- Department of Neurology, University of Colorado, Aurora, CO, United States
Movement disorders are a common feature of many antibody-associated neurological disorders. In fact, cerebellar ataxia is one of the most common manifestations of autoimmune neurological diseases. Some of the first autoantibodies identified against antigen targets include anti-neuronal nuclear antibody type 1 (ANNA-1 or anti-Hu) and Purkinje cell cytoplasmic antibody (PCA-1) also known as anti-Yo have been identified in paraneoplastic cerebellar degeneration. Historically these antibodies have been associated with an underlying malignancy; however, recently discovered antibodies can occur in the absence of cancer as well, resulting in the clinical syndrome of autoimmune cerebellar ataxia. The pace of discovery of new antibodies associated with autoimmune or paraneoplastic cerebellar ataxia has increased rapidly over the last few years, and pathogenesis and potential treatment options remains to be explored. Here we will review the literature on recently discovered antibodies associated with autoimmune and paraneoplastic cerebellar ataxia including adaptor protein-3B2 (AP3B2); inositol 1,4,5-trisphophate receptor type 1 (ITPR1); tripartite motif-containing (TRIM) proteins 9, 67, and 46; neurochondrin; neuronal intermediate filament light chain (NIF); septin 5; metabotropic glutamate receptor 2 (mGluR2); seizure-related 6 homolog like 2 (SEZ6L2) and homer-3 antibodies. We will review their clinical characteristics, imaging and CSF findings and treatment response. In addition, we will discuss two clinical case examples of autoimmune cerebellar ataxia.
Introduction
Cerebellar ataxia has a broad differential diagnosis including both acquired and genetic causes, however autoimmune etiologies or paraneoplastic cerebellar degeneration (PCD) should be considered in most case, particularly in adults (1). The first autoantibodies to be identified against neuronal targets include anti-neuronal nuclear antibody type 1 (Anti-Hu) in 1965 (2) and Purkinje cell cytoplasmic antibody (anti-Yo) in 1983 (3) which both represent classic paraneoplastic neurological syndromes. It is hypothesized that cross-reactivity between proteins expressed on the tumor and neuronal antigens is responsible for the development of neurologic symptoms, of which cerebellar ataxia is one of the most common manifestations (4). Since those two autoantibodies were described, the rate of discovery of new autoantibodies to other neuronal targets has been rapidly increasing and there are now several antibodies known to cause cerebellar ataxia both in the setting of malignancy (i.e. paraneoplastic) and in its absence (i.e. idiopathic autoimmune).
Paraneoplastic cerebellar degeneration is a diagnosis under the umbrella of immune-mediated cerebellar ataxias (IMCAs), which also includes post-infectious cerebellitis and gluten ataxia (5). Patients who present with subacute onset cerebellar ataxia but in the absence of malignancy or a known pathogenic antibody are now deemed to have primary autoimmune cerebellar ataxia (PACA) (6). This is in contrast to neuronal antibodies that have already been shown to be directly involved in the pathogenesis of ataxia (e.g. dipeptidyl-peptidase-like-6 [DPPX], metabotropic glutamate receptor 1 [mGluR1], glutamic acid decarboxylase [GAD]-65). See Table 1 for clinical criteria for PACA. These patients tend to present with a subacute onset of gait and limb ataxia as well as dysarthria or nystagmus and they may have objective findings such as inflammatory cerebrospinal fluid (CSF) or magnetic resonance imaging (MRI) showing abnormalities in cerebellum. Patients with suspected PACA should receive immunotherapy trials such as steroids, intravenous immunoglobulins (IVIg) or plasmapheresis (PLEX), although the likelihood of improvement seems to depend on whether the antibody target is a cell surface or an intracellular antigen (5, 6).
The aim of this review article is to focus on recently discovered autoantibodies against neuronal targets identified in syndromes cerebellar ataxia and includes two clinical case examples of adaptor protein-3B2 (AP3B2) and tripartite motif-containing (TRIM)-46. In these the majority cases, an associated antibody has been identified, but it is not yet clear whether there is a direct pathogenic effect with the exception of Metabotropic glutamate receptor-2 (mGluR2).
Table 2 includes an expanded summary on recently described autoantibodies including seen in PACA as well as prominent extracerebellar phenotypes (e.g. Caspr2).
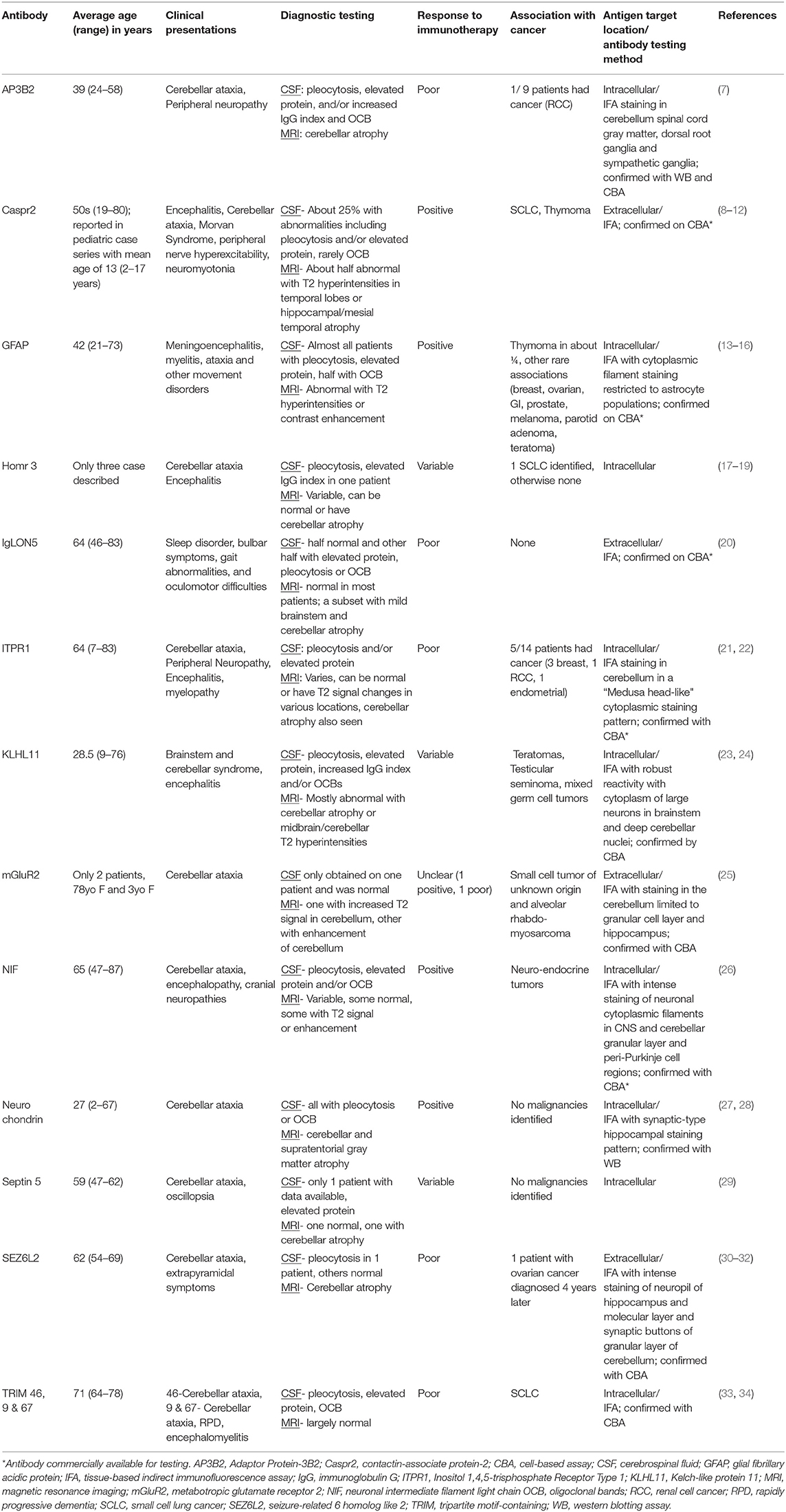
Table 2. Summary table of autoantibodies against neuronal targets identified in syndromes of cerebellar ataxia.
