
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Med. , 19 March 2025
Sec. Family Medicine and Primary Care
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1513272
This article is part of the Research Topic The Increasing Relevance of Traditional Medicine Systems for the Primary Health Care Sector and General Practice: Global Research Perspectives – Volume II View all 10 articles
 Meng-Yuan Shen1†
Meng-Yuan Shen1† Ze-Jiong Li1†
Ze-Jiong Li1† Shu-Han Wang1†
Shu-Han Wang1† Tian-Chen Lin1
Tian-Chen Lin1 Qin-Yi Lou1
Qin-Yi Lou1 Shan Liu1
Shan Liu1 Dan-Dan Feng2
Dan-Dan Feng2 Dong-Dong Yang1
Dong-Dong Yang1 Chen-Juan Wang1
Chen-Juan Wang1 Zhe-Kai Ying1
Zhe-Kai Ying1 Rong Zhou1
Rong Zhou1 Jian-Nong Wu2*
Jian-Nong Wu2*Introduction: This study aims to conduct a systematic review and meta-analysis of randomized controlled trials to evaluate the efficacy and safety of auricular acupoint therapy (AAT) for functional gastrointestinal disorders (FGIDs).
Methods: We conducted a thorough search across eight databases, including PubMed, EMBASE, Web of Science, the Cochrane Library, CNKI, Wanfang, VIP, and CBM. The search covered the period from the inception of each database up to June 30, 2024. The authors independently reviewed all the references, evaluated the risk of bias, and extracted the data. GRADEpro software was utilized to calculate overall strength of evidence. A random effects or fixed effects model was selected on the basis of the p-value and I2. RevMan 5.3, Stata/MP 18.0, R 4.3.1 and R Studio 2023.09.0 were used for data processing. TSA 0.9.5.10 beta software was used to evaluate data stability.
Results: The review included 19 randomized controlled trials with a total of 1,681 patients (895 in the treatment group and 886 in the control group). The treatment duration ranged from 2–12 weeks. The meta-analysis revealed that, compared with the control, AAT was significantly more effective at treating FGIDs (RR: 1.35; 95% CI: 1.21–1.51; p < 0.001), reducing the symptom score (MD: −1.94; 95% CI: −3.06 to −0.85; p < 0.001; five trials), improving the SAS score (MD: −12.47; 95% CI: −13.92 to −11.01; p < 0.001; five trials), and improving the SDS score (MD: −4.97; 95% CI: −9.23 to −0.72; p = 0.02; six trials). A total of two articles mentioned relatively significant adverse reactions (MD: 2.98; 95% CI: 0.51–17.26; p = 0.009). Sensitivity and trial sequential analyses confirmed the stability of these results.
Discussion: While our meta-analysis suggests that AAT may offer benefits for FGIDs, these results must be interpreted with caution due to methodological limitations in the included trials. Further investigations in high-quality trials are warranted.
Systematic review registration: https://clinicaltrials.gov/, identifier CRD42024558786.
Functional gastrointestinal disorders (FGIDs), including functional dyspepsia (FD), functional constipation (FC), and irritable bowel syndrome (IBS). It is characterized by chronic gastrointestinal symptoms without any identifiable organic pathology (1–3). According to the Rome IV criteria, FGIDs are classified as gut-brain interaction disorders. They are divided into eight categories based on the basis of different gastrointestinal symptoms, encompassing a total of 32 distinct conditions (4, 5). FD, IBS, and FC are present worldwide, and their incidence is increasing worldwide. Studies show that FD affects approximately 16% of the global population, significantly impacting patients’ quality of life (6). The prevalence of IBS ranges from 5 to 10%, and that of FC is around 15.3%. These disorders significantly affect individuals’ quality of life, work productivity, and society through frequent medical consultations, medication use, and over-the-counter treatments (7, 8). Thus, effective prevention and treatment strategies for FGIDs are crucial in clinical practice.
The pathophysiological mechanism of FGIDs is complex, and its development is the result of the interaction of physiological, psychological and social factors. At present, the pathogenesis of FGIDs has not been fully elucidated (9, 10). Currently, FGID management relies primarily on gastrointestinal motility agents, along with antianxiety and antidepression treatments. However, its clinical use is limited by long-term side effects, unclear efficacy, and safety concerns associated with various treatments. However, no approach fully addresses the complex pathological changes associated with FGIDs.
Auricular acupoint therapy (AAT), also known as auriculotherapy or auricular therapy, is a key component of traditional Chinese medicine (TCM). AAT has been used in TCM for thousands of years to treat a variety of conditions. It is a non-invasive and drug-free treatment that has gained increasing attention in recent years for its potential to improve symptoms and quality of life in patients with FGIDs. According to TCM theory, specific auricular points (e.g., “large intestine,” “spleen,” and “liver”) corresponding to internal organs and physiological functions. Stimulating these points is thought to regulate qi and blood flow, harmonize Zang-Fu organ systems, and restore gastrointestinal homeostasis (11–13). Modern studies further suggest that AAT can enhance gastric hypersensitivity in rats with FD by balancing sympathetic and vagal nerve activity, suggesting its potential for alleviating gastric pain in FD patients (14). Additionally, stimulating ear points can activate the brainstem to release dopamine, reduce food intake, promote stomach relaxation, and regulate mood (15, 16). These studies show that AAT modulates the brain-gut axis via vagal nerve activation, reducing visceral hypersensitivity and normalizing gut motility—key mechanisms disrupted in FGIDs (17, 18). Additionally, the non-invasive nature and minimal adverse effects of AAT make it an appealing alternative to pharmacological therapies, particularly for chronic conditions requiring long-term management (12). Recent RCTs have demonstrated its efficacy in alleviating FGID symptoms, supporting its integration into evidence-based practice (19, 20).
Considering the current lack of high-quality meta-analyses and the specific impact of AAT on FGID, it is still not fully understood. The purpose of this article is to analyze and summarize the efficacy and safety of AAT in the treatment of FGID thoroughly.
