
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Med. , 03 April 2025
Sec. Intensive Care Medicine and Anesthesiology
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1573112
Objectives: Multimodal analgesia (MA) is a cornerstone in perioperative pain management, enhancing pain relief and minimizing opioid consumption by targeting various pain pathways. This study conducts a bibliometric analysis of MA research from 2013 to 2023 to understand its development and impact on perioperative care.
Methods: A comprehensive literature search of the Web of Science Core Collection (WOSCC) was conducted, covering publications from January 2013 to December 2023. Data were analyzed using VOSviewer and other bibliometric tools to identify publication trends, key contributors, and emerging research themes.
Results: The analysis identified 1,939 studies on MA, with a notable increase in annual publications since 2020. The USA, China, and Canada were the leading contributors. Key terms like Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), Enhanced Recovery After Surgery (ERAS), and Patient-Controlled Analgesia (PCA) were frequently associated with MA. Significant journals included the Cureus Journal of Medical Science and Anesthesia and Analgesia. Influential authors such as Richard D. Urman and Henrik Kehlet were highlighted for their contributions. The research showed significant advancements and growing global interest in MA.
Conclusion: The study underscores the growing importance of MA in perioperative pain management, with significant contributions from leading countries and researchers. Future research should focus on optimizing pain management protocols, enhancing patient recovery, and reducing opioid dependency through MA.
Multimodal analgesia (MA) has become a cornerstone in perioperative pain management (PPM), recognized for its ability to enhance pain relief while minimizing opioid consumption (1–3). This approach utilizes a combination of analgesics targeting various pain pathways, providing a synergistic effect that improves pain control and reduces the adverse effects associated with high doses of single agents (4). This paradigm shift aligns with contemporary trends toward personalized and patient-centered care in medical practice.
Despite the established benefits of MA, there is a lack of comprehensive analysis regarding its current state in perioperative care, specifically the trends in research, key contributors, and emerging areas of focus. The rationale behind MA is rooted in the multifactorial nature of pain (5). By employing a mix of non-opioid analgesics (NOA) such as Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), acetaminophen, local anesthetics (LA), and adjuvant drugs like gabapentinoids and N-Methyl-D-Aspartate receptor antagonists, healthcare providers can achieve more effective pain management (6). This strategy reduces reliance on opioids, thereby mitigating risks such as respiratory depression, nausea, constipation, and the potential for opioid dependence (7).
In the perioperative setting, effective pain management is essential not only for patient comfort but also for optimal recovery (8, 9). Inadequate pain control can lead to prolonged hospital stays, delayed mobilization, and an increased risk of developing chronic pain (10, 11). Consequently, MA protocols are integral to ERAS pathways, which emphasize early mobilization, shortened hospital stays, and improved overall outcomes (12, 13).
Bibliometrics, the statistical analysis of written publications, is vital for understanding scientific knowledge development (14). In medical research, it examines publication trends, citation patterns, and identifies key researchers and research areas. This method highlights important contributions and emerging themes, guiding future research and fostering collaboration. One of the key strengths of bibliometric analysis is its ability to systematically track the evolution of research in a specific field. In the context of MA, it offers valuable insights by mapping research trends over time, identifying influential authors, key publications, and emerging areas of interest. Additionally, bibliometrics can uncover gaps in the literature and provide guidance for future research in MA and its application in perioperative care.
Several bibliometric studies have explored pain management, including opioid-free pain strategies and the use of multimodal analgesia in various clinical settings. For example, Zin et al. (15) provided a bibliometric overview of opioid-related research trends, while Robert et al. (16) focused on the evolution of pain management protocols in surgical settings. However, these studies generally overlook the specific perioperative context of MA.
This bibliometric analysis aims to explore the landscape of multimodal analgesia in the perioperative period by identifying key research trends, influential authors, and major advancements in the field. Through mapping research contributions, this study seeks to offer insights into the current state of MA and provide direction for future research in perioperative pain management.
To ensure comprehensive coverage of relevant studies, a literature search was conducted in the Web of Science Core Collection (WOSCC) using the following search terms: TS = (“multimodal analgesia” OR “multimodal pain management” OR “combined analgesia”) AND TS = (“perioperative” OR “surgery” OR “surgical”). The search was limited to publications in English and covered articles and reviews published between January 1, 2013, and December 31, 2023. The search was performed on July 4, 2024, and the data were downloaded in TEXT format. The search was restricted to the Web of Science Core Collection (WOSCC) database to ensure the quality and relevance of the data used in the bibliometric analysis.
Studies were included if they were peer-reviewed original research articles or systematic reviews focused on multimodal analgesia in the perioperative period, specifically addressing pain management in surgical settings. Studies were excluded if they were case reports, case series, editorials, non-peer-reviewed publications, or published in languages other than English. The inclusion and exclusion criteria are shown in Figure 1.
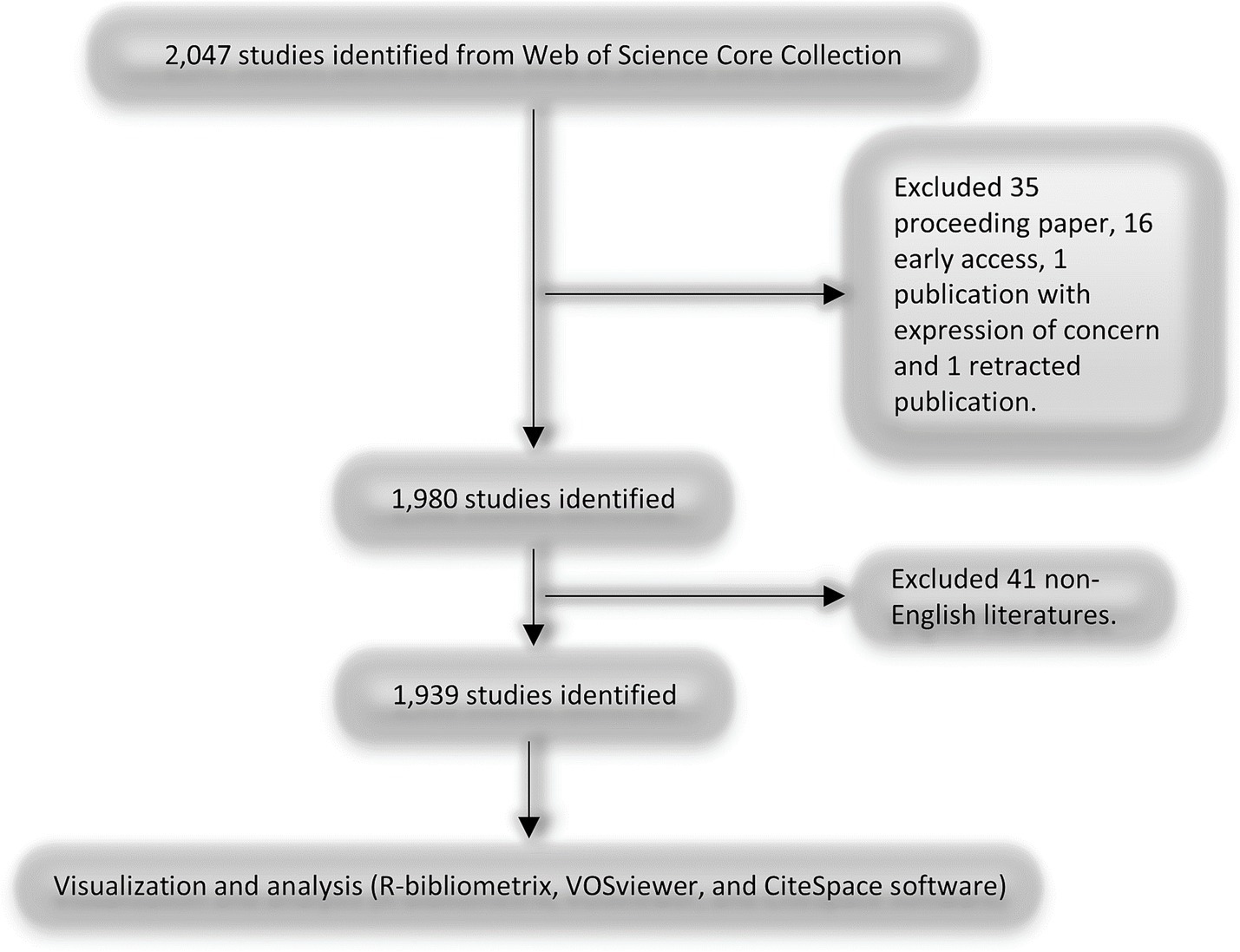
Figure 1. Flowchart of the search strategy and selection process for studies on multimodal analgesia in the perioperative period. This figure presents the step-by-step process of the literature search, screening, and selection criteria applied to identify relevant studies on multimodal analgesia during the perioperative period.
For our bibliometric analysis, we utilized the following software tools known for their robust methodologies:
1. VOSviewer (version 1.6.19): Developed by the Centre for Science and Technology Studies at Leiden University, VOSviewer is a free tool for constructing and visualizing bibliometric networks (17). We applied VOSviewer to construct collaboration networks, co-citation analyses, and keyword co-occurrence networks, which helped us identify emerging research themes and key patterns in multimodal analgesia during the perioperative period (18–20).
