- 1Department of Abdominal Oncology, Cancer Center, West China Hospital, Sichuan University, Chengdu, Sichuan, China
- 2Abdominal Oncology Ward, Division of Medical Oncology, Cancer Center, State Key Laboratory of Biotherapy, West China Hospital, Sichuan University, Chengdu, Sichuan, China
Introduction: The identification of risk factors for regional lymph node (r-LN) metastasis in rectal neuroendocrine tumors (R-NETs) remains challenging. Our objective was to investigate the risk factors associated with patients diagnosed with R-NETs exhibiting r-LN metastasis.
Methods: Patient information was obtained from the Surveillance, Epidemiology, and End Results (SEER) database, complemented by data from the West China Hospital (WCH) databases. The construction cohort comprised patients diagnosed with R-NETs from the SEER database, while cases from the WCH database were utilized as the validation cohort. A novel nomogram was developed to predict the probability of r-LN metastasis, employing a logistic regression model.
Results: Univariate analysis identified four independent risk factors associated with poor r-LN metastasis: age (HR = 1.027, p < 0.05), grade (HR = 0.010, p < 0.05), T stage (HR = 0.010, p < 0.05), and tumor size (HR = 0.005, p < 0.05). These factors were selected as predictors for nomogram construction.
Discussion: The novel nomogram serves as a reliable tool for predicting the risk of r-LN metastasis, providing clinicians with valuable assistance in identifying high-risk patients and tailoring individualized treatments.
Introduction
Neuroendocrine tumors (NETs), which account for just 0.5% of all neoplasms (1, 2), typically develop in various organs, including the gastro-entero-pancreatic tract, lungs, gallbladder, thymus, thyroid, testicles, ovaries, and skin (3–5). Among the Asian population, R-NETs have the highest incidence rate of all gastroenteropancreatic neuroendocrine tumors (6, 7). Previous studies have established an association between r-LN metastasis in R-NETs and poorer prognosis (8). In addition to tumor size, the presence or absence of r-LN metastasis significantly influences treatment options. The European Neuroendocrine Tumor Society consensus guidelines recommend endoscopic resection for R-NETs ≤10 mm, provided there is no invasion of the intrinsic muscle layer or r-LN involvement (9). However, approximately 10% of patients present with r-LN metastasis, which necessitates radical resection (10–12). Diagnosis is typically made through computed tomography (CT) or endoscopic ultrasound (EUS). The Italian Society of Gastroenterology recommends that local staging for tumors larger than 10 mm, or in cases where complete resection is not feasible, should be evaluated using EUS (13). While 68Ga-DOTANOC PET-CT demonstrates satisfactory sensitivity in early r-LN metastasis detection, its widespread clinical application is hindered by high examination costs (14). Thus, identifying high-risk factors for r-LN metastasis is crucial for surgical planning. Given the existing controversies surrounding the prognostic significance and operative procedures for R-NETs with r-LN metastasis, our study conducts a comprehensive evaluation. We aim to determine preoperative factors predicting r-LN metastasis and analyze its impact on R-NETs, utilizing data from the SEER database.
Our study presents a detailed nomogram, developed based on the SEER database, to predict both r-LN metastasis and distant metastasis. This nomogram has been validated using a cohort from the WCH database.
Patients and methods
Patient characteristics and study design
In the construction cohort, the data of patients were extracted from the SEER database, which collected information on cancer. SEER cohort Data from patients with pathologically confirmed R-NETs were retrieved from the SEER database (from 1988 to 2014) using SEER*Stat version 8.3.4 software. In the validation cohort, cases were retrospectively obtained from the WCH database. The collected data included patient demographics, histology, T, N and M stage of the primary tumor, tumor size, r-LN metastasis and distant metastasis. Patients were retrieved according to International Classification of Diseases for Oncology (third revision) codes: carcinoid tumor (8240), argentaffin carcinoid tumor (8241), enterochromaffin cell tumor (8242), mucocarcinoid tumor (8243), mixed adenoneuroendocrine carcinoma (8244), adenocarcinoid tumor (8245), neuroendocrine carcinoid (8246), and atypical carcinoid tumor (8249). We included patients with primary tumors of the rectum. The exclusion criteria were as follows: patients with unknown TNM stage, unknown tumor size, unknown metastatic r-LN, unknown distant metastasis or unknown tumor grade by pathologic examination. We analyzed all patient data for demographic characteristics, including sex and age. Tumor-specific variables, including grade, size, metastatic r-LN, and distant metastasis, were evaluated.
Statistical analysis
Student’s t test and the Chi-squared test (or Fisher’s exact test) were performed in univariate analysis depending on the categorical and ordinal variables. Multivariate analysis was performed using logistic regression analysis to evaluate potential risk factors, including age, sex, tumor size, TNM stage and histologic grade. Hazard ratios (HR) and 95% confidence intervals (CIs) were calculated. Statistical analysis was accomplished using SPSS 26.0 software (IBM, Armonk, NY, United States). For all analyses, p values <0.05 were defined as statistically significant.
A nomogram based on the results of multivariate analysis was constructed using the rms package in R version 4.1.3,1 which estimated r-LN metastasis and distant metastasis. The concordance index (C-index) was used to assess the predictive performance of this nomogram. Calibration curves were utilized to evaluate the agreement between the predicted and actual r-LN metastasis rates.
