
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Microbiol. , 27 June 2019
Sec. Food Microbiology
Volume 10 - 2019 | https://doi.org/10.3389/fmicb.2019.01384
This article is part of the Research Topic Interactions Between Diets, Gut Microbiota and Host Metabolism View all 56 articles
 Liang Gong†
Liang Gong† Haocheng He†
Haocheng He† Dongjie Li
Dongjie Li Lina Cao
Lina Cao Tahir Ali Khan
Tahir Ali Khan Yanping Li
Yanping Li Lifei Pan
Lifei Pan Liang Yan
Liang Yan Xuezhi Ding
Xuezhi Ding Yunjun Sun
Yunjun Sun Youming Zhang
Youming Zhang Ganfeng Yi
Ganfeng Yi Shengbiao Hu*
Shengbiao Hu* Liqiu Xia*
Liqiu Xia*Probiotic-feeding continues to be a promising strategy to control the bacterial pathogens in aquaculture. A new Pediococcus pentosaceus strain (SL001) was isolated from 1000s of soil samples, which exhibited wide antimicrobial spectrum of against fish pathogens, involving Aeromonas hydrophila, Aeromonas veronii, Aeromonas sobria, Edwardsiella tarda, Lactococcus garvieae, and Plesiomonas shigelloide. The challenge test against A. hydrophila showed that the survival rate of SL001-supplemented group was significantly higher than that of control group (P < 0.05). Moreover, SL001 could stably colonize in gut of grass carp and increased mucus-secreting goblet cells and extended intestinal villi could be observed in SL001-supplemented group (P < 0.05). Feeding with SL001 supplemented diet could significantly enhance the growth rate (P < 0.05) and markedly affect gut microbiota structure of grass carps, resulting in reduced potential pathogens and increased potential probiotics. Furthermore, feeding grass carps with SL001 caused the up-regulated expression of insulin-like growth factor (IGF-1 and IGF-2) and down-regulated expression of myostatin (MSTN-1 and MSTN-2) (P < 0.05), which probably also account for the increased growth rate of SL001-fed group. Meanwhile, relative mRNA expression levels of immune-related genes in liver, spleen, and head kidney were analyzed in grass carps after feeding for 30 days with SL001 supplemented diets. In all three immune organs, the expression levels of immunoglobulin M (IgM) and complement 3 (C3) were significantly increased (P < 0.05), whereas the interleukin-8 (IL-8) was down-regulated (P < 0.05). Besides, whole genome sequencing revealed several probiotics properties of SL001, including organic acid synthesis, bacteriocin synthesis (coagulin), superoxide dismutase, and digestive enzymes. In conclusion, P. pentosaceus SL001 which could enhance immunity and promoter growth rate of grass carps, is prospective to be used as a dietary probiotic in freshwater fish aquaculture.
High density of culture and increase of feeding amount have caused long-term crowding stress that aggravates fish susceptibility to pathogens, and outbreaks of fish diseases have become increasingly serious (Athanassopoulou et al., 2004; DiMaggio et al., 2014; Lin et al., 2018). Usually, fishes are frequently infected by microorganisms associated with viruses, bacteria, and parasites under extremely intensive culture conditions (Song et al., 2014; Tang et al., 2018; Zhou et al., 2018). In the past many years, control strategies on fish pathogens relied mostly on the application of chemicals, such as antibiotics and disinfectants. However, extensive use of medicines has caused antibiotic resistance of pathogenic bacteria (Chandrarathna et al., 2018). Therefore, overuse of antibiotics in aquaculture should be strictly controlled, and alternative methods of controlling fish pathogens must be developed, such as using antimicrobial peptides, vaccines, and probiotics.
Recently, probiotic-feeding has been proved to be a promising strategy to control diseases. Probiotics are beneficial microorganisms introduced by implantation or colonization in specific host’s gut to reinforce the intestinal barrier, boost the immune system, or produce antimicrobial substances to suppress pathogen growth (Balcazar et al., 2006; Akhter et al., 2015). Besides, probiotics could promote digestion and enhance nutrient absorption by altering the intestinal microflora, resulting in better growth performance (Balcazar et al., 2006; Falcinelli et al., 2015). Lactic acid bacteria (LAB) and Bacillus species are among the most commonly used probiotic candidates (Cao et al., 2011; Banerjee and Ray, 2017; Alonso et al., 2018; Yi et al., 2018). LAB strains that have been used for fish pathogens control usually include the genera Lactobacillus, Leuconostoc, Streptococcus, and Pediococcus (Perez-Ramos et al., 2018). Pediococcus parvulus 2.6 produces β-glucan, which could benefit colonization of its producer in the fish gut and competition with the pathogen Vibrio anguillarum (Perez-Ramos et al., 2018). Feeding orange-spotted grouper with Pediococcus pentosaceus strain 4012 could not only enhance the growth rate of the grouper and increase the number of red blood cells, but also regulate the gene expression of the pro-/anti-inflammatory cytokines (Huang et al., 2014). Similarly, Pediococcus acidilactici-supplemented diet significantly increases the expression levels of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) in juvenile beluga (Ghiasi et al., 2018).
In aim to screen probiotics which could antagonize bacterial fish pathogens, 100s of bacterial strains have been isolated from soil samples collected from different regions of China. Among them, a novel P. pentosaceus strain (SL001) exhibited excellent antibacterial activity against several important fish pathogens has been added to fish diet and its probiotics properties have been studied. The impact of SL001 feeding on the gut microbiota and growth ability of grass carps was analyzed. Subsequently, the expression of growth-related and immune-related genes of grass carps was measured after feeding with SL001-supplemented diet. Our results indicated that P. pentosaceus SL001 could serve as potential probiotic strain in freshwater aquaculture.
Soil samples from different regions of China were collected and 10-fold serially diluted in glass tubes. 0.1 mL dilutions was spread on Man Ragosa Sharpe (MRS) agar plates and anaerobic cultured on DG250 Anaerobic workstation (DWS, United Kingdom) at 37°C for 24 h. Single colonies appeared on MRS agar plates were purified twice and stored in 25% (v/v) glycerol at -80°C. The P. pentosaceus SL001 was grown on MRS liquid medium at 37°C for 12 h under anaerobic condition, and then scanning electron microscopy (SEM, Hitachi Su8010, Japan) were used to observe the morphology of the bacterial cell. Biochemical characterization of SL001 was done subsequently. Genomic DNA was extracted from a SL001 using a genomic DNA extraction kit according to the manufacturer’s instructions (Sangon, China). The DNA fragments carrying 16S rRNA gene were amplified using primer pair 27F (5′ to 3′: AGAGTTTGATCCTGGCTCAG) and 1492R (5′ to 3′: CGGTTACCTTGTTACGACTT) (Wu et al., 2010; Yi et al., 2018). The polymerase chain reaction (PCR) products were purified and cloned into pMD18-T vector (TaKaRa, Japan). Single colonies were picked up and sent for sequencing. Phylogenetic tree was constructed on the basis of 16S rRNA genes by the neighbor-joining method using MEGA6.06 software and evolutionary distances were computed using the Maximum Composite Likelihood method (Tamura et al., 2004).
SL001 was incubated in MRS liquid medium at 37°C for 24 h under anaerobic conditions. Six fish pathogenic bacteria (Supplementary Table S1) were incubated in Luria-Bertani (LB) liquid medium at 30°C for 12 h. Overnight cultures of pathogenic bacteria were diluted to 107 CFU/mL by fresh LB broth, and 200 μL dilutions were spread on LB agar plates respectively. Then, sterile oxford cups were placed on the plates dried for 30 min on vertical flow clean cabinet. 100 μL of cell-free supernatant (CFS, pH 3.48) of SL001 saturated culture was added into oxford cups. MRS and phosphate-buffered saline (PBS, need pH 3.48 with lactic acid, PL) were used as control. After 18 h of incubation at 30°C, antimicrobial activity was evaluated by measuring inhibition zones (Wang et al., 2018; Yi et al., 2018).
Hepatic L8824 cell line derived from liver of grass carp was cultured at 37°C with 5% CO2 in DMEM (Gibico/Thermo Fisher Scientific, United States) supplemented with 10% fetal calf serum (FCS, BI, Israel) and 1% penicillin-streptomycin solution (BI, Israel). Cells were pre-incubated for 12 h in 24-well flat-bottomed plates (5 × 105 cells/well) as previous described (Matsuda et al., 2017). Both SL001 CFS (10 μL) and bacterial cells (1 × 107 CFU) were applied to L8824 cells and cell morphological changes were analyzed using an inverted light microscope (Leica Microsystems S.p.A, Italy). CFS of Aeromonas hydrophila which has been proved to be toxic to L8824 cells was used as positive control and MRS was used as negative control.
Healthy grass carps (32.1 ± 9 g) obtained from Wangcheng fish pond (Changsha, China) were transferred plastic tanks (volume 60 L) equipped with air pump in laboratory and fed with the basal diet for 1 week. Grass carps were randomly divided into six plastic tanks (38 fishes per tank) and fed twice with different diets per day at 1% of the body weight: three tanks fed with basal diet, three tanks fed with SL001 (1 × 109 CFU/g) supplemented diet (Huang et al., 2014; Yi et al., 2018).
Twelve grass carps were sampled randomly from every tank after 30 days of feeding. Three grass carps were taken for immune-related and growth-related genes expression analysis. Immune organs (liver, spleen, and head kidney) and muscle were collected, sliced, and immediately frozen at -80°C until RNA isolation. Two grass carps’ gut were taken for colonization identification. Two grass carps’ gut were stored in 4% paraformaldehyde fix solution and sent for histological analysis. Five grass carps were taken for intestinal bacterial community diversity analysis by using high-throughput sequencing (Wuhan Nextomics Biotechnology, Co., Ltd., China). The rest 26 grass carps from every tank were used for challenge test against A. hydrophila. Liver, spleen, and head kidney from grass carps after 6 h post-infection (6 hpi) or 12 h post-infection (12 hpi) were collected for immune-related genes expression analysis.
Polymerase chain reaction amplification of the variable region (V3–V4) of tract bacterial 16S rRNA gene was carried out using primer pair 338F (5′ to 3′: ACTCCTACGGGAGGCAGCAG) and 806R (5′ to 3′: GGACTACHVGGGTWTCTAAT). Amplifications were performed in ABI GeneAmp® 9700 (Applied Biosystems, United States) with initial denaturation at 95°C for 3 min, 28 cycles of 30 s at 95°C, 30 s at 58°C, 45 s at 72°C and final extension at 72°C for 10 min. PCR products were purified by using AxyPrepDNA PCR Purification Kit (Axygen, United States) and quantified using QuantiFluorTM-ST Fluorometer (Promega, United States). Sequencing was performed by Wuhan Nextomics Biotechnology, Co., Ltd., with Illumina MiSeq system (Illumina, United States).
Paired-end (PE) reads were procured and then assembled according to the overlap relationship (Huang et al., 2016). The operational taxonomic units (OTUs) picking was completed with a minimum pairwise identity of 97% with USEARCH software (version 7.1) (Edgar, 2010) after removing chimeras. Taxonomic analyses of sequence reads were processed using Quantitative Insights Into Microbial Ecology (QIIME) pipeline (Caporaso et al., 2010). The most abundant sequence in each OTUs was selected to perform a taxonomic classification based on the Silva database (Quast et al., 2013) using the RDP classifier (Wang et al., 2007), clustering the sequences at 97% similarity with a 0.7 confidence thresholds. Alpha diversity indices were calculated with Mothur (version v.1.30.1) (Schloss et al., 2011). ACE and Chao1 were estimated to indicate the community richness, as well as Simpson and Shannon indices were reckoned to reveal the community diversity, and Good’s coverage represented the sequencing depth. Rarefaction (Amato et al., 2013) and Shannon–Wiener curve (Wang et al., 2012) were constructed with R software. Two dimensional principal coordinates analysis (PcoA) characterized the similarities and differences of the community composition. The similarities and differences in overall bacterial community structure between each of intestine samples were detected using the UniFrac metric, and then phylogenetic tree was constructed using the unweighted pair group method with arithmetic mean (UPGMA).
