
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Mar. Sci. , 27 March 2025
Sec. Aquatic Physiology
Volume 12 - 2025 | https://doi.org/10.3389/fmars.2025.1567872
This article is part of the Research Topic Nutritional Metabolism and Immunity of Aquatic Animals View all 8 articles
 Qin-Cheng Huang1,2†
Qin-Cheng Huang1,2† Li Wang1
Li Wang1 Zhi-Min Gu1*
Zhi-Min Gu1* Cui Liu1
Cui Liu1 Tian-Tian Ye1
Tian-Tian Ye1 Jun-Jun Yan1
Jun-Jun Yan1 Ji-Lun Meng1
Ji-Lun Meng1 Yu-Tong Zheng1
Yu-Tong Zheng1 Xian-Ping Ge2*
Xian-Ping Ge2* Zhi-Deng Lin3
Zhi-Deng Lin3 Guo-Yuan Zou4
Guo-Yuan Zou4The effect of dietary retinoic acid (RA) on the growth, lipid deposition, oxidation resistance, immunity, hepatopancreatic and intestinal health of Macrobrachium rosenbergii was evaluated. A total of 1200 prawns (0.22 ± 0.00 g) were divided into six groups and fed their corresponding feed containing 4, 132, 296, 562, 1206 or 2562 mg/kg dietary RA. The weight gain rate, specific growth rate and final body weight of the prawns changed linearly and quadratically, with the maximum observed in those fed 296 mg/kg dietary RA. Increasing dietary RA linearly and quadratically raised the RA content in the whole body. Compared to the prawns fed 4 mg/kg dietary RA, there was significantly lower lipid deposition in the muscle, hepatopancreas and whole body of prawns fed 296 mg/kg dietary RA. Prawns fed 296 mg/kg dietary RA had lower triglyceride (TG) content in the hepatopancreas with upregulated gene expression of retinoid X receptor (RXR), diacylglycerol acyltransferase 1 (dgat1) and carnitine palmitoyltransferase 1 (cpt1). Additionally, 296 mg/kg dietary RA increased the protein expression of RXR, CAMKKβ and phospho-AMPK. A dietary RA level of 296 mg/kg could decrease oxidative stress by upregulating the expression of peroxiredoxin 5 (prx5) and improve immunity by upregulating expression of toll-like receptor 2 (toll2), myeloid differentiation factor 88 (myd88) and dosal in the hepatopancreas. The intestinal health related genes (crustin 2/3/4, anti-lipopolysaccharide factor 1/7, prx5, peritrophin-1, myosin light chain kinase, claudin, myd88 and dosal) and morphological structure were also positively affected by dietary RA. Furthermore, dietary RA could relieve the immune responses induced by lipopolysaccharide, thus leading to lower transcription of antimicrobial peptides. In summary, dietary RA could improve the growth, lipid utilisation, antioxidant capacity and immunity of M. rosenbergii. To avoid the negative effects of excessive addition and obtain optimal growth, a diet containing 296 mg/kg dietary RA was suggested by the present study.
Aquaculture has developed rapidly and played an increasingly important role in the global food supply (Islam et al., 2024). However, human production activities and variations in bacteria and viruses increase the risk of decline in immunity which translates into high mortality in aquatic animals (Tao et al., 2024; Zhu, 2020). Antibiotics and other chemical medications have long been applied to control the occurrence of disease (Assefa and Abunna, 2018). While the usage of antibiotics and other chemical interventions are usually partially effective, they pose a threat to the environment and public health (Diwan et al., 2023). Thus, it is important to develop a safe and effective strategy to strengthen the immune status of aquatic animals for sustainable development.
Currently, complete formula feeds are widely used in aquaculture, and feed additives are a popular method to enhance the immunity of aquatic animals (Salam et al., 2024). The dietary nutritional supplement strategy has gained considerable attention (Zhan et al., 2024). Interestingly, retinoic acid (RA), a derivative of retinol (vitamin A) (Nhieu et al., 2020), has been demonstrated to be a good immunological enhancement factor (Farazuddin et al., 2024). RA has been shown to provoke toll-like receptor (TLR)-mediated polyclonal activation and differentiation of B-cells (Eriksen et al., 2012). In airway epithelial cells, RA drove the immune response by inducing the expression of antimicrobial peptides and cytokines (Jacobo-Delgado et al., 2021). In aquatic animals, RA affected germ cell development and meiotic initiation related genes expression in orange-spotted grouper Epinephelus coioides (Peng et al., 2020). RA injection impacted blood glucose homeostasis in fiddler crab Uca pugilator (Zou and Bonvillain, 2003). In vitro experiments showed that RA participated in the regulation of gonad development in edible crab Oziotelphusa senex senex (Girish et al., 2018). Although RA has been researched in some aquatic animals, there is a paucity of data on their influence on growth and immunity. However, as excessive RA intake may cause teratogenic and toxic effects in aquatic animals, the appropriate dietary supplement amount must be determined (Wang et al., 2023; Yeung et al., 2020).
The health of aquatic animals is usually related to lipid metabolism (Li et al., 2019). Abnormal lipid deposition can cause oxidative stress and induce tissue damage (Zhong et al., 2020). Our previous study in crustaceans demonstrated that proper dietary vitamin A could improve lipid utilisation and reduce tissue lipid content and oxidative stress (Huang et al., 2022b). However, the role of retinoids—especially RA—in lipid metabolism is complicated and still not fully understood (Bonet et al., 2012). It has been showed that dietary RA reduced lipid deposition in the muscle of red sea bream Pagrus major (Alsop et al., 2001). There was limited study on the effect of dietary RA on lipid metabolism in aquatic animals and it needs more exploration.
The giant freshwater prawn Macrobachium rosenbergii is economically important and an excellent protein source for many nations. The freshwater farmed yield of M. rosenbergii exceeded 190,000 tons in 2023 (China Fishery Statistical Yearbook, 2024). Worryingly, this species is facing the problem of disease due to viral (Decapod iridescent virus 1) and bacterial pathogenic infection such as Vibrio and environmental degradation (Dong et al., 2020; Qian et al., 2025; Rattanavichai and Cheng, 2014). Therefore, developing a method for enhancing the immunity and health of M. rosenbergii would lessen the economic losses caused by disease outbreaks. The aim of this study was to explore the effect of diverse dietary RA levels on the growth, lipid metabolism, oxidation resistance and immunity of M. rosenbergii. This enabled a better understanding of the regulatory effect of RA in crustaceans and provided insights into the exploitation of new efficient feed additives and artificial compound feed.
Fish meal, casein, gelatin and soybean protein concentrate were used as the main protein sources (Table 1). Soybean lecithin, cholesterol and oil mixture (fish oil: soybean oil = 1:1) were the main lipid sources. Six isonitrogenous and isolipidic diets were designed, which included different RA levels: 0 (control), 125, 250, 500, 1000 and 2000 mg/kg). The actual RA content in the feed was determined to be 4, 132, 296, 562, 1206 and 2562 mg/kg. The experimental RA supplement level was designed based on a previous experiment (Ogata and Oku, 2001). The RA (LH-081) was from Shanxi Lanhe Biotechnology Co., Lld. All the raw materials were passed through a 60-mesh strainer before being weighed. The feed ingredients, oil and distilled water were mixed to homogeneity and then processed into pellets (2 mm diameter) through an SYSLG30-IV double-screw extruder. Finally, the different diets were air-dried, packaged in marked ziplock bag and stored at –20°C before use.
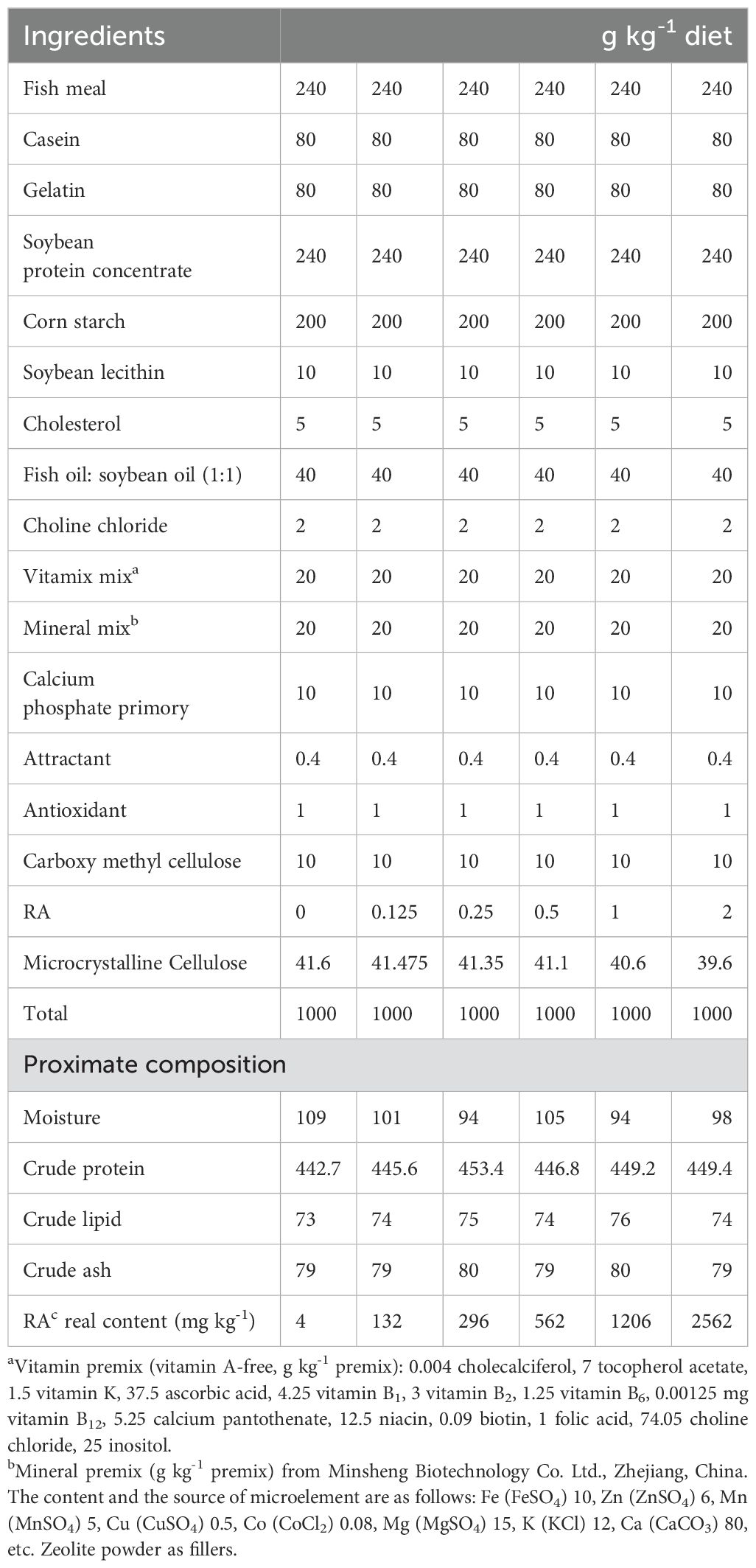
Table 1. Formulation components and nutritional composition of the foundational diet (dry matter basis).
The present study was authorised by the Committee on Experimental Animal Welfare of Zhejiang Academy of Agricultural Sciences (2024ZAASLA046).
