- 1Department of Clinical Medical Sciences, Faculty of Medicine, Yarmouk University, Irbid, Jordan
- 2Faculty of Medicine, Yarmouk University, Irbid, Jordan
- 3Faculty of Medicine, Jordan University of Science and Technology, Irbid, Jordan
- 4Department of Anesthesia and Intensive Care, Al-Maqasid Charity Hospital, Amman, Jordan
- 5Department of Anesthesia and Intensive Care, University of Foggia, Foggia, Italy
- 6Department of Intensive Care, Hospital São Lucas da PUCRS, Porto Alegre, Brazil
Sepsis is the main cause of death among patients admitted to intensive care units. Management of sepsis includes fluid resuscitation, vasopressors, intravenous antimicrobials, source control, mechanical ventilation, and others. New insights into the potential benefits of non-antimicrobial drugs in sepsis have evolved based on the pathophysiology of the disease and the mechanism of action of some drugs, but the findings are still controversial. In this study, we aimed to evaluate the effect of beta-blockers, aspirin, statins, and heparin as adjunctive treatments in septic patients under mechanical ventilation with non-cardiovascular diseases and their effect on mortality. We searched PubMed with relevant keywords (beta-blockers, aspirin, statins, or heparin, and critically ill or sepsis) for the last 10 years and some personal collection of relevant articles, and then we assessed studies according to prespecified inclusion and exclusion criteria. Our results show that beta-blockers, aspirin, and heparin may have promising feedback on reducing mortality. However, new well-controlled, randomized, multicenter studies are needed to confirm that, and multiple issues regarding their usage need to be addressed. On the other hand, the feedback regarding the effectiveness of statins was not as strong as that of the other drugs studied, and we suggest that further research is needed to confirm these results.
Introduction
Modern sepsis definition is characterized as a severe organ dysfunction caused by an impaired host immune response to infection. Clinically, sepsis is defined by a documented focus of infection with at least 2 points or more in the Sequential Organ Failure Assessment (SOFA) (1) score. On the other hand, septic shock is an advanced sepsis stage with circulatory and metabolic derangements enough to increase mortality (2). Sepsis represented 19.7% of all global deaths with approximately 49 million incident cases in 2017 (3). In developing countries, sepsis incidence is estimated to be from 22 to 240/100 000 with a mortality rate of up to 30 %, which depends on the setting and disease severity (4). The cornerstone of sepsis management is supportive measures along with the administration of antimicrobial agents, source control, in addition to hemodynamic support when shock is present (5). Despite advances in care and recognition, sepsis remains a global public health concern due to its high mortality, high health-related risk, and economic burden (6).
As sepsis causes intense immunological, inflammatory, and coagulation derangements, in the last few years, therapies that could ameliorate organ dysfunction/failure in this setting were tested in these patient groups. Studies aimed at modulating and restoring the coagulation, immunological, and inflammatory response were conducted utilizing specific molecules, such as recombinant tissue factor pathway (7), recombinant C-protein (8), or synthetic endotoxin-like immunomodulators (9) but the results were disappointing.
Facing this scenario, successful therapies directed at other diseases were examined in septic patients, such as beta-blockers, aspirin, heparin, and statins, considering their biological plausibility in this setting.
As inflammation can augment myocardial oxygen consumption by excessive adrenergic stimulation, the role of beta-blockade has been studied focusing abate cardiovascular stress. However, there is a scarcity of data to confirm that these drugs could improve survival (10).
Platelets have an important role in sepsis pathophysiology, leading researchers to examine the role of drugs that act in the coagulation system, such as aspirin and heparin (11, 12).
In an in vivo study, pretreatment with simvastatin blunted the effects of TLR4 and TLR2 after a challenge with intravenous LPS in 20 healthy male subjects. These results suggested that statins may have a suppressive effect on these receptors and reduce cytokine actions (13) and that 80 mg of simvastatin for 7 days is associated with better outcomes in patients with community-acquired pneumonia (CAP) (14). Considering these possible beneficial effects, statins are studied in critically ill or septic patients to test if these results could be reproduced in the clinical scenario. The results were mixed (15, 16), and two recent meta-analyses showed no beneficial effect of this drug category in sepsis (17, 18).
Despite these findings, clinical research on beta-blocker, aspirin, heparin, and statins therapies in septic patients is still controversial, as the outcomes remain unclear and require further study. This study aimed to summarize the evidence on the use of these drugs on the outcome and prognosis in septic adult patients.
Methods
Data source and searches
Literature searches were performed using PubMed on 8 August 2022 and included the literature since 2012 as well as a personal collection of relevant articles.
Study selection
Titles were scanned and abstracts were reviewed in duplicate (AAH and HA). In the second stage, full-texts were examined. Any disagreements were adjudicated using a third reviewer (FSD).
Studies that compared heparin, aspirin, statins, and or beta-blockers to standard treatment in septic ICU patients were included. Search terms included: beta-blockers OR aspirin OR statins OR heparin AND critically ill OR sepsis; 141 primary references were obtained. The inclusion criteria were as follows:
(1) study population consisted of septic patients with non-cardiovascular diseases treated in the ICU, (2) studies published in the time frame between 2012 and 2022, (3) outcome of illness was measured, and (4) the language of studies in English only. The exclusion criteria were as follows: pregnant women and those less than 18 years of age were excluded.
Retrospective investigations, prospective observational studies, meta-analysis, systematic review, and randomized controlled trials were included; literature reviews were excluded.
It is worth mentioning that our study design, narrative review, does not follow a specific protocol, and no recommendations for clinical practice are needed.
Data extraction and quality assessment
The data were extracted and tabulated in duplicate (AAH and HA) using data collection headings as follows: article title, first author, place and year, study type/design, outcome, and intervention.
The certainty of evidence for each outcome was assessed using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) framework (19).
