
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
MINI REVIEW article
Front. Neurol. , 17 April 2024
Sec. Epilepsy
Volume 15 - 2024 | https://doi.org/10.3389/fneur.2024.1385468
This article is part of the Research Topic Epilepsy Mortality: Leading Causes of Death, Co-morbidities, Cardiovascular Risk and Prevention, Volume II View all 9 articles
The risk of sudden unexpected death in epilepsy (SUDEP) increases with the frequency of generalized tonic–clonic seizures. Carbamazepine (CBZ) and lamotrigine (LTG) have been suggested to increase the risk. However, the prevailing viewpoint is that the choice of antiseizure medication (ASM) does not influence the occurrence. We have explored the approach to addressing this question in relevant studies to evaluate the validity of the conclusions reached. A systematic search was performed in PubMed to identify all controlled studies on SUDEP risk in individuals on CBZ or LTG. Studies were categorized according to whether idiopathic generalized epilepsy (IGE) or females were considered separately, and whether data were adjusted for seizure frequency. Eight studies on CBZ and six studies on LTG were identified. For CBZ, one study showed a significantly increased risk of SUDEP without adjustment for seizure frequency. Another study found significantly increased risk after statistical adjustment for seizure frequency and one study found increased risk with high blood levels. Five other studies found no increase in risk. For LTG, one study showed a significantly increased risk in patients with IGE as opposed to focal epilepsy, and another study showed a significantly increased risk in females. None of the subsequent studies on LTG and none of the studies on CBZ considered females with IGE separately. Taken together the available studies suggest that LTG, and possibly CBZ, may increase occurrence of SUDEP when used in females with IGE. Additional studies with sub-group analysis of females with IGE are needed.
Sudden unexpected death in epilepsy (SUDEP) primarily affects young adults and, among neurological diseases, only stroke causes more loss of life-years (1). In most observed documented cases, it has occurred in relation to a generalized tonic–clonic seizure (GTCS) (2). Typical GTCSs rarely occur in infants and toddlers and are characterized by a shorter tonic phase and by shorter postictal generalized EEG suppression (PGES) providing an explanation why SUDEP is so extremely rare among the youngest individuals with epilepsy (3). The incidence of SUDEP increases with the frequency of GTCSs and seizure control is probably the most effective preventive measure (4, 5). The highest incidence of SUDEP has been found in untreated patients (6) and the use of effective antiseizure medication (ASM) has resulted in the incidence of SUDEP being reduced by more than seven times (7).
The mechanisms underlying this devastating outcome of epilepsy are still incompletely understood. However, there is considerable evidence that central or obstructive apnea or ictal cardiac arrhythmia, alone or in combination with apnea, may be involved (8). In addition, some of these deaths may be explained by ictal electrocerebral shutdown, where persistent epileptiform activity in the electroencephalogram (EEG) is followed by PGES (9). Whether or not treatment with ASM can play a role in causing or triggering SUDEP and whether gender can play a role for the risk has been discussed for several years (10–14). To date, only two ASMs, carbamazepine (CBZ) and lamotrigine (LTG), have been suggested to be implicated. Still, the present prevailing opinion is that choice of ASM does not affect the risk of SUDEP. However, conclusions from studies investigating this question depend on the methods used, the variables taken into consideration, and the way in which the results have been interpreted. The aim of this review is to explore how relevant studies on this topic have approached the question and whether all relevant variables have been taken into consideration. By applying this critical evaluation of these studies, it will be easier to determine the validity of any conclusions that have been reached.
A systematic search was performed in PubMed restricted to English language articles only.
The inclusion criteria were controlled studies that estimated the relative risk of SUDEP in individuals treated with CBZ or LTG as compared with other ASM. No studies fulfilling the inclusion criteria were excluded. The search for relevant studies was concluded October 5, 2023.
We used the search terms «lamotrigine sudden unexpected death epilepsy», «carbamazepine sudden unexpected death epilepsy» and «risk factors sudden unexpected death epilepsy».
The studies fulfilling the inclusion criteria (Figure 1) were categorized according to whether or not separate analyses were made for patients with idiopathic generalized epilepsy (IGE), whether analyses were divided by patient sex, and whether adjustment of results according to seizure frequency was performed.
