
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Mar. Sci. , 21 March 2025
Sec. Coastal Ocean Processes
Volume 12 - 2025 | https://doi.org/10.3389/fmars.2025.1564173
This article is part of the Research Topic Processes, Mechanisms and Solutions in Coastal Wetland to Adapt to Changing Environment View all 10 articles
Unraveling the relationships between archaea and factors influencing their diversity and distribution is a critical issue in marine ecosystems. Here, the archaeal diversity and community structure in 0 - 20 cm soils from freshwater influenced wetlands (FIW), flooding freshwater and underground seawater influenced wetlands (MIW) and seawater influenced wetlands (SIW) in the Yellow River Nature Reserve were examined utilizing high-throughput sequencing of 16S rRNA gene sequencing. Based on the comparison of the alpha diversity indices, the abundance and diversity of the archaeal community in wetlands with varying hydrologic conditions did not significantly change (p > 0.05), with Thaumarchaeota and Marine_Group_I as the predominant archaeal phylum and class in all the three sampled sites, respectively. Thaumarchaeota, Woesearchaeota and Euryarchaeota constituted more than 90% of the total soil archaeal community in all wetlands. However, beta diversity indices revealed that significantly different distribution patterns of archaea were found among the three wetlands (p < 0.05). And the archaeal community structure in different wetlands varied as the hydrologic conditions changed. Less discriminated archaeal taxa were found in MIW (1 taxon) than in FIW (24 taxa) and SIW (18 taxa). Furthermore, statistical analysis confirmed that the difference in soil salinity caused by different hydrologic conditions was the major driver of archaeal community structure. Overall, this study highlights the role of hydrologic conditions in structuring the soil archaeal community in coastal wetlands.
As an abundant, ubiquitous and diverse community, archaea on the earth widely inhabit not only the terrestrial and marine ecosystems, but also the gut systems of insects or termites (Pfeifer et al., 2021). The archaeal lineages featured with thermophility and/or halophilism can survive in extreme environments with harsh salinity, acidity, alkalinity, and temperature conditions (Naitam et al., 2023; Saavedra-Bouza et al., 2023). Because they can withstand harsh conditions, archaea make up the third domain of life, different from bacteria and eukaryotes (Meng et al., 2022). Archaea are ecologically important and essentially involve in various biogeochemical processes, playing a vital role in global elements (i.e. carbon, nitrogen, sulfur, iron) cycles and energy flow (Baker et al., 2020). For instance, archaea in the phylum Thaumarchaeota are representatives of ammonium oxidizers, while methanogenic archaea (methanogens) are renowned for producing methane after energy metabolism and their influence on global climate changes. Both Thaumarchaeota and methanogens constitute important links of food webs in anaerobic and marine environments (Pfeifer et al., 2021). Besides, archaea hold great promise for a wide range of biotechnological uses, since thy can produce active enzymes those can that withstand challenging industrial settings and additional bioproducts such as lipids (Saavedra-Bouza et al., 2023). Moreover, archaea are also reported to potentially degrade hydrocarbons and dissolved proteins, remediate metals, perform dehalogenation reaction, drain acid mine (Lazar et al., 2016; Krzmarzick et al., 2018). And some archaea can be used as bioremediators for appropriate waste treatment, as well as a feed supplement for growth promotion and disease control (Jifiriya et al., 2023). Thus, the community composition of archaea profoundly affects biogeochemical processes. Moreover, information pertaining to the composition of the archaeal community structure and its dynamic diversity holds significant importance. This information not only contributes substantially to the in - depth understanding of the microbial ecology of wetlands and the refinement of global climate change modeling, but also serves as an indicator of environmental stress and ecosystem changes occurring within wetland soils (Chambers et al., 2016; Cheung et al., 2018). Considering the role of wetlands in the global carbon cycling and the microbial control of wetlands’ functions and services (Wu et al., 2021), understanding both the community structure and diversity of archaea and associated influencing factors in wetlands is imperative.
As an essential element of the sedimentary ecosystems, archaea have a high diverse population in estuaries (Zou et al., 2020a). The ecological functions of estuaries largely depend on the composition of microbes (Xie et al., 2014). And the biogeochemical processes in marine sediments are profoundly affected by the structure and metabolic activities of microbes (Wang et al., 2020a). For example, wetland microbial communities act as key determinants in controlling both the production and release of greenhouse gases (Maietta et al., 2020). The composition and dynamics of microbial populations also serve as critical biomarkers for assessing ecosystem health in estuarine environments (Wang et al., 2025). Thus the investigation of potential drivers of archaeal community is necessary to understand the possible changes in ecological services provided by estuaries. Besides, estuaries connect the land and ocean, exhibiting distinct gradient changes of soil physiochemical properties due to the variations in freshwater inputs, tidal heights and geomorphologic changes along the mouth of the estuaries (Webster et al., 2015; Zou et al., 2020b). The hydrology in estuarine wetlands not only determines basic structures and ecological functions, but also controls physicochemical conditions such as soil salinity due to interactions among tidal flooding, precipitation induced runoff, groundwater flow, and evapotranspiration processes (Zedler, 2000; White and Madsen, 2016; Zhao et al., 2023). The variability of hydrologic conditions affects the community structure of soil and methane cycling microbes and the gene abundances of both ammonia-oxidizing archaea and ammonia-oxidizing bacteria (Liu et al., 2021; Maietta et al., 2020). However, the impact of hydrologic variability on microbial community composition was differently presented by several researches (Peralta et al., 2014; Maietta et al., 2020). Moreover, how archaeal communities respond to salinization caused by salt water intrusion, especially how the shifts of methanogenic archaea affect the global greenhouse gas budget, still exists as uncertainty (Wen et al., 2017). Despite the fact that salinity has been documented as a primary determinant of the archaeal community composition, there remains a lack of consensus regarding the impacts of salinity on archaea. Some researchers found that rising salinity could increase the abundance and diversity of archaeal community (Liu et al., 2016; Wei et al., 2020), while Webster et al. (2015) reported that the diversity of archaea decreased as salinity increased. Furthermore, Chambers et al. (2016) found that the increased inundation frequency played a more decisive role than salinity in determining microbial community structure of brackish soil more strongly. The sea level rise and the intensified global climate warming could introduce seawater high up to the estuaries, which inevitably causes changes of wetland hydrology (i.e. water table and flooding duration) and soil salinity (Chambers et al., 2016). Thus, the response of estuarine archaea to various hydrologic conditions needs further investigation.
Here, to clarify variations in the diversity and community structure of soil archaea in wetlands under various hydrologic conditions, we collected soil samples of 0 - 20 cm depth in wetlands affected by freshwater influenced wetlands (FIW), flooding freshwater and underground seawater influenced wetlands (MIW) and seawater influenced wetlands (SIW). The inundation frequency in the three sampling sites ranked in the order SIW > FIW > MIW. The study aims at: (1) identifying the distribution pattern of soil archaea in wetlands under various hydrologic conditions and (2) clarifying the effects of hydrologic conditions on the archaeal community composition and diversity of archaea in coastal wetlands. Overall, this study will provide information for the management of coastal wetlands.
The study area is located in the Yellow River Nature Reserve, and the selected sampling sites (FIW, MIW and SIW), soil collection (0 - 20 cm depth) for the measurements of soil physicochemical properties and the corresponding analysis have been previous described in the previous researches (Zhao et al., 2020a, b). Soil EC, Na+, K+, Mg2+, Cl− and SO42− increased and soil moisture decreased as the influence of tides intensified and reached the highest level in SIW along the sampling belt (FIW-MIW-SIW) (Zhao et al., 2020b). Thus, FIW, MIW and SIW were also defined as low salinity wetlands, medium salinity wetlands and high salinity wetlands, respectively. Their dominant plants are Phragmites australis, P. australis and Suaeda salsa. And the comparison of other soil properties among three wetlands can also be found in the previous research (Zhao et al., 2020b). Soil samples designated for 16S rRNA sequencing were initially positioned in an ice - cooled box. Subsequently, they were dispatched to Majorbio Bio - pharm Technology Co., Ltd. (Shanghai, China) to undergo further sequencing procedures. In addition, Majorbio was also responsible for conducting the data processing.
