
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Mar. Sci. , 01 April 2025
Sec. Marine Biology
Volume 12 - 2025 | https://doi.org/10.3389/fmars.2025.1567441
The age composition of Todaropsis eblanae from the Sardinian waters (western Mediterranean Sea) was studied for the first time through the analysis of growth increments and the comparison of three structures: statoliths, beaks and eye lenses. The analysis was performed on 270 wild specimens of both sexes at different sizes (45-200 mm of mantle length; 6.98-443 g of total weight) and maturity stages (immature, maturing and mature) caught from July to September by trawl net. Significant differences in growth and length-weight relationship were observed between sexes, due to females reach a larger size than males. All the three structures had dimensions positively correlated with the size of the animals and showed clearly readable growth increments. Low values of IAPE, CV and PA confirmed the accuracy and good reproducibility of age readings. Eye lenses showed a very high number of growth increments (106-640), and a daily deposition was excluded. In contrast, beaks and statoliths showed NI values (70-316 and 73-310, respectively) always consistent with the size and maturity of the specimens, then a daily deposition has been suggested and their value compared. Moreover, the Mann-Whitney W-test confirmed a highly significant relationship between the number of growth increments in beaks and statoliths, suggesting that the beak can be considered a valid alternative to statoliths for age estimation in T. eblanae. The absolute growth rates confirm that females grow faster than males. Both sexes showed a higher initial growth rate, which gradually decreases, with the highest values at the age of 101-151 days, before reaching sexual maturity. According to a semelparous cycle, the estimated ages for the largest mature female (310-316 days) and male (288-292 days) suggest a lifespan of less than one year. Overall, the age and growth results reported, although referred to a specific area and a short sampling, could represent useful knowledge for a correct evaluation and management of this important commercial species in the future.
The lesser flying squid Todaropsis eblanae (Ball, 1841) is a neritic demersal species occurring in the eastern Atlantic Ocean from 61° north to 36° south latitude, in the Mediterranean Sea, the western Indian Ocean, the western Pacific Ocean, the South China Sea and Australian waters. Recent findings extend its distribution north to the Arctic (Roper et al., 2010). The distribution and abundance of T. eblanae (Hastie et al., 2009; Oesterwind et al., 2014; Lauria et al., 2016) and some reproductive aspects related mainly to the Atlantic and Indian populations (Hernández-García, 2002; Sabirov et al., 2012; Robin et al., 2002; Petroni, 2012) have been investigated. A genetic study on this species indicated the existence of at least 3 genetically isolated populations in the Eastern Atlantic (Dillane et al., 2005). Within the ICES European areas, commercial catches of T. eblanae together with other Ommastrephid squids such as Illex coindetii, Ommastrephes caroli and Todarodes sagittatus account for at least 49.8%-65.9% of total squid catches from 2015 to 2020 (FAO, 2020; Lishchenko et al., 2021). In the central western Mediterranean and in particular in Italian waters, T. eblanae represents a conspicuous fraction of bottom trawl commercial catches, forming the commercial category “totani” together with Illex coindetii (Belcari, 2017; Roper et al., 2010). The need to properly identify landings of Ommastrephid species has been emphasized because many stocks have not yet been assessed (Belcari et al., 2015 and Arkhipkin et al., 2015). Knowledge of biological aspects such as reproduction, lifespan, age and growth are essential elements for stock assessment and sustainable exploitation of cephalopods species (Arkhipkin et al., 2021). Generalizing, cephalopods are known for having short life cycles (max 2-3 years) and rapid and continuous growth that ends rather abruptly with the maturation of the gonads. The time allotted for growth can be modified mainly by temperature and available food (Forsythe and Van Heukelem, 1987).
To date, the analysis of the hard structures (i.e. statolith, beak, gladius, stylet, cuttlefish bone and eye lens) is the most widely used method to assess the age and growth of cephalopods. These structures can indeed ‘remember’ ontogenetic events by forming periodic growth increments. Daily deposition in beak has been validated for Octopus vulgaris (Perales-Raya et al., 2014) and then, this structure has been used for studying aging in several species (Xavier et al., 2022). Until now, the deposition of growth increments in squid beaks has been validated only on some Ommastrephid species such as Dosidicus gigas, Ommastrephes bartramii, Illex argentinus and Todarodes pacificus (Sakai et al., 2007; Liu et al., 2015; Fang et al., 2016).
The most common structures used to study age in cephalopods are the statoliths, despite their preparation being laborious and time-consuming; however, their reliability and daily periodicity of microincrements have been demonstrated in several cephalopods (Arkhipkin et al., 2018; Agus et al., 2024) and also in Ommastrephid squids (Hurley et al., 1985; Nakamura and Sakurai, 1991). Other hard structures, such as stylets, gladius, and eye lenses, have also been investigated to assess their potential use in age estimation (Arkhipkin and Bizikov, 1991; Hermosilla et al., 2010; Rodríguez-Domínguez et al., 2013; Agus et al., 2018). Growth patterns in the eye lens have been studied in Sepia officinalis, Enteroctopus megalocyatus, Octopus maya, Loligo vulgaris, Loligo forbesii and Ommastrephes caroli (Clarke, 1993; Baqueiro-Cárdenas et al., 2011; Rodríguez-Domínguez et al., 2013; Agus et al., 2018, 2021) but results were not always satisfactory (Rodríguez-Domínguez et al., 2013). Focusing on T. eblanae, preliminary data on the age and growth have so far been investigated by analyzing the statoliths of specimens from the north-east Atlantic (Robin et al., 2002), African waters (Arkhipkin and Laptikhovsky, 2000) as well as in Mediterranean individuals from Ligurian (Cavanna et al., 2008) and Ionian waters (Fotiadis et al., 2015). In view of the fragmentary knowledge of the biology of this species in the Mediterranean Sea, age and growth are addressed through the analysis and comparison of three hard structures (statolith, beak and eye lens) in the two sexes at different sexual maturity providing a complete picture of the life history of T. eblanae.
Overall, 270 individuals of T. eblanae were collected at depths between 30 to 570 m from the waters around Sardinia (Central-Western Mediterranean Sea, FAO GFCM Geographical subarea GSA 11) from July to September 2023 during Mediterranean International Trawl Survey activities (MEDITS, Spedicato et al., 2020) (Supplementary Figure 1). Dead specimens were immediately frozen on board and then transported to the laboratory for analysis. Dorsal mantle length (DML) was measured to the nearest 1 mm and total weight (TW) was recorded to the nearest 0.01g. For each squid, sex and maturity stage were assigned following the macroscopic scale in Follesa and Carbonara (2019). Length–weight relationships were calculated separately for each sex, according to the function TW = a × DMLb, where TW is the total weight (g), DML is the Dorsal mantle length (mm), a is the intercept of the regression and b is the regression coefficient or slope. The growth was assessed by comparing the slope b to the isometric value of 3, by Student’s t-test, between males and females. Moreover, The Kolmogorov-Smirnov (KS) two-sample test was used to investigate differences between the length-frequency and total weight distributions by sexes (Zar, 1999). All statistical tests were performed using the software Statgraphics Centurion XVI.
