
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Med. , 20 March 2025
Sec. Infectious Diseases: Pathogenesis and Therapy
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1560016
Trichosporon asahii (T. asahii) has been increasingly recognized as the causative pathogen of invasive fungal infection, whereas intracranial infection caused by T. asahii are extremely scarce. Here, we report a rare case of intracranial T. asahii infection in an immunocompetent woman from China. She was hospitalized for obstructive hydrocephalus and experienced two brain surgeries. One week after the second surgery, the patient developed fever, vomiting, and elevated infection-related indicators. Cerebrospinal fluid (CSF) cultures yielded T. asahii and its morphology was demonstrated by Gram staining. The patient initially received empiric antifungal therapy with voriconazole (VCZ), and the subsequent drug sensitivity results supported the continuation of this therapy. Finally, 15 days of VCZ administration successfully achieved satisfactory therapeutic effects. This case highlights that T. asahii has emerged as an infectious cause of intracranial fungal infection in immunocompetent people. Early recognition and adequate antifungal treatment are paramount to ensure a favorable prognosis.
Invasive fungal infection is a serious life-threatening disease in humans, and their attributable mortality rates continue to rise with the expanding use of immunosuppressive agents (1). Trichosporon species are opportunistic fungal pathogens that are widespread in nature and colonize human skin, mucosal surfaces, and gastrointestinal tract, causing fungal infection not only in immunocompromised hosts but also in immunocompetent hosts (2). Noteworthy, Trichosporon asahii (T. asahii), the most common etiologic agent of Trichosporon species, is of increasing concern due to its ability to cause fatal infection with high mortality (3, 4). Until now, numerous studies have emphasized that T. asahii infection occurs in multiple organs, resulting in fungemia, urinary tract infection, subcutaneous mycoses, and keratitis, imposing a heavy infection burden on patients (4–7). However, T. asahii has rarely been considered the causative pathogen of intracranial infection. The first brain trichosporonosis was reported in 1970, and the patient died 4 weeks after hospital admission (8). To date, only five available cases of CNS T. asahii infection have been published, and the current treatment experience is still insufficient (9–13).
T. asahii exhibits high resistance to certain antifungal drugs, a characteristic that poses a serious challenge to clinical management, particularly for patients receiving long-term antibiotic therapy (4, 14). It is reported that T. asahii has intrinsic resistance to echinocandins and therefore echinocandins are not advocated for treating T. asahii infections (2). Triazoles can successfully treat T. asahii infection in different tissues and organs, with VCZ therapy resulting in higher survival rates than other antifungal drugs (1, 6, 15, 16). As for intracranial T. asahii infection, only two cases have been reported in which patients ultimately cured after receiving antifungal therapy (11, 13). One was treated with amphotericin B (AMB) followed by itraconazole (ITC) (11). The other was initially treated with AMB + voriconazole (VCZ) and then continued with VCZ (13). These existing studies provide limited therapeutic experiences in immunocompromised patients, but not in immunocompetent populations. Considering the potentially lethal consequences of brain infection, we believe intracranial T. asahii infection deserves more attention and more clinical practice is required to optimize the treatment strategies. In this report, we present a case of intracranial T. asahii infection successfully treated with VCZ in an immunocompetent patient who had undergone brain surgeries. We hope this study can provide references for accurate recognition and rational treatment of T. asahii infection in clinical practice.
In July 23, 2024, a 66-year-old woman was admitted to our hospital with a 2-month history of obstructive hydrocephalus. Before this patient arrived at our hospital, she was diagnosed with a ruptured and hemorrhaged left posterior inferior cerebellar artery aneurysm and underwent intracranial surgery at another hospital. Postoperatively, the patient developed obstructive hydrocephalus and fever on June 4, 2024. Carbapenem-resistant Acinetobacter baumannii was later identified in blood and CSF cultures on June 11, 2024. After about 2 months of successive treatment with different antibiotics such as meropenem, vancomycin, polymyxin, and amikacin, her infection was eventually well controlled. Afterward, she presented to our hospital for further control of obstructive hydrocephalus.
