
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Oncol. , 18 December 2020
Sec. Neuro-Oncology and Neurosurgical Oncology
Volume 10 - 2020 | https://doi.org/10.3389/fonc.2020.588260
Objective: We retrospectively evaluated the oncological and functional effectiveness of revision surgery for recurrent or remnant vestibular schwannoma (rVS).
Methods: We included 29 consecutive patients with unilateral hearing loss (16 women; mean age: 42.2 years) that underwent surgery for rVS. Previous surgeries included gross total resections (GTRs, n=11) or subtotal resections (n=18); mean times to recurrence were 9.45 and 4.15 years, respectively. House–Brackmann (HB) grading of facial nerve (FN) weakness (grades II-IV) indicated that 22 (75.9%) patients had deep, long-lasting FN paresis (HB grades: IV-VI). The mean recurrent tumor size was 23.3 mm (range: 6 to 51). Seven patients had neurofibromatosis type 2.
Results: All patients received revision GTRs. Fourteen small- to medium-sized tumors located at the bottom of the internal acoustic canal required the translabyrinthine approach (TLA); 12 large and small tumors, predominantly in the cerebellopontine angle, required the retrosigmoid approach (RSA); and 2 required both TLA and RSA. One tumor that progressed to the petrous apex required the middle fossa approach. Fifteen patients underwent facial neurorrhaphy. Of these, 11 received hemihypoglossal–facial neurorrhaphies (HHFNs); nine with simultaneous revision surgery. In follow-up, 10 patients (34.48%) experienced persistent deep FN paresis (HB grades IV-VI). After HHFN, all patients improved from HB grade VI to III (n=10) or IV (n=1). No tumors recurred during follow-up (mean, 3.46 years).
Conclusions: Aggressive microsurgical rVS treatment combined with FN reconstruction provided durable oncological and neurological effects. Surgery was a reasonable alternative to radiosurgery, particularly in facial neurorrhaphy, where it provided a one-step treatment.
Vestibular schwannoma (VS) surgery has changed beyond recognition since the early 20th century, when it was associated with mortality rates as high as 86% (1). The subsequent drop to 15.4% mortality, due to contributions from Harvey Cushing, was considered a milestone in VS surgery (2). In contrast, contemporary skull base surgery includes gross total resections (GTRs), but also aims for a good functional outcome, mostly by preserving facial nerve (FN) and other cranial nerve functions (3–6) in addition to reducing the mortality rate (7, 8).
Treatment options for VS include observation, surgery, via a retrosigmoid approach (RSA) (9–11), middle fossa approach (12), or translabyrinthine approach (TLA) (13), and stereotactic radiosurgery (SRS) (14–17). Additionally, some physicians prefer a combination of treatment methods, including an intentional partial resection, followed by SRS (18–20). Unfortunately, some patients experience tumor progression after a subtotal resection or recurrence after a GTR. Recurrences are uncommon; rates range from 0.3% to 9.2% (21). However, regrowth of a residual tumor occurs in up to 44% of cases (22).
Tumor recurrence or progression management remains controversial. In our department, we follow the VS management principles described by Samii, namely a GTR with the preservation of facial and cochlear nerve functions (23). A second microsurgery is much more difficult to perform than an initial GTR, due to adhesions and scarring, and it is even more difficult after a radiosurgery (24). In this retrospective case study, we described a series of patients that underwent revision surgery for VS and, in some cases, they received different methods of FN reanimation (25–27). In this study, we aimed to evaluate the oncological and functional effectiveness of revision surgery for recurrent and residual VS (together referred to as rVS).
This study was designed as a retrospective, single-center, consecutive case series, undertaken in an academic setting. All participants met the following criteria:
● Previous history of surgery for a VS, with or without radiation therapy;
● Histopathologic diagnosis of a VS, during the first and second surgeries;
● Regrowth, diagnosed in radiological imaging, or a residual tumor in patients with FN palsy;
● Revision surgery required for VS.
No specific exclusion criteria were applied. Patients underwent reoperations from 2002 to 2018. Data were collected between 2018 and 2019. We analyzed 29 patients (16 women, 13 men; mean age: 42.2 years) that underwent surgery for rVS. The mean time to revision surgery was 6.16 years (Table 1).
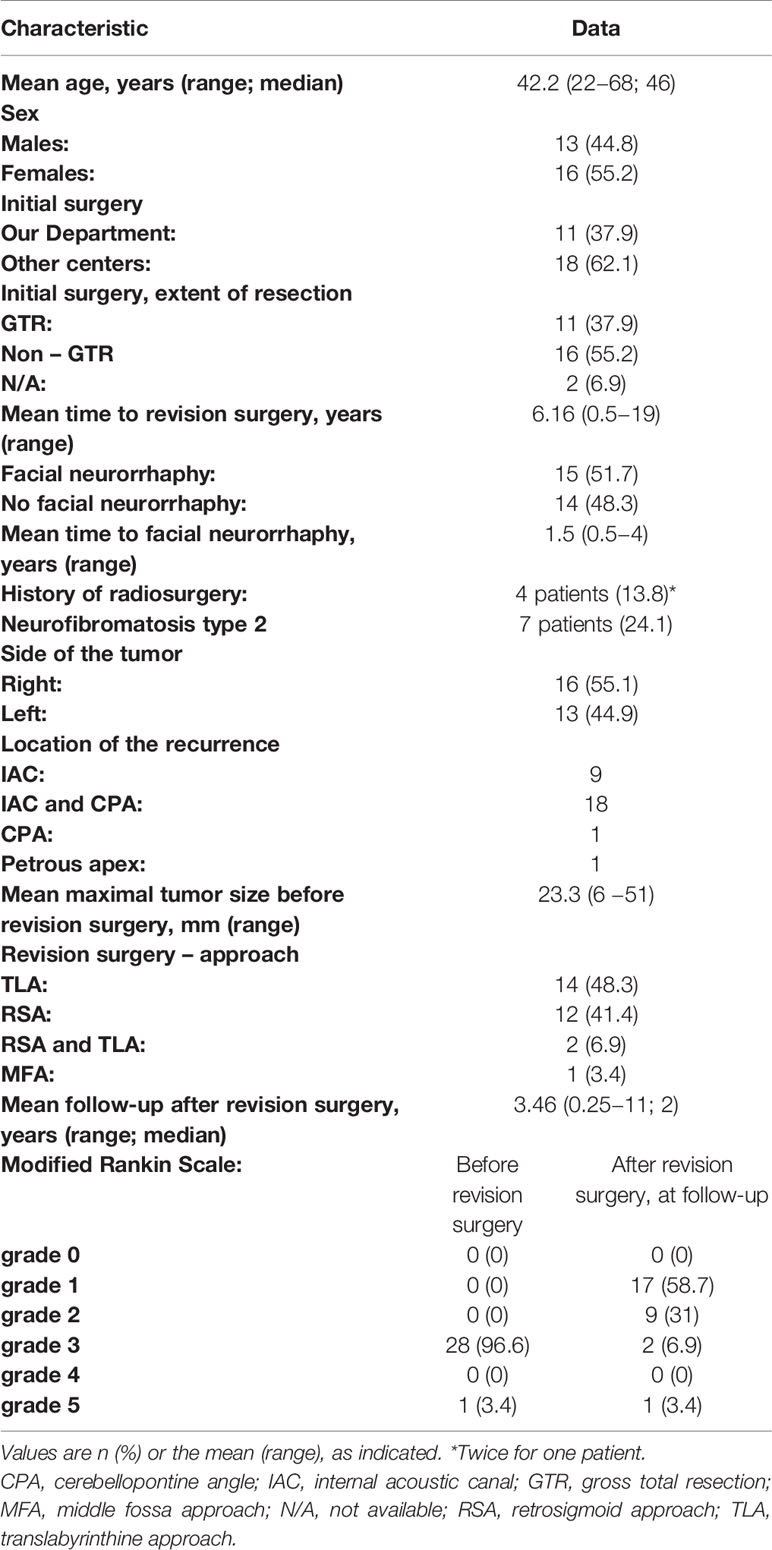
Table 1 Surgical and postoperative characteristics of 29 patients treated for vestibular schwannoma.