Statoliths were surgically extracted from the cephalic cartilage and their maximum length (SL) was taken, with an accuracy of 0.01mm from the end of the dorsal dome to the top of the rostrum (Supplementary Figure 2). According to Ceriola and Milone (2007), the concave side of statolith was mounted on a microscope slide using Crystal Bond TM thermoplastic resin dissolved at 110–140°C. Then, both sides of the statoliths were grinded and polished using lapping film sheets with 30, 12, 5 and 0.5 μm grades. The total number of growth increments (NI) was counted from the natal ring towards the edge of the lateral dome (Ceriola and Milone, 2007). Eye lenses, once extracted, were cleaned with distilled water, measured using a digital gauge (accurate to a tenth of a millimeter) (Supplementary Figure 3) and fixed in 5% formalin, according to Baqueiro-Cárdenas et al. (2011). Lenses were kept under running water for 24 h and then put under a laminar flow hood for a few hours, to allow the complete drying of the structure (Agus et al., 2018). Then, they were cut into two parts, incorporated into epoxy resin and ground using a lapping machine with waterproof sandpaper at different grit (320, 600, 800 and 1000) until a section of 1 mm thickness was attained. The sections were mounted on a microscope slide and stained using Gill’s Ematoxilin and Eosin and examined using an optical microscope equipped with a CANON EOS 1100 D camera to take digital images (magnification: 100× and 400×). Growth increments (NI) were counted starting from the center (nucleus) until the outer end of the lens. To avoid double counting, each eye lens increment was marked and numbered using the Tps_Dig2 software. Beaks were removed, cleaned with distilled water and stored in 75% ethanol and according to Clarke (1986) the following measurements were taken: upper crest length (uCL) and the upper rostral length (uRL) (Supplementary Figure 4). Following Liu et al. (2015, 2017) only the upper beak was considered for age analysis. Upper beaks were sectioned following the sagittal axis along the posterior edge of the hood and crest to the rostral tip, keeping the cutting line slightly to one side, to avoid any possible damage to the Rostrum Sagittal Section (RSS). One of the two sections was embedded in epoxy resin, left to harden for 24 h and glued to a microscope slide. Then, it was ground parallel to the cutting plane using a lapping machine with waterproof sandpaper 320, 600, 800 and 1000 grit, to approach the rostrum sagittal section. Beak increments (NI) were observed and photographed using the same optical microscope already described (magnifications:100x). NI in the RSS were counted slightly further from the Internal Rostral Axis (IRA). The morphometric measurements of all the three hard structures were related to the size of the animals (DML), through a regression analysis.
The increment counting (NI) in the three structures were performed twice by three experienced readers independently, assuming a daily deposition of the bands (Arkhipkin et al., 2018).
The precision and accuracy of the overall readings were assessed by the Index of Average Percent Error (IAPE) (Beamish and Fournier, 1981), calculated as follows:
where N equals the number of samples aged, R represents the number of readings, Xij is the ith age determination of the jth fish and Xj equals the average age calculated for the jth fish. In addition, the Coefficient of Variation (%CV) (Chang, 1982) and the Percentage of Agreement (%A) were also calculated.
The relationship between NI in the three different structures and the size of the animals (DML) were tested with 4 different curve models: linear, exponential, power and logarithmic and the Akaike Information Criterion (AIC) was calculated to choose which model best describes the growth of the sampled specimens (Akaike, 1974).
The absolute growth rate (AGR; mm d−1) was calculated separately for beaks and statoliths for each 50-day interval by sex using the following equation (Gonzalez et al., 1996):
where R1 and R2 are the average ML (mm) at the beginning (T1) and end (T2) of the time interval, respectively. NI counted in both statoliths and beaks were correlated using a linear regression. Before being compared with the Mann-Whitney W-test to check whether significant differences existed between the increments of the two structures, the Shapiro-Wilk normality test was performed. All statistical analysis were made using R (4.4.2 version).
The sample was composed of 110 males (DML range = 61-142 mm; TW range = 17.56-179.83 g), 120 females (DML range = 62-200 mm; TW range = 21.30-443.00 g) at different maturity stages (immature, developing, maturing and mature), whereas the sex of forty specimens was not easily distinguishable macroscopically and they were classified as undetermined (DML range = 45-60 mm; TW range = 6.98- 20.05 g) (Table 1). The frequency distribution of length and weight, by sex are shown in Figure 1.
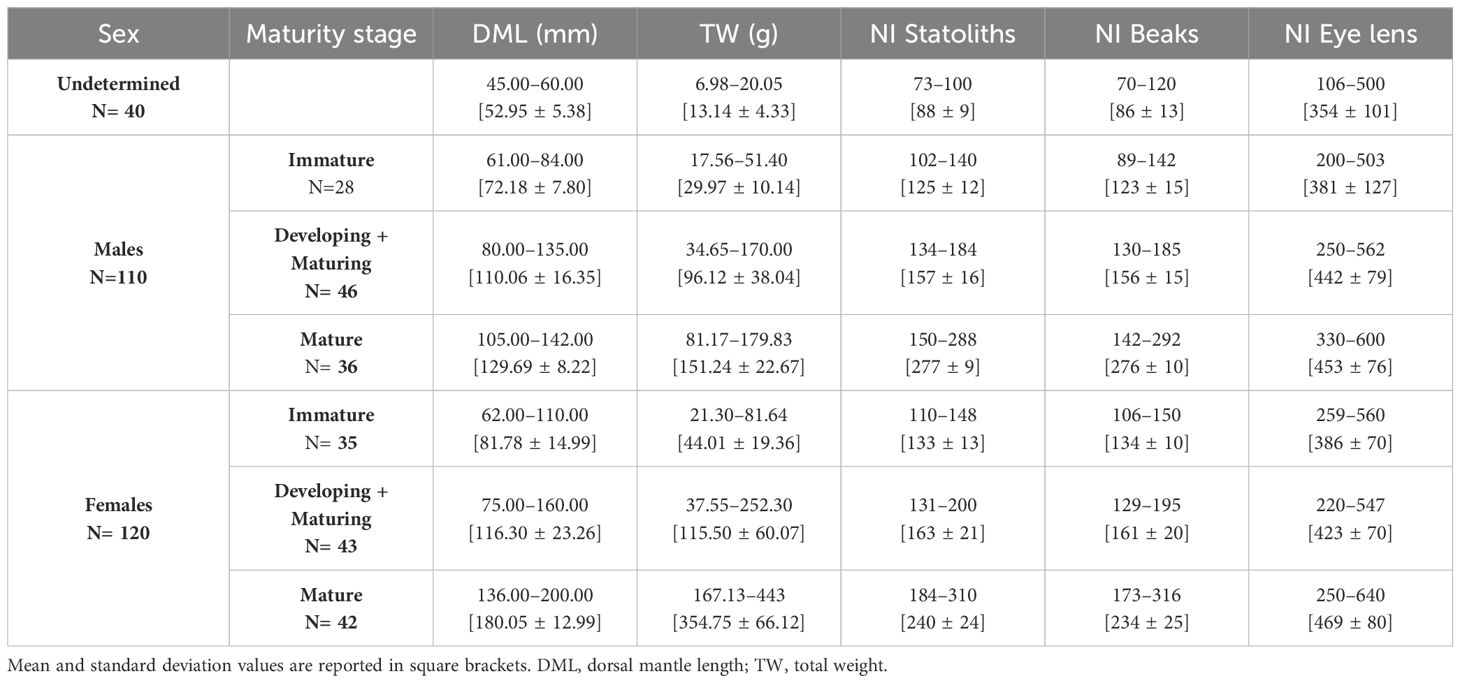
Table 1. Composition of sample per sex and number of increments (NI) counted in statoliths, beaks and eye lenses of 270 T. eblanae specimens, at different sexual maturity stages.