Results
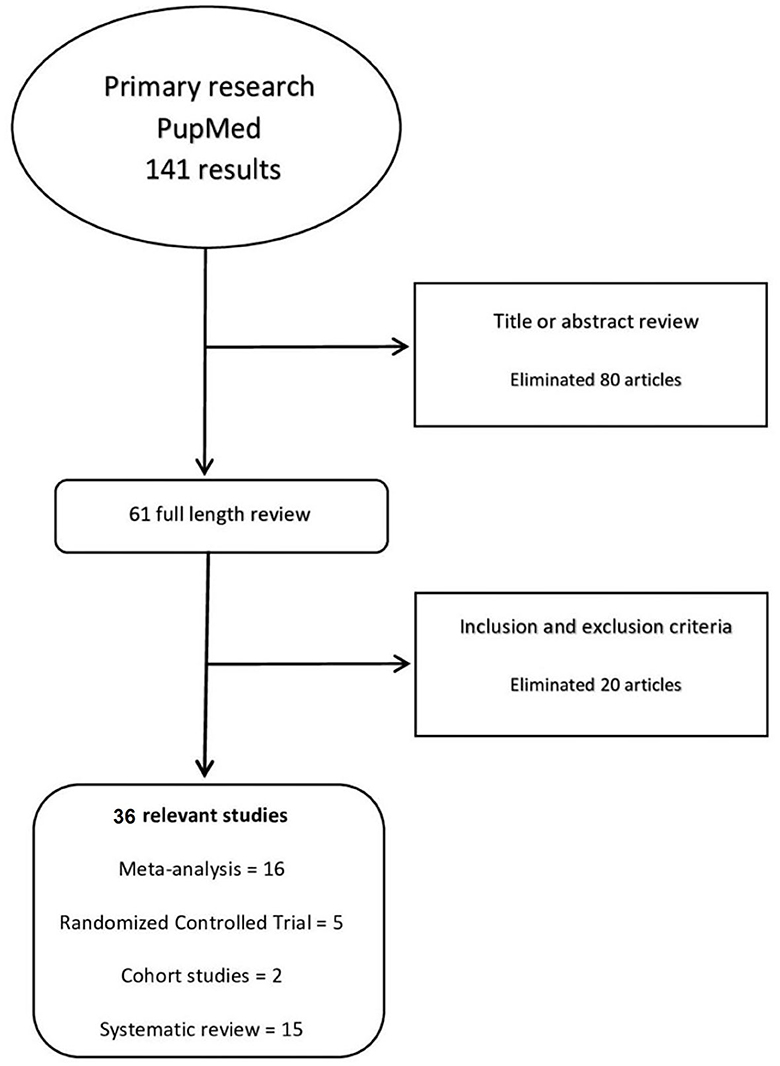
We reviewed 141 citations, reviewed 61 full-texts, and included 36 studies (n = 1,969,965) which showed in Figure 1.
