
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Mar. Sci. , 27 March 2025
Sec. Coral Reef Research
Volume 12 - 2025 | https://doi.org/10.3389/fmars.2025.1539066
Mesophotic coral ecosystems (MCEs)—reefs below 30m depth—represent distinct ecological communities that are under threat from local (e.g., fishing) and global (e.g., climate change) disturbances. However, most MCEs remain unexplored, and their ecological communities are not well characterized. MCEs on remote offshore seamounts are further unexplored and provide the opportunity to assess assembly rules from comparatively less disturbed MCEs, given their remoteness from settlements with high human population densities. Here, we characterize the fish community from the remote offshore seamount of Pickle Bank in the Central Caribbean Sea, exploring differences in the fish community at a 25m depth compared to the mesophotic zone at a 45m depth. We found differences in species composition between the depths, with a significantly higher abundance of fish at mesophotic depths, while species diversity and species richness were significantly higher at the shallow sites. Species from Labridae and Scaridae dominated the biomass at the shallow sites, while species in the family Carangidae dominated biomass in the mesophotic zone. There were also differences in the community composition of trophic guilds between depths, with higher macrocarnivore biomass, macrocarnivore abundance, and omnivore abundance at deep sites compared to shallow sites. Despite the logistical challenges and limitations associated with accessing MCEs on offshore seamounts, these data provide compelling evidence to the growing body of literature documenting MCEs as unique habitats warranting further data collection to obtain a holistic understanding of shallow and mesophotic seamount ecosystems.
Remote offshore seamounts represent globally distributed marine habitats harboring unique mesophotic coral ecosystems (MCE) that remain comparatively unexplored. Seamounts can be defined as underwater mountains greater than 1,000m from the seafloor (Yesson et al., 2011), usually of volcanic origin (Staudigel and Clague, 2010). Because many seamounts are located in remote ocean regions, the characterization of seamount biodiversity is sparse, with data predominantly occurring from fisheries statistics (Clark et al., 2010; Stock et al., 2021). For MCEs, which occur between depths of approximately 30 to 150m (Pyle and Copus, 2019), and remain largely unknown (Kahng et al., 2010; Turner et al., 2019), remote operated vehicles and submersibles are used (Colin, 1974, 1976; Bryan et al., 2013). However, recent advances and accessibility with closed-circuit rebreather technology allows for more comprehensive in situ assessments of MCE biological communities (Garcia-Sais, 2010; Lesser and Slattery, 2011; Bejarano et al., 2014; Pinheiro et al., 2016; Le Gall et al., 2024).
Previous assessments of MCE fish communities have highlighted distinct ecological processes and changes across the depth gradient. For example, changes in the fish community are often related to different topographic features (Lesser et al., 2009), which are heavily influenced by light availability (Laverick et al., 2017; Carpenter et al., 2022; Pérez-Castro et al., 2022). Such changes include a higher prevalence of generalist fish species on mesophotic reefs (>30m) compared to photic reefs (<30m), and an increase in macrocarnivore biomass with depth compared to other trophic guilds (Pinheiro et al., 2016). Additionally, the influence of geographic factors related to isolation (such as allopatry) is far less prevalent on mesophotic reef fish communities compared to their shallow water counterparts (Pinheiro et al., 2023). In turn, differences in species richness between mesophotic reefs within the same bioregions are far less pronounced compared to regional differences for shallow water reefs (Pinheiro et al., 2016, 2019, 2023; Pyle and Copus, 2019), indicating that mesophotic fish assemblages are governed by ecological processes distinct from shallow water coral reefs (Rocha et al., 2018; Pinheiro et al., 2023).
While the characterization of MCEs continues to grow (Pyle and Copus, 2019), MCEs associated with remote offshore seamounts remain comparatively unexplored (Soares et al., 2019). Predominantly within the Atlantic, in situ characterization of tropical MCEs fish communities has occurred on the Vitória-Trindade Seamount Chain (Pinheiro et al., 2011, 2015), and the Flower Banks Garden Banks (Voss et al., 2014; Sanchez et al., 2023) in the Gulf of Mexico. Meanwhile, seamounts in the Caribbean Sea are generally uncharacterized, hampering efforts to gain meaningful insight into ecological processes across depth gradients from remote and less disturbed MCEs. Given that assembly rules for fish are different for mesophotic compared to shallow water reefs, such as convergent filtering of species that occur on mesophotic reefs (Pinheiro et al., 2023), it is important to understand whether MCEs on remote seamounts adhere to the same principles as nearshore MCEs. Additionally, seamounts often act as stepping stones for coral and fish communities, suggesting they are important for genetic connectivity over large spatial scales (Clark et al., 2010; Rogers, 2018; Galbraith et al., 2024), analogous to the role of islands as stepping stones for land-based species (Pinheiro et al., 2017). Remote seamounts are also magnets for higher trophic level fish, including carnivorous mesophotic reef fish (Morato et al., 2010; Cresswell et al., 2023) through to predatory megafauna associated with open seas (Morato et al., 2010; Weber et al., 2025) Finally, remote seamounts also tend to be less exploited than nearshore MCEs fisheries (Clark et al., 2010), hence, a deeper insight into ecological processes from a comparatively undisturbed environment can be obtained.
Finally, effective policy to protect ecosystems cannot be enacted without sufficient understanding of the ecological community (Turner et al., 2019). Thus, there is an urgent requirement to characterize the mesophotic fish community and understand ecological processes across depth gradients in reef fish communities (Turner et al., 2019). Such characterization becomes especially urgent as seamounts become more widely recognized as fishery resources (Clark et al., 2010; Stock et al., 2021). To address this critical knowledge gap, we performed the first documented in situ fish surveys on the remote offshore seamount of Pickle Bank, located in the Caribbean Sea. We conducted surveys in the photic zone (25m) and the upper mesophotic zone (45m) to provide further insights into the ecological processes of mesophotic fish communities.
Pickle Bank seamount is a remote offshore seamount located in the Central Caribbean Sea, 78km north-north-west of Little Cayman and 120km south-west of Cuba (Figure 1A; 20.417°, -80.417°). The seamount is occasionally visited by recreational and commercial fishermen as a prime location for catching large pelagic species, yet there is no formal characterization of the fish communities. The seamount is approximately 5.2 square km and is characterized by a central lagoon, with large patch reef outcroppings rising to 14m in depth. These outcroppings transition to spur and groove reef structures, leading to a deep reef edge at roughly 35m depth before descending with near vertical walls to depths exceeding 1,000m (Figures 1B, C).