Methods
This review focuses on newly described antibodies in the literature seen in PACA and antibody syndromes in which are newly commercially available for testing – thus our understanding of these antibodies will continue to evolve and these syndrome will likely become more readily recognized. We performed a systematic literature search in PubMed to identify autoantibodies reported against neuronal targets in autoimmune and paraneoplastic cerebellar ataxia after 2014. We also included antibody syndromes that are more recently commercially available in the United States for testing since 2021 (35, 36).
Adaptor Protein-3B2 (AP3B2)
β-neuronal adaptin-like protein, located in neuronal cytoplasm, mediates synaptic vesicle coat assembly. Darnell and colleagues first identified it as an antigen target in 1991 in a 32-year-old woman with subacute progressive ataxia (5). At the time, this was the only known case, but more recently, Honorat and colleagues identified a series of patients with this novel but identical sera and CSF immunofluorescence assay (IFA) staining patterns on mouse nervous system tissue (7). This antigen was identified as β-neuronal adaptin-like protein, now named adaptor protein-3B2 (AP2B2).
All patients reported presented clinically with subacute progressive gait disturbances, including both cerebellar and sensory ataxias. These clinical presentations coincide with the areas of the nervous system that were shown to have greatest staining by IFA; namely the cerebellar Purkinje cells, the dorsal spinal column and dorsal root ganglia. Malignancy in general did not seem to be a precipitant of this autoimmune cerebellar ataxia, with only one patient out of nine found to have an underlying cancer (renal cell carcinoma) (7).
In terms of objective data, CSF was abnormal in all patients. Slight pleocytosis was present in a few patients (median WBC count 7/μL) and others had elevated protein (median 46mg/dL) and increased immunoglobulin-G (IgG) index or oligoclonal bands (7). In the patients who presented with ataxia, brain MRIs showed cerebellar atrophy. Unfortunately, patients treated with immunotherapies did not report improvement, and some were noted to have progressive symptoms despite treatment (7).
Given its intracellular location, AP3B2 antibody is unlikely pathogenic, rather simply a biomarker for CD8+ cytotoxic T cell-mediated damage.
Clinical Case Vignette #1
A 15-year-old female with no significant past medical history presented with progressive left sided weakness and ataxia. One month prior, she noticed her left hand became clumsy, her handwriting became illegible (she is left-handed) and she had difficulty completing fine motor tasks such as painting her nails. She also developed a feeling of “heaviness” in her left leg and was unable to keep up with her teammates at practice. With the progression of her symptoms including worsening gait, she was referred to see a neurologist.
On exam, the patient demonstrated focal left-sided abnormalities including dysmetria with finger-to-nose and heel to shin, dysrhythmia of finger taps and difficulty with rapid alternating movements. Cranial nerves were all intact and there was no dysarthria or truncal ataxia. Brain MRI brain showed mild left cerebellar atrophy as well as a 6 mm T2 hyperintense lesion to the left of the cerebellar vermis (Figure 1). Lumbar puncture revealed inflammatory CSF with WBC 23 (lymphocytic predominance) and 13 unique oligoclonal bands. An extensive infectious workup was unrevealing including Next-Generation sequencing (NGS) (UCSF center for Next-Gen precision for diagnostics). She was started on empiric IV methylprednisolone for treatment of a presumed autoimmune disorder. Positron emission tomography and computed tomography (PET-CT) scan was negative for malignancy, although notably three weeks prior to the onset her neurologic symptoms she did have a pilomatrixoma removed from her neck, which was of unclear significance. Autoantibody testing on her CSF performed at Mayo Clinic Laboratories was positive for an unclassified antibody with synaptic antibody features.
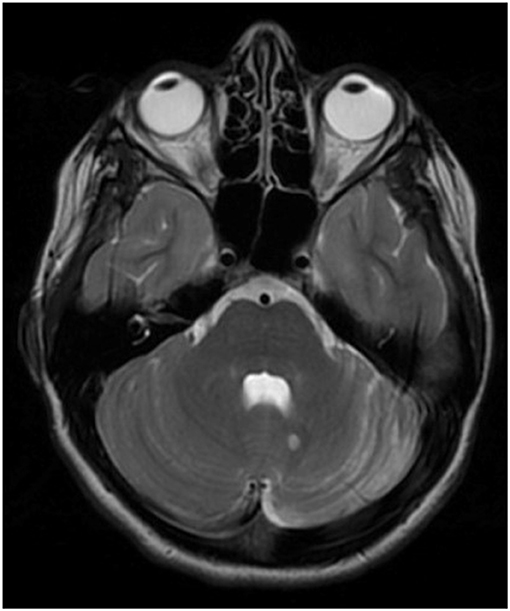
Figure 1. T2 sequence of brain MRI demonstrating hemi-atrophy of the left cerebellum in addition to a 6 mm T2-hyperintense lesion near the cerebellar vermis on the left. Additional brain imaging not show any further T2/FLAIR (fluid-attenuated inversion recovery) abnormalities in the cerebellum, cerebrum or corpus callosum (sequences not shown).
