- 1Laboratory of Food Biotechnology, Institute of Food Nutrition and Health, Department of Health Science and Technology, ETH Zurich, Zurich, Switzerland
- 2Department of Pathology, Radboud Institute for Molecular Life Sciences, Radboudumc, Nijmegen, Netherlands
The Streptococcus bovis/Streptococcus equinus complex (SBSEC) comprises several species inhabiting the animal and human gastrointestinal tract (GIT). They match the pathobiont description, are potential zoonotic agents and technological organisms in fermented foods. SBSEC members are associated with multiple diseases in humans and animals including ruminal acidosis, infective endocarditis (IE) and colorectal cancer (CRC). Therefore, this review aims to re-evaluate adhesion and colonization abilities of SBSEC members of animal, human and food origin paired with genomic and functional host-microbe interaction data on their road from colonization to infection. SBSEC seem to be a marginal population during GIT symbiosis that can proliferate as opportunistic pathogens. Risk factors for human colonization are considered living in rural areas and animal-feces contact. Niche adaptation plays a pivotal role where Streptococcus gallolyticus subsp. gallolyticus (SGG) retained the ability to proliferate in various environments. Other SBSEC members have undergone genome reduction and niche-specific gene gain to yield important commensal, pathobiont and technological species. Selective colonization of CRC tissue is suggested for SGG, possibly related to increased adhesion to cancerous cell types featuring enhanced collagen IV accessibility. SGG can colonize, proliferate and may shape the tumor microenvironment to their benefit by tumor promotion upon initial neoplasia development. Bacteria cell surface structures including lipotheichoic acids, capsular polysaccharides and pilus loci (pil1, pil2, and pil3) govern adhesion. Only human blood-derived SGG contain complete pilus loci and other disease-associated surface proteins. Rumen or feces-derived SGG and other SBSEC members lack or harbor mutated pili. Pili also contribute to binding to fibrinogen upon invasion and translocation of cells from the GIT into the blood system, subsequent immune evasion, human contact system activation and collagen-I-binding on damaged heart valves. Only SGG carrying complete pilus loci seem to have highest IE potential in humans with significant links between SGG bacteremia/IE and underlying diseases including CRC. Other SBSEC host-microbe combinations might rely on currently unknown mechanisms. Comparative genome data of blood, commensal and food isolates are limited but required to elucidate the role of pili and other virulence factors, understand pathogenicity mechanisms, host specificity and estimate health risks for animals, humans and food alike.
General Introduction to the Relevance of the Streptococcus bovis/Streptococcus equinus Complex
Streptococcus bovis/Streptococcus equinus complex (SBSEC) bacteria are Gram-positive species that inhabit the gastrointestinal tract (GIT) of animals and humans. Most SBSEC have been described as commensal bacteria, but some cause serious infections such as bacteremia and infective endocarditis (IE) in humans and animals and match the pathobiont description (Chow et al., 2011; Boleij and Tjalsma, 2013; Jans et al., 2015, 2016). They are associated with underlying conditions including occult colorectal cancer (CRC) (Boleij et al., 2011c), which highlights the importance of SBSEC members in public- and animal health alike.
Furthermore, SBSEC members are detected in food products including fermented milk in sub-Saharan Africa, Asia and Southern Europe, fermented fish in Asia and fermented plants in sub-Saharan Africa and Latin America suggesting a range of habitats and adaptability to different environments for these bacteria (Jans et al., 2015, 2017). Recent advances in phenotypic and molecular technologies provide more detailed classification abilities at various levels from species to sequence type. This advanced classification scheme helps to elucidate the SBSEC population structure, disease associations, transmission routes and host specificity (Dumke et al., 2014; Shibata et al., 2014; Jans et al., 2016). It is still unclear how SBSEC members establish from commensal organisms to pathogens, particularly relating to survival, colonization, adhesion, invasion and interaction with the host immune system. Furthermore, the causality of SBSEC in CRC is not yet proven which leaves the bacterial-driver-passenger model as important theory to describe the potential mechanisms of host-microbe interaction (Tjalsma et al., 2012).
Therefore, this narrative review aims to provide a comprehensive overview for the following questions in relation to SBSEC members on their road to infection regarding prevalence, transmission, niche colonization and mechanisms for adhesion, invasion and infection establishment within the human-animal-food system (Figure 1):
(1) What abilities help SBSEC members to colonize different body sites or ecological niches and facilitate transmission?
(2) Which factors determine SBSEC to evolve from commensal to pathogen?
(3) What is the prevalence of SBSEC members in different habitats in relation to clinical manifestations and infections?
(4) Which genetic factors are known to encode for these abilities and can be linked to experimental studies?
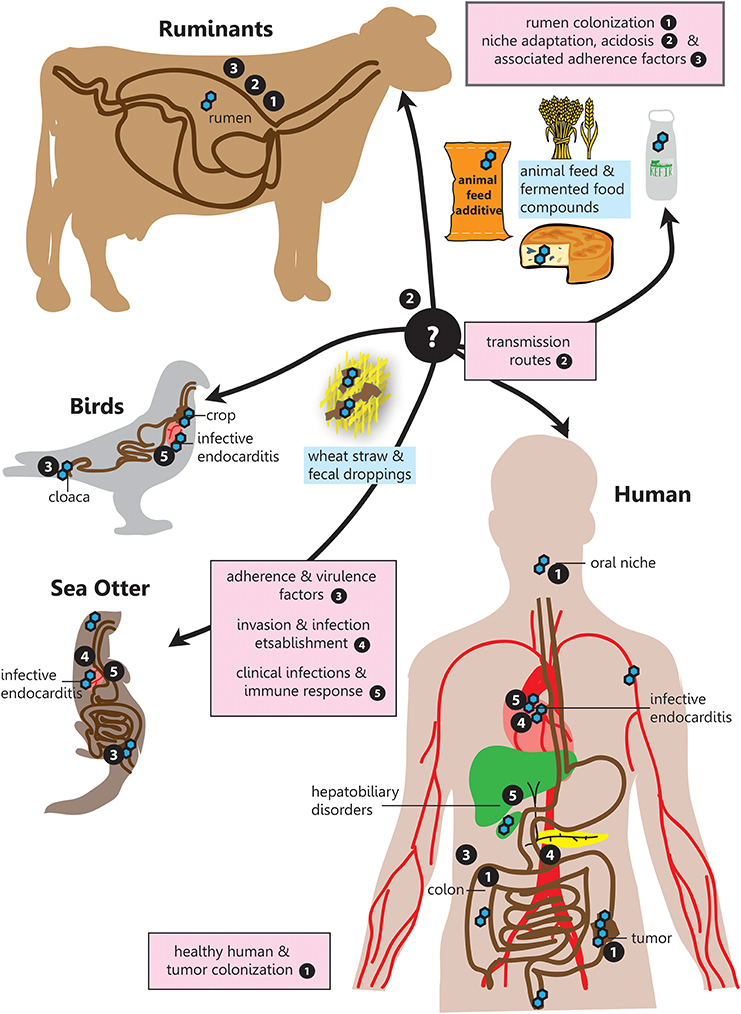
Figure 1. SBSEC - the road to infection. Graphical overview of the different niches inhabited by SBSEC members as well as relevant aspects of host colonization, adherence, invasion and infection covered in the corresponding chapters indicated by bullet point numbers. 1. Prevalence and colonization of SBSEC in animals and humans, 2. Transmission and niche adaptation of SBSEC members, 3. Mechanisms and virulence factors responsible for adhesion and host colonization by SBSEC members, 4. Invasion and infection establishment, 5. Clinical infections and host-immune response due to SBSEC in animals and humans.
Basic Taxonomy and Identification of the SBSEC
SBSEC members are group D streptococci, although the Lancefield group D antiserum reaction is not ubiquitous (Beck et al., 2008). The SBSEC is comprised of several different species, which in this review will be used according to following nomenclature: Streptococcus equinus (SE), Streptococcus infantarius subsp. infantarius (SII), Streptococcus lutetiensis (SL), Streptococcus alactolyticus and three subspecies of the clade Streptococcus gallolyticus, namely gallolyticus (SGG), macedonicus (SGM) and pasteurianus (SGP). The taxonomic assignment of SL as separate species is not fully agreed upon and also referred to as Streptococcus infantarius subsp. coli (Dekker and Lau, 2016). When no distinction was made between subspecies, the old nomenclature Streptococcus bovis (SB) was used (Schlegel et al., 2000, 2003; Poyart et al., 2002; Jans et al., 2015). Biotype differentiation is based on the ability to produce acid from mannitol (biotype I = SGG) or not (biotype II = SE, SGP, SL, and SII) (Schlegel et al., 2003; Jans et al., 2015). For full phenotype descriptions, we refer to Bergey's Manual and their implementations in API and VITEK identification approaches (Whiley and Hardie, 2009).
Alternative identification approaches utilize Matrix Assisted Laser Desorption/Ionization Time Of Flight Mass Spectrometry (MALDI-TOF MS) to identify and discriminate species of the SBSEC but results in unreliable identification of SGM and a roughly 80% identification rate for SGG, SGP and SL (Hinse et al., 2011a; Ben-Chetrit et al., 2017). Furthermore, DNA-based approaches are widely applied using single gene PCR and qPCR assays on 16S rRNA gene (Jans et al., 2012b), sodA (Poyart et al., 1998), groES/groEL (Chen et al., 2008; Lazarovitch et al., 2013; Sheng et al., 2014), recN and gyrB (Lopes et al., 2014) as well as multi locus sequence typing (Dumke et al., 2014; Shibata et al., 2014; Jans et al., 2016).
Prevalence and Colonization of SBSEC in Animals and Humans
Prevalence and Colonization in Animals
SBSEC are mainly described as colonizers of the rumen, crop and cloaca of animals and colon of humans. SBSEC members have been isolated from the GIT or blood system of birds, companion animals, livestock (ruminants, poultry and pigs), marsupials, aquatic mammals and game (Jans et al., 2015), but prevalence data is limited to birds, cattle and lamb. SGG was found in over 90% of fecal droppings in turkey flocks and reached up to 80% prevalence in pigeon crop and cloaca samples (De Herdt et al., 1994a,b; Schulz et al., 2015). SBSEC members were also isolated from chicken crops, but less frequently and not as predominant bacteria (Baele et al., 2001).
In ruminants, SBSEC members are considered as aerotolerant components of the rumen epithelial surface (epimural) microbiota (Mead and Jones, 1981). SBSEC members are estimated to contribute 106 to 107 cells per milliliter of rumen content (Hudson et al., 2000). Their prevalence in cattle is estimated between 20 and 90% with lowest prevalence at early ages (Jans et al., 2015) while in lambs an early live predominance of SBSEC is suggested (Mueller et al., 1984). Colonization and predominance is likely affected by feed composition as shown for reindeers where high SBSEC colonization correlates with higher quantities of starch-rich feed during the summer months (Orpin et al., 1985). SBSEC members are also supplied directly to young calves and goats as probiotics to support the establishment of an anaerobic rumen microbiota and to benefit from consistent α-amylase activity for feed digestion (Kmet et al., 1993; Kumar et al., 2016). SBSEC members therefore seem to form an integral part of the GIT microbiota of birds whereas ruminants feature age-dependent and possible host-associated SBSEC prevalence.
Prevalence in Healthy Humans
The fecal carriage rate of SBSEC member in humans is varying from five to over 60% (Table 1) (Klein et al., 1977; Noble, 1978; Potter et al., 1998; Abdulamir et al., 2010; Al-Jashamy et al., 2010; Chirouze et al., 2013; Lopes et al., 2014; Boltin et al., 2015; Dumke et al., 2017; Kaindi et al., 2017). This variation might depend on detection techniques and regional differences. Furthermore, most studies were conducted using a hospital-derived population with possible differences in age, sex and underlying diseases that limits extrapolation to the general population. (Huang et al., 2008; Chirouze et al., 2013; Lopes et al., 2014; Dumke et al., 2017; Harris et al., 2017). Oral niche colonization in humans seems to be infrequent, but isolation of SII, SGP, and SE was confirmed from dental plaques and root caries lesions (Sissons et al., 1988; Shen et al., 2005; Arul and Palanivelu, 2014).
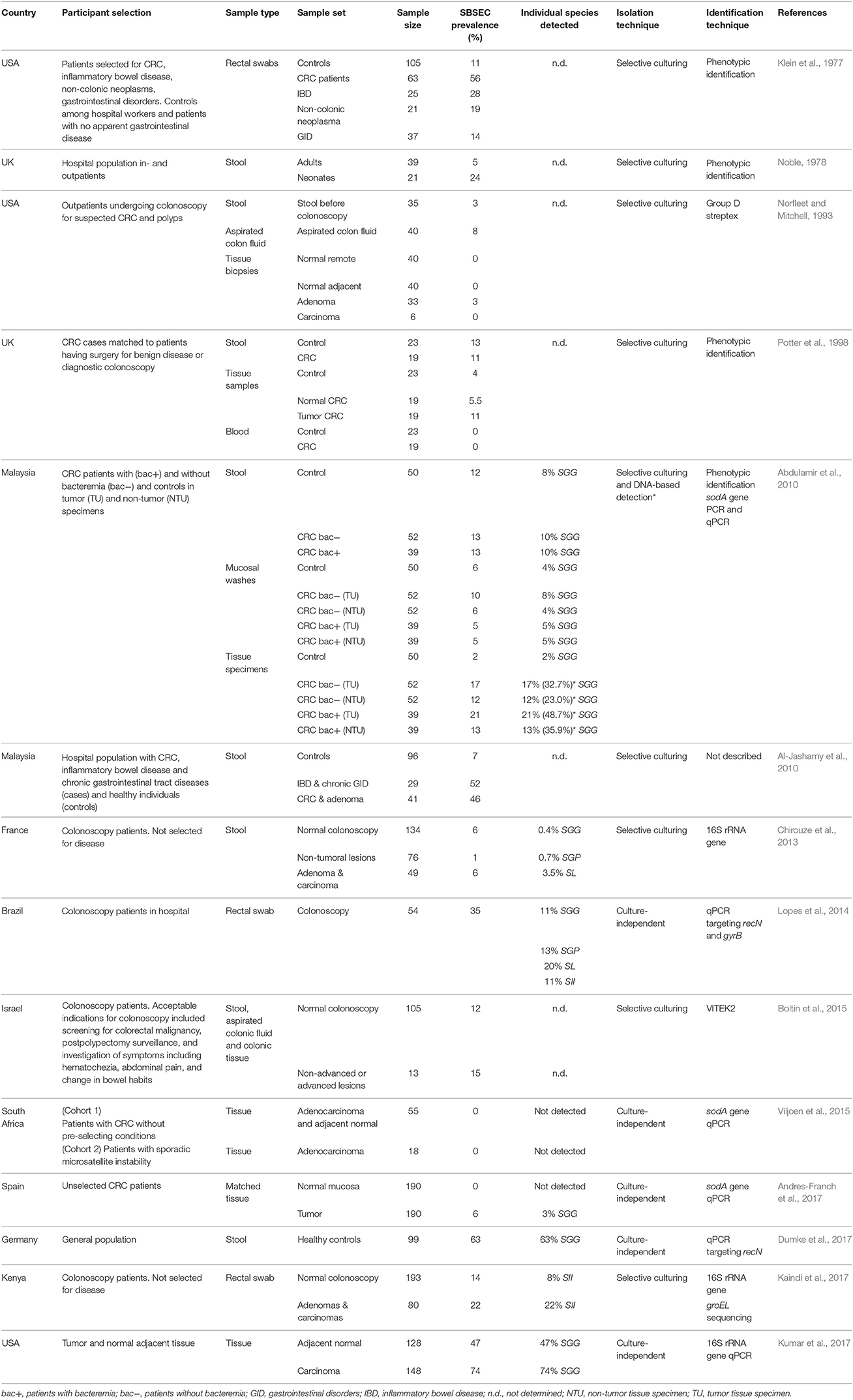
Table 1. SBSEC prevalence in the GIT in studies including healthy people and patients with gastrointestinal conditions sorted by year of publication.
Selective Colonization of CRC Patients and Preliminary Causal Evidence
Selective Colonization of CRC Patients
Fecal carriage of SBSEC members associated with CRC was initially reported to be 56% in 63 CRC patients compared to 11% in 105 healthy controls (Table 1) (Klein et al., 1977). This association has since been reported in a range of 6–46% in patients with adenomas and CRC vs. 7–14% in control patients (Abdulamir et al., 2010; Al-Jashamy et al., 2010; Chirouze et al., 2013; Boltin et al., 2015; Kaindi et al., 2017). Of these five studies, only two observed a significant carriage difference between healthy and neoplasia patients but one suggested a novel association of SII prevalence with hemorrhoids (Al-Jashamy et al., 2010; Kaindi et al., 2017).
The selective association with CRC tissue is also controversial. While no specific association of SBSEC members with CRC tissue was observed on small sample sets based on phenotypic identification (Norfleet and Mitchell, 1993; Potter et al., 1998), more recent DNA-based approaches approve of such an association ranging from 0 to 2% in controls, 47% in normal tissue of cancer patients and 3–74% in tumor tissues (Table 1) (Abdulamir et al., 2010; Andres-Franch et al., 2017; Kumar et al., 2017). Interestingly, tumor tissue of patients with bacteremia was more often positive for SGG (48.7%) than that of patients without bacteremia (32.7%) based on sodA gene PCR, which increased the detection rate of SGG from 27% and 16%, respectively, based on selective culturing. (Abdulamir et al., 2010). Tumor tissues harboring SGG were also significantly associated with co-infection by Epstein-Barr virus (OR: 9.49; 95% CI: 1.1–82.9) (Andres-Franch et al., 2017). Nevertheless, two other studies using a culture-based and qPCR-based approach still suggested no association between tumorous (0–15%) and non-tumorous tissues (0–12%) for SB/SGG (Boltin et al., 2015; Viljoen et al., 2015).
These contradicting data on SBSEC tumor tissue colonization might relate to patients with clinical infections of SBSEC (Abdulamir et al., 2010) or without any clinical symptoms (Viljoen et al., 2015; Andres-Franch et al., 2017), differences in detection techniques (culture-independent vs. selective culturing) or study population. This also indicates a non-obligatory relationship of SGG or SBSEC with CRC. SBSEC might only proliferate if certain requirements to facilitate colonization are met to become passengers as hypothesized in the bacterial-driver-passenger model (Tjalsma et al., 2012).
Preliminary Causality of SBSEC in CRC
Only minimal evidence for a causal relationship between SBSEC and CRC exists. Wall-extracted antigens from SGG NCTC8133, a human fecal isolate with controversial earlier classification as SII or SE, and its active S300 fraction are suggested to induce cell proliferation, polyamines and aberrant crypt foci in the distal colon of azoxymethane-treated rats (Ellmerich et al., 2000b; Biarc et al., 2004; Jans et al., 2016). In human CRC cell lines, stationary SGG strains TX20005 and TX20030 increased cell proliferation of HCT116, HT-29 and LoVo but not of SW480, SW1116, human lung carcinoma cell line A549, kidney epithelial cell line HEK293, human normal colon epithelial cell lines CCD841, CoN, and FHC suggesting the need for specific conditions to facilitate cell proliferation (Kumar et al., 2017).
The adenomatous polyposis coli tumorsuppressor gene APC is inactivated in many CRCs. This leads to an accumulation of β-catenin in the nucleus (He et al., 1998) thereby activating the c-Myc oncogene. Increased levels of β-catenin and c-Myc were detected in CRC cells after SGG incubation. This effect was not observed for SII, SGP, and SGM or for live SGG separated by trans-well membranes from cells, nor with bacterial supernatants, heat-killed bacteria or bacterial lysates suggesting a necessity of live SGG and direct SGG-cell contact (Kumar et al., 2017).