In April 2024, 4000 juvenile prawns were purchased from a local farm in WuXing district (Huzhou, Zhejiang). The prawns were reared in square pools (8 m (L) × 1.1 m (W) × 1.5 m (H)) for about 3 weeks until they conformed to the specifications of the feeding trial. Then, 1200 uniformly-sized prawns (0.22 ± 0.00 g, mean ± SD) were randomly transported into 24 independent 300 L tanks (0.97 m (L) × 0.57 m (W) × 0.61m (H)) with four tanks per treatment (50 prawns per tank). Each diet was fed to four randomly selected tanks at 8:00 and 18:00 for 8 weeks. The daily feed amount was 8% of the body weight and was adjusted according to the measured weight per week. During the culture period, the environmental parameters were monitored. The temperature of the water was 23–27°C and the pH was 7.4–8.3. The dissolved oxygen was no less than 7 mg/L, and the ammonia nitrogen was lower than 0.05 mg/L.
Based on the effect of dietary RA on growth performance, prawns fed the control diet, 296 and 2562 mg/kg dietary RA were used for the LPS (sigma, L2630) challenge test. The concentration of the LPS injection (5 μg LPS/g prawn) was based on a previous study and suitably adjusted (Lu et al., 2009). The injection site was the caudal segment of the abdomen. Before the formal experiment, a preliminary experiment was conducted to guarantee the effectiveness and obtain the final injection concentration. The LPS solvent was phosphate-buffered saline (PBS). At the end of the 8-week feeding trial, at least four prawns per tank were randomly selected and injected with LPS. And at least 4 prawns fed the control diet, 296 and 2562 mg/kg dietary RA were randomly selected and gathered for PBS injection (control group, CN). After 12 h of the LPS challenge test (Lu et al., 2009), the hepatopancreas of each surviving prawn was collected and stored at –80°C for RNA isolation.
After being fasted for 24 h, the prawns per tank were counted and weighed at the end of the 8-week feeding trial. The hepatopancreas and intestines of two prawns from each tank were collected and preserved in 4% paraformaldehyde for morphological observation. Haemolymph (0.25 mL) was extracted from the first abdominal segment of at least three prawns with a 0.5 mL injector containing 0.25 mL of sodium citrate. After centrifugation at 10000 × g for 10 min at 4°C, the resultant plasma (haemocyte-free) was used for enzymatic determination. The hepatopancreas from 5 prawns, 3 prawns and 3 prawns per tank were collected for enzymatic determination, RNA isolation and protein extraction respectively. The intestines of at least three prawns per tank were collected for RNA isolation. Samples for enzyme activities assay and molecular experiments were first frozen with liquid nitrogen and then kept in a –80°C refrigerator before use.
The nutritional components of the experimental diets and prawns were determined. Moisture detection was according to the AOAC (1995) standard method and was 930.15 for feed and 950.46 for tissue. The powdery feed was dried in an oven at 105°C until a constant weight (± 0.1%) was obtained. The Kjeldahl method (K1100 automatic Kjeldahl nitrogen analyser, Hanon, Shandong, China) was used to detect the crude protein content (990.03). Chloroform-methanol was used to determine the total lipid content according to a previously reported method (Huang et al., 2022a). The crude ash content was measured by combustion in a muffle furnace (KSW-12-12, Beijing, China) at 550°C for 5 h (942.05).
The RA content in the diets and prawn body was detected by Wuhan Pronets Testing Technology Co., Ltd (Wuhan, China) through the combination of high-performance liquid chromatography (HPLC, LC-20AD, Shimadzu, Japan) and mass spectroscopy (QTRAP 5500, AB). The chromatographic conditions were as follows: column, Agilent Poroshell 120 SB-C18 (2.1 × 150 mm, 2.7 μm); temperature, 35°C; sample volume, 5 µL; flow rate, 0.35 mL/min; mobile phase A, 0.1% formic acid aqueous solution; mobile phase B, acetonitrile.
Hepatopancreas samples from each tank were homogenised (tissue weight: PBS = 1:9) at 4°C, and the supernatant was collected after centrifugation (3000 × g, 10 min, 4°C). Commercial kits from Nanjing Jiancheng Biological Engineering Institute (Jiangsu, China) were used for the detection of total antioxidant capacity (T-AOC; serial number A015-2-1), glutathione (GSH; A006-2-1), glutathione peroxidase (GPx; A005-1-2), superoxide dismutase (SOD; A001-3), malondialdehyde (MDA; A003-1-1) and acid/alkaline phosphatase (ACP; A060-2-1/AKP; A059-2-1) in the hepatopancreas, and aspartate/alanine aminotransferase (AST; C010-2-1/ALT; C009-2-1) in plasma, according to the manufacturer’s protocols. Specifically, the homogenate for T-AOC detection was centrifuged at 10000 × g. The hepatopancreas homogenate was further diluted because an ‘inhibition ratio’ needed to be guaranteed for GPx (about 50%) and SOD (about 40–60%) for accurate determination.
First, hepatopancreas and intestine samples were fixed using a 4% paraformaldehyde solution. Subsequently, a gradient concentration of ethanol was applied to dehydrate the samples, which were then embedded in paraffin to make solid wax blocks. Then, the trimmed wax blocks were sliced into 4 µm sections using a paraffin slicer. After staining with haematoxylin and eosin (H & E), images were obtained under microscopic examination (Nikon Eclipse E100, Nikon, Japan). Additionally, BODIPY staining and image acquisition of hepatopancreas frozen sections was performed by Wuhan Servicebio Technology Co., Ltd (Wuhan, China). The green areas of the BODIPY staining represent lipid droplets, and the blue areas represent cell nuclei.
Given the impact of dietary RA on the growth performance of prawns, the hepatopancreas or intestines from prawns fed the control diet, 296 and 2562 mg/kg dietary RA were selected for gene expression determination. The total RNA in the prawn hepatopancreas and intestine was extracted using Trizol reagent (R401, Vazyme Biotech Co., Ltd). Agarose gel electrophoresis (4%) and spectrophotometry (NanoDrop 2000, Thermo, USA) were used to check the RNA quality and concentration. Reverse transcription into cDNA was achieved using HiScript III All-in-one RT SuperMix Perfect for qPCR (R333-01, Vazyme Biotech Co., Ltd). The procedure for reverse transcription was 50°C for 15 min and then 85°C for 5 s.
Quantitative real-time PCR was performed using the Quantstudio Real-Time PCR system (Thermo Fisher Scientific Inc.). The 20 μL reaction system comprised 10 μL SYBR qPCR reaction reagent (Q711-02, Vazyme Biotech Co., Ltd), 0.4 μL each of forward and reverse primers, 1 μL of cDNA template and 8.2 μL of DEPC-water. The procedure was: 95°C for 30 sec (pre-degeneration), 40 cycles of 95°C for 10 sec and 60°C for 30 sec (circular reaction), and 95°C for 15 sec, 60°C for 60 sec and 95°C for 15 sec (melting curve). The primer sequences are provided in Supplementary Table S1. The amplification efficiency was verified using the formula E = 10(−1/Slope) – 1 and was about 90–101%. 18s and β-actin were used as reference genes. The 2−ΔΔCt method was used for gene expression calculation.
Prawns fed 4 and 296 mg/kg dietary RA were selected for western blotting according to the phenotype for lipid deposition. Immunoblot analyses were implemented according to a previously reported method (Bu et al., 2022). Protein homogenates were prepared from about 100 mg of hepatopancreas tissue in cell lysis buffer (Beyotime Biotechnology, P0013) containing 1 mM phenylmethanesulfonyl fluoride (Beyotime Biotechnology, ST505). After centrifugation (10400 × g) for 10 min, the supernatant was blended with 5 × SDS loading buffer and boiled via an electromagnetic oven at 95°C for 5 min. A bicinchoninic acid protein determination commercial kit (Beyotime Biotechnology, P0012S) was used to determine the protein concentration. First, separation gel and spacer gel were prepared, and then the protein was separated using SDS−PAGE. The protein was transferred to nitrocellulose membranes at a constant current (200 mA). After being blocked with 5% non-fat milk in Tris-buffered saline (TBS) containing 0.05% Tween 20 for 2 h, the membranes were incubated overnight at 4°C with primary antibodies against RXR (Abways, CY8640), CAMKKβ (Proteintech, 11549-1-AP), phospho-AMPKαT172 (Affinity, AF3423) and β-actin (Proteintech, 66009-1-Ig). After washing through TBST three times, the membranes were incubated with goat anti-rabbit or mouse IgG (SA00001-2 or SA00001-1, Proteintech) for 1 h. The western blot images were captured using Odyssey CLx Imager (Li-Cor) and quantitatively analysed using ImageJ software.
Data homogeneity of variance was tested before performing one-way ANOVA and Duncan’s multiple comparison (SPSS 27). Significant differences among means were when P< 0.05. The linear or quadratic effect of dietary RA against different indices was evaluated through polynomial regression analysis. The independent sample t-test (* means significant difference) was applied to compare the difference in protein expression between prawns fed 4 and 296 mg/kg dietary RA and the effect of dietary RA on immune gene expression after the LPS challenge. The growth performance parameters were calculated as follows:
Here, WG represents to weight gain rate, SGR to specific growth rate, FBW to final body weight, IBW to initial body weight and SR to survival rate.
Different dietary RA levels significantly affected the FBW, WG and SGR of prawns with a linear and quadratic effect (P< 0.05, Table 2). The values of FBW, WG and SGR were significantly lower in prawns fed 562, 1206 and 2562 mg/kg dietary RA compared to those fed 296 mg/kg dietary RA (P< 0.05). There were no obvious differences between the FBW, WG and SGR of prawns fed dietary RA levels of 4, 132 and 296 mg/kg (P > 0.05).
The crude protein, ash and moisture content in the whole body were not influenced by dietary RA levels (P > 0.05, Table 3). The crude lipid content in the whole body decreased linearly and quadratically as the dietary RA level increased (P< 0.05). Prawns fed any dietary RA supplement had significantly reduced crude lipid content in the whole body relative to those fed the control diet (4 mg/kg, P< 0.05).
