
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Immunol. , 19 March 2025
Sec. Autoimmune and Autoinflammatory Disorders : Autoimmune Disorders
Volume 16 - 2025 | https://doi.org/10.3389/fimmu.2025.1473190
Immune thrombocytopenia(ITP)is a common clinical manifestation of systemic lupus erythematosus(SLE). Drug therapy includes glucocorticoids(GCs),disease-modifying anti-rheumatic drugs (DMARDs) and biologics. Refractory thrombocytopenia can be life-threatening, and the use of effective medications plays a crucial role in disease improvement. Here, we report a case of ITP secondary to SLE. The use of dexamethasone(DEX), cyclosporine A(CsA), and hetrombopag resulted in drug-induced liver injury. Subsequently, telitacicept was chosen and successfully controlled the patient’s condition. It suggests that telitacicept may be a new treatment option for refractory SLE-ITP.
Systemic lupus erythematosus (SLE) is a chronic autoimmune disease that can cause damage to multiple organs, with hematological involvement being a common systemic manifestation. Studies have shown that B-cell hyperactivity and the production of anti-platelet-associated antibodies are the main mechanisms of immune thrombocytopenia(ITP) secondary to SLE (1). Conventional treatments are based on glucocorticoids(GCs) and immunologic agents, among others. Here, we describe a case of an SLE-ITP patient, who developed hepatic impairment after various medications treatments and was successfully treated with a combination of GCs and telitacicept. The relevant literatures are discussed to reveal the disease characteristics and potential therapeutic targets of SLE-ITP.
We searched the literature through major scientific databases, including PubMed, ScienceDirect, SpringerLink, and Web of Science,and using the keywords:”telitacicept”. We found only 12 case reports articles reporting telitacicept treatment. The search results are shown in Table 1.
On June 3, 2023, a 31-year-old female patient with cyanotic skin and ecchymosis for three days was admitted to the hematology department. The patient had no special previous medical history. Laboratory tests showed platelet(PLT) counts of 4×10^9/L, positive anti-nuclear antibodies (ANA) (1:1000 granular type),positive anti-SSA/Ro60 antibodies, positive anti-u1-SnRNP antibodies. Liver and kidney function, complement, immunoglobulin, rheumatoid factor, haemagglutination function, AT-III activity test, thromboelastography test, antiphospholipid antibody, anti-ADAMTS13 antibody, and antiplatelet antibody were normal. Bone marrow (BM) cytology revealed increased nucleated cells, impaired maturation of megakaryocytes (MKs), and decreased PLTs.Treatment included PLT transfusion, subcutaneous injection of recombinant human thrombopoietin (TPO), oral dexamethasone (DEX; 40mg/day for 3 days), and intravenous immunoglobulin (IVIG) (20g/day for 2 days).
After treatment, the patient’s PLT counts were rechecked at 86×10^9/L on June 8th. Then, she was transferred to the Department of Rheumatology and Immunology for further treatment, considering thrombocytopenia secondary to connective tissue disease.After transferring to our department, she received methylprednisolone (MP) 40mg qd and hydroxychloroquine (HCQ) 0.2g bid. On June 15, the PLT counts were normal, but alanine aminotransferase (ALT) and aspartate aminotransferase (AST) were significantly elevated (ALT471.70U/L, AST90.72U/L).Analyzing the cause of acute hepatic impairment, the possibility of drug induced liver injury was deemed high. Upon reviewing the patient’s medications, it was discovered that oral DEX 40mg for 3 days was likely the cause of the hepatic damage. As a result, other suspected medications were discontinued, and only MP along with basic hepatoprotective drugs (magnesium isoglycyrrhizinate 200 mg/day and glutathione 1800mg/day) were continued.On July 3rd, the liver function returned to normal, but the PLT count decreased again to 6×10^9/L. Therefore, MP was given to increase the dosage to 200mg qd, and interleukin-11, PLT transfusion and IVIG 10g infusion were also given.The next day, hetrombopag (2.5mg qd) and CsA (50mg bid) orally were added.