Eleven patients (37.9%) had undergone previous GTR surgeries in our department with the retrosigmoid approach (RSA). In this group, the mean time to revision surgery was 9.45 years (range 4 to 19). Eighteen other patients (62.1%) were referred from other centers, due to tumor progression after subtotal resections (all RSA); among these, 4 patients had undergone additional radiosurgery. The mean time to revision surgery was 4.15 years (range 0.5 to 16). Over one third (37.9%; 11/29) of the initial surgeries were GTRs; 55.2% (16/29) were subtotal resections; and 2 were unknown, due to the lack of available data.
All patients presented with unilateral hearing loss (American Academy of Otolaryngology-Head and Neck Surgery class D). Patients had different grades of FN weakness (House–Brackmann[HB] grades: II–VI). Most patients (75.8%) experienced deep FN paresis (HB grades: IV–VI; Tables 1, 2). Recurrent tumor sizes ranged from 6 to 51 mm (mean: 23.3 mm). Seven patients (24.1%) had neurofibromatosis type 2 (NF2). In those cases, surgery notes were reviewed to ensure that the tumor did not originate from a location other than the eighth cranial nerve.
The following pre-intervention procedures were included specifically for this study: when possible, descriptions of previous surgeries were acquired, and current scans were performed with magnetic resonance imaging (MRI), electromyography for the FN, and a bone window computed tomography (CT) for the temporal bone. Otherwise, all patients were prepared for elective surgery in a routine fashion. Hearing was not tested, because all patients were unilaterally deaf.
The interventions included revision surgery, tumor removal, and facial neurorrhaphy (n=15). One patient had undergone a facial neurorrhaphy prior to revision surgery, and 14 patients underwent tumor resections simultaneous with facial reanimation surgeries. Most facial reanimation procedures were hemihypoglossal-facial neurorrhaphies (80%, 12/15). The classic hypoglossal-to-facial nerve neurorrhaphy technique was applied in 3 (20%, 3/15) cases.
Each surgery was performed under general anesthesia, with the patient positioned on the back, with the head rotated contralaterally. The TLA was used for 14 small- to medium-sized tumors (median maximal size: 15.63 mm) that arose from the bottom of the internal acoustic canal (IAC). The RSA was used for larger tumors (median maximal size: 28.33 mm) and for smaller tumors (n=12) that were predominantly located in the CPA. In two cases, a combination of TLA and RSA was performed. The middle fossa approach was employed for one tumor that had progressed to the petrous apex (Figure 1).
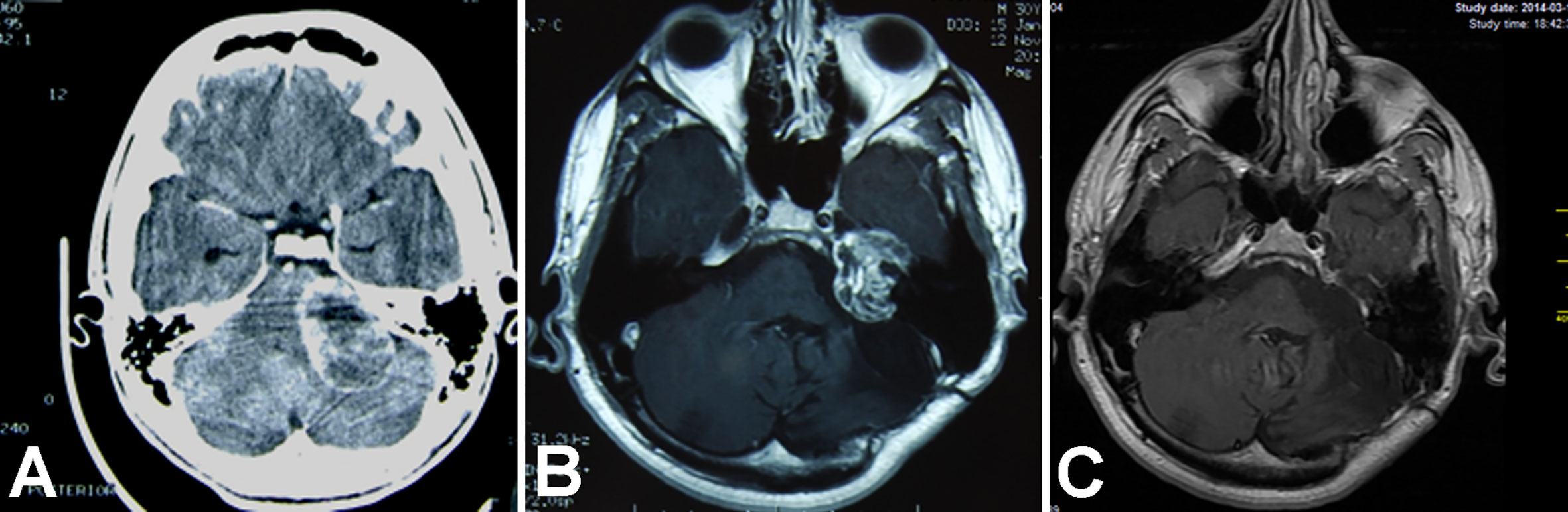
Figure 1 Magnetic resonance T1WI contrast enhanced axial images show treatment results in an 18-year-old man with vestibular schwannoma. (A) Brain scan before the initial surgery; (B) image after the initial surgery; (C) image after revision surgery via a middle fossa approach.
