
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Med. , 10 March 2025
Sec. Obstetrics and Gynecology
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1529470
 Tzyy-Guey Tseng1†
Tzyy-Guey Tseng1† Che-Yen Kuan2†
Che-Yen Kuan2† Yun-Ping Lo3
Yun-Ping Lo3 Yun-Shiuan Chuang1,4,5
Yun-Shiuan Chuang1,4,5 Chun-Ying Lee1,4
Chun-Ying Lee1,4 Yi-Ting Lin1,4,6
Yi-Ting Lin1,4,6 Ing-Luen Shyu2,7,8
Ing-Luen Shyu2,7,8 Ming-Tsang Wu1,4,9*
Ming-Tsang Wu1,4,9* Chi-Jung Tai1,10,11,12*
Chi-Jung Tai1,10,11,12*Background: The mechanism of primary dysmenorrhea remains unraveled. Body symptoms not related to menstrual cycle may indicate the potential mechanism of primary dysmenorrhea, albeit the association has not been proven. Furthermore, we hypothesize that the cumulative burden of these symptoms may influence the incidence of primary dysmenorrhea. Therefore, we aim to design a study to identify bodily symptoms potentially related to primary dysmenorrhea and test the hypothesis in understanding and managing primary dysmenorrhea.
Methods: A total of 3,140 female participants aged 30–50 years were enrolled from the Taiwan Biobank. Stepwise logistic regression was used to select potential body symptoms associated with primary dysmenorrhea from a training dataset. Selected body symptoms were validated in a test dataset. Female participants without dysmenorrhea in the baseline survey were divided into two groups (with and without body symptoms) in a baseline survey. Cox regression and Kaplan-Meier survival analyses were used to evaluate the risk of incident dysmenorrhea.
Results: Women with body symptoms such as cold extremities (adjusted odds ratio [AdjOR], 1.53, 95% confidence interval [CI], 1.12–2.21), dull abdominal pain (AdjOR, 1.45, 95% CI, 1.03–2.04), and edema (AdjOR, 1.43, 95% CI, 1.02–1.99) were significantly associated with dysmenorrhea. Women with the three body symptoms had a significantly higher risk of dysmenorrhea (adjusted hazard ratio, 2.74, 95%CI, 1.18–6.31; log-rank test, p = 0.0017) than those without body symptoms. Trend analysis showed that the risk of dysmenorrhea increased with the number of body symptoms (p-trend = 0.025).
Conclusion: This study identified cold extremities, dull abdominal pain, and edema as predictors of primary dysmenorrhea, with their accumulation associated with a higher risk of developing dysmenorrhea. We propose that further research explore pharmacological and non-pharmacological interventions targeting these symptoms, as they may provide long-term benefits in the management of primary dysmenorrhea.
Dysmenorrhea is characterized by a painful sensation in the lower abdomen that occurs before and/or during menstruation. Affecting 16%−91% of women during their reproductive age (1), this prevalent gynecological condition poses a significant burden on individuals, healthcare systems, and society at large (2). Notably, dysmenorrhea-related absenteeism and presenteeism among women of reproductive age lead to reduced productivity in both educational and workplace settings, resulting in substantial economic losses. In the United States alone, these losses are estimated at $2 billion annually (3). Consequently, effective management of dysmenorrhea is crucial to mitigating its wide-ranging impacts.
Dysmenorrhea is classified into two types: primary dysmenorrhea, which occurs without an underlying gynecological condition, while secondary dysmenorrhea generally has underlying gynecological disease, such as endometriosis, adenomyosis, or uterine myoma (4). Unlike secondary dysmenorrhea, the pathophysiology of primary dysmenorrhea remains unraveled in modern times. The most widely recognized cause of primary dysmenorrhea is the excessive production of uterine leukotrienes and prostaglandins via the cyclooxygenase (COX) pathway, leading to myometrial contraction and ischemia (5). Despite this knowledge, current treatments targeting prostaglandin and its derivatives are still unsatisfactory, not only because of their possible side effects and allergic reactions. For example, although nonsteroidal anti-inflammatory drugs and hormone therapy are effective in alleviating symptoms for many women (6), they cannot offer a cure or prevent symptom recurrence. Other treatment options aimed at inducing myometrial relaxation, such as glyceryl trinitrate (7), magnesium, and calcium channel blockers remain controversial due to inconsistent clinical evidence (8). Therefore, a variety of nonmedical remedies have been proposed to treat dysmenorrhea. However, the efficacy of herbal remedies (e.g., ginger, rose tea, and sweet fennel seed extract) and dietary supplements (e.g., vitamin E, vitamin B1, and fish oil) for primary dysmenorrhea is supported by limited clinical data (9). This suggests the potential involvement of alternative etiological factors in primary dysmenorrhea.
Mechanoreceptors sensitization is another generally recognized pathophysiological mechanism underlying primary dysmenorrhea (6). To address this, transcutaneous electrical nerve stimulation (TENs) has been employed as a treatment modality, functioning by gating ascending pain signals at the spinal cord level (10, 11). A Cochrane review suggests that both high-frequency and low-frequency TENS can effectively alleviate dysmenorrhea symptoms compared to placebo or no treatment (12). Similarly, acupuncture and acupressure have demonstrated comparable efficacy in providing pain relief for patients with dysmenorrhea (13). Manual therapy and pelvic floor exercises have also been shown to be effective in reducing pain (14).