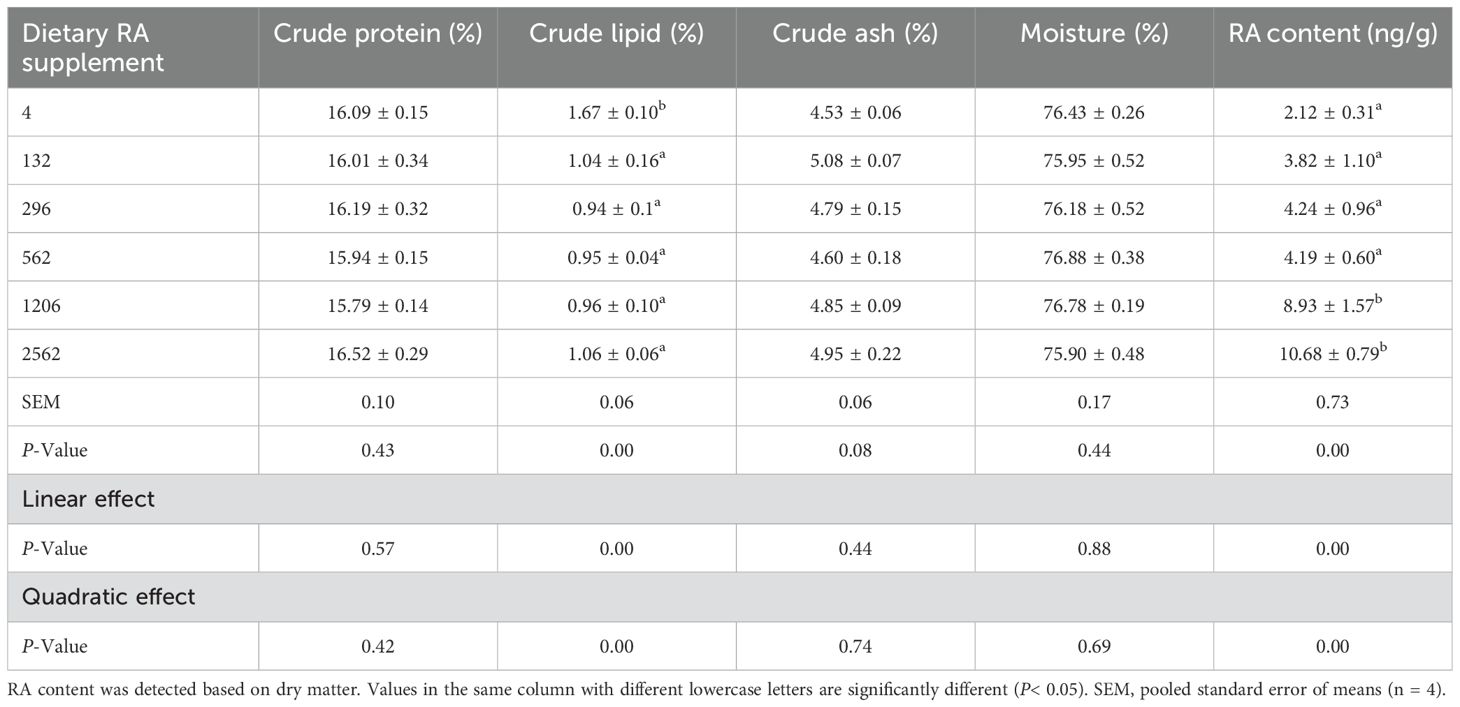
Table 3. Body composition and RA concentration of juvenile M. rosenbergii fed dietary RA for 8 weeks.
The content of RA in the whole body changed linearly and quadratically and was highest in prawns fed 2562 mg/kg dietary RA (P< 0.05). Prawns fed 4, 132, 296 and 562 mg/kg dietary RA had significantly lower body RA concentrations compared to those fed 1206 and 2562 mg/kg dietary RA (P< 0.05).
The hepatopancreas index (HSI) decreased linearly and quadratically as the level of dietary RA increased (P< 0.05, Figure 1A). Prawns fed 4 and 132 mg/kg dietary RA had higher HSI values than those fed 562–2562 mg/kg dietary RA (P< 0.05). The HSI of prawns fed 296 mg/kg dietary RA was higher than that of prawns fed 2562 mg/kg dietary RA (P< 0.05).
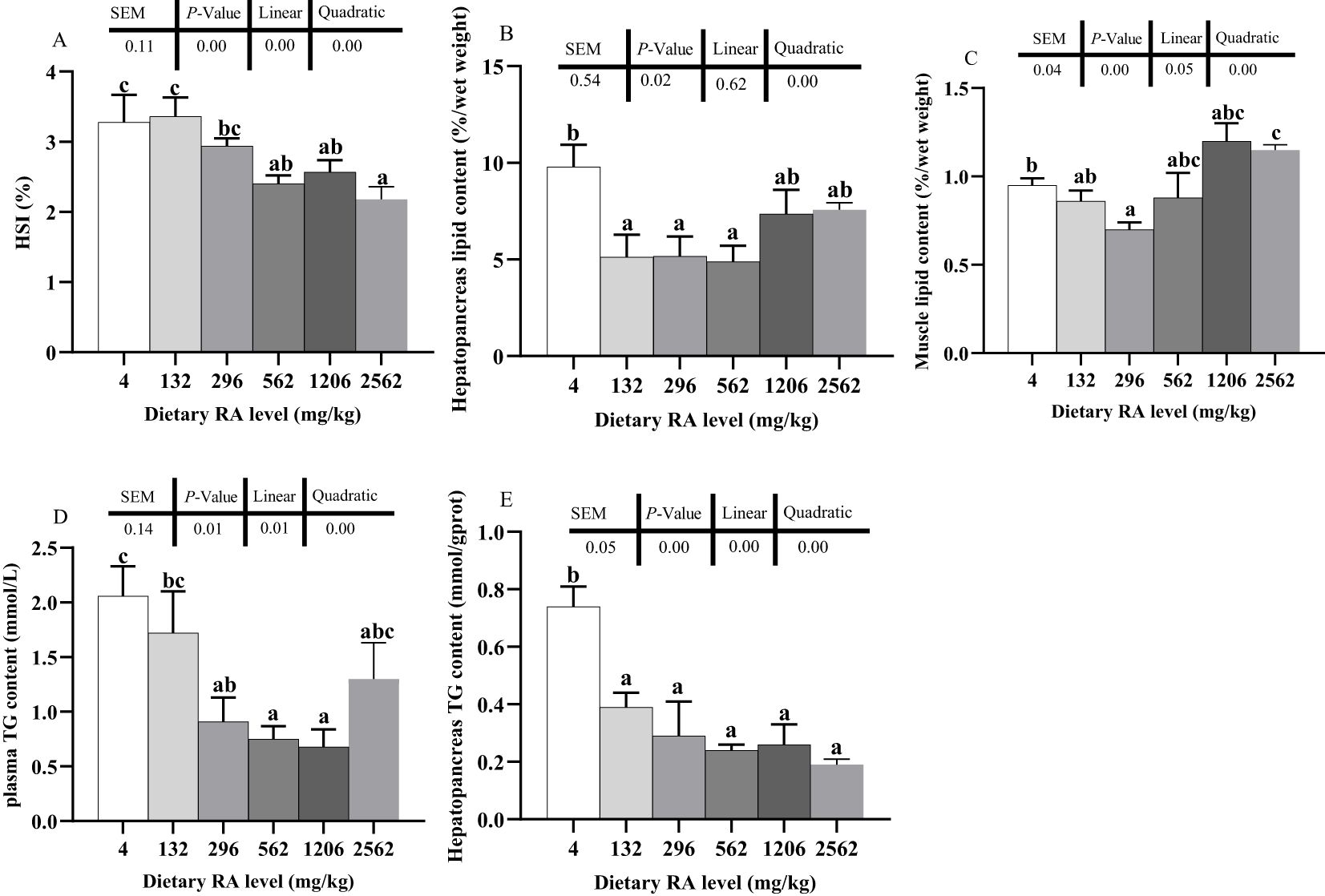
Figure 1. Effect of various dietary RA levels on the hepatopancreas index (HSI, A), hepatopancreas (B) and muscle (C) lipid content, plasma (D) and hepatopancreas (E) triglyceride (TG) content in prawns. Different lowercase letters on different columns indicate a significant difference (P < 0.05). Data are mean ± SEM (n = 4). The pooled SEM, P values from one-way ANOVA and trend analysis are provided above each figure.
The lipid content in the hepatopancreas changed quadratically at different dietary RA levels (P< 0.05, Figure 1B). Prawns fed 4 mg/kg dietary RA had higher lipid content in the hepatopancreas than those fed 132–562 mg/kg dietary RA (P< 0.05). The muscle lipid content decreased linearly and quadratically as the dietary RA level changed (P< 0.05, Figure 1C). Prawns fed 296 mg/kg dietary RA had lower muscle lipid content than those fed 4 and 2562 mg/kg dietary RA (P< 0.05) and was not significantly different to those fed other diets (P > 0.05).
The hepatopancreas and plasma TG content changed linearly and quadratically as the dietary RA level changed (P< 0.05, Figures 1D, E). The plasma TG content in prawns fed 4 mg/kg dietary RA was dramatically higher than in prawns fed 296–1206 mg/kg dietary RA (P< 0.05) and was not significantly different to those fed other diets (P > 0.05). There was higher hepatopancreas TG content in prawns fed 4 mg/kg dietary RA compared to other treatments (P< 0.05).
The green areas of the hepatopancreas following BODIPY staining in prawns fed different dietary RA levels changed quadratically (P< 0.05, Figure 2). Prawns fed 4 and 2562 mg/kg dietary RA had significantly larger green areas of hepatopancreas BODIPY staining than prawns fed 132 and 296 mg/kg dietary RA (P< 0.05).

Figure 2. BODIPY staining of the hepatopancreas in prawns fed different dietary RA levels and quantification of the green area ratio in the picture. The scale represents 1000 μm. Different lowercase letters on different columns indicate a significant difference (P< 0.05). Data are mean ± SEM (n = 4). The pooled SEM, P values of one-way ANOVA and trend analysis are provided above the histogram.
Hepatopancreas T-AOC was not significantly affected by dietary RA supplementation (P > 0.05, Figure 3A). Hepatopancreas SOD and GPx, GSH activity and MDA content in the prawns changed linearly and quadratically at different dietary RA levels (P< 0.05, Figures 3B–E). There was lower hepatopancreas SOD activity in prawns fed 2562 mg/kg dietary RA compared to prawns fed 4–296 mg/kg dietary RA (P< 0.05). Lower SOD activity was also observed in prawns fed 562 and 1206 mg/kg dietary RA than in prawns fed 132 mg/kg dietary RA (P< 0.05). Significantly lower hepatopancreas GPx activity was found in prawns fed 4 and 562–2562 mg/kg dietary RA compared to prawns fed 296 mg/kg dietary RA (P< 0.05). There was lower hepatopancreas GPx activity in prawns fed 2562 mg/kg dietary RA compared to in prawns fed 4 and 132 mg/kg dietary RA (P< 0.05). Hepatopancreas GSH content in prawns fed 296 mg/kg dietary RA was significantly higher than in prawns fed other dietary RA levels (P< 0.05). There was lower hepatopancreas GSH content in prawns fed 562 and 2562 mg/kg dietary RA compared to in prawns fed 4 and 132 mg/kg dietary RA (P< 0.05). A dietary RA level of 4 mg/kg significantly increased the hepatopancreas MDA content compared to levels of 296 and 562 mg/kg (P< 0.05). Additionally, the hepatopancreas MDA content was significantly lower in prawns fed 296–2562 mg/kg dietary RA compared to in prawns fed 132 mg/kg dietary RA (P< 0.05).
