
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Med. , 02 April 2025
Sec. Dermatology
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1550640
 Rose Alraddadi1,2
Rose Alraddadi1,2 Mulham Kalantan1,2
Mulham Kalantan1,2 Yara Aljefri1,2
Yara Aljefri1,2 Hadeel Maaddawi3
Hadeel Maaddawi3 Abdulrahman Alsamadani1,2
Abdulrahman Alsamadani1,2 Athoub Kadasa1,2
Athoub Kadasa1,2 Abdulrahman Softah1,2
Abdulrahman Softah1,2 Baraa Tabbakh1,2
Baraa Tabbakh1,2 Rahaf Alturkistani4
Rahaf Alturkistani4 Abdulhadi Jfri1,2,4*
Abdulhadi Jfri1,2,4*Introduction: Atopic dermatitis (AD) is a chronic inflammatory skin condition affecting 5%−20% of children and 2%−10% of adults worldwide. Treatment for moderate-to-severe AD includes biologics like dupilumab, tralokinumab, lebrikizumab, and JAK inhibitors (abrocitinib, upadacitinib). However, upper respiratory tract infections (URTIs) are commonly reported adverse events for these therapies. This meta-analysis aims to estimate the pooled incidence of URTIs associated with these treatments compared to topicals.
Methods: A systematic search was conducted across PubMed, MEDLINE, DOAJ, and ClinicalTrials.gov for randomized controlled trials (RCTs) involving AD patients treated with dupilumab, tralokinumab, lebrikizumab, abrocitinib, or upadacitinib, excluding studies of patients treated with topicals, Studies on other dermatitis types and biologics. Data on URTI events, sample sizes, and incidence were extracted. Study quality was assessed using the Cochrane Risk of Bias Tool (RoB 2). A random-effects meta-analysis was conducted using the Netmeta package in R, calculating odds ratios (ORs) with 95% confidence intervals (CIs).
Results: From 413 retrieved records, 21 studies met the inclusion criteria. URTI incidence of the treatment group in the included studies ranged from 0.35% to 41.5%, while control groups showed rates between 0% and 40%. Across all studies, URTI incidence was 9.70% in intervention groups and 8.03% in placebo groups (MH OR = 1.18, 95% CI: 0.98–1.42). Heterogeneity was low (I2 = 20.14%), with no evidence of publication bias (p = 0.83). There were no significant subgroup differences between patients taking different biological therapies (Q = 3.90, p = 0.42).
Conclusion: While URTIs are common adverse events for AD therapies, their incidence in intervention groups is similar to control, suggesting no significant increase in risk. These findings provide critical insights for clinicians in balancing efficacy and safety when selecting therapies for AD patients. Further research should explore patient-specific risk factors for URTIs.
Systematic review registration: Prospero registration code: [392093]. PROSPERO, Centre for Reviews and Dissemination: CRD42023392093.
Atopic dermatitis (AD) is a chronic inflammatory skin disease with significant patient and population burden (1) affecting 5%−20% of children and 2%−10% of adults worldwide. AD is characterized by clinical signs of redness, swelling, excoriation, lichenification, and often, oozing/weeping and xerosis (2, 3). AD is thought to be a multifactorial disease that arises due to both genetic and environmental factors, although the complete pathophysiology has yet to be elucidated (4). For management of moderate-to-severe cases of AD, phototherapy and systemic immunosuppressants can be used (5). Dupilumab (anti-IL4/13) is one biologic that has been approved for AD, and more recently, tralokinumb (anti-IL13), lebrikizumab (anti-IL4/13), abrocitinib and upadacitinib [Janus kinases (JAKs) inhibitors], have been added (6, 7). Nemolizumab which target IL-31 has shown potential in reducing pruritus, although its overall efficacy in achieving EASI-75 responses is similar to placebo (8).
However, biologics targeting IL-22, IL-33, OX40, and thymic stromal lymphopoietin (TSLP) are in various stages of development, with some showing potential in early trials (9, 10). The development of biologics is moving toward a more personalized approach, aiming to address the unique immune profiles of different AD subsets (11).