Early-stage pathophysiological mechanisms may not directly cause primary dysmenorrhea but could lead to mild body symptoms in advance. According to traditional Chinese medicine (TCM) theory, such symptoms may be linked to obstetric and gynecological diseases in women (15). From a TCM perspective, dysmenorrhea is associated with cold accumulation, blood stasis, and Qi stagnation (16), which contribute to composite bodily symptoms and the development of what is termed a TCM body constitution (17). These specific bodily symptoms may serve as risk factors or early indicators of primary dysmenorrhea. To the best of our knowledge, only one previous study reported that lower abdominal pain, which was not related to menstrual periods, was associated with dysmenorrhea with an odds ratio (OR) of 1.80 [95% confidence interval (CI), 1.3–2.3] (18). Identifying bodily symptoms potentially related to primary dysmenorrhea could provide valuable insights for its management. Furthermore, we hypothesize that the cumulative burden of these symptoms may influence the incidence of primary dysmenorrhea. Therefore, we aim to design a study to test this hypothesis and explore the relevance of TCM theory in understanding and managing primary dysmenorrhea.
The Taiwan Biobank has recruited 139,842 community-based volunteers and 6,834 hospital-based volunteers since 2012 (19). The Taiwan Biobank obtained informed consent from all participants or their legal guardians for research use of the collected data and samples. The current study was approved by the Institutional Review Board-II of Kaohsiung Medical University Chung-Ho Memorial Hospital [approval no. KMUHIRB-E(II)-20230266] in 2023 and all methods were performed in accordance with relevant guidelines and regulations. We adhered to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) checklist to ensure comprehensive and transparent reporting of the study findings (20).
In this study, we used a subset of the Taiwan Biobank data by random sampling from 104,451 community-based participants recruited between 2012–2018 (21). The data included basic demographic information, personal healthy behaviors, personal medical history, and body symptoms. Details and regulations of the Taiwan Biobank are described elsewhere (22).
The unique point of the Taiwan Biobank for women study is that the questionnaire included many questions about associated body symptoms, which were not related to menstrual periods. In the Taiwan Biobank, body symptoms included cold extremities, fatigue, oral ulcers, dull abdominal pain, shortness of breath, chest tightness, tinnitus, ecchymosis, dry skin, varicose veins, soreness (over the waist, knee, or heels), muscle spasm, edema (over the face, eyebrow, or extremities), and hot flushes. However, only a few female participants answered the questions regarding body symptoms. Therefore, 4,490 female participants with records of body symptoms in the Taiwan biobank were selected for further analysis (Figure 1).
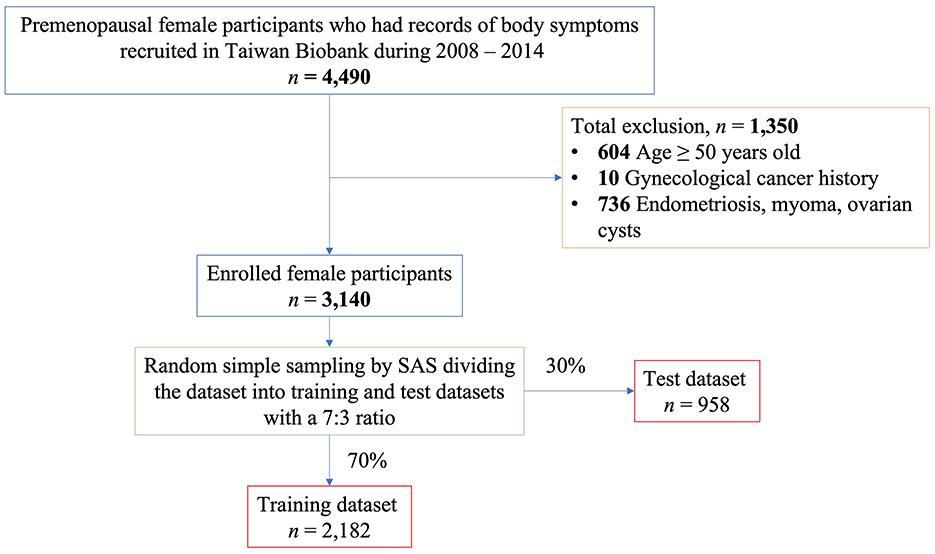
Figure 1. The enrollment process of target female participants and the strategy for selecting training dataset and test dataset.
Targeting at the primary dysmenorrhea, selected female participants went through the exclusion process (Figure 1). First, 604 participants aged ≥50 years were excluded. Second, 10 participants were excluded because of a history of gynecological cancer. Finally, 736 participants with a history of endometriosis, myoma, or ovarian cyst were excluded. After these exclusion, 3,140 female participants were selected using random simple sampling with a 7:3 ratio using SAS (Statistics Analysis System Institute Inc., Cary, NC, USA) software and divided into training (n = 2,182) and test (n = 958) datasets (Figure 1).
Logistic regression analyses were conducted to identify predictors of primary dysmenorrhea. First, stepwise logistic regression was performed on the training dataset to select the primary body symptoms associated with primary dysmenorrhea. Non-significant body symptoms identified during this process were excluded from further analysis. Finally, the selected body symptoms were validated using multivariable logistic regression on the test dataset.
To further investigate whether the selected body symptoms led to dysmenorrhea, we designed a retrospective cohort study. Initially, 2,491 female participants without dysmenorrhea in the first wave of the survey were selected. Next, 1,048 female participants were excluded, owing to a lack of follow-up surveys. The participants were then divided into two groups: with and without selected body symptoms (Figure 2). Moreover, we stratified participants in the group with body symptoms into strata, with different numbers of body symptoms in the first wave of the survey.
Although direct causation of dysmenorrhea has not been definitively established, previous studies have proposed a conceptual framework that highlights associations between dysmenorrhea and its determinants including lifestyle, reproductive, psychological, and other health-related factors (1, 18). In this study, we adopted this framework to select covariates, which were treated as potential confounding factors in the cohort analysis.
In this study, lifestyle factors included exercise habits, smoking, tea, coffee, and alcohol consumption. Previous studies have shown inconsistent evidence regarding the association between dysmenorrhea and cigarette smoking (5, 23), while alcohol consumption was not associated with dysmenorrhea (24). Unexpectedly, a systematic review and meta-analysis showed that exercise reduced pain intensity in patients with primary dysmenorrhea (25).