Results
Patient characteristics
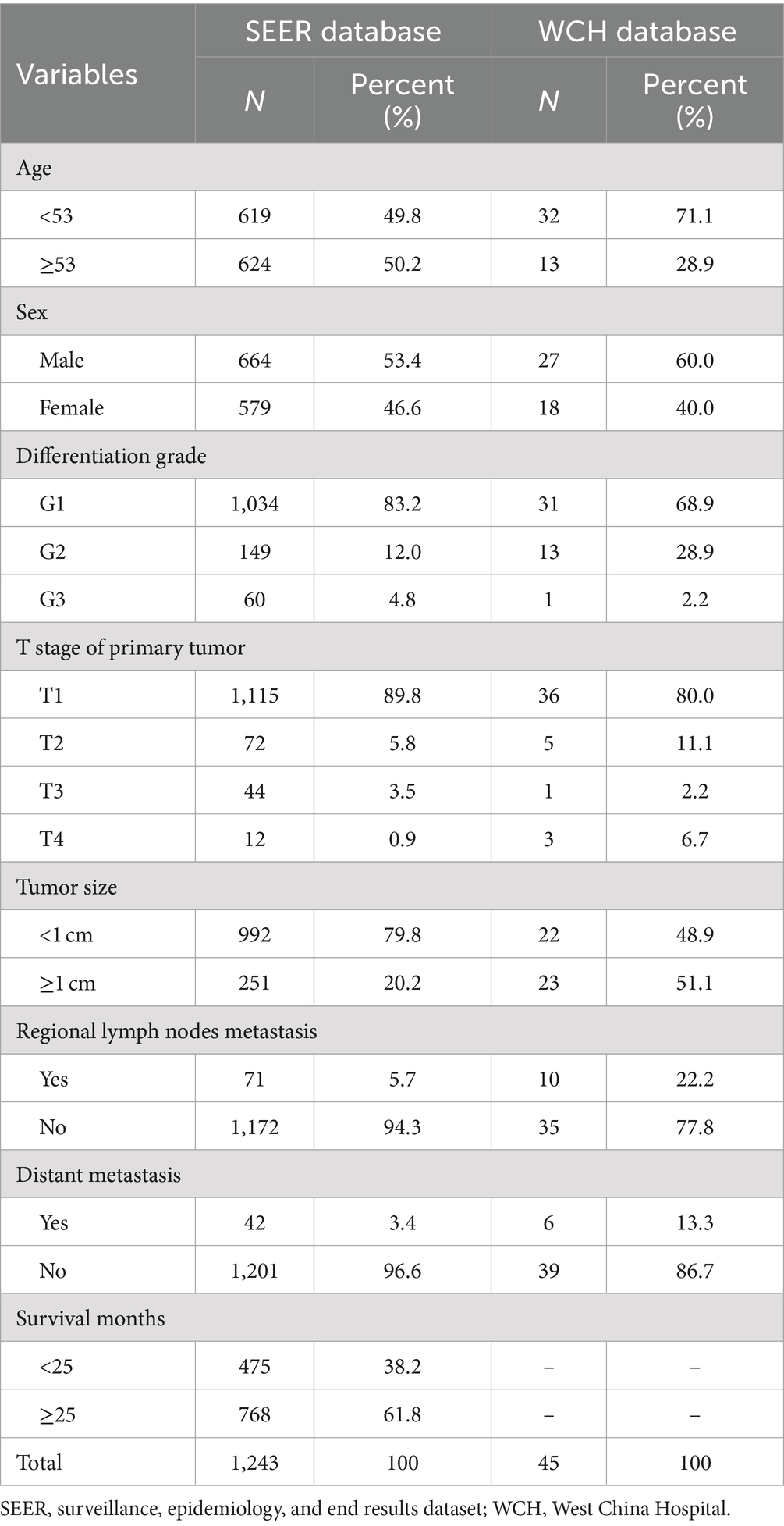
Using the previously mentioned criteria, we screened a total of 5,103 patients with R-NETs, excluding some due to missing data on essential staging, r-LN metastasis, and other factors. Ultimately, 1,243 newly diagnosed R-NET patients from the SEER database were included in the final analysis for the construction cohort (Figure 1). Similarly, 45 patients in the WCH database were assigned to the validation cohort (Figure 1). Clinicopathologic characteristics in the two cohorts regarding age, sex, tumor size, T stage, histologic grade, r-LN metastasis and distant metastasis. The mean age at diagnosis of R-NETs was 53 years old in the SEER cohort and 48 years old in the validation cohort. In the SEER database, more than half of the patients (53.4%) were males. There were 251 (20.2%) cases with tumor sizes greater than 1 cm, 1,115 (89.8%) at the T1 stage, and 1,034 (89.8%) at Grade I. r-LN and distant metastasis have only been shown in a few patients (5.7 and 3.4%, respectively). For the validation cohort, the rate of tumor size greater than 1 cm was 51.1%, and the r-LN and distant metastasis rates were 22.2 and 13.3%, respectively. The details are shown in Table 1. We analyzed the survival curves of patients with or without r-LN metastasis in the SEER cohort. The results revealed that median OS was considerably lower in r-LN metastasis patients than in patients without r-LN metastasis (21 vs. 58 months, 95% CI = 15.06–34.94, p < 0.001) (Figure 2).
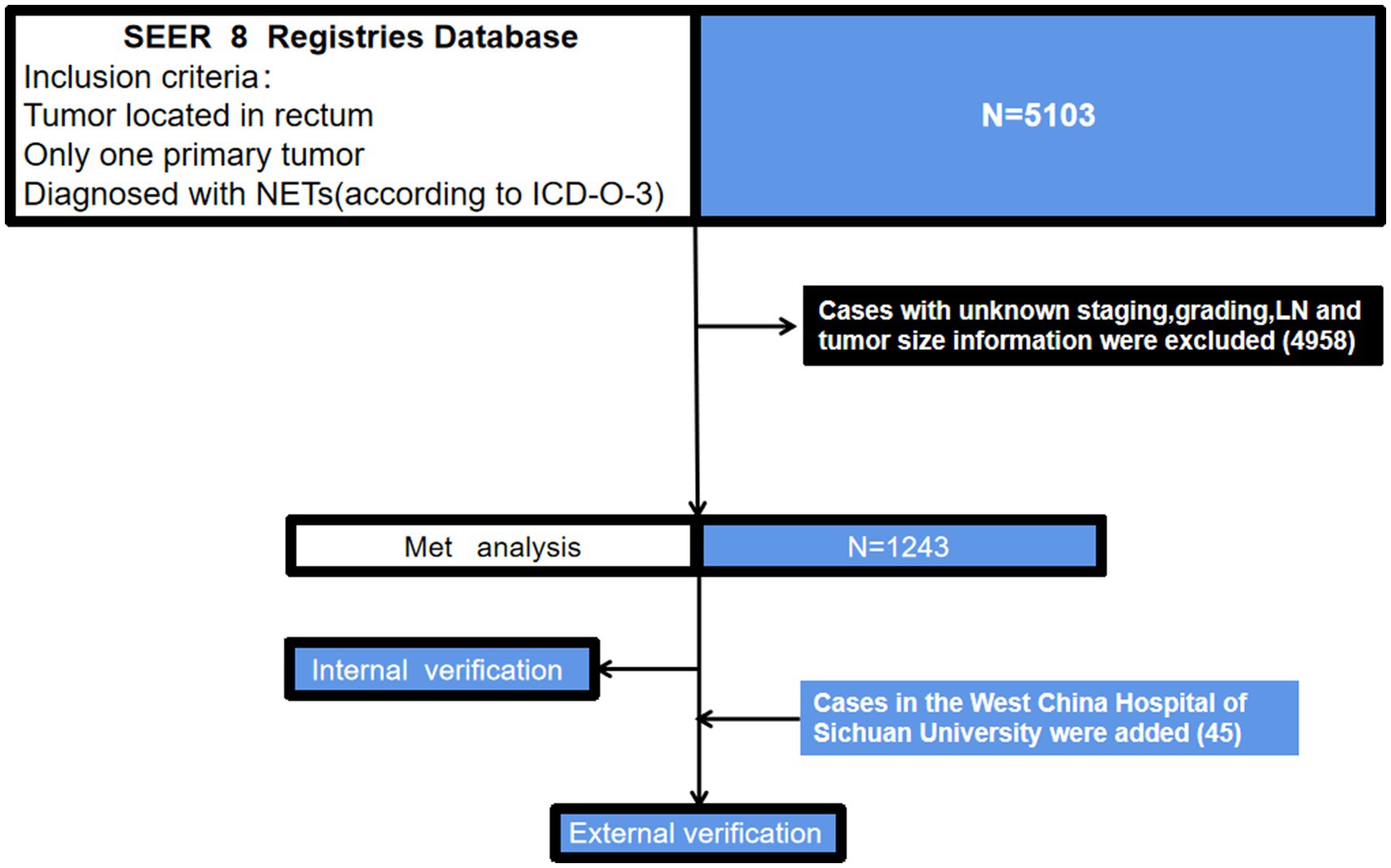
Figure 1. A flowchart of patient selection and study design. NETs, neuroendocrine tumors; ICD-O-3, international classification of diseases for oncology, 3rd edition; SEER, the surveillance, epidemiology, and end results dataset.