While biologics present a promising treatment option for AD, several challenges persist. Limited long-term safety data, particularly in pediatric populations, raise concerns about their potential impact on an immature immune system (11). Additionally, their efficacy varies among patients (8), and the heterogeneous nature of AD—characterized by different phenotypes—necessitates a more personalized approach to treatment (11).
Additionally, concerns regarding adverse effects, particularly the risk of infections, have emerged as key considerations in evaluating these therapies. Upper respiratory tract infections (URTIs) and respiratory symptoms were one of the most frequently reported adverse events associated with dupilumab and the other approved biologics from clinical trials and real-world experience. Prior studies indicate that the incidence of URTIs in dupilumab-treated groups is generally similar to that in placebo groups. It is associated with a reduced risk of serious infections and non-herpetic skin infections, although it may slightly increase the risk of herpesviral infections (12–15). However, Tralokinumab showed URTIs as the most frequent treatment-emergent adverse event. Still, the incidence was similar between the treatment and placebo groups, suggesting no significant increase in risk (16). At the same time, JAK Inhibitors (Upadacitinib and Abrocitinib) have shown a higher incidence of URTIs than other therapies, with upadacitinib having a notably higher risk (17). We aimed to provide pooled incidence estimates using meta-analysis for the incidence of any URTI with dupilumab and other new agents, namely, lebrikizumab, tralokinumab, abrocitinib, and upadacitinib compared to topical.
We systematically searched PubMed, MEDLINE, DOAJ, and ClinicalTrials.gov with no restrictions on language and the last date of searching data for studies assessing the incidence of any URTI with dupilumab and other new agents, namely, lebrikizumab, tralokinumab, abrocitinib, and upadacitinib in the treatment of AD in pediatric or adult populations. The search strategy was “[(Atopic dermatitis) OR (Atopic Eczema) AND (Dupilumab OR dupixent OR lebrikizumab OR Tralokinomab OR Upadacitinib OR Abrocitinib OR IL-4 inhibitors OR IL-13 inhibitors OR Janus Kinase 1 antagonist OR JAK1 inhibitor)].” A sample of the search strategy is detailed (Supplementary Table 1).
The inclusion criteria of this systematic review were randomized controlled trials (RCTs) of adults and pediatrics diagnosed with AD and treated with JAK1 selective inhibitors (Updacitinib, Abrocitinib), or anti-IL4/13 (Dupilumab), or anti-IL13 (Tralokizumab, Lebrikizumab), excluding studies of patients treated with topicals, non-randomized clinical trials, Studies on other dermatitis types and biologics other than Dupilumab (OR) Updacitinib (OR) Abrocitinib (OR) Tralokizumab (OR) Lebrikizumab.
Two reviewers independently conducted data extraction and methodology quality assessment for all included studies. We extracted the type of treatment, the sample size for the treatment group, the sample size for the control group, and the incidence of URTI in the treatment group and the control group. For all studies, the main measure of interest was the incidence of URTI adverse events following the administration of Updacitinib, Abrocitinib, Dupilumab, Tralokizumab, and Lebrikizumab.
Using the Cochrane Risk of Bias Tool (RoB 2), the studies were assessed across seven domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other potential sources of bias (Supplementary Table 2).
The meta-analysis was conducted using the Netmeta statistical package in R. We extracted events and sample sizes for binary outcomes and mean (SD) and sample sizes for continuous outcomes. Odds ratios for binary outcomes and Standardized mean differences for continuous outcomes were calculated. Before NMA, we explored assumptions of transitivity among the studies by using several criteria, including tests for within-designs and between-designs inconsistency. We used a random effects model with the restricted maximum-likelihood estimator for tau2 to account for between-study variability. Odd ratios and 95% confidence intervals (CIs) were calculated to assess the effect sizes. Heterogeneity was assessed using the I2 and Cochran's Q statistic and subgroup analyses were performed to evaluate differences across drugs.