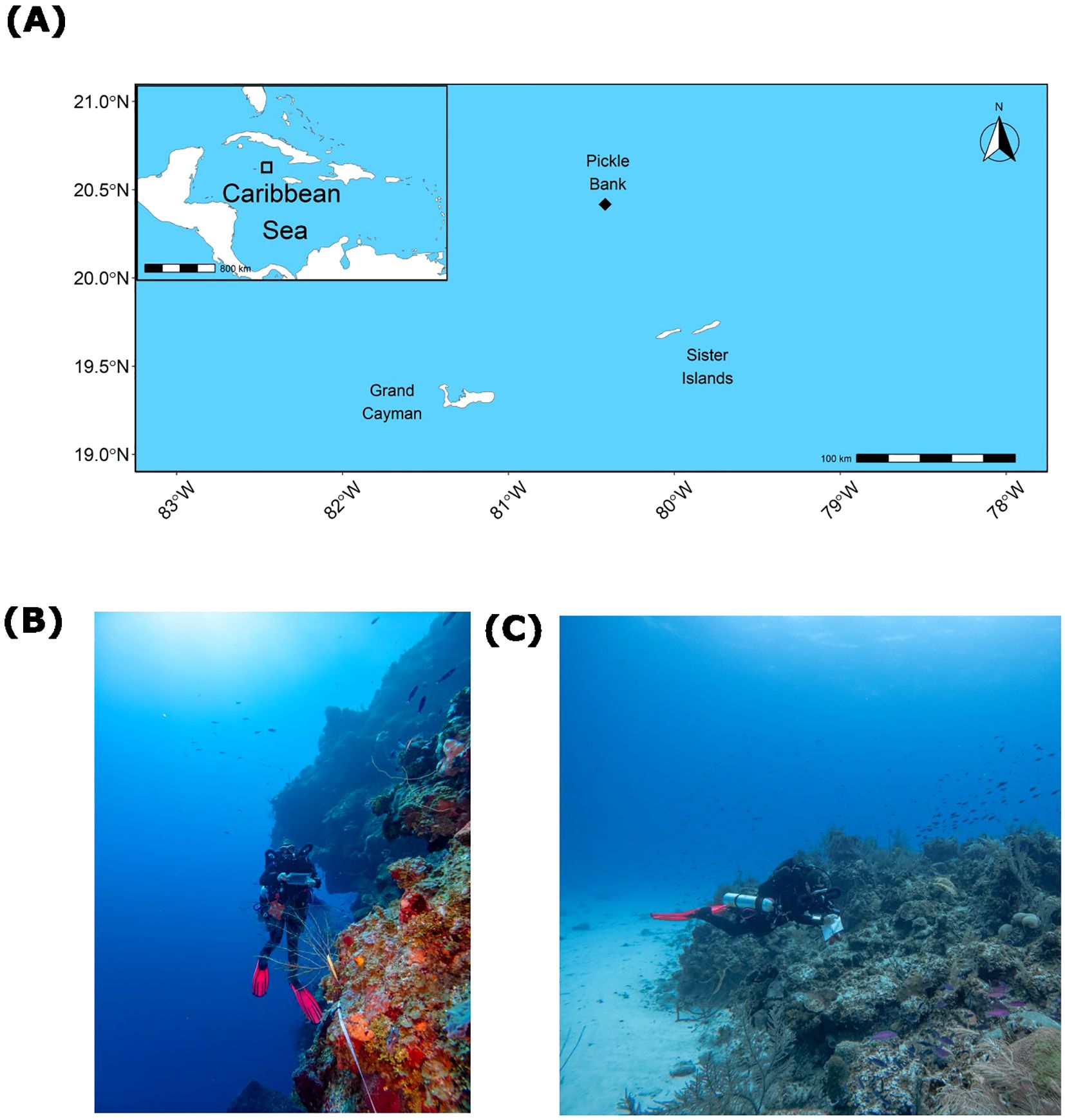
Figure 1. Location of Pickle Bank seamount relative to the Cayman Islands (A). Example of fish transect and habitat type at the 45m depth (B), and an example of habitat type surveyed for shallow sites at the 25m depth (C).
In situ visual fish surveys were conducted on Pickle Bank from 14 July 2024 to 17 July 2024 using closed-circuit rebreathers (Prism2, Hollis Rebreathers) by two experienced technical divers and professional scientists who have extensively published fish data from mesophotic depths in the Western Atlantic (Pinheiro et al., 2016; Andradi-Brown et al., 2017; Goodbody-Gringley et al., 2019, 2023; Le Gall et al., 2024). Due to the time required to reach the seamount each day (8-h round trip) and safety requirements of diving to 45m, a maximum of two dives were conducted each day, limiting the total number of surveys attainable within the logistical constraints of the expedition. A total of 17 fish transects were surveyed at two different depth ranges on the seamount, with shallow surveys (n=8) conducted at 25m, while deep surveys (n=9) were conducted at 45m. At shallow sites, replicate transects (30m x 2m) were haphazardly placed on reef spurs (Figure 1C) that resembled classical benthic assemblages associated with shallow coral reef habitats in the region (Manfrino et al., 2013). Deep transects were attached haphazardly to the benthos and laid horizontally along the reef wall to ensure a constant depth, with benthic assemblages resembling those described for nearshore MCEs in the Cayman Islands (Figure 1B; Slattery and Lesser, 2019; Carpenter et al., 2022; Le Gall et al., 2024). Along each transect, all fish encountered were identified to the species level and their total length visually estimated (Johnson et al., 2023; Le Gall et al., 2024). Fish total length was categorized into size classes (0–5 cm, 6–10 cm, 11–20 cm, 21–30 cm, 31–40 cm, and > 40 cm estimated to the nearest 10cm value) to allow for biomass calculations using the formula:
where W is the weight of the fish and L is the maximum length based on the size classes above. Values for a and b are species-specific constants based on empirical data for calculating fish biomass from size-weight relationships (Bohnsack and Harper, 1988; Torres, 1991; Froese and Pauly, 2010). These constants were obtained from Fish Base (Froese and Pauly, 2010), with values from congenic species used if data for a specific species were not available. Each fish species was subsequently grouped into the appropriate trophic guild based on groupings also derived from Fish Base (Froese and Pauly, 2010).
All data manipulation and statistical analyses were conducted using R 4.1.1 (R Core Team, 2023). Comparisons between depths for fish biomass and species richness of all fish were compared using ANOVAs, as data were normally distributed based on visual inspection of histograms, statistically confirmed with a Shapiro–Wilks test. Mann–Whitney U tests were used to compare the abundance and Shannon diversity of fish between the depths and for comparisons of biomass and abundance for all fish trophic guilds between the depths, as these data did not follow a normal distribution (Shapiro–Wilks, P<0.001). The Kruskal–Wallis test was used to compare the abundance and biomass of fish among trophic guilds as these data were also non-normally distributed (Shapiro–Wilks, P<0.001). Post hoc Dunn’s tests were used for pairwise comparisons of trophic guilds implemented using the FSA package (Ogle et al., 2017). To assess differences in the community composition of all fish species between the depths, and the community composition of trophic guilds, we used a PERMANOVA from the vegan package (Oksanen et al., 2020). Bray–Curtis dissimilarity matrices of the community were calculated from data normalized with a square-root transformation. Ordinations using non-metric multi-dimensional scaling (nMDS) were built also using the vegan package on the Bray–Curtis dissimilarity matrix (Oksanen et al., 2020).
Across all of our transects (n=17), we recorded a total 6,049 individual fish, representing 45 species. Between the two depth zones surveyed (25m vs. 45m), species richness (Figure 2A) was significantly higher at 25m (median = 19), compared to 45m (median = 14) (ANOVA, F=5.929, P=0.028). There was no difference in total fish biomass between the depths (Figure 2B). However, total fish abundance (Figure 2C) was significantly higher at 45m compared to 25m (Mann–Whitney U, W=63, P=0.008), while fish diversity (Figure 2D) was significantly lower at 45m compared to 25m (W=1, P<0.001). Additionally, there is a significant difference in the community composition between the mesophotic and shallow reef depths (Figure 2E; PERMANOVA, F=11.939, df=1, P<0.001).