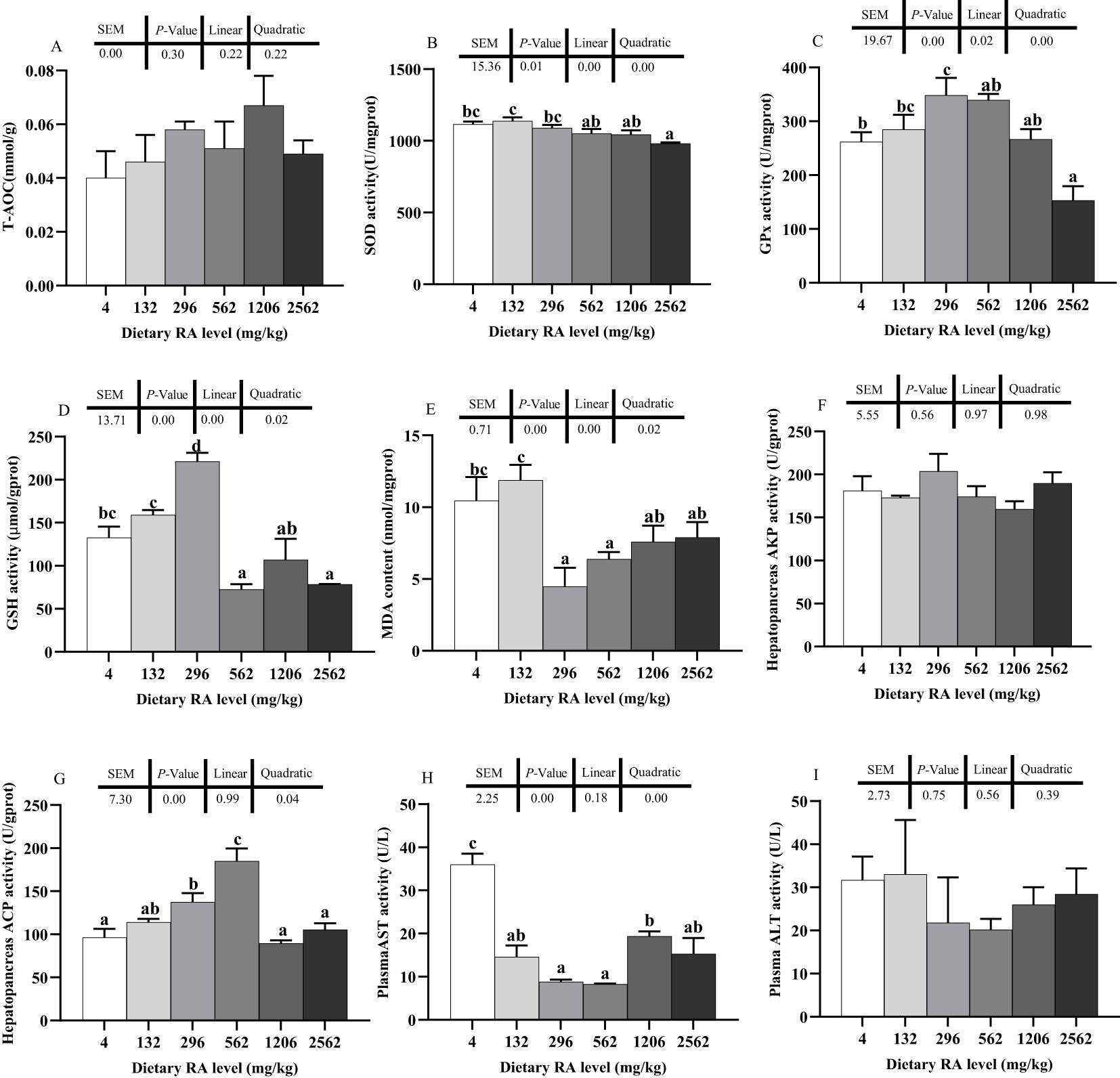
Figure 3. Effect of dietary RA on the hepatopancreas oxidation resistance (A–E), immunity (F, G) and plasma enzyme activity (H, I) in M. rosenbergii. Different lowercase letters on different columns indicate a significant difference (P< 0.05). Data are mean ± SEM (n = 4). The pooled SEM, P values of one-way ANOVA and trend analysis are provided above each figure.
Hepatopancreas AKP activity was not affected by dietary RA (P > 0.05, Figure 3F). Hepatopancreas ACP activity changed quadratically at different dietary RA levels (P< 0.05, Figure 3G). There was significantly higher hepatopancreas ACP activity in prawns fed 296 and 562 mg/kg dietary RA compared to prawns fed 4, 1206 and 2562 mg/kg dietary RA (P< 0.05, Figure 3G). Plasma AST activity also changed quadratically at different dietary RA levels (P< 0.05, Figure 3H). Prawns fed 296, 562 and 1206 mg/kg dietary RA had significantly reduced plasma AST activity relative to prawns fed 4 mg/kg dietary RA (P< 0.05, Figure 3H). There was higher plasma AST activity in prawns fed 1206 mg/kg dietary RA than in prawns fed 296 and 562 mg/kg dietary RA (P< 0.05).
Hepatopancreas gene expression of RXR, cpt1 and dgat1 changed quadratically at different dietary RA levels (P< 0.05, Figures 4A–C, Supplementary Table S2). Prawns fed 296 mg/kg dietary RA had significantly higher RXR and cpt1 expression than prawns fed 4 mg/kg dietary RA (P< 0.05). A dietary RA level of 2562 mg/kg decreased the expression of cpt1 compared to a dietary RA level of 296 mg/kg (P< 0.05). Compared to the control group, there was higher dgat1 expression in prawns fed any dietary RA level (P< 0.05).
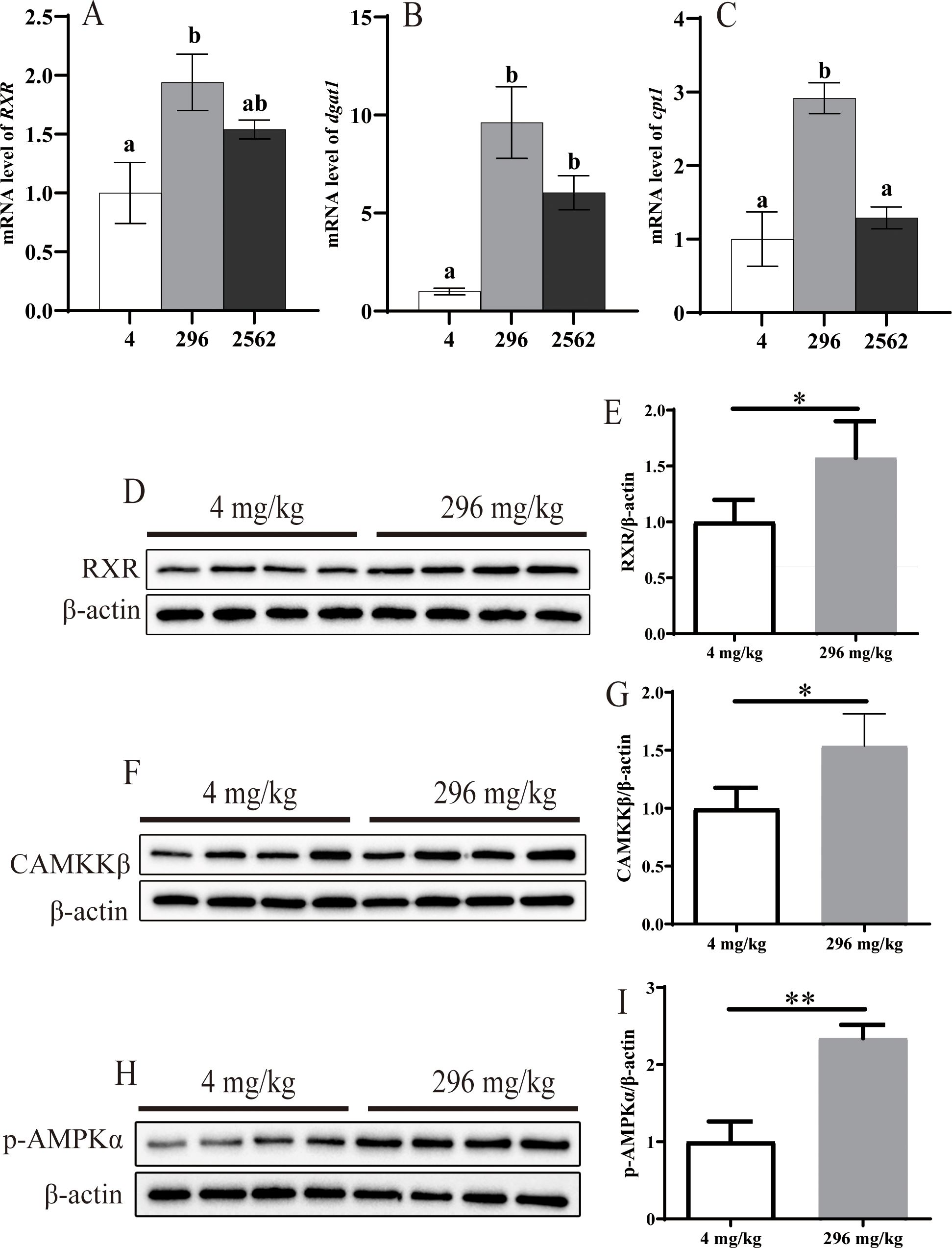
Figure 4. Lipid metabolism–related gene and protein expression. (A) RXR, retinoid X receptor; (B) dgat1, diacylglycerol acyltransferase 1; (C) cpt1, carnitine palmitoyltransferase 1. (D, E), protein expression level of RXR. (F, G), protein expression level of CAMKKβ. (H, I), protein expression level of p-AMPK. Data are mean ± SEM (n = 4). ‘*’ indicates P<0.05; ‘**’ indicates P<0.01.
Compared to prawns fed 4 mg/kg dietary RA, prawns fed 296 mg/kg dietary RA had significantly increased hepatopancreas protein levels of RXR (Figures 4D, E, P< 0.05), CAMKKβ (Figures 4F, G, P< 0.05) and p-AMPK (Figures 4H, I, P< 0.01).
No significant difference in the hepatopancreatic mRNA levels of cru1 and cru2 were observed among prawns fed 4, 296 and 2562 mg/kg dietary RA (P > 0.05, Figure 5A). Hepatopancreas expression of genes alf1 and imd changed quadratically at different dietary RA levels (P< 0.05), and expression of prx5 and iκbα changed linearly (P< 0.05). mRNA levels of myd88 and dosal changed linearly and quadratically at different dietary RA levels, respectively (P< 0.05). Compared with the control group, there were higher hepatopancreas mRNA levels of cru3, prx5, alf1, toll2, myd88 and dosal in prawns fed 296 mg/kg dietary RA (P< 0.05). Prawns fed 2562 mg/kg dietary RA showed higher expression of prx5, myd88 and dosal compared to the control group but lower expression of alf1, imd and dosal relative to those fed 296 mg/kg dietary RA (P< 0.05). There was a higher level of iκbα expression in prawns fed 4 mg/kg dietary RA compared to prawns fed 296 and 2562 mg/kg dietary RA (P< 0.05).