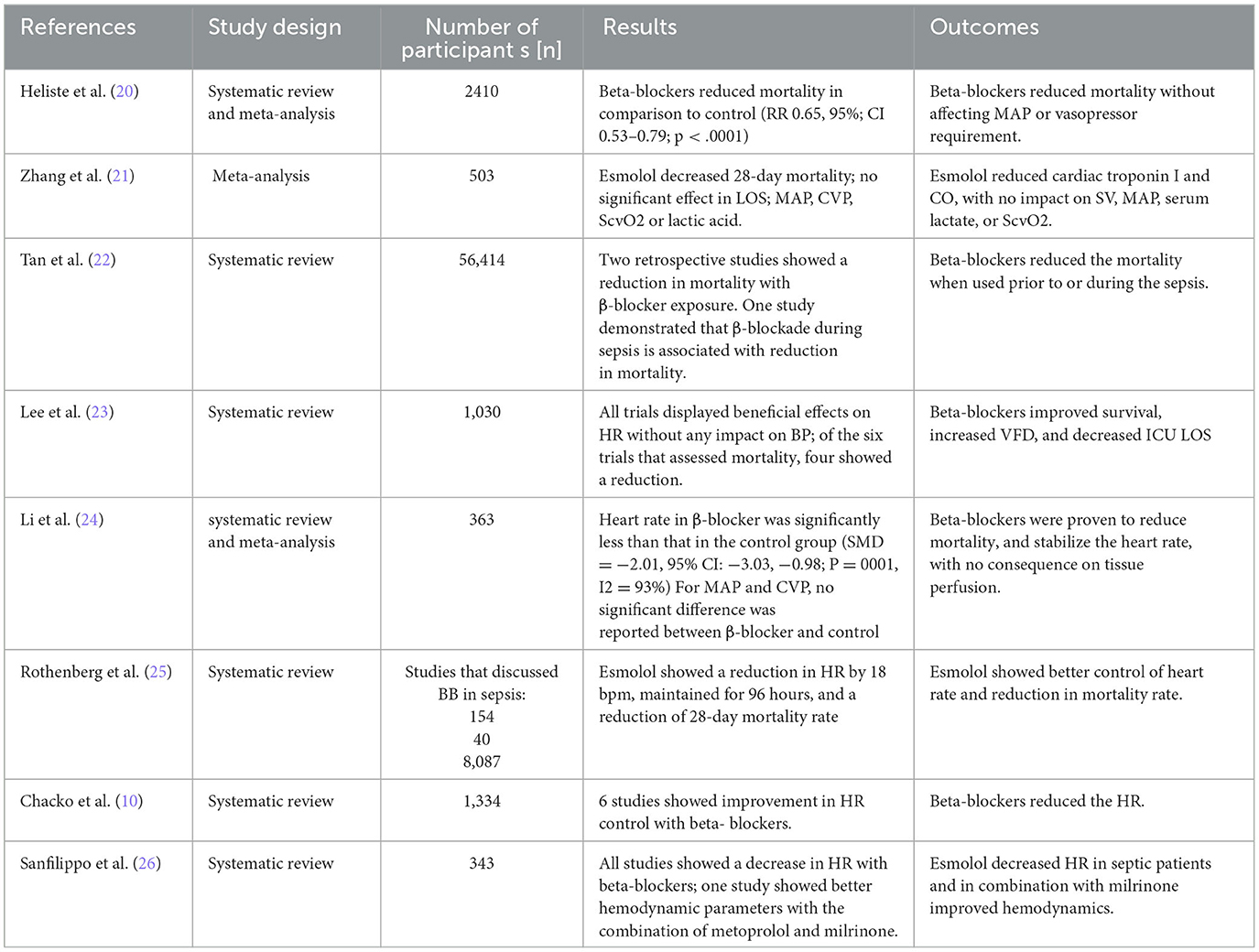
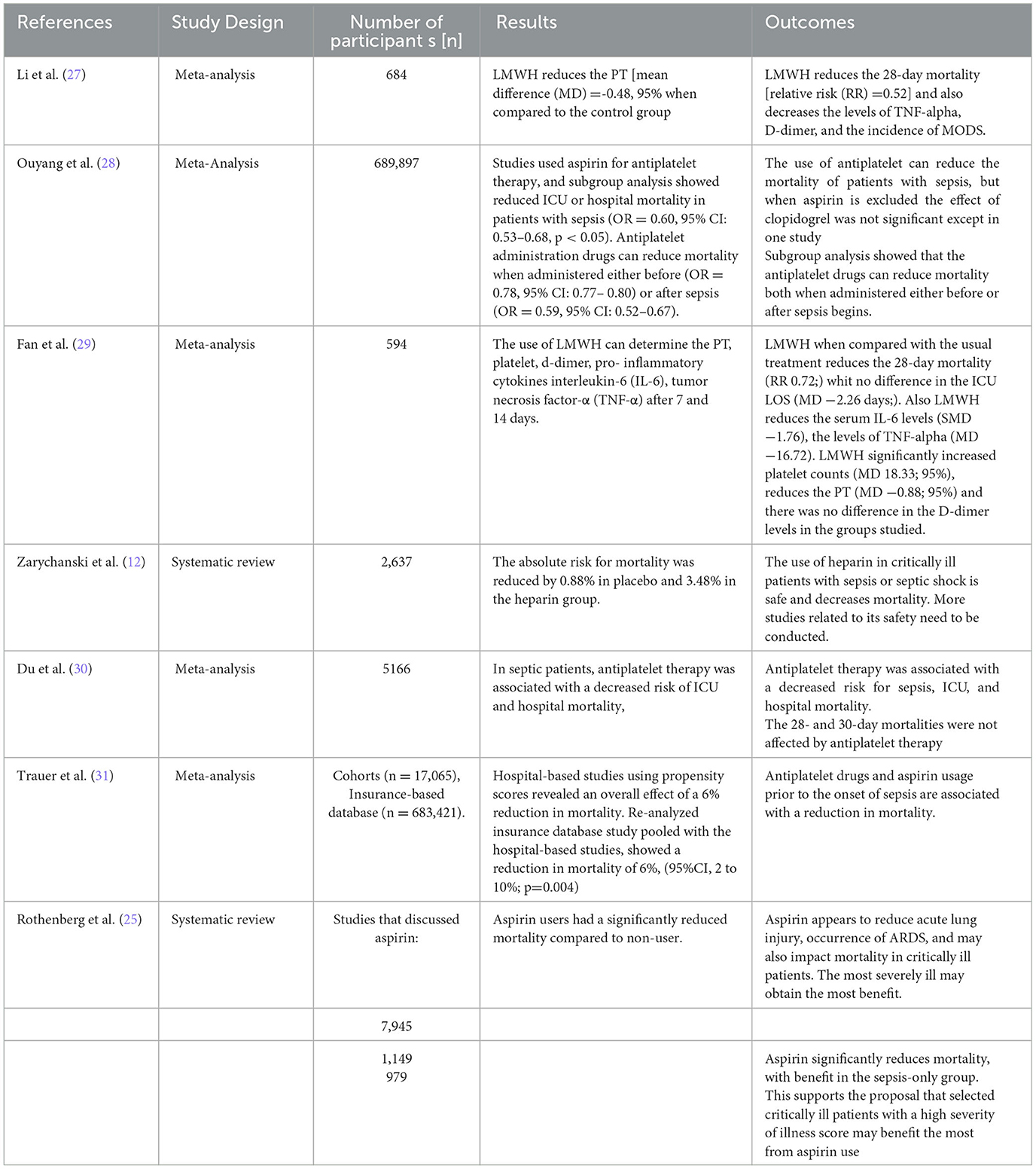
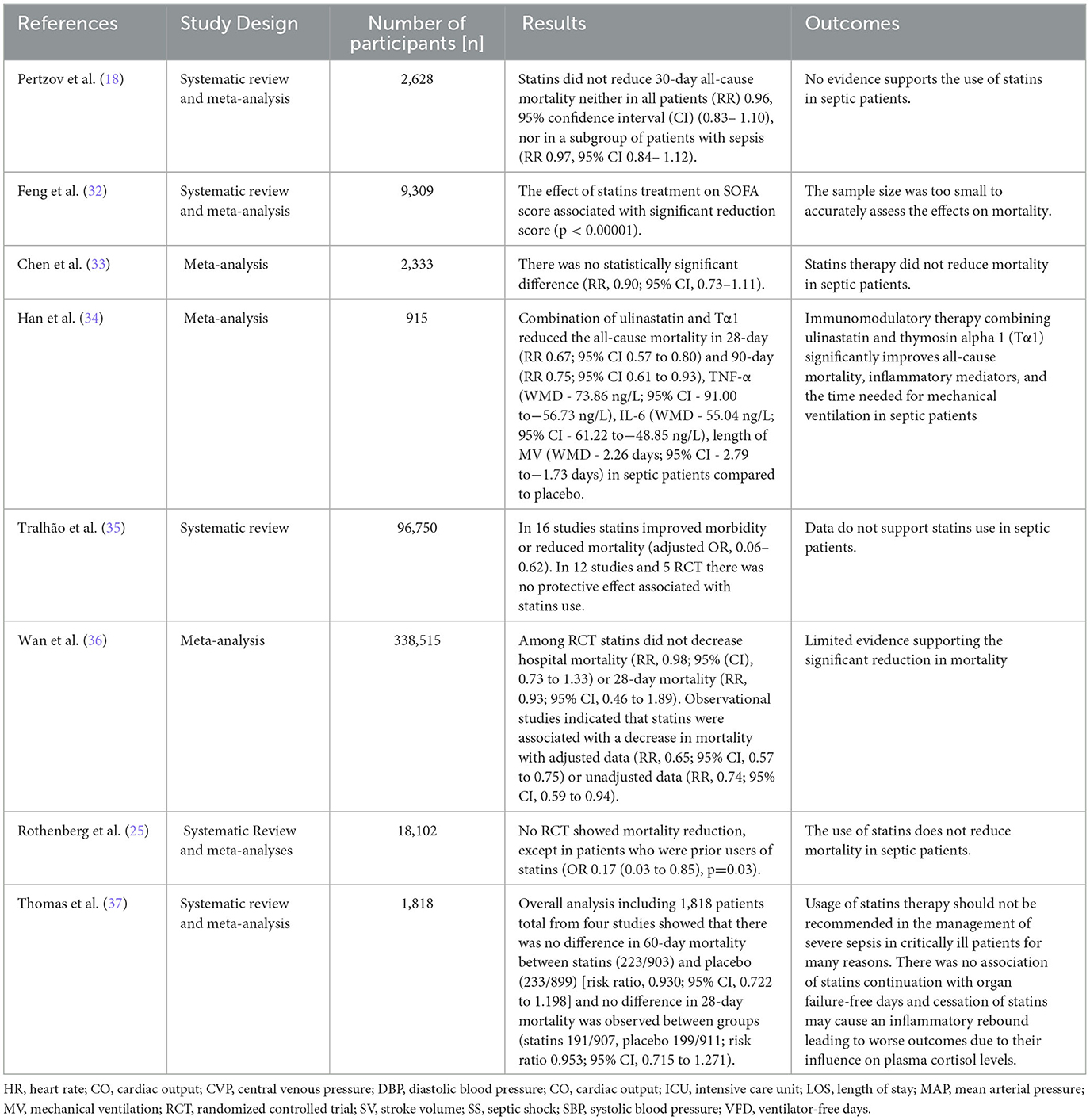
The characteristics of most of the included studies are reported in Tables 1–3.