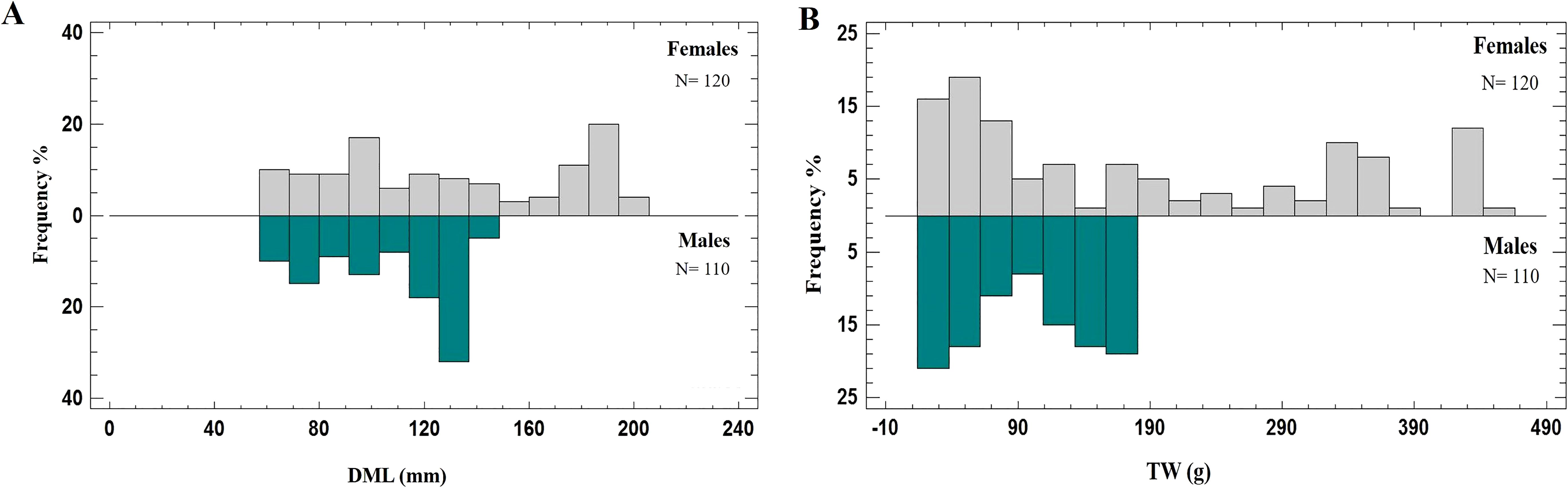
Figure 1. The frequency distributions of length and weight of Todaropsis eblanae for females (grey) and males (green): (A) dorsal mantle length (DML); (B) total weight (TW).
The length-weight relationship in females (TW= 0.0003 × DML 2.6773; r2 = 0.98), differed significantly from males (TW= 0.0002 × DML 2.7883; r2 = 0.97) with t-test P< 0.005. The Kolmogorov-Smirnov two sample test (P<0.05) further confirmed that females reach a statistically significant larger size and weight than males. Macroscopically all mature males had left and right ventral arms hectocotilized and a developed reproductive system with packed spermatophores in the Needham’s Sac (Figures 2A, B). Mature females had large Nedamental’s glands and ripe oocytes in the ovary and oviducts ready to be laid (Figures 2C, D). Mating signs were observed in females for the presence of implanted spermatangia in the buccal mass (Figure 2E), both in mature (36%) and maturing (12%) individuals.
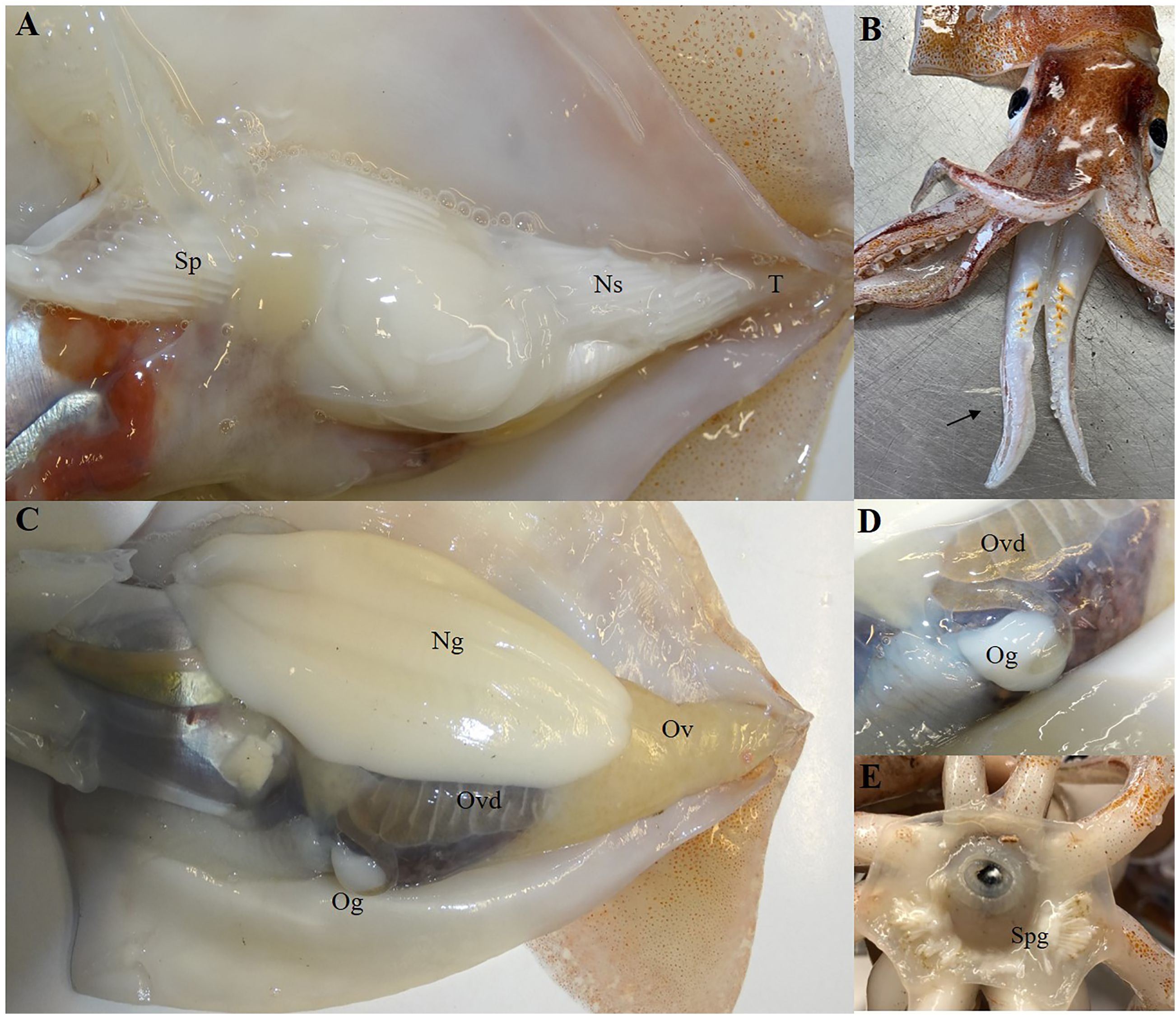
Figure 2. Macroscopic evidence of sexual maturity in male (A, B) and in female (CDE) of Todaropis eblanae. (A) male sexual apparatus; (B) ventral arms hectocotilized; (C) female sexual apparatus; (D) oviduct with ripe eggs and oviducal gland; E Buccal mass with spermatangia implanted. T, Testis; Ns, Needham’s Sac; Sp, spermatophores; Ov, Ovary; Ovd, Oviduct; Og, Oviduct gland; Ng, Nidamental gland; Spg, spermatangia.
The total length of the statolith (SL) ranged from 0.53 mm (45 mm DML, 8.29 TW; undetermined) to 1.35 mm in the biggest mature female (200 mm DML and 443 g TW). In the same specimens we also recorded the smallest (2.6 mm) and the largest (9.83 mm) eye lens diameter. As regard to the beak measurements, the smallest upper beak (uRL=2.11 mm, uCL=7.40 mm) was found in an undetermined specimen of 45 mm DML and the largest (uRL=8.70 mm, uCL=26.0 mm) belonged to the above mentioned biggest female. The linear relationship between the morphometric measurements of the three hard structures (statolith, beak, eye lens) and the size (DML) of the animals showed a significant positive correlation in all samples examined (Figure 3).
