
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Immunol. , 17 March 2025
Sec. Autoimmune and Autoinflammatory Disorders : Autoimmune Disorders
Volume 16 - 2025 | https://doi.org/10.3389/fimmu.2025.1545546
 Angelo Fassio1
Angelo Fassio1 Alvise Berti2,3
Alvise Berti2,3 Alessandro Mantovani4
Alessandro Mantovani4 Giovanni Adami1
Giovanni Adami1 Francesco Pollastri1*
Francesco Pollastri1* Davide Gatti1
Davide Gatti1 Riccardo Bixio1
Riccardo Bixio1 Valeria Messina1
Valeria Messina1 Maurizio Rossini1
Maurizio Rossini1 Davide Bertelle1,5
Davide Bertelle1,5 Eugenia Bertoldo1
Eugenia Bertoldo1 Isotta Galvagni1
Isotta Galvagni1 Roberto Bortolotti3
Roberto Bortolotti3 Ombretta Viapiana1
Ombretta Viapiana1Background/aim: We performed a systematic review and meta-analysis of observational studies aimed at investigating the prevalence of osteoporosis and osteoporotic fractures in subjects affected by systemic vasculitides (SVs) as well as to explore their risk of osteoporosis and osteoporotic fractures when compared to healthy controls.
Methods: Scopus, Web of Science and PubMed were systematically searched from inception to February 2024 for observational studies investigating the prevalence of osteoporosis and fragility fractures in adults with SVs. In addition, when available, we assessed the odd ratios (OR) of prevalent osteoporosis and fragility fractures amongst subjects with SVs vs. healthy controls. Data from eligible studies were extracted, and meta-analysis was performed using a random effects model to obtain ORs with 95% confidence intervals (CIs). Subgroup analyses and meta-regressions were also performed. This study was registered in Open Science Framework (DOI: https://doi.org/10.17605/OSF.IO/3G7RJ).
Results: Forty studies with 23,358 individuals affected by SVs were included. The overall prevalence of osteoporosis and fragility fractures in the SV patients were respectively 14.64% (95%CI 12.21-18.89), and 17.08% (95%CI 11.42-24.78). The ORs for osteoporosis and fragility fractures in SV patients when compared with healthy controls were 2.92 (95%CI 1.72-4.98) and 2.39 (95%CI 1.34-4.26) respectively. The univariable meta-regression analysis showed a significant association between cumulative glucocorticoids’ dosage (total grams) and risk of prevalent osteoporosis (estimate = 0.0995, R2 = 0.24, p=0.0194).
Conclusion: SVs are associated with an increased risk for osteoporosis and fragility fractures, suggesting that active vigilance and pre-emptive screening are recommended.
Systematic review registration: https://archive.org/details/osf-registrations-3g7rj-v1.
Vasculitides represent a group of rare, systemic conditions histologically characterized by the infiltration of leukocytes and inflammation within the blood vessel walls. The Chapel-Hill Consensus Conference Nomenclature of Vasculitides (CHCC) organizes these diseases based on discernible features that differentiate various forms into distinct categories (1). The primary classification criterion is based on the predominant involvement of specific vessel types, namely large, medium, and small vessels.
The varying sizes of these vessel subsets reflect their functions and susceptibility to distinct variants of vasculitis (2). Large vessel vasculitides encompasses giant cell arteritis (GCA) and Takayasu Arteritis (TAK), while polyarteritis nodosa and Kawasaki Disease typically affect medium-sized blood vessels. Small-vessel vasculitides are primarily associated with antineutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV), anti-glomerular basement membrane disease, cryoglobulinemic vasculitis, IgA vasculitis (Henoch-Schonlein), and hypocomplementemic urticarial vasculitis (anti-C1q vasculitis). Furthermore, this classification revision includes other forms of vasculitis, such as single-organ vasculitis or vasculitis involving vessels of any size (small, medium, and large) and type (arteries, veins, and capillaries).
The management of systemic vasculitis (SVs) is tailored to the specific vasculitis type but traditionally entails a regimen of high-dose glucocorticoids (GCs) either alone or alongside other immunosuppressive agents. Current guidelines support the use of high doses of GCs are generally used to induce remission in vasculitides, in the first months of therapy, while suggesting low-dose GCs therapy for maintenance of remission, during the follow-up.
Because of GCs, subjects with small and large vessel vasculitis face heightened fracture risks, in addition to other GC complications. However, GC-induced etiology is not the only risk factor of osteoporosis: chronic inflammation, nutritional inadequacy leading to vitamin D and calcium deficiency, impaired renal function, or other pharmacological interventions may increase the risk of impaired bone health (3).
Interestingly, over the past decade, there has been a notable increase in observational studies focusing on osteoporosis and fractures in idiopathic inflammatory vasculitides. Even if these studies globally showed an increase in osteoporosis prevalence in the different cohorts, different study designs, different populations (i.e. patients with large versus small vessels) and case ascertainment strategies may explain the heterogeneous findings among these different studies, often hampering a direct comparison among them and thus definitive conclusions.
Based on these considerations, we conducted a systematic review and meta-analysis to assess the prevalence of osteoporosis and osteoporotic fractures in individuals with SVs, as well as their risk for these complications compared to healthy controls.
We systematically searched 3 large electronic databases (Scopus, Web of Science and PubMed) from database inception to 01 December 2024, using pre-defined key words, to identify observational studies examining the prevalence of osteoporosis amongst adult individuals with and without SVs, as well as the risk of prevalent osteoporosis amongst those with SVs when compared to healthy controls. The search strategy is reported in the Supplementary Material. We also reviewed references from original papers and review articles to identify further eligible studies not covered by our original database searches. This systematic review was performed according to the updated Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement (4). We also followed the reporting proposed by the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) for the meta-analysis of these studies (5).
Studies were included in the meta-analysis if they met the following criteria: 1) original studies conducted in adults affected by systemic vasculitis; 2) all types of primary SVs as reported in the 2012 CHCC, including IgG4-RD.
Criteria for exclusion of the studies from the meta-analysis were as follows: 1) congress abstracts, case reports, reviews, practice guidelines; 2) studies which did not specifically report any data for the outcome of interest; 3) studies involving secondary forms and those with probable etiology. Searches was confined to randomized controlled trials, retrospective, longitudinal or cross-sectional studies. Studies with less than 10 subjects were excluded. Studies enrolling other forms of non-systemic vasculitides, such as isolated cutaneous or single-organ vasculitides were not included.
