
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Mol. Biosci. , 24 May 2021
Sec. Molecular Diagnostics and Therapeutics
Volume 8 - 2021 | https://doi.org/10.3389/fmolb.2021.559800
Background: Cancer is one of the deadliest diseases at present. Although effective screening and treatment can save lives to a certain extent, our knowledge of the disease is far from sufficient. KIF18B is a member of the kinesin-8 superfamily and plays a conserved regulatory role in the cell cycle. KIF18B reportedly functions as an oncogene in some human cancers, but the correlations between KIF18B and prognosis and immune-infiltrates in different cancers remain unclear.
Methods: Data were collected from the TCGA, GTEx, CCLE, TIMER, and GSEA databases. The expression difference, survival, pathological stage, DNA methylation, tumor mutation burden (TMB), microsatellite instability (MSI), mismatch repairs (MMRs), tumor microenvironment (TME), immune cell infiltration, and gene co-expression of KIF18B were analyzed using the R language software.
Results: KIF18B was widely upregulated in cancers, compared with normal tissues, and high KIF18B expression was associated with unfavorable prognoses. TMB, MSI, MMRs, and DNA methylation correlated with KIF18B dysregulation in cancers. KIF18B correlated closely with tumor immunity and interacted with different immune cells and genes in different cancer types.
Conclusion: KIF18B could be used as a prognostic biomarker for determining prognosis and immune infiltration in pan-cancer.
Tumors have gradually become the leading cause of death in the world (Bray et al., 2018). With the continuous and relentless development of medical technology, tumor screening and treatment are constantly and significantly improving. However, due to the concealment and complexity of the occurrence and development of cancer, almost no malignant tumors can be completely cured. Although drugs targeting specific genes are considerably efficient in certain tumors, no satisfactory outcome has been reached yet. In recent years, the roles of the tumor microenvironment (TME) and host immunity have become increasingly important, and the emergence of immune checkpoint inhibitors (ICIs) has brought promising perspectives to more patients (Topalian et al., 2016). With this increase in clinical applications, more effective targets must be found.
Kinesin superfamily proteins (KIFs) are a group of proteins with highly conserved motor domains that can polymerize and dissociate from microtubules and use the energy released by ATP hydrolysis to promote the directional movement of KIFs (Miki et al., 2005). The motor ability of KIFs plays a vital role in mitosis, vesicle trafficking, and the maintenance of cell polarity, among others (Hirokawa et al., 1998; Sharp et al., 2000; Hirokawa and Takemura, 2005). KIF18B, a member of the kinesin-8 proteins, is involved in the separation of chromosomes and spindle localization during mitosis (Du et al., 2010; McHugh et al., 2018). In recent years, studies have shown that KIF18B participates in the growth and development of a variety of cancers (Wu et al., 2018; Zhong et al., 2019; Li et al., 2020; Yang et al., 2020). However, to date, the expression pattern, prognostic significance, and biological function of KIF18B in cancer have not been elucidated fully.
In this study, we conducted a comprehensive analysis of KIF18B at the transcription level, tumor mutation burden (TMB) level, DNA methylation level, microsatellite instability (MSI) level, mismatch repairs (MMRs) level, TME level, clinical prognosis level, and more.
TCGA is a large-scale sequencing-based genomic analysis technology for the understanding of the molecular mechanisms of cancer (https://cancergenome.nih.gov) (AuthorAnonymous, 2018). We downloaded the expression profile data, TMB data, MSI data, and clinical data of 33 tumors from TCGA.
The GTEx program has collected and studied more than 7,000 autopsy samples from 449 human donors who were healthy during their lifetime, covering 44 organizations (http://commonfund.nih.gov/GTEx/) (Battle et al., 2017). GTEx observed gene expression patterns of almost all transcribed genes. Given that there are fewer normal samples in TCGA, we integrated normal tissue data from GTEx with tumor tissue data from TCGA to analyze the expression differences of KIF18B.
We used CCLE, an online database that provides an analysis and visualization of more than 1,100 cell lines (https://portals.broadinstitute.org/ccle) (Ghandi et al., 2019), to assess the expression level of KIF18B in the tumor cell lines of 21 tissues.
TIMER, a web server used for the comprehensive analysis of tumor-infiltrating immune cells (https://cistrome.shinyapps.io/timer/) (Li et al., 2017), was used to analyze the correlation of KIF18B expression with the abundance of immune infiltrates.
GSEA is based on the existing knowledge of gene location, function, and biological significance and is used to build a database containing multiple functional gene sets (https://www.gsea-msigdb.org/gsea/index.jsp) (Subramanian et al., 2005). Samples were separated into a high group and a low group according to the median expression of KIF18B, and the GSEA software was used to enrich the gene ontology (GO) and Kyoto encyclopedia of genes and genomes (KEGG) pathway.
The Perl script was used to organize and normalize data matrixes, classifying them into cancerous tissues and adjacent tissues and extracting the expression of KIF18B. The analyses of the expression difference, survival, pathological stage, DNA methylation, TMB, MSI, MMRs, TME, immune cell infiltration, and gene co-expression of KIF18B were performed using the R language (Version 3.6.3) (https://www.r-project.org/) for statistical analysis, including the “ggpubr”, “limma”, “survival”, “survminer”, “fmsb”, “ggExtra”, “ggplot2”, “e1071”, “org.Hs.eg.db”, “clusterProfiler”, “enrichplot”, and “forestplot” projects.
We extracted the mRNA levels of KIF18B from 33 cancer types in TCGA and plotted a box diagram of the expression of KIF18B in cancerous and adjacent tissues. As shown in Figure 1A, the expression of KIF18B mRNA in most cancers was different from that in normal tissues. KIF18B mRNA levels increased significantly in bladder urothelial carcinoma (BLCA), breast invasive carcinoma (BRCA), cervical squamous cell carcinoma and endocervical adenocarcinoma (CESC), cholangiocarcinoma (CHOL), colon adenocarcinoma (COAD), esophageal carcinoma (ESCA), glioblastoma multiforme (GBM), head and neck squamous cell carcinoma (HNSC), kidney chromophobe (KICH), kidney renal clear cell carcinoma (KIRC), kidney renal papillary cell carcinoma (KIRP), liver hepatocellular carcinoma (LIHC), lung adenocarcinoma (LUAD), lung squamous cell carcinoma (LUSC), pheochromocytoma and paraganglioma (PCPG), prostate adenocarcinoma (PRAD), rectum adenocarcinoma (READ), sarcoma (SARC), stomach adenocarcinoma (STAD), thyroid carcinoma (THCA), and uterine corpus endometrial carcinoma (UCEC) and increased inconsiderably in pancreatic adenocarcinoma (PAAD). But decrease did not change substantially in thymoma (THYM) and skin cutaneous melanoma (SKCM). Because the transcription levels of the corresponding adjacent tissues in adrenocortical carcinoma (ACC), lymphoid neoplasm diffuse large B-cell lymphoma (DLBC), acute myeloid leukemia (LAML), brain lower grade glioma (LGG), mesothelioma (MESO), ovarian serous cystadenocarcinoma (OV), testicular germ cell tumors (TGCT), uterine carcinosarcoma (UCS), and uveal melanoma (UVM) were unavailable, we integrated normal tissue data into GTEx data. As shown in Figure 1B, KIF18B mRNA levels were also higher in ACC, LGG, OV, TGCT, and UCS. Additionally, KIF18B was generally higher in the tumor cell lines of 21 tissues (Figure 1C).