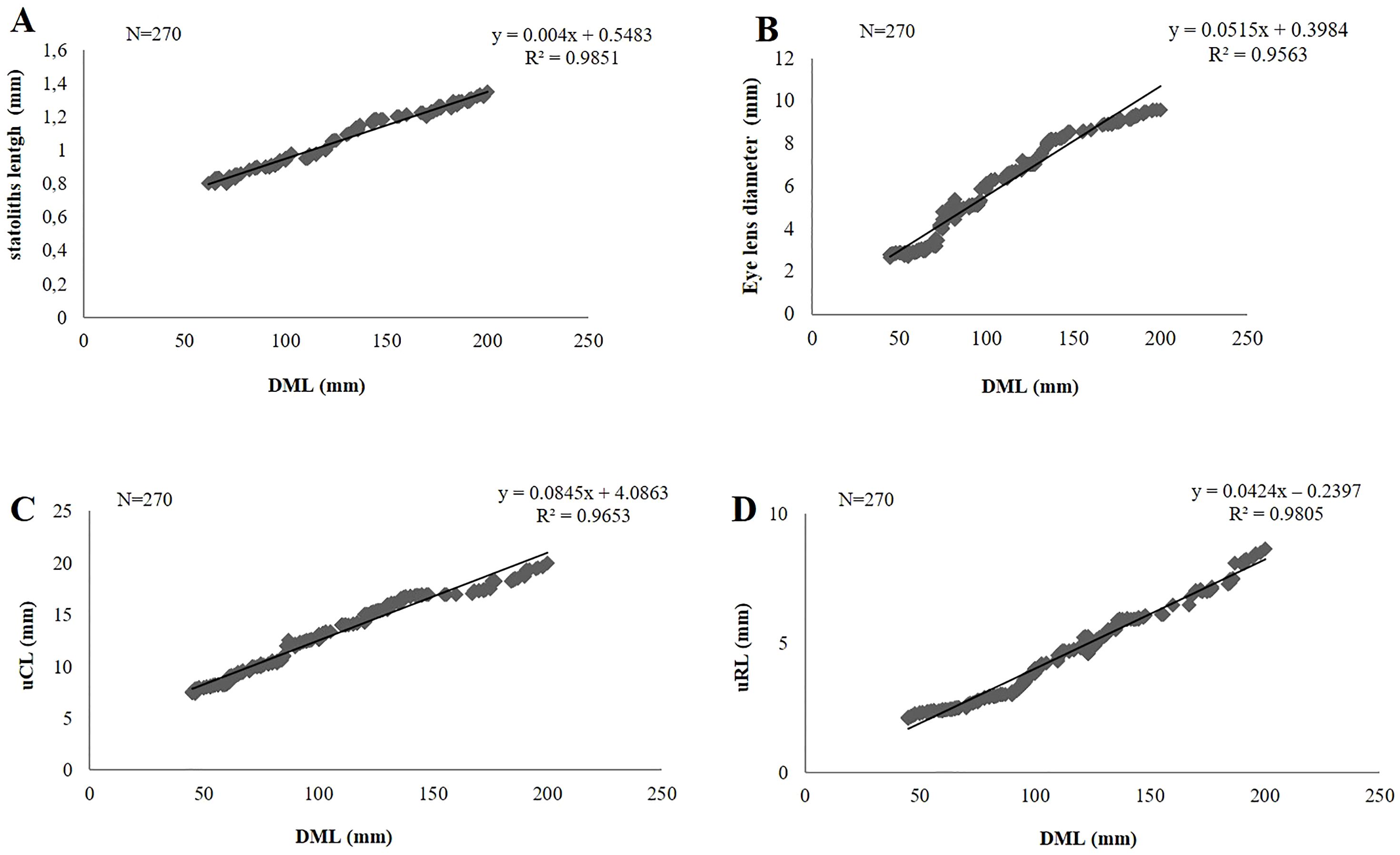
Figure 3. Linear relationship between the measurements of statoliths (A), eye lens (B) and beaks (C, D) vs. dorsal mantle length (DML) in Todaropsis eblanae.
All 270 statoliths, upper beaks and extracted eye lenses, were available for ageing analysis and were legible, clearly showing regular and well-defined growth increments (Figures 4A–C). IAPE, CV and PA values testify to a high level of accuracy and reproducibility of the readings in all structures (Table 2). The count of NI in statoliths showed similar values to those in beaks. The lowest values of 73 NI in the statoliths and 70 NI in the beaks were recorded in the smallest specimen (45 mm DML, 8.29 g TW; undetermined) while the highest values were found in the largest mature female (200 mm DML, 443.0 g TW) with 310 NI and 316 NI in the two structures, respectively. As for males, the highest numbers of NI in statoliths (288) and beaks (292) were observed in a mature specimen of 142 mm DML and 177.0 g TW (Table 1). NI counted in the eye lenses, in both sexes and for all maturity stages, were always considerably higher than those counted in the statoliths and beaks, even in the indeterminate specimens (average of 354 NI) reaching up to 600 NI in the eye lenses of the largest animals (Table 1). For both sexes and for the entire sample, the AIC test (Supplementary Tables 1, 2) showed that the linear model best describes the relationships between NI of statoliths and of beaks, with DML (Figure 5). A good relationship (R2 = 0.9966) was found plotting the increments counted in the statoliths against those observed in the beaks for each specimen (Figure 6). The Shapiro-Wilk test showed a not normal distribution (W=0.93, p-value = 0.0) and the application of the Mann-Whitney test (p-value =0,929878) confirms that there is not a statistically significant difference between the two variables at the 95,0% confidence. Daily growth rates, calculated separately for the two sexes and the two structures (beaks and statoliths), were highest at a young age and then gradually decreased. (Table 3). Females grow faster than males. Regarding the statoliths, the highest rates were recorded in age group II (101-151 days) with values respectively of 1.74 mm/d in females and of 1.67 mm/d in males. Considering the beaks, females showed higher values of 1.71 mm/day and 1.01 mm/day respectively in the age classes II (101-151 day) and III (152-202 days) while males in the I class<100 (1.25 mm/day). Regarding sexual maturity, indeterminate and immature specimens were only present in the first two age classes:<100 and 101-151 days of age, for both structures. Maturing males were in class II from 101 to 151 days of age, while maturing females were between 152 and 202 days of age (class III) for both statoliths and beaks. Mature specimens appear in age class II for males and class III for females, indicating that males reached maturity earlier than females. Considering beaks and statoliths respectively, males begin maturity at 142 and 150 days, while females at 173 and 184 days.

Figure 4. Growth increments marked with numbers (1-5) in statolith (A), upper beak (B) and eye lens (C) belonged to a maturing male of 126 mm DML and 123.14 g TW of Todaropsis eblanae.