In azoxymethane-treated mice, inoculation with SGG led to increased cell proliferation, β-catenin accumulation in colonic crypts and higher numbers of tumors and dysplasia grade supporting a potential tumor-promoting role for SGG (Kumar et al., 2017). This effect was only observed for SGG strains able to induce cell proliferation in vitro (Kumar et al., 2018). The increase in cell proliferation was correlated with increased adhesion abilities in vitro. In vivo such strains also had an increased ability to colonize the mucosa in both C57BL6 as A/J mice. Therefore, the original bacterial driver-passenger model (Tjalsma et al., 2012) might need to be updated regarding the potential driving role of SGG. If SGG contributes to tumor development it would exert its effects probably after colonization of neoplastic sites as a passenger, which might depend on the specific strain and contribute to tumor progression rather than tumor initiation (Boleij and Tjalsma, 2013). In fact, the presence of polyps in the intestinal tract of Notch/APC mice allowed colonization and persistence of SGG UCN34 colonization, which was 1,000-fold higher than Notch control mice (Aymeric et al., 2018). However, no preferential adherence to tumor tissue sites was observed. Colonization was evenly distributed through the ileum and proximal colon, stimulated by secondary bile acids and was dependent on bacteriocins BlpA and BlpB that compete with other enterococci in the gut (Aymeric et al., 2018).
Interestingly, in vitro cultivation experiments suggested a growth advantage for SGG and SGM in the tumor microenvironment. Cultivation of SGG UCN34 and SGM CIP105683T in spent medium from CRC cells HT-29, SW480, HCT116, and Caco-2 was used to simulate utilization of metabolites from the tumor microenvironment (Boleij et al., 2012a). SGG and SGM displayed significantly increased growth rates in spent medium of Caco-2 cells whereas the growth rates of e.g., Salmonella enterica subsp. enterica serovar Typhimurium, Staphylococcus lugdunensis or Enterobacter cloacae were reduced, suggesting a significant advantage for SBSEC to proliferate in the spent CRC metabolites. The major changes in protein expression patterns were related to an upregulation of pyrimidine biosynthesis and glycolysis, particularly glycerolypid, glycosis and fructose utilization; and a downregulation of purine metabolism. Furthermore, SGG seems to be specifically capable to use secondary glucose metabolites fructose 6-phosphate and 3-phosphate glyceric acid (Boleij et al., 2012a). As the tumor microenvironment features increased levels of lactate, glucose derivatives, amino acids, lipids and fatty acids, SGG likely has an advantage to proliferate in this niche (Boleij et al., 2012a). Therefore, SGG can be described as an opportunistic pathobiont benefiting from the favorable oncogenic environment to colonize the host. This eventually promotes its translocation and systemic dissemination, in select cases leading to clinical infections.
Transmission and Niche Adaption of SBSEC Members
Transmission of SBSEC Between Hosts
The prevalence in the GIT of animals and humans facilitates transmission between animals and humans via feces and saliva (Dumke et al., 2015, 2017). Over a duration of 4 weeks, SB counts of an estimated 107 CFU/g broiler feed and 108 CFU/g wheat straw were only reduced by one log unit indicating high transmission possibility (Guy et al., 1980; Mackey and Hinton, 1990). Furthermore, soil clay adhesion of SB from bovine feces is very strong and cannot be desorbed after 24 h whereas long-term persistence seems weak but possibly sufficient to establish transmission within shorter time frames as observed among poultry flocks, surrounding environment and workers. In laying hens, colonization of non-carrier birds introduced into an SGG-positive flock took approximately 35 weeks and occurred likely via feed and feces (Guy et al., 1980; Dumke et al., 2015; Schulz et al., 2015). SGG isolates of identical sequence types where thereby causing infection in one worker and contributing to in-flock and old-young transmission in hens (Dumke et al., 2015). Similarly, sequence types were shared between turkey, pigeons, chicken and humans (Schulz et al., 2015). Rural residency, close animal-human contact and the use of manure as fertilizer were identified as risk factors for colonization with SGG, SGP, SII, SL, and SB in humans (Giannitsioti et al., 2007; Corredoira et al., 2008; Dumke et al., 2017).
Transmission via the fecal-oral or food-oral route requires survival of the gastric passage for colonization of the gut. Survival to simulated gastric conditions is dependent on the SB strain, gastric pH and feed ingestion. At pH < 2.5, survival is minimal whereas at pH 3.0 a reduction of approximately 3 log units was observed for SII while SGM did not survive, suggesting species-specific abilities to survive and reach the intestine in sufficient numbers for colonization (Ripamonti et al., 2011; Jans et al., 2016). The role of transmission via fermented food products that contain SII, SL, or SGM particularly in sub-Saharan Africa, Southern Europe, Asia and Latin America is not yet clear and niche adaptation might play a pivotal role in their ability to colonize the host (Jans et al., 2015, 2017; Kaindi et al., 2017).
Niche Adaptation of SBSEC Members
SBSEC members adapt to multiple niches. In the rumen, the main carbon sources are largely available in the form of plant fibers. Their utilization is a key feature of many SBSEC members. Proteolysis and peptidase activity are important for the breakdown of proteins to ammonia (Wallace and McKain, 1991) that further supports growth of SBSEC members (Sales-Duval et al., 2002). α- galactosidase, β-glucanases, and endoglucanases of SBSEC play important roles for the degradation of complex carbohydrates in ruminants and chicken (Bailey, 1963; Ekinci et al., 1997; Beckmann et al., 2006). Most of these enzymes are adapted to GIT conditions with a narrow pH optimum of pH 5.6–6.3 and a temperature optimum of 37–42°C (Bailey, 1963; Wang et al., 2015; Chen et al., 2016), except for α-amylase and lactate dehydrogenase activity that increases at pH 5.5 vs. pH 6.5 and is possibly part of a self-feeding loop for lactate overproduction (Chen et al., 2016). At pH > 6.0, metabolism is directed toward production of formate, acetate and ethanol (Chen et al., 2016), while below pH 5.5, it is directed toward lactate, particularly in the event of excess glucose sources such as starch (Gunsalus and Niven, 1942; Russell and Hino, 1985; Asanuma and Hino, 1997). Ruminal acidosis results in a drop below pH 5.5 causing bloat in the rumen (Penner et al., 2007). Despite inconclusive causality, evidence suggests initiation by the overgrowth of SB in combination with Prevotella, Ruminococcus, Streptococcus, and Lactobacillus and the parallel inability of lactate utilizers such as Megasphaera elsdenii and Selenomonas ruminantium to metabolize lactate (Palmonari et al., 2010; Wang et al., 2015; McCann et al., 2016).
Particularly SGG has retained the ability to utilize a wide range of carbon sources typical for the rumen. This feature is minimized in SGM and SII or modified to a different variety of carbon sources in SGP (Rusniok et al., 2010; Lin et al., 2011b; Papadimitriou et al., 2014). SGP ATCC43144 in contrast to SGG ATCC43143 harbors a α-L-rhamnosidase, several endo-β-N-acetylglucosaminidase, glucokinase, glucosidases, mannosidases to utilize specific carbon sources available in the gut originating from plant cell walls, biofilms, glycosides, and glycolipids (Lin et al., 2011b). SGG is the only Streptococcus known so far to use malate via the malolactic enzyme (Gallo_2048) and a malate transporter (Gallo_2049) as well as degrade tannins encoded by tanA, that are otherwise toxic to many bacteria (Rusniok et al., 2010; Papadimitriou et al., 2014). SGG UCN34 is also able to hydrolyze bile salts, an important feature to survive in the small intestine. SGG is prototroph for all 20 amino acids. Besides, SGG harbors partial biosynthesis pathways for biotin and thiamine to support growth in varying conditions including the rumen, intestine and also the blood system (Rusniok et al., 2010).
Niche adaptation is also observed in SII, SGM, and SGP via gene loss and gain. They cannot biosynthesize pantothenate and biotin (Lin et al., 2011b; Papadimitriou et al., 2014) but depend on them for growth (Barnes et al., 1961). Dairy variants of SII strains adapted to the dairy environment via a modified lactose (LacS and LacZ instead of phosphotransferase) and peptide metabolism (duplication of oligopeptide transporters) (Jans et al., 2012a, 2013). Dairy SGM lost or harbor truncated genes for degradation of plant carbohydrates and detoxification of substances relevant for survival in the rumen that are likely obsolete in the dairy niche. In addition, dairy SGM gained gene clusters for casein hydrolysis, lactose and galactose metabolism for optimal utilization of these milk components (Papadimitriou et al., 2014). Both dairy SII and SGM possess lactocepin with high sequence similarity to PrtS CEP of S. thermophilus responsible for milk protein degradation. Furthermore, SGP showed gene loss likely related to adaptation to nutrient-rich environments and an overall genome reduction compared to SGG (Papadimitriou et al., 2014). These findings demonstrate the high adaptability of SBSEC members to different carbon and protein sources not only within the GIT, but also within the dairy environment.
Niche adaptability is also reflected by a high genome plasticity among SBSEC members. The pangenome is increasing at a high rate entailing numerous unique genes for each new strain added (Hinse et al., 2011b). Nearly all SBSEC genomes reveal horizontal gene transfer relating to general carbohydrate metabolism, capsular polysaccharides, antimicrobial resistance or tannase as the key discriminator of SGG (Rusniok et al., 2010; Hinse et al., 2011b; Lin et al., 2011b; Jans et al., 2013; Papadimitriou et al., 2014; Kambarev and Caté, 2015; Grimm et al., 2017a; Kambarev et al., 2017). Natural competence operons and pseudopilus identified in SII and SGG likely contribute to the success of horizontal gene transfer and thus to niche adaptation (Rusniok et al., 2010; Lin et al., 2011b; Morrison et al., 2013; Jans et al., 2016). As a consequence, SBSEC members feature highly diverse metabolic abilities and likely also different virulence factors depending on the species and different impact depending on the niche colonized.
Mechanisms and Virulence Factors Responsible for Adhesion and Host Colonization by SBSEC Members
The establishment of bacteria in a niche depends on a multitude of factors relating to adherence, signaling, nutritional adaptation and host modulation. Some of the key factors involved in adhesion and colonization in streptococci include cell-wall anchored factors such as LPXTG-motif proteins, anchorless factors including the cell capsule, two-component signal transduction systems for signaling or released/secreted factors such as exopolysaccharides to form biofilms (Nobbs et al., 2009; Brouwer et al., 2016).
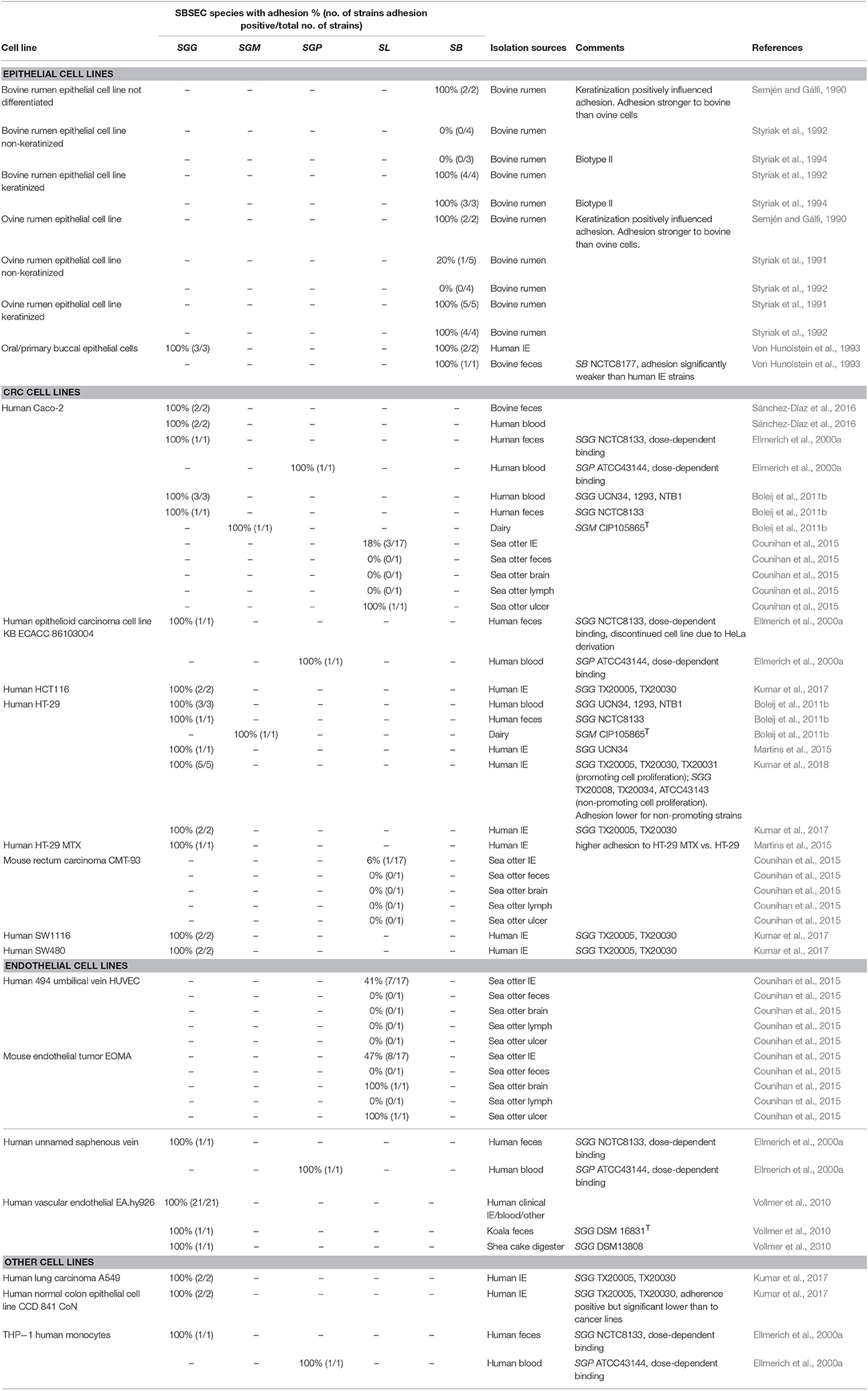
Employing epithelial and endothelial cell lines provides an advanced model to study adhesion via bacteria-cell interactions. Adhesion capabilities can vary depending on the environment and the associated microbiota as well as the cell type present. Given the presence of SBSEC members in GIT and the blood system, adhesion and colonization have to be comprehensively assessed using designated cell lines. This includes cells originating from oral (primary buccal epithelial cells), gastric (rumen epithelial cell line), intestinal (human CRC cell lines Caco-2, HT-29, HCT116 and mucus-producing HT-29 MTX; mouse rectum carcinoma CMT-93) and venous sites (human vascular endothelial EA.hy926, human umbilical vein HUVEC, unnamed saphenous vein and mouse endothelial tumor EOMA).
Adhesion to Epithelial Cell Lines of the GIT
Oral epithelial cells represent the first cells in the GIT to interact with SBSEC members upon ingestion. Adhesion to buccal epithelial cells of human IE-derived SB biotype I and II strains was around 2–3 times higher than that of commensal reference strain SB DSM20480T = NCTC8177 (Von Hunolstein et al., 1993), suggesting that epithelial adhesion is particularly present among IE-derived strains and likely dose-dependent (Ellmerich et al., 2000a).
Adhesion in the rumen seems to be pH and cell type dependent. Highest adhesion of SB strains to rumen epithelial cells was observed between pH 7.0–7.3. Near ruminal pH of 6.5, adhesion was still elevated suggesting that these SB strains adapted to the rumen (Styriak et al., 1994; Wang et al., 2015). An important factor for adhesion in the rumen was related to epithelial keratinization and particularly glycocalix (a glycoprotein and glycolipid cell surface layer) present on differentiated cells (Table 2). Keratinization significantly enhanced adherence of SB and enabled adhesion for those isolates unable to adhere to non-keratinized cells (Semjén and Gálfi, 1990; Styriak et al., 1991, 1992). However, host-specificity of SB strains using sheep and calve rumen epithelial cells was inconclusive. Therefore, it remains unclear whether host-specificity is a driving factor in rumen colonization (Semjén and Gálfi, 1990; Styriak et al., 1992).
In the colon, adhesion of SBSEC members was assessed using epithelial CRC cell lines CMT-93, Caco-2 and HT-29 (Table 2). Adhesion to these CRC cell lines was observed for several SBSEC species including SL, SGG and SGM. Animal-derived SL strains (sea otter IE, feces, brain and lymph node isolates) adhered highly variable to CMT-93 and Caco-2. Significant adhesion (>0.2–0.3% of the inoculum) to CMT-93 and Caco-2 was rare within this strain pool and at slightly lower levels than the S. Typhimurium reference strain (Counihan et al., 2015). Adhesion to CMT-93 was generally lower than and without correlation to that of Caco-2 even for S. Typhimurium (Counihan et al., 2015) suggesting low suitability of CMT-93 cells for SBSEC adhesion assessment and possibly the need to evaluate sea otter-derived SL for host specificity.
Adhesion abilities toward Caco-2 and HT-29 of human-derived SGG and dairy SGM was low (<15% of inoculum) to intermediate (20–50% of inoculum) for SGG and SGM/SE, respectively (Table 2). Low adhesion was comparable with that of S. Typhimurium whereas intermediate adhesion was comparable to that of E. coli and Lb. plantarum reference strains, but significantly lower than the 80–98% observed for E. faecalis. Differences were particularly evident for SGG NCTC8133 that more efficiently adhered to Caco-2 than HT-29 (Boleij et al., 2011b). A comparative assessment of blood-derived SGG of IE patients indicated significantly enhanced adhesion abilities to HT-29 cells, particularly among strains able to promote tumor cell proliferation (Kumar et al., 2018). This differentiation between proliferation-promoting and non-promoting SGG was paralleled by the ability to colonize mice. Interestingly, mice colonization was increased in A/J type mice compared to C57BL/6 mice, which might be related to different host factors required for colonization (Kumar et al., 2018). Therefore, certain tumor-promoting strains might possess enhanced adhesion capabilities and thus a selective advantage particularly in a tumor environment presenting favorable factors. General adhesion of SBSEC members to CRC cell lines however seems limited in comparison to other gut pathogens.
Adhesion to Endothelial Cell Lines
Similar to epithelial cell lines, dose-dependent adhesion behavior was also observed for SGG NCTC8133 (human fecal isolate) and SGP ATCC43144 (human blood isolate) to human saphenous vein endothelial cell lines (Table 2). Especially at low inoculums, binding to endothelial cells was higher than to epithelial cells suggesting a preference toward endothelium (Ellmerich et al., 2000a). This was also observed among primarily human IE SGG isolates adhering to EA.hy926 cells but not with animal feces-derived SGG DSM16831T (Vollmer et al., 2010). Mechanical stress on HUVEC cells had no influence on adherence suggesting that SGG actively colonizes endothelial tissues (Vollmer et al., 2010). Interestingly, only blood isolates of SGG express the blood-group antigen sialyl lewis-X (sLex) on their cell surface. sLex is normally expressed on the cell surface of leukocytes enabling rolling of leukocytes on the endothelium. This might increase SGG adhesion to endothelial cells (Hirota et al., 1996).