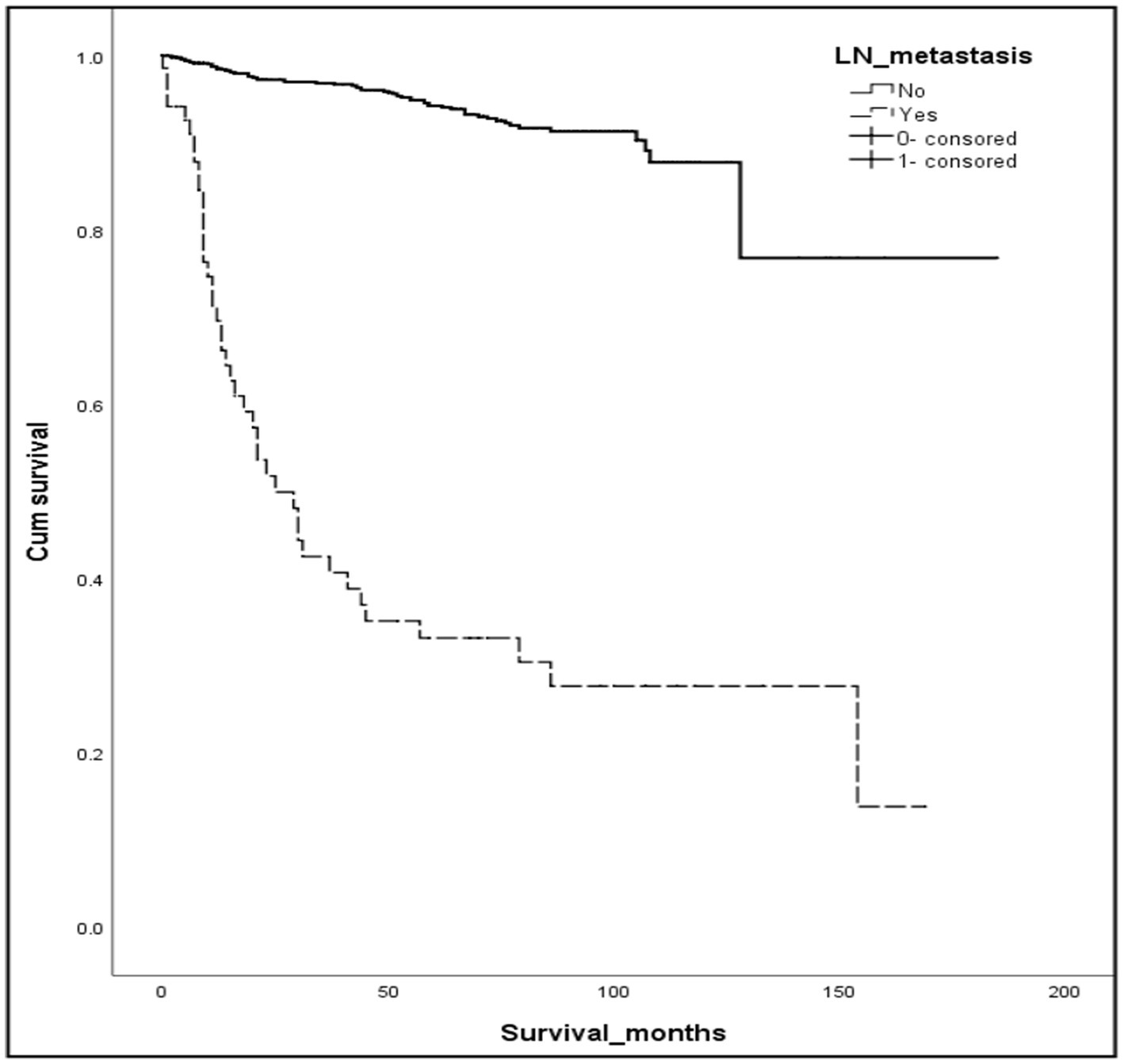
Figure 2. Kaplan–Meier curves of OS for patients with or without r-LN metastasis using the SEER database. Compared to negative r-LN metastasis, positive r-LN metastasis was significantly associated with worse OS (mOS: 21 vs. 58 months, 95%CI = 15.06–34.94, p < 0.001).
Independent risk factors for r-LN and distant metastasis analyzed in the training cohort
In the construction cohort, univariate analysis showed that sex (p > 0.05) was not an independent risk factor, but differentiation age, grade, tumor size and T stage may be risk factors affecting r-LN metastasis (p < 0.05). Following univariate analysis, a multifactorial logistic regression analysis was performed. The results revealed that age (HR = 1.027, 95% CI = 1.005–1.050), grade (HR = 0.010, 95% CI = 0.005–0.020), T stage (HR = 0.010, 95% CI = 0.000–0.012), and tumor size (HR = 0.005, 95% CI = 0.001–0.022) were independently associated with r-LN metastasis (p < 0.05). The results of the univariate and multifactorial analyses are shown in Table 2 and Figure 3A.
