
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Immunol. , 14 February 2025
Sec. Multiple Sclerosis and Neuroimmunology
Volume 16 - 2025 | https://doi.org/10.3389/fimmu.2025.1436246
This article is part of the Research Topic The Relevance of Autoimmune Encephalitis and Autoimmune Psychosis in Psychiatry View all articles
Metabotropic glutamate receptor 5 (mGluR5) antibody encephalitis is an infrequent clinical disorder, initially reported in 2011 among two patients presenting with limbic encephalitis and Hodgkin’s lymphoma. Mental and behavioral abnormalities are prevalent manifestations, accompanied by cognitive impairment, movement disorders, seizures, and other associated symptoms. In this report, we present the case of a young female patient who presented with abnormal mental behavior, seizures, and disturbances of consciousness. A cell-based assay (CBA) showed positive IgG metabotropic glutamate receptor 5 in both her serum and cerebrospinal fluid (CSF), as well as positive IgG N-methyl-D-aspartate receptor (NMDAR) in both her serum and CSF. She was diagnosed with mGluR5 overlapping NMDAR antibody encephalitis and received high-dose intravenous methylprednisolone pulse therapy and immunoglobulin therapy. Tumor screening suggested the presence of bilateral ovarian teratoma. However, unfortunately the prognosis was extremely poor. Clinical results suggested that patients with mGluR5-Abs mostly have good prognoses, excepting our case.
Since the initial identification of anti-N-methyl-D-aspartate receptor (NMDAR) encephalitis in 2007, a plethora of autoantibodies targeting neuronal cell surface or synaptic proteins have been discovered (1). At present, anti-NMDAR encephalitis represents the predominant form, accounting for 54%-80% of AE (2). In 1982, Ian Carr described personality changes and memory loss in his daughter, who was then 15.5 years old. The patient ultimately received a diagnosis of limbic encephalitis and Hodgkin’s lymphoma, which syndrome named “Ophelia syndrome” (3). Metabotropic glutamate receptors (mGluR) are G-protein coupled receptors activated by the binding of glutamate (4). Both mGluR5 and NMDAR are glutamate receptors, which serve as the principal mediators of excitatory synaptic transmission in the brain (5). Metabotropic glutamate receptor 5 (mGluR5) encephalitis is a rare autoimmune encephalitis that was first reported in 2011 in two patients with limbic encephalitis and Hodgkin’s lymphoma (6). Up to now, there have been no reports in China of cases of mGluR5 overlapping NMDAR antibody encephalitis combined with ovarian teratoma. In this report, we present clinical data from a single case of a patient for anti-mGluR5 coexistent anti-NMDAR antibody encephalitis, accompanied by a thorough literature review aiming to enhance clinicians’ understanding of this disease.
