
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Med. , 28 March 2025
Sec. Pulmonary Medicine
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1513370
This article is part of the Research Topic Distinct phenotype but same genotype: Hints for the diversity of phenotypes in ciliopathies View all articles
Kartagener syndrome (KS), a subtype of primary ciliary dyskinesia (PCD), is a rare genetic disorder characterized by situs inversus, chronic sinusitis, bronchiectasis, recurrent respiratory infections, and impaired ciliary function. It is diagnosed through physical examination, imaging techniques such as computed tomography (CT), nasal nitric oxide measurement, genetic testing, and pulmonary function tests. We present a case study of a 15-year-and-11-month-old male patient with KS complicated by sinusitis, secretory otitis media, and bronchiectasis. The patient exhibited situs inversus totalis, affecting the lungs, heart, and abdominal organs. Treatment included antibiotics for infection, mucolytics, and pulmonary rehabilitation. Postural drainage and bronchoscopy were performed for lung lavage. Following treatment, the patient’s respiratory symptoms improved, and lung function tests showed improvement. A literature review identified a high prevalence of lung and heart transpositions in Chinese patients with PCD, while abdominal organ transposition was less commonly reported. Genetic analysis revealed compound heterozygous mutations in the DNAH5 gene, specifically c.12279 + 1 G > A (exon 71, NM_001369) and c.9457 C > T (exon 56, NM_001369), including the newly discovered variant c.9457 C > T (exon 56, NM_001369). This novel mutation expands the genetic landscape associated with KS, providing further insights into the underlying genetic basis of the condition. The study emphasizes the clinical features, the limited reporting of abdominal organ transposition, the genetic basis, and the treatment of KS, thereby contributing to the understanding and management of this condition.
Kartagener syndrome (KS), a subtype of primary ciliary dyskinesia (PCD), is a rare genetic disorder characterized by situs inversus, chronic sinusitis, bronchiectasis, recurrent respiratory infections, and impaired motile ciliary function. It is diagnosed through physical examination, imaging techniques such as computed tomography (CT), nasal nitric oxide measurement, genetic testing, and pulmonary function tests (1).
Motile cilia are microscopic, hair-like structures found on the surface of various cells in the body, including those in the respiratory tract, reproductive system, and other organs. They play a crucial role in mucus movement, pathogen clearance, and the coordination of various cellular processes. Defects in motile ciliary structure or function can lead to a range of clinical manifestations, including chronic respiratory infections, bronchiectasis, sinusitis, and situs inversus totalis (2, 3).
Situs inversus totalis is a condition in which the organs of the chest and abdomen are mirrored or transposed from their usual positions. This occurs due to a developmental abnormality during embryogenesis and is seen in approximately 50% of individuals with PCD (4, 5). This condition arises from the dysfunction of nodal cilia, which disrupts left–right asymmetry during embryonic development, resulting in conditions such as situs inversus. Nodal cilia (6) generate a directed flow of fluid within the node region, initiating a signaling cascade that ultimately orchestrates the accurate alignment of internal organs along the left–right axis. Any malfunction or irregularity in nodal cilia can disrupt this intricate process, leading to situs inversus totalis, where the typical left–right organ positioning is reversed.
The clinical presentation of KS is diverse, but the most common features include chronic respiratory symptoms such as chronic cough, recurrent respiratory infections, chronic secretory otitis media, and infertility. Sinusitis is also a common manifestation, leading to chronic nasal congestion, sinus pressure, and nasal discharge. Additionally, situs inversus totalis affects the positioning of the heart, lungs, and abdominal organs, although the extent of organ transposition can vary among individuals.
Diagnosing KS requires a comprehensive evaluation, including clinical assessment, imaging analysis, and functional tests. Chest X-rays, CT scans, and echocardiography are commonly performed to assess the presence of situs inversus totalis and associated respiratory and cardiac abnormalities. High-resolution electron microscopy carried out on airway respiratory cilia can provide insights into the structural abnormalities of cilia, while genetic testing plays a crucial role in identifying specific gene mutations associated with the condition.