We retrieved 413 records from various databases, including 109 from PubMed, 200 from MEDLINE & DOAJ, and 104 from ClinicalTrials.gov. After removing 98 duplicate records, a total of 315 records were screened based on their title and abstracts. Of these, 281 records were excluded due to irrelevance to the research focus. Subsequently, 34 full-text articles were assessed for eligibility. Among these, 13 articles were excluded as they did not address patient-reported concerns related to URTI. Ultimately, 21 studies were included in the qualitative synthesis (Figure 1).
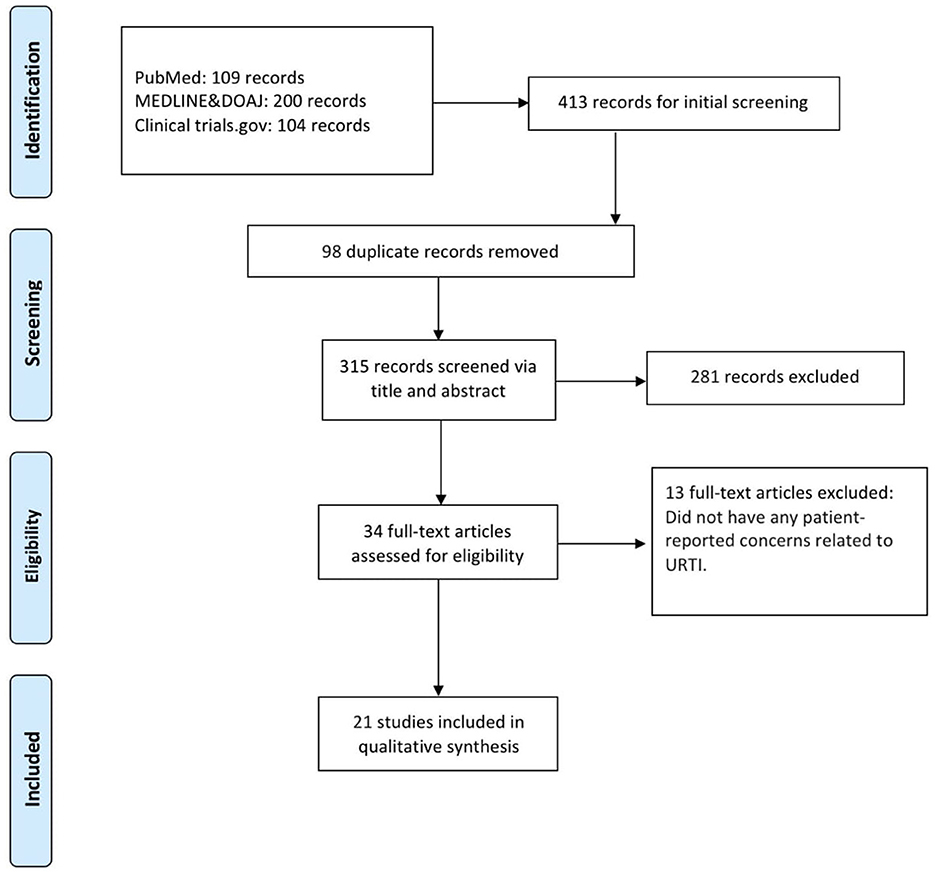
Figure 1. Study flowchart per the preferred reporting items for systematic reviews and meta-analyses (PRISMA) criteria.
Across 21 studies, the incidence of URTI in treatment groups varied widely, with rates as low as 0.35% (26) and as high as 41.5% (27). In control groups, the URTI incidence ranged from 0% (29) to 40% (27). In the study Wollenberg et al. (33), the treatment group had a higher URTI incidence than the control group (23.1% vs. 20.9%). Similarly, Simpson (18), the treatment group had a higher URTI rate than the control group (8.1% vs. 1.8%). Conversely, in studies like Simpson (30), the treatment group showed a slightly higher incidence (19.4%) than the control group (17.6%) (Supplementary Table 3).
