- 1Department of Neuroscience, Imaging and Clinical Sciences, University G. d'Annunzio of Chieti-Pescara, Chieti, Italy
- 2Behavioral Neurology and Molecular Neurology Units, Center for Advanced Studies and Technology–CAST, University G. d'Annunzio of Chieti-Pescara, Chieti, Italy
- 3Institute for Mind Impairments and Neurological Disorders–iMIND, University of California, Irvine, Irvine, CA, United States
- 4Institut du Cerveau et de la Moelle épinière, ICM, INSERM UMRS 1127, CNRS UMR 7225, Pitié Salpêtrière Hospital, Paris, France
- 5AP-HP, GH Pitie-Salpêtrière-Charles Foix, Epilepsy Unit and Neurophysiology Department, Paris, France
- 6Neurology Unit, Department of Clinical and Experimental Sciences, University of Brescia, Brescia, Italy
- 7Parkinson's Disease Rehabilitation Centre, FERB ONLUS–S. Isidoro Hospital, Trescore Balneario, Italy
Agitation is a behavioral syndrome characterized by increased, often undirected, motor activity, restlessness, aggressiveness, and emotional distress. According to several observations, agitation prevalence ranges from 30 to 50% in Alzheimer's disease, 30% in dementia with Lewy bodies, 40% in frontotemporal dementia, and 40% in vascular dementia (VaD). With an overall prevalence of about 30%, agitation is the third most common neuropsychiatric symptoms (NPS) in dementia, after apathy and depression, and it is even more frequent (80%) in residents of nursing homes. The pathophysiological mechanism underlying agitation is represented by a frontal lobe dysfunction, mostly involving the anterior cingulate cortex (ACC) and the orbitofrontal cortex (OFC), respectively, meaningful in selecting the salient stimuli and subsequent decision-making and behavioral reactions. Furthermore, increased sensitivity to noradrenergic signaling has been observed, possibly due to a frontal lobe up-regulation of adrenergic receptors, as a reaction to the depletion of noradrenergic neurons within the locus coeruleus (LC). Indeed, LC neurons mainly project toward the OFC and ACC. These observations may explain the abnormal reactivity to weak stimuli and the global arousal found in many patients who have dementia. Furthermore, agitation can be precipitated by several factors, e.g., the sunset or low lighted environments as in the sundown syndrome, hospitalization, the admission to nursing residencies, or changes in pharmacological regimens. In recent days, the global pandemic has increased agitation incidence among dementia patients and generated higher distress levels in patients and caregivers. Hence, given the increasing presence of this condition and its related burden on society and the health system, the present point of view aims at providing an extensive guide to facilitate the identification, prevention, and management of acute and chronic agitation in dementia patients.
Introduction
Agitation is a common behavioral disturbance featuring exaggerated motor activity and verbal and/or physical aggressiveness, severe enough to impair social relations and daily living activities (1).
Agitation is observed in up to 70% of patients with cognitive decline, and its incidence is higher in moderate to severe stages of the disease (2). The prevalence ranges from 30 to 50% in Alzheimer's disease (AD), 30% in dementia with Lewy bodies (DLB), 40% in frontotemporal dementia (FTD), and 40% in vascular dementia (VaD) (3, 4).
Agitation adversely impacts cognitive performance, functional status, and patients' quality of life and enhances caregiver's distress. Moreover, agitation is associated with a higher admission rate to assisted living facilities, higher use of medications, long-term hospitalization, and higher mortality (5).
Several studies have considered agitation as indicative of the external expression of anxiety (6–8). Thus, some authors have suggested that the presence of anxiety in AD, a typical sign of the disease's preclinical stage, could also be seen as a risk factor for the future ensuing of agitation (9).
The occurrence of agitation is related to frontal lobe dysfunctions (10, 11) and, in particular, to an abnormal activation of the orbitofrontal cortex (OFC) and anterior cingulate cortex (ACC) (12). This notion has been supported by postmortem studies (12, 13) showing the large presence of neurofibrillary tangles in the OFC and ACC of patients with the frontal variant of AD. Indeed, a strict correlation among neuropsychiatric symptoms (NPS), especially aggressiveness, and tau pathology, rather than amyloid, is commonly reported in AD patients (12, 14, 15). A significant tau pathology in the frontal lobes is also a major postmortem finding in FTD, which is notably characterized by disabling behavioral changes (16, 17). As regards DLB and VaD, both characterized by NPS including agitation (18, 19), a clear correlation with frontal lobe dysfunction is lacking.
The involvement of frontal lobe dysfunction in the occurrence of agitation has been also confirmed by functional imaging studies in both cognitively unimpaired subjects (20, 21) and AD patients (22). Furthermore, blood hypoperfusion in the anterior temporal lobe (ATL), dorsolateral pre-frontal cortex (dlPFC), and superior parietal cortex (SPC) is thought to contribute to the production of abnormal emotional responses to external stimuli, thereby causing aggressive or agitated states. The dlPFC is a critical area for thinking and planning, whereas the SPC is involved in sensorimotor integration from external stimuli. A dysfunctional interplay among these cerebral regions may explain abnormal behavioral responses driven by misinterpretation of environmental and social stimuli (23–26). Several studies have also supported the notion of imbalanced neurotransmitter systems in the onset of behavioral impairments (27). Besides the role of cholinergic depletion in AD and DLB, which may also be a mechanism underlying dysregulation of emotions and attention, as suggested by the efficacy of acetylcholinesterase inhibitors (AChEIs) in the control of psychiatric symptoms (28), the noradrenergic system also seems to be directly involved. A compensatory dysfunctional overactivity of noradrenaline, driven by the progressive loss of noradrenergic neurons in the locus coeruleus (LC), may affect the frontal cortex activity, thereby causing a regional up-regulation of adrenergic receptors (29, 30) (Figure 1). In FTD, even though the data are still controversial, agitation appears to be related to increased dopaminergic transmission (31).
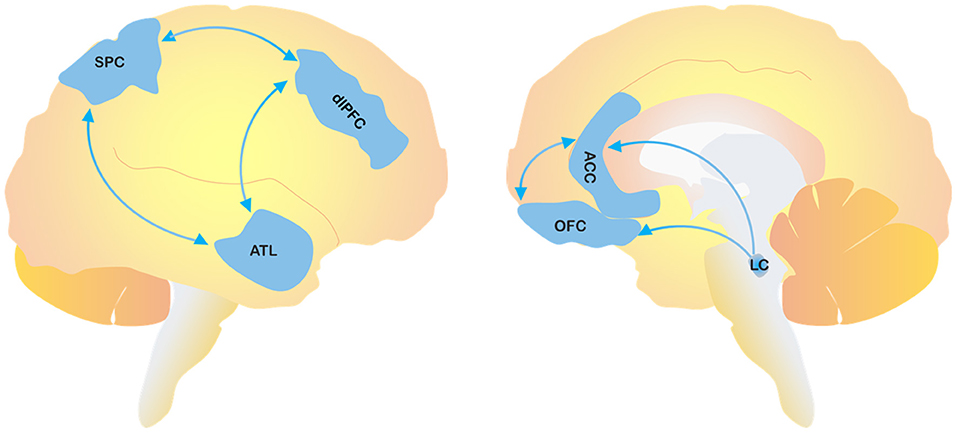
Figure 1. The figure depicts the cerebral areas involved in the pathological process of agitation. SPC, superior parietal cortex; dlPFC, dorsolateral pre-frontal cortex; ATL, anterior temporal lobe; OFC, orbitofrontal cortex; ACC, anterior cingulate cortex; LC, locus coeruleus.
Current managing guidelines for dementia-related agitation recommend implementing non-pharmacological approaches as the first line of intervention (32), whereas considerable research now focuses on prevention strategies. Pharmacological treatments should be initiated whenever behavioral changes may compromise patient safety and produce severe distress to caregivers (33).
The present review provides a toolbox for the detection, prevention, and therapeutical management of acute and chronic agitation in patients suffering from neurodegenerative conditions.
Mild Behavioral Impairment
NPS are common in the prodromal stage of dementia and can precede the onset of cognitive impairment. The presence of NPS in cognitively normal patients or in patients with mild cognitive impairment (MCI) is associated with an increased risk of progression to overt dementia. The need to identify, in the early stages of the disease, the population at risk of cognitive decline has led to the formulation of the concept of mild behavioral impairment (MBI) (34). Building on the prior definitions of a pre-dementia risk state (35, 36) and frontotemporal-MCI (37), the ISTAART NPS-PIA formally described MBI as the emergence of sustained and impactful NPS occurring after the age of 50, which are not encompassed in the psychiatric nosology, persist for at least 6 months, and manifest before or at the onset of MCI (34). Among the NPS associated with MBI, agitation is as frequent as 30%. It is important to understand the prevalence of agitation and impulsivity in pre-dementia syndromes as there is a potential opportunity for early intervention and higher impact in this early stage of disease, even though clinical trials need to be conducted to test and prove that behavioral and pharmacologic treatments in the pre-dementia stage can effectively improve agitation.
Pre-disposing Factors and Conditions to Agitation
Agitation occurs as a result of the interplay between neurobiological and environmental determinants. In addition to neurodegenerative processes, several factors, such as chronic or acute pain (e.g., arthritis), sleep disturbances, sensory impairment, acute medical illness (e.g., infections, respiratory diseases, urinary retention, renal failure), or metabolic changes (38), generate agitated behavior. Psychological factors, depression, or psychological distress may also be involved (39). Sundowning, the ensuing agitation upon the dimming of natural light as happens in the evening, is another predisposing condition for agitation (40). Moreover, acute onset of agitation can be triggered by changes in medication regimens or drug side effects (e.g., with antipsychotics or anticholinergic drugs) (40).
Hospitalization is another well-known trigger of agitation either alone or as a manifestation of hyperkinetic delirium in people with dementia (41).
Finally, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) can be a precipitating factor for agitation because of the related long-term hospitalization and quarantine (42–44) or the ensuing viral encephalitis, critical illness encephalopathy, and systemic inflammation (45).
Agitation in Acute Medical Conditions: Differential Diagnosis With Dementia-Related Agitation
In elderly patients, acute agitation can be the expression of a wide range of conditions, including medical, psychiatric, and substance-induced alterations (46) (Table 1).
In addition to hyperkinetic delirium, which can also arise during the hospitalization of cognitively unimpaired older people (42), other acute neurological conditions, such as stroke (especially in the presence of aphasia), intracranial neoplastic masses or hemorrhages, meningitides, encephalitides, and head injuries, may generate agitation that can be the first or unique symptom of the underlying condition.
The ascertainment of a history of psychiatric illnesses (e.g., schizophrenia or other psychotic disorders, depression, bipolar disorders) is critical, as these conditions may often determine the onset of acute mental alterations, including agitated behavior (47).
Moreover, the presence of substance-induced agitation should always be considered. Older people are particularly susceptible to drug side effects, as co-existing metabolic disturbances can affect drug pharmacokinetics (40). Age-related metabolic alterations include hypoalbuminemia, hepatic or renal failure, and dehydration, conditions that should not be overlooked. Neurotransmitter alterations (e.g., a reduction in dopamine and acetylcholine transmission) can also promote acute agitation in elderly patients (48). Furthermore, the use of benzodiazepines (BZDs) may generate paradoxical response and behavioral disinhibition, as well as worsening of cognitive performances, excessive sedation, and increased risk of falls (49). Therefore, in older people, BZDs should be avoided, and their use or misuse should be taken into account in the event of an acute onset of agitated behavior. Anticholinergic and serotoninergic agonists, steroids, and neuroleptics (46, 50) can also trigger agitation in the elderly. Non-pharmacological toxic agents also need to be considered, especially in suspected carbon monoxide or alcohol intoxications. Substance withdrawal should also be considered as a possible trigger for agitation (48).
Diagnostic Investigations
When dealing with a patient showing agitation signs, the primary goal is to identify cause and severity (51). A diagnostic workup is mandatory to achieve this goal.
The first step is to collect the patient medical history, ideally with caregivers' and relatives' critical help. To that aim, it is critical to identify the time of onset of agitation and the patient's pre-existing mental status. Recent hospitalizations, infections, traumas, or other predisposing factors, including medication history or substance abuse, should also be investigated (48, 51).
Accurate physical examination is key. Vital signs monitoring is mandatory to manage life-threatening conditions, such as cardiocirculatory or respiratory failure. The current mental status and level of consciousness of the patients should be assessed. Neck stiffness, fever, pain, or focal deficits should be evaluated to exclude infections (e.g., meningoencephalitis), stroke, hemorrhages, or central nervous system (CNS) tumors. Moreover, other abnormal findings, such as dehydration, asterixis, or vomiting, should prompt the investigation of underlying metabolic disorders or impairment (e.g., diabetes, renal, or liver failure) (46, 48).
All the agitated patients admitted to the hospital should be assessed with the help of laboratory testing (including the assessment of blood cell counts, glucose levels, electrolytes, creatinine, blood urea nitrogen, transaminases, C-reactive protein, procalcitonin, and urine tests) to rule out certain conditions, such as anemia, intercurrent infections, and liver or kidney failure. Toxicologic examinations should be considered to exclude alcohol or substance abuse. In acute agitation, a non-contrast head computed tomography (CT) should be obtained when an intracranial origin is suspected (48), especially in the presence of focal neurological signs. Lumbar puncture, as well as brain magnetic resonance imaging (MRI) and electroencephalogram (EEG), should be reserved for selected cases (e.g., when encephalitis, or a non-convulsive status epilepticus, is suspected) (48, 52).