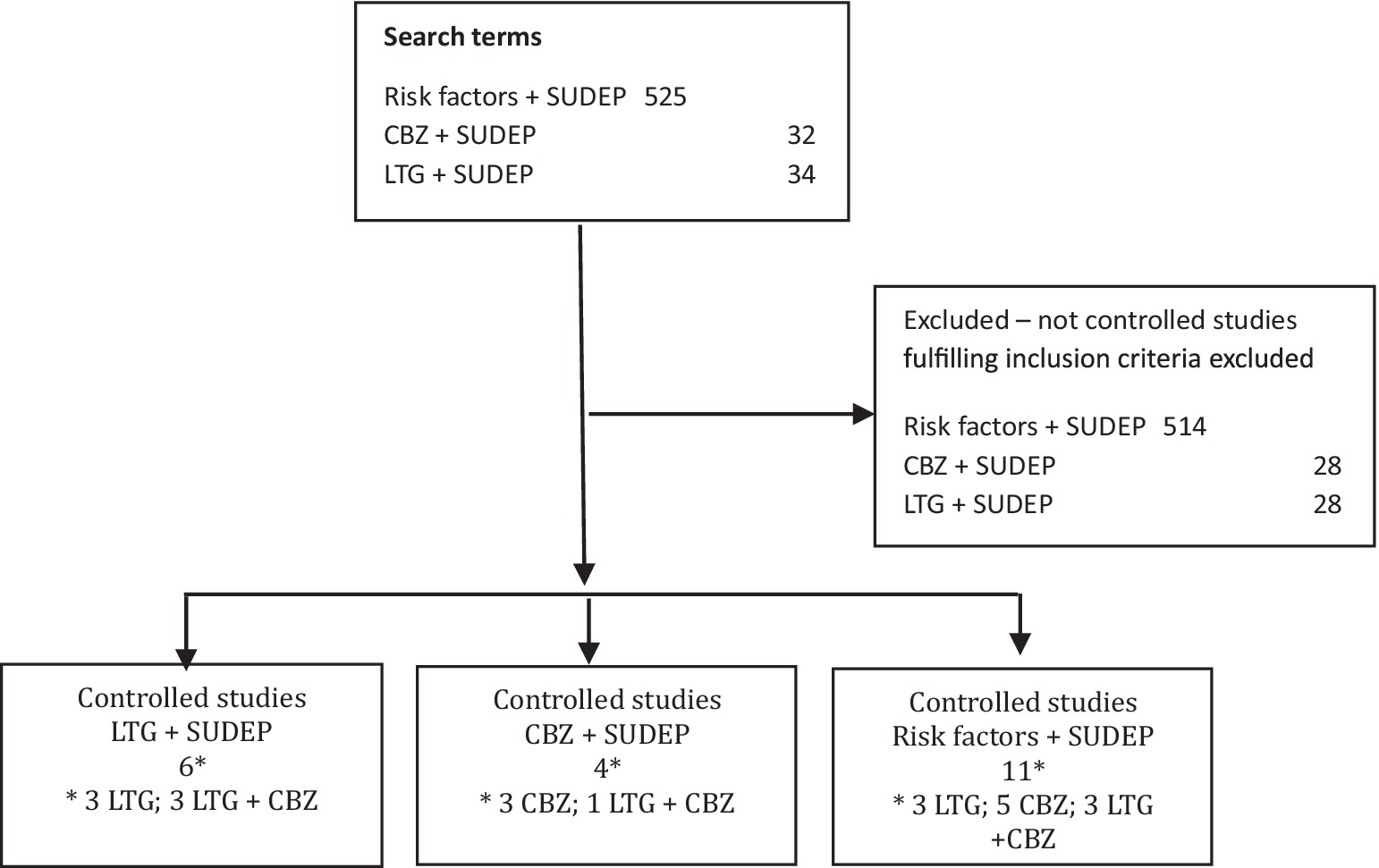
Figure 1. Flow diagram showing the search terms used, number of search results and selected studies. CBZ, carbamazepine; LTG, lamotrigine; SUDEP, sudden unexpected death in epilepsy.
All abstracts from the search results were read, and inclusion of studies fulfilling the inclusion criteria were based on full article texts. The search terms “lamotrigine sudden unexpected death epilepsy” provided 34 results and the search terms “carbamazepine sudden unexpected death epilepsy” provided 32 results of which six and four studies were selected, respectively. The search terms “risk factors sudden unexpected death epilepsy” provided 525 results of which 11 were selected. Only two of these were not identified by the two preceding searches. Five controlled studies fulfilled the inclusion criteria on CBZ, three on LTG, and three on both CBZ and LTG (Figure 1). Table 1 summarizes the selected information researched in the individual selected articles.
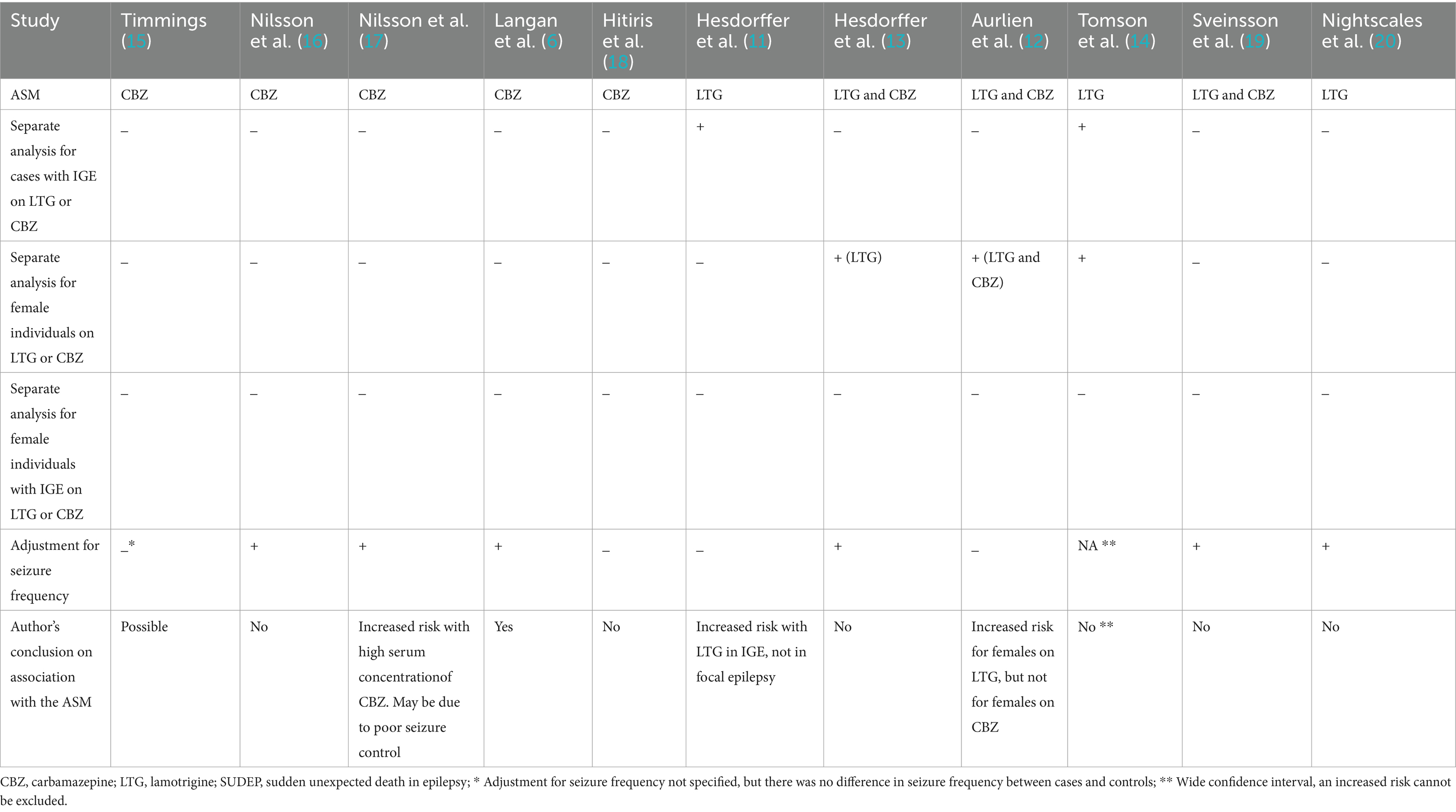
Table 1. Methods and conclusions in studies exploring a possible association between CBZ, LTG and SUDEP.
For CBZ, a study from 1998 showed that treatment with this ASM was associated with a significantly increased risk of SUDEP without adjustment for seizure frequency (15) (Table 1). The majority of those who had died of SUDEP had IGE. A further study found an increased SUDEP risk in those treated with CBZ (6) and another study reported an elevated risk when serum concentrations of CBZ were high (17), with both these studies adjusting for seizure frequency. In contrast, two other studies found no increase in risk of SUDEP associated with CBZ treatment, with or without adjustment for seizure frequency (16, 18). Only one study analyzed females separately and this study found no increase in risk (12). None of the studies included a separate analysis for females with IGE.
For LTG, one study from 2011 showed a significantly increased risk of SUDEP in IGE as opposed to focal epilepsy (11) but did not analyze female gender separately. Another study from 2012 showed a significantly increased risk in females without analyzing females with IGE separately (12). The other four studies on LTG did not include separate analyses for IGE and/or female individuals and/or adjusted for seizure frequency; these studies concluded that there was no association between SUDEP and treatment with LTG (13, 14, 19, 20).