Discussion
Management of sepsis is composed of fluid resuscitation, antimicrobial agents, source control, and organ support. However, recently the possibility of adjunctive treatments came into account depending on the pathophysiology of this disease (5). This study aimed to discuss four drugs basically related to heart disease management, but their mechanism of action brought them into consideration in the literature regarding sepsis management. We will discuss each drug by the use rationale and summarize the latest studies that investigated its use in sepsis.
Beta-blockers
Sinus tachycardia and atrial fibrillation are independent risk factors for mortality in septic patients (20, 38). Septic patients have high sympathetic stimulation due to increased endogenous and exogenous catecholamine concentrations (20). Exogenous catecholamines such as norepinephrine and dobutamine may increase the risk of tachyarrhythmia and thus mortality (39, 40). Decatecholaminisation by beta-blockers inhibits cardiogenic derangements (22), and controlling HR to be less than 95 beats per minute within 24 h improves outcomes (20), while beta-1 receptor blockade reduces HR. Moreover, the chronic use of beta-blockers before sepsis can improve survival (10, 22, 41). Similarly, many studies showed a reduction in mortality when beta-blockers were used during sepsis (20–25, 38, 41). Although a prospective cohort study that recruited 190 patients showed a non-significant reduction in 28-day mortality in the group of esmolol in comparison to the control group, which could be due to the small sample size, there was a significant control of HR, reduction in mechanical ventilation use, and improvement on tissue perfusion (42). Both selective and non-selective beta-blockers were reported to reduce mortality, and the use of ultrashort-acting cardio-selective beta-1 blockers could be important because of their rapid onset and short half-life (20, 25).
Sepsis-induced tachycardia contributes to cardiovascular failure, myocyte death, and dangerous arrhythmias; in this context, beta-blockers can reduce markers of cardiac injury (10) and CO, with no impact on stroke volume (10, 21). The reduction of CO could be due to a better control of HR, and the improvement in SV could be due to increased end-diastolic volume and improvement in contractility, which are also related to a controlled HR (10). Despite these findings, the effects of beta-blockers on cardiac biomarkers are heterogeneous (20). Other benefits attributable to beta-blockers use in sepsis are an increase in VFD (20, 23, 42) and a decrease in ICU LOS (21, 23, 25).
Regarding the concern of the effect of beta-blockers on tissue perfusion and oxygen utilization, two meta-analyses showed that these drugs did not impact lactate levels and ScvO2 (21, 24).
A meta-analysis that included seven RCTs using ultrashort-acting selective beta-blockers in sepsis showed a decrease in serum lactate levels in the esmolol and landiolol groups in comparison to the controls (38). These benefits in tissue perfusion were confirmed by studies with esmolol, showing a decrease in serum lactate and the central venous-to-arterial carbon dioxide difference (PcvaCO2 gap) (30, 42).
Despite beta-blockers did not cause a significant difference in mean arterial pressure in various reports, (20, 21, 30, 38, 42–44), Heliste et al. reported hypotension as an adverse event following beta-blocker administration, and they indicated proper monitoring of BP with their use (20).
An interesting finding is that beta-blockers did not increase the need for vasopressors (20, 21, 38), and surprisingly, an RCT showed a decrease in the vasopressor requirements (45).
Aspirin and heparin
Sepsis is an acute hypercoagulable state associated with platelet consumption and massive thrombosis leading to venous thromboembolism and disseminated intravascular coagulopathy (DIC) (31).
Aspirin is widely used to prevent and treat vascular diseases such as myocardial infarction and stroke, by irreversibly inhibiting cyclooxygenase-1 which is a key component in the activation of platelets aggregation through thromboxane A2 production (11).
A study examined a low-dose aspirin strategy as a method to limit organ failure by interrupting the coagulation cascade. Aspirin reduced mortality in critically ill patients and this could be related to its role in coagulopathy treatment (25).
Similarly, Ouyang et al. and Rothenberg et al. reviewed the effect of antiplatelet use on the prognosis of patients with sepsis and showed that the use of these drugs can reduce mortality in patients with sepsis (25, 28, 46). This supports that selected critically ill patients with a high illness severity may benefit from aspirin use (25). Consistently, the potential benefits of aspirin use in septic patients were further supported by Du et al., who demonstrated that the antiplatelet therapy was associated with a decreased risk of hospital and intensive care unit (ICU) mortality, but 28- and 30-day mortality were not affected by antiplatelet therapy (47). Moreover, a meta-analysis that included 15 studies that described hospital-based cohorts and one that was a large insurance-based database found a reduction in mortality ranging from 2% to 12% among patients hospitalized for sepsis who had prior aspirin treatment. In addition, a combined analysis of all available observational data showed that antiplatelet drugs and aspirin usage in isolation prior to the onset of sepsis are associated with a reduction in mortality. However, we should account that the large sample size in the insurance database study had a considerable impact on this conclusion (31).
Regarding the timing of aspirine usage in sepsis, a subgroup analysis showed that the antiplatelet drugs can reduce mortality both when administered either before “preventive” (OR = 0.78) or after sepsis “additional” (OR = 0.59) (28). A 10-year retrospective population-based cohort study involving approximately 53,000 sepsis hospital admissions revealed that current aspirin users, who had taken aspirin within 90 days prior to admission, had a lower 90-day mortality rate and a longer mean survival time than past users and non-users (48).
On the other hand, it was found that aspirin therapy was associated with negative secondary outcomes such as a higher risk of ICU-acquired sepsis, increased mechanical ventilation duration, ICU LOS, and thus increased morbidity (31). Ouyang et al. found that the secondary outcomes, which included the duration of mechanical ventilation, the incidence of acute respiratory distress syndrome (ARDS), and the incidence of AKI, needed for renal replacement therapy (RRT) and ICU LOS varied and most of them showed no significant difference (28). Nevertheless, other researchers found that aspirin appears to reduce acute lung injury, occurrence of ARDS, and mortality in critically ill patients and that the most severely ill patients may obtain the most benefit (25); however, these results need to be confirmed by further clinical trials.