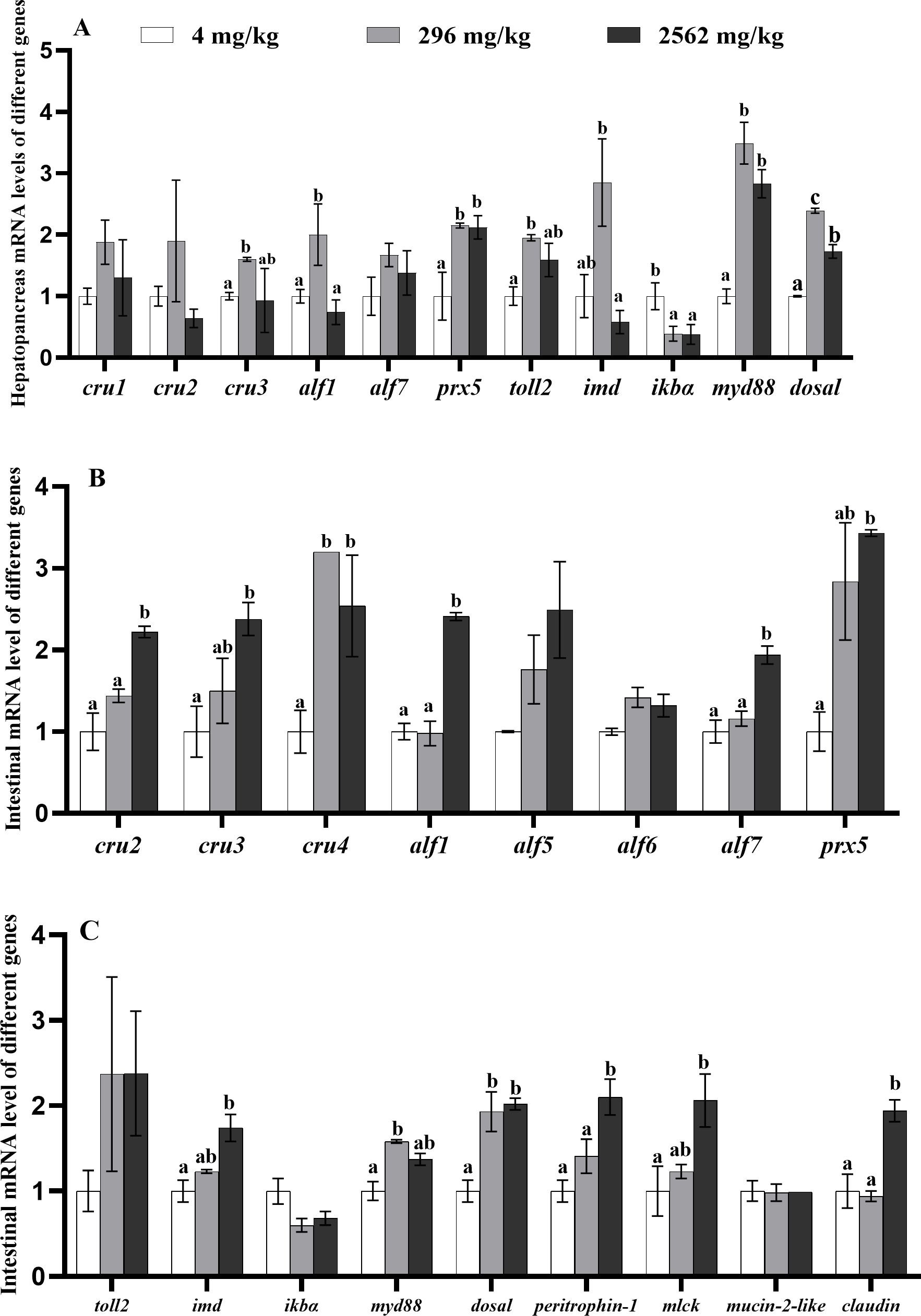
Figure 5. (A) Immunity-related gene expression (cru1/2/3, alf1/7, prx5, imd, iκbα, toll2, myd88 and dosal) in the hepatopancreas of M. rosenbergii fed different dietary levels of RA. (B, C) Immunity (cru2/3/4, alf1/5/6/7, prx5, toll2, imd, iκbα, myd88 and dosal) and intestinal barrier (peritrophin-1, mlck, mucin-2-like and claudin)–related functional gene expression in the intestine of M. rosenbergii fed different dietary levels of RA. Data are mean ± SEM (n = 4). Different lowercase letters on different columns indicate a significant difference (P< 0.05).
Intestinal expression of genes cru2, cru3, alf7 and prx5 increased linearly at different dietary RA levels (P< 0.05, Figure 5B, Supplementary Table S2), while expressions of cru4 and alf1 changed linearly and quadratically, respectively (P<0.05). Compared to prawns fed 4 and 296 mg/kg dietary RA, the prawns fed 2562 mg/kg dietary RA had significantly elevated intestinal mRNA levels of cru2, alf1 and alf7 (P< 0.05). There was significantly lower expression of cru4 in the intestine of prawns fed 4 mg/kg dietary RA relative to prawns fed 296 and 2562 mg/kg dietary RA (P< 0.05). Expression of alf5 and alf6 was not affected by dietary RA (P > 0.05), while expression of IMD and dosal changed linearly, and myd88 changed quadratically (P< 0.05, Figure 5C). Compared to prawns fed 4 mg/kg dietary RA, prawns fed 296 mg/kg dietary RA had increased expression of myd88 and dosal, and prawns fed 2562 mg/kg dietary RA had increased expression of imd and dosal (P< 0.05). The mRNA levels of peritrophin-1 and mlck increased linearly, and claudin changed linearly and quadratically (P< 0.05, Figure 5C). There were significantly higher mRNA levels of peritrophin-1 and claudin in the intestine of prawns fed 296 and 2562 mg/kg dietary RA relative to prawns fed 4 mg/kg dietary RA (P< 0.05). Prawns fed 2562 mg/kg dietary RA had higher mlck expression compared to prawns fed 4 mg/kg dietary RA (P< 0.05).
The hepatic tubule structures in the hepatopancreas of prawns fed 4 and 132 mg/kg dietary RA were unclear and their arrangement was irregular (Figure 6). Wide spaces between hepatic tubules were observed in the hepatopancreas of prawns fed 4, 132, 562 and 1206 mg/kg dietary RA, and there was more vacuolation of hepatocytes in the hepatopancreas of prawns fed 4 and 2562 mg/kg dietary RA. Expanded and deformed hepatic tubules were found in the hepatopancreas of prawns fed 4, 1206 and 2562 mg/kg dietary RA.
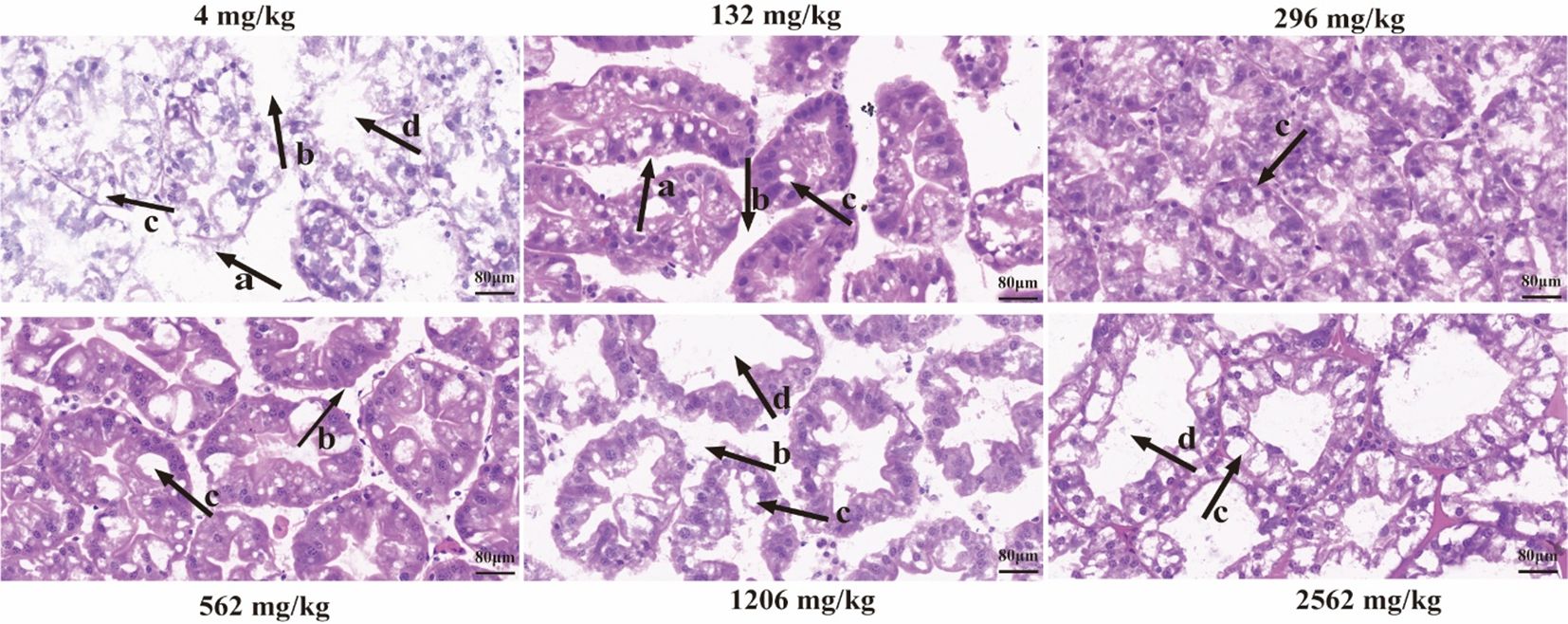
Figure 6. Transverse sections of the hepatopancreas in M. rosenbergii fed different dietary RA levels. The magnification is 400×, the scale is 80 μm, the lowercase (‘a’) indicates that the structure of the hepatic tubules is unclear and the arrangement is irregular, (‘b’) represents wide spaces between hepatic tubules, (‘c’) represents vacuolar degeneration of hepatocytes, (‘d’) represents abnormal expansion of hepatic tubules.
The mucosal thickness in the intestine changed quadratically at different dietary RA levels (P< 0.05, Figure 7). Compared to prawns fed 4 and 2562 mg/kg dietary RA, prawns fed 296 mg/kg dietary RA had a significantly increased intestinal mucosal thickness (P< 0.05, Figure 7). Additionally, there were pronounced deciduous mucosal epithelial cells in prawns fed 4, 132, 1206 and 2562 mg/kg dietary RA (P< 0.05). Prawns fed 296 mg/kg dietary RA had neatly arranged epithelial cells compared to those fed other diets.

Figure 7. Transverse sections of the intestine in M. rosenbergii fed different dietary RA levels (n = 4). The magnification is 400×, the scale is 80 μm, ‘a’ represents the peritrophic membrane, ‘b’ represents epithelial cells and ‘c’ represents the muscular layer.
Following injection with LPS, compared to the prawns injected with PBS, prawns fed 4 mg/kg dietary RA had significantly enhanced transcription levels of cru1, alf1, alf6 (P< 0.05) and alf7 (P< 0.01), and prawns fed 296 mg/kg dietary RA had significantly increased transcription levels of cru1 (P< 0.05, Figure 8). After injection with LPS, prawns fed 4 mg/kg dietary RA showed significantly higher expression of cru1, alf6 (P< 0.05) and alf7 (P< 0.01) compared to prawns fed 2562 mg/kg dietary RA, and higher expression of alf6 and alf7 compared to prawns fed 296 mg/kg dietary RA (P< 0.05). After LPS treatment, prawns fed 2562 mg/kg dietary RA had significantly decreased expression of cru1, cru3 and alf7 compared to prawns fed 296 mg/kg dietary RA (P< 0.05) and reduced expression of alf6 (P< 0.05) and alf7 (P< 0.01) compared to prawns fed 4 mg/kg dietary RA.
