- 1Department of Pediatrics, Division of Infectious Diseases and Immunology, Guerin Children's at Cedars-Sinai Medical Center, Los Angeles, CA, United States
- 2Infectious and Immunologic Diseases Research Center (IIDRC), Department of Biomedical Sciences, Cedars-Sinai Medical Center, Los Angeles, CA, United States
- 3Department of Neurosurgery, Maxine Dunitz Neurosurgical Research Institute, Cedars-Sinai Medical Center, Los Angeles, CA, United States
- 4Department of Biomedical Sciences, Cedars-Sinai Medical Center, Los Angeles, CA, United States
- 5Department of Neurology, Cedars-Sinai Medical Center, Los Angeles, CA, United States
While recent advances in diagnostics and therapeutics offer promising new approaches for Alzheimer's disease (AD) diagnosis and treatment, there is still an unmet need for an effective remedy, suggesting new avenues of research are required. Besides many plausible etiologies for AD pathogenesis, mounting evidence supports a possible role for microbial infections. Various microbes have been identified in the postmortem brain tissues of human AD patients. Among bacterial pathogens in AD, Chlamydia pneumoniae (Cp) has been well characterized in human AD brains and is a leading candidate for an infectious involvement. However, no definitive studies have been performed proving or disproving Cp's role as a causative or accelerating agent in AD pathology and cognitive decline. In this review, we discuss recent updates for the role of Cp in human AD brains as well as experimental models of AD. Furthermore, based on the current literature, we have compiled a list of potential mechanistic pathways which may connect Cp with AD pathology.
Introduction
Alzheimer's disease (AD) is the dominant cause of senile dementia, neuropathologically characterized by the presence of amyloid β-protein (Aβ) plaque and hyperphosphorylated (p)tau protein containing neurofibrillary tangles (NFTs), ultimately leading to neurodegeneration (Jack et al., 2018; Hampel et al., 2021; 2023 Alzheimer's disease facts figures, 2023). Late-onset AD (LOAD) is a progressive neurodegenerative disease that leads to the impairment of memory and cognitive ability, the erosion of social and behavioral skills, and eventual loss of life. The exact cause behind AD remains elusive but studies indicate that AD is ultimately the result of the sum of genetic, environmental, and lifestyle-related insults to the brain.
As AD mostly affects the aging population (Guzman-Martinez et al., 2019), per the National Institute of Aging (NIA), an estimated 6.7 million elderly Americans have been affected by AD dementia. AD is one of the leading causes of death in the elderly in the USA (2023 Alzheimer's disease facts figures, 2023). Worldwide, more than 50 million people have AD and trends suggest that these numbers may triple by 2050 (GBD 2019 Dementia Forecasting Collaborators, 2022). AD is more prevalent over the age of 65 with almost 50% of individuals aged over 85 being AD positive (2023 Alzheimer's disease facts figures, 2023). Significant progress has been made in diagnosis via FDA-approved PET-amyloid and -tau imaging, CSF Aβ, and more recently plasma phosphorylated tau 217 immunoassay (Ashton et al., 2024; Barthélemy et al., 2024) and disease-modifying treatments with anti-Aβ antibodies for AD, aimed at slowing down its progression (Dyck et al., 2022; Boxer and Sperling, 2023). However, despite these efforts, little headway has been achieved in finding both prophylactic and therapeutic AD treatments with substantial effects on preserving cognitive function.
The pathological hallmarks, Aβ plaques, NFTs, synaptic loss, neurodegeneration, vascular impairment, and more recently neuroinflammation, are all known to play a role in AD (Serrano-Pozo et al., 2011; Vickers et al., 2016). Aβ accumulation is believed to occur due to the hyperactivity of secretase enzymes (β-secretase and γ-secretase) which cleaves the amyloid precursor protein (APP) to Aβ40 and Aβ42 monomers. Oligomerization of these Aβ monomers results in the formation of amyloid plaque (Kravenska et al., 2020). Simultaneously, hyperphosphorylation of tau leads to the formation of NFTs in the limbic and cortical regions of the brain (Grundke-Iqbal et al., 1986). Subsequent accumulation of Aβ plaques and NFTs are associated with neurodegeneration affecting neurocognitive decline. Additionally, excessive accumulation of Aβ in the brain parenchyma attracts and activates glial cells, which subsequently potentiate neuroinflammation, vascular damage, and the infiltration of peripheral immune cells, further aggravating the neuroinflammation and neurodegeneration (Cai et al., 2014; Dionisio-Santos et al., 2019). Remarkably, the pathological hallmarks and subsequent inflammatory and neurodegenerative processes start many years before the onset of clinical symptoms (Bateman et al., 2012). This large preclinical gap provides an opportunity for early detection of disease processes, which is of paramount importance to allow effective early interventions before the irreversible loss of neurons.
The National Institute on Aging (NIA) and the Alzheimer's Association (NIA-AA) set a guideline for AD diagnosis and that is termed ATN; “A” stands for Aβ biomarker (amyloid PET or CSF Aβ42); “T” for the tau biomarker (CSF p-tau or tau PET); and “N” for the neurodegeneration biomarker (CSF t-tau, FDG-PET, or structural MRI) (Jack et al., 2016, 2018). Available detection methods limit the capacity to screen for early AD and predict progression and response to therapy in the clinical setting. Recent advances in the development and evaluation of brain PET imaging as well as plasma and cerebrospinal fluid (CSF) biomarkers have revolutionized the prospects of early diagnostic tools for AD (Li et al., 2015; Hameed et al., 2020). Both categories of fluid biomarkers appear superior to most currently available brain scanning tools, which require the administration of unsafe radiolabeled isotopes and have reduced cost-effectiveness, accessibility, and sensitivity. Yet, CSF extraction is an invasive approach which is not a convenient method for patients while plasma markers are affected by other peripheral metabolic processes, therefore plasma markers do not exclusively represent the events happening within the brain.
Recent evidence from experimental, epidemiologic, and clinical reports suggest that AD pathogenesis is not only restricted to the brain but also extends beyond the brain. The pathological hallmarks of AD are also identified in the retina, an accessible CNS tissue, in patients with mild cognitive impairment (MCI) and AD (Koronyo-Hamaoui et al., 2011; Morgia et al., 2016; Koronyo et al., 2017, 2023; Haan et al., 2018; Grimaldi et al., 2019; Shi et al., 2020, 2023; Xu et al., 2022; Walkiewicz et al., 2024). Beyond the CNS, AD pathogenesis is closely associated with several systemic abnormalities. Previous AD research generally focused on the CNS. However, several peripheral and systemic abnormalities, including disorders of systemic immunity, cardiovascular diseases, hepatic dysfunction, metabolic disorder, blood abnormalities, respiratory and sleep disorders, renal dysfunction, intestinal dysfunctions, and systemic inflammation are now understood to be linked to AD and are reported in AD patients (Wang et al., 2017).
Microbial pathogens in AD
The exact cause of AD is unknown, yet several risk factors are thought to play synergistic roles in AD development. These factors include head injuries, age and sex, vascular disease, lifestyle, obesity/diabetes, environmental factors, genetic factors, and infection. With the recent increase in understanding of how inflammation and immune responses play a significant role in AD biology, infection has garnered increased attention in AD research since many of the pathological immune processes observed in AD are also driven by infections. One retrospective study found that severe infections, which are not just limited to the CNS but also systemic inflammation, increased the risk of vascular dementia and AD (Sipilä et al., 2021). This suggests that prophylactic and therapeutic treatment of infectious diseases might slow AD progression and cognitive decline in people with a risk of AD/dementia (Bu and Wang, 2021). Aβ itself is expressed in response to infections and can function as an anti-microbial peptide (Soscia et al., 2010; Kumar et al., 2016). This understanding led to the anti-microbial protection hypothesis of Alzheimer's disease which basically states that Aβ produced in response to infections, can over time become dysregulated leading to improper clearance and induction of inflammation and tangle formation (Moir et al., 2018). This pathway, over time, can then drive the development of AD. Therefore, it is enormously important to investigate the involvement of pathogens in AD progression to better understand AD pathogenesis and therapeutic intervention. Indeed, several studies have identified the presence of different microbes in the post-mortem brain tissue of AD patients, suggesting they might be involved in AD (Renvoize and Hambling, 1984; Miklossy, 1993, 2011, 2015; Itzhaki et al., 1997; Riviere et al., 2002; Alonso et al., 2014; Pisa et al., 2015; Perry et al., 2016; Dominy et al., 2019; Ciaccio et al., 2021; Senejani et al., 2021).
Dr. Oscar Fischer was the first to describe in 1907 that cerebral plaques may be the result of a chronic infection and suggested the infectious etiology of AD (Fisher, 1907; Broxmeyer, 2017). Among the pioneer studies that provided evidence that infections playing a role in AD were conducted by Itzhaki et al. (1997), observing herpes simplex virus 1 (HSV1) in the AD brain and revealing HSV1 as a risk factor for AD. A few other reports also support the presence of HSV1 in AD brains (Chiara et al., 2012; Itzhaki, 2014). Another study observed an association with the presence of serum antibody titers for cytomegalovirus in AD patients (Renvoize and Hambling, 1984). More recently, many studies have suggested that COVID-19 may play a role in the onset of dementia and AD (Ciaccio et al., 2021; Pyne and Brickman, 2021; Gordon et al., 2022). SARS-CoV-2 virus infection can activate AD-like signaling in the human brain and induce neuroinflammation (Reiken et al., 2022; Soung et al., 2022). In addition to viruses, bacteria that can reach the brain generally will induce neuroinflammation, and subsequent neurodegeneration which are the major cascades of AD pathology (Balin et al., 1998; Lim et al., 2014; Tran et al., 2022). A series of investigations found different kinds of spirochetes in human AD brain tissues, possibly revealing a strong connection of these microbes with AD pathogenesis as well (Miklossy, 1993, 2011, 2015). Borrelia burgdorferi, the causative agent of Lyme disease, was detected in AD brains, as a co-localized form with amyloid markers (MacDonald and Miranda, 1987; Senejani et al., 2021). Porphyromonas gingivalis, a pathogen for chronic periodontitis, was also observed in the brain tissues of AD patients (Dominy et al., 2019). Different oral Treponema species have been observed in the trigeminal ganglia, pons, and hippocampus (Riviere et al., 2002). Helicobacter pylori (Hp) IgG titer was higher in AD patients compared with control patients (Malaguarnera et al., 2004), and epidemiological studies suggested that it is involved in the activation of innate immunity pathways that can exacerbate the CNS system leading to AD-like complications (Park and Tsunoda, 2022). Other studies also show a positive relation between Hp infection and AD (Park et al., 2017; Liu et al., 2021). Toxoplasma gondii, a eukaryotic pathogen, has been suggested to be involved in AD (Kusbeci et al., 2011; Mahami-Oskouei et al., 2016; Perry et al., 2016). Other microbes that were detected in the AD brains are Human herpes virus-6, Varicella zoster virus, HCV, and Influenza virus. Additionally, various fungal (e.g., Candida glabrata, Candida famata, Syncephalastrum racemosum, and Candida albicans) pathogens have also been observed in AD brains (Alonso et al., 2014; Pisa et al., 2015). Overall, viruses, bacteria, and fungi, as well as single-cell eukaryotes have been detected in human AD brains suggesting that AD is possibly manifested by multiple microbial infections in the body/brain. Highlighting this, one study found that vaccination for tetanus, diphtheria, and pertussis reduced the risk of dementia in adults (Scherrer et al., 2021). Furthermore, there is ample evidence that the BCG vaccine may provide a protective effect on AD development (Zuo et al., 2017; Gofrit et al., 2019; Greenblatt and Lathe, 2024).
Cp infection and AD pathogenesis
Among the bacterial infections detected in the brain of AD patients, Cp is the most consistent and promising candidate for involvement in AD (Balin et al., 1998; Paradowski et al., 2007). Cp is a gram-negative obligate intracellular bacterium that causes upper and lower respiratory tract infection and is a major contributor to community-acquired pneumonia (Miyashita et al., 2001). Cp has a unique lifecycle which includes a metabolically inactive elementary body that enters the cell forming a compartment termed an inclusion. The inactive elementary body then changes to the metabolically active reticulate body which then replicates, eventually changing back to the elementary body which is released from the cell-by-cell lysis. Chlamydiae all possess type III secretion systems which are required for host cell manipulation and immune system evasion (Peters et al., 2007). Under immune or antibiotic pressure, Cp can transform into a more persistent state, hiding inside the cell until conditions improve (Malinverni et al., 1995). However, little research has been performed in this area. Cp has been suggested to play a key role in several aging-related conditions including AD (Figure 1) (Kuo et al., 1995; Porritt and Crother, 2016).
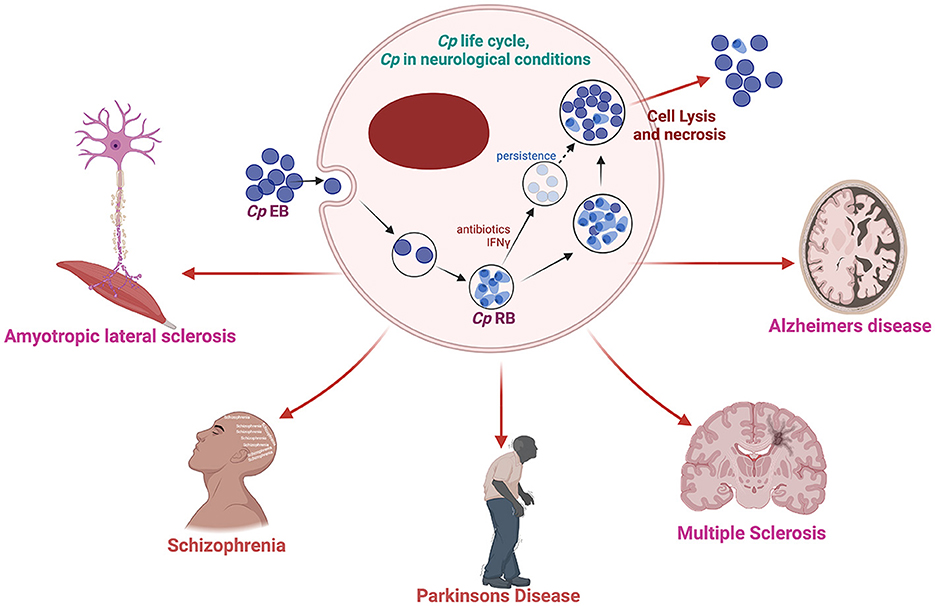
Figure 1. Schematic diagram of Cp life cycle and its possible involvement in several neurological complications. Created with BioRender.com.