On July 7, the PLT counts were repeated at 15 x 10^9/L, however, ALT and AST were again elevated (ALT 454.60 U/L, AST 116.91 U/L).After further exclusion of autoimmune liver disease and viral hepatitis, the patient was evaluated for another short-term sharp increase in liver enzymes, and drug induced liver injury was still suspected. The decision was made to stop CsA and hetrombopag, enhance liver protection and enzyme-lowering therapy, and continue the treatment of MP (80mg qd) and interleukin-11.Considering the patient’s recurrent PLT decrease, despite the effectiveness of GCs and IVIG treatments, severe liver damage occurred when CsA, hetrombopag and other drugs were added. When the GCs was slightly reduced, there was a significant drop in PLTs. In light of the patient’s hepatic impairment from multiple drugs, it was decided to refrain from adding additional conventional disease-modifying antirheumatic drugs (cDMARDs) at this time. Therefore, it was recommended that the patient try using telitacicept. After she agreed, screening was conducted to exclude infectious diseases. On July 26th, PLT counts of 41 x 10^9/L and normal liver function (ALT 51.26 U/L, AST 26.60 U/L)were rechecked before treatment. On July 27th she was started on telitacicept 160 mg subcutaneously and on August 1st she was retested for PLTs 87 x 10^9/L, ALT 43.52 U/L,AST 15.49 U/L.She continued regular telitacicept 160 mg once weekly in combination with MP orally.on August 28, the PLT counts were repeated at 272 x 10^9/L and liver function was normal (Figure 1).
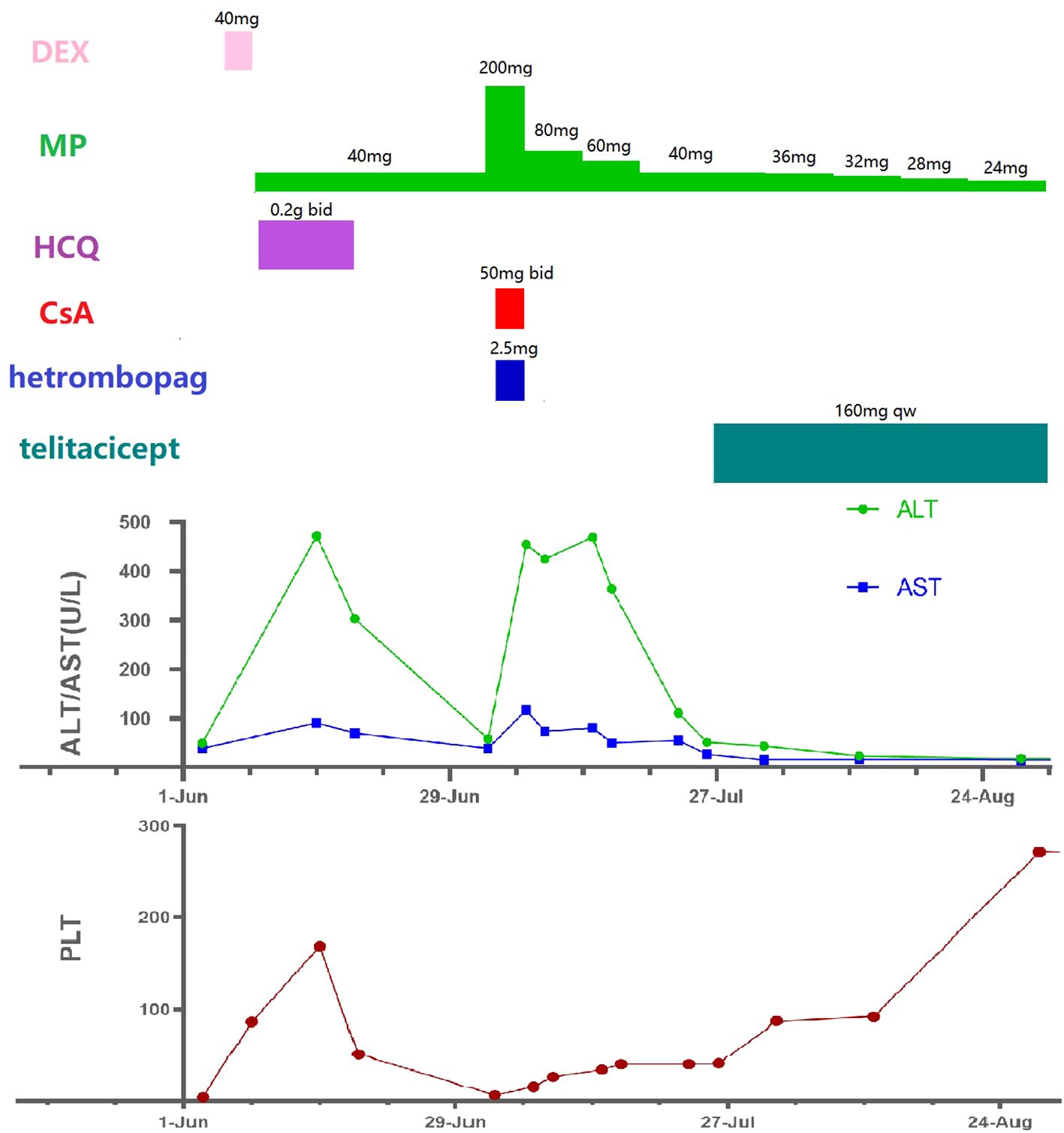
Figure 1. Medication process and laboratory indicators(all timelines are the same). DEX, dexamethasone; MP, methylprednisolone; HCQ, hydroxychloroquine; CsA, cyclosporine A; ALT, alanine aminotransferase; AST, aspartate aminotransferase; PLT, platelet.
In December, the patient discontinued MP and telitacicept on her own. Half a month later, she developed swelling and pain in both elbows and ankles, and a facial rash, which fulfilled the 2019 EULAR/ACR classification criteria for SLE (14), and led to the diagnosis of SLE. On January 18, the patient resumed the use of MP 8mg qd, HCQ 0.2g bid, and telitacicept 160mgqw, with symptomatic relief. On February 25, PLT204×10^9/L was rechecked, and liver and renal functions were normal.