Intraoperative neurophysiological monitoring was performed only in selected cases. Hearing was not monitored, because all patients were unilaterally deaf. FN was monitored in patients that had at least partially preserved FN function after a previous surgery.
Patients that underwent facial neurorrhaphy were instructed by a neurophysiotherapist to perform self-massaging of the facial muscles postoperatively. Then, when the first signs of muscle reinnervation appeared, patients were to perform exercises in front of the mirror. Patients were monitored postoperatively, as follows: outpatient clinic visits at ½, 1, 1.5, and 3.5 years after surgery; and MRIs at ½, 1,5, and 3.5, years after surgery.
There were no deaths. Postoperative complications occurred in four patients (13.8%). These included one subcutaneous hematoma, after an abdominal fat harvest (treated with a wound revision); one otorrhea (treated with a temporary lumbar drainage); one rhinorrhea (treated with duraplasty); and one otorrhea combined with chronic otitis media (treated with temporary lumbar drainage and a petrosectomy). The cerebrospinal fluid (CSF) leak rate was 10.3% (3/29).
In all cases but one, GTR was accomplished during the second surgery. The one unsuccessful GTR was performed in a patient admitted in poor general condition (Modified Rankin Scale 5), and revision of a previous partial VS resection improved his neurological deficits, but not significantly. Among the other 28 patients, no subsequent tumor recurrence was noted during amen follow-up of 3.46 years (range: 0.25–11).
The FN function improved during follow-up in 69.0% (20/29) of patients, and it remained unchanged in 24.1% of patients (7/29). Additionally, two patients (6.9%, 2/29) experienced worse FN function after revision surgery. Both of those patients had been initially treated in our department, 8 and 9 years prior to revision surgery. They initially had HB grades of II and IV, and after revision surgery, their HB grades were IV and VI, respectively. Neither of these patients was eligible for facial neurorrhaphy, due to HB grade IV, in the former patient, and long-lasting FN paralysis, in the latter patient. When analyzed separately, 71.4% of patients (5/7) with NF2 experienced better FN function after revision surgery, and in 28.6% (2/7) it remained unchanged.
Fifteen patients received facial neurorrhaphies. Of these, 12 had hemihypoglossal–facial neurorrhaphies (HHFNs). Of the latter, 9 underwent an HHFN simultaneously with the revision surgery. In follow-up, 10 patients (34.48%) experienced continued deep FN paresis (HB grades IV-VI). Of these, 8 (80%, 8/10) had not received FN reanimation. Of patients with NF2, 42.9% (3/7) had HHFNs: 2 simultaneously with the revision surgery and 1 afterwards. After HHFN, all patients improved from HB grade VI to HB grade III, except one patient that improved to grade IV (Table 3). In summary, HB grades I–III were observed in 24.1% (7/29) of patients before revision surgery and in 65.5% (19/29) during follow-up.
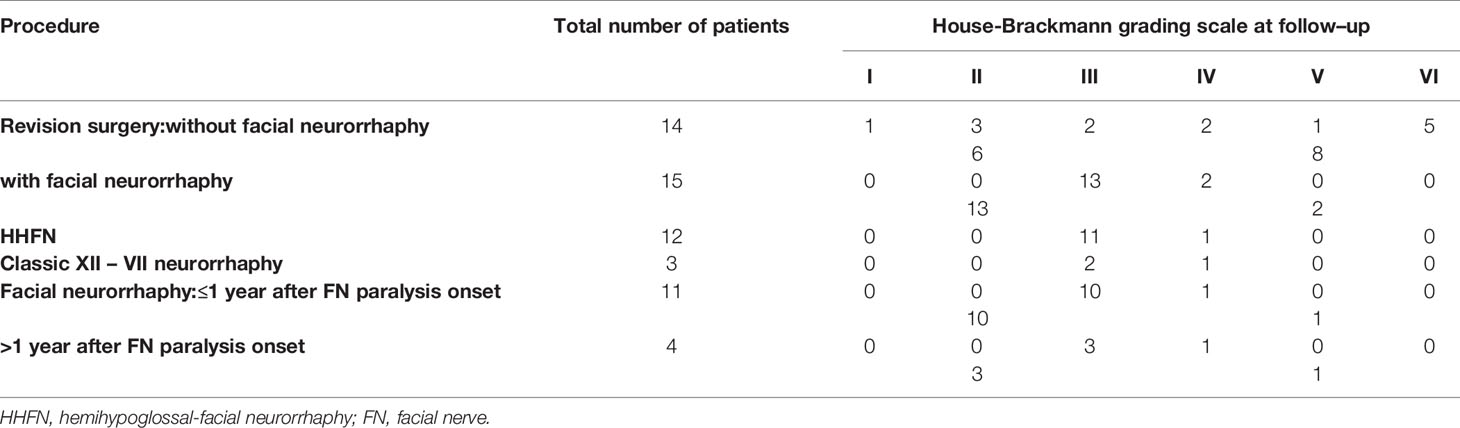
Table 3 Facial nerve function after revision surgery, according to the presence, technique, and time to facial neurorrhaphy.
During the last 5 years of the study (revision surgeries from2013 to 2018), 14 patients had available data. Of these, 9 received the HHFN technique, and the results were satisfactory results in each case. Due to our complex surgical strategy, 11 (78.6%) patients moved from an unsatisfactory functional grade (HB grades VI–VI) to a satisfactory functional grade (HB grades I–III, Table 4).
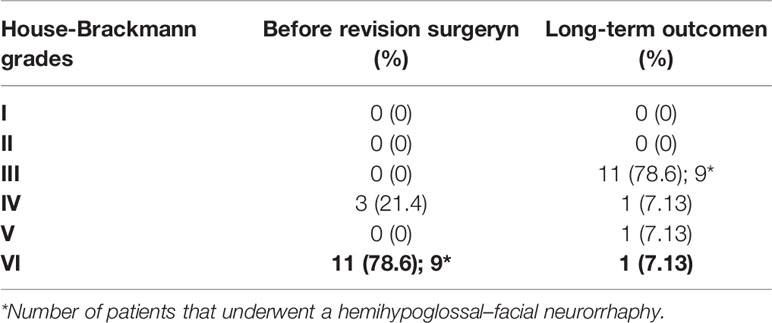
Table 4 Facial nerve outcomes in 14 patients treated for vestibular schwannoma during the last 5 years of the study.