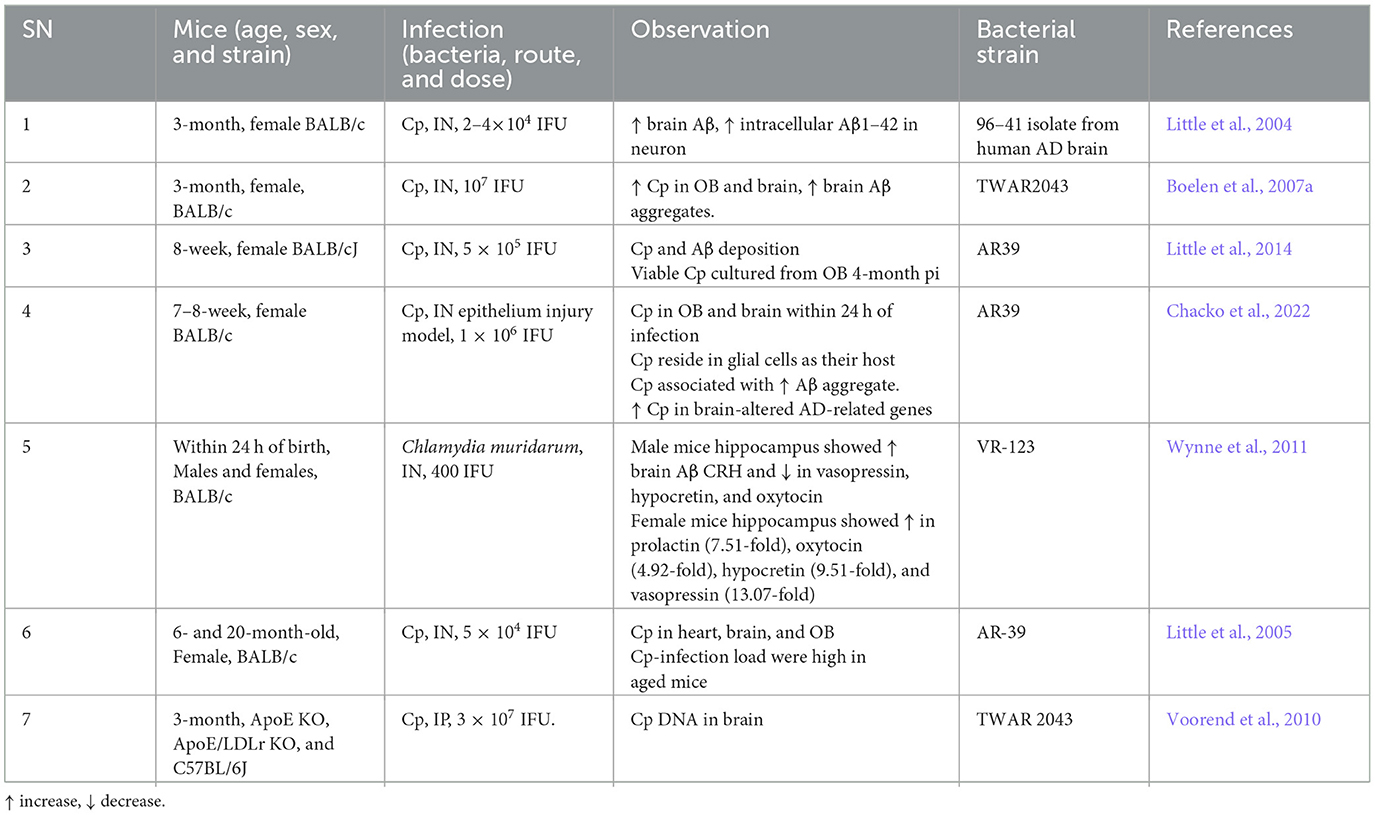
Brian Balin and Alan Hudson first observed Cp in AD brains (Balin et al., 1998) and similarly, other studies also reported the presence and increased expression of Cp-associated markers in AD brains correlating with neuropathology, suggesting a causal link (Chiara et al., 2012; Balin and Hudson, 2014). Cp was observed in close association with both amyloid plaques as well as NFTs (Balin et al., 1998; Gérard et al., 2006; Hammond et al., 2010). Immunohistological studies placed Cp within neurons, microglia, astrocytes, and pericytes (Balin et al., 1998; Gérard et al., 2006; Hammond et al., 2010). In some cases, live Cp was grown from AD patient brain tissue samples by infecting Hep2 cells using the postmortem AD brain tissue lysate (Balin et al., 1998; Gérard et al., 2006; Dreses-Werringloer et al., 2009). An ultrastructural analysis of Cp in AD brains determined that the Cp did not appear as classical inclusions but may have been in a more persistent state (Arking et al., 1999). Additionally, various studies have observed increased anti-Cp titers in AD patients compared with normal controls (Yamamoto et al., 2005; Bu et al., 2015). Meta-analyses of the various studies seeking to determine relationships between infectious organisms and AD have placed Cp as the likeliest organism to play a role in AD pathogenesis (Maheshwari and Eslick, 2015; Ou et al., 2020). As depicted in Table 1, a growing body of studies found Cp as a potential contributor to AD pathogenesis.
While many studies observed the presence of Cp in human AD brains, some studies failed to identify its presence (Nochlin et al., 1999; Gieffers et al., 2000; Ring and Lyons, 2000; Taylor et al., 2002; Wozniak et al., 2003). However, this is understandable as even if Cp does play a role in AD pathogenesis, it is unlikely to be a universal mechanism, as other infections, environmental, and genetic factors will play important roles. Additionally, differences in methodologies could also explain the lack of Cp detection. For example, Paradowski et al. analyzed the level of soluble Aβ in AD patients with Cp infection but were not able to find any difference between AD patients that were Cp-positive and Cp-negative (Paradowski et al., 2007). Several factors including sample location bias, amount of DNA loaded, sample size, etc., might contribute to variation in the results. Overall, there are several limitations in the methods utilized (culture, PCR, IHC, EM etc.) to interrogate the presence of Cp in the human brain, including location bias relative to the large sample size. Some studies used fresh tissue and other used paraffin-embedded tissue. Paraffin embedded tissues, which are generally more common, provide a more difficult route for experimental detection of Cp. Nonetheless, the majority of studies did find a correlation between Cp infection and Aβ oligomers in AD patients (Table 1).
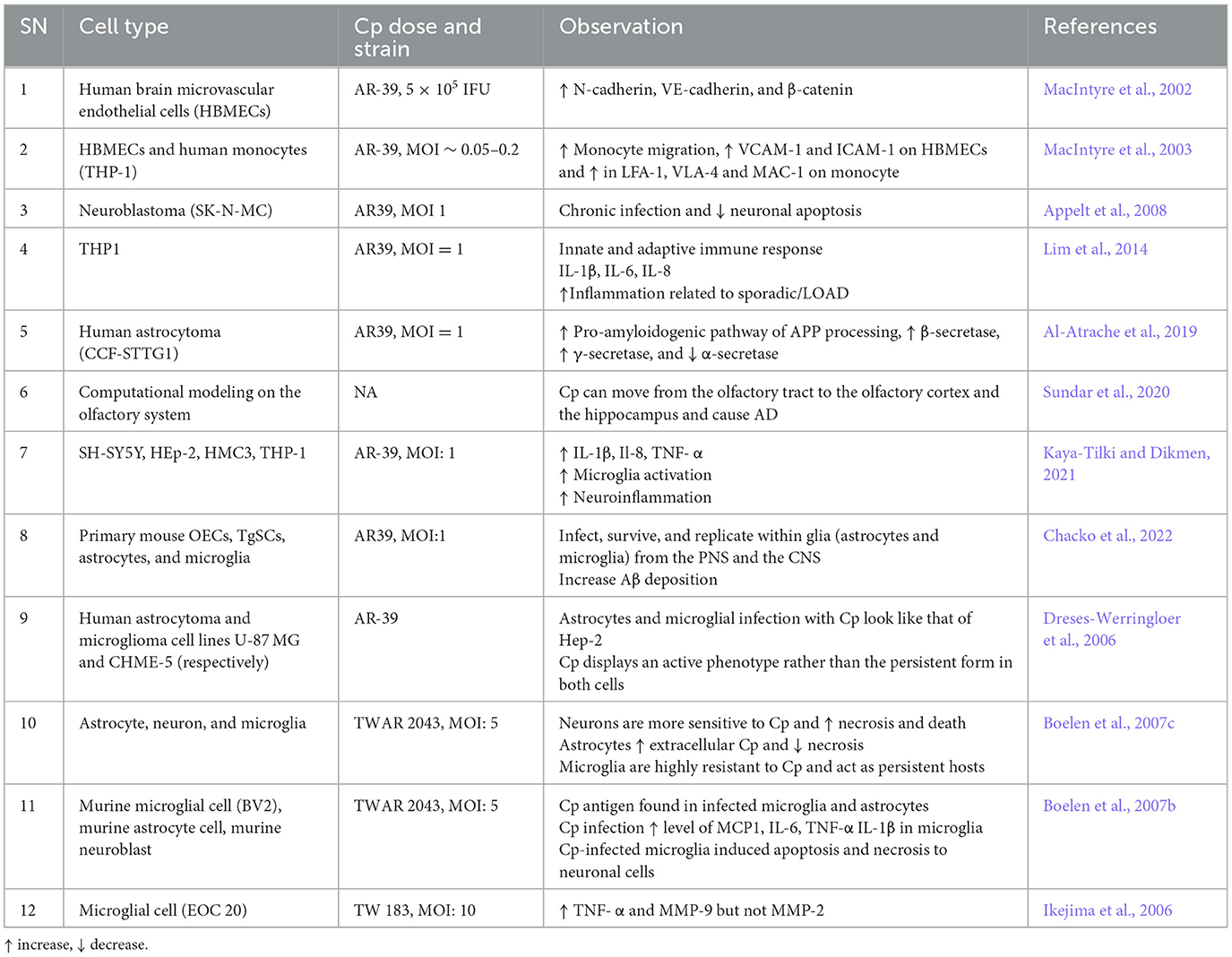
Besides human studies, several in vivo (Table 2) AD mouse model studies obtained evidence for a role of Cp in AD pathogenesis. Most of the in vivo experimental studies employ intranasal administration of Cp to investigate the role of Cp infection in AD-related pathologies in mice. Intranasal Cp infection in mice leads to increased Cp invasion of the brain and accelerated Aβ accumulation in the brain (Little et al., 2004, 2005, 2014; Boelen et al., 2007a; Voorend et al., 2010; Chacko et al., 2022). In addition, a few studies suggested neuronal accumulation of Aβ following Cp infection and glial cells as the host for Cp in the CNS (Little et al., 2004; Chacko et al., 2022). These studies from AD animal models found that intranasal Cp infection can reach different parts of the brain inducing increased amyloid deposition, glial activation, and triggered neuroinflammatory cascades which all could lead to the aggravation of AD pathogenesis. However, to date, no actual cognitive studies have been performed using an AD transgenic model with Cp infection.
In addition to mouse models and post-mortem human tissue samples, many in vitro studies have been performed which support a relationship between Cp and AD (Table 3). Cp infection may lead to increased BBB permeability and inflammatory responses of immune cells to trigger disease pathologies, thereby exacerbating AD. For instance, Cp infection to Human brain microvascular endothelial cells (HBMECs) increased the level of N-cadherin, VE-cadherin, β-catenin, VCAM, and ICAM (MacIntyre et al., 2002, 2003). Monocytes infected by Cp in vitro increased their migration and amounts of adhesive molecules like LFA-1, VLA-4, and MAC-1 (MacIntyre et al., 2003). These molecules are associated with the infiltration of peripheral immune cells into the brain. Additionally, Cp infection of THP1 cells led to increased production of IL-1β, IL-6, and IL-8, which have been closely associated with sporadic/LOAD) (Lim et al., 2014). Human astrocytoma infected with Cp led to the activation of the pro-amyloidogenic pathway by enhancing APP processing, increased the activity of β-secretase and γ-secretase, and reduced α-secretase, which eventually increased Aβ production by cleaving APP (Al-Atrache et al., 2019).
