
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Immunol. , 17 March 2025
Sec. Mucosal Immunity
Volume 16 - 2025 | https://doi.org/10.3389/fimmu.2025.1551445
This article is part of the Research Topic Natural Products and Intestinal Mucosal Immunity View all 7 articles
Immune checkpoint inhibitor-induced colitis (irColitis) is a common and severe adverse reaction to immune checkpoint inhibitors (ICIs), significantly impacting the treatment outcomes and quality of life of cancer patients. Epidemiological studies indicate that the incidence of irColitis is associated with factors such as the type of ICIs, the patient’s gender, age, and medical history. Although the exact pathophysiology remains unclear, irColitis is thought to be related to immune system activation and dysregulation, gut microbiota imbalance, and impaired epithelial barrier function. This review summarized the epidemiology, clinical presentation, diagnostic criteria, and pathogenesis of irColitis. Additionally, the standard and novel therapeutic strategies of irColitis, including corticosteroids, biologics, and gut microbiota interventions, more importantly the potential and application of Traditional Chinese Medicine (TCM). Future researches call for deeper mechanistic investigations, the development of biomarkers, and reveal the integration of TCM therapies within individual immunotherapy frameworks.
Immune checkpoint inhibitors (ICIs) have made significant advancements in cancer treatment, being widely used for non-small cell lung cancer (NSCLC), melanoma, gastric cancer, and other malignancies. This has transformed the treatment landscape for both solid tumors and hematological malignancies. The ICIs are associated with a range of immune-related adverse events (irAEs) that can affect multiple organs, including dermatological reactions, hepatitis, myocarditis, colitis, and others (1). Among these, immune checkpoint inhibitor-induced colitis (irColitis) is one of the most common and severe adverse effects. Patients with irColitis typically experience diarrhea, abdominal pain, and hematochezia, and even bowel perforation in severe cases. The irColitis significantly impacts patients’ quality of life, more seriously, causing drug dose reduction, discontinuation, or even death. The pathogenesis of irColitis remains unclear, but it is likely associated with immune system activation and dysregulation, gut microbiota imbalance, and impaired epithelial barrier function (2–4). Current treatments include corticosteroids and immunosuppressive agents; however, some patients exhibit inadequate responses or develop resistance, highlighting the urgent need for new therapeutic options for irColitis.
Under the principle of treatment based on syndrome differentiation, Traditional Chinese Medicine (TCM) has accumulated extensive experience in treating irAEs. For irColitis, TCM aims to restore the body’s balance and improve immune and intestinal functions (5). This holistic approach, along with syndrome differentiation and individualized treatment, have gradually demonstrated unique advantages.
This review aims to summarize the recent advances in the pathogenesis, clinical features, and TCM-based treatments of irColitis, while also exploring potential future research directions in this field. The goal is to provide new insights into clinical practice and offer reference points for future research endeavors.
Studies have demonstrated that the incidence of irColitis varies significantly among different patient populations. A systematic review and meta-analysis (6) revealed that the incidence of colitis in patients treated with anti-programmed cell death protein 1 (PD-1) antibodies alone was 2%. In contrast, the use of Cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4) inhibitors, such as ipilimumab, resulted in a notable increase in the incidence of colitis to 7%. In CheckMate 920 (7), the combination of nivolumab and ipilimumab led to an 18% incidence rate of all-grade colitis, with a 7% incidence rate of grade 3 or higher. As a consequence, one participant discontinued treatment. These findings suggested that the treatment modality of ICIs significantly influences the incidence of colitis. Other potential risk factors for irColitis have been identified in a cohort study of data from the Explorys, a US population database, indicating that irColitis occurred in 3.6% of patients. The study identified several risk factors associated with an increased likelihood of developing irColitis, including being female, white, older than 65 years, obese, and having a history of alcohol abuse (8).
Furthermore, a retrospective, comparative cohort study focused on the comorbid diseases associated with irColitis. This study showed patients with rheumatoid arthritis (RA) demonstrated a lower incidence of irColitis than those without RA, but the difference was not statistically significant(6 cases [7%] vs. 28 cases [14%]; p = 0.094) (9). This observed difference may be due to the distinct underlying pathological and immune mechanisms between irColitis and RA.
To characterize the epidemiology of irColitis, we reviewed clinical studies published on PubMed between 2020 and 2024 (10–49). The literature search used the keywords “Immune checkpoint inhibitor” and “colitis.” Clinical trials published in the past five years were selected based on the following inclusion criteria: (1) studies included more than 10 participants to ensure statistical robustness, minimized random variation, and reduced biases associated with small sample sizes or case reports; and (2) studies explicitly reported the incidence of irColitis to directly compare, minimizing consistency across different studies. These criteria were established to enhance the reliability of our findings and minimize data heterogeneity. The search results are presented in Table 1, including the year, country, sample size, drug type, and dosage (with treatment cycles and study endpoints detailed in the Supplementary Table), the incidence of irColitis (overall and grades 3–4), and the number of patients who discontinued treatment or died due to irColitis.
As shown in Table 1, these studies focused on the incidence of irColitis across various malignancies. Among the 41 included studies, 39 were prospective, including 28 phase I/II trials and 7 phase III/IV trials. These trials investigated various types of cancer, including melanoma, NSCLC, Renal Cell Carcinoma (RCC), Colorectal Cancer (CRC), Head and Neck Squamous Cell Carcinoma (HNSCC), gastroesophageal adenocarcinoma, classical Hodgkin lymphoma, cervical cancer, bladder cancer, soft tissue sarcoma, and anal cancer. The incidence of colitis varied significantly, ranging from 0 to 32%, with the rate of grade 3 or higher events varying between 0 and 21%.
These differences were primarily influenced by tumor type, the immunotherapy drugs and doses used, treatment frequency, and patient characteristics. Regarding the relationship between immunotherapy drug types and the incidence of irColitis, our findings aligned with previous literature. Compared to treatment with a single drug, the incidence of irColitis was significantly higher in combination therapies. For example, the incidence of irColitis reached 18% in patients treated with nivolumab and ipilimumab (3 mg/kg + 1 mg/kg) (25). In contrast, monotherapy with nivolumab at 3 mg/kg showed a lower incidence of 0.9% compared to ICIs combination therapy (49). Additionally, high-dose and high-frequency application of ICIs appeared to be associated with an increased incidence of irColitis. For instance, pembrolizumab at 10 mg/kg every 2 weeks resulted in an overall irColitis incidence of 11.9% (35), whereas fixed-dose pembrolizumab at 200 mg every 3 weeks led to an overall irColitis incidence of 2%-9.3% (19, 40). Furthermore, patients with different tumor types had varying prevalences of irColitis. For example, in CRC, the incidence of irColitis with pembrolizumab was 5.5%-7% (10, 47), whereas a much lower incidence of 2% in bladder cancer (19). Age was also found to be related to the occurrence of irColitis. In patients with a median age of 13.9 years old, the incidence of irColitis was 27.3% (37), a higher incidence than that in elderly patients (aged over 55 years), ranging from 0.9% to 32% (48, 49). In contrast, the incidence was lower in middle-aged population (30-55 years), ranging from 0% to 12% (12, 17, 18, 44). However, Necchi found age may not be the primary factor compared to other factors (19); in this study, the median age of patients was 72 years old, but the incidence of irColitis reported 2%.
As shown in Table 1, 13 of 41 studies reported discontinuation of immunotherapy due to irColitis (7, 10, 18, 19, 21, 23–25, 28, 37, 39, 42, 49). The ICIs used in these studies included single-agent pembrolizumab, tremelimumab, durvalumab, retifanlimab, tislelizumab, nivolumab, and ipilimumab, as well as combinations of nivolumab with ipilimumab, and pacmilimab with ipilimumab. Cacciotti’s study observed the highest discontinuation rate at 18.2% (37); this study focused on children and young adults with recurrent pediatric high-grade central nervous system (CNS) tumors, which were treated with ipilimumab, nivolumab, and/or pembrolizumab. The high discontinuation rate may be attributed to the combined effects of tumor malignancy, patient age, and the use of combination immunotherapy. Additionally, two studies reported a 6% discontinuation rate due to irColitis (18, 28). Three studies reported irColitis-related deaths. One case involved a female patient with metastatic cervical cancer, who received nivolumab (1 mg/kg) and ipilimumab (3 mg/kg) every 3 weeks for 4 cycles, followed by nivolumab (240 mg) every 2 weeks (17). Another case involved a patient with advanced melanoma treated with ipilimumab (34). Additionally, two cases involved females with receptor-positive, ERBB2-negative metastatic breast cancer, who died of eribulin and pembrolizumab-induced irColitis due to sepsis (41).
However, we acknowledge the potential for selection bias in our review. Focusing on published clinical trials, we have inadvertently excluded smaller retrospective studies and case reports, potentially underestimating rare presentations of irColitis. Additionally, publication bias may have influenced the reported incidence rates. To mitigate these limitations, we conducted a comprehensive keyword search and systematically reviewed the reference lists of relevant articles.
IrColiti typically manifests as diarrhea, the primary clinical symptom. Diarrhea has been found to occur early during immunotherapy. The median onset of diarrhea/colitis typically occurs around 3 to 6 months after treatment with anti-PD-1/Programmed cell death-ligand 1 (PD-L1) agents, while earlier for anti-CTLA-4 agents at around 6 to 8 weeks (50, 51). However, there are also cases of relapse occurring 1-2 years after treatment discontinuation (52). Patients often experience frequent bowel movements, with stools containing mucus or blood. Symptom severity ranges from mild to severe and may significantly impact the patient’s quality of life. Besides diarrhea, patients with irColitis frequently report other gastrointestinal symptoms, including abdominal pain, fever, and, in some cases, upper gastrointestinal symptoms such as dyspepsia, reflux, or heartburn (53, 54).
In addition to the typical symptoms of diarrhea and abdominal pain, irColitis may present with atypical manifestations, including anorexia, significant weight loss or dehydration due to prolonged diarrhea, and fatigue. These systemic symptoms further complicate the clinical management of irColitis (55).
These clinical manifestations may resemble those of other gastrointestinal disorders, making it essential to differentiate irColitis from infections, inflammatory bowel disease (IBD), or ischemic colitis. A careful differential diagnosis is required for proper treatment strategies.