Supplementary Figure 1 illustrates the timeline of the patient’s disease progression during hospitalization. On admission, she presented with a blood pressure of 119/71 mmHg, heart rate of 66 beats/minute, respiratory rate of 19 breaths/minute, body temperature of 35.0°C, and oxygen saturation in arterial blood (SaO2) of 95.3%. Physical examination revealed mild coma with a Glasgow Coma Scale score of 4, decreased pupillary light reflexes in both eyes, neck stiffness, and a positive Babinski sign. The infection markers such as white blood cell count (WBC) and percentage of neutrophilic granulocyte (NEUT%) were within the normal limits. CSF was abnormal which showed glucose 5.36 mmol/L, protein content 2232.5 mg/dL, and WBC 84 × 106/L (Table 1). Computed tomography (CT) of the brain indicated dilation and communicating hydrocephalus in the bilateral ventricles, the third and fourth ventricles (Figure 1A). The primary diagnosis was subsequently confirmed as obstructive hydrocephalus.
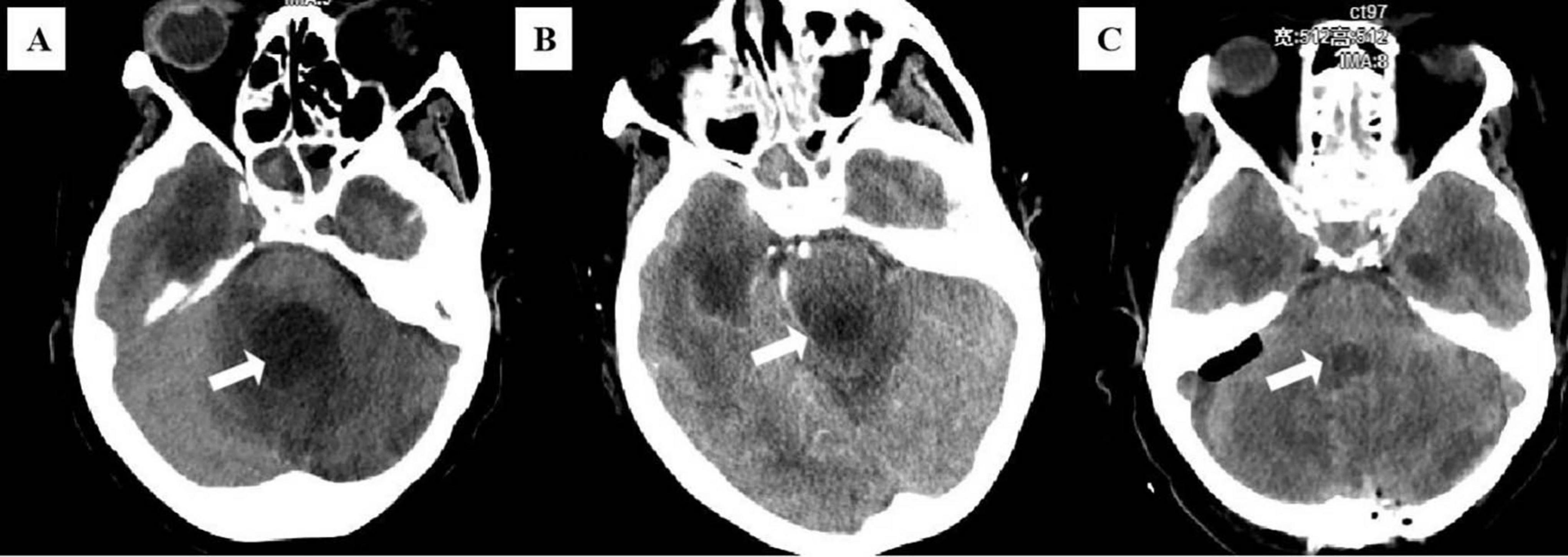
Figure 1. (A) Brain computed tomography (CT) images at the initial diagnosis. (B) Brain CT before the second cranial surgery on 4th day. (C) Brain CT after the second cranial surgery on 9th day. White arrows indicate the lesion in the fourth ventricle.