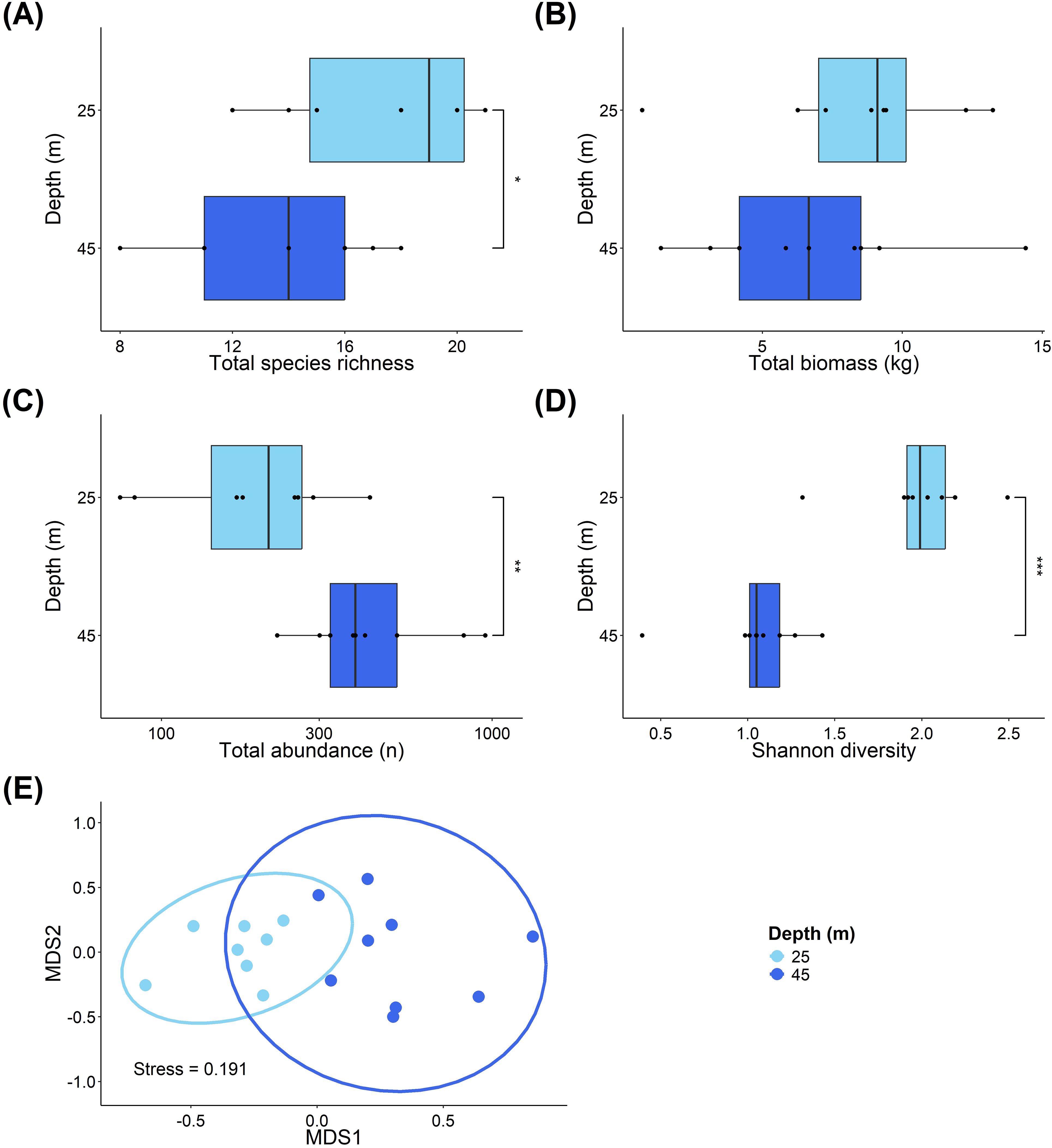
Figure 2. Comparisons of fish community structure at different depths on Pickle Bank Seamount. (A) shows species richness at the two depths, (B) is fish biomass, (C) is total abundance, (D) shows Shannon diversity, while (E) is an ordination of the fish community across depths based on a Bray-Curtis dissimilarity matrix. Each point represents a transect, with depths differentiated by shades of blue. Light blue depicts transects at the shallow depth (25m), while dark blue shows transects from the mesophotic sites (45m).
Overall, we recorded 16 fish families on our transects, with the highest biomass at the shallow depth for the families Labridae, Scaridae, Carangidae, and Balistidae (Figure 3A). Comparatively, Carangidae and Labridae were the two dominant families of biomass at the mesophotic depth (Figure 3B). An overview of species contributions to biomass at the two depths and overall are shown in Figure 3C.
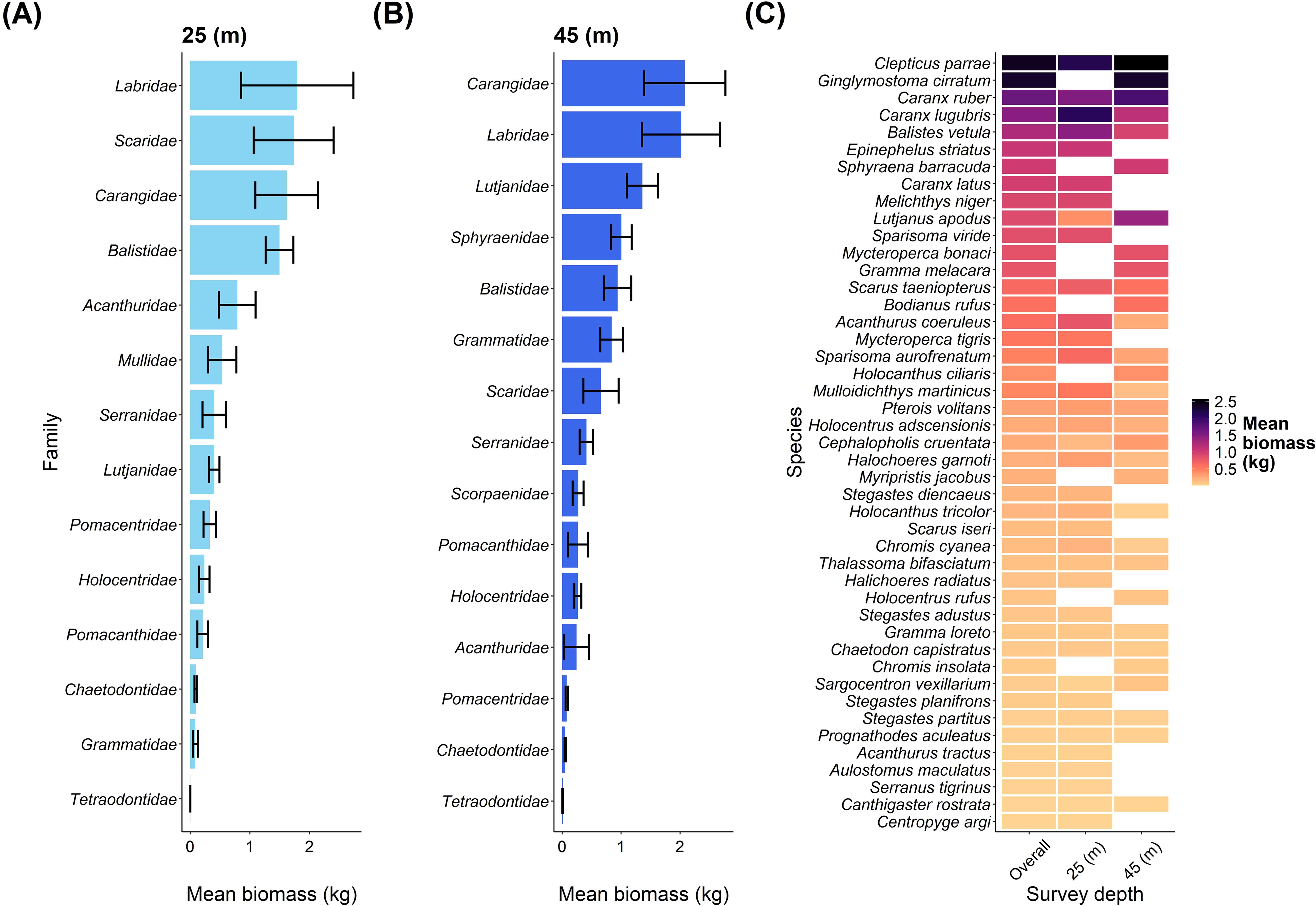
Figure 3. An overview of fish families and fish species biomass between depths at Pickle Bank. (A) shows the mean biomass of fish families at 25m, while (B) shows the mean biomass of fish families at 45m. (C) is the mean biomass of every fish species recorded on our transects for both depths combined (overall) and each depth separately. Note that only 29 of the 33 families are displayed as 4 families were only recorded once, so mean and standard error could not be calculated.