Out of these 21 trials, 11 tested Dupilumab, 4 tested Tralokinumab, 3 tested Upadacitinib, 2 tested Lebrikizumab and one study tested Abrocitinib. Regarding the odds ratios for the incidence of URTI between control and intervention groups, in the Simpson (31) study, the odds ratio for Abrocitinib was 1.10 (95% CI: 0.403–3.004). Several studies using Dupilumab reported odds ratios ranging from 0.354 [95% CI: 0.163–0.772; (32)] to 4.774 [95% CI: 1.677–13.587; (18)]. For Lebrikizumab, odds ratios were as low as 0.164 [95% CI: 0.017–1.588; (16)] and up to 0.454 (95% CI: 0.015–13.708). Tralokinumab studies showed a range of odds ratios from 1.000 [95% CI: 0.344–2.903; (29)] to 2.429 [95% CI: 0.802–7.354; (33)]. Upadacitinib had odds ratios between 1.063 [95% CI: 0.260–4.350; (33)] and 1.628 [95% CI: 0.776–3.416; (33)]. There was minimal heterogeneity across the studies reporting this outcome (Q = 25.04, I2= 20.14%). Out of 5,053 intervention recipients, 490 (9.70%) reported URTI, compared to 182 (8.03%) control recipients. This translated to an MH Odds ratio of 1.18 (95% CI: 0.98–1.42) (Figure 2). There was no evidence of publication bias (p = 0.83) (Figure 3). There were no significant subgroup differences between patients taking different biological therapies (Q = 3.90, p = 0.42). The Risk of Bias assessment using ROB-2 for 19 studies revealed predominantly low risk across most domains, with a mix of low to high risk in the selection of the reported result and one study (28) showing high overall risk (Figure 4).

Figure 2. Forest plot presenting pooled effect size incidence of URTI adverse event with biological therapies in atopic dermatitis.

Figure 4. Risk of bias assessment for the included studies using the cochrane risk of bias tool (RoB 2).
This review aimed to explore the incidence of upper respiratory tract infection in patients with AD treated with different biological therapies. The overall incidence of URTI varied widely across the included studies, ranging from 0.35% to 41.5% among treatment groups compared to 0%−40% among controls. The biological agents used in those individual studies include Dupilumab, Tralokinumab, Upadacitinib, Lebrikizumab and Abrocitinib (19). There were no significant subgroup differences –regarding URTI incidence—between patients taking different biological therapies.
Dupilumab, an IgG4 monoclonal antibody, was the first biological agent described for treating AD. It improved pruritus symptoms and depression and anxiety symptoms. In addition to improving the overall quality of life (18–20).
The effectiveness of biological agents in the setting of AD was established in the literature; a review article demonstrated that many biological agents (Lebrikizumab, tralokinumab, fezakinumab, and nemolizumab) helped decrease disease severity. However, the safety profile of these agents needs to be established. Nevertheless, its use in pediatrics atopic dermatitis is to be investigated (21).
URTI was also reported in psoriasis patients treated with biological agents, as they are immunosuppressive (22). Other side effects of biological agents include conjunctivitis, especially with agents like dupilumab (19). Nemolizumab is currently under investigation. Trials at different stages showed good results in moderate and severe AD with good safety in the long term (10). However, the overall safety profile of these biological agents was said to be favorable, with side effects being mild to moderate (23).
Nevertheless, different biological agents used in the treatment of juvenile idiopathic arthritis were also associated with development of infections (URTI, pneumonia, and pleural effusion). In Rheumatoid arthritis patients treated with biological agents, higher rates of infections and hospitalization were reported especially with infliximab. However, apart from the biological agents which decrease the immunity, there are already patient's risk factors of infections like being immunocompromised, on steroid therapy and other factors that made them already susceptible to opportunistic infections (24, 25).
Updated studies must be included in the analysis, as more evidence might have emerged. Also, more subgroup analysis can be done to differentiate between the sole effect of biological agents vs. the baseline patient's characteristics and the difference in the definition of URTI between different studies.
