
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Med. , 19 March 2025
Sec. Rheumatology
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1552114
This article is part of the Research Topic Sarcoidosis Diagnosis and Treatment Based on Etiology View all 4 articles
 Yuiko Masuda1
Yuiko Masuda1 Hiroko Okabayashi1*
Hiroko Okabayashi1* Kimitaka Akaike1
Kimitaka Akaike1 Shohei Hamada1
Shohei Hamada1 Aiko Masunaga1
Aiko Masunaga1 Hidenori Ichiyasu1
Hidenori Ichiyasu1 Kenichi Ohashi2
Kenichi Ohashi2 Takuro Sakagami1
Takuro Sakagami1Introduction: Tattoo sarcoidosis is characterized by a granulomatous reaction localized to the tattoo site and typical systemic symptoms of sarcoidosis. Herein, we report the case of a patient diagnosed with tattoo sarcoidosis.
Case report: A 28-year-old man presented with subcutaneous nodules at two tattoo sites, bilateral hilar and mediastinal lymphadenopathy, multiple micronodules predominantly in the upper lobes of both lungs, elevated serum angiotensin-converting enzyme and soluble interleukin-2 receptor levels, hypercalcemia, and renal dysfunction. Skin biopsy of a subcutaneous nodule revealed epithelioid cell granulomas. Although periodic acid-Schiff, Grocott methenamine silver, and acid-fast staining showed negative results, antibody staining for Propionibacterium acnes within the epithelioid cell granuloma was positive. Remarkably, all lesions spontaneously resolved, and the systemic manifestations also improved without medical treatment.
Conclusion: The pathogenesis of tattoo sarcoidosis remains unknown, although an immune response to tattoo pigments has been suspected. However, there is a theory that P. acnes is the causative agent of sarcoidosis. In the present case, the detection of P. acnes within the epithelioid cell granuloma suggests that the bacterium may play a role in the etiology of tattoo sarcoidosis.
Sarcoidosis is a rare, multisystem inflammatory disease characterized by granuloma formation in various tissues and organs. While the exact cause remains unknown, it is believed to result from an abnormal immune response triggered by environmental factors, infectious agents, or genetic predispositions (1). A hypothesis suggests that Propionibacterium acnes—currently referred to as Cutibacterium acnes—may play a role in its pathogenesis (2).
Tattoos trigger various dermatological conditions, and localized granulomatous reactions at tattoo sites are referred to as tattoo granulomas. Tattoo sarcoidosis describes cases in which granulomatous reactions are accompanied by systemic manifestations characteristic of sarcoidosis. Although the immune response to tattoo pigments has been proposed as a cause of tattoo sarcoidosis, the exact etiology remains unclear (3). Herein, we present a case of tattoo sarcoidosis in which P. acnes was detected within an epithelioid cell granuloma.
A 28-year-old man was referred to our hospital after abnormal chest shadows were detected during a routine health examination. He noted subcutaneous nodules in the tattooed areas. He had a 1-year history of chronic cough, dyspnea on exertion, and loss of appetite and weight. He had completed a full-body tattoo over a 4-year period, beginning 5 years prior to presentation. The patient was a current smoker, with an approximately 18 pack-year smoking history. On admission, his temperature was 36.4°C, and oxygen saturation was 97% on room air. Subcutaneous nodules were palpable on the reddish-brown tattoos on the lower back and black-inked tattoos on the left thigh (Figure 1).
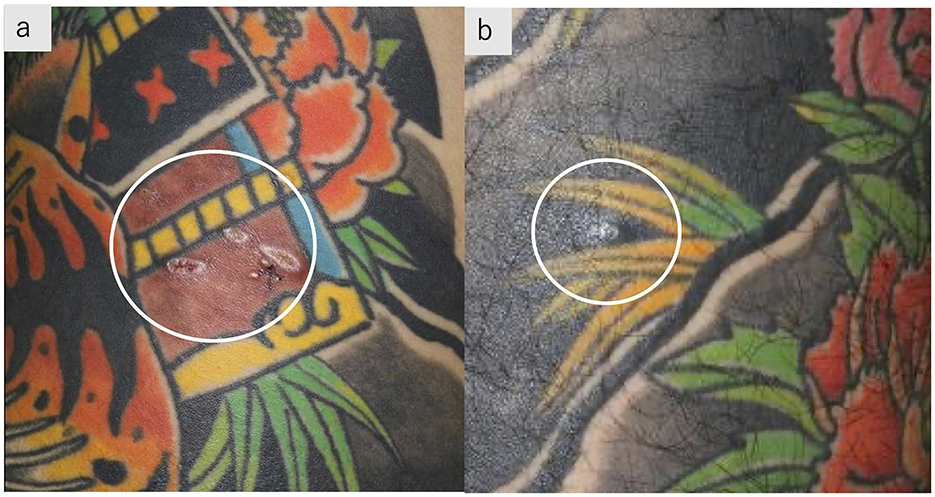
Figure 1. Tattoos with subcutaneous nodules. Subcutaneous nodules palpable on reddish-brown tattoos on the right lower back (a) and black-inked tattoos on the left thigh (b).