Regarding reproductive factors, this study included parity, irregular menstrual cycle, and age at menarche. The association between parity and dysmenorrhea was consistent across previous studies, with reported ORs ranging from 0.2 to 0.74 (1, 5, 24, 26). Irregular menstrual cycle was associated with an increased risk of dysmenorrhea (27). However, the evidence regarding age at menarche as a predictor of dysmenorrhea remains inconclusive. Hu et al. found that an age at menarche ≤ 12 years was a risk factor for dysmenorrhea (OR, 1.16, 95% CI, 1.01–1.33) among Chinese Female University students (28). In contrast, other studies did not observe a higher risk of dysmenorrhea in women with an age at menarche ≤ 12 years (29, 30).
Psychological factors, such as increased levels of stress or depressive mood, were associated with an increased risk of dysmenorrhea (28, 31). This may explain the higher prevalence of dysmenorrhea among adolescent women and those working in healthcare and military services (32, 33). For comorbidities, this study considered personal history of major diseases, including hypertension, asthma, peptic ulcer, diabetes, and low back pain.
For other health factors, body mass index (BMI) was calculated as a person's weight in kilograms divided by the square of their height in meters (kg/m2). BMI was categorized as underweight (BMI < 18.5), normal weight (18.5 ≤ BMI < 24.0), overweight (24.0 ≤ BMI < 27.0), or obese (BMI ≥ 27.0), according to the BMI category defined by the Health Promotion Administration, Taiwan (34).
Propensity score (PS) was calculated using the aforementioned covariates. We then generated a 1:1 PS-matched cohort for sensitivity analysis (35). We used Greedy nearest neighbor matching without replacement. We specified the caliper width of 0.25, which indicated that the difference in PS between the treated unit and its matching control unit must be ≤ 0.25. PS matching were conducted using the PSMATCH procedure provided by SAS. Standardized mean differences were calculated to compare the distribution of baseline covariates between groups with and without body symptoms after PS matching (36). A previous study suggested that a standardized mean difference above 0.1 denoted a meaningful imbalance in the baseline covariates.
Descriptive statistics were used to assess patient demographics, number and percentage for categorical variables, and the mean ± standard deviation for continuous variables. Cox regression analysis was performed to estimate the adjusted hazard ratios (adjHRs) and 95% CIs for dysmenorrhea. A trend analysis was also performed to evaluate the accumulating effect of body symptoms on primary dysmenorrhea. We adjusted these models to account for the confounding factors mentioned above. Statistical analyses were conducted using the SAS version 9.4. Using R (version 4.2.0, R Foundation for Statistical Computing, Vienna, Austria) packages (survival, survminer, and dplyr), the Kaplan-Meier survival analysis and log-rank test were used for time-to-event analysis. A P-value of < 0.05 was considered statistically significant.
In general, women with primary dysmenorrhea exhibited a higher prevalence of associated body symptoms (unrelated to menstrual periods), except for varicose veins, compared to those without dysmenorrhea (Table 1). The most prevalent body symptoms in the dysmenorrhea group were fatigue (70.4%), cold extremities (58.4%), and dizziness (58.4%).

Table 1. The body symptoms which were not related to menstrual cycles in participants with or without dysmenorrhea (N = 3,140).
Using the training dataset, stepwise logistic regression identified cold extremities (AdjOR, 1.38, 95% CI, 1.11–1.71), fatigue (AdjOR, 1.56, 95% CI, 1.22–2.00), dull abdominal pain (AdjOR, 1.61, 95% CI, 1.29–2.02), and edema (AdjOR, 1.56, 95% CI, 1.25–1.96) as major predictors of dysmenorrhea among all associated body symptoms (Table 2). The findings were further validated using multivariable logistic regression on the test dataset, which revealed that only cold extremities (AdjOR, 1.53, 95%CI, 1.12–2.11), dull abdominal pain (AdjOR, 1.45, 95%CI, 1.03–2.04), and edema (AdjOR, 1.45, 95%CI, 1.02–1.99) were significantly associated with an increased risk of dysmenorrhea (Table 2). In the subsequent cohort study, we further investigated whether these selected body symptoms contribute to the incidence of dysmenorrhea.
Participants without dysmenorrhea (n = 1,443) were divided into two groups: 970 (67.2%) participants had at least one of the three selected body symptoms (cold extremities, dull abdominal pain, and edema), while 473 (32.7%) did not suffer from any of these body symptoms (Figure 2). The mean follow-up time was 4.1 ± 1.0 years. Among the group with body symptoms, cold extremities were reported in 68.0% of participants, dull abdominal pain in 44.3%, and edema in 41.4% (Table 3). Over half of the patients (56.4%) reported one body symptom, 33.4% reported two symptoms, and 10.2% reported all three symptoms.