Regarding fish abundance, Labridae was the dominant fish family at the shallow depth, followed by Pomacentridae and Grammatidae (Figure 4A). These families were also dominant at the mesophotic depth, but Grammatidae showed the highest abundance, followed by Labridae and Pomacentridae (Figure 4B). Gramma melacara had the largest contribution out of all the species to overall fish abundance but was only recorded at the mesophotic depth. An overview of all species abundance contributions to each depth is shown in Figure 4C.
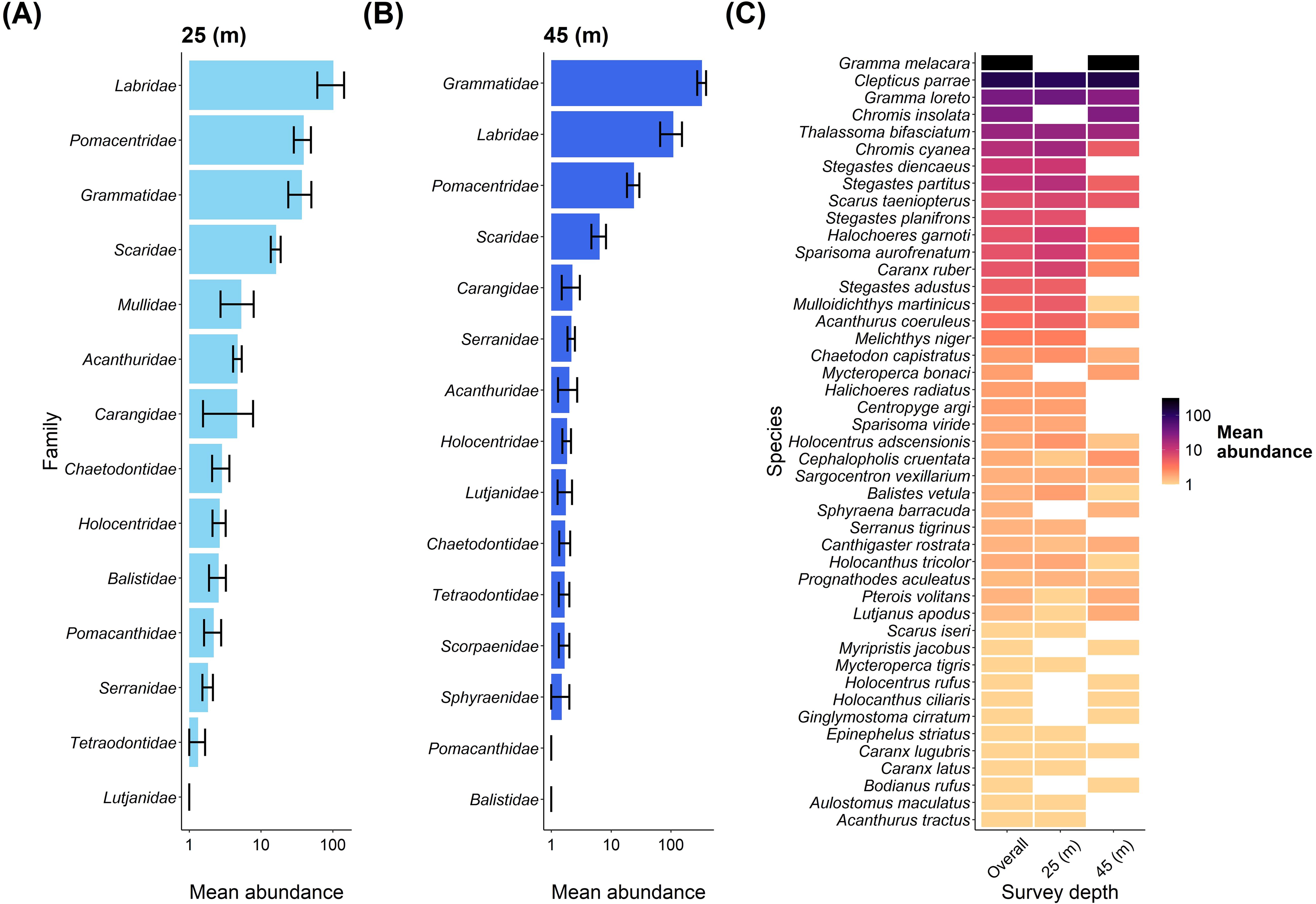
Figure 4. An overview of fish families and fish species abundance between depths at Pickle Bank. (A) shows the mean abundance of fish families at 25m, while (B) shows the mean abundance of fish families at 45m. (C) is the mean abundance of every fish species recorded on our transects for both depths combined (overall) and each depth separately (note log10 scale).
The biomass of reef fish on Pickle Bank was not significantly different across trophic guilds (Figure 5A), however, there were significant differences between the depths (Figure 5B). Macrocarnivores had significantly higher biomass at the mesophotic sites compared to the shallow sites (W=8, P=0.02), while planktivore biomass was significantly higher at the shallow depth compared to the mesophotic depth (W=63, P=0.008).