The pathological changes associated with irColitis are diverse and complex. Although the condition is typically characterized by pancolitis, irColitis may occasionally affect only the descending colon (56, 57). These changes not only influence clinical diagnosis but also have significant implications for treatment strategies and patient prognosis. Common endoscopic findings included erythema, loss of vascular pattern, granular appearance, and ulcerations; however, normal mucosa might also be observed in some cases (57, 58). Therefore, a biopsy has been recommended for suspect irColitis patients.
According to existing literature, the pathological changes associated with irColitis could be classified into several types: (1) Active colitis is the most common histopathological pattern of irColitis. This form is characterized by neutrophil infiltration in the lamina propria, cryptitis, and crypt abscesses. Crypt atrophy or loss was uncommon but could be found Increased epithelial apoptosis and intraepithelial lymphocytes could also be observed (57); (2) Chronic active colitis is commonly observed after long-term ICI treatment. Histopathological findings include persistent lymphocytic infiltration and disruption of intestinal architecture, such as crypt distortion and pseudopyloric or Paneth cell metaplasia (57). Crypt abscesses and ulceration of the intestinal epithelium may also be present (59). (3) Microscopic colitis often appears normal on endoscopy, making it difficult to detect. Histopathological changes do not show significant acute or active inflammation. However, histological examination may reveal increased intraepithelial lymphocytes and lymphoplasmacytic infiltration in the lamina propria (57, 60). Microscopic colitis is potentially required for additional treatment with oral or intravenous corticosteroids and/or nonsteroidal immunosuppressive agents. In addition to these three common types, other patterns, such as increased apoptosis (61), ischemic colitis (58), and nonspecific inflammatory reactive changes (57), have also been observed.
The diagnosis of irColitis primarily depends on clinical manifestations, laboratory tests, imaging studies, and endoscopic evaluation, making it a complex process that integrates various standards and methodologies. Currently, several grading systems are employed to assess clinical symptoms, including the Common Terminology Criteria for Adverse Events (CTCAE) (62), the treatment guidelines for irAEs from the European Society for Medical Oncology (ESMO) (63), the American Society of Clinical Oncology (ASCO) (64), and Chinese Society of Clinical Oncology (CSCO) Guidelines for the management of toxicities related to ICIs (65) (Table 2). However, the ESMO/ASCO/CSCO guidelines do not address irColitis separately; instead, they group irColitis and diarrhea together for grading and management.
Endoscopic examination plays a crucial role in diagnosing irColitis as the golden standard (66, 67). However, due to the similar symptoms between irColitis and other gastrointestinal disorders, additional diagnostic tests are essential for differentiating diagnosis. For example, laboratory blood tests and stool analyses can help differentiate irColitis from infections and IBD. The imaging modalities of CT and MRI are more valuable in evaluating the extent and severity of intestinal inflammation, as well as in managing complex cases of irColitis that may pose risks for endoscopic procedures (66).
Beyond conventional diagnostic methods, recent advancements in irColitis biomarkers have shown promise for predicting and diagnosing its occurrence. Anti-integrin αvβ6, a heterodimeric cell adhesion receptor, has been identified as a potential biomarker. A study revealed that Anti-integrin αvβ6 autoantibodies were significantly more prevalent in irColitis patients than in control groups, including patients with other organ irAEs, cancer patients without irAEs, and healthy volunteers (30.8% vs. 1.9%) (68). Furthermore, the presence of Anti-integrin αvβ6 autoantibodies was associated with disease activity, supported by characteristic endoscopic findings, high-grade irColitis, and steroid resistance (68). A single-cell sequencing study of 13 irColitis patients revealed that increased mucosal Regulatory T cells (Tregs), ITGAEHi CD8 tissue-resident memory T cells (TRMs) expressing C-X-C motif chemokine ligand 13 (CXCL13) and T helper 17 (Th17) gene programs, and recirculating ITGB2Hi CD8 T cells may serve as potential biomarkers for irColitis (69). Another study involving 15 irColitis patients showed that activated CD8+ TRMs cells express high levels of transcripts for checkpoint inhibitors and interferon-gamma and identified Interferon-Gamma-Producing CD8+ TRMs as potential targets (3).
Recent advances in research have illuminated key pathological processes, such as T cell overactivation, pro-inflammatory cytokine production, gut microbiota dysbiosis, and epithelial barrier dysfunction, providing novel insights into irColitis pathogenesis (Table 3, Figure 1).
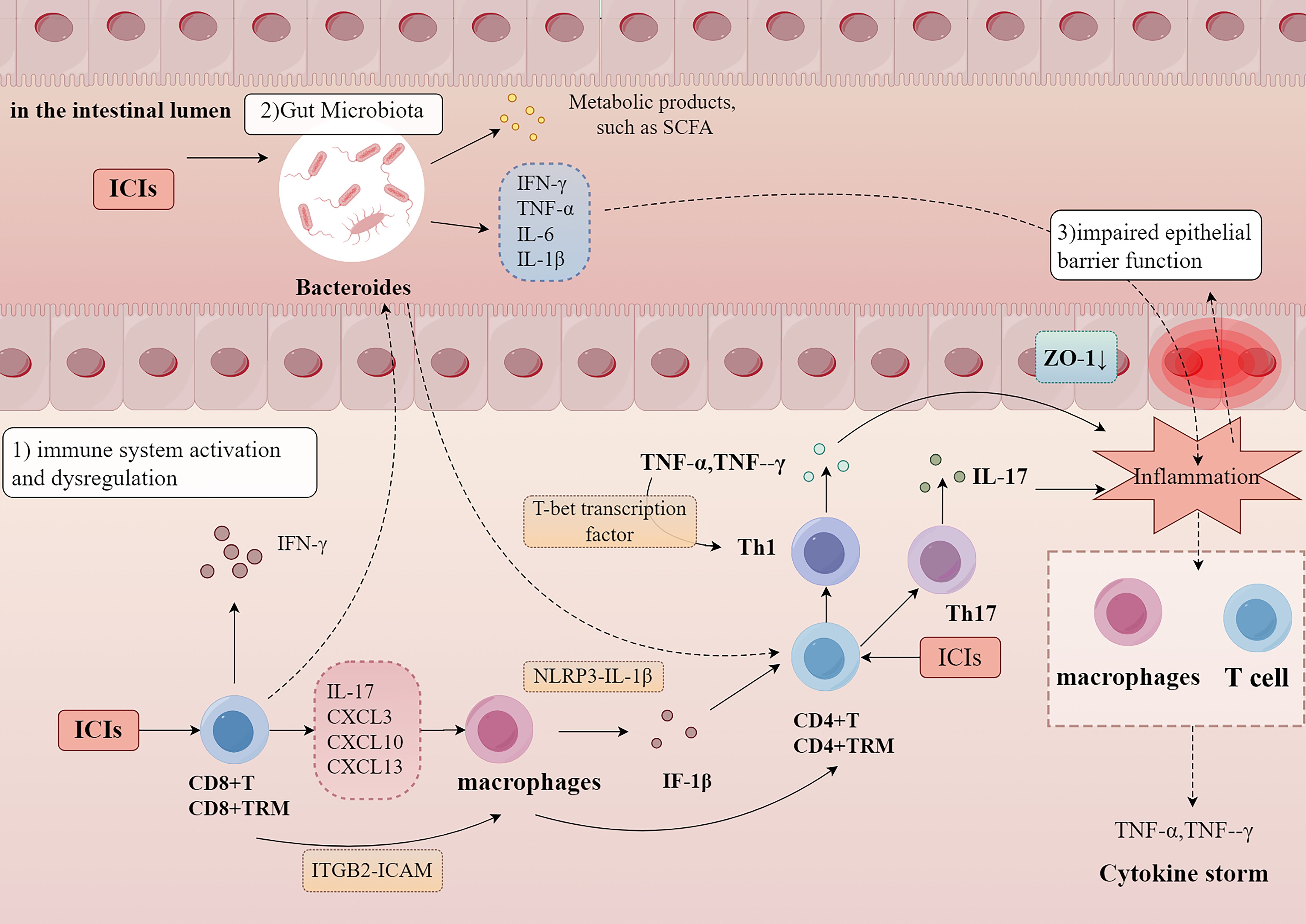
Figure 1. The possible mechanism of irColitis. ICIs induce immune system activation and dysregulation, resulting in excessive T cell and macrophage activation, along with increased pro-inflammatory cytokine release. Gut microbiota, particularly Bacteroides, contribute to inflammation by producing metabolic byproducts and modulating cytokine expression. Additionally, epithelial barrier dysfunction, characterized by reduced ZO-1 expression, further amplifies inflammation, ultimately leading to immune dysregulation and a cytokine storm. (TRM, tissue-resident memory T cells; SCFA, Short-Chain Fatty Acids; ZO-1, Zonula Occludens-1; IFN, Interferon; TH, T helper cells; IL, Interleukin; TNF, Tumor Necrosis Factor. Detailed abbreviations are presented in Supplementary Table 2. Image created with Figdraw.com).
By blocking inhibitory PD-1/CTLA-4 pathways, ICIs activate inhibited T cells thereby enhancing the immune system’s ability to attack tumor cells; however, this activation can cause T cell overactivation, resulting in the loss of immune tolerance, particularly in mucosal tissues, which is a key driver of irColitis (75).
Substantial evidence supports that CD8+ T cells are the key effector cells in irColitis (2). A cross-sectional study of clinical and pathological data analyses from patients with anti-CTLA-4/PD-1 colitis demonstrated that CD8+TRMs constituted the majority of activated TRMs in irColitis, with the degree of activation correlating with clinical and endoscopic severity. Notably, these CD8+ TRMs exhibited significantly higher expression of Interferon-gamma (IFN-γ), which further promoted TRMs activation. This finding was complemented by evidence of upregulation of classic Janus kinase/Signal transducer and activator of transcription (JAK/STAT) components in the IFN-γ signaling pathway in irColitis (3). Collectively, the upregulation of IFN-γ signaling represents a key pathological feature of irColitis, with the JAK/STAT signaling pathway playing a central role in this process, thereby providing a theoretical basis for potential therapeutic strategies, such as JAK inhibitors (74).