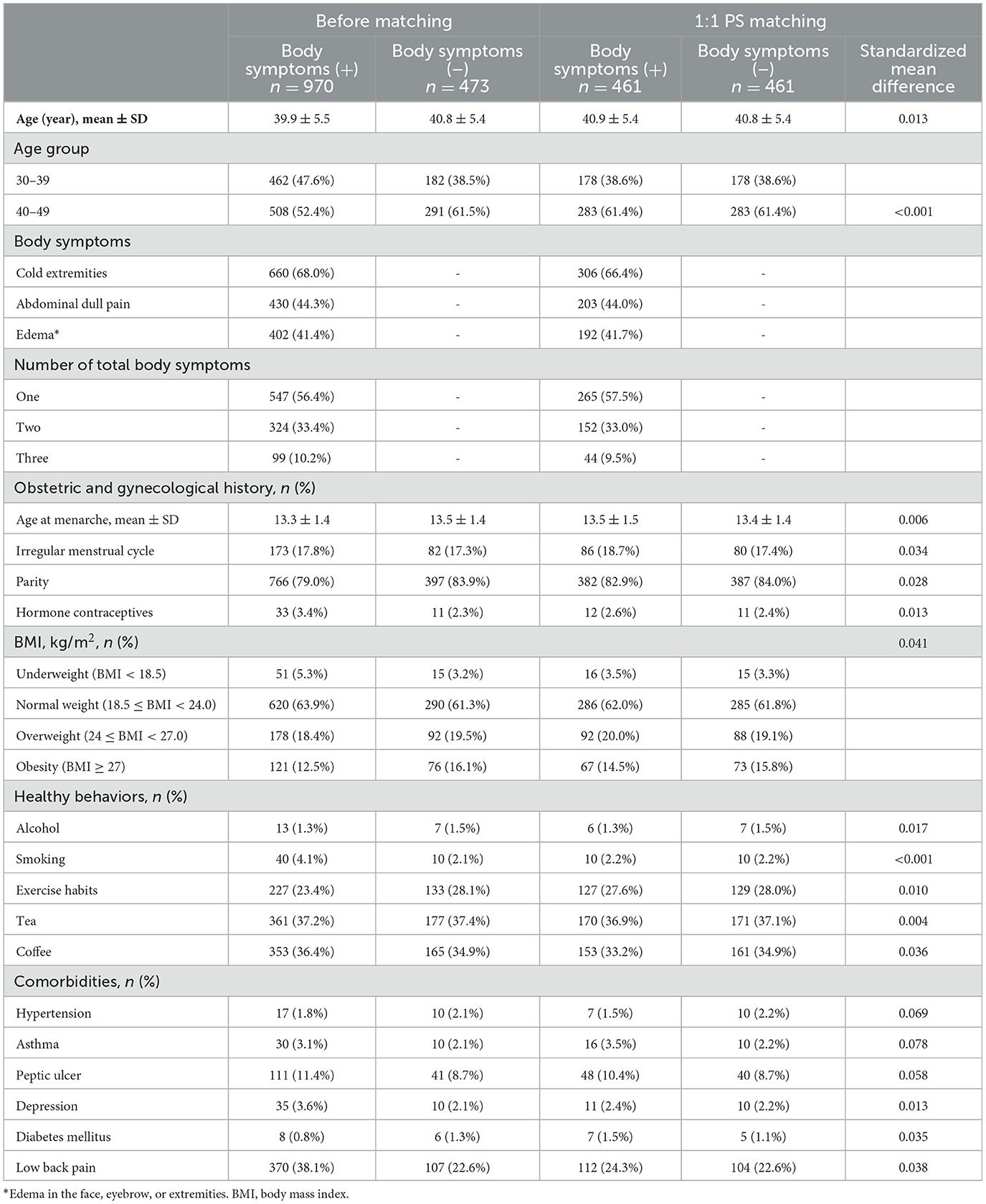
Table 3. Baseline characteristics of target female participants before and after 1:1 propensity-score matching.
The mean age of participants with and without body symptoms was 39.9 (±5.5) and 40.8 (±5.4) years, respectively. In terms of obstetric and gynecological history, both groups had similar ages at menarche (13.3 ± 1.4 vs. 13.5 ± 1.4 years) and comparable prevalence of irregular menstrual cycles (17.8% vs. 17.3%) (Table 3). However, parity was higher among participants without body symptoms (83.9% vs. 79.0%).
Compared to those with body symptoms, a higher proportion of participants without body symptoms had exercise habits. Additionally, participants with body symptoms had a higher prevalence of peptic ulcers (11.4% vs. 8.7%) and low back pain (38.1% vs. 22.6%) (Table 1).
The results showed that the participants with all three body symptoms had a significantly higher risk of incident dysmenorrhea (AdjHR, 2.74, 95% CI, 1.18–6.31) compared to those without any body symptoms (Table 4). While the risk was not statistically significant for participants with one or two symptoms, trend analysis indicated a dose-response relationship, with the risk of dysmenorrhea increasing as the number of body symptoms increased (p trend, 0.025).
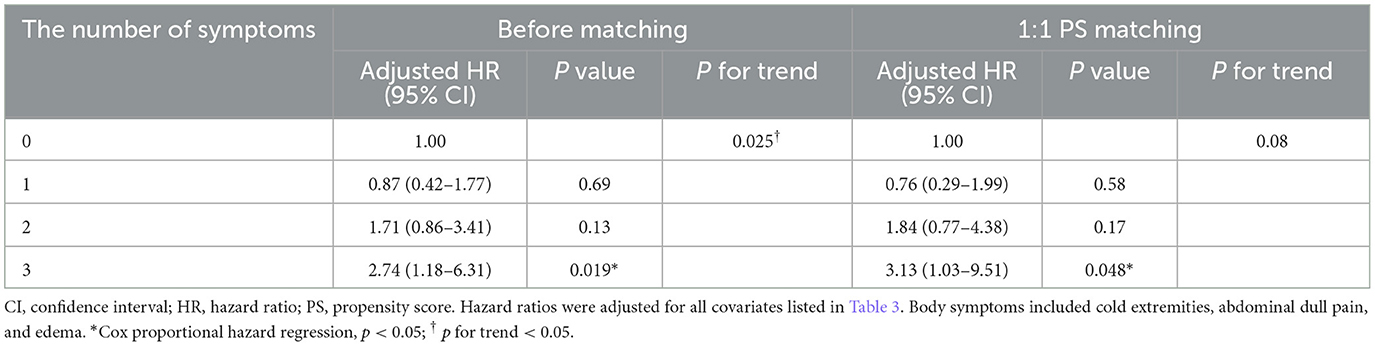
Table 4. Estimates of cox regression investigating associations between the number of body symptoms and dysmenorrhea before and after 1:1 propensity-score matching.