Intestinal contents collected from foregut, midgut, and hindgut were washed with sterile saline (0.85%) and analyzed separately. Colonization of SL001 in intestine was measured by plating the various dilutions of intestinal contents on MRS agar plates. The appeared colonies were identified using 16S rRNA gene sequence.
Intestinal tissues collected for both SL001-supplemented group and control group (six fishes for each group) were preserved in 4% paraformaldehyde fix solution, cleared in xylene and dehydrated in ethanol solutions, and then embedded in paraffin wax. Then, tissues were sectioned to 4 μm and stained with hematoxylin and eosin (H&E) according to standard protocol before examined under a light microscope (Nikon Eclipse E100, Japan).
The serum samples collected at 30 days post-feeding were taken for determining the acid phosphatase (ACP) and alkaline phosphatase (AKP) activity. The measurement was performed with commercial kits from Nanjing Jiancheng Institute (Nanjing, China) according to the manufacturer’s instructions (Yi et al., 2018).
Total RNA was isolated from harvested samples following instructions with modifications by using Trizol reagent (Sangon, China). Briefly, tissue (∼50 mg) was homogenized in 1 mL Trizol reagent. The solution was mixed with 0.2 mL chloroform and in an ice bath for 10 min. After centrifugation at 12000 rpm, 4°C for 10 min, the aqueous phase was transferred to a new 1.5 mL tube and added equal volume of isopropanol with subsequent ice bath for 10 min. The mixture was centrifuged, and then the precipitation was washed twice with 75% ethanol and dissolved in RNase free water. The RNA quality and quantity were assessed using agarose gel (2%) electrophoresis and the ratio of OD260 nm to OD280 nm by NanoDrop 2000 spectrophotometer (Thermo, United States) respectively.
Total RNA (1 μg) was treated with DNase and reversed transcription to synthesize cDNA by PrimeScriptTM RT reagent Kit with gDNA Eraser (TaKaRa, Japan) according to manufacturer’s instructions. For real-time quantitative PCR (qRT-PCR), the primer pair were designed with Primer Premier 5.0 according to the published sequences and listed in Table 1. The amplification was performed with SYBR Permix Ex TagTM GC (TaKaRa, Japan) at 7500 Real-Time PCR system instruments (Applied Biosystems, United States). The PCR cycling conditions were as follows: 2 min at 50°C and 10 min at 95°C, followed by 40 cycles of 15 s at 95°C and 1 min at 60°C. Melting curves were performed from 60 to 95°C to validate the specificity of PCRs (Palazzotto et al., 2015). The β-actin gene was used as internal control gene (Chu et al., 2018).
To monitor the effect of SL001 on the growth of grass carps, the weight gain rate (WGR), specific growth rate (SGR), feed intake (FI), and feed conversion ratio (FCR) of grass carps were calculated as previously described (Shi et al., 2019).
W0 and Wt designates the average weights of grass carps at the start of the experiment and at the termination of the experiment respectively.
As preceding description of Yi et al. (2018). A. hydrophila was inoculated into LB and shaked for 12 h. Overnight culture of A. hydrophila was diluted to 1 × 107 CFU/mL with sterile saline. Grass carps were divided into three groups: in experimental group, grass carps were fed with SL001 supplemented diet for 30 days and then intraperitoneally injected with 100 μL of A. hydrophila suspensions (106 CFU); in challenge control group, grass carps were fed with basal diet and intraperitoneally injected with 100 μL of A. hydrophila suspensions (106 CFU); in negative control group, grass carps were fed with basal diet and intraperitoneally injected with 100 μL sterile saline solution. Protective effect of SL001 against A. hydrophila was evaluated by relative percent survival (RPS) using the following formula: RPS = (1 – mortality in SL001 supplemented group/mortality in challenge control group) × 100%.
Genomic DNA of SL001 was purified using magnetic beads (0.5×) and quantified using NanoDrop 2000 spectrophotometer and Qubit 4 Fluorometer (Thermo, United States). Whole genome sequencing was performed by Wuhan Nextomics Biotechnology, Co., Ltd., using the Nanopore sequencing platform. After filtering out invalid data, the assembly was performed with Canu software (v1.3) (Koren et al., 2017) and corrected using Nanopolish 0.8.4 software (Walker et al., 2014). The assembled genome was annotated with RAST sever (Overbeek et al., 2014). Genome was further analyzed with Anti-SMASH for secondary metabolite and bacteriocin biosynthesis gene clusters. The tRNA, rRNA, and CRISPR were predicted using tRNAscan-SE 1.23, RNAmmer 1.2, and CRISPRS II respectively.
All data were statistically analyzed using SPSS 21.0 software and presented as the mean ± SE of mean (SEM). Independent-Samples t-test and Least-Significant Difference test were used to calculate significant differences. P-value < 0.05 and < 0.01 were considered statistically significant and extremely significant, respectively.
All sampled fish were humanely euthanized by bath immersion using an overdose of MS222 (3-Aminobenzoic acid ethyl ester methanesulfonate, Sigma, United States). This study was carried out in accordance with the recommendations of Animal Management Regulations (Directive 1988/2/CN). This study has been reviewed and approved by the ethics committee of the Hunan Normal University.
Soil samples from different regions of China were collected and subjected to screening of bacterial colonies which could inhibit the growth of bacterial fish pathogens. Among 100s of isolated bacterial strains, the isolate designated as SL001 which was isolated from soil sample collected from Dadonghai beach (located at the south coast of Sanya, China), exhibited excellent antibacterial activity against several important fish pathogens, including Aeromonas hydrophila, Aeromonas veronii, Aeromonas sobria, Edwardsiella tarda, Lactococcus garvieae, and Plesiomonas shigelloide (Figure 1A and Supplementary Figure S1B).
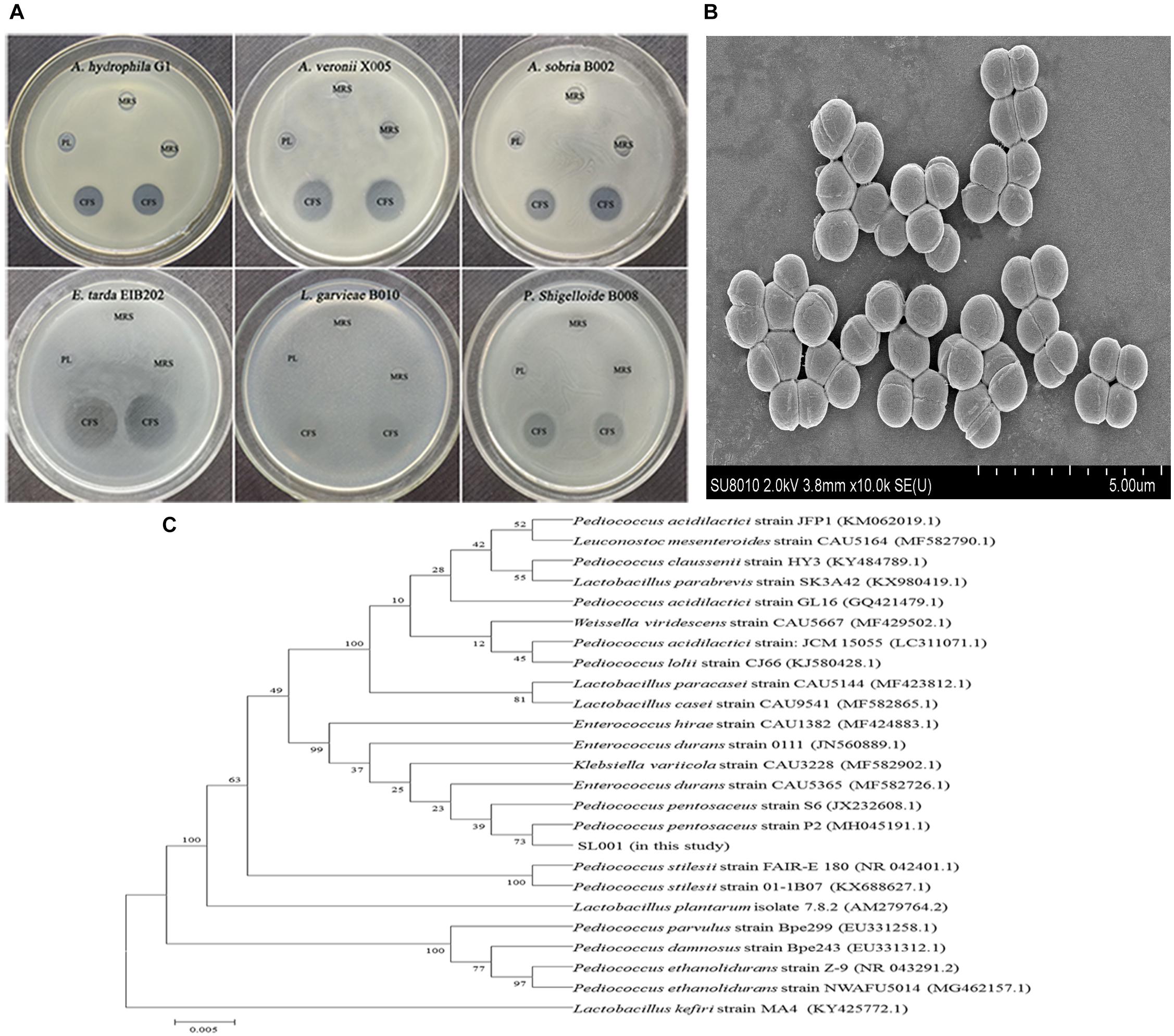
Figure 1. The characterization and antimicrobial activity of Pediococcus pentosaceus SL001. (A) Scanning electron microscope (SEM) of SL001. (B) The inhibition zones of SL001 against fish pathogens. CFS, cell-free supernatant (pH 3.48); MRS, Man Ragosa Sharpe medium; PL, phosphate-buffered saline added lactic acid (pH 3.48). (C) The phylogenetic tree based on 16S rRNA gene sequences inferred evolutionary relationships of strain SL001 by neighbor-joining method.
SL001 cells, which were spherical (0.9–1.1 μm in diameter), Gram-positive, non-spore-forming and appear in pairs or quadruples, could form opalescent and wet colonies on MRS agar plate (Figure 1B and Supplementary Figure S1A). Subsequently, SL001 was characterized on the basis of 16S rRNA gene sequence analysis, which demonstrated that SL001 clustered with P. pentosaceus strain P2 (GenBank Accession No. MH045191.1), with 99% similarity (Figure 1C). The results of biochemical characterization of SL001 was also consistent with that of previously reported P. pentosaceus strains (Supplementary Table S2) (Shukla and Goyal, 2014). Thus, SL001 was identified as a new isolate of P. pentosaceus.
As SL001 showed remarkable potential in controlling fish pathogens, we investigated its biosafety for fish. Cell-free supernatant (CFS) and the cell pellet of SL001 saturated culture was applied to hepatic L8824 cells. After 36 h of incubation, neither CFS nor cell pellet of SL001 exhibited cytotoxicity against L8824 cells. We then evaluated the biosafety of SL001 in vivo by supplementing SL001 (1 × 108 CFU/mL) in fish diet. After 30 days of feeding, neither mortality nor visible adverse effects was observed in grass carps (Figure 2).