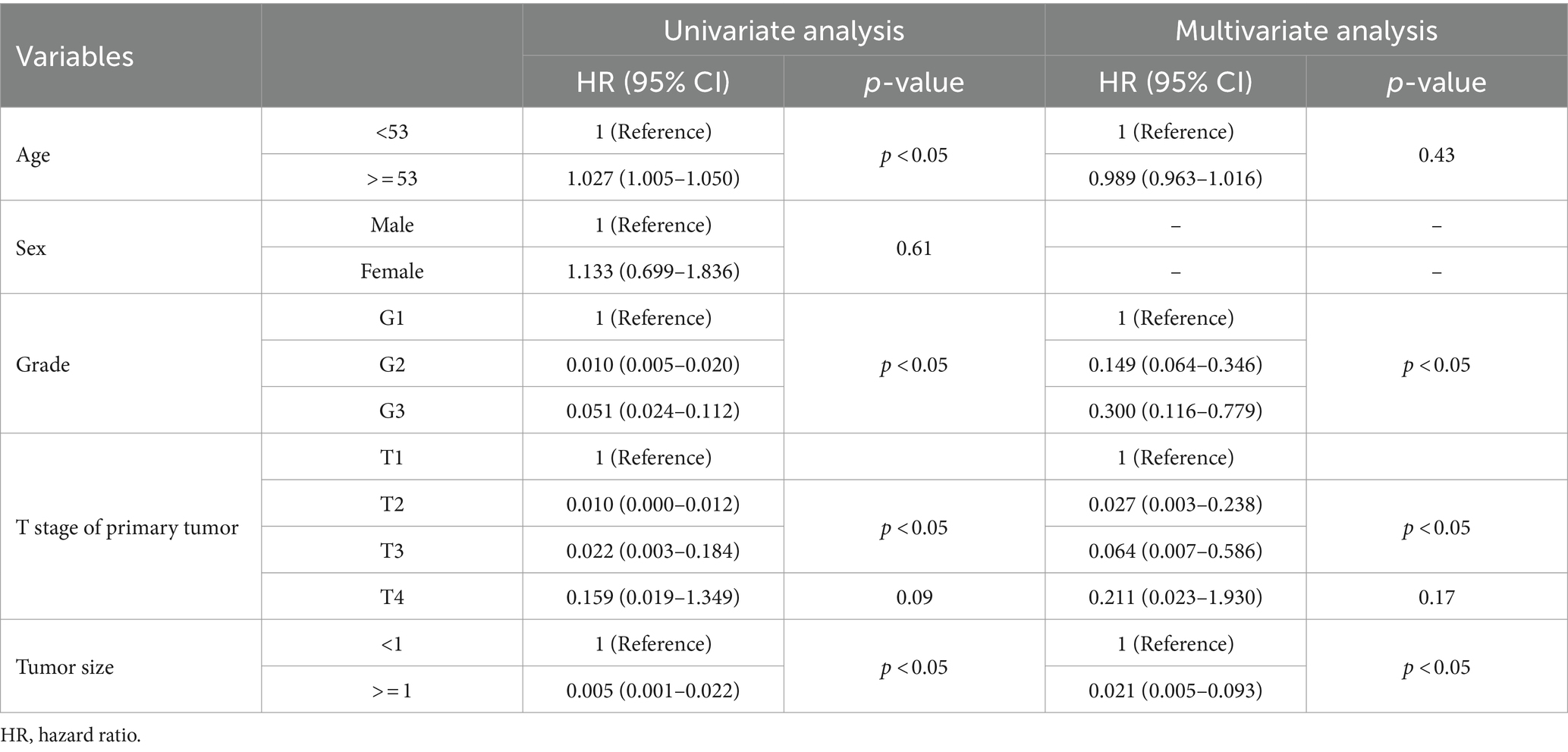
Table 2. Univariate and multivariate logistic analyses of the risk of r-LN metastasis in patients diagnosed with rectal neuroendocrine tumors from the SEER database.
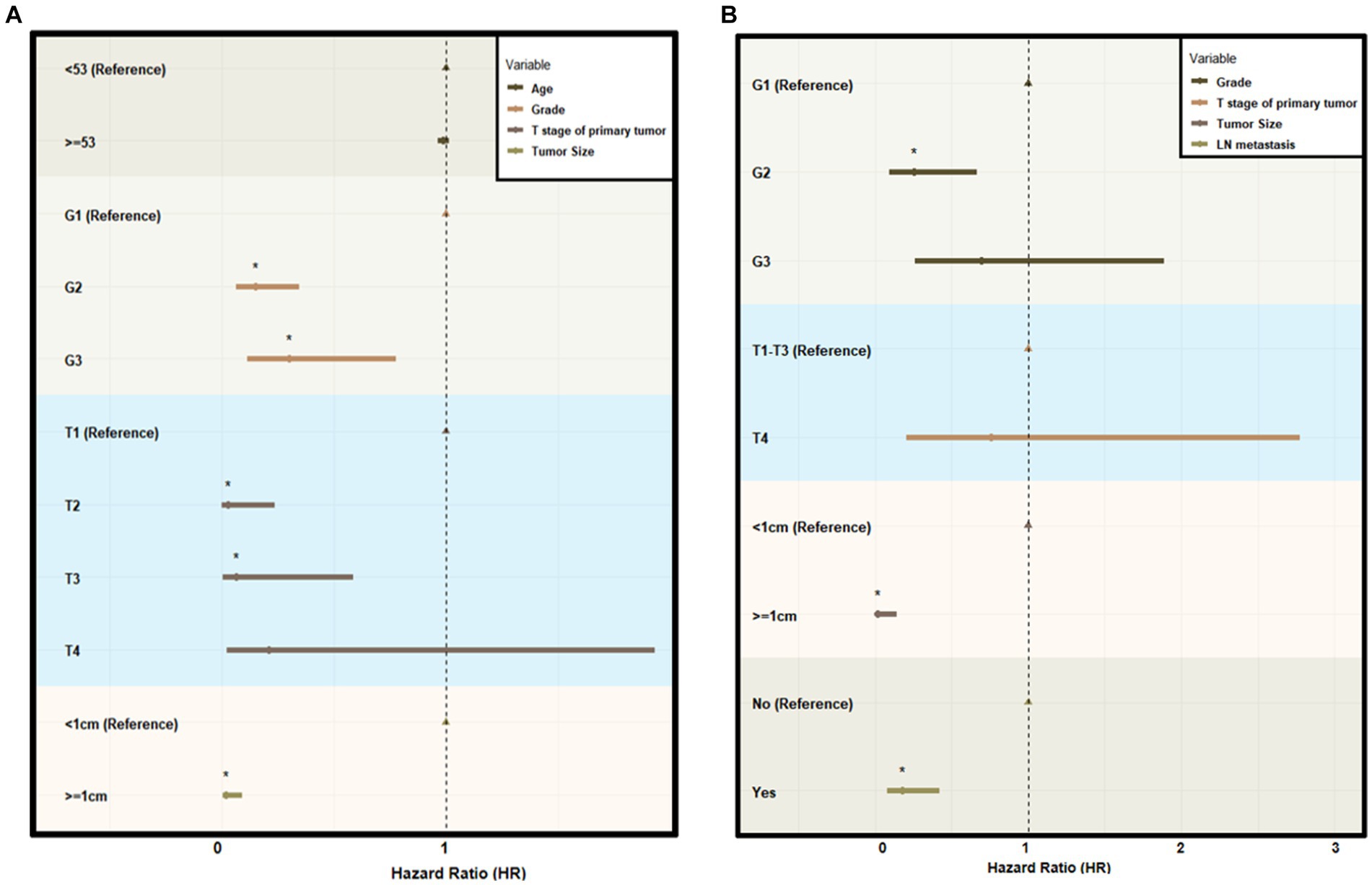
Figure 3. Forest plot of multivariate logistic analysis using the SEER database. (A) Multivariate logistic analysis of the risk of r-LN metastasis for patients diagnosed with rectal neuroendocrine tumors from the SEER database. (B) Multivariate logistic analysis of the risk of distant metastasis for patients diagnosed with rectal neuroendocrine tumors from the SEER database. HR indicates hazard ratio.