Possible mechanisms by which Cp infection affects the brain
There are likely three major routes by which Cp can affect brain biology. (1) The first route is via systemic interactions. Acute infection of the lung can lead to systemic cytokine production, which in the context of the right inflammatory signals, can pass into the brain directly. Additionally, elementary bodies released from infected cells can pass into the circulatory system leading to infections in other tissues and organs. (2) The second route for Cp migration is via a Trojan horse. As an obligate intracellular pathogen, Cp resides in cells, including immune cells such as monocytes/macrophages, T-cells, and neutrophils, and infected cells can migrate to other parts of the body, including the brain, thereby spreading the infection (Kortesoja et al., 2020). Cp-infected neutrophil granulocytes can also be taken up by macrophages silently helping Cp for its replication and hindrance against the immune challenge from the human host (Rupp et al., 2009). These macrophages can migrate to the brain, eventually releasing Cp there. Additionally, while infiltrating macrophages/monocytes can either exacerbate disease via inflammatory pathways, or aid in Aβ clearance (Butovsky et al., 2006; Koronyo-Hamaoui et al., 2009, 2019; Koronyo et al., 2015; Li et al., 2020), infected macrophages are likely to only promote disease pathology. (3) The third route is a direct infection of the nasal cavity. Cp has been shown to directly infect the brains of mice via the nasal route and damage to these areas can lead to enhanced delivery of Cp into the brain (Chacko et al., 2022). The trigeminal nerve extends between the olfactory bulb and the brain and acts as an invasion path through which Cp can readily pass and reach the CNS (Chacko et al., 2022). Cp that reaches the brain can infect the brain's resident cells and trigger proinflammatory signals through activated glial cells (microglia and astrocytes) resulting in secondary brain damage by inducing neuroinflammation. None of these three mechanisms are mutually exclusive and likely all play some role to varying degrees.
Role of Cp in resident brain cells
Under homeostatic conditions resident brain cells work in concert to maintain the optimal CNS environment. However, under neurological disease conditions, such as AD, brain-resident glial cells are activated resulting in a neuroinflammatory milieu, which can subsequently lead to neurodegeneration. Previous studies suggest that Cp can infect both glial and neuronal cells in the brain. Cp can infect and survive in cultured mouse primary microglia, astrocytes, and OECs (Chacko et al., 2022). Furthermore, Cp could also replicate in peripheral (OECs and TgSCs) and well as CNS glial cell types from the peripheral nervous system and the CNS (astrocytes and microglia) (Chacko et al., 2022). Similarly, Cp can infect and reproduce in human microglia and astrocytes in vitro, as well as neuronal cells (Dreses-Werringloer et al., 2006; Boelen et al., 2007c). Neuronal cells were the most permissive to Cp infection, while microglial cells were less but still contained Cp DNA, suggesting these cells may induce a more persistent Cp phenotype (Boelen et al., 2007c). Here, we discuss in greater detail the potential role of Cp in these individual CNS cell types.
Glia (microglia and astrocytes)
A study performed in post-mortem human brain observed the presence of Cp antigens inside both microglia and astrocytes (Balin et al., 1998; Arking et al., 1999; Gérard et al., 2006; Hammond et al., 2010). As an immune cell and a professional macrophage, microglial cells are known to be more resistant to infection, including Cp infection. However, Cp DNA has been found in microglia exposed to Cp (Boelen et al., 2007c). Cp infection in murine microglia and astrocytes both led to an increase in the level of several inflammatory cytokines and chemokines in the culture medium. Cp-infected microglia released a greater amount of TNF-α and IL-1β while Cp-infected astrocytes released a greater amount of monocyte chemoattractant protein 1 (MCP-1), and IL-6 compared with controls (Boelen et al., 2007c). Treatment of neuronal cells with Cp-infected microglial and astrocyte-conditioned media resulted in neurotoxicity in vitro (Boelen et al., 2007b). Microglial supernatant was found to be more neurotoxic than astrocyte supernatant following Cp infection, indicating that microglia can produce neurotoxic factors following Cp infection (Boelen et al., 2007b). Similarly, Cp infection of EOC microglial cells also led to the production of proinflammatory cytokines like TNF-alpha and selectively induced matrix metalloproteinase-9 (MMP9) (Ikejima et al., 2006). Cp-induced microglial activation in the hippocampus was observed in a mouse model of atherosclerosis where Cp was injected i.p. (Voorend et al., 2010). These data support the idea that in addition to the direct nasal route, Cp can access the brain via the BBB likely residing in trafficking immune cells. Previous reports suggested that many toll-like receptors, including TLR1, 2 4, and 6 are expressed in microglia (Laflamme and Rivest, 2001; Laflamme et al., 2001; Bsibsi et al., 2002). These receptors are reported to be involved in the Cp-mediated inflammatory cascades in a MYD88-dependent manner (Porritt and Crother, 2016). Cp elementary bodies can activate these receptors to accelerate the downstream inflammatory pathways through MYD88 resulting in the production of many inflammatory mediators and cytokines including IFN-γ, IL-1α, IL-18, IL-1β, TNF-α, IL-6, IL-12, MCP-1, CXCL1, ICAM1, VCAM-1, E-Selectin, etc. (Porritt and Crother, 2016). These findings suggest that microglia infected with Cp can drive inflammatory responses in the brain (Figure 2).
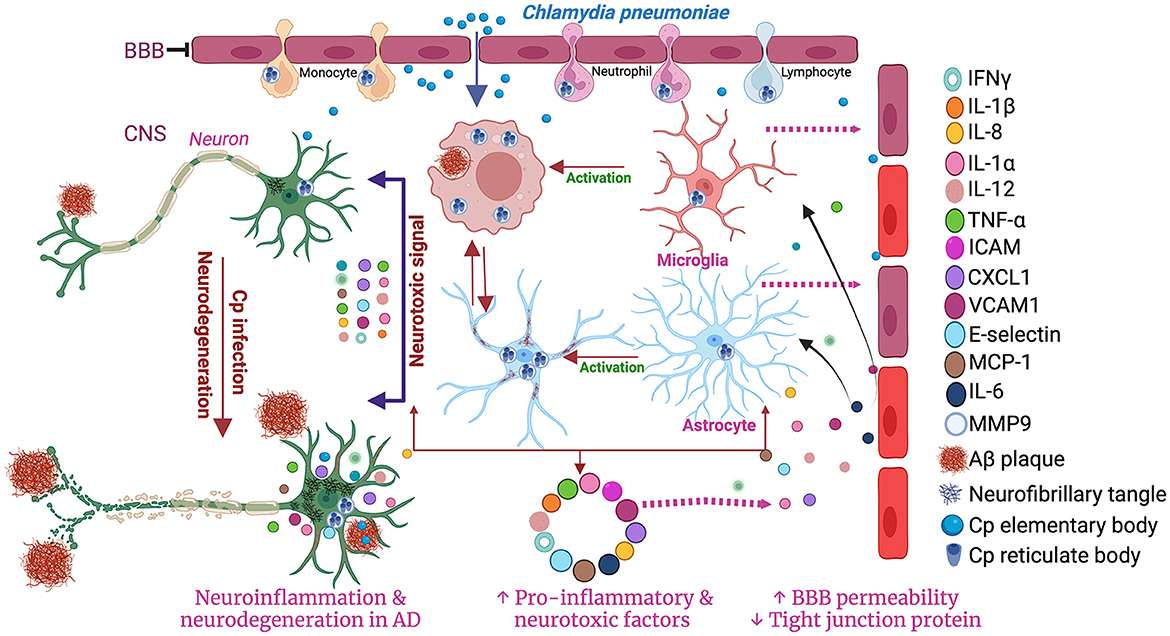
Figure 2. Schematic diagram of the involvement of microglia and astrocytes in Cp-mediated neuroinflammation. Created with BioRender.com.
While less is known about Cp and astrocytes, in vitro studies have found that Cp grow better in astrocytes than in microglia (Boelen et al., 2007b). Astrocytes provide lipids for neurons myelin sheaths which may make these cells more hospitable for Cp growth. Cp-infected astrocytes have altered expression and activity of secretases which are critical to β-amyloid generation in AD (Al-Atrache et al., 2019). Cp infection induced the pro-amyloidogenic pathway of APP processing via increasing the expression and activity of β-secretase and γ-secretase and decreasing the expression and activity of α-secretase. These effects of Cp infection in astrocytes provide evidence for a direct link between Cp and AD pathology (Al-Atrache et al., 2019). Therefore, further studies need to be performed investigating the relationships between Cp infection and astrocytes.
Neurons
Contini et al. suggested that neuronal cells act as the host for the Cp in AD brains and infected neuronal cells were observed to be near NFT and NSP in AD brains (Contini et al., 2010; Balin et al., 2018). In one study, intracellular Cp was observed in almost 1% of the brain cells including neuroglia, different kinds of neurons, and peri-vascular cells (Hammond et al., 2010). Though the presence of Cp was observed in the neuron of humans as well as an animal model of AD its role in the neuron has yet to be explored. In in vitro studies, neuronal cells were most permissive to Cp infection. As mentioned above, supernatants from Cp-infected microglia were found to be neurotoxic (Boelen et al., 2007b). A few studies, however, reported that direct Cp infection can lead to the inhibition of apoptosis in neuronal cells (Appelt et al., 2008). Nonetheless, Cp-infected immune cells can promote neurodegeneration by increasing the production of inflammatory mediators (Boelen et al., 2007b).
Oligodendrocytes and endothelial cells
Unfortunately, no studies to date have been performed investigating Cp infection and oligodendrocytes. However, it is likely that these cells are permissive to some degree for Cp infection. While there is no data regarding Cp and brain endothelial cells, there is large a set of data which support an association between Cp infection and atherosclerosis (Grayston et al., 1995; Belland et al., 2004; Campbell and Kuo, 2004). Growing evidence suggests that in endothelial cells Cp can induce oxidative stress in mitochondria, downregulate MMP3/MMP9 synthesis, and increase TLR2 signaling (Pietro et al., 2013; Wang et al., 2013; Ma et al., 2015; Zhao et al., 2022). These biological events promote the migration of vascular smooth muscle cells to promote atherosclerotic plaque. Cp infection in human brain microvascular endothelial cells induced the expression of adhesion-related proteins, zonula adherent proteins, and reduction of blood-brain barrier tight junction proteins including beta-catenin, N-cadherin, and VE-cadherin, suggesting that Cp can induce the permeability of endothelial cells and possibly leads to BBB permeability (MacIntyre et al., 2002).
Chlamydia pneumoniae in AD: causative or accelerating agent?
The role Cp may play in AD pathogenesis is still a topic of discussion and requires further studies. Among the studies that observed Cp in the AD brain, most found Cp in the brain of late-stage AD patients. Based on emerging research findings, scientists suggest that a persistent form of Cp might play an important role in the induction and progression of AD by increasing the formation and accumulation of senile plaque and NFTs and by increasing neuroinflammation (Balin et al., 1998). Indeed, some studies revealed that Cp can increase the production of Aβ both in animal models and in vitro cultures (Little et al., 2004; Boelen et al., 2007a; Al-Atrache et al., 2019; Chacko et al., 2022). This increase in amyloid production in response to infection supports the antimicrobial protection hypothesis of AD (Soscia et al., 2010; Kagan et al., 2012; Kumar et al., 2016; Moir et al., 2018) Aβ has been found to play a role as an antimicrobial peptide acting as a trap for microbes. However, the multiplication of microbes and the availability of the immune system to defend against these conditions and to drain the complex of Aβ with trapped microbes are limited. Rather, such a condition may lead to the accumulation of Aβ and activation of inflammatory cells leading to aggravated neuroinflammation. This neuroinflammation could cause secondary damage to neurons and other immune cells present in the CNS besides the infection itself. Further, as mentioned above, conditioned media from Cp-infected microglia cell lines possessed neurotoxicity (Boelen et al., 2007b).
As mentioned before, one likely route for Cp to gain access to the brain is through the nasal cavity (Chacko et al., 2022). Once there, it can increase AD-like pathology in the brain characterized by increased Aβ, Tau, and neurodegeneration (Boelen et al., 2007a). Intranasal infection with Cp resulted in the formation of amyloid-like deposits in the brain of the non-AD mouse models. However, Cp infection in the AD brain was correlated with lower CSF AB42 levels and higher CSF tau protein suggesting that the presence of Cp might differentially regulate the Aβ42 and tau in AD brains (Paradowski et al., 2007). Aβ1–42 immunoreactive deposits were observed in brains of Cp-infected mice. The extent of infection was proportionate to the number, size, and density of Aβ1–42 deposits in the infected brains (Little et al., 2004). These findings reaffirm that Cp-mediated infection may be involved in the pathogenesis of sporadic AD. Altogether, these data indicate that Cp can reach and infect cells in the brains of both mice and humans and provide evidence to support the hypothesis that Cp can play a role in AD pathogenesis. This is likely mediated directly by neuronal cell infection and cell death and indirectly by overactivation of glial cells and their responsive inflammatory cascade-mediated neuroinflammation. All these events are capable enough to affect the hippocampus as well as to induce AD-like pathology in the long run. Additionally, this would also open the possibility that CP can initiate as well as aggravate AD.
Potential mechanistic pathways where Cp can affect AD development and progression
APP/Aβ
The presence of Aβ was the first major discovery into the mechanisms of AD pathogenesis. Amyloid precursor protein (APP) is responsible for the production of neurotoxic Aβ via proteolysis which results in AD pathology (O'Brien and Wong, 2011). Accumulation of Ab leads to plaque formation, which is believed to initiate the conversion of tau from the normal to toxic form, which mediates the Aβ toxicity at the synapse (Bloom, 2014). Excessive APP accumulation in/near mitochondrial import channels results in mitochondrial dysfunction, which is a key event of AD pathology (Devi et al., 2006). In contrast, Aβ peptide possesses antimicrobial properties as well and is expressed in response to infections, a key component of the anti-microbial hypothesis of AD (Soscia et al., 2010; Kagan et al., 2012; Kumar et al., 2016; Moir et al., 2018) Cp infection in human astrocytes dramatically increases APP suggesting that Cp infection can trigger APP production and subsequent AD pathology (Al-Atrache et al., 2019). Cp infection can induce Aβ in the brains of mice (Little et al., 2004; Chacko et al., 2022). Several studies identified the presence of Cp in the brain tissue of AD patients and induce the increased production of Aβ plaque and AD-like conditions (Yamamoto et al., 2005; Gérard et al., 2006; Paradowski et al., 2007; Chacko et al., 2022). It is unknown if Aβ plays any role controlling Cp infection, but it is likely that Cp could influence AD development by inducing excessive Ab production.