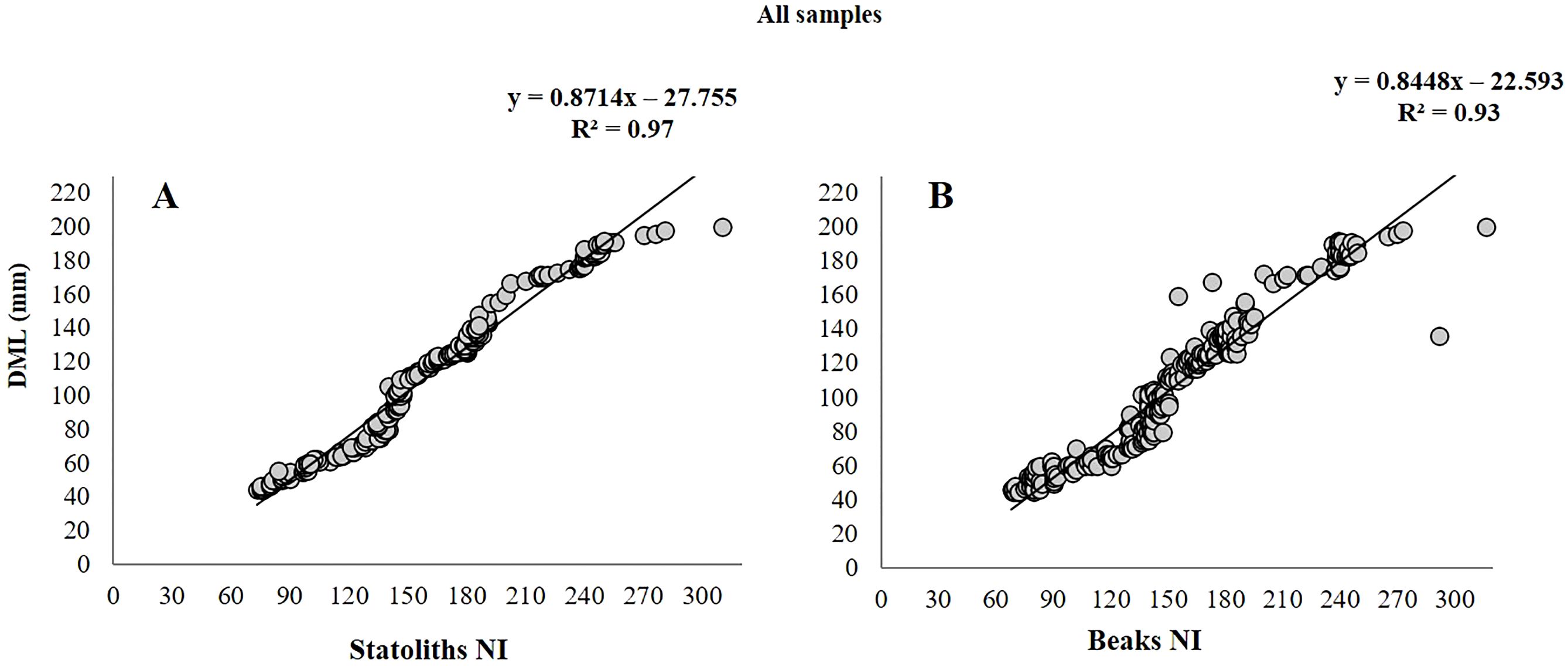
Figure 5. Plots of the number of increments (NI) counted in the statoliths (A) and in the beaks (B) vs. dorsal mantle length (DML) of 270 Todaropsis eblanae.
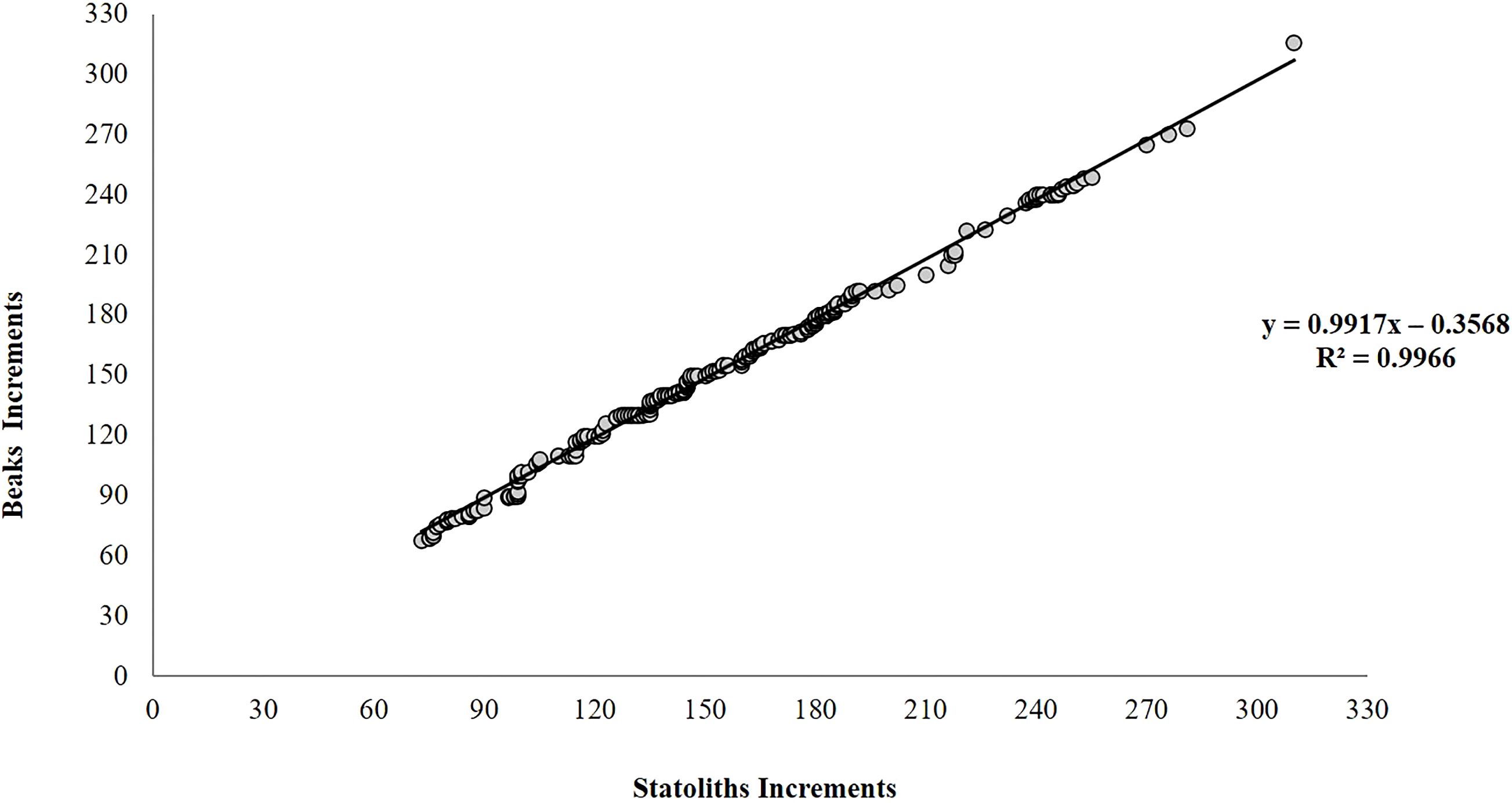
Figure 6. Relationship between the number of increments counted in beaks and statoliths belonging to the same specimens (N= 270) of Todaropsis eblanae.