In patients with SLE,the immune response mediates dysregulation of PLT production and destruction, which in turn leads to ITP (15). Lupus patients can produce a large number of antiplatelet-related antibodies, including anti-GPIbα, anti-gpia/IIa, anti-gpIIb/IIIa, anti-PAIgG, anti-gpib/IX, anti-CD40L antibodies, antiphospholipid antibodies, anti-c-mpl and anti-TPO antibodies (1). The cause of excessive antibody production may be due to the high expression of the stimulatory factors B-cell activation factor (BAFF) and a proliferation-inducing ligand (APRIL), leading to overactivation of B cells (16), and increased autoantibodies on PLT membranes caused by autoimmune B cell clearance disorders. BAFF (also known as B-lymphocyte stimulator,BLyS)and APRIL are members of the tumor necrosis factor (TNF) family (17). They share two receptors, transmembrane activator or calcium-modulating cyclophilin ligand-interactor (TACI) and B cell maturation antigen (BCMA), and BLyS can also bind to a third receptor, B cell activating factor receptor (BAFFR) (18). The process of BAFF/APRIL binding to its receptor is crucial for the proliferation, differentiation and maturation of B cells.It also influences the formation of live and long-lived plasma cells in the spleen (19). PLT antigens in the spleen are mainly delivered to T helper (Th) cells by dendritic cells (DCs)and macrophages, causing increased secretion of BAFF, This further operates through receptors to induce typical and atypical NF-κB activation, which in turn leads to B-cell maturation.The typical NF-κB pathway is activated by TACI receptors, and the sustained expression of Blimp-1 by B cells leads that B cells differentiate into autoantibodies-secreting plasma cells (20) and long-lived plasma cells (21), which causes the secretion of large amounts of pathologic IgG by autoreactive B cells.Furthermore, the high expression of BAFF in the sera of patients with active SLE induces an increase in the proportion of immature regulatory B cells (Bregs), which leads to a decrease in the proportion of mature Bregs subpopulations, thereby exacerbating disease activity (22).