2. CiteSpace (version 6.2.R4): Created by Dr. Chaomei Chen at Drexel University, CiteSpace is a Java application available under a freeware license (21). We specifically used CiteSpace to detect citation bursts and to analyze the structure of the research field through cluster analysis and dual-map overlays. This allowed us to map out significant research milestones and key developments in the field (22, 23). CiteSpace uses several structural metrics, such as betweenness centrality, modularity, and silhouette scores, to analyze co-citation networks. Betweenness centrality measures a node’s role as a bridge in the network, with higher values indicating influential literature. Modularity assesses the division of a network into distinct communities, with values above 0.3 indicating significant division. The silhouette score evaluates the coherence of nodes within clusters, with values above 0.5 indicating credible cohesion (24).
3. Bibliometrix R package (version 3.2.1): This open-source R package is designed for comprehensive bibliometric analysis. It supports various quantitative research activities, including data processing, descriptive analysis, and network analysis. The tool generates essential metrics such as citation counts, collaboration indices, and impact measures, helping to develop a detailed understanding of research dynamics. Additionally, Bibliometrix assesses journal impact factors based on the latest Journal Citation Reports (JCR) (25).
These tools were selected for their ability to handle extensive bibliographic data and their specific analytical strengths, which facilitate a deeper understanding of the patterns, trends, and structural relationships within the field of MA during the perioperative period.
From 2013 to 2023, our bibliometric analysis identified 1,939 studies on MA in the perioperative period, comprising 1,526 articles and 413 reviews. Annual publications have shown an upward trend, with a significant surge since 2020, when they surpassed 300 per year, reflecting the growing prominence of this field (Figure 2).
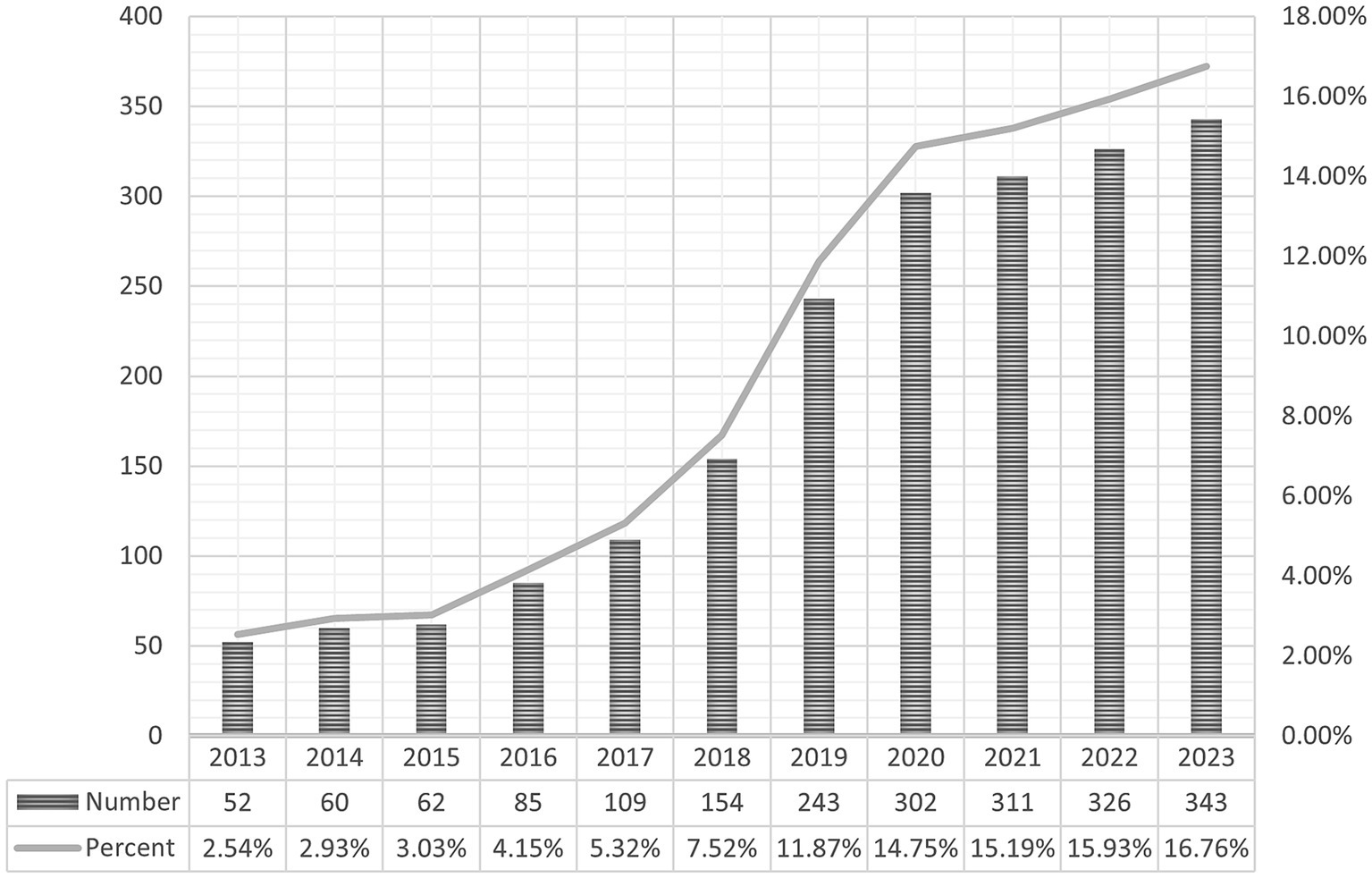
Figure 2. Publication trends in multimodal analgesia research during the perioperative period. The figure illustrates the annual growth in the number of publications related to multimodal analgesia in the perioperative setting, highlighting the increasing interest and research activity in this area over time.
The research spans 84 countries and 2,471 institutions. The USA, China, and Canada are the largest contributors, with 40.25, 8.07, and 5.94% of total publications, respectively. These countries contribute over half of the total publications, underscoring significant interest in perioperative MA. Network analysis (Figure 3A) indicates robust collaboration, particularly between the USA, China, Canada, and England. The USA holds a more influential position in the research network with a centrality index of 0.55, compared to China’s 0.03. Temporal trends (Figure 3B) reveal a shift in research epicenter, with China and India increasing publication volumes in recent years, following earlier dominance by the USA and Canada (Table 1).

Figure 3. Geographical collaboration and publication analysis. (A) A global map depicting the collaboration networks between countries in multimodal analgesia research. (B) An analysis of the number of publications by country over recent years, showing leading contributors. (C) A visual representation of collaborating institutions on a global scale. (D) An analysis of institutional publication outputs over recent years, emphasizing key research centers in the field.
Institutional contributions highlight the dominance of US institutions, with the University of California System leading in document count (5.67%). The University System of Ohio and the University of Toronto, with centrality indices of 0.11 and 0.10, respectively, play significant integrative roles within the research community (Table 2). A collaborative network of 116 institutions, based on a publication threshold of six, shows close cooperation among select universities (Figures 3C,D).
Research concentration in specific scientific journals is apparent (Table 3; Figure 4A). The Cureus Journal of Medical Science leads with 54 publications, followed by the Journal of Arthroplasty and BMC Anesthesiology. These journals have impact factors (IFs) ranging from 1 to 9.1 and are primarily in the first and second quartiles of the JCR 2023, indicating their influence. Co-citation analysis shows Anesthesia and Analgesia as the most cited journal (4,505 citations), followed by Anesthesiology (3,373 citations) and the British Journal of Anaesthesia (3,133 citations), highlighting their pivotal roles in MA research (Figure 4B; Table 4).
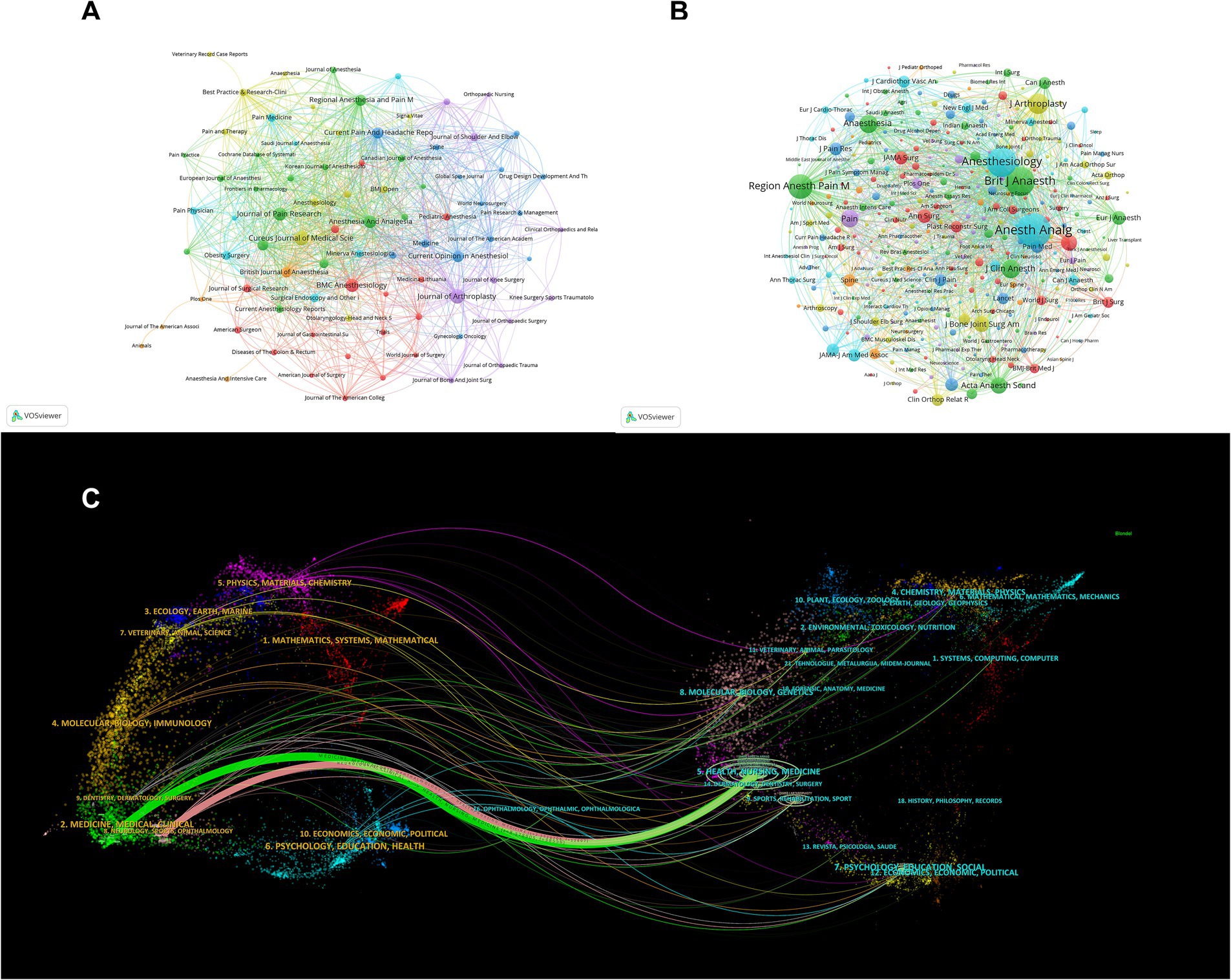
Figure 4. Journal contribution and citation analysis in multimodal analgesia research. (A) Visualization of journals that contribute to research on multimodal analgesia. (B) A map of co-cited journals, reflecting inter-journal citation patterns. (C) Dual-map overlay showing the relationship between citing and cited journals, providing insights into the interdisciplinary nature of the research.