The incidence of upper respiratory tract infections among AD patients treated with biological agents is quite variable among included studies across the literature despite the analysis showing low heterogeneity. Nevertheless, the incidence among controls in these RCTs is arguably similar. Biological agents have a bright future for AD patients with good safety profiles. However, more large-scale trials are needed to prove their efficacy and safety, as well as trials that investigate the use of biological agents in different populations, including pediatrics. However, trials should put more control on patients-specific URTI risk factors both in the research and in the clinical decision-making.
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author.
RAlr: Writing – original draft, Writing – review & editing. MK: Writing – original draft, Writing – review & editing. YA: Writing – original draft, Writing – review & editing. HM: Writing – original draft, Writing – review & editing. AA: Writing – original draft, Writing – review & editing. AK: Writing – original draft, Writing – review & editing. AS: Writing – original draft, Writing – review & editing. BT: Writing – original draft, Writing – review & editing. RAlt: Writing – original draft, Writing – review & editing. AJ: Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2025.1550640/full#supplementary-material
1. Silverberg JI, Gelfand JM, Margolis DJ, Boguniewicz M, Fonacier L, Grayson MH, et al. Patient burden and quality of life in atopic dermatitis in US adults. Ann Aller Asthma Immunol. (2018) 121:340–7. doi: 10.1016/j.anai.2018.07.006
2. Silverberg JI. Public health burden and epidemiology of atopic dermatitis. Dermatol Clin. (2017) 35:283–9. doi: 10.1016/j.det.2017.02.002
3. Yew YW, Thyssen JP, Silverberg JI, A. systematic review and meta-analysis of the regional and age-related differences in atopic dermatitis clinical characteristics. J Am Acad Dermatol. (2018) 80:390–401. doi: 10.1016/j.jaad.2018.09.035
4. Weidinger S, Novak N. Atopic dermatitis. Lancet. (2015) 387:1109–22. doi: 10.1016/S0140-6736(15)00149-X
5. Wong ITY, Tsuyuki RT, Cresswell-Melville A, Doiron P, Drucker AM. Guidelines for the management of atopic dermatitis (eczema) for pharmacists. Revue Des Pharmaciens Du Canada. (2017) 150:285–97. doi: 10.1177/1715163517710958
6. Drucker AM, Lam M, Prieto-Merino D, Malek R, Ellis AG, Yiu ZZN, et al. Systemic immunomodulatory treatments for atopic dermatitis. JAMA Dermatol. (2024) 160:936. doi: 10.1001/jamadermatol.2024.2192
7. Le M, Berman-Rosa M, Ghazawi FM, Bourcier M, Fiorillo L, Gooderham M, et al. Systematic Review on the Efficacy and Safety of oral janus kinase inhibitors for the treatment of atopic dermatitis. Front Med. (2021) 8:682547. doi: 10.3389/fmed.2021.682547
8. Snast I, Reiter O, Hodak E, Friedland R, Mimouni D, Leshem YA. Are biologics efficacious in atopic dermatitis? A systematic review and meta-analysis. Am J Clin Dermatol. (2017) 19:145–65. doi: 10.1007/s40257-017-0324-7
9. Puar N, Chovatiya R, Paller AS. New treatments in atopic dermatitis. Ann Aller Asthma Immunol. (2020) 126:21–31. doi: 10.1016/j.anai.2020.08.016
10. Zhou S, Qi F, Gong Y, Zhang J, Zhu B. Biological therapies for atopic dermatitis: a systematic review. Dermatology. (2021) 237:542–52. doi: 10.1159/000514535
11. Wu J, Guttman-Yassky E. Efficacy of biologics in atopic dermatitis. Expert Opin Biol Ther. (2020) 20:525–38. doi: 10.1080/14712598.2020.1722998