Genomic DNA was extracted from about 0.5 g of each sample’s soil using the soil DNA kit (Omega Bio-tek, Norcross, GA, U.S.). PCR amplification (ABI GeneAmp® 9700) of the V4-V5 region of the 16S rRNA gene was conducted using TransGen AP221-02 (TransStart Fastpfu DNA Polymerase) to analyze archeal community. The forward primer 515F (5′-GTGCCAGCMGCCGCGG-3′) and the reverse primer 915R (5′-GTGCTCCCCCGCCAATTCCT-3′) was used with the following protocol: 95°C for 2 min, 25 cycles of denaturation (95°C; 30 s), annealing (55°C; 30 s), and extension (72°C; 60 s) and a final elongation (72°C; 10 min). The amplified DNA was quantified with a TBS-380 fluorometer (Promega Corporation, CA, USA) after purification using the AxyPrep DNA Gel Extraction Kit (Axygen Biosciences, Union City, CA, USA). Then barcoded pyrosequencing of the 16S rRNA gene was performed on an Illumina MiSeq PE250 system. The sequencing data was deposited in the NCBI database under the accession number SRP099022.
The primers were excluded after alignment and sequences with a length shorter than 200 bp or low quality were removed. Valid sequences were defined as seeds with complete sequence barcodes, and were further classified using Mothur software (http://www.mothur.org) and aligned via the Silva bacterial reference database (http://www.arb-silva.de). The number of operational taxonomic units (OTUs) in all samples was defined at Usearch (version 7.1, http://drive5.com/uparse/) with sequence similarity of 97%. Species classification information represented by each OTU was obtained by RDP Classifier Bayes algorithm (version 2.2 http://sourceforge.net/projects/rdp-classifier/) on the Qiime platform (version 2.2 http://sourceforge.net/projects/rdp-classifier/). The alpha diversity indices determined in this study included the Coverage index, the observed species (Sobs), the richness indices (the abundance-based coverage estimator (ACE) and bias-corrected Chao1 richness estimator), the Shannon diversity index, the community evenness index (Shannoneveness) and Faith’s phylogenetic diversity (PD) index. Within Mothur, they were calculated to examine the sequencing depth, richness and diversity of the archaeal community, respectively. The beta diversity indices, such as principal coordinate analysis (PCoA) and Non-MetricMulti-Dimensional Scaling (NMDS), were determined to evaluate the difference in distribution pattern of archaeal community among the three wetlands. PCoA identifies potential principal components affecting archaeal community through reducing dimensions and was drawn based on the selected distance matrix. NMDS spatially exhibits the dissimilarities among different species and are often used when the accurate similarities or dissimilarities among research objects are hardly obtained.
One-way ANOVA analysis was used to identify the significant differences in archeal community richness and diversity, and in soil properties among three wetlands. Differences were considered to be significant if p < 0.05. Both one-way ANOVA and Pearson correlation analysis between soil properties and the alpha-diversity indices were performed using SPSS v22.0 (SPSS Inc.). The analysis of similarity (ANOSIM) and Adonis test were conducted based on the Bray-Curtis dissimilarity matrix to test significant differences in archaeal community among three wetlands. Additionally, Spearman correlation analysis and Mantel test between archaeal community and environmental factors were performed to identify environmental variables those influenced archaeal community composition. The ANOSIM, Adonis test, Spearman correlation analysis and Mantel test were conducted on the cloud platform of Majorbio Group.
The number of OTUs generated from high-quality sequences was the least in the MIW (1100), and higher in the FIW (2014) and SIW (1902) (Supplementary Figure S1). The number of specific OTUs in FIW, MIW and SIW were 1498, 313 and 1120, respectively. And the shared OTUs’ number of three wetlands was 169. The comparison of alpha diversity indices of archaea is shown in Table 1. The values of Coverage index for soil archaeal community were higher than 99% in all sampling sites, implying the sequencing depth could reflect the archaeal community in the three wetlands. More than 400 archaeal species were found in each of the three wetlands, but the number of archaeal species did no significantly differ among the three wetlands (p > 0.05), as revealed by the Sobs vales. Additionally, the richness indices (ACE and Chao1), the diversity index (Shannon), the Shannoneveness index and PD index showed no significant differences among FIW, MIW and SIW (p > 0.05). In sum, no significant difference in alpha diversity was observed among three types of wetlands (p > 0.05). Statistically, the values of Sobs, ACE, Chao1, Shannon and PD indices were generally lower in MIW than in FIW and SIW.
PCoA and NMDS based on Bray-Curtis distance matrices were performed to identify the relationships among the samples with respect to archaeal community composition, as shown in Figures 1a, b, respectively. In the PCoA and NMDS plots, samples in the FIW, MIW and SIW can be clearly separated, indicating that the distribution patterns of archaea community in the three wetlands were significantly different (p < 0.05). The ANOSIM and Adonis test further verified that archaeal community among FIW, MIW and SIW was significantly dissimilar (p < 0.01) (Supplementary Tables S1, S2).
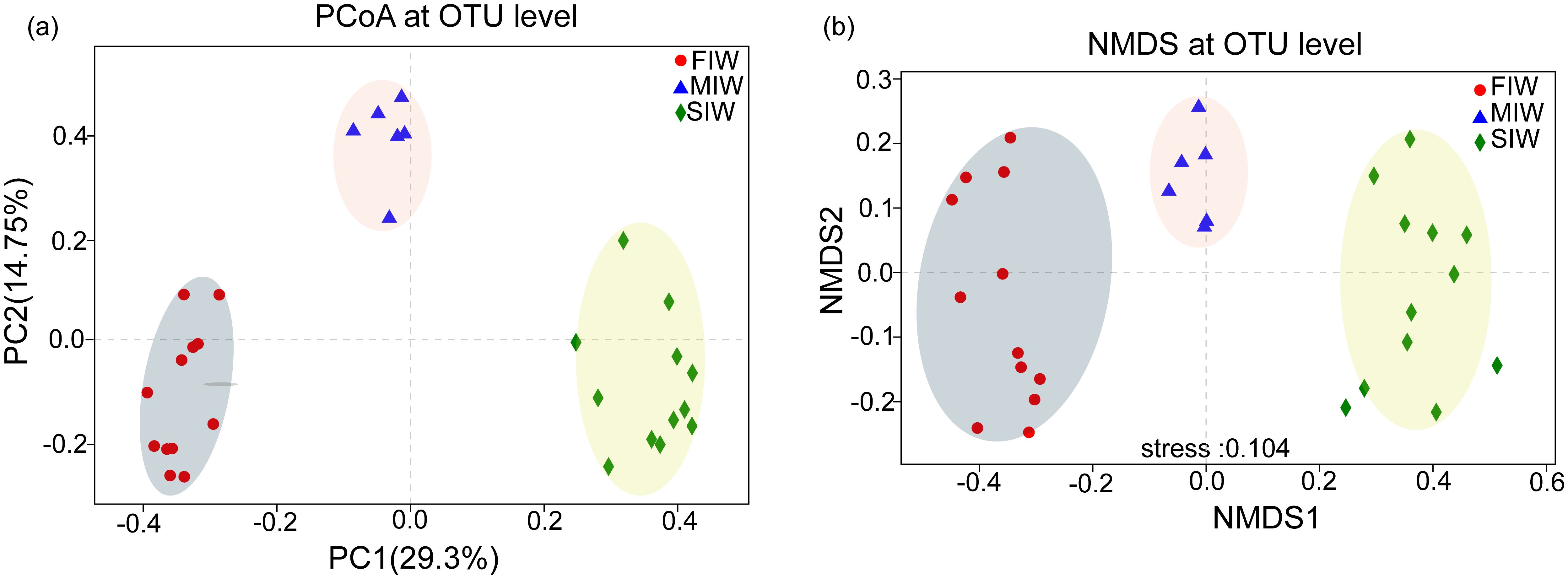
Figure 1. PCoA (a) and NMDS (b) plots of the archaeal communities in wetlands under various hydrologic conditions.