A 21-year-old female patient was first admitted to the hospital on June 16, 2022, with “mental and behavioral abnormalities for a duration of 5 days, and convulsions in all limbs for 2 days.” The patient has no prior medical or familial history of neuropsychiatric disorders. On June 11, 2022, the patient exhibited neuropsychiatric symptoms characterized by disorganized speech, tangentiality, and agitation. On June 14, 2022, limb convulsions accompanied by pyrexia were observed. Subsequently, on June 15, 2022, there was an alteration in consciousness, which should not be referred to. The probability of considering AE is significantly augmented subsequent to admission. Therefore, on June 18, 2022, she received high-dose intravenous methylprednisolone pulse therapy (1000 mg/d for 3 days, 500 mg/d for 3 days, 80 mg/d for 3 days), and subsequent administration of epilepsy control medications (intramuscular phenobarbital sodium injection, sodium valproate, levetiracetam tablets, etc.), along with endotracheal intubation and mechanical ventilation. Unfortunately, the patient’s family members request discharge, and the patient was subsequently discharged on June 27, 2022. The patient was readmitted on July 13, 2022, presenting with the aforementioned symptoms. In the cerebrospinal fluid, leukocytes were 140/mm3, 94% of lymphocytes, the red blood cell count was 100/mm3, the protein concentration was 0.36 g/L, and the glucose concentration was 3.2 mmol/L. However, the outcome of Pandy test is positive. Brain magnetic resonance imaging (MRI) revealed patchy T2WI/FLAIR hyperintensity and abnormally elevated DWI signals in bilateral hippocampus and right temporal parietal lobe (Figure 1). Lumbar puncture was performed again, and the repeated CSF test still indicated leukocytosis (127/mm3), a low protein level (0.11 g/L), and a high glucose concentration (8.4 mmol/L). A cell-based assay (CBA) showed seropositive antibodies of NMDAR (1:100) and mGluR5 (1:30), as well as in the CSF for both NMDAR (1:100) and mGluR5 (1:30) (Figure 2). 24-hours ambulatory electroencephalogram (EEG): multiple sharp slow waves and spike rhythms activity in the right frontal region. The pelvic enhanced CT scan showed that mixed density shadow in bilateral adnexal area and the bottom of the uterus, with an increased size measuring approximately 3.8cm x 4.6cm. Additionally, there was an elevated CA199 level of 50.60U/ml. Unfortunately, the family declined to undergo biopsy and surgical intervention. One month later, the pelvic enhanced CT revealed an increase in size of the lesions compared to previous findings: bilateral adnexal areas exhibited mixed low-density shadows, suggestive of teratoma (right side measuring 3.6 cm x 3.0 cm, left side measuring 4.9 cm x 5.4 cm).

Figure 1. Brain MRI revealed patchy T2WI/FLAIR hyperintensity and abnormally elevated DWI signals in bilateral hippocampus and right temporal parietal lobe (as shown by the arrow). (A, B) T2WI, (T2-weighted imaging); (C, D) FLAIR, (fluid attenuated inversion recovery); (E, F) DWI, (diffusion- weighted imaging).