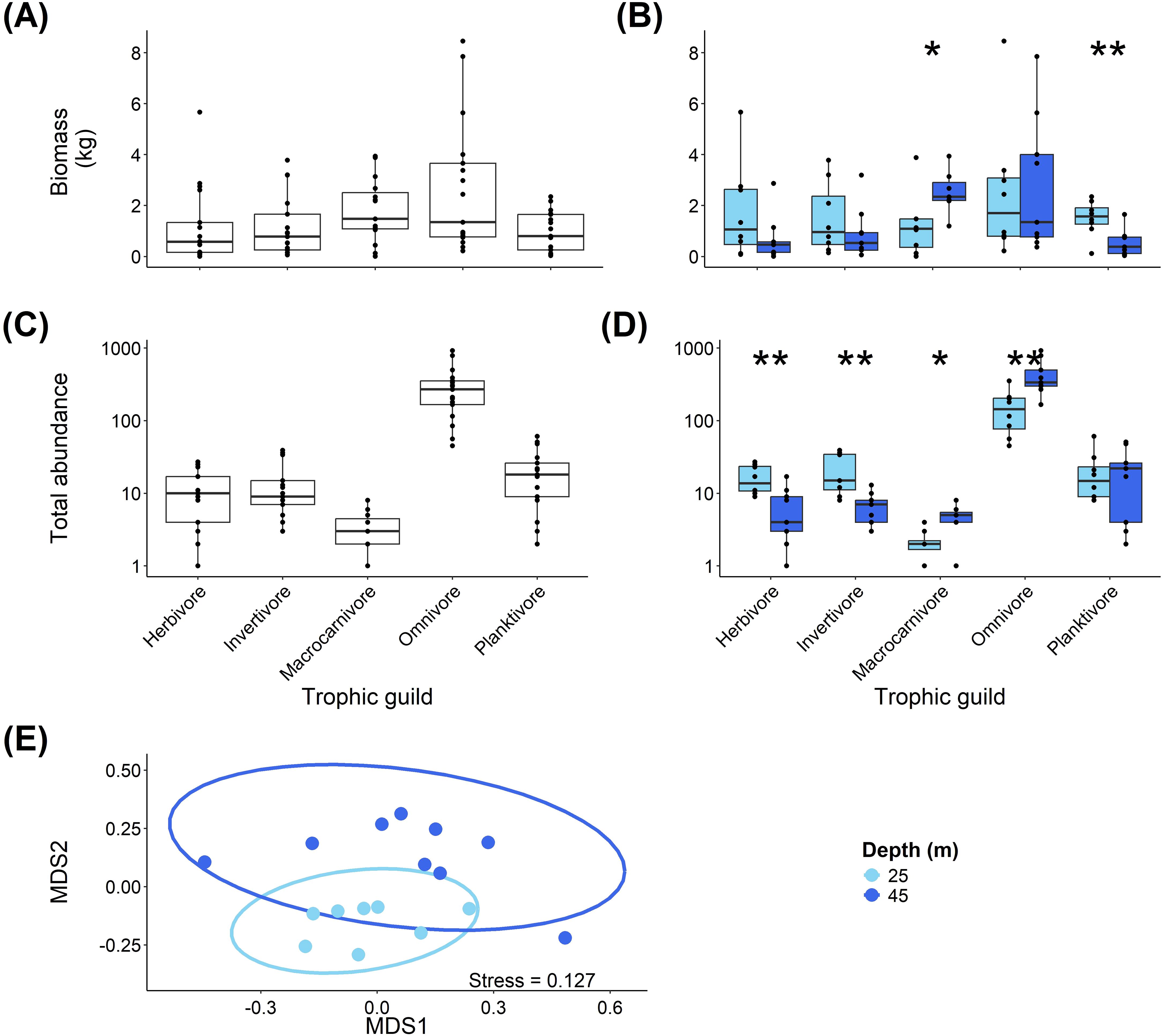
Figure 5. Fish community structure at Pickle Bank seamount based on trophic guilds. (A) shows the biomass of each trophic guild recorded on the seamount, while (B) shows the comparison of biomass between depths. (C) is the abundance of fish in each trophic guild for the entire seamount, while (D) shows the comparison between depths. (E) is an ordination of the trophic guild community structure for each depth.
The shallow and mesophotic sites at Pickle Bank showed a significant difference in the abundance of fish (Figure 5C) across trophic guilds (χ2 = 53.758, df=4, P<0.001). Pairwise comparisons of the difference between trophic guilds are shown in Table 1.
There were significant differences in the abundances of trophic guilds between the shallow (depth of 25m) and mesophotic (depth of 45m) sites (Figure 5D), with higher herbivore abundance (W=64, P=0.008) and higher invertivore abundance (W=66, P=0.004) at the shallow depth compared to the mesophotic sites. Meanwhile, higher macrocarnivore abundance (W=8, P=0.022) and higher omnivore abundance (W=8, P=0.006) were recorded at the mesophotic sites compared to the shallow sites.
Fianlly, there was a significant difference in the trophic community composition between the depths (Figure 5E; PERMANOVA, F=8.894, df=1, P<0.001).
Our findings present the first in situ characterization of Pickle Bank seamount, revealing a depth-dependent partitioning of the reef fish community. While fisheries data from Pickle Bank exists (Stock et al., 2021), catch data fails to reveal detailed ecological insights available from in situ surveys. We discuss these findings in the context of ecological processes observed in reef fish throughout other nearshore and offshore Caribbean MCEs. Additionally, we emphasize the potential importance of Pickle Bank and explore the need for future research to inform adequate protection.
From our 17 transects, we found distinct differences in the fish community between the 25m and 45m depths. Notably, higher species richness and higher diversity of reef fish exist in the shallow water, generally congruent with the higher habitat complexity associated with shallow water reefs that drives fish diversity (Cornell and Karlson, 2000). However, higher fish abundance at greater depths on Pickle Bank is driven by large schools of species such as Gramma melacara, which were absent at the 25m depth. Overall, it appears that while Labridae, Grammatidae, and Pomacentridae dominate the fish communities at both depths (Figure 3), the relative proportions of species in each family drove differences in the fish community.
Such partitioning is reinforced by differences in the biomass and abundance of different trophic groups. For example, higher biomass of typical macrocarniverous reef fish (e.g., species from Carangidae and Lutjanidae) was observed at 45m, congruent with the hypothesis that interspecific competition in shallow water leads to increased carnivore biomass at greater depths (Pinheiro et al., 2023; Grove et al., 2024; Heidmann et al., 2024). However, this hypothesis also applies to planktivorous fish, yet we found higher planktivore biomass at shallow depths. Higher planktivore biomass at shallow depths on Pickle Bank may be an artifact of the limited sample size from our study, which is supported by there being no difference between planktivore abundance between depths. Alternatively, it could be related to the hydrodynamic drivers of fish communities (Galbraith et al., 2023), which are unquantified on Pickle Bank. Herbivore and invertivore abundances are significantly greater at shallow depths on Pickle Bank compared to deeper sites, aligning with the species-energy hypothesis (Whittaker et al., 2001), where light-dependent resources that drive benthic energy availability become limited at greater depths (Pinheiro et al., 2023). Thus, the overall differences in the species and trophic communities between the depth found here appear to follow the prevailing hypotheses regarding fish communities across depth gradients.
Overall, the observed depth partitioning of the Pickle Bank fish community generally aligns with assembly rules that govern fish communities along the depth gradient (Pinheiro et al., 2023), with differences in community composition at 25–27m compared to 44–46m depths. These differences, while preliminary based on our small sample size of 17 transects, also likely exist because of convergent filtering of fish species with depth, where taxonomic groups and trophic strategies are similar at mesophotic depths because of the environmental conditions, placing less emphasis on regional and biogeographic drivers (Pinheiro et al., 2023). For example, our mesophotic sites show similar species compositions as nearby mesophotic locations, including Cuba (Cobián-Rojas et al., 2021), Grand Cayman (Le Gall et al., 2024), and more distant locations such as Puerto Rico (Bejarano et al., 2014), US virgin islands (Grove et al., 2024; Heidmann et al., 2024), Curacao (Pinheiro et al., 2016), and the Mesoamerican Barrier Reef (Andradi-Brown et al., 2016). Additionally, the shallow (25m) fish community appears similar to the adjacent shallow water reefs of Cuba (Navarro-Martínez et al., 2022) and the Cayman Islands (Johnson et al., 2024) based on species composition.
Given that our findings are provisionally congruent with the theories on depth-dependent rules associated with reef fish assemblages and the unexplored nature and the likely importance of Pickle Bank as a stepping stone, our findings incentivize further research to understand the ecological community and enact policy for protection. Pickle Bank likely maintains a healthy fishery given the larger biomass of targeted fishery species (mainly macrocarnivores), such as grouper, in comparison to the rest of the Cayman Islands (Stock et al., 2021). Larger biomass of targeted fishery species is likely associated with reduced harvesting pressure, highlighting the efficacy of reduced harvesting for sustaining fish stocks (Caldwell et al., 2024). Additionally, offshore seamounts provide a habitat for carnivorous reef fish and can act as magnets for predatory fish biomass (Cresswell et al., 2023; Weber et al., 2025), likely also explaining the larger grouper community at Pickle Bank compared to nearshore reefs of Little Cayman and Grand Cayman. Furthermore, Pickle Bank is likely a stepping stone for fish species, contributing to region-wide genetic connectivity, as overlap in species composition between nearshore reefs of Cuba and the Cayman Islands in shallow waters clearly exist (Navarro-Martínez et al., 2022; Johnson et al., 2023). Connectivity between mesophotic seamounts for reef-dwelling fish communities can transcend biogeographic realms, for example, between the Coral Sea, Southwest Pacific, and the Coral Triangle (Galbraith et al., 2024). Such connectivity via either larval dispersal or migrations of pelagic species in the Caribbean appears likely.