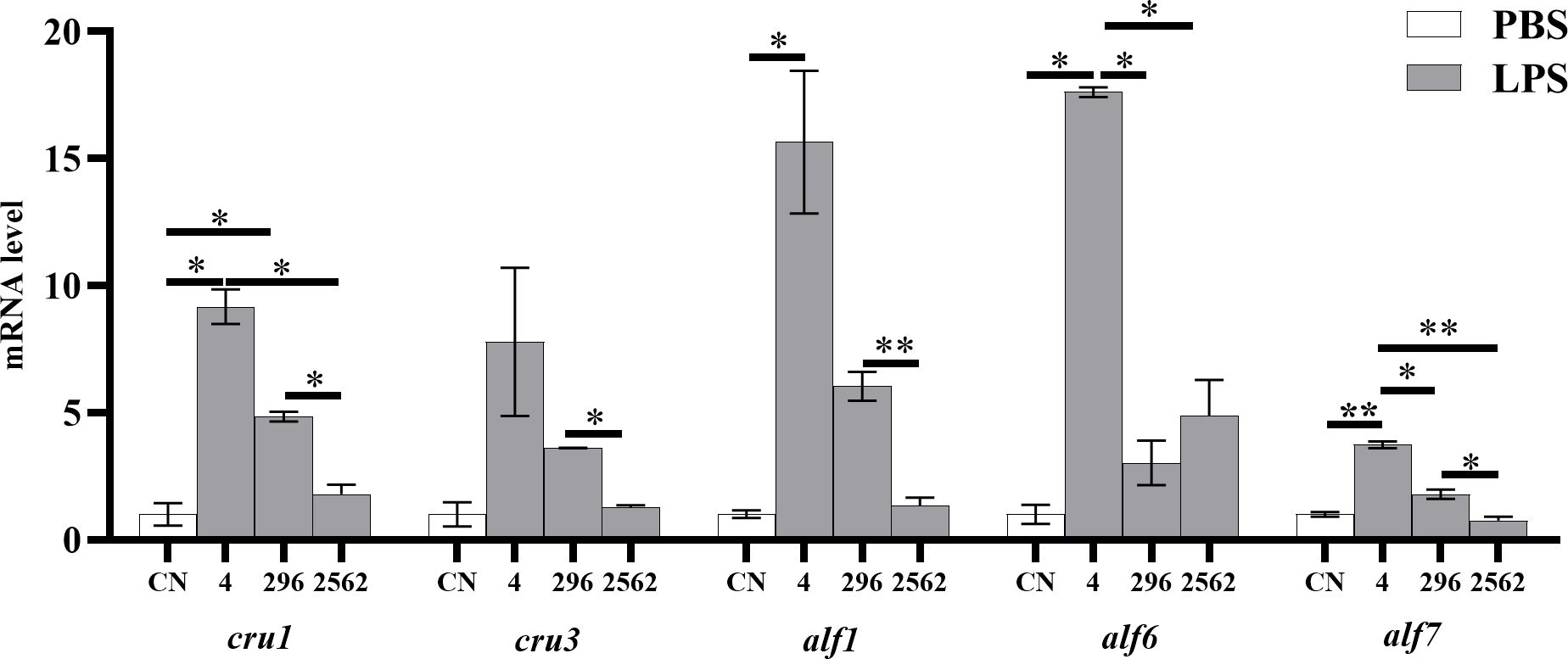
Figure 8. The mRNA levels of immunity-related gene expression in the hepatopancreas of M. rosenbergii fed different dietary levels of RA after the LPS challenge test (n = 4). ‘*’ indicates a significant difference between two different groups (P< 0.05) and ‘**’ indicates a significance level of P<0.01. CN, control group of prawns injected with PBS.
RA, a major active cellular retinoid, is synthesised intracellularly mainly from retinaldehyde produced by retinol (vitamin A) or provitamin A carotenoids (Bonet et al., 2012). In the present study, the significant linear effect of dietary RA on RA content in the whole body suggested that RA was incorporated into the body of prawns during the 8-week feeding experiment. Red sea bream fed 10 or 100 mg/kg also showed higher plasma RA content than those fed the control diet (Ogata and Oku, 2001). Vitamin A exerts its pleiotropic effects and regulates the transcription of numerous genes mainly as RA (Ziad et al., 2013). The growth-promoting effect of vitamin A has been reported in many aquatic animals, such as grass shrimp Penaeus monodon (Shiau and Chen, 2000), Chinese mitten crab Eriocheir sinensis (Jiang et al., 2024) and orange-spotted grouper Epinephelus coioides (Yang et al., 2017). Dietary RA had a quadratic effect on the WG that peaked at 296 mg/kg dietary RA, suggesting that appropriate dietary RA addition was helpful in improving the growth of M. rosenbergii.
Lipid metabolism plays an important role in the growth of crustaceans (Li et al., 2023). The effect of dietary RA on lipid deposition in vivo has rarely been reported in aquatic animals. The current results showed that dietary RA decreased the lipid content in the whole body, hepatopancreas and muscle. This decrease in hepatopancreas lipid content was validated by the reduced green area ratio observed following hepatopancreas BODIPY staining. Red sea bream fed 100 mg/kg of dietary RA had reduced lipid content in the dorsal white muscle and plasma non-esterified fatty acids compared to those fed 10 mg/kg of dietary RA, which contrasted with the results in visceral adipose tissue (Ogata and Oku, 2001). A 16-week feeding experiment in male C57BL/6J mice showed that plasma lipids were unaffected by dietary fish oil plus RA but were significantly decreased by dietary soybean oil plus RA (Tung et al., 2019). Therefore, the results of dietary RA on lipid deposition might vary depending on the experimental animal, lipid source and detected tissues. In addition, there was no significant difference about hepatopancreas lipid content in among prawns fed dietary RA levels of 4, 1206 and 2562 mg/kg. The quadratic effect of dietary RA on the hepatopancreas lipid content indicated that excess dietary RA would show negative effect and elevate lipid deposition. It has been reported that excess dietary vitamin A (200 000 IU/kg) decreased the WG and elevated the lipid content of the whole body and hepatopancreas in the grass shrimp (Shiau and Chen, 2000). Regardless, the present results indicated that appropriate dietary RA could improve lipid utilisation and enhance prawn growth.
In the present study, 296 mg/kg dietary RA decreased hepatopancreatic and plasma TG levels. Carnitine palmitoyltransferase 1 is a mitochondrial rate-limiting enzyme in the β-oxidation of fatty acids, which promotes the transport of long-chain fatty acids into the mitochondrial matrix (Ngo et al., 2023). Four days of RA injection reduced the muscle lipid content by enhancing fatty acid oxidation (Amengual et al., 2012). In the present study, 296 mg/kg of dietary RA induced the expression of hepatopancreas cpt1 in prawns. It has been reported that RA has nongenomic effects that could regulate the activity of important protein kinases through retinoid receptor–dependent or –independent mechanisms (Bonet et al., 2012; Tanoury et al., 2013). AMPK, an energy sensor, plays an important role in lipid metabolism (Bu et al., 2022). Activated AMPK can reduce cellular levels of malonyl-CoA, which is a potent allosteric suppressor of carnitine palmitoyltransferase 1 (Bonet et al., 2012). CAMKKβ is the main kinase upstream of AMPK, which can phosphorylate Thr172 in the catalytic α-subunit of AMPK (Shi et al., 2020). Therefore, the upregulated cpt1 expression was partly due to activation of the CAMKKβ/AMPK signal pathway. Additionally, diacylglycerol acyltransferase 1, a key enzyme in TG synthesis, esterifies TG in the endoplasmic reticulum by utilising substrates diacylglycerol and acyl-coenzyme A (Wang et al., 2024). RA can bind to heterodimeric RXR-liver X receptor (LXR) and directly regulate the expression of genes involved in lipid synthesis (Alsop et al., 2001; Bonet et al., 2012; Karagianni and Talianidis, 2015). It was reported that treatment with RA could activate lipogenesis in HepG2 cells (Roder et al., 2007). The present study showed that prawns fed 296 mg/kg of dietary RA induced the expression of hepatopancreatic gene dgat1, as well as gene and protein expression of RXR. Therefore, a dietary RA level of 296 mg/kg may increase fat synthesis through activating RXR. These results demonstrated that a dietary RA level of 296 mg/kg improved lipid utilisation, possibly through activation of the RXR and CAMKKβ/AMPK signalling pathways.
The antioxidant system plays an important role in protecting organisms from damage by free radicals and oxidant species (Espinosa-Diez et al., 2015). T-AOC, SOD, GPx, GSH and MDA are widely used to reflect the capacity of oxidation resistance of an organism (Liu et al., 2022b; Xu et al., 2024). It has been reported that excessive lipid accumulation could induce oxidative stress (Liu et al., 2022b; Zhong et al., 2020). In the present study, the quadratic change in hepatopancreas SOD, GPx and GSH activity and MDA content was consistent with the varying trends in hepatopancreatic lipid content. The promotion of lipid utilisation, discussed above, may be one reason underpinning the positive role of dietary RA on the antioxidant system observed in the present study. Additionally, it has been reported that RA can enhance the antioxidant system by inhibiting tissue production of reactive oxygen species (ROS) and MDA and activating transcription of antioxidant-related genes and pathways (Lai et al., 2023). Prx family members are small antioxidant proteins that can use thioredoxin as electron donors and scavenge peroxide (Liu et al., 2022a; Wu et al., 2017). Dietary RA showed a positive and linear effect on prx5 expression in different tissues, suggesting that dietary RA could mitigate oxidative stress partly through upregulating prx5 expression.
Crustaceans lack adaptive immunity and combat invading pathogens via non-specific immunity (Hao et al., 2024; Liu et al., 2022a; Si et al., 2024). Both the hepatopancreas and intestine are important immune organs in crustaceans (Zheng et al., 2024). ACPs are important hydrolases within lysosomes, catalysing the hydrolysis of phosphate esters and phosphoproteins (Yohana et al., 2024). It was showed that suitable dietary RA boosts the hepatopancreas immune status by increasing ACP activity in the present study. Antimicrobial peptides (AMPs), including crustins and alfs, are cationic small molecular polypeptides possessing antimicrobial and antiviral ability (Si et al., 2024). Toll-like receptors, the most representative and functional pattern recognition receptors, can activate downstream signal transduction and induce immune responses (Li et al., 2021). Toll-myd88-dosal (NF-κB family member) and imd pathways can regulate the transcription of multiple AMPs and play key roles in the defence against invading pathogens (Li et al., 2021; Niu et al., 2023; Ren et al., 2012; Wen et al., 2022). In the present study, 296 mg/kg dietary RA significantly increased mRNA levels of cru3, alf1, toll2, myd88, dosal and imd in the hepatopancreas and the expression of myd88 and dosal in the intestine. Additionally, dietary RA had positive and linear effects on the expression of cru2/3/4, alf1/7 and imd in the intestine of prawns. Iκbα, specialising in NF-κB inhibition (Liu et al., 2022b), was transcriptionally suppressed by 296 mg/kg dietary RA. These results indicated that dietary RA could improve the immunity of prawns, probably through activating the toll-myd88-dosal and imd pathways. Consistently, suitable dietary retinol levels improved the transcription of toll2, myd88, crustin and alf in Chinese mitten crab, Eriocheir sinensis (Jiang et al., 2024). Additionally, in vitro experiments on mammalian cells demonstrated that RA treatment could promote the nuclear translocation of p-NF-κB and protein expression of p-NF-κB and NF-κB (Guo et al., 2022). Tight junction proteins such as claudin are important components of the intestinal mechanical barrier, which could be regulated by the signal molecule mlck (Zheng et al., 2024). Peritrophin protein, tightly linked to the peritrophic membrane, promoted the anti-bacterial non-specific immunity of crustaceans (Huang et al., 2015). In the present study, a positive and linear effect of dietary RA on the expression of intestinal mlck, claudin and peritrophin-1 was found. More neatly arranged epithelial cells and a greater mucosal thickness in prawns fed 296 mg/kg dietary RA were observed compared to prawns fed the control diet. This suggested that dietary RA could improve the function of the intestinal barrier. Although it has been reported that the upregulation of mlck suppressed the expression of claudin (Zheng et al., 2024), the important role of myosin light chain (mlc) phosphorylation should not be neglected (Qin et al., 2021). Further research is needed to investigate the effect of dietary RA on the posttranslational modification of MLCK.