Thomas et al. applied single-cell multi-omics to analyze approximately 300,000 cells from the colonic mucosa and blood samples of patients with irColitis. Their findings revealed that the expansion of mucosal Tregs, CD8+ TRMs, and recirculating CD8+ T cells are hallmark features of irColitis, particularly T cells expressing pro-inflammatory gene programs, such as CXCL13 and Th17. In patients with irColitis, 1.9% of CD8+ T cells expressed Interleukin-17 (IL-17) A, while IFN-γ and CXCL13 were significantly upregulated in intestinal tissue samples (69). These cytokines synergistically impair epithelial integrity and drive chronic inflammation, further elucidating the mechanisms underlying irColitis. CD4+ T cells can differentiate into various phenotypes, including T helper 1 (Th1), Th17, and Treg cells. Th1 cells primarily secrete IFN-γ, while Th17 cells produce cytokines such as IL-17, which is closely associated with the pathogenesis of intestinal inflammation. Numerous studies have indicated that the overactivation of CD4+ T cells contributes to the development and progression of irColitis (78). To further elucidate the roles of CD4+ T cells in irColitis, Lo et al. established an irColitis mouse model using anti-CTLA-4 treatment. Their findings demonstrated that CTLA-4 blockade significantly promotes the accumulation of IFN-γ+ CD4+ T cells, resulting in intestinal tissue damage, which is considered the initial trigger of irColitis (71). Additionally, they observed that IFN-γ expression was closely linked to the activation of the T-bet transcription factor, which amplified the Th1 response and underscores the Th1-biased inflammatory response in irColitis. Furthermore, a recent study analyzing colon biopsies and blood samples from irColitis patients revealed that CD4+ TRMs were progenitors of cytotoxic effectors such as CD8+ T cells, thereby identifying CD4+ TRMs as novel therapeutic targets (75).
Interactions among immune cells play a pivotal role in disease pathogenesis when irColitis occur. ICIs relieve immune suppression, thereby altering the crosstalk among various immune cell populations within the intestines. These interactions notably include those between T cells and macrophages, dendritic cells, and epithelial cells.
Recent research has shown that activated CD8+ T cells interact with macrophages through C-X-C Chemokine Receptor Type 3 (CXCR3) and macrophage-derived C-X-C Motif Chemokine Ligand 10 (CXCL10), as well as Integrin beta 2-Intercellular Adhesion Molecule (ITGB2-ICAM) pathways. These interactions facilitated macrophage recruitment and activation, initiating localized inflammatory responses in the intestine (55, 69). To further investigate this mechanism, Ankit et al. created an immunodeficient mouse model. Their results demonstrated that conditional deletion of Major Histocompatibility Complex class II (MHC-II) in macrophages reduced the proportion of Granulocyte-macrophage colony-stimulating factor (GM-CSF) + CD4+ T cells by 70%, from 18.3% to 5.5%. This suggests that macrophages present antigens via MHC-II and release Interleukin-1 beta (IL-1β), which promotes the differentiation of CD4+ T cells into GM-CSF+ subsets, contributing to irColitis. Moreover, the study revealed that inhibiting the Nucleotide-binding oligomerization domain (NOD)-like receptor family pyrin domain containing 3 (NLRP3) inflammasome significantly reduced IL-1β production in macrophages, which in turn decreased the population of GM-CSF+ CD4+ T cells (70). These findings underscored the role of macrophages in regulating T cell activation through the NLRP3-IL-1β axis, offering potential therapeutic targets for irColitis.
The interaction between T cells and epithelial cells plays a crucial role in the pathogenesis of irColitis. Epithelial cells enhance the pro-inflammatory activity of CD4+ T cells via MHC-II-mediated antigen presentation. Moreover, the absence of IFN-γ signaling in epithelial cells exacerbates the pathological activation of CD4+ T cells (70). Elevated expression of PD-L1 in intestinal epithelial cells has also been observed; however, this pathway becomes dysregulated in the presence of ICIs, leading to uncontrolled T cell cytotoxic responses. This dysregulation indicates a loss of immune tolerance in patients with irColitis (79).
Recent metabolomics evidence has shown that dysbiosis-induced alterations in lipid and amino acid metabolism contribute to gut inflammation in irColitis.
Ye et al. suggested that certain microbial species, particularly within the Bacteroides genus, exacerbated irColitis through pro-inflammatory effects. Positive correlations between bacteroides and inflammatory factors, such as tumor necrosis factor alpha (TNF-α), interleukin (IL)-6, and IL-1β, highlighted the interactions between microbiota, metabolites, and inflammation. Additionally, gut microbiota dysbiosis intensified TNF-α signaling, further disrupting microbial composition and metabolite production, thereby aggravating irColitis (77).
Activated CD8+ TRMs may contribute to the upregulation of IFN-γ signaling and the development of irColitis by mediating immune responses to both commensal or pathogenic microbes. This suggests that alterations in the microbiome play a critical role in the pathogenesis of irColitis. Therapeutic strategies targeting the gut microbiome, such as fecal microbiota transplantation (FMT), may provide a promising approach to mitigate this condition (3).
The microbiome plays a critical role in the activation of CD4+ T cells and the onset of colitis. Severe intestinal inflammation induced by anti-CTLA-4 treatment occurs only in mice with a fully functional, free-living microbiome. This suggests that the composition of the gut microbiota not only influences CD4+ T cell activation but may also exacerbate inflammatory responses through interactions with the immune system (71).
In conclusion, we find that gut microbiota dysbiosis both drives and results from irColitis. Dysbiosis amplifies local inflammation through pro-inflammatory species and disrupted metabolic pathways, establishing a vicious cycle.
The intestinal epithelial barrier is essential for preventing antigen transfer, and its damage is considered one of the key pathological features of irColitis.
As discussed earlier, the enrichment of pro-inflammatory cytokines, such as IFN-γ and TNF-α, is observed in irColitis. Previous evidence supported that these cytokines could increase epithelial cell apoptosis, thereby impairing the intestinal epithelial barrier and contributing to bowel inflammation, a key factor in the development of irColitis (72). Epithelial defects, including upregulation of apoptosis-related genes (Caspase-1 [CASP1], Caspase-8 [CASP8]), interferon-stimulated genes (STAT1, PD-L1), and pro-inflammatory marker genes (CXCL10), as well as downregulation of aquaporin water channel genes, suggest impaired water and solute transport in the intestines (69). Disruption of tight junction proteins, such as myosin light chain kinase (MLCK) and zonula occludens-1 (ZO-1), increased intestinal permeability and epithelial barrier dysfunction, allowing microbial products to activate immune cells, including T cells and macrophages. This triggered a cytokine storm involving TNF-α and IFN-γ, and further amplified inflammation in irColitis (80).
Interventions targeting the epithelial barrier, such as MLCK inhibitors, show promise in reducing inflammation while preserving the efficacy of tumor immunotherapy. This strategy may effectively manage irColitis by restoring barrier integrity.
Epithelial remodeling, a key feature of irColitis, also represents the precursor to intestinal reconstruction. A clinical study highlighted significant structural alterations in the intestine during irColitis, including epithelial barrier disruption, crypt architectural distortion, and increased immune cell infiltration (4). These changes compromise intestinal integrity, allowing microbial antigens to reach immune cells, which perpetuates inflammation. Chronic inflammation is linked to abnormal epithelial regeneration and the loss of goblet cells, further impairing mucosal defense.
At the cellular and molecular level, evidence suggested that interferon signaling induces changes in epithelial differentiation, leading to a decrease in LGR5Hi stem cells and an increase in transit-amplifying cells, along with the loss of mature epithelial cells and enrichment of interferon-induced epithelial subsets. Additionally, some cases of irColitis feature vascular and fibroblast remodeling, potentially driven by the expansion of angiogenesis-related genes (Hypoxia-inducible factor 1 alpha [HIF1A], Vascular Endothelial Growth Factor A [VEGFA]), highlighting the potential role of neovascularization in the pathological process of irColitis (69, 81).
The recommended treatment of irColitis primarily included corticosteroids, biologic agents, and certain non-pharmacological interventions. Moreover, TCM, as a natural therapeutic approach, has shown a promising potential in the treatment of this condition.
Corticosteroids are the first-line treatment for irColitis, primarily used to suppress T cell activation, thereby inhibiting the production of pro-inflammatory cytokines and regulating overactive innate and adaptive immune responses (82). The NCCN guidelines recommend that patients experiencing grade 2 or higher irColitis should discontinue immunotherapy and receive prednisone or methylprednisolone (1–2 mg/kg/day) until symptoms improve to grade 1 or lower. Corticosteroid tapering should then be performed over 4 to 6 weeks (83). However, some patients may develop resistance to corticosteroids or exhibit poor responses, with relapses occurring during dose reduction, complicating further management (82, 84). A retrospective study reported a clinical response rate of 70% (14/20) to corticosteroid treatment. Among these 14 patients, 2 (14%) experienced relapse during corticosteroid tapering and became steroid-dependent (85). Another retrospective analysis of 49 irColitis patients found that all received glucocorticoids as first-line treatment; however, immunotherapy was discontinued in 21 cases (86). In such cases, it is essential to promptly assess the patient’s clinical condition, differentiate irColitis from other gastrointestinal disorders, and identify potential complications. Alternative therapeutic options should then be considered based on this evaluation.
For patients with corticosteroid resistance or inadequate response, biologic agents are an important therapeutic option. Recent studies have shown that anti-TNF-α monoclonal antibody infliximab and α4β7 integrin immunoglobulin G1 (IgG1) monoclonal antibody vedolizumab exhibited promising efficacy in the treatment of irColitis (82, 83). If no improvement is observed within 2 to 3 days after initiating corticosteroid therapy, infliximab or vedolizumab may be considered within 2 weeks of diarrhea onset while continuing corticosteroid treatment (83). Additionally, patients who relapse during corticosteroid tapering or after completing the corticosteroid regimen may also require additional immunosuppressive therapy (67). Infliximab, a second-line treatment, is widely used in corticosteroid-resistant patients. Clinical data demonstrate its effectiveness in severe acute colitis cases, with a retrospective study reporting a 71.4% remission rate for corticosteroid-resistant irColitis (87). Furthermore, infliximab has been shown to help maintain overall survival (OS) in these patients (88). A study comparing infliximab with corticosteroids demonstrated that infliximab resulted in a shorter median time to diarrhea resolution and corticosteroid tapering compared to the corticosteroid group without negatively affecting OS or time to treatment failure (TTF) (89). Another biologic agent, vedolizumab, is commonly administered intravenously at a dose of 300 mg (90). Although vedolizumab had not been as extensively studied in clinical applications as infliximab; a multicenter retrospective study demonstrated that vedolizumab treatment for corticosteroid-resistant irColitis resulted in sustained clinical remission in 84% of patients (90).