Management of KS focuses on symptomatic treatment, prevention of respiratory infections, and promotion of airway clearance. Antibiotics are prescribed to treat infections, while mucolytics and pulmonary physiotherapy techniques aid in mucus clearance. Physical therapy techniques, such as postural drainage and bronchoscopy with lavage, can also be used to facilitate the removal of secretions from the airways.
In this case study, we present a 15-year-and- 11-month-old male patient with KS, highlighting the clinical features, diagnostic process, genetic findings, and treatment outcomes. The identification of a novel mutation in the DNAH5 gene expands our understanding of the genetic basis of KS. This report contributes to the existing literature on KS, emphasizing the importance of early recognition, accurate diagnosis, and appropriate management of this rare genetic disorder.
The patient, a 15-year-and-11-month-old male, had a history of recurrent respiratory infections since the fourth day after birth, characterized by cough, sputum production, nasal congestion, and yellow nasal discharge without wheezing. The child received repeated antimicrobial treatments locally, resulting in symptom improvement. However, these symptoms have persisted and gradually worsened over the past 14 years, accompanied by decreased exercise tolerance and occasional self-resolving dizziness. At the age of 14 years and 8 months, the patient began experiencing chest tightness, which significantly worsened when lying flat at night but improved when in an upright position. The patient had previously been diagnosed with bronchiectasis and sinusitis. He is the second child of the family, born at full term with normal birth weight and without complications. Both his parents and his siblings are in good health, and there is no family history of genetic or infectious diseases.
On admission, the patient’s vital signs—temperature, pulse rate, respiratory rate, and blood pressure—were within normal ranges. Physical examination revealed that the patient appeared alert and responsive, with no cyanosis observed on the lips. An examination of the throat revealed congestion, and the tonsils were moderately enlarged (grade II). The chest exhibited deformities characterized by asymmetric thoracic structures and localized protrusion on the left side. No nasal flaring was observed, and the three-concave sign was negative. Auscultation of the lungs revealed wet rales bilaterally, and digital clubbing was present.
Laboratory tests showed a mild elevation in white blood cell count during routine blood tests, and C-reactive protein levels were also detected. Other laboratory parameters, such as liver and kidney function, electrolytes, serum proteins, coagulation function, ferritin, and total T lymphocyte analysis, were generally within normal ranges. Tests for hepatitis viruses A, B, C, syphilis spirochetes, and human immunodeficiency virus were all negative. Immunoglobulin A was elevated at 4.74 g/L (normal range: 0.47–2.49 g/L), and immunoglobulin M was elevated at 2.21 g/L (normal range: 0.15–1.88 g/L), while immunoglobulin G levels were normal. Sputum culture confirmed the presence of Pseudomonas aeruginosa. Lung function testing revealed a mixed ventilatory function disorder characterized by significant reductions in lung capacity and expiratory flow rates. The fractional exhaled nitric oxide (FeNO) level was normal.
Chest CT showed bronchiectasis with infection in both lungs (Figure 1A), accompanied by situs inversus totalis (Figure 1B) and sinusitis (Figure 1C). Abdominal CT confirmed situs inversus (Figure 1D). Echocardiography demonstrated dextrocardia with situs inversus (Figure 2A) and a right-sided aortic arch (Figure 2B). Otoscopy revealed congestion in the left tympanic membrane and cloudiness in the right with unclear landmarks.