Heparin is an anticoagulant drug used to prevent venous thromboembolism (VTE) events by augmenting the effect of antithrombin III, which inhibits several clotting factors, mainly the level of Xa and IIa (1:1 ratio) factors (49).
Comparing heparin to placebo, a systematic review concluded that the risk ratio for mortality in septic patients treated with heparin was reduced in comparison to the control group (12). Consistently, some studies demonstrated that the use of heparin decreases the 28-day mortality [odds ratio (OR) = 0.59] and the multi-organ dysfunction syndrome MODS (OR = 0.32,) in septic patients compared to the control group (27, 29). In addition, a clinical trial conducted on an extracorporeal blood filter (Seraph 100) containing heparin resulted in a faster resolution of bloodstream infections when added to antibiotic therapy for patients with bacteremia (50).
However, the positive effect of heparin use in sepsis is debated. Although some authors reported shorter ICU stays of septic patients treated with heparin, others found no significant difference (29). Despite the positive outcomes of heparin use in septic patients' mortality, it increases the incidence of bleeding when compared to the usual method of treatment (27, 29). Moreover, a meta-analysis conducted on 1,340 patients showed no significant differences in mortality and bleeding complications in septic DIC patients treated with anticoagulants, one of which is heparin, compared to placebo (51).
Sepsis-associated DIC is considered a main indication for anticoagulant use in septic patients. A study conducted with concomitant usage of heparin and antithrombin (AT) supplementation in 15.7% of 159 septic patients with DIC found that heparins were related to a higher survival rate than those who were treated without heparins, but the difference was not statistically significant (84.0% vs. 70.9%, P = 0.22) (52).
Moreover, heparin is associated with a lower rate of mortality in DIC positive with high-risk septic patients (SOFA score 13–17), but not in the low-risk to moderate-risk patients (SOFA score ≤ 12). Furthermore, Kazuma et al. demonstrated better outcomes in septic patients with organ dysfunction than those without organ dysfunction treated with anticoagulant. Although the differences were not statistically significant, bleeding was the major unfavorable outcome associated with anticoagulant administration (53).
Statins
Statins, also known as HMG-CoA reductase inhibitors, are a commonly prescribed class of drugs to lower cholesterol levels in the blood. Statins reduce the risk of cardiovascular diseases by lowering low-density lipoprotein cholesterol (LDL-c) and increasing high-density lipoprotein-cholesterol (HDL-c). The main mechanism of statins is by competitively inhibiting the active site of the rate-limiting enzyme in the mevalonate pathway, HMG-CoA reductase, thus preventing the conversion of HMG-CoA to mevalonic acid, which reduces cholesterol synthesis within the liver and consequently the levels in the bloodstream. In addition to their cholesterol-lowering effects, statins have been demonstrated to improve endothelial function, stabilize atherosclerotic plaques, and have anti-inflammatory, immunomodulatory, and antithrombotic effects (54).
On one hand, limited studies revealed the benefits of statins use in septic and critically ill patients. An immunomodulatory therapy combining ulinastatin and thymosin alpha 1 (Tα1) significantly improves all-cause mortality, inflammatory mediators, and the time needed for mechanical ventilation in septic patients (34). A cohort study analyzing data from Taiwan's National Health Insurance Research Database found that simvastatin and atorvastatin were associated with improved 30-day survival in sepsis patients, while rosuvastatin did not show efficacy in preventing mortality. This study suggests that the impact of statins on sepsis outcome is associated with drug-specific effects rather than with statins' lipid-lowering potency (55). A systemic review and meta-analysis suggested a possible benefit of statins use by lowering SOFA among septic patients, as well as an increase in VFD, but mortality and sepsis risk remained the same. Similarly, statins were found to decrease the need for ventilation among ARDS patients. Despite the mentioned results, the sample size was too small to accurately assess the effects on mortality and sepsis (32). Moreover, the pleiotropic benefit effect of statins including antioxidant, anti-inflammatory, and antithrombotic effects may contribute to nephroprotection in AKI in the ICU (56).
On the other hand, several studies showed no effect of statins in reducing mortality when compared to placebo. In one meta-analysis based on nine prospective randomized trials, it was shown that statins therapy did not reduce mortality in septic patients compared with placebo-controls (33). A systemic review concluded that there were insufficient results supporting statins use in septic patients due to inconsistent mortality reduction, which was evident in the studies collected (35). Another systemic review and meta-analysis recommended caution in using statins in treating infections and sepsis as there seems to be limited evidence supporting the significant reduction in mortality (36). Consistently, a prospective, randomized, and controlled trial showed that statins appeared not to have any benefit in reducing mortality or illness severity in septic patients. Thus, recent systemic reviews and meta-analyses do not recommend the use of statins in reducing mortality in patients with sepsis. As a result, this impacts patients who have other indications for statins, such as cardiovascular disease (25).
Along with these findings, other studies recommended against the usage of statins in sepsis therapy. A systemic review and meta-analysis study of 1,818 patients confirmed that the usage of statins therapy should not be recommended in the management of sepsis (33). The use of statins therapy in adults for the indication of sepsis is not recommended, as results showed no reduction in 30-day all-cause mortality neither in all patients nor in a subgroup of patients who used statins in sepsis therapy (18).
Regarding the use of statins in septic patients with organ dysfunction, Joannidis et al. concluded that there was no improvement in clinical outcomes or 60 days mortality between septic patients with ARDS treated using rosuvastatin when compared to a placebo (56). Consistently, a prospective study showed an increased mortality rate and no effect of rosuvastatin on the outcomes in patients with sepsis-associated ARDS (18). Moreover, there were no consistent findings in a study assessing statins for ALI in septic patients using rosuvastatin (57). Additionally, there was no association of statins continuation with organ failure-free days (37), and rosuvastatin use showed small differences in organ failure-free days when compared to a placebo suggesting a specious argument in their effect on organ failure prevention (56). Regarding liver enzymes, it was reported that aspartate aminotransferase (AST) levels in patients taking rosuvastatin for sepsis associated with ARDS were elevated. Although they concluded that rosuvastatin was not associated
with the increase in serum creatinine kinase levels, some patients with elevated AST levels were having related elevations in creatine kinase and alanine aminotransferase (ALT) levels, which suggested the contribution of rosuvastatin in renal and hepatic failure (56). Furthermore, statins therapy was shown not to influence the plasma cortisol profiles in patients with severe sepsis leading to prolonged inflammatory response, hospital stay, and morality (58). However, the cessation of statins may cause an inflammatory rebound leading to worse outcomes due to their influence on plasma cortisol levels (37).