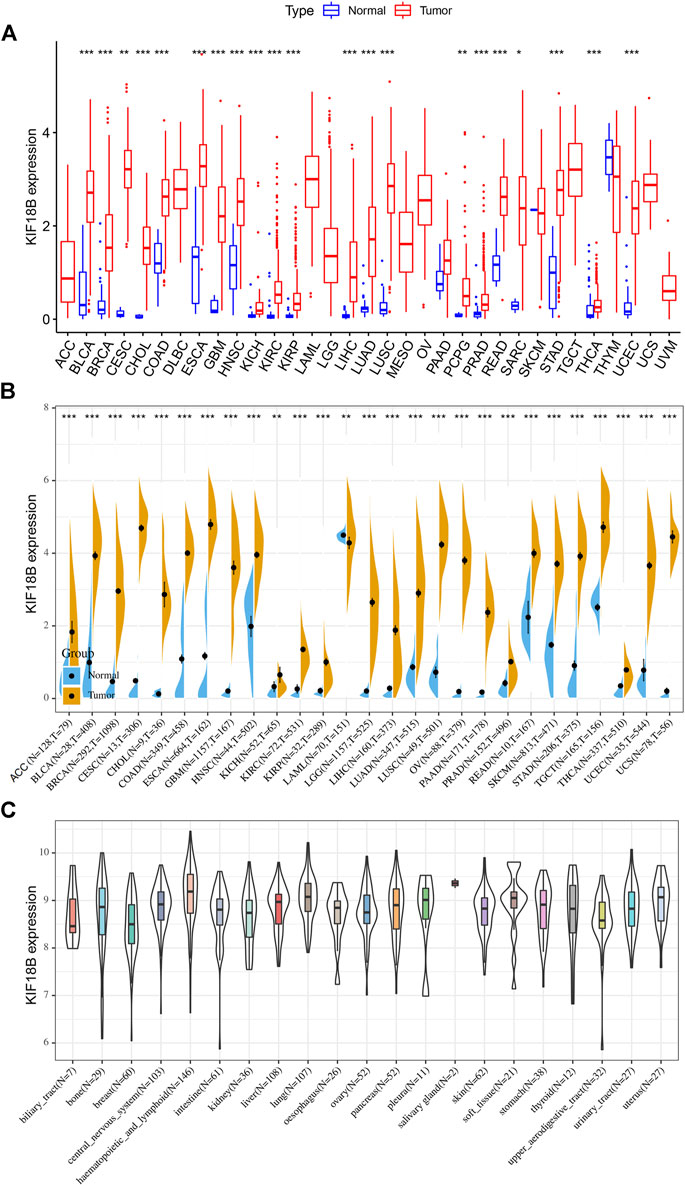
FIGURE 1. The transcription levels of KIF18B in various cancers. (A) The evaluation of KIF18B mRNA in 33 types of cancers from TCGA database. (B) The evaluation of KIF18B mRNA in 27 types of cancers by integrating normal tissue data from GTEx with tumor tissue data from TCGA. (C) The evaluation of KIF18B mRNA in the cancer cell lines of 21 tissues using the CCLE database. * indicates p < 0.05, ** indicates p < 0.01, *** indicates p < 0.001.
We integrated the KIF18B mRNA level with the overall survival (OS), disease-specific survival (DSS), disease-free survival (DFI), and progression-free interval (PFI) of the 33 cancer types and used the Kaplan-Meier (KM) survival curve and Cox proportional hazards models to evaluate the effect of KIF18B on cancer prognosis. Cox regression analyses were displayed in forest charts. The correlations of KIF18B with OS, DSS, DFI, and PFI in different cancers were visually observed. Highly expressed KIF18B correlated negatively with OS in ACC (p < 0.001), KICH (p < 0.001), KIRC (p < 0.001), KIRP (p < 0.001), LGG (p = 0.002), LIHC (p < 0.001), LUAD (p = 0.014), MESO (p < 0.001), PAAD (p < 0.001), PRAD (p = 0.011), SARC (p = 0.018), SKCM (p = 0.042), STAD (p = 0.044), UCEC (p = 0.017), and UVM (p = 0.006) and positively with OS in THYM (p = 0.012) and STAD (p = 0.044) (Figure 2A), negatively with DSS in ACC (p < 0.001), KICH (p < 0.001), KIRC (p < 0.001), KIRP (p < 0.001), LGG (p = 0.002), LIHC (p = 0.001), LUAD (p = 0.033), MESO (p < 0.001), PAAD (p = 0.002), PRAD (p < 0.001), SARC (p = 0.035), SKCM (p = 0.027), UCEC (p = 0.026), and UVM (p = 0.004) (Figure 2B), negatively with DFI in KIRP (p < 0.001), LIHC (p = 0.007), LUAD (p = 0.025), PAAD (p = 0.046), PRAD (p < 0.001), SARC (p = 0.004), and THCA (p < 0.001) (Figure 2C), and negatively with PFI in ACC (p < 0.001), KICH (p < 0.001), KIRC (p < 0.001), KIRP (p < 0.001), LGG (p < 0.001), LIHC (p < 0.001), LUAD (p = 0.042), MESO (p = 0.001), PAAD (p = 0.001), PRAD (p < 0.001), SARC (p = 0.003), SKCM (p = 0.038), THCA (p < 0.001), and UVM (p < 0.001) (Figure 2D).
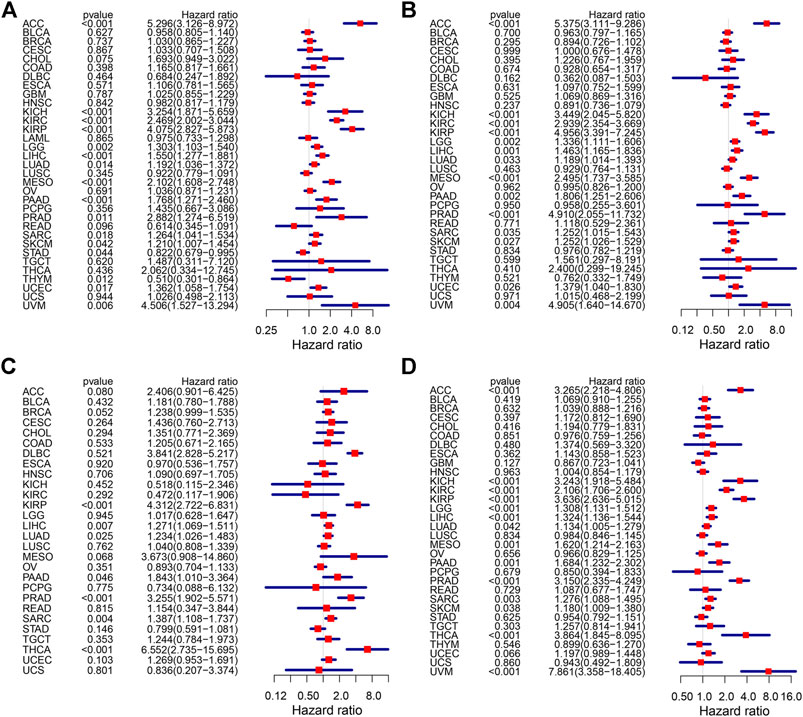
FIGURE 2. The effect of KIF18B on the prognosis of various cancers using Cox proportional hazards models. (A) The effect of KIF18B on OS in 33 types of cancers. (B) The effect of KIF18B on DSS in 33 types of cancers. (C) The effect of KIF18B on DFS in 33 types of cancers. (D) The effect of KIF18B on PFI in 33 types of cancers.