Community structure of archaea at the phylum and class level in the three wetlands is shown in the Circos plots of Figures 2a, b, respectively. The left half circle (small semicircle) indicates the taxonomic composition of soil fungal communities of each group. The outer green, blue and red ribbon represents SIW, MIW and FIW, respectively. Different colors of the inner ribbon characterize different archaeal species at phylum or class level, and the length of the inner ribbon reflects the relative abundance of corresponding archaeal phyla or class. The right half circle (large semicircle) indicates the proportions of the archaea at phylum or class level in different wetlands. Specifically, the outer ribbon shows archaea taxa and the different colors of the inner ribbon represent different wetlands. And length of the inner colored ribbon represents the relative abundance of the certain archea in different wetlands. Symbols p1 - p7 and c1 - c13 represents archeal phyla and class as below: p1: Thaumarchaeota, p2: Woesearchaeota, p3: Euryarchaeota, p4: Bathyarchaeota, p5: others, p6: Aenigmaarchaeota, p7: Miscellaneous_Euryarchaeotic_Group_MEG. C1: Marine_Group_I, c2: norank_p:Woesearchaeota, c3: Thermoplasmata, c4: Halobacteria, c5: Soil_Crenarchaeotic_Group_SCG_incetae_sedis, c6: unclutured_p_Thaumarchaeota, c7: norank_p_Bathyarchaeota, c8: others, c9: Deep_Sea_Euryarchaeotic_Group_DSEG_norank, c10: Methanomicrobia, c11: South_African_Gold_Mine_Gp_1_SAGMCG_1, c12: norank_p_Miscellaneous_Euryarchaeotic_Group_MEG_, c13: Group_C3. The sequencing of 16S rRNA gene amplicons from the soils in the FIW averagely generated 56732 ± 9832 high-quality reads per sample (Supplementary Table S3). These sequences were categorized into 6 archaeal phyla. The archaeal sequences in the FIW were mainly composed of Thaumarchaeota (58.83%), Woesearchaeota (19.76%), Euryarchaeota (11.54%), Bathyarchaeota (5.72%), Aenigmaarchaeota (0.55%) and Miscellaneous_Euryarchaeotic_Group_MEG (1.21%) (Figure 2a). At the class level, a total of 6 archaeal classes were recognized (Figure 3b). Marine_Group_I represented 38.63% of the total archaeal community and was the most abundant archaeal class in the FIW, followed by Soil_Crenarchaeotic_Group_SCG_incetae_sedis (11.80%). The rest identified archaeal classes were Thermoplasmata (8.65%), Methanomicrobia (2.50%), South_African_Gold_Mine_Gp_1_SAGMCG_1 (2.27%) and Group_C3 (1.07%) (Figure 2b).

Figure 2. Circos plots for the community composition at the phylum (a) and class (b) levels in wetlands under various hydrologic conditions.
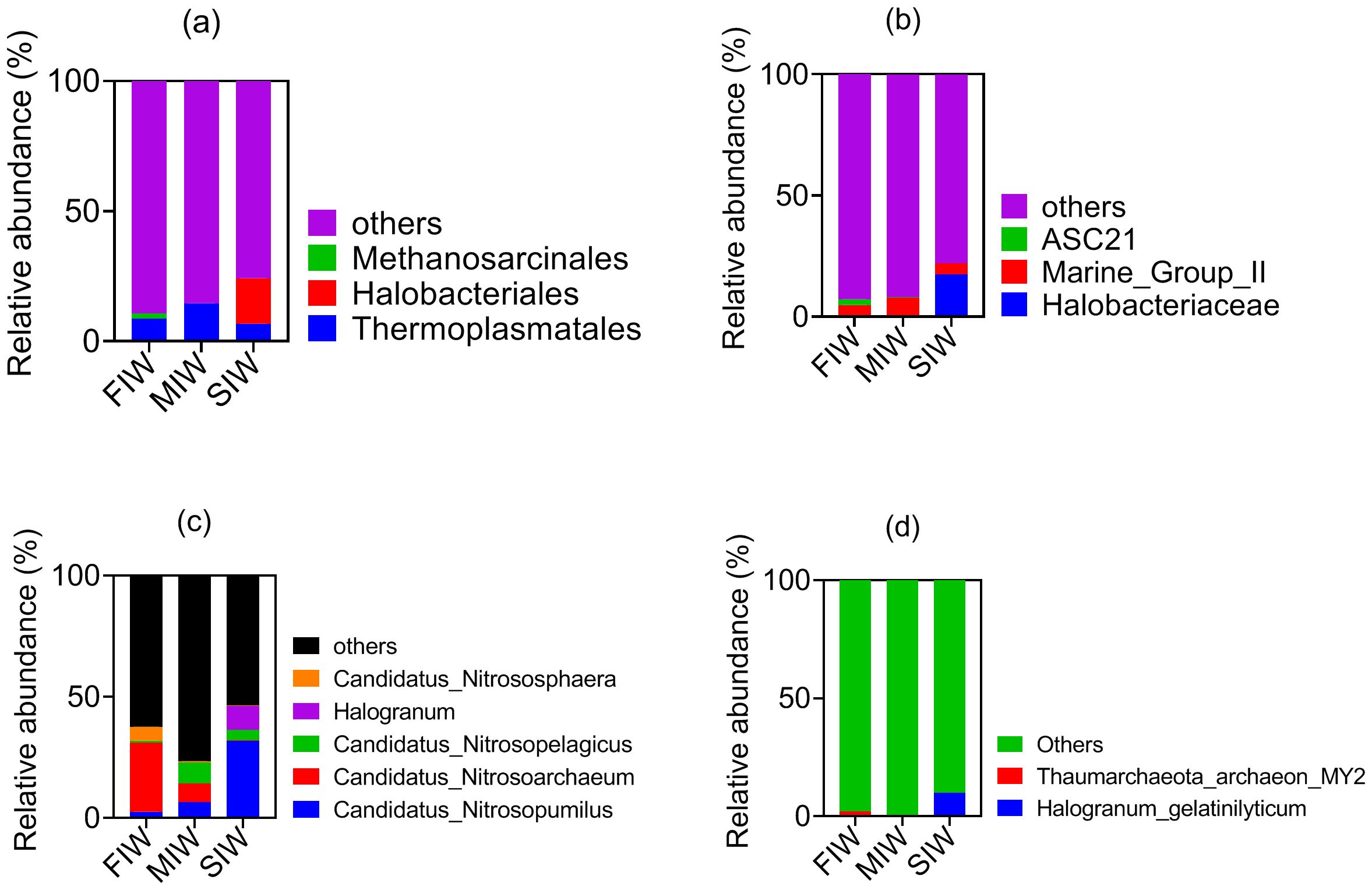
Figure 3. Community structure of archaea at the order (a), family (b), genus (c), and species (d) levels in wetlands under various hydrologic conditions.
An average of 53683 ± 11291 sequences per sample was yielded through 16S rRNA sequencing in the MIW (Supplementary Table S3). Among the 6 archaeal phyla, the relative abundance of Thaumarchaeota ranked the highest with the percentage of 62.19%, and the relative abundance of Woesearchaeota (20.62%) and Euryarchaeota (14.74%) followed by as the second and the third, respectively. However, the relative abundance of Bathyarchaeota (0.10%), Aenigmaarchaeota (1.93%) and Miscellaneous_Euryarchaeotic_Group_MEG (0.01%) was quite lower (Figure 2a). At the class level, archaeal community in the MIW was mostly represented by Marine_Group_I (56.49%), Thermoplasmata (14.61%) and Soil_Crenarchaeotic_Group_SCG_incetae_sedis (3.54%) (Figure 2b).
Sequencing of the soils in the SIW averagely yielded 57307 ± 9826 sequences per sample (Supplementary Table S3). Similar to FIW and MIW, Thaumarchaeota (42.92%), Woesearchaeota (30.64%) and Euryarchaeota (24.30%) occupied much higher proportions than Bathyarchaeota (0.35%), Aenigmaarchaeota (0.58%) and Miscellaneous_Euryarchaeotic_Group_MEG (0.04%) (Figure 2a). At the class level, Marine_Group_I (41.09%), Thermoplasmata (6.83%), Halobacteria (17.35%) and Soil_Crenarchaeotic_Group_SCG_incetae_sedis (1.79%) largely represented the archaeal community in the SIW (Figure 2b).