The patient was discharged home after treatment with IV methylprednisolone and was subsequently treated with weekly IV methylprednisolone 1g (as well as one 3 day course of IVIg which she didn't tolerate due to headache) before being started on rituximab at 1,000 mg day 0 and day 14. She responded well to rituximab therapy and has had moderate improvement in coordination on the left and improvement in her gait. She received rituximab therapy over the course of one year and has since remained off immunotherapy with clinical and radiographic follow up over three years. Repeat MRIs have been stable in terms of degree of atrophy in left cerebellum and size of the T2 hyperintense lesion. She is screened yearly for malignancy, which has been negative over the course of three years. Two years after her diagnosis of autoimmune cerebellar ataxia, her CSF evaluation at Mayo Clinic Laboratories revealed a positive AP3B2 antibody. Two unique features seen in this case, not reported in the prior literature, include the hemi-cerebellar atrophy and the positive response to immune therapy.
Inositol 1,4,5-Trisphosphate Receptor Type 1 (ITPR1)
Inositol 1,4,5-trisphosphate receptor type 1 (ITPR1) is an intracellular ligand-gated calcium channel mainly expressed in membranes surrounding the endoplasmic reticulum. It was first identified as an antibody associated with autoimmune cerebellar ataxia in 2014 by Jarius and colleagues after immunohistochemical testing showed binding of IgG1 antibodies in molecular and Purkinje cell layers on animal cerebellum sections in a pattern similar to, but not matching that of, anti-Ca/anti-RhoGTPase-activating protein 26 (ARHGAP26) antibodies (21, 37).
The largest cohort of patients described with ITPR1-IgG antibodies included five who presented with cerebellar ataxia at an average age of 64 (22). Of the five patients with CSF available, four had abnormalities such as pleocytosis or elevated protein. About half of the patients with ITPR1 IgG antibodies had an underlying malignancy including three breast and one renal carcinoma. Immunotherapy response in these patients is reported to be poor with all ten patients who received immunotherapies in this review failing to improve. Interestingly, there was one case report of a 31-year-old woman with a three-year history of slowly progressive cerebellar ataxia found to have ITPR1 antibodies and BRCA1 mutation. Serial malignancy screenings identified a ductal carcinoma 6 years later, which also had significant ITPR1 expression (38).
The fact that ITPR1 antibodies express such high affinity for Purkinje cells in patients who present with cerebellar ataxia and with such high titers suggests that these antibodies could be pathogenic (37). Additionally, ITPR1 mutations are known to be associated with spinocerebellar ataxias. More studies needed to determine whether ITPR1 may indeed be pathogenic.
Tripartite Motif-Containing (TRIM) Proteins 9, 67, and 46
Tripartite motif (TRIM) containing proteins are part of a large group of E3 ubiquitin ligases involved in many different processes such as cellular signaling, carcinogenesis and immunity. TRIM 9 and TRIM 67 have a large role in neuronal development and axonal growth and are highly concentrated in Purkinje cells both in the hippocampus and cerebellum (33). TRIM 46, expressed more diffusely in the central and peripheral nervous system, plays a role in axon growth (34). All three of these specific TRIM proteins have been described as antigen targets in a handful of patients presenting with paraneoplastic cerebellar ataxia.
The few patients described with TRIM9 and TRIM67 have all presented with a subacute onset of severe cerebellar ataxia (33). Presentations with TRIM 46 antibodies seem to be more varied (which could be explained by more diffuse expression of TRIM 46 in the CNS) and has included encephalitis and rapidly progressive dementia, along with cerebellar ataxia (34). Brain imaging is reported as normal in most of these patients or if abnormal may demonstrate some cerebellar atrophy. CSF is often inflammatory with pleocytosis, elevated protein or the presence of oligoclonal bands. These antibodies are strongly associated with malignancy, specifically small-cell lung cancer (33, 34). In the report of two patients with anti-TRIM9 and anti-TRIM67 antibodies, one patient treated with immunotherapy did not show any improvement, and one other patient who had regression of his cancer continued to have severe ataxia years down the line (33). This finding suggests that TRIM9 and TRIM67 are biomarkers of rare cases of paraneoplastic cerebellar ataxia and prompt diagnosis is necessary to try to initiate early immunotherapy, as the mechanism of damage is likely related to CD8+ T cells that seems to cause irreversible neuronal death (33).
Clinical Case Vignette #2
A 63-year-old woman with a one-year history of metastatic endometrial cancer was recently started on pembrolizumab and lenvatinib for treatment of her cancer. Seven months after initiation of pembrolizumab she developed neurological symptoms of vertigo and ataxia. Pembrolizumab was discontinued and she was started on a short course of high-dose oral steroids with no response in her neurological symptoms. She continued to progress with worsening mobility, gait imbalance, diplopia and vertigo with nausea and vomiting so she was referred to Neurology for further evaluation. Brain MRI at the time of her symptom onset was only remarkable for a small T2 lesion the left medulla and no evidence of cerebellar atrophy. Repeat brain MRI at the time of her neurological evaluation revealed subsequent cerebellar atrophy, worsening T2 signal change in the medulla bilaterally (Figure 2), and new T2/FLAIR signal change in the left thalamus. CSF analysis revealed 0 WBCs, mildly elevated protein at 67 mg/dL (ref <45 mg/dL), 6 unique oligoclonal bands, elevated IgG index of 1.27 (ref 0.28–0.66 ratio), and an unclassified antibody identified on IFA screen at Mayo Clinic Laboratories. Subsequent research antibody testing revealed a positive TRIM46 antibody confirmed on cell-based assay (CBA). Novel autoantibody testing completed on a research basis at UCSF laboratories also confirmed the presence of TRIM46-IgG (Figure 3). The patient was subsequently diagnosed with anti-TRIM46 antibody-associated cerebellar ataxia with suspected paraneoplastic etiology vs. pembrolizumab-associated autoimmune cerebellar ataxia. Notably, immune checkpoint inhibitors (ICIs) can cause a variety of clinical phenotypes and various underlying disease mechanisms have been observed. When classic paraneoplastic associations are seen (i.e. classic phenotypes such as cerebellar ataxia with clear antibody profiles associated with cancer), this could potentially be augmented by the anticancer immune response against onconeural antigens (39). This patient will remain off her pembrolizumab given this complication. She received acute treatment with 5 days of IV methylprednisolone and plasma exchange followed by the initiation of monthly IV cyclophosphamide. Two months after her treatment with immune therapy, she demonstrated mild improvement in her mobility (she is able to walk and transfer with assistance after being wheelchair-bound) and improvement of her vertigo. However, at six months after completion of her cyclophosphamide course she remains severely disabled neurologically.