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-analysis Statement. The review protocol was prospectively registered in the International Prospective Registration of Systematic Reviews and publicly available (CRD42024558786) and followed without deviation. All planned outcomes, analyses, and subgroup assessments were executed as specified in the registration.
We conducted a thorough search across eight databases, including PubMed, EMBASE, Web of Science, Cochrane Library, CNKI, WanFang, VIP, and CBM. The search covered the period from the inception of each database up to June 30, 2024. Two investigators (Shu-Han Wang, Ze-Jiong Li) independently screened all records identified through the search and reviewed the full text for eligibility. For studies that met the inclusion criteria, data were extracted independently by two authors and then reviewed by a third author (Qin-Yi Lou). Any disputes will be resolved by the corresponding author (Jian-Nong Wu). This protocol was applied to all database searches, with some modifications to search terms and operators. The search criteria were based on participants, intervention, comparison, outcome, time, and study design (PICOTS), and the search strategy was structured around the search terms “functional dyspepsia, “Irritable Bowel Syndrome, and “functional constipation, and “functional gastrointestinal disorders, and “Auricular acupoint therapy, and “randomized controlled trials, Subject terms, their synonymous free words, and qualifiers were used to improve search sensitivity: (“Indigestion” OR “postprandial distress syndrome” OR “epigastric pain syndrome” OR “indigestion” OR “functional dyspepsia” OR “functional dyspepsia” OR “irritable bowel syndromes” OR “mucous colitis” OR “mucous colitides” OR “syndrome, irritable bowel” OR “syndromes, irritable bowel” OR “irritable colon” OR “colitis, mucous” OR “colitides, mucous” OR “mucouscolitides” OR “functional constipation” OR “dyschezia” OR “colonic inertia” OR “chronic functional constipation” OR “slow transit constipation” OR “chronic severe functional constipation” OR “functional gastrointestinal disorders” OR “functional bowel disease” OR “functional gastrointestinal disease”) AND (“Acupuncture” OR “electroacupuncture” OR “bloodletting” OR “laser” OR “acupressure” OR acupoint” OR “therapy” OR “stimulate” OR press stick needle” OR “auriculotherapy” OR “otopoint” OR “ototherapy OR “Auricular” OR “ear” OR “acupuncture treatments”) AND (“randomized controlled trial” OR “randomized”). In PubMed, search results were limited to “randomized controlled trials.” The search strategy is detailed in Supplementary Table S1.
The eligibility of studies was determined based on the following criteria: (i) Study Type: RCTs reported in English or Chinese. (ii) Participants: Patients over 18 years old with FGID, diagnosed according to criteria including but not limited to the latest version of the Rome criteria. (iii) Intervention: All forms of AAT that are not combined with other interventions, such as acupuncture, electroacupuncture. (iv) Comparison: The control group includes the regular care group, placebo group, and western medicine group. The placebo group involves the use of non-irritating objects in placebo patches or sham interventions. (v) Outcomes: studies evaluating treatment effectiveness were considered eligible.
Exclusion criteria included the following: (i) Duplication, defined as the same data from subjects in different studies by the same authors; (ii) Unavailability of full text or critical data of the articles was not obtained.
Two authors independently screened all records identified through the search. They then reviewed the full text for eligibility. Data were extracted by two authors and reviewed by a third author. Any discrepancies were resolved by the corresponding author. The recorded study characteristics included author, country, and year of publication; participant characteristics such as diagnostic criteria, age, gender, and number of cases in each group; intervention information including measures of intervention and control, treatment duration, and adverse events; as well as outcome details such as outcomes, adverse events, and follow-ups. Any differences were settled through discussion. Authors of studies with incomplete or ambiguous reported data were contacted.
The quality of RCTs was assessed using the Cochrane Risk-of-Bias Tool (21), which considered randomization methods, allocation concealment, blinding procedures, data integrity, selective result reporting, and other potential sources of bias. These were categorized as “unknown risk,” “low risk,” or “high risk” based on migration criteria.
The GRADE system (22) was used to rate the quality of evidence for the outcomes assessed. For each outcome, we automatically assigned four points to each study because they were RCTs, and downgraded them if there was an increased risk of bias, inconsistency, indirectness, imprecision, and publication bias. We classified the quality of the evidence as A (high), B (moderate), C (low), or D (very low). Two researchers (Tian-Chen Lin and Ze-Jiong Li) independently assessed the quality of the evidence and resolved any disputes by discussion with a third researcher (Jian-Nong Wu).
RevMan 5.3 (Cochrane Collaboration), Stata/MP 18.0 (StataCorp LLC), and R version 4.3.1 (R Foundation for Statistical Computing) with R Studio 2023.09.0 (Posit Software) were used for data analysis. The risk ratio (RR) was used for binary data, while the weighted mean difference (WMD) or standardized mean difference (SMD) was used for continuous data, both with 95% confidence intervals (CI). RR values and their 95% CIs were combined, and routine meta-analyses were performed. Heterogeneity was assessed using the Cochran Q test and I2 statistic (23). If p > 0.1 and I2 ≤ 50%, the fixed-effect model was used; if p < 0.1 or I2 > 50%, the random-effects model was applied along with subgroup and sensitivity analyses to identify sources of heterogeneity (24, 25). Additionally, a cumulative meta-analysis using the “metacum” package in R Studio examined changes in pooled estimates over time to identify trends and assess accuracy while exploring potential sources of heterogeneity. Publication bias was assessed using funnel plots and confirmed through Egger’s test in Stata/MP 18.0. A significance level of p < 0.05 was considered statistically significant (26).
The TSA 0.9.5.10 beta software was used for sequential analysis in the treatment of FGIDs (27). In this study, the Type I error probability was set at α = 0.05, and statistical power was 80%. The relative risk reduction was defined as 20% compared to the control group event rate. If the cumulative Z value reached both the traditional threshold (Z = 1.96) and the TSA threshold, the results were considered conclusive evidence after correction.
The database search identified a total of 673 studies. After the literature screening process, 654 studies were included for further review. Ultimately, 19 RCTs were selected for our analysis (Figure 1).