The gut microbiota plays a crucial role in regulating intestinal immune function. One study found that Faecalibacterium prausnitzii helped reduce intestinal toxicity and boost tumor immunity, enhancing the effectiveness of dual CTLA-4 and PD-1 checkpoint blockade (91). This suggests that supplementary probiotics could potentially reduce the risk of colitis while improving the response to immunotherapy. FMT is an emerging therapeutic approach that has been applied to patients with refractory irColitis. A study involving 12 patients with severe irColitis showed that FMT effectively improved clinical symptoms, with 92% of patients achieving clinical remission after treatment (74). This effect may be mediated through FMT’s influence on gut microbiota diversity and composition. Additionally, a significant reduction in total lymphocytes and CD8+ T cells was observed in complete responders, indicating that FMT mediates inflammation reduction in these patients (74).
TCM has a long and rich history in treating digestive disease, with extensive experience in treating irAEs. It also offers valuable insights into the treatment of irColitis. TCM views the human body as an integrated whole. When the balance of Yin and Yang and the normal function of the internal organs are maintained, as described in the Huangdi Neijing (Yellow Emperor’s Inner Canon), “When Yin is in balance and Yang is hidden, the spirit is healthy,” and “When the vital Qi is preserved within, pathogens cannot invade.” Immunotherapy with ICIs can disrupt this balance while targeting tumors, leading to irAEs. In TCM, irColitis is often classified under categories such as “diarrhea (Xie Xie)” and “intestinal dysentery (Chang Pi).” The occurrence of irColitis is believed to be closely related to pathogenic heat, damp-heat, damage to the intestinal collateral, and spleen-stomach weakness. As the disease progresses, patients may also develop symptoms of Spleen and Kidney Yang deficiency (5). The treatment principles in TCM involve clearing heat and detoxifying, strengthening the spleen and resolving dampness, and warming and tonifying the spleen and kidneys to regulate immune function, balance the internal organs, alleviate symptoms, and promote intestinal recovery. Additionally, TCM offers various methods for treating “diarrhea,” including oral administration, moxibustion, acupoint application, and enemas (5). The rich experience in treating “diarrhea” in TCM can provide valuable insights for future experimental studies and clinical trials on natural product therapies for irColitis.
Since no specific studies have investigated the use of TCM for irColitis, we could retrieved related studies based TCM theory of “treating different diseases with same method”. In TCM, ulcerative colitis, Crohn’s disease, and irColitis share similar symptoms, including diarrhea, abdominal pain, and rectal bleeding. These conditions are classified under the categories of “diarrhea” or “intestinal dysentery”. Therefore, this study conducted a comprehensive review of clinical articles on TCM treatments for diarrhea-related diseases from databases of PubMed and CNKI. This review provides valuable insights for future research (Table 4). The PubMed search utilized the following keywords and MeSH terms: (“Traditional Chinese Medicine” OR “Chinese herbal medicine” OR “Chinese medicine”) AND (“ulcerative colitis” OR “Crohn’s disease” OR “ colitis “), focusing on clinical studies published within the past five years. A similar search strategy was applied to Chinese databases. The inclusion criteria were as follows: (1) studies reported clinical outcomes of TCM in the treatment of irColitis or IBD; (2) studies had a standard research design. Studies were excluded if they lacked specific outcome measures, had an unclear study design, or involved insufficient sample sizes that could compromise statistical validity. To minimize selection bias, representative studies from both English and Chinese databases were included, with a preference for those demonstrating high methodological rigor. However, potential selection bias remains due to publication bias and variations in study design, which is recognized as a limitation.
Fifty articles focused on treatments such as oral Chinese medicine, bamboo scraping therapy, acupoint application, acupuncture, and moxibustion. Due to the limited number of irColitis cases, conducting large-scale clinical trials remains challenging. Therefore, as indicated in Table 4, most of the reviewed studies explore herbal formulas for the treatment of ulcerative colitis and Crohn’s disease. However, a study has explored the use of Chinese patent medicine (105). Given its convenience and cost-effectiveness, further research in this area is warranted.
In TCM, acute inflammatory symptoms are typically associated with Damp-Heat Syndrome. Treatment focuses on clearing heat, detoxifying, and drying dampness, using classic formulas such as Gegen Qinlian decoction, Baitouweng decoction, and Shao Yao decoction. Frequently used herbs include Phellodendri Cortex (Huang Bai), Coptidis Rhizoma (Huang Lian), Scutellariae Radix (Huang Qin), and Rhei Radix et Rhizoma (Da Huang) for their heat-clearing, damp-drying, and detoxifying properties. In TCM theory, it is commonly believed that the patient enters a stage of “deficiency” (Zheng Xu), then treatment focused on tonifying the spleen and stomach for improving patient’s condition. Additionally, TCM emphasizes supporting the body’s vital energy (Zheng Qi) with herbs such as Atractylodis Macrocephalae Rhizoma (Bai Zhu), Glycyrrhizae Radix et Rhizoma (Gan Cao), and Dioscoreae Rhizoma (Shan Yao).
The widespread use of ICIs in oncology has led to increasing clinical and research interest in irColitis. Although increasing findings on irColitis published (8), studies on epidemiological characteristics and underlying mechanisms remain insufficient, needing future researches.
The exploration of mechanisms on the effect Chinese medicine and integrative therapies for irColitis remains limited. Some studies demonstrated the potential interactions between TCM and ICIs, as well as the underlying mechanisms of integrated TCM and Western medicine in the treatment of irColitis. A meta-analysis evaluating the efficacy, safety, and potential mechanisms of TCM as an adjuvant therapy in cancer immunotherapy indicated that TCM influenced PD-1/PD-L1 inhibitors through tumor microenvironment modulation, gut microbiota regulation, inhibition of PD-1 or PD-L1 expression, and cytokine signaling regulation (107). Previous studies on inflammatory bowel diseases IBD with similar symptoms, such as Crohn’s disease and ulcerative colitis, suggested that Chinese medicine might alleviate irColitis through mechanisms including immune regulation, reduction of intestinal inflammation, modulation of gut microbiota, and protection of the intestinal mucosal barrier (2). This highlights the great potential of integrative traditional Chinese and Western medicine in treatment.
For example, the JAK/STAT and NF-κB signaling pathways may serve as important potential targets. PD-1/PD-L1 expression is regulated by JAK/STAT and NF-κB signaling pathways, both of which are targeted by TCM like Baicalin and Gegen Qinlian decoction (108–110). Experimental studies have demonstrated that Baicalin is an effective treatment for IBD. By restoring the Th17/Treg balance through the JAK/STAT signaling pathway and reducing ZO-1 secretion, Baicalin alleviates intestinal inflammation and preserves the integrity of the intestinal epithelial barrier, thereby improving clinical symptoms (108). This signaling pathway is particularly important during the acute phase of irColitis. On the other hand, research suggested that the inhibition of Toll-like receptor 4/Nuclear factor kappa B (TLR4/NF-κB) signaling and the enhancement of gut microbiota abundance might be key mechanisms the therapeutic efficacy of Gegen Qinlian decoction as well as other TCM decoctions in the treatment of IBD (109, 110). Additionally, traditional herbal medicines, such as Sophora flavescens Aiton, P. grandiflorus, and Kuijieling decoction, have also shown significant effects in restoring gut immune function. These herbs achieve this by suppressing excessive T cell activation and rebalancing Th17/Treg (108, 111, 112). Research on P. grandiflorus in the treatment of IBD has revealed that this herbal remedy modulates the homeostasis of colonic immune cells through the mesenteric lymphatic circulation, offering valuable insights for future research directions (113).
Meanwhile, TCM may exert its effects through different targets depending on the progression of irColitis. In the acute phase of irColitis, characterized by severe inflammation and epithelial barrier disruption, TCM interventions targeting rapid immune modulation and inflammation control may be particularly beneficial. For example, Baicalin and Gegen Qinlian decoction have been shown to effectively reduce severe acute bowel inflammation through JAK/STAT and TLR4/NF-κB signaling passageway, regulating the secretion of pro-inflammatory cytokine such as IL-17, IL-6, and IL-1β (108–110). When combined with corticosteroids, they may help achieve faster symptom control. Additionally, the multi-targeted mechanisms of TCM can alleviate the side effects associated with corticosteroid use, thereby improving patient tolerance and adherence to treatment.
The gut microbiome plays a critical role in the pathogenesis of irColitis and represents a promising area for further research. In chronic irColitis, where immune dysregulation and microbiota imbalance persist, TCM’s long-term regulatory effects on gut microbiota and immune homeostasis may be advantageous. Existing studies have demonstrated that various herbal medicines possess the ability to improve gut microbiota abundance (2). While current studies suggested that gut microbiota significantly influences the development of irColitis (3, 71, 77), future investigations should focus on elucidating the complex interactions between ICIs, gut immune function, and the microbiome. A deeper understanding of these interactions is essential for advancing prevention and treatment strategies and clarifying the mechanisms by which Chinese medicine affects chronic irColitis.
Currently, the treatment of irColitis primarily relies on corticosteroids and immunosuppressants such as Infliximab and Vedolizumab. However, some patients show inadequate responses to conventional treatments, and there is even the emergence of drug resistance. Therefore, future research should focus on exploring new therapeutic strategies.
In the treatment of irColitis, TCM emphasizes restoring balance within the body, particularly by normalizing the immune system and gut function. TCM theories, including those related to the gut microbiome, spleen and stomach function, and damp-heat, offer valuable insights for treating irColitis. This approach holds significant potential for future therapeutic strategies. Moreover, TCM emphasizes individualized treatment, tailoring therapeutic strategy according to each patient’s unique symptoms and physical constitution. In the future, combining individualized TCM approaches could enhance the therapeutic efficacy of irColitis.