The dual-map overlay of journals in Figure 4C demonstrates the relationship between citing and cited journals. The most prominent paths indicate that journals in the Health/Nursing/Medicine domain are frequently cited by publications in the Medicine/Medical/Clinical and Neurology/Sports/Ophthalmology domains, effectively representing the interconnectedness between citing and cited journals across various fields.
Our bibliometric analysis mapped the research landscape on MA in the perioperative period, identifying both prolific authors and frequently co-cited ones (Figures 5A,B). Table 5 highlights the top 10 authors by publication count, with Richard D. Urman leading at 18 publications. Notably, seven of these top authors are based in the USA, highlighting the significant contributions from this country. Co-citation data reveals Henrik Kehlet as the most influential author, with 541 citations, emphasizing his substantial impact in this field.
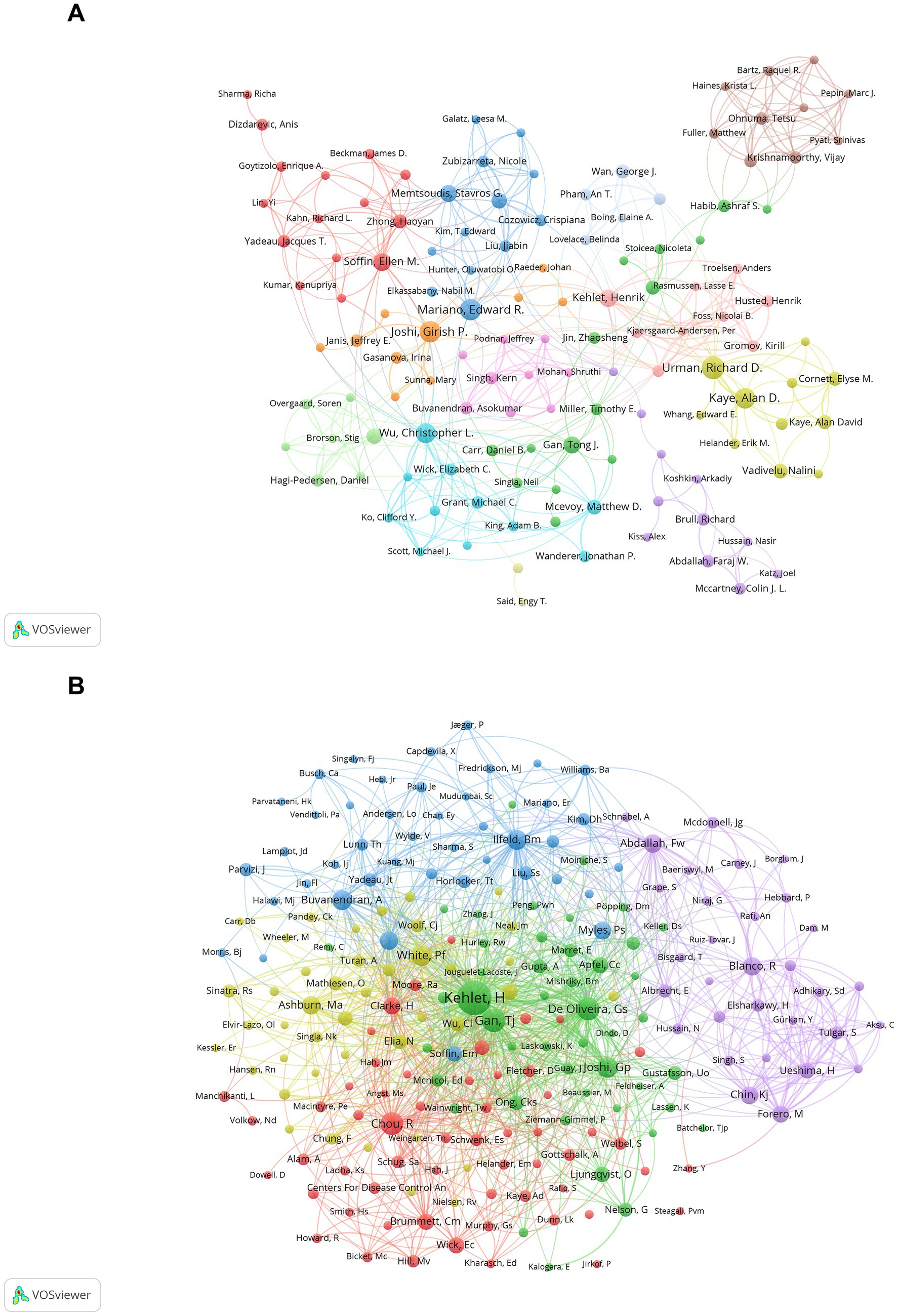
Figure 5. Analysis of authors and co-cited authors in multimodal analgesia research. (A) Visualization of the most prolific authors contributing to perioperative multimodal analgesia research. (B) A map of co-cited authors, illustrating the key contributors whose work has had significant influence in the field.
Our study pinpointed the most influential works in MA during the perioperative period based on co-citation frequencies. Table 6 lists the top 10 co-cited references, led by Roger Chou’s 2016 Journal of Pain article, “Management of Postoperative Pain: A Clinical Practice Guideline,” with 223 citations. This foundational work has significantly shaped subsequent research in the field. These leading articles, many published in prestigious journals such as Lancet and JAMA Surgery, account for four of the top 10 co-cited references (Table 6; Figure 6A).
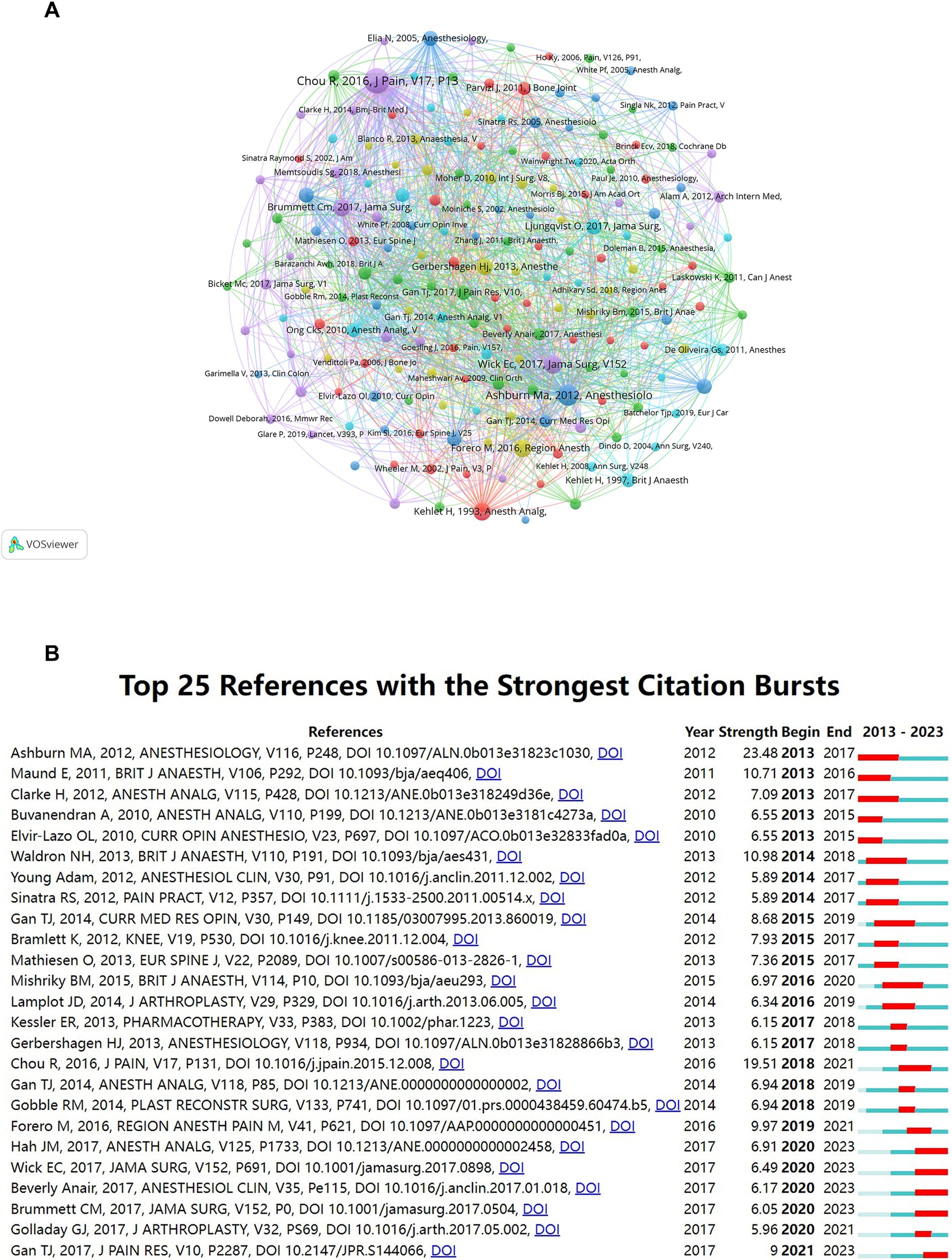
Figure 6. Reference citation analysis in multimodal analgesia research. (A) Visualization of the most frequently co-cited references in the field. (B) A timeline showcasing the top 25 references with the most significant citation bursts, indicating highly influential works over time.