This systematic review included 1,681 individuals diagnosed with FGIDs from 19 RCTs. The participants comprised 895 individuals in the treatment group and 886 in the control group. All included studies, including English articles, were conducted in China. The studies were first published between 2006 and 2023 and the sample size varied between 20 and 300. The mean age of participants ranged from 21.6 to 75.8 years, and there was no significant bias in the gender distribution. The diagnostic criteria for 2 studies were Rome III (28, 29), 5 studies were Rome III (30–34) and 7 studies were Rome IV (35–40). Some studies use non-Rome criteria (41, 42) or are not explicitly stated (43–45). Six studies focused on FD (30, 35–37, 41), five focused on IBS (31, 38, 39, 42, 43), and eight focused on FC (28, 29, 32–34, 40, 44, 45). Auricular acupuncture points were applied unilaterally in one trial, bilaterally in 15 trials, and were not specified in three trials. The most commonly used acupuncture points include the large intestine (11 items), spleen (8 items), and liver (7 items). The interventions primarily involved AAT in the form of acupressure (13 trials) or electroacupuncture (6 trials). The intervention time and frequency of AAT varied according to the specific type, ranging from 2 weeks to 12 weeks, and the frequency of intervention from 3 times weekly (35, 38) to 5 times daily (29–31, 33, 40). 2 RCT (36) did not explicitly explain the frequency of acupuncture therapy interventions. Six studies used usual care, six studies used placebo, including the sham group, and seven studies used conventional Western medicine (include Moxapride, domperidone, etc.). An overview of the characteristics of all included studies and meta-analysis is provided in Table 1. The locations of the acupoints are detailed in Figure 2.
Outcome indicators mainly focus on treatment effect, symptom score, quality of life, psychological status score and safety, and a few studies have explored specific mechanisms. The primary outcome measure was overall response rate, as assessed by improvement in gastrointestinal symptoms. Secondary outcomes included symptom scores, which were assessed using various scales such as IBS-SSS. In addition, quality of life was measured using scales such as IBS-QOL and PAC-QOL. Some studies used the SAS and the SDS to assess changes in participants’ levels of anxiety and depression. In terms of safety, a few studies reported adverse reactions, mostly mild reactions, such as local skin redness. The remaining details are shown in Table 1.
According to the Cochrane Bias risk Assessment tool, the methodological quality items for all included studies are presented in Supplementary Figure S1. All the included studies were verified to be randomized. One randomized controlled trial detailed the method for generating the assigned sequence and was thus evaluated as low risk (30), three randomized controlled trials were categorized as high risk (29, 40, 43), while the remaining studies were classified as unknown risk. 2 RCTs were double-blinded (36, 37) and 1 RCT (39) was single-blinded, all of which were rated as low risk. Other randomized controlled trials did not provide information on whether the assigned hiding and blind procedures were carried out, resulting in an assessment of the risk of unknown bias. The resulting data were complete and therefore assessed as low risk. Since attempts to obtain the protocol or other relevant information from the first author via email, phone, or fax were unsuccessful, the risk of bias from selective outcome reports and other sources was considered low.
The review included 19 studies demonstrating the effectiveness of AAT in treating FGIDs, which showed a significant therapeutic effect (I2 = 78%, p < 0.00001) (Figure 3). Due to substantial heterogeneity, a random effects model was used. The cumulative random effects meta-analysis consistently yielded similar results (RR: 1.35; 95% CI: 1.21–1.51) even with the inclusion of newly published studies (Figure 4). We explored sources of high heterogeneity by subgroup analysis and sensitivity analysis.
The results of the subgroup analysis showed that no significant differences were found between subgroups based on types of stimulation, ear selection, number of acupoints, and intervention providers, indicating that these factors did not significantly affect clinical outcomes. In the control group, there was no heterogeneity between studies (I2 = 0%), and no significant difference (p = 0.79) compared to conventional Western medicine treatment. However, higher inter-group heterogeneity was observed when compared with conventional care and placebo treatments (I2 = 89, 70%), with statistically significant differences. Studies using fewer than five acupoints showed high inter-group heterogeneity (I2 = 85%), while studies using more than five acupoints showed low inter-group heterogeneity (I2 = 19%). For disease types, studies on IBS showed no inter-group heterogeneity (I2 = 0%), whereas studies on FD and FC showed high inter-group heterogeneity (I2 = 86, 85%). In terms of diagnostic criteria, studies using Rome criteria showed high heterogeneity between groups (I2 = 86, 85%), while studies using non-Rome criteria or other criteria showed minimal or no variability (I2 = 25, 0%). These findings suggest that variation in treatment effectiveness may be related to factors such as the number of acupoints, control group measures, types of diseases, and diagnostic criteria (Table 2).
Supplementary Table S2 presents the results of leave-one-out analyses. When one study estimate was removed iteratively, the pooled effect estimates remained consistent, ranging from 1.31 (95% CI: 1.18–1.44) to 1.37 (95% CI: 1.22–1.55).
Five studies assessed changes in patient symptom scores after treatment, involving a total of 376 participants (34–38). The combined data showed significant heterogeneity (I2 = 93%), necessitating the use of a random effects model. The review indicated that auricular stimulation significantly reduced symptom scores in FGID patients (MD: -1.94; 95% CI: −3.06 to −0.85, p < 0.00001) (Figure 5). Due to the observed variability, sensitivity and subgroup analyses were performed to identify potential causes. Subgroup analysis revealed varied results among studies investigating unilateral auricular acupoint compression for ear selection (I2 = 86%), while other unspecified studies showed minimal or no between-group variation (I2 = 0%). Significant differences persisted across other subgroups. Thus, the observed heterogeneity may be attributed to inconsistent practices related to ear selection (Supplementary Table S3). Sensitivity analysis showed minimal impact on effect estimates after sequentially excluding individual studies (Supplementary Table S4).
Five studies examined changes in SAS scores among 296 participants post-treatment (33, 34, 37, 39, 43). With an I2 value of 49%, indicating low heterogeneity, a fixed-effect model was used. The combined results showed that AAT significantly decreased SAS scores in FGID patients (MD: -12.47; 95% CI: −13.92 to −11.01, p < 0.00001) (Figure 6A).