The majority of studies exploring a possible association between ASMs, specifically CBZ and LTG, and SUDEP have concluded that the type of ASM does not influence SUDEP risk (Table 1). Importantly, these studies only examined individuals with epilepsy at the group level and the conclusions were reached without a separate analysis for subgroups (13, 15, 16, 18–20). However, to our knowledge there is no evidence that indicates that the biological vulnerability to ASMs is the same in all individuals with a diagnosis of epilepsy. Indeed, as many epilepsies are caused by mutations in genes encoding for ion channels, and as the mode of action for many ASMs, including CBZ and LTG, is blocking of ion channels (21), there is a need to focus on the subgroups of individuals with IGE. In particular females with IGE are a significant subgroup. Study results have been the subject of considerable debate (10, 12–14). There are important reasons for this:
Discussion on whether particular ASMs could increase SUDEP risk was initiated by Timmings in 1998, when it was found that, among patients who had died of SUDEP, a significantly greater proportion (79%) used CBZ compared to controls (38%; p < 0.01) (15). A separate analysis for IGE was not performed, but a previous report (22) revealed that the majority of the deceased had IGE. There was no difference in seizure frequency between cases and controls. Nevertheless, several studies have documented that the sodium-channel blockers, including CBZ and LTG (21), are of lower efficacy when used in the treatment of IGE (23–25) and may even increase seizure frequency (26). Generally, if a treatment for a potentially lethal disorder has been shown to be less effective than other treatment options, then the argument that choosing the poorer treatment option could increase the risk of premature death is reasonable; the prescribing physician should be aware that this treatment could contribute to the cause of death. In studies comparing the efficacies of two different ASMs, usually only focal epilepsy or generalized epilepsy are included (23, 27, 28), as ASMs can work differently for treating focal epilepsy and generalized epilepsy. It is unclear why this principle is rarely applied in SUDEP research (6, 13, 15–20), and has, to our knowledge, never been explained in the literature.
