
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Med. , 03 February 2025
Sec. Gastroenterology
Volume 12 - 2025 | https://doi.org/10.3389/fmed.2025.1484656
This article is part of the Research Topic Community Series in Gut Microbiota and Immunity in Health and Disease: Dysbiosis and Eubiosis's Effects on the Human Body, Volume II View all articles
Background: Akkermansia muciniphila, as a probiotic, is negatively linked to IBD, obesity, and T2DM. The aim of this study was to comprehensively assess the research status of Akkermansia muciniphila over the past decade and explore the relationships between this bacterium and various health-related aspects.
Methods: Tools VOSviewer, Bibliometrix, and CiteSpace were used to analyze various aspects including publication metrics, contributors, institutions, geography, journals, funding, and keywords.
Results: Over the past decade, research on Akkermansia muciniphila has demonstrated a consistent annual growth in the number of publications, with a notable peak in 2021. China led in the number of publications, totaling 151, whereas the United States exhibited a higher centrality value. Among the 820 institutions involved in the research, the University of California (from the United States) and the Chinese Academy of Sciences (from China) occupied central positions. Willem M. De Vos ranked at the top, with 12 publications and 1,108 citations. The journal GUT, which had 5,125 citations and an Impact Factor of 23.0 in 2024, was the most highly cited. The most cited articles deepened the understanding of the bacterium’s impact on human health, spanning from basic research to translational medicine. Thirty-nine high-frequency keywords were grouped into five clusters, illustrating Akkermansia muciniphila’s associations with metabolic diseases, chronic kidney disease, the gut-brain axis, intestinal inflammation, and Bacteroidetes-Firmicutes shifts.
Conclusion: Given Akkermansia muciniphila’s anti-inflammatory and gut-barrier-strengthening properties, it holds promise as a therapeutic for obesity, metabolic disorders, and inflammatory conditions. Therefore, future research should explore its potential further by conducting clinical trials, elucidating its mechanisms of action, and investigating its efficacy and safety in diverse patient populations.
In the intricate landscape of the gut microbiome, Akkermansia muciniphila stands out as a remarkable microorganism. Native to the gastrointestinal tracts of both humans and various animal species, it is celebrated for its distinctive ability to degrade mucins (1, 2). This bacterium was first identified by Derrien et al. (3) when they successfully cultured it using pure mucin as the sole carbon substrate, a milestone discovery that significantly advanced gut microbiome research. Akkermansia muciniphila is intricately involved in multiple aspects of maintaining gut health from early life. Appearing across different stages of human development, it serves as an important indicator of the vitality and biodiversity of the infant gut microbiota. Its abundance steadily increases from the neonatal period to adulthood (4).
Functionally, Akkermansia muciniphila plays a fundamental role in several key physiological processes within the gut. It is crucial for enhancing intestinal mucus secretion, maintaining the balance of mucosal viscosity, and safeguarding the integrity of the intestinal epithelial barrier (5–7). By degrading mucosal components, Akkermansia muciniphila generates short-chain fatty acids. These fatty acids play a dual role: they effectively reduce inflammatory reactions and prevent an increase in intestinal permeability (8, 9). Additionally, research has shown that it can increase the thickness of the intestinal lining, strengthen barrier functions, and promote the growth of beneficial microbial communities (10, 11). Maintaining gut homeostasis is of utmost importance as disruptions in intestinal physiology are closely linked to the development of various health issues, including metabolic syndromes, immunological disorders, infections, and neoplasias (12, 13). In this context, Akkermansia muciniphila emerges as a key player in preserving gut homeostasis.
Bibliometrics is a discipline that utilizes lexical analysis, citation analysis, and co-occurrence analysis to quantify document attributes and associated processes. Bibliometric analyses can yield valuable insights for guiding future research directions. The aim of this study is to conduct a bibliometric analysis of Akkermansia muciniphila bacteria and its relationship with health from 2013 to 2024, with the goal of identifying research hotspots and key topics for researchers in this field, and providing novel insights and research directions for future investigations.
The Web of Science database was selected as the primary source for this study due to its extensive coverage of over 12,000 academic articles and its regular use by scholars. Compared to other databases such as Scopus, Medline, and PubMed, Web of Science provides the most comprehensive and reliable bibliometric analysis (6). On December 12, 2024, all versions of the database were utilized to search for and export relevant articles from the Web of Science (WoS) Core Collection. The complete records and cited references were then retrieved from the relevant publications and saved in plain text format for future analysis. By applying the following search parameters: disease (search within topic), article (document types), and TI = (“Akkermansia muciniphila”) OR AB = (“Akkermansia muciniphila”), 937 articles were obtained from the WOS database. A plain-text file was exported with all the full records and cited references for further analysis (Figure 1).
We considered all retrieved articles to characterize the different subject areas and document types of Akkermansia literature. For determining the share of each study design, targeted population, and conditions/diseases, we focused on original articles. To assess other bibliometric characteristics, we considered both original articles and reviews, including the year distribution of publications, citation numbers, most productive journals, institutions, sponsors, authors, and countries. Additionally, we examined the collaboration between keywords, terms, authors, and countries.
Subsequently, after employing clustering software to group the keywords into five distinct categories, a meticulous process of literature selection was carried out. Specifically, we sifted through a vast body of literature and ultimately identified 101 relevant documents associated with these categories. These selected 101 papers were then subjected to in-depth discussion and analysis with respect to Akkermansia muciniphila. Each of these literatures was carefully examined to extract valuable insights regarding various aspects of Akkermansia muciniphila, such as its physiological functions and potential applications in different fields. During the discussion phase, we compared and contrasted the findings presented in these papers, aiming to identify commonalities, discrepancies, and emerging trends. This comprehensive analysis enabled us to gain a more profound understanding of Akkermansia muciniphila within the context defined by the five keyword categories.
Deduplication of the obtained data was performed using the CiteSpace (6.1.R1) (14). Two researchers independently extracted the publications, countries, institutions, funding agencies, authors, journals, citations, keywords, and highly cited references. To ensure data accuracy and reliability, discrepancies were reconciled via discussions and negotiations. Microsoft Excel was utilized to open the raw data in.xlxs format. The number of publications, grant money, and journals issued were then statistically compiled and analyzed. Bibliometric programs CiteSpace and VOSviewer (1.6.18) (15), run in a Java language environment, were employed to visualize the keywords, authors, and cited frequency of the literature, countries, issuing journals, issuing institutions, and issuing authors of the raw data from WOS in accordance with the corresponding format. BICOMB (2.02) (16), a tool for biomedical text mining and visualization, was used to extract and visualize medical-related data, such as gene-disease relationships and drug-disease associations, by importing Web of Science data. Cluster analysis was performed using the gCLUTO (1.0) (17) software’s biclustering algorithm, and 3D hill clustering graphs were generated by the multidimensional scaling method.
As of our search date in July 2024, the number of articles published in the field for that year may not be complete. Nevertheless, over the past decade, there has been a consistent annual increase in the number of articles published in the field, with a remarkable surge in 2021 (Figure 2A). Furthermore, Figure 2B illustrates a comparable trend in the predominance of the top 10 countries in the field.
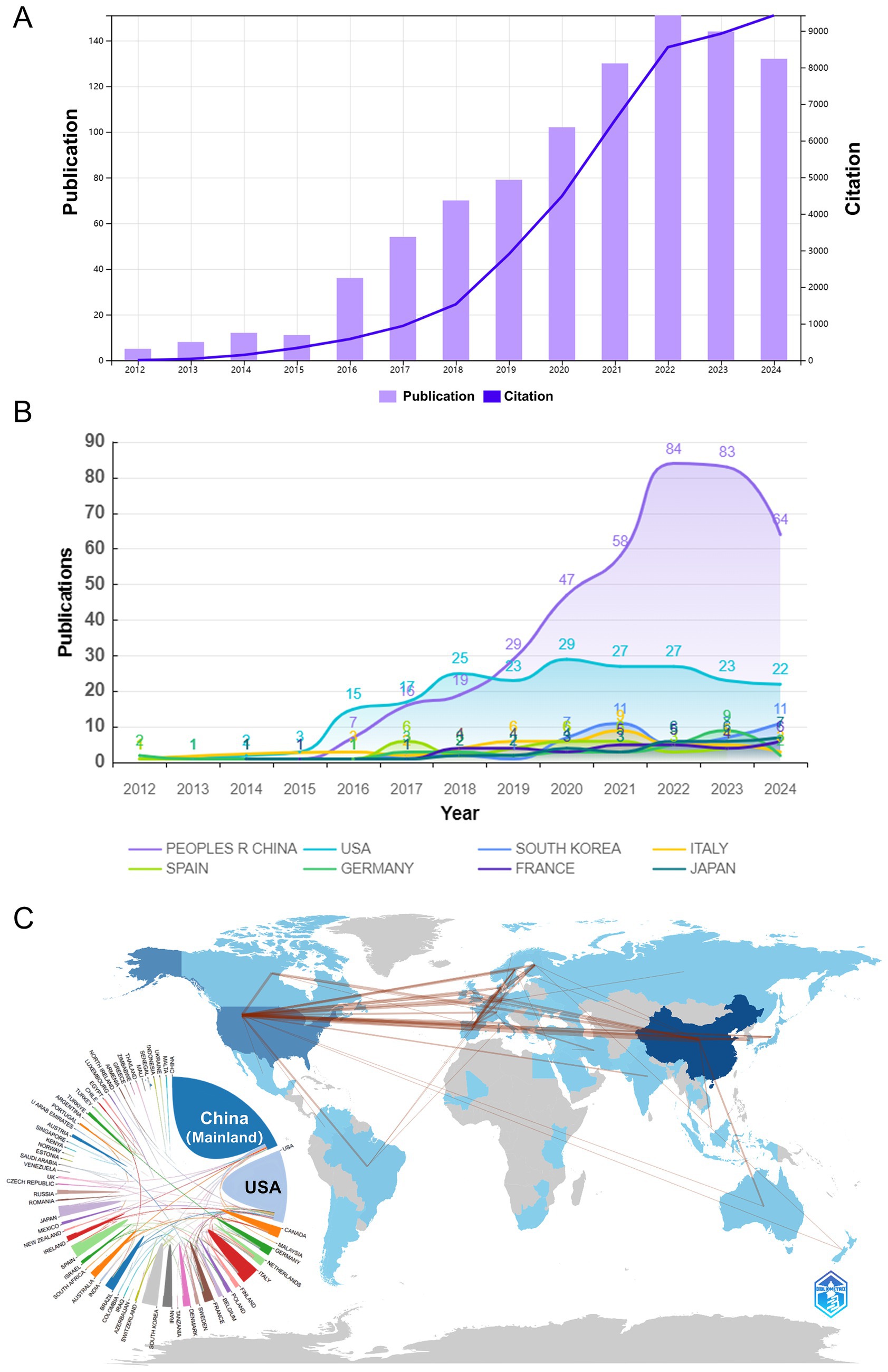
Figure 2. The publication trend of Akkermansia muciniphila. (A) Graph showing the number of publications per year over the past decade. (B) Chart displaying the number of publications issued by the top 10 countries from 2012 to 2024. (C) Map illustrating the cooperation of countries in the field of Akkermansia muciniphila from 2012 to 2024, along with the national and regional geographic distribution of Akkermansia muciniphila.