Conclusion
Our narrative review does not recommend either in favor or against using beta-blockers, aspirin, heparin, or statins in the management of sepsis. However, beta-blockers showed benefits in improving hemodynamics and reducing mortality and their safety on tissue perfusion supporting their use in selected septic patients.
The target hemodynamic parameters and the choice of beta-blocker need further studies. Additionally, aspirin and heparin have promising feedback on improving survival, but more studies are needed to support their use. Most studies recommended against the use of statins in septic patients. In conclusion, there were variable results regarding these drug effects on sepsis outcome, mortality, and prognosis. We strongly suggest further studies be conducted on this field to clear the controversy.
Limitations
This study was limited in several ways: (1) the potential confounding factors such as the varying dose and treatment period of each drug; (2) the combination with other drugs as anticoagulant increase the heterogeneity among the included study; (3) as sepsis is a polygenic disease, we did not approach the individual and genomic influence on pharmacological actions in septic patients; (4) in addition to the language limitations, only databases in English were concluded in this study.
Author contributions
AA, LA-H, and FD conceived and designed the manuscript. AA, HA, BA, MA, OQ, ZA, GF, SH, and IQ analyzed the data and wrote the manuscript. AA, HA, LA-H, FD, and GC reviewed and edited the data analysis and writing. All authors have read and approved the final version of this manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Vincent JL, Moreno R, Takala J, Willatts S, De Mendonça A, Bruining H, et al. The SOFA (Sepsis-related organ failure assessment) score to describe organ dysfunction/failure. Intensive Care Med. (1996) 22:707–10. doi: 10.1007/BF01709751
2. Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. (2016) 315:801–10. doi: 10.1001/jama.2016.0287
3. Rudd KE, Johnson SC, Agesa KM, Shackelford K, Isol D, Kievlan D, et al. Global, regional, and national sepsis incidence and mortality, 1990-2017: analysis for the Global Burden of Disease Study. Lancet. (2020) 395:200–11. doi: 10.1016/S0140-6736(19)32989-7
4. Jawad I, Lukšić I, Rafnsson SB. Assessing available information on the burden of sepsis: global estimates of incidence, prevalence and mortality. J Glob Health. (2012) 2:010404. doi: 10.7189/jogh.01.010404
5. Evans L, Rhodes A, Alhazzani W, Antonelli M, Coopersmith CM, French C, et al. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock 2021. Intensive Care Med. (2021) 47:1181–247. doi: 10.1007/s00134-021-06506-y
6. Reinhart K, Daniels R, Kissoon N, Machado FR, Schachter RD, Finfer S. Recognizing sepsis as a global health priority - a WHO resolution. N Engl J Med. (2017) 377:414–7. doi: 10.1056/NEJMp1707170
7. Abraham E, Reinhart K, Opal S, Demeyer I, Doig C, Rodriguez AL, et al. Efficacy and safety of tifacogin (recombinant tissue factor pathway inhibitor in severe sepsis. JAMA. (2003) 290:238–47. doi: 10.1001/jama.290.2.238
8. Ranieri VM, Thompson BT, Barie PS, Dhainaut J-F, Douglas IS, Finfer S, et al. Drotrecogin alfa (Activated) in adults with septic shock. N Engl J Med. (2012) 366:2055–964. doi: 10.1056/NEJMoa1202290
9. Opal SM, Laterre P-F, Francois B, LaRosa SP, Angus DC, Mira J-P, et al. Effect of eritoran, an antagonist of MD2-TLR4, on mortality in patients with severe sepsis. JAMA. (2013) 309:1154–62. doi: 10.1001/jama.2013.2194
10. Chacko CJ, Gopal S. Systematic review of use of β-blockers in sepsis. J Anaesthesiol Clin Pharmacol. (2015) 31:460–5. doi: 10.4103/0970-9185.169063
11. Hsu J, Donnelly JP, Chaudhary NS, Moore JX, Safford MM, Kim J, et al. Aspirin use and long-term rates of sepsis: a population-based cohort study. PLoS ONE. (2018) 13:e0194829. doi: 10.1371/journal.pone.0194829
12. Zarychanski R, Abou-Setta AM, Kanji S, et al. Canadian Critical Care Trials Group. The efficacy and safety of heparin in patients with sepsis: a systematic review and meta-analysis. Crit Care Med. (2015) 43:511–8. doi: 10.1097/CCM.0000000000000763
13. Niessner A, Steiner S, Speidl WS, Pleiner J, Seidinger D, Maurer G, et al. Simvastatin suppresses endotoxin-induced upregulation of toll-like receptors 4 and 2 in vivo. Atherosclerosis. (2006) 189:408–13. doi: 10.1016/j.atherosclerosis.2005.12.022
14. Sapey E, Patel JM, Greenwood H, Walton GM, Grudzinska F, Parekh D, et al. Simvastatin improves neutrophil function and clinical outcomes in pneumonia. Am J Respir Crit Care Med. (2019) 200:1282–93. doi: 10.1164/rccm.201812-2328OC
15. Harbi SAA, Tamim HM, Arabi YM. Association between statin therapy and outcomes in critically ill patients: a nested cohort study. BMC Clin Pharmacol. (2011) 11:12. doi: 10.1186/1472-6904-11-12