For all eligible studies we extracted data regarding the main characteristics of participants, vasculitis subtypes, number of patients with osteoporosis, number of patients with fragility fractures, values of bone mineral density (BMD) at the lumbar spine, total hip and femoral neck, and proportion of patients treated with GCs.
The primary outcome was the prevalence of osteoporosis and the prevalence of fragility fractures amongst those with and without vasculitis. The secondary outcomes were the risk for osteoporosis (as reported by the Authors, along with the corresponding classification criteria adopted), the standardized and absolute values of BMD at the lumbar spine, total hip and femoral neck and the risk of fragility fractures of subjects affected by systemic vasculitis as compared to healthy controls, the association between cumulative GC dosage (total grams) and risk of osteoporosis. All the analyses were performed in all the studies, when the data was available.
Data from studies eligible for the aggregate data meta-analysis were extracted by two authors independently (R.B. and V.M.). Disagreements at this level were resolved by consensus and a third author if needed (E.B.). For all studies, we extracted data on first author, publication year, study design, study country, population characteristics, methods used for osteoporosis diagnosis, type of vasculitis, BMD data, percentage of patients with any fractures and outcomes of interest. In case of multiple publications, we included the most up-to-date or comprehensive information.
Each eligible study was assessed for quality by using the Newcastle-Ottawa scale (NOS) adapted for cross-sectional studies by two independent reviewers (F.P., I.G.), with disagreements resolved through consensus. The NOS uses a star system to evaluate a study in three domains: selection of participants (assigning a maximum of 5 stars), comparability of study groups (assigning a maximum of 2 stars), and ascertainment of outcomes of interest (assigning a maximum of 3 stars). Therefore, 10 stars reflect the highest quality. We judged studies that received a score of ≥8 to be at low risk of bias, studies that scored 7 stars to be at medium risk, and those that scored ≤6 stars to be at high risk of bias.
In order to estimate the prevalence of osteoporosis and fragility fractures amongst those with and without SVs (when available), the number of patients with osteoporosis and fragility fractures amongst all individuals with vasculitis and/or healthy controls was considered as the effect size for each eligible study. Then, these data were pooled and the overall prevalences of osteoporosis and fragility fractures were calculated using a random-effects model. We used the Score (Wilson) method to compute the confidence intervals. To assess the risk of prevalent osteoporosis and fragility fractures in individuals with and without SVs, the odds ratios (ORs) and 95% CIs were also considered as the effect size for each eligible study. In the case of studies reporting several ORs with varying degrees of covariate adjustment, ORs that reflected the maximum extent of adjustment for potential confounding factors, were extracted. The adjusted ORs of all eligible studies were then pooled, and an overall estimate of the effect-size was calculated using a random-effects model, as this methodology considers any differences between studies, even if there is no statistically significant heterogeneity. To assess the difference of BMD in patients with SVs and healthy controls, BMD data were collected from the eligible studies when available and then weighted mean difference was calculated using a random-effects model.
Visual inspection of the forest plot was used to assess statistical heterogeneity. This was also assessed with the I2-statistics, which provides an estimate of the percentage of variability across eligible studies that is due to heterogeneity rather than chance alone. Heterogeneity was considered to be low if I2 is <25%, moderate if I2 is between 25% and 75%, and high if I2 is >75% (6). The risk of publication bias was examined using the funnel plot and the Egger’s regression test with logit transformed prevalence of osteoporosis (7).
To explore the possible sources of heterogeneity across the studies and to test the robustness of the observed associations, we performed subgroup analyses by study country, modality of osteoporosis diagnosis, and type of vasculitis.
Univariable meta-regression analyses were also performed to test the effect of specific moderator variables (i.e., age, sex, disease duration and cumulative dose of steroids) on the effect size for the risk of osteoporosis in adults with and without SVs. Finally, we tested for possible excessive influence of individual studies using a meta-analysis influence test that eliminated each of the included studies one at a time.
All statistical tests were two-sided, and p-values of <0.05 (two-tailed) was considered statistically significant. For analyses we used R software (version 4.3.3, R Foundation for Statistical Computing, Vienna, Austria) with “meta” and “metafor” packages.
The protocol of this systematic review was registered in advance on Open Science Framework (DOI: https://doi.org/10.17605/OSF.IO/3G7RJ).
We identified 66 potentially relevant papers. After examining the full text of these articles, we excluded 26 studies due to unsatisfactory inclusion criteria or unsatisfactory outcome measures (Supplementary Table 1). A total of 40 studies were considered eligible for the inclusion in this meta-analysis (Figure 1) and were assessed for quality, for a total of 23,358 individuals affected by SVs (mean age 64.8 years, SD: 12.2 years; 39.8% men). Tables 1 and 2 show the characteristics and RoB of the eligible studies and the characteristics of patients and controls included in the final analysis, respectively. Thiry-three out of 40 studies enrolled patients from hospital-based cohorts and 7 from population-based cohorts.