An 18–year old male underwent a GTR with an RSA for a sporadic VS in our department. Then, he underwent a classic hypoglossal – facial neurorrhaphy. His FN function improved to HB grade IV. Eight years later, he was diagnosed with a recurrent tumor, with a maximal size of 33 mm, which had progressed toward the petrous apex. The patient underwent revision surgery via a middle fossa approach, and the tumor was totally resected. The postoperative course was uneventful. No subsequent tumor regrowth was noted in a follow-up of 11 years (Figure 1).
A 54–year old female presented with FN paresis after a subtotal VS resection in another center one year earlier. The neurological examination at admission revealed deafness, right sided dysmmetry, dizziness, subjective diplopia, hypoaesthesia on the right side of the face, and paresis of the soft palate, but no difficulties in swallowing. Her follow-up MRI revealed a tumor remnant, located in the right CPA, intrameatally. She was eligible for revision surgery via RSA with a simultaneous HHFN.
After revision surgery, the patient developed hypoglossal paresis, which subsequently resolved. Two months later, the patient developed otorrhea with chronic otitis media. A CT revealed a fistula between CPA, mastoid air cells and the external acoustic canal. The patient underwent petrosectomy. The subsequent postoperative course was uneventful; her follow-up MRI confirmed the completeness of the VS resection. In a 2–year follow–up, FN function improved to HB grade III (Figure 2).
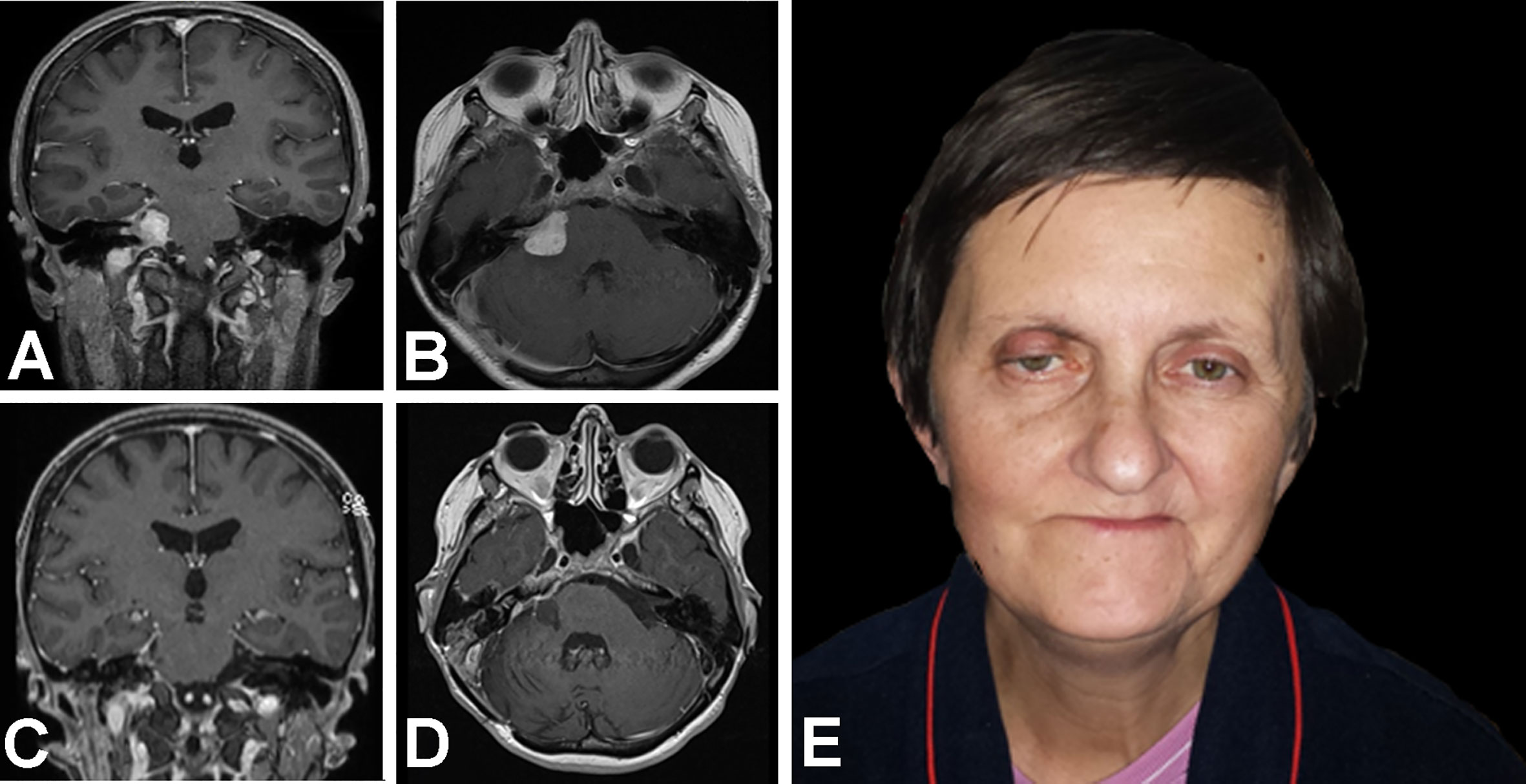
Figure 2 Magnetic resonance T1WI contrast enhanced images and photograph show treatment results in a 54–year old woman with facial nerve paresis after a subtotal resection of a vestibular schwannoma. At one year after the initial surgery, (A) coronal and (B) axial MR images show growth after stereotactic radiosurgery. Then, revision surgery was performed, via a retrosigmoid approach with facial neurorrhaphy; 11 months later, (C) coronal and (D) axial T1WI MR images show the results of a gross total resection with (E) satisfactory facial nerve function.
A 53–year old male presented with right facial and abducens nerve paresis and right-sided deafness after a partial VS removal 6 months earlier in another center. A tumor remnant was located close to the porus acusticus. He was eligible for a tumor remnant resection with a TLA and simultaneous HHFN. The postoperative course was uneventful. He was discharged one week after surgery. At a 1–year follow-up, his FN function improved to HB grade III, and a postoperative MRI revealed no tumor remnant (Figure 3).