On the 5th hospital day, the patient experienced projectile vomiting with a heart rate dropping to 40 beats/minute, which was considered to be a consequence of exaggerated obstructive hydrocephalus. She immediately underwent surgical treatment with brain ventricle puncture and drainage. After surgery, the vomiting improved, but pathological breathing developed and her heart rate decreased to 35–60 beats/minute. A brain CT scan showed obvious enlargement of the fourth ventricle with compression of the medulla oblongata (Figure 1B). As a result, a second surgery (brain ventricle puncture and drainage, fourth ventricular lesion resection, and suboccipital decompression) was performed on the 8th day of hospitalization. Postoperatively, her symptoms gradually subsided, and CT demonstrated evidence of improvement in ventricular dilation and effusion (Figure 1C). On the 15th day of admission, she suffered from vomiting and intermittent fever, with a maximum temperature of 38.2°C. As listed in Table 1, laboratory tests demonstrated a significantly elevated WBC (11.38 × 109/L) and NEUT% (83.92%). CSF analysis revealed glucose of 3.96 mmol/L, protein of 172.2 mg/dL, and WBC of 323 × 106/L, all of which were abnormal. Considering that the obstructive hydrocephalus had significantly improved and that prolonged drain placement may contribute to infection, the ventricular drainage device was removed. Subsequently, her CSF, sputum, and blood were collected for culture. Considering no pathogenic microorganisms were detected in the sputum and blood cultures, and neither the chest CT scan nor the clinical presentation showed signs of lung infection, lung and blood infections were excluded. However, the CSF cultures were positive after 24 h of incubation, and isolates from CSF cultures showed septate hyphae with arthrospores by Gram staining (Figure 2). The species in the CSF cultures was then identified as T. asahii, and the diagnosis of intracranial T. asahii infection was established. On the 17th hospital day, voriconazole (0.4 g/twice daily for two doses, then 0.2g/twice daily for 2 weeks, intravenously) was empirically administered to combat T. asahii invasion. Shortly after, antifungal susceptibility testing proved that voriconazole exhibited potent activity against T. asahii with a MIC of 0.125 μg/mL (Table 2). After 5 days of voriconazole treatment, the patient was afebrile and her serum WBC and NEUT% returned to normal (Table 1). Consequently, the patient completed 15 days of voriconazole monotherapy and was discharged on hospital day 55 in stable condition and without recurrent infection.