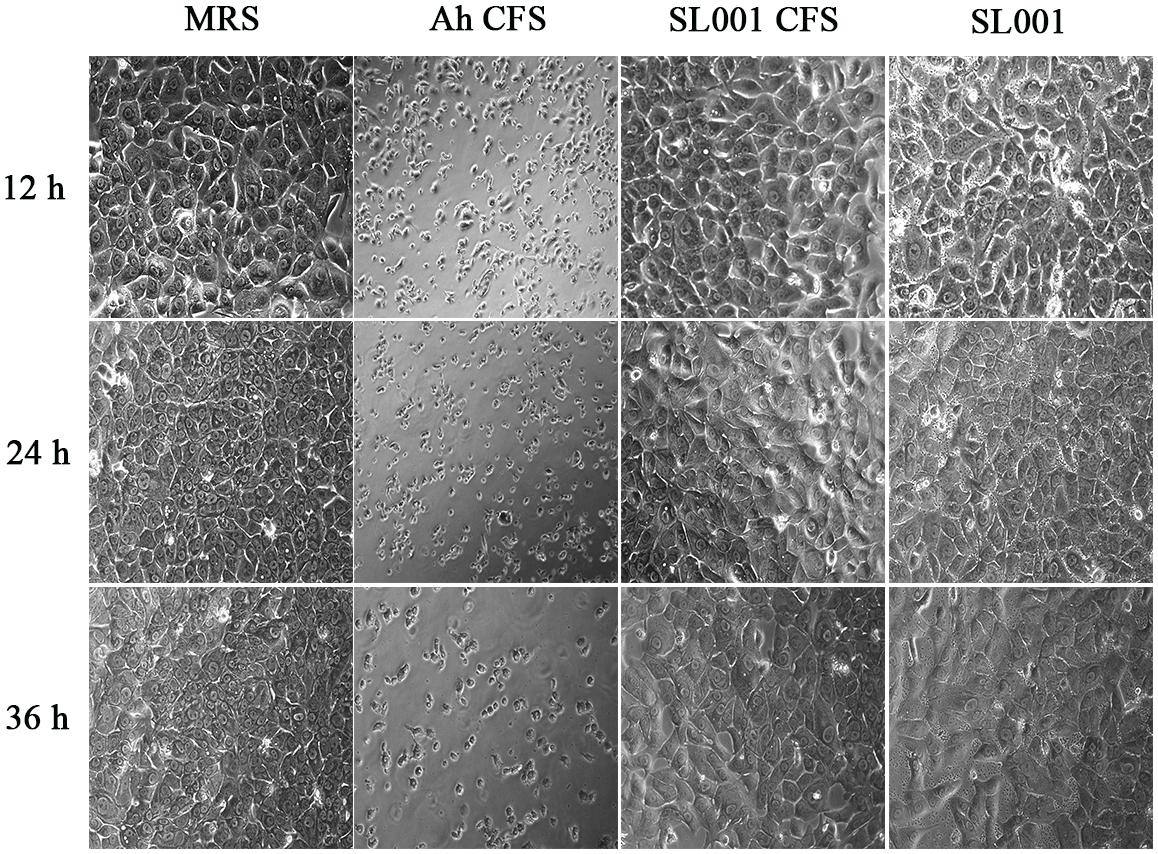
Figure 2. The morphology of grass carp hepatic L8824 cell line after treatment with SL001. Medium or bacterial culture applied to L8824 cells and incubated for 12, 24, and 36 h, respectively. MRS, Man Ragosa Sharpe medium (negative control); Ah CFS, cell free culture supernatant of Aeromonas hydrophila (positive control); SL001 CFS, cell free culture supernatant of SL001; SL001, cell pellet of SL001.
After 30 days of feeding with SL001-supplemented diet, grass carps were intraperitoneally challenged with A. hydrophila (1 × 106 CFU per fish). The cumulative mortality of SL001-supplemented group after 7 days was 51.7%, which was significantly lower than that of without SL001 supplemented group (90%) (Figure 3), with a RPS value of 42.6%. Thus, it indicated that SL001 could protect grass carps from A. hydrophila infection.
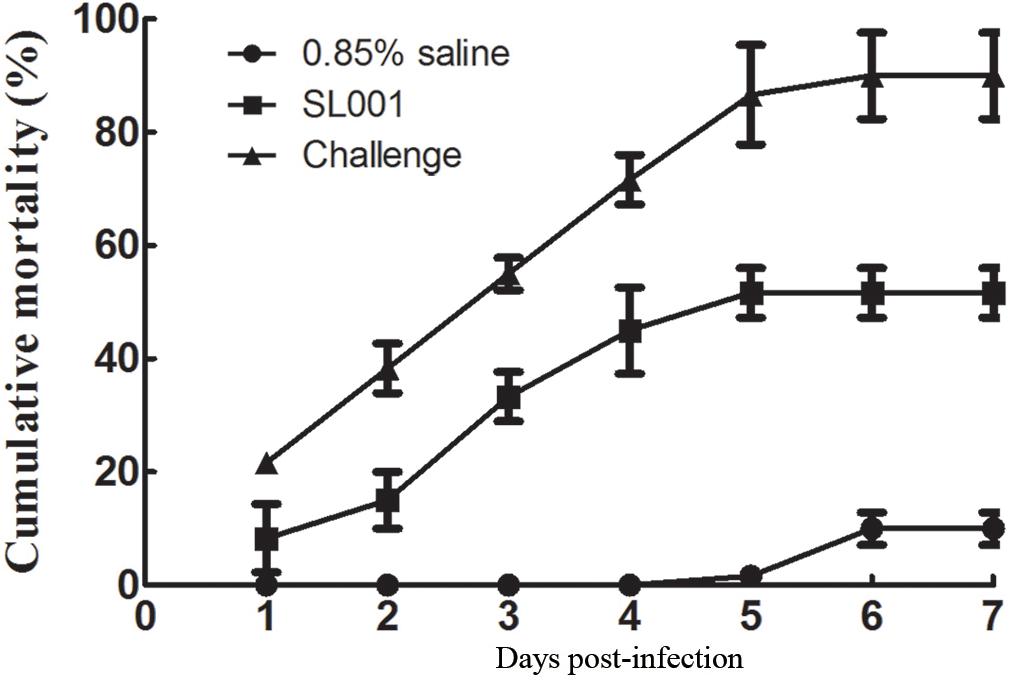
Figure 3. Challenge test to detect the protection of SL001 against A. hydrophila. The survival rates of grass carp after A. hydrophila infection were measured. Each value represents mean ± SEM (n = 3).
After 30 days of feeding, all the detected growth indices including weight gain rate (WGR, 15.06%), specific growth rate (SGR, 0.47%), feed intake (FI, 9.88) and feed conversion ratio (FCR, 2.35) of SL001-supplemented group were significantly increased when compared to those of control group (11.69, 0.37, 7.03, and 2.13%, respectively) (Supplementary Table S3). Histological analysis of intestine of grass carps demonstrated that normal villi, distinct lamina propria, enterocytes, and goblet cells were visible in both SL001-supplemented group and without SL001 supplemented group (n = 6, Figure 4). However, more mucus-secreting goblet cells and longer intestinal villi could be observed in SL001-supplemented group. This observation was more pronounced in the midgut (as the main structure of nutrient absorption), with the increase in length of villi by approximately 53% (P < 0.05, Supplementary Table S4), indicating that SL001 promoted nutrients absorption in grass carps. Moreover, the lamina propria in control group was wider compared with that in SL001-supplemented group (Figure 4), implying that SL001 could reduce inflammatory response in grass carps. After anaerobic cultivated, SL001 dominated in all the dilutions of intestinal contents in SL001-supplemented group and only SL001 colonies appeared on MRS agar plate in the 10-8 dilution, which was confirmed by 16S rRNA gene sequencing (Supplementary Figure S2).
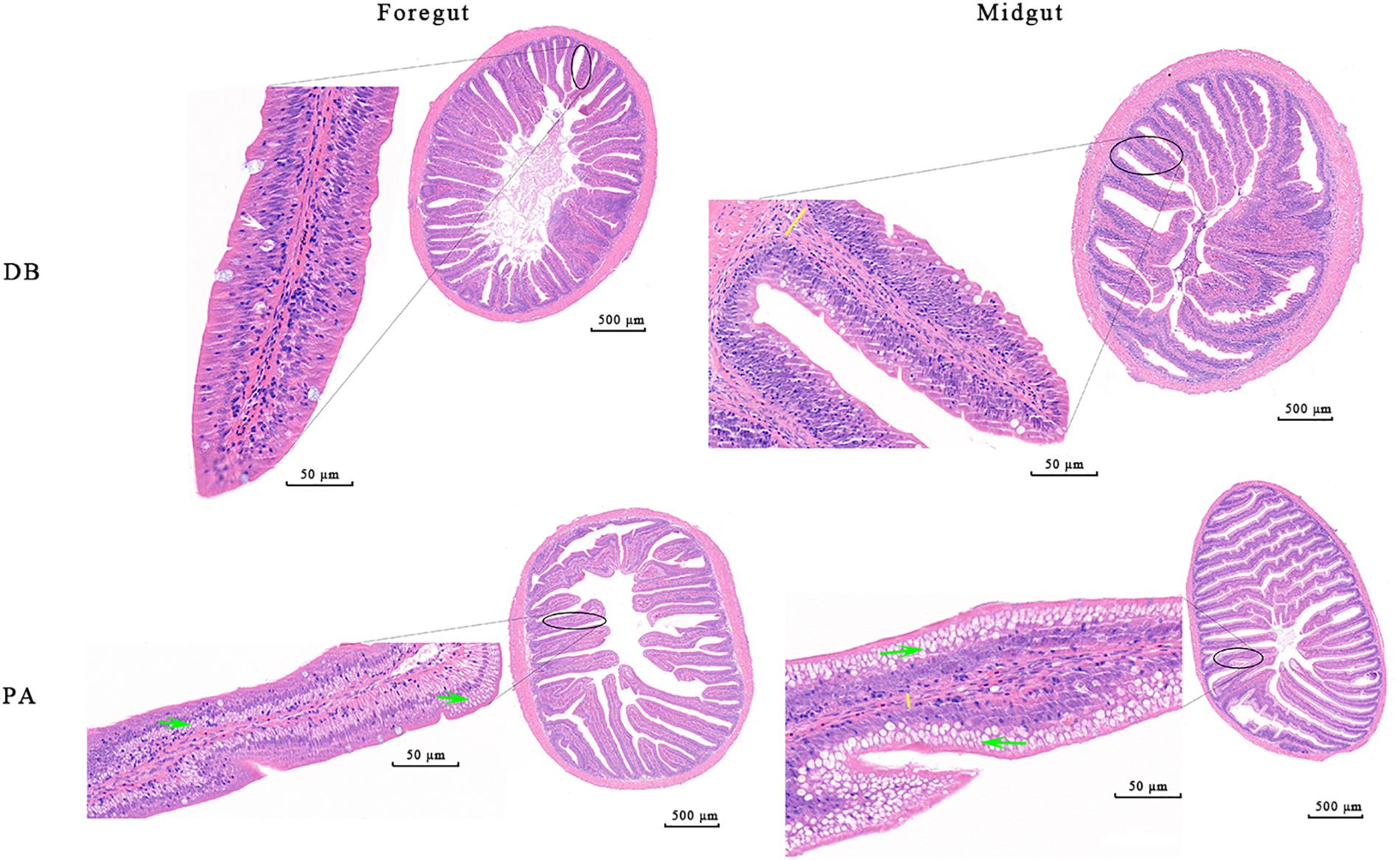
Figure 4. Photomicrographs of the different intestinal site of grass carp after 30 days of feeding with SL001-supplemented diet (n = 6, data from one fish is presented). Enterocytes (white arrow), goblet cells (red arrow), lamina propria (indicated by yellow lines) are shown in the figure. PA, SL001-supplemented group; DB, without SL001 supplemented group.
We subsequently examined the expression of genes involved in muscle growth regulation using qRT-PCR. In SL001-supplemented group, genes involved in restraining muscle growth (myostatin, MSTN-1, and MSTN-2) were significantly down-regulated, whereas genes related to promoting muscle growth (insulin-like growth factor, IGF-1 and IGF-2) were notably up-regulated (Figure 5).