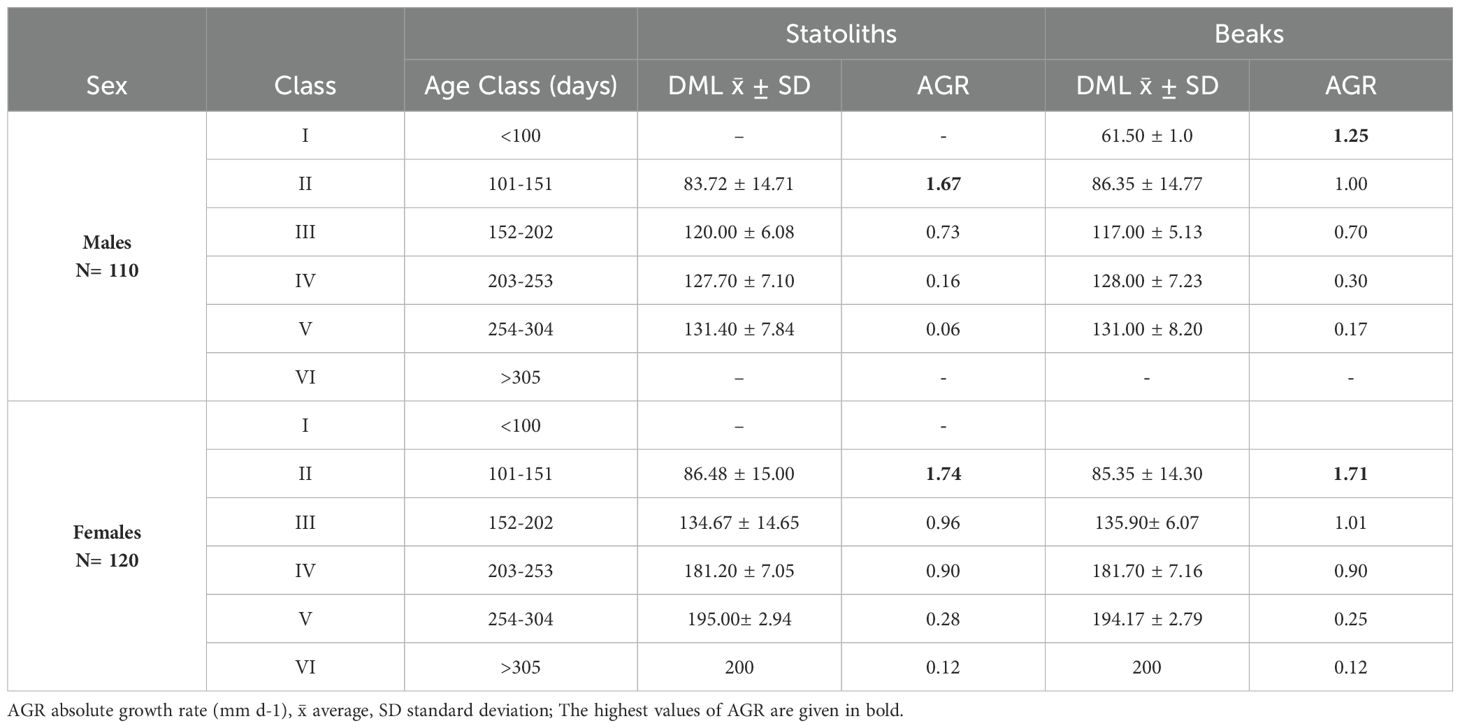
Table 3. Absolute growth rate (AGR) for mantle length for age class of increments in statoliths and beaks of Todaropsis eblanae by sex.
The sample of Todaropsis eblanae analyzed in this study showed that females are significantly larger than males in mantle length and weight, as already reported in literature (Roper et al., 2010). The maximum sizes recorded in Sardinian waters for females (200 mm DML;443 g WT) and males (142 mm DML; 180 g WT) are in agreement with other Mediterranean observations (Mangold-Wirz, 1963; Cavanna et al., 2008), but are smaller than in north Atlantic populations, where females reach a maximum size 290 mm DML against 220 mm DML in males (Robin et al., 2002). The significant differences between sexes found in the length-weight relationship were also observed in most of the studied regions in both the Atlantic and Mediterranean as well as a negative allometry (regression coefficient b<3) (Belcari et al., 2015) The present study represents the first age estimation in T. eblanae through the use of multiple hard structures (i.e., upper beaks, eye lenses, statoliths), implementing the so far available fragmentary information on the age of the species, obtained exclusively through the analysis of statoliths in both the Atlantic (Arkhipkin and Laptikhovsky, 2000; Robin et al., 2002) and the Mediterranean Sea (Cavanna et al., 2008; Fotiadis et al., 2015).
All the hard structures showed measurements positively correlated with the size of the animals and were easily readable with well-defined growth increments suggesting the feasibility of their use for age determination. In these structures, the overall CV, PA and IAPE values related to the counting of growth increments, demonstrate a high level of accuracy and reproducibility of readings (Campana, 2001). Although statoliths are the most common structures used for ageing in cephalopods and their reliability and daily periodicity have been validated or hypothesized in several species (Arkhipkin et al., 2018; Agus et al., 2024), in terms of processing effort, the smaller size of statoliths compared to beaks and eye lenses resulted in more laborious and time-consuming laboratory work. As regards the eye lens, to date, a daily deposition according to the size of the animals was suggested only in L. vulgaris, L. forbesii, and O. caroli (Agus et al., 2018, 2021). A sub-daily deposition has been proposed instead for the lenses of S. officinalis, E. megalocyatus and O. maya (Clarke, 1993; Baqueiro-Cárdenas et al., 2011; Rodríguez-Domínguez et al., 2013). In the present analysis, the considerable number of increments (up to 600) counted in the eye lenses of T. eblanae, significantly higher than those obtained with statoliths and beaks, clashes with a daily deposition, suggesting a different increment periodicity. Considering these results and according to Rodríguez-Domínguez et al. (2013), although this structure is considered attractive as a potential tool for aging, its use is not successful in all cephalopod species, and this variation suggests the need for further studies for its validation. On the contrary, the NI obtained from statoliths and beaks were similar and correlated to the size of the specimens of both sexes, as well as consistent/congruent with sexual maturity, in agreement with a daily deposition. The number of increments obtained from beaks and their positive comparison with statoliths (confirmed by Mann-Whitney test) prove for the first time that this hard structure is a promising and simple tool for age estimation also in T. eblanae, as already observed for other Ommastrephid squids: O. bartrami (Fang et al., 2016), D. gigas, I. argentinus, Stenotheuthis oualaniensis (Liu et al., 2015), O. caroli (Agus et al., 2021). Assuming daily increments in both statoliths and beaks and considering that the samples were taken mostly in summer, it can be assumed that the smallest, non-sexually determinable specimens of T. eblanae (around 13 g and 88 days old) were able to hatch in spring and that the large mature specimens, following a semelparous cycle, were close to death at less than a year old. Furthermore, our results showed a higher initial growth rate for both sexes (higher in females), which gradually decreases, with the highest values at the age of 101-151 days, before reaching sexual maturity. The same trend was observed also in Atlantic (Arkhipkin and Laptikhovsky, 2000). The attainment of maturity, which in males occurs at a smaller size (105 mm DML; 82 g WT) and age (142-150 days) than in females (136 mm DML; 167 g WT; 173-184 days), is in agreement with previous observations made in the Atlantic (Hernández-García, 2002; Gonzales et al., 1994; Rasero, 1996; Zumholz and Piatkowski, 2005) and in the Mediterranean Sea (Mangold-Wirz, 1963; Cavanna et al., 2008; Fotiadis et al., 2015). This precociousness in male maturation prompts males to mate with females that are not yet ready, as confirmed by the presence of implanted spermatangia in 12% of the buccal mass of maturing females. This characteristic (behavior) already observed for T. eblanae off NorthWest Africa (Hernández-García, 2002) is common in other cephalopods, especially octopods (e. g. Mangold, 1983; Cuccu et al., 2013; Agus et al., 2021, 2022).
This study, provides for the first time in T. eblanae the analysis of age at different stages of maturity using beaks, statoliths and eye lenses, and give a complete picture of the biological history of this species in Sardinian waters. Considering that advances in age estimation of cephalopods could enable the identification of seasonal cohorts and age-based assessment (Pierce et al., 2019), our results update to the still fragmentary knowledge on T. eblanae in the Mediterranean Sea that needs to be validated in other regions and environmental conditions in the future.
Overall, the results suggest a daily deposition for beaks and statoliths. The strong relationship between the increments observed in the beaks and statoliths confirms their potential use for determining age in T. eblanae of different sex, size and maturity stage. However, although the beak and the statolith provide similar results, the use of beak for age determination is preferred to the statolith because of the simplicity of its analysis. As for the eye lens, the high number of increments, considerably higher than that of statoliths and beaks, does not support daily deposition suggesting the need for further studies to assess which alternative deposition rate characterizes this structure.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethical approval was not required for the study involving animals in accordance with local legislation and institutional requirements, because the animals analysed were found dead.