For the distant metastasis analysis, univariate analysis showed that age (p > 0.05) and sex (p > 0.05) were not significant risk factors in the construction cohort. The differentiation grade, tumor size, T stage and r-LN metastasis could be risk factors affecting distant metastasis. The logistic regression analysis of distant metastasis might be associated with differentiation grade (HR = 0.253, 95% CI = 0.096–0.668), tumor size (HR = 0.019, 95% CI = 0.002–0.145) and r-LN metastasis (HR = 0.179, 95% CI = 0.076–0.424), while T stage was not regarded as a meaningful factor (p > 0.05). The results of distant metastasis are shown in Table 3 and Figure 3B.
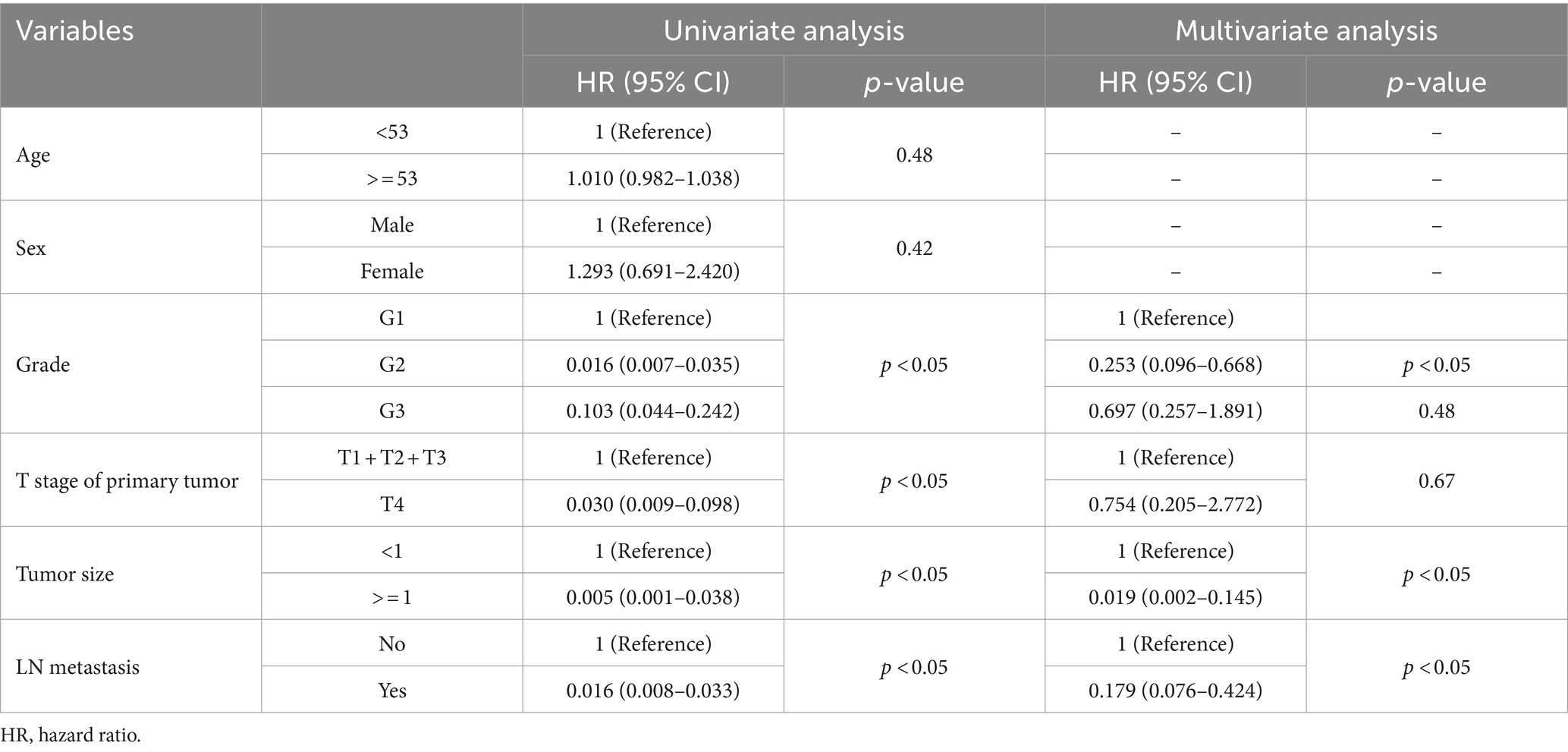
Table 3. Univariate and multivariate logistic analyses of the risk of distant metastasis in patients diagnosed with rectal neuroendocrine tumors from the SEER database.
Prognostic nomogram for r-LN metastasis
Based on the independent prognostic factors indicated in the multivariate logistic analysis, we developed a novel nomogram to estimate r-LN metastasis (Figure 4). Each variable was given a score according to the hazard ratio (HR). Adding up the total scores of each selected variable and locating it onto the total points scale, the probability of r-LN metastasis of an individual patient could be easily estimated. We conducted internal validation of the nomogram and obtained a C-index of 0.968 (95% CI = 0.949–0.986). By bootstrap sampling 1,000 times, the calibration curves of the nomogram were plotted and demonstrated optimal agreement between the predicted and actual survival. In the external validation using cases from the WCH database, the C-index was 0.877 (95% CI = 0.765–0.989), which showed great consistency between the nomogram prediction and the actual prognosis for the training set and validation set. The results are shown in Figures 5A,B. At the same time, the constructed calibration curves reflect the consistency between our predicted and actual values (Figures 5C,D).