Assessment Scales for Agitation
Several assessment scales are currently available to investigate the presence and severity of agitation (53). The Agitated Behavior Scale (ABS) assesses an agitated state's occurrence and duration after brain injury. The scale's primary purpose is to monitor behavioral changes after admission to a hospital ward (54). The Behavioral Activity Rating Scale (BARS) is often used in clinical trials. According to this scale, patients are classified into seven different levels of agitation (55). The Overt Agitation Severity Scale (OASS) offers a helpful approach to detect and rate agitation when spanning from anxiety to aggression (56). The Modified Overt Aggression Scale (MOAS), a scale divided into four sections, examines the frequency and severity of aggressive episodes (57). Another easy-to-use tool is the Pittsburgh Agitation Scale (PAS), which measures the dementia-related agitation severity (58). The Agitated Behavior Mapping Instrument (ABMI) (59) is a diagnostic scale that evaluates 14 different physical and verbal agitated behavior features. Finally, the Cohen-Mansfield Agitation Inventory (CMAI) is a frequency rating scale completed by caregivers (60).
Some assessment scales are primarily administered in high-intensity care wards. The Riker Sedation–Agitation Scale (SAS) (61), the first specific agitation scale developed for the intensive care unit (ICU), identifies seven levels of consciousness ranging from severe and life-threatening agitation to deep sedation. A similar tool is the Motor Activity Assessment Scale (MAAS) (62), which describes, according to the patient's motor behavior, seven different levels of agitation (from unresponsive to dangerously agitated). The Richmond Agitation Sedation Scale (RASS) assesses the level of alertness and agitated behavior in critically ill patients (63).
A screening tool for delirium is the 4AT, in which a score higher than four indicates a high risk for delirium (64). However, the Confusion Assessment Method for the ICU (CAM-ICU) is a more sensitive and specific assessment to diagnose a delirium state (65).
Managing Agitation With Non-pharmacological Strategies
Over the past years, several studies have investigated a wide range of non-pharmacological approaches to treat and prevent dementia-related agitation. Hereby, we discuss the most validated first-line non-pharmacological approaches to chronic and acute agitation in cognitively impaired subjects (32, 66).
Prevention Strategies for Home-Living and Care Home Residents
Person-centered care (PCC) is an operating system in healthcare, which considers the health practitioner and the patients as partners in achieving tailored care that meets patients' needs in a unique way (67, 68). In the PCC framework, the social and historical background, the personality, and the lifestyle of the patients are considered to promote a positive social environment, good compliance, and best outcomes for patients with dementia (67, 68). The PCC approach is considered a successful option to prevent agitation in home-living and care home patients with dementia (69, 70) and reduce antipsychotic use (71). Nonetheless, not every PCC-based strategy is effective. For instance, the Dementia Care Mapping (DCM) (69, 72, 73), which is based on patients' systematic observations, has generated conflicting results. In contrast, the Managing Agitation and Raising Quality of Life (MARQUE) intervention (74) has failed to improve agitation prevention in care home settings.
As indicated by a recent meta-analysis (75), music intervention, especially when employed in groups, can significantly reduce agitation in cognitively impaired patients. Furthermore, passive listening to music has been associated with behavioral improvement and reduction of aggressiveness and agitation (76). This effect is considered to be driven by a positive exploration of repressed feelings (76). Regarding music therapy trials, some studies have explored the effectiveness of white noise to prevent the sundowning syndrome, or the use of personalized music, via headphones, upon daily hygiene care or walking, reporting that these approaches can improve agitated behavior (77). Doll therapy has been shown to improve happiness and engagement but did not significantly reduce agitation (78, 79). Some concerns have been raised on this approach's feasibility in patients who do not have offspring (79). Thus, further investigation and research are required.
Previous studies have indicated a role for boredom in the onset of physical agitation (80). Thus, activity-based strategies have been developed to prevent this condition. The application of combined stimuli (tasks, reading, work-related, manipulation) has shown to be more effective than a single activity (81). However, the most effective activity was live socialization (82).
New technologies, which still need full validation, are being developed to monitor and prevent agitation. Behavioral & Environmental Sensing and Intervention (BESI) collects environmental data, caregivers' information, and patients' behavioral and motion states through wearable tools and then detects early signals of incoming agitation delivering an alert to the caregiver, who can timely prevent the complete escalation of the symptom (83–85). A teleconsultation system based on expert's recommendations, namely, ECHO-AGE, has also provided clinical improvement in nursing home settings when dealing with agitated patients (86).
Several other non-pharmacological interventions are being evaluated for agitation prevention, as virtual and sensory therapy (e.g., aromatherapy, bright light therapy, taking a breath of fresh air, the use of rocking chairs) (77, 79). Lastly, ongoing studies are proposing virtual reality-related approaches that include music, sensory, and mental stimulations to improve cognitive functions (attention, executive function, visual and verbal memory) and psychological domains (agitation, depression, anxiety, and apathy) (87–89).
Data that emerged from other non-pharmacological approaches, such as acupuncture and electroconvulsive therapy, are mostly conflicting. Some ongoing trials could possibly clarify a potential role for these therapies (90, 91).
Therapeutic Strategies for Hospitalized Patients
Unfortunately, the PCC approach is challenging in hospital settings, given the short length of the patient stay and the necessity of rapid interventions (92). However, the presence of pain (93) and intercurrent pathologic conditions (94) (e.g., infections, dehydration, metabolic imbalance) has been associated with the onset of aggressiveness, anxiety, and agitation and should be addressed. Furthermore, the direct assessment of unmet needs in hospitalized patients, which is often less recognized and identified than expected, can provide a more time-effective approach in a clinical environment, according to the standards of “Needs-driven care” (68) based on the Unmet Needs Model (80). Nonetheless, cognitively impaired patients may experience difficulties communicating feelings of discomfort, pain, and loneliness, thereby making the assessment difficult (80). In nursing homes or at the patient home, the Needs-driven care is ancillary to the PCC and the most successful given the presence of familiarity and closed relationships with the patients.
Moreover, as previously reported, patients with dementia, when hospitalized, often present with a more complex clinical picture of hyperkinetic delirium (42). Combined strategies are far more adequate to prevent delirium onset, yet not always effective (95). Careful management of pharmacological therapy, space–time reorientation, early mobilization, minimization of restraint use, and adequate sleep hygiene is the most recommended option for preventing delirium in hospitalized patients (42, 96). Other options include access to a living room with other patients and the caregiver's presence (97). Multiple changes of rooms should be avoided (98).
All the strategies developed in the last 20 years have led to a sophisticated solution, the “Delirium Room” (DR) (99, 100). Elderly patients with cognitive decline and the need for intensive/continuous observation can be monitored, in this dedicated four-bed environment, by a trained nurse. Restraint use is strongly discouraged, whereas non-pharmacological approaches to prevent agitation are indicated (99). A valid option is offered by the Geriatric Monitoring Unit (GMU) (101, 102). The GMU combines, in a five-bed room, typical DR management with a PCC approach and bright therapy and has been shown to achieve significantly shorter lengths of delirium symptoms (102).
Pharmacological Treatments in Acute and Chronic Conditions
The Food and Drug Administration (FDA) warning on the use of antipsychotic drugs (both typical and atypical) has led to better awareness about the risk associated with the use of these classes of drugs in cognitively impaired and older people, including increased mortality (103–105).
The recently published European Academy of Neurology (EAN) recommendation on the management of issues in dementia, including agitation, reports that individuals with dementia and agitation and/or aggression should be treated with atypical antipsychotics only after all non-pharmacological measures have been proven to be without benefit or in the case of severe self-harm or harm to others (weak recommendation). Antipsychotics should be discontinued after cessation of behavioral disturbances and in patients in whom there are side effects (Good Practice statement) (66).
Further suggestions for pharmacological management of acute agitation, such as agitation in delirium and chronic agitation in dementia, are depicted in the following sections. Randomized controlled trials (RCTs) are summarized in Table 2.
In Figure 2, we propose an approach to treat patients with agitation properly.
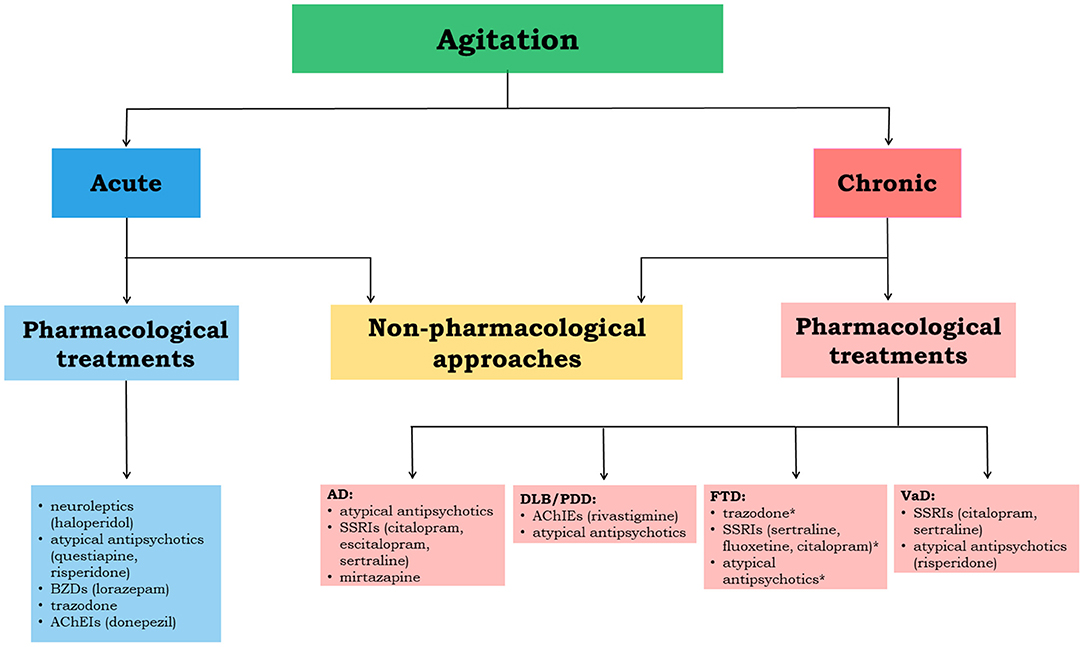
Figure 2. The flowchart depicts the therapeutical management flow to be implemented in acute and chronic agitation. The most relevant evidence-based drug options are reported for each neurodegenerative disease. *indicates not sufficiently supported by clinical evidence; AD, Alzheimer's disease; DLB, dementia with Lewy bodies; PDD, Parkinson's disease with dementia; VaD, vascular dementia; FTD, frontotemporal dementia; AChEIs, acetylcholinesterase inhibitors; BZDs, benzodiazepines; SSRIs, selective serotonin reuptake inhibitors.
Agitation in Delirium
Any pharmacological treatment for agitation in hyperkinetic delirium should be started at the lowest dose and continued for the minimum amount of time (134).
The first step is to consider the patient medical history, including concomitant therapies and comorbidity (135), in order to reduce/minimize the underlying causative factors. In case of an agitated state, neuroleptics represent the first choice, especially haloperidol (starting dose ranging from 0.5 to 1 mg, not to exceed from 3 to 5 mg in 24 h), which also presents the advantage of parenteral formulation (136, 137). Given the drug's well-known parkinsonian side effects, it should be avoided in patients with dementia associated with parkinsonism (138). Indeed, in patients with extrapyramidal signs, pharmacological recommendations suggest using atypical antipsychotics, such as quetiapine (25–50 mg/day) (135). Three previous RCTs showed comparable clinical efficacy of atypical antipsychotics and haloperidol (137, 139, 140). Moreover, except for chlorpromazine, first- and second-generation antipsychotics can be safely administered in case of liver or kidney dysfunction (141).
In general, both typical and atypical antipsychotics should be avoided in patients with known cardiac disease due to pro-arrhythmogenic effects (e.g., QT interval prolongation) (142). In this case, BDZs, per os or via intramuscular administration according to delirium severity, should be preferred in patients without respiratory deficits or in the delirium/agitation associated with alcohol withdrawal (143, 144). Among BDZs, lorazepam (0.5–1 mg per os) seems to be the most suitable drug due to its pharmacokinetic properties (it is directly metabolized through glucuronidation, which potentially reduces drug interactions) (145). When treating patients with dementia and hyperkinetic delirium, it is important to consider the known possible paradoxical effects of BDZs with increased agitation in these patients (49).
Cognitively impaired patients with history of seizures or chronic pain can also be treated with antiepileptic drugs in case of mild delirium (135). Among anticonvulsant drugs, gabapentin and pregabalin present a good safety profile, even if they should be avoided in the presence of severe renal failure (146).
Some findings also supported the use of low doses of trazodone (50–300 mg/day) in treating hyperkinetic delirium (147–149). Indeed, this drug mainly acts through direct serotonin receptor blockade and may produce sedation due to its strong histaminergic effects (149).