Several studies on a possible association between SUDEP and treatment with CBZ and/or LTG have not included a separate analysis for IGE (6, 13, 15–20); they have concluded that there is no increase in SUDEP risk associated with these ASMs, and not made any distinction for the subgroup with IGE. In 2011, the largest study to date on ASM and SUDEP was published by Hesdorffer et al. (11) on commission from the International League Against Epilepsy (ILAE) Commission on Epidemiology; Subcommission on Mortality. This study, which included 289 SUDEP victims from four different countries, revealed a significantly increased risk of SUDEP for LTG users with IGE (odds ratio (OR) 2.2; confidence intervals (CI) 1.14–4.23), but not even a tendency towards an increased SUDEP risk for LTG users with focal epilepsy (OR 1.04; CI 0.57–1.92). The authors stated that independent studies were needed to “support or refute” whether LTG is a risk factor for SUDEP. However, only a few months later, the same authors published new analyses based on the same material and this time concluded that there is no connection between choice of ASM and SUDEP (13). Strangely, their contradictory findings from their previous study, indicating an increased risk of SUDEP when LTG was used in IGE patients only, were not discussed, and a separate analysis on the subgroup of IGE patients was not performed. Thus, it seems possible that dilution of their material, by mixing the smaller subgroup of patients previously shown to have an increased SUDEP risk (IGE), with a much larger subgroup with focal epilepsy without an increased risk, explains their finding of a lack of association of SUDEP with LTG treatment. Furthermore, the 2012 study (13) adjusted for differences in seizure frequency between cases and controls and concluded that increased SUDEP risk depends on the number of seizures, and not on the ASM. However, as seizure frequency itself depends upon the efficacy of the ASM, adjustment for seizure frequency thereby entails adjustment for differences in ASM efficacy. Adjusting for seizure frequency only answers the question of whether there is a difference between SUDEP victims and controls, independent of the GTCSs. The relevance of this issue is dubious, as SUDEP occurs in association with GTCSs (2), and it is unclear why this principle of a statistical correction, which implies adjustment for differences in ASM efficacy, has been so widely accepted (6, 13, 16, 17, 19, 20).
Based on Timmings` (15) findings of a significantly higher proportion of SUDEP deaths in patients on CBZ compared to controls and no differences in seizure frequency in the 1998 study, the author suggested that one possible explanation could be a CBZ-induced elongation of the QT interval in the electrocardiogram (ECG), combined with a mild pro-arrhythmic effect of the seizures (15). CBZ has been associated with atrioventricular conduction delay, including potentially life-threatening bradyarrhythmia (29) and also with a slight, but significant, decrease in the QT interval in a study of elderly epilepsy patients (30). Furthermore, CBZ has been shown to reduce heart-rate variability in individuals with epilepsy (31). However, to our knowledge the arrhythmogenic potential of CBZ in IGE has not been explored in either clinical studies or in vitro studies.
In a publication from 2005, Danielsson et al. (32) demonstrated that LTG inhibits the cardiac rapid delayed rectifier potassium ion current (IKr). The authors suggested that by increasing the QT interval in the ECG, LTG could increase the risk of a fatal arrhythmia, possibly primarily in the presence of seizure-induced acidosis (32). The discussion of whether treatment with LTG could increase SUDEP risk in genetically predisposed individuals was initiated by a case series in 2007, where all four of the deceased were females with genetic epilepsy and all had been treated with LTG in monotherapy (10). Among possible explanations for this clinical observation, one suggestion was that LTG, when used in IGE, could increase the risk of cardiac arrhythmia, or provide inferior efficacy, or that a combination of these could have occurred (10). The following year, a study on LTG in clinically relevant doses in healthy subjects showed no increment of the QT interval, and the authors concluded that there is no convincing evidence that LTG is associated with cardiac arrhythmia (33). However, whether their finding of an apparent lack of association was due to the sole inclusion of healthy individuals (no individuals with IGE were included) was not considered. In 2009, postmortem sequencing of long QT syndrome (LQTS) genes from four SUDEP victims with idiopathic epilepsy (10) had revealed in one of the deceased a novel mutation (R523C) in the SCN5A gene that encodes the cardiac voltage-gated sodium channel, Nav1.5 (34). Mutations in this gene are associated with LQTS type 3 and Brugada syndrome (35). As the gene is co-expressed in heart and brain (36), it was suggested that this mutation could both explain the epilepsy and increase the probability of a pro-arrhythmic side effect of LTG.
Recently, a study used whole-cell voltage clamp electrophysiology to examine the influence of three rare genetic variants of the SCN5A gene found in SUDEP victims on channel function (37). The results revealed that these genetic variants significantly influenced Nav1.5 channel function, suggesting a potential to increase the risk of cardiac arrhythmia. Importantly, therapeutic concentrations of LTG also significantly changed the channel function and the authors suggested that LTG may increase the risk of cardiac arrhythmia in individuals carrying mutations in cardiac arrhythmia genes. This study illustrates the need to consider whether or not SUDEP victims were genetically susceptible to cardiac arrhythmia when investigating a potential link between SUDEP and treatment with the sodium and potassium channel blocker LTG (21, 32).
Clinical studies, case reports, and animal studies (36, 38–47) have suggested that several mutations in genes encoding cardiac arrhythmia are co-expressed in heart and brain (8). This means that individuals with a cardiac channelopathy may have genuine epileptic seizures, in addition to episodes with cardiac arrhythmia. Furthermore, individuals with convulsive epileptic seizures and a diagnosis of assumed genetic epilepsy may be particularly vulnerable to cardiac arrhythmia when treated with ion channel blockers. Idiopathic epilepsy may be caused by a diversity of mutations in ion channels (48). In addition to IGE, there are also focal genetic epilepsies that are also caused by ion channel mutations (49, 50). However, in clinical practice, it is usually unknown which ion channel mutation(s) may be present in patients with presumed genetic epilepsy, and whether or not such a mutation is co-expressed in heart and brain. Nevertheless, as some individuals with presumed genetic epilepsy will have a mutation predisposing them to cardiac arrhythmia, caution in prescribing treatment with ion-channel blockers, like LTG and CBZ, is reasonable as they could increase the risk of a pro-arrhythmic side effect in this subgroup of individuals with epilepsy. Thus, whether a cardiocerebral channelopathy may have been present in SUDEP victims is essential to take into account when investigating possible associations between CBZ or LTG treatment and SUDEP.