The Kaplan-Meier survival curve demonstrated that the group with three body symptoms had a higher cumulative incidence of dysmenorrhea after two patient-years (Figure 3A). Additionally, the incidence of dysmenorrhea increased among participants with two body symptoms after approximately 3.4 patient-years. The p-value for the log-rank test was 0.0017, further supporting these findings.
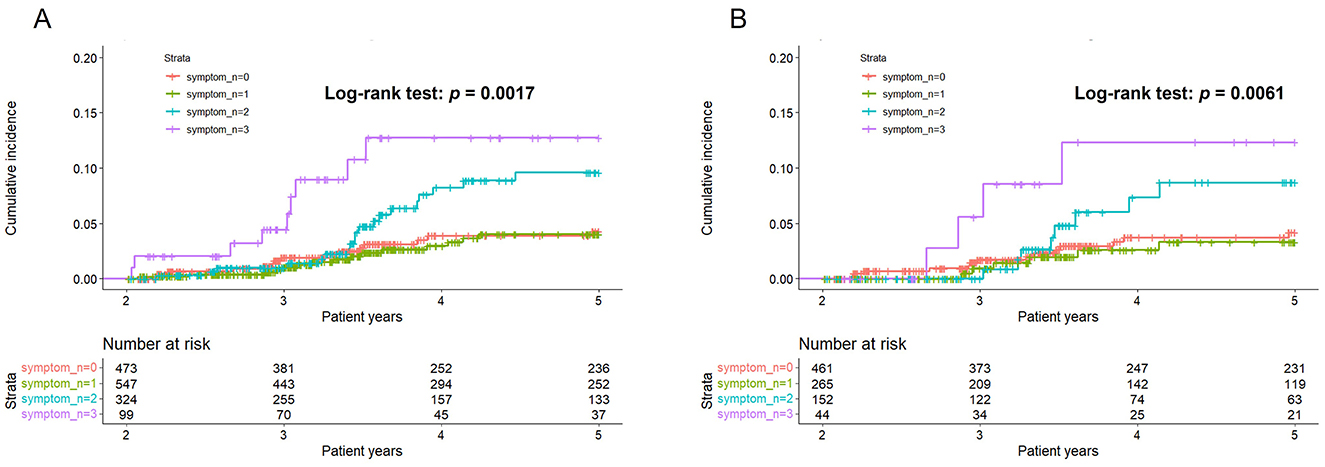
Figure 3. Kaplan-Meier curves of numbers of body symptoms in predicting dysmenorrhea. (A) Original cohort. (B) Post 1:1 propensity-score matching.
After the 1:1 PS matching, 461 participants were included in each group (Table 3). The standard mean difference of all covariates was below 0.1, indicating a well-balanced distribution between the two groups post-matching. Cox regression analysis consistently demonstrated that participants with all three body symptoms had a significant higher risk of incident dysmenorrhea (AdjHR, 3.13, 95% CI, 1.03–9.51) (Table 3). This finding was further supported by Kaplan-Meier survival analysis (log-rank test, p = 0.0061) (Figure 3B). Although the risk of dysmenorrhea increased with the number of body symptoms in the PS-matched cohort, the p-value for trend did not reach statistical significance.
Unlike previous studies that primarily focused on adolescents and young women, this study evaluated risk factors for dysmenorrhea in young and middle-aged women. The prevalence of dysmenorrhea in this study was 20.7%, which, while relatively low, was higher than rates reported in studies from Japan (15.8%) (37), Hungary (15.5%) (29), and the United Kingdom (15%) (38). In comparison to a cross-sectional study conducted among adult and middle-aged women in Brazil, the parity rates were similar (80.6% vs. 82.0%) (39). However, the prevalence of irregular menstrual cycles was lower in our study (17.7% vs. 25.5%).
We designed a cross-sectional study and identified three body symptoms predictive of dysmenorrhea in young and middle-aged women: cold extremities (AdjOR, 1.53), dull abdominal pain (AdjOR, 1.45), and edema in the face, eyebrow, or extremities (AdjOR, 1.43) (Table 2). Interestingly, these findings align closely with TCM theory. Cold extremities are indicative of cold accumulation in the body, dull abdominal pain may arise from Qi stagnation in the abdomen, and edema may correlate with blood stasis according to TCM principles.
In Western medicine, it is well known that the presence of multiple abnormal blood test results increases an individual's susceptibility to cardiovascular diseases (40). For example, someone with multiple morbidities, such as impaired fasting glucose, abnormal lipid profiles, and chronic kidney disease, is at a higher risk of ischemic heart disease compared to those with only one abnormality. Through this study, we demonstrated that the accumulation of three specific body symptoms—cold extremities, dull abdominal pain, and edema)—was significantly associated with a higher risk of incident dysmenorrhea, with an AdjHR of 2.74 (Table 4). This finding was further supported by sensitivity analysis using the 1:1 PS-matched cohort.
However, the concept of accumulating specific body symptoms as predictors of diseases is not widely recognized in Western medicine, though it is a common perspective in TCM. Using real-world data, this study is the first to support this perspective in the context of primary dysmenorrhea. Our findings suggest that the combination of three body symptoms may reflect a specific body condition or represent a TCM body constitution, which predisposes female to primary dysmenorrhea. Therefore, this study provides new insights into the potential mechanisms and approaches underlying primary dysmenorrhea.
The first strength of this study lies in the unique opportunity provided by the Taiwan Biobank to evaluate the association between body symptoms and dysmenorrhea. Additionally, this study utilized a larger cohort than previous research, comprising over 3,000 participants with detailed records of body symptoms. We also conducted a robust cross-sectional study, using a training dataset to identify associated body symptoms and a test dataset to validate these findings. Furthermore, we demonstrated the association between body symptoms and primary dysmenorrhea through a cohort study.
Moreover, Weissman et al. reported that a 1-year increase in age was associated with an 8% decrease in the risk of developing dysmenorrhea (5). As a result, our findings may represent an underestimation, as the risk associated with accumulated body symptoms could be attenuated by aging.