Finally, some case reports and one open-label study (150–153) suggested a promising use of AChEIs, such as donepezil (5 mg daily), in delirium treatment, but further evidence and RCTs are needed.
Alzheimer's Disease
Among typical antipsychotics, haloperidol in a dose of 1.2–3.5 mg/day suppresses aggressiveness effectively but shows lower efficacy on agitation. Because of its remarkable side effects (e.g., extrapyramidal signs, prolongation of the QTc interval, arrhythmias, and increased mortality), haloperidol is not recommended (142). Atypical antipsychotics show comparable effectiveness and higher patient tolerance. For instance, in a multicenter, double-blind, placebo-controlled trial, 421 outpatients suffering from AD with psychosis, aggressiveness, or agitation were randomly assigned to receive olanzapine (mean dose 5.5 mg/day), quetiapine (mean dose 56.5 mg/day), risperidone (mean dose 1.0 mg/day), or placebo. Clinical benefits were observed in 32% of patients assigned to olanzapine, 26% of patients assigned to quetiapine, 29% of patients assigned to risperidone, and 21% of patients assigned to placebo, without significant differences (154). The CATIE-AD study evaluated the effects on NPS of olanzapine, risperidone, and quetiapine, compared with placebo. Among NPS, antipsychotic resulted to be more effective for specific behavioral symptoms, such as agitation (155). Regarding second-generation antipsychotics, high doses are not recommended as the risk of mortality is dose-dependent. The most common causes of death are cardiovascular, cerebrovascular, respiratory, and infectious (especially respiratory) complications (142).
Moreover, selective serotonin reuptake inhibitors (SSRIs), such as citalopram, escitalopram, and sertraline, have demonstrated efficacy in AD patients (111, 156). The use of citalopram is supported by the most compelling evidence for treating AD dementia-related agitation (156). Despite earlier studies not focusing on agitation effects, in the first RCT, citalopram and perphenazine were more effective than placebo in short-term hospital treatments of psychotic disturbances, including agitation (106). Another RCT also demonstrated that citalopram decreases agitation scores compared with risperidone, without relevant side effects (107). Additionally, the Citalopram for Agitation in Alzheimer Disease Study (CitAD), a placebo-controlled double-blind RCT, showed a high acceptability rate and efficacy for citalopram over placebo in the treatment of agitation (108). In a nursing home setting, a longitudinal 6-month study showed similar efficacy against agitation of citalopram, olanzapine, or quetiapine, even though citalopram was associated with a lower occurrence of falls, orthostatic hypotension, and fewer hospitalizations (109). Citalopram should be considered in AD patients below 85 and used at low doses as cardiac conduction disturbances are dose-dependent and more frequent in elderly patients. The FDA recommends a maximum daily dose of citalopram of 20 mg/day in patients above the age of 60 (157). Since the effects of citalopram take 2 weeks to ensue, citalopram should not be considered for the acute treatment of agitation (156).
Escitalopram also showed clinical benefits in agitation treatment (158). For instance, a 6-week RCT compared escitalopram to risperidone and showed that both drugs reduced agitation. Although risperidone revealed efficacy earlier, the drug produced a higher burden of side effects (110). Since escitalopram may also prolong the QT interval in a dose-dependent manner, a low dose should be considered in the elderly (156). In that respect, the maximum daily dose recommended is 10 mg for patients over 60 (159).
Furthermore, sertraline was demonstrated to decrease agitation or aggressiveness and other psychiatric symptoms, in a double-blind, randomized, placebo-controlled study (111). In particular, AD patients with moderate to severe behavioral symptoms seem to respond to sertraline (156).
Mirtazapine, an antidepressant with alpha-2 adrenergic, 5-HT2, and 3-HT2 antagonist properties, showed promising results in open-label studies. For instance, in a 12-week prospective study, AD patients with agitated behavior receiving 15–30 mg/day of mirtazapine showed a significant reduction in CMAI-short form (CMAI-SF) after treatment (112).
Even if AChEIs seem to have positive effects on other behavioral symptoms (such as depression and anxiety) (142), they did not show good efficacy in the treatment of agitation in AD (160–162). Despite the initial positive results of memantine for the treatment of agitation and psychosis in AD (163), two subsequent RCTs failed to achieve statistically significant results in agitation (113, 114).
Small, placebo-controlled studies on carbamazepine found evidence of modest benefits for agitation and aggression in AD patients (115). Moreover, in 103 institutionalized cognitively impaired patients with agitation and aggressiveness, oxcarbazepine did not improve NPS vs. placebo, and side effects occurred more frequently in the treatment group (116). Regarding valproate (daily dose ranging from 480 to 1,500 mg/day), RCTs and subsequent meta-analysis failed to provide any support for its use in agitation (115). Few data, including mainly case reports and small case series, are available for other anticonvulsants (gabapentin, levetiracetam, topiramate, and lamotrigine) (115).
Prazosin, an alpha-adrenoreceptor blocking drug, demonstrated a significant impact on agitation and aggressiveness in a daily dose ranging from 1 to 6 mg (164), but it is not recommended in patients who have experienced orthostatic hypotension (165).
Vascular Dementia
VaD is a neurologic condition due to the occurrence of single or multiple ischemic lesions of the brain (166). However, risk factors for cerebrovascular injuries also contribute to neurodegenerative disease onset. Rather than a “stand-alone” entity, VaD should be regarded as part of a “Mixed dementia” phenotype, particularly in elderly subjects (167). In this perspective, we can easily understand why RCTs have a hard time in disentangling VaD from AD (the most common cause of neurodegenerative dementia). Considering this limitation, as well as the lack of recent studies addressing the issue of agitation in VaD, we report the most relevant approaches identified.
A sequential drug treatment algorithm for agitation and aggressiveness associated with AD and mixed dementia, such as AD/VaD, has been lately proposed (168). The authors indicated that antipsychotic drugs have the highest efficacy for treating behavioral and psychotic symptoms. However, the risk of well-known side effects limits their use to only severe conditions (33).
Among antipsychotic drugs, risperidone seems to present the most substantial benefit for agitation, as well as other behavioral changes (169–171). In a clinical trial comparing dosage and time needed to reach a therapeutic response of three antipsychotics (risperidone, sulpiride, and quetiapine), the risperidone-treated group showed the shortest time to obtain a clinical response (172). The authors recommended a target dose ranging from 0.5 to 1.5 mg/day (173). Despite limited findings, also aripiprazole and quetiapine showed statistically significant effects on agitation (170, 174).
Some meta-analyses also revealed clinical benefits on agitation using SSRIs, especially citalopram and sertraline (175). AChEIs failed to improve the cognitive functions and behavioral symptoms of VaD patients (176–178). In a double-blind study, only rivastigmine showed its superior efficacy over placebo on cognition, but effects were missing when dealing with daily living activities or NPS (124).
Anticonvulsants, such as carbamazepine, gabapentin, and pregabalin, may be useful in patients who did not respond to antipsychotics, although clear evidence is lacking (118–120, 179).
Dementia With Lewy Bodies and Parkinson's Disease With Dementia
The first step in treating agitation of parkinsonian patients should be an accurate revision of current treatment. Dopamine agonists (DAs), monoamine oxidase inhibitors (MAOIs), and catechol-O-methyltransferase (COMT) inhibitors, especially when associated with SSRIs or serotonin and norepinephrine reuptake inhibitors (SNRIs), might increase agitation (180). Thus, careful reduction or redistribution of dopaminergic drugs should represent the first option for these patients.
The management of agitation in DLB and Parkinson's disease with dementia (PDD) is particularly challenging in light of the well-documented side effects of typical and, to a lesser extent, atypical neuroleptics, compounds that worsen parkinsonism and even precipitate the ensuing of the neuroleptic malignant syndrome (181–184). Hence, the treatment with neuroleptics should be avoided in agitated patients with DLB/PDD. Nevertheless, some anecdotal evidence has indicated the efficacy and safety of atypical neuroleptics. In particular, clozapine (average dose 44.6 mg/day) (185) and quetiapine (50–75 mg/day) (117), atypical neuroleptics with weak affinity for dopamine D2 receptors, have achieved benefits in the management of agitation in patients with dementia and parkinsonism (117, 185). However, in line with the recent concerns about an increased risk of death in elderly patients treated with antipsychotic drugs (both atypical and conventional) (186), clozapine and quetiapine should be cautiously employed as a first-line treatment for agitation. AChEIs (118, 179) show modest efficacy in improving the cognitive and behavioral features of DLB/PDD patients and lack the risk of triggering extrapyramidal effects, although increased tremor is sometimes limiting. A sizeable placebo-controlled study on rivastigmine found that the use of rivastigmine in PDD patients significantly decreased the NPS, although an improvement of agitation was not clearly reported (119). Three placebo-controlled studies on donepezil in PDD patients (120–122) produced no effects vs. placebo for psychiatric disturbances.
Frontotemporal Dementia
No FDA-approved therapies are available for FTD (174). Nevertheless, clinicians often liberally implement drugs used in other forms of dementia, even in the absence of evidence-based support. For instance, AChEIs and memantine failed to provide benefits in FTD patients and actually exacerbated behavioral symptoms (187–189).
Alterations of serotoninergic and dopaminergic rather than cholinergic neurotransmission are present in FTD (190), and therefore, SSRIs and antipsychotic drugs have been tested with different results. The clinical effects of sertraline, fluoxetine, and citalopram are controversial. These drugs reduced agitation, anxiety, and impulsiveness and worsened apathy and cognitive functioning (191–193). Some evidence suggested that SSRIs may help treat some behavioral features without affecting cognition, but further studies are needed (187). Trazodone has also been tested in clinical trials, and a study has supported its effectiveness against agitation and other NPS (188). In a randomized, placebo-controlled trial, trazodone (with daily doses ranging from 50 to 300 mg) dramatically reduced behavioral symptoms over 12 weeks of treatment. Common mild side effects included fatigue, dizziness, hypotension, and somnolence (123). Nevertheless, there is insufficient evidence to support the employment of trazodone for agitation in patients with FTD (148).
Among antipsychotic agents, risperidone, quetiapine, olanzapine, aripiprazole, and clozapine showed efficacy on the agitation of FTD patients, but, because of their low safety profile, the drugs are recommended only as a second-line treatment (194–196).
Recently, a low oral dose of lithium was proposed as an add-on therapy to antipsychotic treatment for aggressiveness and agitation in FTD (197). However, only three patients with FTD and three patients with AD were included in the study, and therefore, further RCTs are needed.
Regarding the use of antiepileptic drugs, two studies tested gabapentin for acute onset of agitation, but not a large and robust body of evidence is available to suggest its use in FTD patients (198).
COVID-19 and Agitation in Neurodegenerative Diseases
After the first cases of the novel coronavirus disease 2019 (COVID-19) were reported in Wuhan, China, in December 2019, the spread of SARS-CoV-2 rapidly became a pandemic, involving millions of patients worldwide. Although SARS-CoV-2 poses a risk at all ages, adults aged 65 years and older are at greatest risk of severe disease, hospitalization, intensive care use, and death, accounting for more than 80% of deaths in Western countries. With the increasing number of confirmed cases and the accrued clinical data, it is now well-established that a significant proportion of these patients experience neurological symptoms and syndromes (in addition to the pre-dominant respiratory symptoms) (199, 200). Early COVID-19 studies have reported estimated rates of delirium at 20–50% and agitation at almost 10% at in hospitalized patients with dementia, even in the absence of typical signs and symptoms of SARS-CoV-2 infection, such as fever or cough (201). Different studies have clearly reported that the presence of delirium and/or agitation was associated with increased mortality, even taking into account age and multimorbidity (200, 202, 203).
Acute agitation in patients with delirium evoked by hypoxia and/or fever is the most commonly observed clinical feature, especially in elderly individuals with pre-existing dementia or psychiatric conditions (204). Given the elevated rates of clinical and adverse events associated with dementia and the various NPS potentially associated with COVID-19, physicians need to be mindful of these potential complications when evaluating these patients. A thorough examination of mental and neurological status helps the clinicians evaluate any acute behavioral disorder, which may indicate underlying encephalopathies or encephalitis triggered by the COVID-19 infection (199, 205).
Hospitalized patients with COVID-19, who are acutely agitated, deserve special attention given the risk of poor compliance to mechanical ventilation. Furthermore, the implementation of non-pharmacological and reorientation strategies to treat agitation is compromised by social distancing and isolation measures implemented to minimize the pandemic (206). The physical isolation of COVID-19 patients, either hospitalized or not, could trigger agitation and is still a critical issue in acute and chronic care. Patients with persistent and severe agitation despite attempts to treat the underlying causes require sedative medications. Low doses of first-generation antipsychotics, such as haloperidol, or second-generation antipsychotics, such as olanzapine and risperidone, are equally effective and are considered first-line management strategies in COVID-19 units (103, 207). However, no RCTs have addressed the treatment of COVID-19-related agitation (207). When there are no absolute contraindications to any particular medication class, possible management includes administering alpha-2 agonists and low-potency antipsychotics, whereas BDZs should be avoided given their depressive effects on ventilation.
Finally, the occurrence of hyperkinetic delirium and agitation has been reported also during the recovery phase from COVID 19, in the absence of direct brain infection (208).
Clinical Pharmacological Trials and Future Prospects
Nowadays, dementia-related agitation treatments pose a significant challenge to clinicians as specific pharmacological therapies are lacking.