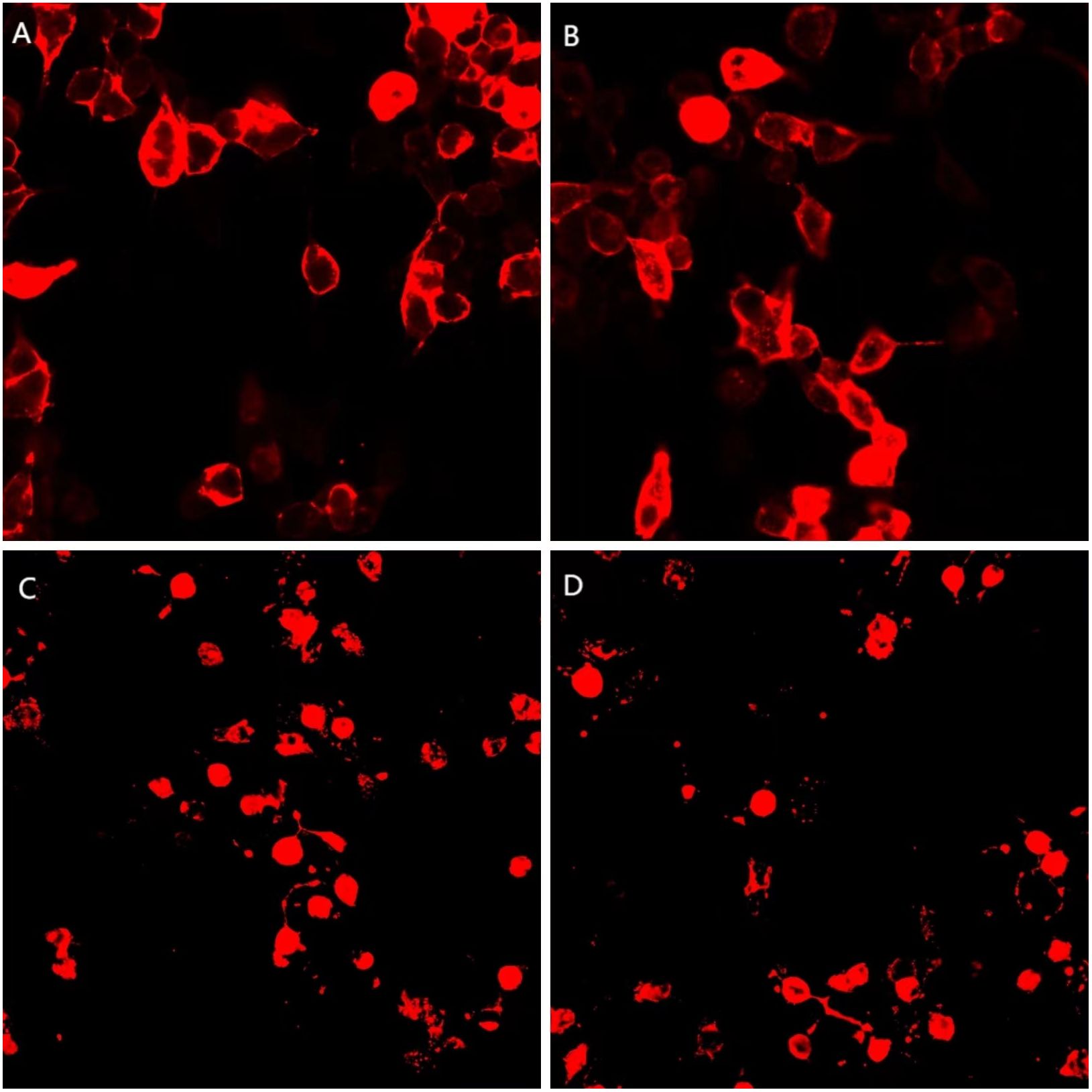
Figure 2. A cell-based assay (CBA) demonstrates positive mGluR5 (1:30, 1:30) and NMDAR (1:100, 1:100) expression in both serum and CSF. (A, C) serum; (B, D) CSF; (A, B) mGluR5 antibody; (C, D) NMDAR antibody.
Based on the patient’s medical history, presenting symptoms, and positive findings of mGluR5 and NMDAR autoantibodies, she has finally been diagnosed with mGluR5 overlap NMDAR antibody-associated AE. Therefore, she received high-dose intravenous methylprednisolone pulse therapy (1000 mg/d for 3 days, 500 mg/d for 3 days, 120mg/d for 3 days, 60mg/d for 3 days, 40mg/d for 3 days, 20mg/d for 3 days), Following, she is prescribed oral prednisolone (12mg/d) for maintenance therapy. The patient’s condition remains critical, and human immunoglobulin (25g/d for 5 days) is being used in combination for immune enhancement. However, the patient’s condition was complicated. Throughout this period, the patient consistently experienced high fever, with a temperature peaking at 40°C, and underwent continuous renal replacement therapy (CRRT) treatment. Subsequently, acute heart failure developed in the patient along with an elevated NT-proBNP level of 14700pg/ml. Consecutive sputum, blood, and ascites cultures demonstrated the presence of Acinetobacter baumannii, Candida albicans, and Pseudomonas aeruginosa strains successively. Based on drug sensitivity testing results and pharmacist recommendations, appropriate antibiotic therapy was administered. Unfortunately, the patient succumbed to acute gastrointestinal hemorrhage during the course of hospitalization. The clinical manifestations, diagnosis and therapeutic interventions of the patient have been summarized in Figure 3.
mGluR5-IgG in serum was tested by a cell-based assay (CBA). HEK293 cells were co-transfected with full-length human mGluR5 and pcDNA3.1-EGFP. After 36 hours of transfection in 96 well plate, the cells were fixed with 4% paraformaldehyde for 20 minutes, washed with phosphate-buffered saline (PBS) and permeabilized with 0.1% Triton X-100 in PBS for 20 minutes, which was ready for antibody detection. Serum diluted at 1:10 in PBS-10% goat serum and incubated on cells for 2 hours at room temperature. Cells were then washed in PBS-0.1% Tween 20 for 3 times, incubated for 30 minutes with goat anti-human IgG (1:500, Thermo Scientific), washed again in PBS-0.1% Tween 20, and evaluated by immunofluorescence microscopy. Two independent masked assessors classified each sample as positive or negative based on the intensity of immunofluorescence in direct comparison with non-transfected cells and control samples. Once confirmed, the positive specimens were then serially diluted by threefold from 1:100 to 1:1000 to determine the titers. The final titer was defined as the sample dilution value for which specific fluorescence was barely but clearly identifiable and expressed as the corresponding dilution value.