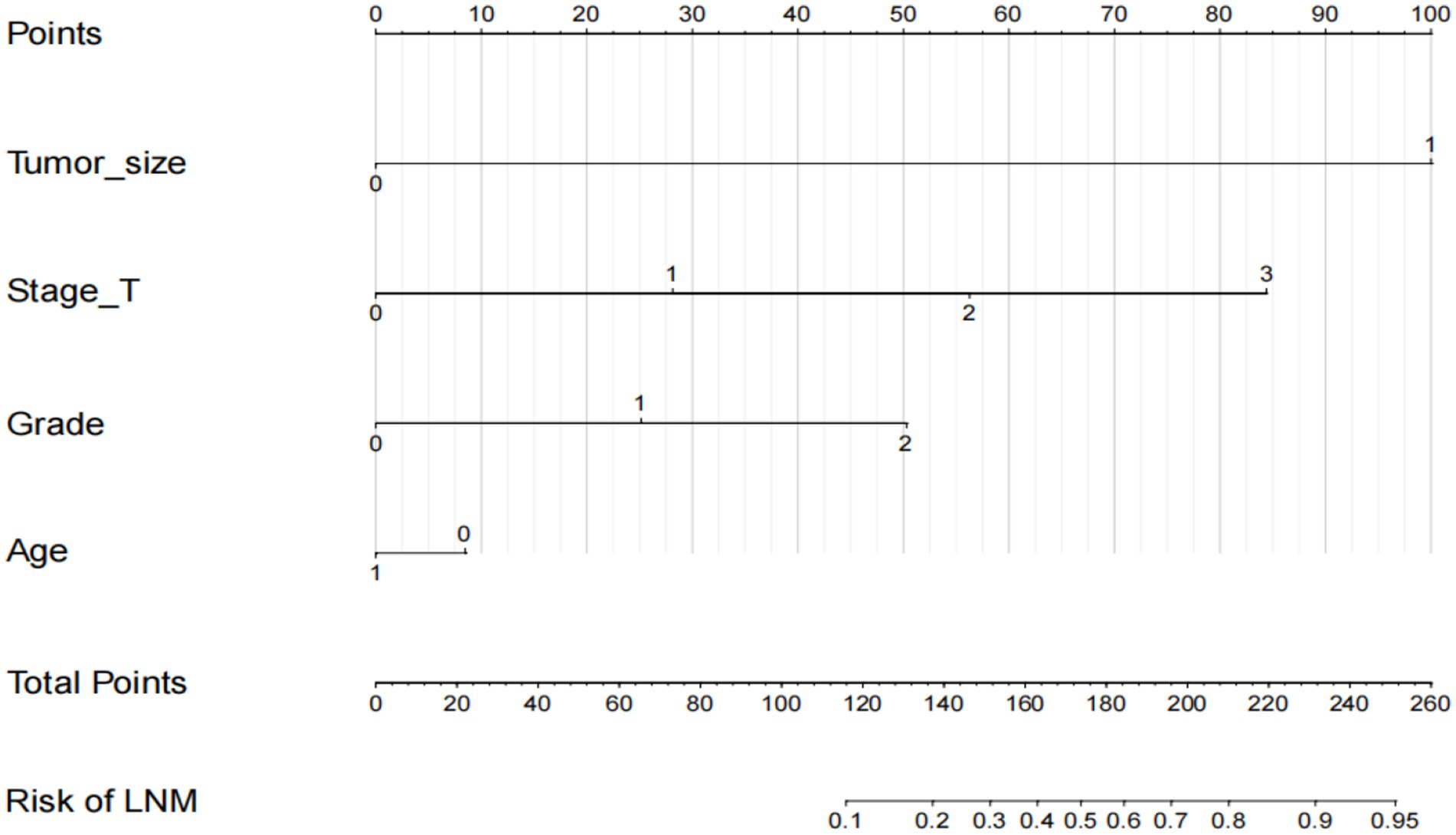
Figure 4. Nomogram to predict the risk of r-LN metastasis in patients with rectal neuroendocrine tumors. Points for age, tumor size, tumor grade and T stage are obtained by drawing a line upward from the corresponding values to the “Points” line. The sum of the points of these 4 factors is located on the “Total points” line.
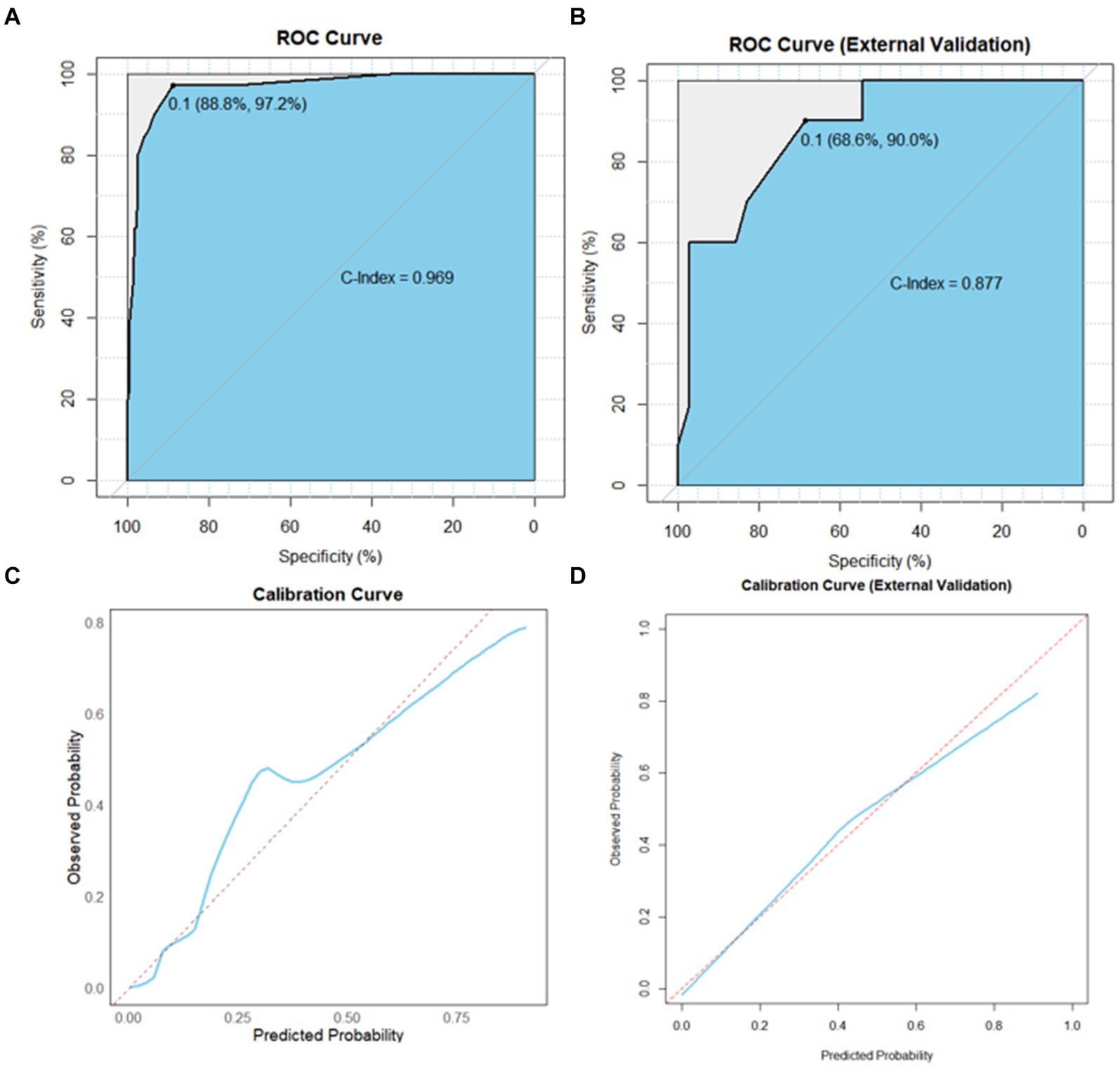
Figure 5. AUC value of ROC prediction for the nomogram using the training and validation sets. (A) Calibration plots of the nomogram for the risk of r-LN metastasis in the training set. (B) Calibration plots of the nomogram for the risk of r-LN metastasis in the validation set. Sky blue area under the curves of the two models to predict risk rates of r-LN metastasis. (C) Calibration curves in the training set. (D) Calibration curves in the validation set. AUC, area under the curve; ROC, receiver operating characteristic.
