
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
EDITORIAL article
Front. Microbiol., 31 March 2023
Sec. Infectious Agents and Disease
Volume 14 - 2023 | https://doi.org/10.3389/fmicb.2023.1191946
This article is part of the Research TopicMetagenomics for epidemiological surveillance in ONE HEALTHView all 11 articles
Editorial on the Research Topic
Metagenomics for epidemiological surveillance in One Health
The highest burden of endemic, neglected, and emerging zoonotic diseases occur in low and middle-income countries (LMICs) (Worsley-Tonks et al., 2022). However, limited resources, surveillance infrastructure and access to advanced molecular techniques in these regions can severely impact the rapid detection and characterization necessary to mitigate and respond to outbreak events. Metagenomics approaches continue to become less expensive, and now represent a potentially cost-effective approach to conduct biosurveillance and monitor molecular epidemiology (Ko et al., 2022). Compared to PCR, which only detects pathogen based on specific primers, metagenomic agnostic sequencing allows for the identification of known and unknown microbes (viruses, bacteria and protozoans) from a single sample (Govender et al., 2021). Forward-facing portable sequencing platforms such as the MinION can provide expansive knowledge of circulating pathogens from a wide array of field-collected samples, including arthropod vectors, soil and water samples and a variety of swabs and bloods from vertebrates, both in the field and in basic laboratory conditions (Gardy and Loman, 2018; Achee et al.). Using metagenomics approaches, detection of known, emergent, and emerging zoonotic pathogens of human and veterinary importance can be made available to decision-makers, researchers, and policymakers in near real-time, to inform effective containment or mitigation efforts to minimize public health impacts.
The nine publications presented here in this Research Topic capture a wide range of settings that metagenomics can be applied to through a One Health lens, including outbreak response, clinical management, biosurveillance, and disease monitoring within the human animal interface. Publication summaries can be found in Table 1.
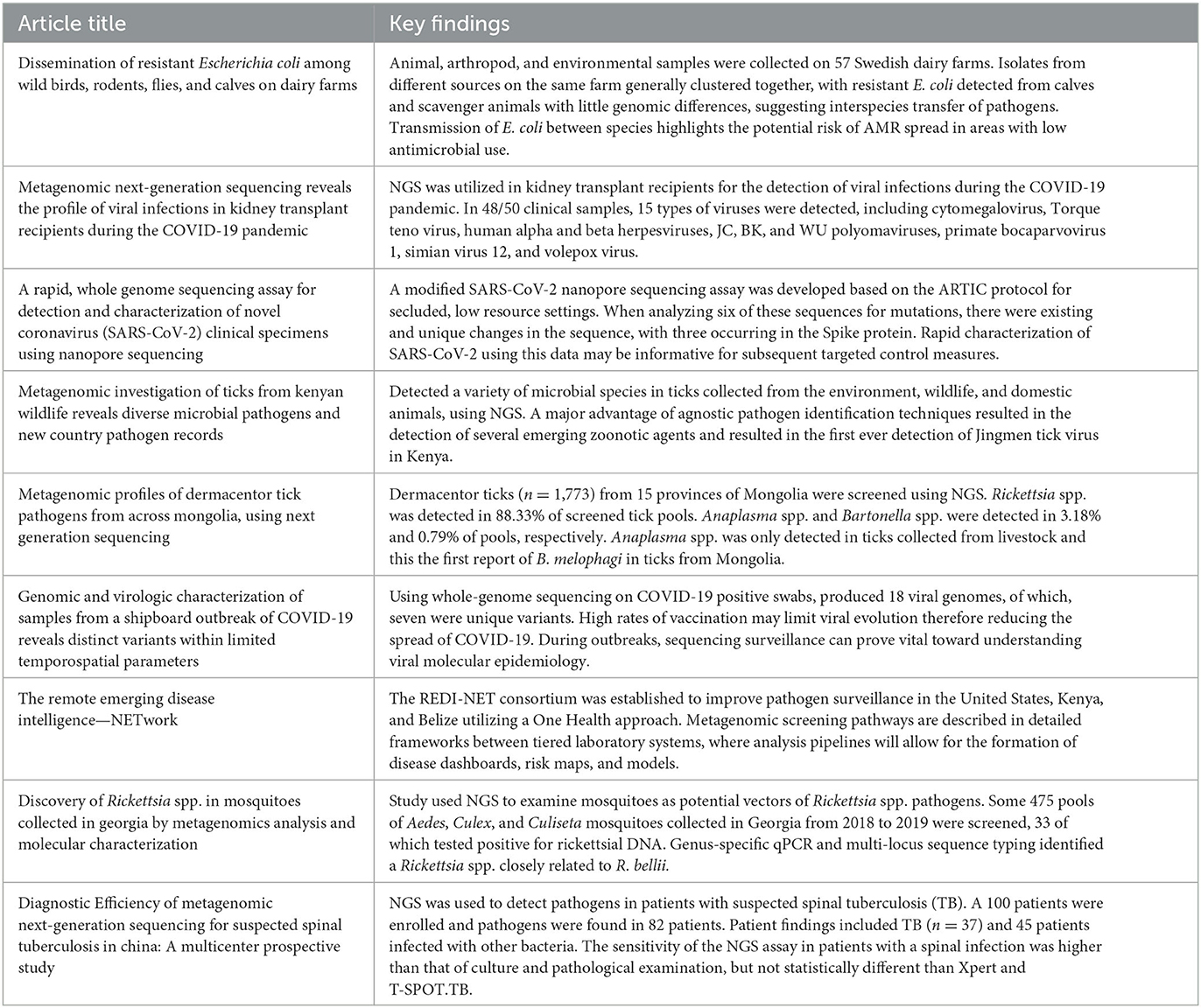
Table 1. Summary of special edition publications as they relate to metagenomics, surveillance, and One Health.