Investigations upon admission showed renal dysfunction, hypercalcemia, and elevated urinary N-acetyl-β-D-glucosaminidase. Serum angiotensin-converting enzyme (ACE), soluble interleukin-2 receptor (sIL-2R), and lysozyme levels were also elevated (Table 1). Chest radiographs showed bilateral hilar lymphadenopathy (Figure 2a), and high-resolution computed tomography (CT) scans revealed multiple nodular and granular opacities along the bronchovascular bundles, predominantly in the upper lobes, forming a galaxy sign in the right upper lobe. Enlarged hilar and mediastinal lymph nodes were observed (Figure 2b). Gallium scintigraphy revealed significant uptake in the hilar and mediastinal lymph nodes, left supraclavicular lymph node, ileocolic lymph node, right inguinal lymph node, and granular shadows of the right upper lobe (Figure 2c). Pulmonary function testing revealed no restrictive impairment (%FVC 86.8%) but did indicate diffusion impairment (%DLCO 73.4%).
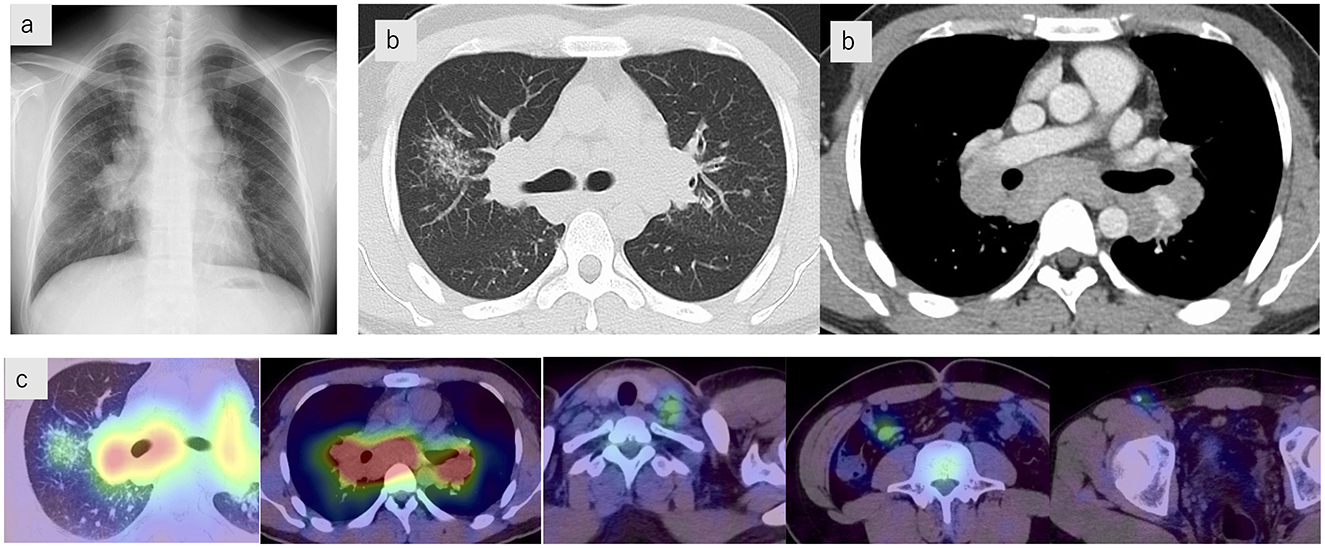
Figure 2. Imaging findings. (a) Chest radiograph showing bilateral hilar lymphadenopathy. (b) Chest computed tomography scans showing multiple nodular and granular opacities along the bronchovascular bundles, predominantly in the upper lobes, forming a galaxy sign in the right upper lobe. Enlarged hilar and mediastinal lymph nodes are also observed. (c) Gallium scintigraphy demonstrates significant uptake in the hilar and mediastinal lymph nodes, the left supraclavicular lymph node, the ileocolic lymph node, the right inguinal lymph node, and the granular shadows of the right upper lobe.