Six studies analyzed post-treatment changes in SDS scores, including 416 participants (28, 33, 35–37, 43). The heterogeneity test revealed an I2 value greater than 50% (I2 = 91%), leading to the use of a random effects model. The overall findings indicated that auricular point stimulation was significantly effective in reducing SDS scores (MD: -4.97; 95% CI: −9.23 to −0.72, p = 0.02) (Figure 6B). Subgroup analyses showed high heterogeneity, resulting in low confidence in the results (Supplementary Table S5). Sensitivity analysis confirmed minimal impact on overall outcomes after excluding individual studies (Supplementary Table S6).
Mild adverse reactions were reported in two studies. The results showed that MD = 2.98, 95% CI = (0.51, 17.26), I2 = 0, p = 0.009. A trial (28) reported 1/37 participant in the experimental group experienced mild local skin redness due to excessive pressure on the ear point. Another trial (36) reported adverse events in 7 participants: 6/200 in the experimental group experienced transient dizziness or localized tenderness, and 1/100 in the control group reported mild headache. All of which were classified as mild and were resolved within 24 h without intervention (Supplementary Figure S2).
The funnel plot for the meta-analysis of AAT for FGIDs showed symmetry. Quantitative assessment using the Egger and Begg tests revealed no significant evidence of publication bias (Egger: β [SE], 4.96 [3.47], p = 0.171) (Figure 7).
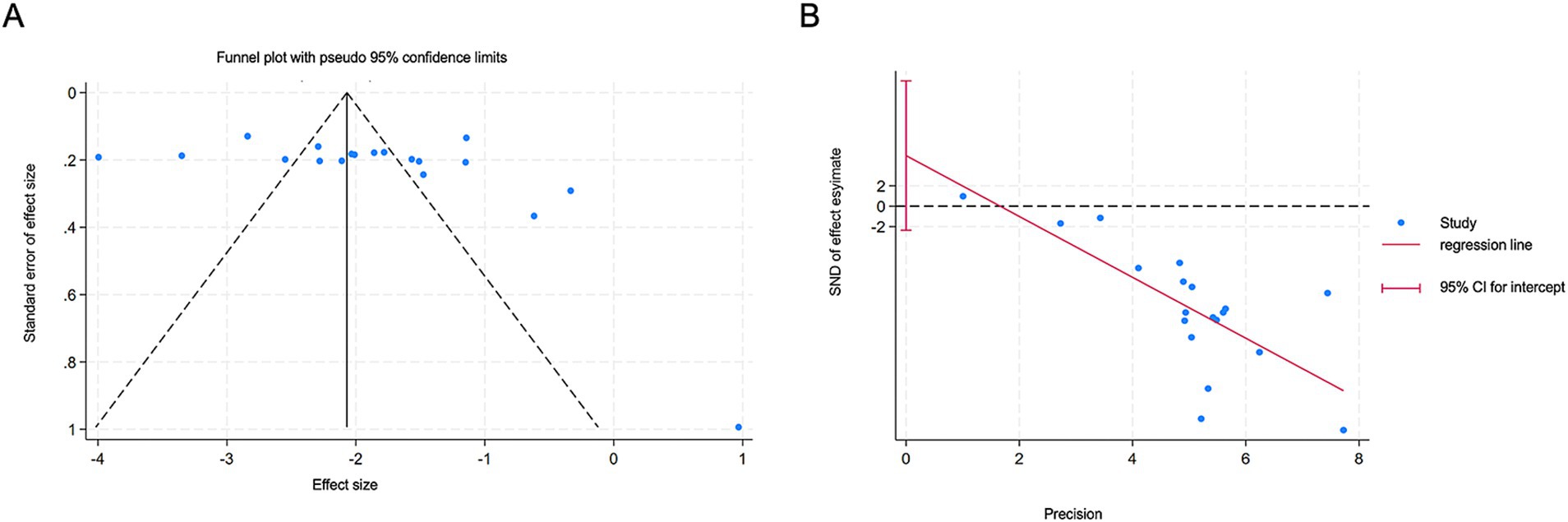
Figure 7. Publication bias on efficacy rate. (A) Funnel Plot of the efficacy rate. (B) Egger’s Regression Test Plot of efficacy rate.
The TSA analysis revealed that the optimal sample size was 992 cases. Results showed that after the fifth study was included (28), the cumulative Z value exceeded both the traditional threshold and the TSA threshold. This finding aligns with the meta-analysis results, indicating that the sample size included in the studies has reached and surpassed the optimal value. Thus, auricular stimulation therapy for FGIDs appears to be more effective than for the control group, providing robust evidence (Figure 8).
The quality of evidence for all outcome measures is assessed with GRADEpro. The quality of evidence for total effective rate was low, the quality of evidence for symptom scores was low, the quality of evidence for effectiveness was rated medium, and the quality of evidence for adverse reactions was low. The quality of evidence for SAS was rated medium, and the quality of evidence for SDS was low. The most common reason for reducing the quality of evidence is high heterogeneity and high risk of bias; this is followed by inconsistency (SoF details, see Supplementary Table S7).
To our knowledge, this systematic review is the first comprehensive analysis of AAT for FGIDs. The results suggest that AAT is effective in treating FGIDs, alleviating symptoms, and addressing anxiety and depressive states, although there is considerable statistical variability across studies. AAT can be used as a treatment option for the treatment of FGIDs.