Significant adhesion to endothelial HUVEC-C cells was observed in multiple IE and septicemia SL strains isolated from sea otters (Counihan et al., 2015). The ability to adhere to cell lines other than HUVEC-C was minimal; only one strain showed adhesion to human epithelial Caco-2, human endothelial HUVEC-C and mouse endothelial tumor EOMA cells (Table 2). The other strains showed minimal adhesion to mouse/human epithelial or endothelial cells, suggesting key differences in adhesion mechanisms to different cell types and possibly host origin. In general, isolates from heart or blood adhered better to intact endothelial cells and support possible IE establishment without previous history of heart disease (Counihan et al., 2015). However, general adhesion of SL strains was in the range of 0.05–0.2% of the inoculum and thus significantly lower than the 2% of human clinical Staphylococcus aureus ATCC25923 used as reference (Counihan et al., 2015). The biological impact to trigger IE despite this significantly lower adhesion ability of SL in contrast to S. aureus will require further evaluation in relation to host specificity and mechanisms responsible for endothelial tissue colonization by SBSEC members in vitro and in vivo.
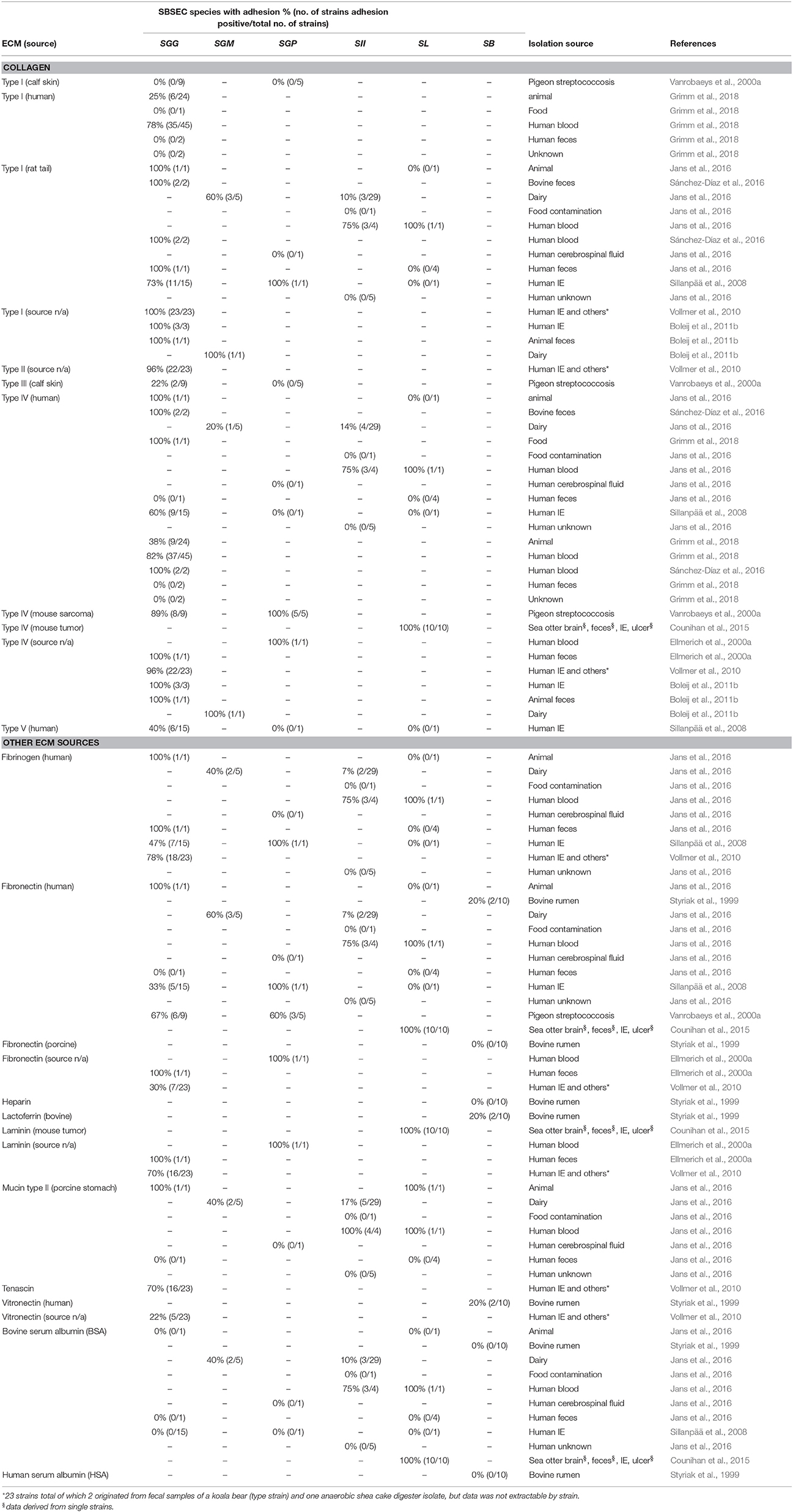
Binding to Extracellular Matrix Proteins
Extracellular matrix proteins (ECM) are an important component to facilitate bacterial binding to epithelial and endothelial cell surfaces and thus niche colonization in humans and animals. Collagen thereby plays an important role. Collagen type I is present in organ capsules and scar tissue, such as on damaged heart valves. Collagen type IV is the main constituent of basement membranes and can become exposed at tumor sites (Tanjore and Kalluri, 2006; Boleij and Tjalsma, 2013).
Adhesion to collagen type I and IV is a key feature of SGG (Table 3). Most SGG strains derived from human blood cultures of IE or bacteremia patients, pigeons suffering from streptococcosis (SGG and SGP) and SL strains derived from sea otters with IE, but also fecal and dairy SBSEC isolates displayed binding to collagen type IV. In contrast, binding to collagen type I was a feature mostly associated with human blood-derived SGG, SGP, SII and SL isolates and partially also with SGM isolates (Vanrobaeys et al., 2000a; Sillanpää et al., 2008; Vollmer et al., 2010; Boleij et al., 2011b; Counihan et al., 2015; Grimm et al., 2018). Among a panel of human and food-derived SII and SL as well as dairy SGM, adhesion to collagen type I and IV was particularly present in human blood isolates (Boleij et al., 2011b; Jans et al., 2016). In contrast, SGG considered as commensals and isolated from human feces, pigeons and ruminants including the SGG type strain rarely bound to collagen type I, III and IV (Table 3). Similar differences were also observed between SGG from infected vs. SGG from healthy humans featuring high vs. low adhesion, respectively (Grimm et al., 2018). SGG NCTC8133 and SGP strain ATCC43144 were only shown to bind collagen type IV whereas SGP DSM15351T strains displayed no adhesion to collagen type I and IV. Other collagen types bound by human IE-derived SGG are collagen type II (96%) and to a lesser extent collagen type V (40%) (Table 3) (Ellmerich et al., 2000a; Sillanpää et al., 2008; Vollmer et al., 2010). Differences in collagen adhesion patterns therefore seem to exist between animal and human-derived strains even within the same species. Whether these different adhesion patterns among SGG and other SBSEC members also translate into different abilities to cause disease remains to be investigated.
Connective tissue and the tumor-microenvironment contain an extensive network of ECM including collagen, laminins, fibronectin, proteoglycans, and hyaluronans (Peddareddigari et al., 2010). SL strains from sea otters adhered to fibronectin, laminin, and hyaluronic acid in all cases (Table 3) (Counihan et al., 2015). Also human blood-derived SII and SL featured high adhesion abilities to fibronectin (Jans et al., 2016). Interestingly, the SL genome features relevant hits to adhesion factors such as pneumococcal cell surface adherence protein A PavA involved in fibronectin-binding and the laminin-binding protein Lmb (Jin et al., 2013). Binding to fibronectin is also observed in the SGG type strain and the majority of pigeon-derived SGG strains (Table 3). Interestingly, fibronectin-binding is less prevalent in human-derived SGG and SGP with the exception of SGG NCTC8133 and human blood strain SGP ATCC43144 (Ellmerich et al., 2000a; Vanrobaeys et al., 2000a; Sillanpää et al., 2008; Vollmer et al., 2010; Jans et al., 2016). Furthermore, rumen-derived SB strains showed low or no binding to both human and porcine fibronectin (Styriak et al., 1999). Similar, the SII type strain, SII dairy and SL human commensal strains showed with a few exceptions only minor adhesion abilities to fibronectin, collagen type I and IV, mucin and fibrinogen (Table 3) (Jans et al., 2016).
Fibrinogen-binding is in contrast to fibronectin-binding a common feature also among human IE SGG strains, dairy SGM, human SII and SL blood isolates (Table 3) (Ellmerich et al., 2000a; Sillanpää et al., 2008; Vollmer et al., 2010; Jans et al., 2016). Human IE-derived SGG also showed interactions with tenascin, laminin and vitronectin (Ellmerich et al., 2000a; Sillanpää et al., 2008; Vollmer et al., 2010). In contrast, rumen SB strains showed mostly moderate or weak adhesion to bovine lactoferrin, vitronectin, heparin, and BSA. None of the SB strains bound to human serum albumin (Table 3) (Styriak et al., 1999). These differences in ECM adhesion patterns between animal and human strains might therefore be important for their colonization abilities of different body sites. These patterns furthermore suggest different adhesion mechanisms in SL, SGG and other SBSEC members (Lin et al., 2011b; Papadimitriou et al., 2014). Particularly human and animal blood isolates seem to have the ability to bind fibrinogen, while fibronectin-binding is variable, which implies different adhesion abilities regarding fibronectin in the tumor-microenvironment and fibrinogen at damaged sites requiring blood clotting.
Biofilm Formation, Exopolysaccharides, Dextran Production and Capsular Polysaccharides
Adhesion and biofilm-forming abilities are linked to colonization and persistence in the GIT. SB produce at least two types of polysaccharides: (1) water-soluble glucans, often dextrans, comprised of α-1:6 linked glucose units (Bailey, 1959); and (2) capsular polysaccharide (Bailey and Oxford, 1958). GtfA in SGG was found to produce water-insoluble α-1,3-linked glucosidic polymers whereas GtfB encoded for α-1,3-linked water-insoluble and α-1,6-linked glucosidic water-soluble polymers (Lin et al., 2011b). Both types of polysaccharides have specific roles in adhesion, colonization and host immune evasion (Nobbs et al., 2009; Isenring et al., 2018). The ability to form biofilms is however not directly correlated to virulence and needs to be carefully distinguished (Vollmer et al., 2010).
Biofilm formation was observed with SBSEC strains from GIT, blood and food origin (Vollmer et al., 2010; Boleij et al., 2011b; Jans et al., 2016). SII and other SBSEC members were also observed to form biofilms on human teeth featuring various degrees of auto- and co-aggregation with other oral microbes (Shen et al., 2005; Arul and Palanivelu, 2014). Even outside a host, biofilm formation to uncoated plastic and stainless steel surfaces was observed for all SBSEC species of blood, animal and dairy origin (Flint et al., 1997, 1999; Jans et al., 2016).
Polysaccharides are major constituents of biofilms (Christensen, 1989; Nobbs et al., 2009). SGG, SL and many SBSEC members are known to produce extracellular glucan encoded by glycosyl-transferases similar to GtfA, GtfB, and GtfC of S. mutans but lacking in SGP ATCC43144 (Rusniok et al., 2010; Lin et al., 2011b). Instead, SGP harbored a strain-specific exopolysaccharide biosynthesis gene cluster featuring sequence identity highest with those of Bacillus cereus and Clostridium thermocellum (Lin et al., 2011b). Generally, this suggests that from a common SBSEC ancestor, SGG likely kept most biofilm-related loci while the respective loci were either absent or comprised of pseudogenes in SGP, SII and SGM potentially reducing or abrogating biofilm formation capabilities in comparison to SGG (De Vuyst and Tsakalidou, 2008; Lin et al., 2011b; Papadimitriou et al., 2014). Biofilm production might therefore be SBSEC-species dependent but older data are inconclusive in this respect.
Dextran production from rumen SB biotype II isolates of sheep, calve and cow is particularly dependent on available sugar compounds and a CO2 source. In contrast to capsular polysaccharides, the production of dextran is limited and directly correlated with the available sucrose concentration (Bailey and Oxford, 1958; Barnes et al., 1961; Cheng et al., 1976). The CO2 source can include , which is readily available in the rumen (Bailey and Oxford, 1958; Barnes et al., 1961). For dextran production by SB, three different growth requirements are suggested: (i) biotin and ammonium chloride as sole vitamin and N-source, respectively, (ii) calcium, pantothenate, adenine, biotin, thiamine and arginine or glutamic acid, or (iii) xanthine and additional amino acids (Barnes et al., 1961). Prototroph SGG in contrast to SB biotype II likely possess the metabolic capabilities to produce biofilm even in niches not meeting these growth requirements (Rusniok et al., 2010).
Dextran production furthermore seems to play a role in ruminal acidosis (Humer et al., 2018). It is hypothesized that the higher sucrose content of grain feed boosts dextran production in ruminants to form a slime in the rumen (Cheng et al., 1976; Kulp and Ponte, 2000; Humer et al., 2018). This slime, comprised of proteins and polysaccharides of other bacteria, increases viscosity and produces a froth foam eventually leading to bloat (Cheng et al., 1976). A key role in the slime production process is attributed to SB via acidification and dextran production via its rumen-adapted dextran sucrase (Bailey and Oxford, 1958; Min et al., 2006).
Virulence Factors of SBSEC Members Related to Adhesion and Colonization
General Aspects of the Cell Surface in Relation to Adhesion
The bacterial cell surface has important roles in the interaction with the environment, the host and for pathogenesis (Nobbs et al., 2009; Isenring et al., 2018). Lipoproteins featuring a serine-rich motif following a cysteine residue are frequently present on the surface of SGG UCN34, possibly linked to specific interactions with polysaccharides from the environment (Rusniok et al., 2010). Wall-extracted antigens from the cell surface of SBSEC members bound equally well to epithelial and endothelial cell lines as whole SBSEC cells, supporting a role for cell surface factors in adhesion (Ellmerich et al., 2000a). Among surface proteins, SB surface protein Sbs6, Sbs10, Sbs13, and Sbs16 as well as the histone-like protein HlpA, autolysin AtlA and the cell surface protein antigen C PaC are currently characterized SBSEC virulence factors besides pili. While HlpA is present in most SBSEC members, Sbs13, Sbs16, AtlA and PaC are mainly limited to SGG whereas Sbs6 and Sbs10 are also regularly observed in SE. Among SGG, only blood-derived SGG usually feature all seven surface proteins in contrast to rumen or fecal isolates (Table 4 and Supplementary Data 1). This suggests reduced or different virulence characteristics of the other SBSEC members in comparison to blood-derived SGG.
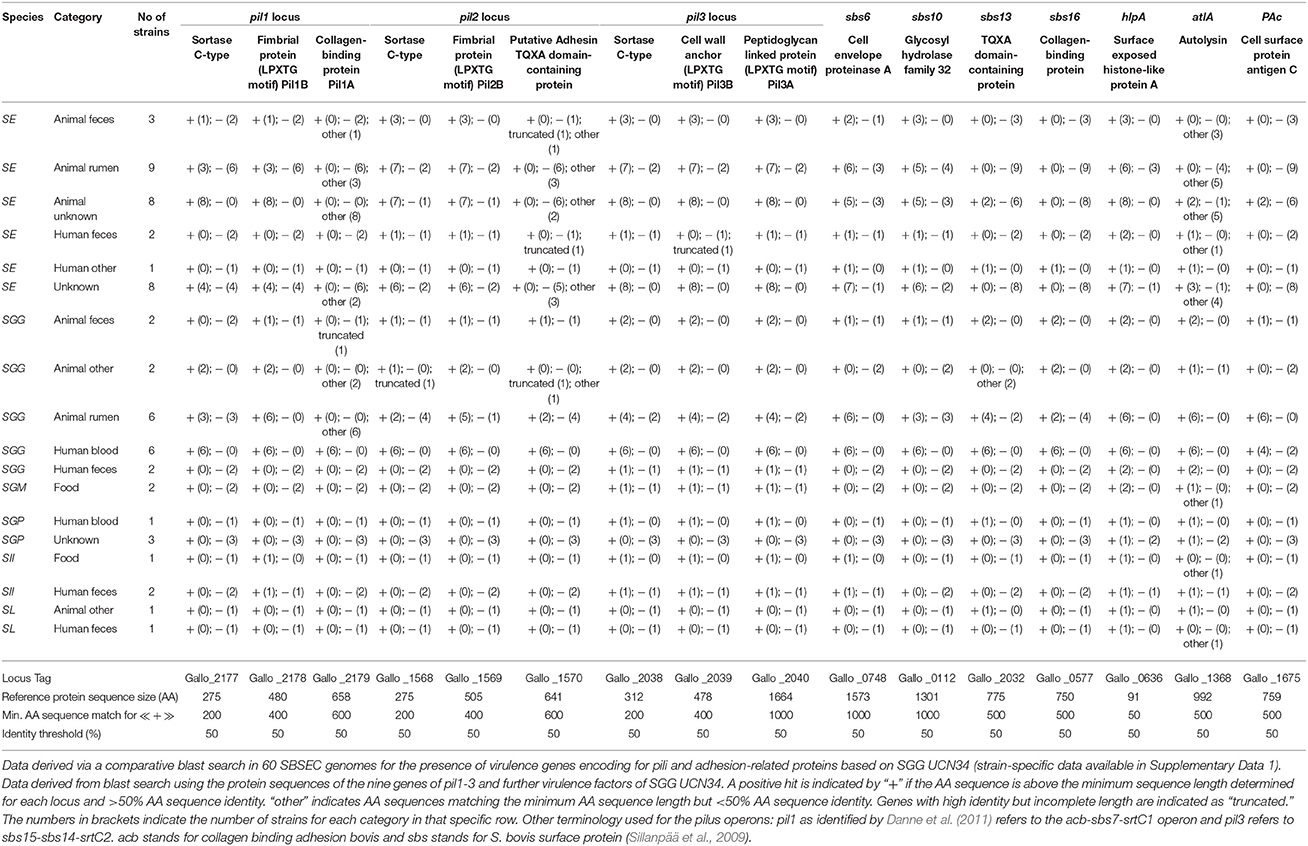
Table 4. Presence of major known virulence factors of SBSEC members in relation to adhesion, colonization and immune system interaction for SBSEC strains of human, animal and food origin.
Specific studies were performed on enolase and HlpA. Enolase is a conserved anchorless surface protein involved in cross-linking of SGG UCN34 and human epithelial cells (Boleij et al., 2011a). The main interaction partner was identified as cytokeratin 8. Cytokeratin 8 is constantly expressed by epithelial cells, but at increased levels by CRC and could therefore play a role in the association of SGG with CRC (Boleij et al., 2011a).
HlpA is highly prevalent among SBSEC members possibly involved in adhesion (Table 4) (Boleij et al., 2009a; Lin et al., 2011b; Papadimitriou et al., 2014). HlpA is an anchorless bacterial surface protein that binds to lipoteichoic acid at the Gram-positive cell wall. Lipoteichoic acid was previously suggested to be involved in adhesion in cooperation with surface proteins (Von Hunolstein et al., 1993; Styriak et al., 1994). Binding to colon tumor cells is then further established via heparan sulfate proteoglycans (Boleij et al., 2009a). However, heparin (a heparan sulfate proteoglycan) and lipoteichoic acid compete for the same binding sites in HlpA which cannot efficiently bind simultaneously to both structures (Boleij et al., 2009a) supporting earlier observations that heparin treatment of rumen SB isolates inhibited lactoferrin-binding (Styriak et al., 1999). ARH-77syn myeloma cells overexpressing syndecan-1 (the predominant heparan sulfate proteoglycan on epithelial cells) displayed increased adherence of SB, other streptococci and staphylococci in contrast to ARH77 cells without syndecan-1 as well as E. faecalis or E. coli and other Gram-negative bacteria (Henry-Stanley et al., 2005). Therefore, heparan sulfate proteoglycans might play a significant role in epithelial interactions for staphylococci and streptococci to modulate interactions with tumor epithelial cells (Henry-Stanley et al., 2005; Boleij et al., 2009a).