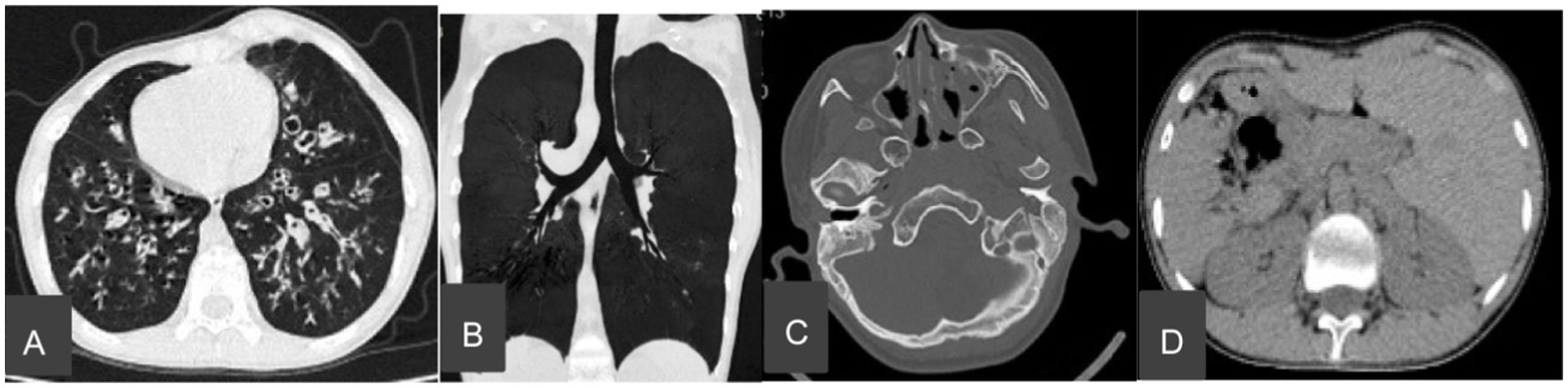
Figure 1. Computed tomography (CT) image of chest and nasal. (A) Bronchiectasis and bronchiolitis. (B) Situs inversus totalis. (C) Sinusitis. (D) Abdominal organ displacement.
Preliminary diagnosis: pneumonia, bronchiectasis, and associated visceral situs inversus. The treatment plan included ceftazidime for infection, acetylcysteine for mucolysis, and mechanical assistance for sputum clearance. An ENT specialist consultation revealed bilateral abundant purulent nasal discharge, hypertrophied inferior turbinates, and signs of rhinitis and sinusitis. Aggressive anti-inflammatory and decongestant treatments were initiated, such as saline nasal irrigation and nasal steroid sprays. Otoscopic examination indicated intact bilateral tympanic membranes, with congestion in the left and opacity in the right, suggestive of acute otitis media (left) and serous otitis media (right). Ofloxacin ear drops were administered to the left ear.
Following the treatment, the child’s cough improved, and there was a noticeable change in the color of sputum and nasal discharge, transitioning from yellow to white. Subsequent lung function tests revealed an improvement compared to previous results. However, the cough with sputum and nasal discharge persisted. Due to the patient’s history of recurrent respiratory infections, rhinitis, and sinusitis since childhood, along with worsening symptoms, decreased exercise tolerance, and occasional dizziness, the pulmonary CT scan findings of bronchiectasis and visceral situs inversus raised the suspicion of KS.
Bronchoscopy lavage treatment was performed, revealing a significant amount of yellow-white thread-like secretions in the airway, along with fishbone-like changes. Microscopic examination of the bronchial mucosa showed thinning and shedding of epithelium, fibrous hyperplasia underneath, mild infiltration of inflammatory cells, and fibrin exudation. Imunohistochemistry of the bronchial mucosa showed positive staining for cytokeratin, smooth muscle actin, and vimentin, while showing negative results for acid-fast and periodic acid-Schiff staining, indicating the presence of epithelial and smooth muscle cells, mesenchymal cells, and the absence of acid-fast bacteria or glycogen in the tissue. Electron microscopy examination of cilia structure revealed a clear 9 + 2 microtubule structure in some cilia cross-sections, with a significant absence of outer dynein arms in all evaluable cilia (Figure 3).