Although our study generated important findings, these results should be interpreted with caution. First, similar to other observational studies, the current study may be subject to residual confounding factors and cannot establish causality. Second, certain clinical data, such as the severity, duration, and frequency of dysmenorrhea, were not available from the Taiwan Biobank. Additionally, medical records between two waves of survey were absent, preventing us from accounting for the influence of time-dependent covariates. In terms of generalizability, the findings of this study are applicable only to young and middle-aged women and cannot be extended to adolescents. Furthermore, the cohort results were limited to an Asian population and may not apply to Caucasian, Latin, or Black populations.
This study proposes a novel approach to dysmenorrhea management by addressing specific body symptoms—such as cold extremities, abdominal dull pain, and edema—rather than solely focusing on pain relief during dysmenorrhea attacks.
From a TCM perspective, cold extremities indicate cold accumulation in the body, while edema may correlate with blood stasis. These symptoms, reflecting poor circulation and overall health, align with Western medicine's understanding of systemic circulatory dysfunction. Notably, peripheral circulation has been linked to conditions like erectile dysfunction in men (41), suggesting a potential connection between peripheral circulation and the circulatory function of internal genital organs in women. Consequently, vasodilators such as Sidenafil citrate (42), a selective phosphodiesterase type-5 inhibitor known to improve genital circulation in men, could be a promising candidate for dysmenorrhea treatment. From another perspective, diuretics, which reduce overall body water volume and alleviate edema, may also decrease the water content within the uterus, potentially relieving dysmenorrhea. However, these hypotheses necessitate further basic research and clinical studies to validate their efficacy.
On the other hand, improving peripheral and general circulation may enhance uterine circulation, potentially preventing, or alleviating primary dysmenorrhea. Previous studies have demonstrated that localized heat application to the suprapubic region provides pain relief and improves quality of life for dysmenorrhea patients (43). Ancient TCM literature highlights the benefits of hot springs for various gynecological conditions, including dysmenorrhea. Similarly, Yang et al. have shown that spa therapy can alleviate general health issues (44). However, as these treatments were studied over relatively short durations, we recommend further research through cohort studies to evaluate their long-term effects on dysmenorrhea prevention and recurrence.
We believe that enhancing general health status and modifying TCM body constitution can contribute positively to dysmenorrhea management. Although previous studies have shown limited evidence for the effectiveness of exercise and yoga in treating dysmenorrhea (45, 46), high-quality cohort studies are needed to clarify their role. In contrast, local manual therapy and pelvic floor exercises have demonstrated beneficial effects on pain relief and reducing pain sensitivity (14). These insights collectively underscore the potential of non-pharmacological approaches in dysmenorrhea management.
This study identified cold extremities, dull abdominal pain, and edema as predictors of primary dysmenorrhea. The accumulation of these symptoms was associated with an increased risk of incident dysmenorrhea. Both pharmacological and non-pharmacological approaches aimed at alleviating these symptoms may offer long-term benefits in managing primary dysmenorrhea. We believe these findings can pave the way for innovative treatments and help reduce the socio-economic burden associated with primary dysmenorrhea.
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding authors.
The studies involving humans were approved by the Institutional Review Board-II of Kaohsiung Medical University Chung-Ho Memorial Hospital. The studies were conducted in accordance with the local legislation and institutional requirements. The Ethics Committee/institutional review board waived the requirement of written informed consent for participation from the participants or the participants' legal guardians/next of kin because this study is a database research which meets the criteria for a waiving of written informed consent.
T-GT: Conceptualization, Project administration, Writing – original draft. C-YK: Conceptualization, Writing – original draft. Y-PL: Conceptualization, Writing – review & editing. Y-SC: Data curation, Writing – review & editing. C-YL: Methodology, Writing – review & editing. Y-TL: Methodology, Software, Writing – review & editing. I-LS: Writing – review & editing. M-TW: Funding acquisition, Writing – review & editing. C-JT: Conceptualization, Formal analysis, Supervision, Writing – original draft.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was partially supported by the Research Center for Precision Environmental Medicine, Kaohsiung Medical University, Kaohsiung, Taiwan from the Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan and by Kaohsiung Medical University Research Center Grant (KMU-TC112A01) awarded to M-TW. The sponsor has no role in the design, methods, subject recruitment, data collections, analysis and preparation of paper.
We thank our colleagues from the Division of Medical Statistics and Bioinformatics, Department of Medical Research, Kaohsiung Medical University Hospital, the Research Center for Precision Environmental Medicine, and the Center for Big Data Research, Kaohsiung Medical University, Kaohsiung, Taiwan for providing support in the statistical analysis.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
BMI, body mass index; CI, confidence interval; COX, cyclooxygenase; HR, hazard ratio; OR, odds ratio; PS, propensity score; STROBE, Strengthening the Reporting of Observational Studies in Epidemiology; TCM, traditional Chinese medicine; TENs, transcutaneous electrical nerve stimulation.