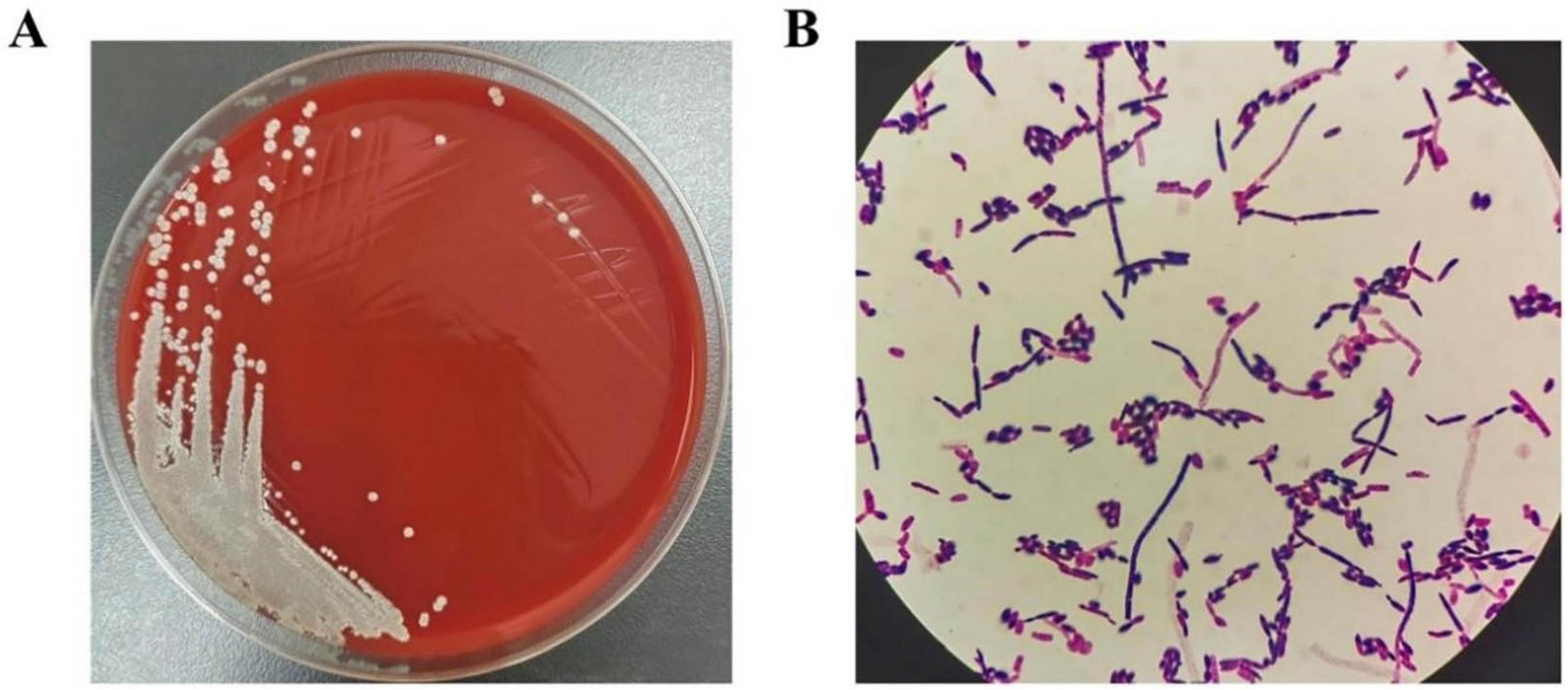
Figure 2. (A) T. asahii in CSF appears as white yeast-like fungus after 24 h of incubation on blood agar plates. (B) Microscopy of isolates (Gram staining) showing T. asahii septate hyphae with arthrospores.
Cerebrospinal fluid (CSF) was inoculated onto blood agar plates for 24 h at 35°C under aerobic conditions (Figure 2A). Yeast-like fungi were isolated from the CSF cultures and showed septate hyphae with arthrospores by Gram staining under microscopy (Figure 2B). The fungus was further identified by Matrix-Assisted Laser Desorption/Ionization Time-of-Flight (MALDI-TOF) technology (Zybio, China, EXS 2600). According to the interpretation criteria, identification scores <1.7 indicate unreliable identification, 1.7–2.0 indicate genus-level identification, and ≥2.0 indicate species-level identification. The isolate in our case was identified at the species-level with an acceptable score of 2.06, thus the results provide a reliable T. asahii identification. Antifungal drug susceptibility analysis was performed by broth microdilution method with VITEK-2 Compact fully automated microbial susceptibility analysis system (France bioMérieux). The minimum inhibitory concentrations (MICs) of the antifungal drugs are listed in Table 2.