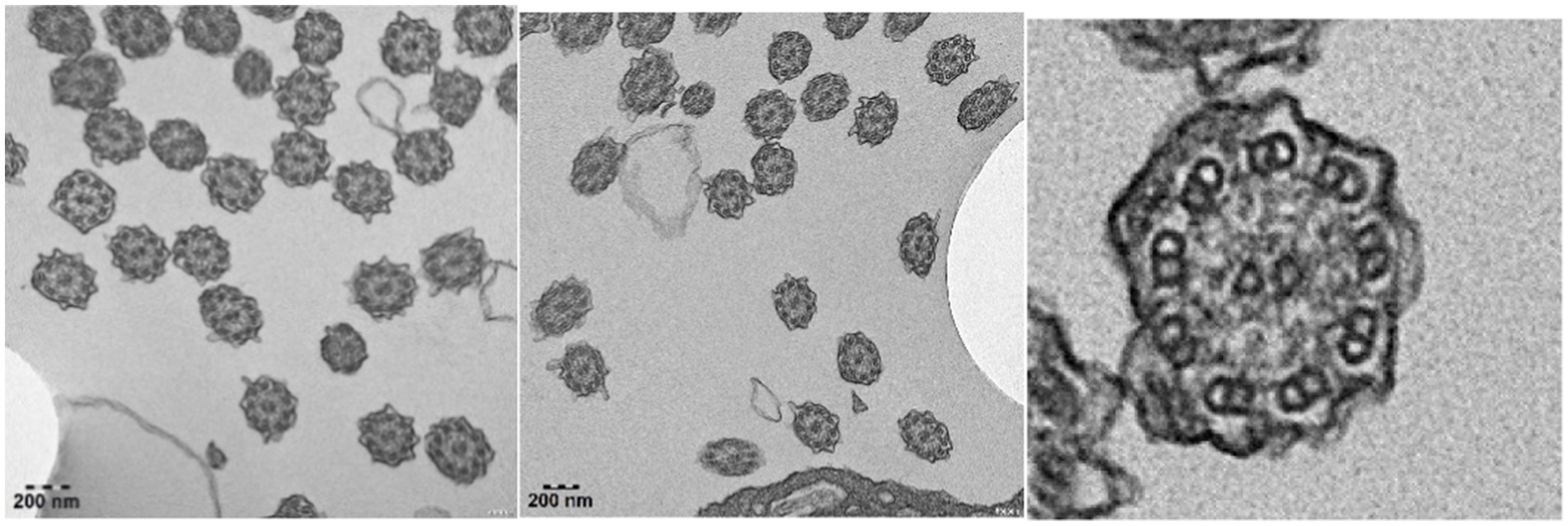
Figure 3. Electron microscopy imaging revealed respiratory cilia with a notable absence of outer dynein arms.
Furthermore, genetic testing was performed. Ethical approval was obtained from our hospital, and informed consent was obtained from the parents. Peripheral blood samples were collected from both the patient and his mother. The child’s father was unable to provide a blood sample due to COVID-19 prevention and control measures. He is working in another city and is unable to return for the blood collection. The samples were sent to the Medical Laboratory of Mygenostics (Mygenostics Co. Ltd.) for whole-exome sequencing. Pathogenicity analysis was conducted according to the guidelines of the American College of Medical Genetics and Genomics (ACMG). The following variants were identified in the DNAH5 (NM_001369) gene of the child (Table 1; Figure 4).
