
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Neurol. , 27 March 2025
Sec. Endovascular and Interventional Neurology
Volume 16 - 2025 | https://doi.org/10.3389/fneur.2025.1552610
This article is part of the Research Topic Advances in the Understanding, Diagnosis, and Management of Intracranial and Extracranial Arterial Dissections View all 5 articles
Background: The Tubridge flow diverter (TFD) has become a widely used device for reconstructing parent vessels and sealing complicated aneurysms in China. However, its application to managing distal small to medium-sized dissecting aneurysms has not been extensively investigated. This study aimed to evaluate the safety and effectiveness of the TFD and the factors affecting healing in patients with dissecting aneurysms in the middle cerebral artery (MCA).
Methods: Patients with dissecting aneurysms in the MCA who were treated with the TFD from 2019 to 2023 were included. According to the O’Kelly–Marotta (OKM) scale, OKM grade A was defined as dense embolism, while OKM grades B to D were defined as non-dense embolism in this study. Clinical information, the degree of aneurysm occlusion, and clinical outcomes were retrospectively analyzed.
Results: A total of 25 patients with 25 MCA dissecting aneurysms were identified. The average age was 52.4 years (age range 20–76), with 5 patients (20%) experiencing subarachnoid hemorrhage. In total, 12 aneurysms were located in the M1 segment, 11 involved the M2 bifurcation, and two were in the M3 segment or above. The median aneurysm length was 17.3 mm (range 4.2–27), and the average width was 5.4 mm (range 2.3–7.6). A total of 12 cases had one or more branches in the MCA. Furthermore, four cases (16%) showed asymptomatic in-stent stenosis, and five cases (20%) had main branch injuries during the angiographic follow-up. A total of three patients experienced acute ischemic events, and one had not fully recovered during the follow-up. There were no deaths related to procedure complications. According to the single-factor analysis, the patients in the non-dense embolism group during the follow-up had more strong branches before the operation (χ2 = 9.001, p=
Conclusion: The TFD is a safe and effective flow diverter for distal dissecting MCA aneurysms. The aneurysms with strong branches were associated with a higher rate of non-dense embolism during the follow-up.
As we know, it remains challenging to manage dissecting aneurysms in the middle cerebral artery (MCA) with complex anatomies, particularly those with wide-neck configurations that involve small, critical branches (1, 2). In the past, many neurosurgeons preferred clipping, wrapping, or ligating MCA dissecting aneurysms as the primary treatment option (3–5). Recently, flow diverters (FDs) have been regarded as a promising treatment option for treating complex and distorted wide-neck aneurysms, including those with many small branches. However, the efficacy and safety of this technique are not yet fully understood (6, 7). The Tubridge flow diverter (TFD, MicroPort Neurotech, Shanghai, China), an innovative self-expanding stent made of nickel-titanium braiding, has been proven to be a feasible and effective treatment for complex, large, and giant intracranial aneurysms in China (8, 9). It could also be applied to small internal carotid artery (ICA) (10) dissecting aneurysms (11, 12).
In this study, we conducted a retrospective analysis focusing on the treatment of MCA dissecting aneurysms using the TFD, with the goal of elucidating the following aspects: (1) the occlusion rate and its relevant factors, (2) treatment-related complications and clinical outcomes, and (3) the outcome for small branches after TFD deployment.
The study was approved by our institutional review board, and all patients provided general informed consent. We reviewed our database to identify dissecting aneurysms in the MCA treated with flow diversion between May 2019 and May 2023.
The inclusion criteria were as follows: (1) Dissecting aneurysms located in the middle cerebral artery, as identified using cerebral digital subtraction angiography (DSA) and high-definition magnetic resonance imaging; (2) patients over 18 years old; and (3) treatment with TFD. The exclusion criteria were as follows: (1) aneurysms associated with trauma, mycosis or infection, and inflammation, and (2) previous surgical clipping or stent-assisted coiling.
Based on the Sacho’ study, dissecting aneurysms were defined as those with an aneurysmal dilation exceeding 50% of the vessel wall circumference (13). The aneurysms were classified into three types: (1) located in the M1 segment only; (2) involving the M2 bifurcation; and (3) located in the distal MCA, from M3 to M4. Small cortical branches arising before the division of the MCA into a superior and inferior trunk, or three divisions, and their relationship with the aneurysm were noted.
Clinical data on patient demographics, symptoms at presentation, postoperative angiograms, complications, clinical outcomes, and follow-up imaging were all collected (see Table 1).