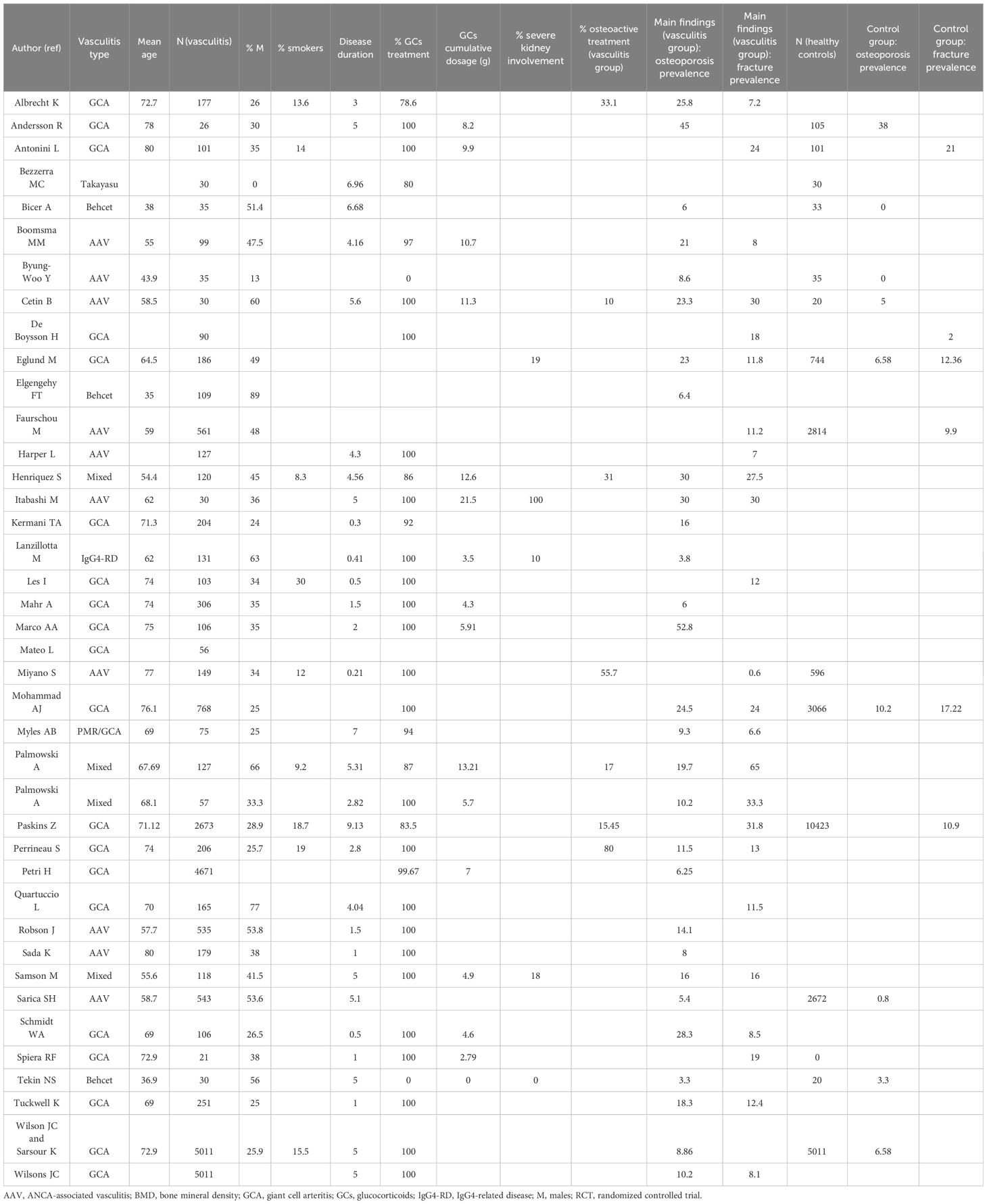
Table 2. Characteristics of the patients and healthy controls (when available) enrolled in the studies included in the final analysis.
The data on the prevalence of GCs was available in 34 studies, with 27 studies including cohorts with a >90% prevalence of steroid treatment. The mean cumulative GC dosage was 7.88 grams, SD 5.22 grams.
The criteria for the diagnosis of “osteoporosis” were rather heterogeneous: only 33% of the eligible studies based the definition on dual-energy X-ray absorptiometry (DXA) assessment (of which 13% without reporting the BMD values), with only four studies reporting standardized BMD data. 5% of the studies enrolled subjects based on self-reported history of “osteoporosis”, 50% based on medical records (unspecified criteria), and 12% based on history of fractures extracted from the medical records.
Overall, the pooled estimate for prevalence of “osteoporosis” was 14.64% (95%CI 11.21-18.89) in the whole SVs cohort (Figure 2) reporting overall analysis and grouped according to the different types of SVs), with IgG4-RD and Behcet’s being the SVs with the lower estimates and large vessel vasculitides (LVVs), and AAV that with the higher estimate (test for subgroup difference p<0.01). When focusing on the risk of “osteoporosis” in SVs versus healthy controls, the OR was significantly increased: 2.92 (95%CI 1.72-4.98) (Figure 3). European and American studies have the higher prevalence of “osteoporosis” as compared to Asia (Supplementary Figure 1, results are grouped according to the different countries in which the studies were conducted).
We performed the analysis on the forest plot and pooled estimates for total BMD for the SVs versus control, that yielded non-significant results (weighted mean difference: -0.03, 95%CI -0.07;0.01) (Supplementary Figure 2). Given the scarcity of data and unclear technique methodology, it was decided not to proceed with the meta-analysis for Z and T-scores.
Univariable meta-regression analyses to examine the effects of potential moderators on the risk of “osteoporosis”, including age, percentage of men, disease duration and cumulative dose of GCs were performed (Supplementary Table 2).
When focusing on the prevalence of osteoporotic fractures in the SVs groups, we observed an overall prevalence of 17.08% (95%CI 11.42-24.78) (Figure 4a). The OR for osteoporotic fractures in SVs versus healthy controls was OR: 2.39 (95%CI 1.34-4.26) (Figure 4b).
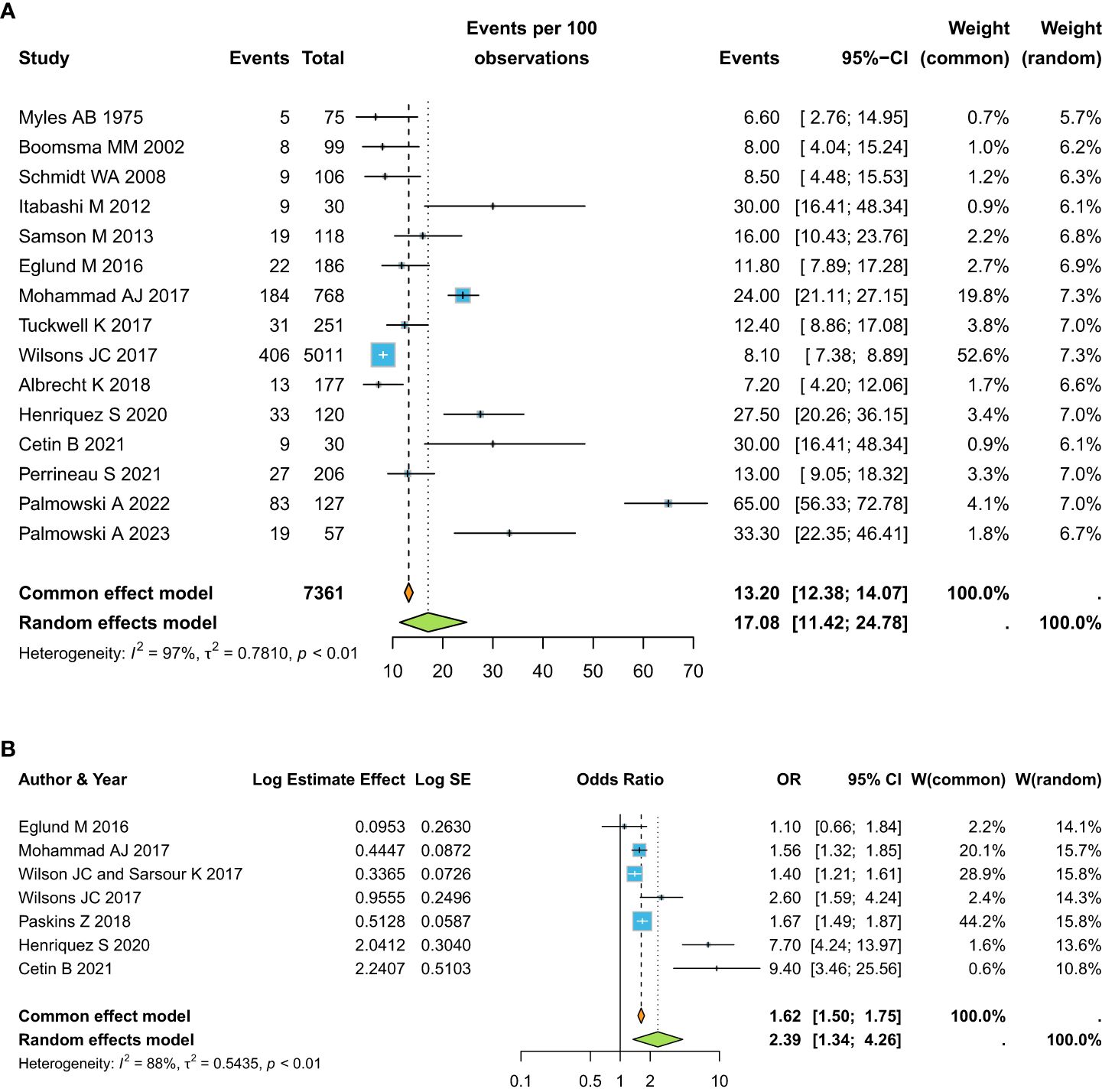
Figure 4. Pooled estimate for the prevalence of osteoporotic fractures in the SVs groups (a), and OR for osteoporotic fractures in SVs versus healthy controls (b).