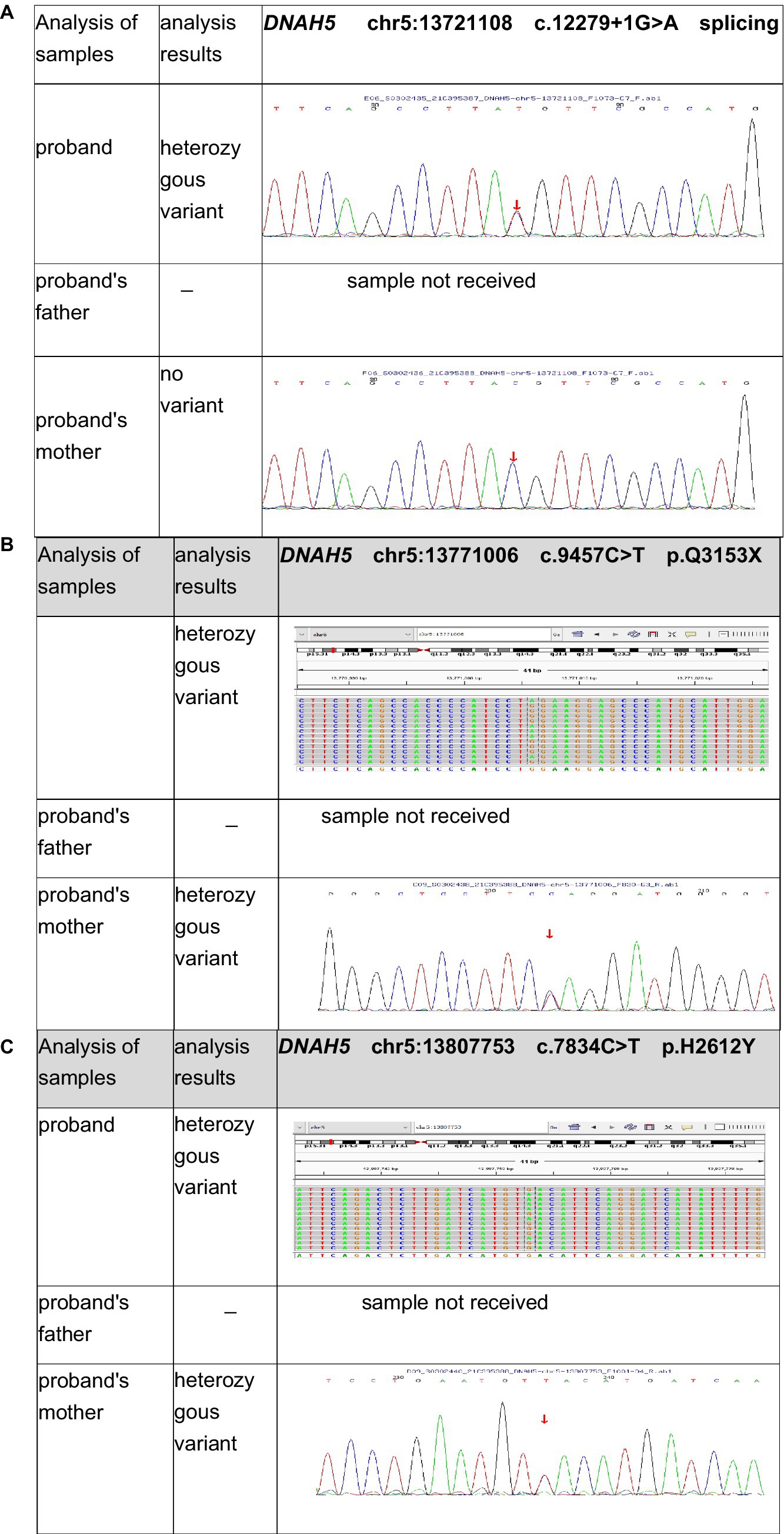
Figure 4. Test results for the patient’s first and second generation revealed the following mutations in DNAH5 (NM_001369): (A) Exon 71: c.12279 + 1 G > A. (B) Exon 56: c.9457 C > T. (C) Exon 47: c.7834C > T.
Based on the diagnostic analysis provided, the child was ultimately diagnosed with Kartagener syndrome. Specifically, the child exhibited situs inversus totalis, affecting the lungs, heart, and abdominal organs.
Exon 71: c.12279 + 1 G > A (Figure 4A), resulting in an amino acid splice site mutation. This variant was preliminarily classified as a pathogenic variant with strong evidence (PVS1 + PM2_Supporting+PM3_Strong).
Exon 56: c.9457 C > T (Figure 4B), resulting in an amino acid change p.Q3153X, a nonsense mutation. This variant was preliminarily classified as a likely pathogenic variant with supporting evidence (PVS1 + PM2_Supporting). No relevant reports on this variant were found in the literature databases.
Exon 47: c.7834C > T (Figure 4C), resulting in an amino acid change p.H2612Y, a missense mutation. This variant was preliminarily classified as a variant of uncertain clinical significance with supporting evidence (PM2_Supporting+BP4).