Bronchoalveolar lavage fluid analysis revealed a lymphocyte ratio of 6.5% and an elevated CD4/CD8 ratio of 7.2. Endobronchial ultrasonography-guided transbronchial needle aspiration was performed on the subcarinal lymph nodes; however, the specimen was of poor quality. Skin biopsy of the reddish-brown tattoo on the right lower back revealed numerous epithelioid cell granulomas in the upper dermis, with some areas of fibrinoid necrosis (Figure 3). Staining for pathogens, such as periodic acid-Schiff, Grocott's methenamine silver, and acid-fast bacilli, was negative. Immunohistochemistry with the P. acnes-specific monoclonal antibody (PAB antibody) revealed fine red granules, indicating P. acnes within the epithelioid cells and multinucleated giant cells of the granulomatous lesion (Figure 4). Based on these findings, the patient was diagnosed with tattoo-associated sarcoidosis. In this case, an elevated serum creatinine level and an increased urinary NAG/creatinine ratio (44.7 IU/g·Cr) were observed, raising the possibility of interstitial nephritis. However, Ga scintigraphy showed no renal uptake, and renal biopsy could not be performed due to the patient's lack of consent.
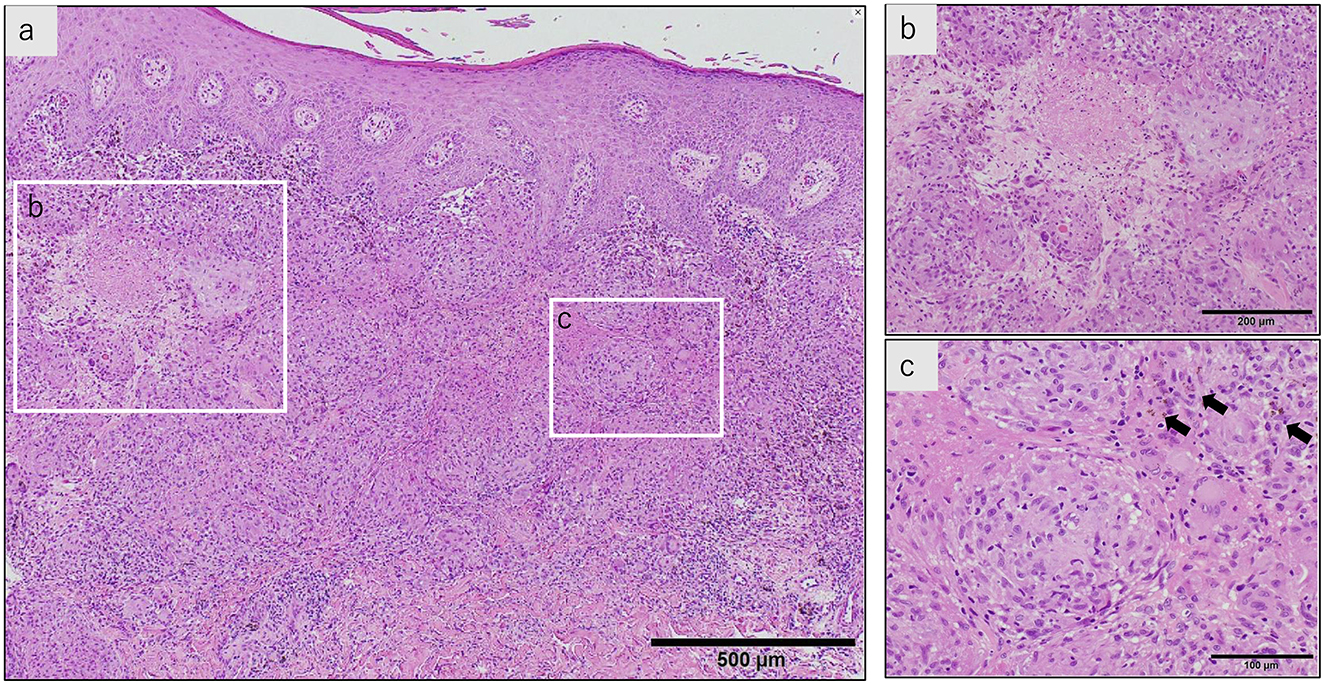
Figure 3. A skin biopsy of the reddish-brown tattoo on the right lower back (hematoxylin-eosin stain). (a) The stratum corneum shows parakeratosis, and the epidermis is thickened with spongiosis. There is a marked infiltration of inflammatory cells just below the epidermis, and multiple granulomas composed of epithelioid cells and lymphocytes are present. Langerhans giant cells are also seen. (b) Fibrinoid necrosis is observed in some areas. (c) Within the epithelioid cell granulomas, reddish-brown granular pigments, presumed to be tattoo ink, are observed (arrow).