The main drugs used in the clinic for the treatment of SLE-ITP are GCs, IVIG, cDMARDs, biologic agents and thrombopoietin-receptor agonists. For refractory thrombocytopenia, biologics like rituximab (RTX) are also a viable choice. In pharmacology, RTX depletes CD20-expressing B cells instead of Pro-B cells or plasma cells. It has been found that B-cell depletion leads to increased levels of BAFF in the spleen and serum, which contributes to plasma cell survival and differentiation into long-lived plasma cells (23). In BM hyperactivation of BAFF/APRIL has also been found (24), and in the spleen BAFF/APRIL can also increase long-lived plasma cells (25). This may account for some of the RTX treatment failures.
With the use of belimumab, there are continued case reports (26) of effective treatment of SLE-ITP.Sequential treatment of RTX and belimumab decreases the formation of long-lived plasma cells, enabling long-term disease management, offering a new approach for treating SLE-ITP (26). Belimumab is a recombinant humanized IgG2-λ monoclonal antibody, which can bind to BAFF to inhibit the differentiation, survival and antibody secretion of naive B lymphocytes, memory cells, marginal B lymphocytes, follicular B lymphocytes and plasma cells (27). However, because APRIL can still bind to TACI and BCMA, belimumab treatment is more commonly used to inhibit naive B lymphocytes (28), resulting in a relatively weak effect on plasma cells and a less rapid response to therapy for life-threatening thrombocytopenia.
In 2021, telitacicept was approved as a treatment for active SLE in China. It is a new type of fusion protein biologic that is not a traditional monoclonal antibody (18). It is fusion protein comprising a TACI (the common ligand of BLys and APRIL) receptor fused to the fragment crystallizable (Fc) domain of human IgG, which can simultaneously block the binding of BLyS and APRIL to TACI, inhibit plasma cell differentiation and survival, and affect the secretion of autoantibodies and pro-inflammatory cytokines (29). Because APRIL regulates the survival and function of long-lived plasma cells, it offers superior control of disease activity compared to belimumab and achieves multi-stage, multifaceted inhibition of B-cells.
With clinical application, telitacicept has demonstrated positive therapeutic outcomes. Wu et al. have reported that a phase 2b clinical trial of telitacicept in SLE demonstrated a notably higher percentage of patients achieving an SRI-4 response compared to the placebo group at week 48 (30). A group of 30 LN patients reported by Huang et al., with poor response or adverse reaction to conventional GCs showed a significant reduction in disease severity after receiving telitacicept treatment (31). Fan et al. described a case of lupus hepatitis in a patient with persistent liver damage despite treatment with various cDMARDs and belimumab. The patient’s liver function returned to normal after switching to telitacicept for one month (13). In addition to SLE, telitacicept has been tried for other immune system disorders, including primary sjögren’s syndrome, systemic vasculitis, neuromyelitis optica, MG, IgG4-associated diseases, and many other immune system disorders (5, 12, 32–34).
Our patient had persistent severely low platelets and a very high risk of bleeding, necessitating mega doses of MP to maintain platelets at a relatively safe level. Based on past experience, we introduced CsA and hetrombopag in the hope of raising platelets and helping MP decrease. However, the patient experienced recurrent severe drug-induced hepatitis. Inflammation, oxidative stress, and apoptosis for a variety of reasons (35), resulting in hepatic impairment frequently seen in treatment with DMARDs (36). Considering the drug-induced liver injury and time to take effect, we did not choose rituximab, belimumab, or other cDMARDs.Instead, we tried combining MP directly with telitacicept and achieved a positive therapeutic outcome. Platelet levels normalized rapidly without inducing liver damage, ensuring the patient’s safety.