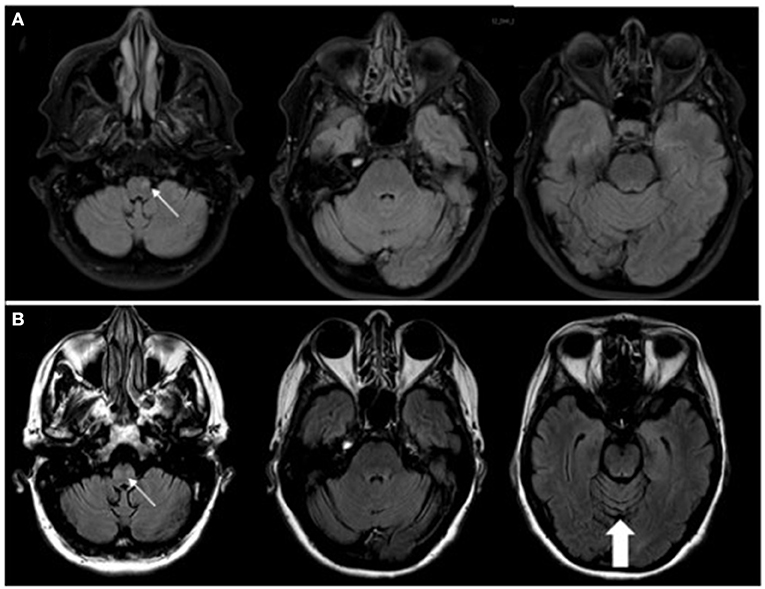
Figure 2. (A) MRI sequences of T2/FLAIR (fluid-attenuated inversion recovery) with STIR (short tau inversion recovery; i.e. fat suppression) at the onset of neurological symptoms of vertigo and ataxia demonstrating a small T2, non-enhancing lesion (contrast imaging not shown) in the left medulla (thin, white arrow). (B) with T2/FLAIR MRI 3 months later demonstrating T2 changes seen bilaterally within the medulla (thin, white arrow) with no associated contrast enhancement. There is noted cerebellar atrophy, particularly seen in the superior cerebellum (thick, white arrow), compared to the MRI from 3 months prior.

Figure 3. Images of (A) 10x magnification and (B) 60x magnification of cerebellar tissue demonstrating antigen staining of the patient's Trim46 antibody (in green) and nuclei are in blue. (C) There are axon initial segments staining in green in the dentate gyrus and (D) in the cortex as well. Images courtesy of Dr. Christopher Bartley and team at University of San Francisco (USCF) Weill Institute for Neurosciences laboratory.
Neurochondrin
Neurochondrin is an intracellular protein expressed in neurons in a somato-dendritic distribution. In concert with G-protein-coupled receptors such as mGluR1 and mGluR5, it has been shown to have an important role in synaptic plasticity, particularly in the cerebellum (27).
There have been two small cohorts of patients described that were found to have anti-neurochondrin antibodies. They all presented with a rapidly progressive cerebellar ataxia, and a few demonstrated brainstem findings such as eye movement abnormalities or dysphagia. CSF had an inflammatory profile in all patients, including pleocytosis, elevated protein, oligoclonal bands and/or increased IgG index. While some MRIs were initially normal or showed increased T2/FLAIR signal (mainly in pons, midbrain, and hippocampus), almost all patients eventually developed significant cerebellar atrophy on subsequent MRI scans (27, 28). Malignancy was only identified in one patient (uterine carcinoma) (28). Unfortunately, none of the patients treated with immunotherapy showed any signs of improvement.
Given its intracellular expression, damage related to anti-neurochondrin IgG antibodies is likely mediated by a cytotoxic T-cell response.
Neuronal Intermediate Filament Light Chain
Neuronal intermediate filaments (NIF) are a group of proteins that are integral to the structure and function of neurons in the central and peripheral nervous systems. Neurofilament light chain proteins are a subtype of NIF proteins that have been implicated as antigen targets in patients with cerebellar ataxia and encephalopathy (26).
Subacute but rapidly progressive cerebellar ataxia as well as encephalopathy were the two most common clinical presentations in a large cohort of patients described in 2018 (26). Of the 9 patients with data available, 5 had an abnormal MRI consisting of cerebellar atrophy and/or T2 hyperintensities in patients with ataxia (26). Most patients had CSF abnormalities including lymphocytic pleocytosis (median 41.5 WBCs in patients with CA) and/or CSF-restricted oligoclonal bands and increased protein (median 116 mg/dL) (26). NIF-IgG was strongly associated with malignancy, with cancer in 77% of these patients, most commonly neuroendocrine tumors including small-cell lung cancer. Patients with NIF antibodies with reported immunotherapy treatment tended to have improvement of their neurologic symptoms, which is less common for paraneoplastic neurologic disorders mediated by antibodies to intracellular antigens (26).
Septin 5
Septins are a group of cytoskeletal GTP-binding proteins with many different functions; although in the CNS they seem to have an important role in synaptic vesicle formation and exocytosis (40). The anti-Septin 5 antibody was identified by Honorat and colleagues at Mayo Clinic when indirect IFA of patient's sera and CSF demonstrated a novel staining pattern of synaptic regions of mouse cerebrum and cerebellum (29).
Anti-septin 5 antibodies have been identified in only a handful of patients. Clinically these patients all presented with a rapidly progressive cerebellar syndrome, including two with oscillopsia. CSF was only available for one patient and was significant for increase IgG synthesis rate. MRI imaging was only available for two patients, one of which was normal and the other showing cerebellar atrophy. There were no malignancies identified in any of the patients. In terms of treatment, only two patients were treated with immunotherapies which resulted in significant improvement for one patient, but only transient improvement in the other patient who ended up dying 6 months later (29).