To further assess the impact of dietary RA on the antibacterial ability of prawns, LPS was applied for the challenge test. The expression of crustins and alfs was detected due to their evolutionarily conserved antimicrobial activity in many organisms (Si et al., 2024). In agreement with previous results in M. rosenbergii (Lu et al., 2009), injection with LPS could markedly enhance the transcription of AMPs such as cru1 and alf1/6/7 in prawns compared to those injected with PBS alone, indicating the successful establishment of the LPS stimulating model. Interestingly, there was higher expression of hepatopancreas AMPs in prawns fed 4 mg/kg dietary RA compared to those fed 296 or 2562 mg/kg dietary RA. This result was supported by previous findings showing that RA could suppress inflammation induced by LPS and lead to lower expression of pro-inflammatory factors such as tumour necrosis-α (TNF-α), interleukin (IL)-18 and IL-1β (Han et al., 2018). Thus, dietary RA could mitigate the drastic immune responses induced by LPS, protecting the health of the organism.
Notably, it has been reported that exogenous RA may cause toxic effects and impair organism health. This was because RA exposure led to neurodevelopmental toxicity in zebrafish Danio rerio embryos (Wang et al., 2023). Plasma AST and ALT are markers of hepatopancreas injury (Lai et al., 2023). The quadratic effect of dietary RA on plasma AST activity suggested that excessive dietary RA levels would endanger hepatopancreatic health. This was supported by the unusual hepatic tubule structures with abnormal expansion observed in prawns fed 1206 and 2562 mg/kg dietary RA. Furthermore, 2562 mg/kg dietary RA may have caused intestinal injury because of the significantly lower mucosal thickness relative to that of prawns fed 296 mg/kg dietary RA. Considering the growth performance impairment of prawns fed 562 mg/kg dietary RA, the optimal dietary RA level suggested by this study was 296 mg/kg.
In conclusion, suitable dietary RA positively affected prawn growth, lipid utilisation, antioxidant capacity, immunity and hepatopancreatic and intestinal health. A dietary RA level of 296 mg/kg had a lipid-lowering effect and enhanced expression of key genes involved in fatty acid catabolism and TG synthesis in the hepatopancreas, possibly through activating the RXR and CAMKKβ/AMPK pathways. Furthermore, dietary RA decreased oxidative stress and improved the immune status in the hepatopancreas, as well as mitigated the immune impairment induced by LPS. Dietary RA showed a positive influence on intestinal immunity, barrier function and health. Based on optimal prawn growth, 296 mg/kg of dietary RA appeared to be the most suitable level. This study suggests that RA has potential as a functional additive in aquatic feed.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author/s.
The animal studies were approved by Committee on Experimental animal Welfare of Zhejiang Academy of Agricultural Sciences. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent was obtained from the owners for the participation of their animals in this study.
HQC: Data curation, Formal analysis, Funding acquisition, Investigation, Software, Writing – original draft, Writing – review & editing. WL: Investigation, Supervision, Writing – original draft. GZM: Funding acquisition, Resources, Validation, Supervision, Project administration, Writing – review & editing. LC: Investigation, Supervision, Validation, Writing – original draft. YTT: Software, Writing – original draft. YJJ: Investigation, Supervision, Validation, Writing – original draft. MJL: Investigation, Methodology, Writing – original draft. ZYT: Investigation, Supervision, Writing – original draft. GXP: Conceptualization, Resources, Supervision, Validation, review & editing. LZD: Investigation, Resources, Writing – original draft. ZGY: Resources, Writing – original draft.
The author(s) declare that financial support was received for the research and/or publication of this article. Supported by “Pioneer” and “Leading Goose” R&D Program of Zhejiang (2024SSYS0101), China Postdoctoral Science Foundation (2024M751454), National Subsidization Program for Postdoctoral Researcher (GZC20231128) and Xianghu lab project (2023C2S02002).
Author GZ was employed by the company Zhejiang Lanke Breeding Biotechnology Co., LTD.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2025.1567872/full#supplementary-material
Alsop D., Brown S., Kraak G. V. D. (2001). Development of a retinoic acid receptor-binding assay with rainbow trout tissue: characterization of retinoic acid binding, receptor tissue distribution, and developmental changes. Gen. Comp. Endocrinol. 123, 254–267. doi: 10.1006/gcen.2001.7659
Amengual J., Ribot J., Bonet M. L., Palou A. (2012). Retinoic acid treatment increases lipid oxidation capacity in skeletal muscle of mice. Obesity 16, 585–591. doi: 10.1038/oby.2007.104
AOAC (1995). Official Methods of Analysis of the Association of Official Analytical Chemists. 16th ed (Arlington, VA, USA: ACOC).
Assefa A., Abunna F. (2018). Maintenance of fish health in aquaculture: review of epidemiological approaches for prevention and control of infectious disease of fish. Vet. Med. Int. 2018, 5432497. doi: 10.1155/2018/5432497
Bonet M. L., Ribot J., Palou A. (2012). Lipid metabolism in mammalian tissues and its control by retinoic acid. Biochim. Biophys. Acta (BBA)-Molecular Cell Biol. lipids. 1821, 177–189. doi: 10.1016/j.bbalip.2011.06.001
Bu X., Wang X., Lin Z., Wang C., Li L., Liu S., et al. (2022). Myo-inositol improves growth performance and regulates lipid metabolism of juvenile Chinese mitten crab (Eriocheir sinensis) fed different percentage of lipid. Brit J. Nutr. 127, 666–678. doi: 10.1017/S0007114521001409
China Fishery Statistical Yearbook (2024). China Fishery Statistical Yearbook 2024 (Beijing, China: China Agriculture Press), 24.
Diwan A., Harke S. N., Panche A. N. (2023). Studies on exploring the potentials of gut microbiomes to mitigate the bacterial and viral diseases of fish and shellfish in aquaculture farming. Microbe. 2, 100031. doi: 10.1016/j.microb.2023.100031
Dong X., Wang J., Ji P., Gao X., Sun L., Miao S., et al. (2020). Dietary betaine supplementation promotes growth, n-3 LC-PUFA content and innate immunity in Macrobrachium rosenbergii. Aquaculture. 525, 735308. doi: 10.1016/j.aquaculture.2020.735308
Eriksen A. B., Indrevær R. L., Holm K. L., Landskron J., Blomhoff H. K. (2012). TLR9-signaling is required for turning retinoic acid into a potent stimulator of RP105 (CD180)-mediated proliferation and IgG synthesis in human memory B cells. Cell. Immunol. 279, 87–95. doi: 10.1016/j.cellimm.2012.09.003
Espinosa-Diez C., Miguel V., Mennerich D., Kietzmann T., Sánchez-Pérez P., Cadenas S., et al. (2015). Antioxidant responses and cellular adjustments to oxidative stress. Redox Biol. 6, 183–197. doi: 10.1016/j.redox.2015.07.008
Farazuddin M., Acker G., Zourob J., O’Konek J. J., Wong P. T., Morris S., et al. (2024). Inhibiting retinoic acid signaling in dendritic cells suppresses respiratory syncytial virus infection through enhanced antiviral immunity. iScience 27 (7), 110103. doi: 10.1016/j.isci.2024.110103
Girish B., Swetha C., Srilatha M., Hemalatha M., Reddy P. S. (2018). Evidence for retinoic acid involvement in the regulation of vitellogenesis in the fresh water edible crab, Oziotelphusa senex senex. Comp. Biochem. Physiol. Part A: Mol. Integr. Physiol. 222, 1–6. doi: 10.1016/j.cbpa.2018.04.003
Guo L., Zhang Y., Liu H., Cheng Q., Yang S., Yang D. (2022). All-trans retinoic acid inhibits the osteogenesis of periodontal ligament stem cells by promoting IL-1β production via NF-κB signaling. Int. Immunopharmacol. 108, 108757. doi: 10.1016/j.intimp.2022.108757
Han L., Liu Y., Lu M., Wang H., Tang F. (2018). Retinoic acid modulates iron metabolism imbalance in anemia of inflammation induced by LPS via reversely regulating hepcidin and ferroportin expression. BBRC. 507, 280–285. doi: 10.1016/j.bbrc.2018.11.022
Hao R., Zhao M., Tayyab M., Lin Z., Zhang Y. (2024). The mucosal immunity in crustaceans: Inferences from other species. Fish Shellfish Immunol. 152, 109785. doi: 10.1016/j.fsi.2024.109785
Huang Y., Ma F., Wang W., Ren Q. (2015). Identification and molecular characterization of a peritrophin-like gene, involved in the antibacterial response in Chinese mitten crab, Eriocheir sinensis. Dev. Comp. Immunol. 50, 129–138. doi: 10.1016/j.dci.2015.01.002
Huang Q., Wang X., Bu X., Song Y., You J., Zhang C., et al. (2022a). Dietary vitamin A affects growth performance, immunity, antioxidant capacity, and lipid metabolism of juvenile Chinese mitten crab Eriocheir sinensis. Aquaculture. 548, 737556. doi: 10.1016/j.aquaculture.2021.737556
Huang Q., You J., Wang X., Bu X., Song Y., Zhang C., et al. (2022b). Effect of vitamin A supplement on the growth performance, antioxidant status, and lipid accumulation of Chinese mitten crab Eriocheir Sinensis fed different lipid levels. Aquaculture. 554, 738123. doi: 10.1016/j.aquaculture.2022.738123
Islam S. I., Mahfuj S., Baqar Z., Asadujjaman M., Islam M. J., Alsiwiehri N., et al. (2024). Bacterial diseases of Asian sea bass (Lates calcarifer): A review for health management strategies and future aquaculture sustainability. Heliyon. 10, 9. doi: 10.1016/j.heliyon.2024.e29793
Jacobo-Delgado Y. M., Torres-Juarez F., Rodríguez-Carlos A., Santos-Mena A., Enciso-Moreno J. E., Rivas-Santiago C., et al. (2021). Retinoic acid induces antimicrobial peptides and cytokines leading to Mycobacterium tuberculosis elimination in airway epithelial cells. Peptides. 142, 170580. doi: 10.1016/j.peptides.2021.170580
Jiang W., Jia X., Zhang M., Qiang W., Shen X., Jiang G., et al. (2024). The effect of dietary vitamin A supplementation on growth, gonadal development, molting, immunity, and ammonia nitrogen resistance in sub-adult male Chinese mitten crab, Eriocheir sinensis. Aquaculture. 581, 740451. doi: 10.1016/j.aquaculture.2023.740451
Karagianni P., Talianidis I. (2015). Transcription factor networks regulating hepatic fatty acid metabolism. Biochim. Biophys. Acta 1851, 2–8. doi: 10.1016/j.bbalip.2014.05.001
Lai X., Wu A., Bing Y., Liu Y., Luo J., Yan H., et al. (2023). Retinoic acid protects against lipopolysaccharide-induced ferroptotic liver injury and iron disorders by regulating Nrf2/HO-1 and RARβ signaling. Free Radical Biol. Med. 205, 202–213. doi: 10.1016/j.freeradbiomed.2023.06.003