Generally, Thaumarchaeota, Woesearchaeota and Euryarchaeota represented higher than 90% of the total archaeal community in the soils of all three wetlands. An obviously increasing trend was observed in the relative abundance of Woesearchaeota and Euryarchaeota as soil salinity increased, while the relative abundance of Bathyarchaeota and Miscellaneous_Euryarchaeotic_Group_MEG decreased with increasing soil salinity. Thaumarchaeota and Aenigmaarchaeota are more abundant in MIW than in FIW and SIW. Marine_Group_I absolutely dominated the archaeal community in all three wetlands, showing a general increasing trend as salinity increased. The relative abundance of Thermoplasmata is highest in MIW than in FIW and SIW, showing an initial increasing then decreasing tend. The relative abundance of Halobacteria increased with increasing salinity, while Methanomicrobia, South_African_Gold_Mine_Gp_1_SAGMCG_1, Group_C3 and Soil_Crenarchaeotic_Group_SCG_incetae_sedis showed an absolutely decreasing tend as salinity increased. As for Thermoplasmata, the peak value of its relative abundance was observed in MIW, showing an initial increasing then decreasing tend.
The community composition of archaea at the order, family, genus and species level are shown in Figures 3a–d, respectively. As shown in Figure 3, three order (Thermoplasmatales, Halobacteriales and Methanosarcinales), three family (Halobacteriaceae, Marine_Group_II and ASC21), five genus (Candidatus_Nitrosopumilus, Candidatus_Nitrosoarchaeum, Candidatus_Nitrosopelagicus, Halogranum and Candidatus_Nitrososphaera), and two species (Halogranum_gelatinilyticum and Thaumarchaeota_archaeon_MY2) were recognized. All the recognized archaeal taxa only occupied a small proportion. Generally, the relative abundance of Halobacteriales, Halobacteriaceae, Candidatus_Nitrosopumilus, Halogranum and Halogranum_gelatinilyticum increased as salinity increased, while the relative abundance of Methanosarcinales, ASC21, Candidatus_Nitrosoarchaeum, Candidatus_Nitrososphaera and Thaumarchaeota_archaeon_MY2 decreased with increasing salinity. As for Thermoplasmatales, Marine_Group_II and Candidatus_Nitrosopelagicus, the peak value of their relative abundance occurred in MIW.
Discriminated archaeal taxa significantly affected the difference between groups are revealed by Linear Discriminant Analysis (LDA) effect size (LEfSe) in Figure 4 (norank, unclassified archaeal taxa and those with relative abundance < 1% were not shown in Figure 4). The all-against-all strategy was adopted in the recognition of discriminated archaeal taxa, which is more strict than one-against-all strategy. Totally, FIW, MIW and SIW had 14, 1 and 18 significantly enriched archaeal taxa, respectively (p < 0.05). Notably, Thaumarchaeota was the only taxa that had significantly higher relative abundance of in the soils of MIW than in FIW and SIW (p < 0.05). At the phylum level, Miscellaneous_Euryarchaeotic_Group_MEG_, Bathyarchaeota, Marine_Benthic_Group_E, Aenigmarchaeota_Incertae_Sedis, Methanomicrobia and the order (Methanomicrobiales), the families (Methanoregulaceae, Methanocorpusculaceae, GOM_Arc_I, Methanosarcinaceae, and Methanosaetaceae), the genra (Methanosaeta, Methanosarcina, Candidatus_Methanoperedens, Methanoregula, Methanobacterium, Methanocorpusculum, Methanosphaerula) belonging to this class, Soil_Crenarchaeotic_Group_SCG_, South_African_Gold_Mine_Gp_1_SAGMCG_1_, Terrestrial_Miscellaneous_Gp_TMEG_, ASC21 and CCA47 were significantly enriched in FIW (p < 0.05), while Halobacteria, the order (Halobacteriales), the family (Halobacteriaceae) and the genra (Halogranum, Halorussus, Natronomonas, Halomicroarcula, Halorubellus, Halomarina, Haloarcula, Halorientalis, Haloplanus, Halarchaeum, Haloferax, Salinigranum, Haloarchaeobius, Halolamina, Halogeometricum) belonging to this class were significantly enriched in the soils of SIW (p < 0.05).
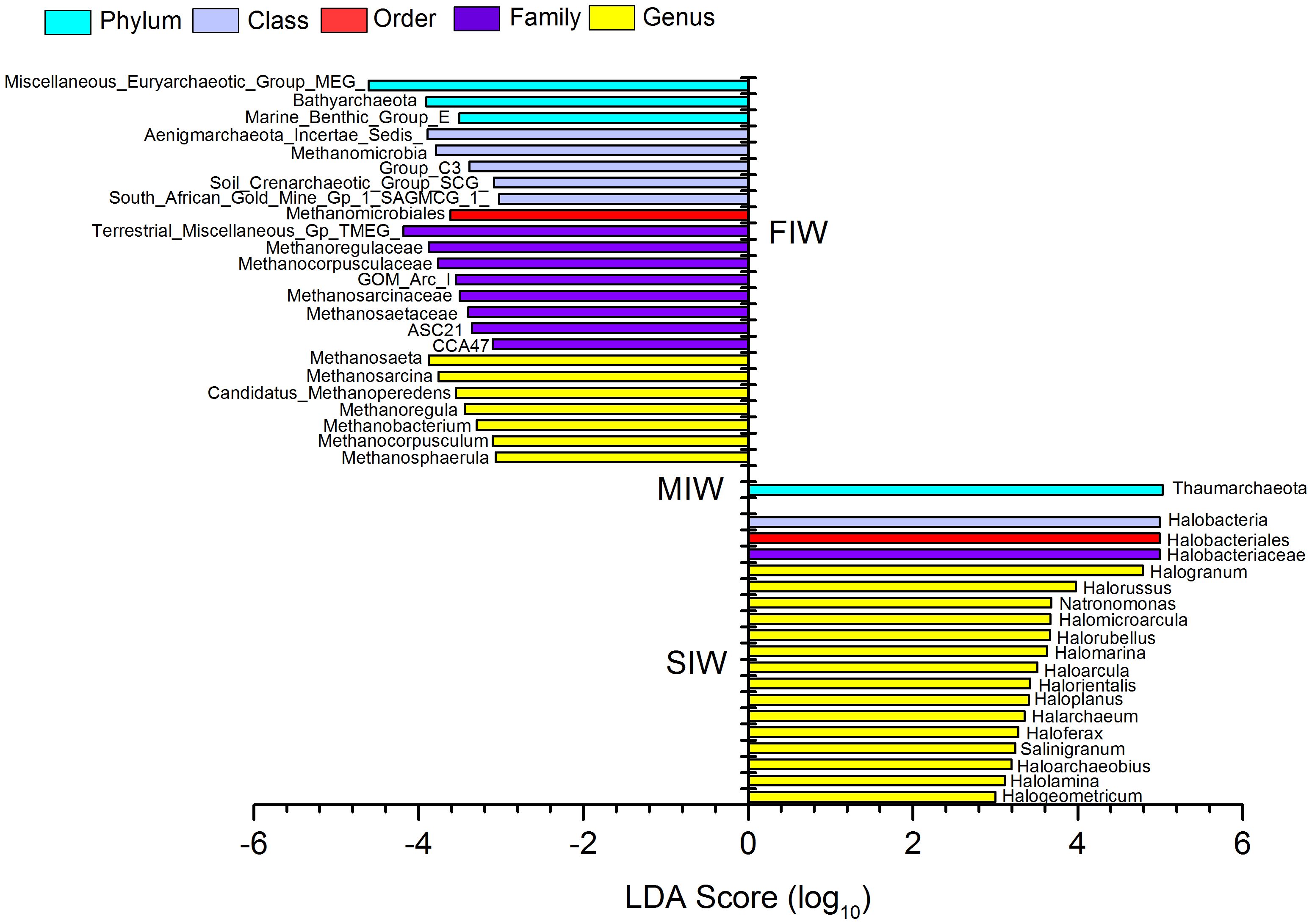
Figure 4. Archaeal taxa with Linear discriminant analysis (LDA) score greater than 3 present in wetlands under various hydrologic conditions.