APOE4
The apolipoprotein E (APOE) ε4 allele (APOE4) is a leading risk determinant for AD with a host of detrimental interactions (Yamazaki et al., 2019; Raulin et al., 2022), the APOE4 derived from astrocytes are reported to be responsible for the alteration of BBB integrity during AD pathology (Jackson et al., 2021). APOE4 has been associated with an increased burden of Cp in human AD brains (Gérard et al., 2005). Additionally, the presence of APOE4 enhances the attachment of Cp elementary bodies to human cells (Gérard et al., 2008). Another study observed that Cp infection was associated with worse memory among APOE ε4 carriers, but not among non–ε4 carriers (Zhao et al., 2020). APOE4 has been shown to increase the accumulation of lipid droplets in microglial cells and negatively affect cholesterol efflux (Leeuw et al., 2022; Victor et al., 2022). Cp requires host-derived lipids for growth and limits cholesterol efflux in macrophages (Ooij et al., 2000; Tumurkhuu et al., 2018). As APOE4 is highly expressed in astrocytes, this may allow Cp to grow more readily in the astrocytes of APOE4 carriers (Tcw et al., 2022). Thus, the presence of APOE4 may facilitate enhanced Cp growth beyond the increased attachment to cells observed in vitro.
SIRT 1
Sirtuin 1 is known to be beneficial to multiple age-related neurological disorders including AD. SIRT1 plays a critical role at the nexus between many cellular pathways including metabolism, energy production, and stress responses (Liu et al., 2008). Low levels of SIRT1 are associated with accumulation of Aβ and tau in the brain cerebral cortex of AD patients and SIRT1 has become a target for therapeutic efforts (Julien et al., 2009; Liu et al., 2022). Upon infection by Cp, the SIRT1 pathway is reduced in macrophages (Taavitsainen-Wahlroos et al., 2022). Resveratrol, the potent SIRT1 inducer, inhibits the growth of the closely related organism, Chlamydia trachomatis (Petyaev et al., 2017). Levels of SIRT1 is reported to be downregulated in AD conditions. Chen et al. observed that SIRT1 protects against microglia-dependent Aβ toxicity by inhibiting NF-κB signaling (Chen et al., 2005). Supportively, another group suggested that SIRT1 activation in neurons controls calorie restriction which can prevent amyloid neuropathy and AD (Qin et al., 2006). Kim et al. also found a neuroprotective effect of SIRT1 against AD (Kim et al., 2007). Low levels of SIRT1 was associated with a higher amount of accumulated tau in AD (Julien et al., 2009). SIRT1 deletion resulted in increased acetylated-tau phosphorylated-tau, possibly by blocking proteolysis (Min et al., 2010). SIRT1 inhibition is also reported to be associated with a higher bacterial load during infection (Hajra et al., 2023). Additionally, SIRT1 activation leads to increased cholesterol efflux via upregulation of ABCA1 (Feng et al., 2018). Overall, the sirtuin-1 pathway appears to act against both infectious processes as well as AD pathogenesis and its downregulation may be a key step in infection exacerbated AD.
MMP-9
MMP-9 is a critical pathogenic mediator in several CNS disorders, including AD (Gu et al., 2020). MMP9 is associated with Aβ transport and abnormal tau cleavages thus facilitating the brain Aβ accumulation and tau oligomerization (Nübling et al., 2012; Shackleton et al., 2019; Hernandes-Alejandro et al., 2020). The plasma level of MMP-9 is increased in AD patients, and its expression is elevated in postmortem AD brain tissues (Lorenzl et al., 2003). MMP-9 plays an important role in C. trachomatis-induced trachoma (Natividad et al., 2006). Cp Heat Shock Protein 60 regulates the expression of MMP-9 expression in macrophages (Kol et al., 1998). Microglial cells infected with Cp express MMP-9 and TNF-α which can further trigger the production of inflammatory cytokines (Ikejima et al., 2006). Upregulation of extracellular MMP inducers and gelatinases were reported in human atherosclerotic patients infected with Cp (Choi et al., 2002) and MMP-9 expression is associated with the presence of Cp in human coronary atherosclerotic plaques (Arno et al., 2005). Collectively, Cp infection induced the production of MMP-9 which subsequently triggered inflammatory cascades in AD.
NLRP3 inflammasome
The nucleotide binding domain and leucine-rich repeat (NLR) pyrin domain containing three (NLRP3) inflammasome is a major driver of diverse inflammatory cascades in CNS diseases, including AD (Shao et al., 2018; Jha et al., 2023). Activation of the NLRP3 inflammasome generally requires two distinct steps: (1) NF-κB signaling to upregulate inflammasome components, and (2) activation and formation of the inflammasome via danger signals (Evavold and Kagan, 2019). The formation of the NLRP3 inflammasome leads to caspase-1 activation which can then cleave pro-IL-1β and pro-IL-18 into their active mature forms and subsequently secreted. In AD, NLRP3 inflammasome signaling is triggered by multiple factors such as Aβ, tau, activated glial cells, impaired autophagy, and endoplasmic reticulum stress that ultimately leads to the release of pro-inflammatory cytokines such as IL-1β and IL-18 (Jha et al., 2023). These inflammatory molecules are associated with neuroinflammation, neurodegeneration, and cognitive deficit in AD (Liang et al., 2022). Most pathogens promote NLRP3 inflammasome activation by increasing NF-κB downstream signaling and phagolysosome impairment and or mitochondrial dysfunction (Anand et al., 2011). Cp can directly activate the NLRP3/ASC inflammasome in Cp-infected bone marrow-derived macrophages, leading to the release of biologically active IL-1β (He et al., 2010; Itoh et al., 2014) Caspase-1 and IL-1β are required to control acute pulmonary Cp infection in mice, and modulation of inflammasome activity via autophagy can lead to increased pathology (Shimada et al., 2011; Crother et al., 2019). While there is lack of studies examining the role of Cp in NLRP3 inflammasome signaling in AD, Cp triggering inflammasome activation in other disease/cell types indicate that Cp infection may promote NLRP3 mediated inflammatory responses in AD as well.
Other biomolecules
In addition to those listed above, there are several other biomolecules which may provide a link between Cp infection and AD pathologies. These include: TREM 2 (N'Diaye et al., 2009; Gratuze et al., 2018), Lipoprotein receptor-related protein 1 (Liu et al., 2017; Al-Atrache et al., 2019), Homocysteine inducible ER protein with ubiquitin like domain (Li et al., 2021; Wen et al., 2021), inducible nitric oxide synthases (Igietseme et al., 1998; Shao et al., 2010; Kummer et al., 2011), MAP1B (Al-Younes et al., 2011; Ma et al., 2014), Midkine (Muramatsu, 2011; Sanino et al., 2020), aldolase A (Ende and Derré, 2020; Li et al., 2021), gamma-actin 1 (Bagnicka et al., 2021; Li et al., 2021), DDX5 (Li et al., 2021; Hu et al., 2022), Osteopontin (Filippis et al., 2017; Rentsendorj et al., 2018; Kessler et al., 2019), and ICAM-1 (Igietseme et al., 1999; Vielma et al., 2003). However, their direct relationship with Cp infection in AD is yet to be established.
Conclusion
Accumulation of abnormal Aβ and tau forms, alongside neurodegeneration are the defining events making up AD pathogenesis (Jack et al., 2016, 2018). These events are further underpinned by other adjunctive ones, such as inflammation, vascular dysfunction, synaptic loss, and functional impairments. The exact cause of late-onset AD is still not understood but it is likely multifactorial. The concept that infections are a key element in AD has gained support recently. The data presented here in this review article provides strong correlative support for Cp playing a contributory role in AD. However, further investigations need to be performed in both human and animal studies to understand what role Cp infection might actually play and by what mechanism Cp induces or exacerbates AD. The optimization of experimental protocols and methods of testing will also help to better understand and confirm the presence of Cp in the AD brain. In addition to the standardized retrospective studies, we should also perform multi-center studies with increased cohorts. Controlled animal studies using AD transgenic models with Cp infection will provide valuable insight into this question. In addition to Cp infection, several other studies confirmed the presence of different microbes in AD and other dementia types, hence multi-pathogen studies including both viruses and bacteria should also be taken into consideration in future studies. The gap in finding appropriate therapeutic approaches against AD can be resolved with the utilization of specific therapy or preventative measures against the responsible microbes in AD pathogenesis. Overall, among the various microbes that have been studied to have a role in AD, Cp appears to be the most plausible agent therefore warranting further studies.
Author contributions
LS: Conceptualization, Writing – original draft, Writing – review & editing. BG: Conceptualization, Writing – review & editing. YK: Conceptualization, Writing – review & editing. MK-H: Conceptualization, Writing – original draft, Writing – review & editing. TC: Writing – original draft, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported by National Institutes of Health (NIH) grants R01AG075998 to MK-H and TC, R01AG055865 to MK-H, and Alzheimer's Association grant AARG-NTF-21-846586 to TC.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
2023 Alzheimer's disease facts and figures (2023). Alzheimers Dement. 19, 1598–1695. doi: 10.1002/alz.13016
Al-Atrache, Z., Lopez, D. B., Hingley, S. T., and Appelt, D. M. (2019). Astrocytes infected with Chlamydia pneumoniae demonstrate altered expression and activity of secretases involved in the generation of β-amyloid found in Alzheimer disease. BMC Neurosci. 20:6. doi: 10.1186/s12868-019-0489-5
Alonso, R., Pisa, D., Marina, A. I., Morato, E., Rábano, A., and Carrasco, L. (2014). Fungal infection in patients with Alzheimer's disease. J. Alzheimers Dis. 41, 301–311. doi: 10.3233/JAD-132681
Al-Younes, H. M., Al-Zeer, M. A., Khalil, H., Gussmann, J., Karlas, A., Machuy, N., et al. (2011). Autophagy-independent function of MAP-LC3 during intracellular propagation of Chlamydia trachomatis. Autophagy 7, 814–828. doi: 10.4161/auto.7.8.15597
Anand, P. K., Malireddi, R. K. S., and Kanneganti, T.-D. (2011). Role of the Nlrp3 inflammasome in microbial infection. Front. Microbiol. 2:12. doi: 10.3389/fmicb.2011.00012
Appelt, D. M., Roupas, M. R., Way, D. S., Bell, M. G., Albert, E. V., Hammond, C. J., et al. (2008). Inhibition of apoptosis in neuronal cells infected with Chlamydophila (Chlamydia) pneumoniae. BMC Neurosci. 9:13. doi: 10.1186/1471-2202-9-13
Arking, E. J., Appelt, D. M., Abrams, J. T., Kolbe, S., Hudson, A. P., and Balin, B. J. (1999). Ultrastructural analysis of Chlamydia pneumoniae in the Alzheimer's brain. Pathog. J. Mech. Dis. Process. 1, 201–211.