The Capsule
The capsular polysaccharide of SBSEC members primarily consists of galactose, rhamnose and uronic acid. It is produced from glucose or other carbohydrates and in contrast to exopolysaccharides does not need CO2 for production (Bailey and Oxford, 1958). Capsule properties are however strain dependent. Highly virulent strains, in this case only SGG strains, possessed a significantly thicker capsule whereas truncated genes in dairy isolate of SII or SGM might inhibit capsule production (Vanrobaeys et al., 1999; Boleij et al., 2011b; Papadimitriou et al., 2014). Genome data suggests a high diversity of capsular polysaccharides in SGG, SGP and SII varying in length between 12 and 19 genes with a conserved start followed by a strain-specific genetic variability. It is thus related to capsule heterogeneity and varying antigenic properties (Rusniok et al., 2010; Hinse et al., 2011b; Lin et al., 2011b; Jans et al., 2013; Jin et al., 2013; Papadimitriou et al., 2014).
The capsule of many streptococci also comprises hyaluronic acid, which is important for adhesion to host cells, colonization and phagocytic killing and infection (Hynes, 2004). Capsule degradation by hyaluronidase was significantly correlated with decreasing adherence to epithelial and endothelial cells (Counihan et al., 2015). Conversely, SL is also able to use host-derived hyaluronic acid to boost adherence and invasion suggesting a key role for hyaluronic acid in SBSEC pathogenesis (Counihan et al., 2015).
Pili and Their Role in Adhesion
A key surface structure involved in the interaction of SBSEC members with their environment and hosts are fimbriae or pili. Initially, these specific surface structures were described as fimbriae among highly virulent SGG of pigeon origin (Vanrobaeys et al., 1999). However, it seems that currently recognized pili in SBSEC encompass these fimbriae. Fimbriae were therefore interpreted accordingly for this review.
In general, SGG harbors three pilus loci termed pil1, pil2, and pil3, each of which is comprised of three genes (Danne et al., 2011). The main exceptions among SGG seem to be animal-derived SGG including the type strain and human fecal SGG, which harbor incomplete pilus loci or significant mutations in the genes encoding for collagen-binding and adhesion in pil1 and pil2 (Table 4). In other SBSEC members, the three pilus loci display signs of genome decay through various mutated, truncated or completely absent loci and genes (Table 4 and Supplementary Data 1). Most SE, SII and SGM harbor only pil3 as a complete locus with significant sequence identity to SGG. pil1 and pil2 loci are frequently incomplete due to truncated or completely absent genes. Similar to animal-derived SGG, the genes encoding for collagen-binding and adhesion in pil1 and pil2 are regularly mutated in SE and SII, which leaves open questions regarding their functional properties. SL and SGP do not seem to possess any structures resembling the pilus loci of SGG (Table 4 and Supplementary Data 1) (Jans et al., 2013; Papadimitriou et al., 2014). This might explain the absence of pili in low virulent SGP (Vanrobaeys et al., 1999) and suggests species-specific pilus organization. However, genome data is limited to animal commensal strains but not animal pathogens such as SL from sea otters or SGP from pigeons, which will be required to determine the pili and virulence factor repertoire in relation to SBSEC species and host.
The pil1 locus in SGG is among the most investigated virulence factors of SGG. The three genes of pil1 encode for two LPXTG-motif proteins and one sortase C. The two LPXTG-motif proteins [Gallo_2179 (Pil1A) and Gallo_2178 (Pil1B)] represent the typical features of pilin subunits with a pilin motif PK centralized in the protein containing a structural CnaB domain. Only Pil1A harbors a putative collagen-binding domain (Danne et al., 2011). The core pilus itself is comprised of heteropolymers of the two LPXTG-motif proteins while other parts were only comprised of Pil1A or Pil1B (Danne et al., 2011). A comparative blast search using the pil1-3 locus proteins of SGG UCN34 to 60 available SBSEC genomes revealed that the collagen-binding protein Pil1A is unique among human blood SGG (Table 4 and Supplementary Data 1). This suggests that Pil1A is a key feature of SGG capable of causing IE that might have been lost or modified in other SGG lineages and SBSEC members. This highlights further that only some virulence factors specific for SGG have been unraveled whereas those specific for other SBSEC members remain mostly unknown.
Each pilus loci and the individual proteins seem to have specific roles of which only that of pil1 and pil3 have been elucidated. The role of the pil1 pilus was confirmed to be a collagen-binding adhesin, important for biofilm formation and virulence, particularly in IE rat models using SGG UCN34 as model strain (Sillanpää et al., 2009; Danne et al., 2011). Adhesion assays confirmed preferred collagen I adhesion of pil1 over collagen IV, fibronectin and fibrinogen. This finding is relevant for the establishment of biofilms by SGG on collagen-rich surfaces, which are observed in CRC tissues and on damaged heart valves where it may lead to IE development (Danne et al., 2011). pil3 is a key factor in binding to mucus, which covers the intestinal epithelium (Lichtenberger, 1995). In vivo assays using SGG UCN34 displayed impaired colonization of Δpil3 mutants in the distal colon of mice (Martins et al., 2015). pil3 also binds to human stomach mucins and human fibrinogen. This supports the importance of pil3 in host adhesion by SGG UCN34 (Martins et al., 2016) and likely other SGG, SII and SL harboring pil3 (Table 4), the presence of which correlated directly with fibrinogen and mucin adhesion in vitro (Jans et al., 2016; Isenring et al., 2018). This would give pil1 and pil3-carrying SGG an advantage for the adhesion to CRC tissue featuring mislocalized MUC5AC mucin and exposed collagen type IV, the adhesion to fibrinogen at injured sites in the blood system as well as the colonization of collagen type I exposing surfaces such as damaged heart valves (Martins et al., 2016). This would subsequently initiate the first stage of IE.
Invasion and Infection Establishment
SBSEC as pathobionts are opportunistic invasive pathogens able to colonize secondary sites via the bloodstream. SBSEC possess different mechanisms to establish disease by bacterial translocation, survival in the blood stream and adhesion to endothelial cell surfaces. In the case of underlying conditions such as damaged heart valves or CRC, SBSEC possess a selective advantage and cause severe bacteremia or IE in humans and animals.
Epithelial Cell Response to Host-Microbe Interaction
The intestinal epithelial barrier is an important line of defense of the GIT, as it is proposed to be the main point of entry to cause infection. Several studies have looked at the ability of SBSEC to be recognized by the epithelium and result in a local immune activation and chemoattraction. For example, SGG NCTC8133 and SGP ATCC 43144 have been shown to induce IL-8 secretion in buccal epithelial and endothelial cells in a dose-dependent manner, but not in intestinal Caco-2 cells. While SGG was more effective in the induction of IL-8 from buccal epithelial cells, SGP released higher levels of IL-8 from endothelial cells. Furthermore, SE and SGM induce IL-8 also at mRNA level, while SGG was not able to induce an IL-8 response in HT-29 and Caco-2 cells. None of the strains induced IL1-β at mRNA level in intestinal epithelial colon cells (Boleij et al., 2011b).
In contrast, wall-extracted antigens from SE are very effective in inducing IL-8 from buccal epithelial cells, endothelial cells and intestinal epithelial cells (Caco-2). Heat inactivation ablated the effect of wall-extracted antigens on IL-8 secretion, suggesting that proteinaceous components of the cell wall are involved in the IL-8 response (Ellmerich et al., 2000a). Especially the S300 fraction of the wall-extracted antigens was capable of inducing IL-8 and prostaglandin secretion in Caco-2 cells. Proteins in the wall-extracted antigens that could be responsible for these effects were GroEL, SOD, Dpr, Aldolase, Enolase and L-lactate dehydrogenase (Biarc et al., 2004). In contrast, colonization of pre-sensitized Balb/C mice with the SB HC5 strain producing the lantibiotic bovicin HC5, resulted in an increase influx of eosinofils and reduction of brushborder and goblet cells in the small intestine, with a 4-fold increase in cell proliferation, while the large intestine was not affected by this HC5 strain (Paiva et al., 2012).
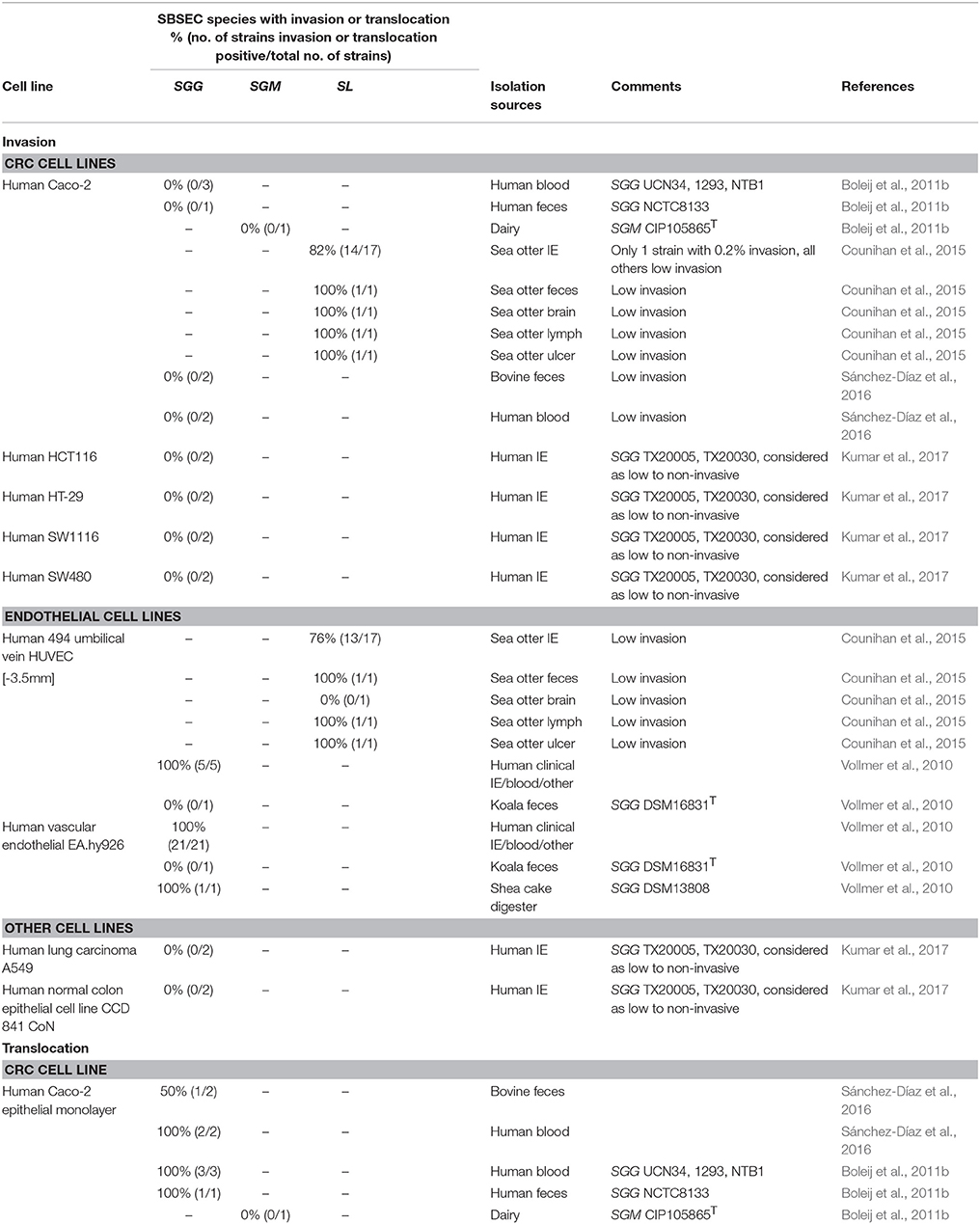
Epithelial/Endothelial Translocation and Invasion
For bacterial invasion, either epithelial invasion or translocation is necessary to enter the human body. While adherence to epithelial cells ranged from 2 to 10% for SGG, 2 to 50% for SE, and 25 to 30% for SGM depending on the cell line used, internalization was below 0.1% for all strains (Boleij et al., 2011b; Sánchez-Díaz et al., 2016). SGG strains rarely showed significant invasion into epithelial cells (Table 5) (Sánchez-Díaz et al., 2016). The adhesion of SL to Caco-2 cells varied between 0.05 and 0.3%, and only one of 21 SL strains had a high invasion of 0.2% while all others were below 0.1% invasion in Caco-2 cells (Table 5) (Counihan et al., 2015). However, SGG, but not SGM and SE, was able to translocate across differentiated epithelial monolayers (Boleij et al., 2011b), suggesting species and strain-dependent abilities to invade and translocate epithelial cells as well as invade endothelial cells (Vollmer et al., 2010). Similarly, several SL isolates were able to invade HUVEC endothelial cells. In general, encapsulated strains were found to be more invasive, which further correlated with the presence of a hyaluronic acid capsule (Counihan et al., 2015). Some SL strains even have a mucoid appearance, which in other streptococci is associated with greater degrees of tissue necrosis and bacteremia (Counihan et al., 2015).
Survival in Blood and Macrophages and Activation of the Human Contact System
Survival in blood, activation of the human contact system and escape from the immune system are pivotal for infection establishment. For survival in blood, hemolysis can be a beneficial ability for bacteria to gain access to iron (Malachowa and DeLeo, 2011). Genome analysis of SBSEC members suggests the presence of multiple virulence factors related to hemolysis including hemolytic toxin CylZ as well as hemolysins TLY, III and A family protein (Jin et al., 2013; Papadimitriou et al., 2014). In vitro experiments on survival in blood however featured controversial data likely linked to SBSEC-species dependency. While serum isolated from heparin-treated blood had little bactericidal effect on most SBSEC strains, filament-forming SB strains had a much higher sensitivity to serum (Lorian and Atkinson, 1978). In contrast to heparin-treated blood, SGG and some SII demonstrate high survival and growth in citrate-treated blood similar to Streptococcus pyogenes AP1. Pil1 and Pil3 mutants seemed to have little effect on survival in blood whereas a capsule deficient mutant (ΔcpsD) showed reduced but not significant lower survival (Isenring et al., 2018).
SGG, but not SII, is able to activate the human contact system at the bacterial surface. The human contact system is comprised of a cascade of factors including serine proteases factor XI, XII and plasma prekallikrein. The co-factor high molecular weight kininogen and later plasma kallikrein are involved in degrading kininogen to liberate pro-inflammatory bradykinin (Isenring et al., 2018). This ability is linked to the presence of the pil1 locus in the bacteria and the bacteria cell capsule encoded by multiple genes, one of which being cpsD. Pil1, particularly the Pil1A protein of this pilus, is able to bind factor XII and alter the host blood coagulation cascade. Mutants deprived of pil1 or pil3 showed decreased activity on factor XII and plasma kallikrein. An inverse correlation was observed for cpsD. SGG capsule mutant ΔcpsD showed increased activity on factor XII, plasma kallikrein and activated partial thromboplastin-time prolongation suggesting enhanced interaction between Pil1 and these factors if not hindered by the capsule. The interference with the human contact system and coagulation cascade by SGG suggests that Pil1 is an important factor for the establishment of IE in humans (Isenring et al., 2018).
Intracellular survival in macrophages and multiplication are important additional virulence mechanisms of multiple pathogens. SGG was able to survive in macrophages for up to 24 h and SL was shown to survive up to 48 h in macrophages (Counihan et al., 2015) whereas most Lactobacillus, Lactococcus and Bacillus subtilis are efficiently killed within this time-frame (Boleij et al., 2011b; Counihan et al., 2015). The presence of pil1 and cpsD seems to affect macrophage survival. Non-piliated SGG mutants had high survival rates to phagocytosis whereas pil1 overexpressing mutants and the ΔcpsD mutant were killed more efficiently (Isenring et al., 2018). This supports the protective effect of the capsule against phagocytosis and the impact of pilus expression and degree of piliation on immune system interactions (Danne et al., 2014; Martins et al., 2016). The addition of purified Pil1 antibodies increased the uptake of pil1 overexpressing but not of Δpil1 bacteria by THP-1 macrophages. The opsonized SGG were mainly the pil1 high expressing bacteria and not the weakly piliated cells. This suggests phase variation of pil1 an important immune evasion mechanism to reach the blood system (Danne et al., 2014; Martins et al., 2015, 2016). Phase variation of pil1 has important practical implications regarding silent infections. Patients can be asymptomatically affected by SBSEC bacteremia including the potential risk for undetected underlying CRC or adenomas (Haimowitz et al., 2005; Lin et al., 2011a; Lee et al., 2013). Pili are however not the only factor involved in immune system interaction. The rapid killing and cytokine induction of a non-piliated low virulent SGG strain lacking pil1 and pil3 (DSM16831T isolated from koala feces) in contrast to the survival of SGG UCN34Δpil1 suggests that further factors besides pil1 are involved in immune system interactions and the establishment of infection (Grimm et al., 2017b).
Pigeon and duckling infection models allow correlating the in vitro and in vivo findings. In the pigeon infection models, the highly virulent SGG strain STR357 was found intracellularly in 2–20% of macrophages counted in the spleen 96 h post-infection. SGG seemed to be actively multiplying. However, the exact mechanism of SGG to avoid killing by macrophages is not yet clear. Observations of free SGG outside phagosomes suggest a phagosome escape mechanism rather than the inhibition of phagosome-lysosome fusion (De Herdt et al., 1995).
In a duckling infection model, SGP resulted in meningitis and neurological symptoms 3 days post-infection. Macrophages were hypertrophic with abundant replicating SGP within phagosomes of degenerating macrophages causing necroptosis (Li et al., 2013). Based on these data SGG, SGP, and SL are capable of surviving in the blood stream, resisting phagocytosis and evading the immune system with strong dependency on the extracellular capsule, pili phase variation and so far unknown virulence factors.
Causation of Infective Endocarditis
Native-valve IE is caused by the colonization of damaged endothelial tissue of heart valves by bacteria such as Staphylococcus aureus, enterococci or SBSEC members (Hoen and Duval, 2013). SBSEC members are suspected to cause significant endocardial endothelial damage (Schoemaker et al., 1994). The minimal infectious dose to reach a lethal dose (LD90) in an experimental rat IE model was determined to be 104 CFU and is comparable to the LD90 of other IE pathogens (Danne et al., 2011). Important factors for the establishment of IE are binding of the bacteria to the endocardium and formation of a biofilm to shield microbes from immune attacks. The injured heart valve is recruited by platelets and fibrin, but also collagen was shown to be present in sterile injured valves. These sterile plaques (nonbacterial thrombotic endocarditis) can be colonized by pathogens during bacteremia. SB have a high adherence capability to normal valves, but an even 5-times increased binding to damaged aortic valves. Pil1 and Pil3 likely mediate this binding. Pil1, a collagen-binding adhesin, was shown to be important for IE establishment in a rat IE model and required for the initial establishment of an endocardial vegetation (Danne et al., 2011). Pil3 is of importance due to its ability to bind to fibrinogen. Due to platelet recruitment to injured sites, pil3-expressing strains have significantly improved ability to colonize the injured endocardium (Martins et al., 2016). Glucan-producing abilities of strains might further enhance binding to the endocardium. Treatment with dextranase inhibited the binding of glucan-positive strains, but not of glucan-negative strains, showing that the capsular glucan presence can be of significant importance for the binding efficiency of SB to injured sites (Ramirez-Ronda, 1978).