Invasive infection induced by T. asahii is considered extremely fatal and difficult to treat, as it usually progresses rapidly, with 78% of patients dying even after receiving antifungal therapy (2, 17). Therefore, delayed diagnosis and inadequate treatment indeed aggravate patient suffering and increase mortality rates. Despite numerous reports indicating that T. asahii is responsible for infections in multiple tissues and organs, it has rarely been reported as the etiological agent of intracranial infection (18, 19). We believe its invasiveness and pathogenicity in the CNS cannot be ignored. Supplementary Table 1 summarizes the five existing cases of CNS T. asahii infection, among which only two reported that the patients were ultimately recovered following antifungal therapy. The first case was reported in 2007 involving a non-immunocompromised host who partially responded to fluconazole therapy but finally lost to follow-up (9). In 2011, a second case of Trichosporon meningitis and cerebral abscess was described in a burn patient who succumbed to multiorgan dysfunction prior to receiving appropriate antifungal therapy (10). Subsequently, the first case of successful treatment of CNS T. asahii infection was published, in which a patient undergoing immunosuppressive therapy for autoimmune hepatitis was diagnosed with a Trichosporon brain abscess, which resolved after treatment with AMB and ITC (11). In 2015, an immunocompetent patient presenting with chronic meningo-ventriculitis and intra ventricular fungal ball caused by T. asahii was reported (12). This patient received empirical treatment with liposomal amphotericin B but unfortunately died rapidly from cardiac arrest shortly thereafter. Soon after, another study detailed an allogeneic hematopoietic stem cell transplant (allo-HSCT) recipient suffering from invasive CNS trichosporonosis who achieved recovered after administering VCZ (13). It is noteworthy that both cured patients were immunosuppressed, and considering that there are no references on the successful management of intracranial T. asahii infection in immunocompetent populations, we report a rare intracranial T. asahii infection in a 66-year-old immunocompetent female in China.
To date, the exact mechanism by which T. asahii infects humans is not fully understood. The available data suggest that the infection mechanisms of T. asahii may be related to its adherence proteins binding to host proteins (e.g., serum albumin, α-1antitrypsin, and vitronectin) (20). Nowadays, increasing studies have discovered that T. asahii primarily affects immunocompromised patients and rarely infects individuals with normal immunity (1, 21, 22). Interestingly, the patient in our case was immunocompetent. It was found that other factors besides immune status can also increase the risk of T. asahii infection, especially antibiotic use and invasive medical devices (23). Among them, it was demonstrated that T. asahii can escape the immune response by adhering to implanted devices to form biofilms, which can facilitate its invasion (2). We speculate that this feature may contribute to the T. asahii infection in immunocompetent patients. Consistent with these findings, our patient had an underlying history of prolonged exposure to antibiotics, brain surgeries, and drainage device placement, all of which are recognized as existing risk factors contributing to intracranial T. asahii infection. Furthermore, T. asahii is a commensal of human skin and can infect humans from colonized skin via catheters or central lines (1, 24). We suspect that T. asahii in this case was transferred from human skin to the brain through ventricular drainage devices.
Notably, T. asahii is inherently resistant to multiple antifungal drugs, and its limited therapeutic options have become a matter of concern. To date, there are no specific therapeutic recommendations for T. asahii infection as the MIC breakpoints have not been defined and convincing evidence from randomized clinical trials is lacking (25). Since echinocandins exhibit no antifungal activities against T. asahii, they are not advocated for treating T. asahii infection (25, 26). The 2021 global guideline for the diagnosis and management of rare yeast infections recommends VCZ as a first-line agent for initial therapy against T. asahii (25). However, this recommendation is primarily based on animal studies, in vitro experiments, and case reports, and the optimal antifungal therapy for T. asahii infection remains uncertain. An in-depth study comparing the effects of various triazoles on clinically isolated T. asahii showed that VCZ had the highest fungistatic activity, followed by itraconazole, posaconazole, and isavuconazole, which had equal activity against T. asahii, whereas fluconazole showed the lowest antifungal activity (26). From the above, we can see the remarkable efficacy of VCZ on T. asahii infection. When compared to polyenes, guidelines suggest that VCZ is superior to AMB because T. asahii is usually resistant to AMB (MICs ≥ 2 μg/mL) (25, 27). Furthermore, some case reports have demonstrated that switching to VCZ resulted in satisfactory patient outcomes when AMB-based regimens failed to control T. asahii infection (17, 28). Based on this information, VCZ appears to be a better therapeutic choice for invasive trichosporiasis than AMB. Moreover, combination therapy with VCZ and AMB was also utilized in some studies, but the results were not superior to monotherapy (23). Referring to the 2021 guidelines, azole-polyene combinations are recommended as salvage therapy since it has no advantage as initial therapy (25). Therefore, a combination regimen was not adopted for the initial treatment of our patient. It is worth noting that although there is sufficient evidence that azoles are the best first-line antifungal therapy agents for T. asahii infection, some findings indicate that long-term use of azoles therapy may lead to mutations in the ERG11 gene, resulting in the emergence of azole-resistant isolates (3).