For the visualization of the impact of KIF18B on prognosis using the KM curve, and the filter condition was p < 0.05. As shown in Figure 3A, higher levels of KIF18B mRNA indicated worse OS in ACC (p < 0.001), CHOL (p = 0.015), KIRC (p < 0.001), KIRP (p = 0.001), LIHC (p = 0.001), LUAD (p = 0.001), MESO (p < 0.001), PAAD (p = 0.005), SARC (p = 0.014), SKCM (p = 0.017), and UCEC (p = 0.046), while the opposite result was observed in THYM (p = 0.017).
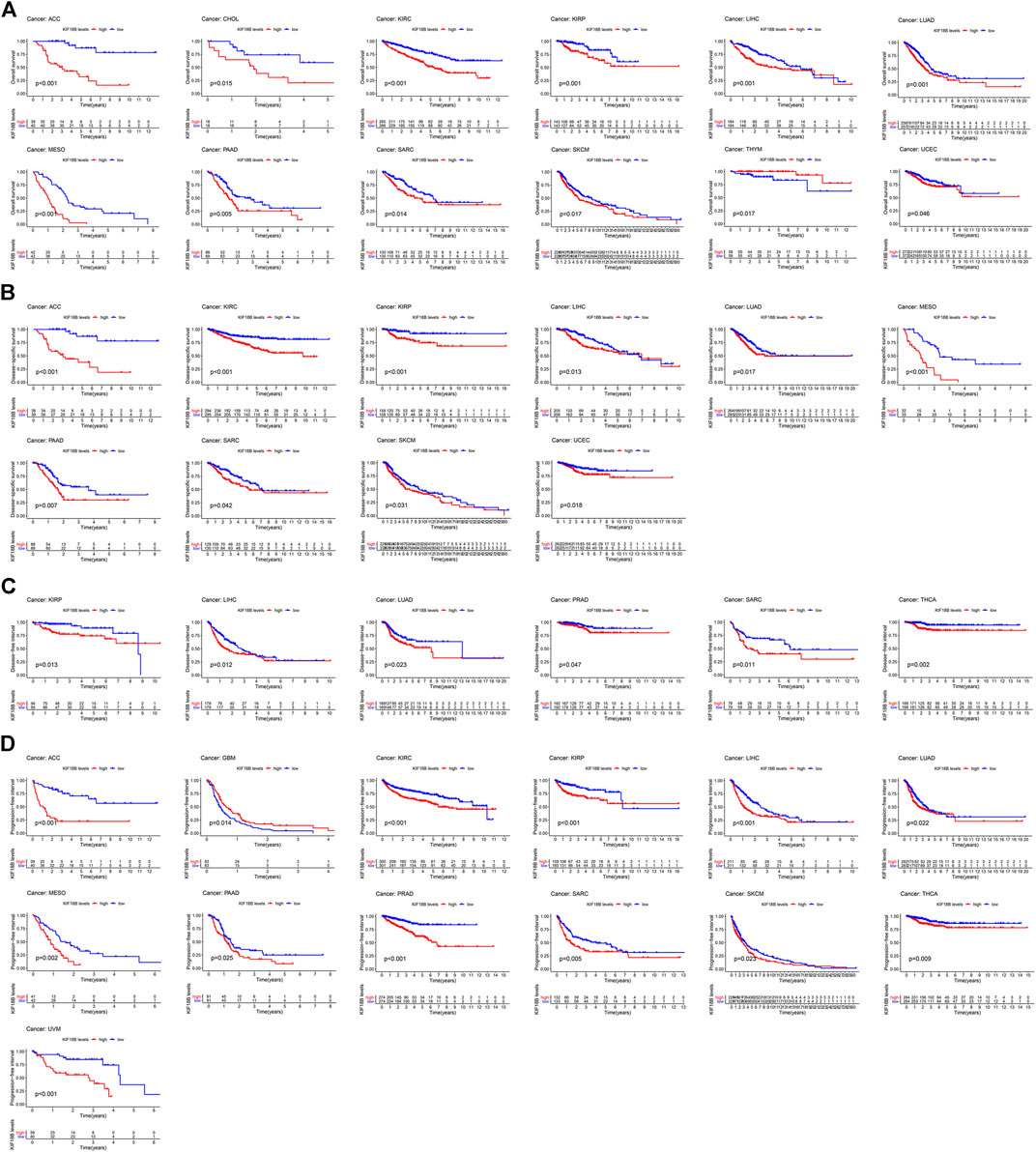
FIGURE 3. The survival curve of KIF18B in various cancers using Kaplan-Meier methods and the log-rank test. Values of p < 0.05 were considered and displayed. (A) The survival curve of KIF18B for OS in 12 types of cancers. (B) The survival curve of KIF18B for DSS in 10 types of cancers. (C) The survival curve of KIF18B for DFS in 6 types of cancers. (D) The survival curve of KIF18B for PFI in 13 types of cancers.
Higher levels of KIF18B mRNA were also linked with worse DSS in ACC (p < 0.001), KIRC (p < 0.001), KIRP (p < 0.001), LIHC (p = 0.013), LUAD (p = 0.017), MESO (p < 0.001), PAAD (p = 0.007), SARC (p = 0.042), SKCM (p = 0.031), and UCEC (p = 0.018) (Figure 3B), worse DFI in KIRP (p = 0.013), LIHC (p = 0.012), LUAD (p = 0.023), PRAD (p = 0.047), SARC (p = 0.011), and THCA (p = 0.002) (Figure 3C), and worse PFI in ACC (p < 0.001), KIRC (p < 0.001), KIRP (p < 0.001), LIHC (p < 0.001), LUAD (p = 0.022), MESO (p = 0.002), PAAD (p = 0.025), PRAD (p < 0.001), SARC (p = 0.005), SKCM (p = 0.023), THCA (p = 0.009), and UVM (p < 0.001), while better PFI in GBM (p = 0.014) (Figure 3D).
To assess whether KIF18B mRNA was expressed at different levels at various cancer stages, we extracted pathological stages, including stages I, II, III, and IV, from 33 tumors and integrated them with KIF18B mRNA levels. Figure 4 shows that there were differences in KIF18B expression between stages I and II in BRCA, ESCA, LUAD, LUSC, SKCM, TGCT, and THCA, between stages I and III in ACC, KIRC, KIRP, LIHC, SKCM, and TGCT, between stages I and IV in ACC, ESCA, KICH, KIRC, KIRP, and SKCM, between stages II and III in ACC, KIRC, and STAD, between stages II and IV in ACC, KICH, KIRC, and THCA, and between stages III and IV in ACC, ESCA, and KICH. However, there was no difference in KIF18B expression between different pathological stages in BLCA, CHOL, COAD, HNSC, MESO, PAAD, READ, and UVM. In addition, no pathological staging records were detected in CESC, DLBC, GBM, LAML, LGG, OV, PCPG, PRAD, SARC, THYM, UCEC, and UCS.
TMB refers to the number of somatic mutations that occur on the average 1 Mb base in the coding region of the tumor cell genome (Campbell et al., 2017). MSI represents the random changes in the length of microsatellites in tumor cells due to the insertion or deletion of repeating units, compared to normal cells, and the emergence of new microsatellite alleles (Cortes-Ciriano et al., 2017). We calculated the TMB and MSI of each sample in all tumors and then analyzed the relationship between KIF18B expression and TMB and MSI in different tumors using the Spearman rank correlation coefficient and displayed it as a radar chart.