Arno, G., Kaski, J. C., Smith, D. A., Akiyu, J. P., Hughes, S. E., and Baboonian, C. (2005). Matrix metalloproteinase-9 expression is associated with the presence of Chlamydia pneumoniae in human coronary atherosclerotic plaques. Heart 91:521. doi: 10.1136/hrt.2003.012054
Ashton, N. J., Brum, W. S., Molfetta, G. D., Benedet, A. L., Arslan, B., Jonaitis, E., et al. (2024). Diagnostic accuracy of a plasma phosphorylated Tau 217 immunoassay for Alzheimer disease pathology. JAMA Neurol. 81, 255–263. doi: 10.1001/jamaneurol.2023.5319
Bagnicka, E., Kawecka-Grochocka, E., Pawlina-Tyszko, K., Zalewska, M., Kapusta, A., Kościuczuk, E., et al. (2021). MicroRNA expression profile in bovine mammary gland parenchyma infected by coagulase-positive or coagulase-negative staphylococci. Vet. Res. 52:41. doi: 10.1186/s13567-021-00912-2
Balin, B. J., Gérard, H. C., Arking, E. J., Appelt, D. M., Branigan, P. J., Abrams, J. T., et al. (1998). Identification and localization of Chlamydia pneumoniae in the Alzheimer's brain. Méd. Microbiol. Immunol. 187, 23–42. doi: 10.1007/s004300050071
Balin, B. J., Hammond, C. J., Little, C. S., Hingley, S. T., Al-Atrache, Z., Appelt, D. M., et al. (2018). Chlamydia pneumoniae: an etiologic agent for late-onset dementia. Front. Aging Neurosci. 10:302. doi: 10.3389/fnagi.2018.00302
Balin, B. J., and Hudson, A. P. (2014). Etiology and pathogenesis of late-onset Alzheimer's disease. Curr. Allergy Asthma Rep. 14:417. doi: 10.1007/s11882-013-0417-1
Barthélemy, N. R., Salvadó, G., Schindler, S. E., He, Y., Janelidze, S., Collij, L. E., et al. (2024). Highly accurate blood test for Alzheimer's disease is similar or superior to clinical cerebrospinal fluid tests. Nat. Med. 30, 1085–1095. doi: 10.1038/s41591-024-02869-z
Bateman, R. J., Xiong, C., Benzinger, T. L. S., Fagan, A. M., Goate, A., Fox, N. C., et al. (2012). Clinical and biomarker changes in dominantly inherited Alzheimer's disease. N. Engl. J. Med. 367, 795–804. doi: 10.1056/NEJMoa1202753
Belland, R. J., Ouellette, S. P., Gieffers, J., and Byrne, G. I. (2004). Chlamydia pneumoniae and atherosclerosis. Cell. Microbiol. 6, 117–127. doi: 10.1046/j.1462-5822.2003.00352.x
Bloom, G. S. (2014). Amyloid-β and tau: the trigger and bullet in Alzheimer disease pathogenesis. JAMA Neurol. 71, 505–508. doi: 10.1001/jamaneurol.2013.5847
Boelen, E., Stassen, F. R., van der Ven, A. J., Lemmens, M. A., Steinbusch, H. P., Bruggeman, C. A., et al. (2007a). Detection of amyloid beta aggregates in the brain of BALB/c mice after Chlamydia pneumoniae infection. Acta Neuropathol. 114, 255–261. doi: 10.1007/s00401-007-0252-3
Boelen, E., Steinbusch, H. W. M., Pronk, I., Grauls, G., Rennert, P., Bailly, V., et al. (2007b). Inflammatory responses following Chlamydia pneumoniae infection of glial cells. Eur. J. Neurosci. 25, 753–760. doi: 10.1111/j.1460-9568.2007.05339.x
Boelen, E., Steinbusch, H. W. M., Ven, A. J. A. M., van der, Grauls, G., Bruggeman, C. A., and Stassen, F. R. M. (2007c). Chlamydia pneumoniae infection of brain cells: an in vitro study. Neurobiol. Aging 28, 524–532. doi: 10.1016/j.neurobiolaging.2006.02.014
Boxer, A. L., and Sperling, R. (2023). Accelerating Alzheimer's therapeutic development: the past and future of clinical trials. Cell 186, 4757–4772. doi: 10.1016/j.cell.2023.09.023
Broxmeyer, L. (2017). Are the infectious roots of Alzheimer's buried deep in the past. Mol. Pathol. Epidemiol. 3, 1–19. doi: 10.5281/zenodo.894140
Bsibsi, M., Ravid, R., Gveric, D., and Noort, J. M. van (2002). Broad expression of toll-like receptors in the human central nervous system. J. Neuropathol. Exp. Neurol. 61, 1013–1021. doi: 10.1093/jnen/61.11.1013
Bu, X.-L., and Wang, Y.-J. (2021). Should infectious diseases be targeted to prevent dementias? Lancet Infect. Dis. 21, 1477–1478. doi: 10.1016/S1473-3099(21)00258-9
Bu, X. -L., Yao, X. -Q., Jiao, S. -S., Zeng, F., Liu, Y. -H., Xiang, Y., et al. (2015). A study on the association between infectious burden and Alzheimer's disease. Eur. J. Neurol. 22, 1519–1525. doi: 10.1111/ene.12477
Butovsky, O., Koronyo-Hamaoui, M., Kunis, G., Ophir, E., Landa, G., Cohen, H., et al. (2006). Glatiramer acetate fights against Alzheimer's disease by inducing dendritic-like microglia expressing insulin-like growth factor 1. Proc. Natl. Acad. Sci. 103, 11784–11789. doi: 10.1073/pnas.0604681103
Cai, Z., Hussain, M. D., and Yan, L.-J. (2014). Microglia, neuroinflammation, and beta-amyloid protein in Alzheimer's disease. Int. J. Neurosci. 124, 307–321. doi: 10.3109/00207454.2013.833510
Campbell, L. A., and Kuo, C. (2004). Chlamydia pneumoniae — an infectious risk factor for atherosclerosis? Nat. Rev. Microbiol. 2, 23–32. doi: 10.1038/nrmicro796
Chacko, A., Delbaz, A., Walkden, H., Basu, S., Armitage, C. W., Eindorf, T., et al. (2022). Chlamydia pneumoniae can infect the central nervous system via the olfactory and trigeminal nerves and contributes to Alzheimer's disease risk. Sci. Rep. 12:2759. doi: 10.1038/s41598-022-06749-9
Chen, J., Zhou, Y., Mueller-Steiner, S., Chen, L.-F., Kwon, H., Yi, S., et al. (2005). SIRT1 protects against microglia-dependent amyloid-β toxicity through inhibiting NF-κB signaling*. J. Biol. Chem. 280, 40364–40374. doi: 10.1074/jbc.M509329200
Chiara, G. D., Marcocci, M. E., Sgarbanti, R., Civitelli, L., Ripoli, C., Piacentini, R., et al. (2012). Infectious agents and neurodegeneration. Mol. Neurobiol. 46, 614–638. doi: 10.1007/s12035-012-8320-7
Choi, E. Y., Kim, D., Hong, B. K., Kwon, H. M., Song, Y. G., Byun, K. H., et al. (2002). Upregulation of extracellular matrix metalloproteinase inducer (EMMPRIN) and gelatinases in human atherosclerosis infected with Chlamydia pneumoniae: the potential role of Chlamydia pneumoniae infection in the progression of atherosclerosis. Exp. Mol. Med. 34, 391–400. doi: 10.1038/emm.2002.56
Ciaccio, M., Sasso, B. L., Scazzone, C., Gambino, C. M., Ciaccio, A. M., Bivona, G., et al. (2021). COVID-19 and Alzheimer's disease. Brain Sci. 11:305. doi: 10.3390/brainsci11030305
Contini, C., Seraceni, S., Cultrera, R., Castellazzi, M., Granieri, E., and Fainardi, E. (2010). Chlamydophila pneumoniae infection and its role in neurological disorders. Interdiscip. Perspect. Infect. Dis. 2010:273573. doi: 10.1155/2010/273573
Crother, T. R., Porritt, R. A., Dagvadorj, J., Tumurkhuu, G., Slepenkin, A. V., Peterson, E. M., et al. (2019). Autophagy limits inflammasome during Chlamydia pneumoniae infection. Front Immunol 10:754. doi: 10.3389/fimmu.2019.00754
Devi, L., Prabhu, B. M., Galati, D. F., Avadhani, N. G., and Anandatheerthavarada, H. K. (2006). Accumulation of amyloid precursor protein in the mitochondrial import channels of human Alzheimer's disease brain is associated with mitochondrial dysfunction. J. Neurosci. 26, 9057–9068. doi: 10.1523/JNEUROSCI.1469-06.2006
Dionisio-Santos, D. A., Olschowka, J. A., and O'Banion, M. K. (2019). Exploiting microglial and peripheral immune cell crosstalk to treat Alzheimer's disease. J. Neuroinflammation 16:74. doi: 10.1186/s12974-019-1453-0
Dominy, S. S., Lynch, C., Ermini, F., Benedyk, M., Marczyk, A., Konradi, A., et al. (2019). Porphyromonas gingivalis in Alzheimer's disease brains: evidence for disease causation and treatment with small-molecule inhibitors. Sci. Adv. 5:eaau3333. doi: 10.1126/sciadv.aau3333
Dreses-Werringloer, U., Bhuiyan, M., Zhao, Y., Gérard, H. C., Whittum-Hudson, J. A., and Hudson, A. P. (2009). Initial characterization of Chlamydophila (Chlamydia) pneumoniae cultured from the late-onset Alzheimer brain. Int. J. Méd. Microbiol. 299, 187–201. doi: 10.1016/j.ijmm.2008.07.002
Dreses-Werringloer, U., Gérard, H. C., Whittum-Hudson, J. A., and Hudson, A. P. (2006). Chlamydophila (Chlamydia) pneumoniae infection of human astrocytes and microglia in culture displays an active, rather than a persistent, phenotype. Am. J. Méd. Sci. 332, 168–174. doi: 10.1097/00000441-200610000-00003
Dyck, C. H., van Swanson, C. J., Aisen, P., Bateman, R. J., Chen, C., Gee, M., et al. (2022). Lecanemab in early Alzheimer's disease. N. Engl. J. Med. 388, 9–21. doi: 10.1056/NEJMoa2212948
Ende, R. J., and Derré, I. (2020). Host and bacterial glycolysis during Chlamydia trachomatis infection. Infect. Immun. 88:e00545-20. doi: 10.1128/IAI.00545-20
Evavold, C. L., and Kagan, J. C. (2019). Inflammasomes: threat-assessment organelles of the innate immune system. Immunity 51, 609–624. doi: 10.1016/j.immuni.2019.08.005
Feng, T., Liu, P., Wang, X., Luo, J., Zuo, X., Jiang, X., et al. (2018). SIRT1 activator E1231 protects from experimental atherosclerosis and lowers plasma cholesterol and triglycerides by enhancing ABCA1 expression. Atherosclerosis 274, 172–181. doi: 10.1016/j.atherosclerosis.2018.04.039
Filippis, A. D., Buommino, E., Domenico, M. D., Feola, A., Brunetti-Pierri, R., and Rizzo, A. (2017). Chlamydia trachomatis induces an upregulation of molecular biomarkers podoplanin, Wilms' tumour gene 1, osteopontin and inflammatory cytokines in human mesothelial cells. Microbiology 163, 654–663. doi: 10.1099/mic.0.000465
Fisher, O. (1907). Miliary necrosis with nodular proliferation of the neurofibrils: a common chnage of the cerebral cortex in senikle dementia. Monatsschr. Psychiatr. Neurol. 22, 361–372.
GBD 2019 Dementia Forecasting Collaborators (2022). Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health 7, e105–e125. doi: 10.1016/S2468-2667(21)00249-8
Gérard, H. C., Dreses-Werringloer, U., Wildt, K. S., Deka, S., Oszust, C., Balin, B. J., et al. (2006). Chlamydophila (Chlamydia) pneumoniae in the Alzheimer's brain. FEMS Immunol. Méd. Microbiol. 48, 355–366. doi: 10.1111/j.1574-695X.2006.00154.x
Gérard, H. C., Fomicheva, E., Whittum-Hudson, J. A., and Hudson, A. P. (2008). Apolipoprotein E4 enhances attachment of Chlamydophila (Chlamydia) pneumoniae elementary bodies to host cells. Microb. Pathog. 44, 279–285. doi: 10.1016/j.micpath.2007.10.002
Gérard, H. C., Wildt, K. L., Whittum-Hudson, J. A., Lai, Z., Ager, J., and Hudson, A. P. (2005). The load of Chlamydia pneumoniae in the Alzheimer's brain varies with APOE genotype. Microb. Pathog. 39, 19–26. doi: 10.1016/j.micpath.2005.05.002
Gieffers, J., Reusche, E., Solbach, W., and Maass, M. (2000). Failure to detect Chlamydia pneumoniae in brain sections of Alzheimer's disease patients. J. Clin. Microbiol. 38, 881–882. doi: 10.1128/JCM.38.2.881-882.2000
Gofrit, O. N., Klein, B. Y., Cohen, I. R., Ben-Hur, T., Greenblatt, C. L., and Bercovier, H. (2019). Bacillus Calmette-Guérin (BCG) therapy lowers the incidence of Alzheimer's disease in bladder cancer patients. PLOS ONE 14:e0224433. doi: 10.1371/journal.pone.0224433
Gordon, M. N., Heneka, M. T., Page, L. M. L., Limberger, C., Morgan, D., Tenner, A. J., et al. (2022). Impact of COVID-19 on the onset and progression of Alzheimer's disease and related dementias: a roadmap for future research. Alzheimers Dement. 18, 1038–1046. doi: 10.1002/alz.12488
Gratuze, M., Leyns, C. E. G., and Holtzman, D. M. (2018). New insights into the role of TREM2 in Alzheimer's disease. Mol. Neurodegener. 13:66. doi: 10.1186/s13024-018-0298-9
Grayston, J. T., Kuo, C., Coulson, A. S., Campbell, L. A., Lawrence, R. D., Lee, M. J., et al. (1995). Chlamydia pneumoniae (TWAR) in Atherosclerosis of the carotid artery. Circulation 92, 3397–3400. doi: 10.1161/01.CIR.92.12.3397
Greenblatt, C. L., and Lathe, R. (2024). Vaccines and dementia: part I. Non-specific immune boosting with BCG: history, ligands, and receptors. J. Alzheimers Dis. 98, 343–360. doi: 10.3233/JAD-231315