To our knowledge, this is the first case to report SLE-ITP treated with only GCs in combination with telitacicept. Given that severe thrombocytopenia can be life-threatening, the selection of effective drugs can significantly aid in achieving disease remission.This case suggests that telitacicept may be a new treatment option when conventional treatment for SLE-ITP is ineffective.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
The studies involving humans were approved by the Institutional Review Boards at Affiliated Hospital of Jiujiang University. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
XL: Writing – original draft, Writing – review & editing. YF: Writing – review & editing. HY: Supervision, Writing – review & editing. HZ: Supervision, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Jiang Y, Cheng Y, Ma S, Li T, Chen Z, Zuo X, et al. Systemic lupus erythematosus-complicating immune thrombocytopenia: From pathogenesis to treatment. J Autoimmun. (2022) 132:102887. doi: 10.1016/j.jaut.2022.102887
2. Ma X, Fu X, Cui B, Lin H. Telitacicept for recalcitrant cutaneous manifestations of systemic lupus erythematosus: A case report and review of the literature. Tohoku J Exp Med. (2022) 258:219–23. doi: 10.1620/tjem.2022.J074
3. Zhang L, Jin H, Wang D, Wang Y. Case report: Successful treatment of refractory membranous nephropathy with telitacicept. Front Immunol. (2023) 14:1268929. doi: 10.3389/fimmu.2023.1268929
4. Chen JW, Zhan JY, Liang SP, Wang XD, Lin CS, Xu Q. A patient with refractory proliferative lupus nephritis treated with telitacicept: A case report. Int J Rheum Dis. (2023) 26:1417–21. doi: 10.1111/1756-185X.14752
5. Huang L, Lin W, Liu Y, Zhu J, Li Y, Zheng Z, et al. Combination treatment with telitacicept, cyclophosphamide and glucocorticoids for severe Granulomatous polyangiitis: a case report and literature review. Front Immunol. (2023) 14:1298650. doi: 10.3389/fimmu.2023.1298650
6. Wang JL, Sun YL, Kang Z, Zhang SK, Yu CX, Zhang W, et al. Anti-phospholipase A2 receptor-associated membranous nephropathy with human immunodeficiency virus infection treated with telitacicept: A case report. World J Clin Cases. (2023) 11:5309–15. doi: 10.12998/wjcc.v11.i22.5309
7. Guo Q, Huang Y, Wang F, Fang L. Case Report: Telitacicept in severe myasthenia gravis: a case study with multiple autoantibodies. Front Immunol. (2023) 14:1270011. doi: 10.3389/fimmu.2023.1270011
8. Ren Y, Chen S, Yang H. Case Report: Telitacicept in treating a patient with NF155+ autoimmune nodopathy: a successful attempt to manage recurrent elevated sero-anti-NF155 antibodies. Front Immunol. (2023) 14:1279808. doi: 10.3389/fimmu.2023.1279808
9. Tian M, Tang L. Good efficacy achieved by telitacicept, corticosteroids and immunosuppressants in the treatment of SLE combined with MOG-AD. Rheumatol Adv Pract. (2023) 7:rkad088. doi: 10.1093/rap/rkad088
10. Li S, Deng S, Wen S, Peng S, Jiang N, Li B, et al. Telitacicept treatment refractory lupus nephritis: A case report. Case Rep Nephrol Dial. (2024) 14:42–7. doi: 10.1159/000538033
11. Huang L, Qian G, Zhang H, Li Q, Chen L, Tang X, et al. Efficacy of Telitacicept in a systemic lupus erythematosus patient with suboptimal response to Belimumab: A case report. Lupus. (2024) 33:172–5. doi: 10.1177/09612033231221911