The integration of TCM and Western medicine holds significant potential for the treatment of irColitis. During the acute phase, irColitis is often characterized by symptoms such as diarrhea, mucus or bloody stools, and abdominal pain. In severe cases, patients may experience serious complications such as intestinal perforation and sepsis, necessitating rapid inflammation control. Western medicine primarily focuses on immediate immunosuppression through glucocorticoids and biologics. Concurrently, anti-inflammatory Chinese herbal medicines, such as Scutellariae Radix (Huang Qin), Coptidis Rhizoma (Huang Lian), and Rhizoma Sinensis (Bai Jiang Cao), may be used in combination (93). Additionally, spleen-tonifying herbs like Codonopsis Radix (Dang Shen)and Atractylodis Macrocephalae Rhizoma (Bai Zhu) may help protect the intestinal barrier (97). Future research should further explore the role of integrative therapy in reducing glucocorticoid dosage, shortening treatment duration, and mitigating drug resistance during the acute phase. In the chronic phase or in cases of prolonged disease due to repeated ICI treatment, Western medicine offers limited strategies. At this stage, TCM presents a promising approach, interpreting the condition as an underlying deficiency with excess manifestations (Ben Xu Biao Shi). TCM emphasizes strengthening and protecting the spleen and stomach, often employing formulas that tonify the spleen and dry dampness, with individualized modifications. Studies have shown that Shenling Baizhu San improves colitis by enhancing intestinal epithelial barrier integrity and reducing inflammation (114). Additionally, a meta-analysis suggested that it alleviates diarrhea symptoms by enhancing immune function (115). Therefore, integrating Chinese medicine into standard Western treatment, tailored to the patient’s specific symptoms at different stages of irColitis represents a highly promising therapeutic approach. Previous studies on ulcerative colitis have demonstrated that integrative treatment combining TCM and Western medicine yields superior efficacy compared to either treatment alone (104, 106).
However, clinical evidence supporting the use of TCM or integrative approaches for irColitis remains limited. Current clinical practices mainly follow the “treating different diseases with similar symptoms (Yi Bing Tong Zhi)” principle, often based on treatment strategies for diseases with analogous symptoms. Therefore, further high-quality clinical trials are required. Future research should focus on evaluating the clinical efficacy of TCM in treating irColitis, assessing its safety, and exploring its synergistic effects with corticosteroids and immunosuppressants. Multicenter, large-scale, randomized controlled, double-blind trials will be essential to verify the therapeutic effects of TCM in irColitis and provide scientific evidence for its broader clinical application.
HD: Conceptualization, Data curation, Writing – original draft. YP: Conceptualization, Writing – original draft, Writing – review & editing. XW: Writing – original draft. HC: Resources, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was funded by National High Level Hospital Clinical Research Funding (2022-NHLHCRF-LX-02-0111), and China-Japan Friendship Hospital.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer HL declared a shared parent affiliation with the authors to the handling editor at the time of review.
The author(s) declare that Generative AI was used in the creation of this manuscript. Generative AI was used for language polishing to enhance the clarity and readability of the text.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1551445/full#supplementary-material
1. Wang SJ, Dougan SK, Dougan M. Immune mechanisms of toxicity from checkpoint inhibitors. Trends Cancer. (2023) 9:543. doi: 10.1016/j.trecan.2023.04.002
2. Wang J, Guo Z, Shen M, Xie Q, Xiang H. Potential application mechanism of traditional Chinese medicine in treating immune checkpoint inhibitor-induced colitis. Front Immunol. (2024) 15:1366489. doi: 10.3389/fimmu.2024.1366489
3. Sasson SC, Slevin SM, Cheung VTF, Nassiri I, Olsson-Brown A, Fryer E, et al. Interferon-gamma-producing CD8+ Tissue resident memory T cells are a targetable hallmark of immune checkpoint inhibitor-colitis. Gastroenterology. (2021) 161:1229–1244.e9. doi: 10.1053/j.gastro.2021.06.025
4. Tomm NK, Szczepanski JM, Fang JM, Choi W-T, Xue Y, Setia N, et al. Follow-up biopsies in gastrointestinal immune checkpoint inhibitor toxicity may show markedly different inflammatory patterns than initial injury. Hum Pathol. (2024) 148:60–5. doi: 10.1016/j.humpath.2024.05.001
5. Liu M, He M, Cao F. Mechanism of gastrointestinal adverse reactions caused bylmmune checkpoint inhibitors and treatment of integratedTraditional chinese and western medicine. J Pract Traditional Chin Internal Med. (2023) 37:88–92. doi: 10.13729/j.issn.1671-7813.Z20220613
6. Nielsen DL, Juhl CB, Chen IM, Kellermann L, Nielsen OH. Immune checkpoint Inhibitor-Induced diarrhea and Colitis: Incidence and Management. A systematic review and Meta-analysis. Cancer Treat Rev. (2022) 109:102440. doi: 10.1016/j.ctrv.2022.102440
7. George DJ, Spigel DR, Gordan LN, Kochuparambil ST, Molina AM, Yorio J, et al. Safety and efficacy of first-line nivolumab plus ipilimumab alternating with nivolumab monotherapy in patients with advanced renal cell carcinoma: the non-randomised, open-label, phase IIIb/IV CheckMate 920 trial. BMJ Open. (2022) 12:e058396. doi: 10.1136/bmjopen-2021-058396
8. Farha N, Alkhayyat M, Lindsey A, Mansoor E, Saleh MA. Immune checkpoint inhibitor induced colitis: A nationwide population-based study. Clin Res Hepatol Gastroenterol. (2022) 46:101778. doi: 10.1016/j.clinre.2021.101778
9. McCarter KR, Wolfgang T, Arabelovic S, Wang X, Yoshida K, Banasiak EP, et al. Mortality and immune-related adverse events after immune checkpoint inhibitor initiation for cancer among patients with pre-existing rheumatoid arthritis: a retrospective, comparative, cohort study. Lancet Rheumatol. (2023) 5:e274–83. doi: 10.1016/s2665-9913(23)00064-4
10. Diaz LA, Shiu K-K, Kim T-W, Jensen BV, Jensen LH, Punt C, et al. Pembrolizumab versus chemotherapy for microsatellite instability-high or mismatch repair-deficient metastatic colorectal cancer (KEYNOTE-177): final analysis of a randomised, open-label, phase 3 study. Lancet Oncol. (2022) 23:659–70. doi: 10.1016/S1470-2045(22)00197-8
11. Shitara K, Rha SY, Wyrwicz LS, Oshima T, Karaseva N, Osipov M, et al. Neoadjuvant and adjuvant pembrolizumab plus chemotherapy in locally advanced gastric or gastro-oesophageal cancer (KEYNOTE-585): an interim analysis of the multicentre, double-blind, randomised phase 3 study. Lancet Oncol. (2024) 25:212–24. doi: 10.1016/S1470-2045(23)00541-7
12. Lynch RC, Ujjani CS, Poh C, Warren EH, Smith SD, Shadman M, et al. Concurrent pembrolizumab with AVD for untreated classic Hodgkin lymphoma. Blood. (2023) 141:2576–86. doi: 10.1182/blood.2022019254
13. Ready NE, Audigier-Valette C, Goldman JW, Felip E, Ciuleanu T-E, Rosario García Campelo M, et al. First-line nivolumab plus ipilimumab for metastatic non-small cell lung cancer, including patients with ECOG performance status 2 and other special populations: CheckMate 817. J Immunother Cancer. (2023) 11:e006127. doi: 10.1136/jitc-2022-006127
14. Tykodi SS, Gordan LN, Alter RS, Arrowsmith E, Harrison MR, Percent I, et al. Safety and efficacy of nivolumab plus ipilimumab in patients with advanced non-clear cell renal cell carcinoma: results from the phase 3b/4 CheckMate 920 trial. J Immunother Cancer. (2022) 10:e003844. doi: 10.1136/jitc-2021-003844
15. Frentzas S, Austria Mislang AR, Lemech C, Nagrial A, Underhill C, Wang W, et al. Phase 1a dose escalation study of ivonescimab (AK112/SMT112), an anti-PD-1/VEGF-A bispecific antibody, in patients with advanced solid tumors. J Immunother Cancer. (2024) 12:e008037. doi: 10.1136/jitc-2023-008037
16. Schoenfeld JD, Giobbie-Hurder A, Ranasinghe S, Kao KZ, Lako A, Tsuji J, et al. Durvalumab plus tremelimumab alone or in combination with low-dose or hypofractionated radiotherapy in metastatic non-small-cell lung cancer refractory to previous PD(L)-1 therapy: an open-label, multicentre, randomised, phase 2 trial. Lancet Oncol. (2022) 23:279–91. doi: 10.1016/S1470-2045(21)00658-6
17. Oaknin A, Moore K, Meyer T, López-Picazo-González J, Devriese LA, Amin A, et al. Nivolumab with or without ipilimumab in patients with recurrent or metastatic cervical cancer (CheckMate 358): a phase 1-2, open-label, multicohort trial. Lancet Oncol. (2024) 25:588–602. doi: 10.1016/S1470-2045(24)00088-3
18. Monge C, Xie C, Myojin Y, Coffman K, Hrones DM, Wang S, et al. Phase I/II study of PexaVec in combination with immune checkpoint inhibition in refractory metastatic colorectal cancer. J Immunother Cancer. (2023) 11:e005640. doi: 10.1136/jitc-2022-005640
