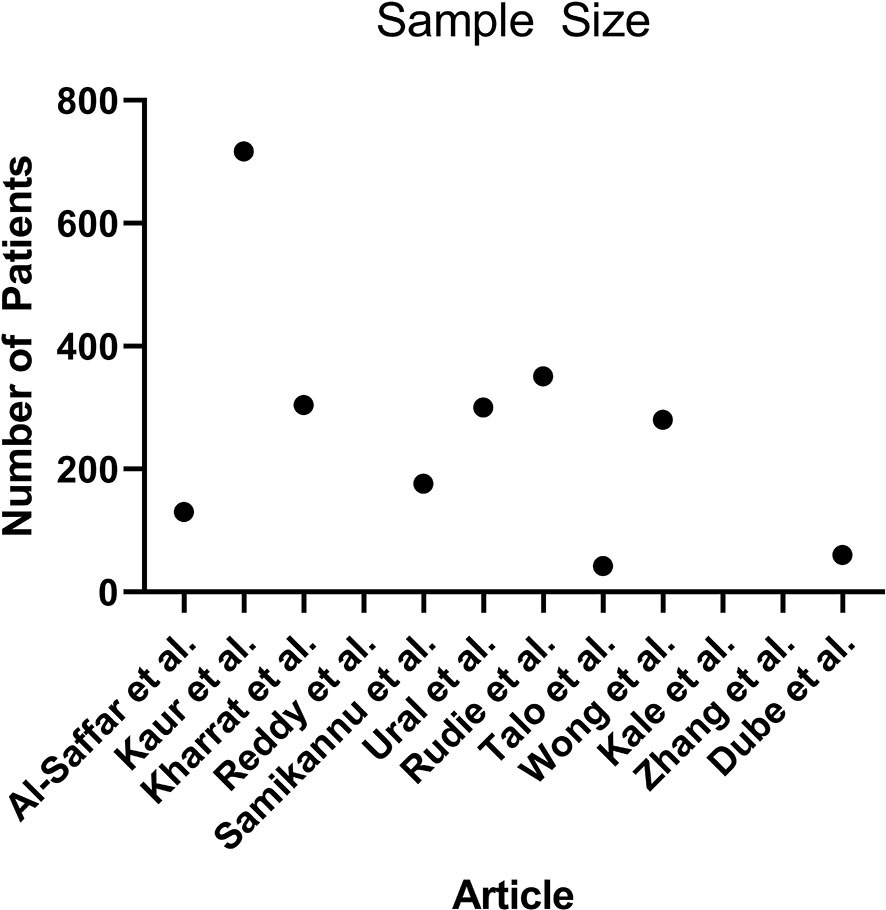
Figure 5 Scatterplot demonstrating the number of patients used in each article (n = 9, 3 articles did not report the number of patients).
A description of the machine learning algorithms is presented in Table 2. The most common algorithm was a neural network used in 10 articles, while two articles used support vector machine algorithms. A wide variety of neural networks were used, including five articles which developed novel algorithms. The quantitative results are demonstrated in Table 3, which summarizes the testing performance of each algorithm, and includes accuracy, sensitivity, specificity, AUC, and Dice coefficient. When multiple algorithms were evaluated in a study, the best performing algorithm was reported. The most commonly reported metric was accuracy, which ranged from 0.75 to 1.00 (median 0.96, 10 articles). When segmentation was investigated, the Dice coefficient was reported, which ranged from 0.92 to 0.98 (2 articles). A random effects meta-analysis was attempted, however could not be performed due to the lack of available data (24). The AUC was reported in only one of 12 articles and therefore not suitable for meta-analysis. Furthermore, for algorithm accuracy the standard deviation or confidence interval was only reported in three articles and therefore also not sufficient to perform an unbiased and generalizable meta-analysis (25).