In the last decade, research on Akkermansia muciniphila bacteria has been conducted by 52 countries and regions (Figure 2C). China has contributed the most with 151 publications, followed by the United States with 102 publications. The United States (USA) has a higher centrality value of 0.89, while China has a centrality value of 0.04.
When analyzing the network hotspots of research institutions (Figure 3), a total of 820 institutions were found to be involved in this field, with 176 links. In terms of centrality, the University of California, Harvard University (both in the USA), and the Chinese Academy of Sciences, Zhejiang University, Jiangnan University, Peking Union Medical College, and Shanghai Jiao Tong University (all in China) have greater centrality values. The majority of the other universities in the top 10 are also from the USA and China. These institutions collaborate to varying degrees within their respective countries as a single unit, as well as with institutions in other countries and regions.
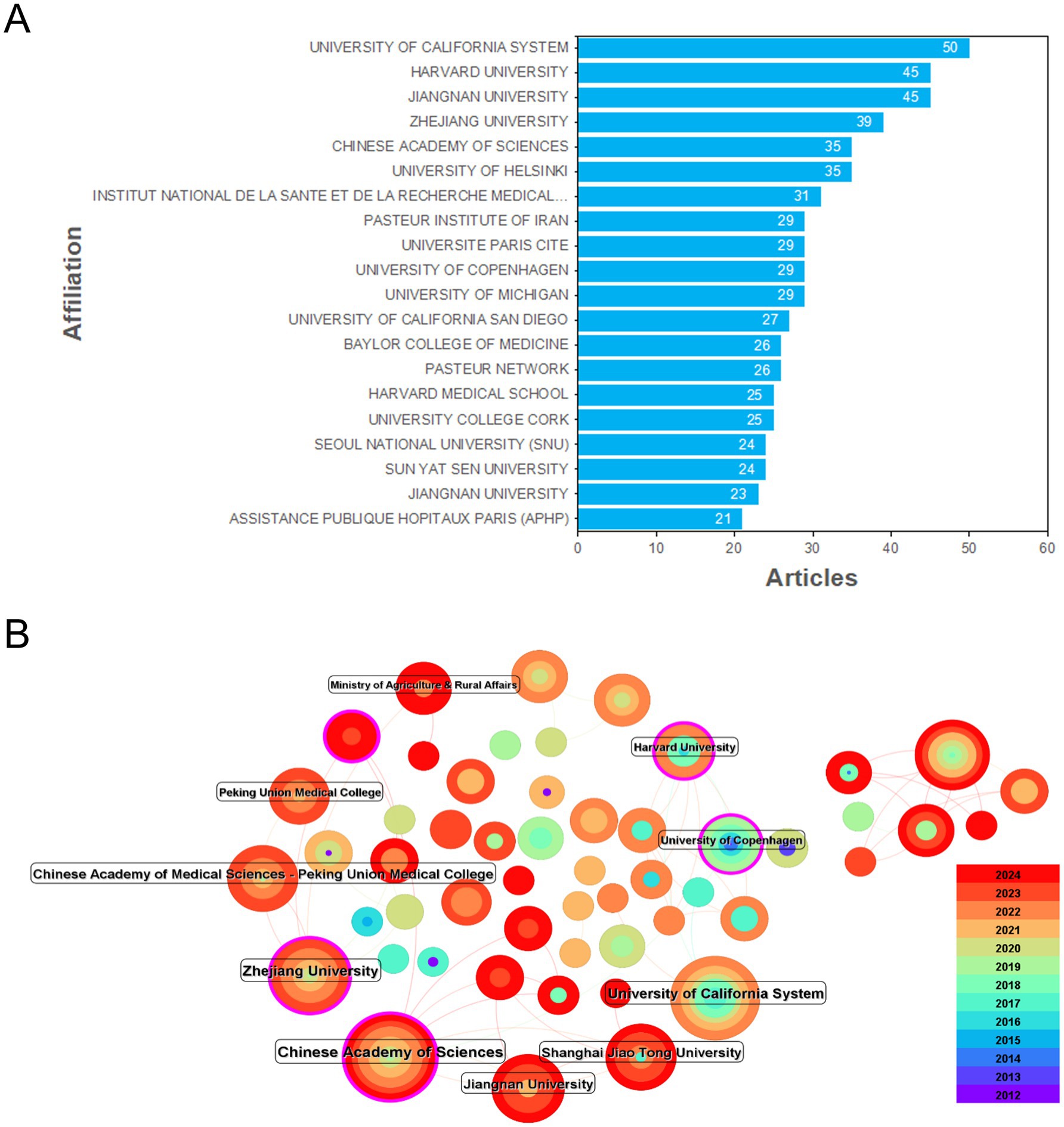
Figure 3. Information on institutional publications and collaborations. (A) The publication status of papers by various institutions. (B) Analysis of cooperation between institutions.
Within the field focusing on the microbiota species Akkermansia muciniphila, our bibliometric assessment identifies a quintet of leading contributors whose works significantly shape the academic discourse (Table 1). Willem M. De Vos leads the ranking with a noteworthy publication count of 12 articles, garnering a cumulative citation tally of 1,108, which showcases his profound impact on Akkermansia muciniphila research. Hao Zhang closely follows with 11 contributions, having amassed 178 citations, reflecting a solid presence in the scientific community dedicated to studying this bacterial strain. Seyed Davar Siadat, also with 11 publications, secures his position with a total of 280 citations, substantially contributing to the knowledge base around Akkermansia muciniphila. Wei Chen, credited with 10 papers, has accumulated 158 citations, evidencing his active engagement in the exploration of this microbial area. Patrice D. Cani, despite sharing the same number of published articles as Chen, stands out with 2,693 citations, indicative of the high regard and broad influence his work receives in the scientific community. These authors collectively represent the forefront of Akkermansia muciniphila-centered investigations, and their combined efforts have substantially enriched the current body of knowledge and influenced future directions in this specialized microbiological domain.
In identifying the most influential journals dedicated to disseminating Akkermansia muciniphila studies over the past decade, a comprehensive analysis reveals a roster of prestigious publications that have significantly contributed to advancing the scientific discourse surrounding this beneficial bacterium (Table 2). GUT stands as the leader in citations with a remarkable 5,125 tallies, holding an Impact Factor (2024) of 23.0, and is categorically placed within Gastroenterology & Hepatology. Its prominence reflects its role as a pivotal venue for cutting-edge research on the human gut ecosystem. Scientific Reports, with 2,106 citations, features an Impact Factor of 3.8 and spans Multidisciplinary Sciences, indicating its broad scope and interdisciplinary appeal in Akkermansia muciniphila studies. Frontiers in Microbiology, securing third place, has amassed 1,996 citations and carries an Impact Factor of 4.0, signaling its specialization in Microbiology and underscoring its dedication to bacterial and microbial sciences relevant to Akkermansia muciniphila. PLOS One, recognized for its focus on Multidisciplinary Sciences, has garnered 1,896 citations and an Impact Factor of 2.9 (2024), contributing to the rich tapestry of knowledge on the microbiome and health interrelations. Frontiers in Cellular and Infection Microbiology, positioned fifth, gathers 829 citations with an Impact Factor of 4.6, concentrating on Microbiology and reinforcing its stance as a prime forum for cellular and infectious diseases studies. These findings delineate the impact and breadth of Akkermansia muciniphila research across various scientific domains, showcasing how these top journals serve as epicenters for knowledge production and dissemination in this vibrant field.
In mapping the intellectual landscape of Akkermansia muciniphila literature over the past decade, a review of the most cited articles reveals pivotal works that have significantly enriched the field through innovative perspectives and empirical evidence (Table 3). These studies stand as beacons in their respective areas, shaping theoretical frameworks and practical approaches toward understanding the multifaceted impacts of this bacterial genus on human health. These articles form the vanguard of Akkermansia muciniphila research, not only by virtue of their citation numbers but also due to their transformative impact on the discourse. Their collective influence spans diverse areas of inquiry, from basic biology to translational medicine, underlining the versatility and significance of Akkermansia muciniphila in human health and disease.