Several experts have agreed that, to optimize the clinical response, patients with dementia should be treated with specific and novel medications interacting with pharmacologically relevant targets (209–211). Therefore, preclinical and clinical studies focused on novel biological targets are needed.
Considering that the serotonin 5-HT2AR polymorphisms and altered functioning of these receptors are associated with psychosis and aggressiveness onset in AD patients, recent studies have been focused on the effects of pimavanserin, a selective inverse agonist of the 5-HT2AR (212) or lumateperone and brexpiprazole, two novel atypical antipsychotics acting as 5-HT2A antagonists. After showing promising efficacy in phase II RCTs, these drugs now face validation through phase III RCTs (213–215).
Regarding pimavanserin, recent phase II RCTs suggested efficacy in AD patients, although the safety profile needs further investigations. For instance, the drug may induce QT prolongation (216). A phase III, double-blind, placebo-controlled, relapse prevention study (NCT03325556) is evaluating the efficacy and safety profile of variable dosages of pimavanserin (125, 216).
Brexpiprazole is a second-generation antipsychotic, occasionally referred to as a third-generation antipsychotic with partial agonist properties at the D2 receptor. The compound is FDA-approved for treating schizophrenia and major depressive disorder as add-on therapy (217). Compared with aripiprazole and brexpiprazole, the molecule shows less activity at the D2 receptor, thereby generating a more tolerable side effects profile. Three phase III clinical trials are ongoing to evaluate the compound's long-term safety and clinical efficacy in AD patients with agitation (NCT03724942, NCT03620981, and NCT03548584) (127–129).
A recent phase III, randomized, double-blind, placebo-controlled, multicenter study (NCT02817906) investigating the efficacy of lumateperone in reducing dementia-related agitation failed (126).
Moreover, molecules acting via the inhibition of serotonin transporter (SERT) are another class of therapeutic options (218, 219). Dextromethorphan seems to be the most promising because of its high affinity to SERT. To improve its unfavorable pharmacokinetics, dextromethorphan has been combined with a cytochrome P450 2D6 inhibitor, quinidine (220). Quinidine reversibly inhibits cytochrome P450 2D6, thereby prolonging dextromethorphan plasma half-life. The first clinical study, a phase II, randomized, multicenter, double-blind, placebo-controlled trial (NCT01584440), evaluated the efficacy of AVP-923 (dextromethorphan/quinidine) at doses ranging from 20/10 mg once daily to 30/10 mg twice daily for 10 weeks (130). The trial revealed significant effects, compared with placebo, in reducing agitation in 220 patients with dementia. Another recently developed formulation contains a subclinical quinidine dose and deuterated dextromethorphan (AVP-786) (221). Dextromethorphan was deuterated to improve its pharmacokinetic profile and reduce its first-pass metabolism in the liver (221). After its first results, the FDA agreed to initiate a follow-up, phase III clinical trial (NCT02446132) to evaluate the long-term safety and efficacy of AVP-786 (131). In this extension study, three different dextromethorphan doses are administered twice a day over 52 weeks.
Cannabinoid receptors are also a potential target for agitation treatment due to their neuroprotective effects when employed at not psychoactive doses (222). Δ-9-tetrahydrocannabinol (THC) (132), dronabinol, and nabilone are the most widely investigated cannabinoid receptor agonists in clinical trials (222). While THC failed to show efficacy in reducing agitation or aggressiveness, dronabinol showed effects on nighttime agitation (223). Nabilone appeared to be the most promising and was reported to reduce agitation in a randomized, double-blind, placebo-controlled crossover trial (NCT02351882), although patients experienced significant sedation (133).
Finally, the metabotropic glutamate receptor 2 (mGluR2), N-methyl-D-aspartate receptor (NMDA), muscarinic acetylcholine receptor M4 (M4R), and M1/M4 receptors (M1/M4R) (217) are also promising targets of future intervention. A mGluR2 agonist, LY2979165, is not investigated in one phase II clinical trial. Similarly, the indirect NMDA agonist, SND-51, is in one phase II clinical trial. A selective M4 receptor agonist, HTL0016878, is being tested in one phase I clinical trial. Several compounds, acting on pharmacologically relevant targets, such as the 5-HT6 receptor (5-HT6R) and M1/M4R, have also progressed to advanced preclinical development stages (217). Therefore, one can envision drugs targeting mGluR, NMDA receptors, cannabinoid receptor type 1 (CB1R), M1/M4R, and 5-HT6R to be soon part of the pharmacological armamentarium employed for dementia-related agitation.
Conclusions
The agitation associated with neurodegenerative diseases can occur at any disease stage and takes a significant toll on patients and caregivers. The symptom also represents a management challenge for clinicians. To date, precise and useful recommendations are still lacking. However, it is safe to say that pharmacological treatments should be implemented as a second choice, whereas non-pharmacological interventions should be person-centered, preferred, and prioritized. Particular attention and research endeavor should be put into the treatment of elderly patients.
Author Contributions
CC, MR, and LB: paper design and conception. CC, MR, FD, FB, MGR, LF, MD, AD, PA, RS, AG, VF, APi, SS, and LB: manuscript writing. AT, APa, SS, LB, and MO: manuscript revision and editing. CC and MR: figures. All authors agree to be accountable for the content of the work.
Funding
This study was funded by the Alzheimer's Association Part the Cloud: Translational Research Funding for Alzheimer's Disease (PTC) PTC-19-602325 and the Alzheimer's Association—GAAIN Exploration to Evaluate Novel Alzheimer's Queries (GEENA-Q-19-596282) (SLS). AG is supported by the European Union's Horizon 2020 Research and Innovation Program under the Marie Skłodowska-Curie grant agreement iMIND—no. 841665. LB and APi are supported by the Italian Ministry of Health, grant RF-2018-12366209.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Cummings J, Mintzer J, Brodaty H, Sano M, Banerjee S, Devanand DP, et al. Agitation in cognitive disorders: International Psychogeriatric Association provisional consensus clinical and research definition. Int Psychogeriatrics. (2015) 27:7–17. doi: 10.1017/S1041610214001963
2. Ringman JM, Schneider L. Treatment options for agitation in dementia. Curr Treat Options Neurol. (2019) 21:30. doi: 10.1007/s11940-019-0572-3
3. Senanaronga V, Cummings JL, Fairbanks L, Mega M, Masterman DM, O'Connor SM, et al. Agitation in Alzheimer's disease is a manifestation of frontal lobe dysfunction. Dement Geriatr Cogn Disord. (2003) 17:14–20: doi: 10.1159/000074080
4. Anor CJ, O'Connor S, Saund A, Tang-Wai DF, Keren R, Tartaglia MC. Neuropsychiatric symptoms in Alzheimer disease, vascular dementia, and mixed dementia. Neurodegener Dis. (2017) 17:127–34. doi: 10.1159/000455127
5. Kongpakwattana K, Sawangjit R, Tawankanjanachot I, Bell JS, Hilmer SN, Chaiyakunapruk N. Pharmacological treatments for alleviating agitation in dementia: a systematic review and network meta-analysis. Br J Clin Pharmacol. (2018) 84:1445–56. doi: 10.1111/bcp.13604
6. Mintzer JE, Brawman-Mintzer O. Agitation as a possible expression of generalized anxiety disorder in demented elderly patients: toward a treatment approach. J Clin Psychiatry. (1996) 57 (Suppl. 7):55.
7. Kwak YT, Yang Y, Koo MS. Anxiety in dementia. Dement Neurocognitive Disord. (2017) 16:33. doi: 10.12779/dnd.2017.16.2.33
8. Twelftree H, Qazi A. Relationship between anxiety and agitation in dementia. Aging Ment Heal. (2006) 10:362–7. doi: 10.1080/13607860600638511
9. Liu KY, Costello H, Reeves S, Howard R. The relationship between anxiety and incident agitation in Alzheimer's disease. J Alzheimer's Dis. (2020) 78:1119–27. doi: 10.3233/jad-200516
10. Chen ST, Sultzer DL, Hinkin CH, Mahler ME, Cummings JL. Executive dysfunction in Alzheimer's disease: association with neuropsychiatric symptoms and functional impairment. J Neuropsychiatry Clin Neurosci. (1998) 10:426–32. doi: 10.1176/jnp.10.4.426
11. Tekin S, Fairbanks LA, O'Connor S, Rosenberg S, Cummings JL. Activities of daily living in Alzheimer's disease: neuropsychiatric, cognitive, and medical illness influences. Am J Geriatr Psychiatry. (2001) 9:81–6. doi: 10.1097/00019442-200102000-00013
12. Tekin S, Mega MS, Masterman DM, Chow T, Garakian J, Vinters HV, et al. Orbitofrontal and anterior cingulate cortex neurofibrillary tangle burden is associated with agitation in Alzheimer disease. Ann Neurol. (2001) 49:355–61. doi: 10.1002/ana.72
13. Johnson JK, Head E, Kim R, Starr A, Cotman CW. Clinical and pathological evidence for a frontal variant of Alzheimer disease. Arch Neurol. (1999) 56:1233–9. doi: 10.1001/archneur.56.10.1233
14. Esteban De Antonio E, López-Álvarez J, Rábano A, Agüera-Ortiz L, Sánchez-Soblechero A, Amaya L, et al. Pathological correlations of neuropsychiatric symptoms in institutionalized people with dementia. J Alzheimer's Dis. (2020) 78:1731–41. doi: 10.3233/JAD-200600
15. Rapp MA, Schnaider-Beeri M, Purohit DP, Perl DP, Haroutunian V, Sano M. Increased neurofibrillary tangles in patients with Alzheimer disease with comorbid depression. Am J Geriatr Psychiatry. (2008) 16:168–74. doi: 10.1097/JGP.0b013e31816029ec
16. Gotovac K, Nikolac Perković M, Pivac N, Borovečki F. Biomarkers of aggression in dementia. Prog Neuro Psychopharmacology Biol Psychiatry. (2016) 69:125–30. doi: 10.1016/j.pnpbp.2016.03.002
17. Ghetti B, Oblak AL, Boeve BF, Johnson KA, Dickerson BC, Goedert M. Invited review: frontotemporal dementia caused by microtubule-associated protein tau gene (MAPT) mutations: a chameleon for neuropathology and neuroimaging. Neuropathol Appl Neurobiol. (2015) 41:24–46. doi: 10.1111/nan.12213
18. Srikanth S, Nagaraja AV, Ratnavalli E. Neuropsychiatric symptoms in dementia-frequency, relationship to dementia severity and comparison in Alzheimer's disease, vascular dementia and frontotemporal dementia. J Neurol Sci. (2005) 236:43–8. doi: 10.1016/j.jns.2005.04.014
19. McKeith IG, Boeve BF, Dickson DW, Halliday G, Taylor J-P, Weintraub D, et al. Diagnosis and management of dementia with lewy bodies: fourth consensus report of the DLB consortium. Neurology. (2017) 89:88–100. doi: 10.1212/WNL.0000000000004058
20. Goyer PF, Andreason PJ, Semple WE, Clayton AH, King AC, Compton-Toth BA, et al. Positron-emission tomography and personality disorders. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol. (1994) 10:21–8. doi: 10.1038/npp.1994.3
21. Delli Pizzi S, Padulo C, Brancucci A, Bubbico G, Edden RA, Ferretti A, et al. GABA content within the ventromedial prefrontal cortex is related to trait anxiety. Soc Cogn Affect Neurosci. (2016) 11:758–66. doi: 10.1093/scan/nsv155
22. Weissberger GH, Melrose RJ, Narvaez TA, Harwood D, Mandelkern MA, Sultzer DL. 18F-Fluorodeoxyglucose positron emission tomography cortical metabolic activity associated with distinct agitation behaviors in Alzheimer disease. Am J Geriatr Psychiatry. (2017) 25:569–79. doi: 10.1016/j.jagp.2017.01.017