Additionally, the overall species richness of fish on Pickle Bank (45 species recorded) being similar to mesophotic reefs in the nearby island of Grand Cayman (48 species recorded), species per transect also being similar (Le Gall et al., 2024), may suggest these mesophotic depths are more sheltered from local anthropogenic stress even in proximity to high human population density. For example, shallow water reef fish are substantially impacted by local-scale activity associated with high human populations in Grand Cayman, such as the presence of mega cruise ships (Johnson et al., 2023). Yet, little difference in fish species richness at mesophotic depths between Pickle Bank and Grand Cayman may suggest the depth of MCEs buffer impacts of such local-scale activity. However, it is important to consider that further quantification of fish communities from both Pickle Bank and Grand Cayman mesophotic reefs are required to reach saturation for measuring species richness. One would expect that the combination of remoteness and depth may shelter fauna associated with Pickle Bank from anthropogenic activity, at least while Pickle Bank is not directly targeted.
For these reasons, ensuring Pickle Bank remains sheltered from unsustainable harvesting activities is likely crucial to avoid ecosystem collapse, given the small size of the seamount. Further characterization of the fish and benthic community will be beneficial for understanding ecological processes in this unique ecosystem and further inform effective management. Such ecological processes can be further understood with a holistic characterization of ecological communities, including genetic connectivity and the role of remote mesophotic reefs as a refuge during climate change (Smith et al., 2014; Bongaerts and Smith, 2019). The role of remote offshore seamounts as refuges during the current rapid period of global warming is especially pertinent given that remoteness is not protection from heatwaves for benthic organisms such as corals (Baumann et al., 2022; Johnson et al., 2022), and depth-dependent survival during thermal stress depends on a variety of factors (Smith et al., 2016; Rocha et al., 2018). Thus, acquiring more data on the fish community and characterizing the benthic community is vital for implementing effective policy to protect the unique habitat of Pickle Bank seamount.
Our work characterizing the fish community of Pickle Bank is the first step toward quantifying the community in this unique habitat. Quantifying mesophotic communities on offshore seamounts with in situ surveys is an incredible logistical challenge, but it is an important challenge to overcome given the insights society can gain from ecological, evolutionary, and biogeographical theories (Pinheiro et al., 2017, 2023; Lesser et al., 2018). Additionally, if we are to effectively protect the biological communities associated with offshore seamounts, we first need a holistic understanding of the community that exists (Lesser et al., 2018), followed by continued monitoring efforts to assess community changes as pressures from climate warming, mining, and overfishing inevitably reach untouched MCEs (Rocha et al., 2018; Rogers, 2018; Pinheiro et al., 2019). Therefore, while we show the differences in the communities between depths, further data to reach saturation points for species accumulation would greatly increase the ecological insights we can infer from the unique habitat of Pickle Bank seamount. Characterizing the benthic community is especially paramount to further understand the influence on the fish community. Such characterization becomes even more important given the threats posed to benthic coral habitats, including MCEs.
In conclusion, our findings provisionally support the notion of assembly rules across the depth gradient for reef fish (Pinheiro et al., 2023), with patterns of assembly on Pickle Bank congruent with that of the rest of the Caribbean (Pinheiro et al., 2016; Le Gall et al., 2024). However, given the logistical challenges associated with offshore mesophotic research, further characterization of the benthic and fish communities is required for a holistic understanding of the ecological process on Pickle Bank seamount. Yet, it remains likely that Pickle Bank acts as a stepping stone for genetic connectivity in the Central Caribbean and maintains a healthy fishery of commercially targeted species (Stock et al., 2021), likely rendering the seamount valuable both economically and ecologically. Further characterization of the physical and biological environment is also required to quantify the economic value and ecological importance of Pickle Bank seamount. Such quantification would enable informed policy to enact effective protection, providing benefits for both nature and society.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://github.com/JackVJohnson/Pickle-Bank-Fish-community.
Ethical approval was not required for the study involving animals in accordance with the local legislation and institutional requirements because the data recorded was purely observational.
JJ: Data curation, Formal Analysis, Investigation, Writing – original draft, Writing – review & editing. AC: Conceptualization, Data curation, Funding acquisition, Investigation, Methodology, Project administration, Resources, Validation, Visualization, Writing – review & editing. GGG: Conceptualization, Data curation, Formal Analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Supervision, Validation, Visualization, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. Funding for this work was provided by the UK Government through Darwin Plus (Ref: DPLUS162) and a matching fund made by an anonymous private donor. The donor was not involved in the study design, collection, analysis, interpretation of data, the writing of this article, or the decision to submit it for publication.
We are grateful to Nat Robb and Chris Nicholson from InDepth Watersports for field assistance, vessel support, and mediocre company during the long days out to Pickle Bank. We thank Arthur Trembanis and his team from the University of Delaware for conducting initial mapping surveys that provided critical guidance for determining the location of each of our dives. Thanks to Nicole Rotelle, Brooke Enright, Leon Schlenger, and Chloe Lee for assistance in the field as deck support and safety divers. Thanks to Ryan Eckert and Ashley Carreiro for joining us for fieldwork, contributing to a formidable rebreather dive team. Thank you to Balt von Huene and Lowell Forbes for their valiant efforts with logistics. We appreciate the support from Little Cayman Beach Resort for allowing us to use their dock after Hurricane Beryl destroyed ours. Thank you to Doug, Grace, and Scottish Davey from Little Cayman Divers for logistical assistance with the boat, and all-around emotional support. We thank our project collaborators for guidance and planning support, including those from the Cayman Islands Department of Environment and the Guy Harvey Ocean Foundation. We are grateful to two fantastic reviewers who’s thoughts and comments have improved our manuscript.