1. Ju H, Jones M, Mishra G. The prevalence and risk factors of dysmenorrhea. Epidemiol Rev. (2014) 36:104–13. doi: 10.1093/epirev/mxt009
2. Ballagh SA, Heyl A. Communicating with women about menstrual cycle symptoms. J Reprod Med. (2008) 53:837–46.
3. Sharghi M, Mansurkhani SM, Larky DA, Kooti W, Niksefat M, Firoozbakht M, et al. An update and systematic review on the treatment of primary dysmenorrhea. JBRA Assist Reprod. (2019) 23:51–7. doi: 10.5935/1518-0557.20180083
4. Proctor M, Farquhar C. Diagnosis and management of dysmenorrhoea. BMJ. (2006) 332:1134–8. doi: 10.1136/bmj.332.7550.1134
5. Weissman AM, Hartz AJ, Hansen MD, Johnson SR. The natural history of primary dysmenorrhoea: a longitudinal study. Bjog. (2004) 111:345–52. doi: 10.1111/j.1471-0528.2004.00090.x
6. Ferries-Rowe E, Corey E, Archer JS. Primary dysmenorrhea: diagnosis and therapy. Obstet Gynecol. (2020) 136:1047–58. doi: 10.1097/AOG.0000000000004096
7. Morgan PJ, Kung R, Tarshis J. Nitroglycerin as a uterine relaxant: a systematic review. J Obstet Gynaecol Can. (2002) 24:403–9. doi: 10.1016/S1701-2163(16)30403-0
8. Andersson KE, Ulmsten U. Effects of nifedipine on myometrial activity and lower abdominal pain in women with primary dysmenorrhoea. Br J Obstet Gynaecol. (1978) 85:142–8. doi: 10.1111/j.1471-0528.1978.tb10469.x
9. Proctor ML, Murphy PA. Herbal and dietary therapies for primary and secondary dysmenorrhoea. Cochrane Database Syst Rev. (2001) 2001:Cd002124. doi: 10.1002/14651858.CD002124
10. Mayer DJ, Hayes RL. Stimulation-produced analgesia: development of tolerance and cross-tolerance to morphine. Science. (1975) 188:941–3. doi: 10.1126/science.1094537
11. Melzack R, Wall PD. Pain mechanisms: a new theory. Science. (1965) 150:971–9. doi: 10.1126/science.150.3699.971
12. Han S, Park KS, Lee H, Kim E, Zhu X, Lee JM, et al. Transcutaneous electrical nerve stimulation (TENS) for pain control in women with primary dysmenorrhoea. Cochrane Database Syst Rev. (2024) 7:Cd013331. doi: 10.1002/14651858.CD013331.pub2
13. Liu T, Yu JN, Cao BY, Peng YY, Chen YP, Zhang L. Acupuncture for primary dysmenorrhea: a meta-analysis of randomized controlled trials. Altern Ther Health Med. (2017) 23:46–53.
14. Deodato M, Grosso G, Drago A, Martini M, Dudine E, Murena L, et al. Efficacy of manual therapy and pelvic floor exercises for pain reduction in primary dysmenorrhea: a prospective observational study. J Bodyw Mov Ther. (2023) 36:185–91. doi: 10.1016/j.jbmt.2023.07.002
15. Jiang QY, Li J, Zheng L, Wang GH, Wang J. Constitution of traditional chinese medicine and related factors in women of childbearing age. J Chin Med Assoc. (2018) 81:358–65. doi: 10.1016/j.jcma.2018.01.005
16. Gou CQ, Gao J, Wu CX, Bai DX, Mou HY, Hou XL, et al. Moxibustion for primary dysmenorrhea at different interventional times: a systematic review and meta-analysis. Evid Based Complement Alternat Med. (2016) 2016:6706901. doi: 10.1155/2016/6706901
17. Hsu MF, Tang PL, Pan TC, Hsueh KC. Different traditional Chinese medicine constitution is associated with dietary and lifestyle behaviors among adults in Taiwan. Medicine. (2022) 101:e30692. doi: 10.1097/MD.0000000000030692
18. Patel V, Tanksale V, Sahasrabhojanee M, Gupte S, Nevrekar P. The burden and determinants of dysmenorrhoea: a population-based survey of 2262 women in Goa, India. BJOG. (2006) 113:453–63. doi: 10.1111/j.1471-0528.2006.00874.x
19. Feng YA, Chen CY, Chen TT, Kuo PH, Hsu YH, Yang HI, et al. Taiwan Biobank: a rich biomedical research database of the Taiwanese population. Cell Genom. (2022) 2:100197. doi: 10.1016/j.xgen.2022.100197
20. von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol. (2008) 61:344–9. doi: 10.1016/j.jclinepi.2007.11.008
21. Chiu TH, Huang YC, Chiu H, Wu PY, Chiou HC, Huang JC, et al. Comparison of various obesity-related indices for identification of metabolic syndrome: a population-based study from Taiwan biobank. Diagnostics. (2020) 10:1081. doi: 10.3390/diagnostics10121081
22. Fan CT, Hung TH, Yeh CK. Taiwan regulation of biobanks. J Law, Med Ethics. (2015) 43:816–26. doi: 10.1111/jlme.12322
23. Burnett MA, Antao V, Black A, Feldman K, Grenville A, Lea R, et al. Prevalence of primary dysmenorrhea in Canada. J Obstetr Gynaecol Canada. (2005) 27:765–70. doi: 10.1016/S1701-2163(16)30728-9
24. Nohara M, Momoeda M, Kubota T, Nakabayashi M. Menstrual cycle and menstrual pain problems and related risk factors among Japanese female workers. Ind Health. (2011) 49:228–34. doi: 10.2486/indhealth.MS1047
25. Carroquino-Garcia P, Jiménez-Rejano JJ, Medrano-Sanchez E, de la Casa-Almeida M, Diaz-Mohedo E, Suarez-Serrano C. Therapeutic exercise in the treatment of primary dysmenorrhea: a systematic review and meta-analysis. Phys Ther. (2019) 99:1371–80. doi: 10.1093/ptj/pzz101
26. Pitts MK, Ferris JA, Smith AM, Shelley JM, Richters J. Prevalence and correlates of three types of pelvic pain in a nationally representative sample of Australian women. Med J Aust. (2008) 189:138–43. doi: 10.5694/j.1326-5377.2008.tb01945.x