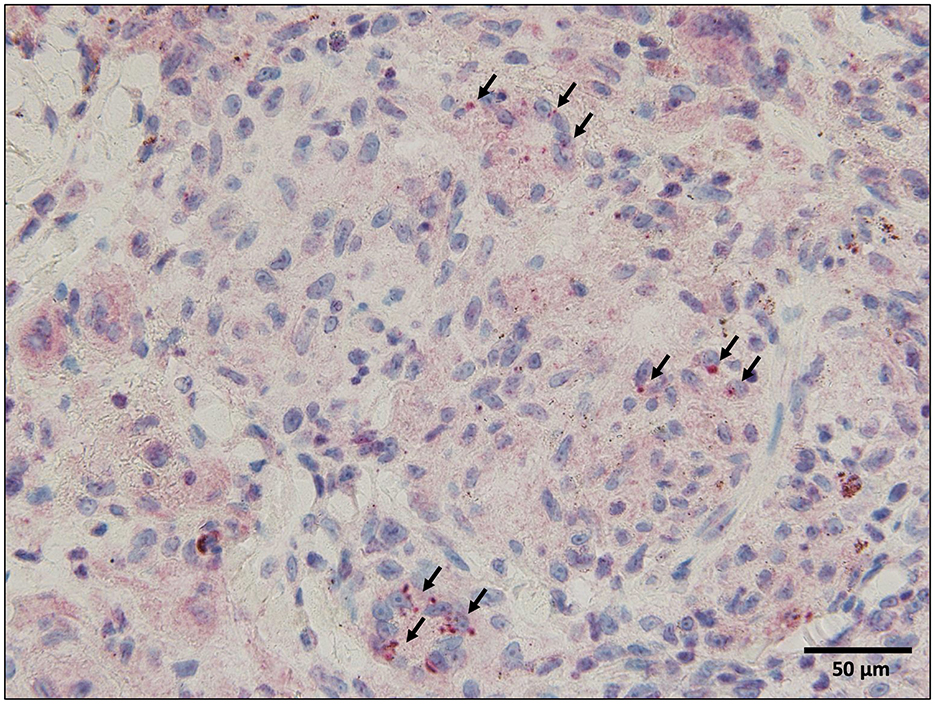
Figure 4. Immunohistochemistry with the Propionibacterium acnes-specific monoclonal antibody (PAB antibody) Since the tattoo pigment is reddish-brown, the chromogen 3-amino-9-ethylcarbazole (AEC) was used for immunohistochemical staining to differentiate between positive findings and tattoo pigment, and P. acnes is stained red. Immunohistochemistry with the PAB antibody reveals fine red granules (arrows), indicating P. acnes within the epithelioid cells, and multinucleated giant cells of the granulomatous lesion.
Although systemic steroid therapy was planned, the patient's symptoms, renal dysfunction, hypercalcemia, and bilateral hilar lymphadenopathy improved during the investigation period; therefore, pharmacological treatment was deferred. One year later, chest CT revealed reduced hilar and mediastinal lymphadenopathy and pulmonary lesions, and the serum ACE and sIL-2R levels also normalized. Subcutaneous nodules at tattoo sites resolved spontaneously. The patient continues with outpatient follow-up with no signs of recurrence.
Tattoo sarcoidosis is rare, and its incidence and prevalence remain unclear. According to a study by Ohtsuka et al. examining tattoo sarcoidosis in the Japanese population, the condition predominantly affected men and showed a bimodal age distribution, peaking in the 20s−30s and again in the 60s. The median time from tattoo application to onset was 13.7 years. In most cases, tattoo-related reactions were the initial manifestations of sarcoidosis. The possible reasons for the long duration between tattoo application and symptom onset include the slow degradation of tattoo pigments, chronic exposure to ultraviolet light, and the role of additional factors, such as hepatitis C virus infection or interferon therapy (4).