Further propagation of IE is linked again to the human contact system involving the coagulation cascade and the potent pro-inflammatory peptide bradykinin (Isenring et al., 2018). SGG UCN34 seems to prolong the intrinsic coagulation time by binding and activating factors of the human contact system on its surface. These findings were strongly related to Pil1. SII usually only possesses Pil3 explaining its inability to bind factor XII or plasma kallikrein, possibly reducing the overall impact of SII vs. SGG on coagulation time prolongation in the blood system. Likely, only wildtype SGG possess the capability to fully degrade kininogen in contrast to mutants with deletion of pili (Δpil1 or Δpil3) or capsule (ΔcpsD) and overexpression of pil1 (Δterm). Since kininogen degradation leads to the release of bradykinin, also bradykinin release was significantly reduced in all mutant strains although not completely inhibited (Isenring et al., 2018). This supports the major role of Pil1 in the pathogenesis of SGG. Pathogenesis is likely also influenced by other factors including Pil3, glucan producing abilities and the capsule. Additional factors and their roles in pathogenesis in SGG and other SBSEC members, particularly that of Pil2, are still unknown. Based on current knowledge, this resulted in the postulation of the following IE model for SGG: SGG upon entering into the bloodstream can survive and multiply. Subsequently, the coagulation cascade is activated leading to a procoagulant state. The bacteria can then adhere to exposed collagen, most likely type I, on heart valves via Pil1-binding. This further activates contact factors at the bacterial surface and via the contact system cascade, leads to a release of bradykinin that binds to its receptor to trigger IE (Isenring et al., 2018).
Clinical Infections and Host-Immune Response Due to SBSEC in Animals and Humans
Causation of IE and Bacteremia
SBSEC members are placed among the top-five causes for human IE globally and responsible for up to 6% of confirmed IE cases (Hoen et al., 2005; Vogkou et al., 2016). SBSEC as causal agents of IE increased from 10% to over 20% of all streptococcal IE cases in Europe and the USA with a particular hotspot in France (Hoen et al., 2005). Of the SBSEC bacteremia cases, 47% developed into IE (Barnham and Weightman, 2004). SBSEC IE is particularly common in patients with congenital heart defects, prosthetic valves, heart-care associated cross infections, diabetes, living in rural areas and also increasingly affects healthy valves of subjects >65 years (Barrau et al., 2004; Corredoira et al., 2008; Vogkou et al., 2016).
SGG seems to be the main causative agent of IE in humans in comparison with SGP, SII and SL. Between seven to over 50% of cases are attributed to SGG followed by 17–30% to SGP and roughly 30% to SL with strong regional differences (Beck et al., 2008; Boleij et al., 2011c; Romero et al., 2011; Lazarovitch et al., 2013; Sheng et al., 2014; Marmolin et al., 2016; Ben-Chetrit et al., 2017).
SBSEC members are also of high importance for bacteremia and IE in animals (Jans et al., 2015). In pigeons, SGG and SGP were found as the etiological agent for bacteremia and IE causing spontaneous infections that result in liver-, kidney- and spleen-swelling (Devriese et al., 1990, 1998). The preferred route of infection is suggested to be the GIT (Kimpe et al., 2003). SL has been associated with bacteremia and IE in sea otters (Counihan-Edgar et al., 2012; Counihan et al., 2015) and is often isolated from the GIT, cardiac valve lesions, heart, blood, brain, and other organs of sea otters that died due to IE, but hardly ever from the GIT of healthy sea otters. Still, the GIT is suggested as the preferred route of entry for SL (Counihan et al., 2015). The disease shows high similarity with IE in humans, which suggests similar patterns in infection establishment, and thus the need for common concepts that help to identify SBSEC-specific virulence factors.
Association of IE With Underlying CRC and Hepatobiliary Disorders
SBSEC bacteremia and IE have a much stronger association with colon neoplasia than tumor colonization of SBSEC. A meta-analysis clearly indicated that especially SGG IE has a very high association with neoplasia (OR 7.26, CI 3.94–13.36) compared to SB biotype II species. The median prevalence of neoplasia in SB infected patients was 60%, and was higher for adenomas (43%) than for carcinomas (18%). SB infection could therefore be predominantly associated with premalignant colonic lesions, which underscores the importance of colon examination for colon pathologies (Boleij et al., 2009b, 2011c). This is supported by the fact that most neoplasias are identified during the episode of bacteremia in surveillance studies. Surveillance of SGG bacteremia patients resulted in 57 new neoplasias (seven carcinomas and 23 advanced adenomas) in 232 patients (24.6%) with a mean follow-up of 41.8 months. In Clostridium septicum bacteremia patients, no new neoplasias were found with a mean follow-up of 37.4 months (Corredoira et al., 2017). Retrospective analysis of patients with a previous SB bacteremia revealed the detection of three CRCs and seven adenomas in 14 patient that underwent colonoscopy (72% detection rate) (McKenna et al., 2011) strongly supporting the association of SBSEC IE with gut pathologies. This might support the theory that SGG bacteremia frequently has an intestinal source and is a marker for intestinal SGG colonization.
Stratified by SBSEC species (Table 6), SGG associates with colon pathology in 28.6–70.7% of cases varying from 16.1 to 52.5% for adenomas and 3.6–33.3% for carcinomas (Tripodi et al., 2004; Beck et al., 2008; Romero et al., 2011; Corredoira et al., 2013b, 2017; Lazarovitch et al., 2013; Sheng et al., 2014; Ben-Chetrit et al., 2017), SGP associates with 0.0–45.8% colon pathologies in 200 cases of which 126 were reported from Asia (Beck et al., 2008; Romero et al., 2011; Lazarovitch et al., 2013; Sheng et al., 2014; Ben-Chetrit et al., 2017). For SL, the data is more controversial ranging from 0.0 to 50.0% for colon pathologies, 21.4 and 33.3% reported by 2 studies for adenomas and only 1 study reported a rate of 7.1% for carcinomas. Similarly, data for SII and SGM are very limited showing an overall neoplasia rate of 14.3% for SII and 100% for SGM, often relying on a few single cases (Table 6) (Beck et al., 2008; Romero et al., 2011; Lazarovitch et al., 2013; Sheng et al., 2014; Ben-Chetrit et al., 2017). Meta-analysis of these studies together revealed colon pathology for SBSEC in 40.7% (n = 786) and divided by subspecies for SGG in 51.7% (n = 462), SGP in 24.5% (n = 200) and SL in 24.2% (n = 33) (Ben-Chetrit et al., 2017). However, in all these studies there was no control group reflecting the prevalence of colorectal adenomas in the general population >65 years of age that is estimated to be between 10 and 25% (Boleij et al., 2011c).
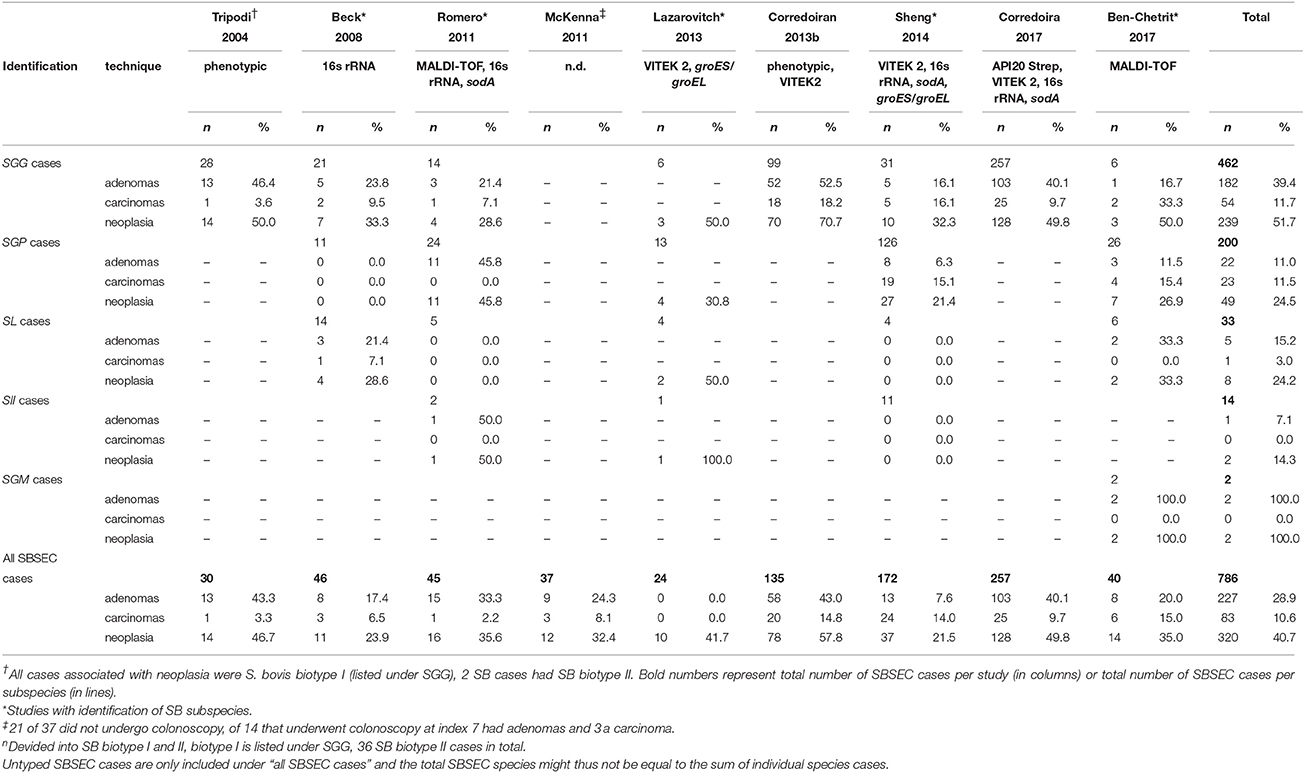
Table 6. Association of SBSEC subspecies clinical infections (bacteremia and IE) with underlying colon pathology.
Interestingly, it is suggested that SII has a different association with neoplasia than SGG. SGG is more often associated with smaller asymptomatic non-advanced and advanced adenomas, while SII is generally an uncommon observation but more likely associated with larger tumors at a more advanced stage (Corredoira et al., 2013a; Stein, 2013). Based on this meta-analysis, this seems not the case as no carcinomas were found in any of the SII cases and adenomas and carcinomas were found almost equally in SGP (11.0 vs. 11.5%, respectively), although the association of SGG with adenomas was confirmed in five of seven studies (39.4 vs. 11.7% in carcinomas; Table 6) (Tripodi et al., 2004; Beck et al., 2008; Romero et al., 2011; Corredoira et al., 2013b, 2017).
Besides the obvious association with neoplasia, hepatobiliary disorders such as chronic liver diseases and liver cirrhosis seem to be highly prevalent among SBSEC IE patients (Gonzlez-Quintela et al., 2001; Tripodi et al., 2004). About 20% of all SB bacteremia cases have an association with underlying biliary origin of which 59% was isolated from polymicrobial infections, most frequently in combination with Escherichia coli and Enterococcus spp (Corredoira et al., 2014). The specific species associations with hepatobiliary disorders vary for SGP (10–83%) and SL (25–60%) followed by SGG (2–35%) and rarely for SII, SGM and S. alactolyticus (Beck et al., 2008; Romero et al., 2011; Lazarovitch et al., 2013; Corredoira et al., 2014; Sheng et al., 2014; Toepfner et al., 2014; Almeida et al., 2016). In rare cases, SB was obtained from cerebrospinal fluid and blood culture of patients with liver cirrhosis that developed bacterial meningitis (Barahona-Garrido et al., 2010). Urinary tract infections are a further infection caused by SBSEC members, particularly SGP, which are responsible for nearly 10% of cases in Vietnam (Poulsen et al., 2012).
Immunogenic Molecules and Antibody Response
Lessons From Animal Vaccination
SBSEC colonization and infection leads to immune responses by the host recognizing immunogenic molecules on SBSEC. Some of these immunogenic molecules have been discovered by the development of vaccines for SB in cattle. Parental immunization with SB using three boosters reduced clinical signs of lactic acidosis in cattle and has been a partly effective strategy. Feed intake in immunized sheep and steers was higher, there was a lower prevalence of diarrhea and they all survived grain challenge that results in the drop of rumen pH (Shu et al., 1999, 2000a; Gill et al., 2000). This immunization strategy resulted in a long-lasting serum and saliva anti-SB IgG response in steers and sheep. Although effective antibody levels were generated, it did not result in complete removal of SB from the rumen (Gill et al., 2000; Shu et al., 2000a,b, 2001). Feeding trials in mice using mozzarella cheese prepared from SGM or SGM in combinations with S. thermophilus and Lactobacillus bulgaricus as well as only S. thermophilus and Lb. bulgaricus also increased IL1-β levels for all combinations. However, only cheese prepared with SGM increased TNF-α and IL6 suggesting immune stimulation by SGM without inducing significant differences in mice body weight gain, spleen index and thymus index (Cho et al., 2012).
The bacteria cell surface likely plays a crucial role in SBSEC antigenicity. SB of CRC patients harbor an immunogenic surface molecule comparable to human choriogonadotropin. Vaccines prepared with chemically killed SB containing this human choriogonadotropin-like material on the cell surface have immunogenic properties and elicited antibody responses in rabbits. These antibodies also reacted with the human trophoblastic hormone and were similar to antibodies produced by human choriogonadotropin (Domingue et al., 1986; Acevedo et al., 1987). Furthermore, capsule and cell wall may contribute to the antigenicity of SB. This was observed via a lower cross-reactivity index for encapsulated strains (range 9.4–12.4%) than non-encapsulated strains (range 28.9–56.1%). This suggested antigenicity of capsule components and therefore a potential contribution of capsule and cell wall to the antigenicity of SB. In fact, colonization and antibody response of pigeons immunized with an SGP strain containing a thin irregular capsule (PDH 827) did not protect against a high-virulent strain SGG strain with a thick capsule (STR357). These findings indicate that supernatant proteins, glycans, fimbriae and the capsule may be involved in the induction of protective immunity against SGG infections but likely not across SBSEC species (Kane and Karakawa, 1969; Kane et al., 1972; Pazur and Forsberg, 1978; Pazur et al., 1978; Vanrobaeys et al., 1997; Kimpe et al., 2002).
Humoral Reactions in IE
During establishment of IE, the immune system tries to clear the infection with SB. In several studies, the specific humoral response to SB has been investigated by crossing isolated pathogens or sterilized antigen compounds with the patients serum. By using SGG NCTC8133, 11 antigens between 41 and 130 kDa were detected via immunoblotting of two IE patient sera. Both IE patients produced strong IgG responses to many bands with a limited cross-reaction to enterococci (Burnie et al., 1987), but also considerable variation was found between SE and SB biotype II (SII, SL, or SGP) (Darjee and Gibb, 1993). The presence of antibody responses to common (c) antigen was specific to the SB isolates, but not to other Gram-positive IE isolates. This antigen did not lose its antigenicity by trypsin treatment or boiling, but changes in the pH from 7 to 5 had a significant reversible impact on antigenicity (Kaplan et al., 1983). In pigeons, a similar approach identified a 114-kDa immunogenic protein that was only recognized on highly virulent SGGs and two proteins (115 and 207 kDa) that were only recognized on low virulent SGG strains (Vanrobaeys et al., 2000b). Furthermore, antibodies to SGG are also common in the low molecular weight proteome. In the wall-extracted antigens, HlpA and RPL7/L12 have been identified as potential immunogenic proteins. These anchorless surface proteins might have important functions for host-microbe interactions as observed for the surface-exposed enolase from Streptococcus pneumoniae in tissue invasion (Tjalsma et al., 2007).
SBSEC Humoral Response for Detection of CRC
Because of the specific antibody response to SBSEC and the strong correlation with colorectal adenomas and carcinomas, several studies have tried to identify adenoma and CRC patients utilizing the specific antibody response to SBSEC in general or to specific SBSEC surface proteins. The IgG antibody responses to whole cell extracted protein preparations from SE and SB were higher in patients with CRC than controls, but not for IgM titers (Darjee and Gibb, 1993). A similar approach with immunoblot of whole antigen extracts showed specific proteins at 22 kDa band that was associated with neoplasia (OR 7.98; 95% CI: 3.54–17.93) and in combination with a 30-kDa band resulted in an OR of 22.37 (CI 3.77–131.64) with a very high specificity (84.9% 22 kDa and 98.1% 30 kDa) and lower sensitivity (58.6% 22kDa and 30.1% 30 kDa) (Garza-González et al., 2012). These data were confirmed for wall-extracted antigen extracts from SGG showing increased seroprevalence in CRC and adenoma patients compared to colonoscopy controls (Abdulamir et al., 2009).
Specific approaches to antigenic proteins revealed that HlpA and RpL7/L12 wall-extracted antigen proteins from SGG were diagnostic for CRC (Tjalsma et al., 2006; Tjalsma, 2010). A detailed analysis revealed a positive correlation of anti-RpL7/L12 levels with age and the presence of colon polyps. Both adenoma and stage I/II CRC patients contained the highest anti-RpL7/L12 titers, which were significantly different from those of healthy individuals and advanced stage CRC patients, suggesting a temporal relation with early stage colonic lesions. A drawback of RpL7/L12 is the conserved nature within the bacterial kingdom and significant cross-reactivity resulting in significant overlap in serum from healthy controls and CRC patients (Boleij et al., 2010).
The IgG response to four SGG pili proteins (Pil1A, Pil1B, Pil2B, and Pil3B) was tested in single plex and multiplex assays. The IgG response to Pil1B was the best predictor for tumor presence, but did not result in the same response in all IE infected patients. CRC patients generally had response to only one of the four antigens with a maximum sensitivity of 20–43% by combination of four antigens (Boleij et al., 2012b). In the multiplex assay, the strongest association with CRC seropositivity was found by a combination of Pil1A and B (OR 3.54; 95% CI 1.49–8.44) and it was more predictive in patients with an age below 65 years. Although very specific, only a limited number of CRC cases could be identified using this approach (Butt et al., 2016). The association with Pil1B was confirmed in an independent cohort (OR 4.3; CI 2.14–8.65), but not for Pil1A. Interestingly, a specific association with non-advanced adenomas was found using a 6-marker panel with at least a positive seroresponse to two of six markers (OR 2.98; 95% CI: 1.18–7.57). With this marker panel, 27% of SGG-positives cases among non-advanced adenomas were identified compared to 11% of controls. Additionally, 7% of non-advanced adenomas were double-positive to Pil1A-Pil1B, compared to 0% of controls (p-value < 0.0001) (Butt et al., 2017). Data from a recent nested case-control study confirm previous observations that single marker panels are not sufficient to identify CRC patients and adenomas (Butt et al., 2017, 2018). This is potentially due to significant heterogeneity in IgG response between patients and depends on the antigenicity of the strain triggering the immune response. Nevertheless, these three studies confirm the strong and specific association of SGG with CRC and adenomas of the colon.
Interpretation and Conclusion
SBSEC pathobionts inhabit the GIT of animals and humans as well as food products featuring significant niche adaptation. While SGG retained most genetic and functional properties for a large variety of niches, SGP, SL, SII and particularly SGM have adapted more specifically to certain niches by gene gain and loss. SBSEC colonization as part of the rumen and gut microbiota is not ubiquitous and confers only a small sub-population of the overall microbiota during symbiosis. However, their ability to proliferate and colonize in dysbiosis, e.g., ruminal acidosis or in the tumor microenvironment is significantly enhanced and defines their pathogenicity.
Although SBSEC are not often found in association with tumors in microbiome studies, they likely have increased colonization levels at tumor sites. A potential tumor promoting ability of SGG may shape their microenvironment further and support the association with neoplastic sites. Increased immune responses in CRC patients toward SGG suggests a strong association with these sites. Considering the bacterial driver-passenger model, it seems that particularly SGG could be considered as a neoplastic site hijacker. SGG only excels when colonization factors and growth conditions in the environment are favorable. SGG is then able to further shape the microenvironment to its benefit.