The term “gut microbiota” was the most frequently occurring term (179 occurrences), while “inflammation,” “obesity,” and “insulin resistance” also appeared multiple times (Figure 4A). To conduct biclustering analysis, high-frequency keywords with a frequency greater than or equal to 5 were selected (Figure 4B). A total of 39 eligible high-frequency keywords were identified and divided into five groups to construct a visualization matrix of high-frequency keywords and source literature. Cluster 0 suggests Akkermansia muciniphila has a relationship with metabolic diseases such as the gut-liver axis and NAFLD. Cluster 1 indicates a relationship between Akkermansia muciniphila bacteria and chronic kidney disease, insulin resistance, obesity, diabetes, metabolic syndrome, lipid metabolism, and a high-fat diet. Cluster 2 suggests a relationship with the gut-brain axis, Parkinson’s disease, and Alzheimer’s disease. Cluster 3 indicates a relationship between Akkermansia muciniphila bacteria and intestinal inflammation. Cluster 4 suggests a relationship with alterations in Bacteroidetes and Firmicutes. These findings provide valuable insights into the potential roles of Akkermansia muciniphila bacteria in various physiological processes and diseases.
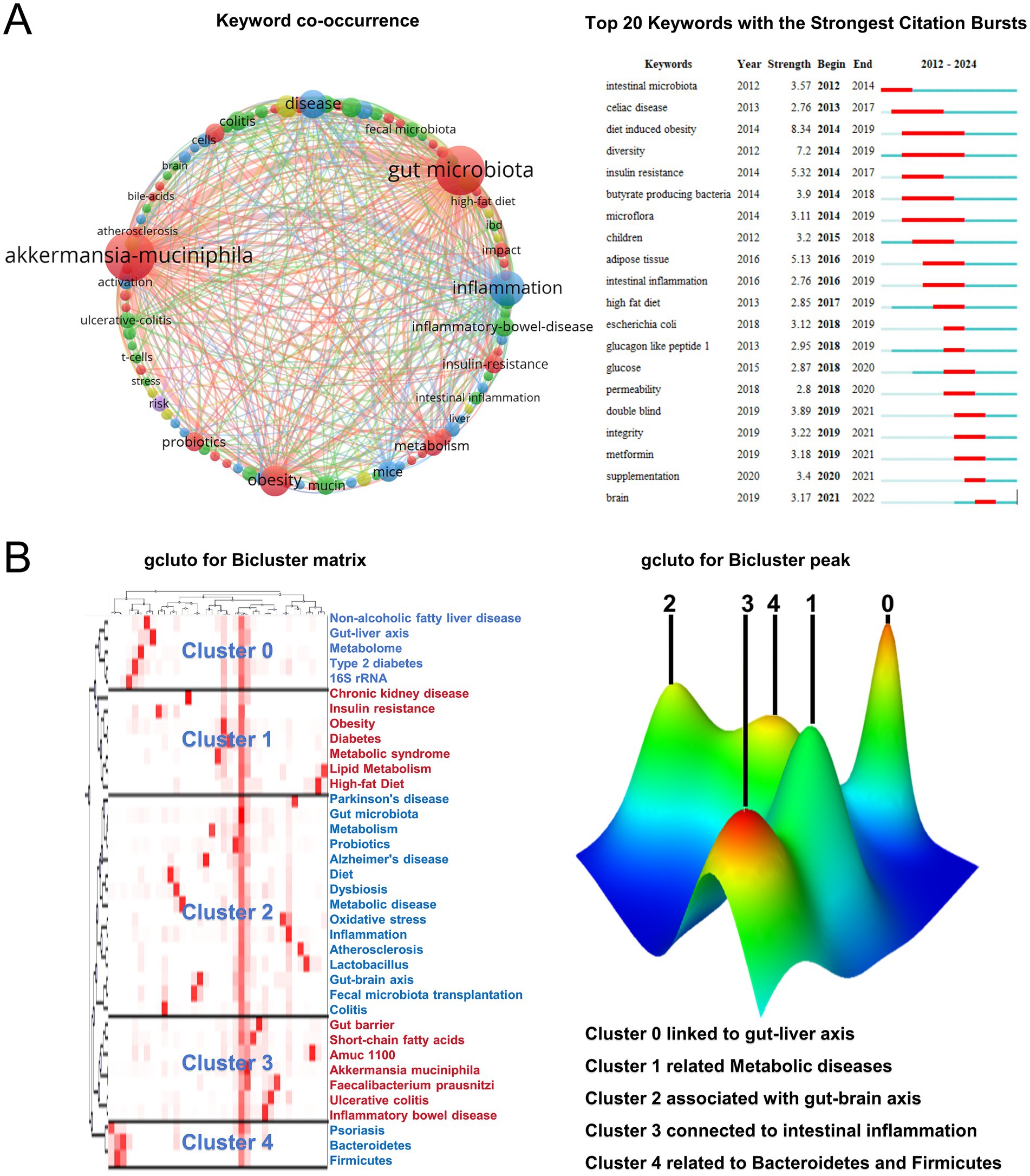
Figure 4. Keyword analysis. (A) Keyword co-occurrence diagram and the top 20 most cited keywords. (B) Bicluster matrix visualization of 31 high-frequency keywords and source articles, along with bicluster peak visualization of the same keywords and articles.
Research on Akkermansia muciniphila has indeed witnessed rapid advancements. As documented in a bibliometric analysis from 2020, obesity (71 publications) and type 2 diabetes (39 publications) emerged as focal points of interest. More recently, in a 2022 bibliometric review, recurrent keywords highlighted the themes of oxidative stress, diet, metformin, fecal microbiota transplantation, short-chain fatty acids, polyphenols, and bacterial metabolites, underscoring multifaceted avenues of investigation. Our current study advances this body of knowledge by synthesizing subsequent developments and employing cluster analysis to elucidate connections. Specifically, we uncover Akkermansia muciniphila’s association with intestinal mucosal barrier function and its link to gut inflammation. Moreover, our findings reveal its relationship with inflammatory bowel disease, diabetes, obesity, non-alcoholic fatty liver disease and other metabolic disorders. Notably, Akkermansia muciniphila demonstrates intriguing ties to the gut-brain axis and neurological conditions, including Alzheimer’s disease, broadening its potential impact on health beyond traditional realms of metabolic syndromes. This comprehensive exploration underscores the evolving landscape of Akkermansia muciniphila’s physiological roles, illuminating its potential influences on both gut-centric and systemic health phenomena.
Within the scope of this study, our analytical framework hinged primarily on the Web of Science (WoS) database, where we meticulously evaluated research spanning from 2012 to 2024. The findings before 2022 echoed those delineated in previous bibliometric analyses, affirming the continuity and evolution of research trajectories within the field (18, 19). We complemented this core dataset with exhaustive examinations drawn from CNKI (a Chinese database), Scopus, and PubMed. This endeavor yielded 177 publications identified in CNKI, 897 in Scopus, and 605 in PubMed. Acknowledging the intrinsic variances across different data sources that may introduce systematic variations, dedicated analyses were conducted separately for each repository, as illustrated in Supplementary Figure S1. Across these databases, a remarkable coherence was observed, notably between Chinese and English repositories, indicating harmonized trends in topical directions. Despite navigational complexities posed by the multiplicity of databases, the consistent outcomes attest to the resilience and integrity of our discoveries.
High-frequency word analysis reveals that Akkermansia muciniphila influences the structure of the gut microbiota. Studies have shown that its abundance is often associated with a healthier gut environment, characterized by a diverse and balanced microbiota (20, 21). This bacterium’s role in maintaining gut barrier integrity and its anti-inflammatory properties contribute to the stabilization of the gut microbiome (22, 23). Research indicates that Akkermansia muciniphila can promote the growth of beneficial bacteria such as Bifidobacterium and Lactobacillus (24), while also influencing the levels of Faecalibacterium prausnitzii, another key species in gut health (25, 26). Moreover, Akkermansia muciniphila’s influence on the gut microbiome extends beyond direct effects on bacterial populations; it also affects the gut’s physical niche, such as mucin production, which can alter microbial community composition and functionality (27, 28). Therefore, Akkermansia muciniphila plays a critical role in shaping the gut microbiota’s diversity and structure of the gut microbiota, contributing to a balanced and healthy gut environment.
Furthermore, our analysis highlighted that Akkermansia muciniphila has an impact on gut microbiota metabolites. The metabolic activities of Akkermansia muciniphila not only directly affect the gut environment but also have indirect effects on the host’s systemic metabolism (29). Akkermansia muciniphila’s metabolism of mucin results in the production of short-chain fatty acids, particularly butyrate, which is a critical nutrient for colonic epithelial cells, promoting gut integrity and reducing inflammation (29–31). Through cross-feeding mechanisms, Akkermansia muciniphila influences the metabolism of other gut microbiota members, shaping the overall gut microbiome and metabolome (32). Akkermansia muciniphila can modulate tryptophan metabolism, an essential pathway with implications for gut health and beyond (33). The presence of Akkermansia muciniphila influences tryptophan derivatives, including indoleamine 2,3-dioxygenase 1 (IDO1) products, affecting gut-brain signaling and neuroprotection (34, 35). Akkermansia muciniphila’s impact extends to the host’s bile acid pool, where it can modify bile acid profiles, influencing host lipid and glucose metabolism (36, 37). These modifications in bile acid metabolism can help in the prevention of metabolic disorders, such as obesity and type 2 diabetes, by regulating the host’s energy homeostasis. Additionally, Akkermansia muciniphila’s influence on the production of gamma-aminobutyric acid (GABA), a neurotransmitter crucial for brain function, highlights its role in the gut-brain axis (31, 38). The modulation of GABA and other neurotransmitters by Akkermansia muciniphila may have implications for neurological conditions, such as depression and anxiety, suggesting potential therapeutic avenues (38, 39). Therefore, the metabolic activities of Akkermansia muciniphila play a crucial role in shaping the gut microbiome’s metabolic landscape by influencing short-chain fatty acids production, tryptophan metabolism, bile acid profiles, neurotransmitter levels, and other metabolic pathways.