We dedicate this paper to the late Emily Christian Lopez, otherwise known as Ms Em, a renowned figure within the Sister Islands community, who committed 16 years of her life to supporting researchers and education groups at CCMI.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Andradi-Brown D. A., Gress E., Wright G., Exton D. A., Rogers A. D. (2016). Reef fish community biomass and trophic structure changes across shallow to upper-mesophotic reefs in the mesoamerican barrier reef, Caribbean. PLoS One 11, e0156641. doi: 10.1371/journal.pone.0156641
Andradi-Brown D. A., Vermeij M. J., Slattery M., Lesser M., Bejarano I., Appeldoorn R., et al. (2017). Large-scale invasion of western Atlantic mesophotic reefs by lionfish potentially undermines culling-based management. Biol. Invasions 19, 939–954. doi: 10.1007/s10530-016-1358-0
Baumann J. H., Zhao L. Z., Stier A. C., Bruno J. F. (2022). Remoteness does not enhance coral reef resilience. Global Change Biol. 28, 417–428. doi: 10.1111/gcb.15904
Bejarano I., Appeldoorn R., Nemeth M. (2014). Fishes associated with mesophotic coral ecosystems in La Parguera, Puerto Rico. Coral reefs 33, 313–328. doi: 10.1007/s00338-014-1125-6
Bohnsack J. A., Harper D. E. (1988). Length-weight relationships of selected marine reef fishes from the southeastern United States and the Caribbean. NOAA Technical Memorandum NMFS-SEFC-215, 31 p. doi: 10.1007/978-3-319-92735-0_45
Bongaerts P., Smith T. B. (2019). Beyond the “Deep Reef Refuge” hypothesis: a conceptual framework to characterize persistence at depth. Mesophotic coral Ecosyst., 881–895. doi: 10.1007/978-3-319-92735-0_1
Bryan D. R., Kilfoyle K., Gilmore R. Jr., Spieler R. E. (2013). Characterization of the mesophotic reef fish community in south F lorida, USA. J. Appl. Ichthyology 29, 108–117. doi: 10.1111/j.1439-0426.2012.02055.x
Caldwell I. R., McClanahan T. R., Oddenyo R. M., Graham N. A. J., Beger M., Vigliola L., et al. (2024). Protection efforts have resulted in ~10% of existing fish biomass on coral reefs. Proc. Natl. Acad. Sci. 121, e2308605121. doi: 10.1073/pnas.2308605121
Carpenter G. E., Chequer A. D., Weber S., Mass T., Goodbody-Gringley G. (2022). Light and photoacclimatization drive distinct differences between shallow and mesophotic coral communities. Ecosphere 13, e4200. doi: 10.1002/ecs2.4200
Clark M. R., Rowden A. A., Schlacher T., Williams A., Consalvey M., Stocks K. I., et al. (2010). The ecology of seamounts: structure, function, and human impacts. Annu. Rev. Mar. Sci. 2, 253–278. doi: 10.1146/annurev-marine-120308-081109
Cobián-Rojas D., Navarro-Martínez Z., García-Rodríguez A., David A., Chevalier-Monteagudo P., Drummond F., et al. (2021). Characterization of fish assemblages in mesophotic reefs of Cuba. Bull. Mar. Sci. 97, 443–472. doi: 10.5343/bms.2020.0067
Colin P. L. (1974). Observation and collection of deep-reef fishes off the coasts of Jamaica and British Honduras (Belize). Mar. Biol. 24, 29–38. doi: 10.1007/BF00402844
Colin P. L. (1976). Observations of deep-reef fishes in the Tongue-of-the-Ocean, Bahamas. Bull. Mar. Sci. 26, 603–605. doi: 10.1007/978-3-319-92735-0_52
Cornell H., Karlson R. (2000). Coral species richness: ecological versus biogeographical influences. Coral reefs 19, 37–49. doi: 10.1007/s003380050224
Cresswell B. J., Galbraith G. F., Harrison H. B., McCormick M. I., Jones G. P. (2023). Coral reef pinnacles act as ecological magnets for the abundance, diversity and biomass of predatory fishes. Mar. Ecol. Prog. Ser. 717, 143–156. doi: 10.3354/meps14377
Galbraith G. F., Cresswell B. J., McClure E. C., Hoey A. S. (2024). Tropical seamounts as stepping-stones for coral reef fishes: range extensions and new regional distributions from mesophotic ecosystems in the Coral Sea, Australia. Mar. Biodivers. 54, 17. doi: 10.1007/s12526-024-01404-0
Galbraith G. F., Cresswell B. J., McCormick M. I., Jones G. P. (2023). Strong hydrodynamic drivers of coral reef fish biodiversity on submerged pinnacle coral reefs. Limnology Oceanography 68, 2415–2430. doi: 10.1002/lno.12431
Garcia-Sais J. R. (2010). Reef habitats and associated sessile-benthic and fish assemblages across a euphotic–mesophotic depth gradient in Isla Desecheo, Puerto Rico. Coral Reefs 29, 277–288. doi: 10.1007/s00338-009-0582-9
Goodbody-Gringley G., Chequer A., Grincavitch C., Noyes T., Dowell R., Lundberg A., et al. (2023). Impacts of recurrent culling of invasive lionfish on mesophotic reefs in Bermuda. Coral Reefs 42, 443–452. doi: 10.1007/s00338-023-02354-y
Goodbody-Gringley G., Eddy C., Pitt J. M., Chequer A. D., Smith S. R. (2019). Ecological Drivers of Invasive Lionfish (Pterois volitans and Pterois miles) Distribution Across Mesophotic Reefs in Bermuda. Front. Mar. Sci. 6. doi: 10.3389/fmars.2019.00258
Grove L. J. W., Blondeau J., Swanson D. W., Heidmann S. L., Smith S. G., Johnson M. W., et al. (2024). Expansion of an established fishery-independent survey into the US Virgin Islands’ upper mesophotic zone: feasibility and management implications. Bull. Mar. Sci. 100, 113–132. doi: 10.5343/bms.2023.0013
Heidmann S. L., Olinger L. K., Brandtneris V. W., Ennis R. S., Blondeau J., Grove L. J. W., et al. (2024). Depth and structure as environmental drivers of fish communities across a shallow to mesophotic gradient in the northern US Virgin Islands. Bull. Mar. Sci. 100, 133–154. doi: 10.5343/bms.2023.0022
Johnson J. V., Bruno J. F., Gall L. L., Doherty M. L., Chequer A., Gringley G. G. (2024). Creation of complex reef structures through coral restoration does not affect associated fish populations on a remote, well-protected, Caribbean reef. PeerJ 12, e17855. doi: 10.7717/peerj.17855
Johnson J. V., Chequer A. D., Goodbody-Gringley G. (2023). Insights from the 2-year-long human confinement experiment in Grand Cayman reveal the resilience of coral reef fish communities. Sci. Rep. 13, 21806. doi: 10.1038/s41598-023-49221-y
Johnson J. V., Dick J. T. A., Pincheira-Donoso D. (2022). Local anthropogenic stress does not exacerbate coral bleaching under global climate change. Global Ecol. Biogeography 31, 1228–1236. doi: 10.1111/geb.13506
Kahng S., Garcia-Sais J., Spalding H., Brokovich E., Wagner D., Weil E., et al. (2010). Community ecology of mesophotic coral reef ecosystems. Coral Reefs 29, 255–275. doi: 10.1007/s00338-010-0593-6
Laverick J. H., Andradi-Brown D. A., Rogers A. D. (2017). Using light-dependent scleractinia to define the upper boundary of mesophotic coral ecosystems on the reefs of Utila, Honduras. PloS One 12, e0183075. doi: 10.1371/journal.pone.0183075
Le Gall L., Johnson J. V., Chequer A., Doherty M. L., Goodbody-Gringley G. (2024). Benthic and fish community composition on mesophotic reefs in Grand Cayman. PeerJ 12, e17763. doi: 10.7717/peerj.17763
Lesser M. P., Slattery M. (2011). Phase shift to algal dominated communities at mesophotic depths associated with lionfish (Pterois volitans) invasion on a Bahamian coral reef. Biol. Invasions 13, 1855–1868. doi: 10.1007/s10530-011-0005-z
Lesser M. P., Slattery M., Leichter J. J. (2009). Ecology of mesophotic coral reefs. J. Exp. Mar. Biol. Ecol. 375, 1–8. doi: 10.1016/j.jembe.2009.05.009
Lesser M. P., Slattery M., Mobley C. D. (2018). Biodiversity and functional ecology of mesophotic coral reefs. Annu. Rev. Ecology Evolution Systematics 49, 49–71. doi: 10.1146/annurev-ecolsys-110617-062423
Manfrino C., Jacoby C. A., Camp E., Frazer T. K. (2013). A positive trajectory for corals at little Cayman island. PLoS One 8, e75432. doi: 10.1371/journal.pone.0075432
Morato T., Hoyle S. D., Allain V., Nicol S. J. (2010). Seamounts are hotspots of pelagic biodiversity in the open ocean. Proc. Natl. Acad. Sci. 107, 9707–9711. doi: 10.1073/pnas.0910290107
Navarro-Martínez Z. M., Armenteros M., Espinosa L., Lake J. J., Apprill A. (2022). Taxonomic and functional assemblage structure of coral reef fishes from Jardines de la Reina (Caribbean Sea, Cuba). Mar. Ecol. Prog. Ser. 690, 113–132. doi: 10.3354/meps14049
Ogle D. H., Wheeler P., Dinno A. (2017). FSA: fisheries stock analysis. R package version 0.8, Vol. 17. 636.