BA: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Software, Writing – original draft, Writing – review & editing. PB: Data curation, Investigation, Methodology, Writing – review & editing. AB: Data curation, Formal Analysis, Investigation, Methodology, Writing – review & editing. RC: Data curation, Formal Analysis, Writing – review & editing. EC: Data curation, Formal Analysis, Writing – review & editing. DC: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Resources, Supervision, Writing – original draft, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. Data for the purposes of the analyses were obtained from the International Mediterranean Trawl survey MEDITS within the Data Collection Framework (EU Reg. 199/2008).
We would like to thank the crew of the ship ‘Gisella’ employed for the sampling.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2025.1567441/full#supplementary-material
Agus B., Carbonara P., Melis R., Cannas R., Carugati L., Cera J., et al. (2022). First Integrative Morphological and Genetic Characterization of Tremoctopus violaceus sensu stricto in the Mediterranean Sea. Animals 12, 80. doi: 10.3390/ani12010080
Agus B., Carugati L., Bellodi A., Cannas R., Cau A., Cera J., et al. (2021). Molecular and biological analysis on ommastrephes caroli findings in the central western mediterranean sea (Sardinian waters) including first age investigation using eye lenses and beaks. Front. Mar. Sci. 8. doi: 10.3389/fmars.2021.683856
Agus B., Mereu M., Cannas R., Cau A., Coluccia E., Follesa M. C., et al. (2018). Age determination of Loligo vulgaris and Loligo forbesii using eye lens analysis. Zoomorphology 137, 63–70. doi: 10.1007/s00435-017-0381-8
Agus B., Ruiu S., Cera J., Bellodi A., Pasquini V., Cuccu D. (2024). Age estimation in sepia officinalis using beaks and statoliths. Animals. 14, 2230. doi: 10.3390/ani14152230
Akaike H. (1974). A new look at the statistical model identification. IEEE Trans. Autom Control 19, 716–723. doi: 10.1109/TAC.1974.1100705
Arkhipkin A. I., Bizikov V. A. (1991). Comparative analysis of age and growth rates estimation using statoliths and gladius in squids. In: Jereb P., Ragonese S., von Boletzky S., editors. Squid age determination using statoliths. Mazara del Vallo, Italy: N.T.R.-I. T.P.P. (Spec. Publ. No. I.), 19–37.
Arkhipkin A. I., Bizikov V. A., Doubleday Z. A., Laptikhovsky V. V., Lishchenko F. V., Perales-Raya C., et al. (2018). Techniques for estimating the age and growth of Molluscs: Cephalopoda. J. Shellfish Res. 37, 783–793. doi: 10.2983/035.037.0409
Arkhipkin A. I., Hendrickson L. C., Payá I., Pierce G. J., Roa-Ureta R. H., Robin J. P., et al. (2021). Stock assessment and management of cephalopods: advances and challenges for short-lived fishery resources. ICES J. Mar. Sci. 78, 714–730. doi: 10.1093/icesjms/fsaa038
Arkhipkin A. I., Laptikhovsky V. V. (2000). Age and growth of the squid Todaropsis eblanae (Cephalopoda: Ommastrephidae) on the north-west African shelf. J. Mar. Biol. Assoc. UK. 80, 747–748. doi: 10.1017/S0025315400002642
Arkhipkin A. I., Rodhouse P. G. K., Pierce G. J., Sauer W., Sakai M., Allcock L., et al. (2015). World squid fisheries. Rev. Fisheries Sci. Aquaculture 23, 92–252. doi: 10.1080/23308249.2015.1026226
Baqueiro-Cárdenas E. R., Medrano Correa S., Contreras Guzman R., Barahona N., Briceño F., Villegas M. J., et al. (2011). Eye lens structure of the octopus Enteroctopus megalocyathus: evidence of growth. J. Shell. Res. 30, 199–204. doi: 10.2983/035.030.0201
Beamish R. J., Fournier D. A. (1981). A method for comparing the precision of a set of age determinations. Can. J. Fish. Aquat. Sci. 38, 982–983. doi: 10.1139/f81-132
Belcari P. (2017). “Illex coindettii,” in Sintesi delle conoscenze di biologia, ecologia e pesca delle specie ittiche dei mari italiani/Synthesis of the knowledge on biology, ecology and fishery of the halieutic resoursec of the Italian seas. Biol. Mar. Mediterr, vol. 24 . Eds. Sartor P., Mannini A., Carlucci R., Massaro E., Queirolo S., Sabatini A., et al (Genova, IT), 90–98.
Belcari P., Piatkowski U., Zumholz K., Sartor P., Lefkaditou E., Pierce G. J., et al. (2015). “Todaropsis eblanae (Ball 1841),” in Cephalopod biology and fisheries in Europe: II. Species Accounts. ICES Cooperative Research Report No, vol. 325 . Eds. Jereb P., Allcock A. L., Lefkaditou E., Piatkowski U., Hastie L. C., Pierce G. J. (Copenhagen, DK), 207–218.
Campana S. E. (2001). Accuracy, precision and quality control in age determination, including a review of the use and abuse of age validation methods. J. Fish Biol. 59, 197–242. doi: 10.1111/j.1095-8649.2001.tb00127.x
Cavanna P., Lanteri L., Beccornia E., Relini G. (2008). Growth of illex coindetii (Verany 1839) and todaropsis eblanae (Ball 1841) in the ligurian sea. Biol. Mar. Mediterr. 15, 320–321.
Ceriola L., Milone N. (2007). “Cephalopods age determination by statolith reading: A technical manual,” in Scientific Cooperation to Support Responsible Fisheries in the Adriatic Sea; GCP/RER/010/ITA/TD-22 (AdriaMed Technical Documents, Rome, Italy). No 22.
Chang W. Y. B. (1982). A statistical method for evaluating the reproducibility of age determination. Can. J. Fish. Aquat. Sci. 39, 1208–1210. doi: 10.1139/f82-158
Clarke M. R. (1986). A Handbook for the Identification of Cephalopod Beaks (Marine Biology Association U.K. Plymouth: Clarendon Press Oxford).
Clarke M. R. (1993). “Age determination and common sense - a free discussion on difficulties encountered by the author,” in Recent Advances in Cephalopod Fisheries Biology. Eds. Okutani T., O’Dor R. K., Kubodera T. (Tokai University Press, Tokyo), 670–678.
Cuccu D., Mereu M., Porcu C., Follesa M. C., Cau Al., Cau A. (2013). Development of sexual organs and fecundity in Octopus vulgaris Cuvier 1797 from the Sardinian waters (Mediterranean Sea). Mediterr. Mar. Sci. 14, 270–277. doi: 10.12681/mms.412
Dillane E., Galvin P., Coughlan J., Lipinski M., Cross T. F. (2005). Genetic variation in the lesser flying squid Todaropsis eblanae (Cephalopoda, Ommastrephidae) in east Atlantic and Mediterranean waters. Mar. Ecol. Prog. Ser. 292, 225–232. doi: 10.3354/meps292225
Fang Z., Li J., Thompson K., Hu F., Chen X., Liu B. L., et al. (2016). Age, growth, and population structure of the red flying squid (Ommastrephes bartramii) in the North Pacific Ocean, determined from beak microstructure. Fishery Bull. 114, 34–44. doi: 10.7755/FB.114.1.3
FAO. (2020). Global Capture Production (online query). Available online at: http://www.fao.org/fishery/statistics/global-capture-production/e (Accessed October 17, 2024).
Follesa M. C., Carbonara P. (Eds.) (2019). Atlas of the Maturity Stages of Mediterranean Fishery Resources; Studies and Reviews N. 99 Vol. 2019 (Rome, Italy: FAO), 268p.
Forsythe J. W., Van Heukelem W. F. (1987). “Growth,” in Cephalopod life cycles vol II. Ed. Boyle P. R. (Academic press University of Michigan), 135–156.
Fotiadis N., Lefkaditou E., Koitsoubas D. (2015). “The biology of the lesser flying squid Todaropsis eblanae in the Ionian Sea,” in 11th Panhellenic Symposium on Oceanography and Fisheries, Mytilene Greece. 1–4.