Grimaldi, A., Pediconi, N., Oieni, F., Pizzarelli, R., Rosito, M., Giubettini, M., et al. (2019). Neuroinflammatory processes, A1 astrocyte activation and protein aggregation in the retina of Alzheimer's disease patients, possible biomarkers for early diagnosis. Front. Neurosci. 13:925. doi: 10.3389/fnins.2019.00925
Grundke-Iqbal, I., Iqbal, K., Quinlan, M., Tung, Y. C., Zaidi, M. S., and Wisniewski, H. M. (1986). Microtubule-associated protein tau. A component of Alzheimer paired helical filaments. J. Biol. Chem. 261, 6084–6089. doi: 10.1016/S0021-9258(17)38495-8
Gu, D., Liu, F., Meng, M., Zhang, L., Gordon, M. L., Wang, Y., et al. (2020). Elevated matrix metalloproteinase-9 levels in neuronal extracellular vesicles in Alzheimer's disease. Ann. Clin. Transl. Neurol. 7, 1681–1691. doi: 10.1002/acn3.51155
Guzman-Martinez, L., Maccioni, R. B., Farías, G. A., Fuentes, P., and Navarrete, L. P. (2019). Biomarkers for Alzheimer's disease. Curr. Alzheimer Res. 16, 518–528. doi: 10.2174/1567205016666190517121140
Haan, J., den Morrema, T. H. J., Verbraak, F. D., Boer, J. F., de Scheltens, P., Rozemuller, A. J., et al. (2018). Amyloid-beta and phosphorylated tau in post-mortem Alzheimer's disease retinas. Acta Neuropathol. Commun. 6:147. doi: 10.1186/s40478-018-0650-x
Hajra, D., Rajmani, R. S., Chaudhary, A. D., Gupta, S. K., and Chakravortty, D. (2023). Salmonella-induced SIRT1 and SIRT3 are crucial for maintaining the metabolic switch in bacteria and host for successful pathogenesis. bioRxiv. doi: 10.1101/2022.11.21.517246
Hameed, S., Fuh, J.-L., Senanarong, V., Ebenezer, E. G. M., Looi, I., Dominguez, J. C., et al. (2020). Role of fluid biomarkers and PET imaging in early diagnosis and its clinical implication in the management of Alzheimer's disease. J. Alzheimers Dis. Rep. 4, 21–37. doi: 10.3233/ADR-190143
Hammond, C. J., Hallock, L. R., Howanski, R. J., Appelt, D. M., Little, C. S., and Balin, B. J. (2010). Immunohistological detection of Chlamydia pneumoniae in the Alzheimer's disease brain. BMC Neurosci. 11:121. doi: 10.1186/1471-2202-11-121
Hampel, H., Cummings, J., Blennow, K., Gao, P., Jack, C. R., and Vergallo, A. (2021). Developing the ATX(N) classification for use across the Alzheimer disease continuum. Nat. Rev. Neurol. 17, 580–589. doi: 10.1038/s41582-021-00520-w
He, X., Mekasha, S., Mavrogiorgos, N., Fitzgerald, K. A., Lien, E., and Ingalls, R. R. (2010). Inflammation and fibrosis during Chlamydia pneumoniae infection is regulated by IL-1 and the NLRP3/ASC inflammasome. J. Immunol. 184, 5743–5754. doi: 10.4049/jimmunol.0903937
Hernandes-Alejandro, M., Montaño, S., Harrington, C. R., Wischik, C. M., Salas-Casas, A., Cortes-Reynosa, P., et al. (2020). Analysis of the relationship between metalloprotease-9 and tau protein in Alzheimer's disease. J. Alzheimers Dis. 76, 553–569. doi: 10.3233/JAD-200146
Hu, M., Zheng, H., Wu, J., Sun, Y., Wang, T., and Chen, S. (2022). DDX5: an expectable treater for viral infection- a literature review. Ann. Transl. Med. 10, 712–712. doi: 10.21037/atm-22-2375
Igietseme, J. U., Ananaba, G. A., Bolier, J., Bowers, S., Moore, T., Belay, T., et al. (1999). The intercellular adhesion molecule type-1 is required for rapid activation of T helper type 1 lymphocytes that control early acute phase of genital chlamydial infection in mice. Immunology 98, 510–519. doi: 10.1046/j.1365-2567.1999.00926.x
Igietseme, J. U., Perry, L. L., Ananaba, G. A., Uriri, I. M., Ojior, O. O., Kumar, S. N., et al. (1998). Chlamydial infection in inducible nitric oxide synthase knockout mice. Infect. Immun. 66, 1282–1286. doi: 10.1128/IAI.66.4.1282-1286.1998
Ikejima, H., Friedman, H., and Yamamoto, Y. (2006). Chlamydia pneumoniae infection of microglial cells in vitro: a model of microbial infection for neurological disease. J. Méd. Microbiol. 55, 947–952. doi: 10.1099/jmm.0.46348-0
Itoh, R., Murakami, I., Chou, B., Ishii, K., Soejima, T., Suzuki, T., et al. (2014). Chlamydia pneumoniae harness host NLRP3 inflammasome-mediated caspase-1 activation for optimal intracellular growth in murine macrophages. Biochem. Biophys. Res. Commun. 452, 689–694. doi: 10.1016/j.bbrc.2014.08.128
Itzhaki, R. F. (2014). Herpes simplex virus type 1 and Alzheimer's disease: increasing evidence for a major role of the virus. Front. Aging Neurosci. 6:202. doi: 10.3389/fnagi.2014.00202
Itzhaki, R. F., Lin, W.-R., Shang, D., Wilcock, G. K., Faragher, B., and Jamieson, G. A. (1997). Herpes simplex virus type 1 in brain and risk of Alzheimer's disease. Lancet 349, 241–244. doi: 10.1016/S0140-6736(96)10149-5
Jack, C. R., Bennett, D. A., Blennow, K., Carrillo, M. C., Dunn, B., Haeberlein, S. B., et al. (2018). NIA-AA research framework: toward a biological definition of Alzheimer's disease. Alzheimers Dement. 14, 535–562. doi: 10.1016/j.jalz.2018.02.018
Jack, C. R., Bennett, D. A., Blennow, K., Carrillo, M. C., Feldman, H. H., Frisoni, G. B., et al. (2016). A/T/N: an unbiased descriptive classification scheme for Alzheimer disease biomarkers. Neurology 87, 539–547. doi: 10.1212/WNL.0000000000002923
Jackson, R. J., Meltzer, J. C., Nguyen, H., Commins, C., Bennett, R. E., Hudry, E., et al. (2021). APOE4 derived from astrocytes leads to blood–brain barrier impairment. Brain 145:awab478. doi: 10.1093/brain/awab478
Jha, D., Bakker, E. N. T. P., and Kumar, R. (2023). Mechanistic and therapeutic role of NLRP3 inflammasome in the pathogenesis of Alzheimer's disease. J. Neurochem. 1–25. doi: 10.1111/jnc.15788
Julien, C., Tremblay, C., Émond, V., Lebbadi, M., Salem, N., Bennett, D. A., et al. (2009). Sirtuin 1 reduction parallels the accumulation of tau in Alzheimer disease. J. Neuropathol. Exp. Neurol. 68, 48–58. doi: 10.1097/NEN.0b013e3181922348
Kagan, B. L., Jang, H., Capone, R., Arce, F. T., Ramachandran, S., Lal, R., et al. (2012). Antimicrobial properties of amyloid peptides. Mol. Pharm. 9, 708–717. doi: 10.1021/mp200419b
Kaya-Tilki, E., and Dikmen, M. (2021). Neuroprotective effects of some epigenetic modifying drugs' on Chlamydia pneumoniae-induced neuroinflammation: a novel model. PLoS ONE 16:e0260633. doi: 10.1371/journal.pone.0260633
Kessler, M., Hoffmann, K., Fritsche, K., Brinkmann, V., Mollenkopf, H.-J., Thieck, O., et al. (2019). Chronic Chlamydia infection in human organoids increases stemness and promotes age-dependent CpG methylation. Nat. Commun. 10:1194. doi: 10.1038/s41467-019-09144-7
Kim, D., Nguyen, M. D., Dobbin, M. M., Fischer, A., Sananbenesi, F., Rodgers, J. T., et al. (2007). SIRT1 deacetylase protects against neurodegeneration in models for Alzheimer's disease and amyotrophic lateral sclerosis. EMBO J. 26, 3169–3179. doi: 10.1038/sj.emboj.7601758
Kol, A., Sukhova, G. K., Lichtman, A. H., and Libby, P. (1998). Chlamydial heat shock protein 60 localizes in human atheroma and regulates macrophage tumor necrosis factor-α and matrix metalloproteinase expression. Circulation 98, 300–307. doi: 10.1161/01.CIR.98.4.300
Koronyo, Y., Biggs, D., Barron, E., Boyer, D. S., Pearlman, J. A., Au, W. J., et al. (2017). Retinal amyloid pathology and proof-of-concept imaging trial in Alzheimer's disease. JCI Insight 2:e93621. doi: 10.1172/jci.insight.93621
Koronyo, Y., Rentsendorj, A., Mirzaei, N., Regis, G. C., Sheyn, J., Shi, H., et al. (2023). Retinal pathological features and proteome signatures of Alzheimer's disease. Acta Neuropathol. 145, 409–438. doi: 10.1007/s00401-023-02548-2
Koronyo, Y., Salumbides, B. C., Sheyn, J., Pelissier, L., Li, S., Ljubimov, V., et al. (2015). Therapeutic effects of glatiramer acetate and grafted CD115+ monocytes in a mouse model of Alzheimer's disease. Brain 138, 2399–2422. doi: 10.1093/brain/awv150
Koronyo-Hamaoui, M., Ko, M. K., Koronyo, Y., Azoulay, D., Seksenyan, A., Kunis, G., et al. (2009). Attenuation of AD-like neuropathology by harnessing peripheral immune cells: local elevation of IL-10 and MMP-9. J Neurochem 111, 1409–1424. doi: 10.1111/j.1471-4159.2009.06402.x
Koronyo-Hamaoui, M., Koronyo, Y., Ljubimov, A. V., Miller, C. A., Ko, M. K., Black, K. L., et al. (2011). Identification of amyloid plaques in retinas from Alzheimer's patients and noninvasive in vivo optical imaging of retinal plaques in a mouse model. NeuroImage 54, S204–S217. doi: 10.1016/j.neuroimage.2010.06.020
Koronyo-Hamaoui, M., Sheyn, J., Hayden, E. Y., Li, S., Fuchs, D.-T., Regis, G. C., et al. (2019). Peripherally derived angiotensin converting enzyme-enhanced macrophages alleviate Alzheimer-related disease. Brain 143, 336–358. doi: 10.1093/brain/awz364
Kortesoja, M., Trofin, R. E., and Hanski, L. (2020). A platform for studying the transfer of Chlamydia pneumoniae infection between respiratory epithelium and phagocytes. J. Microbiol. Methods 171, 105857. doi: 10.1016/j.mimet.2020.105857
Kravenska, Y., Nieznanska, H., Nieznanski, K., Lukyanetz, E., Szewczyk, A., and Koprowski, P. (2020). The monomers, oligomers, and fibrils of amyloid-β inhibit the activity of mitoBKCa channels by a membrane-mediated mechanism. Biochim. Biophys. Acta (BBA) - Biomembr. 1862:183337. doi: 10.1016/j.bbamem.2020.183337
Kumar, D. K. V., Choi, S. H., Washicosky, K. J., Eimer, W. A., Tucker, S., Ghofrani, J., et al. (2016). Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer's disease. Sci. Transl. Med. 8:340ra72. doi: 10.1126/scitranslmed.aaf1059
Kummer, M. P., Hermes, M., Delekarte, A., Hammerschmidt, T., Kumar, S., Terwel, D., et al. (2011). Nitration of tyrosine 10 critically enhances amyloid β aggregation and plaque formation. Neuron 71, 833–844. doi: 10.1016/j.neuron.2011.07.001
Kuo, C. C., Jackson, L. A., Campbell, L. A., and Grayston, J. T. (1995). Chlamydia pneumoniae (TWAR). Clin. Microbiol. Rev. 8, 451–461. doi: 10.1128/CMR.8.4.451
Kusbeci, O. Y., Miman, O., Yaman, M., Aktepe, O. C., and Yazar, S. (2011). Could Toxoplasma gondii have any role in Alzheimer disease? Alzheimer Dis. Assoc. Disord. 25, 1–3. doi: 10.1097/WAD.0b013e3181f73bc2
Laflamme, N., and Rivest, S. (2001). Toll-like receptor 4: the missing link of the cerebral innate immune response triggered by circulating gram-negative bacterial cell wall components. FASEB J. 15, 155–163. doi: 10.1096/fj.00-0339com
Laflamme, N., Soucy, G., and Rivest, S. (2001). Circulating cell wall components derived from gram-negative, not gram-positive, bacteria cause a profound induction of the gene-encoding Toll-like receptor 2 in the CNS. J. Neurochem. 79, 648–657. doi: 10.1046/j.1471-4159.2001.00603.x
Leeuw, S. M., de Kirschner, A. W. T., Lindner, K., Rust, R., Budny, V., Wolski, W. E., et al. (2022). APOE2, E3, and E4 differentially modulate cellular homeostasis, cholesterol metabolism, and inflammatory response in isogenic iPSC-derived astrocytes. Stem Cell Rep. 17, 110–126. doi: 10.1016/j.stemcr.2021.11.007
Lehrer, S., and Rheinstein, P. H. (2022). Circulating Chlamydia trachomatis antigens in subjects with Alzheimer's disease. Vivo Athens Greece 36, 2650–2653. doi: 10.21873/invivo.12999
Li, M., Geng, R., Li, C., Meng, F., Zhao, H., Liu, J., et al. (2021). Dysregulated gene-associated biomarkers for Alzheimer's disease and aging. Transl. Neurosci. 12, 83–95. doi: 10.1515/tnsci-2021-0009
Li, Q.-X., Villemagne, V. L., Doecke, J. D., Rembach, A., Sarros, S., Varghese, S., et al. (2015). Alzheimer's disease normative cerebrospinal fluid biomarkers validated in PET amyloid-β characterized subjects from the australian imaging, biomarkers and lifestyle (AIBL) study. J. Alzheimers Dis. 48, 175–187. doi: 10.3233/JAD-150247