Akkermansia muciniphila, particularly through its outer membrane protein Amuc_1100, has emerged as a versatile therapeutic agent with broad potential in promoting health and managing diseases (40). Amuc_1100 has demonstrated promise in stimulating GLP-1 secretion, improving metabolic health, and attenuating intestinal mucositis, thereby demonstrating potential in treating diabetes and chemotherapy-induced complications (41). Amuc_1100 has also been found to alleviate depression-like behavior in mice, suggesting benefits for mental health (42–45). Furthermore, its role in modulating CD8+ T cells to mitigate colitis-associated tumorigenesis and enhance immunotherapy efficacy highlights their potential in cancer treatment (46–48). Surface-displayed Amuc_1100 on lactic acid bacteria has shown potential in improving hepatic steatosis and gut health, offering a novel delivery platform (49). With its ability to regulate immune responses, gut barrier function, and metabolism (50–52), Akkermansia muciniphila and Amuc_1100 are promising candidates for novel interventions in a range of diseases, from metabolic disorders to neuropsychiatric conditions.
Akkermansia muciniphila plays a crucial role in maintaining the integrity of the gut barrier. Recent studies have highlighted its influence on barrier function through various mechanisms. In alcoholic steatohepatitis, conventional type 1 dendritic cells preserve gut barrier integrity by sustaining populations of Akkermansia muciniphila (53). Akkermansia muciniphila upregulates genes critical for the intestinal barrier via activation of the ALPK1/TIFA pathway, which is dependent on ADP-heptose (54). Binding by intelectin-1 alters the localization of Akkermansia muciniphila, impacting mucus barrier modification (55). Grape polyphenols have been shown to positively influence Akkermansia muciniphila and enhance the gut barrier (56). Pasteurized Akkermansia muciniphila mitigates LPS-induced intestinal barrier dysfunction by modulating AMPK and NF-κB through TLR2 (57). Andrographolide strengthens intestinal barrier function and boosts the abundance of Akkermansia muciniphila, exerting antihyperglycemic effects (58). Akkermansia muciniphila colonization alleviates jejunal mucosal barrier disruption caused by high fructose and stress (59). These findings underscore the significant contribution of Akkermansia muciniphila to gut barrier maintenance and its potential therapeutic value in gut-related disorders.
Mucin, a critical component of the gut’s protective mucus layer, plays a central role in the ecology and function of Akkermansia muciniphila. Recent studies have elucidated how Akkermansia muciniphila’s specialized mucin degradation enzymes enable it to colonize and thrive within this niche (60–63). Binding specificity to O-glycans on mucin is key to its adhesion and utilization (64). The bacterium’s adaptation to high mucin environments, through energy homeostasis regulation, showcases its metabolic flexibility (65). Berberine indirectly promotes Akkermansia muciniphila growth by stimulating gut mucin secretion, highlighting the importance of diet-microbiome interactions (66). A deficiency in Akkermansia muciniphila and mucin depletion are linked to intestinal barrier dysfunction and inflammation (67), while excessive mucin consumption can lead to barrier damage (10). Mucin acts as a functional niche, significantly impacting gut microbiota composition and functionality, and its depletion can have detrimental effects beyond mere Akkermansia muciniphila supplementation (28). These findings underscore the pivotal role of mucin in gut health and highlight Akkermansia muciniphila’s potential as a therapeutic target for gut-related disorders.
Akkermansia muciniphila has been identified as a significant contributor to the modulation of the host’s immune response. This Gram-negative anaerobe is known to interact with the host’s mucus layer, promoting gut barrier integrity and influencing the balance of the gut microbiota. Recent studies have highlighted Akkermansia muciniphila’s ability to reduce inflammation in various disease models. For instance, it has been demonstrated to decrease Porphyromonas gingivalis-induced inflammation and periodontal bone destruction, suggesting a protective role in periodontal disease (68). Furthermore, the tripeptide derived from Akkermansia muciniphila has been reported to mitigate sepsis, inflammation, and mortality, highlighting its broad anti-inflammatory effects (69).
Akkermansia muciniphila, a beneficial gut bacterium, exerts its effects on immune homeostasis and inflammation by influencing Toll-like receptors (TLRs) signaling. This bacterium can directly engage TLRs, such as TLR2 and TLR4, to regulate the activation of immune cells and the release of cytokines (70, 71). Its phospholipid components have been demonstrated to possess immunomodulatory properties, which can dampen inflammation and promote immune balance (72, 73). Additionally, Amuc_1100 activates TLR2 signaling, enhancing the intestinal barrier function and modulating the gut microbiota (74, 75). This protein can also influence the production of serotonin, thereby affecting mood and behavior (76). Akkermansia muciniphila protects against nonalcoholic steatohepatitis by modulating macrophage polarization and TLR2-activated γδT17 cells (77) and improves cognitive function in aged mice (78). These effects are partly mediated through the regulation of TLR signaling pathways. Furthermore, Akkermansia muciniphila can alleviate the effects of high-fat diet-induced obesity and insulin resistance by modulating gut TLR signaling (79). Its extracellular vesicles have been shown to modulate TLRs and tight junctions, thereby improving intestinal barrier function (80). These findings underscore the importance of Akkermansia muciniphila in maintaining immune balance and health through its interactions with TLRs.
High-frequency word analysis reveals that Akkermansia muciniphila is strongly associated with intestinal inflammation. Akkermansia muciniphila has been implicated in both protective and pathogenic effects in inflammatory bowel disease (IBD). In mice with dextran sulfate sodium (DSS)-induced colitis, administration of Akkermansia has been shown to ameliorate colitis symptoms, suggesting a beneficial role in IBD (81). However, in gnotobiotic interleukin-10-deficient mice, the Akkermansia muciniphila strain ATCC BAA-835 does not promote short-term intestinal inflammation, indicating that its effects may be strain-specific (82). In experimental models of colitis, Akkermansia muciniphila has been shown to reduce peritonitis and improve intestinal tissue wound healing after a colonic transmural defect through a MyD88-dependent mechanism (83). Additionally, the bacterium’s outer membrane protein Amuc_1100 has been identified as regulating tryptophan metabolism in colitis, highlighting its potential as a therapeutic target (84). Akkermansia may protect against colitis-associated tumorigenesis by modulating CD8+ T cells (48). Clinical studies have also further indicated the potential of Akkermansia muciniphila as a therapeutic agent. In an open-labeled study, an increased relative abundance of Akkermansia muciniphila was associated with improvement in abdominal pain in irritable bowel syndrome patients (85). These findings suggest that Akkermansia muciniphila could be a valuable target for the development of new treatments for intestinal disorders.
High-frequency word analysis reveals that research on Akkermansia muciniphila in metabolic disorders has primarily concentrated on metabolic dysregulation and obesity. Akkermansia muciniphila, a mucin-degrading bacterium with a growing reputation, has emerged as a key player in the regulation of metabolic diseases (86). The following review highlights recent studies that have elucidated its role in obesity, type 2 diabetes, and related disorders. Akkermansia muciniphila has been shown to play a role in the improvement of metabolic profiles by reducing inflammation in chow diet-fed mice (87). A study by Shen et al. (88) reported that capsaicin’s anti-obesity effect in mice fed a high-fat diet was associated with an increase in Akkermansia muciniphila population. In humans, higher stool salt levels were associated with obesity and Akkermansia muciniphila depletion (89). Akkermansia muciniphila cross-talk with the intestinal epithelium controlled diet-induced obesity (90). The bacterium was found to be lower in severe obesity (91) and to be inversely correlated with the onset of inflammation and metabolic disorders during obesity in mice (92). In a study using NOD mice, Hansen et al. found that early-life treatment with vancomycin propagated Akkermansia muciniphila and reduced diabetes incidence (4). Akkermansia muciniphila has been found to be negatively correlated with hemoglobin A1c in refractory diabetes (93). A decreased abundance of Akkermansia muciniphila led to impaired insulin secretion and glucose homeostasis in lean individuals with type 2 diabetes (49). In a randomized, double-blind, placebo-controlled trial, Roshanravan et al. (94) found that sodium butyrate and inulin supplementation affected angiotensin signaling by promoting of Akkermansia muciniphila abundance in type 2 diabetes. Furthermore, it has been demonstrated that Akkermansia muciniphila can effectively mitigate metabolic dysfunction-associated fatty liver disease by regulating L-aspartate metabolism through the gut-liver axis (95). The Ophiopogon japonicus polysaccharide MDG has also been shown to inhibit non-alcoholic fatty liver disease by modulating the abundance of Akkermansia muciniphila (96). Additionally, Akkermansia muciniphila has been found to reduce fat accumulation by activating the nhr-49-mediated nuclear hormone signaling pathway in Caenorhabditis elegans (97). These findings collectively suggest that Akkermansia muciniphila represents a promising therapeutic target for the treatment of metabolic syndrome and related disorders.