During an outbreak investigation, Cer et al., showed the utilization of metagenomics by comparing genomic variants to measure human–human spread and mutation rates of the COVID-19 virus. Comparing two different outbreaks of COVID-19—one in a group prior to vaccine development and one in a group with high vaccination rates—a larger number of variants were observed within the unvaccinated group. The study highlighted further questions and discussion on the impacts of sampling bias, the impact of purifying selection in creating clonal viral populations in vaccinated individuals, and how that can inform and support vaccination efforts.
Pandemics such as the Ebola in 2014–2016 and COVID-19 have shown that global security is compromised when response requires advanced training, special tools, and established infrastructure to accurately survey and assess the outbreak situation, where delays ultimately cost lives. It was during the Ebola outbreak that portable sequencing technology (MinION) was piloted in the field to speed up detection and public health response with a simpler, pared down protocol and analysis workflow. COVID-19 sequencing protocol using the ARTIC protocol from Illumina can be time consuming, limiting the potential use of molecular epidemiology for control measures. To reduce sequencing time during an outbreak, Arévalo et al., modified a COVID-19 whole genome sequencing assay that produced high quality metagenomics data relatively quickly in an isolated, low resource setting. Adaptation of current protocols to LMIC infrastructure and resources while providing a quick turnaround for results will be invaluable in controlling future outbreaks.
Clinically, metagenomics can be used to identify unknown or rare pathogens that might go undetected using traditional methods. An example of this was outlined by Tian et al., to detect viral infections in kidney transplant recipients (KTRs) during the COVID-19 pandemic. An NGS approach detected rare viruses in multiple sample types including bronchoalveolar lavage fluid (BALF), urine and blood. This method identified infections or co-infections in vulnerable immunosuppressed kidney transplant recipients, which was then used to inform treatment strategies.
Li et al., used NGS on clinical samples of patients who were suspected of having spinal tuberculosis (TB). Over half of the samples (45/82) tested had bacteria that was not Mycobacterium tuberculosis. This approach quickly and simultaneously confirmed patients with spinal infection if they had TB or non-TB infections. This approach also distinguished and identified other pathogens in these patients, which helped guide patient's clinical treatment. This study evaluated the efficacy and application of NGS as clinical diagnostic assay for suspected spinal TB infection in clinical laboratories.
Altantogtokh et al., tested pools of Dermacentor nuttalli ticks (n = 1,773) from 15 provinces of Mongolia using NGS. Rickettsia spp. was detected in 88.33% of screened tick pools, with Khentii aimag having the highest detection rate for Rickettsia spp. Rickettsia raoultii and Rickettsia sibirica were detected within this study, aligning with previous findings that applied NGS to livestock samples in Mongolia (Chaorattanakawee et al., 2022). Anaplasma spp. and Bartonella spp. were detected in 3.18 and 0.79% of pools, respectively, with Anaplasma spp. only detected in ticks collected from livestock, as seen elsewhere (von Fricken et al., 2020). This study represents the first detection of Bartonella melophagi in ticks tested from Mongolia.
Pollio et al., discovered a novel Rickettsia spp. in mosquitoes which shared a close identity with Rickettsia bellii based on data from NGS. While it is unknown if R. bellii is pathogenic in humans, detecting this microorganism in mosquitoes may hold value down the road should this disease emerge. The use of metagenomics to screen mosquito samples elsewhere in the world may hold value for the identification and characterization of potential pathogenic Rickettsia spp. within an atypical vector host.
Human-livestock transmission is one mode for infection of zoonotic pathogens. The likelihood of infection is increased as animals pass pathogens between species similar to the spread of Escherichia coli in Hickman et al., This interspecies exchange is conducive to pathogen mutations, and passage of mobile genetic elements or plasmids via clonal or horizontal gene transfer, increasing the risk of spreading to humans potentially increasing the severity of infection, and resistance to antibiotic interventions.