The mantel test between soil archaea and soil physiochemical properties at the OTU, phylum and class level is shown in Table 2. At the OTU level, DOC, pH and BD significantly influences soil archaeal community structure (p < 0.05). DOC, TN, BD, clay and Ca2+ positively affected the relative abundance of archaeal phyla (p < 0.05), but silt negatively affected the archaeal community structure at the phylum level (p < 0.05). DOC, TN and pH showed a positively significant relationship with archaeal community structure at the class level (p < 0.05), and SOC and silt showed a negatively significant relationship with archaeal community structure (p < 0.05).
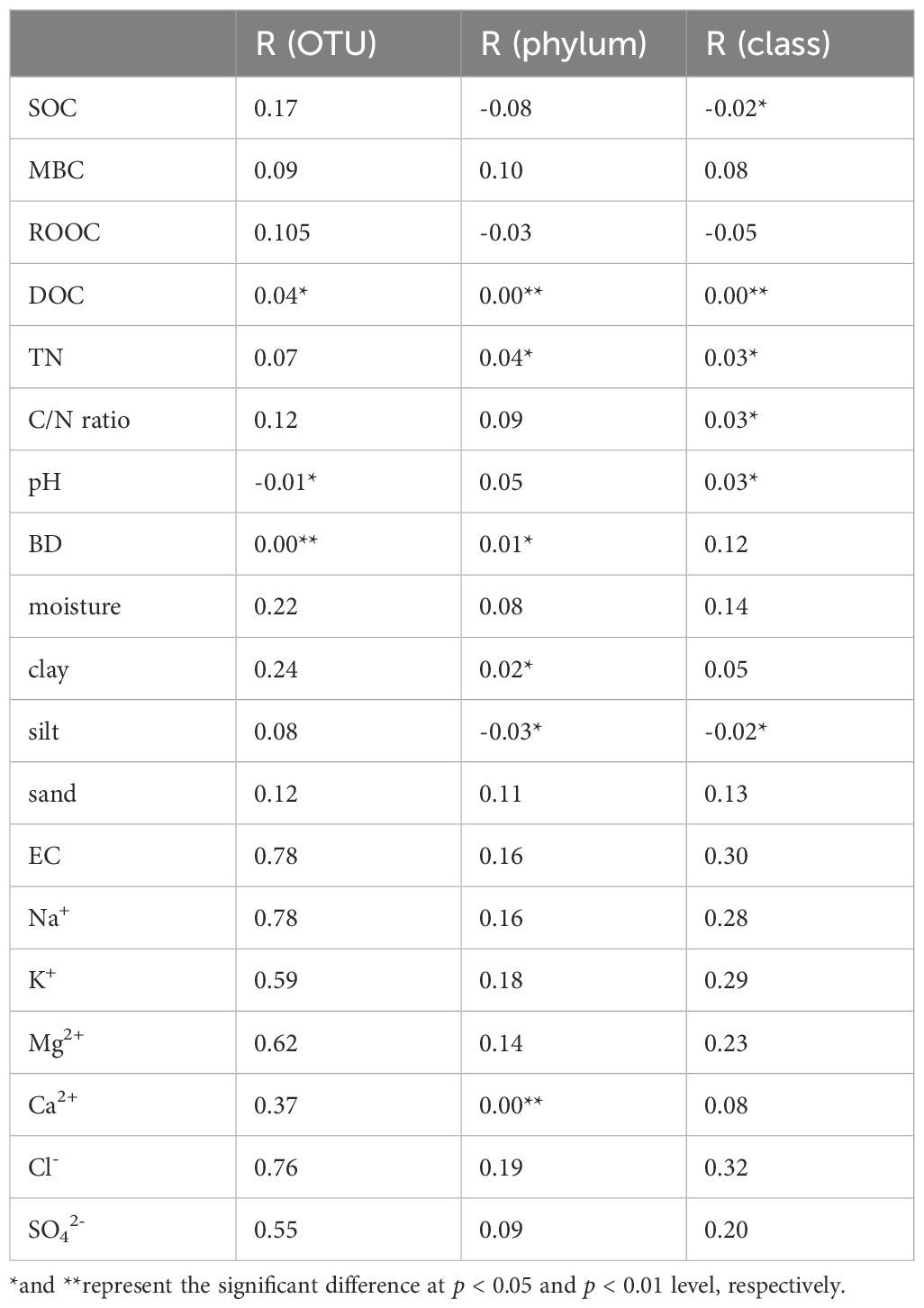
Table 2. The mantel test between archaeal community and soil properties (at the OTU, phylum and class level).
As shown in Figure 5, soil properties except for pH and BD affected the archaeal community structure at both phylum (Figure 5a) and class (Figure 5b) levels. Specifically, soil salinity parameters including EC, Na+, K+, Mg2+, Ca2+, Cl- and SO42- significantly and positively influenced the relative abundance of Diapherotrites, Euryarchaeota, Nanohaloarchaeota, Woesearchaeota, Deep_Sea_Euryarchaeotic_Group_DSEG_, Diapherotrites_Incertae_Sedis_ and Halobacteria (p < 0.05), but negatively and significantly influenced the relative abundance of Altiarchaeales, Bathyarchaeota, Candidate_division_YNPFFA, Hadesarchaea, Lokiarchaeota, Marine_Benthic_Group_E, Miscellaneous_Euryarchaeotic_Group_MEG_, Parvarchaeota, Group_C3, Methanobacteria, Methanomicrobia, Soil_Crenarchaeotic_Group_SCG_, South_African_Gold_Mine_Gp_1_SAGMCG-1_ and WCHA1-57 (p < 0.05). Moreover, Supplementary Table S4 shows the correlation relationships between alpha diversity indices and soil properties, which indicates that the alpha diversity indices of archaeal community were not significantly affected by soil properties (p > 0.05).