The “citation burst” analysis identified articles that experienced a rapid increase in citations over specific periods, indicating the emergence of significant research topics (26). Our analysis highlighted the top 25 references with notable citation bursts (Figure 6B), signaling growing interest from 2013 to 2023. Michael A. Ashburn’s paper, “Practice guidelines for acute pain management in the perioperative setting,” showed the strongest citation burst (strength = 23.48) from 2013 to 2017. This comprehensive guideline offers evidence-based recommendations for acute pain management during the perioperative period. Its widespread adoption has significantly influenced pain management protocols. Roger Chou et al.’s work, “Management of Postoperative Pain,” ranked second (strength = 19.51) from 2018 to 2021. This multidisciplinary guideline provides recommendations for postoperative pain management, emphasizing multimodal approaches. Its citation increase reflects its role in standardizing postoperative pain care. N.H. Waldron’s paper, “Impact of Perioperative Dexamethasone on Postoperative Analgesia and Side-Effects: Systematic Review and Meta-Analysis,” demonstrated a significant citation burst (strength = 10.98) from 2014 to 2018. This meta-analysis evaluates the efficacy of dexamethasone in enhancing postoperative analgesia and reducing side effects. The surge in citations highlights its substantial contribution to refining postoperative care strategies. The significant rise in citations of these articles underscores their pivotal role in advancing MA research. They have not only influenced clinical practices and informed guidelines but have also spurred further research in the field. Analyzing such articles provides insight into key developments and shifts in research focus within MA.
Using VOSviewer and CiteSpace, we analyzed 2,773 author keywords across 1,939 documents, identifying 134 keywords appearing in at least nine documents each. Our analysis, which included keyword co-occurrence and temporal progression mapping, revealed evolving trends in MA during the perioperative period. “MA “emerged as a central node in the co-occurrence network, frequently cited in the literature. Significant adjacent terms included “opioids,” “total knee arthroplasty (TKA),” “sternotomy,” and “ERAS,” indicating their importance in the field (Figure 7A). The temporal progression map highlighted emerging research areas, with terms like “erector spinae plane block,” “dexmedetomidine,” and “thyroidectomy” appearing in orange, marking them as frontier topics (Figure 7B). In contrast, foundational terms such as “paracetamol” and “tramadol” were depicted in green, providing historical context to these evolving trends. CiteSpace cluster analysis identified 11 clusters, ranging from “#0 ERAS “to “#10 relief,” with a modularity (Q) value above 0.3 and a silhouette (S) value above 0.5 (Q = 0.51, S = 0.66), confirming the robustness and significance of these clusters (Figure 7C).
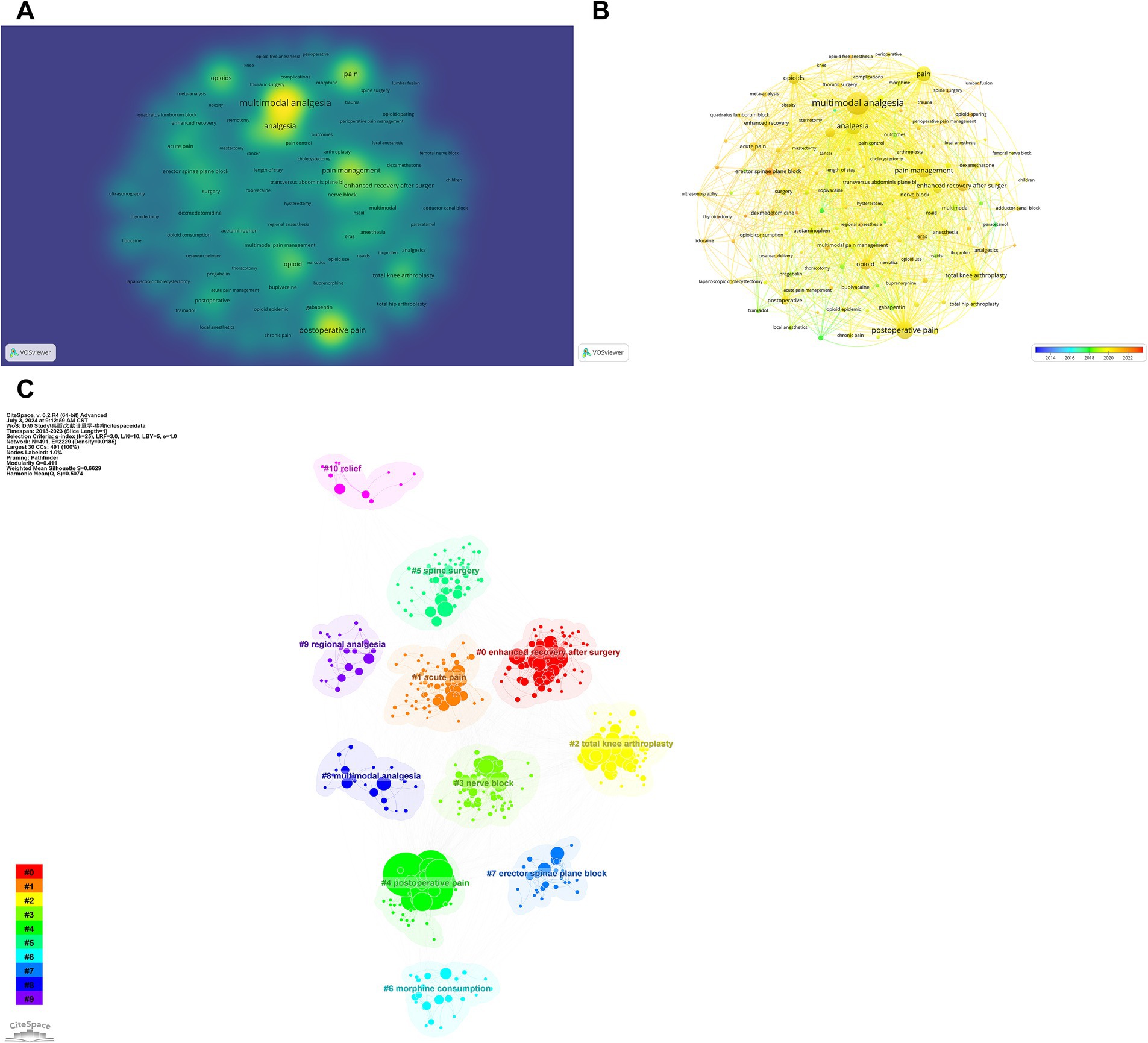
Figure 7. Keyword analysis in multimodal analgesia research. (A) Visualization of the most frequently used keywords in multimodal analgesia studies. (B) A temporal map showing the evolution of keyword usage over time, reflecting trends and shifts in research focus. (C) Clustering of related keywords, revealing key thematic areas and research topics within the field.
The annual publication trend, as illustrated in Figure 2, highlights a growing academic interest in this field. Since 2016, there has been a consistent rise in publications, with the last 5 years accounting for 74.5% of the total output, signifying rapid advancement. This trend can be attributed to several key factors. In particular, the opioid crisis in the United States has led to significant policy changes, such as the implementation of the 2016 “Opioid Prescribing Guidelines” by the Centers for Disease Control and Prevention (CDC) (27), which has driven research into opioid alternatives and MA. Additionally, the introduction of the “Comprehensive Addiction and Recovery Act” (CARA) (28) in 2016, which promotes non-opioid pain management strategies, further stimulated research in this area. The increasing adoption of ERAS protocols, which emphasize opioid-sparing strategies including MA, has also contributed to the surge in publications related to perioperative pain management.