Li X. S., Cui K., Fang W., Chen Q., Xu D., Mai K. S., et al. (2019). High level of dietary olive oil decreased growth, increased liver lipid deposition and induced inflammation by activating the p38 MAPK and JNK pathways in large yellow croaker (Larimichthys crocea). Fish Shellfish Immunol. 94, 157–165. doi: 10.1016/j.fsi.2019.08.062
Li Y., Xu Q., Gepan Y., Liu H., Su J., Dai X. (2021). Molecular cloning, characterization and RNA interference assay of two toll-like receptors in giant freshwater prawn, Macrobrachium rosenbergii. Aquaculture Rep. 20, 100643. doi: 10.1016/j.aqrep.2021.100643
Li Z., Zhang G., Pan K., Niu X., Shu-Chien A. C., Chen T., et al. (2023). Functional transcriptome reveals hepatopancreatic lipid metabolism during the molting cycle of the Chinese mitten crab Eriocheir sinensis. Comp. Biochem. Physiol. Part A: Mol. Integr. Physiol. 284, 111474. doi: 10.2139/ssrn.4353414
Liu M., Gao Q., Sun C., Liu B., Liu X., Zhou Q., et al. (2022a). Effects of dietary tea tree oil on the growth, physiological and non-specific immunity response in the giant freshwater prawn (Macrobrachium rosenbergii) under high ammonia stress. Fish Shellfish Immunol. 120, 458–469. doi: 10.1016/j.fsi.2021.12.025
Liu Y., Zhou X., Liu B., Gao Q., Sun C., Zhou Q., et al. (2022b). Effects of high fat in the diet on growth, antioxidant, immunity and fat deposition of Macrobrachium rosenbergii post-larvae. Fish Shellfish Immunol. 129, 13–21. doi: 10.1016/j.fsi.2022.08.027
Lu K. Y., Sung H. J., Liu C. L., Sung H. H. (2009). Differentially enhanced gene expression in hemocytes from Macrobrachium rosenbergii challenged in vivo with lipopolysaccharide. J. Invertebr. Pathol. 100, 9–15. doi: 10.1016/j.jip.2008.09.005
Ngo J., Choi D. W., Stanley I. A., Stiles L., Molina A. J., Chen P. H., et al. (2023). Mitochondrial morphology controls fatty acid utilization by changing CPT1 sensitivity to malonyl-CoA. EMBO J. 42, e111901. doi: 10.15252/embj.2022111901
Nhieu J., Lin Y. L., Wei L. N. (2020). Noncanonical retinoic acid signaling. Methods Enzymol. 637, 261–281. doi: 10.1016/bs.mie.2020.02.012
Niu S., Zhu Y., Geng R., Chen N., Luo M., Zuo H., et al. (2023). A novel Toll receptor from Pacific white shrimp Penaeus vannamei is involved in immune defense against Vibrio parahaemolyticus infection. Aquaculture. 569, 739396. doi: 10.1016/j.aquaculture.2023.739396
Ogata H. Y., Oku H. (2001). The effects of dietary retinoic acid on body lipid deposition in juvenile red sea bream (Pagrus major); a preliminary study. Aquaculture. 193, 271–279. doi: 10.1016/S0044-8486(00)00496-8
Peng C., Wang Q., Chen J., Yang H., Zhang W., Wang D., et al. (2020). Retinoic acid and androgen influence germ cells development and meiotic initiation in juvenile orange-spotted grouper, Epinephelus coioides. Gen. Comp. Endocrinol. 289, 113379. doi: 10.1016/j.ygcen.2019.113379
Qian Q., Zhou L., Chen A., Tang X., Gao X., Zhang Y., et al. (2025). Decapod iridescent virus 1 replication dynamics in Macrobrachium rosenbergii at different water temperatures and the host immune response. Aquaculture 599, 742113. doi: 10.1016/j.aquaculture.2024.742113
Qin C., Jiang Y., Chen X., Bian Y., Wang Y., Xie K., et al. (2021). Dexmedetomidine protects against burn-induced intestinal barrier injury via the MLCK/p-MLC signalling pathway. Burns. 47, 1576–1585. doi: 10.1016/j.burns.2021.01.017
Rattanavichai W., Cheng W. (2014). Effects of hot-water extract of banana (Musa acuminata) fruit’s peel on the antibacterial activity, and anti-hypothermal stress, immune responses and disease resistance of the giant freshwater prawn, Macrobrachium rosenbegii. Fish Shellfish Immunol. 39, 326–335. doi: 10.1016/j.fsi.2014.05.031
Ren Q., Zhang Z., Li X. C., Jie D., Hui K. M., Zhang C. Y., et al. (2012). Three different anti-lipopolysaccharide factors identified from giant freshwater prawn, Macrobrachium rosenbergii. Fish Shellfish Immunol. 33, 766–774. doi: 10.1016/j.fsi.2012.06.032
Roder K., Zhang L., Schweizer M. (2007). SREBP-1c mediates the retinoid-dependent increase in fatty acid synthase promoter activity in HepG2. FEBS Lett. 581, 2715–2720. doi: 10.1016/j.febslet.2007.05.022
Salam M. A., Das T. R., Paul S. I., Islam F., Baidya A., Rahman M. L., et al. (2024). Dietary chitosan positively influences the immunity and reproductive performances of mature silver barb (Barbonymus gonionotus). Aquaculture Rep. 36, 102155. doi: 10.1016/j.aqrep.2024.102155
Shi B., Jin M., Jiao L., Betancor M. B., Tocher D. R., Zhou Q. (2020). Effects of dietary zinc level on growth performance, lipolysis and expression of genes involved in the calcium/calmodulin-dependent protein kinase kinase-beta/amp-activated protein kinase pathway in juvenile pacific white shrimp. Brit J. Nutr. 124, 773–784. doi: 10.1017/S0007114520001725
Shiau S. Y., Chen Y. (2000). Estimation of the dietary vitamin A requirement of juvenile grass shrimp, Penaeus monodon. J. Nutr. 130, 90–94. doi: 10.1093/jn/130.1.90
Si Q., Min X., Dai X., Gao Q., Jiang Q., Ren Q. (2024). Diversity of MrTolls and their regulation of antimicrobial peptides expression during Enterobacter cloacae infection in Macrobrachium rosenbergii. Fish Shellfish Immunol. 144, 109279. doi: 10.1016/j.fsi.2023.109279
Tanoury Z. A., Piskunov A., Rochette-Egly C. (2013). Vitamin A and retinoid signaling: genomic and nongenomic effects. J. Lipid Res. 54, 1761–1775. doi: 10.1194/jlr.R030833
Tao L., Lu H., Xiong J., Zhang L., Sun W., Shan X. (2024). The application and potential of postbiotics as sustainable feed additives in aquaculture. Aquaculture. 592, 741237. doi: 10.1016/j.aquaculture.2024.741237
Tung Y. T., Lyu K. N., Chun Chen Y., Chien Y. W. (2019). Supplementation comprising dietary fish oil with all-trans retinoic acid decreased blood lipids and fat accumulation in C57BL/6J mice. J. Funct. Foods. 52, 310–315. doi: 10.1016/j.jff.2018.11.010
Wang X., Ma T., Wei C., Liu J., Yu T., Zou Y., et al. (2023). Toxic effects of exogenous retinoic acid on the neurodevelopment of zebrafish (Danio rerio) embryos. Neurotoxicol. Teratol. 100, 107291. doi: 10.1016/j.ntt.2023.107291
Wang L., Xu S., Zhou M., Hu H., Li J. (2024). The role of DGAT1 and DGAT2 in tumor progression via fatty acid metabolism: A comprehensive review. Int. J. Biol. Macromol. 278, 134835. doi: 10.1016/j.ijbiomac.2024.134835
Wen Q., Wang W., Shi L., Wang C., Chan S. F. (2022). Molecular and functional characterization of an astakine cDNA from the giant freshwater prawn Macrobrachium rosenbergii. Aquaculture Rep. 24, 101165. doi: 10.1016/j.aqrep.2022.101165
Wu L., Zhou Y., Abbas M. N., Kausar S., Chen Q., Jiang C. X., et al. (2017). Molecular structure and functional characterization of the peroxiredoxin 5 in Procambarus clarkii following LPS and Poly I:C challenge. Fish Shellfish Immunol. 71, 28–34. doi: 10.1016/j.fsi.2017.09.072
Xu J., Peng X., Li F., Zhang S., Wang T., Dong X., et al. (2024). Effects of astaxanthin in diet on antioxidant capacity and molting-related genes expression of larvae giant freshwater prawn (Macrobrachium rosenbergii). Aquaculture Rep. 35, 101956. doi: 10.1016/j.aqrep.2024.101956
Yang Q. H., Ding M. Y., Tan B. P., Dong X. H., Chi S. Y., Zhang S., et al. (2017). Effects of dietary vitamin A on growth, feed utilization, lipid metabolism enzyme activities, and fatty acid synthase and hepatic lipase mRNA expression levels in the liver of juvenile orange spotted grouper, Epinephelus coioides. Aquaculture. 479, 501–507. doi: 10.1016/j.aquaculture.2017.06.024
Yeung K. W. Y., Zhou G. J., Hilscherová K., Giesy J. P., Leung K. M. Y. (2020). Current understanding of potential ecological risks of retinoic acids and their metabolites in aquatic environments. Environ. Int. 136, 105464. doi: 10.1016/j.envint.2020.105464
Yohana M. A., Ray G. W., Yang Q., Kou S., Tan B., Wu J., et al. (2024). Protective effects of butyric acid during heat stress on the survival, immune response, histopathology, and gene expression in the hepatopancreas of juvenile pacific shrimp (L. Vannamei). Fish Shellfish Immunol. 150, 109610. doi: 10.1016/j.fsi.2024.109610
Zhan W., Peng H., Xie S., Deng Y., Zhu T., Cui Y., et al. (2024). Dietary lauric acid promoted antioxidant and immune capacity by improving intestinal structure and microbial population of swimming crab (Portunus trituberculatus). Fish Shellfish Immunol. 151, 109739. doi: 10.1016/j.fsi.2024.109739
Zheng X., Xu X., Liu M., Yang J., Yuan M., Sun C., et al. (2024). Bile acid and short chain fatty acid metabolism of gut microbiota mediate high-fat diet induced intestinal barrier damage in Macrobrachium rosenbergii. Fish Shellfish Immunol. 146, 109376. doi: 10.1016/j.fsi.2024.109376
Zhong Y., Pan Y., Liu L., Li H., Li Y., Jiang J., et al. (2020). Effects of high fat diet on lipid accumulation, oxidative stress and autophagy in the liver of Chinese softshell turtle (Pelodiscus sinensis). Comp. Biochem. Physiol. Part B: Biochem. Mol. Biol. 240, 110331. doi: 10.1016/j.cbpb.2019.110331
Zhu F. (2020). A review on the application of herbal medicines in the disease control of aquatic animals. Aquaculture. 526, 735422. doi: 10.1016/j.aquaculture.2020.735422
Ziad A. T., Piskunov A., Rochette-Egly C. (2013). Vitamin A and retinoid signaling: genomic and nongenomic effects: thematic review series: fat-soluble vitamins: vitamin A. J. Lipid Res. 54, 1761–1775. doi: 10.1194/jlr.R030833
Keywords: retinoic acid, Macrobrachium rosenbergii, immunity, lipid metabolism, growth
Citation: Huang Q-C, Wang L, Gu Z-M, Liu C, Ye T-T, Yan J-J, Meng J-L, Zheng Y-T, Ge X-P, Lin Z-D and Zou G-Y (2025) Dietary retinoic acid improved the growth, lipid metabolism and immune status in Macrobrachium rosenbergii. Front. Mar. Sci. 12:1567872. doi: 10.3389/fmars.2025.1567872
Received: 28 January 2025; Accepted: 04 March 2025;
Published: 27 March 2025.
Edited by:
Xueshan Li, Jimei University, ChinaReviewed by:
Xiaohui Dong, Guangdong Ocean University, ChinaCopyright © 2025 Huang, Wang, Gu, Liu, Ye, Yan, Meng, Zheng, Ge, Lin and Zou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhi-Min Gu, Z3V6aGltaW4yMDA2QDE2My5jb20=; Xian-Ping Ge, Z2V4cEBmZnJjLmNu
†ORCID: Qin-Cheng Huang, orcid.org/0009-0009-8181-8587
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.