23. Wolpert DM, Goodbody SJ, Husain M. Maintaining internal representations: the role of the human superior parietal lobe. Nat Neurosci. (1998) 1:529–33. doi: 10.1038/2245
24. Delli Pizzi S, Franciotti R, Tartaro A, Caulo M, Thomas A, Onofrj M, et al. Structural alteration of the dorsal visual network in DLB patients with visual hallucinations: a cortical thickness MRI study. PLoS ONE. (2014) 9:e86624. doi: 10.1371/journal.pone.0086624
25. Onofrj M, Bonanni L, Albani G, Mauro A, Bulla D, Thomas A. Visual hallucinations in Parkinson's disease: clues to separate origins. J Neurol Sci. (2006) 248:143–50. doi: 10.1016/j.jns.2006.05.025
26. Onofrj M, Taylor JP, Monaco D, Franciotti R, Anzellotti F, Bonanni L, et al. Visual hallucinations in PD and lewy body dementias: old and new hypotheses. Behav Neurol. (2013) 27:479–93. doi: 10.3233/BEN-129022
27. Sarter M, Bruno JP, Parikh V. Abnormal neurotransmitter release underlying behavioral and cognitive disorders: toward concepts of dynamic and function-specific dysregulation. Neuropsychopharmacology. (2007) 32:1452–61. doi: 10.1038/sj.npp.1301285
28. Trinh N, Hoblyn J, Mohanty S, Yaffe K. Efficacy of cholinesterase inhibitors in the treatment of neuropsychiatric symptoms and functional impairment in Alzheimer disease: a meta-analysis. J Am Med Assoc. (2003) 289:210–16. doi: 10.1001/jama.289.2.210
29. Liu KY, Stringer AE, Reeves SJ, Howard RJ. The neurochemistry of agitation in Alzheimer's disease: a systematic review. Ageing Res Rev. (2018) 43:99–107. doi: 10.1016/j.arr.2018.03.003
30. Szot P, White SS, Greenup JL, Leverenz JB, Peskind ER, Raskind MA. Compensatory changes in the noradrenergic nervous system in the locus ceruleus and hippocampus of postmortem subjects with Alzheimer's disease and dementia with lewy bodies. J Neurosci. (2006) 11:467–78. doi: 10.1523/JNEUROSCI.4265-05.2006
31. Engelborghs S, Vloeberghs E, Le Bastard N, Van Buggenhout M, Mariën P, Somers N, et al. The dopaminergic neurotransmitter system is associated with aggression and agitation in frontotemporal dementia. Neurochem Int. (2008) 52:1052–60. doi: 10.1016/j.neuint.2007.10.018
32. Ooi CH, Yoon PS, How CH, Poon NY. Managing challenging behaviours in dementia. Singapore Med J. (2018) 59:514–8. doi: 10.11622/smedj.2018125
33. Reus VI, Fochtmann LJ, Eyler AE, Hilty DM, Horvitz-Lennon M, Jibson MD, et al. The American psychiatric association practice guideline on the use of antipsychotics to treat agitation or psychosis in patients with dementia. Am J Psychiatry. (2016) 173:543–6. doi: 10.1176/appi.ajp.2015.173501
34. Ismail Z, Smith EE, Geda Y, Sultzer D, Brodaty H, Smith G, et al. Neuropsychiatric symptoms as early manifestations of emergent dementia: provisional diagnostic criteria for mild behavioral impairment. Alzheimers Dement. (2016) 12:195–202. doi: 10.1016/j.jalz.2015.05.017
35. Taragano FE, Allegri RF, Lyketsos C. Mild behavioral impairment: a prodromal stage of dementia. Dement Neuropsychol. (2008) 2:256–60. doi: 10.1590/s1980-57642009dn20400004
36. Taragano FE, Allegri RF, Krupitzki H, Sarasola DR, Serrano CM, Loñ L, et al. Mild behavioral impairment and risk of dementia: a prospective cohort study of 358 patients. J Clin Psychiatry. (2009) 70:584–92. doi: 10.4088/jcp.08m04181
37. De Mendonça A, Ribeiro F, Guerreiro M, Garcia C. Frontotemporal mild cognitive impairment. J Alzheimers Dis. (2004) 6:1–9. doi: 10.3233/jad-2004-6101
38. Frederiksen K.S., Waldemar G. Aggression, agitation, hyperactivity, and irritability. In: Verdelho A, Gonçalves-Pereira M, editors. Neuropsychiatric Symptoms of Cognitive Impairment and Dementia. Cham: Springer (2017). p. 199–236. doi: 10.1007/978-3-319-39138-0_9
39. Kong EH. Agitation in dementia: concept clarification. JAN. (2005) 52:526–36. doi: 10.1111/j.1365-2648.2005.03613.x
40. Bachman D, Rabins P. “Sundowning” and other temporally associated agitation states in dementia patients. Annu Rev Med. (2006) 57:499–511. doi: 10.1146/annurev.med.57.071604.141451
41. Goldberg TE, Chen C, Wang Y, Jung E, Swanson A, Ing C, et al. Association of delirium with long-term cognitive decline: a meta-analysis. JAMA Neurol. (2020) 10032:1–9. doi: 10.1001/jamaneurol.2020.2273
42. Bellelli G, Morandi A, Di Santo SG, Mazzone A, Cherubini A, Mossello E, et al. “Delirium Day”: a nationwide point prevalence study of delirium in older hospitalized patients using an easy standardized diagnostic tool. BMC Med. (2016) 14:106. doi: 10.1186/s12916-016-0649-8
43. D'Ardes D, Carrarini C, Russo M, Dono F, Speranza R, Digiovanni A, et al. Low molecular weight heparin in COVID-19 patients prevents delirium and shortens hospitalization. Neurol Sci. (2020) 42:1527–30. doi: 10.1007/s10072-020-04887-4
44. Cagnin A, Di Lorenzo R, Marra C, Bonanni L, Cupidi C, Laganà V, et al. Behavioral and psychological effects of coronavirus disease-19 quarantine in patients with dementia. Front Psychiatry. (2020) 11:578015. doi: 10.3389/fpsyt.2020.578015
45. Beach SR, Praschan NC, Hogan C, et al. Delirium in COVID-19: a case series and exploration of potential mechanisms for central nervous system involvement. Gen Hosp Psychiatry. (2020) 65:47–53. doi: 10.1016/j.genhosppsych.2020.05.008
46. Han JH, Wilber ST. Altered mental status in older patients in the emergency department. Clin Geriatr Med. (2013) 29:101–36. doi: 10.1016/j.cger.2012.09.005
47. Aftab A, Shah AA. Behavioral emergencies: special considerations in the geriatric psychiatric patient. Psychiatr Clin North Am. (2017) 40:449–62. doi: 10.1016/j.psc.2017.05.010
48. Gottlieb M, Long B, Koyfman A. Approach to the agitated emergency department patient. J Emerg Med. (2018) 54:447–57. doi: 10.1016/j.jemermed.2017.12.049
49. Tampi RR, Tampi DJ. Efficacy and tolerability of benzodiazepines for the treatment of behavioral and psychological symptoms of dementia: a systematic review of randomized controlled trials. Am J Alzheimers Dis Other Demen. (2014) 29:565–74. doi: 10.1177/1533317514524813
50. Ables AZ, Nagubilli R. Prevention, diagnosis, and management of serotonin syndrome. Am Fam Phys. (2010) 81:1139–42.
51. Wolf MU, Goldberg Y, Freedman M. Aggression and agitation in dementia. Continuum. (2018) 24:783–803. doi: 10.1212/CON.0000000000000605
52. Nordstrom K, Zun LS, Wilson MP, Stiebel V, Ng AT, Bregman B, et al. Medical evaluation and triage of the agitated patient: consensus statement of the American association for emergency psychiatry project BETA medical evaluation workgroup. West J Emerg Med. (2012) 13:3–10. doi: 10.5811/westjem.2011.9.6863
53. Zun L, Downey L. Comparison of self-reported to observational agitation scales. Eur Psychiatry. (2017) 41:S151. doi: 10.1016/j.eurpsy.2017.01.2005
54. Bogner JA, Corrigan JD, Fugate L, Mysiw WJ, Clinchot D. Role of agitation in prediction of outcomes after traumatic brain injury. Am J Phys Med Rehabil. (2001) 80:636–44. doi: 10.1097/00002060-200109000-00002
55. Simpson S, Pidgeon M, Nordstrom KD. Using the behavioural activity rating scale as a vital sign in the psychiatric emergency service. Mountain Scholar. (2017). doi: 10.25677/10968/4333
56. Yudofsky SC, Kopecky HJ, Kunik M, Silver JM, Endicott J. The overt agitation severity scale for the objective rating of agitation. J Neuropsychiatry Clin Neurosci. (1997) 9:541–8. doi: 10.1176/jnp.9.4.541
57. Huang HC, Wang YT, Chen KC, Yeh TL, Lee IH, Chen PS, et al. The reliability and validity of the Chinese version of the modified overt aggression scale. Int J Psychiatry Clin Pract. (2009) 13:303–6. doi: 10.3109/13651500903056533
58. Rosen J, Burgio L, Kollar M, Cain M, Allison M, Fogleman M, et al. The pittsburgh agitation scale: a user-friendly instrument for rating agitation in dementia patients. Am J Geriatr Psychiatry. (1994) 2:52–9. doi: 10.1097/00019442-199400210-00008
59. Cohen-Mansfield J. Conceptualization of agitation: results based on the cohen-mansfield agitation inventory and the agitation behavior mapping instrument. Int Psychogeriatrics. (1996) 8:309–15. doi: 10.1017/S1041610297003530
60. Cohen-Mansfield J, Libin A. Assessment of agitation in elderly patients with dementia: correlations between informant rating and direct observation. Int J Geriatr Psychiatry. (2004) 19:881–91. doi: 10.1002/gps.1171
61. Khan BA, Guzman O, Campbell NL, Walroth T, Tricker JL, Hui SL, et al. Comparison and agreement between the richmond agitation-sedation scale and the riker sedation-agitation scale in evaluating patients' eligibility for delirium assessment in the ICU. Chest. (2012) 142:48–54. doi: 10.1378/chest.11-2100
62. Devlin JW, Boleski G, Mlynarek M, Nerenz DR, Peterson E, Jankowski M, et al. Motor activity assessment scale: a valid and reliable sedation scale for use with mechanically ventilated patients in an adult surgical intensive care unit. Crit Care Med. (1999) 27:1271–5. doi: 10.1097/00003246-199907000-00008
63. Sessler CN, Gosnell MS, Grap MJ, Brophy GM, O'Neal P V, Keane KA, et al. The richmond agitation-sedation scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. (2002) 166:1338–44. doi: 10.1164/rccm.2107138
64. Bellelli G, Morandi A, Davis DHJ, Mazzola P, Turco R, Gentile S, et al. Validation of the 4AT, a new instrument for rapid delirium screening: a study in 234 hospitalised older people. Age Ageing. (2014) 43:496–502. doi: 10.1093/ageing/afu021
65. Ely EW, Margolin R, Francis J, May L, Truman B, Dittus R, et al. Evaluation of delirium in critically ill patients: validation of the confusion assessment method for the intensive care unit (CAM-ICU). Crit Care Med. (2001) 29:1370–9. doi: 10.1097/00003246-200107000-00012
66. Frederiksen KS, Cooper C, Frisoni GB, Frölich L, Georges J, Kramberger MG, et al. A European academy of neurology guideline on medical management issues in dementia. Eur J Neurol. (2020) 27:1805–20. doi: 10.1111/ene.14412
67. Brooker D. What is person-centred care in dementia? Rev Clin Gerontol. (2004) 13:215–22. doi: 10.1017/S095925980400108X
68. Keszycki RM, Fisher DW, Dong H. The hyperactivity-impulsivity-irritiability-disinhibition-aggression-agitation domain in Alzheimer's disease: current management and future directions. Front Pharmacol. (2019) 10:1109. doi: 10.3389/fphar.2019.01109
69. Chenoweth L, King MT, Jeon YH, Brodaty H, Stein-Parbury J, Norman R, et al. Caring for aged dementia care resident study (CADRES) of person-centred care, dementia-care mapping, and usual care in dementia: a cluster-randomised trial. Lancet Neurol. (2009) 8:317–25. doi: 10.1016/S1474-4422(09)70045-6
70. Sloane PD, Hoeffer B, Mitchell CM, McKenzie DA, Barrick AL, Rader J, et al. Effect of person-centered showering and the towel bath on bathing-associated aggression, agitation, and discomfort in nursing home residents with dementia: a randomized, controlled trial. J Am Geriatr Soc. (2004) 52:1795–804. doi: 10.1111/j.1532-5415.2004.52501.x
71. Fossey J, Ballard C, Juszczak E, James I, Alder N, Jacoby R, et al. Effect of enhanced psychosocial care on antipsychotic use in nursing home residents with severe dementia: cluster randomised trial. BMJ. (2006) 332:756–61. doi: 10.1136/bmj.38782.575868.7C
72. Surr CA, Holloway I, Walwyn REA, Griffiths AW, Meads D, Martin A, et al. Effectiveness of dementia care mappingTM to reduce agitation in care home residents with dementia: an open-cohort cluster randomised controlled trial. Aging Ment Health. (2020) 1–14. doi: 10.1080/13607863.2020.1745144. [Epub ahead of print].