High-frequency word analysis reveals that Akkermansia muciniphila is strongly associated with the gut-brain axis. Akkermansia muciniphila is increasingly recognized for its pivotal role in modulating the gut-brain axis, impacting various neurological conditions. Research has demonstrated that Akkermansia muciniphila ameliorates depression-like behaviors in mice exposed to chronic stress (45, 98), with its outer membrane protein, Amuc_1100, specifically alleviating stress-induced anxiety and depression (44). Notably, this probiotic strain also improves depressive symptoms by regulating 5-HT neurotransmitter levels in both the gut and brain (99), and its small extracellular vesicles suppress microglial activation, thereby attenuating cognitive dysfunction post-surgery (74). The benefits of Akkermansia muciniphila extend to Alzheimer’s disease, where it mitigates cognitive deficits and amyloid pathology in mouse models (100), Supplementation with Akkermansia muciniphila can prevent cognitive impairment in sleep-deprived mice (101). Moreover, it reverses cognitive impairment associated with high-fat diets in rats (102), and its abundance correlates with reduced anxiety and depression in socially defeated mice (103). The diversity of Akkermansia muciniphila in the gut microbiota is linked to Parkinson’s disease dementia (104), and its presence in the gut microbiota, as modulated by metformin, improves cognitive function in aged mice (105). These findings reinforce the critical role of Akkermansia muciniphila in maintaining neurological health, offering potential therapeutic avenues for cognitive and neurodegenerative disorders. The emerging evidence on Akkermansia muciniphila’s influence on the gut-brain axis underscores its potential as a therapeutic target for neurological conditions.
Akkermansia muciniphila has garnered acclaim as a prospective probiotic, a notion largely substantiated by studies in animal models. Our bibliometric analysis, however, predominantly highlighted associations between Akkermansia muciniphila and inflammatory conditions and metabolic disorders, with less pronounced evidence regarding its relevance to other diseases. This discrepancy may stem from several factors: In many disease cohorts, the abundance of Akkermansia muciniphila does not demonstrate statistically significant fluctuations, implying minimal impact or involvement in certain pathologies. The probiotic benefits of Akkermansia muciniphila in contexts beyond metabolism and inflammation remain underexplored, potentially due to a scarcity of targeted functional assays evaluating its activity in diverse disease states. Comprehensive clinical assessments featuring large patient populations are lacking, hindering definitive conclusions about Akkermansia muciniphila’s widespread efficacy and safety profile. Notably, some studies illustrate a close association between elevated Akkermansia muciniphila levels and disease onset, underscoring the complexity of its role in health and disease. For Akkermansia muciniphila to genuinely transition into a mainstream health-promoting organism, extensive large-scale clinical trials are warranted, coupled with vigilant monitoring for potential adverse effects. Such endeavors will provide critical insights necessary for establishing Akkermansia muciniphila’s utility and safety across various health spectra.
Akkermansia muciniphila emerges as a cornerstone probiotic species, demonstrating potent anti-inflammatory properties by fortifying the gut barrier, generating short-chain fatty acids (SCFAs), and orchestrating immunological responses. Its potential in mitigating obesity is evidenced by improvements in insulin sensitivity, adiposity reduction, and body weight regulation. Akkermansia muciniphila fosters gut health through diverse strategies, encompassing barrier optimization, modulation of energy metabolism, facilitation of GLP-1 secretion, and biosynthesis of advantageous metabolites, which synergistically support weight management and metabolic homeostasis. By leveraging mucin utilization, competitive exclusion of detrimental microbes, promotion of commensal bacteria, and production of SCFAs alongside additional bioactive compounds, Akkermansia muciniphila plays a pivotal role in preserving gut health and augmenting overall welfare, thereby affecting neurological and metabolic disorders. Prospectively, Akkermansia muciniphila’s anti-inflammatory and gut-shielding characteristics position it at the forefront of therapeutic innovation. It holds promise in ameliorating inflammation-associated conditions, including malignancies and cardiovascular ailments. These hypotheses require validation through large-scale clinical cohort investigations to ascertain its efficacy. Concurrently, mechanistic insights can be sought through cell-based and animal model experiments to unravel underlying molecular interactions. Collectively, such endeavors will solidify Akkermansia muciniphila’s standing as a novel therapeutic candidate for a wider array of health challenges, underpinning its transformative potential in biomedical research and healthcare delivery.
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author.
FG: Conceptualization, Data curation, Formal analysis, Writing – original draft. CC: Conceptualization, Investigation, Methodology, Writing – original draft. RL: Methodology, Project administration, Writing – original draft. ZC: Methodology, Project administration, Resources, Writing – original draft. KT: Resources, Writing – original draft. GD: Software, Validation, Visualization, Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmed.2025.1484656/full#supplementary-material
SUPPLEMENTARY FIGURE S1 | International cooperation information analyzed using PubMed and Scopus databases.
1. Sparfel, L, Ratodiarivony, S, Boutet-Robinet, E, Ellero-Simatos, S, and Jolivet-Gougeon, A. Akkermansia muciniphila and alcohol-related liver diseases. A systematic review. Mol Nutr Food Res. (2024) 68:e2300510. doi: 10.1002/mnfr.202300510
2. Keane, JM, Cazzaniga, M, and Gahan, CGM. Akkermansia muciniphila in infectious disease: a new target for this next-generation probiotic? Sci Prog. (2024) 107:368504241231159. doi: 10.1177/00368504241231159
3. Derrien, M, Vaughan, EE, Plugge, CM, and de Vos, WM. Akkermansia muciniphila gen. nov., sp. nov., a human intestinal mucin-degrading bacterium. Int J Syst Evol Microbiol. (2004) 54:1469–76. doi: 10.1099/ijs.0.02873-0
4. Hansen, CH, Krych, L, Nielsen, DS, Vogensen, FK, Hansen, LH, Sorensen, SJ, et al. Early life treatment with vancomycin propagates Akkermansia muciniphila and reduces diabetes incidence in the NOD mouse. Diabetologia. (2012) 55:2285–94. doi: 10.1007/s00125-012-2564-7
5. Niu, H, Zhou, M, Zogona, D, Xing, Z, Wu, T, Chen, R, et al. Akkermansia muciniphila: a potential candidate for ameliorating metabolic diseases. Front Immunol. (2024) 15:1370658. doi: 10.3389/fimmu.2024.1370658
6. Cheng, T, Li, C, Shen, L, Wang, S, Li, X, Fu, C, et al. The intestinal effect of atorvastatin: Akkermansia muciniphila and barrier function. Front Microbiol. (2022) 12:797062. doi: 10.3389/fmicb.2021.797062
7. Mo, C, Lou, X, Xue, J, Shi, Z, Zhao, Y, Wang, F, et al. The influence of Akkermansia muciniphila on intestinal barrier function. Gut Pathog. (2024) 16:41. doi: 10.1186/s13099-024-00635-7
8. Ottman, N, Geerlings, SY, Aalvink, S, de Vos, WM, and Belzer, C. Action and function of Akkermansia muciniphila in microbiome ecology, health and disease. Best Pract Res Clin Gastroenterol. (2017) 31:637–42. doi: 10.1016/j.bpg.2017.10.001
9. Zhai, R, Xue, X, Zhang, L, Yang, X, Zhao, L, and Zhang, C. Strain-specific anti-inflammatory properties of two Akkermansia muciniphila strains on chronic colitis in mice. Front Cell Infect Microbiol. (2019) 9:239. doi: 10.3389/fcimb.2019.00239
10. Qu, S, Zheng, Y, Huang, Y, Feng, Y, Xu, K, Zhang, W, et al. Excessive consumption of mucin by over-colonized Akkermansia muciniphila promotes intestinal barrier damage during malignant intestinal environment. Front Microbiol. (2023) 14:1111911. doi: 10.3389/fmicb.2023.1111911
11. Trastoy, B, Naegeli, A, Anso, I, Sjogren, J, and Guerin, ME. Structural basis of mammalian mucin processing by the human gut O-glycopeptidase OgpA from Akkermansia muciniphila. Nat Commun. (2020) 11:4844. doi: 10.1038/s41467-020-18696-y
12. Zheng, M, Han, R, Yuan, Y, Xing, Y, Zhang, W, Sun, Z, et al. The role of Akkermansia muciniphila in inflammatory bowel disease: current knowledge and perspectives. Front Immunol. (2022) 13:1089600. doi: 10.3389/fimmu.2022.1089600