Recently, increased attention has been given to auricular acupuncture therapy. Compared with traditional acupuncture, the use of disposable stainless steel needles will inevitably cause damage to the human body, AAT, as a non-invasive non-drug therapy, has been widely used to treat FGIDs (46). According to the “dose-effect” principle of acupuncture, higher doses of acupoint stimulation (higher frequency, multiple acupoints) are likely to produce better results (47). Consistent with this, subgroup analysis in our meta-analysis revealed significant clinical differences based on the number of acupoints used. It is worth noting that no significant difference was found in the effectiveness of interventions based on the type of AAT provider. Previous studies have utilized ear acupressure managed by nurses (48), and our results further support the effectiveness of interventions conducted by trained professionals, including nurses or collaborative teams of professionals and patients. Additionally, some studies have applied ear acupuncture to only one ear, suggesting that bilateral application might increase discomfort during treatment (49, 50). However, our study indicates that bilateral application is more common in treating FGIDs, potentially enhancing treatment intensity. Furthermore, the selection of acupoints varied across studies. Among the 19 RCTs analyzed, the most frequently used acupoints were the large intestine (n = 11), followed by the spleen (n = 8) and the liver (n = 7). The large intestine and liver acupoints are located in the cavum cymba conchae area, whereas the spleen acupoint is located in the cavum conchae area. These areas are commonly targeted in treatment. According to traditional Chinese medicine theory, the large intestine helps with bowel movements and diarrhea (51), while the spleen and liver regulate qi and blood, aiding in restoring organ function (52). Therefore, clinicians using AAT for FGIDs may consider bilateral application and the inclusion of these acupoints.
Different stimulation regimens may also contribute to the high heterogeneity observed in the results. Acupressure and electroacupuncture share the same stimulation mechanism-vagus nerve stimulation, but differences in nervous system activation and stimulation intensity, among others, may lead to different treatment mechanisms and effects (53). Acupressure may be more effective in promoting neurotransmitter release and mood improvement. It may be particularly appropriate for patients with mild disease who prefer noninvasive treatment. Electroacupuncture, on the other hand, may be more effective in modulating the autonomic nervous system and reducing stress, provides more consistent, controlled stimulation, and may be more appropriate for severe patients who require more intense stimulation (54). In addition, there are some differences in the efficacy of auriculotherapy for FD, IBS and FC. Our results showed that FD had the highest heterogeneity, suggesting that efficacy stability may be low, possibly due to large differences in electroacupuncture parameters (e.g., frequency, intensity) used in different studies, and differences in treatment response by different subtypes of FD (e.g., postprandial discomfort syndrome and epigastric pain syndrome) were not adequately stratified. However, IBS had the most consistent efficacy of AAT and a relatively stable improvement in anxiety and depression, which may be closely related to the central mechanism. The pooled effect size of FC was significant, but the heterogeneity was high, especially in the subgroup of “auricular number ≥ 5,” the effect was more stable, and the improvement in bowel motor function was more direct, but the effect on emotional symptoms was weaker. This may be due to the inconsistent definition of “effective” in different studies and the fact that this article includes a large number of older patients who have a weak response to treatment due to the natural decline in bowel motility.
At present, there are more and more researches on AAT in the treatment of FGIDs. Our results show that since Liu et al.’s research in 2011, with the gradual addition of new research, the 95% CI has gradually narrowed, the accuracy has improved, and the direction of evidence is consistent. Sensitivity analysis and TSA also show that the results of this meta-analysis are stable, which shows that the statistical evidence of the effectiveness of AAT in the treatment of FGIDs is sufficient. At the same time, our research results are basically consistent with other meta-analyses (55, 56). Although the differences in inclusion criteria and statistical methods may result in slightly different reported effects, they all confirm the efficacy of AAT in the treatment of FGIDs. At present, the treatment of FGIDs remains a major challenge. The existing drugs, such as prokinetic drugs and proton pump inhibitors, only treat one symptom at a time, and the combined use is easy to increase the side effects, so many problems such as long-term recurrent attacks and drug side effects have attracted more and more attention. Compared with drug therapy, AAT provides a non-invasive, safe, and minimal side-effect alternative therapy (57). Some non-drug therapies, such as cognitive behavioral therapy and hypnotherapy, have some effects, but their clinical efficacy and safety still need to be verified by large-scale clinical research (58). AAT is cost-effective, has low treatment costs, and has the potential for self-medication, reducing the need for frequent medical treatment. In conclusion, AAT is a promising alternative to medications and psychotherapy for the treatment of FGIDs, providing a safe, effective, and economical choice for clinicians and patients. Future research should further compare the efficacy, safety, and cost-effectiveness of AAT with other treatment methods to provide more comprehensive guidance for clinical practice.
However, the methodological limitations of the included studies, such as the lack of blinding and allocation concealment, high heterogeneity, and small sample sizes, resulted in most outcomes being rated as low or very low quality. These limitations restrict the generalizability of the findings to a broader population. Therefore, when interpreting the results of this study, these limitations should be taken into account, and further well-designed trials are needed to address these limitations and provide more robust and reliable evidence.
Recently, the brain-gut axis has received increasing attention. The brain-gut axis is a two-way communication system between the central nervous system and the gastrointestinal tract (59). Previous studies have shown that AAT (such as the auricular distribution area of the vagus nerve) can directly regulate autonomic nervous function by activating the vagus afferent pathway. Vagal activation can inhibit sympathetic excitability and increase parasympathetic tone, thereby reducing the systemic stress response (e.g., decreasing cortisol release) and improving visceral hypersensitivity and gastrointestinal motility disorders (60). In addition, AAT may also affect the release of neurotransmitters such as serotonin, dopamine, and norepinephrine. These neurotransmitters are involved in the regulation of mood (61). Our results suggest that AAT may not only improve symptoms in FGID patients, but also effectively alleviate anxiety and depression in FGID patients, and ear selection and intervention providers may be one of the reasons for the high heterogeneity. This study provides an evidence-based basis for non-drug intervention for the “brain-gut axis” theory. More studies combined with neuroimaging (such as fMRI) should be included in the future to explore the specific mechanism of AAT in the treatment of FGID from the perspective of the gut-brain axis.
This systematic review has several notable advantages. To our knowledge, this is the first comprehensive meta-analysis that systematically summarizes and analyzes the effectiveness of AAT for FGIDs. Unlike traditional meta-analyses, the cumulative meta-analysis approach provides insights into how the pooled estimates and their precision evolve over time as new studies are added. Additionally, we performed subgroup analyses to investigate the source of heterogeneity and validated the stability of the results through sensitivity and TSA.