Assessment of the quality of reporting using TRIPOD criteria yielded a mean individual TRIPOD ratio of 0.50 (standard deviation 0.14, range 0.37 to 0.85). Individual TRIPOD scores are depicted in Figure 6 and feature TRIPOD adherence scores are depicted in Figure 7. Due to the inherent nature of the articles, no study created risk groups or discussed model updating. Both subitems of model specification were also not fully discussed in any article. In addition, both subitems of model development were fully included in only two articles. The maximum possible points for an individual article ranged from 26 to 29 when accounting for non-applicable features (the theoretical maximum points with all features included would be 37). Of the eligible features, the poorest adherence was seen with the title (0 adherent articles), abstract (1 adherent article), missing data (1 adherent article), results - participants (0 adherent articles) and model performance (2 adherent articles).
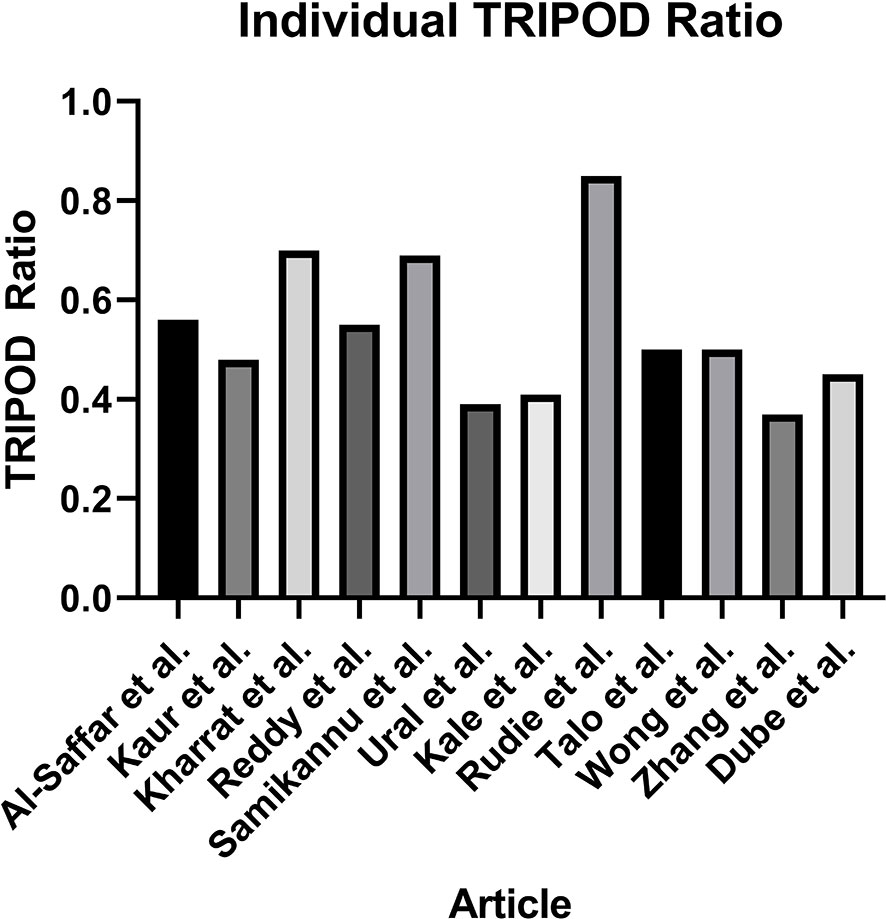
Figure 6 Individual TRIPOD Ratio, calculated for each article as the ratio of the TRIOPD score to the maximum possible score.