Although septin-5 is largely expressed intracellularly, it is revealed extracellularly during exocytosis. It is therefore unclear whether septin-5 antibodies directly pathogenic or if they mediate damage via cytotoxic T-cells.
Metabotropic Glutamate Receptor 2 (mGluR2)
Metabotropic glutamate receptors (mGluR) are a family of cell surface G-protein-coupled receptors that bind glutamate and therefore play a significant role in synaptic transmission and neuronal excitability. Metabotropic glutamate receptor 1 was the first to be identified in patients with cerebellar ataxia in the early 2000s. More recently in 2019, mGluR2 was described in two patients with subacute onset cerebellar ataxia (25). There are 8 subtypes of mGluRs divided into 3 subgroups: Group I includes mGluR1 and 5 in which both are autoimmune targets in cerebellar ataxia and encephalitis associated with Hodgkin lymphoma (41, 42); Group II comprises of mGluR2 and 3; and Group III includes the remaining subtypes. The main physiologic role of mGluR2 is to modulate glutamatergic and GABAergic synaptic transmission and mGluR2 antibodies could potentially alter these functions (25, 43). Immunohistochemical studies in rat cerebellar cortex demonstrated mGluR2 and 3 immunoreactivity in the cerebellar cortex localizing to the Golgi cells with the majority of the Golgi cells distributed mainly in the Purkinje cell layer and superficial part of the granular layer (44).
The two patients described with anti-mGluR2 antibodies are on opposite ends of the spectrum in terms of age; one patient was a 78-year-old woman and the other a 3-year-old girl. The older woman presented initially with intermittent episodes of ataxia that eventually became progressive and constant. The young girl developed ataxia and dysarthria after a 3-day prodrome of fever, nausea and vomiting. Both patient's MRIs were abnormal with hyperintense T2 cerebellar lesions including patchy enhancement of cerebellum in the 3-year-old. CSF was unavailable for the older patient but was normal in the young girl. Both received immunotherapy with drastically different outcomes. The older woman had progressive symptoms unresponsive to therapies while the young girl had complete recovery after receiving IV methylprednisolone and IVIg. Malignancy was identified in both patients after the development of neurologic symptoms. Small-cell neuroendocrine cancer of unknown origin was discovered on inguinal node biopsy in the 78 year-old woman three years after symptom onset, and the young girl was found to have alveolar rhabdomyosarcoma one year later (25).
Unlike other paraneoplastic cerebellar ataxias described here, mGluR2 antibodies target an extracellular antigen. Like anti-mGluR1 antibodies, mGluR2 antibodies likely have a direct pathogenic effect.
Seizure-Related 6 Homolog Like 2 (SEZ6L2)
SEZ6L2 is highly expressed in the hippocampus and cerebellar cortex and is an auxiliary subunit of the α-Amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor (30). SEZ6L2 modulates AMPA receptor function by binding to adducin and glutamate receptor 1 (45). In 2014, SEZ6L2 antibodies were identified in a patient with subacute cerebellar ataxia and retinopathy, and later in another patient with cerebellar ataxia associated with hypomimia, bradykinesia, and postural instability (30–32). Since then, Landa and colleagues described four more cases in 2020 with SEZ6L2 antibodies identified in serum and CSF by immunohistochemistry on rate brain sections and immunoprecipitation from rat cerebellar neurons; all patients were then identified on CBA with unclassified neuropil antibodies (32). The median age of these four patients was 62 years old and patients presented with a subacute gait ataxia, dysarthria and mild extrapyramidal symptoms (32). Only one out of four patients had a CSF pleocytosis and three patients had evidence of moderate cerebellar atrophy with no evidence of contrast enhancement or other inflammatory features (32). In these series, none of the patients improved with immunotherapy (32).
Homer-3
Homer-3 is expressed at a high level in Purkinje cells (17, 37). Anti-Homer-3 antibody-associated cerebellar ataxia is rare and only reported in three cases (17–19, 37). The first case was presented in a 65-year-old female with vertigo, vomiting, dysarthria and severe subacute limb and gait ataxia (18). A second case was then reported in a 38-year-old man with a pancerebellar syndrome followed by encephalitis, seizures, and papilledema (19). Later a third case was described in a 51-year-old women with a pancerebellar syndrome (17). CSF analysis was abnormal in all cases including lymphocytic pleocytosis in two and oligoclonal bands in the last case. Follow up MRI was available in two patients, both reporting cerebellar atrophy. The 65-year-old woman had no improvement with steroids, while the other two patients were reported to have a partial improvement with immunotherapy which included a combination of steroids, IVIg, and mycophenolate mofetil (MMF) (17–19, 37).
Other Antibodies Associated With Cerebellar Ataxia
There are a handful of other autoantibodies that are associated with symptoms of cerebellar ataxia, although clinical manifestation of these antibody syndromes predominately present with extracerebellar phenotypes (Table 2). IgLON5 has a unique presentation consisting mainly of sleep disorders, bulbar symptoms and gait abnormalities (not necessarily all attributed to cerebellar dysfunction). Cerebellar ataxia in anti-IgLON5 disease is rare, however it has been described along with MRI findings of cerebellar atrophy (20). In anti-contactin-associated protein-like 2(Caspr2) antibody syndrome, unique symptoms such as peripheral nerve hyperexcitability and neuromyotonia tend to accompany the encephalitis and ataxia (8, 46). In a cohort of 37 patients with anti-Caspr2 antibodies (20 with encephalitis and 17 with neuromyotonia), 5 patients with transient symptoms suggestive of cerebellar impairment suggesting that cerebellar ataxia could be observed in up to 25% of patients (9). Kelch-like protein 11 (KLHL11) have been identified as a biomarker of a paraneoplastic brainstem cerebellar syndrome associated with testicular seminoma (23). While often presenting with cerebellar ataxia, more recently expanded phenotypes in KLHL11 have been described including co-existing anti-NMDAR encephalitis, brainstem diencephalic encephalitis, opsoclous-myoclonus, limbic and extralimbic encephalitis and chronic psychosis in the setting of teratomas (24). Autoimmune glial fibrillary acidic protein (GFAP) astrocytopathy is a newly described autoimmune meningoencephalomyelitis syndrome associated with ataxia in 40% of reported cases (13).