13. Watanabe, Y, Fujisaka, S, Morinaga, Y, Watanabe, S, Nawaz, A, Hatta, H, et al. Isoxanthohumol improves obesity and glucose metabolism via inhibiting intestinal lipid absorption with a bloom of Akkermansia muciniphila in mice. Mol Metab. (2023) 77:101797. doi: 10.1016/j.molmet.2023.101797
14. Chen, C. CiteSpace II: detecting and visualizing emerging trends and transient patterns in scientific literature. J Am Soc Inf Sci Technol. (2006) 57:359–77. doi: 10.1002/asi.20317
15. Arruda, H, Silva, ER, Lessa, M, Proença, D Jr, and Bartholo, R. VOSviewer and Bibliometrix. J Med Libr Assoc. (2022) 110:392–5. doi: 10.5195/jmla.2022.1434
16. Li, M, and Cheng, Y. Bibliometric analysis of researches of Orem self-care model in China based on BICOMB. TMR Integr Nurs. (2021) 5:21. doi: 10.53388/TMRIN20191214
17. Rasmussen, M, and Karypis, G. gCLUTO—an interactive clustering, visualization, and analysis system In: Technical Report; 04-021 (2004)
18. Li, Z, Ke, H, Wang, Y, Chen, S, Liu, X, Lin, Q, et al. Global trends in Akkermansia muciniphila research: a bibliometric visualization. Front Microbiol. (2022) 13:1037708. doi: 10.3389/fmicb.2022.1037708
19. Dehghanbanadaki, H, Aazami, H, Keshavarz Azizi Raftar, S, Ashrafian, F, Ejtahed, HS, Hashemi, E, et al. Global scientific output trend for Akkermansia muciniphila research: a bibliometric and scientometric analysis. BMC Med Inform Decis Mak. (2020) 20:291. doi: 10.1186/s12911-020-01312-w
20. Remely, M, Hippe, B, Geretschlaeger, I, Stegmayer, S, Hoefinger, I, and Haslberger, A. Increased gut microbiota diversity and abundance of Faecalibacterium prausnitzii and Akkermansia after fasting: a pilot study. Wien Klin Wochenschr. (2015) 127:394–8. doi: 10.1007/s00508-015-0755-1
21. Dao, MC, Everard, A, Aron-Wisnewsky, J, Sokolovska, N, Prifti, E, Verger, EO, et al. Akkermansia muciniphila and improved metabolic health during a dietary intervention in obesity: relationship with gut microbiome richness and ecology. Gut. (2016) 65:426–36. doi: 10.1136/gutjnl-2014-308778
22. Anhe, FF, Roy, D, Pilon, G, Dudonne, S, Matamoros, S, Varin, TV, et al. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut. (2015) 64:872–83. doi: 10.1136/gutjnl-2014-307142
23. Shang, Q, Sun, W, Shan, X, Jiang, H, Cai, C, Hao, J, et al. Carrageenan-induced colitis is associated with decreased population of anti-inflammatory bacterium, Akkermansia muciniphila, in the gut microbiota of C57BL/6J mice. Toxicol Lett. (2017) 279:87–95. doi: 10.1016/j.toxlet.2017.07.904
24. Shang, Q, Wang, Y, Pan, L, Niu, Q, Li, C, Jiang, H, et al. Dietary polysaccharide from Enteromorpha Clathrata modulates gut microbiota and promotes the growth of Akkermansia muciniphila, Bifidobacterium spp. and Lactobacillus spp. Mar Drugs. (2018) 16:167. doi: 10.3390/md16050167
25. Remely, M, Hippe, B, Zanner, J, Aumueller, E, Brath, H, and Haslberger, AG. Gut microbiota of obese, type 2 diabetic individuals is enriched in Faecalibacterium prausnitzii, Akkermansia muciniphila and Peptostreptococcus anaerobius after weight loss. Endocr Metab Immune Disord Drug Targets. (2016) 16:99–106. doi: 10.2174/1871530316666160831093813
26. Demirci, M, Tokman, HB, Uysal, HK, Demiryas, S, Karakullukcu, A, Saribas, S, et al. Reduced Akkermansia muciniphila and Faecalibacterium prausnitzii levels in the gut microbiota of children with allergic asthma. Allergol Immunopathol. (2019) 47:365–71. doi: 10.1016/j.aller.2018.12.009
27. Van Herreweghen, F, De Paepe, K, Roume, H, Kerckhof, FM, and Van de Wiele, T. Mucin degradation niche as a driver of microbiome composition and Akkermansia muciniphila abundance in a dynamic gut model is donor independent. FEMS Microbiol Ecol. (2018) 94:fiy186. doi: 10.1093/femsec/fiy186
28. Van Herreweghen, F, De Paepe, K, Marzorati, M, and Van de Wiele, T. Mucin as a functional niche is a more important driver of in vitro gut microbiota composition and functionality than supplementation of Akkermansia muciniphila. Appl Environ Microbiol. (2021) 87:e02647. doi: 10.1128/AEM.02647-20
29. Lukovac, S, Belzer, C, Pellis, L, Keijser, BJ, de Vos, WM, Montijn, RC, et al. Differential modulation by Akkermansia muciniphila and Faecalibacterium prausnitzii of host peripheral lipid metabolism and histone acetylation in mouse gut organoids. mBio. (2014) 5:e01438. doi: 10.1128/mBio.01438-14
30. Zhang, H, Zhang, S, Chen, L, Xu, R, and Zhu, J. LC-HRMS-based metabolomics and lipidomics analyses of a novel probiotic Akkermansia Muciniphila in response to different nutritional stimulations. J Microbiol Methods. (2024) 223:106975. doi: 10.1016/j.mimet.2024.106975
31. Konstanti, P, Ligthart, K, Fryganas, C, Constantinos, P, Smidt, H, de Vos, WM, et al. Physiology of gamma-aminobutyric acid production by Akkermansia muciniphila. Appl Environ Microbiol. (2024) 90:e0112123. doi: 10.1128/aem.01121-23
32. Chia, LW, Hornung, BVH, Aalvink, S, Schaap, PJ, de Vos, WM, Knol, J, et al. Deciphering the trophic interaction between Akkermansia muciniphila and the butyrogenic gut commensal Anaerostipes caccae using a metatranscriptomic approach. Antonie Van Leeuwenhoek. (2018) 111:859–73. doi: 10.1007/s10482-018-1040-x
33. Gu, Z, Pei, W, Shen, Y, Wang, L, Zhu, J, Zhang, Y, et al. Akkermansia muciniphila and its outer protein Amuc_1100 regulates tryptophan metabolism in colitis. Food Funct. (2021) 12:10184–95. doi: 10.1039/D1FO02172A
34. Xiong, S, Sun, H, Lu, C, He, J, Wu, Z, Wang, Y, et al. Kuqin ameliorates lipopolysaccharide-induced acute lung injury by regulating indoleamine 2, 3-dioxygenase 1 and Akkermansia muciniphila. Biomed Pharmacother. (2023) 158:114073. doi: 10.1016/j.biopha.2022.114073
35. Chen, Y, Chen, J, Wei, H, Gong, K, Meng, J, Long, T, et al. Akkermansia muciniphila-Nlrp 3 is involved in the neuroprotection of phosphoglycerate mutase 5 deficiency in traumatic brain injury mice. Front Immunol. (2023) 14:1172710. doi: 10.3389/fimmu.2023.1172710
36. Gao, W, Xiao, M, Gu, Z, Fu, X, Ren, X, Yu, Y, et al. Genome analysis and 2′-fucosyllactose utilization characteristics of a new Akkermansia muciniphila strain isolated from mice feces. Mol Gen Genomics. (2022) 297:1515–28. doi: 10.1007/s00438-022-01937-8
37. Jiang, L, Yuan, C, Ye, W, Huang, Q, Chen, Z, Wu, W, et al. Akkermansia and its metabolites play key roles in the treatment of campylobacteriosis in mice. Front Immunol. (2022) 13:1061627. doi: 10.3389/fimmu.2022.1061627
38. Xu, R, Zhang, Y, Chen, S, Zeng, Y, Fu, X, Chen, T, et al. The role of the probiotic Akkermansia muciniphila in brain functions: insights underpinning therapeutic potential. Crit Rev Microbiol. (2023) 49:151–76. doi: 10.1080/1040841X.2022.2044286
39. Zou, R, Shen, G, Wu, Y, Guo, M, Chen, J, Yang, S, et al. Akkermansia muciniphila plays a neuroprotective role in HMC3 cells through the 'gut-brain' axis. Future Microbiol. (2023) 18:255–66. doi: 10.2217/fmb-2022-0007
40. Zhang, F, and Wang, D. Potential of Akkermansia muciniphila and its outer membrane proteins as therapeutic targets for neuropsychological diseases. Front Microbiol. (2023) 14:1191445. doi: 10.3389/fmicb.2023.1191445
41. Cani, PD, and Knauf, C. A newly identified protein from Akkermansia muciniphila stimulates GLP-1 secretion. Cell Metab. (2021) 33:1073–5. doi: 10.1016/j.cmet.2021.05.004
42. Sun, Y, Zhu, H, Cheng, R, Tang, Z, and Zhang, M. Outer membrane protein Amuc_1100 of Akkermansia muciniphila alleviates antibiotic-induced anxiety and depression-like behavior in mice. Physiol Behav. (2023) 258:114023. doi: 10.1016/j.physbeh.2022.114023
43. Qu, D, Chen, M, Zhu, H, Liu, X, Cui, Y, Zhou, W, et al. Akkermansia muciniphila and its outer membrane protein Amuc_1100 prevent high-fat diet-induced nonalcoholic fatty liver disease in mice. Biochem Biophys Res Commun. (2023) 684:149131. doi: 10.1016/j.bbrc.2023.149131
44. Cheng, R, Zhu, H, Sun, Y, Hang, T, and Zhang, M. The modified outer membrane protein Amuc_1100 of Akkermansia muciniphila improves chronic stress-induced anxiety and depression-like behavior in mice. Food Funct. (2022) 13:10748–58. doi: 10.1039/D2FO01198K
45. Cheng, R, Xu, W, Wang, J, Tang, Z, and Zhang, M. The outer membrane protein Amuc_1100 of Akkermansia muciniphila alleviates the depression-like behavior of depressed mice induced by chronic stress. Biochem Biophys Res Commun. (2021) 566:170–6. doi: 10.1016/j.bbrc.2021.06.018
46. Zhu, Z, Huang, J, Zhang, Y, Hou, W, Chen, F, Mo, YY, et al. Landscape of tumoral ecosystem for enhanced anti-PD-1 immunotherapy by gut Akkermansia muciniphila. Cell Rep. (2024) 43:114306. doi: 10.1016/j.celrep.2024.114306