Sanger sequencing was performed for validation. Parental origin analysis revealed that the child’s mother carried heterozygous variants at c.9457C > T (p.Q3153X) and c.7834C > T (p.H2612Y), while the variant at 12279 + 1G > A (splicing) was not detected. The child’s father was not sampled. Consequently, the child was found to have compound heterozygous mutations in the DNAH5 (NM_001369) gene: exon 71:c.12279 + 1G > A (splicing) and exon 56:c.9457 C > T (p.Q3153X).
In this paper, we explored the presentation and clinical characteristics of PCD or KS in China. We conducted a literature review using the following keywords in the Chinese databases Wanfang and CNKI: “Primary Ciliary Dyskinesia,” “Kartagener Syndrome,” “DNAH5,” and “China.” We selected cases with visceral transposition from the literature search. In the PubMed database, we used the keywords “primary ciliary dyskinesia,” “Kartagener syndrome,” “situs inversus,” “DNAH5,” and “China” for the search.
Based on our literature review, we identified 15 relevant articles involving 17 patients. The articles selected pertain to PCD or KS in China, specifically focusing on cases with visceral transposition and DNAH5 mutations. Among these, 16 patients (94.1%) were reported to have situs inversus of the lungs, while information on lung situs inversus was not provided for 1 patient (5.9%). Additionally, all 17 patients (100%) had situs inversus of the heart. However, only 6 patients (35.3%) were reported to have abdominal organ transposition, while information on abdominal organ transposition was not mentioned for 11 patients (64.7%).
KS, also known as a subtype of PCD, is a rare autosomal recessive disorder characterized by chronic sinusitis, bronchiectasis, and situs inversus totalis (1, 4). The patient in this case study exhibited typical clinical features of KS, including recurrent respiratory infections, chronic cough, nasal congestion, and bronchiectasis. The presence of situs inversus totalis, dextrocardia, and right-sided aortic arch further supported the diagnosis. Additionally, physical examination findings, such as digital clubbing, wet rales on lung auscultation, and chest deformities, reinforced the presence of chronic respiratory disease.
These research findings (7, 8) highlight a high prevalence of lung and heart transpositions in Chinese patients with PCD or KS. However, it should be noted that while abdominal organ transposition appears to be less commonly reported in these patients, it does not necessarily indicate a low incidence of the condition. The lack of mention in the article could suggest insufficient attention from healthcare professionals toward abdominal organ transposition in KS. Further studies and case reports are needed to fully understand the prevalence and clinical implications of abdominal organ transposition in these patients. By increasing awareness of this aspect, clinicians can improve the understanding and treatment provided to individuals with KS.
KS is primarily caused by defects in the structure or function of motile cilia (1), which can result from mutations in various genes (9, 10). Among these genes, DNAH5 plays a crucial role in ciliary function (11). The DNAH5 gene encodes dynein axonemal heavy chain 5, a protein crucial for the structure and function of motile cilia (12). Mutations in the DNAH5 gene disrupt the normal ciliary function, leading to impaired mucociliary clearance and subsequent respiratory and reproductive tract abnormalities seen in KS (13).
In the present case, whole-exome sequencing identified compound heterozygous mutations in the DNAH5 gene. The variants identified, including the newly discovered mutation in exon 56 (c.9457 C > T), contribute to the development of KS. These mutations were classified based on the ACMG guidelines, with the splice site mutation in exon 71 (c.12279 + 1 G > A) being classified as pathogenic and the mutation in exon 56 (c.9457 C > T) as likely pathogenic. Understanding the genetic basis of KS, particularly the role of DNAH5 gene mutations, is crucial for accurate diagnosis, genetic counseling, and potential targeted therapies in the future.
In this study, electron microscopy analysis revealed significant structural abnormalities in the ciliated epithelial cells of patients with PCD. Specifically, there was a notable absence of outer dynein arms in all evaluated cilia, which is consistent with previous reports linking PCD to the loss of outer dynein arms. This observation further supports the understanding that the absence of outer dynein arms impairs the coordinated movement of cilia, leading to compromised mucociliary clearance and the accumulation of mucus in the respiratory tract. Consequently, our findings suggest a potential pathological basis for bronchiectasis and chronic respiratory symptoms in PCD. Based on the literature (6, 7), DNAH5 mutations are associated with cilia structural abnormalities, such as outer dynein arm loss, consistent with our observations. However, further studies are needed to confirm these findings.