In 2021, the American Food and Drug Administration (FDA) issued an update to LTG labels, with a warning of an increased risk of life-threatening cardiac arrhythmia in individuals with heart disease, including «cardiac channelopathies such as Brugada syndrome» (Lamictal (lamotrigine): Drug Safety Communication - Studies Show Increased Risk of Heart Rhythm Problems in Patients with Heart Disease | FDA). The warning was based on in vitro studies and mentioned the sodium channel-blocking properties of LTG; however, inhibition of the cardiac potassium ion current IKr (32) was not noted. In response to this FDA warning, the ILAE and the American Epilepsy Society (AES) Task Force published an ad hoc advisory to health professionals to reduce the risk of cardiac arrhythmia in association with LTG treatment (51). Although they reported that they found no clinical studies suggesting a greater risk from LTG compared with other ASM, the studies that were cited had been performed solely on individuals without any known heart conditions (30, 33, 51, 52). In addition, the ad hoc advisory did not emphasize that that cardiac channelopathies are among the conditions associated with an increased risk. A 2022 review of studies on SUDEP, sudden cardiac death, and new or worsened ECG abnormalities in individuals that had been treated with LTG (53) reported that none of the identified studies had discussed subgroups of people with epilepsy or included individuals with heart disease. This study concluded that there is insufficient evidence to state whether LTG treatment increases the risk of these outcomes in these individuals, compared with other ASM or placebo (53). Furthermore, a Danish registry-based study (54) found no evidence that LTG elevates the risk of cardiac conduction disorders in individuals without known cardiac morbidity or increases the mortality from all causes in individuals with pre-existing cardiac disorders. The study included individuals with a prescription for LTG, but a diagnosis of epilepsy was not a prerequisite for inclusion. Consequently, the study was not designed to answer the question of whether LTG increases the risk of SUDEP in patients with IGE.
Similarly, there is a paucity of relevant studies addressing the subgroup of females with IGE (Table 1). An ILAE study from 2012 (13) made a separate analysis for female and male SUDEP victims using LTG and found that the OR for SUDEP in females on LTG in monotherapy was 6.6 (CI 0.3–174.9), whereas the OR was 0.4 in males. Compared with controls, the difference was not statistically significant and therefore the authors concluded that there was no elevated risk in this group. However, they did not consider whether the very wide confidence interval could be due to dilution of their material by combining a smaller subgroup with an increased risk (IGE) with a much larger subgroup with focal epilepsy, and thus without an increased risk of SUDEP. It is possible that a difference could have been detected if the subgroup with IGE had been analyzed separately. A study from Norway (12), published at about the same time as the 2012 study (13), provided further evidence suggesting that the risk of SUDEP differed according to patient sex. In concordance with the ILAE study, there were no increase in OR regarding risk in males, but the incidence of SUDEP was five times higher in women that had been treated with LTG than in women that had not been treated with LTG (p = 0.007). Furthermore, a significantly higher proportion of the female SUDEP victims were being treated with LTG (58%) than the proportion in controls, matched on age and gender (24.4%) (p = 0.038). Whether these findings are mainly due to an inferior efficacy of LTG regarding seizure reduction in this group or whether they reflect a pro-arrhythmic adverse effect in IGE is unclear. However, it could be relevant that the incidence of drug-induced torsade de pointes arrhythmia is up to three times higher in females than in males (56 – 57). The possibility of LTG efficacy being affected by patient sex has not, to our knowledge, been studied.
The majority of studies exploring a possible association between treatment with CBZ or LTG and SUDEP have concluded that there is no such association but have not taken the possibility of important subgroups in the patient material into consideration. However, for LTG, results from studies that have included subgroup analyses, when taken together, suggest that female patients with IGE may be at an increased risk of SUDEP. This could be because of poorer seizure protection or due to a pro-arrhythmic effect in IGE, or both. For CBZ, only one study analyzed females separately, and did not identify any increase in risk of SUDEP. For future studies, the scientific evidence indicates that it would be of value to conduct a separate analysis for female individuals with IGE. Statistical adjustment for differences in seizure frequency between cases and controls means correction for differences in ASM efficacy. As the efficacy of ASM at reducing seizure frequency is crucial for protection against SUDEP, the usefulness of such as an adjustment is questionable.