Oksanen J., Blanchet F. G., Friendly M., Kindt R., Legendre P., McGlinn D., et al. (2020). vegan: Community Ecology Package. Available online at: https://CRAN.R-project.org/package=vegan (Accessed August 23, 2021).
Pérez-Castro M.Á., Schubert N., Ang-Montes-de-Oca G., Leyte-Morales G. E., Eyal G., Hinojosa-Arango G. (2022). Mesophotic Coral Ecosystems in the Eastern Tropical Pacific: The current state of knowledge and the spatial variability of their depth boundaries. Sci. Total Environ. 806, 150576. doi: 10.1016/j.scitotenv.2021.150576
Pinheiro H. T., Bernardi G., Simon T., Joyeux J.-C., Macieira R. M., Gasparini J. L., et al. (2017). Island biogeography of marine organisms. Nature 549, 82–85. doi: 10.1038/nature23680
Pinheiro H. T., Eyal G., Shepherd B., Rocha L. A. (2019). Ecological insights from environmental disturbances in mesophotic coral ecosystems. Ecosphere 10, e02666. doi: 10.1002/ecs2.2666
Pinheiro H. T., Ferreira C. E. L., Joyeux J.-C., Santos R. G., Horta P. A. (2011). Reef fish structure and distribution in a south-western Atlantic Ocean tropical island. J. Fish Biol. 79, 1984–2006. doi: 10.1111/j.1095-8649.2011.03138.x
Pinheiro H. T., Goodbody-Gringley G., Jessup M. E., Shepherd B., Chequer A. D., Rocha L. A. (2016). Upper and lower mesophotic coral reef fish communities evaluated by underwater visual censuses in two Caribbean locations. Coral Reefs 35, 139–151. doi: 10.1007/s00338-015-1381-0
Pinheiro H. T., MacDonald C., Quimbayo J. P., Shepherd B., Phelps T. A., Loss A. C., et al. (2023). Assembly rules of coral reef fish communities along the depth gradient. Curr. Biol. 33, 1421–1430.e4. doi: 10.1016/j.cub.2023.02.040
Pinheiro H. T., Mazzei E., Moura R. L., Amado-Filho G. M., Carvalho-Filho A., Braga A. C., et al. (2015). Fish biodiversity of the vitória-trindade seamount chain, Southwestern Atlantic: an updated database. PLoS One 10, e0118180. doi: 10.1371/journal.pone.0118180
Pyle R. L., Copus J. M. (2019). Mesophotic coral ecosystems: introduction and overview. Mesophotic coral Ecosyst., 3–27. doi: 10.1007/978-3-319-92735-0_1
R Core Team (2023). R: A language and environment for statistical computing. Available online at: https://www.R-project.org/.
Rocha L. A., Pinheiro H. T., Shepherd B., Papastamatiou Y. P., Luiz O. J., Pyle R. L., et al. (2018). Mesophotic coral ecosystems are threatened and ecologically distinct from shallow water reefs. Science 361, 281–284. doi: 10.1126/science.aaq1614
Rogers A. D. (2018). “Chapter four - the biology of seamounts: 25 years on,” in Advances in Marine Biology. Ed. Sheppard C. (Academic Press), 137–224. doi: 10.1016/bs.amb.2018.06.001
Sanchez P. J., Dance M. A., Kraus R. T., Hill R. L., Rooker J. R. (2023). Fish community characterization of mid-shelf and shelf- edge mesophotic coral ecosystems in the expanded Flower Garden Banks National Marine Sanctuary. Bull. Mar. Sci. 99, 41–50. doi: 10.5343/bms.2022.0014
Slattery M., Lesser M. P. (2019). The Bahamas and Cayman Islands. In: Loya Y., Puglise K., Bridge T. (eds) Mesophotic Coral Ecosystems. Coral Reefs of the World, vol 12. Springer, Cham. Available at: https://doi.org/10.1007/978-3-319-92735-0_3
Smith T. B., Glynn P. W., Maté J. L., Toth L. T., Gyory J. (2014). A depth refugium from catastrophic coral bleaching prevents regional extinction. Ecology 95, 1663–1673. doi: 10.1890/13-0468.1
Smith T. B., Gyory J., Brandt M. E., Miller W. J., Jossart J., Nemeth R. S. (2016). Caribbean mesophotic coral ecosystems are unlikely climate change refugia. Global Change Biol. 22, 2756–2765. doi: 10.1111/gcb.2016.22.issue-8
Soares M.de O., Tavares T. C. L., Carneiro P.B.de M. (2019). Mesophotic ecosystems: Distribution, impacts and conservation in the South Atlantic. Diversity Distributions 25, 255–268. doi: 10.1111/ddi.12846
Staudigel H., Clague D. A. (2010). The geological history of deep-sea volcanoes: Biosphere, hydrosphere, and lithosphere interactions. Oceanography 23, 58–71. doi: 10.5670/oceanog.2010.62
Stock B. C., Heppell S. A., Waterhouse L., Dove I. C., Pattengill-Semmens C. V., McCoy C. M., et al. (2021). Pulse recruitment and recovery of Cayman Islands Nassau Grouper (Epinephelus striatus) spawning aggregations revealed by in situ length-frequency data. ICES J. Mar. Sci. 78, 277–292. doi: 10.1093/icesjms/fsaa221
Torres (1991). Tabular data on marine fishes from Southern Africa. Part 1: Length-weight relationships. Fishbyte 9, 50–53.
Turner J. A., Andradi-Brown D. A., Gori A., Bongaerts P., Burdett H. L., Ferrier-Pagès C., et al. (2019). Key questions for research and conservation of mesophotic coral ecosystems and temperate mesophotic ecosystems. Mesophotic coral Ecosyst., 989–1003. doi: 10.1007/978-3-319-92735-0_52
Voss J., Williams M., Reed J., Clark R. (2014). “Benthic and fish communities in the mid and lower mesophotic zone of the sanctuary,” in Fish and benthic communities of the Flower Garden Banks National Marine Sanctuary: science to support sanctuary management. (Silver Spring, MD: NOAA Technical Memorandum NOS NCCOS 179), 179, 200.
Weber S. B., Richardson A. J., Thompson C. D., Brown J., Campanella F., Godley B. J., et al. (2025). Shallow seamounts are “oases” and activity hubs for pelagic predators in a large-scale marine reserve. PloS Biol. 23, e3003016. doi: 10.1371/journal.pbio.3003016
Whittaker R. J., Willis K. J., Field R. (2001). Scale and species richness: towards a general, hierarchical theory of species diversity. J. biogeography 28, 453–470. doi: 10.1046/j.1365-2699.2001.00563.x
Keywords: Caribbean, Cayman Islands, Cuba, coral reef, remote, offshore, rebreather, fisheries
Citation: Johnson JV, Chequer AD and Goodbody-Gringley G (2025) Depth partitioning of mesophotic reef fish communities on Pickle Bank seamount. Front. Mar. Sci. 12:1539066. doi: 10.3389/fmars.2025.1539066
Received: 03 December 2024; Accepted: 04 March 2025;
Published: 27 March 2025.
Edited by:
Tyler Burton Smith, University of the Virgin Islands, US Virgin IslandsReviewed by:
Savanna Barry, University of Florida, United StatesCopyright © 2025 Johnson, Chequer and Goodbody-Gringley. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jack V. Johnson, amFja3Zqb2huc29uQGhvdG1haWwuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.