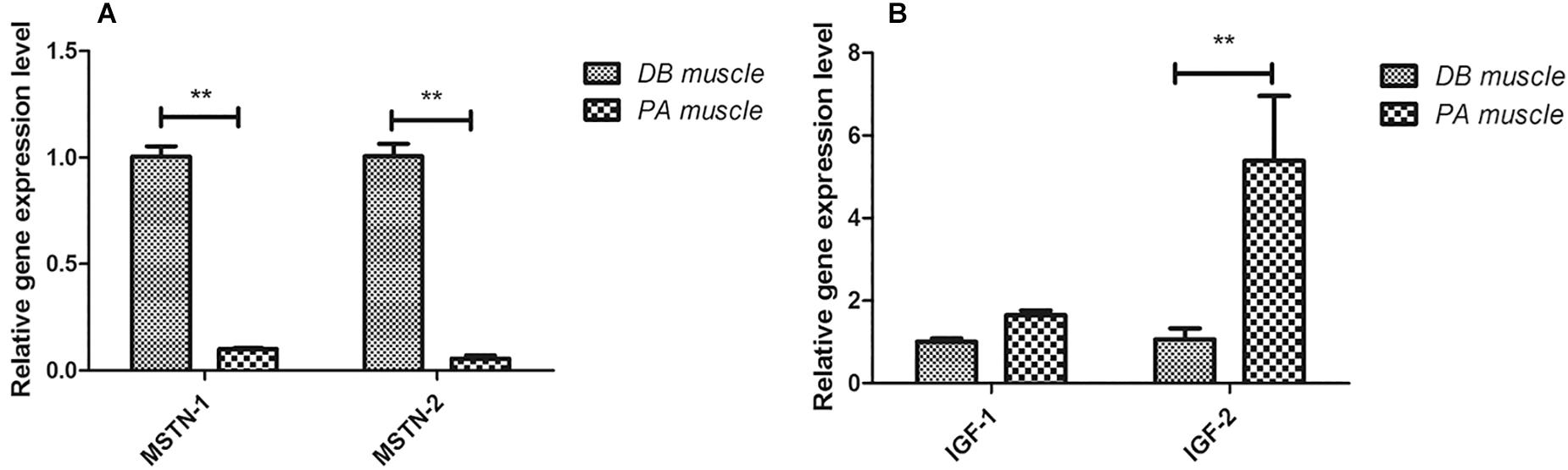
Figure 5. The expression levels of genes involved in muscle growth in grass carp. (A) Genes involved in restraining muscle growth (MSTN-1 and MSTN-2). (B) Genes involved in promoting muscle growth (IGF-1 and IGF-2). Values represented mean ± SEM. Each gene expression level in control group was regarded as 1. ∗P < 0.05, ∗∗P < 0.01 (n = 4). PA, SL001-supplemented group; DB, without SL001 supplemented group.
In order to evaluate the influence of SL001 on gut microbiota structure of grass carps, high-throughput sequencing was conducted to sequence 16S rRNA gene (V3–V4 region) of bacterial community from intestine of grass carps at 30 days post-feeding (dpf). A total of 431493 valid sequences and 441 OTUs were obtained. The rarefaction curves reached a saturation phase at approximately 360 OUTs, which indicated that the sequencing data were reasonable and could reliably describe the full microbial diversity (Figure 6A). This conclusion was further verified by the Good’s coverage (Table 2) and Shannon–Wiener curve (Supplementary Figure S3A). The LDA Effect Size (LEf Se) also revealed that the abundance of gut microbiota between SL001-supplemented group and without SL001-supplemented group exhibited significant differences. Pediococcus, Lactobacillaceae, Lactobacillales and bacilli were the over-represented taxon in SL001-supplemented group (Supplementary Figure S3B). Alpha diversity indices showed a higher microbial diversity and lower community richness in SL001-supplemented group compared to without SL001-supplemented group (Table 2). PCoA plots based on weighted UniFrac distances were used to evaluate bacterial community composition, which showed a clear clustering pattern that samples were largely partitioned based on the SL001-feeding (Figure 6D). The existence of statistically significant differences in both abundance and community composition between SL001-supplemented group and without SL001-supplemented group (Figure 6F) indicated that SL001 could affect, to a great extent, gut microbiota of grass carps.
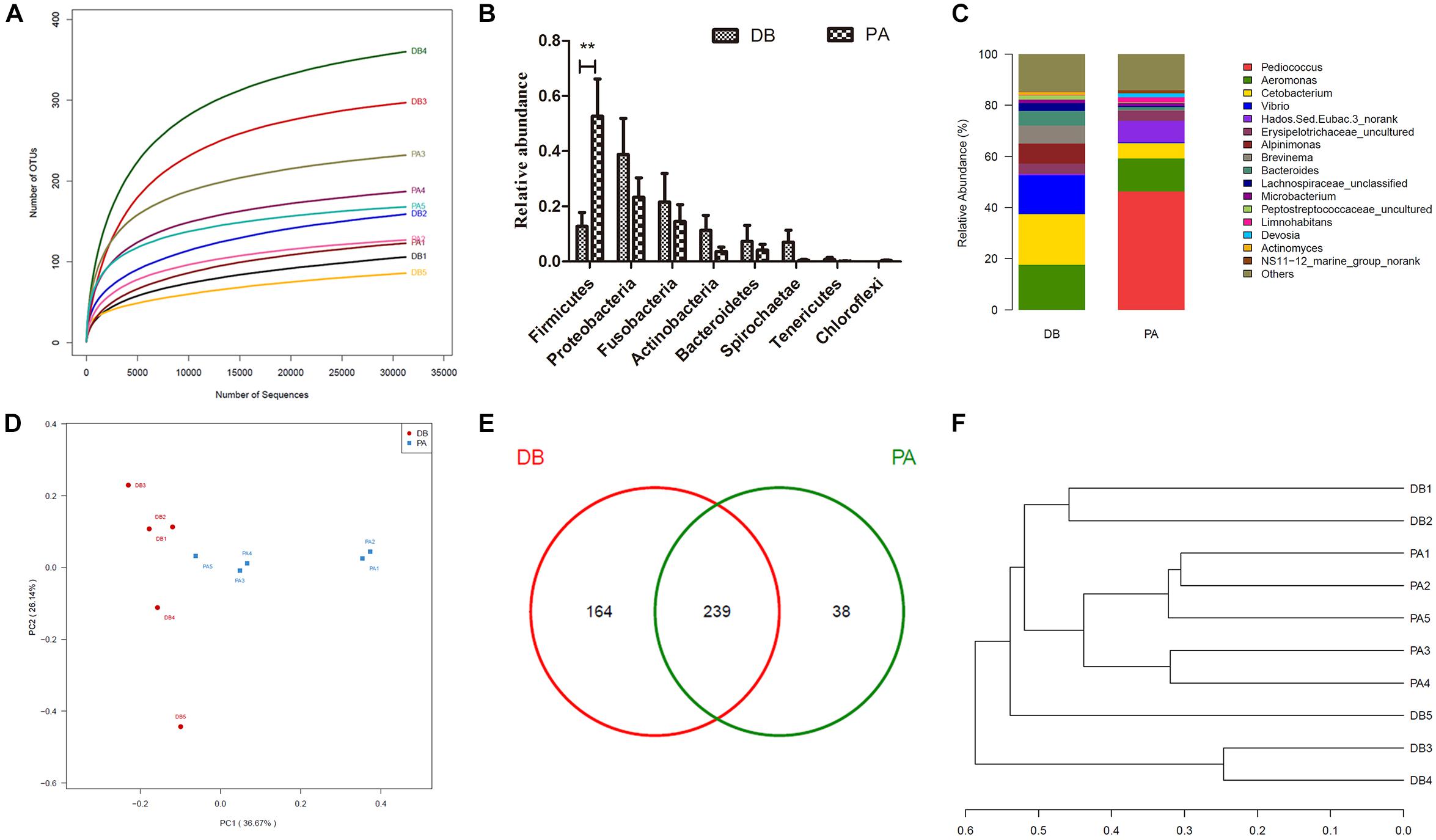
Figure 6. The influence of SL001 on gut microbiota of grass carps. V3–V4 region of bacterial 16S rRNA genes were sequenced and analyzed on 30 dpf grass carps. (A) The rarefaction curves plot of all samples. (B) Relative abundance in phyla level. (C) Stacked bar chart about the relative abundance in genus level. (D) Two dimensional principal coordinates analysis (PCoA) graph based on weighted UniFrac distances. (E) Venn diagram on shared and unique OTUs in SL001-supplemented group and without SL001 supplemented group. (F) The phylogenetic tree was constructed using unweighted pair group method with arithmetic mean (UPGMA). PA, SL001-supplemented group; DB, without SL001 supplemented group.
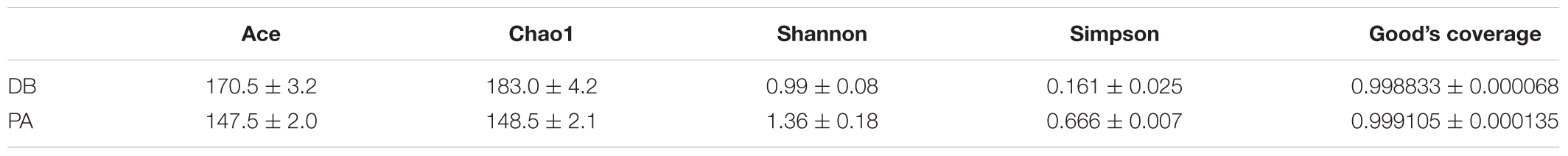
Table 2. Alpha diversity metrics of Ace, Chao1, Shannon’s diversity, Simpson, and Good’s coverage of grass carps of 30 dpf (n = 5).
All sequences were classified into 20 phyla and 285 genera. As a whole, the phyla with relative abundance of above 0.1% were clearly observed in the bar graph (Figure 6B). In general, the dominant phyla (abundance > 10%) in both groups were Firmicutes, Proteobacteria, and Fusobacteria. However, the relative abundance of Firmicutes in SL001-supplemented group was around five times higher than that in without SL001-supplemented group (Figure 6B). In genus level, Pediococcus, which was the dominant genus in SL001-supplemented group (46.3% of all reads), could hardly be detected in non-SL001-supplemented group (0.007% of all reads), indicating that SL001 could inhabit the gut of grass carps (P < 0.05, Figure 6C). Importantly, two fish pathogens, namely Aeromonas and Vibrio showed a higher abundance in without SL001-supplemented group (17.6 and 15.1%, respectively) than that in SL001-supplemented group (12.9 and 0.3%, respectively) (Figure 6C and Supplementary Figure S3C). The shared microbiome, discovered in both groups, was identified and comprised 239 OTUs; 38 OTUs were unique in SL001-supplemented group and 164 OTUs were unique in without SL001 supplemented group (Figure 6E).
The expression levels of genes involved in fish immunity were determined. After 30 days of feeding with SL001-supplemented diet or basal diet. In the liver, the expression levels of all detected genes but lysozyme (LSZ) gene showed significant differences between SL001-supplemented group and without SL001-supplemented group (Figure 7A). The gene expression levels of immunoglobulin M (IgM), complement 3 (C3), and interleukin-1β (IL-1β) in SL001-supplemented group elevated 3-, 2.6-, and 2.5-fold respectively (Figure 7A). The expression level of interleukin-8 (IL-8) decreased and maintained 1/3 level of without SL001-supplemented group (Figure 7A). In spleen, the gene expression of IgM, C3, and LSZ were significantly upregulated by SL001 after 30 days of feeding, with 1.5-, 1.4-, and 1.5-fold increases respectively (Figure 7B). By contrast, the gene expression levels of both genes from the interleukin (IL) family were down-regulated (Figure 7B), among which IL-1β gene expression showed a considerable decrease in SL001-supplemented group (Figure 7B). The down-regulation of two pro-inflammatory cytokine (IL-1β, IL-8) indicated the reduction of host’s inflammatory response, and up-regulated expression of IgM, C3, and LSZ suggested that both specific and non-specific immunity of grass carps were improved in SL001-supplemented group.