We found that only the cumulative GCs dosage (total grams) modulated the risk of “osteoporosis” in patients with and without SVs (p=0.0194, estimate = 0.0995, standard error = 0.0426, R2 = 0.24), corresponding approximately to an increase of 1% in the prevalence of osteoporosis with each additional gram of cumulative GC. Considering that over the years treatment regimens for SVs have increasingly aimed at reducing the cumulative corticosteroid dose, and given that not all studies reported this data, a sensitivity analysis was performed using an invariable meta-regression analysis with the year of publication as a possible surrogate, and a multivariable analysis including both the year of publication and the cumulative dose of GCs.
The univariable analysis, using the year of publication as a predictor of “osteoporosis” prevalence, was not significant: estimate per publication year -0.015 (95% CI -0.046; 0.154, p=0.32), as was the multivariable analysis: estimate per publication year -0.025 (95% CI -0.083; 0.032, p=0.38) and estimate per GCs cumulative dosage (grams) 0.085 (95% CI -0.004; 0.175, p=0.06).
Regarding fracture prevalence, the univariable analysis was statistically significant: estimate per publication year 0.041 (95% CI 0.022; 0.080, p=0.0022). Similarly, the multivariable analysis also showed statistical significance: estimate per publication year 0.11 (95% CI 0.055; 0.165, p<0.001) and estimate per GCs cumulative dosage (grams) 0.0697 (95% CI -0.0059; 0.145, p=0.070).
In addition, since only 33% of osteoporosis diagnoses appear to be based on DXA and given the potential for misclassification when using patient-reported data or medical records, we performed a sensitivity analysis including only studies that used DXA assessment as an inclusion criterion. The resulting pooled estimate was similar to the original (Supplementary Figure 3): 15.91% (95% CI, 9.60–25.51).
The risk of bias for each eligible studies was assessed by the NOS tool and is reported in Table 1. Overall, there was a moderate-high risk of bias across the studies included in our meta-meta-analysis. In addition, the Egger’s regression test (p-value=0.0541) did not show any statistically significant asymmetry of the funnel plot, suggesting that the publication bias was unlikely (Supplementary Figure 4).
We conducted a systematic review and meta-analysis of observational studies to investigate the prevalence of osteoporosis and osteoporotic fractures among patients with different types of SV. We also examined the odds for either condition between patients with SVs versus healthy controls, when this data was available. Our analysis revealed a considerable prevalence of osteoporosis (14.6%) and osteoporotic fractures (17.1%) in patients affected by SVs, with an increased ORs both for osteoporosis (2.92) and fragility fractures (2.39) To our knowledge, this is the first data summarizing comprehensively (by means of a systematic review and meta-analysis) the prevalence of osteoporosis in SVs.
Osteoporosis and fragility fractures are associated with a significant disease burden, including limited mobility, chronic pain, loss of independence, and reduced quality of life (8). Indeed, available evidence show a remarkable increased risk of mortality following osteoporotic vertebral or hip fractures (9, 10), with a 20% one-year mortality rate after a hip fracture (11).
Several autoimmune rheumatic diseases have been associated with systemic bone loss and fragility, including rheumatoid arthritis (RA), spondyloarthritis (SpA), and systemic lupus erythematosus (SLE) (12–15). While there is good evidence suggesting a direct role for conditions such as RA and SpA (12, 13, 16), net of potential confounders such as GC treatment, the direct role of the rheumatic disease in others, such as SLE, remains unclear (17). This may also be the case for SVs, given the magnitude of GC treatments required in these patients and the relevance of associated complications (e.g., chronic kidney disease/end-stage kidney disease, sarcopenia, prevalence of solid-organ transplants, etc.).
The findings of the meta-regression univariate analysis showed that roughly 1% increase in the prevalence of osteoporosis was associated with each additional gram of cumulative GC dosage. Given that the mean cumulative dose in the studies was approximately 8 grams, this finding confirms the significant role of well-known GC-induced toxicity in this context. However, due to the scarcity of studies reporting other relevant factors associated with impaired bone health, the independent impact of SVs remains unclear. Recent vasculitis trials, including GIACTA for GCA (18), ADVOCATE (19) and PEXIVAS (20) for AAV, have focused on reducing glucocorticoid use to minimize treatment-related complications. The tapering glucocorticoid schemes of these trials have been included in the clinical guidelines and are now clinical practice. For instance, the dose of glucocorticoid recommended by the last update of the 2022 EULAR AAV recommendations (21) advise to target 5 mg prednisolone equivalent/day within 4-5 months as compared to 7.5-10 mg of previous guidelines of 2016 (22). By tapering glucocorticoid doses, these trials aim to reduce long-term risks including osteoporosis and other steroid-induced adverse effects. Our findings, obtained with a sensitivity analyses, do not suggest a decline in the reported prevalence of osteoporosis over time. On the contrary there appears to be an increasing trend, even after adjusting for cumulative GC dose. However, this finding should be interpreted with caution, as it may simply reflect a progressively greater awareness and interest in the comorbidities associated with SVs, including osteometabolic complication, thereby influencing reported prevalence rates.