In terms of paraclinical data, the majority of patients with GFAP and KLHL11 antibodies had an inflammatory CSF profile (including pleocytosis, elevated protein, OCBs and/or elevated IgG index), while only about half of patients with IgLON5 and a quarter of patients with Caspr2 antibodies had these same abnormalities. Cerebellar atrophy was one of the more common MRI findings along with T2/FLAIR signal changes in the cerebellum, brainstem, or hippocampi. In GFAP astrocytopathy the majority of patients have characteristic T1 postgadolinium enhancement including patterns of radial periventricular, leptomeningeal and punctate, and serpiginous and periependymal enhancement; spinal cord enhancement can also be seen often with a central cord pattern (13).
Discussion
Here we have reviewed recently described autoantibodies associated with cerebellar ataxia including anti-AP3B2, anti-ITPR1, anti-TRIM, anti-neurochondrin, anti-NIF, anti-septin 5,anti-mGluR2, anti-SEZ6L2, and anti-homer 3. Clinically, patients present with a subacute to rapidly progressive cerebellar ataxia. The majority of cases reported an inflammatory CSF profile with a lymphocytic pleocytosis and/or the presence of unique oligoclonal bands. Imaging abnormalities are more variable but when present often involve T2-hyperintensities or atrophy of the cerebellum.
Main differences between the antibodies described lies in their associations with malignancy and reported responses to treatment. Malignancy was less common in patients with anti-AP3B2 antibody-associated cerebellar ataxia with only 1 in 9 patients identified as having an underlying cancer. On the other hand, anti-NIF antibodies are strongly associated with neuroendocrine tumors and the few patients described with anti-TRIM antibodies were all found to have lung adenocarcinomas. ITPR1 antibodies seem to be associated with breast cancer although reports of malignancy types were more variable. Both patients with mGluR2 antibodies had malignancies of different types. No malignancies have been reported with anti-neurochondrin and with anti-SEZ6L2 only 1 patient was identified with a ovarian cancer, diagnosed 4 years later. In contrast, KLHL11 has a strong association with testicular seminoma in men.
Most of the described antibodies in this review, except for mGluR2 and SEZ6L2 (see details in Table 2), are directed against intracellular antigens. It is widely hypothesized, that antibodies against intracellular targets are not directly pathogenic and rather mediate damage via cytotoxic T-cells, although there has been some evidence to dispute this assumption. For example, studies have shown that neurons are able to take up IgG containing anti-Yo, which subsequently resulted in Purkinje cell death (39). The same direct cytotoxic effect has also been found with anti-Hu antibodies (27), although it remains unclear exactly how these antibodies cause destruction of neurons. Regardless of whether the antibodies themselves or cytotoxic T-cells are mediating the damage, these conditions are often reported to be less responsive to immunotherapies. This appears to hold true of the newly discovered antibodies described in this review with the only exception being anti-NIF antibody cerebellar ataxias, where most patients had improvement of symptoms with immunotherapy. Furthermore, while SEZ6L2 is an extracellular antibody, the immunotherapy response in the 4 reported patients was poor.
In general, autoimmune and paraneoplastic cerebellar ataxias result in progressive and debilitating symptoms, therefore, it is crucial for clinicians to be aware of this diagnosis so treatment can be started as early as possible, ideally before significant, irreversible neuronal cell death has taken place. Any patient presenting with a subacute onset of cerebellar ataxia without a family history of genetic ataxia, an autoimmune pathogenesis should be considered. The absence of an antibody does not exclude neurological autoimmunity and if suspected immunotherapy should be considered, especially if the diagnostic work up reveals an inflammatory CSF profile. Equally important, these patients need screening for underlying malignancy as the neurologic syndrome is often the first indication of a neoplasm. There remains much to learn regarding pathogenesis and exact mechanisms of neuronal destruction so more effective therapies can be investigated, ideally through robust randomized-controlled trials.
Author Contributions
MG and AP drafted and revised the manuscript for intellectual content and direct patient care. Both authors contributed to the article and approved the submitted version.
Funding
This work was supported in part by the Drake family in the name of Susan Drake and through the Rocky Mountain MS Center at the University of Colorado.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
We would like to thank Dr. Christopher Bartley and colleagues in Dr. Michael Wilson's laboratory at the University of San Francisco (USCF) Weill Institute for Neurosciences for providing novel autoantibody testing and the images provided in Figure 3. The authors would also like to thank Dr. Divyanshu Dubey and colleagues at the Mayo Clinic Neuroimmunology Laboratory for providing identification of the unclassified antibodies in both clinical cases presented in this manuscript.