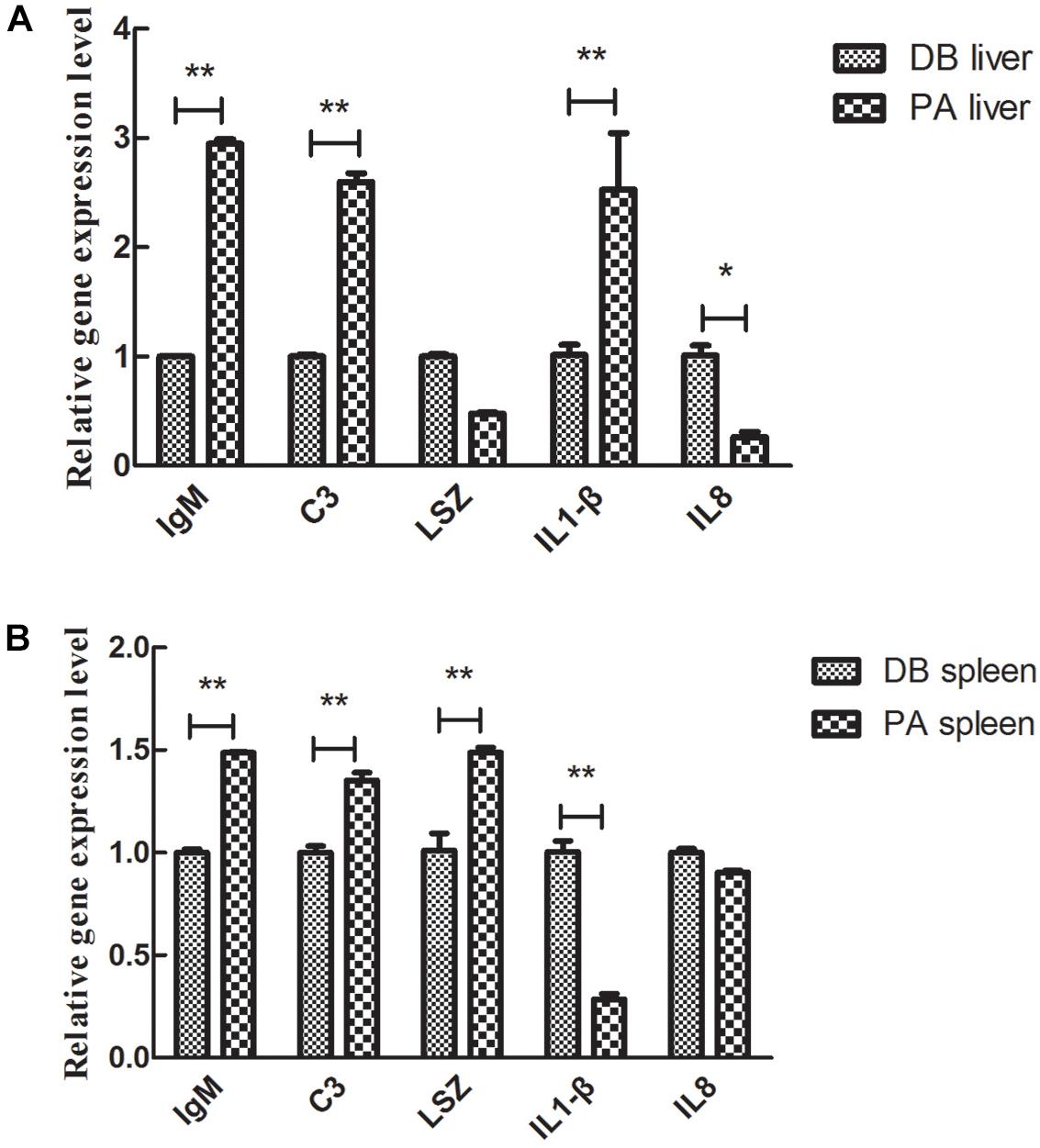
Figure 7. The effect of SL001-feeding on the gene expression of immune-related cytokines in the grass carp. The gene expression levels of immune-related cytokines in (A) liver and (B) spleen were measured by qRT-PCR. Values represented mean ± SEM. Each gene expression level in control group was regarded as 1. ∗P < 0.05, ∗∗P < 0.01 (n = 4). PA, SL001-supplemented group; DB, without SL001 supplemented group.
We further analyzed the influence of 30 days-feeding with SL001-supplemented diet on the expression of immune genes in grass carps post A. hydrophila infection. After feeding grass carps with SL001-supplemented diet for 30 days, the grass carps were challenged with 1 × 106 CFU/mL A. hydrophila, and genes involved in fish immunity were determined. In the liver, the gene expression levels of LSZ, IL-1β, and IL-8 in the SL001-supplemented group were significantly higher than those of the non-supplement group at 6 hpi (Figure 8A). IL-8 gene expression in the SL001-supplemented group at 12 hpi was significantly higher than that of the non-supplement group. However, the gene expression levels of IgM, C3, LSZ, and IL-1β in the SL001-fed group at 12 hpi were prominently down-regulated compared with those of the control group (Figure 8D). In the spleen, the C3 gene expression levels in the SL001-supplemented grass carps exhibited notable decrease at 6 hpi, and then increased remarkably at 12 hpi (Figures 8B,E). By contrast, the gene expression levels of LSZ, IL-1β, and IL-8 in the SL001-fed grass carps were significantly higher at 6 hpi and became considerably lower at 12 hpi (Figures 8B,E). In addition, the expression of IgM in the SL001-supplemented group was evidently down-regulated at 12 hpi (Figure 8E). In the head-kidney, the gene expression levels of C3 and IL-1β in the SL001-fed grass carps at 6 and 12 hpi were higher than those of the non-SL001-fed grass carps (Figures 8C,F). LSZ gene expression in SL001-fed grass carps was notably upregulated at 6 hpi and then significantly down-regulated at 12 hpi (Figures 8C,F). The gene expression levels of IgM in the SL001-fed grass carps were evidently lower at 12 hpi, compared to that of the control grass carps (Figure 8F). In the early stage of infection (6 hpi), pro-inflammatory cytokines (IL1β, IL8) and LSZ were significantly up-regulated in the immune organ of grass carp fed with SL001-diets, which was beneficial to the body against pathogens. In the late infected stage (12 hpi), the higher expression level of C3 indicated that could eliminate pathogens, because of SL001 stimulating the body to improve its immunity.
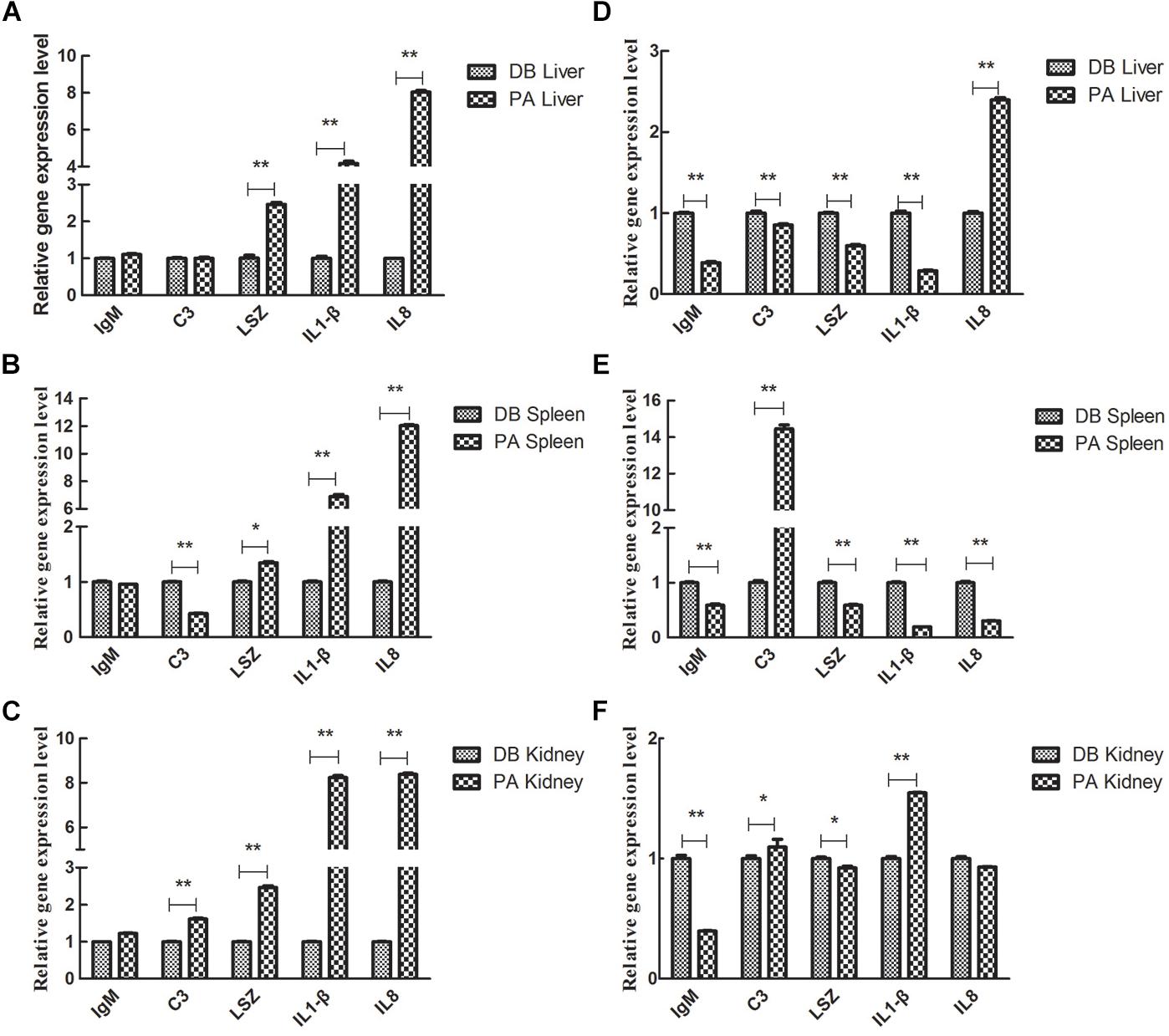
Figure 8. The relative gene expression level of immune-related cytokines in grass carps in challenge test. The grass carp were fed with or without SL001-supplemented diet for 30 days and then infected with A. hydrophila. The gene expression levels of immune-related cytokines in liver (A,D) and spleen (B,E) and head-kidney (C,F) were analyzed (A–C) were at 6 dpi (D–F) were 12 dpi. Values represented mean ± SEM. Each gene expression level in control group was regarded as 1. ∗P < 0.05, ∗∗P < 0.01 (n = 4). PA, SL001-supplemented group; DB, without SL001 supplemented group.
Besides, alkaline phosphatase (AKP) activity in SL001-supplemented groups was significantly increased compared with the control group after 30 dpf (P < 0.05). The activity of ACP was also increased by SL001 supplemented diets albeit not statistically significant (Figure 9).
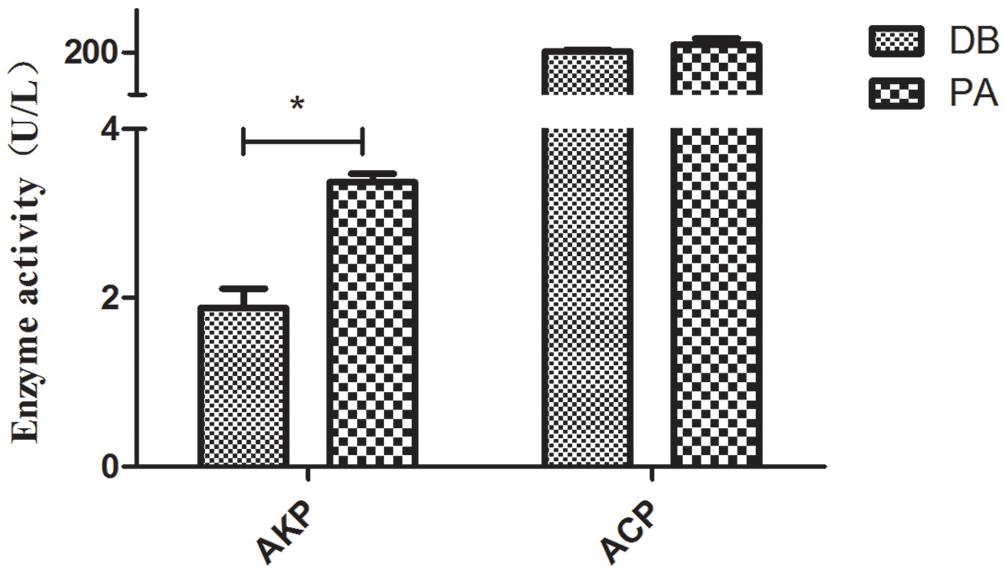
Figure 9. The enzyme activity of non-specific immunological factors from serum of grass carp. Values represented mean ± SEM. ∗P < 0.05, ∗∗P < 0.01 (n = 3). PA, SL001-supplemented group; DB, without SL001 supplemented group.