Interestingly, from the present analysis, the mean prevalence of osteoporosis (14.64%) was lower than that of osteoporotic fractures (17.08%), with largely overlapping confidence intervals. This is a limitation and reflects the heterogeneity of the available studies for the systematic review, using different criteria to define osteoporosis and to collected this data, as subjects with history of fragility fractures are classified as osteoporotic regardless of BMD values (23). This led us to conclude that the published data on osteoporosis likely underestimate its true prevalence, which may indeed be higher in patients with SVs.
Additionally, within the general population, approximately 6% of men and 21% of women aged 50–84 years are classified as having osteoporosis according to WHO criteria (24). Therefore, it is unexpected for a high-risk population such as the present one to have a comparable prevalence of this condition, especially after observing a significantly increased OR for fractures in the SV population when compared to healthy controls (OR: 2.39). Similarly, when considering only vertebral fractures, data from the European Vertebral Osteoporosis Study (EVOS) have shown age-standardized population prevalence rates of 12.2% for men and 12.0% for women aged 50 to 79 years (25). Considering that the vast majority of included cohorts received large doses of GCs, with expected fracture events occurring in as many as 30–50% of patients receiving chronic GC therapy (26), led us to speculate that the diagnosis of osteoporosis is probably overlooked, and the fragility fractures largely underestimated. This could be explained by the lack of systematic assessment for bone mineral density impairment in SVs, of the recording of fracture events, and of a systematic search for subclinical vertebral fractures.
This study has strengths and limitations. The primary strength of the current study is that this is currently the first assessment on the state-of the art of the present topic, made with a well-established comprehensive approach (i.e. systematic review) to identify all pertinent studies that fulfil pre-defined inclusion criteria.
Among the limitations, we acknowledge that most eligible studies have relatively small sample sizes. Second, the group of SVs include patients with different forms of vasculitis, resulting in a very heterogeneous group in terms of vasculitis forms and consequently treatment types. To compensate this potential bias, we performed subanalyses based on vessels size, assessing the outcome on more homogeneous groups of SVs. Third, treatments changed significantly over time, including glucocorticoid regimens. Since our study extend on more decades, we performed subanalyses to assess for any time-trends, without significant impact on the outcomes.
Fourth, the methodologies employed for assessing BMD and investigating prevalent fractures are heterogenous and in general not always standardized. Third, the underreporting of significant risk factors such as family history, vitamin D status, kidney function, and other chronic conditions, as well as relevant biochemical data (i.e. bone turnover markers), may have impacted on our findings. Last, data focusing on fracture incidence over time following the diagnosis of SVs are lacking, and the article included in this specific analysis is low.
In conclusion, the current systematic review and meta-analysis provides a comprehensive overview of the prevalence of osteoporosis and osteoporotic fractures in SVs, indicating that SVs are associated with an increased risk of skeletal fragility. Moreover, this study points out a clear dose-dependence of cumulative GC and risk of osteoporosis.
It remains unclear to what extent this risk is attributed to the SV itself or influenced by subsequent treatments, advanced age, female predominance, and other comorbidities, suggesting the need for further research to investigate these aspects using rigorous methodology. Given the substantial burden associated with fragility fractures, it is imperative to prioritize pre-emptive screening, diagnosis, and treatment of osteoporosis in patients with SVs, including the identification of patients at risk. If all the subjects affected by SVs should or should not undergo to DXA and calcium-phosphate metabolism exam at baseline to avoid porotic fractures needs to be tested in future studies.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
AF: Conceptualization, Writing – original draft. AB: Conceptualization, Writing – review & editing. AM: Conceptualization, Formal Analysis, Methodology, Software, Writing – review & editing. GA: Conceptualization, Writing – review & editing. FP: Data curation, Writing – review & editing. DG: Project administration, Supervision, Writing – review & editing. RBi: Data curation, Visualization, Writing – review & editing. VM: Data curation, Writing – review & editing. MR: Project administration, Supervision, Writing – review & editing. DB: Data curation, Writing – review & editing. EB: Data curation, Writing – review & editing. IG: Data curation, Writing – review & editing. RBo: Writing – review & editing. OV: Supervision, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests, outside the submitted work: AF has received advisory board honoraria, consultancy fees and/or speaker fees from Boehringer Ingelheim. AB has received advisory board honoraria, consultancy fees and/or speaker fees from GSK. DG has received advisory board honoraria, consultancy fees and/or speaker fees from Amgen, Celgene, Eli-Lilly, MSD-Italia, Organon, UCB. MR has received advisory board honoraria, consultancy fees and/or speaker fees from Abiogen, Amgen, Abbvie, BMS, Celgene, Eli-Lilly, Galapagos, Grunenthal, MSD, Novartis, Pfizer, Sanofi, Sandoz, Theramex, UCB.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1545546/full#supplementary-material
ANCA, anti-neutrophil cytoplasmic antibody; AAV, ANCA-associated vasculitis BMD, bone mineral density; CHCC, Chapel-Hill Consensus Conference; CI, confidence interval; GCs, glucocorticoids GCA, Giant-cell arteritis; IgG4-RD, IgG4-related disease; LVV, large vessel vasculitis; MOOSE, Meta-analysis Of Observational Studies in Epidemiology; NOS, Newcastle-Ottawa Scale; OR, odds ratio; PRISMA, Preferred Reporting Items for Systematic Reviews and Meta-Analysis; RA, Rheumatoid Arthritis; SD, standard deviation; SLE, systemic lupus erythematosus; SpA, spondyloarthritis; SV, systemic vasculitis; TAK, Takayasu arteritis.