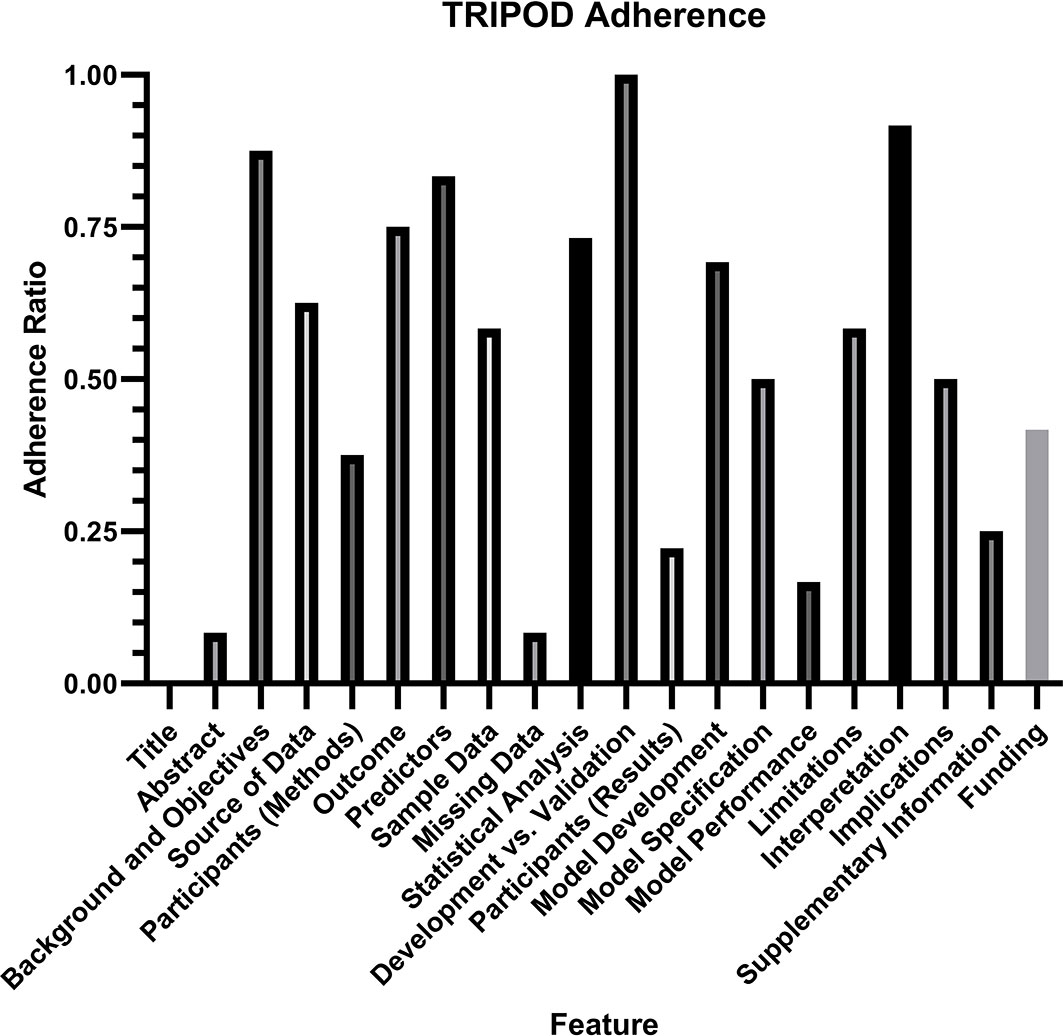
Figure 7 TRIPOD Adherence Ratio, calculated for each feature as the ratio of the total points scored to the total possible points for that feature. Notably, two features (risk groups and model updating) were not assessed in any article and therefore not included in the analysis.
Additional linear regression analysis was performed to identify any predictor of algorithm accuracy. A comparison of algorithm accuracy and the sample size is demonstrated in Supplementary Figure S2, and shows no significant relationship, with an R2 of 0.1204 (P = 0.45). A comparison of algorithm accuracy and the individual TRIPOD ratio is demonstrated in Supplementary Figure S3, and shows no significant relationship, with an R2 of 0.01578 (P = 0.73).
A systematic review of the literature identified 12 studies which investigate the use of machine learning to identify gliomas in datasets which include non-glioma images. This scenario most closely simulates routine clinical practice, where gliomas are intermixed with normal examinations and non-oncologic pathologies. Moreover, these algorithms may have the potential to support a screening program for gliomas in the future. The studies were all published between 2006 and 2020, with nine published from 2019 to 2020, reflecting the increasing popularity of machine learning research in recent years. The five studies using BRATS or TCIA datasets included only normal images or glioma images. These datasets are more generalizable to clinical practice than those containing only glioma images, however still lack other routinely encountered pathologies. The remaining seven studies utilizing other single institution or multicenter datasets included a mix of normal, glioma, and other pathologic images. The other pathologies included stroke, Alzheimer’s disease, multiple sclerosis, and meningioma, among others. This data is more representative of routine clinical practice, however still comes with limitations. There are a wide variety of healthcare settings, such as a tertiary or academic medical center, small hospital, or outpatient practice, each with different patient populations and pathologies. Additionally, datasets from different locations around the world will demonstrate different heterogeneity based on regional variations.