The United States stands out as the most prolific country, with five of the 10 most cited authors, establishing its leading position in this domain. Anesthesia and Analgesia emerges as the most cited journal and the fifth most published, serving as a significant reference for scholars.
The chord diagram reveals that the United States, the United Kingdom, and Canada have the widest chords, indicating their strong collaborative ties with a diverse range of countries (Supplementary Figure 1). This observation is further supported by the analysis of the top 10 co-cited references, half of which were authored by researchers affiliated with these nations. This substantial overlap underscores a significant correlation between the intensity of international collaboration and the quality of research. The close ties among these countries suggest that enhanced cooperation not only facilitates the exchange of knowledge and resources but also contributes to the production of high-impact research, reinforcing the notion that collaboration is a key driver of academic excellence.
As depicted in Figure 5B and Table 5, five of the top 10 co-cited authors and nine of the top 10 authors hail from the United States. Notably, Henrik Kehlet of Copenhagen University, the most co-cited author, led the seminal work “The Value of ‘Multimodal’ or ‘Balanced Analgesia’ in Postoperative Pain Treatment” (29). Tong J. Gan of Stony Brook University Renaissance School of Medicine is renowned for his pivotal contributions to perioperative MA through research, education, and clinical practice, significantly enhancing postoperative pain management and patient recovery experiences (30–32). Roger Chou of Oregon Health & Science University, as the lead author of the influential article “Management of Postoperative Pain: A Clinical Practice Guideline” from the American Pain Society and other organizations, has extensively contributed to perioperative MA through research, systematic reviews, and guidelines that have shaped clinical practice and improved patient outcomes (1, 33, 34).
Table 5 reveals that top co-cited references predominantly focus on perioperative and postoperative pain management, particularly MA strategies. These references explore combining various NOA and techniques to enhance pain control, reduce opioid usage and its side effects, and improve patient recovery outcomes.
In the first cluster, ‘Surgical Applications’, the emphasis is placed on specific surgical procedures where MA has demonstrated a significant impact. Noteworthy procedures include TKA, Spine Surgery, and protocols under ERAS. TKA, a prevalent orthopedic surgery, involves the replacement of the knee joint with prosthetic components (35). The complexity of TKA necessitates effective pain management to facilitate early mobilization and rehabilitation, which are pivotal for favorable patient outcomes. MA, incorporating a variety of analgesic methods and medications, has been effective in mitigating postoperative pain and enhancing functional recovery in TKA patients (1, 36). Over time, research has evolved to emphasize opioid-sparing strategies in TKA, particularly with the integration of nerve blocks and regional anesthesia techniques, reducing dependency on systemic analgesics (37).
Similarly, spine surgery has gained prominence in pain management discussions due to the increasing complexity of surgical interventions and their associated postoperative pain challenges. The range from minimally invasive techniques to extensive spinal fusions, all of which can potentially cause significant postoperative pain (38, 39). Effective pain management is crucial for facilitating early rehabilitation, reducing the risk of developing chronic pain, and improving patient satisfaction (40, 41). MA in spine surgery typically involves the use of LA, systemic analgesics (SA), and regional techniques such as epidural analgesia (42). Recent literature has highlighted the role of multimodal approaches in spine surgery, shifting from traditional opioid-centric regimens to strategies incorporating non-opioid analgesics and regional blocks, reflecting a broader trend toward personalized pain control. One study examined the efficacy of a combination of NOA (acetaminophen and gabapentin) and intraoperative infusions (ketamine and lidocaine), finding that it did not significantly improve recovery or reduce opioid consumption in patients undergoing multilevel spine surgery (43). Furthermore, advancements in regional anesthesia techniques, such as ultrasound-guided methods, have been shown to decrease the risks associated with local anesthetic systemic toxicity and nerve injury (44). These findings highlight the importance of precision in regional anesthesia and underscore the necessity of ongoing research to determine the most effective strategies for reducing opioid-related adverse effects in spine surgery.
ERAS protocols offer a holistic framework for perioperative care, designed to enhance the surgical experience through a blend of evidence-based interventions (45, 46). These protocols aim to reduce the physiological impact of surgery, hasten recovery, and improve overall outcomes by integrating comprehensive care steps that span patient education, nutrition optimization, precise analgesic and anesthetic plans, and early mobilization (46). The incorporation of MA into ERAS protocols has gained traction as a central component of pain management, demonstrating a paradigm shift in perioperative care. A key pillar of ERAS is the implementation of MA, which is central to effective pain management strategies (47). Effective implementation of MA under ERAS protocols significantly enhances patient comfort, facilitates a faster return to function, and allows for earlier engagement in physical therapy, reducing discomfort and expediting recovery (48). This active participation is crucial in preventing post-surgical complications such as deep vein thrombosis (DVT), which are common in patients with prolonged immobility (49). In the framework of ERAS, the tailored approach to MA is continuously refined based on ongoing research and feedback from clinical practice. This adaptive methodology ensures that each patient’s pain management regimen is optimized to their specific surgical procedure and individual needs, promoting faster healing and reducing the overall burden of surgery (50, 51). The increasing emphasis on ERAS in recent years signifies a transition toward structured, protocol-driven perioperative care, where MA plays a pivotal role in ensuring enhanced recovery and opioid stewardship.
In the second cluster, “Pain Management Techniques and Strategies,” the focus shifts to various methods that enhance PPM through MA. Key techniques within this cluster include Nerve Block Techniques, Quadratus Lumborum Block, Regional Analgesia, and MA, reflecting a shift from systemic opioid reliance to targeted, site-specific pain control approaches.
Nerve block techniques are integral to the MA paradigm, offering targeted analgesia that significantly enhances PPM (52). These techniques involve the precise administration of anesthetics adjacent to specific nerves, thereby attenuating pain signals at their origin. The use of brachial plexus blocks in upper extremity surgeries exemplifies their efficacy, reducing reliance on systemic opioids and significantly improving postoperative analgesia (53), reflects an increasing emphasis on precision pain control and opioid minimization. In the perioperative context, brachial plexus blocks facilitate quicker recovery by enabling effective pain control, which is critical for patient mobilization and rehabilitation (54). Combining femoral and sciatic nerve blocks, commonly employed in lower extremity surgeries, supports early mobilization and rehabilitation by providing superior pain control compared to other methods, thus playing a crucial role in perioperative care strategies, though more research is needed on rehabilitation parameters (55). Thoracic interfascial block is effective for alleviating thoracic cage pain in intensive care unit patients, contributing to enhanced functional recovery post-surgery (56). These nerve blocks exemplify targeted interventions that align with the objectives of reducing opioid use and expediting patient recovery, critical components of modern perioperative care. The Erector Spinae Plane Block (ESPB) is used in thoracic, abdominal, and hip surgeries to mitigate visceral and somatic pain by anesthetizing the abdominal wall (57, 58). This regional technique significantly lowers postoperative pain scores and decreases opioid requirements, facilitating enhanced recovery protocols (59). The inclusion of these techniques in recent research suggests a paradigm shift toward less invasive yet highly effective regional anesthesia methods, driven by technological advancements and clinical demand.
Regional analgesia encompasses a range of techniques that provide extensive pain relief by targeting central and peripheral nerve pathways directly associated with the surgical site (60). Epidural analgesia and spinal anesthesia are pivotal in managing pain for lower extremity and lower abdominal surgeries, offering profound pain relief and reducing the surgical stress response (61). In the perioperative phase, these techniques are essential for optimizing patient comfort and minimizing stress-related complications. Continuous peripheral nerve blocks provide sustained analgesia, crucial for managing pain in procedures such as orthopedic surgeries or surgeries involving major joints (62). Their integration into perioperative care protocols aligns with the goals of MA to improve patient outcomes and satisfaction, ensuring a holistic approach to pain management.
Over the years, the conceptualization of MA has evolved from a simple multimodal approach to a highly individualized, evidence-driven strategy aimed at optimizing recovery and minimizing opioid-related complications. MA approach involves using multiple analgesic medications and techniques targeting different pain pathways to maximize pain relief while minimizing reliance on any single type of medication, particularly opioids. Combining various analgesics, such as acetaminophen, NSAIDs, and selective COX-2 inhibitors, with patient-controlled analgesia (PCA) morphine, has been shown through meta-analyses to offer superior pain relief and reduce opioid-related side effects compared to morphine alone (1). Advocated by guidelines from the American Pain Society and related organizations (1), this strategy aligns with the principles of ERAS, aiming to expedite recovery and improve overall patient outcomes. By addressing different aspects of pain through pharmacological and non-pharmacological methods, MA leads to more effective and balanced pain control, contributing significantly to the overarching goals of perioperative care and facilitating faster patient recovery and improved surgical outcomes (63, 64).
In the third cluster, “Pain and Medication Consumption,” the focus is on managing perioperative pain and optimizing medication use through MA. This section includes key areas such as Acute Pain, Postoperative Pain, and Morphine Consumption.
Acute pain, occurring immediately after surgery, significantly impacts patient recovery and satisfaction (65–67). As clinical guidelines continue to evolve, there has been a notable transition from opioid-centric acute pain management to multimodal and patient-specific strategies, incorporating regional analgesia, non-opioid analgesics, and integrative pain management techniques (48). Effective management of acute pain is essential to prevent the development of chronic pain and to promote early mobilization and rehabilitation (68). MA plays a crucial role in this context, as it provides balanced and comprehensive pain relief by combining different analgesic agents and techniques. This approach enhances patient comfort, facilitates faster recovery, and reduces postoperative complications by addressing pain through various mechanisms.