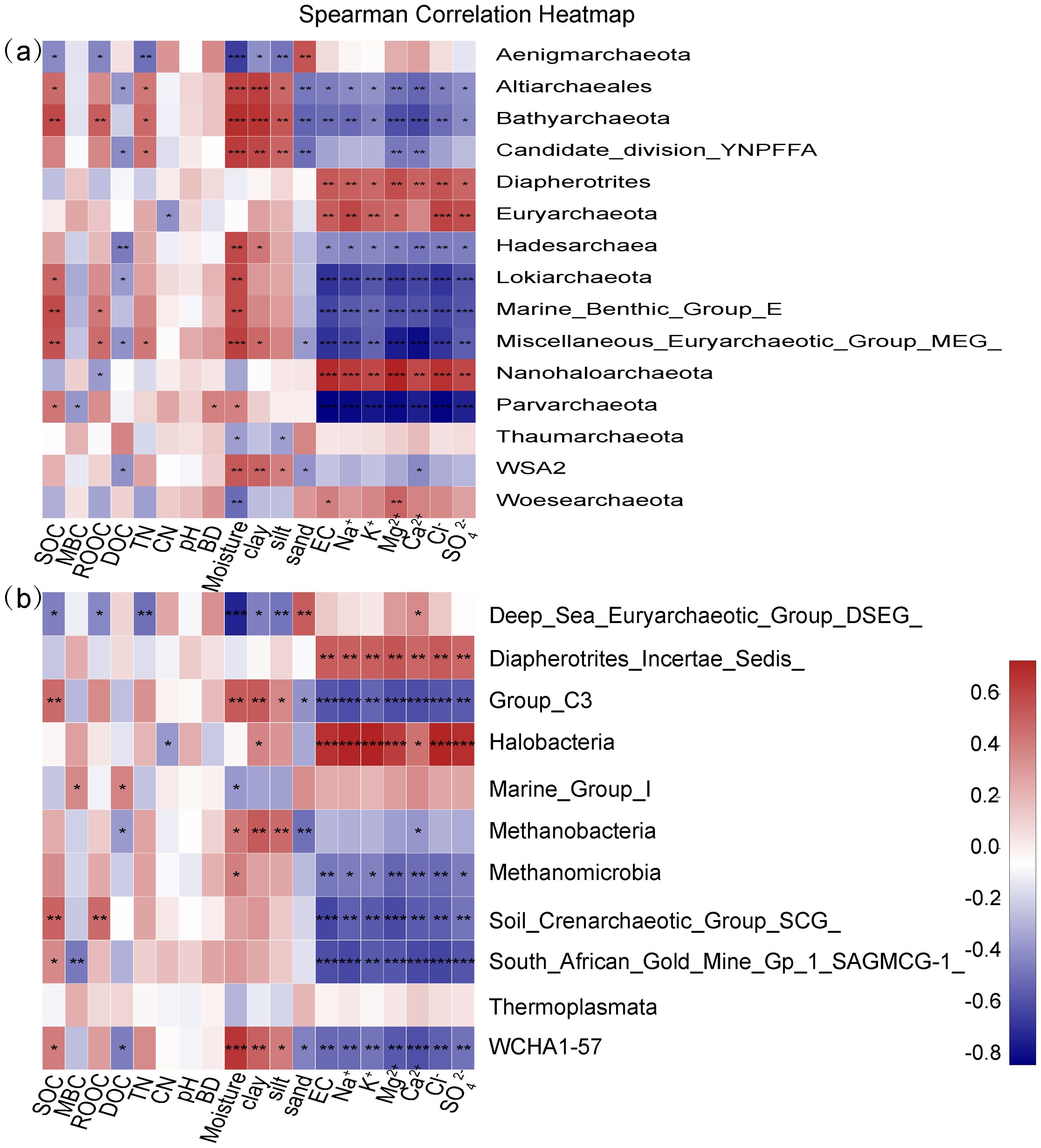
Figure 5. Heatmap for Spearman correlation coefficients between archaeal phyla (a), class (b) and soil properties.
The redox gradients caused by hydrologic variations result in the difference in the abundance and composition of archaeal community (Maietta et al., 2020; Wang et al., 2020a). It has been reported that the members of Marine Group I were more enriched in aerobic environments, while Woesearchaeota and Bathyarchaeota were generally found in the oxygen depleted zones (Wang et al., 2020a). The different hydrologic conditions created the significant difference in some soil properties besides soil salinity in 0 - 20 cm soils of the three wetlands, as presented in our previous work (Zhao et al., 2020a; Zhao et al., 2020b). Specifically, soil SOC was significantly higher in FIW than in MIW, while pH showed a significantly higher value in SIW than in MIW. Soil silt content was significantly lower in MIW than in FIW and SIW, while soil sand content showed an opposite trend. Previous researches showed that salinity differently affected the diversity of archaea in various environments. For example, Webster et al. (2015) found that the abundance of archaeal community was higher in brackish sediments with low salinity than in the high-salinity marine sediments. Liu et al. (2016) found that the archaeal diversity increased with salinity in Tibetan Plateau lake sediments. Wei et al. (2020) reported that archaeal diversity increased with rising salinity in soils covered with wheat, cotton, paddy, wild plant Suaeda salsa and barren fields without any plants. In this study, both the abundance and diversity of archaea showed no significant differences among soils with low, medium and high salinity (p > 0.05) and also were not significantly affected by soil properties (p > 0.05). Similar results were found by Zou et al. (2020b) in surface sediments of the Pearl River, which showed that no significant differences of archeal diversity index were found in sample groups with different salinity, and no strong correlations between the 16S rRNA gene abundance of archaea and the parameters investigated were observed.
Maietta et al. (2020) presented that soil microbial composition in inundated or saturated soils (more than 50% of the year) were quitely different from soils that were rarely flooded, which is consistent with this current study. Chen et al. (2022) found that tide-driven hydrodynamic disturbance control the composition of microbial community. It was concluded that the different hydrologic conditions in three wetlands resulted in the varying archaeal community structure in this study. Furthermore, the variations in plant species caused by different hydrologic conditions can lead to differences in the archaeal community (Gao et al., 2024). Except for the direct influence of hydrologic conditions on soil archeal community, the indirect effects of hydrologic conditions which are mainly fulfilled though influencing soil properties should also be highlighted. Numerous researches have reported that soil physiochemical properties largely determined the community structure of archaea, especially soil salinity, which played a dominant role in shaping the composition of archaea (Xie et al., 2014; Zhang et al., 2015; Liu et al., 2017; Wei et al., 2020; Zou et al., 2020a, b). Although no apparent effects of salinity on the abundance and diversity of archaea, the community structure of archaea was significantly affected by salinity as revealed by spearman correlation analysis (Figure 5). LEfSe analysis showed that archaea distributed in the phyla including Aenigmarchaeota, Bathyarchaeota, Euryarchaeota, Halobacterota, Marine_Benthic_Group_E, Miscellaneous_Euryarchaeotic_Group_MEG_ and Thaumarchaeota showed significant differences among FIW, MIW and SIW (p < 0.05).
Bathyarchaeota essentially involve in the global carbon cycle due to their utilization of various organic substrates, capacity of methane metabolism and carbon fixation (Zou et al., 2020a; Liang et al., 2023). Additionally, some members of Bathyarchaeota were reported to potentially participate in nitrogen and sulfur metabolism (Chen et al., 2020).The relative abundance of Bathyarchaeota was positively and significantly correlated with SOC (Figure 5), which is also reported by Ma et al. (2016) and Zou et al. (2020a). Besides, consistent with other studies (Chen et al., 2020; Zou et al., 2020a), salinity posed significantly negative effects on the relative abundance of Bathyarchaeota in the current study (p < 0.05, Figure 5). Soil texture has been proved to affect the distribution of microbes (Maier and Pepper, 2009), and modulate the influence of response biochemical quality of organic inputs on ammonia-oxidizing bacterial and archaeal communities (Muema et al., 2016). The relative abundance of Bathyarchaeota was significantly and positively related with clay and silt contents and soil moisture (WC) (p < 0.01), but showed significantly negative relationship with sand content (p < 0.01). Thus, the significantly higher SOC content and silt content, and lower salinity and sand content in soils of FIW than in MIW and SIW (p < 0.05, Zhao et al., 2020b) contributed to the significantly higher relative abundance of Bathyarchaeota in FIW. Similar to Bathyarchaeota, members of Miscellaneous_Euryarchaeotic_Group_MEG_ and Marine Benthic_Group_E (recently named Candidatus Hydrothermarchaeota) were more enriched in soils of FIW due to the positive effects of higher SOC content and lower salinity in FIW than in MIW and SIW (p < 0.05, Figure 5).
Euryarchaeota not only essentially involve in the global carbon cycling through methane production, anaerobic methane oxidation, the transformation of hydrocarbons, but also involve in the sulfur, nitrogen and iron cycling (Baker et al., 2020). 17 species of Euryarchaeota identified by LEfSe analysis were more enriched in soils of FIW than in soils of MIW and SIW in this study, distributing in three classes: Thermoplasmata (3 families), Methanomicrobia (14 taxa) and Methanobacteria (1 family). Thermoplasmata, Methanomicrobia and Methanobacteria comprise methanogens and essentially involve in methane production. The relative abundance of Euryarchaeota, Methanomicrobia and Methanobacteria were significantly and negatively affected by salinity (p < 0.05, Figure 5), thereby showing significantly higher values in FIW than in MIW and SIW (p < 0.05).