This could also explain controversies between the relation of SBSEC with CRC in IE matched and unmatched patients. As SBSEC members are not strong ubiquitous gut colonizers and nonobligatory passengers on CRC, their prevalence among the general CRC population is low in contrast to patients with SBSEC IE. This also supports the proposed route of infection in humans and animals alike: SBSEC members, mostly SGG, are only able to colonize neoplastic tissues selectively. Subsequently, some strains have the capacity to translocate across the epithelial barrier into the blood system. Human blood-derived SGG possess the full pil1, pil2, and pil3 loci, which enable adhesion to endothelial cells, exposed collagen and fibrinogen on heart valves and activate the human contact system. Via pili phase variation and expression of a capsular polysaccharide they can survive phagocytosis and minimize activation of the humoral immune response. SGG strains of animal origin and other SBSEC members without full pilus loci or different pilus proteins likely do not possess the same degree of infection ability and might depend on other factors to establish infection. Thus, animal and food-derived strains often adhere best to collagen type IV, whereas human-derived strains, particularly those of bacteremia or IE origin, predominantly show highest adhesion toward collagen type I. However, data is so far limited to presumed commensal animal strains whereas animal pathogens are not characterized leaving open question regarding virulence factors, host specificity but also zoonosis. In the light of SBSEC zoonotic potential, association with rural areas and prevalence in food, it will be relevant to determine the factors that enable colonization and establishment in animal and human hosts alike and provide comparison to food-derived strains. This will greatly contribute to a better understanding of transmission routes and development of intervention strategies to mitigate health risks associated with individual SBSEC members.
Author Contributions
CJ and AB equally shared all aspects of topic development, analysis, interpretation, manuscript writing and revision. Both authors have read and approved the submitted version of the manuscript. Both authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Funding
AB is funded by the Dutch scientific organization (NWO; Veni-scheme grant no. 016.166.089). CJ was funded in part via the ERAfrica New Ideas grant.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
ERAfrica Safe Dairy Consortium for scientific advice received: Dasel Wambua Mulwa Kaindi, Jan Hattendorf, Leo Meile, Bernd Kreikemeyer, Pierre Renault, Bassirou Bonfoh, Wambui Kogi-Makau, Godfrey N. Lule, Christophe Lacroix, Esther Schelling, and Jakob Zinsstag. Harold Tjalsma, Shaynoor Dramsi, Julia Butt, Michael Pawlita, Ikuko Kato, Juan Corredoira, and Cynthia Sears for great advise, discussion, and collaborations regarding SBSEC.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmicb.2018.00603/full#supplementary-material
References
Abdulamir, A. S., Hafidh, R. R., and Bakar, F. (2010). Molecular detection, quantification, and isolation of Streptococcus gallolyticus bacteria colonizing colorectal tumors: inflammation-driven potential of carcinogenesis via IL-1, COX-2, and IL-8. Mol. Cancer 9:249. doi: 10.1186/1476-4598-9-249
Abdulamir, A. S., Hafidh, R. R., Mahdi, L. K., Al-Jeboori, T., and Abubaker, F. (2009). Investigation into the controversial association of Streptococcus gallolyticus with colorectal cancer and adenoma. BMC Cancer 9:403. doi: 10.1186/1471-2407-9-403
Acevedo, H. F., Pardo, M., Campbell-Acevedo, E., and Domingue, G. J. (1987). Human choriogonadotropin-like material in bacteria of different species: electron microscopy and immunocytochemical studies with monoclonal and polyclonal antibodies. Microbiology 133, 783–791. doi: 10.1099/00221287-133-3-783
Al-Jashamy, K., Murad, A., Zeehaida, M., Rohaini, M., and Hasnan, J. (2010). Prevalence of colorectal cancer associated with Streptococcus bovis among inflammatory bowel and chronic gastrointestinal tract disease patients. Asian Pac. J. Cancer Prev. 11, 1765–1768.
Almeida, P., Railsback, J., and Gleason, J. B. (2016). A rare case of Streptococcus alactolyticus infective endocarditis complicated by septic emboli and mycotic left middle cerebral artery aneurysm. Case Rep. Infect. Dis. 2016:9081352. doi: 10.1155/2016/9081352
Andres-Franch, M., Galiana, A., Sanchez-Hellin, V., Ochoa, E., Hernandez-Illan, E., Lopez-Garcia, P., et al. (2017). Streptococcus gallolyticus infection in colorectal cancer and association with biological and clinical factors. PLoS ONE 12:e0174305. doi: 10.1371/journal.pone.0174305
Arul, A. S., and Palanivelu, P. (2014). Biofilm forming ability of a new bacterial isolate from dental caries: an atomic force microscopic study. J. Nat. Sci. Biol. Med. 5, 278–283. doi: 10.4103/0976-9668.136162
Asanuma, N., and Hino, T. (1997). Tolerance to low pH and lactate production in rumen bacteria. Anim. Sci. Technol. 68, 367–376.
Aymeric, L., Donnadieu, F., Mulet, C., du Merle, L., Nigro, G., Saffarian, A., et al. (2018). Colorectal cancer specific conditions promote Streptococcus gallolyticus gut colonization. Proc. Natl. Acad. Sci. U.S.A. 115, e283–e291. doi: 10.1073/pnas.1715112115
Baele, M., Devriese, L. A., and Haesebrouck, F. (2001). Lactobacillus agilis is an important component of the pigeon crop flora. J. Appl. Microbiol. 91, 488–491. doi: 10.1046/j.1365-2672.2001.01407.x
Bailey, R. W. (1959). Transglucosidase activity of rumen strains of Streptococcus bovis; structure of the dextran produced from sucrose. Biochem. J. 71, 23–26. doi: 10.1042/bj0710023
Bailey, R. W. (1963). The intracellular alpha-galactodidase of a rumen strain of Streptococcus bovis. Biochem. J. 86, 509–514. doi: 10.1042/bj0860509
Bailey, R. W., and Oxford, A. E. (1958). A quantitative study of the production of dextran from sucrose by rumen strains of Streptococcus bovis. J. Gen. Microbiol. 19, 130–145. doi: 10.1099/00221287-19-1-130
Barahona-Garrido, J., Hernández-Calleros, J., Téllez-Ávila, F. I., Chávez-Tapia, N. C., Remes-Troche, J. M., and Torre, A. (2010). Bacterial meningitis in cirrhotic patients. J. Clin. Gastroenterol. 44, e218–e223. doi: 10.1097/MCG.0b013e3181d88d53
Barnes, I. J., Seeley, H. W., and Vandemark, P. J. (1961). Nutrition of Streptococcus bovis in relation to dextran formation. J. Bacteriol. 82, 85–93.
Barnham, M. R., and Weightman, N. C. (2004). Changing incidence of detected streptococcal bacteraemia in North Yorkshire, England. Indian J. Med. Res. 119(Suppl.), 160–163.
Barrau, K., Boulamery, A., Imbert, G., Casalta, J. P., Habib, G., Messana, T., et al. (2004). Causative organisms of infective endocarditis according to host status. Clin. Microbiol. Infect. 10, 302–308. doi: 10.1111/j.1198-743X.2004.00776.x
Beck, M., Frodl, R., and Funke, G. (2008). Comprehensive study of strains previously designated Streptococcus bovis consecutively isolated from human blood cultures and emended description of Streptococcus gallolyticus and Streptococcus infantarius subsp. coli. J. Clin. Microbiol. 46, 2966–2972. doi: 10.1128/JCM.00078-08
Beckmann, L., Simon, O., and Vahjen, W. (2006). Isolation and identification of mixed linked beta -glucan degrading bacteria in the intestine of broiler chickens and partial characterization of respective 1,3-1,4-beta -glucanase activities. J. Basic Microbiol. 46, 175–185. doi: 10.1002/jobm.200510107
Ben-Chetrit, E., Wiener-Well, Y., Kashat, L., Yinnon, A. M., and Assous, M. V. (2017). Streptococcus bovis new taxonomy: does subspecies distinction matter? Eur. J. Clin. Microbiol. Infect. Dis. 36, 387–393. doi: 10.1007/s10096-016-2814-6
Biarc, J., Nguyen, I. S., Pini, A., Gossé, F., Richert, S., Thiersé, D., et al. (2004). Carcinogenic properties of proteins with pro-inflammatory activity from Streptococcus infantarius (formerly S. bovis). Carcinogenesis 25, 1477–1484. doi: 10.1093/carcin/bgh091
Boleij, A., Dutilh, B. E., Kortman, G. A., Roelofs, R., Laarakkers, C. M., Engelke, U. F., et al. (2012a). Bacterial responses to a simulated colon tumor microenvironment. Mol. Cell Proteomics 11, 851–862. doi: 10.1074/mcp.M112.019315
Boleij, A., Laarakkers, C. M., Gloerich, J., Swinkels, D. W., and Tjalsma, H. (2011a). Surface-affinity profiling to identify host-pathogen interactions. Infect. Immun. 79, 4777–4783. doi: 10.1128/IAI.05572-11
Boleij, A., Muytjens, C. M., Bukhari, S. I., Cayet, N., Glaser, P., Hermans, P. W. M., et al. (2011b). Novel clues on the specific association of Streptococcus gallolyticus subsp gallolyticus with colorectal cancer. J. Infect. Dis. 203, 1101–1109. doi: 10.1093/infdis/jiq169
Boleij, A., Roelofs, R., Danne, C., Bellais, S., Dramsi, S., Kato, I., et al. (2012b). Selective antibody response to Streptococcus gallolyticus pilus proteins in colorectal cancer patients. Cancer Prev. Res. (Phila) 5, 260–265. doi: 10.1158/1940-6207.CAPR-11-0321
Boleij, A., Roelofs, R., Schaeps, R. M., Schülin, T., Glaser, P., Swinkels, D. W., et al. (2010). Increased exposure to bacterial antigen RpL7/L12 in early stage colorectal cancer patients. Cancer 116, 4014–4022. doi: 10.1002/cncr.25212
Boleij, A., Schaeps, R. M., De Kleijn, S., Hermans, P. W., Glaser, P., Pancholi, V., et al. (2009a). Surface-exposed histone-like protein A modulates adherence of Streptococcus gallolyticus to colon adenocarcinoma cells. Infect. Immun. 77, 5519–5527. doi: 10.1128/IAI.00384-09
Boleij, A., Schaeps, R. M. J., and Tjalsma, H. (2009b). Association between Streptococcus bovis and colon cancer. J. Clin. Microbiol. 47:516. doi: 10.1128/JCM.01755-08
Boleij, A., and Tjalsma, H. (2013). The itinerary of Streptococcus gallolyticus infection in patients with colonic malignant disease. Lancet Infect. Dis. 13, 719–724. doi: 10.1016/S1473-3099(13)70107-5
Boleij, A., van Gelder, M., Swinkels, D. W., and Tjalsma, H. (2011c). Clinical importance of Streptococcus gallolyticus infection among colorectal cancer patients: systematic review and meta-analysis. Clin. Infect. Dis. 53, 870–878. doi: 10.1093/cid/cir609
Boltin, D., Goldberg, E., Bugaevsky, O., Kelner, E., Birkenfeld, S., Gingold-Belfer, R., et al. (2015). Colonic carriage of Streptococcus bovis and colorectal neoplasia: a prospective 17-year longitudinal case-control study. Eur. J. Gastroenterol. Hepatol. 27, 1449–1453. doi: 10.1097/MEG.0000000000000466
Brouwer, S., Barnett, T. C., Rivera-Hernandez, T., Rohde, M., and Walker, M. J. (2016). Streptococcus pyogenes adhesion and colonization. FEBS Lett. 590, 3739–3757. doi: 10.1002/1873-3468.12254
Burnie, J. P., Holland, M., Matthews, R. C., and Lees, W. (1987). Role of immunoblotting in the diagnosis of culture negative and enterococcal endocarditis. J. Clin. Pathol. 40, 1149–1158. doi: 10.1136/jcp.40.10.1149
Butt, J., Jenab, M., Willhauck-Fleckenstein, M., Michel, A., Pawlita, M., Kyrø, C., et al. (2018). Prospective evaluation of antibody response to Streptococcus gallolyticus and risk of colorectal cancer. Int. J. Cancer. doi: 10.1002/ijc.31283. [Epub ahead of print].
Butt, J., Romero-Hernández, B., Pérez-Gómez, B., Willhauck-Fleckenstein, M., Holzinger, D., Martin, V., et al. (2016). Association of Streptococcus gallolyticus subspecies gallolyticus with colorectal cancer: serological evidence. Int. J. Cancer 138, 1670–1679. doi: 10.1002/ijc.29914
Butt, J., Werner, S., Willhauck-Fleckenstein, M., Michel, A., Waterboer, T., Zörnig, I., et al. (2017). Serology of Streptococcus gallolyticus subspecies gallolyticus and its association with colorectal cancer and precursors. Int. J. Cancer 141, 897–904. doi: 10.1002/ijc.30765
Chen, H.-J., Tsai, J.-C., Chang, T.-C., Hung, W.-C., Tseng, S.-P., Hsueh, P.-R., et al. (2008). PCR-RFLP assay for species and subspecies differentiation of the Streptococcus bovis group based on groESL sequences. J. Med. Microbiol. 57, 432–438. doi: 10.1099/jmm.0.47628-0
Chen, L., Liu, S., Wang, H., Wang, M., and Yu, L. (2016). Relative significances of pH and substrate starch level to roles of Streptococcus bovis S1 in rumen acidosis. AMB Express 6:80. doi: 10.1186/s13568-016-0248-2
Cheng, K.-J., Hironaka, R., Jones, G. A., Nicas, T., and Costerton, J. W. (1976). Frothy feedlot bloat in cattle: production of extracellular polysaccharides and development of viscosity in cultures of Streptococcus bovis. Can. J. Microbiol. 22, 450–459. doi: 10.1139/m76-071
Chirouze, C., Patry, I., Duval, X., Baty, V., Tattevin, P., Aparicio, T., et al. (2013). Streptococcus bovis/Streptococcus equinus complex fecal carriage, colorectal carcinoma, and infective endocarditis: a new appraisal of a complex connection. Eur. J. Clin. Microbiol. Infect. Dis. 32, 1171–1176. doi: 10.1007/s10096-013-1863-3
Cho, S.-A., Kim, K.-S., and Lim, S.-D. (2012). Effects of mozzarella cheese manufactured by S. macedonicus LC743 on the immune status of mouse. Korean J. Food Sci. Anim. Resour. 32, 40–44. doi: 10.5851/kosfa.2012.32.1.40
Chow, J., Tang, H., and Mazmanian, S. K. (2011). Pathobionts of the gastrointestinal microbiota and inflammatory disease. Curr. Opin. Immunol. 23, 473–480. doi: 10.1016/j.coi.2011.07.010
Christensen, B. E. (1989). The role of extracellular polysaccharides in biofilms. J. Biotechnol. 10, 181–202. doi: 10.1016/0168-1656(89)90064-3
Corredoira, J., Alonso, M. P., García-Garrote, F., García-Pais, M. J., Coira, A., Rabuñal, R., et al. (2014). Streptococcus bovis group and biliary tract infections: an analysis of 51 cases. Clin. Microbiol. Infect. 20, 405–409. doi: 10.1111/1469-0691.12333
Corredoira, J., Alonso, M. P., Pita, J., and Alonso-Mesonero, D. (2008). Association between rural residency, group D streptococcal endocarditis and colon cancer? Clin. Microbiol. Infect. 14, 190–190. doi: 10.1111/j.1469-0691.2007.01913.x
Corredoira, J., Coira, A., Iñiguez, I., Pita, J., Varela, J., and Alonso, M. P. (2013a). Advanced intestinal cancer associated with Streptococcus infantarius (former S. bovis II/1) sepsis. Int. J. Clin. Pract. 67, 1358–1359. doi: 10.1007/s10096-013-1940-7
Corredoira, J. C., Alonso, M. P., García-País, M. J., Rabuñal, R., García-Garrote, F., López-Roses, L., et al. (2013b). Is colonoscopy necessary in cases of infection by Streptococcus bovis biotype II? Eur. J. Clin. Microbiol. Infect. Dis. 33, 171–177. doi: 10.1111/ijcp.12190
Corredoira, J., Grau, I., Garcia-Rodriguez, J. F., García-País, M. J., Rabuñal, R., Ardanuy, C., et al. (2017). Colorectal neoplasm in cases of Clostridium septicum and Streptococcus gallolyticus subsp. gallolyticus bacteraemia. Eur. J. Intern. Med. 41, 68–73. doi: 10.1016/j.ejim.2017.02.009
Counihan, K. L., Gill, V. A., Miller, M. A., Burek-Huntington, K. A., LeFebvre, R. B., and Byrne, B. A. (2015). Pathogenesis of Streptococcus infantarius subspecies coli isolated from sea otters with infective endocarditis. Comp. Immunol., Microbiol. Infect. Dis. 40, 7–17. doi: 10.1016/j.cimid.2015.03.002
Counihan-Edgar, K. L., Gill, V. A., Doroff, A. M., Burek, K. A., Miller, W. A., Shewmaker, P. L., et al. (2012). Genotypic characterization of Streptococcus infantarius subsp. coli isolates from sea otters with infective endocarditis and/or septicemia and from environmental mussel samples. J. Clin. Microbiol. 50, 4131–4133. doi: 10.1128/JCM.02581-12
Danne, C., Dubrac, S., Trieu-Cuot, P., and Dramsi, S. (2014). Single cell stochastic regulation of pilus phase variation by an attenuation-like mechanism. PLoS Pathog. 10:e1003860. doi: 10.1371/journal.ppat.1003860
Danne, C., Entenza, J. M., Mallet, A., Briandet, R., Débarbouillé, M., Nato, F., et al. (2011). Molecular characterization of a Streptococcus gallolyticus genomic island encoding a pilus involved in endocarditis. J. Infect. Dis. 204, 1960–1970. doi: 10.1093/infdis/jir666
Darjee, R., and Gibb, A. P. (1993). Serological investigation into the association between Streptococcus bovis and colonic cancer. J. Clin. Pathol. 46, 1116–1119. doi: 10.1136/jcp.46.12.1116
De Herdt, P., Haesebrouck, F., Charlier, G., Ducatelle, R., Devriese, L. A., and Vandenbossche, G. (1995). Intracellular survival and multiplication of virulent and less virulent strains of Streptococcus bovis in pigeon macrophages. Vet. Microbiol. 45, 157–169. doi: 10.1016/0378-1135(95)00035-9
De Herdt, P., Haesebrouck, F., Devriese, L. A., and Ducatelle, R. (1994a). Prevalence of Streptococcus bovis in racing pigeons. Vet. Q. 16, 71–74.
De Herdt, P., Haesebrouck, F., Ducatelle, R., Degroote, B., and Devriese, L. A. (1994b). Streptococcus bovis infections in pigeons - virulence of different serotypes. Vet. Microbiol. 41, 321–332.
Dekker, J. P., and Lau, A. F. (2016). An update on the Streptococcus bovis group: classification, identification, and disease associations. J. Clin. Microbiol. 54, 1694–1699. doi: 10.1128/JCM.02977-15
Devriese, L. A., Uyttebroek, E., Gevaert, D., Vandekerckhove, P., and Ceyssens, K. (1990). Streptococcus bovis infections in Pigeons. Avian Pathol. 19, 429–434. doi: 10.1080/03079459008418697
Devriese, L. A., Vandamme, P., Pot, B., Vanrobaeys, M., Kersters, K., and Haesebrouck, F. (1998). Differentiation between Streptococcus gallolyticus strains of human clinical and veterinary origins and Streptococcus bovis strains from the intestinal tracts of ruminants. J. Clin. Microbiol. 36, 3520–3523.