However, our study has several limitations. First, although most studies used uniform criteria, a few employed different efficiency standards, which might have affected the meta-analysis results. Second, our search was limited to articles in English and Chinese, which may have introduced publication bias by excluding relevant studies in other languages. Third, inadequate adjustment for key confounding variables during subgroup analyses may affect the credibility of our conclusions. Finally, all included studies were conducted in developing countries, so the generalizability of the results requires further evaluation.
In conclusion, AAT shows promise as a complementary therapy for FGIDs, with preliminary evidence supporting its efficacy and safety. However, the clinical implications of these findings remain provisional due to the heterogeneity and methodological limitations of existing trials. Future large-scale, rigorously designed RCTs—with standardized protocols, robust blinding, and diverse populations—are essential to confirm these results and establish evidence-based guidelines.
The original contributions presented in this study are included in this article/Supplementary material, further inquiries can be directed to the corresponding authors.
M-YS: Conceptualization, Writing – original draft, Writing – review & editing. Z-JL: Writing – review & editing. S-HW: Writing – review & editing. Q-YL: Writing – review & editing. SL: Methodology, Writing – review & editing. D-DF: Writing – review & editing. D-DY: Writing – review & editing. C-JW: Writing – review & editing. Z-KY: Writing – review & editing. T-CL: Writing – review & editing. RZ: Writing – review & editing. J-NW: Conceptualization, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the National Natural Science Foundation of China (82174178) and Zhejiang Provincial Administration of Traditional Chinese Medicine Major Project (GZY-ZJ-KJ-23015).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Gen AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2025.1513272/full#supplementary-material
1. Sayuk, GS, and Gyawali, CP. Functional dyspepsia: diagnostic and therapeutic approaches. Drugs (2020) 80:1319–1336. doi: 10.1007/s40265-020-01362-4
2. Ford, AC, Sperber, AD, Corsetti, M, and Camilleri, M. Irritable bowel syndrome. Lancet. (2020) 396:1675–88. doi: 10.1016/S0140-6736(20)31548-8
3. Vriesman, MH, Koppen, IJN, Camilleri, M, Di Lorenzo, C, and Benninga, MA. Management of functional constipation in children and adults. Nat Rev Gastroenterol Hepatol. (2020) 17:21–39. doi: 10.1038/s41575-019-0222-y
4. Drossman, DA, and Hasler, WL. Rome IV-functional gi disorders: disorders of gut-brain interaction. Gastroenterology. (2016) 150:1257–61. doi: 10.1053/j.gastro.2016.03.035
5. Black, CJ, Drossman, DA, Talley, NJ, Ruddy, J, and Ford, AC. Functional gastrointestinal disorders: advances in understanding and management. Lancet. (2020) 396:1664–74. doi: 10.1016/S0140-6736(20)32115-2
6. Ford, AC, Mahadeva, S, Carbone, MF, Lacy, BE, and Talley, NJ. Functional dyspepsia. Lancet. (2020) 396:1689–702. doi: 10.1016/S0140-6736(20)30469-4
7. Barberio, B, Judge, C, Savarino, EV, and Ford, AC. Global prevalence of functional constipation according to the Rome criteria: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. (2021) 6:638–48. doi: 10.1016/S2468-1253(21)00111-4
8. Jaafari, H, Houghton, LA, West, RM, Agrawal, A, Aziz, I, Black, CJ, et al. The national prevalence of disorders of gut brain interaction in the United Kingdom in comparison to their worldwide prevalence: results from the Rome foundation global epidemiology study. Neurogastroenterol Motil. (2023) 35:e14574. doi: 10.1111/nmo.14574
9. Șchiopu, CG, Ștefănescu, C, Boloș, A, Diaconescu, S, Gilca-Blanariu, GE, and Ștefănescu, G. Functional gastrointestinal disorders with psychiatric symptoms: involvement of the microbiome-gut-brain Axis in the pathophysiology and case management. Microorganisms. (2022) 10:2199. doi: 10.3390/microorganisms10112199
10. Tack, J, Carbone, F, Chang, L, and Lacy, BE. Patient-reported outcomes in disorders of gut-brain interaction. Gastroenterology. (2024) 166:572–587.e1. doi: 10.1053/j.gastro.2023.11.307
11. Zhang, YQ, Jing, X, and Guyatt, G. Improving acupuncture research: progress, guidance, and future directions. BMJ. (2022) 376:o487. doi: 10.1136/bmj.o487
12. Allen, J, Mak, SS, Begashaw, M, Begashaw, M, Larkin, J, Miake-Lye, I, et al. Use of acupuncture for adult health conditions, 2013 to 2021: a systematic review. JAMA Netw Open. (2022) 5:e2243665. doi: 10.1001/jamanetworkopen.2022.43665
13. Zou, N, Zhou, Q, Zhang, Y, Xin, C, Wang, Y, Claire-Marie, R, et al. Transcutaneous auricular vagus nerve stimulation as a novel therapy connecting the central and peripheral systems: a review. Int J Surg. (2024) 110:4993–5006. doi: 10.1097/JS9.0000000000001592
14. Zhou, J, Li, S, Wang, Y, Lei, Y, Foreman, RD, Yin, J, et al. Effects and mechanisms of auricular electroacupuncture on gastric hypersensitivity in a rodent model of functional dyspepsia. PLoS One. (2017) 12:e0174568. doi: 10.1371/journal.pone.0174568
15. Wang, Y, Li, SY, Wang, D, Wu, MZ, He, JK, Zhang, JL, et al. Transcutaneous auricular Vagus nerve stimulation: from concept to application. Neurosci Bull. (2021) 37:853–62. doi: 10.1007/s12264-020-00619-y
16. Teckentrup, V, Neubert, S, Santiago, JCP, Hallschmid, M, Walter, M, and Kroemer, NB. Non-invasive stimulation of vagal afferents reduces gastric frequency. Brain Stimul. (2020) 13:470–3. doi: 10.1016/j.brs.2019.12.018
17. Guo, K, Lu, Y, Wang, X, Duan, Y, Li, H, Gao, F, et al. Multi-level exploration of auricular acupuncture: from traditional Chinese medicine theory to modern medical application. Front Neurosci. (2024) 18:1426618. doi: 10.3389/fnins.2024.1426618
18. Hou, PW, Hsu, HC, Lin, YW, Tang, NY, Cheng, CY, and Hsieh, CL. The history, mechanism, and clinical application of auricular therapy in traditional Chinese medicine. Evid Based Complement Alternat Med. (2015) 2015:495684:1–13. doi: 10.1155/2015/495684
19. de Oliveira Rodrigues, DM, Menezes, PR, Machado Ribeiro Silotto, AE, Heps, A, Pereira Sanches, NM, Schveitzer, MC, et al. Efficacy and safety of auricular acupuncture for depression: a randomized clinical trial. JAMA Netw Open. (2023) 6:e2345138. doi: 10.1001/jamanetworkopen.2023.45138
20. Qu, F, Zhang, D, Chen, LT, Wang, FF, Pan, JX, Zhu, YM, et al. Auricular acupressure reduces anxiety levels and improves outcomes of in vitro fertilization: a prospective, randomized and controlled study. Sci Rep. (2014) 4:5028. doi: 10.1038/srep05028
21. Higgins, JPT, Thomas, J, Chandler, J, Cumpston, M, Li, T, Page, MJ, et al. (Eds.), Cochrane handbook for systematic reviews of interventions version 6.4 (updated August 2023). (2011). Available at: https://training.cochrane.org/handbook (Accessed August, 2024).