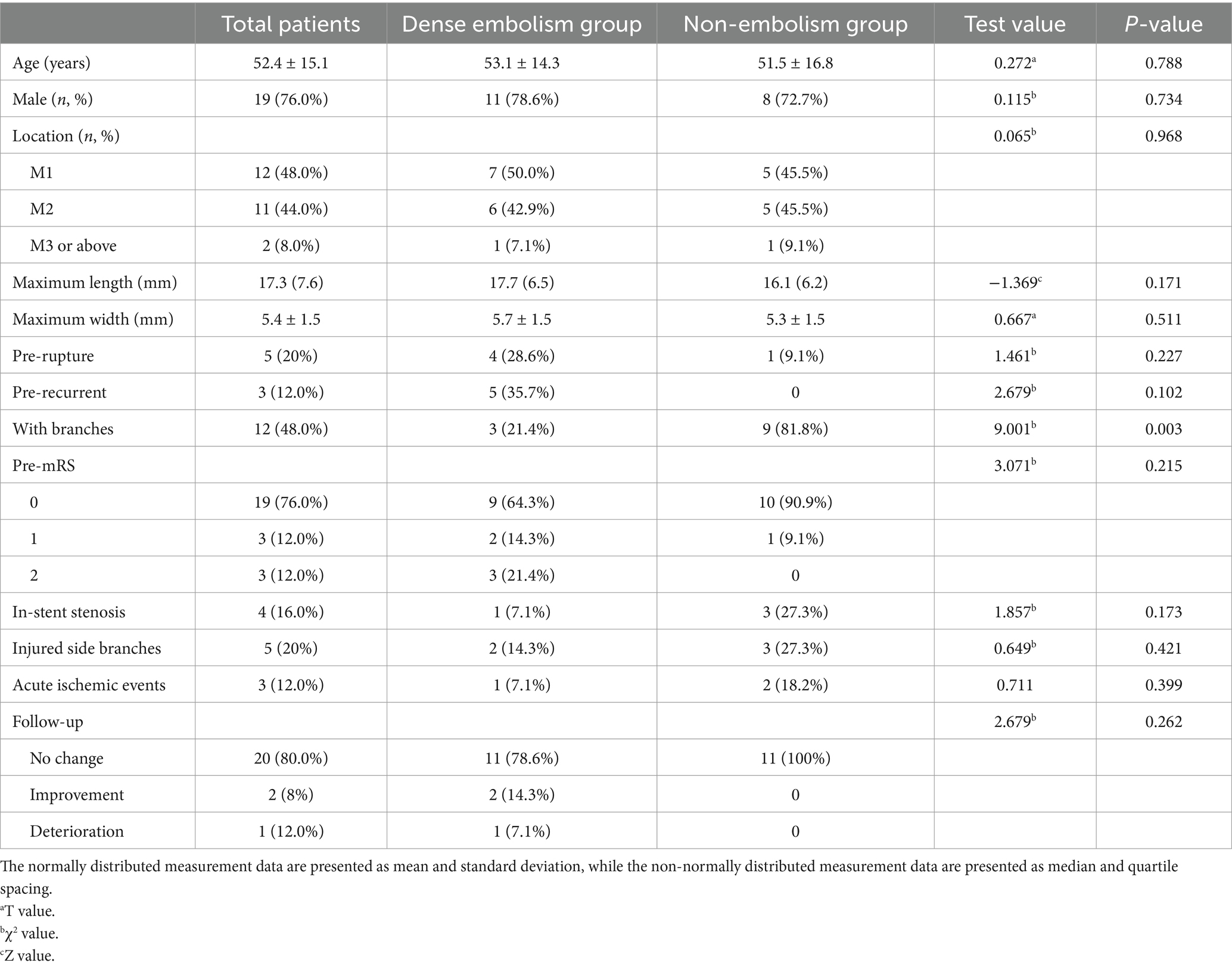
Table 1. Clinical information of the 25 patients and comparison between the dense embolism group and non-embolism group.
The patients received two dual antiplatelet therapy regimens: 100 mg/d aspirin and 75 mg/d clopidogrel (from March 2019 to February 2021), and 100 mg/d aspirin and 90 mg 90 mg/bid ticagrelor (from March 2021 to March 2023). The dual antiplatelet therapy was carried out 5 days before the procedure and continued for 3 months. The monotherapy of aspirin was continued for the next 9 months.
All procedures were performed under general anesthesia. Systemic heparinization was administered following sheath placement to prevent blood clotting during the procedure. An 8F femoral artery sheath with a 6F long sheath (80 or 90 cm) was used. After the long sheath reached the origin of the common carotid artery (CCA), an intracranial support catheter was navigated to the distal internal carotid artery. The Fastrack microcatheter was advanced over a 0.014-inch guidewire into the M3 segment or beyond in preparation for flow diverter delivery. The appropriate size and length of the TFD were selected based on the diameter of the parent artery and the length of the aneurysm. Under fluoroscopic guidance, for patients treated with adjunctive coil embolization, an Echelon−10 microcatheter (Medtronic, Minneapolis, MN, United States) was advanced into the aneurysm via contralateral femoral artery access for coiling after the implantation of the flow diverter.