1. Jennette JC, Falk RJ, Andrassy K, Bacon PA, Churg J, Gross WL, et al. Nomenclature of systemic vasculitides. Proposal of an international consensus conference. Arthritis Rheumatol. (1994) 37:187–92. doi: 10.1002/art.1780370206
2. Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F, et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. (2013) 65:1–11. doi: 10.1002/art.37715
3. Box CD, Cronin O, Hauser B. The impact of high dose glucocorticoids on bone health and fracture risk in systemic vasculitides. Front Endocrinol (Lausanne). (2022) 13:806361. doi: 10.3389/fendo.2022.806361
4. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. (2021) 372:n71. doi: 10.1136/bmj.n71
5. Stroup DF, Berlin JA, Morton SC, Olkin I, Williamson GD, Rennie D, et al. Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA. (2000) 283:2008–12. doi: 10.1001/jama.283.15.2008
6. Higgins JPT, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. (2002) 21:1539–58. doi: 10.1002/sim.v21:11
7. Lin L, Chu H. Quantifying publication bias in meta-analysis. Biometrics. (2018) 74:785–94. doi: 10.1111/biom.12817
8. Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos Int. (2006) 17:1726–33. doi: 10.1007/s00198-006-0172-4
9. Son HJ, Park S-J, Kim J-K, Park J-S. Mortality risk after the first occurrence of osteoporotic vertebral compression fractures in the general population: A nationwide cohort study. PloS One. (2023) 18:e0291561. doi: 10.1371/journal.pone.0291561
10. Haentjens P, Magaziner J, Colón-Emeric CS, Vanderschueren D, Milisen K, Velkeniers B, et al. Meta-analysis: excess mortality after hip fracture among older women and men. Ann Intern Med. (2010) 152:380–90. doi: 10.7326/0003-4819-152-6-201003160-00008
11. Mundi S, Pindiprolu B, Simunovic N, Bhandari M. Similar mortality rates in hip fracture patients over the past 31 years. Acta Orthop. (2014) 85:54–9. doi: 10.3109/17453674.2013.878831
12. Adami G, Fassio A, Rossini M, Caimmi C, Giollo A, Orsolini G, et al. Osteoporosis in rheumatic diseases. Int J Mol Sci. (2019) 20. doi: 10.3390/ijms20235867
13. Fassio A, Atzeni F, Rossini M, D’Amico V, Cantatore F, Chimenti MS, et al. Osteoimmunology of spondyloarthritis. Int J Mol Sci. (2023) 24:14924. doi: 10.3390/ijms241914924
14. Gu C, Zhao R, Zhang X, Gu Z, Zhou W, Wang Y, et al. A meta-analysis of secondary osteoporosis in systemic lupus erythematosus: prevalence and risk factors. Arch Osteoporos. (2019) 15:1. doi: 10.1007/s11657-019-0667-1
15. Adami G. Regulation of bone mass in inflammatory diseases. Best Pract Res Clin Endocrinol Metab. (2022) 36:101611. doi: 10.1016/j.beem.2021.101611
16. Adami G, Fassio A, Rossini M, Benini C, Pistillo F, Viapiana O, et al. Bone loss in inflammatory rheumatic musculoskeletal disease patients treated with low-dose glucocorticoids and prevention by anti-osteoporosis medications. Arthritis Rheumatol. (2023) 75:1762–9. doi: 10.1002/art.v75.10
17. Adami G, Gatti D, Rossini M, Giollo A, Gatti M, Bertoldo F, et al. Risk of fracture in women with glucocorticoid requiring diseases is independent from glucocorticoid use: An analysis on a nation-wide database. Bone. (2024) 179:116958. doi: 10.1016/j.bone.2023.116958
18. Stone JH, Tuckwell K, DiMonaco S, Klearman M, Aringer M, Blockmans D, et al. Trial of tocilizumab in giant-cell arteritis. N Engl J Med. (2017) 377:317–28. doi: 10.1056/NEJMoa1613849
19. Jayne DRW, Merkel PA, Schall TJ, Bekker P, ADVOCATE Study Group. Avacopan for the treatment of ANCA-associated vasculitis. N Engl J Med. (2021) 384:599–609. doi: 10.1056/NEJMoa2023386
20. Walsh M, Merkel PA, Peh C-A, Szpirt WM, Puéchal X, Fujimoto S, et al. Plasma exchange and glucocorticoids in severe ANCA-associated vasculitis. N Engl J Med. (2020) 382:622–31. doi: 10.1056/NEJMoa1803537
21. Hellmich B, Sanchez-Alamo B, Schirmer JH, Berti A, Blockmans D, Cid MC, et al. EULAR recommendations for the management of ANCA-associated vasculitis: 2022 update. Ann Rheum Dis. (2024) 83:30–47. doi: 10.1136/ard-2022-223764
22. Yates M, Watts RA, Bajema IM, Cid MC, Crestani B, Hauser T, et al. EULAR/ERA-EDTA recommendations for the management of ANCA-associated vasculitis. Ann Rheum Dis. (2016) 75:1583–94. doi: 10.1136/annrheumdis-2016-209133
23. Kanis JA, Cooper C, Rizzoli R, Reginster J-Y, Scientific advisory board of the European Society for Clinical and Economic Aspects of Osteoporosis (ESCEO) and the committees of scientific advisors and national societies of the International Osteoporosis Foundation (IOF). European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Eur Guidance Diagnosis Manage Osteoporosis Postmenopausal Women Osteoporos Int. (2019) 30:3–44. doi: 10.1007/s00198-018-4704-5
24. Kanis JA, McCloskey EV, Johansson H, Oden A, Melton LJ, Khaltaev N. A reference standard for the description of osteoporosis. Bone. (2008) 42:467–75. doi: 10.1016/j.bone.2007.11.001
25. O’Neill TW, Felsenberg D, Varlow J, Cooper C, Kanis JA, Silman AJ. The prevalence of vertebral deformity in european men and women: the European Vertebral Osteoporosis Study. J Bone Miner Res. (1996) 11:1010–8. doi: 10.1002/jbmr.5650110719
26. Canalis E, Mazziotti G, Giustina A, Bilezikian JP. Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int. (2007) 18:1319–28. doi: 10.1007/s00198-007-0394-0