22. Guyatt, GH, Oxman, AD, Kunz, R, Vist, GE, Falck-Ytter, Y, Schünemann, HJ, et al. Rating quality of evidence and strength of recommendations: what is "quality of evidence" and why is it important to clinicians? BMJ. (2008) 336:995–8. doi: 10.1136/bmj.39490.551019.BE
23. Higgins, JP, Thompson, SG, Deeks, JJ, and Altman, DG. Measuring inconsistency in meta-analyses. BMJ. (2003) 327:557–60. doi: 10.1136/bmj.327.7414.557
24. DerSimonian, R, and Laird, N. Meta-analysis in clinical trials. Control Clin Trials. (1986) 7:177–88. doi: 10.1016/0197-2456(86)90046-2
25. Mantel, N, and Haenszel, W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst. (1959) 22:719–48.
26. Egger, M, Davey Smith, G, Schneider, M, and Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ. (1997) 315:629–34. doi: 10.1136/bmj.315.7109.629
27. Wetterslev, J, Thorlund, K, Brok, J, and Gluud, C. Trial sequential analysis may establish when firm evidence is reached in cumulative meta-analysis. J Clin Epidemiol. (2008) 61:64–75. doi: 10.1016/j.jclinepi.2007.03.013
28. Ji, XP, Zhang, YL, Cai, JP, and Chen, FL. Effects of auricular application therapy on elderly functional constipation. Shanghai J Trad Chin Med. (2010) 44:85–7.
29. Wang, L. Nursing observation of auricular acupuncture point burying beans intervention for functional constipation in Alzheimer's disease. Shanxi. J Tradit Chin Med. (2019) 39:599–608.
30. Wang, D, Yang, J, Shi, Z, and Yang, DY. Observation on the efficacy of auricular point application in treating postprandial distress syndrome of functional dyspepsia with incoordination between liver and stomach. China J Trad Chin Med Pharm. (2018)
31. Huang, YJ, and Chen, JY. Therapeutic efficacy of auricular point sticking for diarrhea-predominant irritable bowel syndrome and its effect on serum 5-HT. Shanghai J Acup Moxibust. (2013)
32. Liu, S, Lin, F, and Wang, L. Clinical observation on the treatment of functional constipation by auricular pressure pills. Chin Gen Pract Nurs. (2011) 9:210–1. doi: 10.3969/j.issn.1674-4748.2011.03.015
33. Liu, X. Clinical observation on auricular acupressure treatment of functional constipation of liver Qi stagnation (in chinese) [master’s thesis]. Hubei, China: Hubei University of Chinese Medicine (2018).
34. Wang, XX, Liu, X, and Ke, MH. Clinical observation on auricular point sticking therapy for functional constipation combined with anxiety. Chin. Med. Mod. Distance Educ. China. (2020) 18:89–91. doi: 10.3969/j.issn.1672-2779.2020.10.036
35. Wu, D, Rong, PJ, Wang, HC, Wei, W, Hou, LW, Wang, Y, et al. Efficacy observation on 90 cases of functional dyspepsia treated by Electroacupuncture at concha Auriculae. World Chin Med. (2020)
36. Shi, X, Zhao, L, Luo, H, Deng, H, Wang, XP, Ren, G, et al. Transcutaneous auricular vagal nerve stimulation is effective for the treatment of functional dyspepsia: a multicenter, randomized controlled study. Am J Gastroenterol. (2024) 119:521–31. doi: 10.14309/ajg.0000000000002548
37. Zhou, S. Effect and central mechanism via MRI of transcutaneous auricular Vagus nerve stimulation on functional dyspepsia (in chinese) [master’s thesis]. Guangdong, China: Guangzhou University of Chinese Medicine (2021).