Ergunay et al., used NGS to screen 75 blood-fed ticks (Rhipicephalus spp. and Amblyomma spp.) collected from wild and domestic animals in Kenya for pathogens. Fifty-six human or veterinary pathogenic bacterial species were detected including Escherichia coli (62.8%), Proteus mirabilis (48.5%), Coxiella burnetii (45.7%), and Francisella tularensis (14.2%). Additional pathogens detected include fungal species, filarial pathogens, protozoa, and environmental and water/foodborne pathogens. Jingmen tick virus was detected within 13% of these samples, representing the first report of this virus in Kenya. Interestingly, in these wild and domestic animal blood-bags, human pathogens, including Plasmodium falciparum were detected, indicating back spill-over of pathogens of human pathogens to animals.
Finally, the adoption of a One Health focused surveillance strategy is described in detail by Achee et al., through the Remote Emerging Disease Intelligence—NETwork (REDI-NET), which has implemented MinION-based xenosurveillance strategies in the United States, Kenya and Belize. Comprehensive standard operating procedures (SOPs) were developed for the deployment of metagenomic surveillance at high-risk disease envelopes. The ultimate goal of REDI-NET is to apply agnostic pathogen detection to a variety of sample types including water, ticks, soil, and leeches across Gold- (reach-back laboratories with greater infrastructure) and Silver- (remote, field-forward laboratories) tiered laboratory systems. This study outlined the framework required for working with metagenomic data, efficient data pipeline strategies, and systematic processes for collection and testing of samples for deeper analysis.
This Research Topic highlights the diversity of metagenomic application across disciplines, geographical regions, and both research and clinical settings. The field of metagenomics is growing rapidly, with all papers in this Research Topic published within the past year. Utilizing metagenomics from a One Health perspective will expand surveillance and characterization of existing and new pathogens in humans and animals, which will likely continue to be adopted into existing systems as next generation sequencing becomes more accessible and cost-effective.
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Chaorattanakawee, S., Wofford, R. N., Takhampunya, R., Katherine Poole-Smith, B., Boldbaatar, B., Lkhagvatseren, S., et al. (2022). Tracking tick-borne diseases in Mongolian livestock using next generation sequencing (NGS). Ticks Tick Borne Dis. 13, 101845. doi: 10.1016/j.ttbdis.2021.101845
Gardy, J. L., and Loman, N. J. (2018). Towards a genomics-informed, real-time, global pathogen surveillance system. Nat. Rev. Genet. 19, 9–20. doi: 10.1038/nrg.2017.88
Govender, K. N., Street, T. L., Sanderson, N. D., and Eyre, D. W. (2021). Metagenomic sequencing as a pathogen-agnostic clinical diagnostic tool for infectious diseases: a systematic review and meta-analysis of diagnostic test accuracy studies. J. Clin. Microbiol. 59, e0291620. doi: 10.1128/JCM.02916-20
Ko, K. K. K., Chang, K. R., and Nagarajan, N. (2022). Metagenomics-enabled microbial surveillance. Nat. Microbiol. 7, 486–496. doi: 10.1038/s41564-022-01089-w
von Fricken, M. E., Qurollo, B. A., Boldbaatar, B., Wang, Y. W., Jiang, R. R., Lkhagvatseren, S., et al. (2020). Genetic diversity of Anaplasma and Ehrlichia bacteria found in Dermacentor and Ixodes ticks in Mongolia. Ticks Tick Borne Dis. 11, 101316. doi: 10.1016/j.ttbdis.2019.101316
Worsley-Tonks, K. E. L., Bender, J. B., Deem, S. L., Ferguson, A. W., Fèvre, E. M., Martins, D. J., et al. (2022). Strengthening global health security by improving disease surveillance in remote rural areas of low-income and middle-income countries. Lancet Glob. Health. 10, e579–e584. doi: 10.1016/S2214-109X(22)00031-6
Keywords: metagenomics, biosurveillance, One Health, next generation sequencing (NGS), vector-borne diseases
Citation: von Fricken ME, Melendrez MC, Linton Y-M and Takhampunya R (2023) Editorial: Metagenomics for epidemiological surveillance in One Health. Front. Microbiol. 14:1191946. doi: 10.3389/fmicb.2023.1191946
Received: 22 March 2023; Accepted: 24 March 2023;
Published: 31 March 2023.
Edited and reviewed by: Axel Cloeckaert, Institut National de Recherche pour l'Agriculture, l'Alimentation et l'Environnement (INRAE), France
Copyright © 2023 von Fricken, Melendrez, Linton and Takhampunya. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Michael E. von Fricken, bXZvbmZyaWNAZ211LmVkdQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.