16. Novack V, Eisinger M, Frenkel A, Terblanche M, Adhikari NKJ, Douvdevani A, et al. The effects of statin therapy on inflammatory cytokines in patients with bacterial infections: a randomized double-blind placebo controlled clinical trial. Intensive Care Med. (2009) 35:1255–60. doi: 10.1007/s00134-009-1429-0
17. Pasin L, Landoni G, Castro ML, Cabrini L, Belletti A, Feltracco P, et al. The effect on statins on mortality in septic patients: a meta- analysis of randomized controlled trials. PLoS ONE. (2013) 8:e82775. doi: 10.1371/journal.pone.0082775
18. Pertzov B, Eliakim-Raz N, Atamna H, Trestioreanu AZ, Yahav D, Leibovici L. Hydroxymethylglutaryl-CoA reductase inhibitors (statins) for the treatment of sepsis in adults—a systematic review and meta-analysis. Clin Microbiol Infect. (2019) 25:280–9. doi: 10.1016/j.cmi.2018.11.003
19. Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE working group. GRADE: an emerging consensus on rating quality of evidence and strength of recommendations. BMJ. (2008) 336:924–6. doi: 10.1136/bmj.39489.470347.AD
20. Heliste M, Pettilä V, Berger D, Jakob SM, Wilkman E. Beta-blocker treatment in the critically ill: a systematic review and meta-analysis. Ann Med. (2022) 54:1994–2010. doi: 10.1080/07853890.2022.2098376
21. Zhang J, Chen C, Liu Y, Yang Y, Yang X, Yang J. Benefits of esmolol in adults with sepsis and septic shock: an updated meta-analysis of randomized controlled trials. Medicine. (2022) 101:e29820. doi: 10.1097/MD.0000000000029820
22. Tan K, Harazim M, Tang B, Mclean A, Nalos M. The association between premorbid beta blocker exposure and mortality in sepsis-a systematic review. Crit Care. (2019) 23:298. doi: 10.1186/s13054-019-2562-y
23. Lee YR, Seth MS, Soney D, Dai H. Benefits of Beta-Blockade In Sepsis And Septic Shock: A Systematic Review. Clin Drug Investig. (2019) 39:429–40. doi: 10.1007/s40261-019-00762-z
24. Li J, Sun W, Guo Y, Ren Y, Li Y, Yang Z. Prognosis of β-adrenergic blockade therapy on septic shock and sepsis: a systematic review and meta-analysis of randomized controlled studies. Cytokine. (2020) 126:154916. doi: 10.1016/j.cyto.2019.154916
25. Rothenberg FG, Clay MB, Jamali H, Vandivier-Pletsch RH. Systematic review of β blocker, aspirin, and statin in critically ill patients: importance of severity of illness and cardiac troponin. J Investig Med. (2017) 65:747–53. doi: 10.1136/jim-2016-000374
26. Sanfilippo F, Santonocito C, Morelli A, Foex P. Beta-blocker use in severe sepsis and septic shock: a systematic review. Curr Med Res Opin. (2015) 31:1817–25. doi: 10.1185/03007995.2015.1062357
27. Li X, Liu Z, Luo M, Xi Y, Li C, Wang S, et al. Therapeutic effect of low-molecular-weight heparin on adult sepsis: a meta- analysis. Ann Palliat Med. (2021) 10:3115–27. doi: 10.21037/apm-21-169
28. Ouyang Y, Wang Y, Liu B, Ma X, Ding R. Effects of antiplatelet therapy on the mortality rate of patients with sepsis: a meta-analysis. J Crit Care. (2019) 50:162–8. doi: 10.1016/j.jcrc.2018.12.004
29. Fan Y, Jiang M, Gong D, Zou C. Efficacy and safety of low-molecular-weight heparin in patients with sepsis: a meta-analysis of randomized controlled trials. Sci Rep. (2016) 6:25984. doi: 10.1038/srep25984
30. Du W, Wang XT, Long Y, Liu DW. Efficacy and safety of esmolol in treatment of patients with septic shock. Chin Med J (Engl). (2016) 129:1658–65. doi: 10.4103/0366-6999.185856
31. Trauer J, Muhi S, McBryde ES, Harbi SAA, Arabi YM, Boyle AJ, et al. Quantifying the effects of prior acetyl-salicylic acid on sepsis- related deaths: an individual patient data meta-analysis using propensity matching. Crit Care Med. (2017) 45:1871–9. doi: 10.1097/CCM.0000000000002654
32. Feng Y. Efficacy of statin therapy in patients with acute respiratory distress syndrome/acute lung injury: a systematic review and meta-analysis. Eur Rev Med Pharmacol Sci. (2018) 22:3190–8.
33. Chen M, Ji M, Si X. The effects of statin therapy on mortality in patients with sepsis: a meta-analysis of randomized trials. Medicin. (2018) 97:e11578. doi: 10.1097/MD.0000000000011578
34. Han D, Shang W, Wang G, Sun L, Zhang Y, Wen H, et al. Ulinastatin- and thymosin α1-based immunomodulatory strategy for sepsis: a meta-analysis. Int Immunopharmacol. (2015) 29:377–82. doi: 10.1016/j.intimp.2015.10.026
35. Tralhão AF, Cés de. Souza-Dantas V, Salluh JI, Póvoa PM. Impact of statins in outcomes of septic patients: a systematic review. Postgrad Med. (2014) 126:45–58. doi: 10.3810/pgm.2014.11.2832
36. Wan YD, Sun TW, Kan QC, Guan FX, Zhang SG. Effect of statin therapy on mortality from infection and sepsis: a meta-analysis of randomized and observational studies. Crit Care. (2014) 18:R71. doi: 10.1186/cc13828
37. Thomas G, Hraiech S, Loundou A, Truwit J, Kruger P, Mcauley DF, et al. Statin therapy in critically-ill patients with severe sepsis: a review and meta-analysis of randomized clinical trials. Minerva Anestesiol. (2015) 81:921−30.