Discussion
R-NETs are generally considered tumors with a favorable prognosis, with a five-year survival rate of up to 88% in North America (15, 16). Surgery remains the primary treatment for R-NETs, with endoscopic resection often being sufficient (17). However, the choice of endoscopic surgical approach is heavily influenced by tumor size and r-LN metastasis status. Current guidelines suggest that endoscopic treatment alone is appropriate for tumors <1 cm without r-LN metastasis (18, 19). Conversely, when the tumor >1 cm, achieving R0 resection becomes challenging based on tumor size alone, requiring greater attention to r-LN involvement. Previous studies have reported that it is challenging to predict the presence of r-LN metastasis through CT scans (20). In recent years, 68Ga-DOTANOC PET-CT has been introduced in the diagnosis and treatment of neuroendocrine tumors, offering a more efficient preoperative screening for patients with positive r-LN metastasis (14). However, due to the high cost of this examination, widespread clinical implementation poses certain challenges. Therefore, our aim is to capture clinical characteristics of patients, identify those at high risk for positive r-LN metastasis, and tailor more suitable diagnostic and therapeutic strategies, thus avoiding unnecessary expenses.
Various studies have explored characteristics and risk factors for r-LN metastasis in R-NETs. Wei et al. conducted a retrospective study involving 419 patients, revealing G grade (95% CI = 2.122–32.103, p < 0.001), depth of tumor invasion (95% CI = 4.586–566.595, p < 0.001), and tumor size (95% CI = 2.798–307.298, p < 0.001) as independent risk factors (21). Beonghoon and his colleagues found tumor size and tumor grade to be predictive factors for LN metastasis in R-NET patients (15). A recent multicenter retrospective study in 199 patients identified tumor size >11.5 mm and vascular invasion as independent poor prognostic factors (22). Consistent with these findings, our results highlighted four factors: age, differentiation grade, T stage of primary tumor, and tumor size.
To our knowledge, this is the most extensive study on r-LN metastatic R-NETs to date, encompassing both American and Chinese cases. Despite differences between the two populations, including variations in r-LN metastasis rates (5.7% vs. 22.2%), the predictive model we developed performed well in the validation cohort. Our study identified four independent prognostic factors and introduced a novel nomogram. Current guidelines for R-NETs recommend a formal oncologic low anterior resection with total mesorectal excision based on patients’ clinical and genetic characteristics (9, 23). Management should be guided by predictors of nodal involvement (24, 25). Our study aims to enhance the recognition of r-LN metastasis, aiding clinicians in estimating optimal treatment. Additionally, this is the first study constructing an r-LN metastasis nomogram based on both American and Chinese R-NET patients.
However, some limitations exist. Firstly, due to its retrospective nature, our study is susceptible to selection bias. Secondly, unavailable data in the SEER database might lead to the oversight of crucial factors such as depth of tumor invasion, vascular invasion, ki-67, etc. Thirdly, owing to the low incidence of R-NETs, the validation cohort’s sample size was small. Despite the limitations of this study, we advocate for larger prospective studies to further validate our findings. Additionally, incorporating more clinical features, such as genetic markers and lifestyle factors, into the model assessment is essential to enhance the nomogram’s adaptability to real-time clinical practice.
Conclusion
We identified four independent LN metastasis risk factors in a large cohort of R-NET patients, including age, differentiation grade, T stage of primary tumor and tumor size. In addition, a novel nomogram was developed based on these variables. Internal validation of the nomogram exhibited satisfactory performance. Further research is needed to verify the practicality of this model.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
RL: Investigation, Writing – original draft. XiaofL: Conceptualization, Writing – original draft. YW: Data curation, Formal analysis, Writing – original draft. CC: Software, Writing – original draft. WL: Validation, Writing – original draft. XiaoyL: Project administration, Writing – original draft. DC: Conceptualization, Funding acquisition, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study was supported by the National Key Research and Development Program of China (2023YFB330850103).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Footnotes
References
1. Moertel, CG. Karnofsky memorial lecture. An odyssey in the land of small tumors. J Clin Oncol. (1987) 5:1502–22. doi: 10.1200/JCO.1987.5.10.1502
2. Young, K, Starling, N, and Sadanandam, A. The molecular biology of pancreatic neuroendocrine neoplasms: challenges and translational opportunities. Semin Cancer Biol. (2020) 61:132–8. doi: 10.1016/j.semcancer.2019.09.024
3. Ahmed, M. Gastrointestinal neuroendocrine tumors in 2020. World J Gastrointest Oncol. (2020) 12:791–807. doi: 10.4251/wjgo.v12.i8.791
4. Mohamed, A, Wu, S, Hamid, M, Mahipal, A, Cjakrabarti, S, Bajor, D, et al. Management of Appendix Neuroendocrine Neoplasms: insights on the current guidelines. Cancers (Basel). (2022) 15:295. doi: 10.3390/cancers15010295
5. Iabichino, G, Di Leo, M, Arena, M, Passoni, GG, Morandi, E, Turpini, F, et al. Diagnosis, treatment, and current concepts in the endoscopic management of gastroenteropancreatic neuroendocrine neoplasms. World J Gastroenterol. (2022) 28:4943–58. doi: 10.3748/wjg.v28.i34.4943
6. Li, AF, Hsu, CY, Li, A, Tai, LC, Liang, WY, Li, WY, et al. A 35-year retrospective study of carcinoid tumors in Taiwan: differences in distribution with a high probability of associated second primary malignancies. Cancer. (2008) 112:274–83. doi: 10.1002/cncr.23159