As shown in Figure 5A, the high expressions of KIF18B correlated significantly positively with TMB in ACC, BLCA, BRCA, CESC, COAD, HNSC, KICH, KIRC, LGG, LUAD, LUSC, MESO, OV, PAAD, PRAD, READ, SARC, SKCM, STAD, TGCT, and UCEC but markedly negatively in THYM (Supplementary Table S1). Figure 5B shows that the high expressions of KIF18B correlated significantly positively with MSI in ACC, BLCA, BRCA, CESC, ESCA, GBM, LIHC, LUAD, LUSC, PRAD, SARC, STAD, and UCEC but considerably negatively in DLBC, LAML, and READ (Supplementary Table S2).

FIGURE 5. The correlations between KIF18B expression and tumor mutation burden (TMB), microsatellite instability (MSI), DNA methylation, and mismatch repairs (MMRs) in various cancers. (A) The correlations between KIF18B expression and TMB in 33 types of cancers. (B) The correlation between KIF18B and MSI in 33 types of cancers. (C) The correlations between KIF18B and four methyltransferases (DNMT1: red, DNMT2: blue, DNMT3A: green, and DNMT3B: purple) in 33 types of cancers. (D) The correlation between KIF18B and five MMRs related genes in 33 types of cancers.
DNA methylation is a form of chemical modification of DNA that can change genetic performance without changing the DNA sequence (Perri et al., 2017). We evaluated the relationship between four methyltransferases, including DNMT1: red, DNMT2: blue, DNMT3A: green, and DNMT3B: purple, and KIF18B expression. As shown in Figure 5C, KIF18B expressions associated positively with all four methyltransferases in 20 types of cancer but did not correlate with any of the four methyltransferases in PCPG or with DNMT2 in PRAD, PAAD, LUAD, LAML, KIRC, COAD, CHOL, UVM, UCS, UCEC, and THCA, DNMT3A in PAAD, LAML, DLBC, CHOL, and UCS, or DNMT3B in LAML and CHOL. MMRs are intracellular mismatch repair mechanisms. The loss of key gene functions in these mechanisms can lead to DNA replication errors that cannot be repaired, eventually causing higher somatic mutations (Kunkel and Erie, 2005). We evaluated the relationship between five MMRs-related genes, including MLH1, MSH2, MSH6, PMS2, and EPCAM, and KIF18B expression. As shown in Figure 5D, KIF18B expressions correlated significantly positively with MLH1 in 23 types of cancer, with MSH2 in 30 types of cancer, with MSH6 in 30 types of cancer, with PMS2 in 21 types of cancer, and with EPCAM in 14 types of cancer. Additionally, KIF18B expressions correlated significantly negatively with EPCAM in LGG, THCA, and THYM.
TME is the local biological environment where tumors are located. Abnormal alterations to this environment can induce changes, such as promoting tumor cell proliferation, invasion, metastasis, and inhibiting apoptosis, to the biological behavior of tumor cells (Pitt et al., 2016; Locy et al., 2018). We obtained the stromal and immune scores of each sample in all tumors and assessed the relationship between KIF18B expression and both scores in TME using the Spearman rank correlation coefficient (Supplementary Table S3). We visualized the results at p < 0.001. As shown in Figure 6, the high expressions of KIF18B correlated significantly negatively with stromal cells in BLCA, BRCA, CESC, COAD, GBM, HNSC, LIHC, LUAD, LUSC, PAAD, SARC, SKCM, STAD, THYM, and UCEC, and immune cells in CESC, COAD, ESCA, GBM, LUSC, SARC, STAD, TGCT, and UCEC but the correlations between high KIF18B levels and stromal and immune scores in KIRC and THCA were positive.
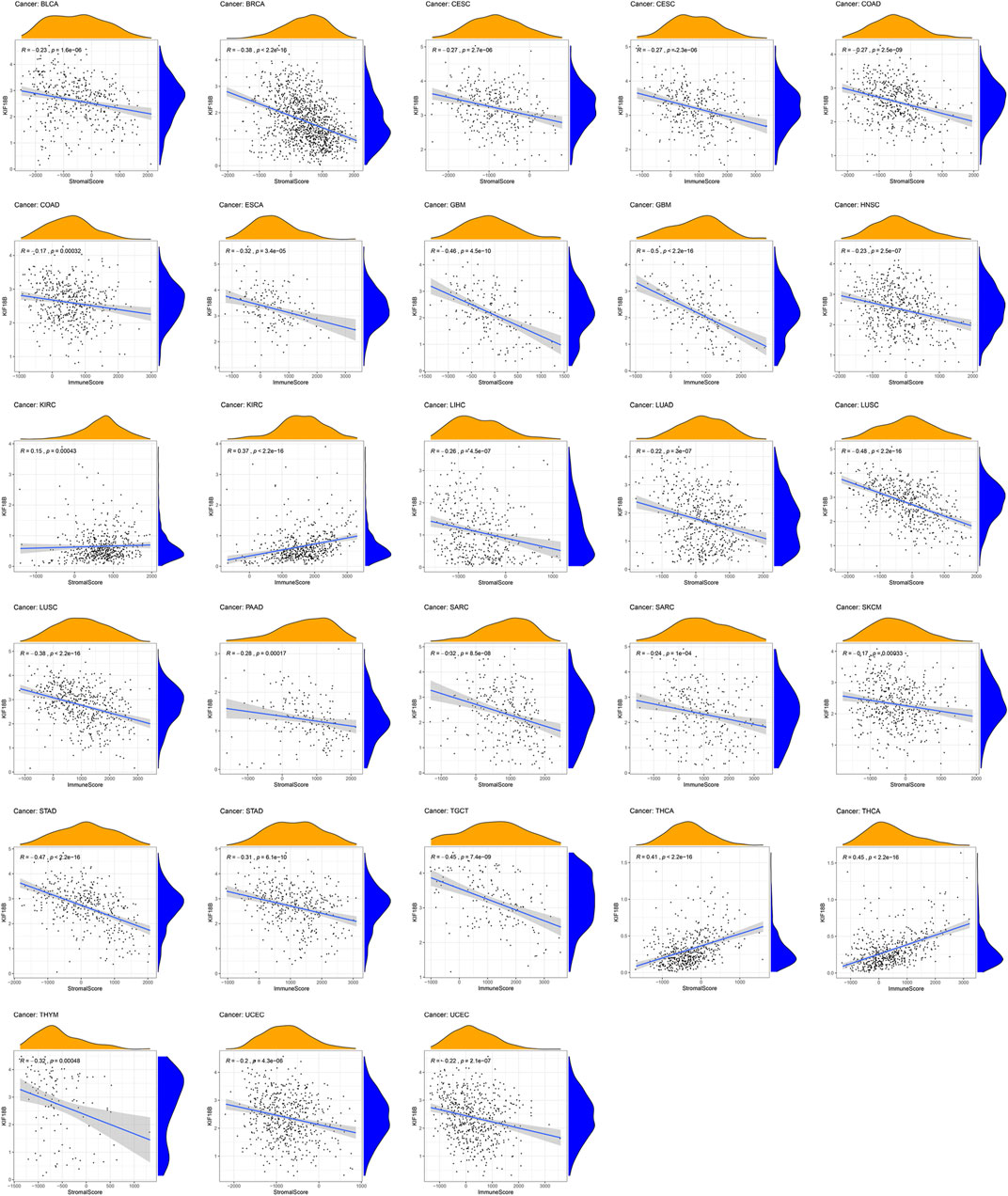
FIGURE 6. The correlations between KIF18B expression and the stromal and immune scores of tumor microenvironments in various cancers. Values of p < 0.001 were considered and displayed.