In our case, the patient was empirically started on VCZ according to accessible guidelines and literature. Subsequently, the results of antifungal susceptibility testing revealed that the isolate had low MICs for VCZ (MIC = 0.125 μg/mL), AMB (MIC ≤ 0.5 μg/mL), itraconazole (MIC = 0.25 μg/mL), and fluconazole (MIC = 2 μg/mL). Combining previous studies and our data, we consider that VCZ monotherapy may be the preferred regimen for our patient, and the patient continued administering VCZ 400 mg/day for an adequate duration. After 15 days of VCZ monotherapy, the patient’s infection symptoms resolved completely without serious complications or recurrence until discharge. Indeed, the duration of antifungal therapy required for recovery in our patients was relatively short compared to other reported patients with CNS T. asahii infection (11, 13). We hypothesize that one possible explanation is that our patient had normal immune function that facilitates microbial pathogen clearance. On the other hand, we provided rapid diagnosis and effective treatment to control the infection before it worsened successfully. Based on these factors, her intracranial T. asahii infection was eventually cured rapidly.
We describe a case of intracranial T. asahii infection in an immunocompetent patient who was ultimately cured by VCZ. It is worth reflecting that timely diagnosis and appropriate clinical treatment are key to a favorable prognosis. Initiation of azole antifungal therapy, particularly VCZ, may be an optimal choice to control intracranial T. asahii infection. Moreover, clinicians should remain vigilant that T. asahii infection is not restricted only to immunosuppressed populations. Long-term antibiotic use, surgical operation, or drainage device placement may increase the risk of T. asahii infection in immunocompetent individuals, and close monitoring of these populations can prevent the occurrence of T. asahii infection.
The original contributions presented in this study are included in this article/Supplementary material, further inquiries can be directed to the corresponding authors.
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
CH: Writing – original draft, Conceptualization, Data curation. DZ: Data curation, Methodology, Writing – review and editing. XW: Methodology, Resources, Writing – review and editing. HW: Resources, Data curation, Writing – review and editing. LH: Formal Analysis, Writing – review and editing. ML: Conceptualization, Data curation, Project administration, Writing – original draft, Writing – review and editing. EC: Conceptualization, Formal Analysis, Writing – review and editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2025.1560016/full#supplementary-material
1. Boutin CA, Luong ML. Update on therapeutic approaches for invasive fungal infections in adults. Ther Adv Infect Dis. (2024) 11:1–21. doi: 10.1177/20499361231224980
2. Mehta V, Nayyar C, Gulati N, Singla N, Rai S, Chandar JA. Comprehensive review of trichosporon spp.: An invasive and emerging fungus. Cureus. (2021) 13:e17345. doi: 10.7759/cureus.17345
3. Padovan A, Rocha W, Toti A, Freitas de Jesus DF, Chaves GM, Colombo AL. Exploring the resistance mechanisms in Trichosporon asahii: Triazoles as the last defense for invasive trichosporonosis. Fungal Genet Biol. (2019) 133:103267. doi: 10.1016/j.fgb.2019.103267
4. Hu L, Liu G, Chen X. Rhinocerebral mucormycosis and Trichosporon asahii fungemia in a pediatric patient with acute lymphoblastic leukemia: A rare coinfection. Rev Inst Med Trop Sao Paulo. (2024) 66:e41. doi: 10.1590/S1678-9946202466041