DA: Writing – review & editing, Writing – original draft. ET: Writing – review & editing, Writing – original draft.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. David, J, Thurman, DJ, Hesdorffer, DC, and French, JA. Sudden unexpected death in epilepsy: assessing the public health burden. Epilepsia. (2014) 55:1479–85. doi: 10.1111/epi.12666
2. Langan, Y, Nashef, L, and Sander, JW. Sudden unexpected death in epilepsy: a series of witnessed deaths. J Neurol Neurosurg Psychiatry. (2000) 68:211–3. doi: 10.1136/jnnp.68.2.211
3. Trivisano, M, Muccioli, L, Ferretti, A, Lee, H-F, Chi, C-S, and Bisulli, F. Risk of SUDEP during infancy. Epilepsy Behav. (2022) 131:107896. doi: 10.1016/j.yebeh.2021.107896
4. Harden, C, Tomson, T, Gloss, D, Buchhalter, J, Cross, JH, Donner, E, et al. Practice Guideline Summary: Sudden Unexpected Death in Epilepsy Incidence Rates and Risk Factors: Report of the Guideline Development, Dissemination, and Implementation Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Epilepsy Curr. (2017) 17:180–7. doi: 10.5698/1535-7511.17.3.180
5. DeGiorgio, CM, Curtis, A, Hertling, D, and Moseley, BD. Sudden unexpected death in epilepsy: risk factors, biomarkers, and prevention. Acta Neurol Scand. (2019) 139:220–30. doi: 10.1111/ane.13049
6. Langan, Y, Nashef, L, and Sander, JW. Case-control study of SUDEP. Neurology. (2005) 64:1131–3. doi: 10.1212/01.WNL.0000156352.61328
7. Ryvlin, P, Cucherat, M, and Rheims, S. Risk of sudden unexpected death in epilepsy in patients given adjunctive antiepileptic treatment for refractory seizures: a meta-analysis of placebo-controlled randomised trials. Lancet Neurol. (2011) 10:961–8. doi: 10.1016/S1474-4422(11)70193-4
8. Pensel, MC, Nass, RD, Taubøll, E, Aurlien, D, and Surges, R. Prevention of sudden unexpected death in epilepsy: current status and future perspectives. Expert Rev Neurother. (2020) 20:497–508. doi: 10.1080/14737175.2020.1754195
9. Kang, JY, Rabiei, AH, Myint, L, and Nei, M. Equivocal significance of post-ictal generalized EEG suppression as a marker of SUDEP risk. Seizure. (2017) 48:28–32. doi: 10.1016/j.seizure.2017.03.017
10. Aurlien, D, Taubøll, E, and Gjerstad, L. Lamotrigine in idiopathic epilepsy - increased risk of cardiac death? Acta Neurol Scand. (2007) 115:199–203. doi: 10.1111/j.1600-0404.2006.00730.x
11. Hesdorffer, DC, Tomson, T, Benn, E, Sander, JW, Nilsson, L, Langan, Y, et al. Combined analysis of risk factors for SUDEP. Epilepsia. (2011) 52:1150–9. doi: 10.1111/j.1528-1167.2010.02952
12. Aurlien, D, Larsen, JP, Gjerstad, L, and Taubøll, E. Increased risk of sudden unexpected death in epilepsy in females using lamotrigine: a nested, case-control study. Epilepsia. (2012) 53:258–66. doi: 10.1111/j.1528-1167.2011.03334.x
13. Hesdorffer, DC, Tomson, T, Benn, E, Sander, JW, Nilsson, L, Langan, Y, et al. Do antiepileptic drugs or generalized tonic-clonic seizure frequency increase SUDEP risk? A combined analysis. Epilepsia. (2012) 53:249–52. doi: 10.1111/j.1528-1167.2011.03354
14. Tomson, T, Hirsch, LJ, Friedman, D, Bester, N, Hammer, A, Irizarry, M, et al. Sudden unexpected death in epilepsy in lamotrigine randomized-controlled trials. Epilepsia. (2013) 54:135–40. doi: 10.1111/j.1528-1167.2012.03689.x
15. Timmings, PL. Sudden unexpected death in epilepsy: is carbamazepine implicated? Seizure. (1998) 7:289–91. doi: 10.1016/s1059-1311(98)80020-4
16. Nilsson, L, Farahmand, BY, Persson, PG, Thiblin, I, and Tomson, T. Risk factors for sudden unexpected death in epilepsy: a case-control study. Lancet. (1999) 353:888–93. doi: 10.1016/s0140-6736(98)05114-9
17. Nilsson, L, Bergman, U, Diwan, V, Farahmand, BY, Persson, PG, and Tomson, T. Antiepileptic drug therapy and its management in sudden unexpected death in epilepsy: a case-control study. Epilepsia. (2001) 42:667–73. doi: 10.1046/j.1528-1157.2001.22000
18. Hitiris, N, Suratman, S, Kelly, K, Stephen, LJ, Sills, GJ, and Brodie, MJ. Sudden unexpected death in epilepsy: a search for risk factors. Epilepsy Behav. (2007) 10:138–41. doi: 10.1016/j.yebeh.2006.11.010
19. Sveinsson, O, Andersson, T, Mattsson, P, Carlsson, S, and Tomson, T. Pharmacologic treatment and SUDEP risk a nationwide, population-based, case-control study. Neurology. (2020) 95:e2509–18. doi: 10.1212/WNL.0000000000010874
20. Nightscales, R, Barnard, S, Laze, J, Chen, Z, Tao, G, Auvrez, C, et al. Risk of sudden unexpected death in epilepsy (SUDEP) with lamotrigine and other sodium channel-modulating antiseizure medications. Epilepsia Open. (2023) 8:334–45. doi: 10.1002/epi4.12693