38. Hasegawa D, Sato R, Prasitlumkum N, Nishida K, Takahashi K, Yatabe T, et al. Effect of ultrashort-acting β-blockers on mortality in patients with sepsis with persistent tachycardia despite initial resuscitation: a systematic review and meta-analysis of randomized controlled trials. Chest. (2021) 159:2289–300. doi: 10.1016/j.chest.2021.01.009
39. Schmittinger CA, Torgersen C, Luckner G, Schröder DC, Lorenz I, Dünser MW. Adverse cardiac events during catecholamine vasopressor therapy: a prospective observational study. Intensive Care Med. (2012) 38:950–8. doi: 10.1007/s00134-012-2531-2
40. Wilkman E, Kaukonen KM, Pettilä V, Kuitunen A, Varpula M. Association between inotrope treatment and 90-day mortality in patients with septic shock. Acta Anaesthesiol Scand. (2013) 57:431–42. doi: 10.1111/aas.12056
41. Hasegawa D, Sato R, Prasitlumkum N, Nishida K. Effect of premorbid beta-blockers on mortality in patients with sepsis: a systematic review and meta-analysis. J Intensive Care Med. (2022) 3:908–16. doi: 10.1177/08850666211052926
42. Shang X, Wang K, Xu J, Gong S, Ye Y, Chen K, et al. The effect of esmolol on tissue perfusion and clinical prognosis of patients with severe sepsis: a prospective cohort study. Biomed Res Int. (2016) 2016:1038034. doi: 10.1155/2016/1038034
43. Morelli A, Donati A, Ertmer C, Rehberg S, Kampmeier T, Orecchioni A, et al. Microvascular effects of heart rate control with esmolol in patients with septic shock: a pilot study. Crit Care Med. (2013) 41:2162–8. doi: 10.1097/CCM.0b013e31828a678d
44. Liu P, Wu Q, Tang Y, Zhou Z, Feng M. The influence of esmolol on septic shock and sepsis: a meta- analysis of randomized controlled studies. Am J Emerg Med. (2018) 36:470–4. doi: 10.1016/j.ajem.2017.11.013
45. Wang Z, Wu Q, Nie X, Guo J, Yang C. Combination therapy with milrinone and esmolol for heart protection in patients with severe sepsis: a prospective, randomized trial. Clin Drug Investig. (2015) 35:707–16. doi: 10.1007/s40261-015-0325-3
46. Dewitte A, Lepreux S, Villeneuve J, Rigotheir C, Combe C, Ouattara A, et al. Blood platelets and sepsis pathophysiology: a new therapeutic prospect in critically [corrected] ill patients? Ann Intensive Care. (2017) 7:115. doi: 10.1186/s13613-017-0337-7
47. Du F, Jiang P, He S, Song D, Xu F. Antiplatelet therapy for critically ill patients: a pairwise and bayesian network meta-analysis. Shock. (2018) 49:616–24. doi: 10.1097/SHK.0000000000001057
48. Hsu W-T, Porta L, Chang I-J, Dao Q-L, Tehrani BM, Hsu T-C, et al. Association between aspirin use and sepsis outcomes: a national cohort study. Anesth Analg. (2022) 135:110–7. doi: 10.1213/ANE.0000000000005943
49. Harter K, Levine M, Henderson SO. Anticoagulation drug therapy: a review. West J Emerg Med. (2015) 16:11–7. doi: 10.5811/westjem.2014.12.22933
50. Eden G, Schmidt JJ, Büttner S, Kümpers P, Hafer C, Rovas A, et al. Safety and efficacy of the Seraph 100 Microbind Affinity Blood Filter to remove bacteria from the blood stream: results of the first in human study. Crit Care. (2022) 26:181. doi: 10.1186/s13054-022-04044-7
51. Yatabe T, Inoue S, Sakamoto S, Sumi Y, Nishida O, Hayashida K, et al. The anticoagulant treatment for sepsis induced disseminated intravascular coagulation; network meta-analysis. Thromb Res. (2018) 171:136–42. doi: 10.1016/j.thromres.2018.10.007
52. Iba T, Gando S, Saitoh D, Ikeda T, Anan H, Oda S, et al. Efficacy and bleeding risk of antithrombin supplementation in patients with septic disseminated intravascular coagulation: a third survey. Clin Appl Thromb Hemost. (2017) 23:422–8. doi: 10.1177/1076029616648405
53. Yamakawa K, Umemura Y, Hayakawa M, Kudo D, Sanui M, Takahashi H, et al. Japan septic disseminated intravascular coagulation (J-Septic DIC) study group. Benefit profile of anticoagulant therapy in sepsis: a nationwide multicentre registry in Japan. Crit Care. (2016) 20:229. doi: 10.1186/s13054-016-1415-1
54. Eladawy S, Bazan N. Future of statins in sepsis: a review on its safety and efficacy. Arch Pharm Sci ASU. (2020) 4:20–35. doi: 10.21608/APS.2020.2001.1022
55. Lee CC, Lee MG, Hsu TC, Porta L, Chang SS, Yo CH, et al. A population-based cohort study on the drug-specific effect of statins on sepsis outcome. Chest. (2018) 153:805–15. doi: 10.1016/j.chest.2017.09.024
56. Joannidis M, Druml W, Forni LG, Groeneveld ABJ, Honore PM, Hoste E, et al. Prevention of acute kidney injury and protection of renal function in the intensive care unit: update 2017: expert opinion of the Working Group on Prevention, AKI section, European Society of Intensive Care Medicine. Intensive Care Med. (2017) 43:730–49. doi: 10.1007/s00134-017-4832-y
57. Hashem M, Hopkins R, Colantuoni E, Dinglas VD, Sinhas P, Friedman LA, et al. Six-month and 12-month patient outcomes based on inflammatory subphenotypes in sepsis-associated ARDS: secondary analysis of SAILS-ALTOS trial. Thorax. (2022) 77:22–30. doi: 10.1136/thoraxjnl-2020-216613
58. Venkatesh B, Imeson L, Kruger P, Cohen J, Jones M, Bellomo R. Australian and New Zealand Intensive Care Society Clinical Trials Group. STATInS Trial Investigators Elevated plasma-free cortisol concentrations and ratios are associated with increased mortality even in the presence of statin therapy in patients with severe sepsis. Crit Care Med. (2015) 43:630–5. doi: 10.1097/CCM.0000000000000750
Keywords: sepsis, aspirin, beta-blocker, statin, heparin
Citation: Al-Husinat L, Abu Hmaid A, Abbas H, Abuelsamen B, Albelbisi M, Haddad S, Qamileh I, Quneis O, Al Modanat ZJ, Ferrara G, Dias FS and Cinnella G (2023) Role of aspirin, beta-blocker, statins, and heparin therapy in septic patients under mechanical ventilation: a narrative review. Front. Med. 10:1143090. doi: 10.3389/fmed.2023.1143090
Received: 12 January 2023; Accepted: 22 June 2023;
Published: 10 July 2023.
Edited by:
Simone Gattarello, University Medical Center Göttingen, GermanyReviewed by:
Meity Ardiana, Airlangga University, IndonesiaFan Zhang, Shandong University, China
Wan-Ting Hsu, Harvard University, United States
Copyright © 2023 Al-Husinat, Abu Hmaid, Abbas, Abuelsamen, Albelbisi, Haddad, Qamileh, Quneis, Al Modanat, Ferrara, Dias and Cinnella. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lou'i Al-Husinat, bG91aS5odXNpbmF0QHl1LmVkdS5qbw==
 Lou'i Al-Husinat
Lou'i Al-Husinat Amer Abu Hmaid
Amer Abu Hmaid Hadeel Abbas
Hadeel Abbas Batool Abuelsamen2
Batool Abuelsamen2 Mutaz Albelbisi
Mutaz Albelbisi Said Haddad
Said Haddad Ibrahim Qamileh
Ibrahim Qamileh Ossaid Quneis
Ossaid Quneis Zaid Jehad Al Modanat
Zaid Jehad Al Modanat Giuseppe Ferrara
Giuseppe Ferrara Fernando Suparregui Dias
Fernando Suparregui Dias Gilda Cinnella
Gilda Cinnella