Clinical evaluation took place immediately after each procedure, as well as at discharge and 3-, 6-, and 12-months post-procedure. The modified Rankin Scale (mRS) was used to assess clinical outcomes (14). Morbidity and mortality were defined as any deterioration in the mRS after the procedure and death related to treatment, respectively. CT angiography was usually performed 3 months after the procedure. Subsequent angiographies were performed initially at 6 months and then again at 12 months during the follow-up. Aneurysm occlusion was evaluated based on the O’Kelly–Marotta (OKM) scale: A—total filling, B—subtotal filling, C—entry remnant, and D—no filling (15). In this study, OKM grade A was defined as dense embolism, while OKM grades B to D were defined as non-dense embolism. The jailed side branch by the TFD was described as follows: (1) patent (unchanged), (2) narrowing of the diameter (stenosed) or orifice, and (3) occluded.
The data were analyzed using Statistical Package for the Social Sciences Version 25.0 for Windows (SPSS, Chicago, Illinois, United States). Normally distributed data were presented as mean and standard deviation and analyzed using the independent samples t-test. Non-normally distributed data were presented as median and quartile spacing and analyzed using the Mann–Whitney U-test. Categorical variables were presented as percentages and analyzed using Pearson’s χ2 test. A p-value of <0.05 was considered statistically significant.
A total of 25 cases of dissecting aneurysms in the MCA treated with the TFD were included. The average age was 52.4 years (age range 20 ~ 76), and 19 patients (76.0%) were female. The aneurysms were found incidentally in nine patients (36.0%), while five patients (20%) experienced hemorrhage due to the rupture of the aneurysm (Figure 1). The remaining patients exhibited symptoms such as headache and dizziness (Figure 2).
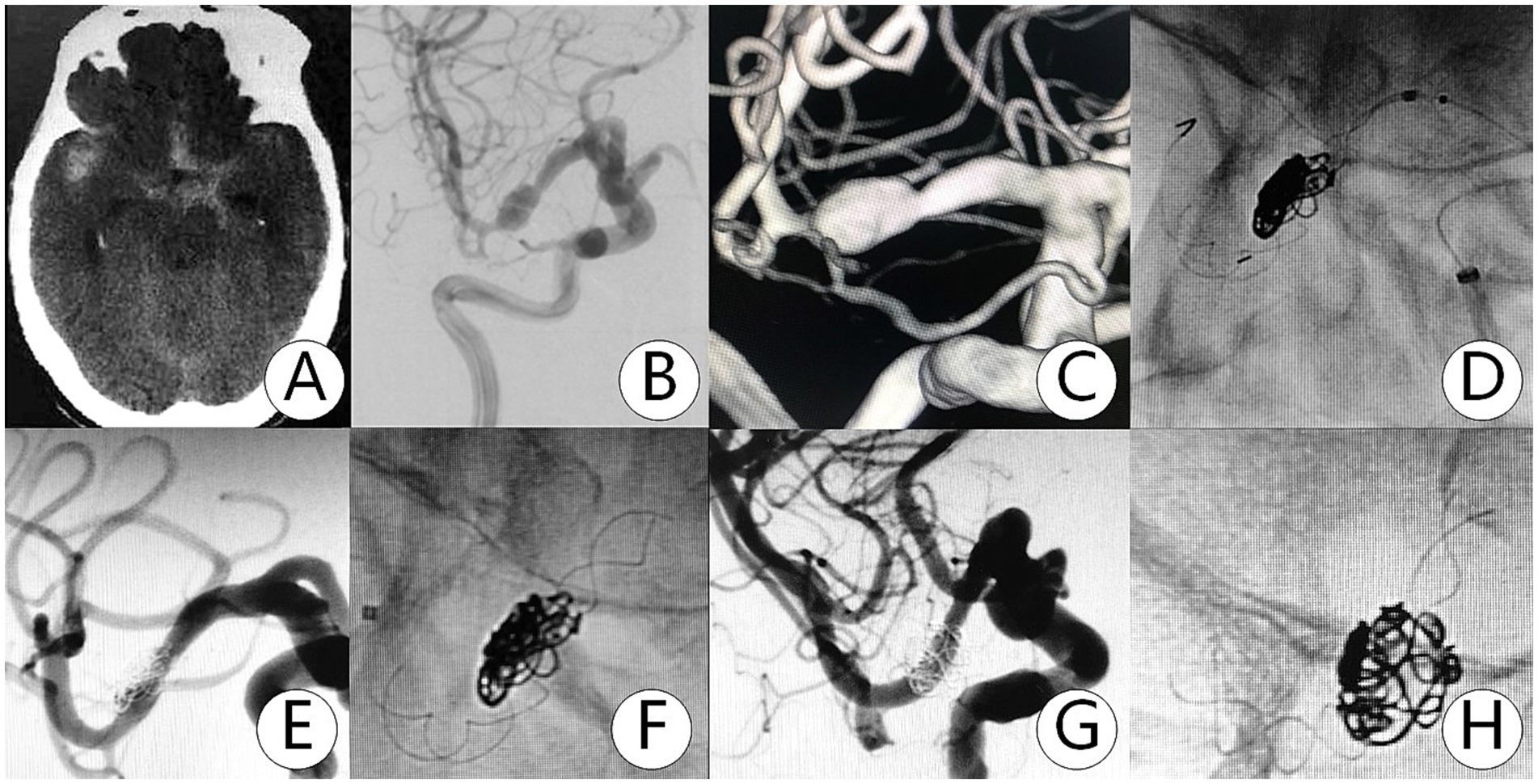
Figure 1. Patient experienced a subarachnoid hemorrhage with a Hunt–Hess grade I for 5 h. (A) CT showed subarachnoid hemorrhage in the Sylvian cistern and pericallosal cistern. (B,C) DSA revealed a dissecting aneurysm in the M1 segment. (D–F) The TFD (3.0×25 mm) and coils were successfully deployed in the aneurysm. (G,H) DSA at 13 months follow-up showed that the dissecting aneurysm was completely embolized.