De Vuyst, L., and Tsakalidou, E. (2008). Streptococcus macedonicus, a multi-functional and promising species for dairy fermentations. Int. Dairy J. 18, 476–485. doi: 10.1016/j.idairyj.2007.10.006
Domingue, G. J., Acevedo, H. F., Powell, J. E., and Stevens, V. C. (1986). Antibodies to bacterial vaccines demonstrating specificity for human choriogonadotropin (hCG) and immunochemical detection of hCG-like factor in subcellular bacterial fractions. Infect. Immun. 53, 95–98.
Dumke, J., Hinse, D., Vollmer, T., Knabbe, C., and Dreier, J. (2014). Development and application of a multilocus sequence typing scheme for Streptococcus gallolyticus subsp. gallolyticus. J. Clin. Microbiol. 52, 2472–2478. doi: 10.1128/JCM.03329-13
Dumke, J., Hinse, D., Vollmer, T., Schulz, J., Knabbe, C., and Dreier, J. (2015). Potential transmission pathways of Streptococcus gallolyticus subsp. gallolyticus. PLoS ONE 10:e0126507. doi: 10.1371/journal.pone.0126507
Dumke, J., Vollmer, T., Akkermann, O., Knabbe, C., and Dreier, J. (2017). Case-control study: determination of potential risk factors for the colonization of healthy volunteers with Streptococcus gallolyticus subsp. gallolyticus. PLoS ONE 12:e0176515. doi: 10.1371/journal.pone.0176515
Ekinci, M. S., McCrae, S. I., and Flint, H. J. (1997). Isolation and overexpression of a gene encoding an extracellular β- (1,3-l,4)-glucanase from Streptococcus bovis JB1. Appl. Environ. Microbiol. 63, 3752–3756.
Ellmerich, S., Djouder, N., Schöller, M., and Klein, J. P. (2000a). Production of cytokines by monocytes, epithelial and endothelial cells activated by Streptococcus bovis. Cytokine 12, 26–31.
Ellmerich, S., Schöller, M., Duranton, B., Gossé, F., Galluser, M., Klein, J. P., et al. (2000b). Promotion of intestinal carcinogenesis by Streptococcus bovis. Carcinogenesis 21, 753–756.
Flint, S. H., Brooks, J. D., and Bremer, P. J. (1997). Use of the Malthus conductance growth analyser to determine numbers of thermophilic streptococci on stainless steel. J. Appl. Microbiol. 83, 335–339. doi: 10.1046/j.1365-2672.1997.00233.x
Flint, S. H., Ward, L. J., and Brooks, J. D. (1999). Streptococcus waius sp, nov., a thermophilic streptococcus from a biofilm. Int. J. Syst. Bacteriol. 49, 759–767. doi: 10.1099/00207713-49-2-759
Garza-González, E., Ríos, M., Bosques-Padilla, F. J., Francois, F., Cho, I., González, G. M., et al. (2012). Immune response against Streptococcus gallolyticus in patients with adenomatous polyps in colon. Int. J. Cancer 131, 2294–2299. doi: 10.1002/ijc.27511
Giannitsioti, E., Chirouze, C., Bouvet, A., Béguinot, I., Delahaye, F., Mainardi, J. L., et al. (2007). Characteristics and regional variations of group D streptococcal endocarditis in France. Clin. Microbiol. Infect. 13, 770–776. doi: 10.1111/j.1469-0691.2007.01753.x
Gill, H. S., Shu, Q., and Leng, R. A. (2000). Immunization with Streptococcus bovis protects against lactic acidosis in sheep. Vaccine 18, 2541–2548. doi: 10.1016/S0264-410X(00)00017-7
Gonzlez-Quintela, A., Martínez-Rey, C., Castroagudín, J. F., Rajo-Iglesias, M. C., Domínguez-Santalla, M. J., Gonzalez-Quintela, A., et al. (2001). Prevalence of liver disease in patients with Streptococcus bovis bacteraemia. J. Infect. 42, 116–119. doi: 10.1053/jinf.2001.0799
Grimm, I., Dumke, J., Dreier, J., Knabbe, C., and Vollmer, T. (2018). Biofilm formation and transcriptome analysis of Streptococcus gallolyticus subsp. gallolyticus in response to lysozyme. PLoS ONE 13:e0191705. doi: 10.1371/journal.pone.0191705
Grimm, I., Dumke, J., Vollmer, T., Hinse, D., Rückert, C., Kalinowski, J., et al. (2017a). Complete genome sequence of the Streptococcus gallolyticus subsp. gallolyticus strain DSM 16831. Genome Announc. 5, e00108–e00117. doi: 10.1128/genomeA.00108-17
Grimm, I., Garben, N., Dreier, J., Knabbe, C., and Vollmer, T. (2017b). Transcriptome analysis of Streptococcus gallolyticus subsp. gallolyticus in interaction with THP-1 macrophage-like cells. PLoS ONE 12:e0180044. doi: 10.1371/journal.pone.0180044
Gunsalus, I. C., and Niven, C. F. (1942). The effect of pH on the lactic acid fermentation. J. Biol. Chem. 145, 131–136.
Guy, E. M., Theng, B. K. G., and Walker, G. D. (1980). Interactions of Escherichia coli and Streptococcus bovis with soil clay surfaces as revealed by scanning electron microscopy. J. Plant Nutr. Soil Sci. 143, 194–199.
Haimowitz, M. D., Hernandez, L. A., and Herron, R. M. (2005). A blood donor with bacteraemia. Lancet 365, 1596–1596. doi: 10.1016/S0140-6736(05)66462-8
Harris, V. C., Armah, G., Fuentes, S., Korpela, K. E., Parashar, U., Victor, J. C., et al. (2017). Significant correlation between the infant gut microbiome and rotavirus vaccine response in rural Ghana. J. Infect. Dis. 215, 34–41. doi: 10.1093/infdis/jiw518
He, T. C., Sparks, A. B., Rago, C., Hermeking, H., Zawel, L., Da Costa, L. T., et al. (1998). Identification of c-MYC as a target of the APC pathway. Science 281, 1509–1512. doi: 10.1126/science.281.5382.1509
Henry-Stanley, M. J., Hess, D. J., Erlandsen, S. L., and Wells, C. L. (2005). Ability of the heparan sulfate proteoglycan syndecan-1 to participate in bacterial translocation across the intestinal epithelial barrier. Shock 24, 571–576. doi: 10.1097/01.shk.0000184286.95493.78
Hinse, D., Vollmer, T., Erhard, M., Welker, M., Moore, E. R. B., Kleesiek, K., et al. (2011a). Differentiation of species of the Streptococcus bovis/equinus-complex by MALDI-TOF Mass Spectrometry in comparison to sodA sequence analyses. Syst. Appl. Microbiol. 34, 52–57. doi: 10.1016/j.syapm.2010.11.010
Hinse, D., Vollmer, T., Rückert, C., Blom, J., Kalinowski, J., Knabbe, C., et al. (2011b). Complete genome and comparative analysis of Streptococcus gallolyticus subsp gallolyticus, an emerging pathogen of infective endocarditis. BMC Genomics 12:400. doi: 10.1186/1471-2164-12-400
Hirota, K., Osawa, R., Nemoto, K., Ono, T., and Miyake, Y. (1996). Highly expressed human sialyl Lewis antigen on cell surface of Streptococcus gallolyticus. Lancet 347:760.
Hoen, B., Chirouze, C., Cabell, C. H., Selton-Suty, C., Duchêne, F., Olaison, L., et al. (2005). Emergence of endocarditis due to group D streptococci: findings derived from the merged database of the International Collaboration on Endocarditis. Eur. J. Clin. Microbiol. Infect. Dis. 24, 12–16. doi: 10.1007/s10096-004-1266-6
Hoen, B., and Duval, X. (2013). Infective endocarditis. N. Engl. J. Med. 369:785. doi: 10.1056/NEJMcp1206782
Huang, C. C., Chang, S.-C., Shen, M.-H., Wong, J.-U., Wu, H.-Y., and Lee, H. H.-C. (2008). Streptococcus bovis fecal carriage is not associated with an Increased Risk of colorectal neoplasm. J. Soc. Colon Rectal Surgeon (Taiwan) 19, 71–77.
Hudson, J. A., Cai, Y., Corner, R. J., Morvan, B., and Joblin, K. N. (2000). Identification and enumeration of oleic acid and linoleic acid hydrating bacteria in the rumen of sheep and cows. J. Appl. Microbiol. 88, 286–292. doi: 10.1046/j.1365-2672.2000.00968.x
Humer, E., Petri, R. M., Aschenbach, J. R., Bradford, B. J., Penner, G. B., Tafaj, M., et al. (2018). Invited review: practical feeding management recommendations to mitigate the risk of subacute ruminal acidosis in dairy cattle. J. Dairy Sci. 101, 872–888. doi: 10.3168/jds.2017-13191
Hynes, W. (2004). Virulence factors of the group A streptococci and genes that regulate their expression. Front. Biosci. 9, 3399–3433. doi: 10.2741/1491
Isenring, J., Kohler, J., Nakata, M., Frank, M., Jans, C., Renault, P., et al. (2018). Streptococcus gallolyticus subsp. gallolyticus endocarditis isolate interferes with coagulation and activates the contact system. Virulence 9, 248–261. doi: 10.1080/21505594.2017.1393600
Jans, C., de Wouters, T., Bonfoh, B., Lacroix, C., Kaindi, D. W. M., Anderegg, J., et al. (2016). Phylogenetic, epidemiological and functional analyses of the Streptococcus bovis/Streptococcus equinus complex through an overarching MLST scheme. BMC Microbiol. 16:117. doi: 10.1186/s12866-016-0735-2
Jans, C., Follador, R., Hochstrasser, M., Lacroix, C., Meile, L., and Stevens, M. J. A. (2013). Comparative genome analysis of Streptococcus infantarius subsp. infantarius CJ18, an African fermented camel milk isolate with adaptations to dairy environment. BMC Genomics 14:200. doi: 10.1186/1471-2164-14-200
Jans, C., Gerber, A., Bugnard, J., Njage, P. M., Lacroix, C., and Meile, L. (2012a). Novel Streptococcus infantarius subsp. infantarius variants harboring lactose metabolism genes homologous to Streptococcus thermophilus. Food Microbiol. 31, 33–42. doi: 10.1016/j.fm.2012.02.001
Jans, C., Lacroix, C., and Meile, L. (2012b). A novel multiplex PCR/RFLP assay for the identification of Streptococcus bovis/Streptococcus equinus complex members from dairy microbial communities based on the 16S rRNA gene. FEMS Microbiol. Lett. 326, 144–150. doi: 10.1111/j.1574-6968.2011.02443.x
Jans, C., Meile, L., Kaindi, D. W. M., Kogi-Makau, W., Lamuka, P., Renault, P., et al. (2017). African fermented dairy products – overview of predominant technologically important microorganisms focusing on African Streptococcus infantarius variants and potential future applications for enhanced food safety and security. Int. J. Food Microbiol. 250, 27–36. doi: 10.1016/j.ijfoodmicro.2017.03.012
Jans, C., Meile, L., Lacroix, C., and Stevens, M. J. A. (2015). Genomics, evolution, and molecular epidemiology of the Streptococcus bovis/Streptococcus equinus complex (SBSEC). Infect. Genet. Evol. 33, 419–436. doi: 10.1016/j.meegid.2014.09.017
Jin, D., Chen, C., Li, L., Lu, S., Li, Z., Zhou, Z., et al. (2013). Dynamics of fecal microbial communities in children with diarrhea of unknown etiology and genomic analysis of associated Streptococcus lutetiensis. BMC Microbiol. 13:141. doi: 10.1186/1471-2180-13-141
Kaindi, D. W. M., Kogi-Makau, W., Lule, G. N., Kreikemeyer, B., Renault, P., Bonfoh, B., et al. (2017). Investigating the association between African spontaneously fermented dairy products, faecal carriage of Streptococcus infantarius subsp. infantarius and colorectal adenocarcinoma in Kenya. Acta Trop. 178, 10–18. doi: 10.1016/j.actatropica.2017.10.018
Kambarev, S., and Caté, C. (2015). Draft genome sequence of erythromycin-resistant Streptococcus gallolyticus subsp. gallolyticus NTS 31106099 isolated from a patient with infective endocarditis and colorectal cancer. Genome Announc. 3, 2–3. doi: 10.1128/genomeA.00370-15
Kambarev, S., Pecorari, F., and Corvec, S. (2017). Draft genome sequences of two highly erythromycin-resistant Streptococcus gallolyticus subsp. gallolyticus isolates containing a novel Tn916-like element, Tn6331. Genome Announc. 5, 17–19. doi: 10.1128/genomeA.00226-17
Kane, J. A., and Karakawa, W. W. (1969). Immunochemical analysis of Streptococcus bovis, strain s19, cell walls. J. Gen. Microbiol. 56, 157–164. doi: 10.1099/00221287-56-2-157
Kane, J. A., Karakawa, W. W., and Pazur, J. H. (1972). Glycans from streptococcal cell walls: structural features of a diheteroglycan isolated from the cell wall of Streptococcus bovis. J. Immunol. 108, 1218–1226.
Kaplan, M. H., Chmel, H., Stephens, A., Hsieh, H. C., Tenenbaum, M. J., Rothenberg, I. R., et al. (1983). Humoral reactions in human endocarditis due to Streptococcus bovis: evidence for a common S bovis antigen. J. Infect. Dis. 148, 266–274. doi: 10.1093/infdis/148.2.266
Kimpe, A., Decostere, A., Hermans, K., Mast, J., and Haesebrouck, E. (2003). Association of Streptococcus gallolyticus strains of high and low virulence with the intestinal tract of pigeons. Avian Dis. 47, 559–565. doi: 10.1637/6081
Kimpe, A., Hermans, K., De Herdt, P., and Haesebrouck, F. (2002). Failure of a low virulence Streptococcus gallolyticus serotype 1 strain to immunize pigeons against streptococcosis. Avian Pathol. 31, 421–423. doi: 10.1080/0307945021000005770
Klein, R. S., Recco, R. A., Catalano, M. T., Edberg, S. C., Casey, J. I., and Steigbigel, N. H. (1977). Association of Streptococcus bovis with carcinoma of colon. New Engl. J. Med. 297, 800–802. doi: 10.1056/NEJM197710132971503
Kmet, V., Flint, H. J., and Wallace, R. J. (1993). Probiotics and manipulation of rumen development and function. Arch Tierernahr. 44, 1–10. doi: 10.1080/17450399309386053
Kulp, K., and Ponte, J. G. J. (2000). Handbook of Cereal Science and Technology, Second Edition, Revised and Expanded. New York, NY: Marcel Dekker, Inc.
Kumar, K., Chaudhary, L. C., Agarwal, N., and Kamra, D. N. (2016). Effect of supplementing Streptococcus gallolyticus strain TDGB 406 on rumen fermentation and microbial profile of goats fed on oak leaves. Anim. Nutr. Feed Technol. 16:495. doi: 10.5958/0974-181X.2016.00046.9
Kumar, R., Herold, J. L., Schady, D., Davis, J., Kopetz, S., Martinez-Moczygemba, M., et al. (2017). Streptococcus gallolyticus subsp. gallolyticus promotes colorectal tumor development. PLoS Pathog. 13:e1006440. doi: 10.1371/journal.ppat.1006440
Kumar, R., Herold, J. L., Taylor, J., Xu, J., and Xu, Y. (2018). Variations among Streptococcus gallolyticus subsp. gallolyticus strains in connection with colorectal cancer. Sci. Rep. 8:1514. doi: 10.1038/s41598-018-19941-7
Lazarovitch, T., Shango, M., Levine, M., Brusovansky, R., Akins, R., Hayakawa, K., et al. (2013). The relationship between the new taxonomy of Streptococcus bovis and its clonality to colon cancer, endocarditis, and biliary disease. Infection 41, 329–337. doi: 10.1007/s15010-012-0314-x
Lee, C. K., Chan, H. M. H., Ho, P. L., Wong, H. K., Leung, J. N., Tsoi, W. C., et al. (2013). Long-term clinical outcomes after Streptococcus bovis isolation in asymptomatic blood donors in Hong Kong. Transfusion 53, 2207–2210. doi: 10.1111/trf.12087
Li, M., Gu, C., Zhang, W., Li, S., Liu, J., Qin, C., et al. (2013). Isolation and characterization of Streptococcus gallolyticus subsp. pasteurianus causing meningitis in ducklings. Vet. Microbiol. 162, 930–936. doi: 10.1016/j.vetmic.2012.11.038
Lichtenberger, L. M. (1995). The hydrophobic barrier properties of gastrointestinal mucus. Annu. Rev. Physiol. 57, 565–583. doi: 10.1146/annurev.ph.57.030195.003025
Lin, C. -Y., Tseng, S.-B., Lu, P.-L., Chen, T.-C., Lin, W.-R., Chen, Y.-H., et al. (2011a). Isolation of Streptococcus bovis from apheresis platelets of asymptomatic donor warranted colonoscopy investigation: case report and literature review. Transfusion 51, 2023–2027. doi: 10.1111/j.1537-2995.2011.03088.x
Lin, I. -H., Liu, T.-T., Teng, Y.-T., Wu, H.-L., Liu, Y.-M., Wu, K.-M., et al. (2011b). Sequencing and comparative genome analysis of two pathogenic Streptococcus gallolyticus subspecies: genome plasticity, adaptation and virulence. PLoS ONE 6:e20519. doi: 10.1371/journal.pone.0020519
Lopes, P. G., Cantarelli, V. V., Agnes, G., Costabeber, A. M., and d'azevedo, P. A. (2014). Novel real-time PCR assays using TaqMan minor groove binder probes for identification of fecal carriage of Streptococcus bovis/Streptococcus equinus complex from rectal swab specimens. J. Clin. Microbiol. 52, 974–976. doi: 10.1128/JCM.03253-13
Lorian, V., and Atkinson, B. (1978). Effect of serum on Gram-positive cocci grown in the presence of penicillin. J. Infect. Dis. 138, 865–871. doi: 10.1093/infdis/138.6.865
Mackey, E. T., and Hinton, M. (1990). The survival of streptococci and enterococci in animal feed and on straw with particular reference to Streptococcus uberis. J. Appl. Bacteriol. 68, 345–348. doi: 10.1111/j.1365-2672.1990.tb02884.x
Malachowa, N., and DeLeo, F. R. (2011). Staphylococcus aureus survival in human blood. Virulence 2, 567–569. doi: 10.4161/viru.2.6.17732
Marmolin, E. S., Hartmeyer, G. N., Christensen, J. J., Nielsen, X. C., Dargis, R., Skov, M. N., et al. (2016). Bacteremia with the bovis group streptococci: species identification and association with infective endocarditis and with gastrointestinal disease. Diagn. Microbiol. Infect. Dis. 85, 239–242. doi: 10.1016/j.diagmicrobio.2016.02.019
Martins, M., Aymeric, L., Du Merle, L., Danne, C., Robbe-Masselot, C., Trieu-Cuot, P., et al. (2015). Streptococcus gallolyticus Pil3 pilus is required for adhesion to colonic mucus and for colonization of mouse distal colon. J. Infect. Dis. 212, 1646–1655. doi: 10.1093/infdis/jiv307
Martins, M., Porrini, C., Du Merle, L., Danne, C., Robbe-Masselot, C., Trieu-Cuot, P., et al. (2016). The Pil3 pilus of Streptococcus gallolyticus binds to intestinal mucins and to fibrinogen. Gut Microb. 7, 526–532. doi: 10.1080/19490976.2016.1239677
McCann, J. C., Luan, S., Cardoso, F. C., Derakhshani, H., Khafipour, E., and Loor, J. J. (2016). Induction of subacute ruminal acidosis affects the ruminal microbiome and epithelium. Front. Microbiol. 7:701. doi: 10.3389/fmicb.2016.00701
McKenna, A. J., O'Donnell, M. E., McMullan, R., and Irwin, T. (2011). Long-term gastrointestinal outcomes after Streptococcus bovis bacteraemia. Int. J. Clin. Pract. 65, 1203–1205. doi: 10.1111/j.1742-1241.2011.02754.x
Mead, L. J., and Jones, G. A. (1981). Isolation and presumptive identification of adherent epithelial bacteria ('epimural' bacteria) from the ovine rumen wall. Appl. Environ. Microbiol. 41, 1020–1028.