73. Rokstad AMM, Røsvik J, Kirkevold Ø, Selbaek G, Saltyte Benth J, Engedal K. The effect of person-centred dementia care to prevent agitation and other neuropsychiatric symptoms and enhance quality of life in nursing home patients: a 10-month randomized controlled trial. Dement Geriatr Cogn Disord. (2013) 36:340–53. doi: 10.1159/000354366
74. Livingston G, Barber J, Marston L, Stringer A, Panca M, Hunter R, et al. Clinical and cost-effectiveness of the managing agitation and raising quality of life (MARQUE) intervention for agitation in people with dementia in care homes: a single-blind, cluster-randomised controlled trial. Lancet Psychiatry. (2019) 6:293–304. doi: 10.1016/S2215-0366(19)30045-8
75. Pedersen SKA, Andersen PN, Lugo RG, Andreassen M, Sütterlin S. Effects of music on agitation in dementia: a meta-analysis. Front Psychol. (2017) 8:742. doi: 10.3389/fpsyg.2017.00742
76. Lin Y, Chu H, Yang C-Y, Chen C-H, Chen S-G, Chang H-J, et al. Effectiveness of group music intervention against agitated behavior in elderly persons with dementia. Int J Geriatr Psychiatry. (2011) 26:670–8. doi: 10.1002/gps.2580
77. Lin LW, Weng SC, Wu HS, Tsai LJ, Lin YL, Yeh SH. The effects of white noise on agitated behaviors, mental status, and activities of daily living in older adults with dementia. J Nurs Res. (2018) 26:2–9. doi: 10.1097/JNR.0000000000000211
78. Moyle W, Murfield J, Jones C, Beattie E, Draper B, Moyle W, et al. Can lifelike baby dolls reduce symptoms of anxiety, agitation, or aggression for people with dementia in long-term care? Findings from a pilot randomised controlled trial. Aging Ment Health. (2018) 23:1442–50. doi: 10.1080/13607863.2018.1498447
79. Braden BA, Gaspar PM. Implementation of a baby doll therapy protocol for people with dementia: innovative practice. Dementia. (2015) 14:696–706. doi: 10.1177/1471301214561532
80. Cohen-Mansfield J, Dakheel-Ali M, Marx MS, Thein K, Regier NG. Which unmet needs contribute to behavior problems in persons with advanced dementia? Psychiatry Res. (2015) 228:59–64. doi: 10.1016/j.psychres.2015.03.043
81. Cohen-Mansfield J, Marx MS, Dakheel-Ali M, Regier NG, Thein K, Freedman L. Can agitated behavior of nursing home residents with dementia be prevented with the use of standardized stimuli? J Am Geriatr Soc. (2010) 58:1459–64. doi: 10.1111/j.1532-5415.2010.02951.x
82. Cohen-Mansfield J, Thein K, Marx MS, Dakheel-Ali M, Freedman L. Efficacy of nonpharmacologic interventions for agitation in advanced dementia: a randomized, placebo-controlled trial. J Clin Psychiatry. (2012) 73:1255–61. doi: 10.4088/JCP.12m07918
83. Homdee N, Alam R, Hayes JA, Hamid T, Park J, Wolfe S, et al. Agitation monitoring and prevention system for dementia caregiver empowerment. Computer. (2019) 52:30–9. doi: 10.1109/MC.2019.2933192
84. Alam R, Homdee N, Wolfe S, Hayes J, Lach J. Besi: behavior learning and tracking with wearable and in-home sensors - a dementia case-study: poster abstract. In: IoTDI 2019 - Proceeding 2019 Internet Things Des Implement. Montreal, QC, (2019). p. 281–2. doi: 10.1145/3302505.3312595
85. Anderson MS, Homdee N, Bankole A, Alam R, Mitchell B, Hayes J, et al. Behavioral interventions for alzheimer's management using technology: home-based monitoring. Curr Geriatr Rep. (2020) 9:90–100. doi: 10.1007/s13670-020-00312-y
86. Catic AG, Mattison MLP, Bakaev I, Morgan M, Monti SM, Lipsitz L. ECHO-AGE: an innovative model of geriatric care for long-term care residents with dementia and behavioral issues. J Am Med Dir Assoc. (2014) 15:938–42. doi: 10.1016/j.jamda.2014.08.014
87. Strong J. Immersive virtual reality and persons with dementia: a literature review. J Gerontol Soc Work. (2020) 63:209–26. doi: 10.1080/01634372.2020.1733726
88. D'Cunha NM, Nguyen D, Naumovski N, McKune AJ, Kellett J, Georgousopoulou EN, et al. A mini-review of virtual reality-based interventions to promote well-being for people living with dementia and mild cognitive impairment. Gerontology. (2019) 65:430–40. doi: 10.1159/000500040
89. Hirt J, Beer T. Use and impact of virtual reality simulation in dementia care education: a scoping review. Nurse Educ Today. (2020) 84:104207. doi: 10.1016/j.nedt.2019.104207
90. Tampi RR, Tampi DJ, Young J, Hoq R, Resnick K. The place for electroconvulsive therapy in the management of behavioral and psychological symptoms of dementia. Neurodegener Dis Manag. (2019). doi: 10.2217/nmt-2019-0018. [Epub ahead of print].
91. Levy I, Attias S, Gavrieli S, Oliven R, Neerland BE, Devlin JW, et al. The study protocol for a randomized, controlled trial of acupuncture versus a sham procedure versus standard care alone for the treatment of delirium in acutely hospitalized older adults with a medical illness. Maturitas. (2019) 124:72–80. doi: 10.1016/j.maturitas.2019.03.001
92. Gkioka M, Schneider J, Kruse A, Tsolaki M, Moraitou D, Teichmann B. Evaluation and effectiveness of dementia staff training programs in general hospital settings: a narrative synthesis with holton's three-level model applied. J Alzheimer's Dis. (2020) 78:1089–1108. doi: 10.3233/jad-200741
93. Sampson EL, White N, Lord K, Leurent B, Vickerstaff V, Scott S, et al. Pain, agitation, and behavioural problems in people with dementia admitted to general hospital wards: a longitudinal cohort study. Pain. (2015) 156:675–83. doi: 10.1097/j.pain.0000000000000095
94. Rossi G, Oh JC. Management of agitation in huntington's disease: a review of the literature. Cureus. (2020) 12:e9784. doi: 10.7759/cureus.9748
95. Glass OM, Hermida AP, Hershenberg R, Schwartz AC. Considerations and current trends in the management of the geriatric patient on a consultation-liaison service. Curr Psychiatry Rep. (2020) 22:21. doi: 10.1007/s11920-020-01147-2
96. Regan NAO, Fitzgerald J, Timmons S, Connell HO, Meagher D. Delirium: a key challenge for perioperative care. Int J Surg. (2013) 11:136–44. doi: 10.1016/j.ijsu.2012.12.015
97. Van Velthuijsen EL, Zwakhalen SMG, Mulder WJ, Verhey FRJ, Kempen GIJM. Detection and management of hyperactive and hypoactive delirium in older patients during hospitalization: a retrospective cohort study evaluating daily practice. Int J Geriatr Psychiatry. (2018) 33:1521–9. doi: 10.1002/gps.4690
98. Goldberg A, Straus SE, Hamid JS, Wong CL. Room transfers and the risk of delirium incidence amongst hospitalized elderly medical patients: a case-control study. BMC Geriatr. (2015) 15:69. doi: 10.1186/s12877-015-0070-8
99. Flaherty JH, Little MO. Matching the environment to patients with delirium: lessons learned from the delirium room, a restraint-free environment for older hospitalized adults with delirium. J Am Geriatr Soc. (2011) 59 (Suppl. 2):S295–300. doi: 10.1111/j.1532-5415.2011.03678.x
100. Flaherty JH, Tariq SH, Raghavan S, Bakshi S, Moinuddin A, Morley JE. A model for managing delirious older inpatients. J Am Geriatr Soc. (2003) 51:1031–5. doi: 10.1046/j.1365-2389.2003.51320.x
101. Chong MS, Chan MPC, Kang J, Han HC, Ding YY, Tan TL. A new model of delirium care in the acute geriatric setting: geriatric monitoring unit. BMC Geriatr. (2011) 11:41. doi: 10.1186/1471-2318-11-41
102. Chong MS, Chan M, Tay L, Ding YY. Outcomes of an innovative model of acute delirium care: the geriatric monitoring unit (GMU). Clin Interv Aging. (2014) 9:603–12. doi: 10.2147/CIA.S60259
103. Steinberg M, Lyketsos CG. Atypical antipsychotic use in patients with dementia: managing safety concerns. Am J Psychiatry. (2012) 169:900–6. doi: 10.1176/appi.ajp.2012.12030342
104. Sheldon T. Dutch doctors should tackle female genital mutilation. BMJ. (2005) 330:922. doi: 10.1136/bmj.330.7497.922-a
105. Kuehn BM. FDA warns antipsychotic drugs may be risky for elderly. JAMA. (2005) 293:2462. doi: 10.1001/jama.293.20.2462
106. Pollock BG, Mulsant BH, Rosen J, Sweet RA, Mazumdar S, Bharucha A, et al. Comparison of citalopram, perphenazine, and placebo for the acute treatment of psychosis and behavioral disturbances in hospitalized, demented patients. Res Pract Alzheimers Dis. (2003) 7:180–6. doi: 10.1176/appi.ajp.159.3.460
107. Pollock BG, Mulsant BH, Rosen J, Mazumdar S, Blakesley RE, Houck PR, et al. A double-blind comparison of citalopram and risperidone for the treatment of behavioral and psychotic symptoms associated with dementia. Am J Geriatr Psychiatry. (2007) 15:942–52. doi: 10.1097/JGP.0b013e3180cc1ff5
108. Porsteinsson AP, Drye LT, Pollock BG, Devanand DP, Frangakis C, Ismail Z, et al. Effect of citalopram on agitation in Alzheimer disease: the CitAD randomized clinical trial. JAMA. (2014) 311:682–91. doi: 10.1001/jama.2014.93
109. Viscogliosi G, Chiriac IM, Ettorre E. Efficacy and safety of citalopram compared to atypical antipsychotics on agitation in nursing home residents with alzheimer dementia. J Am Med Dir Assoc. (2017) 18:799–802. doi: 10.1016/j.jamda.2017.06.010
110. Barak Y, Plopski I, Tadger S, Paleacu D. Escitalopram versus risperidone for the treatment of behavioral and psychotic symptoms associated with Alzheimer's disease: a randomized double-blind pilot study. Int Psychogeriatrics. (2011) 23:1515–9. doi: 10.1017/S1041610211000743
111. Lanctôt KL, Herrmann N, Van Reekum R, Eryavec G, Naranjo CA. Gender, aggression and serotonergic function are associated with response to sertraline for behavioral disturbances in Alzheimer's disease. Int J Geriatr Psychiatry. (2002) 17:531–41. doi: 10.1002/gps.636
112. Cakir S, Kulaksizoglu IB. The efficacy of mirtazapine in agitated patients with Alzheimer's disease: a 12-week open-label pilot study. Neuropsychiatr Dis Treat. (2008) 4:963–6. doi: 10.2147/ndt.s3201
113. Fox C, Crugel M, Maidment I, Auestad BH, Coulton S, Treloar A, et al. Efficacy of memantine for agitation in Alzheimer's dementia: a randomised double-blind placebo controlled trial. PLoS ONE. (2012) 7:e35185. doi: 10.1371/journal.pone.0035185
114. Ballard C, Thomas A, Gerry S, Yu LM, Aarsland D, Merritt C, et al. A double-blind randomized placebo-controlled withdrawal trial comparing memantine and antipsychotics for the long-term treatment of function and neuropsychiatric symptoms in people with Alzheimer's disease (MAIN-AD). J Am Med Dir Assoc. (2015) 16:316–22. doi: 10.1016/j.jamda.2014.11.002
115. Gallagher D, Herrmann N. Antiepileptic drugs for the treatment of agitation and aggression in dementia: Do they have a place in therapy? Drugs. (2014) 74:1747–55. doi: 10.1007/s40265-014-0293-6
116. Sommer OH, Aga O, Cvancarova M, Olsen IC, Selbaek G, Engedal K. Effect of oxcarbazepine in the treatment of agitation and aggression in severe dementia. Dement Geriatr Cogn Disord. (2009) 27:155–63. doi: 10.1159/000199236
117. Kurlan R, Cummings J, Raman R, Thal L. Quetiapine for agitation or psychosis in patients with dementia and parkinsonism. Neurology. (2007) 68:1356–63. doi: 10.1212/01.wnl.0000260060.60870.89
118. McKeith I, Del Ser T, Spano P, Emre M, Wesnes K, Anand R, et al. Efficacy of rivastigmine in dementia with lewy bodies: a randomised, double-blind, placebo-controlled international study. Lancet. (2000) 356:2031–6. doi: 10.1016/S0140-6736(00)03399-7
119. Emre M, Aarsland D, Albanese A, Byrne EJ, Deuschl G, De Deyn PP, et al. Rivastigmine for dementia associated with Parkinson's disease. N Engl J Med. (2004) 351:2509–18. doi: 10.1056/NEJMoa041470
120. Aarsland D, Laake K, Larsen JP, Janvin C. Donepezil for cognitive impairment in Parkinson's disease: a randomised controlled study. J Neurol Neurosurg Psychiatry. (2002) 72:708–12. doi: 10.1136/jnnp.72.6.708