21. Macdonald, RL, and Kelly, KM. Antiepileptic drug mechanisms of action. Epilepsia. (1995) 36:S2–S12. doi: 10.1111/j.1528-1157.1995.tb05996.x
22. Timmings, PL. Sudden unexpected death in epilepsy: a local audit. Seizure. (1993) 2:287–90. doi: 10.1016/s1059-1311(05)80142-6
23. Tang, L, Ge, L, Wu, W, Yang, X, Rui, P, Wu, Y, et al. Lamotrigine versus valproic acid monotherapy for generalised epilepsy: a meta-analysis of comparative studies. Seizure. (2017) 51:95–101. doi: 10.1016/j.seizure.2017.08.001
24. Mohanraj, R, and Brodie, MJ. Pharmacological outcomes in newly diagnosed epilepsy. Epilepsy Behav. (2005) 6:382–7. doi: 10.1016/j.yebeh.2005.01.008
25. Marson, AG, Al-Kharusi, AM, Alwaidh, M, Appleton, R, Baker, GA, Chadwick, DW, et al. The SANAD study of effectiveness of valproate, lamotrigine, or topiramate for generalised and unclassifiable epilepsy: an unblinded randomised controlled trial. Lancet. (2007) 369:1016–26. doi: 10.1016/S0140-6736(07)60461-9
26. Crespel, A, Genton, P, Berramdane, M, Coubes, P, Monicard, C, Baldy-Moulinier, M, et al. Lamotrigine associated with exacerbation or de novo myoclonus in idiopathic generalized epilepsies. Neurology. (2005) 65:762–4. doi: 10.1212/01.wnl.0000174517.21383.36
27. Privitera, M, Fincham, R, Penry, J, Reife, R, Kramer, L, Pledger, G, et al. Topiramate placebo-controlled dose-ranging trial in refractory partial epilepsy using 600-, 800-, and 1,000-mg daily dosages. Topiramate YE study group. Neurology. (1996) 46:1678–83. doi: 10.1212/wnl.46.6.1678
28. Slater, J, Chung, S, Huynh, L, Duh, MS, Gorin, B, McMicken, C, et al. Efficacy of antiepileptic drugs in the adjunctive treatment of refractory partial-onset seizures: meta-analysis of pivotal trials. Epilepsy Res. (2018) 143:120–9. doi: 10.1016/j.eplepsyres.2017.10.004
29. Kasarskis, EJ, Kuo, CS, Berger, R, and Nelson, KR. Carbamazepine-induced cardiac dysfunction: characterization of two distinct clinical syndromes. Arch Intern Med. (1992) 152:186–91. doi: 10.1001/archinte.152.1.186
30. Saetre, E, Abdelnoor, M, Amlie, JP, Tossebro, M, Perucca, E, Taubøll, E, et al. Cardiac function and antiepileptic drug treatment in the elderly: a comparison between lamotrigine and sustained-release carbamazepine. Epilepsia. (2009) 50:1841–9. doi: 10.1111/j.1528-1167.2009.02069.x
31. Tomson, T, and Kenneback, G. Arrhythmia, heart rate variability, and antiepileptic drugs. Epilepsia. (1997) 38:S48–51. doi: 10.1111/j.1528-1157.1997.tb06128.x
32. Danielsson, BR, Lansdell, K, Patmore, L, and Tomson, T. Effects of the antiepileptic drugs lamotrigine, topiramate and gabapentin on hERG potassium currents. Epilepsy Res. (2005) 63:17–25. doi: 10.1016/j.eplepsyres.2004.10.002
33. Dixon, R, Job, S, Oliver, R, Tompson, D, Wright, JG, Maltby, K, et al. Lamotrigine does not prolong QTc in a thorough QT/QTc study in healthy subjects. Br J Clin Pharmacol. (2008) 66:396–404. doi: 10.1111/j.1365-2125.2008.03250.x
34. Aurlien, D, Leren, TP, Taubøll, E, and Gjerstad, L. New SCN5A mutation in a SUDEP victim with idiopathic epilepsy. Seizure. (2009) 18:158–60. doi: 10.1016/j.seizure.2008.07.008
35. Beaufort-Krol, GC, van den Berg, MP, Wilde, AA, van Tintelen, JP, Viersma, JW, Bezzina, CR, et al. Developmental aspects of long QT syndrome type 3 and Brugada syndrome on the basis of a single SCN5A mutation in childhood. J Am Coll Cardiol. (2005) 46:331–7. doi: 10.1016/j.jacc.2005.03.066
36. Hartmann, HA, Colom, LV, Sutherland, ML, and Noebels, JL. Selective localization of cardiac SCN5A sodium channels in limbic regions of rat brain. Nat Neurosci. (1999) 2:593–5. doi: 10.1038/10147
37. Soh, MS, Bagnall, RD, Semsarian, C, Scheffer, IE, Berkovic, SF, and Reid, CA. Rare sudden unexpected death in epilepsy SCN5A variants cause changes in channel function implicating cardiac arrhythmia as a cause of death. Epilepsia. (2022) 63:e57–62. doi: 10.1111/epi.17254
38. González, A, Aurlien, D, Larsson, PG, Olsen, KB, Dahl, IT, Edvardsen, T, et al. Seizure-like episodes and EEG abnormalities in patients with long QT syndrome. Seizure. (2018) 61:214–20. doi: 10.1016/j.seizure.2018.08.020
39. Johnson, JN, Hofman, N, Haglund, CM, Cascino, GD, Wilde, AA, and Ackerman, MJ. Identification of a possible pathogenic link between congenital long QT syndrome and epilepsy. Neurology. (2009) 72:224–31. doi: 10.1212/01.wnl.0000335760.02995.ca