There are major limitations with the algorithm training and testing strategies. The description of algorithm training, validation, and testing strategies is heterogenous across studies. Often in machine learning research, validation and testing are used interchangeably, however this leads to confusion in the evaluation of algorithm performance. Validation should be reserved for the description of algorithm finetuning using data separate from the training data. Testing should be used to describe the unbiased evaluation of an algorithm using data separate from the training and validation sets. Each study reported training and testing data, however many studies used the term validation for what should actually be described as testing. Only two studies performed a true validation in addition to training and testing, Al-Saffar et al. used 5-fold cross validation for training and validation followed by a separate set of images within the same dataset for testing, and Rudie et al. used 10-fold cross validation for training and validation followed by a separate set of images within the same dataset for testing. None of the 12 studies tested their algorithms on external data. This poses a major limitation to the generalizability of these algorithms. In the United States, this also hinders the ability for approval by the Food and Drug Administration, which recommends algorithms be tested on external datasets.
Overall, there appears to be limited availability of high-quality data to train these machine learning algorithms. The number of patients in the datasets was low, with no study reaching 1,000 patients, and one study dropping as low as 42 patients. As a result of low sample sizes, the k-fold cross validation technique was commonly used for algorithm training, and five studies even used k-fold cross validation to test their algorithms. This technique is optimal for providing more data with a small sample size, but comes with the drawback of increased overtraining and decreased generalizability when applying the algorithm to an outside dataset. Additionally, nine studies used the same three datasets: BRATS, TCIA, and Harvard Medical School. Only two studies used datasets compiled from multiple institutions. This highlights a need to develop larger and more clinically applicable datasets to perform more robust machine learning research. Moreover, it will be critical to develop datasets that closely represent the mix of pathology encountered in each individual hospital, because this will vary between different institutions and practice settings. This will potentially fabricate the need for hospital specific dataset creation for the translation of algorithms.
Risk of bias analysis using TRIPOD criteria revealed that the quality of reporting was insufficient to draw any conclusion about algorithm generalizability. On average, there was adherence to only half of the reporting standards, with a large variation between studies. The poorest adherence was noted with the title and abstract, the method for handling missing data, the description of study participants within the results section, and the reporting of model performance. Specifically for model performance, the confidence interval of the discrimination measure was reported in only two studies. It is important to note that the TRIOPD criteria were primary developed for studies that used conventional multivariate regression prediction models rather than machine learning models, and TRIPOD-AI criteria are currently in development to specifically address the reporting of artificial intelligence and machine learning models (26). Poor quality of reporting also limited the ability to perform a meta-analysis, as AUC was reported in only one study, and the standard deviation for accuracy was reported in only three studies. Overall, the current analysis demonstrates that a substantial portion of information needed for translating algorithms to clinical practice is not available.
Systematic review of the literature identified machine learning algorithms which can identify gliomas in datasets containing non-glioma images, which are the most suitable algorithms for integration into general clinical workflow. Such algorithms may also serve as the basis for a potential brain tumor screening program. Severe limitations hindering the application of these algorithms to clinical practice were identified, including limited datasets, the lack of generalizable algorithm training and testing strategies, and poor quality of reporting. There is a need to develop more robust and heterogeneous datasets, which can be applied to individual clinical practice settings. Future studies would benefit from using external datasets for algorithm testing as well as placing increased attention on quality of reporting standards.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
This publication was made possible by KL2 TR001862 (MA) from the National Center for Advancing Translational Science (NCATS), components of the National Institutes of Health (NIH), and NIH roadmap for Medical Research. This publication also received support from the American Society of Neuroradiology Fellow Award 2018 (MA).
Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NIH.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2021.788819/full#supplementary-material
Supplementary Figure 1 | Search strategy and syntax. A total of four databases were searched: Embase, Ovid MEDLINE(R), Cochrane CENTRAL (trials), and Web of Science.
Supplementary Figure 2 | Linear regression analysis demonstrates no significant relationship between algorithm accuracy and sample size (R2 of 0.1204, P = 0.45).
Supplementary Figure 3 | Linear regression analysis demonstrates no significant relationship between algorithm accuracy and individual TRIOPD ratio (R2 of 0.01578, P = 0.73).
1. Wang S, Summers RM. Machine Learning and Radiology. Med Image Anal (2012) 16:933–51. doi: 10.1016/j.media.2012.02.005
2. Badillo S, Banfai B, Birzele F, Davydov II, Hutchinson L, Kam-Thong T, et al. An Introduction to Machine Learning. Clin Pharmacol Ther (2020) 107(4):871–85. doi: 10.1002/cpt.1796
3. Zaharchuk G, Gong E, Wintermark M, Rubin D, Langlotz CP. Deep Learning in Neuroradiology. AJNR Am J Neuroradiol (2018) 39:1776–84. doi: 10.3174/ajnr.A5543
4. Lotan E, Jain R, Razavian N, Fatterpekar GM, Lui YW. State of the Art: Machine Learning Applications in Glioma Imaging. AJR Am J Roentgenol (2019) 212:26–37. doi: 10.2214/AJR.18.20218
5. Moher D, Shamseer L, Clarke M, Ghersi D, Liberati A, Petticrew M, et al. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015 Statement. Syst Rev (2015) 4(1):1. doi: 10.1186/2046-4053-4-1
6. Collins GS, Reitsma JB, Altman DG, Moons KG. Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD): The TRIPOD Statement. BMJ (2015) 350:g7594. doi: 10.1161/CIRCULATIONAHA.114.014508
7. Al-Saffar ZA, Yildirim T. A Novel Approach to Improving Brain Image Classification Using Mutual Information-Accelerated Singular Value Decomposition. IEEE Access (2020) 8:52575–87. doi: 10.1109/ACCESS.2020.2980728
8. Dube S, El-Saden S, Cloughesy TF, Sinha U. Content Based Image Retrieval for MR Image Studies of Brain Tumors. Conf Proc IEEE Eng Med Biol Soc (2006) 2006:3337–40. doi: 10.1109/IEMBS.2006.260262
9. Kale VV, Hamde ST, Holambe RS. Multi Class Disorder Detection of Magnetic Resonance Brain Images Using Composite Features and Neural Network. BioMed Eng Lett (2019) 9:221–31. doi: 10.1007/s13534-019-00103-1
10. Kaur T, Gandhi TK. Deep Convolutional Neural Networks With Transfer Learning for Automated Brain Image Classification. Mach Vision Appl Vol (2020) 31:20. doi: 10.1007/s00138-020-01069-2
11. Kharrat A, Neji M. A System for Brain Image Segmentation and Classification Based on Three-Dimensional Convolutional Neural Network. Computacion Y Sistemas (2020) 24:1617–26. doi: 10.13053/cys-24-4-3058
12. Reddy AVN, Krishna CP, Mallick PK. An Image Classification Framework Exploring the Capabilities of Extreme Learning Machines and Artificial Bee Colon. Neural Computing Appl (2020) 32:3079–99. doi: 10.1007/s00521-019-04385-5
13. Rudie JD, Weiss DA, Saluja R, Rauschecker AM, Wang J, Sugrue L, et al. Multi-Disease Segmentation of Gliomas and White Matter Hyperintensities in the BraTS Data Using a 3D Convolutional Neural Network. Front Comput Neurosci (2019) 13:84. doi: 10.3389/fncom.2019.00084
14. Samikannu R, Ravi R, Murugan S, Diarra B. An Efficient Image Analysis Framework for the Classification of Glioma Brain Images Using CNN Approach. Computers Mater & Continua (2020) 63:1133–42. doi: 10.32604/cmc.2020.08578