27. Latthe P, Mignini L, Gray R, Hills R, Khan K. Factors predisposing women to chronic pelvic pain: systematic review. BMJ. (2006) 332:749–55. doi: 10.1136/bmj.38748.697465.55
28. Hu Z, Tang L, Chen L, Kaminga AC, Xu H. Prevalence and risk factors associated with primary dysmenorrhea among Chinese female university students: a cross-sectional study. J Pediatr Adolesc Gynecol. (2020) 33:15–22. doi: 10.1016/j.jpag.2019.09.004
29. László KD, Gyorffy Z, Adám S, Csoboth C, Kopp MS. Work-related stress factors and menstrual pain: a nation-wide representative survey. J Psychosom Obstet Gynaecol. (2008) 29:133–8. doi: 10.1080/01674820701804423
30. Pawłowski B. Prevalence of menstrual pain in relation to the reproductive life history of women from the Mayan rural community. Ann Hum Biol. (2004) 31:1–8. doi: 10.1080/03014460310001602072
31. Faramarzi M, Salmalian H. Association of psychologic and nonpsychologic factors with primary dysmenorrhea. Iran Red Crescent Med J. (2014) 16:e16307. doi: 10.5812/ircmj.16307
32. Chiu MH, Hsieh HF, Yang YH, Chen HM, Hsu SC, Wang HH. Influencing factors of dysmenorrhoea among hospital nurses: a questionnaire survey in Taiwan. BMJ Open. (2017) 7:e017615. doi: 10.1136/bmjopen-2017-017615
33. Gordley LB, Lemasters G, Simpson SR, Yiin JH. Menstrual disorders and occupational, stress, and racial factors among military personnel. J Occupat Environ Med. (2000) 42:871–81. doi: 10.1097/00043764-200009000-00005
34. Chang HT, Hsu NW, Chen HC, Tsao HM, Lo SS, Chou P. Associations between body mass index and subjective health outcomes among older adults: findings from the yilan study, Taiwan. Int J Environ Res Public Health. (2018) 15:2645. doi: 10.3390/ijerph15122645
35. Tai CJ, Wu CC, Lee KT, Tseng TG, Wang HC, Chang FR, et al. The impact of urate-lowering therapy in post-myocardial infarction patients: insights from a population-based, propensity score-matched analysis. Clin Pharmacol Ther. (2022) 111:655–63. doi: 10.1002/cpt.2473
36. Austin PC. Balance diagnostics for comparing the distribution of baseline covariates between treatment groups in propensity-score matched samples. Stat Med. (2009) 28:3083–107. doi: 10.1002/sim.3697
37. Ohde S, Tokuda Y, Takahashi O, Yanai H, Hinohara S, Fukui T. Dysmenorrhea among Japanese women. Int J Gynaecol Obstet. (2008) 100:13–7. doi: 10.1016/j.ijgo.2007.06.039
38. Santer M, Warner P, Wyke S. A Scottish postal survey suggested that the prevailing clinical preoccupation with heavy periods does not reflect the epidemiology of reported symptoms and problems. J Clin Epidemiol. (2005) 58:1206–10. doi: 10.1016/j.jclinepi.2005.02.026
39. Santos LBD, Barbosa IR, Dantas THM, Araujo CM, Dantas JH, Ferreira CWS, et al. Prevalence of primary dysmenorrhea and associated factors in adult women. Rev Assoc Med Bras. (2022) 68:31–36. doi: 10.1590/1806-9282.20210341
40. Chang HY, Fang HL, Huang CY, Chiang CY, Chuang SY, Hsu CC, et al. Developing and validating risk scores for predicting major cardiovascular events using population surveys linked with electronic health insurance records. Int J Environ Res Public Health. (2022) 19:1319. doi: 10.3390/ijerph19031319
41. Stuckey BG, Walsh JP, Ching HL, Stuckey AW, Palmer NR, Thompson PL, et al. Erectile dysfunction predicts generalised cardiovascular disease: evidence from a case-control study. Atherosclerosis. (2007) 194:458–64. doi: 10.1016/j.atherosclerosis.2006.08.043
42. Vicari E, Malaguarnera M, La Vignera S, Calzavara F, Battiato C, Calogero AE. Efficacy and limits of sildenafil citrate in patients with arterial erectile dysfunction: role of peripheral arterial disease and cardiovascular comorbidities. Asian J Androl. (2008) 10:847–53. doi: 10.1111/j.1745-7262.2008.00435.x
43. Jo J, Lee SH. Heat therapy for primary dysmenorrhea: a systematic review and meta-analysis of its effects on pain relief and quality of life. Sci Rep. (2018) 8:16252. doi: 10.1038/s41598-018-34303-z
44. Yang B, Qin QZ, Han LL, Lin J, Chen Y. Spa therapy (balneotherapy) relieves mental stress, sleep disorder, and general health problems in sub-healthy people. Int J Biometeorol. (2018) 62:261–72. doi: 10.1007/s00484-017-1447-5
45. Brown J, Brown S. Exercise for dysmenorrhoea. Cochrane Database Syst Rev. (2010) 2010:Cd004142. doi: 10.1002/14651858.CD004142.pub2
Keywords: body symptoms, cold extremities, edema, primary dysmenorrhea, risk factors
Citation: Tseng T-G, Kuan C-Y, Lo Y-P, Chuang Y-S, Lee C-Y, Lin Y-T, Shyu I-L, Wu M-T and Tai C-J (2025) Clinical association of body symptoms and primary dysmenorrhea among young and middle-aged women: an observational study. Front. Med. 12:1529470. doi: 10.3389/fmed.2025.1529470
Received: 17 November 2024; Accepted: 24 February 2025;
Published: 10 March 2025.
Edited by:
Manuela Deodato, University of Trieste, ItalyReviewed by:
Giacomo Rossettini, University of Verona, ItalyCopyright © 2025 Tseng, Kuan, Lo, Chuang, Lee, Lin, Shyu, Wu and Tai. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chi-Jung Tai, dGFpY2hpanVuZ0BnbWFpbC5jb20=; Ming-Tsang Wu, bWluZ3Rzd3VAZ21haWwuY29t
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.