References
1. Lopez-Chiriboga AS, McKeon A. Autoimmune and paraneoplastic movement disorders. In: Piquet A, Alvarez E, editors. Neuroimmunology: Multiple Sclerosis, Autoimmune Neurology and Related Disease. 1. Switzerland: Springer Nature. (2021). p. 207–20. doi: 10.1007/978-3-030-61883-4_14
2. Wilkinson PC, Zeromski J. Immunofluorescent detection of antibodies against neurones in sensory carcinomatous neuropathy. Brain. (1965) 88:529–83. doi: 10.1093/brain/88.3.529
3. Greenlee JE, Brashear HR. Antibodies to cerebellar Purkinje cells in patients with paraneoplastic cerebellar degeneration and ovarian carcinoma. Ann Neurol. (1983) 14:609–13. doi: 10.1002/ana.410140603
4. Galli J, Greenlee J. Paraneoplastic diseases of the central nervous system. F1000Res. (2020) 9:F1000. doi: 10.12688/f1000research.21309.1
5. Mitoma H, Hadjivassiliou M, Honnorat J. Guidelines for treatment of immune-mediated cerebellar ataxias. Cerebellum Ataxias. (2015) 2:14. doi: 10.1186/s40673-015-0034-y
6. Hadjivassiliou M, Graus F, Honnorat J, Jarius S, Titulaer M, Manto M, et al. Diagnostic criteria for primary autoimmune cerebellar ataxia-guidelines from an international task force on immune-mediated cerebellar ataxias. Cerebellum. (2020) 19:605–10. doi: 10.1007/s12311-020-01132-8
7. Honorat JA, Lopez-Chiriboga AS, Kryzer TJ, Komorowski L, Scharf M, Hinson SR, et al. Autoimmune gait disturbance accompanying adaptor protein-3B2-IgG. Neurology. (2019) 93:e954–e63. doi: 10.1212/WNL.0000000000008061
8. Boyko M, Au KLK, Casault C, de Robles P, Pfeffer G. Systematic review of the clinical spectrum of CASPR2 antibody syndrome. J Neurol. (2020) 267:1137–46. doi: 10.1007/s00415-019-09686-2
9. Joubert B, Gobert F, Thomas L, Saint-Martin M, Desestret V, Convers P, et al. Autoimmune episodic ataxia in patients with anti-CASPR2 antibody-associated encephalitis. Neurol Neuroimmunol Neuroinflamm. (2017) 4:e371. doi: 10.1212/NXI.0000000000000371
10. López-Chiriboga AS, Klein C, Zekeridou A, McKeon A, Dubey D, Flanagan EP, et al. LGI1 and CASPR2 neurological autoimmunity in children. Ann Neurol. (2018) 84:473–80. doi: 10.1002/ana.25310
11. Syrbe S, Stettner GM, Bally J, Borggraefe I, Bien CI, Ferfoglia RI, et al. CASPR2 autoimmunity in children expanding to mild encephalopathy with hypertension. Neurology. (2020) 94:e2290–e301. doi: 10.1212/WNL.0000000000009523
12. Lancaster E, Huijbers MG, Bar V, Boronat A, Wong A, Martinez-Hernandez E, et al. Investigations of caspr2, an autoantigen of encephalitis and neuromyotonia. Ann Neurol. (2011) 69:303–11. doi: 10.1002/ana.22297
13. Kunchok A, Zekeridou A, McKeon A. Autoimmune glial fibrillary acidic protein astrocytopathy. Curr Opin Neurol. (2019) 32:452–8. doi: 10.1097/WCO.0000000000000676
14. Flanagan EP, Hinson SR, Lennon VA, Fang B, Aksamit AJ, Morris PP, et al. Glial fibrillary acidic protein immunoglobulin G as biomarker of autoimmune astrocytopathy: analysis of 102 patients. Ann Neurol. (2017) 81:298–309. doi: 10.1002/ana.24881
15. Dubey D, Hinson SR, Jolliffe EA, Zekeridou A, Flanagan EP, Pittock SJ, et al. Autoimmune GFAP astrocytopathy: Prospective evaluation of 90 patients in 1 year. J Neuroimmunol. (2018) 321:157–63. doi: 10.1016/j.jneuroim.2018.04.016
16. Fang B, McKeon A, Hinson SR, Kryzer TJ, Pittock SJ, Aksamit AJ, et al. Autoimmune Glial Fibrillary Acidic Protein Astrocytopathy: A Novel Meningoencephalomyelitis. JAMA Neurol. (2016) 73:1297–307. doi: 10.1001/jamaneurol.2016.2549
17. Xu X, Ren H, Li L, Wang J, Fechner K, Guan H. Anti-Homer-3 antibody associated cerebellar ataxia: a rare case report and literature review. J Neuroimmunol. (2019) 330:155–8. doi: 10.1016/j.jneuroim.2019.01.002
18. Höftberger R, Sabater L, Ortega A, Dalmau J, Graus F. Patient with homer-3 antibodies and cerebellitis. JAMA Neurol. (2013) 70:506–9. doi: 10.1001/jamaneurol.2013.1955
19. Zuliani L, Sabater L, Saiz A, Baiges JJ, Giometto B, Graus F. Homer 3 autoimmunity in subacute idiopathic cerebellar ataxia. Neurology. (2007) 68:239–40. doi: 10.1212/01.wnl.0000251308.79366.f9
20. Gaig C, Graus F, Compta Y, Högl B, Bataller L, Brüggemann N, et al. Clinical manifestations of the anti-IgLON5 disease. Neurology. (2017) 88:1736–43. doi: 10.1212/WNL.0000000000003887
21. Jarius S, Scharf M, Begemann N, Stöcker W, Probst C. Serysheva, II, et al. Antibodies to the inositol 1,4,5-trisphosphate receptor type 1 (ITPR1) in cerebellar ataxia. J Neuroinflammation. (2014) 11:206. doi: 10.1186/s12974-014-0206-3
22. Alfugham N, Gadoth A, Lennon VA, Komorowski L, Scharf M, Hinson S, et al. ITPR1 autoimmunity: Frequency, neurologic phenotype, and cancer association. Neurol Neuroimmunol Neuroinflamm. (2018) 5:e418. doi: 10.1212/NXI.0000000000000418
23. Mandel-Brehm C, Dubey D, Kryzer TJ, O'Donovan BD, Tran B, Vazquez SE, et al. Kelch-like Protein 11 Antibodies in Seminoma-Associated Paraneoplastic Encephalitis. N Engl J Med. (2019) 381:47–54. doi: 10.1056/NEJMoa1816721
24. Maudes E, Landa J, Muñoz-Lopetegi A, Armangue T, Alba M, Saiz A, et al. Clinical significance of Kelch-like protein 11 antibodies. Neurol Neuroimmunol Neuroinflammation. (2020) 7:e666. doi: 10.1212/NXI.0000000000000666
25. Ruiz-García R, Martínez-Hernández E, Joubert B, Petit-Pedrol M, Pajarón-Boix E, Fernández V, et al. Paraneoplastic cerebellar ataxia and antibodies to metabotropic glutamate receptor 2. Neurol Neuroimmunol Neuroinflamm. (2019) 7:e658. doi: 10.1212/NXI.0000000000000658