15. Talo M, Baloglu UB, Yıldırım Ö, Acharya UR. Application of Deep Transfer Learning for Automated Brain Abnormality Classification Using MR Images. Cogn Syst Res (2019) 54:176–88. doi: 10.1016/j.cogsys.2018.12.007
16. Ural B, Özışık P, Hardalaç F. An Improved Computer Based Diagnosis System for Early Detection of Abnormal Lesions in the Brain Tissues With Using Magnetic Resonance and Computerized Tomography Images. Multimedia Tools Appl (2020) 79:15613–34. doi: 10.1007/s11042-019-07823-7
17. Wong KCL, Syeda-Mahmood T, Moradi M. Building Medical Image Classifiers With Very Limited Data Using Segmentation Networks. Med Image Anal (2018) 49:105–16. doi: 10.1016/j.media.2018.07.010
18. Zhang Y, Wang S, Ji G, Dong Z. An MR Brain Images Classifier System via Particle Swarm Optimization and Kernel Support Vector Machine. ScientificWorldJournal (2013) 2013:130134. doi: 10.1155/2013/130134
19. Menze BH, Jakab A, Bauer S, Kalpathy-Cramer J, Farahani K, Kirby J, et al. The Multimodal Brain Tumor Image Segmentation Benchmark (BRATS). IEEE Trans Med Imaging (2015) 34(10):1993–2024. doi: 10.1109/TMI.2014.2377694
20. Bakas S, Akbari H, Sotiras A, Bilello M, Rozycki M, Kirby JS, et al. Advancing The Cancer Genome Atlas Glioma MRI Collections With Expert Segmentation Labels and Radiomic Features. Sci Data (2017) 4:170117. doi: 10.1038/sdata.2017.117
21. Bakas S, Reyes M, Jakab A, Bauer S, Rempfler M, Crimi A, et al. Identifying the Best Machine Learning Algorithms for Brain Tumor Segmentation, Progression Assessment, and Overall Survival Prediction in the BRATS Challenge. xrXiv (2018). preprint arXiv:1811.02629
22. Clark K, Vendt B, Smith K, Freymann J, Kirby J, Koppel P, et al. The Cancer Imaging Archive (TCIA): Maintaining and Operating a Public Information Repository. J Digit Imaging (2013) 26(6):1045–57. doi: 10.1007/s10278-013-9622-7
23. Johnson KA, Becker JA. The Whole Brain Atlas (1999). Available at: https://www.med.harvard.edu/aanlib/home.html.
24. Riley RD, Higgins JP, Deeks JJ. Interpretation of Random Effects Meta-Analyses. BMJ (2011) 342:d549. doi: 10.1136/bmj.d549
25. Liu Z, Yao Z, Li C, Liu X, Chen H, Gao C. A Step-by-Step Guide to the Systematic Review and Meta-Analysis of Diagnostic and Prognostic Test Accuracy Evaluations. Br J Cancer (2013) 108:2299–303. doi: 10.1038/bjc.2013.185
Keywords: artificial intelligence, bias, brain tumor, diagnostic imaging, glioma, machine learning, Magnetic Resonance Imaging, segmentation
Citation: Subramanian H, Dey R, Brim WR, Tillmanns N, Cassinelli Petersen G, Brackett A, Mahajan A, Johnson M, Malhotra A and Aboian M (2021) Trends in Development of Novel Machine Learning Methods for the Identification of Gliomas in Datasets That Include Non-Glioma Images: A Systematic Review. Front. Oncol. 11:788819. doi: 10.3389/fonc.2021.788819
Received: 03 October 2021; Accepted: 07 December 2021;
Published: 23 December 2021.
Edited by:
Alireza Mansouri, The Pennsylvania State University (PSU), United StatesReviewed by:
Bahram Mohajer, Johns Hopkins University, United StatesCopyright © 2021 Subramanian, Dey, Brim, Tillmanns, Cassinelli Petersen, Brackett, Mahajan, Johnson, Malhotra and Aboian. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mariam Aboian, bWFyaWFtLmFib2lhbkB5YWxlLmVkdQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.