As a ubiquitous archaeal phylum in water and soil/sediment environments, Thaumarchaeota represent a large prokaryotic biomass involved in nitrification process (Webster et al., 2015) and have been reported to obviously predominate in the freshwater wetlands in the Yangtze estuary (Liu et al., 2017), in sediments of the Pearl River Estuary (Wang et al., 2020a) and estuary water of Chesapeake Bay, United States (Wang et al., 2020b). Webster et al. (2015) found that salinity importantly impacted distribution and composition of Thaumarchaeota. Yang et al. (2018) reported that the Thaumarchaeota populations in lake sediments were not significantly influenced by salinity. Kuznetsova et al. (2020) revealed that the occurrence of Thaumarchaeota in the saline soils in the Lake Elton area was driven by both salinity and pH. In this current study, Thaumarchaeota was also found to be the dominant archaea in all the three wetlands. However, the relative abundance of Thaumarchaeota was only negatively and significantly correlated with soil WC and silt content instead of soil salinity (p < 0.05, Figure 5a). The significantly lower soil moisture in soils of MIW than in FIW and lower slit content in MIW than in FIW and SIW explained why Thaumarchaeota was significantly enriched in MIW. Consistently, an estuarine sediment-seawater microcosm experiment confirmed that AOA showed the most abundant and the highest transcriptional activity under moderate salinity (Zhang et al., 2015). The differences in salinity range of such different environments might contribute to the different effects of salinity on the relative abundance of Thaumarchaeota. Besides, three classes (Group_C3, Soil_Crenarchaeotic_Group_SCG_ and South_African_Gold_Mine_Gp_1_SAGMCG-1_) belonging to Thaumarchaeota showed significantly higher relative abundance in soils of FIW than MIW and SIW. Thus, the methane emissions in FIW and ammonia oxidation in MIW would be affected in the scenario of seawater intrusion caused by sea level rise.
The class Halobacteria and 17 archaeal species belonging to Halobacteria were significantly enriched in the soils of SIW and all belong to the phylum Halobacterota. Members in the phylum Halobacterota can survive in environments with high salinity due to their high tolerance to salt (Xiao et al., 2021), showing a positively significant relationship with salinity (p < 0.05, Figure 5). Haloarchaea naturally inhabit and thrive under saline and hypersaline conditions (Torregrosa-Crespo et al., 2018; Naitam et al., 2023) and the high salinity in soils of SIW provided suitable habitats for archaea belonging to Halobacteria. Some members of Haloarcula and Haloferax were recognized as denitrifiers, contributing significantly to NO and N2O emissions and consequently contributing to ozone depletion and climate change (Torregrosa-Crespo et al., 2018). Therefore, the possible increased area of salt marshes might affect the emission of greenhouse gases.
This study highlighted the changes of archaeal community in wetlands under various hydrologic conditions. The different hydrologic conditions created differences in SOC, salinity, pH, WC and silt and sand contents in coastal wetlands, which consequently affected the archaeal community. Although the abundance and diversity of archaea were not affected by hydrologic conditions, the community structure of archaea differed among coastal wetlands with various hydrologic conditions. The relative abundance of Bathyarchaeota, Marine_Benthic_Group_E, Miscellaneous_Euryarchaeotic_Group_MEG_ and methanogens in the phylum Euryarchaeota decreased as the influence of saltwater intrusion enhanced. As representatives of ammonium oxidizers, Thaumarchaeota showed preference for soils of wetland with medium salinity, although they were detected as the dominant archaea in all sampling sites. The halophilic archaea in the class Halobacteria showed preference for high-salinity soils affected by tides. In the scenario saltwater intrusion due to sea level rise or climate change, the nitrification process and methanogenesis would be changed in coastal wetlands.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://www.ncbi.nlm.nih.gov/sra/?term=SRP099022.
QZ: Writing – original draft, Writing – review & editing, Formal Analysis, Investigation. JJ: Funding acquisition, Investigation, Writing – original draft, Writing – review & editing. FS: Funding acquisition, Visualization, Writing – review & editing. TL: Methodology, Software, Writing – original draft. WZ: Supervision, Validation, Writing – review & editing. YH: Project administration, Supervision, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This study was financially supported by National Natural Science Foundation (42207527), Major Innovative Projects in the Pilot Program for the Integration of Science, Education, and Industry of Qilu University of Technology (2024ZDZX10), and National Natural Science Foundation (U22A20615).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fmars.2025.1564173/full#supplementary-material
Baker B. J., De Anda V., Seitz K. W., Dombrowski N., Santoro A. E., Lloyd K. G. (2020). Diversity, ecology and evolution of Archaea. Nat. Microbiol. 5, 887–900. doi: 10.1038/s41564-020-0715-z
Chambers L. G., Guevara R., Boyer J. N., Troxler T. G., Davis S. E. (2016). Effects of salinity and inundation on microbial community structure and function in a mangrove peat soil. Wetlands 36, 361–371. doi: 10.1007/s13157-016-0745-8
Chen Y. J., Leung P. M., Cook P. L. M., Wong W. W., Hutchinson T., Eate V., et al. (2022). Hydrodynamic disturbance controls microbial community assembly and biogeochemical processes in coastal sediments. ISME J. 16, 750–763. doi: 10.1038/s41396-021-01111-9
Chen Y., Li S., Yu Z., Chen Y., Mi T., Zhen Y. (2020). Characteristics of the Bathyarchaeota community in surface sediments from the southern Yellow Sea and northern East China sea. Estuarine Coast. Shelf Sci. 235, 106595. doi: 10.1016/j.ecss.2020.106595
Cheung M. K., Wong C. K., Chu K. H., Kwan H. S. (2018). Community structure, dynamics and interactions of bacteria, archaea and fungi in subtropical coastal wetland sediments. Sci. Rep. 8, 14397. doi: 10.1038/s41598-018-32529-5
Gao X., Wang S., Kong W., Li G., Zhang L., Yin X. (2024). Floristic changes and environmental drivers of soil fungi and archaea in different salt-tolerant plant communities in the intertidal habitat of coastal wetlands. Environ. Geochem. Health 46, 167. doi: 10.1007/s10653-024-01951-2
Jifiriya M. J., Preena P. G., Rejish Kumar V. J., Nair A. J., Joseph V. (2023). Role of archaea in aquaculture: prospects and challenges. Aquacult. Int. 32, 3169–3194. doi: 10.1007/s10499-023-01317-y
Krzmarzick M. J., Taylor D. K., Fu X., McCutchan A. L. (2018). Diversity and niche of archaea in bioremediation. Archaea-An Int. Microbiol. J. 2018, 1–17. doi: 10.1155/2018/3194108
Kuznetsova A. I., Ivanova E. A., Samylina O. S., Kurbanova F. G., Gruzdev D. S., Kanapatskiy T. A., et al. (2020). Prokaryotic communities in saline soils of the lake Elton area in a soil catena along the Khara river. Microbiology 89, 670–684. doi: 10.1134/S0026261720060119
Lazar C. S., Baker B. J., Seitz K., Hyde A. S., Dick G. J., Hinrichs K., et al. (2016). Genomic evidence for distinct carbon substrate preferences and ecological niches of Bathyarchaeota in estuarine sediments. Environ. Microbial. 18, 1200–1211. doi: 10.1111/emi.2016.18.issue-4
Liang W., Tiantian Y. U., Dong L., Jia Z., Wang F. (2023). Determination of carbon-fixing potential of Bathyarchaeota in marine sediment by DNA stable isotope probing analysis. Sci. CHINA: Earth Sci. 66, 8. doi: 10.1007/s11430-022-1002-4
Liu K., Luo X., Jimmy Jiao J., Gu J.-D., Aravena R. (2021). Gene abundances of AOA, AOB, and anammox controlled by groundwater chemistry of the Pearl River Delta, China. China Geol. 4, 1–14. doi: 10.31035/cg2021081
Liu Y., Priscu J. C., Xiong J., Conrad R., Vick-Majors T., Chu H., et al. (2016). Salinity drives archaeal distribution patterns in high altitude lake sediments on the Tibetan Plateau. FEMS Microbiol. Ecol. 92. doi: 10.1093/femsec/fiw033
Liu X. Y., Tao K. Y., Sun J., He C. Q., Cui J., Chen X. P. (2017). The introduction of woody plants for freshwater wetland restoration alters the archaeal community structure in soil. Land Degrad. Dev. 28, 1933–1942. doi: 10.1002/ldr.v28.7
Ma Y., Liu F., Kong Z., Yin J., Kou W., Wu L., et al. (2016). The Distribution Pattern of Sediment Archaea Community of the Poyang Lake, the Largest Freshwater Lake in China. Archaea 2016, 9278929.