19. Necchi A, Roumiguié M, Kamat AM, Shore ND, Boormans JL, Esen AA, et al. Pembrolizumab monotherapy for high-risk non-muscle-invasive bladder cancer without carcinoma in situ and unresponsive to BCG (KEYNOTE-057): a single-arm, multicentre, phase 2 trial. Lancet Oncol. (2024) 25:720–30. doi: 10.1016/S1470-2045(24)00178-5
20. Grimm M-O, Esteban E, Barthélémy P, Schmidinger M, Busch J, Valderrama BP, et al. Tailored immunotherapy approach with nivolumab with or without nivolumab plus ipilimumab as immunotherapeutic boost in patients with metastatic renal cell carcinoma (TITAN-RCC): a multicentre, single-arm, phase 2 trial. Lancet Oncol. (2023) 24:1252–65. doi: 10.1016/S1470-2045(23)00449-7
21. Lakhani N, Cosman R, Banerji U, Rasco D, Tomaszewska-Kiecana M, Garralda E, et al. A first-in-human phase I study of the PD-1 inhibitor, retifanlimab (INCMGA00012), in patients with advanced solid tumors (POD1UM-101). ESMO Open. (2024) 9:102254. doi: 10.1016/j.esmoop.2024.102254
22. Schöffski P, Bahleda R, Wagner AJ, Burgess MA, Junker N, Chisamore M, et al. Results of an open-label, phase ia/b study of pembrolizumab plus olaratumab in patients with unresectable, locally advanced, or metastatic soft-tissue sarcoma. Clin Cancer Res. (2023) 29:3320–8. doi: 10.1158/1078-0432.CCR-23-0742
23. Zhao Z-R, Liu S-L, Zhou T, Chen G, Long H, Su X-D, et al. Stereotactic body radiotherapy with sequential tislelizumab and chemotherapy as neoadjuvant therapy in patients with resectable non-small-cell lung cancer in China (SACTION01): a single-arm, single-centre, phase 2 trial. Lancet Respir Med. (2024) 12:988–96. doi: 10.1016/S2213-2600(24)00215-7
24. Saba NF, Wong SJ, Nasti T, McCook-Veal AA, McDonald MW, Stokes WA, et al. Intensity-modulated reirradiation therapy with nivolumab in recurrent or second primary head and neck squamous cell carcinoma: A nonrandomized controlled trial. JAMA Oncol. (2024) 10(7):896–904. doi: 10.1001/jamaoncol.2024.1143
25. Emamekhoo H, Olsen MR, Carthon BC, Drakaki A, Percent IJ, Molina AM, et al. Safety and efficacy of nivolumab plus ipilimumab in patients with advanced renal cell carcinoma with brain metastases: CheckMate 920. Cancer. (2022) 128:966–74. doi: 10.1002/cncr.34016
26. Morano F, Raimondi A, Pagani F, Lonardi S, Salvatore L, Cremolini C, et al. Temozolomide followed by combination with low-dose ipilimumab and nivolumab in patients with microsatellite-stable, O6-methylguanine-DNA methyltransferase-silenced metastatic colorectal cancer: the MAYA trial. J Clin Oncol. (2022) 40:1562–73. doi: 10.1200/JCO.21.02583
27. Kim S, Ghiringhelli F, de la Fouchardière C, Evesque L, Smith D, Badet N, et al. Atezolizumab plus modified docetaxel, cisplatin, and fluorouracil as first-line treatment for advanced anal cancer (SCARCE C17-02 PRODIGE 60): a randomised, non-comparative, phase 2 study. Lancet Oncol. (2024) 25:518–28. doi: 10.1016/S1470-2045(24)00081-0
28. Ferris RL, Moskovitz J, Kunning S, Ruffin AT, Reeder C, Ohr J, et al. Phase I trial of cetuximab, radiotherapy, and ipilimumab in locally advanced head and neck cancer. Clin Cancer Res. (2022) 28:1335–44. doi: 10.1158/1078-0432.CCR-21-0426
29. Xiao A. Evaluation of dose distribution to organs-at-risk in a prospective phase 1 trial of pembrolizumab and multisite stereotactic body radiation therapy (SBRT). Pract Radiat Oncol. (2022) 12(1):68–77. doi: 10.1016/j.prro.2021.09.005
30. Marabelle A, Le DT, Ascierto PA, Di Giacomo AM, De-Jesus-Acosta A, Delord J-P, et al. Efficacy of pembrolizumab in patients with noncolorectal high microsatellite instability/mismatch repair-deficient cancer: results from the phase II KEYNOTE-158 study. J Clin Oncol. (2020) 38:1–10. doi: 10.1200/JCO.19.02105
31. Stratigos AJ, Sekulic A, Peris K, Bechter O, Prey S, Kaatz M, et al. Cemiplimab in locally advanced basal cell carcinoma after hedgehog inhibitor therapy: an open-label, multi-centre, single-arm, phase 2 trial. Lancet Oncol. (2021) 22:848–57. doi: 10.1016/S1470-2045(21)00126-1
32. Qian DC, Kleber T, Brammer B, Xu KM, Switchenko JM, Janopaul-Naylor JR, et al. Effect of immunotherapy time-of-day infusion on overall survival among patients with advanced melanoma in the USA (MEMOIR): a propensity score-matched analysis of a single-centre, longitudinal study. Lancet Oncol. (2021) 22:1777–86. doi: 10.1016/S1470-2045(21)00546-5
33. Tawbi HA, Forsyth PA, Hodi FS, Algazi AP, Hamid O, Lao CD, et al. Long-term outcomes of patients with active melanoma brain metastases treated with combination nivolumab plus ipilimumab (CheckMate 204): final results of an open-label, multicentre, phase 2 study. Lancet Oncol. (2021) 22:1692–704. doi: 10.1016/S1470-2045(21)00545-3
34. Ascierto PA, Del Vecchio M, Mandalá M, Gogas H, Arance AM, Dalle S, et al. Adjuvant nivolumab versus ipilimumab in resected stage IIIB-C and stage IV melanoma (CheckMate 238): 4-year results from a multicentre, double-blind, randomised, controlled, phase 3 trial. Lancet Oncol. (2020) 21:1465–77. doi: 10.1016/S1470-2045(20)30494-0
35. Goldberg SB, Schalper KA, Gettinger SN, Mahajan A, Herbst RS, Chiang AC, et al. Pembrolizumab for management of patients with NSCLC and brain metastases: long-term results and biomarker analysis from a non-randomised, open-label, phase 2 trial. Lancet Oncol. (2020) 21:655–63. doi: 10.1016/S1470-2045(20)30111-X
36. Campbell MT, Matin SF, Tam AL, Sheth RA, Ahrar K, Tidwell RS, et al. Pilot study of Tremelimumab with and without cryoablation in patients with metastatic renal cell carcinoma. Nat Commun. (2021) 12:6375. doi: 10.1038/s41467-021-26415-4
37. Cacciotti C, Choi J, Alexandrescu S, Zimmerman MA, Cooney TM, Chordas C, et al. Immune checkpoint inhibition for pediatric patients with recurrent/refractory CNS tumors: a single institution experience. J Neurooncol. (2020) 149:113–22. doi: 10.1007/s11060-020-03578-6
38. Gao J, Navai N, Alhalabi O, Siefker-Radtke A, Campbell MT, Tidwell RS, et al. Neoadjuvant PD-L1 plus CTLA-4 blockade in patients with cisplatin-ineligible operable high-risk urothelial carcinoma. Nat Med. (2020) 26:1845–51. doi: 10.1038/s41591-020-1086-y
39. Brastianos PK, Strickland MR, Lee EQ, Wang N, Cohen JV, Chukwueke U, et al. Phase II study of ipilimumab and nivolumab in leptomeningeal carcinomatosis. Nat Commun. (2021) 12:5954. doi: 10.1038/s41467-021-25859-y
40. Kawazoe A, Yamaguchi K, Yasui H, Negoro Y, Azuma M, Amagai K, et al. Safety and efficacy of pembrolizumab in combination with S-1 plus oxaliplatin as a first-line treatment in patients with advanced gastric/gastroesophageal junction cancer: Cohort 1 data from the KEYNOTE-659 phase IIb study. Eur J Cancer. (2020) 129:97–106. doi: 10.1016/j.ejca.2020.02.002
41. Tolaney SM, Barroso-Sousa R, Keenan T, Li T, Trippa L, Vaz-Luis I, et al. Effect of eribulin with or without pembrolizumab on progression-free survival for patients with hormone receptor-positive, ERBB2-negative metastatic breast cancer: A randomized clinical trial. JAMA Oncol. (2020) 6:1598–605. doi: 10.1001/jamaoncol.2020.3524
42. Sanborn RE, Hamid O, de Vries EG, Ott PA, Garcia-Corbacho J, Boni V, et al. CX-072 (pacmilimab), a Probody PD-L1 inhibitor, in combination with ipilimumab in patients with advanced solid tumors (PROCLAIM-CX-072): a first-in-human, dose-finding study. J Immunother Cancer. (2021) 9:e002446. doi: 10.1136/jitc-2021-002446
43. Desai J, Deva S, Lee JS, Lin C-C, Yen C-J, Chao Y, et al. Phase IA/IB study of single-agent tislelizumab, an investigational anti-PD-1 antibody, in solid tumors. J Immunother Cancer. (2020) 8:e000453. doi: 10.1136/jitc-2019-000453
44. Diefenbach CS, Hong F, Ambinder RF, Cohen JB, Robertson MJ, David KA, et al. Ipilimumab, nivolumab, and brentuximab vedotin combination therapies in patients with relapsed or refractory Hodgkin lymphoma: phase 1 results of an open-label, multicentre, phase 1/2 trial. Lancet Haematol. (2020) 7:e660–70. doi: 10.1016/S2352-3026(20)30221-0
45. Apolo AB, Nadal R, Girardi DM, Niglio SA, Ley L, Cordes LM, et al. Phase I study of cabozantinib and nivolumab alone or with ipilimumab for advanced or metastatic urothelial carcinoma and other genitourinary tumors. J Clin Oncol. (2020) 38:3672–84. doi: 10.1200/JCO.20.01652
46. Yap TA, Nakagawa K, Fujimoto N, Kuribayashi K, Guren TK, Calabrò L, et al. Efficacy and safety of pembrolizumab in patients with advanced mesothelioma in the open-label, single-arm, phase 2 KEYNOTE-158 study. Lancet Respir Med. (2021) 9:613–21. doi: 10.1016/S2213-2600(20)30515-4
47. McDermott DF, Lee J-L, Bjarnason GA, Larkin JMG, Gafanov RA, Kochenderfer MD, et al. Open-label, single-arm phase II study of pembrolizumab monotherapy as first-line therapy in patients with advanced clear cell renal cell carcinoma. J Clin Oncol. (2021) 39:1020–8. doi: 10.1200/JCO.20.02363