12. Zhang Z, Wang Z, Du X, Huang X, Zhang Y. Refractory generalized myasthenia gravis treated successfully with telitacicept: two cases report. J Neurol. (2024) 271:584–8. doi: 10.1007/s00415-023-12036-y
13. Fan Q, Ji H, Liu Y, Jia C, Zou L, Yang H. Refractory lupus hepatitis successfully treated with telitacicept who failed to belimumab: A case report and literature review. Lupus. (2024) 33:414–9. doi: 10.1177/09612033241233021
14. Aringer M, Costenbader K, Daikh D, Brinks R, Mosca M, Ramsey-Goldman R, et al. European league against rheumatism/American college of rheumatology classification criteria for systemic lupus erythematosus. Arthritis Rheumatol. (2019) 71:1400–12. doi: 10.1002/art.40930
15. Xu XR, Zhang D, Oswald BE, Carrim N, Wang X, Hou Y, et al. Platelets are versatile cells: New discoveries in hemostasis, thrombosis, immune responses, tumor metastasis and beyond. Crit Rev Clin Lab Sci. (2016) 53:409–30. doi: 10.1080/10408363.2016.1200008
16. Tangye SG, Bryant VL, Cuss AK, Good KL. BAFF, APRIL and human B cell disorders. Semin Immunol. (2006) 18:305–17. doi: 10.1016/j.smim.2006.04.004
17. Jonsson MV, Szodoray P, Jellestad S, Jonsson R, Skarstein K. Association between circulating levels of the novel TNF family members APRIL and BAFF and lymphoid organization in primary Sjögren’s syndrome. J Clin Immunol. (2005) 25:189–201. doi: 10.1007/s10875-005-4091-5
18. Dhillon S. Telitacicept: first approval. Drugs. (2021) 81:1671–5. doi: 10.1007/s40265-021-01591-1
19. Kamhieh-Milz J, Ghosoun N, Sterzer V, Salama A. Effect of glucocorticoid treatment on BAFF and APRIL expression in patients with immune thrombocytopenia (ITP). Clin Immunol. (2018) 188:74–80. doi: 10.1016/j.clim.2017.12.010
20. Tsuji S, Cortesão C, Bram RJ, Platt JL, Cascalho M. TACI deficiency impairs sustained Blimp-1 expression in B cells decreasing long-lived plasma cells in the bone marrow. Blood. (2011) 118:5832–9. doi: 10.1182/blood-2011-05-353961
21. Kuwana M, Okazaki Y, Ikeda Y. Splenic macrophages maintain the anti-platelet autoimmune response via uptake of opsonized platelets in patients with immune thrombocytopenic purpura. J Thromb Haemost. (2009) 7:322–9. doi: 10.1111/j.1538-7836.2008.03161.x
22. Salazar-Camarena DC, Ortiz-Lazareno PC, Cruz A, Oregon-Romero E, Machado-Contreras JR, Muñoz-Valle JF, et al. Association of BAFF, APRIL serum levels, BAFF-R, TACI and BCMA expression on peripheral B-cell subsets with clinical manifestations in systemic lupus erythematosus. Lupus. (2016) 25:582–92. doi: 10.1177/0961203315608254
23. Mahévas M, Michel M, Vingert B, Moroch J, Boutboul D, Audia S, et al. Emergence of long-lived autoreactive plasma cells in the spleen of primary warm auto-immune hemolytic anemia patients treated with rituximab. J Autoimmun. (2015) 62:22–30. doi: 10.1016/j.jaut.2015.05.006
24. Yu TS, Wang HY, Zhao YJ, Yu YF, Hou Y, Liu S, et al. Abnormalities of bone marrow B cells and plasma cells in primary immune thrombocytopenia. Blood Adv. (2021) 5:4087–101. doi: 10.1182/bloodadvances.2020003860
25. Godeau B. B-cell depletion in immune thrombocytopenia. Semin Hematol. (2013) 50 Suppl 1:S75–82. doi: 10.1053/j.seminhematol.2013.03.014