47. Xu, Y, Tan, X, Yang, Q, Fang, Z, and Chen, W. Akkermansia muciniphila outer membrane protein regulates recruitment of CD8+ T cells in lung adenocarcinoma and through JAK-STAT signalling pathway. Microb Biotechnol. (2024) 17:e14522. doi: 10.1111/1751-7915.14522
48. Wang, L, Tang, L, Feng, Y, Zhao, S, Han, M, Zhang, C, et al. A purified membrane protein from Akkermansia muciniphila or the pasteurised bacterium blunts colitis associated tumourigenesis by modulation of CD8+ T cells in mice. Gut. (2020) 69:1988–97. doi: 10.1136/gutjnl-2019-320105
49. Zhang, FL, Yang, YL, Zhang, Z, Yao, YY, Xia, R, Gao, CC, et al. Surface-displayed Amuc_1100 from Akkermansia muciniphila on Lactococcus lactis ZHY1 improves hepatic steatosis and intestinal health in high-fat-fed zebrafish. Front Nutr. (2021) 8:726108. doi: 10.3389/fnut.2021.726108
50. Liu, S, Zhao, S, Cheng, Z, Ren, Y, Shi, X, Mu, J, et al. Akkermansia muciniphila protects against antibiotic-associated diarrhea in mice. Probiotics Antimicrob Proteins. (2024) 16:1190–204. doi: 10.1007/s12602-023-10101-6
51. Wade, H, Pan, K, Duan, Q, Kaluzny, S, Pandey, E, Fatumoju, L, et al. Akkermansia muciniphila and its membrane protein ameliorates intestinal inflammatory stress and promotes epithelial wound healing via CREBH and mi R-143/145. J Biomed Sci. (2023) 30:38. doi: 10.1186/s12929-023-00935-1
52. Segers, A, and de Vos, WM. Mode of action of Akkermansia muciniphila in the intestinal dialogue: role of extracellular proteins, metabolites and cell envelope components. Microbiome Res Rep. (2023) 2:6. doi: 10.20517/mrr.2023.05
53. Hao, L, Zhong, W, Woo, J, Wei, X, Ma, H, Dong, H, et al. Conventional type 1 dendritic cells protect against gut barrier disruption via maintaining Akkermansia muciniphila in alcoholic steatohepatitis. Hepatology. (2023) 78:896–910. doi: 10.1097/HEP.0000000000000019
54. Martin-Gallausiaux, C, Garcia-Weber, D, Lashermes, A, Larraufie, P, Marinelli, L, Teixeira, V, et al. Akkermansia muciniphila upregulates genes involved in maintaining the intestinal barrier function via ADP-heptose-dependent activation of the ALPK1/TIFA pathway. Gut Microbes. (2022) 14:2110639. doi: 10.1080/19490976.2022.2110639
55. Matute, JD, Duan, J, Flak, MB, Griebel, P, Tascon-Arcila, JA, Doms, S, et al. Intelectin-1 binds and alters the localization of the mucus barrier-modifying bacterium Akkermansia muciniphila. J Exp Med. (2023) 220:e20211938. doi: 10.1084/jem.20211938
56. Mezhibovsky, E, Wu, Y, Bawagan, FG, Tveter, KM, Szeto, S, and Roopchand, D. Impact of grape polyphenols on Akkermansia muciniphila and the gut barrier. AIMS Microbiol. (2022) 8:544–65. doi: 10.3934/microbiol.2022035
57. Shi, M, Yue, Y, Ma, C, Dong, L, and Chen, F. Pasteurized Akkermansia muciniphila ameliorate the LPS-induced intestinal barrier dysfunction via modulating AMPK and NF-kappa B through TLR2 in Caco-2 cells. Nutrients. (2022) 14:764. doi: 10.3390/nu14040764
58. Su, H, Mo, J, Ni, J, Ke, H, Bao, T, Xie, J, et al. Andrographolide exerts antihyperglycemic effect through strengthening intestinal barrier function and increasing microbial composition of Akkermansia muciniphila. Oxid Med Cell Longev. (2020) 2020:6538930. doi: 10.1155/2020/6538930
59. Yu, J, Liu, T, Gao, Z, Liu, R, Wang, Z, Chen, Y, et al. Akkermansia muciniphila colonization alleviating high fructose and restraint stress-induced jejunal mucosal barrier disruption. Nutrients. (2022) 14:3164. doi: 10.3390/nu14153164
60. Kosciow, K, and Deppenmeier, U. Characterization of a phospholipid-regulated beta-galactosidase from Akkermansia muciniphila involved in mucin degradation. Microbiology. (2019) 8:e00796. doi: 10.1002/mbo3.796
61. Lee, JY, Jin, HS, Kim, KS, Baek, JH, Kim, BS, and Lee, DW. Nutrient-specific proteomic analysis of the mucin degrading bacterium Akkermansia muciniphila. Proteomics. (2022) 22:e2100125. doi: 10.1002/pmic.202100125
62. Qian, K, Yang, W, Chen, X, Wang, Y, Zhang, M, and Wang, M. Functional and structural characterization of a GH3 beta-N-acetylhexosaminidase from Akkermansia muciniphila involved in mucin degradation. Biochem Biophys Res Commun. (2022) 589:186–91. doi: 10.1016/j.bbrc.2021.12.022
63. Xu, W, Yang, W, Wang, Y, Wang, M, and Zhang, M. Structural and biochemical analyses of β-N-acetylhexosaminidase Am0868 from Akkermansia muciniphila involved in mucin degradation. Biochem Biophys Res Commun. (2020) 529:876–81. doi: 10.1016/j.bbrc.2020.06.116
64. Elzinga, J, Narimatsu, Y, de Haan, N, Clausen, H, de Vos, WM, and Tytgat, HLP. Binding of Akkermansia muciniphila to mucin is O-glycan specific. Nat Commun. (2024) 15:4582. doi: 10.1038/s41467-024-48770-8
65. Liu, X, Zhao, F, Liu, H, Xie, Y, Zhao, D, and Li, C. Transcriptomics and metabolomics reveal the adaption of Akkermansia muciniphila to high mucin by regulating energy homeostasis. Sci Rep. (2021) 11:9073. doi: 10.1038/s41598-021-88397-z
66. Dong, C, Yu, J, Yang, Y, Zhang, F, Su, W, Fan, Q, et al. Berberine, a potential prebiotic to indirectly promote Akkermansia growth through stimulating gut mucin secretion. Biomed Pharmacother. (2021) 139:111595. doi: 10.1016/j.biopha.2021.111595
67. Lopez-Cauce, B, Puerto, M, Garcia, JJ, Ponce-Alonso, M, Becerra-Aparicio, F, Del Campo, R, et al. Akkermansia deficiency and mucin depletion are implicated in intestinal barrier dysfunction as earlier event in the development of inflammation in interleukin-10-deficient mice. Front Microbiol. (2022) 13:1083884. doi: 10.3389/fmicb.2022.1083884
68. Huck, O, Mulhall, H, Rubin, G, Kizelnik, Z, Iyer, R, Perpich, JD, et al. Akkermansia muciniphila reduces Porphyromonas gingivalis-induced inflammation and periodontal bone destruction. J Clin Periodontol. (2020) 47:202–12. doi: 10.1111/jcpe.13214
69. Cani, PD, Van Hul, M, and Bachmann, R. Akkermansia muciniphila derived tripeptide jams the gear of sepsis, inflammation and mortality. Gut. (2023) 73:3–4. doi: 10.1136/gutjnl-2023-331092
70. Liu, Y, Yang, M, Tang, L, Wang, F, Huang, S, Liu, S, et al. TLR4 regulates RORγt+ regulatory T-cell responses and susceptibility to colon inflammation through interaction with Akkermansia muciniphila. Microbiome. (2022) 10:98. doi: 10.1186/s40168-022-01296-x
71. Luo, Y, Lan, C, Xie, K, Li, H, Devillard, E, He, J, et al. Active or autoclaved Akkermansia muciniphila relieves TNF-alpha-induced inflammation in intestinal epithelial cells through distinct pathways. Front Immunol. (2021) 12:788638. doi: 10.3389/fimmu.2021.788638
72. Bae, M, Cassilly, CD, Liu, X, Park, SM, Tusi, BK, Chen, X, et al. Akkermansia muciniphila phospholipid induces homeostatic immune responses. Nature. (2022) 608:168–73. doi: 10.1038/s41586-022-04985-7
73. Ghosh, S, and Mani, S. A phospholipid immunomodulator from Akkermansia muciniphila. Cell Host Microbe. (2022) 30:1191–3. doi: 10.1016/j.chom.2022.08.014
74. Gao, X, Lin, C, Feng, Y, You, Y, Jin, Z, Li, M, et al. Akkermansia muciniphila-derived small extracellular vesicles attenuate intestinal ischemia-reperfusion-induced postoperative cognitive dysfunction by suppressing microglia activation via the TLR2/4 signaling. Biochim Biophys Acta, Mol Cell Res. (2024) 1871:119630. doi: 10.1016/j.bbamcr.2023.119630
75. Wang, J, Xu, W, Wang, R, Cheng, R, Tang, Z, and Zhang, M. The outer membrane protein Amuc_1100 of Akkermansia muciniphila promotes intestinal 5-HT biosynthesis and extracellular availability through TLR2 signalling. Food Funct. (2021) 12:3597–610. doi: 10.1039/D1FO00115A
76. Yaghoubfar, R, Behrouzi, A, Ashrafian, F, Shahryari, A, Moradi, HR, Choopani, S, et al. Modulation of serotonin signaling/metabolism by Akkermansia muciniphila and its extracellular vesicles through the gut-brain axis in mice. Sci Rep. (2020) 10:22119. doi: 10.1038/s41598-020-79171-8
77. Han, Y, Ling, Q, Wu, L, Wang, X, Wang, Z, Chen, J, et al. Akkermansia muciniphila inhibits nonalcoholic steatohepatitis by orchestrating TLR2-activated γδT17 cell and macrophage polarization. Gut Microbes. (2023) 15:2221485. doi: 10.1080/19490976.2023.2221485
78. Xie, S, Li, J, Lyu, F, Xiong, Q, Gu, P, Chen, Y, et al. Novel tripeptide RKH derived from Akkermansia muciniphila protects against lethal sepsis. Gut. (2023) 73:78–91. doi: 10.1136/gutjnl-2023-329996
79. Yang, Y, Zhong, Z, Wang, B, Xia, X, Yao, W, Huang, L, et al. Early-life high-fat diet-induced obesity programs hippocampal development and cognitive functions via regulation of gut commensal Akkermansia muciniphila. Neuropsychopharmacology. (2019) 44:2054–64. doi: 10.1038/s41386-019-0437-1
80. Ashrafian, F, Behrouzi, A, Shahriary, A, Ahmadi Badi, S, Davari, M, Khatami, S, et al. Comparative study of effect of Akkermansia muciniphila and its extracellular vesicles on toll-like receptors and tight junction. Gastroenterol Hepatol Bed Bench. (2019) 12:163–8.