12. Eichenfield LF, Bieber T, Beck LA, Simpson EL, Thaçi D, De Bruin-Weller M, et al. Infections in dupilumab clinical trials in atopic dermatitis: a comprehensive pooled analysis. Am J Clin Dermatol. (2019) 20:443–56. doi: 10.1007/s40257-019-00445-7
13. Ou Z, Chen C, Chen A, Yang Y, Zhou W. Adverse events of Dupilumab in adults with moderate-to-severe atopic dermatitis: a meta-analysis. Int Immunopharmacol. (2017) 54:303–10. doi: 10.1016/j.intimp.2017.11.031
14. Paller AS, Siegfried EC, Sidbury R, Lockshin B, Cork M, Pinter A, et al. 437 Treatment-emergent adverse events in patients aged 6 months to 5 years with moderate-to-severe atopic dermatitis treated with dupilumab in an open-label extension clinical trial. Br J Dermatol. (2023) 188:ljad162-057. doi: 10.1093/bjd/ljad162.057
15. Paller AS, Beck LA, Blauvelt A, Siegfried EC, Cork MJ, Wollenberg A, et al. Infections in children and adolescents treated with dupilumab in pediatric clinical trials for atopic dermatitis—A pooled analysis of trial data. Pediatr Dermatol. (2022) 39:187–96. doi: 10.1111/pde.14909
16. Wollenberg A, Howell MD, Guttman-Yassky E, Silverberg JI, Kell C, Ranade K, et al. Treatment of atopic dermatitis with tralokinumab, an anti-IL-13 mAb. J Allergy Clin Immunol. (2019) 143:135–41. doi: 10.1016/j.jaci.2018.05.029
17. Zhang Y, Hong P, Rai S, Liu R, Liu B. Upadacitinib is a better choice than abrocitinib for patients with moderate-to-severe atopic dermatitis: an updated meta-analysis. J Clin Pharm Ther. (2023) 2023:1–11. doi: 10.1155/2023/9067797
18. Simpson EL, Bieber T, Guttman-Yassky E, Beck LA, Blauvelt A, Cork MJ, et al. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. (2016) 375:2335–48. doi: 10.1056/NEJMoa1610020
19. Jappe U, Beckert H, Bergmann KC, Gülsen A, Klimek L, Philipp S, et al. Biologics for atopic diseases: indication, side effect management, and new developments. Allergologie Select. (2021) 5:1–25. doi: 10.5414/ALX02197E
20. Blauvelt A, De Bruin-Weller M, Gooderham M, Cather JC, Weisman J, Pariser D, et al. Long-term management of moderate-to-severe atopic dermatitis with dupilumab and concomitant topical corticosteroids (LIBERTY AD CHRONOS): a 1-year, randomised, double-blinded, placebo-controlled, phase 3 trial. Lancet. (2017) 389:2287–303. doi: 10.1016/S0140-6736(17)31191-1
21. Uppal SK, Kearns DG, Chat VS, Han G, Wu JJ. Review and analysis of biologic therapies currently in phase II and phase III clinical trials for atopic dermatitis. J Dermatol Treat. (2020) 33:626–36. doi: 10.1080/09546634.2020.1775775
22. Wan MT, Shin DB, Winthrop KL, Gelfand JM. The risk of respiratory tract infections and symptoms in psoriasis patients treated with interleukin 17 pathway–inhibiting biologics: A meta-estimate of pivotal trials relevant to decision making during the COVID-19 pandemic. J Am Acad Dermatol. (2020) 83:677–9. doi: 10.1016/j.jaad.2020.05.035
23. Alvarenga JM, Bieber T, Torres T. Emerging biologic therapies for the treatment of atopic dermatitis. Drugs. (2024) 84:1379–94. doi: 10.1007/s40265-024-02095-4
24. Aygun D, Sahin S, Adrovic A, Barut K, Cokugras H. The frequency of infections in patients with juvenile idiopathic arthritis on biologic agents : 1-year prospective study. Clin Rheumatol. (2019) 38:1025–30. doi: 10.1007/s10067-018-4367-9
25. Curtis JR, Xie F, Chen L, Baddley JW, Beukelman T, Saag KG, et al. The comparative risk of serious infections among rheumatoid arthritis patients starting or switching biological agents. Ann Rheum Dis. (2011) 70:1401–6. doi: 10.1136/ard.2010.146365