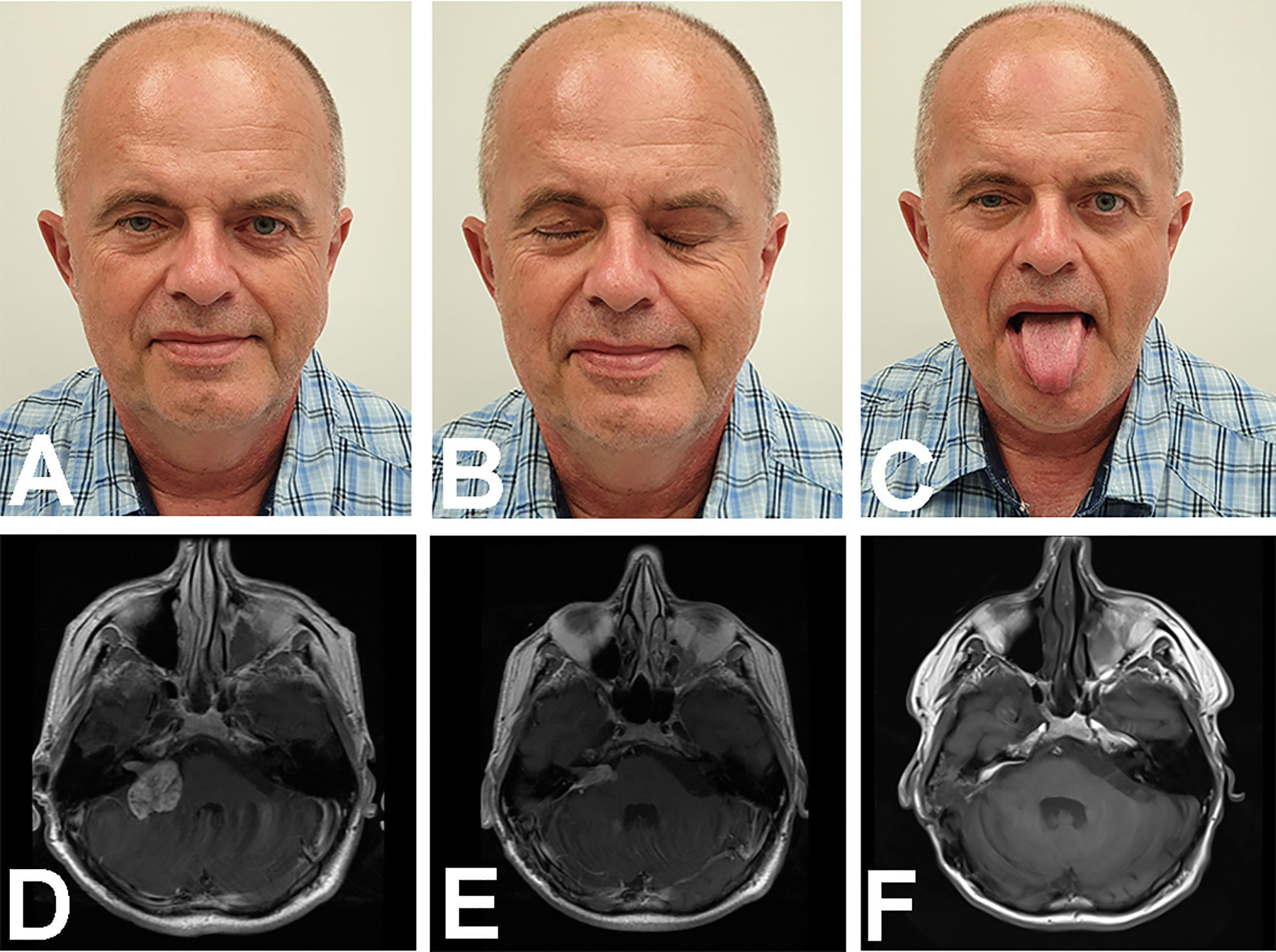
Figure 3 Images show treatment results in a 53–year old male with right facial nerve and abducens nerve pareses and right-sided deafness, after a partial VS removal. Photographs show the patient with the face (A) at rest, (B) with closed eyes, and (C) when asked to put out his tongue. The latter image is significant, because it shows no visible muscular atrophy and a straight position. (D−F) Magnetic resonance T1WI images with contrast enhancement show the brain (D) before, and (E) after the first surgery, which resulted in a remnant tumor. (F) The brain after revision surgery via a translabyrinthine approach.
The source of a VS recurrence might be a microscopic tumor remnant on the cranial nerves or in the fundus of the internal auditory canal (24). To address these possibilities, various attempts have been made to maximize the VS resection via a retrosigmoid or middle fossa approach; i.e., with endoscopy (28–30). However, in a previous retrospective study, Panigrahi et al. found that the Ki-67 labeling index, rather than the extent of resection, was associated with VS recurrences (31). Additionally, Freeman et al. hypothesized that a recurrence or regrowth after a GTR was more likely to reflect the biological behavior of the individual tumor, rather than the size of a tumor residue (32).
On the other hand, the number and relative contribution of revision surgeries for growing tumor remnants will probably increase, due to the current popularity of less invasive approaches, like non-total “functional” resections, with or without radiosurgery (20, 33). Therefore, complex solutions should be developed for the most demanding resections.
There are three therapeutic options for treating a VS, including observation, revision surgery, and radiosurgery. According to Tomita et al., a small remnant VS after microsurgery could be managed conservatively without additional treatment (34). Similarly, Troude et al. preferred a “wait and scan policy” over a postoperative SRS (35). Although the “wait and scan” policy is advantageously non-invasive, it was previously observed that, in certain patients with VS, the quality of life was mostly improved after microsurgery. However, one potential explanation for that result might be that the patient was not told that he/she had an intracranial tumor; this knowledge appeared to cause an intolerable burden, regardless of the tumor size, even though tumors often remained stable in observation groups (36, 37). Revision surgery is the only method that actually removes a tumor, but the trade-off is the high risk of postoperative complications, compared to other alternatives. Stereotactic radiosurgery is a less invasive alternative to microsurgery, but it does not actually eliminate the tumor. Moreover, large or giant tumors usually cannot be treated with SRS; therefore, they require microsurgical resection (38, 39). Furthermore, it should not be forgotten that, although rare and controversial, a VS can be malignantly transformed after a SRS, and radiation can induce a VS (40–44).
The aim of the second surgery should be an oncological cure and preserving FN function. Alternatively, in cases with FN paralysis, neurorrhaphy techniques should be carried out alongside the tumor resection, when the time period after paralysis onset allows. In contrast to the initial VS treatment, a second surgery does not usually aim to preserve hearing, because the patients are unilaterally deaf.
Several previous investigations of microsurgery for rVS showed satisfactory results and low complication rates (45). The rate of postoperative complications in our series was 10.3%, and the CSF leak rate was 13.8%. Three patients required different surgical revisions without intradural CPA inspections. Perry et al. described two cases in a 6-patient series where a CSF leak (33%) was successfully treated with a lumbar drain and surgical revision. Their summary of a previous series indicated a 4% CSF leak rate (45). Apart from that study, no other study reported major complications (24, 32, 45, 46).