38. Wu, D, Peng, T, Ring, PJ, Lian, HH, Zhang, JL, and Wang, Y. Clinical effects of concha Auriculae Electroacupuncture in the treatment of diarrhea irritable bowel syndrome : World Chinese Medicine (2021)
39. Shi, X, Hu, Y, Zhang, B, Li, W, Chen, JD, and Liu, F. Ameliorating effects and mechanisms of transcutaneous auricular vagal nerve stimulation on abdominal pain and constipation. JCI Insight. (2021) 6:e150052. doi: 10.1172/jci.insight.150052
40. Wang, ZY, Wang, XX, Jiao, WN, and Hua, SW. Observation on therapeutic effect of auricular Acupoint pressing in the treatment of community elderly functional constipation. Guangming. J Chin Med. (2022)
41. Lin, Y, and Chen, XJ. Clinical observations on treatment of functional dyspepsia by auricular plaster therapy. Shanghai J Acup Moxibusti. (2007)
42. Kang, H, and Li, M. Auricular acupressure for the treatment of irritable bowel syndrome in 54 cases. J Emerg Trad Chin Med. (2006)
43. Han, Z, Ren, L, Ruan, C, Shan, CQ, and Hu, YJ. Clinical observation on 40 cases of diarrhoea-type irritable bowel syndrome treated by auricular pressure-patching. Zhejiang. J Tradit Chin Med. (2018)
44. Huang, X. Clinical observation on the treatment of functional constipation by auricular pressure bean. Chin Sci Technol J Database. (2021)
45. Zhou, A, Liang, M, and Wei, Y. Application of auricular acupressure technique in patients with constipation. Electr J Pract Clin Nurs Sci. (2020)
46. Karst, M, and Li, C. Acupuncture-a question of culture. JAMA Netw Open. (2019) 2:e1916929. doi: 10.1001/jamanetworkopen.2019.16929
47. Bauer, M, McDonald, JL, and Saunders, N. Is acupuncture dose dependent? Ramifications of acupuncture treatment dose within clinical practice and trials. Integr Med Res. (2020) 9:21–7. doi: 10.1016/j.imr.2020.01.003
48. Chang, LH, Hsu, CH, Jong, GP, Ho, S, Tsay, SL, and Lin, KC. Auricular acupressure for managing postoperative pain and knee motion in patients with total knee replacement: a randomized sham control study. Evid Based Complement Alternat Med. (2012) 2012:528452. doi: 10.1155/2012/528452
49. Cha, NH, and Sok, SR. Effects of auricular acupressure therapy on primary dysmenorrhea for female high school students in South Korea. J Nurs Scholarsh. (2016) 48:508–16. doi: 10.1111/jnu.12238
50. Yeh, ML, Hung, YL, Chen, HH, and Wang, YJ. Auricular acupressure for pain relief in adolescents with dysmenorrhea: a placebo-controlled study. J Altern Complement Med. (2013) 19:313–8. doi: 10.1089/acm.2011.0665
51. Go, GY, and Park, H. Effects of auricular acupressure on women with irritable bowel syndrome. Gastroenterol Nurs. (2020) 43:E24–34. doi: 10.1097/SGA.0000000000000332
52. Aminizadeh, M, Tirgari, B, Rashtabadi, OR, Jahani, Y, and Tajadini, H. Effectiveness of auricular acupressure on constipation and related quality of life among the older people in the residential care home: a randomized clinical trial. BMC Geriatr. (2023) 23:171. doi: 10.1186/s12877-023-03881-7
53. Liu, S, Wang, Z, Su, Y, Qi, L, Yang, W, Fu, M, et al. A neuroanatomical basis for electroacupuncture to drive the vagal-adrenal axis. Nature. (2021) 598:641–5. doi: 10.1038/s41586-021-04001-4
54. Zhang, L, Yu, C, Chen, B, Chao, Y, Zhang, H, Zhao, Q, et al. Modulation of colonic function in irritable bowel syndrome rats by electroacupuncture at ST25 and the neurobiological links between ST25 and the colon. Front Neurosci. (2022) 16:930489. doi: 10.3389/fnins.2022.930489
55. Hu, Y, Cheng, X, Su, X, and Fu, Y. Auricular therapy improves gastrointestinal function in patients with gynecological laparoscopic surgery: a protocol for systematic review and meta analysis. Medicine (Baltimore). (2020) 99:e23421. doi: 10.1097/MD.0000000000023421
56. Xiong, J, Zhou, X, Luo, X, Gong, X, Jiang, L, Luo, Q, et al. Acupuncture therapy on myofascial pain syndrome: a systematic review and meta-analysis. Front Neurol. (2024) 15:1374542. doi: 10.3389/fneur.2024.1374542
57. Jin, R, Wang, X, Lv, Y, Xu, G, Yang, C, Guo, Y, et al. The efficacy and safety of auricular point combined with moxibustion for insomnia: a protocol for systematic review and meta-analysis. Medicine. (2020) 99:e22107. doi: 10.1097/MD.0000000000022107
58. Zhou, X, Zhang, J, Jiang, L, Zhang, S, Gu, Y, Tang, J, et al. Therapeutic efficacy of acupuncture point stimulation for stomach cancer pain: a systematic review and meta-analysis. Front Neurol. (2024) 15:1334657. doi: 10.3389/fneur.2024.1334657
59. Agirman, G, Yu, KB, and Hsiao, EY. Signaling inflammation across the gut-brain axis. Science. (2021) 374:1087–92. doi: 10.1126/science.abi6087
60. Li, H, He, T, Xu, Q, Li, Z, Liu, Y, Li, F, et al. Acupuncture and regulation of gastrointestinal function. World J Gastroenterol. (2015) 21:8304–13. doi: 10.3748/wjg.v21.i27.8304
Keywords: auricular acupoint therapy, functional gastrointestinal disorders, systematic review, meta-analysis, randomized controlled trials
Citation: Shen M-Y, Li Z-J, Wang S-H, Lin T-C, Lou Q-Y, Liu S, Feng D-D, Yang D-D, Wang C-J, Ying Z-K, Zhou R and Wu J-N (2025) Auricular acupoint therapy for functional gastrointestinal disorders: a systematic review and meta-analysis of randomized clinical trials. Front. Med. 12:1513272. doi: 10.3389/fmed.2025.1513272
Received: 18 October 2024; Accepted: 21 February 2025;
Published: 19 March 2025.
Edited by:
Pathirage Kamal Perera, University of Colombo, Sri LankaReviewed by:
Guanhu Yang, Ohio University, United StatesCopyright © 2025 Shen, Li, Wang, Lin, Lou, Liu, Feng, Yang, Wang, Ying, Zhou and Wu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jian-Nong Wu, d2VpeWlkaWFuZGlhbkB6Y211LmVkdS5jbg==
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.