27. Albrecht K, Huscher D, Buttgereit F, Aringer M, Hoese G, Ochs W, et al. Long-term glucocorticoid treatment in patients with polymyalgia rheumatica, giant cell arteritis, or both diseases: results from a national rheumatology database. Rheumatol Int. (2018) 38:569–77. doi: 10.1007/s00296-017-3874-3
28. Andersson R, Rundgren A, Rosengren K, Bengtsson BA, Malmvall BE, Mellström D. Osteoporosis after long-term corticosteroid treatment of giant cell arteritis. J Intern Med. (1990) 227:391–5. doi: 10.1111/j.1365-2796.1990.tb00177.x
29. Antonini L, Dumont A, Lavergne A, Castan P, Barakat C, Gallou S, et al. Real-life analysis of the causes of death in patients consecutively followed for giant cell arteritis in a French centre of expertise. Rheumatol (Oxford). (2021) 60:5080–8. doi: 10.1093/rheumatology/keab222
30. Bezerra MC, Calomeni GD, Caparbo VF, Gebrim ES, Rocha MS, Pereira RMR. Low bone density and low serum levels of soluble RANK ligand are associated with severe arterial calcification in patients with Takayasu arteritis. Rheumatol (Oxford). (2005) 44:1503–6. doi: 10.1093/rheumatology/kei045
31. Bicer A, Tursen U, Kaya TI, Ozer C, Camdeviren H, Ikizoglu G, et al. Bone mineral density in patients with Behçet’s disease. Rheumatol Int. (2004) 24:355–8. doi: 10.1007/s00296-003-0381-5
32. Boomsma MM, Stegeman CA, Kramer AB, Karsijns M, Piers DA, Tervaert JWC. Prevalence of reduced bone mineral density in patients with anti-neutrophil cytoplasmic antibody associated vasculitis and the role of immunosuppressive therapy: a cross-sectional study. Osteoporos Int. (2002) 13:74–82. doi: 10.1007/s198-002-8341-z
33. Yoo B-W, Jung SM, Song JJ, Park Y-B, Lee S-W. Prevalence of osteopenia in drug-naive patients with antineutrophil cytoplasmic antibody-associated vasculitis: A monocentric study. J Clin Rheumatol. (2021) 27:e330–5. doi: 10.1097/RHU.0000000000001413
34. Cetin B, Cetin EA, Arikan H, Velioglu A, Alibaz-Oner F, Direskeneli H, et al. FRAX scores are increased in patients with ANCA-associated vasculitis. Int Urol Nephrol. (2021) 53:2333–9. doi: 10.1007/s11255-021-02857-z
35. de Boysson H, Barakat C, Dumont A, Boutemy J, Martin Silva N, Maigné G, et al. Tolerance of glucocorticoids in giant cell arteritis: a study of patient-reported adverse events. Rheumatol (Oxford). (2022) 61:3567–75. doi: 10.1093/rheumatology/keab921
36. Englund M, Merkel PA, Tomasson G, Segelmark M, Mohammad AJ. Comorbidities in patients with antineutrophil cytoplasmic antibody-associated vasculitis versus the general population. J Rheumatol. (2016) 43:1553–8. doi: 10.3899/jrheum.151151
37. Elgengehy FT, Gamal SM, Sobhy N, Siam I, Soliman AM, Elhady GW, et al. Vasculitis damage index in Behçet’s disease. Adv Rheumatol. (2021) 61:33. doi: 10.1186/s42358-021-00193-5
38. Faurschou M, Baslund B, Obel N. Pronounced risk of fractures among elderly men affected by granulomatosis with polyangiitis. J Rheumatol. (2015) 42:1667–71. doi: 10.3899/jrheum.141566
39. Harper L, Morgan MD, Walsh M, Hoglund P, Westman K, Flossmann O, et al. Pulse versus daily oral cyclophosphamide for induction of remission in ANCA-associated vasculitis: long-term follow-up. Ann Rheum Dis. (2012) 71:955–60. doi: 10.1136/annrheumdis-2011-200477
40. Henriquez S, Dunogué B, Porcher R, Régent A, Cohen P, Berezne A, et al. Handgrip strength is a comorbidity marker in systemic necrotizing vasculitides and predicts the risk of fracture and serious adverse events. Rheumatol (Oxford). (2020) 59:2581–90. doi: 10.1093/rheumatology/kez680
41. Itabashi M, Takei T, Moriyama T, Shiohira S, Shimizu A, Tsuruta Y, et al. Long-term damage assessment in patients with microscopic polyangiitis and renal-limited vasculitis using the Vasculitis Damage Index. Mod Rheumatol. (2014) 24:112–9. doi: 10.3109/14397595.2013.852836
42. Kermani TA, Sreih AG, Cuthbertson D, Carette S, Hoffman GS, Khalidi NA, et al. Evaluation of damage in giant cell arteritis. Rheumatol (Oxford). (2018) 57:322–8. doi: 10.1093/rheumatology/kex397
43. Lanzillotta M, Campochiaro C, Mancuso G, Ramirez GA, Capurso G, Falconi M, et al. Clinical phenotypes of IgG4-related disease reflect different prognostic outcomes. Rheumatol (Oxford). (2020) 59:2435–42. doi: 10.1093/rheumatology/keaa221
44. Les I, Pijoán JI, Rodríguez-Álvarez R, Ruiz-Irastorza G, Martínez-Berriotxoa A. Effectiveness and safety of medium-dose prednisone in giant cell arteritis: a retrospective cohort study of 103 patients. Clin Exp Rheumatol. (2015) 33:S–90-97.
45. Mahr A, Hachulla E, de Boysson H, Guerroui N, Héron E, Vinzio S, et al. Presentation and real-world management of giant cell arteritis (Artemis study). Front Med (Lausanne). (2021) 8:732934. doi: 10.3389/fmed.2021.732934
46. Alba MA, García-Martínez A, Prieto-González S, Tavera-Bahillo I, Corbera-Bellalta M, Planas-Rigol E, et al. Relapses in patients with giant cell arteritis: prevalence, characteristics, and associated clinical findings in a longitudinally followed cohort of 106 patients. Med (Baltimore). (2014) 93:194–201. doi: 10.1097/MD.0000000000000033
47. Mateo L, Nolla JM, Rozadilla A, Rodríguez-Moreno J, Niubó R, Valverde J, et al. Bone mineral density in patients with temporal arteritis and polymyalgia rheumatica. J Rheumatol. (1993) 20:1369–73.