We obtained the content of 22 specific immune cells in each sample of all tumors and observed the relationship between KIF18B expression and these contents using the Spearman rank correlation coefficient (Supplementary Table S4). We visualized the results at p < 0.001. As shown in Supplementary Figure S1, the high expressions of KIF18B correlated significantly negatively with T cells CD4 memory resting in UCEC (R = −0.18), TGCT (R = −0.27), LUSC (R = −0.22), LUAD (R = −0.29), KIRC (R = −0.19), GBM (R = −0.3), COAD (R = −0.18), and BRCA (R = −0.19), but significantly positively with T cells CD4 memory activated in UCEC (R = 0.26), THCA (R = 0.37), STAD (R = 0.21), LUAD (R = 0.36), LIHC (R = 0.21), KIRP (R = 0.23), KIRC (R = 0.2), COAD (R = 0.19), and BRCA (R = 0.26). Highly expressed KIF18B also correlated significantly positively with T cells follicular helper in UCEC (R = 0.25), THCA (R = 0.34), STAD (R = 0.35), OV (R = 0.2), LUSC (R = 0.2), LUAD (R = 0.2), LIHC (R = 0.37), KIRP (R = 0.25), KIRC (R = 0.21), HNSC (R = 0.15), BRCA (R = 0.18), and BLCA (R = 0.22) and T cells regulatory (Tregs) in PRAD and KIRC but significantly negatively with Tregs in UCEC (R = −0.21), SKCM (R = −0.19), DLBC (R = −0.51), and BLCA (R = −0.26). Furthermore, the high expressions of KIF18B correlated significantly negatively with macrophages M0 in THYM (R = −0.55) and KIRP (R = −0.23) but substantially positively in STAD (R = 0.35), SARC (R = 0.26), PAAD (R = 0.28), LUSC (R = 0.18), LUAD (R = 0.4), LIHC (R = 0.22), BRCA (R = 0.3), and ACC (R = 0.6), significantly negatively with macrophages M1 in THYM (R = −0.44) but markedly positively in UCEC (R = 0.32), THCA (R = 0.34), STAD (R = 0.22), LUSC (R = 0.2), LUAD (R = 0.42), KIRP (R = 0.25), KIRC (R = 0.17), BRCA (R = 0.26), and BLCA (R = 0.2), significantly negatively with macrophages M2 in THYM (R = −0.63), THCA (R = −0.32), STAD (R = −0.17), LIHC (R = −0.33), KIRP (R = −0.3), and KIRC (R = −0.21), but considerably positively in TGCT (R = 0.45) and GBM (R = 0.33), significantly negatively with dendritic cells resting in STAD (R = −0.24), LUSC (R = −0.23), LUAD (R = −0.37), HNSC (R = −0.31), COAD (R = −0.2), and BRCA (R = −0.13), but remarkably positively in THYM (R = 0.65) and LIHC (R = 0.19), and significantly negatively with mast cells resting in THYM (R = −0.56), STAD (R = −0.3), LUAD (R = −0.43), KIRC (R = −0.27), HNSC (R = −0.16), BRCA (R = −0.34), BLCA (R = −0.23), and ACC (R = −0.5).
To further explore whether KIF18B could be used as a potential target for immune checkpoints in cancer, we examined the relationship between KIF18B expression and some known immune-related factors, displaying the outcome as a heat map. As shown in Figure 7, KIF18B correlated negatively with 9 immune-related factors and positively with 27 immune-related factors in BRCA, negatively with 21 immune-related factors and positively with 9 immune-related factors in COAD, negatively with 24 immune-related factors and positively with 6 immune-related factors in GBM, positively with 29 immune-related factors and negatively with 1 immune-related factor in HNSC, positively with 33 immune-related factors and negatively with 4 immune-related factors in KIRC, positively with 39 immune-related factors and negatively with 1 immune-related factor in LIHC, positively with 31 immune-related factors and negatively with 5 immune-related factors in PRAD, positively with 8 immune-related factors and negatively with 31 immune-related factors in TGCT, positively with 42 immune-related factors in THCA, and positively with 14 immune-related factors and negatively with 21 immune-related factors in THCA.
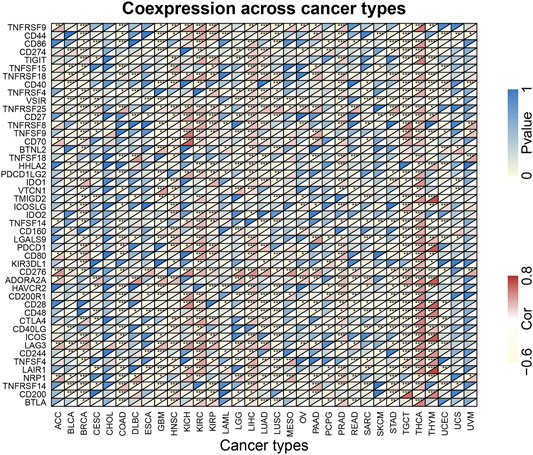
FIGURE 7. The correlations between KIF18B expression and immune-related factors in various cancers. * indicates p < 0.05, ** indicates p < 0.01, *** indicates p < 0.001.
GSEA was used for the GO and KEGG analyses of KIF18B positively co-expressed genes in pan-cancers. GO enrichment analysis predicted the functional roles of target host genes based on three aspects: biological processes, cellular components, and molecular functions. Results at p < 0.05 were screened out, and the enrichment results higher than five were visualized. GO analysis suggested that these genes were mainly concentrated in the chromosome, spindle, and microtubule and that they were primarily involved in cell cycle-related activities and regulation (Supplementary Figure S2; Supplementary Tables S5–S37). Per the KEGG analysis, KIF18B in pan-cancer tissues participated largely in antigen processing and presentation, cell cycle, cytosolic DNA sensing pathway, regulation of autophagy, and toll-like receptor signaling pathway (Figure 8; Supplementary Tables S38–S70).
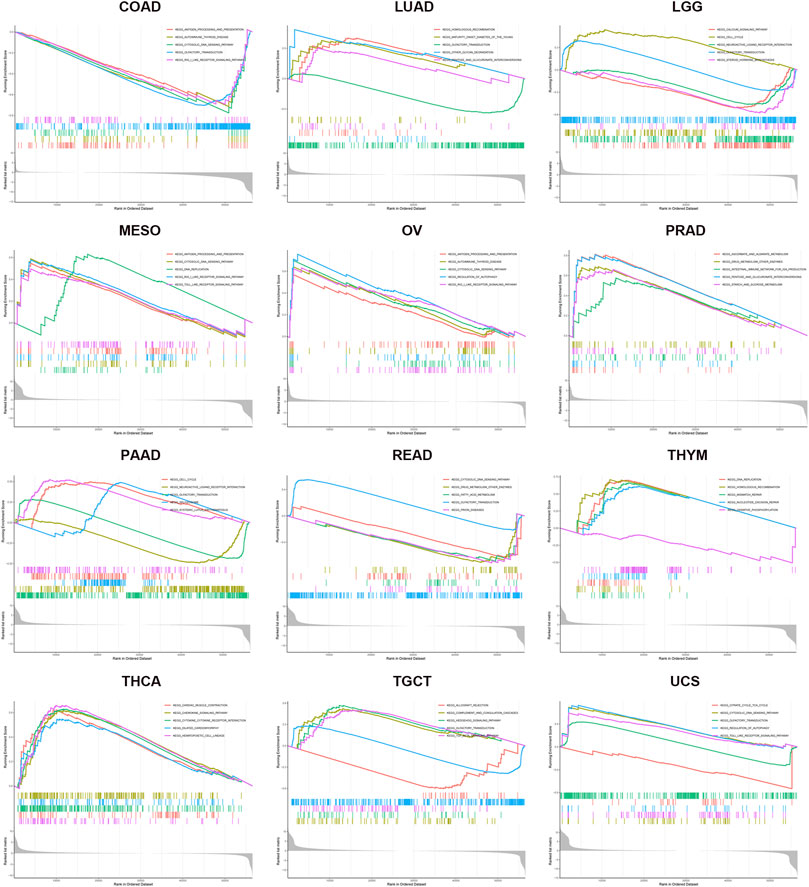
FIGURE 8. KEGG enrichment analysis of KIF18B in various cancers. Values of p < 0.05 and results higher than 5 were considered and displayed.