26. Basal E, Zalewski N, Kryzer TJ, Hinson SR, Guo Y, Dubey D, et al. Paraneoplastic neuronal intermediate filament autoimmunity. Neurology. (2018) 91:e1677–e89. doi: 10.1212/WNL.0000000000006435
27. Miske R, Gross CC, Scharf M, Golombeck KS, Hartwig M, Bhatia U, et al. Neurochondrin is a neuronal target antigen in autoimmune cerebellar degeneration. Neurol Neuroimmunol Neuroinflamm. (2017) 4:e307. doi: 10.1212/NXI.0000000000000307
28. Shelly S, Kryzer TJ, Komorowski L, Miske R, Anderson MD, Flanagan EP, et al. Neurochondrin neurological autoimmunity. Neurol Neuroimmunol Neuroinflamm. (2019) 6:e612. doi: 10.1212/NXI.0000000000000612
29. Honorat JA, Lopez-Chiriboga AS, Kryzer TJ, Fryer JP, Devine M, Flores A, et al. Autoimmune septin-5 cerebellar ataxia. Neurol Neuroimmunol Neuroinflamm. (2018) 5:e474. doi: 10.1212/NXI.0000000000000474
30. Yaguchi H, Yabe I, Takahashi H, Watanabe M, Nomura T, Kano T, et al. Anti-Sez6l2 antibody detected in a patient with immune-mediated cerebellar ataxia inhibits complex formation of GluR1 and Sez6l2. J Neurol. (2018) 265:962–5. doi: 10.1007/s00415-018-8785-z
31. Borsche M, Hahn S, Hanssen H, Münchau A, Wandinger KP, Brüggemann N. Sez6l2-antibody-associated progressive cerebellar ataxia: a differential diagnosis of atypical parkinsonism. J Neurol. (2019) 266:522–4. doi: 10.1007/s00415-018-9115-1
32. Landa J, Guasp M, Petit-Pedrol M, Martínez-Hernández E, Planagumà J, Saiz A, et al. Seizure-related 6 homolog like 2 autoimmunity: Neurologic syndrome and antibody effects. Neurol Neuroimmunol Neuroinflamm. (2021) 8. doi: 10.1212/NXI.0000000000000916
33. Do LD, Gupton SL, Tanji K, Bastien J, Brugière S, Couté Y, et al. TRIM9 and TRIM67 are new targets in paraneoplastic cerebellar degeneration. Cerebellum. (2019) 18:245–54. doi: 10.1007/s12311-018-0987-5
34. van Coevorden-Hameete MH, van Beuningen SFB, Perrenoud M, Will LM, Hulsenboom E, Demonet JF, et al. Antibodies to TRIM46 are associated with paraneoplastic neurological syndromes. Ann Clin Transl Neurol. (2017) 4:680–6. doi: 10.1002/acn3.396
35. Honorat JA, McKeon A. Autoimmune movement disorders: a clinical and laboratory approach. Curr Neurol Neurosci Rep. (2017) 17:4. doi: 10.1007/s11910-017-0709-2
36. Movement Disorder Evaluation Algorithm Mayo Foundation for Medical Education and Research (2021). Available online at: https://www.mayocliniclabs.com/it-mmfiles/Movement_Disorder_Evaluation_Algorithm__Serum.pdf. (accessed July 1, 2021)
37. Jarius S, Wildemann B. ‘Medusa head ataxia': the expanding spectrum of Purkinje cell antibodies in autoimmune cerebellar ataxia. Part 1: Anti-mGluR1, anti-Homer-3, anti-Sj/ITPR1 and anti-CARP VIII. J Neuroinflammation. (2015) 12:166. doi: 10.1186/s12974-015-0356-y
38. Berzero G, Hacohen Y, Komorowski L, Scharf M, Dehais C, Leclercq D, et al. Paraneoplastic cerebellar degeneration associated with anti-ITPR1 antibodies. Neurol Neuroimmunol Neuroinflamm. (2017) 4:e326. doi: 10.1212/NXI.0000000000000326
39. Sechi E, Markovic SN, McKeon A, Dubey D, Liewluck T, Lennon VA, et al. Neurologic autoimmunity and immune checkpoint inhibitors: Autoantibody profiles and outcomes. Neurology. (2020) 95:e2442–e52. doi: 10.1212/WNL.0000000000010632
40. Dolat L, Hu Q, Spiliotis ET. Septin functions in organ system physiology and pathology. Biol Chem. (2014) 395:123–41. doi: 10.1515/hsz-2013-0233
41. Spatola M, Sabater L, Planagumà J, Martínez-Hernandez E, Armangué T, Prüss H, et al. Encephalitis with mGluR5 antibodies: Symptoms and antibody effects. Neurology. (2018) 90:e1964–e72. doi: 10.1212/WNL.0000000000005614
42. Sillevis Smitt P, Kinoshita A, De Leeuw B, Moll W, Coesmans M, Jaarsma D, et al. Paraneoplastic cerebellar ataxia due to autoantibodies against a glutamate receptor. N Engl J Med. (2000) 342:21–7. doi: 10.1056/NEJM200001063420104
43. Ferraguti F, Shigemoto R. Metabotropic glutamate receptors. Cell Tissue Res. (2006) 326:483–504. doi: 10.1007/s00441-006-0266-5
44. Ohishi H, Ogawa-Meguro R, Shigemoto R, Kaneko T, Nakanishi S, Mizuno N. Immunohistochemical localization of metabotropic glutamate receptors, mGluR2 and mGluR3, in rat cerebellar cortex. Neuron. (1994) 13:55–66. doi: 10.1016/0896-6273(94)90459-6
45. Yaguchi H, Yabe I, Takahashi H, Watanabe M, Nomura T, Kano T, et al. Sez6l2 regulates phosphorylation of ADD and neuritogenesis. Biochem Biophys Res Commun. (2017) 494:234–41. doi: 10.1016/j.bbrc.2017.10.047
Keywords: autoimmune cerebellar ataxia, paraneoplastic cerebellar ataxia, AP3B2 antibody, ITPR1 antibody, TRIM9 antibody, TRIM67 antibody, TRIM46 antibody, mGluR2 antibody
Citation: Garza M and Piquet AL (2021) Update in Autoimmune Movement Disorders: Newly Described Antigen Targets in Autoimmune and Paraneoplastic Cerebellar Ataxia. Front. Neurol. 12:683048. doi: 10.3389/fneur.2021.683048
Received: 19 March 2021; Accepted: 28 July 2021;
Published: 18 August 2021.
Edited by:
Christian Vedeler, University of Bergen, NorwayReviewed by:
Bruno Giometto, Azienda Provinciale per i Servizi Sanitari (APSS), ItalyHiroshi Mitoma, Tokyo Medical University, Japan
Takayoshi Shimohata, Gifu University, Japan
Mario U. Manto, University of Mons, Belgium
Lidia Sabater, Institut de Recerca Biomèdica August Pi i Sunyer (IDIBAPS), Spain
Copyright © 2021 Garza and Piquet. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Amanda L. Piquet, YW1hbmRhLnBpcXVldEBjdWFuc2NodXR6LmVkdQ==
 Madeline Garza
Madeline Garza Amanda L. Piquet
Amanda L. Piquet