The whole genome of P. pentosaceus strain SL001 was sequenced using third-generation DNA sequencing, which generated 1,842,476 bp longest contig and 76,699 bp shortest contig, resulting in a genome assembly with total size of 1,917,175 bp (GenBank accession no. CP039378 and CP039379) and G+C content 37.4%. 1,905 coding DNA sequences (CDSs), 72 RNA (including 57 tRNAs and 15 rRNAs), and 22 CRISPRs were obtained by the method of NCBI prokaryotic genome annotation pipeline. Online software RAST was used to annotate the genome, the results of which showed that 41% of the annotated CDS were assorted to the subsystem (Figure 10A). Among the CDS, protein metabolism was the most-enriched metabolic category. In addition, a full-genome comparison analysis of P. pentosaceus SL001 with already-sequenced P. pentosaceus strains showed significant differences for carbohydrates and DNA metabolism (in genes 10–59, 118–181, 348–398, 608–633, 813–860, and 1794-1812) (Figure 10B).
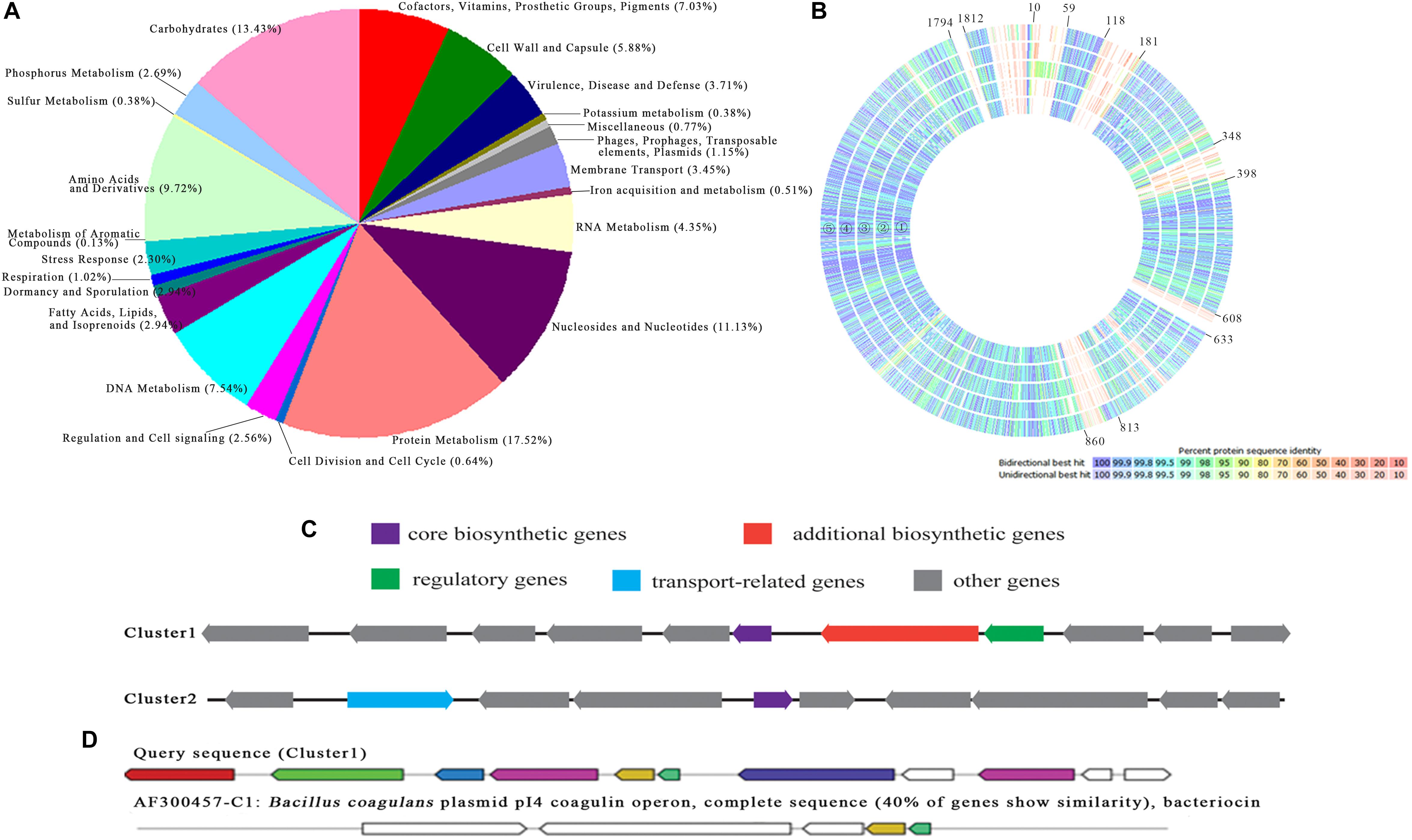
Figure 10. The annotation and comparison of P. pentosaceus SL001 genome. (A) The distribution of predicted CDSs of SL001 in different categories of metabolic function. (B) A full genome comparison analysis of P. pentosaceus SL001 with P. pentosaceus strains.  P. pentosaceus wikim20;
P. pentosaceus wikim20;  P. pentosaceus SL4;
P. pentosaceus SL4;  P. pentosaceus SRCM100892;
P. pentosaceus SRCM100892;  P. pentosaceus KCCM 40703;
P. pentosaceus KCCM 40703;  P. pentosaceus ATCC 25745. (C) Bacteriocin synthesis gene clusters identified in the genome of P. pentosaceus SL001. (D) The homology analysis of the gene cluster 1 in genome of SL001 and coagulin biosynthetic gene cluster from B. coagulans.
P. pentosaceus ATCC 25745. (C) Bacteriocin synthesis gene clusters identified in the genome of P. pentosaceus SL001. (D) The homology analysis of the gene cluster 1 in genome of SL001 and coagulin biosynthetic gene cluster from B. coagulans.
The SL001 genome includes numerous genes involved in organic acid synthesis [e.g., garR (ORF540), pgk (ORF1671), glxk (ORF1746), alsD (ORF490), and ilvB (ORF491)], as well as superoxide dismutase [SOD, e.g., perR (ORF558) and npx (ORF643, ORF1158, ORF1182)], and digestive enzymes [e.g., pepE (ORF865, ORF1344, ORF1780) and IV86 (ORF1278)]. In addition, two bacteriocin synthesis gene clusters were present in SL001 genome (Figure 10C). DNA sequence of cluster 1 (833557 to 842637) shares 40% identity with coagulin biosynthetic gene cluster from Bacillus coagulans plasmid pI4 (Figure 10D), while cluster 2 (853776 to 863958) was an unknown bacteriocin that possibility synthesize new antimicrobial compound (Figure 10C).
P. pentosaceus which can antagonize against several important pathogens (Listeria, Enterococcus faecium, Pseudomonas aeruginosa, and Klebsiella pneumoniae) has been applied in the food industry to control foodborne pathogenic bacteria (Bajpai et al., 2016; Meira et al., 2017; Sriphochanart and Skolpap, 2018), as well as probiotic in animals and humans (Todorov and Dicks, 2009; Bajpai et al., 2016; Cavicchioli et al., 2017). Although P. pentosaceus had a great potential as a probiotic in marine fishes, its probiotics property in freshwater fish has seldom been touched (Xing et al., 2013; Huang et al., 2014). In this study, a new P. pentosaceus strain SL001 was isolated from soil sample of Dadonghai beach in Hainan Province. SL001 exhibited broad-spectrum antibacterial activity against fish pathogens. qRT-PCR analysis demonstrated that feeding with SL001 could not only protect grass carps from A. hydrophila infection, but also promote growth rate. In SL001-supplemented group, more mucus-secreting goblet cells and longer intestinal villi were observed and up-regulated expression of insulin-like growth factor (IGF-1 and IGF-2) and down-regulated expression of myostatin (MSTN-1 and MSTN-2). Relative mRNA expression levels of immune-related genes in liver, spleen and head kidney were analyzed. Genome sequencing of SL001 has been performed to reveal its probiotics properties.
Fish probiotics with good colonization in the gut are an important element in growth and immune regulation (Hai, 2015; Lazado et al., 2015). Here, we demonstrated that SL001 could not only stably colonize in the intestine of grass carps but also prominently affect gut microbiota structure. The composition of gut microbiota was tightly associated with the growth of grass carps, the higher proportion relative abundance of Firmicutes over Bacteroidetes the faster growth of fish (Li et al., 2013). The relative abundance of Firmicutes in SL001-supplemented group was around five times higher than that in control group, which resulted in the increased proportion relative abundance of Firmicutes over Bacteroidetes. The influence of SL001 on the gut microbiota structure of grass carps could be responsible for the increased growth rate. Intestine goblet cells are responsible for the production of the protective mucus layers by synthesizing and secreting mucins. Goblet cells are polarized mucus-secreting cells, which could serve as a source of several digestive enzymes, including lipase, amylase, and trypsin (O’Neill et al., 2011). Efficient digestion and absorption of nutrients by the intestine require a very large apical surface area, a feature that is enhanced by the presence of villi (Freddo et al., 2016). We inferred that elevated mucus-secreting goblet cells and elongated intestinal villi in the SL001-fed group were also key factors for increased growth rate of grass carps.
Commensal probiotics can not only promoter growth of host but also provide protection from pathogenic bacteria, by creating inhibitory compounds, competing for adhesion sites or modulating immune responses (Perez et al., 2010). It was reported that P. fluorescens strain AH2 was able to inhibit the growth of V. anguillarum and increased the survival rate of infected fish (Gram et al., 1999). Bacillus P64 showed both probiotic and immuno-stimulatory features, which could promote growth and improve immune factors activity of host (Gullian et al., 2004). According to previous reports (Isolauri et al., 2002; Ramesh et al., 2017; Meng et al., 2019), IgM, a earliest immune molecule, was produced in fish-specific immune response. C3, LSZ, IL1β, and IL-8 played an important role in fish infection prevention and inflammatory response, which could quickly counterwork against invading pathogens and participate in non-specific immunity of fish. In our study, up-regulated expression of IgM, C3, and LSZ encoding genes and down-regulated expression of pro-inflammatory cytokines (IL-1β and IL-8) in SL001-supplemented group were detected. We speculated that SL001 which could produce prebiotics (exopolysaccharides, SOD, etc.) caused the downregulation of two pro-inflammatory cytokine genes and activated the host’s immune system, so that the significant upregulation of LSZ and C3 (involved in non-specific immunity) and the IgM (involved in specific immunity). Previous studies also showed that probiotic Bacillus aerophilus KADR3 significantly enhanced the levels of serum lysozyme and serum IgM and elevated the activity of the complement pathway, while P. pentosaceus, L. plantarum, and L. brevis reduced the expression levels of IL-1β and IL-8 (Isolauri et al., 2002; Huang et al., 2014; Ramesh et al., 2017). In addition, ACP localized within lysosomes, is a marker for determining whether the macrophages are activated (Song et al., 2006). AKP is an extracellular enzyme, which could hydrolyzes phosphate group within various organic compounds like proteins, nucleic acid (El-Ebiarie, 2012). ACP and AKP as important markers were usually used to evaluate the function of probiotic bacteria in aquaculture. Yi reported that the levels of ACP and AKP of Carassius auratus fed with B. velezensis JW supplemented diets were higher compared to the control group (Yi et al., 2018), which was verified in our study. Besides, the lamina propria in control group was wider than that in SL001-supplemented group. Vasanth et al. (2015) reported that the width of lamina propria could indirectly reflect the host’s health, in which a smaller width indicated no onset of inflammation and reflected healthier host. After challenged against A. hydrophila, LSZ was significantly up-regulated and then down-regulated in all three immune organs, which are consistent with Zhang’s results (Zhang et al., 2018). IL-1β and IL-8 are an immune-associated cytokines that play important roles in innate immune responses (Harada et al., 1994). Feeding grass carps with SL001-supplemenged diet resulted in a significant increase in expression of IL1β and IL8 at 6 h after infection.