Kinesin is a motor molecule that exists in all eukaryotes. Most newly synthesized proteins in cells move through molecular motors along directional cytoskeletal fibers to their appropriate destinations. Substances are precisely transported and unloaded to the corresponding parts to perform specific functions, which is essential to maintain the morphology and function of cells, especially during cell division (Mazumdar and Misteli, 2005; Wu et al., 2006). Kinesin is precise because it plays a vital role in the regulation of cytokinesis, spindle formation, and chromosome assembly and arrangement; so, its abnormal expression can easily cause abnormal cell replication and differentiation, which can lead to cancer. A variety of KIFs are abnormally expressed in various tumor cells, and their role in promoting cancer has also been studied to varying degrees (Wang et al., 2017; Hu et al., 2019; Jungwirth et al., 2019; Li et al., 2019; Xu et al., 2019). Since tumors are gene-related diseases with abnormal cell proliferation, targeting kinesin therapy could be a new strategy for tumor treatment. KIF18B is located on chromosome 17q21.31 and consists of 17 exons. The protein is a novel dynamics regulatory protein that regulates astral microtubule length and life in space conformation (Walczak et al., 2016). The underlying molecular mechanisms of KIF18B dysregulation in tumors have not been elucidated fully. Considering the potential prognostic value and biological function of KIF18B, it is essential to explore the fundamental mechanism of its dysregulation in pan-cancer.
In this study, we first revealed that KIF18B was upregulated at mRNA levels in ACC, BLCA, BRCA, CESC, CHOL, COAD, ESCA, GBM, HNSC, KICH, KIRC, KIRP, LIHC, LUAD, LUSC, LGG, PCPG, PRAD, READ, SARC, STAD, THCA, UCEC, OV, TGCT, and UCS cancer tissues, compared with corresponding normal tissues, suggesting that KIF18B might act as an oncogene in pan-cancers. Based on survival curves, including OS, DFI, DSS, and PFI, KIF18B mRNA expression demonstrated reliable diagnostic value, indicating that KIF18B is a potentially promising biomarker for pan-cancer diagnosis. We further explored the clinical significance of KIF18B mRNA expression in pan-cancers and revealed that KIF18B mRNA expressions were higher in advanced pathological stages (III and IV) in ACC, KIRC, KIRP, KICH, LIHC, SKCM, TGCT, ESCA, and STAD, which pointed to KIF18B playing a role in the carcinogenesis and progression of pan-cancer.
TMB and MSI are very common in cancer and may have a profound impact on tumor phenotype and patient survival (Hause et al., 2016; Samstein et al., 2019). In the current study, the upregulation of KIF18B mRNA correlated significantly positively with TMB and MSI in ACC, BLCA, BRCA, CESC, LUAD, LUSC, PRAD, SARC, STAD, and UCEC, but negatively in THYM. Therefore, we speculate that the high neo-antigen load causing the aberrant KIF18B expression and dysregulation in pan-cancer is a positive note for biomarkers of new antigens. DNA methylation and MMRs are linked to the activation of oncogenes and the inhibition of tumor suppressor genes. In our study, KIF18B expression correlated positively with the methylation and MMRs of most cancers, suggesting that DNA methylation and MMRs may change the expression of KIF18B.
There are several immune cells in the TME, and these can be further divided into immune-killer cells linked to tumor-killing and immunosuppressive cells associated with tumor-escape immune surveillance. Tumor immune escape is an important mechanism that imparts malignant features to tumors. Based on the principle of immune escape, the development of specific tumor immunotherapy has gradually become a research hotspot in the field of cancer. Immunotherapy of tumors, including monoclonal antibody-type ICIs, therapeutic antibodies, cancer vaccines, cell therapy, small molecule inhibitors, among others (O'Donnell et al., 2019; Yang, 2015), is a treatment that restarts and maintains the tumor-immune cycle, restores the body’s anti-tumor immune response, thus, controlling and clearing tumors. The higher the stromal score and immune score, the fewer the cancer cells. Here, our results showed that KIF18B expression correlated significantly negatively with the purity of stromal cells and immune cells in CESC, COAD, GBM, LUSC, SARC, STAD, and UCEC. KIF18B expression also correlated markedly negatively with infiltrating levels of T cells CD4 memory resting in eight types of cancer, macrophages M2 in six types of cancer, dendritic cells resting in six types of cancer, and mast cells resting in eight types of cancer. Moreover, KIF18B expression correlated considerably positively with infiltrating levels of T cells CD4 memory activated in nine types of cancer, T cells follicular helper in 12 types of cancer, macrophages M0 in eight types of cancer, and macrophages M1 in nine types of cancer. The correlation between KIF18B and tumor-associated macrophages (TAMs) revealed that KIF18B possibly plays an immunosuppressive role in cancers by promoting the presentation of tumor antigens rather than secreting immune-inflammatory mediators and that KIF18B could potentially also activate Tregs and induce T cell exhaustion, pointing to a likely regulatory role of KIF18B in tumor immunology. These findings require further validation and experimentation.
To further evaluate whether KIF18B may have an immune-related role in cancer, we collected 47 common immune-related genes. Among them, CD274 (PD-L1) is the hottest immune checkpoint currently studied. As an inhibitory ligand, after binding to its receptor PD-1, PD-L1 negatively regulates T cell proliferation and cytokine secretion at a certain threshold of T cell receptor attack, impairs T cell immune function in the TME, and maintains peripheral tolerance (Parsa et al., 2007; Ansell et al., 2015). According to our findings, the PD-L1 function correlated highly with KIF18B expression in BLCA, BRCA, KIRC, LIHC, LUAD, OV, PAAD, PCPG, STAD, and THCA. Lag-3 acts as a negative regulator of T cell activation, binding major histocompatibility complex class II. Blocking Lag-3 can improve cytotoxic T lymphocyte proliferation and effector function. Lag-3 is also co-expressed with PD-1 in tumor-infiltrating lymphocytes in tumors and synergizing with PD-1 blockade may improve tumor control or regression (Grosso et al., 2007; Woo et al., 2012). In this study, Lag-3 correlated highly with KIF18B expression in ACC, BLCA, BRCA, COAD, GBM, HNSC, KICH, KIRC, KIRP, LGG, LIHC, LUAD, OV, PAAD, PRAD, READ, STAD, THCA, and UCEC. KIF18B, PD-L1, and Lag-3 showed the same expression trends in tumors, suggesting that they may share some pathways that promote tumor aggressiveness. HAVCR2 (Tim-3), a crucial surface protein on exhausted T cells and anti-Tim-3 antibodies can affect the phenotype of myeloid cells in the TME (Huang et al., 2015). Tim-3 can also suppress immune response indirectly by abrogating the acquisition of an M2-like phenotype in TAMs (Dardalhon et al., 2010; Kurtulus et al., 2019). KIF18B expression and Tim-3 are mutually exclusive in CESC, COAD, ESCA, GBM, KIRP, LAML, LUSC, PCPG, SARC, STAD, and TGCT. Therefore, targeting KIF18B may be an option for patients who are ineligible for Tim-3 immunotherapy.