26. Blauvelt A, Thyssen JP, Guttman-Yassky E, Bieber T, Serra-Baldrich E, Simpson E, et al. Efficacy and safety of lebrikizumab in moderate-to-severe atopic dermatitis: 52-week results of two randomized double-blinded placebo-controlled phase III trials. Br J Dermatol. (2023) 188:740–8. doi: 10.1093/bjd/ljad022
27. Guttman-Yassky E, Thaçi D, Pangan AL, Hong HC, Papp KA, Reich K, et al. Upadacitinib in adults with moderate to severe atopic dermatitis: 16-week results from a randomized, placebo-controlled trial. J Am Acad Dermatol. (2020) 82:1–10. doi: 10.1016/j.jaci.2019.11.025
28. Guttman-Yassky E, Bissonnette R, Ungar B, Suarez-Fariñas M, Ardeleanu M, Esaki H, et al. Dupilumab progressively improves systemic and cutaneous abnormalities in patients with atopic dermatitis. J Allergy Clin Immunol. (2019) 143:155–72. doi: 10.1016/j.jaci.2018.08.022
29. Guttman-Yassky E, Blauvelt A, Eichenfield LF, Paller AS, Armstrong AW, Drew J, et al. Efficacy and safety of lebrikizumab, a high-affinity interleukin 13 inhibitor, in adults with moderate to severe atopic dermatitis: a phase 2b randomized clinical trial. JAMA Dermatol. (2020) 156:411–20. doi: 10.1001/jamadermatol.2020.0079
30. Simpson EL, Paller AS, Siegfried EC, Boguniewicz M, Sher L, Gooderham MJ, et al. Efficacy and safety of dupilumab in adolescents with uncontrolled moderate to severe atopic dermatitis: a phase 3 randomized clinical trial. JAMA Dermatol. (2020) 156:44–56. doi: 10.1001/jamadermatol.2019.3336
31. Simpson EL, Sinclair R, Forman S, Wollenberg A, Aschoff R, Cork M, et al. Efficacy and safety of abrocitinib in adults and adolescents with moderate-to-severe atopic dermatitis (JADE MONO-1): a multicentre, double-blind, randomised, placebo-controlled, phase 3 trial. Lancet. (2020) 396:255–66. doi: 10.1016/S0140-6736(20)30732-7
32. Thaçi D, Simpson EL, Beck LA, Bieber T, Blauvelt A, Papp K, et al. Efficacy and safety of dupilumab in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical treatments: a randomised, placebo-controlled, dose-ranging phase 2b trial. Lancet. (2016) 387:40–52. doi: 10.1016/S0140-6736(15)00388-8
33. Wollenberg A, Blauvelt A, Guttman-Yassky E, Worm M, Lynde C, Lacour J-P, et al. Tralokinumab for moderate-to-severe atopic dermatitis: results from two 52-week, randomized, double-blind, multicentre, placebo-controlled phase III trials (ECZTRA 1 and ECZTRA 2). J Am Acad Dermatol. (2021) 84:70–9. doi: 10.1111/bjd.19574
Keywords: atopic dermatitis, biologics, dupilumab, IL4/13 inhibitor, tralokinumab, lebrikizumab, IL13 inhibitors, abrocitinib
Citation: Alraddadi R, Kalantan M, Aljefri Y, Maaddawi H, Alsamadani A, Kadasa A, Softah A, Tabbakh B, Alturkistani R and Jfri A (2025) Incidence of upper respiratory tract infections with biological therapies in moderate to severe atopic dermatitis: a systematic review and meta-analysis. Front. Med. 12:1550640. doi: 10.3389/fmed.2025.1550640
Received: 23 December 2024; Accepted: 17 March 2025;
Published: 02 April 2025.
Edited by:
Joel Correa Da Rosa, Icahn School of Medicine at Mount Sinai, United StatesReviewed by:
Robert Stewart Dawe, NHS Tayside, United KingdomCopyright © 2025 Alraddadi, Kalantan, Aljefri, Maaddawi, Alsamadani, Kadasa, Softah, Tabbakh, Alturkistani and Jfri. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Abdulhadi Jfri, QWJkdWxoYWRpamZyaUBnbWFpbC5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.