7. Zhang, Y, Zhang, Y, Yang, Y, Xu, Z, Gao, C, Liu, M, et al. Case report: indocyanine green fluorescence-guided imaging in laparoscope, a more sensitive detection technique of lateral lymph nodes metastases from rectal neuroendocrine tumors. Front Oncol. (2022) 12:1101990. doi: 10.3389/fonc.2022.1101990
8. Hiyoshi, Y, Mukai, T, Nagasaki, T, Yamaguchi, T, Akiyoshi, T, and Fukunaga, Y. ASO author reflections: rectal neuroendocrine tumors have strong potential for lymph node metastasis in accordance with an increase of risk factors, and lymph node metastasis is associated with a poor prognosis. Ann Surg Oncol. (2023) 30:3954–5. doi: 10.1245/s10434-023-13350-5
9. Ramage, JK, De Herder, WW, Delle Fave, G, Ferolla, P, Ferone, D, Ito, T, et al. Vienna consensus conference participants. ENETS consensus guidelines update for colorectal neuroendocrine neoplasms. Neuroendocrinology. (2016) 103:139–43. doi: 10.1159/000443166
10. Goretzki, PE, Mogl, MT, Akca, A, and Pratschke, J. Curative and palliative surgery in patients with neuroendocrine tumors of the gastro-entero-pancreatic (GEP) tract. Rev Endocr Metab Disord. (2018) 19:169–78. doi: 10.1007/s11154-018-9469-9
11. Andreasi, V, Partelli, S, Muffatti, F, Manzoni, MF, Capurso, G, and Falconi, M. Update on gastroenteropancreatic neuroendocrine tumors. Dig Liver Dis. (2021) 53:171–82. doi: 10.1016/j.dld.2020.08.031
12. Zhang, DG, Luo, S, Xiong, F, Xu, ZL, Li, YX, Yao, J, et al. Endoloop ligation after endoscopic mucosal resection using a transparent cap: a novel method to treat small rectal carcinoid tumors. World J Gastroenterol. (2019) 25:1259–65. doi: 10.3748/wjg.v25.i10.1259
13. Panzuto, F, Parodi, MC, Esposito, G, Massironi, S, Fantin, A, Cannizzaro, R, et al. Endoscopic management of gastric, duodenal and rectal NETs: position paper from the Italian Association for Neuroendocrine Tumors (ITANET), Italian Society of Gastroenterology (SIGE), Italian Society of Digestive Endoscopy (SIED). Dig Liver Dis. (2024) 56:589–600. doi: 10.1016/j.dld.2023.12.015
14. Zhou, Z, Wang, Z, Zhang, B, Wu, Y, Li, G, and Wang, Z. Comparison of 68Ga-DOTANOC and 18F-FDG PET-CT scans in the evaluation of primary tumors and lymph node metastasis in patients with rectal neuroendocrine tumors. Front Endocrinol (Lausanne). (2021) 12:727327. doi: 10.3389/fendo.2021.727327
15. Sohn, B, Kwon, Y, Ryoo, SB, Song, I, Kwon, YH, Lee, DW, et al. Predictive factors for lymph node metastasis and prognostic factors for survival in rectal neuroendocrine tumors. J Gastrointest Surg. (2017) 21:2066–74. doi: 10.1007/s11605-017-3603-y
16. Yao, JC, Hassan, M, Phan, A, Dagohoy, C, Leary, C, Mares, JE, et al. One hundred years after "carcinoid": epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. (2008) 26:3063–72. doi: 10.1200/JCO.2007.15.4377
17. Bertani, E, Ravizza, D, Milione, M, Massironi, S, Grana, CM, Zerini, D, et al. Neuroendocrine neoplasms of rectum: a management update. Cancer Treat Rev. (2018) 66:45–55. doi: 10.1016/j.ctrv.2018.04.003
18. Gallo, C, Rossi, RE, Cavalcoli, F, Barbaro, F, Boškoski, I, Invernizzi, P, et al. Rectal neuroendocrine tumors: current advances in management, treatment, and surveillance. World J Gastroenterol. (2022) 28:1123–38. doi: 10.3748/wjg.v28.i11.1123
19. Rossi, RE, and Massironi, S. NEN gain or NEN loss: endoscopic resection for rectal neuroendocrine neoplasms < 15 mm. Dig Dis Sci. (2023) 68:2793–5. doi: 10.1007/s10620-023-07927-z
20. Hiyoshi, Y, Daitoku, N, Mukai, T, Nagasaki, T, Yamaguchi, T, Akiyoshi, T, et al. Risk factors for lymph node metastasis of rectal neuroendocrine tumor and its prognostic impact: a single-center retrospective analysis of 195 cases with radical resection. Ann Surg Oncol. (2023) 30:3944–53. doi: 10.1245/s10434-023-13348-z
21. Wei, G, Feng, X, Wang, W, Zhang, Y, Zeng, Y, Chen, M, et al. Analysis of risk factors of lymph node metastasis in rectal neuroendocrine neoplasms using multicenter data. Future Oncol. (2018) 14:1817–23. doi: 10.2217/fon-2018-0059
22. Ricci, AD, Pusceddu, S, Panzuto, F, Gelsomino, F, Massironi, S, de Angelis, CG, et al. Assessment of the risk of nodal involvement in rectal neuroendocrine neoplasms: the NOVARA score, a multicentre retrospective study. J Clin Med. (2022) 11:713. doi: 10.3390/jcm11030713
23. de Mestier, L, Lepage, C, Baudin, E, Coriat, R, Courbon, F, Couvelard, A, et al. Digestive neuroendocrine neoplasms (NEN): French intergroup clinical practice guidelines for diagnosis, treatment and follow-up (SNFGE, GTE, RENATEN, TENPATH, FFCD, GERCOR, UNICANCER, SFCD, SFED, SFRO, SFR). Dig Liver Dis. (2020) 52:473–92. doi: 10.1016/j.dld.2020.02.011
24. Zilli, A, Arcidiacono, PG, Conte, D, and Massironi, S. Clinical impact of endoscopic ultrasonography on the management of neuroendocrine tumors: lights and shadows. Dig Liver Dis. (2018) 50:6–14. doi: 10.1016/j.dld.2017.10.007
25. Nuñez-Valdovinos, B, Carmona-Bayonas, A, Jimenez-Fonseca, P, Capdevila, J, Castaño-Pascual, Á, Benavent, M, et al. Neuroendocrine tumor heterogeneity adds uncertainty to the World Health Organization 2010 classification: real-world data from the Spanish tumor registry (R-GETNE). Oncologist. (2018) 23:422–32. doi: 10.1634/theoncologist.2017-0364
Keywords: regional lymph node metastasis, rectal neuroendocrine tumors, SEER, West China hospital databases, tumor size
Citation: Li R, Li X, Wang Y, Chang C, Lv W, Li X and Cao D (2024) Risk factors for regional lymph node metastasis in rectal neuroendocrine tumors: a population-based study. Front. Med. 11:1383047. doi: 10.3389/fmed.2024.1383047
Edited by:
Yifei Zheng, Huazhong University of Science and Technology, ChinaReviewed by:
Sara Massironi, San Gerardo Hospital, ItalyBenjian Gao, The Affiliated Hospital of Southwest Medical University, China
Copyright © 2024 Li, Li, Wang, Chang, Lv, Li and Cao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dan Cao, Y2FvZGFuQHNjdS5lZHUuY24=
†These authors share first authorship
 Ruizhen Li
Ruizhen Li Xiaofen Li
Xiaofen Li Yan Wang
Yan Wang Chen Chang
Chen Chang Wanrui Lv
Wanrui Lv Xiaoying Li
Xiaoying Li Dan Cao
Dan Cao