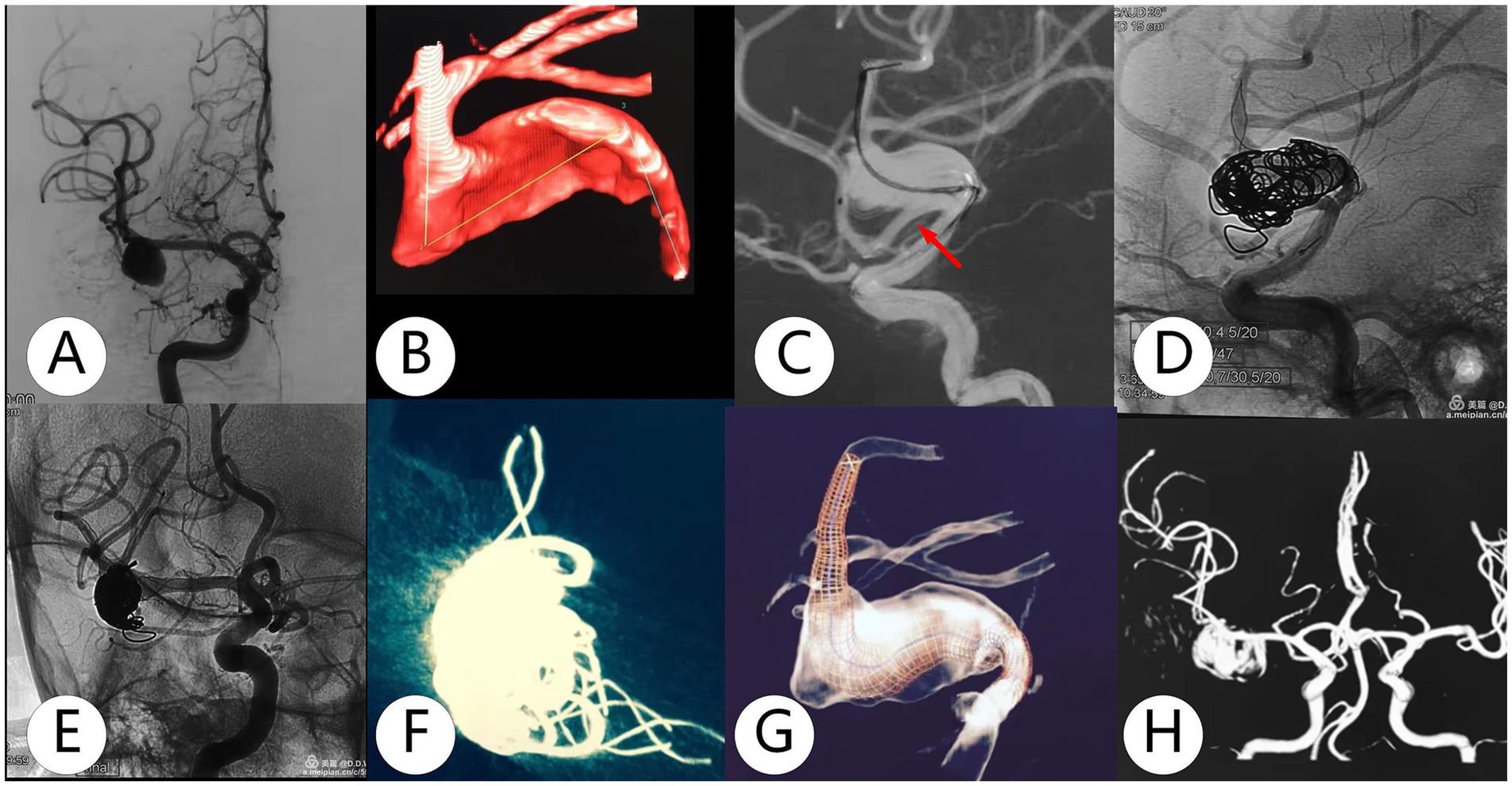
Figure 2. Adult was diagnosed with a right dissecting aneurysm during a health check. (A,B) The dissecting aneurysm extended from M1 to M2 with a strong branch. (C,D) The TFD (3.0×30 mm) with coils was deployed. (E–G) The reconstruction showed that the TFD unfolded well. (H). MRA at 1-year follow-up showed that the dissecting aneurysm remained at O’Kelly–Marotta grade C.