Li, S., Hayden, E. Y., Garcia, V. J., Fuchs, D.-T., Sheyn, J., Daley, D. A., et al. (2020). Activated bone marrow-derived macrophages eradicate Alzheimer's-related Aβ42 oligomers and protect synapses. Front. Immunol. 11:49. doi: 10.3389/fimmu.2020.00049
Liang, T., Zhang, Y., Wu, S., Chen, Q., and Wang, L. (2022). The role of NLRP3 inflammasome in Alzheimer's disease and potential therapeutic targets. Front. Pharmacol. 13, 845185. doi: 10.3389/fphar.2022.845185
Lim, C., Hammond, C. J., Hingley, S. T., and Balin, B. J. (2014). Chlamydia pneumoniae infection of monocytes in vitro stimulates innate and adaptive immune responses relevant to those in Alzheimer's disease. J. Neuroinflammation 11:217. doi: 10.1186/s12974-014-0217-0
Lindman, K. L., Weidung, B., Olsson, J., Josefsson, M., Johansson, A., Eriksson, S., et al. (2021). Plasma amyloid-β in relation to antibodies against herpes simplex virus, cytomegalovirus, and chlamydophila pneumoniae. J. Alzheimers Dis. Rep. 5, 229–235. doi: 10.3233/ADR-210008
Little, C. S., Bowe, A., Lin, R., Litsky, J., Fogel, R. M., Balin, B. J., et al. (2005). Age Alterations in extent and severity of experimental intranasal infection with Chlamydophila pneumoniae in BALB/c Mice. Infect Immun 73, 1723–1734. doi: 10.1128/IAI.73.3.1723-1734.2005
Little, C. S., Hammond, C. J., MacIntyre, A., Balin, B. J., and Appelt, D. M. (2004). Chlamydia pneumoniae induces Alzheimer-like amyloid plaques in brains of BALB/c mice. Neurobiol. Aging 25, 419–429. doi: 10.1016/S0197-4580(03)00127-1
Little, C. S., Joyce, T. A., Hammond, C. J., Matta, H., Cahn, D., Appelt, D. M., et al. (2014). Detection of bacterial antigens and Alzheimer's disease-like pathology in the central nervous system of BALB/c mice following intranasal infection with a laboratory isolate of Chlamydia pneumoniae. Front. Aging Neurosci. 6:304. doi: 10.3389/fnagi.2014.00304
Liu, C.-C., Hu, J., Zhao, N., Wang, J., Wang, N., Cirrito, J. R., et al. (2017). Astrocytic LRP1 mediates brain aβ clearance and impacts amyloid deposition. J. Neurosci. 37, 4023–4031. doi: 10.1523/JNEUROSCI.3442-16.2017
Liu, H., Xu, S., Wang, C., Deng, Y., Xu, B., Yang, T., et al. (2022). The beneficial role of sirtuin 1 in preventive or therapeutic options of neurodegenerative diseases. Neuroscience 504, 79–92. doi: 10.1016/j.neuroscience.2022.09.021
Liu, N.-Y., Sun, J.-H., Jiang, X.-F., and Li, H. (2021). Helicobacter pylori infection and risk for developing dementia: an evidence-based meta-analysis of case-control and cohort studies. Aging 13, 22571–22587. doi: 10.18632/aging.203571
Liu, Y., Dentin, R., Chen, D., Hedrick, S., Ravnskjaer, K., Schenk, S., et al. (2008). A fasting inducible switch modulates gluconeogenesis via activator/coactivator exchange. Nature 456, 269–273. doi: 10.1038/nature07349
Loeb, M. B., Molloy, D. W., Smieja, M., Standish, T., Goldsmith, C. H., Mahony, J., et al. (2004). A randomized, controlled trial of doxycycline and rifampin for patients with Alzheimer's disease. J. Am. Geriatr. Soc. 52, 381–387. doi: 10.1111/j.1532-5415.2004.52109.x
Lorenzl, S., Albers, D. S., Relkin, N., Ngyuen, T., Hilgenberg, S. L., Chirichigno, J., et al. (2003). Increased plasma levels of matrix metalloproteinase-9 in patients with Alzheimer's disease. Neurochem. Int. 43, 191–196. doi: 10.1016/S0197-0186(03)00004-4
Ma, L., Zhang, L., Wang, B., Wei, J., Liu, J., and Zhang, L. (2015). Berberine inhibits Chlamydia pneumoniae infection-induced vascular smooth muscle cell migration through downregulating MMP3 and MMP9 via PI3K. Eur. J. Pharmacol. 755, 102–109. doi: 10.1016/j.ejphar.2015.02.039
Ma, Q.-L., Zuo, X., Yang, F., Ubeda, O. J., Gant, D. J., Alaverdyan, M., et al. (2014). Loss of MAP function leads to hippocampal synapse loss and deficits in the morris water maze with aging. J. Neurosci. 34, 7124–7136. doi: 10.1523/JNEUROSCI.3439-13.2014
MacDonald, A. B., and Miranda, J. M. (1987). Concurrent neocortical borreliosis and Alzheimer's disease. Hum. Pathol. 18, 759–761. doi: 10.1016/S0046-8177(87)80252-6
MacIntyre, A., Abramov, R., Hammond, C. J., Hudson, A. P., Arking, E. J., Little, C. S., et al. (2003). Chlamydia pneumoniae infection promotes the transmigration of monocytes through human brain endothelial cells. J. Neurosci. Res. 71, 740–750. doi: 10.1002/jnr.10519
MacIntyre, A., Hammond, C. J., Little, C. S., Appelt, D. M., and Balin, B. J. (2002). Chlamydia pneumoniae infection alters the junctional complex proteins of human brain microvascular endothelial cells. FEMS Microbiol. Lett. 217, 167–172. doi: 10.1111/j.1574-6968.2002.tb11470.x
Mahami-Oskouei, M., Hamidi, F., Talebi, M., Farhoudi, M., Taheraghdam, A. A., Kazemi, T., et al. (2016). Toxoplasmosis and Alzheimer: can Toxoplasma gondii really be introduced as a risk factor in etiology of Alzheimer? Parasitol. Res. 115, 3169–3174. doi: 10.1007/s00436-016-5075-5
Maheshwari, P., and Eslick, G. D. (2015). Bacterial infection and Alzheimer's disease: a meta-analysis. J. Alzheimers Dis. 43, 957–966. doi: 10.3233/JAD-140621
Mahony, J. B., Woulfe, J., Munoz, D., Browning, D., Chong, S., and Smieja, M. (2000). Identification of Chlamydia pneumoniae in the Alzheimer's brain. Neurobiol. Aging 21:245. doi: 10.1016/S0197-4580(00)83440-5
Malaguarnera, M., Bella, R., Alagona, G., Ferri, R., Carnemolla, A., and Pennisi, G. (2004). Helicobacter pylori and Alzheimer's disease: a possible link. Eur. J. Intern. Med. 15, 381–386. doi: 10.1016/j.ejim.2004.05.008
Malinverni, R., Kuo, C. C., Campbell, L. A., Lee, A., and Grayston, J. T. (1995). Effects of two antibiotic regimens on course and persistence of experimental Chlamydia pneumoniae TWAR pneumonitis. Antimicrob. Agents Chemother. 39, 45–49. doi: 10.1128/AAC.39.1.45
Miklossy, J. (1993). Alzheimer's disease—a spirochetosis? NeuroReport 4, 841–848. doi: 10.1097/00001756-199307000-00002
Miklossy, J. (2011). Alzheimer's disease - a neurospirochetosis. Analysis of the evidence following Koch's and Hill's criteria. J. Neuroinflammation 8:90. doi: 10.1186/1742-2094-8-90
Miklossy, J. (2015). Historic evidence to support a causal relationship between spirochetal infections and Alzheimer's disease. Front. Aging Neurosci. 7:46. doi: 10.3389/fnagi.2015.00046
Min, S.-W., Cho, S.-H., Zhou, Y., Schroeder, S., Haroutunian, V., Seeley, W. W., et al. (2010). Acetylation of tau inhibits its degradation and contributes to tauopathy. Neuron 67, 953–966. doi: 10.1016/j.neuron.2010.08.044
Miyashita, N., Niki, Y., Nakajima, M., Fukano, H., and Matsushima, T. (2001). Prevalence of asymptomatic infection with Chlamydia pneumoniae in subjectively healthy adults. Chest 119, 1416–1419. doi: 10.1378/chest.119.5.1416
Moir, R. D., Lathe, R., and Tanzi, R. E. (2018). The antimicrobial protection hypothesis of Alzheimer's disease. Alzheimers Dement. 14, 1602–1614. doi: 10.1016/j.jalz.2018.06.3040
Morgia, C. L., Ross-Cisneros, F. N., Koronyo, Y., Hannibal, J., Gallassi, R., Cantalupo, G., et al. (2016). Melanopsin retinal ganglion cell loss in Alzheimer disease. Ann. Neurol. 79, 90–109. doi: 10.1002/ana.24548
Muramatsu, T. (2011). Midkine: a promising molecule for drug development to treat diseases of the central nervous system. Curr. Pharm. Des. 17, 410–423. doi: 10.2174/138161211795164167
Natividad, A., Cooke, G., Holland, M. J., Burton, M. J., Joof, H. M., Rockett, K., et al. (2006). A coding polymorphism in matrix metalloproteinase 9 reduces risk of scarring sequelae of ocular Chlamydia trachomatisinfection. BMC Méd. Genet. 7:40. doi: 10.1186/1471-2350-7-40
N'Diaye, E.-N., Branda, C. S., Branda, S. S., Nevarez, L., Colonna, M., Lowell, C., et al. (2009). TREM-2 (triggering receptor expressed on myeloid cells 2) is a phagocytic receptor for bacteria. J. Cell Biol. 184, 215–223. doi: 10.1083/jcb.200808080
Nochlin, D., Shaw, C. M., Campbell, L. A., and Kuo, C.-C. (1999). Failure to detect Chlamydia pneumoniae in brain tissues of Alzheimer's disease. Neurology 53:1888. doi: 10.1212/WNL.53.8.1888-a
Nübling, G., Levin, J., Bader, B., Israel, L., Bötzel, K., Lorenzl, S., et al. (2012). Limited cleavage of tau with matrix-metalloproteinase MMP-9, but not MMP-3, enhances tau oligomer formation. Exp. Neurol. 237, 470–476. doi: 10.1016/j.expneurol.2012.07.018
O'Brien, R. J., and Wong, P. C. (2011). Amyloid precursor protein processing and Alzheimer's disease. Annu. Rev. Neurosci. 34, 185–204. doi: 10.1146/annurev-neuro-061010-113613
Ooij, C. V., Kalman, L., Ijzendoorn, S. V., Nishijima, M., Hanada, K., Mostov, K., et al. (2000). Host cell-derived sphingolipids are required for the intracellular growth of Chlamydia trachomatis. Cell. Microbiol. 2, 627–637. doi: 10.1046/j.1462-5822.2000.00077.x
Ou, H., Chien, W.-C., Chung, C.-H., Chang, H.-A., Kao, Y.-C., Wu, P.-C., et al. (2021). Association between antibiotic treatment of Chlamydia pneumoniae and reduced risk of Alzheimer dementia: a nationwide cohort study in Taiwan. Front. Aging Neurosci. 13:701899. doi: 10.3389/fnagi.2021.701899
Ou, Y.-N., Zhu, J.-X., Hou, X.-H., Shen, X.-N., Xu, W., Dong, Q., et al. (2020). Associations of infectious agents with Alzheimer's disease: a systematic review and meta-analysis. J. Alzheimers Dis. 75, 299–309. doi: 10.3233/JAD-191337
Paradowski, B., Jaremko, M., Dobosz, T., Leszek, J., and Noga, L. (2007). Evaluation of CSF-Chlamydia pneumoniae, CSF-tau, and CSF-Abeta42 in Alzheimer's disease and vascular dementia. J. Neurol. 254, 154–159. doi: 10.1007/s00415-006-0298-5
Park, A., Omura, S., Fujita, M., Sato, F., and Tsunoda, I. (2017). Helicobacter pylori and gut microbiota in multiple sclerosis versus Alzheimer's disease: 10 pitfalls of microbiome studies. Clin. Exp. Neuroimmunol. 8, 215–232. doi: 10.1111/cen3.12401
Park, A.-M., and Tsunoda, I. (2022). Helicobacter pylori infection in the stomach induces neuroinflammation: the potential roles of bacterial outer membrane vesicles in an animal model of Alzheimer's disease. Inflamm. Regen. 42:39. doi: 10.1186/s41232-022-00224-8
Perry, C. E., Gale, S. D., Erickson, L., Wilson, E., Nielsen, B., Kauwe, J., et al. (2016). Seroprevalence and serointensity of latent Toxoplasma gondii in a sample of elderly adults with and without Alzheimer disease. Alzheimer Dis. Assoc. Disord. 30, 123–126. doi: 10.1097/WAD.0000000000000108
Peters, J., Wilson, D. P., Myers, G., Timms, P., and Bavoil, P. M. (2007). Type III secretion à la Chlamydia. Trends Microbiol. 15, 241–251. doi: 10.1016/j.tim.2007.04.005
Petyaev, I. M., Zigangirova, N. A., Morgunova, E. Y., Kyle, N. H., Fedina, E. D., and Bashmakov, Y. K. (2017). Resveratrol inhibits propagation of Chlamydia trachomatis in McCoy cells. Biomed. Res. Int. 2017:4064071. doi: 10.1155/2017/4064071
Pietro, M. D., Filardo, S., Santis, F. D., and Sessa, R. (2013). Chlamydia pneumoniae infection in atherosclerotic lesion development through oxidative stress: a brief overview. Int. J. Mol. Sci. 14, 15105–15120. doi: 10.3390/ijms140715105
Pisa, D., Alonso, R., Rábano, A., Rodal, I., and Carrasco, L. (2015). Different brain regions are infected with fungi in Alzheimer's disease. Sci. Rep. 5:15015. doi: 10.1038/srep15015
Porritt, R. A., and Crother, T. R. (2016). Chlamydia pneumoniae infection and inflammatory diseases. Forum Immunopathol. Dis. Ther. 7, 237–254. doi: 10.1615/ForumImmunDisTher.2017020161
Pyne, J. D., and Brickman, A. M. (2021). The impact of the COVID-19 pandemic on dementia risk: potential pathways to cognitive decline. Neurodegener. Dis. 21, 1–23. doi: 10.1159/000518581