5. Vianello M, Jesus D, Sampaio J, de Oliveira G, Lincopan N, Ishida K. Possible Trichosporon asahii urinary tract infection in a critically ill COVID-19 patient. Rev Iberoam Micol. (2022) 39:54–6. doi: 10.1016/j.riam.2022.05.001
6. Kruschewsky W, Massaroni-Peçanha P, Maifrede S, Leite M, Pôssa T, Alberto-Lei F, et al. Trichosporon asahii causing subcutaneous mycoses in an immunocompetent patient: Case report and a minireview. Braz J Microbiol. (2022) 53:1221–9. doi: 10.1007/s42770-022-00737-x
7. Shin J, Son W, Cho K, Rho C. Bilateral Trichosporon asahii keratitis after ptosis correction: A case report. Medicine (Baltimore). (2021) 100:e26688. doi: 10.1097/MD.0000000000026688
8. Watson K, Kallichurum S. Brain abscess due to Trichosporon cutaneum. J Med Microbiol. (1970) 3:191–3. doi: 10.1099/00222615-3-1-191
9. Rastogi V, Nirwan P. Invasive trichosporonosis due to Trichosporon asahii in a non-immunocompromised host: A rare case report. Indian J Med Microbiol. (2007) 25:59–61. doi: 10.4103/0255-0857.31065
10. Heslop O, Nyi Nyi M, Abbott S, Rainford L, Castle D, Coard K. Disseminated trichosporonosis in a burn patient: Meningitis and cerebral abscess due to Trichosporon asahii. J Clin Microbiol. (2011) 49:4405–8. doi: 10.1128/JCM.05028-11
11. Basiri K, Meidani M, Rezaie F, Soheilnader S, Fatehi F. A rare case of Trichosporon brain abscess, successfully treated with surgical excision and antifungal agents. Neurol Neurochir Pol. (2012) 46:92–5. doi: 10.5114/ninp.2012.27180
12. Kumar A, Udayakumaran S, Babu R, Rajamma B, Prakash A, Panikar D, et al. Trichosporon asahii infection presenting as chronic meningo-ventriculitis and intra ventricular fungal ball: A case report and literature review. Mycoses. (2015) 58:99–103. doi: 10.1111/myc.12282
13. Thien S, Chung S, Tan A, Hwang W, Tan B, Tan T. Recurrent trichosporonosis with central nervous system involvement in an allogeneic hematopoietic stem cell transplant recipient. Transpl Infect Dis. (2016) 18:768–72. doi: 10.1111/tid.12577
14. Li H, Wang C, Chen Y, Zhang S, Yang R. Integrated transcriptomic analysis of Trichosporon Asahii uncovers the core genes and pathways of fluconazole resistance. Sci Rep. (2017) 7:17847. doi: 10.1038/s41598-017-18072-9
15. Al Momani M, Yusef D, Hamasha D, Hamad M, Farran S. Complicated Trichosporon asahii mastoiditis in immunocompetent child. BMC Infect Dis. (2021) 21:1229. doi: 10.1186/s12879-021-06915-w
16. Lee E, Koh MJA. A rare case of cutaneous Trichosporon asahii infection in an immunocompromised child. Pediatr Dermatol. (2020) 37:962–3. doi: 10.1111/pde.14251
17. Fournier S, Pavageau W, Feuillhade M, Deplus S, Zagdanski A, Verola O, et al. Use of voriconazole to successfully treat disseminated Trichosporon asahii infection in a patient with acute myeloid leukaemia. Eur J Clin Microbiol Infect Dis. (2002) 21:892–6. doi: 10.1007/s10096-002-0841-y
18. Mulč A, Rossini F, Sollima A, Lenzi A, Fumarola B, Amadasi S, et al. Trichosporon asahii infective endocarditis of prosthetic valve: A case report and literature review. Antibiotics (Basel). (2023) 12:1181. doi: 10.3390/antibiotics12071181
19. Mello López A, da Silva Neto LA, de Oliveira VF, de Carvalho VC, de Oliveira PRD, Lima A. Orthopedic infections due to Trichosporon species: Case series and literature review. Med Mycol. (2022) 61:myad001. doi: 10.1093/mmy/myad001
20. Ichikawa T, Okumura M, Mizumura T, Hayashi M, Watanabe M, Ikeda R, et al. Interaction of host proteins with cell surface molecules of the pathogenic yeast Trichosporon asahii. Med Mycol J. (2023) 64:29–36.