The incidence, clinical symptoms, and severity of sarcoidosis vary according to race and sex, likely owing to the combined effects of genetic and environmental factors. In a study of Japanese patients with tattoo-associated sarcoidosis by Ohtsuka et al., features such as sex, age distribution, median tattoo duration, and frequency of intrathoracic lymphadenopathy were similar to those reported by Kluger in cases involving other racial groups. However, the frequency of uveitis is higher in Japanese patients with tattoo sarcoidosis than in patients of other ethnicities (5). This may reflect a genetic predisposition of Japanese individuals, as ocular involvement is more prevalent in Japanese patients with sarcoidosis than in Western populations. In previous reports, the treatment of tattoo sarcoidosis has frequently involved oral steroids, immunosuppressants, local treatment, and tattoo removal. However, there are also reports in which the condition naturally improved without treatment (6–11).
Although the etiology of tattoo sarcoidosis remains poorly understood, several mechanisms have been proposed. A key factor in the pathogenesis is the activation of the immune system, particularly a heightened Th1 immune response. Tattoo pigments are thought to act as exogenous antigens that trigger granulomatous reactions. This reaction may be exacerbated by the persistence of tattoo pigments in the dermis, which can serve as a continuous stimulus to the immune system even years after tattooing.
A theory suggests that immune reactions to tattoo pigments may trigger sarcoidosis, but there is also a theory that postulates that tattoos are not the cause, but rather a target for sarcoidosis. Tattoos may trigger an enhanced immune response in patients predisposed to sarcoidosis (5). Scar sarcoidosis is a condition in which skin sarcoidosis occurs in scars formed by trauma, surgical procedures, shingles, or other infections, and tattoos and permanent makeup can also be precursor conditions. Repeated skin microinjuries may also be involved in the development of Köbner's phenomenon (12, 13).
The etiology of sarcoidosis remains incompletely understood, although it is believed to arise from a complex interplay between genetic predisposition, environmental exposure, and immune dysregulation. Sarcoidosis is characterized by the formation of non-caseating granulomas that seem to result from an exaggerated immune response to unidentified antigens.
In Japan, the leading theory is that P. acnes is the causative bacterium of sarcoidosis. P. acnes DNA is frequently detected in sarcoidosis granulomas (14). PAB antibody is a monoclonal antibody that reacts with a glycolipid antigen called lipoteichoic acid distributed throughout the cell wall of P. acnes bacterial cell membranes, and when immunostaining is performed on sarcoidosis lesions, a large percentage of positivity is seen in the intracellular spaces of epithelioid cells and giant cells within the granulomas (15). P. acnes enters the body of healthy people via the respiratory tract, causing subclinical infection at the cellular level of the lungs and hilar lymph nodes, but an environmental factor or host predisposition may cause endogenous relapse in some people, resulting in chronic granuloma formation.
In the present case, P. acnes was detected within the granuloma of the skin lesion. To the best of our knowledge, this is the first case report of P. acnes detection in tattoo sarcoidosis. Whether this patient had a predisposition to sarcoidosis, whether tattooing exacerbated the condition, or whether the tattooing procedure caused P. acnes, which is normally present in the stratum corneum, to enter the subcutaneous tissue and cause sarcoidosis remained unknown. However, the fact that P. acnes was detected in the granuloma suggests that P. acnes may be involved in the pathogenesis of tattoo sarcoidosis. Until now, the suspected trigger for tattoo sarcoidosis was repeated microscopic damage to the pigment particles or to the skin itself, but we believe that the examination of increasing numbers of future cases for the presence of P. acnes will lead to a better understanding of the etiology of tattoo sarcoidosis.
In conclusion, this case of tattoo sarcoidosis highlights the potential role of P. acnes in the pathogenesis of this rare form of sarcoidosis. Detection of P. acnes within granulomatous lesions on tattooed skin suggests that this bacterium may play a role in the initiation or exacerbation of sarcoidosis in predisposed individuals.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
YM: Writing – original draft, Writing – review & editing. HO: Writing – original draft, Writing – review & editing. KA: Writing – review & editing. SH: Writing – review & editing. AM: Writing – review & editing. HI: Writing – review & editing. KO: Writing – review & editing. TS: Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
We would like to thank Editage (http://www.editage.com/) for the English language editing.