In total, 12 dissecting aneurysms (48%) were in the M1 segment, 11 (44%) involved the M2 bifurcation, and 2 (8%) were in the M3 or above. The median aneurysm length was 17.3 mm (range 4.2–27), and the average width was 5.4 mm (range 2.3–7.6). In addition, 12 (48%) cases had one or more branches in the MCA segment.
A total of 10 (40%) cases were treated with the TFD assisted by coils, including all five ruptured dissecting aneurysms. The remaining 15 cases (60%) were treated with the TFD alone, and 2 cases (80%) were treated with 2 TFD stents deployed tandemly.
The immediate postoperative angiographic outcome showed that 4 cases (16%) of ruptured dissecting aneurysms were completely occluded (OKM A), 4 (16%) cases were assessed as OKM grade B (near complete occlusion), and 12 (48%) and 5 (20%) cases were assessed as OKM C and OKM D, respectively.
During the angiographic follow-up (13-month median), 14 cases (44%) showed complete occlusion (OKM A) and were in the dense embolism group. A total of 11 cases (56%) were in the non-dense embolism group. Of these, eight (32%) were in OKM grade B (near complete occlusion), three (12%) were in OKM C, and there were no cases in OKM D. In total, 22 cases (88%) showed progress according to the embolization assessment during the follow-up. From another point of view, the rate of complete (OKM A) and near-complete (OKM B) occlusion reached 88%.
Differences in age, aneurysm size, aneurysm location, history of subarachnoid hemorrhage, complications, and prognosis were not statistically significant between the dense embolism group and the non-dense embolism group. According to the single-factor analysis, the patients in the non-dense embolism group had a higher rate of having branches in the MCA segment, as shown by preoperative DSA images (χ2 = 9.001, p=).
A total of four cases (16%) showed asymptomatic in-stent stenosis, while five cases (20%) displayed branch injuries during the angiographic follow-up. Of those with branch injuries, one anterior cerebral artery (ACA) covered by the TFD showed narrowing, and four M1 to M2 branches covered by the TFD exhibited occlusion or a reduced caliber. In addition, three patients (12.0%) experienced procedure-related ischemic events during the follow-up. There were no deaths related to procedure complications.
A total of 24 patients (96%) had a good clinical outcome at the last follow-up (11 months to 36 months), and only 1 (4%) experienced a decline in neurological function from mRS 0 before the operation to mRS 1 at 23 months after the operation during the last follow-up.
Our study demonstrated that the TFD was also a safe and effective treatment for dissecting aneurysms in the MCA. The overall rate of complete occlusion (OKM A) and near complete (OKM B) was 88% (22/25), and 96% of the patients (24/25) received a good clinical outcome at the last follow-up. A total of 22 cases (88%) showed progress in embolization assessment during the follow-up. There were no cases of rupture or death. In the non-dense embolism group, the patients had a higher rate of having branches, which might have been an important factor contributing to incomplete healing during the follow-up.
Despite similar clinical safety and efficacy in terms of obliteration rates and complications between the Pipeline embolization device (PED) and TFD (16, 17), the TFD, being made of nickel–titanium alloys, offers high shape-holding memory and super-elasticity. Its radiopaque microfilaments improve the visualization of the stent during deployment (15, 16).
A random-effects meta-analysis of FDs showed that the obliteration rate was satisfactory (>70%) and the risk of branch occlusion-related complications was low (incidence rate < 5%) after 1-year follow-up (18). Xie et al. (11) reported that the TFD showed satisfying results for vertebrobasilar artery dissecting aneurysms, with a complete occlusion rate of 78.26% and a mild asymptomatic cerebral infarction rate of 13.04%. However, there is still limited literature on the use of FDs for treating distal dissecting aneurysms in the MCA or above. Soydemir et al. (19) reported that in 76 MCA aneurysms treated with different FD stents, including the flow re-direction endoluminal device (FRED), FRED Jr., and PED, the branches were occluded in 10% of cases and/or 30.1% had in-stent stenosis, but none of the patients had neurological deterioration during follow-up. Gai’s et al. (12) study of the PED and TFD showed that the complete occlusion rate was 75% in eight cases of M1 dissecting aneurysms. However, 10% of the cases experienced procedure-related morbidity.