When considering the rVS treatment approach, the surgeon should bear in mind both the location of the tumor and its size. Typically, the approach should be different than that previously used to avoid scar tissue, which can adhere tightly to the cranial nerves, brainstem, or cerebellum (32). In our experience, the TLA was most suitable for resecting minor remnants, when the primary aim was an HHFN, because, after skeletonizing the FN in the Fallopian canal, limited expansion of the bony drilling was required to access the tumor. On the other hand, for major tumor progressions toward the brain stem or petrous apex, the RSA and middle fossa approach are also reasonable choices.
In a literature review of microsurgical treatments for rVS, Perry et al. reported no recurrences after a mean follow-up of 55 months. That finding was consistent with our present findings with revision surgery; we found no recurrences after a mean follow-up of 41.5 months. In comparison, Huang et al. recently reported a 94% tumor control rate with SRS for rVS after a median clinical follow–up of 74 months (47).
In our series, the rate of deep FN deficits (HB grades IV-VI) was reduced from 75.9% to 34.5% in the long-term follow-up (p <0.05, Fisher’s exact test, Table 2). Considering only the results from the last 5 years of our study, 82% (9/11) of patients with complete FN paralysis recovered satisfactory function. According to Samii et al., the main predictor of FN outcome in a second VS surgery was the level of facial function before revision surgery (24). Postsurgical results can be further improved by combining revision surgery with facial neurorrhaphy. In our series, after an HHFN, all patients improved from HB grade VI to HB grade III, except one, who improved to grade IV.
Fourteen of our patients did not undergo facial neurorrhaphy, partly because the timeframe was exceeded for performing this procedure (n=7). Prolonged times between the FN paralysis onset and neurorrhaphy was correlated with worse FN outcomes (27). The remaining 7 patients had initial HB grades of II to IV, which excluded facial neurorrhaphy during revision surgery. In follow–up, the HB grades of this subgroup improved, except in 3 patients: the HB grade remained unchanged in 1 patient, and it worsened in 2 patients. For patients with unsatisfactory long–term FN function, plastic surgery was advised.
A substantial number of our patients had unsatisfactory FN function, particularly during the early part of the study period. The two main explanations were that some patients did not consent to receive face reanimation procedures and some surgeons underestimated the significance of postoperative FN weakness. Nineteen patients presented with HB grade VI after the first surgery; of these, 13 (68.4%) were treated initially in centers with relatively low surgical volumes. In other words, as much as 72.2% (13/18) of patients treated in other centers had HB grade VI after surgery. This finding suggested that patients with VS should be treated preferentially in experienced, large volume centers. High-volume departments have shorter lengths of stay, lower complication rates, and reduced hospital-related costs (48, 49). Patients treated initially in our department, apart from experiencing better FN outcomes, had longer mean times to revision surgery (9.45 years) compared to other centers (4.15 years). Similarly, Sanna et al. concluded that patients with VS should be treated by highly specialized centers, which limit subtotal resection to only few selected cases without compromising postoperative morbidity (50).
We performed GTRs during revision surgery in 97%of patients, and we observed FN functional improvements in 69% of patients. Based on these results, we suggest that revision surgery should be considered when: (1) a patient requires facial neurorrhaphy and has a stable residual tumor; or (2) the tumor has regrown (here, the HHFN should be performed, when necessary). Moreover, we prefer neurorrhaphy techniques, rather than plastic surgery, up to the end of the 3rd year after paresis onset (27). Additionally, it is important to provide a multidisciplinary, complex approach, from the diagnosis to late follow-up, with close surveillance of postoperative FN function.
This study was limited by its retrospective, single–center design. Additionally, patients with and without NF2 were analyzed together, despite differences in the management of these two groups.
This study demonstrated that aggressive microsurgical rVS treatment, together with modern FN reconstruction techniques provided an acceptable risk profile, yielded durable oncological effects, and could restore satisfactory FN function. We found that surgery was a reasonable alternative to SRS, particularly in patients that required facial neurorrhaphy, because it offered a one-step treatment.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
The studies involving human participants were reviewed and approved by The Bioethics Committee of the Medical University of Warsaw. The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
PK designed the project and critically reviewed it. ŁP prepared the manuscript, collected and reviewed data. WR and TD collected case data. AM did final approval of the version to be published. All authors have participated sufficiently in the conception and design of this work or the analysis and interpretation of the data, as well as the writing of the manuscript, to take public responsibility for it. All authors contributed to the article and approved the submitted version.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
This study was presented at the 30th Annual North American Skull Base Society Meeting, San Antonio, TX, USA, February 7-9, 2020.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2020.588260/full#supplementary-material
CPA, cerebellopontine angle; CSF, cerebrospinal fluid; CT, computed tomography; FN, facial nerve; GTR, gross total resection; HB, House–Brackmann; HHFN, hemihypoglossal-facial neurorrhaphy; IAC, internal acoustic canal; MFA, middle fossa approach; MRI, magnetic resonance imaging; NF2, neurofibromatosis type 2; RSA, retrosigmoid approach; rVS, recurrent and residual vestibular schwannoma; SRS, stereotactic radiosurgery; TLA, translabyrinthine approach; VS, vestibular schwannoma.
1. Machinis TG, Fountas KN, Dimopoulos V, Robinson JS. History of acoustic neurinoma surgery. Neurosurg Focus (2005) 18(4):e9. doi: 10.3171/foc.2005.18.4.10
2. Harvey Cushing. Tumors of the Nervus Acusticus and the Syndrome of the Cerebellopontine Angle. Philadelphia and London: WB Saunders Company (1917).