81. Bian, X, Wu, W, Yang, L, Lv, L, Wang, Q, Li, Y, et al. Administration of Akkermansia muciniphila ameliorates dextran sulfate sodium-induced ulcerative colitis in mice. Front Microbiol. (2019) 10:2259. doi: 10.3389/fmicb.2019.02259
82. Ring, C, Klopfleisch, R, Dahlke, K, Basic, M, Bleich, A, and Blaut, M. Akkermansia muciniphila strain ATCC BAA-835 does not promote short-term intestinal inflammation in gnotobiotic interleukin-10-deficient mice. Gut Microbes. (2019) 10:188–203. doi: 10.1080/19490976.2018.1511663
83. Bachmann, R, Van Hul, M, Baldin, P, Leonard, D, Delzenne, NM, Belzer, C, et al. Akkermansia muciniphila reduces peritonitis and improves intestinal tissue wound healing after a colonic transmural defect by a MyD88-dependent mechanism. Cells. (2022) 11:2666. doi: 10.3390/cells11172666
84. Gu, ZY, Pei, WL, Zhang, Y, Zhu, J, Li, L, and Zhang, Z. Akkermansia muciniphila in inflammatory bowel disease and colorectal cancer. Chin Med J. (2021) 134:2841–3. doi: 10.1097/CM9.0000000000001829
85. Cruz-Aguliar, RM, Wantia, N, Clavel, T, Vehreschild, M, Buch, T, Bajbouj, M, et al. An open-labeled study on fecal microbiota transfer in irritable bowel syndrome patients reveals improvement in abdominal pain associated with the relative abundance of Akkermansia Muciniphila. Digestion. (2019) 100:127–38. doi: 10.1159/000494252
86. Cani, PD, and Everard, A. Akkermansia muciniphila: a novel target controlling obesity, type 2 diabetes and inflammation? Med Sci. (2014) 30:125–7. doi: 10.1051/medsci/20143002003
87. Zhao, S, Liu, W, Wang, J, Shi, J, Sun, Y, Wang, W, et al. Akkermansia muciniphila improves metabolic profiles by reducing inflammation in chow diet-fed mice. J Mol Endocrinol. (2017) 58:1–14. doi: 10.1530/JME-16-0054
88. Shen, W, Shen, M, Zhao, X, Zhu, H, Yang, Y, Lu, S, et al. Anti-obesity effect of capsaicin in mice fed with high-fat diet is associated with an increase in population of the gut bacterium Akkermansia muciniphila. Front Microbiol. (2017) 8:272. doi: 10.3389/fmicb.2017.00272
89. Seck, EH, Senghor, B, Merhej, V, Bachar, D, Cadoret, F, Robert, C, et al. Salt in stools is associated with obesity, gut halophilic microbiota and Akkermansia muciniphila depletion in humans. Int J Obes. (2019) 43:862–71. doi: 10.1038/s41366-018-0201-3
90. Everard, A, Belzer, C, Geurts, L, Ouwerkerk, JP, Druart, C, Bindels, LB, et al. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci USA. (2013) 110:9066–71. doi: 10.1073/pnas.1219451110
91. Dao, MC, Belda, E, Prifti, E, Everard, A, Kayser, BD, Bouillot, JL, et al. Akkermansia muciniphila abundance is lower in severe obesity, but its increased level after bariatric surgery is not associated with metabolic health improvement. Am J Physiol Endocrinol Metab. (2019) 317:E446–59. doi: 10.1152/ajpendo.00140.2019
92. Schneeberger, M, Everard, A, Gomez-Valades, AG, Matamoros, S, Ramirez, S, Delzenne, NM, et al. Akkermansia muciniphila inversely correlates with the onset of inflammation, altered adipose tissue metabolism and metabolic disorders during obesity in mice. Sci Rep. (2015) 5:16643. doi: 10.1038/srep16643
93. Shih, CT, Yeh, YT, Lin, CC, Yang, LY, and Chiang, CP. Akkermansia muciniphila is negatively correlated with hemoglobin A1c in refractory diabetes. Microorganisms. (2020) 8:1360. doi: 10.3390/microorganisms8091360
94. Roshanravan, N, Mahdavi, R, Alizadeh, E, Ghavami, A, Rahbar Saadat, Y, Mesri Alamdari, N, et al. The effects of sodium butyrate and inulin supplementation on angiotensin signaling pathway via promotion of Akkermansia muciniphila abundance in type 2 diabetes; a randomized, double-blind, placebo-controlled trial. J Cardiovasc Thorac Res. (2017) 9:183–90. doi: 10.15171/jcvtr.2017.32
95. Rao, Y, Kuang, Z, Li, C, Guo, S, Xu, Y, Zhao, D, et al. Gut Akkermansia muciniphila ameliorates metabolic dysfunction-associated fatty liver disease by regulating the metabolism of L-aspartate via gut-liver axis. Gut Microbes. (2021) 13:1–19. doi: 10.1080/19490976.2021.1927633
96. Zhang, L, Wang, Y, Wu, F, Wang, X, Feng, Y, and Wang, Y. MDG, an Ophiopogon japonicus polysaccharide, inhibits non-alcoholic fatty liver disease by regulating the abundance of Akkermansia muciniphila. Int J Biol Macromol. (2022) 196:23–34. doi: 10.1016/j.ijbiomac.2021.12.036
97. Wu, Z, Xiao, Y, Zhou, F, Chen, J, Chen, X, Hou, A, et al. Pasteurized Akkermansia muciniphila reduces fat accumulation via nhr-49-mediated nuclear hormone signaling pathway in Caenorhabditis elegans. Molecules. (2022) 27:6159. doi: 10.3390/molecules27196159
98. Guo, D, Park, C, Li, Y, Li, B, Yang, Q, Deng, Y, et al. Akkermansia muciniphila ameliorates depressive disorders in a murine alcohol-LPS (mALPS) model. Food Funct. (2022) 13:12766–76. doi: 10.1039/D2FO01478E
99. Guo, H, Liu, X, Chen, T, Wang, X, and Zhang, X. Akkermansia muciniphila improves depressive-like symptoms by modulating the level of 5-HT neurotransmitters in the gut and brain of mice. Mol Neurobiol. (2024) 61:821–34. doi: 10.1007/s12035-023-03602-6
100. Ou, Z, Deng, L, Lu, Z, Wu, F, Liu, W, Huang, D, et al. Protective effects of Akkermansia muciniphila on cognitive deficits and amyloid pathology in a mouse model of Alzheimer’s disease. Nutr Diabetes. (2020) 10:12. doi: 10.1038/s41387-020-0115-8
101. Li, N, Tan, S, Wang, Y, Deng, J, Wang, N, Zhu, S, et al. Akkermansia muciniphila supplementation prevents cognitive impairment in sleep-deprived mice by modulating microglial engulfment of synapses. Gut Microbes. (2023) 15:2252764. doi: 10.1080/19490976.2023.2252764
102. Higarza, SG, Arboleya, S, Arias, JL, Gueimonde, M, and Arias, N. Akkermansia muciniphila and environmental enrichment reverse cognitive impairment associated with high-fat high-cholesterol consumption in rats. Gut Microbes. (2021) 13:1–20. doi: 10.1080/19490976.2021.1880240
103. McGaughey, KD, Yilmaz-Swenson, T, Elsayed, NM, Cruz, DA, Rodriguiz, RM, Kritzer, MD, et al. Relative abundance of Akkermansia spp. and other bacterial phylotypes correlates with anxiety- and depressive-like behavior following social defeat in mice. Sci Rep. (2019) 9:3281. doi: 10.1038/s41598-019-40140-5
104. Arikan, M, Kahraman Demir, T, Yildiz, Z, Helvaci Yilmaz, N, Sen, A, Hanoglu, L, et al. Investigation of the relationship between Akkermansia genomic diversity in gut microbiota and Parkinson’s disease dementia. Mikrobiyol Bul. (2024) 58:13–28. doi: 10.5578/mb.20249951
Keywords: Akkermansia muciniphila, bibliometrics, gut microbiota, gut-brain axis, metabolic disorder
Citation: Gao F, Cheng C, Li R, Chen Z, Tang K and Du G (2025) The role of Akkermansia muciniphila in maintaining health: a bibliometric study. Front. Med. 12:1484656. doi: 10.3389/fmed.2025.1484656
Received: 22 August 2024; Accepted: 21 January 2025;
Published: 03 February 2025.
Edited by:
Veronica I. Dodero, Bielefeld University, GermanyReviewed by:
Yina Huang, Anhui Polytechnic University, ChinaCopyright © 2025 Gao, Cheng, Li, Chen, Tang and Du. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Guankui Du, ZHVndWFua3VpQDE2My5jb20=
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.