The treatment approach for KS focuses on managing respiratory symptoms, preventing infections, and improving mucociliary clearance (14). In the present case, the patient received cephalosporin antibiotics to address the Pseudomonas aeruginosa infection, acetylcysteine for mucolysis, and pulmonary rehabilitation. Postural drainage and bronchoscopy with lavage were performed to facilitate clearance of secretions from the airways. These interventions resulted in clinical improvement, with the resolution of cough and improvements in sputum and nasal discharge. In advanced stages of PCD, lung transplantation can be considered. Marro et al. (15) described the outcomes of the largest lung transplant population for PCD and Kartagener Syndrome, suggesting that lung transplantation is an acceptable treatment option in this population.
The patient’s mother expressed dissatisfaction with the previous treatments for recurrent respiratory infections. They sought care at a leading children’s hospital, where, despite efforts, the child’s condition persisted. While a definitive diagnosis was made, the parents felt that a cure had not been achieved. They believed the worsening of the child’s condition was linked to factors at the boarding school, where the child refrained from coughing forcefully, resulting in mucus retention and bronchiectasis. Following effective communication with the doctors, the parents and child embraced pulmonary rehabilitation and preventive measures, leading to a reduction in hospitalizations. Psychological counseling supported the child’s engagement in outdoor activities.
In conclusion, this case report highlights the clinical features, the underrepresentation of abdominal organ transposition, the genetic basis, and the treatment of KS in a 15-year-and-11-month-old male patient. The identification of compound heterozygous mutations in the DNAH5 gene further elucidates the pathogenesis of the syndrome. By increasing awareness of KS and its various aspects, clinicians can improve diagnosis, management, genetic counseling, and potential therapeutic approaches for affected patients. Parental feedback underscores the importance of ongoing communication, psychological support, and societal assistance for patients with chronic illnesses.
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding author.
Written informed consent was obtained from the individual(s), and minor(s)’ legal guardian/next of kin, for the publication of any potentially identifiable images or data included in this article.
JZ: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. LG: Data curation, Software, Writing – review & editing. YX: Data curation, Resources, Writing – review & editing. HW: Conceptualization, Visualization, Writing – review & editing. XL: Formal analysis, Validation, Writing – review & editing. YZ: Investigation, Project administration, Supervision, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
We would like to extend our heartfelt thanks to Dr. Richard Chavasse from the Department of Pediatrics at St. George’s Hospital, United Kingdom, for his valuable contributions and expert guidance in reviewing and enhancing this article. His insights and attention to detail have significantly improved the quality and clarity of the content. While Dr. Chavasse is not a co-author of this paper, his dedicated support and feedback throughout the writing process were invaluable.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Shapiro, AJ, Davis, SD, Polineni, D, Manion, M, Rosenfeld, M, Dell, SD, et al. Diagnosis of primary ciliary dyskinesia. An official American Thoracic Society clinical practice guideline. Am J Respir Crit Care Med. (2018) 197:e24–39. doi: 10.1164/rccm.201805-0819ST