3. Kunert P, Smolarek B, Marchel A. Facial nerve damage following surgery for cerebellopontine angle tumours. Prevention and comprehensive treatment. Neurol Neurochir Pol (2011) 45(5):480–8. doi: 10.1016/S0028-3843(14)60317-0
4. Bernardo A, Evins AI, Visca A, Stieg PE. The intracranial facial nerve as seen through different surgical windows: an extensive anatomosurgical study. Neurosurgery (2013) 72(2 Suppl Operative):ons194–207. doi: 10.1227/NEU.0b013e31827e5844
5. Samii M, Matthies C. Hearing preservation in acoustic tumour surgery. Adv Tech Stand Neurosurg (1995) 22:343–73. doi: 10.1007/978-3-7091-6898-1_6
6. Gharabaghi A, Samii A, Koerbel A, Rosahl SK, Tatagiba M, Samii M. Preservation of function in vestibular schwannoma surgery. Neurosurgery (2007) 60(2 Suppl 1):ONS124–127; discussion ONS127-128. doi: 10.1227/01.NEU.0000249245.10182.0D
7. Niknafs YS, Wang AC, Than KD, Etame AB, Thompson BG, Sullivan SE. Hemorrhagic vestibular schwannoma: review of the literature. World Neurosurg (2014) 82(5):751–6. doi: 10.1016/j.wneu.2013.02.069
8. Yamakami I, Kubota S, Higuchi Y, Ito S. Challenging Anterior Inferior Cerebellar Artery in Retrosigmoid Vestibular Schwannoma Removal. World Neurosurg (2019) 121:e370–8. doi: 10.1016/j.wneu.2018.09.111
9. Zhou C, Evins AI, Boschi A, Tang Y, Li S, Przepiorka L, et al. Preoperative identification of the initial burr hole site in retrosigmoid craniotomies: A teaching and technical note. Int J Med Robot Comput Assist Surg MRCAS (2019) 15(3):e1987. doi: 10.1002/rcs.1987
10. Bernardo A, Boeris D, Evins AI, Anichini G, Stieg PE. A combined dual-port endoscope-assisted pre- and retrosigmoid approach to the cerebellopontine angle: an extensive anatomo-surgical study. Neurosurg Rev (2014) 37(4):597–608. doi: 10.1007/s10143-014-0552-8
11. Samii M, Matthies C. Management of 1000 vestibular schwannomas (acoustic neuromas): surgical management and results with an emphasis on complications and how to avoid them. Neurosurgery (1997) 40(1):11–21; discussion 21-23. doi: 10.1227/00006123-199701000-00002
12. Jean WC, Mueller K, Kim HJ. Middle Fossa Approach for Resection of an Intracanalicular Vestibular Schwannoma. J Neurol Surg Part B Skull Base (2019) 80(Suppl 3):S287. doi: 10.1055/s-0039-1677859
13. Feng Y, Patel NS, Gompel JJV, Carlson ML. Translabyrinthine Approach for Resection of Vestibular Schwannoma. J Neurol Surg Part B Skull Base (2019) 80(Suppl 3):S286. doi: 10.1055/s-0039-1677856
14. Shinya Y, Hasegawa H, Shin M, Sugiyama T, Kawashima M, Takahashi W, et al. Long-Term Outcomes of Stereotactic Radiosurgery for Vestibular Schwannoma Associated with Neurofibromatosis Type 2 in Comparison to Sporadic Schwannoma. Cancers (2019) 11(10):1498. doi: 10.3390/cancers11101498
15. Karam SD, Tai A, Strohl A, Steehler MK, Rashid A, Gagnon G, et al. Frameless fractionated stereotactic radiosurgery for vestibular schwannomas: a single-institution experience. Front Oncol (2013) 3:121. doi: 10.3389/fonc.2013.00121
16. Patel MA, Marciscano AE, Hu C, Jusué-Torres I, Garg R, Rashid A, et al. Long-term Treatment Response and Patient Outcomes for Vestibular Schwannoma Patients Treated with Hypofractionated Stereotactic Radiotherapy. Front Oncol (2017) 7:200. doi: 10.3389/fonc.2017.00200
17. Huang C-W, Tu H-T, Chuang C-Y, Chang CS, Chou HH, Lee MT, et al. Gamma Knife radiosurgery for large vestibular schwannomas greater than 3 cm in diameter. J Neurosurg (2018) 128(5):1380–7. doi: 10.3171/2016.12.JNS161530
18. Suero Molina E, van Eck ATCJ, Sauerland C, Schipmann S, Horstmann G, Stummer W, et al. Local Tumor Control and Clinical Symptoms After Gamma Knife Radiosurgery for Residual and Recurrent Vestibular Schwannomas. World Neurosurg (2019) 122:e1240–6. doi: 10.1016/j.wneu.2018.11.022
19. Bailo M, Boari N, Gagliardi F, Franzin A, Piloni M, Spina A, et al. Gamma Knife Radiosurgery for Residual and Recurrent Vestibular Schwannomas After Previous Surgery: Clinical Results in a Series of 90 Patients and Review of the Literature. World Neurosurg (2017) 98:60–72. doi: 10.1016/j.wneu.2016.10.066
20. Iwai Y, Ishibashi K, Watanabe Y, Uemura G, Yamanaka K. Functional Preservation After Planned Partial Resection Followed by Gamma Knife Radiosurgery for Large Vestibular Schwannomas. World Neurosurg (2015) 84(2):292–300. doi: 10.1016/j.wneu.2015.03.012
21. Shelton C. Unilateral acoustic tumors: how often do they recur after translabyrinthine removal? Laryngoscope (1995) 105(9 Pt 1):958–66. doi: 10.1288/00005537-199509000-00016
22. El-Kashlan HK, Zeitoun H, Arts HA, Hoff JT, Telian SA. Recurrence of acoustic neuroma after incomplete resection. Am J Otol (2000) 21(3):389–92. doi: 10.1016/S0196-0709(00)80049-6
23. Samii M, Matthies C. Management of 1000 vestibular schwannomas (acoustic neuromas): the facial nerve–preservation and restitution of function. Neurosurgery (1997) 40(4):684–94; discussion 694-695. doi: 10.1097/00006123-199704000-00006
24. Samii M, Metwali H, Gerganov V. Microsurgical management of vestibular schwannoma after failed previous surgery. J Neurosurg (2016) 125(5):1198–203. doi: 10.3171/2015.8.JNS151350
25. Asaoka K, Sawamura Y, Nagashima M, Fukushima T. Surgical anatomy for direct hypoglossal-facial nerve side-to-end “anastomosis.” J Neurosurg (1999) 91(2):268–75. doi: 10.3171/jns.1999.91.2.0268
26. Kunert P, Podgórska A, Bartoszewicz R, Marchel A. Hemihypoglossal-facial nerve anastomosis for facial nerve palsy. Neurol Neurochir Pol (2011) 45(5):452–60. doi: 10.1016/S0028-3843(14)60313-3
27. Dziedzic TA, Kunert P, Marchel A. Hemihypoglossal-Facial Nerve Anastomosis for Facial Nerve Reanimation: Case Series and Technical Note. World Neurosurg (2018) 118:e460–7. doi: 10.1016/j.wneu.2018.06.217