Maier R. M., Pepper I. L. (2009). Chapter 4 - Earth Environments. in Maier R. M., Pepper I. L., Gerba C. P., editors. Environmental Microbiology (Second Edition). (San Diego: Academic Press), 57–82. doi: 10.1016/B978-0-12-370519-8.00004-3
Maietta C. E., Hondula K. L., Jones C. N., Palmer M. A. (2020). Hydrological conditions influence soil and methane-cycling microbial populations in seasonally saturated wetlands. Front. Environ. Sci. 8. doi: 10.3389/fenvs.2020.593942
Meng K., Chung C. Z., Soll D., Krahn N. (2022). Unconventional genetic code systems in archaea. Front. Microbiol. 13, 1007832. doi: 10.3389/fmicb.2022.1007832
Muema E. K., Cadisch G., Rasche F. (2016). Soil texture modulates the response of ammonia-oxidizing prokaryotes to biochemical quality of organic inputs in tropical agricultural soils. Soil Biol. Biochem. 100, 218–228. doi: 10.1016/j.soilbio.2016.06.027
Naitam M. G., Ramakrishnan B., Grover M., Kaushik R. (2023). Rhizosphere-dwelling halophilic archaea: a potential candidate for alleviating salinity-associated stress in agriculture. Front. Microbiol. 14, 1212349. doi: 10.3389/fmicb.2023.1212349
Peralta A. L., Ludmer S., Matthews J. W., Kent A. D. (2014). Bacterial community response to changes in soil redox potential along a moisture gradient in restored wetlands. Ecol. Eng. 73, 246–253. doi: 10.1016/j.ecoleng.2014.09.047
Pfeifer K., Ergal İ., Koller M., Basen M., Schuster B., Rittmann S. K. M. R. (2021). Archaea biotechnology. Biotechnol. Adv. 47, 107668. doi: 10.1016/j.biotechadv.2020.107668
Saavedra-Bouza A., Escuder−Rodríguez J.-J., deCastro M.-E., Becerra M., González-Siso M.-I. (2023). Xylanases from thermophilic archaea: A hidden treasure. Curr. Res. Biotechnol. 5, 100116. doi: 10.1016/j.crbiot.2022.11.003
Torregrosa-Crespo J., Bergaust L., Pire C., Martinez-Espinosa R. M. (2018). Denitrifying haloarchaea: sources and sinks of nitrogenous gases. FEMS Microbiol. Lett. 365. doi: 10.1093/femsle/fnx270
Wang H., Bier R., Zgleszewski L., Peipoch M., Omondi E., Mukherjee A., et al. (2020b). Distinct distribution of archaea from soil to freshwater to estuary: implications of archaeal composition and function in different environments. Front. Microbiol. 11, 576661. doi: 10.3389/fmicb.2020.576661
Wang Z., Fuad M. T. I., Liu J., Lin K., Liu L., Gao C., et al. (2025). Spatial patterns of microbial communities in intertidal sediments of the yellow river estuary, China. Microb. Ecol. 87, 173. doi: 10.1007/s00248-025-02494-4
Wang W., Tao J., Liu H., Li P., Chen S., Wang P., et al. (2020a). Contrasting bacterial and archaeal distributions reflecting different geochemical processes in a sediment core from the Pearl River Estuary. AMB Express 10, 16. doi: 10.1186/s13568-020-0950-y
Webster G., O’Sullivan L. A., Meng Y., Williams A. S., Sass A. M., Watkins A. J., et al. (2015). Archaeal community diversity and abundance changes along a natural salinity gradient in estuarine sediments. FEMS Microbiol. Ecol. 91, 1–18. doi: 10.1093/femsec/fiu025
Wei G., Li M., Shi W., Tian R., Chang C., Wang Z., et al. (2020). Similar drivers but different effects lead to distinct ecological patterns of soil bacterial and archaeal communities. Soil Biol. Biochem. 144, 107759. doi: 10.1016/j.soilbio.2020.107759
Wen X., Yang S., Horn F., Winkel M., Wagner D., Liebner S. (2017). Global biogeographic analysis of methanogenic archaea identifies community-shaping environmental factors of natural environments. Front. Microbiol. 8, 1339. doi: 10.3389/fmicb.2017.01339
White S. M., Madsen E. A. (2016). Tracking tidal inundation in a coastal salt marsh with Helikite airphotos: influence of hydrology on ecological zonation at Crab Haul Creek, South Carolina. Remote Sens. Environ. 184, 605–614. doi: 10.1016/j.rse.2016.08.005
Wu D., Zhao C., Bai H., Feng F., Sui X., Sun G. (2021). Characteristics and metabolic patterns of soil methanogenic archaea communities in the high-latitude natural forested wetlands of China. Ecol. Evol. 11, 10396–10408. doi: 10.1002/ece3.v11.15
Xiao F., Li Y., Li G., He Y., Lv X., Zhuang L., et al. (2021). High throughput sequencing-based analysis of the soil bacterial community structure and functions of Tamarix shrubs in the lower reaches of the Tarim River. PeerJ 9, e12105. doi: 10.7717/peerj.12105
Xie W., Zhang C., Zhou X., Wang P. (2014). Salinity-dominated change in community structure and ecological function of Archaea from the lower Pearl River to coastal South China Sea. Appl. Microbiol. Biotechnol. 98, 7971–7982. doi: 10.1007/s00253-014-5838-9
Yang J., Jiang H., Wu G., Liu W. (2018). Phylum-level archaeal distributions in the sediments of Chinese lakes with a large range of salinity. Geomicrobiol. J. 35, 404–410. doi: 10.1080/01490451.2017.1382611
Zedler J. B. (2000). Progress in wetland restoration ecology. Trends Ecol. Evol. (Amst.) 15, 402–407. doi: 10.1016/S0169-5347(00)01959-5
Zhang Y., Chen L., Dai T., Tian J., Wen D. (2015). The influence of salinity on the abundance, transcriptional activity, and diversity of AOA and AOB in an estuarine sediment: a microcosm study. Appl. Microbiol. Biotechnol. 99, 9825–9833. doi: 10.1007/s00253-015-6804-x
Zhao Q., Bai J., Gao Y., Zhao H., Zhang G., Cui B. (2020b). Shifts in the soil bacterial community along a salinity gradient in the Yellow River Delta. Land Degrad. Dev. 31, 2255–2267. doi: 10.1002/ldr.v31.16
Zhao Q., Bai J., Wang X., Zhang W., Huang Y., Wang L., et al. (2020a). Soil organic carbon content and stock in wetlands with different hydrologic conditions in the Yellow River Delta, China. Ecohydrol. Hydrobiol. 20, 537–547. doi: 10.1016/j.ecohyd.2019.10.008
Zhao Z., Zhang L., Zhang G., Gao H., Chen X., Li L., et al. (2023). Hydrodynamic and anthropogenic disturbances co-shape microbiota rhythmicity and community assembly within intertidal groundwater-surface water continuum. Water Res. 242, 120236. doi: 10.1016/j.watres.2023.120236
Zou D., Liu H., Li M. (2020b). Community, distribution, and ecological roles of estuarine archaea. Front. Microbiol. 11, 2060. doi: 10.3389/fmicb.2020.02060
Keywords: archaea, diversity and community structure, soil salinity, hydrologic conditions, coastal wetlands
Citation: Zhao Q, Jia J, Song F, Li T, Zhang W and Huang Y (2025) Variations in the archaeal community in wetlands soils under various hydrologic conditions in the Yellow River Estuary. Front. Mar. Sci. 12:1564173. doi: 10.3389/fmars.2025.1564173
Received: 21 January 2025; Accepted: 03 March 2025;
Published: 21 March 2025.
Edited by:
Qin Zhu, Southern Marine Science and Engineering Guangdong Laboratory, ChinaCopyright © 2025 Zhao, Jia, Song, Li, Zhang and Huang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Qingqing Zhao, cWluZ3Fpbmd6aGFvQHFsdS5lZHUuY24=; Jia Jia, amlhamlhc3RAMTI2LmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.