Gonzales A. F., Rasero M., Guerra A. (1994). Preliminary study of Illex coindetii and Todaropsis eblanae (Cephalopoda: Ommastrephidae) in the northern Spanish Atlantic waters. Fish. Res. 21, 115–126.
Gonzalez A. F., Castro B. G., Guerra A. (1996). Age and growth of the short-finned squid Illex coindetii in Galician waters (NW Spain) based on statolith analysis. ICES J. Mar. Sci. 53, 802–810. doi: 10.1006/jmsc.1996.0101
Hastie L. C., Pierce G. J., Wang J., Bruno I., Moreno A., Piatkowski U., et al. (2009). Cephalopods in the north-eastern Atlantic: Species, biogeography, ecology, exploitation and conservation. Oceanography Mar. Biology: Annu. Rev. 47, 111–190.
Hermosilla C. A., Rocha F., Fiorito G., Gonzalez A. F., Guerra A. (2010). Age validation in common octopus Octopus vulgaris using stylet increment analysis. ICES J. Mar. Sci. 67, 1458–1463. doi: 10.1093/icesjms/fsq054
Hernández-García V. (2002). Reproductive biology of Illex coindetii and Todaropsis eblanae (Cephalopoda: Ommastrephidae) off Northwest Africa (4°N, 35°N). Bull. Mar. Sci. 71, 347–366.
Hurley G. V., Odense P. H., O’Dor R. K., Dawe E. G. (1985). Strontium labelling for verifying daily growth increments in the statolith of the short-finned squid (Illex illecebrosus). Can. J. Fish. aquat. Sci. 42, 380–383. doi: 10.1139/f85-050
Lauria V., Garofalo G., Gristina M., Fiorentino F. (2016). Contrasting habitat selection amongst cephalopods in the Mediterranean Sea: When the environment makes the difference. Mar. Environ. Res. 119, 252–266. doi: 10.1016/j.marenvres.2016.06.011
Lishchenko F., Perales-Raya C., Barrett C., Oesterwind D., Power A. M., Larivain A., et al. (2021). A review of recent studies on the life history and ecology of European cephalopods with emphasis on commercial species with the greatest fishery and culture potential. Fish. Res. 236, 105847. doi: 10.1016/j.fishres.2020.105847
Liu B. L., Chen X. J., Chen Y., Hu G. Y., Yu W., et al. (2017). Periodic increments in the jumbo squid (Dosidicus gigas) beak: a potential tool for determining age and investigating regional difference in growth rates. Hydrobiologia 790, 83–92. doi: 10.1007/s10750-016-3020-3
Liu B. L., Chen X. J., Chen Y., Hu G. Y. (2015). Determination of squid age using upper beak rostrum sections: technique improvement and comparison with the statolith. Mar. Biol. 162, 1685–1693. doi: 10.1007/s00227-015-2702-0
Mangold K. (1983). “Octopus vulgaris,” in In Cephalopod Life Cycles. 1. Species Accounts. Ed. Boyle P. R. (Academic Press, London), 335–364.
Mangold-Wirz K. (1963). Biologie des Céphalopodes benthiques et nectoniques de la Mer Catalane. Vie Milieu Suppl. 13, 285.
Nakamura Y., Sakurai Y. (1991). Validation of daily growth increments in statoliths of Japanese common squid Todarodes pacificus. Nippon Suisan Gatiaishi 57, 2007–2011. doi: 10.2331/suisan.57.2007
Oesterwind D., Piatkowski U., Heinz B. (2014). On distribution, size and maturity of shortfin squids (Cephalopoda, Ommastrephidae) in the North Sea. Mar. Biol. Res. 11, 188–196. doi: 10.1080/17451000.2014.894246
Perales-Raya C., Almansa E., Bartolome A., Felipe B. C., Iglesias J., Sanchez F. J., et al. (2014). Age validation in Octopus vulgaris beaks across the full ontogenetic range: Beaks as recorders of life events in octopuses. J. Shellfish Res. 33, 481–493. doi: 10.2983/035.033.0217
Petroni G. (2012). Comparative morphology of early stages of ommastrephid squids from the Mediterranean Sea (Barcelona: Interuniversity master of aquaculture, Institut de Ciències del Mar, CSIC).
Pierce G., Robin J. P., Montero-Castaño C., Barrett C., Laptikhovsky V., González Á., et al. (2019). Assessment of cephalopods in European waters: state of the art and ways forward. Cephs & Chef ICES Annual Conference 2019. doi: 10.13140/RG.2.2.28386.96969
Rasero M. (1996). Short finned squid Todaropsis eblanae (Ball,1841) (Chephalopoda, Ommastephidae): life cycle and ecological aspects in the coastal waters of Galicia (NW Iberian Peninsula). University of Santiago de Compostela, Spain, Santiago de Compostela, Spain. PhD Dissertation.
Robin J. P., Denis V., Royer J., Challier L. (2002). Recruitment, growth and reproduction in Todaropsis eblanae (Ball 1841) in the area fished by French Atlantic trawlers. Bull. Mar. Sci. 71, 711–724.
Rodríguez-Domínguez A., Rosas C., Méndez-Loeza I., Markaida U. (2013). Validation of growth increments in stylets, beaks and lenses as ageing tools in Octopus maya. J. Exp. Mar. Biol. Ecol. 449, 194–199. doi: 10.1016/j.jembe.2013.10.001
Roper C. F. E., Nigmatullin C., Jereb P. (2010). “Family ommastrephidae,” in Cephalopods of the world. An annotated and illustrated catalogue of cephalopod species known to date. Myopsid and Oegopsid Squids. FAO Species catalogue for fishery purposes, vol. 2 . Eds. Jereb P., Roper C. F. E. (Roma, IT), 269–347.
Sabirov R. M., Golikov A. V., Nigmatullin C., Lubin P. A. (2012). Structure of the reproductive system and hectocotylus in males of lesser flying squid Todaropsis eblanae (Cephalopoda: Ommastrephidae). J. Natural History 46-issue 29-30, 1761–1778. doi: 10.1080/00222933.2012.700335
Sakai M., Brunetti N., Bower J., Elena B., Ichii T., Ivanovic, et al. (2007). Daily growth increments in upper beak of five ommastrephid paralarvae, Illex argentinus, Ommastrephes bartramii, Dosidicus gigas, Sthenoteuthis oualaniensis, Todarodes pacificus. Squids Resour. Res. Conf. 9, 1–7.
Spedicato M. T., Massutí E., Mérigot B., Tserpes G., Jadaud A., Relini G. (2020). The MEDITS trawl survey specifications in an ecosystem approach to fishery management. Scientia Marina 83, 9–20. doi: 10.3989/scimar.04915.11X
Xavier J. C., Golikov A. V., Queirós J. P., Perales-Raya C., Rosas-Luis R., Abreu J., et al. (2022). The significance of cephalopod beaks as a research tool: An update. Front. Physiol. 13, 1038064. doi: 10.3389/fphys.2022.1038064
Keywords: Todaropsis eblanae, age, growth, statoliths, beaks, eye lens, Mediterranean
Citation: Agus B, Battaglia P, Bellodi A, Cannas R, Coluccia E and Cuccu D (2025) Age and growth of Todaropsis eblanae (Ommastrephidae) through comparison of statoliths, beaks and eye lenses. Front. Mar. Sci. 12:1567441. doi: 10.3389/fmars.2025.1567441
Received: 27 January 2025; Accepted: 17 March 2025;
Published: 01 April 2025.
Edited by:
Simon Morley, British Antarctic Survey (BAS), United KingdomReviewed by:
Letizia Sion, University of Bari Aldo Moro, ItalyCopyright © 2025 Agus, Battaglia, Bellodi, Cannas, Coluccia and Cuccu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Danila Cuccu, Y3VjY3VAdW5pY2EuaXQ=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.