In animal research, Castle et al. performed whole-exome sequencing on B16F10 murine melanoma and inoculated the identified mutant gene-encoding peptide into mice for tumor immunization. They found that the mice immunized with mutant KIF18B peptide had weaker tumor progression and higher survival rates, which provided an experimental basis for our research results (Castle et al., 2012). In short, these correlations could indicate potential mechanisms and suggest that KIF18B plays a vital role in the recruitment and regulation of the immune system in pan-cancer. Furthermore, we explored the basic biological function and molecular mechanisms that underpin the potential clinical value of KIF18B in pan-cancers. In addition to the known “cell cycle” as the most important signaling pathway for KIF18B co-expressed genes, we determined that KIF18B was involved in antigen processing and presentation, cytosolic DNA sensing pathway, regulation of autophagy, toll-like receptor signaling pathway, and many more in various cancers. These findings could help predict the possibility of the use of KIF18B as a new immune target. Of course, our results were based on bioinformatics analysis, meaning that there were no experiments to verify our claims, and transcriptomics levels do not reflect protein expression levels or activity. However, our findings provided new insights into the development and treatment of KIF18B in cancer. We intend to perform in-depth studies to validate our current conclusions in future research.
In summary, KIF18B was upregulated in pan-cancer tissues, compared with corresponding normal tissues. Additionally, the high expression of KIF18B was associated with unfavorable clinicopathological features and independently predicted poor prognosis in pan-cancer. Also, TMB, MSI, MMRs, and DNA methylation might contribute to KIF18B dysregulation in cancers, and KIF18B is closely linked to tumor immunity and may be a potential therapeutic target for immunotherapy.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
Z-fX and S-lY conceived and designed the experiments. M-jQ, Q-sW, and Q-tL carried out the research. M-jQ, L-sZ, and Y-nL contributed the analysis tools and analyzed the data for the study. M-jQ wrote the paper. All authors have read and approved the final manuscript.
This study was supported by the National Key Research and Development Program of China (2018YFC2002000).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmolb.2021.559800/full#supplementary-material
Ansell, S. M., Lesokhin, A. M., Borrello, I., Halwani, A., Scott, E. C., Gutierrez, M., et al. (2015). PD-1 Blockade with Nivolumab in Relapsed or Refractory Hodgkin's Lymphoma. N. Engl. J. Med. 372, 311–319. doi:10.1056/nejmoa1411087
Battle, A., Brown, C. D., Engelhardt, B. E., and Montgomery, S. B.Laboratory, Data Analysis &Coordinating Center (LDACC)Statistical Methods groups (2017). Genetic Effects on Gene Expression across Human Tissues. Nature 550, 204–213. doi:10.1038/nature24277
Bray, F., Ferlay, J., Soerjomataram, I., Siegel, R. L., Torre, L. A., and Jemal, A. (2018). Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: A Cancer J. Clinicians 68, 394–424. doi:10.3322/caac.21492
Campbell, B. B., Light, N., Fabrizio, D., Zatzman, M., Fuligni, F., de Borja, R., et al. (2017). Comprehensive Analysis of Hypermutation in Human Cancer. Cell 171, 1042–1056. doi:10.1016/j.cell.2017.09.048
Castle, J. C., Kreiter, S., Diekmann, J., Löwer, M., van de Roemer, N., de Graaf, J., et al. (2012). Exploiting the Mutanome for Tumor Vaccination. Cancer Res. 72, 1081–1091. doi:10.1158/0008-5472.can-11-3722
Cortes-Ciriano, I., Lee, S., Park, W. Y., Kim, T. M., and Park, P. J. (2017). A Molecular Portrait of Microsatellite Instability across Multiple Cancers. Nat. Commun. 8, 15180. doi:10.1038/ncomms15180
Dardalhon, V., Anderson, A. C., Karman, J., Apetoh, L., Chandwaskar, R., Lee, D. H., et al. (2010). Tim-3/galectin-9 Pathway: Regulation of Th1 Immunity through Promotion of CD11b+Ly-6G+ Myeloid Cells. J. Immunol. 185, 1383–1392. doi:10.4049/jimmunol.0903275
Du, Y., English, C. A., and Ohi, R. (2010). The Kinesin-8 Kif18A Dampens Microtubule Plus-End Dynamics. Curr. Biol. 20, 374–380. doi:10.1016/j.cub.2009.12.049
Ghandi, M., Huang, F. W., Jané-Valbuena, J., Kryukov, G. V., Lo, C. C., McDonald, E. R., et al. (2019). Next-generation Characterization of the Cancer Cell Line Encyclopedia. Nature 569, 503–508. doi:10.1038/s41586-019-1186-3
Grosso, J. F., Kelleher, C. C., Harris, T. J., Maris, C. H., Hipkiss, E. L., De Marzo, A., et al. (2007). LAG-3 Regulates CD8+ T Cell Accumulation and Effector Function in Murine Self- and Tumor-Tolerance Systems. J. Clin. Invest. 117, 3383–3392. doi:10.1172/jci31184
Hause, R. J., Pritchard, C. C., Shendure, J., and Salipante, S. J. (2016). Classification and Characterization of Microsatellite Instability across 18 Cancer Types. Nat. Med. 22, 1342–1350. doi:10.1038/nm.4191
Hirokawa, N., Noda, Y., and Okada, Y. (1998). Kinesin and Dynein Superfamily Proteins in Organelle Transport and Cell Division. Curr. Opin. Cel Biol. 10, 60–73. doi:10.1016/s0955-0674(98)80087-2
Hirokawa, N., and Takemura, R. (2005). Molecular Motors and Mechanisms of Directional Transport in Neurons. Nat. Rev. Neurosci. 6, 201–214. doi:10.1038/nrn1624
Hu, G., Yan, Z., Zhang, C., Cheng, M., Yan, Y., Wang, Y., et al. (2019). FOXM1 Promotes Hepatocellular Carcinoma Progression by Regulating KIF4A Expression. J. Exp. Clin. Cancer Res. 38, 188. doi:10.1186/s13046-019-1202-3
Huang, Y.-H., Zhu, C., Kondo, Y., Anderson, A. C., Gandhi, A., Russell, A., et al. (2015). CEACAM1 Regulates TIM-3-Mediated Tolerance and Exhaustion. Nature 517, 386–390. doi:10.1038/nature13848
Jungwirth, G., Yu, T., Moustafa, M., Rapp, C., Warta, R., Jungk, C., et al. (2019). Identification of KIF11 As a Novel Target in Meningioma. Cancers (Basel), 11, 545. doi:10.3390/cancers11040545
Kunkel, T. A., and Erie, D. A. (2005). DNA Mismatch Repair. Annu. Rev. Biochem. 74, 681–710. doi:10.1146/annurev.biochem.74.082803.133243