Postoperative pain management is critical for recovery, mobility, and overall patient outcomes (40, 69). Uncontrolled postoperative pain can lead to delayed recovery, increased risk of complications, and prolonged hospital stays (10, 11). MA combines various analgesic techniques and medications to offer effective pain control, reduce opioid consumption, and improve patient satisfaction (6). Strategies for managing postoperative pain include the use of LA, regional analgesia techniques, SA, and non-pharmacologic interventions such as physical therapy and cognitive-behavioral therapy (7). These comprehensive pain management approaches promote better functional recovery and overall patient outcomes.
Reducing morphine consumption is a central focus within the context of MA, given the risks associated with opioid use, such as respiratory depression, nausea, vomiting, and the potential for addiction (6, 7). Studies have demonstrated that MA protocols can significantly reduce opioid consumption, decrease the incidence of opioid-related side effects, and improve overall patient outcomes (70, 71). A growing body of literature also supports the role of patient education and shared decision-making in optimizing perioperative pain control while minimizing unnecessary opioid exposure. By optimizing pain control while reducing opioid use, MA promotes safer and more effective perioperative care.
In this study, we conducted a bibliometric analysis of MA research published between 2013 and 2023. Our findings reveal both consistencies and deviations when compared to previous studies in the field. Our focus on the 2013–2023 period aligns with certain studies emphasizing pain management across various medical contexts. For instance, a bibliometric analysis covering a broader timeframe identified significant growth in research, with recent trends focusing on e-health, telemedicine, virtual reality, and peripheral nerve blocks (72). However, by concentrating on a specific decade, our study offers a more nuanced understanding of the evolution of MA, enabling detailed observation of trends and characteristics during this period. Our findings indicate that the United States, China, and Canada are the primary contributors to MA research. This aligns with the dominant role of developed countries in medical research, as highlighted in previous studies. For example, in bibliometric analyses of labor analgesia, the United States is also one of the countries with the highest number of publications (73). However, our study further underscores the significant role of China in MA research, likely reflecting the country’s increasing emphasis and investment in pain management in recent years. Compared to bibliometric analyses in other fields, the contributions of different countries to MA research may vary due to differences in medical resources, research priorities, and policy support. The frequent occurrence of terms such as NSAIDs, ERAS, and PCA in our analysis aligns with their established importance in pain management literature (74). For example, multimodal analgesia protocols have been associated with improved outcomes in various surgical procedures, including total knee arthroplasty (75). Moreover, advances in molecular mechanisms have led to the development of new pharmaceutical products to treat postoperative pain, highlighting the ongoing evolution of pain management strategies. Our study uniquely identifies the expanding application of the ERAS concept within MA, likely driven by the global emphasis on postoperative recovery and complication reduction. The prominence of journals like the Cureus Journal of Medical Science and Anesthesia and Analgesia in our findings mirrors their recognized role in disseminating pain management research. Influential authors such as Richard D. Urman and Henrik Kehlet have significantly contributed to the advancement of MA, as reflected in our analysis. Their work aligns with the broader academic community’s efforts to enhance pain management practices. However, our focused examination of MA research allows for a more precise assessment of their contributions within this specific domain.
By comparing our findings with previous bibliometric analyses, this study demonstrates its unique contributions and value in the field of multimodal analgesia. Additionally, the cited literature provides readers with resources for further exploration of this research area. Future studies could build on our findings to delve deeper into optimizing multimodal analgesia, fostering international collaborations, and exploring the application of emerging technologies in pain management.
This study provides a comprehensive analysis of MA research from 2013 to 2023 but has several limitations. Reliance on the WOSCC may introduce selection bias by excluding significant research from other databases, potentially leading to an incomplete representation of the field. Additionally, the 10-year time frame might miss recent trends or emerging studies not yet widely cited. Language bias is another concern, as the majority of analyzed publications are likely in English, excluding significant contributions from non-English-speaking countries, limiting the global perspective. The analysis identifies major contributing countries and institutions but lacks a detailed examination of the nature and impact of these collaborations. Furthermore, the study points out emerging research areas but does not comprehensively analyze how these areas are evolving and their potential future impact. Another limitation is the potential underestimation of multidisciplinary contributions, as it may not fully capture the interconnectedness of MA research with other fields such as pharmacology, nursing, and physical therapy, leading to a narrower understanding of collaborative efforts that enhance patient care.
This bibliometric analysis provides an in-depth overview of MA research in the perioperative period from 2013 to 2023. By examining data from the WOSCC, the study identifies key trends, influential authors, leading institutions, and contributing countries. The USA, China, and Canada emerge as the primary contributors, reflecting a global focus on this topic. Richard D. Urman and Henrik Kehlet are recognized as prominent figures in the field. Key journals, such as the Cureus Journal of Medical Science and Anesthesia and Analgesia, play crucial roles in disseminating research. Research hotspots identified include “opioids,” “TKA,” and “ERAS.” The analysis underscores the significance of MA in improving postoperative outcomes and emphasizes the need for further research, particularly in optimizing rehabilitation parameters and enhancing patient recovery protocols.
Future research should focus on optimizing rehabilitation within MA protocols to better integrate pain management with recovery. Additionally, studying the long-term effects of opioid-sparing strategies on quality of life and evaluating new regional anesthesia techniques, such as ultrasound-guided nerve blocks, would offer valuable insights. Further exploration is needed on adapting MA protocols to diverse patient populations, including those with comorbidities, to ensure personalized care. Lastly, fostering international collaborations to assess the global applicability of MA strategies, especially in developing healthcare settings, will help create universal frameworks for perioperative pain management.
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
WJ: Conceptualization, Data curation, Formal analysis, Supervision, Visualization, Writing – original draft, Writing – review & editing. YQ: Formal analysis, Investigation, Validation, Writing – review & editing. LC: Investigation, Visualization, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2025.1573112/full#supplementary-material
SUPPLEMENTARY FIGURE 1 | Visualization of international cooperation between countries. Chord diagram illustrating the patterns of international cooperation between countries. Each sector in the diagram represents a country, and the color coding distinguishes different entities. The width of the chords is proportional to the strength of the cooperative relationship between the countries, with wider chords indicating stronger cooperation. The diagram highlights both bilateral and multilateral cooperation, visualizing the flow of interactions across nations.
MA, Multimodal Analgesia; NSAIDs, Non-Steroidal Anti-Inflammatory Drugs; ERAS, Enhanced Recovery After Surgery; WOSCC, Web of Science Core Collection; PCA, Patient-Controlled Analgesia; JCR, Journal Citation Reports; PPM, Perioperative Pain Management; NOA, Non-Opioid Analgesics; DVT, Deep Vein Thrombosis; TKA, Total Knee Arthroplasty; ESPB, Erector Spinae Plane Block; LA, Local Anesthetics; SA, Systemic Analgesics; CDC, Disease Control and Prevention; CARA, Comprehensive Addiction and Recovery Act.