48. Boutros C, Chaput-Gras N, Lanoy E, Larive A, Mateus C, Routier E, et al. Dose escalation phase 1 study of radiotherapy in combination with anti-cytotoxic-T-lymphocyte-associated antigen 4 monoclonal antibody ipilimumab in patients with metastatic melanoma. J Immunother Cancer. (2020) 8:e000627. doi: 10.1136/jitc-2020-000627
49. Felip E, Ardizzoni A, Ciuleanu T, Cobo M, Laktionov K, Szilasi M, et al. CheckMate 171: A phase 2 trial of nivolumab in patients with previously treated advanced squamous non-small cell lung cancer, including ECOG PS 2 and elderly populations. Eur J Cancer. (2020) 127:160–72. doi: 10.1016/j.ejca.2019.11.019
50. Geukes Foppen MH, Rozeman EA, van Wilpe S, Postma C, Snaebjornsson P, van Thienen JV, et al. Immune checkpoint inhibition-related colitis: symptoms, endoscopic features, histology and response to management. ESMO Open. (2018) 3:e000278. doi: 10.1136/esmoopen-2017-000278
51. Samaan MA, Pavlidis P, Papa S, Powell N, Irving PM. Gastrointestinal toxicity of immune checkpoint inhibitors: from mechanisms to management. Nat Rev Gastroenterol Hepatol. (2018) 15:222–34. doi: 10.1038/nrgastro.2018.14
52. Gong Z, Wang Y. Immune checkpoint inhibitor-mediated diarrhea and colitis: A clinical review. JCO Oncol Pract. (2020) 16:453–61. doi: 10.1200/OP.20.00002
53. Zoghbi M, Burk KJ, Haroun E, Saade M, Carreras MTC. Immune checkpoint inhibitor-induced diarrhea and colitis: an overview. Support Care Cancer. (2024) 32:680. doi: 10.1007/s00520-024-08889-2
54. Losurdo G, Angelillo D, Favia N, Sergi MC, Di Leo A, Triggiano G, et al. Checkpoint inhibitor-induced colitis: an update. Biomedicines. (2023) 11:1496. doi: 10.3390/biomedicines11051496
55. Giesler S, Riemer R, Lowinus T, Zeiser R. Immune-mediated colitis after immune checkpoint inhibitor therapy. Trends Mol Med. (2024) 29:S1471-4914(24)00266-1. doi: 10.1016/j.molmed.2024.09.009
56. Karamchandani DM, Chetty R. Immune checkpoint inhibitor-induced gastrointestinal and hepatic injury: pathologists’ perspective. J Clin Pathol. (2018) 71:665–71. doi: 10.1136/jclinpath-2018-205143
57. Patil PA, Zhang X. Pathologic manifestations of gastrointestinal and hepatobiliary injury in immune checkpoint inhibitor therapy. Arch Pathol Lab Med. (2021) 145:571–82. doi: 10.5858/arpa.2020-0070-RA
58. Gonzalez RS, Salaria SN, Bohannon CD, Huber AR, Feely MM, Shi C. PD-1 inhibitor gastroenterocolitis: case series and appraisal of “immunomodulatory gastroenterocolitis. Histopathology. (2017) 70:558–67. doi: 10.1111/his.13118
59. Ma C, Pai RK, Schaeffer DF, Krell J, Guizzetti L, McFarlane SC, et al. Recommendations for standardizing biopsy acquisition and histological assessment of immune checkpoint inhibitor-associated colitis. J Immunother Cancer. (2022) 10:e004560. doi: 10.1136/jitc-2022-004560
60. Fredrick TW, Ramos GP, Braga Neto MB, Kane S, Faubion WA, Loftus EV, et al. Clinical course and impact of immune checkpoint inhibitor colitis resembling microscopic colitis. Crohns Colitis 360. (2022) 4:otac008. doi: 10.1093/crocol/otac008
61. Zhang ML, Neyaz A, Patil D, Chen J, Dougan M, Deshpande V. Immune-related adverse events in the gastrointestinal tract: diagnostic utility of upper gastrointestinal biopsies. Histopathology. (2020) 76(2):233–43. doi: 10.1111/his.13963
62. Common terminology criteria for adverse events (CTCAE) | Protocol development | CTEP . Available online at: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm (Accessed December 18, 2024).
63. Haanen J, Obeid M, Spain L, Carbonnel F, Wang Y, Robert C, et al. Management of toxicities from immunotherapy: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. (2022) 33:1217–38. doi: 10.1016/j.annonc.2022.10.001
64. Schneider BJ, Naidoo J, Santomasso BD, Lacchetti C, Adkins S, Anadkat M, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: ASCO guideline update. J Clin Oncol. (2021) 39:4073–126. doi: 10.1200/JCO.21.01440
65. Chinese society of clinical oncology . Available online at: https://www.csco.org.cn/cn/index.aspx (Accessed December 24, 2024).
66. Kou F, Li J, Cao Y, Peng Z, Xu T, Shen L, et al. Immune checkpoint inhibitor-induced colitis with endoscopic evaluation in Chinese cancer patients: a single-centre retrospective study. Front Oncol. (2023) 13:1285478. doi: 10.3389/fonc.2023.1285478
67. Dougan M, Wang Y, Rubio-Tapia A, Lim JK. AGA clinical practice update on diagnosis and management of immune checkpoint inhibitor colitis and hepatitis: expert review. Gastroenterology. (2021) 160:1384–93. doi: 10.1053/j.gastro.2020.08.063
68. Yokode M, Shiokawa M, Kawakami H, Kuwada T, Nishikawa Y, Muramoto Y, et al. Anti-integrin αvβ6 autoantibodies are a potential biomarker for ulcerative colitis-like immune checkpoint inhibitor-induced colitis. Br J Cancer. (2024) 130:1552–60. doi: 10.1038/s41416-024-02647-1
69. Thomas MF, Slowikowski K, Manakongtreecheep K, Sen P, Samanta N, Tantivit J, et al. Single-cell transcriptomic analyses reveal distinct immune cell contributions to epithelial barrier dysfunction in checkpoint inhibitor colitis. Nat Med. (2024) 30:1349–62. doi: 10.1038/s41591-024-02895-x
70. Malik A, Sharma D, Aguirre-Gamboa R, McGrath S, Zabala S, Weber C, et al. Epithelial IFNγ signalling and compartmentalized antigen presentation orchestrate gut immunity. Nature. (2023) 623:1044–52. doi: 10.1038/s41586-023-06721-1
71. Lo BC, Kryczek I, Yu J, Vatan L, Caruso R, Matsumoto M, et al. Microbiota-dependent activation of CD4+ T cells induces CTLA-4 blockade-associated colitis via Fcγ receptors. Science. (2024) 383:62–70. doi: 10.1126/science.adh8342
72. Zeng L, Wang Y, Shen J, Wei X, Wu Y, Chi X, et al. TIPE2 aggravates experimental colitis and disrupts intestinal epithelial barrier integrity by activating JAK2/STAT3/SOCS3 signal pathway. Exp Cell Res. (2024) 443:114287. doi: 10.1016/j.yexcr.2024.114287
73. Reschke R, Shapiro JW, Yu J, Rouhani SJ, Olson DJ, Zha Y, et al. Checkpoint blockade-induced dermatitis and colitis are dominated by tissue-resident memory T cells and th1/tc1 cytokines. Cancer Immunol Res. (2022) 10:1167–74. doi: 10.1158/2326-6066.CIR-22-0362
74. Halsey TM, Thomas AS, Hayase T, Ma W, Abu-Sbeih H, Sun B, et al. Microbiome alteration via fecal microbiota transplantation is effective for refractory immune checkpoint inhibitor–induced colitis. Sci Transl Med. (2023) 15:eabq4006. doi: 10.1126/scitranslmed.abq4006
75. He JY, Kim Y-J, Mennillo E, Rusu I, Bain J, Rao AA, et al. Dysregulation of CD4+ and CD8+ resident memory T, myeloid, and stromal cells in steroid-experienced, checkpoint inhibitor colitis. J Immunother Cancer. (2024) 12:e008628. doi: 10.1136/jitc-2023-008628
76. Ghosh S, Singh R, Goap TJ, Sunnapu O, Vanwinkle ZM, Li H, et al. Inflammation-targeted delivery of Urolithin A mitigates chemical- and immune checkpoint inhibitor-induced colitis. J Nanobiotechnol. (2024) 22:701. doi: 10.1186/s12951-024-02990-8
77. Ye R, Zheng H, Yang D, Lin J, Li L, Li Y, et al. irAE-colitis induced by CTLA-4 and PD-1 blocking were ameliorated by TNF blocking and modulation of gut microbial. BioMed Pharmacother. (2024) 177:116999. doi: 10.1016/j.biopha.2024.116999
78. Keam S, Turner N, Kugeratski FG, Rico R, Colunga-Minutti J, Poojary R, et al. Toxicity in the era of immune checkpoint inhibitor therapy. Front Immunol. (2024) 15:1447021. doi: 10.3389/fimmu.2024.1447021
79. Lin X, Kang K, Chen P, Zeng Z, Li G, Xiong W, et al. Regulatory mechanisms of PD-1/PD-L1 in cancers. Mol Cancer. (2024) 23:108. doi: 10.1186/s12943-024-02023-w
80. Huang J, Xiong L, Tang S, Zhao J, Zuo L. Balancing tumor immunotherapy and immune-related adverse events: unveiling the key regulators. Int J Mol Sci. (2024) 25:10919. doi: 10.3390/ijms252010919
81. Reinhardt C, Bergentall M, Greiner TU, Schaffner F, Ostergren-Lundén G, Petersen LC, et al. Tissue factor and PAR1 promote microbiota-induced intestinal vascular remodelling. Nature. (2012) 483:627–31. doi: 10.1038/nature10893
82. Ohwada S, Ishigami K, Akutsu N, Nakase H. Pharmacological treatments available for immune-checkpoint-inhibitor-induced colitis. Biomedicines. (2022) 10:1334. doi: 10.3390/biomedicines10061334
83. Thompson JA, Schneider BJ, Brahmer J, Andrews S, Armand P, Bhatia S, et al. NCCN guidelines insights: management of immunotherapy-related toxicities, version 1.2020. J Natl Compr Canc Netw. (2020) 18:230–41. doi: 10.6004/jnccn.2020.0012
84. Daetwyler E, Wallrabenstein T, König D, Cappelli LC, Naidoo J, Zippelius A, et al. Corticosteroid-resistant immune-related adverse events: a systematic review. J Immunother Cancer. (2024) 12:e007409. doi: 10.1136/jitc-2023-007409
85. Collins M, Michot JM, Danlos FX, Mussini C, Soularue E, Mateus C, et al. Inflammatory gastrointestinal diseases associated with PD-1 blockade antibodies. Ann Oncol. (2017) 28:2860–5. doi: 10.1093/annonc/mdx403