121. Leroi I, Brandt J, Reich SG, Lyketsos CG, Grill S, Thompson R, et al. Randomized placebo-controlled trial of donepezil in cognitive impairment in Parkinson's disease. Int J Geriatr Psychiatry. (2004) 19:1–8. doi: 10.1002/gps.993
122. Ravina B, Putt M, Siderowf A, Farrar JT, Gillespie M, Crawley A, et al. Donepezil for dementia in Parkinson's disease: a randomised, double blind, placebo controlled, crossover study. J Neurol Neurosurg Psychiatry. (2005) 76:934–9. doi: 10.1136/jnnp.2004.050682
123. Lebert F, Stekke W, Hasenbroekx C, Pasquier F. Frontotemporal dementia: a randomised, controlled trial with trazodone. Dement Geriatr Cogn Disord. (2004) 17:355–9. doi: 10.1159/000077171
124. Ballard C, Sauter M, Scheltens P, He Y, Barkhof F, van Straaten ECW, et al. Efficacy, safety and tolerability of rivastigmine capsules in patients with probable vascular dementia: the VantagE study. Curr Med Res Opin. (2008) 24:2561–74. doi: 10.1185/03007990802328142
125. Yunusa I, El Helou ML, Alsahali S. Pimavanserin: a novel antipsychotic with potentials to address an unmet need of older adults with dementia-related psychosis. Front Pharmacol. (2020) 11:87. doi: 10.3389/fphar.2020.00087
126. ClinicalTrials.gov. ITI-007 for the Treatment of Agitation in Patients With Dementia, Including Alzheimer's Disease. Available online at: https://clinicaltrials.gov/ct2/show/NCT02817906
127. ClinicalTrials.gov. A Trial to Evaluate the Safety, Efficacy, and Tolerability of Brexpiprazole in Treating Agitation Associated With Dementia of the Alzheimer's Type. Available online at: https://clinicaltrials.gov/ct2/show/NCT03548584
128. ClinicalTrials.gov. Brexpiprazole for the Long-term Treatment of Patients With Agitation Associated With Dementia of the Alzheimer's Type. Availablee online at: https://clinicaltrials.gov/ct2/show/NCT03724942
129. ClinicalTrials.gov. Brexpiprazole for the Treatment of Patients With Agitation Associated With Dementia of the Alzheimer's Type. Available online at: https://clinicaltrials.gov/ct2/show/NCT03620981
130. ClinicalTrials.gov. Efficacy, Safety and Tolerability Study of AVP-923 (Dextromethorphan/Quinidine) for Treatment of Symptoms of Agitation in Alzheimer's Patients. Available online at: https://clinicaltrials.gov/ct2/show/NCT01584440
131. ClinicalTrials.gov. Long Term, Extension Study of the Safety and Efficacy of AVP-786 for the Treatment of Agitation in Patients With Dementia of the Alzheimer's Type. Available online at: https://clinicaltrials.gov/ct2/show/NCT02446132
132. ClinicalTrials.gov. The Effect of Cannabis on Dementia Related Agitation and Aggression. Available online at: https://clinicaltrials.gov/ct2/show/NCT03328676
133. ClinicalTrials.gov. Safety and Efficacy of Nabilone in Alzheimer's Disease. Available online at: https://clinicaltrials.gov/ct2/show/NCT02351882
134. Oh ES, Fong TG, Hshieh TT, Inouye SK. Delirium in older persons: advances in diagnosis and treatment. JAMA. (2017) 318:1161–1174. doi: 10.1001/jama.2017.12067
135. Lauretani F, Bellelli G, Pelà G, Morganti S, Tagliaferri S, Maggio M. Treatment of delirium in older persons: what we should not do! Int J Mol Sci. (2020) 21:2397. doi: 10.3390/ijms21072397
136. Breitbart W, Marotta R, Platt MM, Weisman H, Derevenco M, Grau C, et al. A double-blind trial of haloperidol, chlorpromazine, and lorazepam in the treatment of delirium in hospitalized AIDS patients. Am J Psychiatry. (1996) 153:231–7. doi: 10.1176/ajp.153.2.231
137. Jain R, Arun P, Sidana A, Sachdev A. Comparison of efficacy of haloperidol and olanzapine in the treatment of delirium. Indian J Psychiatry. (2017) 59:451–6. doi: 10.4103/psychiatry.IndianJPsychiatry_59_17
138. Satterthwaite TD, Wolf DH, Rosenheck RA, Gur RE, Caroff SN. A meta-analysis of the risk of acute extrapyramidal symptoms with intramuscular antipsychotics for the treatment of agitation. J Clin Psychiatry. (2008) 69:1869–79. doi: 10.4088/jcp.v69n1204
139. Han C-S, Kim Y-K. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. (2004) 45:297–301. doi: 10.1016/S0033-3182(04)70170-X
140. Kim JY, Jung IK, Han C, Cho SH, Kim L, Kim SH, et al. Antipsychotics and dopamine transporter gene polymorphisms in delirium patients. Psychiatry Clin Neurosci. (2005) 59:183–8. doi: 10.1111/j.1440-1819.2005.01355.x
141. Telles-Correia D, Barbosa A, Cortez-Pinto H, Campos C, Rocha NBF, Machado S. Psychotropic drugs and liver disease: a critical review of pharmacokinetics and liver toxicity. World J Gastrointest Pharmacol Ther. (2017) 8:26–38. doi: 10.4292/wjgpt.v8.i1.26
142. Masopust J, Protopopová D, Vališ M, Pavelek Z, Klímová B. Treatment of behavioral and psychological symptoms of dementias with psychopharmaceuticals: a review. Neuropsychiatr Dis Treat. (2018) 14:1211–1220. doi: 10.2147/NDT.S163842
143. Baillargeon J, Singh G, Kuo Y-F, Raji MA, Westra J, Sharma G. Association of opioid and benzodiazepine use with adverse respiratory events in older adults with chronic obstructive pulmonary disease. Ann Am Thorac Soc. (2019) 16:1245–51. doi: 10.1513/AnnalsATS.201901-024OC
144. Sachdeva A, Choudhary M, Chandra M. Alcohol withdrawal syndrome: benzodiazepines and beyond. J Clin Diagn Res. (2015) 9:VE01–7. doi: 10.7860/JCDR/2015/13407.6538
145. Griffin CE III, Kaye AM, Bueno FR, Kaye AD. Benzodiazepine pharmacology and central nervous system-mediated effects. Ochsner J. (2013) 13:214–23.
146. Baghdady NT, Banik S, Swartz SA, McIntyre RS. Psychotropic drugs and renal failure: translating the evidence for clinical practice. Adv Ther. (2009) 26:404–24. doi: 10.1007/s12325-009-0021-x
147. Fong TG, Tulebaev SR, Inouye SK. Delirium in elderly adults: diagnosis, prevention and treatment. Nat Rev Neurol. (2009) 5:210–20. doi: 10.1038/nrneurol.2009.24
148. Martinón-Torres G, Fioravanti M, Grimley Evans J. Trazodone for agitation in dementia. Cochrane Database Syst Rev. (2004) CD004990. doi: 10.1002/14651858.cd004990
149. López-Pousa S, Garre-Olmo J, Vilalta-Franch J, Turon-Estrada A, Pericot-Nierga I. Trazodone for Alzheimer's disease: a naturalistic follow-up study. Arch Gerontol Geriatr. (2008) 47:207–15. doi: 10.1016/j.archger.2007.07.010
150. Sampson EL, Raven PR, Ndhlovu PN, Vallance A, Garlick N, Watts J, et al. A randomized, double-blind, placebo-controlled trial of donepezil hydrochloride (Aricept) for reducing the incidence of postoperative delirium after elective total hip replacement. Int J Geriatr Psychiatry. (2007) 22:343–9. doi: 10.1002/gps.1679
151. Noyan MA, Elbi H, Aksu H. Donepezil for anticholinergic drug intoxication: a case report. Prog Neuropsychopharmacol Biol Psychiatry. (2003) 27:885–7. doi: 10.1016/s0278-5846(03)00119-2
152. Wengel SP, Roccaforte WH, Burke WJ. Donepezil improves symptoms of delirium in dementia: implications for future research. J Geriatr Psychiatry Neurol. (1998) 11:159–61. doi: 10.1177/089198879801100308
153. Liptzin B, Laki A, Garb JL, Fingeroth R, Krushell R. Donepezil in the prevention and treatment of post-surgical delirium. Am J Geriatr Psychiatry. (2005) 13:1100–6. doi: 10.1097/00019442-200512000-00010
154. Maher AR, Theodore G. Summary of the comparative effectiveness review on off-label use of atypical antipsychotics. J Manag Care Pharm. (2012) 18:5 (Supp. B):S1–20. doi: 10.18553/jmcp.2012.18.s5-b.1
155. Sultzer DL, Davis SM, Tariot PN, Dagerman KS, Lebowitz BD, Lyketsos CG, et al. Clinical symptom responses to atypical antipsychotic medications in Alzheimer's disease: phase1outcomes from the CATIE-AD effectiveness trial. Am J Psychiatry. (2008) 165:844–54. doi: 10.1176/appi.ajp.2008.07111779
156. Aga VM. When and how to treat agitation in alzheimer's disease dementia with citalopram and escitalopram. Am J Geriatr Psychiatry. (2019) 27:1099–107. doi: 10.1016/j.jagp.2019.04.016
157. Marcum ZA VG. Clinical considerations for older adults. Am J Geriatr Pharmacother. (2013) 10:264–71. doi: 10.1016/j.amjopharm.2012.05.002.FDA
158. Žmuc Veranič L, Pregelj P, Jerin A. Treating agitation and aggression in patients with Alzheimer's disease with escitalopram. Psychogeriatrics. (2016) 16:384–5. doi: 10.1111/psyg.12171
159. Marcum ZA, Vande Griend JP, Linnebur SA. FDA drug safety communications: a narrative review and clinical considerations for older adults. Am J Geriatr Pharmacother. (2012) 10:264–71. doi: 10.1016/j.amjopharm.2012.05.002
160. Howard RJ, Juszczak E, Ballard CG, Bentham P, Brown RG, Bullock R, et al. Donepezil for the treatment of agitation in Alzheimer's disease. N Engl J Med. (2007) 357:1382–92. doi: 10.1056/nejmoa066583
161. Ballard C, Margallo-Lana M, Juszczak E, Douglas S, Swann A, Thomas A, et al. Quetiapine and rivastigmine and cognitive decline in Alzheimer's disease: randomised double blind placebo controlled trial. Br Med J. (2005) 330:874–7. doi: 10.1136/bmj.38369.459988.8F
162. Nakayama S, Suda A, Nakanishi A, Motoi Y, Hattori N. Galantamine response associates with agitation and the prefrontal cortex in patients with Alzheimer's disease. J Alzheimer's Dis. (2017) 57:267–73. doi: 10.3233/JAD-160902
163. Creese B, Da Silva MV, Johar I, Ballard C. The modern role of antipsychotics for the treatment of agitation and psychosis in Alzheimer's disease. Expert Rev Neurother. (2018) 18:461–7. doi: 10.1080/14737175.2018.1476140
164. Wang LY, Shofer JB, Rohde K, Hart KL, Hoff DJ, McFall YH, et al. Prazosin for the treatment of behavioral symptoms in patients with Alzheimer disease with agitation and aggression. Am J Geriatr Psychiatry. (2009) 17:744–51. doi: 10.1097/JGP.0b013e3181ab8c61
165. Thein T, Koene RA, Wijdeveld PG. Orthostatic hypotension due to prazosin. Lancet. (1977) 1:363. doi: 10.1016/s0140-6736(77)91163-1
166. O'Brien JT, Thomas A. Vascular dementia. Lancet. (2015) 386:1698–706. doi: 10.1016/S0140-6736(15)00463-8
167. Lam SM, Wang Y, Duan X, Wenk MR, Kalaria RN, Chen CP, et al. The brain lipidomes of subcortical ischemic vascular dementia and mixed dementia. Neurobiol Aging. (2014) 35:2369–81. doi: 10.1016/j.neurobiolaging.2014.02.025
168. Davies SJC, Burhan AM, Kim D, Gerretsen P, Graff-Guerrero A, Woo VL, et al. Sequential drug treatment algorithm for agitation and aggression in Alzheimer's and mixed dementia. J Psychopharmacol. (2018) 32:509–23. doi: 10.1177/0269881117744996
169. Durán JC, Greenspan A, Diago JI, Gallego R, Martinez G. Evaluation of risperidone in the treatment of behavioral and psychological symptoms and sleep disturbances associated with dementia. Int psychogeriatrics. (2005) 17:591–604. doi: 10.1017/S104161020500219X
170. Schneider LS, Dagerman K, Insel PS. Efficacy and adverse effects of atypical antipsychotics for dementia: meta-analysis of randomized, placebo-controlled trials. Am J Geriatr Psychiatry. (2006) 14:191–210. doi: 10.1097/01.JGP.0000200589.01396.6d
171. Suh G-H, Greenspan AJ, Choi SK. Comparative efficacy of risperidone versus haloperidol on behavioural and psychological symptoms of dementia. Int J Geriatr Psychiatry. (2006) 21:654–60. doi: 10.1002/gps.1542
172. Lin YT, Hwang TJ, Shan JC, Chiang HL, Sheu YH, Hwu HG. Dosage and duration of antipsychotic treatment in demented outpatients with agitation or psychosis. J Formos Med Assoc. (2015) 114:147–53. doi: 10.1016/j.jfma.2012.09.002
173. Jeste D V, Blazer D, Casey D, Meeks T, Salzman C, Schneider L, et al. ACNP white paper: update on use of antipsychotic drugs in elderly persons with dementia. Neuropsychopharmacol. (2008) 33:957–70. doi: 10.1038/sj.npp.1301492
174. Cheung G, Stapelberg J. Quetiapine for the treatment of behavioural and psychological symptoms of dementia (BPSD): a meta-analysis of randomised placebo-controlled trials. N Z Med J. (2011) 124:39–50.