28. Corrivetti F, Cacciotti G, Giacobbo Scavo C, Roperto R, Mastronardi L. Flexible Endoscopic-Assisted Microsurgical Radical Resection of Intracanalicular Vestibular Schwannomas by a Retrosigmoid Approach: Operative Technique. World Neurosurg (2018) 115:229–33. doi: 10.1016/j.wneu.2018.04.108
29. Montaser AS, Todeschini AB, Harris MS, Adunka OF, Prevedello DM. Role of Endoscopy in Resection of Intracanalicular Vestibular Schwannoma via Middle Fossa Approach: Technical Nuances. World Neurosurg (2018) 120:395–9. doi: 10.1016/j.wneu.2018.08.215
30. Wackym PA, King WA, Poe DS, Meyer GA, Ojemann RG, Barker FG, et al. Adjunctive use of endoscopy during acoustic neuroma surgery. Laryngoscope (1999) 109(8):1193–201. doi: 10.1097/00005537-199908000-00003
31. Panigrahi M, Kumar D, Vooturi S, Madigubba S. MIB Index as Predictor of Recurrence in Sporadic Vestibular Schwannomas. World Neurosurg (2018) 120:e1203–7. doi: 10.1016/j.wneu.2018.09.039
32. Freeman SRM, Ramsden RT, Saeed SR, Alzoubi FQ, Simo R, Rutherford SA, et al. Revision surgery for residual or recurrent vestibular schwannoma. Otol Neurotol Off Publ Am Otol Soc Am Neurotol Soc Eur Acad Otol Neurotol (2007) 28(8):1076–82. doi: 10.1097/MAO.0b013e318159e76a
33. Iwai Y, Yamanaka K, Ishiguro T. Surgery combined with radiosurgery of large acoustic neuromas. Surg Neurol (2003) 59(4):283–289; discussion 289-291. doi: 10.1016/S0090-3019(03)00025-9
34. Tomita Y, Tosaka M, Aihara M, Horiguchi K, Yoshimoto Y. Growth of Primary and Remnant Vestibular Schwannomas: A Three-Year Follow-Up Study. World Neurosurg (2015) 83(6):937–44. doi: 10.1016/j.wneu.2015.02.005
35. Troude L, Boucekine M, Montava M, Lavieille J-P, Régis J-M, Roche P-H. Adjunctive Gamma Knife Surgery or Wait and Scan Policy After Optimal Resection of Large Vestibular Schwannomas: Clinical and Radiologic Outcomes. World Neurosurg (2018) 118:e895–905. doi: 10.1016/j.wneu.2018.07.093
36. Di Maio S, Akagami R. Prospective comparison of quality of life before and after observation, radiation, or surgery for vestibular schwannomas. J Neurosurg (2009) 111(4):855–62. doi: 10.3171/2008.10.JNS081014
37. Malhotra PS, Sharma P, Fishman MA, Grumbine FL, Tholey R, Dam VQ, et al. Clinical, radiographic, and audiometric predictors in conservative management of vestibular schwannoma. Otol Neurotol Off Publ Am Otol Soc Am Neurotol Soc Eur Acad Otol Neurotol (2009) 30(4):507–14. doi: 10.1097/MAO.0b013e31819d3465
38. Silva J, Cerejo A, Duarte F, Silveira F, Vaz R. Surgical removal of giant acoustic neuromas. World Neurosurg (2012) 77(5–6):731–5. doi: 10.1016/j.wneu.2011.08.019
39. Sekhar LN, Tariq F, Ferreira M. Giant acoustic neuromas and their treatment. World Neurosurg (2012) 77(5–6):629–30. doi: 10.1016/j.wneu.2011.10.011
40. Yanamadala V, Williamson RW, Fusco DJ, Eschbacher J, Weisskopf P, Porter RW. Malignant transformation of a vestibular schwannoma after gamma knife radiosurgery. World Neurosurg (2013) 79(3–4):593.e1–8. doi: 10.1016/j.wneu.2012.03.016
41. Peker HO, Gok H, Altay T. Malignant Transformation of Vestibular Schwannoma after Stereotactic Radiosurgery. World Neurosurg (2019) 124:81–3. doi: 10.1016/j.wneu.2018.12.149
42. Yamanaka R, Hayano A. Radiation-Induced Malignant Peripheral Nerve Sheath Tumors: A Systematic Review. World Neurosurg (2017) 105:961–970.e8. doi: 10.1016/j.wneu.2017.06.010
43. Yamanaka R, Hayano A. Radiation-Induced Schwannomas and Neurofibromas: A Systematic Review. World Neurosurg (2017) 104:713–22. doi: 10.1016/j.wneu.2017.05.066
44. Akamatsu Y, Murakami K, Watanabe M, Jokura H, Tominaga T. Malignant peripheral nerve sheath tumor arising from benign vestibular schwannoma treated by gamma knife radiosurgery after two previous surgeries: a case report with surgical and pathological observations. World Neurosurg (2010) 73(6):751–4. doi: 10.1016/j.wneu.2010.04.009
45. Perry A, Graffeo CS, Copeland WR, Carlson ML, Neff BA, Driscoll CL, et al. Microsurgery for Recurrent Vestibular Schwannoma After Previous Gross Total Resection. Otol Neurotol Off Publ Am Otol Soc Am Neurotol Soc Eur Acad Otol Neurotol (2017) 38(6):882–8. doi: 10.1097/MAO.0000000000001402
46. Roche P-H, Khalil M, Thomassin J-M. Microsurgical removal of vestibular schwannomas after failed previous microsurgery. Prog Neurol Surg (2008) 21:158–62. doi: 10.1159/000156903
47. Huang MJ, Kano H, Mousavi SH, Niranjan A, Monaco EA, Arai Y, et al. Stereotactic radiosurgery for recurrent vestibular schwannoma after previous resection. J Neurosurg (2017) 126(5):1506–13. doi: 10.3171/2016.5.JNS1645
48. Hatch JL, Bauschard MJ, Nguyen SA, Lambert PR, Meyer TA, McRackan TR. Does Hospital Volume Affect Outcomes in Patients Undergoing Vestibular Schwannoma Surgery? Otol Neurotol Off Publ Am Otol Soc Am Neurotol Soc Eur Acad Otol Neurotol (2018) 39(4):481–7. doi: 10.1097/MAO.0000000000001718
49. Ward BK, Gourin CG, Francis HW. Vestibular schwannoma surgical volume and short-term outcomes in Maryland. Arch Otolaryngol Head Neck Surg (2012) 138(6):577–83. doi: 10.1001/archoto.2012.877
Keywords: vestibular schwannoma, surgery, revision, facial nerve, gross total resection
Citation: Przepiórka Ł, Kunert P, Rutkowska W, Dziedzic T and Marchel A (2020) Surgery After Surgery for Vestibular Schwannoma: A Case Series. Front. Oncol. 10:588260. doi: 10.3389/fonc.2020.588260
Received: 28 July 2020; Accepted: 03 November 2020;
Published: 18 December 2020.
Edited by:
Theodore Nicolaides, New York University, United StatesReviewed by:
Kaleb Yohay, New York University, United StatesCopyright © 2020 Przepiórka, Kunert, Rutkowska, Dziedzic and Marchel. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Przemysław Kunert, cHJ6ZW15c2xhdy5rdW5lcnRAd3VtLmVkdS5wbA==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.