By analyzing the genome of SL001, we obtained some clues about its probiotics properties. Genes related to organic acids, bacteriocins, SOD, and digestive enzymes which were thought to be linked to disease-resistance, immune response, and growth were identified in the genome of SL001. It was well-known that organic acid and bacteriocin could inhibit the growth of pathogenic bacteria and regulated the micro-ecological balance of the gastrointestinal and reproductive tract of animals (Muhialdin et al., 2018). Previous report demonstrated that the SOD enhanced humoral and cellular immunity in animals and improved disease resistance (Picchietti et al., 2009). In addition, the digestive enzymes from SL001 secretion promoted the digestion and absorption of nutrients by fish, thereby promoting fish growth (Suzer et al., 2008). Besides, two putative gene clusters responsible for bacteriocins synthesis were present in SL001 genome. One gene cluster was predicated to produce coagulin, which was reported to exhibite antimicrobial activity against pathogenic bacteria (Hyronimus et al., 1998). Coagulin, a new member of the pediocin-like family protein, was firstly discovered in B. coagulans. Afterward, coagulin was also identified in P. pentosaceus MZF16 (Zommiti et al., 2018). The difference between coagulin and pediocin was only a single amino acid residue at their C terminus (Le Marrec et al., 2000).
Our work showed that feeding with SL001-supplemented diet can promote growth on grass carps and enhance its immune. We envisioned that this can be expanded to other freshwater fish aquaculture. It would undoubtedly provide an important strain for freshwater fish aquaculture and broaden the application of P. pentosaceus. Encapsulation of P. pentosaceus Li05 in an alginate-gelatin microgels significantly enhanced their viability under different conditions (Yao et al., 2018). Formulated and encapsulated probiotics can improve their survival and colonization and avoid loss of probiotics owing to the reduced probiotic activity during food storage and gastrointestinal transit during feeding. We will then focus on the microecological preparation P. pentosaceus for its practical application in future.
This manuscript contains previously unpublished data. The name of the repository and accession number are not available.
LG and LP performed the bacterial strain isolation and identification. LC, HH, and LY performed the analysis of antimicrobial activity and determination of non-specific immunological factors activity. DL, LC, HH, and XD performed the cellular culture, challenge test, and weight analysis. YL and YS performed the collection of gut, liver, spleen, kidney, and muscle. GY performed the H&E staining. LG, HH, DL, and SH performed the Total RNA isolation and qRT-PCR analysis. LG, YZ, and LX analyzed the data of high-throughput sequence and genome. LG, HH, SH, and LX designed the study and wrote the draft of manuscript. TK, SH, and LX corrected the manuscript. All authors discussed the results and approved the final manuscript.
This present work was supported by the National Natural Science Foundation of China (Grant Nos. 31570125 and 31770106), the National Basic Research Program of China (“973” program; Grant No. 2012CB722301), and the “Hunan Province Biological Development Engineering and New Product Development Collaborative Innovation Center” project (Grant No. 20134486).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2019.01384/full#supplementary-material
Akhter, N., Wu, B., Memon, A. M., and Mohsin, M. (2015). Probiotics and prebiotics associated with aquaculture: a review. Fish Shellfish Immunol. 45, 733–741. doi: 10.1016/j.fsi.2015.05.038
Alonso, S., Carmen Castro, M., Berdasco, M., de la Banda, I. G., Moreno-Ventas, X., and de Rojas, A. H. (2018). Isolation and partial characterization of lactic acid bacteria from the gut microbiota of marine fishes for potential application as probiotics in aquaculture. Probiotics Antimicrob. Proteins 11, 569–579. doi: 10.1007/s12602-018-9439-2
Amato, K. R., Yeoman, C. J., Kent, A., Righini, N., Carbonero, F., Estrada, A., et al. (2013). Habitat degradation impacts black howler monkey (Alouatta pigra) gastrointestinal microbiomes. ISME J. 7, 1344–1353. doi: 10.1038/ismej.2013.16
Athanassopoulou, F., Billinis, C., and Prapas, T. (2004). Important disease conditions of newly cultured species in intensive freshwater farms in Greece: first incidence of nodavirus infection in Acipenser sp. Dis. Aquat. Organ. 60, 247–252. doi: 10.3354/dao060247
Bajpai, V. K., Han, J. H., Rather, I. A., Park, C., Lim, J., Paek, W. K., et al. (2016). Characterization and antibacterial potential of lactic acid bacterium Pediococcus pentosaceus 4I1 isolated from freshwater fish zacco koreanus. Front. Microbiol. 7:2037. doi: 10.3389/fmicb.2016.02037
Balcazar, J. L., de Blas, I., Ruiz-Zarzuela, I., Cunningham, D., Vendrell, D., and Muzquiz, J. L. (2006). The role of probiotics in aquaculture. Vet. Microbiol. 114, 173–186. doi: 10.1016/j.vetmic.2006.01.009
Banerjee, G., and Ray, A. K. (2017). The advancement of probiotics research and its application in fish farming industries. Res. Vet. Sci. 115, 66–77. doi: 10.1016/j.rvsc.2017.01.016
Cao, H., He, S., Wei, R., Diong, M., and Lu, L. (2011). Bacillus amyloliquefaciens G1: a potential antagonistic bacterium against eel-pathogenic Aeromonas hydrophila. Evid. Based Complement. Alternat. Med. 2011:824104. doi: 10.1155/2011/824104
Caporaso, J. G., Kuczynski, J., Stombaugh, J., Bittinger, K., Bushman, F. D., Costello, E. K., et al. (2010). QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 7, 335–336. doi: 10.1038/nmeth.f.303
Cavicchioli, V. Q., Camargo, A. C., Todorov, S. D., and Nero, L. A. (2017). Novel bacteriocinogenic Enterococcus hirae and Pediococcus pentosaceus strains with antilisterial activity isolated from Brazilian artisanal cheese. J. Dairy Sci. 100, 2526–2535. doi: 10.3168/jds.2016-12049
Chandrarathna, H. P. S. U., Nikapitiya, C., Dananjaya, S. H. S., Wijerathne, C. U. B., Wimalasena, S. H. M. P., Kwon, H. J., et al. (2018). Outcome of co-infection with opportunistic and multidrug resistant Aeromonas hydrophila and Aeromonas veronii in zebrafish: identification, characterization, pathogenicity and immune responses. Fish Shellfish Immunol. 80, 573–581. doi: 10.1016/j.fsi.2018.06.049
Chu, P., He, L., Li, Y., Huang, R., Liao, L., Li, Y., et al. (2018). Molecular cloning and functional characterisation of NLRX1 in grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol. 81, 276–283. doi: 10.1016/j.fsi.2018.07.031
DiMaggio, M. A., Ohs, C. L., Broach, J. S., and Sink, T. D. (2014). Effects of stocking density on growth, survival, and stress physiology of Pigfish. North Am. J. Aquacult. 76, 201–210. doi: 10.1080/15222055.2013.867000
Edgar, R. C. (2010). Search and clustering orders of magnitude faster than BLAST. Bioinformatics 26, 2460–2461. doi: 10.1093/bioinformatics/btq461
El-Ebiarie, A. S. (2012). Studying the activity of alkaline phosphatase, digestive proteases and some carbohydrate enzymes in the mid-gut of the third instar larvae of Gasterophilus intestinalis and comparing some of them with pupae. Life Sci. J. 9, 2076–2085.
Falcinelli, S., Picchietti, S., Rodiles, A., Cossignani, L., Merrifield, D. L., Taddei, A. R., et al. (2015). Lactobacillus rhamnosus lowers zebrafish lipid content by changing gut microbiota and host transcription of genes involved in lipid metabolism. Sci. Rep. 5:9336. doi: 10.1038/srep09336
Freddo, A. M., Shoffner, S. K., Shao, Y., Taniguchi, K., Grosse, A. S., Guysinger, M. N., et al. (2016). Coordination of signaling and tissue mechanics during morphogenesis of murine intestinal villi: a role for mitotic cell rounding. Integr. Biol. 8, 918–928. doi: 10.1039/c6ib00046k
Ghiasi, M., Binaii, M., Naghavi, A., Rostami, H. K., Nori, H., and Amerizadeh, A. (2018). Inclusion of Pediococcus acidilactici as probiotic candidate in diets for beluga (Huso huso) modifies biochemical parameters and improves immune functions. Fish Physiol. Biochem. 44, 1099–1107. doi: 10.1007/s10695-018-0497-x
Gram, L., Melchiorsen, J., Spanggaard, B., Huber, I., and Nielsen, T. F. (1999). Inhibition of Vibrio anguillarum by Pseudomonas fluorescens AH2, a possible probiotic treatment of fish. Appl. Environ. Microbiol. 65, 969–973.
Gullian, M., Thompson, F., and Rodriguez, J. (2004). Selection of probiotic bacteria and study of their immunostimulatory effect in Penaeus vannamei. Aquaculture 233, 1–14. doi: 10.1016/j.aquaculture.2003.09.013
Hai, N. V. (2015). The use of probiotics in aquaculture. J. Appl. Microbiol. 119, 917–935. doi: 10.1111/jam.12886
Harada, A., Sekido, N., Akahoshi, T., Wada, T., Mukaida, N., and Matsushima, K. (1994). Essential involvement of interleukin-8 (IL-8) in acute inflammation. J. Leukoc. Biol. 56, 559–564. doi: 10.1002/jlb.56.5.559
Huang, J. B., Wu, Y. C., and Chi, S. C. (2014). Dietary supplementation of Pediococcus pentosaceus enhances innate immunity, physiological health and resistance to Vibrio anguillarum in orange-spotted grouper (Epinephelus coioides). Fish Shellfish Immunol. 39, 196–205. doi: 10.1016/j.fsi.2014.05.003
Huang, Z. T., Wan, R., Song, X. F., Liu, Y., Hallerman, E., Dong, D. P., et al. (2016). Metagenomic analysis shows diverse, distinct bacterial communities in biofilters among different marine recirculating aquaculture systems. Aquacult. Int. 24, 1393–1408. doi: 10.1007/s10499-016-9997-9
Hyronimus, B., Le Marrec, C., and Urdaci, M. C. (1998). Coagulin, a bacteriocin-like inhibitory substance produced by Bacillus coagulans I4. J. Appl. Microbiol. 85, 42–50. doi: 10.1046/j.1365-2672.1998.00466.x
Isolauri, E., Kirjavainen, P. V., and Salminen, S. (2002). Probiotics: a role in the treatment of intestinal infection and inflammation? Gut 50(Suppl. 3), III54–III59.