26. Hanai S, Kobayashi Y, Harama K, Ito R, Mabuchi N, Nakagomi D. Early glucocorticoid withdrawal by combining rituximab with belimumab in two patients with lupus-associated thrombocytopenia. Rheumatol (Oxford). (2023) 62:e315–6. doi: 10.1093/rheumatology/kead171
27. Wise LM, Stohl W. Belimumab and rituximab in systemic lupus erythematosus: A tale of two B cell-targeting agents. Front Med (Lausanne). (2020) 7:303. doi: 10.3389/fmed.2020.00303
28. Regola F, Piantoni S, Lowin T, Archetti S, Reggia R, Kumar R, et al. Association between changes in BLyS levels and the composition of B and T cell compartments in patients with refractory systemic lupus erythematosus treated with belimumab. Front Pharmacol. (2019) 10:433. doi: 10.3389/fphar.2019.00433
29. Cornelis R, Chang HD, Radbruch A. Keeping up with the stress of antibody production: BAFF and APRIL maintain memory plasma cells. Curr Opin Immunol. (2021) 71:97–102. doi: 10.1016/j.coi.2021.06.012
30. Wu D, Li J, Xu D, Merrill JT, van Vollenhoven RF, Liu Y, et al. Telitacicept in patients with active systemic lupus erythematosus: results of a phase 2b, randomised, double-blind, placebo-controlled trial. Ann Rheum Dis. (2024) 83:475–87. doi: 10.1136/ard-2023-224854
31. Huang X, Lin F, Chen H. Efficacy and safety of telitacicept in patients with lupus nephritis: a single-center, real-world retrospective study. Clin Exp Nephrol. (2024) 28(9):902–9. doi: 10.1007/s10157-024-02501-x
32. Xu D, Fang J, Zhang S, Huang C, Huang C, Qin L, et al. Efficacy and safety of telitacicept in primary Sjögren’s syndrome: a randomized, double-blind, placebo-controlled, phase 2 trial. Rheumatol (Oxford). (2024) 63:698–705. doi: 10.1093/rheumatology/kead265
33. Ding J, Jiang X, Cai Y, Pan S, Deng Y, Gao M, et al. Telitacicept following plasma exchange in the treatment of subjects with recurrent neuromyelitis optica spectrum disorders: A single-center, single-arm, open-label study. CNS Neurosci Ther. (2022) 28:1613–23. doi: 10.1111/cns.13904
34. Cai S, Hu Z, Chen Y, Chen Y, Ming B, Gao R, et al. BLyS/APRIL dual inhibition for IgG4-RD: a prospective single-arm clinical trial of telitacicept. Ann Rheum Dis. (2023) 82:881–3. doi: 10.1136/ard-2022-223529
35. El-Magd MA, Zedan AMG, Zidan NS, Sakran MI, Bahattab O, Oyouni AAA, et al. Avocado seeds-mediated alleviation of cyclosporine A-induced hepatotoxicity involves the inhibition of oxidative stress and proapoptotic endoplasmic reticulum stress. Molecules. (2022) 27:7859. doi: 10.3390/molecules27227859
Keywords: immune thrombocytopenia, systemic lupus erythematosus, drug induced liver injury, telitacicept, refractory
Citation: Li X, Fu Y, Yin H and Zhu H (2025) Successful treatment of refractory systemic lupus erythematosus-associated immune thrombocytopenia with drug-induced liver injury with telitacicept: a case report and review. Front. Immunol. 16:1473190. doi: 10.3389/fimmu.2025.1473190
Received: 01 August 2024; Accepted: 27 February 2025;
Published: 19 March 2025.
Edited by:
Giacomo Cafaro, University of Perugia, ItalyReviewed by:
Rahul Kakalij, University of Nebraska Medical Center, United StatesCopyright © 2025 Li, Fu, Yin and Zhu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xuefei Li, bGl4dWVmZWkxMjA3QHFxLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.