The risk of thromboembolic events caused by clopidogrel resistance is taken seriously (20), and since 2021, clopidogrel has been replaced by ticagrelor (21), in combination with aspirin, as the conventional antiplatelet regimen for the perioperative period at our center. There was no direct evidence that four patients with in-stent stenosis were associated with antiplatelet therapy in this cohort; however, two patients with in-stent stenosis were observed after ticagrelor was discontinued. For the small-vessel PED, we tend to apply assured higher-potency antiplatelet agents, such as ticagrelor, to decrease the risk of acute stent occlusion (22). Ge’s et al. (23) report indicated that a higher maximum platelet aggregation rate predicted higher rates of incomplete occlusion and in-stent stenosis and that the dose levels of ticagrelor showed no significant difference in clinical prognosis and aneurysm occlusion rates. Antiplatelet aggregation materials or medications coated on FDs, such as heparin, hydrophilic polymers, or others, will be promising therapeutic targets in the future (24, 25). We will further explore the associated risks and complications of FDs with the expansion of sample size and longer follow-up.
In this cohort, 11 cases (44%) had one or more branches in the MCA segment, which is similar to the rate (66.6%) of Pervinder’ saccular aneurysms (26). It is admittedly recognized that MCA segment aneurysms with a deeper dissection have a close relationship with many small branches, leading to unpredictable outcomes. MCA aneurysms are readily accessible through an endovascular approach, and their morphology may make them suitable for flow diversion, either alone or with coil assistance (27, 28).
During the follow-up, we found that the branches hindered the dissecting aneurysm from achieving complete occlusion. The main reason was that continuous blood flow in the branches impeded stent reendothelialization. It was our basic concept that preserving the perforating vessels was more important than occluding the aneurysms itself. As we all know, perforating vessels in the MCA, such as lenticulostriate perforators, have irreplaceable functions, and if injured, they can lead to disastrous consequences. Bender reported a patient with a huge right MCA aneurysm who, after receiving the PED with adjunctive coiling, experienced a severe stroke. The injury to branches in the MCA was considered the pivotal cause of the stroke (27). Cagnazzo et al. (29) reported a thromboembolic complication rate of 16.3%, with symptomatic stroke related to jailed branch occlusion and slow flow occurring in approximately 5% of cases. In Gawltza’s et al. (30) study of FDs in the MCA bifurcation or anterior communicating artery complex, after cortical branches were covered, 41.2% of patients showed symptomatic or asymptomatic lesions in the perforator territories. In our cohort, we observed that five cases (20%) showed branch injuries, and three of them experienced mild stroke and hemiplegia after the operation. Based on our experience, if possible, it is important to avoid jailing the M2 branch (31), while ensuring adequate coverage of the aneurysm neck. We also agree that adjunctive coil embolization with a loose density (<12%) is necessary for large or giant dissecting aneurysms (32).
Our study has several limitations. First, the inherent bias associated with a retrospective, single-center design must be acknowledged. Second, the results from the subgroup analyses within this small population need to be confirmed through multi-center studies with larger sample sizes and longer follow-ups.
The TFD is a safe and effective flow diverter for distal dissecting aneurysms in the MCA. The aneurysms with strong branches were associated with a higher rate of non-dense embolism during the follow-up.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by the Ethics Committee of Huadong Hospital. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants' legal guardians/next of kin. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