The authors declare that this study was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Grunewald J, Grutters JC, Arkema EV, Saketkoo LA, Moller DR, Müller-Quernheim J. Sarcoidosis. Nat Rev Dis Primers. (2019) 5:45. doi: 10.1038/s41572-019-0096-x
2. Eishi Y. Etiologic aspect of sarcoidosis as an allergic endogenous infection caused by Propionibacterium acnes. BioMed Res Int. (2013) 2013:935289. doi: 10.1155/2013/935289
3. Hanada K, Chiyoya S, Katabira Y. Systemic sarcoidal reaction in tattoo. Clin Exp Dermatol. (1985) 10:479–84. doi: 10.1111/j.1365-2230.1985.tb00609.x
4. Ohtsuka M, Matsumura N, Yamamoto T. Sarcoidal granuloma presenting on tattoo: a report of a Japanese female patient and a review of Japanese published work. Sarcoidosis Vasc Diffuse Lung Dis. (2016) 33:83–9.
5. Kluger N. Sarcoidosis on tattoos: a review of the literature from 1939 to 2011. Sarcoidosis Vasc Diffuse Lung Dis. (2013) 30:86–102.
6. Weidman AI, Andrade R, Franks AG. Sarcoidosis. Report of a case of sarcoid lesions in a tattoo and subsequent discovery of pulmonary sarcoidosis. Arch Dermatol. (1966) 94:320–5. doi: 10.1001/archderm.1966.01600270070014
7. Kennedy C. Sarcoidosis presenting in tattoos. Clin Exp Dermatol. (1976) 1:395–9. doi: 10.1111/j.1365-2230.1976.tb01448.x
8. McGavin CR, Gaber K, Davies MG. Sarcoidosis in a gecko. Lancet. (2005) 365:1876. doi: 10.1016/S0140-6736(05)66620-2
9. Toulemonde A, Quereux G, Dréno B. Sarcoidosis granuloma on a tattoo induced by interferon alpha. Ann Dermatol Venereol. (2004) 131:49–51. doi: 10.1016/S0151-9638(04)93541-7
10. Baumgartner M, Feldmann R, Breier F, Steiner A. Sarcoidal granulomas in a cosmetic tattoo in association with pulmonary sarcoidosis. J Dtsch Dermatol Ges. (2010) 8:900–2. doi: 10.1111/j.1610-0387.2010.07391.x
11. Ikeda M, Suzuki K, Shirai T, Furuhashi K, Suda T, Chida K, et al. male case with granulomatous lung disease associated with a whole body tattoo. Jpn J Sarcoidosis Granulomatous Disord. (2008) 28:63–8.
12. Yamamoto T. Viewpoints of infiltrative scar and scar sarcoidosis in and outside Japan: proposal of a new classification of cutaneous manifestation of systemic sarcoidosis. JJSOGD. (2022) 42:12–5. doi: 10.7878/jjsogd.42.1_2_12
13. Marcoval J, Iriarte A, Rocamora G, Mañá J. Scar sarcoidosis: clinical features and prognostic significance. Sarcoidosis Vasc Diffuse Lung Dis. (2024) 41:e2024046.
14. Yamada T, Eishi Y, Ikeda S, Ishige I, Suzuki T, Takemura T, et al. In situ localization of Propionibacterium acnes DNA in lymph nodes from sarcoidosis patients by signal amplification with catalysed reporter deposition. J Pathol. (2002) 198:541–7. doi: 10.1002/path.1243
Keywords: tattoo sarcoidosis, sarcoidosis, tattoo, Propionibacterium acnes, Cutibacterium acnes
Citation: Masuda Y, Okabayashi H, Akaike K, Hamada S, Masunaga A, Ichiyasu H, Ohashi K and Sakagami T (2025) Case Report: Tattoo sarcoidosis with epithelioid cell granuloma positive for Propionibacterium acnes. Front. Med. 12:1552114. doi: 10.3389/fmed.2025.1552114
Received: 27 December 2024; Accepted: 28 February 2025;
Published: 19 March 2025.
Edited by:
Michiru Sawahata, Jichi Medical University, JapanReviewed by:
Mansoor C. Abdulla, Sultan Qaboos Hospital, OmanCopyright © 2025 Masuda, Okabayashi, Akaike, Hamada, Masunaga, Ichiyasu, Ohashi and Sakagami. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hiroko Okabayashi, aGlyb2tva2FiYUBrdWgua3VtYW1vdG8tdS5hYy5qcA==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.