175. Bui Q. Antidepressants for agitation and psychosis in patients with dementia. Am Fam Phys. (2012) 85:20–2.
176. Malouf R, Birks J. Donepezil for vascular cognitive impairment. Cochrane database Syst Rev. (2004) CD004395. doi: 10.1002/14651858.CD004395.pub2
177. Birks J, McGuinness B, Craig D. Rivastigmine for vascular cognitive impairment. Cochrane database Syst Rev. (2013) CD004744. doi: 10.1002/14651858.CD004744.pub3
178. Craig D, Birks J. Galantamine for vascular cognitive impairment. Cochrane database Syst Rev. (2006) CD004746. doi: 10.1002/14651858.CD004746.pub2
179. Matsunaga S, Kishi T, Yasue I, Iwata N. Cholinesterase inhibitors for lewy body disorders: a meta-analysis. Int J Neuropsychopharmacol. (2016) 19:1–15. doi: 10.1093/ijnp/pyv086
180. Boot BP, McDade EM, McGinnis SM, Boeve BF. Treatment of dementia with lewy bodies. Curr Treat Options Neurol. (2013) 15:738–64. doi: 10.1007/s11940-013-0261-6
181. Aarsland D, Perry R, Larsen JP, McKeith IG, O'Brien JT, Perry EK, et al. Neuroleptic sensitivity in Parkinson's disease and parkinsonian dementias. J Clin Psychiatry. (2005) 66:633–37. doi: 10.4088/JCP.v66n0514
182. Shea YF, Chu L-W. Neuroleptic malignant syndrome caused by quetiapine in an elderly man with lewy body dementia. J Am Geriatr Soc. (2016) 64:e55–6. doi: 10.1111/jgs.14184
183. Teng PR, Yeh CH, Lin CY, Lai TJ. Olanzapine-induced neuroleptic malignant syndrome in a patient with probable dementia with lewy bodies. J Neuropsychiatry Clin Neurosci. (2012) 24:E1–2. doi: 10.1176/appi.neuropsych.11090212
184. Bonanni L, Di Giacomo R, D'Amico A, Frazzini V, Franciotti R, Manzoli L, et al. Akinetic crisis in dementia with lewy bodies. J Neurol Neurosurg Psychiatry. (2016) 87:1123–6. doi: 10.1136/jnnp-2015-312914
185. Lee HB, Hanner JA, Yokley JL, Appleby B, Hurowitz L, Lyketsos CG. Clozapine for treatment-resistant agitation in dementia. J Geriatr Psychiatry Neurol. (2007) 20:178–82. doi: 10.1177/0891988707303335
186. Teri L, Logsdon RG, Peskind E, Raskind M, Weiner MF, Tractenberg RE, et al. Treatment of agitation in AD: a randomized, placebo-controlled clinical trial. Neurology. (2000) 55:1271–8. doi: 10.1212/WNL.55.9.1271
187. O'Brien JT, Holmes C, Jones M, Jones R, Livingston G, McKeith I, et al. Clinical practice with anti-dementia drugs: a revised (third) consensus statement from the British association for psychopharmacology. J Psychopharmacol. (2017) 31:147–68. doi: 10.1177/0269881116680924
188. Mendez MF. Frontotemporal dementia: therapeutic interventions. Front Neurol Neurosci. (2009) 24:168–78. doi: 10.1159/000197896
189. Logroscino G, Imbimbo BP, Lozupone M, Sardone R, Capozzo R, Battista P, et al. Promising therapies for the treatment of frontotemporal dementia clinical phenotypes: from symptomatic to disease-modifying drugs. Expert Opin Pharmacother. (2019) 20:1091–107. doi: 10.1080/14656566.2019.1598377
190. Murley AG, Rowe JB. Neurotransmitter deficits from frontotemporal lobar degeneration. Brain. (2018) 141:1263–85. doi: 10.1093/brain/awx327
191. Swartz JR, Miller BL, Lesser IM, Darby AL. Frontotemporal dementia: treatment response to serotonin selective reuptake inhibitors. J Clin Psychiatry. (1997) 58:212–6.
192. Herrmann N, Lanctôt KL, Hogan DB. Pharmacological recommendations for the symptomatic treatment of dementia: the Canadian consensus conference on the diagnosis and treatment of dementia 2012. Alzheimers Res Ther. (2013) 5:S5. doi: 10.1186/alz193
193. Prodan CI, Monnot M, Ross ED. Behavioural abnormalities associated with rapid deterioration of language functions in semantic dementia respond to sertraline. J Neurol Neurosurg Psychiatry. (2009) 80:1416–7. doi: 10.1136/jnnp.2009.173260
194. Freedman M. Frontotemporal dementia: recommendations for therapeutic studies, designs, and approaches. Can J Neurol Sci. (2007) 34 (Suppl. 1):S118–24. doi: 10.1017/s0317167100005680
195. Boxer AL, Boeve BF. Frontotemporal dementia treatment: current symptomatic therapies and implications of recent genetic, biochemical, and neuroimaging studies. Alzheimer Dis Assoc Disord. (2007) 21:S79–87. doi: 10.1097/WAD.0b013e31815c345e
196. Panza F, Solfrizzi V, Seripa D, Imbimbo BP, Santamato A, Lozupone M, et al. Progresses in treating agitation: a major clinical challenge in Alzheimer's disease. Expert Opin Pharmacother. (2015) 16:2581–8. doi: 10.1517/14656566.2015.1092520
197. Devanand DP, Pelton GH, D'Antonio K, Strickler JG, Kreisl WC, Noble J, et al. Low-dose lithium treatment for agitation and psychosis in alzheimer disease and frontotemporal dementia. Alzheimer Dis Assoc Disord. (2017) 31:73–5. doi: 10.1097/WAD.0000000000000161
198. Sheldon LJ, Ancill RJ, Holliday SG. Gabapentin in geriatric psychiatry patients. Can J Psychiatry. (1998) 43:422–3.
199. Ellul MA, Benjamin L, Singh B, Lant S, Michael BD, Easton A, et al. Neurological associations of COVID-19. Lancet Neurol. (2020) 19:767–83. doi: 10.1016/S1474-4422(20)30221-0
200. Pilotto A, Benussi A, Libri I et al. COVID-19 impact on consecutive neurological patients admitted to the emergency department. J Neurol Neurosurg Psychiatry. (2020) 92:218–20. doi: 10.1136/jnnp-2020-323929
201. Bianchetti A, Rozzini R, Guerini F, Boffelli S, Ranieri P, Minelli G, et al. Clinical presentation of COVID19 in dementia patients. J Nutr Health Aging. (2020) 24:560–2. doi: 10.1007/s12603-020-1389-1
202. Kennedy M, Helfand BKI, Gou RY, Gartaganis SL, Webb M, Moccia JM, et al. Delirium in older patients with COVID-19 presenting to the emergency department. JAMA Netw open. (2020) 3:e2029540. doi: 10.1001/jamanetworkopen.2020.29540
203. Chen T, Wu D, Chen H, Yan W, Yang D, Chen G, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. (2020) 368:m1091. doi: 10.1136/bmj.m1091
204. Wong AH, Roppolo LP, Chang BP, Yonkers KA, Wilson MP, Powsner S, et al. Management of agitation during the COVID-19 pandemic. West J Emerg Med. (2020) 21:795–800. doi: 10.5811/westjem.2020.5.47789
205. Pilotto A, Masciocchi S, Volonghi I, Crabbio M, Magni E, De Giuli V, et al. Clinical presentation and outcomes of SARS-CoV-2 related encephalitis: the ENCOVID multicentre study. J Infect Dis. (2020) 223:28–37. doi: 10.1093/infdis/jiaa609
206. Inouye SK, Westendorp RGJ, Saczynski JS. Delirium in elderly people. Lancet. (2014) 383:911–22. doi: 10.1016/S0140-6736(13)60688-1
207. Baller EB, Hogan CS, Fusunyan MA, Ivkovic A, Luccarelli JW, Madva E, et al. Neurocovid: pharmacological recommendations for delirium associated with COVID-19. Psychosomatics. (2020) 61:585–96. doi: 10.1016/j.psym.2020.05.013
208. Han D, Wang C, Feng X, Wu J. Delirium during recovery in patients with severe COVID-19: two case reports. Front Med. (2020) 7:573791. doi: 10.3389/fmed.2020.573791
209. Kales HC, Lyketsos CG, Miller EM, Ballard C. Management of behavioral and psychological symptoms in people with Alzheimer's disease: an international delphi consensus. Int psychogeriatrics. (2019) 31:83–90. doi: 10.1017/S1041610218000534
210. Magierski R, Sobow T, Schwertner E, Religa D. Pharmacotherapy of behavioral and psychological symptoms of dementia: state of the art and future progress. Front Pharmacol. (2020) 11:1168. doi: 10.3389/fphar.2020.01168
211. Garay RP, Citrome L, Grossberg GT, Cavero I, Llorca PM. Investigational drugs for treating agitation in persons with dementia. Expert Opin Investig Drugs. (2016) 25:973–83. doi: 10.1080/13543784.2016.1193155
212. Magierski R, Sobow T. Serotonergic drugs for the treatment of neuropsychiatric symptoms in dementia. Expert Rev Neurother. (2016) 16:375–87. doi: 10.1586/14737175.2016.1155453
213. Kumar B, Kuhad A, Kuhad A. Lumateperone: a new treatment approach for neuropsychiatric disorders. Drugs Today. (2018) 54:713–9. doi: 10.1358/dot.2018.54.12.2899443
214. Blair HA. Lumateperone: first approval. Drugs. (2020) 80:417–23. doi: 10.1007/s40265-020-01271-6
215. Stummer L, Markovic M, Maroney M. Brexpiprazole in the treatment of schizophrenia and agitation in Alzheimer's disease. Neurodegener Dis Manag. (2020) 10:205–17. doi: 10.2217/nmt-2020-0013
216. Üçok A, Gaebel W. Side effects of atypical antipsychotics: a brief overview. World Psychiatry. (2008) 7:58–62. doi: 10.1002/j.2051-5545.2008.tb00154.x
217. Marcinkowska M, Sniecikowska J, Fajkis N, Paśko P, Franczyk W, Kołaczkowski M. Management of dementia-related psychosis, agitation and aggression: a review of the pharmacology and clinical effects of potential drug candidates. CNS Drugs. (2020) 34:243–68. doi: 10.1007/s40263-020-00707-7
218. Sukonick DL, Pollock BG, Sweet RA, Mulsant BH, Rosen J, Klunk WE, et al. The 5-HTTPR*S/*L polymorphism and aggressive behavior in Alzheimer disease. Arch Neurol. (2001) 58:1425–8. doi: 10.1001/archneur.58.9.1425
219. Sweet RA, Pollock BG, Sukonick DL, Mulsant BH, Rosen J, Klunk WE, et al. The 5-HTTPR polymorphism confers liability to a combined phenotype of psychotic and aggressive behavior in Alzheimer disease. Int Psychogeriatr. (2001) 13:401–9. doi: 10.1017/s1041610201007827
220. Taylor CP, Traynelis SF, Siffert J, Pope LE, Matsumoto RR. Pharmacology of dextromethorphan: relevance to dextromethorphan/quinidine (Nuedexta®) clinical use. Pharmacol Ther. (2016) 164:170–82. doi: 10.1016/j.pharmthera.2016.04.010
221. Garay RP, Grossberg GT. AVP-786 for the treatment of agitation in dementia of the Alzheimer's type. Expert Opin Investig Drugs. (2017) 26:121–32. doi: 10.1080/13543784.2017.1267726
222. Charernboon T, Lerthattasilp T, Supasitthumrong T. Effectiveness of cannabinoids for treatment of dementia: a systematic review of randomized controlled trials. Clin Gerontol. (2020) 44:16–24. doi: 10.1080/07317115.2020.1742832
Keywords: agitation, dementia, hyperkinetic delirium, Alzheimer's Disease, Dementia with Lewy Bodies, Frontotemporal Dementia, Vascular Dementia, COVID-19
Citation: Carrarini C, Russo M, Dono F, Barbone F, Rispoli MG, Ferri L, Di Pietro M, Digiovanni A, Ajdinaj P, Speranza R, Granzotto A, Frazzini V, Thomas A, Pilotto A, Padovani A, Onofrj M, Sensi SL and Bonanni L (2021) Agitation and Dementia: Prevention and Treatment Strategies in Acute and Chronic Conditions. Front. Neurol. 12:644317. doi: 10.3389/fneur.2021.644317
Received: 20 December 2020; Accepted: 12 March 2021;
Published: 16 April 2021.
Edited by:
David Gordon Lichter, University at Buffalo, United StatesReviewed by:
Antonina Luca, University of Catania, ItalyJudith Aharon Peretz, Rambam Health Care Campus, Israel
Copyright © 2021 Carrarini, Russo, Dono, Barbone, Rispoli, Ferri, Di Pietro, Digiovanni, Ajdinaj, Speranza, Granzotto, Frazzini, Thomas, Pilotto, Padovani, Onofrj, Sensi and Bonanni. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Laura Bonanni, bC5ib25hbm5pQHVuaWNoLml0
 Claudia Carrarini
Claudia Carrarini Mirella Russo
Mirella Russo Fedele Dono1
Fedele Dono1 Martina Di Pietro
Martina Di Pietro Paola Ajdinaj
Paola Ajdinaj Alberto Granzotto
Alberto Granzotto Astrid Thomas
Astrid Thomas Alessandro Padovani
Alessandro Padovani Marco Onofrj
Marco Onofrj Stefano L. Sensi
Stefano L. Sensi Laura Bonanni
Laura Bonanni