YD: Data curation, Writing – original draft. GH: Formal analysis, Investigation, Writing – review & editing. JS: Funding acquisition, Methodology, Writing – review & editing. ZX: Formal analysis, Writing – review & editing. ZL: Methodology, Project administration, Writing – review & editing. JL: Investigation, Writing – review & editing. DD: Funding acquisition, Methodology, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. Funding for this study was provided by National Natural Science Foundation of China (No. 82301355); China 234 Discipline Peak Climbing Program of naval military medical University, (No. 2020YXK046); National Key Research and Development Program. Application Demonstration Project of Medical and Nutritional Service Models and Norms, (No. 2020YFC2006005).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Brunozzi, D, Shakur, SF, Hussein, AE, Charbel, FT, and Alaraj, A. Middle cerebral artery flow velocity increases more in patients with delayed intraparenchymal hemorrhage after pipeline. J Neurointerv Surg. (2018) 10:249–51. doi: 10.1136/neurintsurg-2017-013042
2. Saleme, S, Iosif, C, Ponomarjova, S, Mendes, G, Camilleri, Y, Caire, F, et al. Flow-diverting stents for intracranial bifurcation aneurysm treatment. Neurosurgery. (2014) 75:623–31. doi: 10.1227/NEU.0000000000000522
3. Ning, L, Kato, Y, Sano, H, Nair, RB, Yoneda, M, Watanabe, S, et al. Spontaneous dissecting aneurysm of middle cerebral artery: a case report with review of the literature. Minim Invasive Neurosurg. (2003) 46:357–60. doi: 10.1055/s-2003-812503
4. Iida, H, Kusumi, M, Sagiuchi, T, and Mitomi, T. Cerebral infarction and fluid collection due to an enlarged dissecting aneurysm of the middle cerebral artery-case report. Neurol Med Chir. (2004) 44:535–9. doi: 10.2176/nmc.44.535
5. Chuang, MJ, Lu, CH, and Cheng, MH. Management of middle cerebral artery dissecting aneurysm. Asian J Surg. (2012) 35:42–8. doi: 10.1016/j.asjsur.2012.04.007
6. Cagnazzo, F, Perrini, P, Dargazanli, C, Lefevre, PH, Gascou, G, Morganti, R, et al. Treatment of Unruptured distal anterior circulation aneurysms with flow-diverter stents: a meta-analysis. AJNR. (2019) 40:687–93. doi: 10.3174/ajnr.A6002
7. Sayin, B, Dereli, B, Senol, YC, Akmangit, I, Karaman, A, Daglioglu, E, et al. The evaluation of low-profile surpass evolve(TM) flow diverter for endovascular treatment of distal cerebral artery aneurysms: a single-center experience. Turk Neurosurg. (2023) 33:477–87. doi: 10.5137/1019-5149.JTN.42071-22.1
8. Li, Q, Lv, N, Li, L, Gu, Y, Xu, L, Lv, M, et al. Intracranial aneurysms managed by parent artery reconstruction using Tubridge flow diverter study: 1-year outcomes. J Neurosurg. (2024) 141:1697–704. doi: 10.3171/2024.3.JNS232116
9. Li, Q, Zhu, D, Lv, N, Yang, P, Zhou, Y, Zhao, R, et al. Clinical outcomes on Tubridge flow diverter in Treatmenting intracranial aneurysms: a retrospective multicenter registry study. Clin Neuroradiol. (2024) 34:465–74. doi: 10.1007/s00062-024-01393-4
10. Xie, D, Yang, H, Zhao, L, Ye, X, Yang, S, Gao, C, et al. Tubridge flow diverter for the treatment of small and medium aneurysms. Front Neurol. (2023) 14:1054631. doi: 10.3389/fneur.2023.1054631
11. Xie, D, Zhao, L, Liu, H, Wang, J, Lu, P, Ye, X, et al. Tubridge flow diverter for the treatment of Unruptured dissecting cerebral aneurysms. World Neurosurg. (2023) 172:e343–8. doi: 10.1016/j.wneu.2023.01.024
12. Gai, Y, Nuerdong, M, Jiang, Y, Wang, W, Pu, B, Xu, F, et al. Flow diversion for unruptured fusiform aneurysms of the proximal middle cerebral artery. Front Neurol. (2023) 14:1325983. doi: 10.3389/fneur.2023.1325983
13. Sacho, RH, Saliou, G, Kostynskyy, A, Menezes, R, Tymianski, M, Krings, T, et al. Natural history and outcome after treatment of unruptured intradural fusiform aneurysms. Stroke. (2014) 45:3251–6. doi: 10.1161/STROKEAHA.114.006292
14. Bamford, JM, Sandercock, PA, Warlow, CP, and Slattery, J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke. (1989) 20:828. doi: 10.1161/01.STR.20.6.828
15. O'Kelly, CJ, Krings, T, Fiorella, D, and Marotta, TR. A novel grading scale for the angiographic assessment of intracranial aneurysms treated using flow diverting stents. Interv Neuroradiol. (2010) 16:133–7. doi: 10.1177/159101991001600204
16. Qi, P, Tong, X, Liang, X, Xue, X, Wu, Z, Feng, X, et al. Flow diversion for posterior circulation aneurysms: a multicenter retrospective study. Ther Adv Neurol Disord. (2023) 16:17562864231176187. doi: 10.1177/17562864231176187
17. Huang, C, Ma, G, Tong, X, Feng, X, Wen, Z, Huang, M, et al. Comparison of pipeline embolization device versus Tubridge embolization device in unruptured intracranial aneurysms: a multicenter, propensity score matched study. J Neurointerv Surg. (2024):jnis-2024-021623. doi: 10.1136/jnis-2024-021623