1. Chou, R, Gordon, DB, de Leon-Casasola, OA, Rosenberg, JM, Bickler, S, Brennan, T, et al. Management of postoperative pain: a clinical practice guideline from the American Pain Society, the American Society of Regional Anesthesia and Pain Medicine, and the American Society of Anesthesiologists’ Committee on Regional Anesthesia, Executive Committee, and Administrative Council. J Pain. (2016) 17:131–57. doi: 10.1016/j.jpain.2015.12.008
2. American Society of Anesthesiologists Task Force on Acute Pain Management. Practice guidelines for acute pain management in the perioperative setting: an updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. (2012) 116:248–73. doi: 10.1097/ALN.0b013e31823c1030
3. Sala, E, Ferrari, F, Lanza, M, Milia, C, Sabatini, C, Bonazzi, A, et al. Improved efficacy, tolerance, safety, and abuse liability profile of the combination of CR4056 and morphine over morphine alone in rodent models. Br J Pharmacol. (2020) 177:3291–308. doi: 10.1111/bph.15049
4. Elvir-Lazo, OL, and White, PF. The role of multimodal analgesia in pain management after ambulatory surgery. Curr Opin Anaesthesiol. (2010) 23:697–703. doi: 10.1097/ACO.0b013e32833fad0a
5. Zheng, X, Feng, X, and Cai, X-J. Effectiveness and safety of continuous wound infiltration for postoperative pain management after open gastrectomy. World J Gastroenterol. (2016) 22:1902–10. doi: 10.3748/wjg.v22.i5.1902
6. Lunn, TH, Husted, H, Laursen, MB, Hansen, LT, and Kehlet, H. Analgesic and sedative effects of perioperative gabapentin in total knee arthroplasty: a randomized, double-blind, placebo-controlled dose-finding study. Pain. (2015) 156:2438–48. doi: 10.1097/j.pain.0000000000000309
7. Schwenk, ES, and Mariano, ER. Designing the ideal perioperative pain management plan starts with multimodal analgesia. Korean J Anesthesiol. (2018) 71:345–52. doi: 10.4097/kja.d.18.00217
8. Ishida, K, Shibanuma, N, Matsumoto, T, Tei, K, Kuroda, R, and Kurosaka, M. Periarticular multimodal drug injection improves post-operative pain and functional recovery after total knee arthroplasty. J Orthop Sci. (2016) 21:178–83. doi: 10.1016/j.jos.2015.12.004
9. Koh, IJ, Kim, MS, Sohn, S, Song, KY, Choi, NY, and In, Y. Duloxetine reduces pain and improves quality of recovery following total knee arthroplasty in centrally sensitized patients: a prospective, randomized controlled study. J Bone Joint Surg Am. (2019) 101:64–73. doi: 10.2106/JBJS.18.00347
10. Garimella, V, and Cellini, C. Postoperative pain control. Clin Colon Rectal Surg. (2013) 26:191–6. doi: 10.1055/s-0033-1351138
11. Kehlet, H, Jensen, TS, and Woolf, CJ. Persistent postsurgical pain: risk factors and prevention. Lancet. (2006) 367:1618–25. doi: 10.1016/S0140-6736(06)68700-X
12. Nelson, G, Altman, AD, Nick, A, Meyer, LA, Ramirez, PT, Achtari, C, et al. Guidelines for postoperative care in gynecologic/oncology surgery: enhanced recovery after surgery (ERAS®) society recommendations--part II. Gynecol Oncol. (2016) 140:323–32. doi: 10.1016/j.ygyno.2015.12.019
13. Mark, J, Argentieri, DM, Gutierrez, CA, Morrell, K, Eng, K, Hutson, AD, et al. Ultrarestrictive opioid prescription protocol for pain management after gynecologic and abdominal surgery. JAMA Netw Open. (2018) 1:e185452. doi: 10.1001/jamanetworkopen.2018.5452
14. Hicks, D, Wouters, P, Waltman, L, de Rijcke, S, and Rafols, I. Bibliometrics: the Leiden manifesto for research metrics. Nature. (2015) 520:429–31. doi: 10.1038/520429a
15. Akbar, HF, Siddiq, K, and Nusrat, S. Citation classics and trends in the field of opioids: a bibliometric analysis. Cureus. (2019) 11:e5055. doi: 10.7759/cureus.5055
16. Robert, C, and Wilson, CS. Thirty-year survey of bibliometrics used in the research literature of pain: analysis, evolution, and pitfalls. Front Pain Res (Lausanne). (2023) 4:1071453. doi: 10.3389/fpain.2023.1071453
17. Xin, X, Xiang, X, Xin, Y, Li, Q, Ma, H, Liu, X, et al. Global trends in research on oxidative stress associated with periodontitis from 1987 to 2022: a bibliometric analysis. Front Immunol. (2022) 13:979675. doi: 10.3389/fimmu.2022.979675
18. van Eck, NJ, and Waltman, L. Software survey: VOSviewer, a computer program for bibliometric mapping. Scientometrics. (2010) 84:523–38. doi: 10.1007/s11192-009-0146-3
19. Yeung, AWK, and Mozos, I. The innovative and sustainable use of dental panoramic radiographs for the detection of osteoporosis. Int J Environ Res Public Health. (2020) 17:2449. doi: 10.3390/ijerph17072449
20. van Eck, NJ, and Waltman, L. Citation-based clustering of publications using CitNetExplorer and VOSviewer. Scientometrics. (2017) 111:1053–70. doi: 10.1007/s11192-017-2300-7
21. Wei, N, Xu, Y, Li, Y, Shi, J, Zhang, X, You, Y, et al. A bibliometric analysis of T cell and atherosclerosis. Front Immunol. (2022) 13:948314. doi: 10.3389/fimmu.2022.948314
22. Zhang, XL, Zheng, Y, Xia, ML, Wu, YN, Liu, XJ, Xie, SK, et al. Knowledge domain and emerging trends in vinegar research: a bibliometric review of the literature from WoSCC. Food Secur. (2020) 9:166. doi: 10.3390/foods9020166
23. Chen, C. Searching for intellectual turning points: progressive knowledge domain visualization. Proc Natl Acad Sci USA. (2004) 101:5303–10. doi: 10.1073/pnas.0307513100
24. Yan, WT, Lu, S, Yang, YD, Ning, WY, Cai, Y, Hu, XM, et al. Research trends, hot spots and prospects for necroptosis in the field of neuroscience. Neural Regen Res. (2021) 16:1628–37. doi: 10.4103/1673-5374.303032
25. Aria, M, and Cuccurullo, C. bibliometrix: an R-tool for comprehensive science mapping analysis. J Informetr. (2017) 11:959–75. doi: 10.1016/j.joi.2017.08.007
26. Sharma, P, Hu-Lieskovan, S, Wargo, JA, and Ribas, A. Primary, adaptive, and acquired resistance to cancer immunotherapy. Cell. (2017) 168:707–23. doi: 10.1016/j.cell.2017.01.017
27. Dowell, D, Haegerich, TM, and Chou, R. CDC guideline for prescribing opioids for chronic pain--United States, 2016. JAMA. (2016) 315:1624–45. doi: 10.1001/jama.2016.1464
28. Murphy, J, Goodman, D, Johnson, MC, and Terplan, M. The comprehensive addiction and recovery act: opioid use disorder and midwifery practice. Obstet Gynecol. (2018) 131:542–4. doi: 10.1097/AOG.0000000000002493
29. Kehlet, H, and Dahl, JB. The value of “multimodal” or “balanced analgesia” in postoperative pain treatment. Anesth Analg. (1993) 77:1048–56. doi: 10.1213/00000539-199311000-00030
30. Gan, TJ. Poorly controlled postoperative pain: prevalence, consequences, and prevention. J Pain Res. (2017) 10:2287–98. doi: 10.2147/JPR.S144066
31. Gan, TJ, Habib, AS, Miller, TE, White, W, and Apfelbaum, JL. Incidence, patient satisfaction, and perceptions of post-surgical pain: results from a US national survey. Curr Med Res Opin. (2014) 30:149–60. doi: 10.1185/03007995.2013.860019
32. Gan, TJ, Diemunsch, P, Habib, AS, Kovac, A, Kranke, P, Meyer, TA, et al. Consensus guidelines for the management of postoperative nausea and vomiting. Anesth Analg. (2014) 118:85–113. doi: 10.1213/ANE.0000000000000002
33. Page, MJ, Moher, D, Bossuyt, PM, Boutron, I, Hoffmann, TC, Mulrow, CD, et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. Br Med J. (2021) 372:n160. doi: 10.1136/bmj.n160
34. Page, MJ, McKenzie, JE, Bossuyt, PM, Boutron, I, Hoffmann, TC, Mulrow, CD, et al. Statement: an updated guideline for reporting systematic reviews. Int J Surg. (2020) 88:105906. doi: 10.1016/j.ijsu.2021.105906
35. Escobar Ivirico, JL, Bhattacharjee, M, Kuyinu, E, Nair, LS, and Laurencin, CT. Regenerative engineering for knee osteoarthritis treatment: biomaterials and cell-based technologies. Engineering (Beijing). (2017) 3:16–27. doi: 10.1016/j.eng.2017.01.003
36. Berend, ME, Berend, KR, and Lombardi, AV. Advances in pain management: game changers in knee arthroplasty. Bone Joint J. (2014) 96-B:7–9. doi: 10.1302/0301-620X.96B11.34514
37. Vendittoli, P-A, Makinen, P, Drolet, P, Lavigne, M, Fallaha, M, Guertin, M-C, et al. A multimodal analgesia protocol for total knee arthroplasty. A randomized, controlled study. J Bone Joint Surg Am. (2006) 88:282–9. doi: 10.2106/JBJS.E.00173
38. Deyo, RA, Mirza, SK, Martin, BI, Kreuter, W, Goodman, DC, and Jarvik, JG. Trends, major medical complications, and charges associated with surgery for lumbar spinal stenosis in older adults. JAMA. (2010) 303:1259–65. doi: 10.1001/jama.2010.338
39. Cho, JH, Hwang, CJ, Kim, H, Joo, Y-S, Lee, D-H, and Lee, CS. Effect of osteoporosis on the clinical and radiological outcomes following one-level posterior lumbar interbody fusion. J Orthop Sci. (2018) 23:870–7. doi: 10.1016/j.jos.2018.06.009
40. Chen, X, Zhang, J, Yu, Y, Wang, H, Ma, G, Wang, D, et al. Ultrasound-triggered on demand lidocaine release relieves postoperative pain. Front Bioeng Biotechnol. (2022) 10:925047. doi: 10.3389/fbioe.2022.925047
41. Kao, H-W, Lin, Y-Y, Gwathney, WJ, and Hong, K. Formulation and evaluation of multilamellar vesicles ropivacaine in pain management. Int J Nanomedicine. (2019) 14:7891–901. doi: 10.2147/IJN.S215952