86. Burla J, Bluemel S, Biedermann L, Barysch MJ, Dummer R, Levesque MP, et al. Retrospective analysis of treatment and complications of immune checkpoint inhibitor-associated colitis: histological ulcerations as potential predictor for a steroid-refractory disease course. Inflammation Intest Dis. (2020) 5:109–16. doi: 10.1159/000507579
87. Alexander JL, Ibraheim H, Sheth B, Little J, Khan MS, Richards C, et al. Clinical outcomes of patients with corticosteroid refractory immune checkpoint inhibitor-induced enterocolitis treated with infliximab. J Immunother Cancer. (2021) 9:e002742. doi: 10.1136/jitc-2021-002742
88. Alorfi NM, Alourfi MM. Biologic therapy for refractory immune checkpoint inhibitor colitis. Biologics. (2022) 16:119–27. doi: 10.2147/BTT.S367675
89. Johnson DH, Zobniw CM, Trinh VA, Ma J, Bassett RL, Abdel-Wahab N, et al. Infliximab associated with faster symptom resolution compared with corticosteroids alone for the management of immune-related enterocolitis. J Immunother Cancer. (2018) 6:103. doi: 10.1186/s40425-018-0412-0
90. Abu-Sbeih H, Ali FS, Alsaadi D, Jennings J, Luo W, Gong Z, et al. Outcomes of vedolizumab therapy in patients with immune checkpoint inhibitor–induced colitis: a multi-center study. J Immunother Cancer. (2018) 6:142. doi: 10.1186/s40425-018-0461-4
91. Gao Y, Xu P, Sun D, Jiang Y, Lin X-L, Han T, et al. Faecalibacterium prausnitzii abrogates intestinal toxicity and promotes tumor immunity to increase the efficacy of dual CTLA4 and PD-1 checkpoint blockade. Cancer Res. (2023) 83:3710–25. doi: 10.1158/0008-5472.CAN-23-0605
92. Li Y, Li Z-X, Xie C-Y, Fan J, Lv J, Xu X-J, et al. Gegen Qinlian decoction enhances immunity and protects intestinal barrier function in colorectal cancer patients via gut microbiota. World J Gastroenterol. (2020) 26:7633–51. doi: 10.3748/wjg.v26.i48.7633
93. Shen H, Zhang S, Zhao W, Ren S, Ke X, Gu Q, et al. Randomised clinical trial: Efficacy and safety of Qing-Chang-Hua-Shi granules in a multicenter, randomized, and double-blind clinical trial of patients with moderately active ulcerative colitis. BioMed Pharmacother. (2021) 139:111580. doi: 10.1016/j.biopha.2021.111580
94. Xu J, Ma C. Effect of baitouweng decoction enema in the treatment of ulcerative colitis patients and its influence on T cell subsets. J Nanjing Univ Traditional Chin Med. (2019) 35:29–31 + 62. doi: 10.14148/j.issn.1672-0482.2019.0029
95. Wu D, Niu M, Zhen H. Effects of qingre zaoshi jiedu huyin decoction in patients with ulcerative colitis. Henan Med Res. (2024) 33:4165–8. doi: 10.3969/j.issn.1004-437X.2024.22.033
96. Peng C, Li M, Wang J, Wang R. Clinical effect of Wentongjingluo bamboo scraping therapy combined with acupoint application in patients with ulcerative colitis of large intestine damp-heat: Based on the theory of “TongYinTongYong. Hebei J Traditional Chin Med. (2024) 46:1871–1875 + 1880. doi: 10.3969/j.issn.1002-2619.2024.11.024
97. Peng B, Wen Y, Zhang S, Lin W, Li H, Cai J, et al. N-of-1 trial of jianpiqingrehuashi granular decoction with mesalamine in the treatment of ulcerative colitis in remission with spleen deficiency-induced damp-heat syndrome. Chin Gen Pract. (2022) 25:293–297 + 304. doi: 10.12114/j.issn.1007-9572.2021.02.106
98. He H, Shen H. Chinese medicine prescription of paniculate swallowwort root as sovereign drug in the treatment of active ulcerative colitis. Chin Med Modern Distance Educ China. (2024) 22:149–52. doi: 10.3969/j.issn.1672⁃2779.2024.18.048
99. Ben-Horin S, Salomon N, Karampekos G, Viazis N, Lahat A, Ungar B, et al. Curcumin-qingDai combination for patients with active ulcerative colitis: A randomized, double-blinded, placebo-controlled trial. Clin Gastroenterol Hepatol. (2024) 22:347–356.e6. doi: 10.1016/j.cgh.2023.05.023
100. Erol Doğan Ö, Karaca Çelik KE, Baş M, Alan EH, Çağın YF. Effects of mediterranean diet, curcumin, and resveratrol on mild-to-moderate active ulcerative colitis: A multicenter randomized clinical trial. Nutrients. (2024) 16:1504. doi: 10.3390/nu16101504
101. Guo S, Zhou J, Zhang L, Bao C-H, Zhao J-M, Gao Y-L, et al. Acupuncture and moxibustion inhibited intestinal epithelial-mesenchymal transition in patients with crohn’s disease induced by TGF- β 1/smad3/snail pathway: A clinical trial study. Chin J Integr Med. (2022) 28:823–32. doi: 10.1007/s11655-022-2888-1
102. Qi Q, Im H, Li K-S, Gu M, Wu H-G, Yang L, et al. Influence of herb-partitioned moxibustion at Qihai (CV6) and bilateral Tianshu (ST25) and Shangjuxu (ST37) acupoints on toll-like receptors 4 signaling pathways in patients with ulcerative coliti. J Tradit Chin Med. (2021) 41:479–85. doi: 10.19852/j.cnki.jtcm.20210310.001
103. Guo W, Li L, Mei Q, Li H, Dai W, Wang J. Efficacy of huanglian wendan decoction in the treatment of ulcerative colitis of large intestine with damp-heat type. Eval Anal Drug-Use Hospitals China. (2024) 24:455–8. doi: 10.14009/j.issn.1672-2124.2024.04.016
104. Fan J, Zhao J, Yan H, Sun D, Wang S, Liang J, et al. Effects of Anchang Yuyang Decoction combined with mesalazine in the treatment of ulcerative colitis with syndrome of spleen deficiency and dampness retention and its influence on inflammatory factors. China J Traditional Chin Med Pharm. (2020) 35:3765–70.
105. Xiaoling D, Anming Z, Hui L, Bei S, Yi R, Hongzhu W, et al. Qingchang suppositry induced remission in patients with mild-to-moderate ulcerative proctitis: a multicenter, prospective, randomized, parallel-controlled clinical trial. J Tradit Chin Med. (2024) 44:156–62. doi: 10.19852/j.cnki.jtcm.20231121.004
106. Zhou C, Wang Y, Chen J, Liu F. Clinical observation on treatment of ulcerative colitis with retention enema of baitouweng decoction. Liaoning J Traditional Chin Med. (2021) 48:121–4. doi: 10.13192/j.issn.1000-1719.2021.09.033
107. Li J, Fan S, Li H, Hu Z, Hu Q. Evaluation of efficacy, safety and underlying mechanism on Traditional Chinese medicine as synergistic agents for cancer immunotherapy: A preclinical systematic review and meta-analysis. J Ethnopharmacol. (2025) 338:119035. doi: 10.1016/j.jep.2024.119035
108. Zhu L, Xu L-Z, Zhao S, Shen Z-F, Shen H, Zhan L-B. Protective effect of baicalin on the regulation of Treg/Th17 balance, gut microbiota and short-chain fatty acids in rats with ulcerative colitis. Appl Microbiol Biotechnol. (2020) 104:5449–60. doi: 10.1007/s00253-020-10527-w
109. Li R, Chen Y, Shi M, Xu X, Zhao Y, Wu X, et al. Gegen Qinlian decoction alleviates experimental colitis via suppressing TLR4/NF-κB signaling and enhancing antioxidant effect. Phytomedicine. (2016) 23:1012–20. doi: 10.1016/j.phymed.2016.06.010
110. Yun H-F, Liu R, Han D, Zhao X, Guo J-W, Yan F-J, et al. Pingkui enema alleviates TNBS-induced ulcerative colitis by regulation of inflammatory factors, gut bifidobacterium, and intestinal mucosal barrier in rats. Evid Based Complement Alternat Med. (2020) 2020:3896948. doi: 10.1155/2020/3896948
111. Li Z, Lin M, Li Y, Shao J, Huang R, Qiu Y, et al. Total flavonoids of Sophora flavescens and kurarinone ameliorated ulcerative colitis by regulating Th17/Treg cell homeostasis. J Ethnopharmacol. (2022) 297:115500. doi: 10.1016/j.jep.2022.115500
112. Xiao S, Yan Y, Shao M, Zhou X, Niu Z, Wu Y, et al. Kuijieling decoction regulates the Treg/Th17 cell balance in ulcerative colitis through the RA/RARα signaling pathway. J Ethnopharmacol. (2024) 318:116909. doi: 10.1016/j.jep.2023.116909
113. Liu Y, Dong Y, Shen W, Du J, Sun Q, Yang Y, et al. Platycodon grandiflorus polysaccharide regulates colonic immunity through mesenteric lymphatic circulation to attenuate ulcerative colitis. Chin J Nat Med. (2023) 21:263–78. doi: 10.1016/S1875-5364(23)60435-2
114. Rao K, Qin S, Yang Y, Zhan K, Wu H, Zheng H, et al. Shenling baizhu powder alleviates TNBS-induced colitis in rats by improving intestinal epithelial permeability and inhibiting inflammation through the TLR5/myD88/NF-κB pathway. Front Pharmacol. (2022) 13:883918. doi: 10.3389/fphar.2022.883918
Keywords: immune checkpoint inhibitors (ICIs), immune checkpoint inhibitor induced colitis (irColitis), traditional Chinese medicine (TCM), gut microbiota, review
Citation: Dong H, Peng Y, Wang X and Cui H (2025) An updated review on immune checkpoint inhibitor-induced colitis: epidemiology, pathogenesis, treatment strategies, and the role of traditional Chinese medicine. Front. Immunol. 16:1551445. doi: 10.3389/fimmu.2025.1551445
Received: 25 December 2024; Accepted: 26 February 2025;
Published: 17 March 2025.
Edited by:
Chunping Wan, First Affiliated Hospital of Yunnan University of Traditional Chinese Medicine, ChinaReviewed by:
Hou Li, Beijing University of Chinese Medicine, ChinaCopyright © 2025 Dong, Peng, Wang and Cui. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Huijuan Cui, Y2hqenJ5aHl5QHNpbmEuY29t
†These authors share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.