48. Miyano S, Michihata N, Sada K-E, Uda K, Matsui H, Fushimi K, et al. Comparison of fracture risk between proton pump inhibitors and histamine-2 receptor antagonists in ANCA-associated vasculitis patients: a nested case-control study. Rheumatol (Oxford). (2021) 60:1717–23. doi: 10.1093/rheumatology/keaa594
49. Mohammad AJ, Englund M, Turesson C, Tomasson G, Merkel PA. Rate of comorbidities in giant cell arteritis: A population-based study. J Rheumatol. (2017) 44:84–90. doi: 10.3899/jrheum.160249
50. Myles AB. Polymyalgia rheumatica and giant cell arteritis: a seven-year survey. Rheumatol Rehabil. (1975) 14:231–5. doi: 10.1093/rheumatology/14.4.231
51. Palmowski A, Wiebe E, Muche B, Hermann S, Dejaco C, Matteson EL, et al. Glucocorticoids are not associated with bone mineral density in patients with polymyalgia rheumatica, giant cell arteritis and other vasculitides-cross-sectional baseline analysis of the prospective Rh-GIOP cohort. Cells. (2022) 11:536. doi: 10.3390/cells11030536
52. Palmowski A, Akahoshi M, Muche B, Boyadzhieva Z, Hermann S, Terao C, et al. No association between methotrexate and impaired bone mineral density in a cohort of patients with polymyalgia rheumatica, giant cell arteritis, granulomatosis with polyangiitis and other vasculitides-a cross-sectional analysis with dose-response analyses. Rheumatol Int. (2023) 43:903–9. doi: 10.1007/s00296-023-05286-6
53. Paskins Z, Whittle R, Sultan AA, Muller S, Blagojevic-Bucknall M, Helliwell T, et al. Risk of fracture among patients with polymyalgia rheumatica and giant cell arteritis: a population-based study. BMC Med. (2018) 16:4. doi: 10.1186/s12916-017-0987-1
54. Perrineau S, Ghesquière T, Charles P, Paule R, Samson M, Gayraud M, et al. A French cohort of patients with giant cell arteritis: glucocorticoid treatment and its associated side effects. Clin Exp Rheumatol. (2021) 39 Suppl 129:155–60. doi: 10.55563/clinexprheumatol/0nd4kk
55. Petri H, Nevitt A, Sarsour K, Napalkov P, Collinson N. Incidence of giant cell arteritis and characteristics of patients: data-driven analysis of comorbidities. Arthritis Care Res (Hoboken). (2015) 67:390–5. doi: 10.1002/acr.22429
56. Quartuccio L, Isola M, Bruno D, Treppo E, Gigante L, Angelotti F, et al. Treatment strategy introducing immunosuppressive drugs with glucocorticoids ab initio or very early in giant cell arteritis: A multicenter retrospective controlled study. J Transl Autoimmun. (2020) 3:100072. doi: 10.1016/j.jtauto.2020.100072
57. Robson J, Doll H, Suppiah R, Flossmann O, Harper L, Höglund P, et al. Damage in the anca-associated vasculitides: long-term data from the European vasculitis study group (EUVAS) therapeutic trials. Ann Rheum Dis. (2015) 74:177–84. doi: 10.1136/annrheumdis-2013-203927
58. Sada K-E, Ohashi K, Asano Y, Hayashi K, Morishita M, Watanabe H, et al. Treatment-related damage in elderly-onset ANCA-associated vasculitis: safety outcome analysis of two nationwide prospective cohort studies. Arthritis Res Ther. (2020) 22:236. doi: 10.1186/s13075-020-02341-6
59. Samson M, Puéchal X, Devilliers H, Ribi C, Cohen P, Bienvenu B, et al. Long-term follow-up of a randomized trial on 118 patients with polyarteritis nodosa or microscopic polyangiitis without poor-prognosis factors. Autoimmun Rev. (2014) 13:197–205. doi: 10.1016/j.autrev.2013.10.001
60. Sarica SH, Gallacher PJ, Dhaun N, Sznajd J, Harvie J, McLaren J, et al. Multimorbidity in antineutrophil cytoplasmic antibody-associated vasculitis: results from a longitudinal, multicenter data linkage study. Arthritis Rheumatol. (2021) 73:651–9. doi: 10.1002/art.41557
61. Schmidt WA, Moll A, Seifert A, Schicke B, Gromnica-Ihle E, Krause A. Prognosis of large-vessel giant cell arteritis. Rheumatol (Oxford). (2008) 47:1406–8. doi: 10.1093/rheumatology/ken258
62. Spiera RF, Mitnick HJ, Kupersmith M, Richmond M, Spiera H, Peterson MG, et al. A prospective, double-blind, randomized, placebo controlled trial of methotrexate in the treatment of giant cell arteritis (GCA). Clin Exp Rheumatol. (2001) 19:495–501.
63. Tekin NS, Ozdolap S, Sarikaya S, Esturk E, Gumustas S. Bone mineral density and bone turnover markers of patients with Behçet’s disease. J Eur Acad Dermatol Venereol. (2007) 21:25–9. doi: 10.1111/j.1468-3083.2006.01845.x
64. Tuckwell K, Collinson N, DiMonaco S, Klearman M, Blockmans D, Brouwer E, et al. Newly diagnosed vs. relapsing giant cell arteritis: Baseline data from the GiACTA trial. Semin Arthritis Rheumatol. (2017) 46:657–64. doi: 10.1016/j.semarthrit.2016.11.002
65. Wilson JC, Sarsour K, Collinson N, Tuckwell K, Musselman D, Klearman M, et al. Incidence of outcomes potentially associated with corticosteroid therapy in patients with giant cell arteritis. Semin Arthritis Rheumatol. (2017) 46:650–6. doi: 10.1016/j.semarthrit.2016.10.001
66. Wilson JC, Sarsour K, Collinson N, Tuckwell K, Musselman D, Klearman M, et al. Serious adverse effects associated with glucocorticoid therapy in patients with giant cell arteritis (GCA): A nested case-control analysis. Semin Arthritis Rheumatol. (2017) 46:819–27. doi: 10.1016/j.semarthrit.2016.11.006
Keywords: vasculitis < rheumatic diseases, osteoporosis, fractures - bone, giant cell (temporal) arteritis, ANCA - associated vasculitis, glucocorticoids, DEXA - duel-energy X-ray absorptiometry, Takayasu ‘ s arteritis
Citation: Fassio A, Berti A, Mantovani A, Adami G, Pollastri F, Gatti D, Bixio R, Messina V, Rossini M, Bertelle D, Bertoldo E, Galvagni I, Bortolotti R and Viapiana O (2025) Osteoporosis and fractures in systemic vasculitides: a systematic review and meta-analysis. Front. Immunol. 16:1545546. doi: 10.3389/fimmu.2025.1545546
Received: 15 December 2024; Accepted: 27 February 2025;
Published: 17 March 2025.
Edited by:
Davide Firinu, University of Cagliari, ItalyReviewed by:
Simone Negrini, University of Turin, ItalyCopyright © 2025 Fassio, Berti, Mantovani, Adami, Pollastri, Gatti, Bixio, Messina, Rossini, Bertelle, Bertoldo, Galvagni, Bortolotti and Viapiana. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Francesco Pollastri, ZnJhbnBvbGxhc3RyaUB5YWhvby5pdA==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.