21. Kourti M, Roilides E. Invasive Trichosporonosis in neonates and pediatric patients with malignancies or hematologic disorders. Pathogens. (2022) 11:242.
22. Colombo A, Padovan A, Chaves G. Current knowledge of Trichosporon spp. and Trichosporonosis. Clin Microbiol Rev. (2011) 24:682–700.
23. Li H, Guo M, Wang C, Li Y, Fernandez A, Ferraro T, et al. Epidemiological study of Trichosporon asahii infections over the past 23 years. Epidemiol Infect. (2020) 148:e169. doi: 10.1017/S0950268820001624
24. Cordeiro R, Aguiar A, da Silva B, Pereira L, Portela F, de Camargo Z, et al. Trichosporon asahii and Trichosporon inkin biofilms produce antifungal-tolerant persister cells. Front Cell Infect Microbiol. (2021) 11:645812. doi: 10.3389/fcimb.2021.645812
25. Chen S, Perfect J, Colombo A, Cornely O, Groll A, Seidel D, et al. Global guideline for the diagnosis and management of rare yeast infections: An initiative of the ECMM in cooperation with ISHAM and ASM. Lancet Infect Dis. (2021) 21: e375–86.
26. Hazirolan G, Canton E, Sahin S, Arikan-Akdagli S. Head-to-head comparison of inhibitory and fungicidal activities of fluconazole, itraconazole, voriconazole, posaconazole, and isavuconazole against clinical isolates of Trichosporon asahii. Antimicrob Agents Chemother. (2013) 57:4841–7. doi: 10.1128/AAC.00850-13
27. Arendrup M, Boekhout T, Akova M, Meis J, Cornely O, Lortholary O. ESCMID and ECMM joint clinical guidelines for the diagnosis and management of rare invasive yeast infections. Clin Microbiol Infect. (2014) 20:76–98.
Keywords: intracranial infection, Trichosporon asahii, immunocompetent patient, antifungal treatment, treatment efficacy
Citation: He C, Zhao D, Wang X, Wang H, Huang L, Liu M and Chen E (2025) Voriconazole successfully treats intracranial Trichosporon asahii infection in an immunocompetent patient: a rare case report and literature review. Front. Med. 12:1560016. doi: 10.3389/fmed.2025.1560016
Received: 15 January 2025; Accepted: 27 February 2025;
Published: 20 March 2025.
Edited by:
Sam Donta, Falmouth Hospital, United StatesReviewed by:
Tong-Bao Liu, Southwest University, ChinaCopyright © 2025 He, Zhao, Wang, Wang, Huang, Liu and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Enqiang Chen, Y2hlbmVucWlhbmcxOTgzQGhvdG1haWwuY29t; Maozhu Liu, bWFvemh1MjAyMUAxMjYuY29t
†These authors have contributed equally to this work
‡ORCID: Cuilin He, orcid.org/0009-0008-1682-0624; Maozhu Liu, orcid.org/0009-0004-4747-9263
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.