Qin, W., Yang, T., Ho, L., Zhao, Z., Wang, J., Chen, L., et al. (2006). Neuronal SIRT1 activation as a novel mechanism underlying the prevention of Alzheimer disease amyloid neuropathology by calorie restriction. J. Biol. Chem. 281, 21745–21754. doi: 10.1074/jbc.M602909200
Raulin, A.-C., Doss, S. V., Trottier, Z. A., Ikezu, T. C., Bu, G., and Liu, C.-C. (2022). ApoE in Alzheimer's disease: pathophysiology and therapeutic strategies. Mol. Neurodegener. 17:72. doi: 10.1186/s13024-022-00574-4
Reiken, S., Sittenfeld, L., Dridi, H., Liu, Y., Liu, X., and Marks, A. R. (2022). Alzheimer's-like signaling in brains of COVID-19 patients. Alzheimers Dement. 18, 955–965. doi: 10.1002/alz.12558
Rentsendorj, A., Sheyn, J., Fuchs, D.-T., Daley, D., Salumbides, B. C., Schubloom, H. E., et al. (2018). A novel role for osteopontin in macrophage-mediated amyloid-β clearance in Alzheimer's models. Brain Behav. Immun. 67, 163–180. doi: 10.1016/j.bbi.2017.08.019
Renvoize, E. B., and Hambling, M. H. (1984). Cytomegalovirus infection and Alzheimer's disease. Age Ageing 13, 205–209. doi: 10.1093/ageing/13.4.205
Ring, R. H., and Lyons, J. M. (2000). Failure To Detect Chlamydia pneumoniae in the late-onset Alzheimer's brain. J. Clin. Microbiol. 38, 2591–2594. doi: 10.1128/JCM.38.7.2591-2594.2000
Riviere, G. R., Riviere, K. H., and Smith, K. S. (2002). Molecular and immunological evidence of oral Treponema in the human brain and their association with Alzheimer's disease. Oral Microbiol. Immunol. 17, 113–118. doi: 10.1046/j.0902-0055.2001.00100.x
Rupp, J., Pfleiderer, L., Jugert, C., Moeller, S., Klinger, M., Dalhoff, K., et al. (2009). Chlamydia pneumoniae hides inside apoptotic neutrophils to silently infect and propagate in macrophages. PLoS ONE 4:e6020. doi: 10.1371/journal.pone.0006020
Sanino, G., Bosco, M., and Terrazzano, G. (2020). Physiology of midkine and its potential pathophysiological role in COVID-19. Front. Physiol. 11:616552. doi: 10.3389/fphys.2020.616552
Scherrer, J. F., Salas, J., Wiemken, T. L., Jacobs, C., Morley, J. E., and Hoft, D. F. (2021). Lower risk for dementia following adult tetanus, diphtheria, and pertussis (TDAP) vaccination. J. Gerontol.: Ser. A 76, 1436–1443. doi: 10.1093/gerona/glab115
Senejani, A. G., Maghsoudlou, J., El-Zohiry, D., Gaur, G., Wawrzeniak, K., Caravaglia, C., et al. (2021). Borrelia burgdorferi co-localizing with amyloid markers in Alzheimer's disease brain tissues. J. Alzheimers Dis. 85, 889–903. doi: 10.3233/JAD-215398
Serrano-Pozo, A., Frosch, M. P., Masliah, E., and Hyman, B. T. (2011). Neuropathological alterations in Alzheimer disease. Cold Spring Harb. Perspect. Med. 1:a006189. doi: 10.1101/cshperspect.a006189
Shackleton, B., Ringland, C., Abdullah, L., Mullan, M., Crawford, F., and Bachmeier, C. (2019). Influence of matrix metallopeptidase 9 on beta-amyloid elimination across the blood-brain barrier. Mol. Neurobiol. 56, 8296–8305. doi: 10.1007/s12035-019-01672-z
Shao, B.-Z., Cao, Q., and Liu, C. (2018). Targeting NLRP3 inflammasome in the treatment of CNS diseases. Front Mol Neurosci 11:320. doi: 10.3389/fnmol.2018.00320
Shao, R., Zhang, S. X., Weijdegård, B., Zou, S., Egecioglu, E., Norström, A., et al. (2010). Nitric oxide synthases and tubal ectopic pregnancies induced by Chlamydia infection: basic and clinical insights. MHR: Basic Sci. Reprod. Med. 16, 907–915. doi: 10.1093/molehr/gaq063
Shi, H., Koronyo, Y., Fuchs, D., Sheyn, J., Jallow, O., Mandalia, K., et al. (2023). Retinal arterial Aβ40 deposition is linked with tight junction loss and cerebral amyloid angiopathy in MCI and AD patients. Alzheimers Dement. 19, 5185–5197. doi: 10.1002/alz.13086
Shi, H., Koronyo, Y., Rentsendorj, A., Regis, G. C., Sheyn, J., Fuchs, D.-T., et al. (2020). Identification of early pericyte loss and vascular amyloidosis in Alzheimer's disease retina. Acta Neuropathol. 139, 813–836. doi: 10.1007/s00401-020-02134-w
Shimada, K., Crother, T. R., Karlin, J., Chen, S., Chiba, N., Ramanujan, V. K., et al. (2011). Caspase-1 dependent IL-1β secretion is critical for host defense in a mouse model of Chlamydia pneumoniae lung infection. PLoS ONE 6:e21477. doi: 10.1371/journal.pone.0021477
Sipilä, P. N., Heikkilä, N., Lindbohm, J. V., Hakulinen, C., Vahtera, J., Elovainio, M., et al. (2021). Hospital-treated infectious diseases and the risk of dementia: a large, multicohort, observational study with a replication cohort. Lancet Infect. Dis. 21, 1557–1567. doi: 10.1016/S1473-3099(21)00144-4
Soscia, S. J., Kirby, J. E., Washicosky, K. J., Tucker, S. M., Ingelsson, M., Hyman, B., et al. (2010). The Alzheimer's disease-associated amyloid β-protein is an antimicrobial peptide. PLoS ONE 5:e9505. doi: 10.1371/journal.pone.0009505
Soung, A. L., Vanderheiden, A., Nordvig, A. S., Sissoko, C. A., Canoll, P., Mariani, M. B., et al. (2022). COVID-19 induces CNS cytokine expression and loss of hippocampal neurogenesis. Brain 145, awac270. doi: 10.1093/brain/awac270
Sundar, S., Battistoni, C., McNulty, R., Morales, F., Gorky, J., Foley, H., et al. (2020). An agent-based model to investigate microbial initiation of Alzheimer's via the olfactory system. Theor. Biol. Méd. Model. 17:5. doi: 10.1186/s12976-020-00123-w
Taavitsainen-Wahlroos, E., Miettinen, I., Ylätalo, M., Reigada, I., Savijoki, K., Nyman, T. A., et al. (2022). Chlamydia pneumoniae interferes with macrophage differentiation and cell cycle regulation to promote its replication. Cell. Microbiol. 2022, 1–19. doi: 10.1155/2022/9854449
Taylor, G. S., Vipond, I. B., Paul, I. D., Matthews, S., Wilcock, G. K., and Caul, E. O. (2002). Failure to correlate C. pneumoniae with late onset Alzheimer's disease. Neurology 59, 142–143. doi: 10.1212/WNL.59.1.142
Tcw, J., Qian, L., Pipalia, N. H., Chao, M. J., Liang, S. A., Shi, Y., et al. (2022). Cholesterol and matrisome pathways dysregulated in astrocytes and microglia. Cell 185, 2213–2233.e25. doi: 10.1016/j.cell.2022.05.017
Tran, V. T. A., Lee, L. P., and Cho, H. (2022). Neuroinflammation in neurodegeneration via microbial infections. Front. Immunol. 13:907804. doi: 10.3389/fimmu.2022.907804
Tumurkhuu, G., Dagvadorj, J., Porritt, R. A., Crother, T. R., Shimada, K., Tarling, E. J., et al. (2018). Chlamydia pneumoniae hijacks a host autoregulatory IL-1β loop to drive foam cell formation and accelerate atherosclerosis. Cell Metab. 28, 432–448.e4. doi: 10.1016/j.cmet.2018.05.027
Vickers, J. C., Mitew, S., Woodhouse, A., Fernandez-Martos, C. M., Kirkcaldie, M. T., Canty, A. J., et al. (2016). Defining the earliest pathological changes of Alzheimer's disease. Curr. Alzheimer Res. 13, 281–287. doi: 10.2174/1567205013666151218150322
Victor, M. B., Leary, N., Luna, X., Meharena, H. S., Scannail, A. N., Bozzelli, P. L., et al. (2022). Lipid accumulation induced by APOE4 impairs microglial surveillance of neuronal-network activity. Cell Stem Cell 29, 1197–1212.e8. doi: 10.1016/j.stem.2022.07.005
Vielma, S. A., Krings, G., and Lopes-Virella, M. F. (2003). Chlamydophila pneumoniae Induces ICAM-1 expression in human aortic endothelial cells via protein kinase C–dependent activation of nuclear factor-κB. Circ. Res. 92, 1130–1137. doi: 10.1161/01.RES.0000074001.46892.1C
Voorend, M., Ven, A. J. A. M., van der Mulder, M., Lodder, J., Steinbusch, H. W. M., and Bruggeman, C. A. (2010). Chlamydia pneumoniae infection enhances microglial activation in atherosclerotic mice. Neurobiol. Aging 31, 1766–1773. doi: 10.1016/j.neurobiolaging.2008.09.022
Walkiewicz, G., Ronisz, A., Ginderdeuren, R. V., Lemmens, S., Bouwman, F. H., Hoozemans, J. J. M., et al. (2024). Primary retinal tauopathy: a tauopathy with a distinct molecular pattern. Alzheimers Dement. 20, 330–340. doi: 10.1002/alz.13424
Wang, B., Zhang, L., Zhang, T., Wang, H., Zhang, J., Wei, J., et al. (2013). Chlamydia pneumoniae infection promotes vascular smooth muscle cell migration through a toll-like receptor 2-related signaling pathway. Infect. Immun. 81, 4583–4591. doi: 10.1128/IAI.01087-13
Wang, J., Gu, B. J., Masters, C. L., and Wang, Y.-J. (2017). A systemic view of Alzheimer disease — insights from amyloid-β metabolism beyond the brain. Nat. Rev. Neurol. 13, 612–623. doi: 10.1038/nrneurol.2017.111
Wen, Y., Luo, F., Zhao, L., Su, S., Lei, W., Liu, Y., et al. (2021). Long non-coding RNA FGD5-AS1 induced by Chlamydia trachomatis infection inhibits apoptosis via Wnt/β-catenin signaling pathway. Front. Cell. Infect. Microbiol. 11:701352. doi: 10.3389/fcimb.2021.701352
Wozniak, M. A., Cookson, A., Wilcock, G. K., and Itzhaki, R. F. (2003). Absence of Chlamydia pneumoniae in brain of vascular dementia patients. Neurobiol. Aging 24, 761–765. doi: 10.1016/S0197-4580(02)00236-1
Wynne, O., Horvat, J. C., Osei-Kumah, A., Smith, R., Hansbro, P. M., Clifton, V. L., et al. (2011). Early life infection alters adult BALB/c hippocampal gene expression in a sex specific manner. Stress 14, 247–261. doi: 10.3109/10253890.2010.532576
Xu, Q. A., Boerkoel, P., Hirsch-Reinshagen, V., Mackenzie, I. R., Hsiung, G.-Y. R., Charm, G., et al. (2022). Müller cell degeneration and microglial dysfunction in the Alzheimer's retina. Acta Neuropathol. Commun. 10:145. doi: 10.1186/s40478-022-01448-y
Yamamoto, H., Watanabe, T., Miyazaki, A., Katagiri, T., Idei, T., Iguchi, T., et al. (2005). High prevalence of Chlamydia pneumoniae antibodies and increased high-sensitive C-reactive protein in patients with vascular dementia. J. Am. Geriatr. Soc. 53, 583–589. doi: 10.1111/j.1532-5415.2005.53204.x
Yamazaki, Y., Zhao, N., Caulfield, T. R., Liu, C.-C., and Bu, G. (2019). Apolipoprotein E and Alzheimer disease: pathobiology and targeting strategies. Nat. Rev. Neurol. 15, 501–518. doi: 10.1038/s41582-019-0228-7
Zhao, C., Strobino, K., Moon, Y. P., Cheung, Y. K., Sacco, R. L., Stern, Y., et al. (2020). APOE ϵ4 modifies the relationship between infectious burden and poor cognition. Neurol. Genet. 6:e462. doi: 10.1212/NXG.0000000000000462
Zhao, X., Miao, G., Zhang, L., Zhang, Y., Zhao, H., Xu, Z., et al. (2022). Chlamydia pneumoniae infection induces vascular smooth muscle cell migration and atherosclerosis through mitochondrial reactive oxygen species-mediated JunB-Fra-1 activation. Front. Cell Dev. Biol. 10:879023. doi: 10.3389/fcell.2022.879023
Zuo, Z., Qi, F., Yang, J., Wang, X., Wu, Y., Wen, Y., et al. (2017). Immunization with Bacillus Calmette-Guérin (BCG) alleviates neuroinflammation and cognitive deficits in APP/PS1 mice via the recruitment of inflammation-resolving monocytes to the brain. Neurobiol. Dis. 101, 27–39. doi: 10.1016/j.nbd.2017.02.001
Keywords: Alzheimer's disease, amyloid beta-protein (Aβ) plaque, infection, microbes, Chlamydia pneumoniae
Citation: Subedi L, Gaire BP, Koronyo Y, Koronyo-Hamaoui M and Crother TR (2024) Chlamydia pneumoniae in Alzheimer's disease pathology. Front. Neurosci. 18:1393293. doi: 10.3389/fnins.2024.1393293
Received: 28 February 2024; Accepted: 22 April 2024;
Published: 06 May 2024.
Edited by:
Willayat Yousuf Wani, Northwestern University, United StatesReviewed by:
Xian-Le Bu, Third Military Medical University, ChinaDeepak Kumar Vijaya Kumar, Massachusetts General Hospital and Harvard Medical School, United States
Copyright © 2024 Subedi, Gaire, Koronyo, Koronyo-Hamaoui and Crother. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Timothy R. Crother, dGltb3RoeS5jcm90aGVyQGNzaHMub3Jn
 Lalita Subedi
Lalita Subedi Bhakta Prasad Gaire
Bhakta Prasad Gaire Yosef Koronyo3
Yosef Koronyo3 Maya Koronyo-Hamaoui
Maya Koronyo-Hamaoui Timothy R. Crother
Timothy R. Crother