Min, B. R., Pinchak, W. E., Anderson, R. C., and Hume, M. E. (2006). In vitro bacterial growth and in vivo ruminal microbiota populations associated with bloat in steers grazing wheat forage. J. Anim. Sci. 84, 2873–2882. doi: 10.2527/jas.2005-399
Morrison, D. A., Guédon, E., and Renault, P. (2013). Competence for natural genetic transformation in the S. bovis group streptococci, S. infantarius and S. macedonicus. J. Bacteriol. 195, 2612–2620. doi: 10.1128/JB.00230-13
Mueller, R. E., Iannotti, E. L., and Asplund, J. M. (1984). Isolation and identification of adherent epimural bacteria during succession in young lambs. Appl. Environ. Microbiol. 47, 724–730.
Nobbs, A. H., Lamont, R. J., and Jenkinson, H. F. (2009). Streptococcus adherence and colonization. Microbiol. Mol. Biol. Rev. 73, 407–450. doi: 10.1128/MMBR.00014-09
Noble, C. J. (1978). Carriage of group D streptococci in the human bowel. J. Clin. Pathol. 31, 1182–1186. doi: 10.1136/jcp.31.12.1182
Norfleet, R. G., and Mitchell, P. D. (1993). Streptococcus bovis does not selectively colonize colorectal cancer and polyps. J. Clin. Gastroenterol. 17, 25–28. doi: 10.1097/00004836-199307000-00008
Orpin, C. G., Mathiesen, S. D., Greenwood, Y., and Blix, A. S. (1985). Seasonal changes in the ruminal microflora of the high-arctic Svalbard reindeer (Rangifer tarandus platyrhynchus). Appl. Environ. Microbiol. 50, 144–151.
Paiva, A. D., Fernandes, K. M., Dias, R. S., Rocha, A. S., de Oliveira, L. L., Neves, C. A., et al. (2012). Effects of the oral administration of viable and heat-killed Streptococcus bovis HC5 cells to pre-sensitized BALB/c mice. PLoS ONE 7:e48313. doi: 10.1371/journal.pone.0048313
Palmonari, A., Stevenson, D. M., Mertens, D. R., Cruywagen, C. W., and Weimer, P. J. (2010). pH dynamics and bacterial community composition in the rumen of lactating dairy cows. J. Dairy Sci. 93, 279–287. doi: 10.3168/jds.2009-2207
Papadimitriou, K., Anastasiou, R., Mavrogonatou, E., Blom, J., Papandreou, N. C., Hamodrakas, S. J., et al. (2014). Comparative genomics of the dairy isolate Streptococcus macedonicus ACA-DC 198 against related members of the Streptococcus bovis/Streptococcus equinus complex. BMC Genomics 15:272. doi: 10.1186/1471-2164-15-272
Pazur, J. H., Dropkin, D. J., and Scott Forsberg, L. (1978). Glycans from streptococcal cell-walls: the molecular structure of an antigenic diheteroglycan of D-glucose and L-rhamnose from Streptococcus bovis. Carbohydr. Res. 66, 155–166. doi: 10.1016/S0008-6215(00)83248-6
Pazur, J. H., and Forsberg, L. S. (1978). Determination of the sugar sequences and the glycosidic-bond arrangements of immunogenic heteroglycans. Carbohydr. Res. 60, 167–178. doi: 10.1016/S0008-6215(00)83474-6
Peddareddigari, V. G., Wang, D., and Dubois, R. N. (2010). The tumor microenvironment in colorectal carcinogenesis. Cancer Microenviron. 3, 149–166. doi: 10.1007/s12307-010-0038-3
Penner, G. B., Beauchemin, K. A., and Mutsvangwa, T. (2007). Severity of ruminal acidosis in primiparous Holstein cows during the periparturient period. J. Dairy Sci. 90, 365–375. doi: 10.3168/jds.S0022-0302(07)72638-3
Potter, M. A., Cunliffe, N. A., Smith, M., Miles, R. S., Flapan, A. D., and Dunlop, M. G. (1998). A prospective controlled study of the association of Streptococcus bovis with colorectal carcinoma. J. Clin. Pathol. 51, 473–474. doi: 10.1136/jcp.51.6.473
Poulsen, L. L., Bisgaard, M., Son, N. T., Trung, N. V., An, H. M., and Dalsgaard, A. (2012). Enterococcus and Streptococcus spp. associated with chronic and self-medicated urinary tract infections in Vietnam. BMC Infect. Dis. 12:320. doi: 10.1186/1471-2334-12-320
Poyart, C., Quesne, G., Coulon, S., Berche, P., and Trieu-Cuot, P. (1998). Identification of streptococci to species level by sequencing the gene encoding the manganese-dependent superoxide dismutase. J. Clin. Microbiol. 36, 41–47.
Poyart, C., Quesne, G., and Trieu-Cuot, P. (2002). Taxonomic dissection of the Streptococcus bovis group by analysis of manganese-dependent superoxide dismutase gene (sodA) sequences: reclassification of 'Streptococcus infantarius subsp. coli' as Streptococcus lutetiensis sp. nov. and of Streptococcus bovis biotype II.2 as Streptococcus pasteurianus sp. nov. Int. J. Syst. Evol. Microbiol. 52, 1247–1255. doi: 10.1099/ijs.0.02044-0
Ramirez-Ronda, C. H. (1978). Adherence of glucan-positive and glucan-negative streptococcal strains to normal and damaged heart valves. J. Clin. Invest. 62, 805–814. doi: 10.1172/JCI109192
Ripamonti, B., Agazzi, A., Bersani, C., De Dea, P., Pecorini, C., Pirani, S., et al. (2011). Screening of species-specific lactic acid bacteria for veal calves multi-strain probiotic adjuncts. Anaerobe 17, 97–105. doi: 10.1016/j.anaerobe.2011.05.001
Romero, B., Morosini, M.-I., Loza, E., Rodríguez-Baños, M., Navas, E., Cantón, R., et al. (2011). Reidentification of Streptococcus bovis isolates causing bacteremia according to the new taxonomy criteria: still an issue? J. Clin. Microbiol. 49, 3228–3233. doi: 10.1128/JCM.00524-11
Rusniok, C., Couvé, E., Da Cunha, V., El Gana, R., Zidane, N., Bouchier, C., et al. (2010). Genome sequence of Streptococcus gallolyticus: insights into its adaptation to the bovine rumen and its ability to cause endocarditis. J. Bacteriol. 192, 2266–2276. doi: 10.1128/JB.01659-09
Russell, J. R., and Hino, T. (1985). Regulation of lactate production in Streptococcus bovis: a spiraling effect that contributes to rumen acidosis. J. Dairy Sci. 68, 1712–1721.
Sales-Duval, M., Lucas, F., and Blanchart, G. (2002). Effects of exogenous ammonia or free amino acids on proteolytic activity and protein breakdown products in Streptococcus bovis, Prevotella albensis, and Butyrivibrio fibrisolvens. Curr. Microbiol. 44, 435–443. doi: 10.1007/s00284-001-0013-9
Sánchez-Díaz, A. M., Romero-Hernández, B., Conde-Moreno, E., Kwak, Y. K., Zamora, J., Colque-Navarro, P., et al. (2016). New insights into the Enterococcus faecium and Streptococcus gallolyticus subsp. gallolyticus host interaction mechanisms. PLoS ONE 11:e0159159. doi: 10.1371/journal.pone.0159159
Schlegel, L., Grimont, F., Ageron, E., Grimont, P. A. D., and Bouvet, A. (2003). Reappraisal of the taxonomy of the Streptococcus bovis/Streptococcus equinus complex and related species: description of Streptococcus gallolyticus subsp. gallolyticus subsp. nov., S. gallolyticus subsp. macedonicus subsp. nov. and S. gallolyticus subsp. pasteurianus subsp. nov. Int. J. Syst. Evol. Microbiol. 53, 631–645. doi: 10.1099/ijs.0.02361-0
Schlegel, L., Grimont, F., Collins, M. D., Régnault, B., Grimont, P. A. D., and Bouvet, A. (2000). Streptococcus infantarius sp. nov., Streptococcus infantarius subsp. infantarius subsp. nov. and Streptococcus infantarius subsp. coli subsp. nov., isolated from humans and food. Int. J. Syst. Evol. Microbiol. 50, 1425–1434. doi: 10.1099/00207713-50-4-1425
Schoemaker, I. E., Sys, S. U., Andries, L. J., Meyers, J. M., Pattyn, S. R., and Brutsaert, D. L. (1994). Positive inotropic effect of Streptococcus in isolated cardiac muscle faecalis. Am J Physiol. 267, H2450–H2461. doi: 10.1152/ajpheart.1994.267.6.H2450
Schulz, J., Dumke, J., Hinse, D., Dreier, J., Habig, C., and Kemper, N. (2015). Organic turkey flocks: a reservoir of Streptococcus gallolyticus subspecies gallolyticus. PLoS ONE 10:e0144412. doi: 10.1371/journal.pone.0144412
Semjén, G., and Gálfi, P. (1990). Factors influencing the adherence of strains of Streptococcus bovis and Escherichia coli isolated from ruminal epithelium. Vet. Res. Commun. 14, 181–191. doi: 10.1007/BF00347736
Shen, S., Samaranayake, L. P., and Yip, H. K. (2005). Coaggregation profiles of the microflora from root surface caries lesions. Arch. Oral Biol. 50, 23–32. doi: 10.1016/j.archoralbio.2004.07.002
Sheng, W. H., Chuang, Y. C., Teng, L. J., and Hsueh, P. R. (2014). Bacteraemia due to Streptococcus gallolyticus subspecies pasteurianus is associated with digestive tract malignancies and resistance to macrolides and clindamycin. J. Infect. 69, 145–153. doi: 10.1016/j.jinf.2014.03.010
Shibata, Y., Tien le, H. T., Nomoto, R., and Osawa, R. (2014). Development of a multilocus sequence typing scheme for Streptococcus gallolyticus. Microbiology 160, 113–122. doi: 10.1099/mic.0.071605-0
Shu, Q., Bir, S. H., Gill, H. S., Duan, E., Xu, Y., Hiliard, M. A., Rowe, J. B., et al. (2001). Antibody response in sheep following immunization with Streptococcus bovis in different adjuvants. Vet. Res. Commun. 25, 43–54. doi: 10.1023/A:1026757917968
Shu, Q., Gill, H. S., Hennessy, D. W., Leng, R. A., Bird, S. H., and Rowe, J. B. (1999). Immunisation against lactic acidosis in cattle. Res. Vet. Sci. 67, 65–71. doi: 10.1053/rvsc.1998.0284
Shu, Q., Gill, H. S., Leng, R. A., and Rowe, J. B. (2000a). Immunization with a Streptococcus bovis vaccine administered by different routes against lactic acidosis in sheep. Vet. J. 159, 262–269. doi: 10.1053/tvjl.1999.0400
Shu, Q., Hillard, M. A., Bindon, B. M., Duan, E., Xu, Y., Bird, S. H., et al. (2000b). Effects of various adjuvants on efficacy of a vaccine against Streptococcus bovis and Lactobacillus spp in cattle. Am. J. Vet. Res. 61, 839–843.
Sillanpää, J., Nallapareddy, S. R., Qin, X., Singh, K. V., Muzny, D. M., Kovar, C. L., et al. (2009). A collagen-binding adhesin, Acb, and ten other putative MSCRAMM and pilus family proteins of Streptococcus gallolyticus subsp. gallolyticus (Streptococcus bovis group, biotype I). J. Bacteriol. 191, 6643–6653. doi: 10.1128/JB.00909-09
Sillanpää, J., Nallapareddy, S. R., Singh, K. V., Ferraro, M. J., and Murray, B. E. (2008). Adherence characteristics of endocarditis-derived Streptococcus gallolyticus ssp. gallolyticus (Streptococcus bovis biotype I) isolates to host extracellular matrix proteins. FEMS Microbiol. Lett. 289, 104–109. doi: 10.1111/j.1574-6968.2008.01378.x
Sissons, C. H., Hancock, E. M., Perinpanayagam, H. E., and Cutress, T. W. (1988). The bacteria responsible for ureolysis in artificial dental plaque. Arch. Oral Biol. 33, 727–733. doi: 10.1016/0003-9969(88)90006-4
Stein, R. A. (2013). Streptococcus infantarius and carcinogenesis: a new chapter in colorectal pathology. Int. J. Clin. Pract. 67, 1220–1224. doi: 10.1111/ijcp.12295
Styriak, I., Gálfi, P., and Kmet, V. (1991). Preliminary observations of interaction between bacteriophages and Streptococcus bovis bacteria on ruminal epithelium primoculture. Vet. Microbiol. 29, 281–287. doi: 10.1016/0378-1135(91)90135-3
Styriak, I., Gálfi, P., and Kmet, V. (1992). Adherence of ruminal Streptococcus bovis and Lactobacillus strains to primary and secondary cultures of rumen epithelium. Acta Microbiol. Hung. 39, 323–325.
Styriak, I., Galfi, P., and Kmet, V. (1994). The adherence of three Streptococcus bovis strains to cells of rumen epithelium primoculture under various conditions. Arch Tierernahr. 46, 357–365. doi: 10.1080/17450399409381786
Styriak, I., Lauková, A., Fallgren, C., and Wadström, T. (1999). Binding of selected extracellular matrix proteins to enterococci and Streptococcus bovis of animal origin. Curr. Microbiol. 39, 327–0335. doi: 10.1007/s002849900467
Tanjore, H., and Kalluri, R. (2006). The role of type IV collagen and basement membranes in cancer progression and metastasis. Am. J. Pathol. 168, 715–717. doi: 10.2353/ajpath.2006.051321
Tjalsma, H. (2010). Identification of biomarkers for colorectal cancer through proteomics-based approaches. Exp. Rev. Proteomics 7, 879–895. doi: 10.1586/epr.10.81
Tjalsma, H., Boleij, A., Marchesi, J. R., and Dutilh, B. E. (2012). A bacterial driver-passenger model for colorectal cancer: beyond the usual suspects. Nat. Rev. Microbiol. 10, 575–582. doi: 10.1038/nrmicro2819
Tjalsma, H., Lasonder, E., Schöller-Guinard, M., and Swinkels, D. W. (2007). Shotgun immunoproteomics to identify disease-associated bacterial antigens: application to human colon cancer. Proteomics Clin. Appl. 1, 429–434. doi: 10.1002/prca.200601018
Tjalsma, H., Schöller-Guinard, M., Lasonder, E., Ruers, T. J., Willems, H. L., and Swinkels, D. W. (2006). Profiling the humoral immune response in colon cancer patients: diagnostic antigens from Streptococcus bovis. Int. J. Cancer 119, 2127–2135. doi: 10.1002/ijc.22116
Toepfner, N., Shetty, S., Kunze, M., Orlowska-Volk, M., Krüger, M., Berner, R., et al. (2014). Fulminant neonatal sepsis due to Streptococcus alactolyticus- A case report and review. APMIS 122, 654–656. doi: 10.1111/apm.12219
Tripodi, M. F., Adinolfi, L. E., Ragone, E., Durante Mangoni, E., Fortunato, R., Iarussi, D., et al. (2004). Streptococcus bovis endocarditis and its association with chronic liver disease: an underestimated risk factor. Clin. Infect. Dis. 38, 1394–1400. doi: 10.1086/392503
Vanrobaeys, M., De Herdt, P., Charlier, G., Ducatelle, R., and Haesebrouck, F. (1999). Ultrastructure of surface components of Streptococcus gallolyticus (S. bovis) strains of differing virulence isolated from pigeons. Microbiology 145, 335–342. doi: 10.1099/13500872-145-2-335
Vanrobaeys, M., De Herdt, P., Ducatelle, R., Creten, W., and Haesebrouck, F. (1997). Extracellular proteins and virulence in Streptococcus bovis isolates from pigeons. Vet. Microbiol. 59, 59–66. doi: 10.1016/S0378-1135(97)00174-0
Vanrobaeys, M., Haesebrouck, F., Ducatelle, R., and De Herdt, P. (2000a). Adhesion of Streptococcus gallolyticus strains to extracellular matrix proteins. Vet. Microbiol. 74, 273–280. doi: 10.1016/S0378-1135(00)00180-2
Vanrobaeys, M., Haesebrouck, F., Ducatelle, R., and De Herdt, P. (2000b). Identification of virulence associated markers in the cell wall of pigeon Streptococcus gallolyticus strains. Vet. Microbiol. 73, 319–325. doi: 10.1016/S0378-1135(00)00159-0
Viljoen, K. S., Dakshinamurthy, A., Goldberg, P., and Blackburn, J. M. (2015). Quantitative profiling of colorectal cancer-associated bacteria reveals associations between Fusobacterium spp., enterotoxigenic Bacteroides fragilis (ETBF) and clinicopathological features of colorectal cancer. PLoS ONE 10:e0119462. doi: 10.1371/journal.pone.0119462
Vogkou, C. T., Vlachogiannis, N. I., Palaiodimos, L., and Kousoulis, A. A. (2016). The causative agents in infective endocarditis: a systematic review comprising 33,214 cases. Eur. J. Clin. Microbiol. Infect. Dis. 35, 1227–1245. doi: 10.1007/s10096-016-2660-6
Vollmer, T., Hinse, D., Kleesiek, K., and Dreier, J. (2010). Interactions between endocarditis-derived Streptococcus gallolyticus subsp gallolyticus isolates and human endothelial cells. BMC Microbiol. 10:78. doi: 10.1186/1471-2180-10-78
Von Hunolstein, C., Ricci, M. L., and Orefici, G. (1993). Adherence of glucan-positive and glucan-negative strains of Streptococcus bovis to human epithelial cells. J. Med. Microbiol. 39, 53–57. doi: 10.1099/00222615-39-1-53
Wallace, R. J., and McKain, N. (1991). A survey of peptidase activity in rumen bacteria. J Gen Microbiol 137, 2259–2264. doi: 10.1099/00221287-137-9-2259
Wang, H., Pan, X., Wang, C., Wang, M., and Yu, L. (2015). Effects of different dietary concentrate to forage ratio and thiamine supplementation on the rumen fermentation and ruminal bacterial community in dairy cows. Anim. Prod. Sci. 55, 189–193. doi: 10.1071/AN14523
Keywords: Streptococcus gallolyticus, Streptococcus lutetiensis, Streptococcus infantarius, virulence, infective endocarditis, colorectal cancer, pilus, microbiota
Citation: Jans C and Boleij A (2018) The Road to Infection: Host-Microbe Interactions Defining the Pathogenicity of Streptococcus bovis/Streptococcus equinus Complex Members. Front. Microbiol. 9:603. doi: 10.3389/fmicb.2018.00603
Received: 14 December 2017; Accepted: 15 March 2018;
Published: 10 April 2018.
Edited by:
Sven Hammerschmidt, University of Greifswald, GermanyReviewed by:
Konstantinos Papadimitriou, Agricultural University of Athens, GreeceMarcelo Gottschalk, Université de Montréal, Canada
Copyright © 2018 Jans and Boleij. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Christoph Jans, Y2hyaXN0b3BoLmphbnNAaGVzdC5ldGh6LmNo
 Christoph Jans
Christoph Jans Annemarie Boleij
Annemarie Boleij