2. Wallmeier, J, Nielsen, KG, Kuehni, CE, Lucas, JS, Leigh, MW, Zariwala, MA, et al. Motile ciliopathies. Nat Rev Dis Primers. (2020) 6:77. doi: 10.1038/s41572-020-0209-6
3. Leigh, MW, Horani, A, Kinghorn, B, O'Connor, MG, Zariwala, MA, and Knowles, MR. Primary ciliary dyskinesia (PCD): a genetic disorder of motile cilia. Transl Sci Rare Dis. (2019) 4:51–75. doi: 10.3233/TRD-190036
4. Goutaki, M, Meier, AB, Halbeisen, FS, Lucas, JS, Dell, SD, Maurer, E, et al. Clinical manifestations in primary ciliary dyskinesia: systematic review and meta-analysis. Eur Respir J. (2016) 48:1081–95. doi: 10.1183/13993003.00736-2016
5. Best, S, Shoemark, A, Rubbo, B, Patel, MP, Fassad, MR, Dixon, M, et al. Risk factors for situs defects and congenital heart disease in primary ciliary dyskinesia. Thorax. (2019) 74:203–5. doi: 10.1136/thoraxjnl-2018-212104
6. Little, RB, and Norris, DP. Right, left and cilia: how asymmetry is established. Semin Cell Dev Biol. (2021) 110:11–8. doi: 10.1016/j.semcdb.2020.06.003
7. Zhang, S, Wang, C, Zhang, Y, Hu, Y, Li, X, and Zhi, C. Genetic analysis of a child with Kartagener syndrome due to novel compound heterozygous variants of DNAH5 gene. Zhonghua Yi Xue Yi Chuan Xue Za Zhi. (2023) 40:71–5. doi: 10.3760/cma.j.cn511374-20220110-00020
8. Zhang, L, Feng, X, Zhang, J, Hao, Y, and Wang, Y. Co-occurrence of Moyamoya syndrome and Kartagener syndrome caused by the mutation of DNAH5 and DNAH11: a case report. BMC Neurol. (2020) 20:314. doi: 10.1186/s12883-020-01895-x
9. Hannah, WB, Seifert, BA, Truty, R, Zariwala, MA, Ameel, K, Zhao, Y, et al. The global prevalence and ethnic heterogeneity of primary ciliary dyskinesia gene variants: a genetic database analysis. Lancet Respir Med. (2022) 10:459–68. doi: 10.1016/S2213-2600(21)00453-7
10. Guan, Y, Yang, H, Yao, X, Xu, H, Liu, H, Tang, X, et al. Clinical and genetic Spectrum of children with primary ciliary dyskinesia in China. Chest. (2021) 159:1768–81. doi: 10.1016/j.chest.2021.02.006
11. Zhao, X, Bian, C, Liu, K, Xu, W, Liu, Y, Tian, X, et al. Clinical characteristics and genetic spectrum of 26 individuals of Chinese origin with primary ciliary dyskinesia. Orphanet J Rare Dis. (2021) 16:293. doi: 10.1186/s13023-021-01840-2
12. Hornef, N, Olbrich, H, Horvath, J, Zariwala, MA, Fliegauf, M, Loges, NT, et al. DNAH5 mutations are a common cause of primary ciliary dyskinesia with outer dynein arm defects. Am J Respir Crit Care Med. (2006) 174:120–6. doi: 10.1164/rccm.200601-084OC
13. Nöthe-Menchen, T, Wallmeier, J, Pennekamp, P, Höben, IM, Olbrich, H, Loges, NT, et al. Randomization of left-right asymmetry and congenital heart defects: the role of DNAH5 in humans and mice. Circ Genom Precis Med. (2019) 12:e002686. doi: 10.1161/CIRCGEN.119.002686
14. Paff, T, Omran, H, Nielsen, KG, and Haarman, EG. Current and future treatments in primary ciliary dyskinesia. Int J Mol Sci. (2021) 22:9834. doi: 10.3390/ijms22189834
Keywords: primary ciliary dyskinesia, Kartagener syndrome, ciliary function, situs inversus, genetic mutations, DNAH5
Citation: Zhang J, Gao L, Xing Y, Wu H, Liu X and Zhang Y (2025) Analysis of clinical and genetic features in a pediatric patient with Kartagener syndrome caused by compound heterozygous mutations in the DNAH5 gene: a case study and literature review. Front. Med. 12:1513370. doi: 10.3389/fmed.2025.1513370
Received: 18 October 2024; Accepted: 03 March 2025;
Published: 28 March 2025.
Edited by:
Mark O. Wielpütz, Heidelberg University, GermanyReviewed by:
Rute Pereira, University of Porto, PortugalCopyright © 2025 Zhang, Gao, Xing, Wu, Liu and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yingqian Zhang, emhhbmd5aW5ncWlhbjY2NkAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.