Kurtulus, S., Madi, A., Escobar, G., Klapholz, M., Nyman, J., Christian, E., et al. (2019). Checkpoint Blockade Immunotherapy Induces Dynamic Changes in PD-1−CD8+ Tumor-Infiltrating T Cells. Immunity 50, 181–194. doi:10.1016/j.immuni.2018.11.014
Li, B., Liu, B., Zhang, X., Liu, H., and He, L. (2020). KIF18B Promotes the Proliferation of Pancreatic Ductal Adenocarcinoma via Activating the Expression of CDCA8. J. Cell Physiol. 235, 4227–4238. doi:10.1002/jcp.29201
Li, G., Xie, Z. K., Zhu, D. S., Guo, T., Cai, Q. L., and Wang, Y. (2019). KIF20B Promotes the Progression of clear Cell Renal Cell Carcinoma by Stimulating Cell Proliferation. J. Cell Physiol. 234, 16517–16525. doi:10.1002/jcp.28322
Li, T., Fan, J., Wang, B., Traugh, N., Chen, Q., Liu, J. S., et al. (2017). TIMER: A Web Server for Comprehensive Analysis of Tumor-Infiltrating Immune Cells. Cancer Res. 77, e108–e110. doi:10.1158/0008-5472.can-17-0307
Locy, H., de Mey, S., de Mey, W., De Ridder, M., Thielemans, K., and Maenhout, S. K. (2018). Immunomodulation of the Tumor Microenvironment: Turn Foe Into Friend. Front. Immunol. 9, 2909. doi:10.3389/fimmu.2018.02909
Mazumdar, M., and Misteli, T. (2005). Chromokinesins: Multitalented Players in Mitosis. Trends Cel Biol. 15, 349–355. doi:10.1016/j.tcb.2005.05.006
McHugh, T., Gluszek, A. A., and Welburn, J. P. I. (2018). Microtubule End Tethering of a Processive Kinesin-8 Motor Kif18b Is Required for Spindle Positioning. J. Cell Biol. 217, 2403–2416. doi:10.1083/jcb.201705209
Miki, H., Okada, Y., and Hirokawa, N. (2005). Analysis of the Kinesin Superfamily: Insights into Structure and Function. Trends Cel Biol. 15, 467–476. doi:10.1016/j.tcb.2005.07.006
O'Donnell, J. S., Teng, M. W. L., and Smyth, M. J. (2019). Cancer Immunoediting and Resistance to T Cell-Based Immunotherapy. Nat. Rev. Clin. Oncol. 16, 151–167. doi:10.1038/s41571-018-0142-8
Parsa, A. T., Waldron, J. S., Panner, A., Crane, C. A., Parney, I. F., Barry, J. J., et al. (2007). Loss of Tumor Suppressor PTEN Function Increases B7-H1 Expression and Immunoresistance in Glioma. Nat. Med. 13, 84–88. doi:10.1038/nm1517
Perri, F., Longo, F., Giuliano, M., Sabbatino, F., Favia, G., Ionna, F., et al. (2017). Epigenetic Control of Gene Expression: Potential Implications for Cancer Treatment. Crit. Rev. Oncology/Hematology 111, 166–172. doi:10.1016/j.critrevonc.2017.01.020
Pitt, J. M., Marabelle, A., Eggermont, A., Soria, J.-C., Kroemer, G., and Zitvogel, L. (2016). Targeting the Tumor Microenvironment: Removing Obstruction to Anticancer Immune Responses and Immunotherapy. Ann. Oncol. 27, 1482–1492. doi:10.1093/annonc/mdw168
Samstein, R. M., Lee, C.-H., Shoushtari, A. N., Hellmann, M. D., Shen, R., Janjigian, Y. Y., et al. (2019). Tumor Mutational Load Predicts Survival after Immunotherapy across Multiple Cancer Types. Nat. Genet. 51, 202–206. doi:10.1038/s41588-018-0312-8
Sharp, D. J., Rogers, G. C., and Scholey, J. M. (2000). Microtubule Motors in Mitosis. Nature 407, 41–47. doi:10.1038/35024000
Subramanian, A., Tamayo, P., Mootha, V. K., Mukherjee, S., Ebert, B. L., Gillette, M. A., et al. (2005). Gene Set Enrichment Analysis: a Knowledge-Based Approach for Interpreting Genome-wide Expression Profiles. Proc. Natl. Acad. Sci. 102, 15545–15550. doi:10.1073/pnas.0506580102
Topalian, S. L., Taube, J. M., Anders, R. A., and Pardoll, D. M. (2016). Mechanism-Driven Biomarkers to Guide Immune Checkpoint Blockade in Cancer Therapy. Nat. Rev. Cancer 16, 275–287. doi:10.1038/nrc.2016.36
Walczak, C. E., Zong, H., Jain, S., and Stout, J. R. (2016). Spatial Regulation of Astral Microtubule Dynamics by Kif18B in PtK Cells. Mol. Biol. Cell 27, 3021–3030. doi:10.1091/mbc.e16-04-0254
Wang, J., Guo, X., Xie, C., and Jiang, J. (2017). KIF15 Promotes Pancreatic Cancer Proliferation via the MEK-ERK Signalling Pathway. Br. J. Cancer 117, 245–255. doi:10.1038/bjc.2017.165
Woo, S.-R., Turnis, M. E., Goldberg, M. V., Bankoti, J., Selby, M., Nirschl, C. J., et al. (2012). Immune Inhibitory Molecules LAG-3 and PD-1 Synergistically Regulate T-Cell Function to Promote Tumoral Immune Escape. Cancer Res. 72, 917–927. doi:10.1158/0008-5472.can-11-1620
Wu, X., Xiang, X., and Hammer, J. A. (2006). Motor Proteins at the Microtubule Plus-End. Trends Cel Biol. 16, 135–143. doi:10.1016/j.tcb.2006.01.004
Wu, Y., Wang, A., Zhu, B., Huang, J., Lu, E., Xu, H., et al. (2018). KIF18B Promotes Tumor Progression through Activating the Wnt/β-Catenin Pathway in Cervical Cancer. Onco. Targets Ther. 11, 1707–1720. doi:10.2147/ott.s157440
Xu, H., Zhao, G., Zhang, Y., Jiang, H., Wang, W., Zhao, D., et al. (2019). Long Non-coding RNA PAXIP1-AS1 Facilitates Cell Invasion and Angiogenesis of Glioma by Recruiting Transcription Factor ETS1 to Upregulate KIF14 Expression. J. Exp. Clin. Cancer Res. 38, 486. doi:10.1186/s13046-019-1474-7
Yang, B., Wang, S., Xie, H., Wang, C., Gao, X., Rong, Y., et al. (2020). KIF18B Promotes Hepatocellular Carcinoma Progression through Activating Wnt/β-Catenin-Signaling Pathway. J. Cell Physiol. 235, 6507. doi:10.1002/jcp.29444
Yang, Y. (2015). Cancer Immunotherapy: Harnessing the Immune System to Battle Cancer. J. Clin. Invest. 125, 3335–3337. doi:10.1172/jci83871
Keywords: KIF18B, tumor immunity, prognosis, bioinformatics, immunecheckpoint
Citation: Qiu M-j, Wang Q-s, Li Q-t, Zhu L-s, Li Y-n, Yang S-l and Xiong Z-f (2021) KIF18B is a Prognostic Biomarker and Correlates with Immune Infiltrates in Pan-Cancer. Front. Mol. Biosci. 8:559800. doi: 10.3389/fmolb.2021.559800
Received: 13 May 2020; Accepted: 21 April 2021;
Published: 24 May 2021.
Edited by:
William C. Cho, QEH, ChinaReviewed by:
Mahendra K. Singh, University of Miami, United StatesCopyright © 2021 Qiu, Wang, Li, Zhu, Li, Yang and Xiong. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sheng-li Yang, eWFuZ3NoZW5nbGkyMDE0QHlhaG9vLmNvbQ==; Zhi-fan Xiong, eGlvbmd6aGlmYW5AMTI2LmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.