Koren, S., Walenz, B. P., Berlin, K., Miller, J. R., Bergman, N. H., and Phillippy, A. M. (2017). Canu: scalable and accurate long-read assembly via adaptive k-mer weighting and repeat separation. Genome Res. 27, 722–736. doi: 10.1101/gr.215087.116
Lazado, C. C., Caipang, C. M., and Estante, E. G. (2015). Prospects of host-associated microorganisms in fish and penaeids as probiotics with immunomodulatory functions. Fish Shellfish Immunol. 45, 2–12. doi: 10.1016/j.fsi.2015.02.023
Le Marrec, C., Hyronimus, B., Bressollier, P., Verneuil, B., and Urdaci, M. C. (2000). Biochemical and genetic characterization of coagulin, a new antilisterial bacteriocin in the pediocin family of bacteriocins, produced by Bacillus coagulans I(4). Appl. Environ. Microbiol. 66, 5213–5220. doi: 10.1128/aem.66.12.5213-5220.2000
Li, X. M., Yan, Q. Y., Xie, S. Q., Hu, W., Yu, Y. H., and Hu, Z. H. (2013). Gut microbiota contributes to the growth of fast-growing transgenic common carp (Cyprinus carpio L.). PLoS One 8:e64577. doi: 10.1371/journal.pone.0064577
Lin, W., Li, L., Chen, J., Li, D., Hou, J., Guo, H., et al. (2018). Long-term crowding stress causes compromised nonspecific immunity and increases apoptosis of spleen in grass carp (Ctenopharyngodon idella). Fish Shellfish Immunol. 80, 540–545. doi: 10.1016/j.fsi.2018.06.050
Matsuda, Y., Imamura, R., and Takahara, S. (2017). Evaluation of antigen-specific IgM and IgG production during an in vitro peripheral blood mononuclear cell culture assay. Front. Immunol. 8:794. doi: 10.3389/fimmu.2017.00794
Meira, N. V. B., Holley, R. A., Bordin, K., Macedo, R. E. F., and Luciano, F. B. (2017). Combination of essential oil compounds and phenolic acids against Escherichia coli O157:H7 in vitro and in dry-fermented sausage production. Int. J. Food Microbiol. 260, 59–64. doi: 10.1016/j.ijfoodmicro.2017.08.010
Meng, X., Shen, Y., Wang, S., Xu, X., Dang, Y., Zhang, M., et al. (2019). Complement component 3 (C3): an important role in grass carp (Ctenopharyngodon idella) experimentally exposed to Aeromonas hydrophila. Fish Shellfish Immunol. 88, 189–197. doi: 10.1016/j.fsi.2019.02.061
Muhialdin, B. J., Hassan, Z., and Saari, N. (2018). In vitro antifungal activity of lactic acid bacteria low molecular peptides against spoilage fungi of bakery products. Ann. Microbiol. 68, 557–567. doi: 10.1007/s13213-018-1363-x
O’Neill, M. J., Bourre, L., Melgar, S., and O’Driscoll, C. M. (2011). Intestinal delivery of non-viral gene therapeutics: physiological barriers and preclinical models. Drug Discov. Today 16, 203–218. doi: 10.1016/j.drudis.2011.01.003
Overbeek, R., Olson, R., Pusch, G. D., Olsen, G. J., Davis, J. J., Disz, T., et al. (2014). The SEED and the rapid annotation of microbial genomes using subsystems technology (RAST). Nucleic Acids Res. 42, D206–D214. doi: 10.1093/nar/gkt1226
Palazzotto, E., Renzone, G., Fontana, P., Botta, L., Scaloni, A., Puglia, A. M., et al. (2015). Tryptophan promotes morphological and physiological differentiation in Streptomyces coelicolor. Appl. Microbiol. Biotechnol. 99, 10177–10189. doi: 10.1007/s00253-015-7012-4
Perez, T., Balcazar, J. L., Ruiz-Zarzuela, I., Halaihel, N., Vendrell, D., de Blas, I., et al. (2010). Host-microbiota interactions within the fish intestinal ecosystem. Mucosal Immunol. 3, 355–360. doi: 10.1038/mi.2010.12
Perez-Ramos, A., Mohedano, M. L., Pardo, M. A., and Lopez, P. (2018). Beta-glucan-producing Pediococcus parvulus 2.6: test of probiotic and immunomodulatory properties in zebrafish models. Front. Microbiol. 9:1684. doi: 10.3389/fmicb.2018.01684
Picchietti, S., Fausto, A. M., Randelli, E., Carnevali, O., Taddei, A. R., Buonocore, F., et al. (2009). Early treatment with Lactobacillus delbrueckii strain induces an increase in intestinal T-cells and granulocytes and modulates immune-related genes of larval Dicentrarchus labrax (L.). Fish Shellfish Immunol. 26, 368–376. doi: 10.1016/j.fsi.2008.10.008
Quast, C., Pruesse, E., Yilmaz, P., Gerken, J., Schweer, T., Yarza, P., et al. (2013). The SILVA ribosomal RNA gene database project: improved data processing and web-based tools. Nucleic Acids Res. 41, D590–D596. doi: 10.1093/nar/gks1219
Ramesh, D., Souissi, S., and Ahamed, T. S. (2017). Effects of the potential probiotics Bacillus aerophilus KADR3 in inducing immunity and disease resistance in Labeo rohita. Fish Shellfish Immunol. 70, 408–415. doi: 10.1016/j.fsi.2017.09.037
Schloss, P. D., Gevers, D., and Westcott, S. L. (2011). Reducing the effects of PCR amplification and sequencing artifacts on 16S rRNA-based studies. PLoS One 6:e27310. doi: 10.1371/journal.pone.0027310
Shi, C. M., Zhao, H., Zhai, X. L., Chen, Y. J., and Lin, S. M. (2019). Linseed oil can decrease liver fat deposition and improve antioxidant ability of juvenile largemouth bass, Micropterus salmoides. Fish Physiol. Biochem. doi: 10.1007/s10695-019-00636-3 [Epub ahead of print].
Shukla, R., and Goyal, A. (2014). Probiotic potential of Pediococcus pentosaceus CRAG3: a new isolate from fermented cucumber. Probiotics Antimicrob Proteins 6, 11–21. doi: 10.1007/s12602-013-9149-8
Song, X. H., Zhao, J., Bo, Y. X., Liu, Z. J., Wu, K., and Gong, C. L. (2014). Aeromonas hydrophila induces intestinal inflammation in grass carp (Ctenopharyngodon idella): an experimental model. Aquaculture 434, 171–178. doi: 10.1016/j.aquaculture.2014.08.015
Song, Z. F., Wu, T. X., Cai, L. S., Zhang, L. J., and Zheng, X. D. (2006). Effects of dietary supplementation with clostridium butyricum on the growth performance and humoral immune response in Miichthys miiuy. J. Zhejiang Univ. Sci. B 7, 596–602. doi: 10.1631/jzus.2006.B0596
Sriphochanart, W., and Skolpap, W. (2018). Modeling of starter cultures growth for improved Thai sausage fermentation and cost estimating for sausage preparation and transportation. Food Sci. Nutr. 6, 1479–1491. doi: 10.1002/fsn3.708
Suzer, C., Coban, D., Kamaci, H. O., Saka, S., Firat, K., Otgucuoglu, O., et al. (2008). Lactobacillus spp. bacteria as probiotics in gilthead sea bream (Sparus aurata, L.) larvae: effects on growth performance and digestive enzyme activities. Aquaculture 280, 140–145. doi: 10.1016/j.aquaculture.2008.04.020
Tamura, K., Nei, M., and Kumar, S. (2004). Prospects for inferring very large phylogenies by using the neighbor-joining method. Proc. Natl. Acad. Sci. U.S.A. 101, 11030–11035. doi: 10.1073/pnas.0404206101
Tang, Y., Han, L., Chen, X., Xie, M., Kong, W., and Wu, Z. (2018). Dietary supplementation of probiotic Bacillus subtilis affects antioxidant defenses and immune response in grass carp under Aeromonas hydrophila challenge. Probiotics Antimicrob. Proteins 11, 545–558. doi: 10.1007/s12602-018-9409-8
Todorov, S. D., and Dicks, L. M. (2009). Bacteriocin production by Pediococcus pentosaceus isolated from marula (Scerocarya birrea). Int. J. Food Microbiol. 132, 117–126. doi: 10.1016/j.ijfoodmicro.2009.04.010
Vasanth, G., Kiron, V., Kulkarni, A., Dahle, D., Lokesh, J., and Kitani, Y. (2015). A microbial feed additive abates intestinal inflammation in Atlantic Salmon. Front. Immunol. 6:409. doi: 10.3389/fimmu.2015.00409
Walker, B. J., Abeel, T., Shea, T., Priest, M., Abouelliel, A., Sakthikumar, S., et al. (2014). Pilon: an integrated tool for comprehensive microbial variant detection and genome assembly improvement. PLoS One 9:e112963. doi: 10.1371/journal.pone.0112963
Wang, J., Zeng, Y., Wang, S., Liu, H., Zhang, D., Zhang, W., et al. (2018). Swine-derived probiotic Lactobacillus plantarum inhibits growth and adhesion of enterotoxigenic Escherichia coli and mediates host defense. Front. Microbiol. 9:1364. doi: 10.3389/fmicb.2018.01364
Wang, Q., Garrity, G. M., Tiedje, J. M., and Cole, J. R. (2007). Naive bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 73, 5261–5267. doi: 10.1128/Aem.00062-07
Wang, Y., Sheng, H. F., He, Y., Wu, J. Y., Jiang, Y. X., Tam, N. F. Y., et al. (2012). Comparison of the levels of bacterial diversity in freshwater, intertidal wetland, and marine sediments by Uusing millions of Illumina tags. Appl. Environ. Microbiol. 78, 8264–8271. doi: 10.1128/Aem.01821-12
Wu, J. Y., Jiang, X. T., Jiang, Y. X., Lu, S. Y., Zou, F., and Zhou, H. W. (2010). Effects of polymerase, template dilution and cycle number on PCR based 16S rRNA diversity analysis using the deep sequencing method. BMC Microbiol. 10:255. doi: 10.1186/1471-2180-10-255
Xing, C. F., Hu, H. H., Huang, J. B., Fang, H. C., Kai, Y. H., Wu, Y. C., et al. (2013). Diet supplementation of Pediococcus pentosaceus in cobia (Rachycentron canadum) enhances growth rate, respiratory burst and resistance against photobacteriosis. Fish Shellfish Immunol. 35, 1122–1128. doi: 10.1016/j.fsi.2013.07.021
Yao, M. F., Li, B., Ye, H. W., Huang, W. H., Luo, Q. X., Xiao, H., et al. (2018). Enhanced viability of probiotics (Pediococcus pentosaceus Li05) by encapsulation in microgels doped with inorganic nanoparticles. Food Hydrocoll. 83, 246–252. doi: 10.1016/j.foodhyd.2018.05.024
Yi, Y., Zhang, Z., Zhao, F., Liu, H., Yu, L., Zha, J., et al. (2018). Probiotic potential of Bacillus velezensis JW: antimicrobial activity against fish pathogenic bacteria and immune enhancement effects on Carassius auratus. Fish Shellfish Immunol. 78, 322–330. doi: 10.1016/j.fsi.2018.04.055
Zhang, C., Zhang, J., Liu, M., and Huang, M. (2018). Molecular cloning, expression and antibacterial activity of goose-type lysozyme gene in Microptenus salmoides. Fish Shellfish Immunol. 82, 9–16. doi: 10.1016/j.fsi.2018.07.058
Zhou, S. Y., Liu, Y. M., Zhang, Q. Z., Fu, Y. W., and Lin, D. J. (2018). Evaluation of an antiparasitic compound extracted from Polygonum cuspidatum against Ichthyophthirius multifiliis in grass carp. Vet. Parasitol. 253, 22–25. doi: 10.1016/j.vetpar.2018.02.005
Zommiti, M., Bouffartigues, E., Maillot, O., Barreau, M., Szunerits, S., Sebei, K., et al. (2018). In vitro assessment of the probiotic properties and bacteriocinogenic potential of Pediococcus pentosaceus MZF16 isolated from artisanal tunisian meat ”Dried Ossban. Front. Microbiol. 9:2607. doi: 10.3389/Fmicb.2018.02607
Keywords: Pediococcus pentosaceus, antibacterial activity, grass carps, gut microbiota, fish immunity, growth rate
Citation: Gong L, He H, Li D, Cao L, Khan TA, Li Y, Pan L, Yan L, Ding X, Sun Y, Zhang Y, Yi G, Hu S and Xia L (2019) A New Isolate of Pediococcus pentosaceus (SL001) With Antibacterial Activity Against Fish Pathogens and Potency in Facilitating the Immunity and Growth Performance of Grass Carps. Front. Microbiol. 10:1384. doi: 10.3389/fmicb.2019.01384
Received: 24 February 2019; Accepted: 03 June 2019;
Published: 27 June 2019.
Edited by:
Jie Yin, Institute of Subtropical Agriculture (CAS), ChinaReviewed by:
Yun-Zhang Sun, Jimei University, ChinaCopyright © 2019 Gong, He, Li, Cao, Khan, Li, Pan, Yan, Ding, Sun, Zhang, Yi, Hu and Xia. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Shengbiao Hu, c2hlbmdiaWFvaHVAaHVubnUuZWR1LmNu; Liqiu Xia, eGlhbHFAaHVubnUuZWR1LmNu
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.