40. MacCormick, JM, Crawford, JR, Chung, SK, Shelling, AN, Evans, CA, Rees, MI, et al. Symptoms and signs associated with syncope in young people with primary cardiac arrhythmias. Heart Lung Circ. (2011) 20:593–8. doi: 10.1016/j.hlc.2011.04.036
41. González, A, Aurlien, D, Haugaa, KH, and Taubøll, E. Epilepsy in patients with long QT syndrome type 1: a Norwegian family. Epilepsy Behav Case Rep. (2018) 10:118–21. doi: 10.1016/j.ebcr.2018.09.006
42. Heron, SE, Hernandez, M, Edwards, C, Edkins, E, Jansen, FE, Scheffer, IE, et al. Neonatal seizures and long QT syndrome: a cardiocerebral channelopathy. Epilepsia. (2010) 51:293–6. doi: 10.1111/j.1528-1167.2009.02317.x
43. Omichi, C, Momose, Y, and Kitahara, S. Congenital long QT syndrome presenting with a history of epilepsy: misdiagnosis or relationship between channelopathies of the heart and brain. Epilepsia. (2010) 51:289–92. doi: 10.1111/j.1528-1167.2009.02267.x
44. Hindocha, N, Nashef, L, Elmslie, F, Birch, R, Zuberi, S, Al-Chalabi, A, et al. Two cases of sudden unexpected death in epilepsy in a GEFS+ family with an SCN1A mutation. Epilepsia. (2008) 49:360–5. doi: 10.1111/j.1528-1167.2007.01439_2.x
45. Le Gal, F, Korff, CM, Monso-Hinard, C, Mund, MT, Morris, M, Malafosse, A, et al. A case of SUDEP in a patient with Dravet syndrome with SCN1A mutation. Epilepsia. (2010) 51:1915–8. doi: 10.1111/j.1528-1167.2010.02691.x
46. Lehnart, SE, Mongillo, M, Bellinger, A, Lindegger, N, Chen, BX, Hsueh, W, et al. Leaky Ca2+ release channel/ryanodine receptor 2 causes seizures and sudden cardiac death in mice. J Clin Invest. (2008) 118:2230–45. doi: 10.1172/JCI35346
47. Goldman, AM, Glasscock, E, Yoo, J, Chen, TT, Klassen, TL, and Noebels, JL. Arrhythmia in heart and brain: KCNQ1 mutations link epilepsy and sudden unexplained death. Sci Transl Med. (2009) 1:2ra6. doi: 10.1126/scitranslmed.3000289
48. Heron, SE, Scheffer, IE, Berkovic, SF, Dibbens, LM, and Mulley, JC. Channelopathies in idiopathic epilepsy. Neurotherapeutics. (2007) 4:295–304. doi: 10.1016/j.nurt.2007.01.009
49. Zuberi, SM, Eunson, LH, Spauschus, A, De Silva, R, Tolmie, J, Wood, NW, et al. A novel mutation in the human voltage-gated potassium channel gene (Kv1.1) associates with episodic ataxia type 1 and sometimes with partial epilepsy. Brain. (1999) 122:817–25. doi: 10.1093/brain/122.5.817
50. Riva, A, Coppola, A, Balagura, G, Scala, M, Lacomino, M, Marchese, F, et al. Temporal-parietal-occipital epilepsy in GEFS+ associated with SCN1A mutation. Epileptic Disord. (2021) 23:397–401. doi: 10.1684/epd.2021.1266
51. French, JA, Perucca, E, Sander, JW, Bergfeldt, L, Baulac, M, Auerbach, DS, et al. FDA safety warning on the cardiac effects of lamotrigine: an advisory from the ad hoc ILAE/AES task force. Epilepsia Open. (2021) 6:45–8. doi: 10.1002/epi4.12475
52. Svalheim, S, Aurlien, D, Amlie, JP, Gjerstad, L, and Taubøll, E. Signal averaged and standard electrocardiography in patients with newly diagnosed epilepsy. Epilepsy Behav. (2012) 25:543–5. doi: 10.1016/j.yebeh.2012.09.023
53. Bunschoten, JW, Husein, N, Devinsky, O, French, JA, Sander, JW, Thijs, RD, et al. Sudden death and cardiac Arrythmia with lamotrigine: a rapid systematic review. Neurology. (2022) 98:e1748–60. doi: 10.1212/WNL.0000000000200164
54. Christensen, J, Trabjerg, BB, and Dreier, JW. Cardiac morbidity and mortality associated with the use of lamotrigine. Epilepsia. (2022) 63:2371–80. doi: 10.1111/epi.17339
55. Makkar, RR, Fromm, BS, Steinman, RT, Meissner, MD, and Lehmann, MH. Female gender as a risk factor for torsades de pointes associated with cardiovascular drugs. JAMA. (1993) 270:2590–7. doi: 10.1001/jama.270.21.2590
Keywords: antiseizure medication (ASM), SUDEP, carbamazepine, lamotrigine, IGE (idiopathic generalized epilepsy), females
Citation: Aurlien DB and Taubøll E (2024) Antiseizure medication and SUDEP – a need for unifying methodology in research. Front. Neurol. 15:1385468. doi: 10.3389/fneur.2024.1385468
Received: 12 February 2024; Accepted: 03 April 2024;
Published: 17 April 2024.
Edited by:
Rohit Shankar, University of Plymouth, United KingdomReviewed by:
Alessandro Ferretti, Sapienza University of Rome, ItalyCopyright © 2024 Aurlien and Taubøll. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Erik Taubøll, ZXJpay50YXVib2xsQG1lZGlzaW4udWlvLm5v
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.