18. Liu, J, Cao, F, Zhenmei, N, Guo, Y, Li, Y, and Yuan, D. Flow-diverter stents in intracranial aneurysm treatment: impact on covered cerebral artery branches. Int J Surg. (2024) 110:53–65. doi: 10.1097/JS9.0000000000000762
19. Soydemir, E, Gündoğmuş, CA, Türeli, D, Andaç Baltacıoğlu, N, Bayri, Y, and Baltacıoğlu, F. Safety and efficacy of flow diverter stents in the treatment of middle cerebral artery aneurysms: a single-center experience and follow-up data. Diagn Interv Radiol. (2023) 29:350–8. doi: 10.4274/dir.2022.211050
20. Hidayat, R, Nabilah, RA, Rasyid, A, Harris, S, Harahap, AR, Herqutanto, H, et al. Clopidogrel resistance among ischemic stroke patients and its risk factors in Indonesia. Acta Med Acad. (2022) 51:29–34. doi: 10.5644/ama2006-124.367
21. Park, KY, Ozaki, T, Kostynskyy, A, Kortman, H, Hilario, A, Nicholson, P, et al. Ticagrelor versus Clopidogrel in the dual antiplatelet regimen for intracranial stenting or flow-diverter treatment for Unruptured cerebral aneurysms: a single-center cohort study. AJNR. (2021) 42:1638–44. doi: 10.3174/ajnr.A7216
22. Bender, MT, Zarrin, DA, Campos, JK, Lin, LM, Huang, J, Caplan, JM, et al. Tiny pipes: 67 cases of flow diversion for aneurysms in distal vessels measuring less than 2.0 mm. World Neurosurg. (2019) 127:e193–201. doi: 10.1016/j.wneu.2019.02.204
23. Ge, R, Lin, J, Feng, X, Huang, C, Huang, J, Li, C, et al. Analysis of the effect of platelet function and different doses of ticagrelor after flow diverter treatment of intracranial aneurysms. Neurosurg Rev. (2025) 48:90. doi: 10.1007/s10143-025-03225-6
24. King, RM, Peker, A, Epshtein, M, Arends, JM, Brochu, AB, Raskett, CM, et al. Active drug-coated flow diverter in a preclinical model of intracranial stenting. J Neurointerv Surg. (2024) 16:731–6. doi: 10.1136/jnis-2023-020391
25. Guzzardi, G, Galbiati, A, Stanca, C, del Sette, B, Paladini, A, Cossandi, C, et al. Flow diverter stents with hydrophilic polymer coating for the treatment of acutely ruptured aneurysms using single antiplatelet therapy: preliminary experience. Interv Neuroradiol. (2020) 26:525–31. doi: 10.1177/1591019920950878
26. Bhogal, P, Martinez, R, Gansladt, O, Bäzner, H, Henkes, H, and Aguilar, M. Management of Unruptured Saccular Aneurysms of the M1 segment with flow diversion: a single Centre experience. Clin Neuroradiol. (2018) 28:209–16. doi: 10.1007/s00062-016-0553-9
27. Bender, MT, Jiang, B, Campos, JK, Lin, LM, Beaty, N, Vo, CD, et al. Single-stage flow diversion with adjunctive coiling for cerebral aneurysm: outcomes and technical considerations in 72 cases. J Neurointerv Surg. (2018) 10:843–50. doi: 10.1136/neurintsurg-2017-013739
28. Lin, LM, Jiang, B, Bender, MT, Westbroek, EM, Campos, JK, Tamargo, RJ, et al. 47 consecutive cases of pipeline flex flow diversion utilizing a novel large-bore intracranial intermediate catheter: nuances and institutional experience with the Syphontrak. Interv Neurol. (2018) 7:153–63. doi: 10.1159/000486538
29. Cagnazzo, F, Mantilla, D, Lefevre, PH, Dargazanli, C, Gascou, G, and Costalat, V. Treatment of middle cerebral artery aneurysms with flow-diverter stents: a systematic review and Meta-analysis. AJNR. (2017) 38:2289–94. doi: 10.3174/ajnr.A5388
30. Gawlitza, M, Januel, AC, Tall, P, Bonneville, F, and Cognard, C. Flow diversion treatment of complex bifurcation aneurysms beyond the circle of Willis: a single-center series with special emphasis on covered cortical branches and perforating arteries. J Neurointerv Surg. (2016) 8:481–7. doi: 10.1136/neurintsurg-2015-011682
31. Burrows, AM, Zipfel, G, and Lanzino, G. Treatment of a pediatric recurrent fusiform middle cerebral artery (MCA) aneurysm with a flow diverter. J Neurointerv Surg. (2013) 5:e47. doi: 10.1136/neurintsurg-2012-010478.rep
Keywords: dissecting aneurysms, flow diverter, middle cerebral artery, branches, stent
Citation: Duan Y, Huang G, Shen J, Xu Z, Li Z, Li J and Dai D (2025) Tubridge flow diverter for the treatment of dissecting aneurysms in the middle cerebral artery. Front. Neurol. 16:1552610. doi: 10.3389/fneur.2025.1552610
Received: 28 December 2024; Accepted: 03 March 2025;
Published: 27 March 2025.
Edited by:
Kaijun Zhao, Tongji University, ChinaReviewed by:
Jianping Deng, Air Force Medical University, ChinaCopyright © 2025 Duan, Huang, Shen, Xu, Li, Li and Dai. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dongwei Dai, aGRzdHJva2VAMTI2LmNvbQ==; Jian Li, bHVuYWxqQHFxLmNvbQ==
†These authors share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.