42. Flowers, KM, Patton, ME, Hruschak, VJ, Fields, KG, Schwartz, E, Zeballos, J, et al. Conditioned open-label placebo for opioid reduction after spine surgery: a randomized controlled trial. Pain. (2021) 162:1828–39. doi: 10.1097/j.pain.0000000000002185
43. Kianian, S, Bansal, J, Lee, C, Zhang, K, and Bergese, SD. Perioperative multimodal analgesia: a review of efficacy and safety of the treatment options. Anesthesiol Perioper Sci. (2024) 2:9. doi: 10.1007/s44254-023-00043-1
44. Maheshwari, K, Avitsian, R, Sessler, DI, Makarova, N, Tanios, M, Raza, S, et al. Multimodal analgesic regimen for spine surgery: a randomized placebo-controlled trial. Anesthesiology. (2020) 132:992–1002. doi: 10.1097/ALN.0000000000003143
45. Meyer, LA, Shi, Q, Lasala, J, Iniesta, MD, Lin, HK, Nick, AM, et al. Comparison of patient reported symptom burden on an enhanced recovery after surgery (ERAS) care pathway in patients with ovarian cancer undergoing primary vs. interval tumor reductive surgery. Gynecol Oncol. (2019) 152:501–8. doi: 10.1016/j.ygyno.2018.10.044
46. Ljungqvist, O, Scott, M, and Fearon, KC. Enhanced recovery after surgery: a review. JAMA Surg. (2017) 152:292–8. doi: 10.1001/jamasurg.2016.4952
47. Vadivelu, N, Mitra, S, Schermer, E, Kodumudi, V, Kaye, AD, and Urman, RD. Preventive analgesia for postoperative pain control: a broader concept. Local Reg Anesth. (2014) 7:17–22. doi: 10.2147/LRA.S62160
48. Wick, EC, Grant, MC, and Wu, CL. Postoperative multimodal analgesia pain management with nonopioid analgesics and techniques: a review. JAMA Surg. (2017) 152:691–7. doi: 10.1001/jamasurg.2017.0898
49. Chakravarthy, VB, Yokoi, H, Coughlin, DJ, Manlapaz, MR, and Krishnaney, AA. Development and implementation of a comprehensive spine surgery enhanced recovery after surgery protocol: the Cleveland Clinic experience. Neurosurg Focus. (2019) 46:E11. doi: 10.3171/2019.1.FOCUS18696
50. Soffin, EM, Wetmore, DS, Barber, LA, Vaishnav, AS, Beckman, JD, Albert, TJ, et al. An enhanced recovery after surgery pathway: association with rapid discharge and minimal complications after anterior cervical spine surgery. Neurosurg Focus. (2019) 46:E9. doi: 10.3171/2019.1.FOCUS18643
51. Reif de Paula, T, Nemeth, SK, Kurlansky, PA, Simon, HL, Miller, LK, and Keller, DS. A randomized controlled trial examining the impact of an anorectal surgery multimodal enhanced recovery program on opioid use. Ann Surg. (2022) 275:e22–9. doi: 10.1097/SLA.0000000000004701
52. Memtsoudis, SG, Cozowicz, C, Bekeris, J, Bekere, D, Liu, J, Soffin, EM, et al. Peripheral nerve block anesthesia/analgesia for patients undergoing primary hip and knee arthroplasty: recommendations from the International Consensus on Anesthesia-Related Outcomes after Surgery (ICAROS) group based on a systematic review and meta-analysis of current literature. Reg Anesth Pain Med. (2021) 46:971–85. doi: 10.1136/rapm-2021-102750
53. Grape, S, Pawa, A, Weber, E, and Albrecht, E. Retroclavicular vs supraclavicular brachial plexus block for distal upper limb surgery: a randomised, controlled, single-blinded trial. Br J Anaesth. (2019) 122:518–24. doi: 10.1016/j.bja.2018.12.022
54. Zarnegar, R, Brown, MRD, Henley, M, Tidman, V, and Pathmanathan, A. Patient perceptions and recall of consent for regional anaesthesia compared with consent for surgery. J R Soc Med. (2015) 108:451–6. doi: 10.1177/0141076815604494
55. Terkawi, AS, Mavridis, D, Sessler, DI, Nunemaker, MS, Doais, KS, Terkawi, RS, et al. Pain management modalities after total knee arthroplasty: a network meta-analysis of 170 randomized controlled trials. Anesthesiology. (2017) 126:923–37. doi: 10.1097/ALN.0000000000001607
56. López-Matamala, B, Fajardo, M, Estébanez-Montiel, B, Blancas, R, Alfaro, P, and Chana, M. A new thoracic interfascial plane block as anesthesia for difficult weaning due to ribcage pain in critically ill patients. Med Intensiva. (2014) 38:463–5. doi: 10.1016/j.medin.2013.10.005
57. Salama, ER. Ultrasound-guided bilateral quadratus lumborum block vs. intrathecal morphine for postoperative analgesia after cesarean section: a randomized controlled trial. Korean J Anesthesiol. (2020) 73:121–8. doi: 10.4097/kja.d.18.00269
58. Feray, S, Lubach, J, Joshi, GP, Bonnet, F, and Van de Velde, M. PROSPECT guidelines for video-assisted thoracoscopic surgery: a systematic review and procedure-specific postoperative pain management recommendations. Anaesthesia. (2022) 77:311–25. doi: 10.1111/anae.15609
59. Semkovych, Y, and Dmytriiev, D. Elevated serum TLR4 level as a potential marker for postsurgical chronic pain in pediatric patients with different approaches to analgesia. Front Med (Lausanne). (2022) 9:897533. doi: 10.3389/fmed.2022.897533
60. Li, Y-S, Chang, K-Y, Lin, S-P, Chang, M-C, and Chang, W-K. Group-based trajectory analysis of acute pain after spine surgery and risk factors for rebound pain. Front Med (Lausanne). (2022) 9:907126. doi: 10.3389/fmed.2022.907126
61. Hong, Y, Zhao, J, Chen, Y-R, Huang, Z-H, Hou, L-D, Shen, B, et al. Spinal anesthesia alleviates dextran sodium sulfate-induced colitis by modulating the gut microbiota. World J Gastroenterol. (2022) 28:1239–56. doi: 10.3748/wjg.v28.i12.1239
62. Ilfeld, BM, Morey, TE, Wang, RD, and Enneking, FK. Continuous popliteal sciatic nerve block for postoperative pain control at home: a randomized, double-blinded, placebo-controlled study. Anesthesiology. (2002) 97:959–65. doi: 10.1097/00000542-200210000-00031
63. Shi, Y, and Wu, W. Multimodal non-invasive non-pharmacological therapies for chronic pain: mechanisms and progress. BMC Med. (2023) 21:372. doi: 10.1186/s12916-023-03076-2
64. Helander, EM, Menard, BL, Harmon, CM, Homra, BK, Allain, AV, Bordelon, GJ, et al. Multimodal analgesia, current concepts, and acute pain considerations. Curr Pain Headache Rep. (2017) 21:3. doi: 10.1007/s11916-017-0607-y
65. Valtola, A, Laakso, M, Hakomäki, H, Anderson, BJ, Kokki, H, Ranta, V-P, et al. Intranasal fentanyl for intervention-associated breakthrough pain after cardiac surgery. Clin Pharmacokinet. (2021) 60:907–19. doi: 10.1007/s40262-021-01002-4
66. Kehlet, H, and Dahl, JB. Anaesthesia, surgery, and challenges in postoperative recovery. Lancet. (2003) 362:1921–8. doi: 10.1016/S0140-6736(03)14966-5
67. Pavlin, DJ, Chen, C, Penaloza, DA, Polissar, NL, and Buckley, FP. Pain as a factor complicating recovery and discharge after ambulatory surgery. Anesth Analg. (2002) 95:627–34. doi: 10.1213/00000539-200209000-00025
68. Mifflin, KA, and Kerr, BJ. The transition from acute to chronic pain: understanding how different biological systems interact. Can J Anaesth. (2014) 61:112–22. doi: 10.1007/s12630-013-0087-4
69. Yoon, J-U, Byeon, G-J, Cheon, J-H, Choi, Y-M, Ri, H-S, and Baik, S-W. Post-operative intravenous patient-controlled analgesic efficacy of morphine with ketorolac versus nefopam after laparoscopic gynecologic surgery: a randomized non-inferiority trial. Korean J Anesthesiol. (2016) 69:161–6. doi: 10.4097/kjae.2016.69.2.161
70. De Oliveira, GS, Castro-Alves, LJ, Nader, A, Kendall, MC, and McCarthy, RJ. Transversus abdominis plane block to ameliorate postoperative pain outcomes after laparoscopic surgery: a meta-analysis of randomized controlled trials. Anesth Analg. (2014) 118:454–63. doi: 10.1213/ANE.0000000000000066
71. Brogi, E, Kazan, R, Cyr, S, Giunta, F, and Hemmerling, TM. Transversus abdominal plane block for postoperative analgesia: a systematic review and meta-analysis of randomized-controlled trials. Can J Anaesth. (2016) 63:1184–96. doi: 10.1007/s12630-016-0679-x
72. Demir, E, Doğan, G, Kiraz, M, Akdağli Ekici, A, Kayir, S, Ekici, M, et al. Current trends in pain management: a bibliometric analysis for the 1980-to-2023 period. Medicine (Baltimore). (2025) 104:e41319. doi: 10.1097/MD.0000000000041319
73. Yu, K, Ding, Z, Yang, J, Han, X, Li, T, and Miao, H. Bibliometric analysis on global analgesia in labor from 2002 to 2021. J Pain Res. (2023) 16:1999–2013. doi: 10.2147/JPR.S416142
74. Argoff, CE. Recent management advances in acute postoperative pain. Pain Pract. (2014) 14:477–487. doi: 10.1111/papr.12108
Keywords: perioperative pain management, bibliometric analysis, CiteSpace, VOSviewer, multimodal analgesia
Citation: Jiang W, Qin Y and Chen L (2025) Bibliometric analysis of multimodal analgesia research in the perioperative period: trends, contributions, and emerging areas (2013–2023). Front. Med. 12:1573112. doi: 10.3389/fmed.2025.1573112
Received: 08 February 2025; Accepted: 17 March 2025;
Published: 03 April 2025.
Edited by:
Lalit Gupta, University of Delhi, IndiaReviewed by:
Devang Bharti, University of Delhi, IndiaCopyright © 2025 Jiang, Qin and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wenchen Jiang, Y2FlbGlhd2VuY2hlbkBob3RtYWlsLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.