mGluR5-IgG in CSF was tested by a cell-based assay using HEK293 cells co-transfected with human mGluR5 and pcDNA3.1-EGFP. Thirty-six hours after transfection the HEK293T cells were fixed with 4% paraformaldehyde for 20 minutes and permeabilized with 0.1% Triton X-100 in phosphate-buffered saline (PBS) for 20 minutes. Cells were incubated with patient’s CSF for 2 hours and then immunolabeled with an AlexaFluor 546 secondary antibody against human IgG (1:1000, Thermo Scientific) for 1 hour at room temperature. Images were acquired using a Zeiss Axiovert A1 fluorescence microscope.
In this study, we present a case of AE characterized by the coexistence of double antibodies targeting mGluR5 and NMDAR in conjunction with bilateral ovarian teratoma. The occurrence of Anti-mGluR5 encephalitis is infrequent in clinical practice, and there is a scarcity of reports on the coexistence of mGluR5 with other antibody-associated encephalitis. To date, a total of six cases of mGluR5 overlapping with other antibody encephalitis (Table 1) have been reported (7–11), with four cases demonstrating co-occurrence of NMDAR antibodies (8, 10, 11); among these, one case was positive for MOG, NMDAR, and mGluR5 antibodies (8), while another case showed positivity for NMDAR, AMPAR, and mGluR5 antibodies (10). Additionally, one case tested positive for LGI1 antibody (9), and another case exhibited positivity for SOX1 antibody (7). The injection of IgG from patients with anti-mGluR5 encephalitis into the ventricular system of mice has been demonstrated in animal studies to induce memory impairment and heightened anxiety, while concurrently diminishing mGluR5 levels within the hippocampus (12). Notably, mGluR5 plays a pivotal role in memory acquisition by primarily augmenting NMDAR response (13). Both mGluR5 and NMDAR are glutamate receptors, which serve as the principal mediators of excitatory synaptic transmission in the brain (5, 14). Glutamate receptors (GluRs) can be categorized into ionic (iGluRs) and metabolic receptors (mGluRs) (15). Anti-NMDAR encephalitis is a clinical syndrome characterized by the presence of patients with anti-mGluR5 encephalitis into the ventricular system of mice has been demonstrated in animal studies to induce memory impairment and heightened anxiety, while concurrently diminishing mGluR5 levels within the hippocampus (12). Notably, mGluR5 plays a pivotal role in memory acquisition by primarily augmenting NMDAR response (13). Both mGluR5 and NMDAR are glutamate receptors, which serve as the principal mediators of excitatory synaptic transmission in the brain (5, 14). Glutamate receptors (GluRs) can be categorized into ionic (iGluRs) and metabolic receptors (mGluRs) (15). Anti-NMDAR encephalitis is a clinical syndrome characterized by the presence of iGluRs antibodies (16). Previous studies have indicated that mGluR5 encephalitis commonly coexists with NMDAR encephalitis, suggesting potential shared pathogenic mechanisms or possibly due to the relatively high incidence of anti-NMDAR encephalitis. The rare syndrome of marginal encephalitis associated with iGluRs antibody also encompasses the presence of an uncommon anti-AMPAR antibody (17). The primary clinical presentations observed in patients with mGluR5 overlapping NMDAR antibody encephalitis in this study encompassed cognitive and behavioral abnormalities, seizures, and impaired consciousness, exhibiting no significant deviations from previous studies. It has been reported in the literature that the patient with overlapping LGI1 antibodies showed left faciobrachial dystonic seizures, which represent the primary clinical manifestation of LGI1 antibody encephalitis. The patient ‘s mGluR5 serum titer was 1:10, while CSF analysis negative results, thus LGI1 was considered to be a pathogenic antibody. Patients with overlapping MOG antibodies showed cerebral cortical encephalitis, which was easily misdiagnosed as viral encephalitis. Patients with overlapping SOX1 antibodies have progressive ophthalmoplegia and cerebellar ataxia symptoms, alongside small cell lung cancer. Anti-SOX1 antibodies are regarded as serum biomarkers for small cell lung cancer (18). The overlapping antibody encephalitis of NMDAR, AMPAR and mGluR5 showed refractory epilepsy.
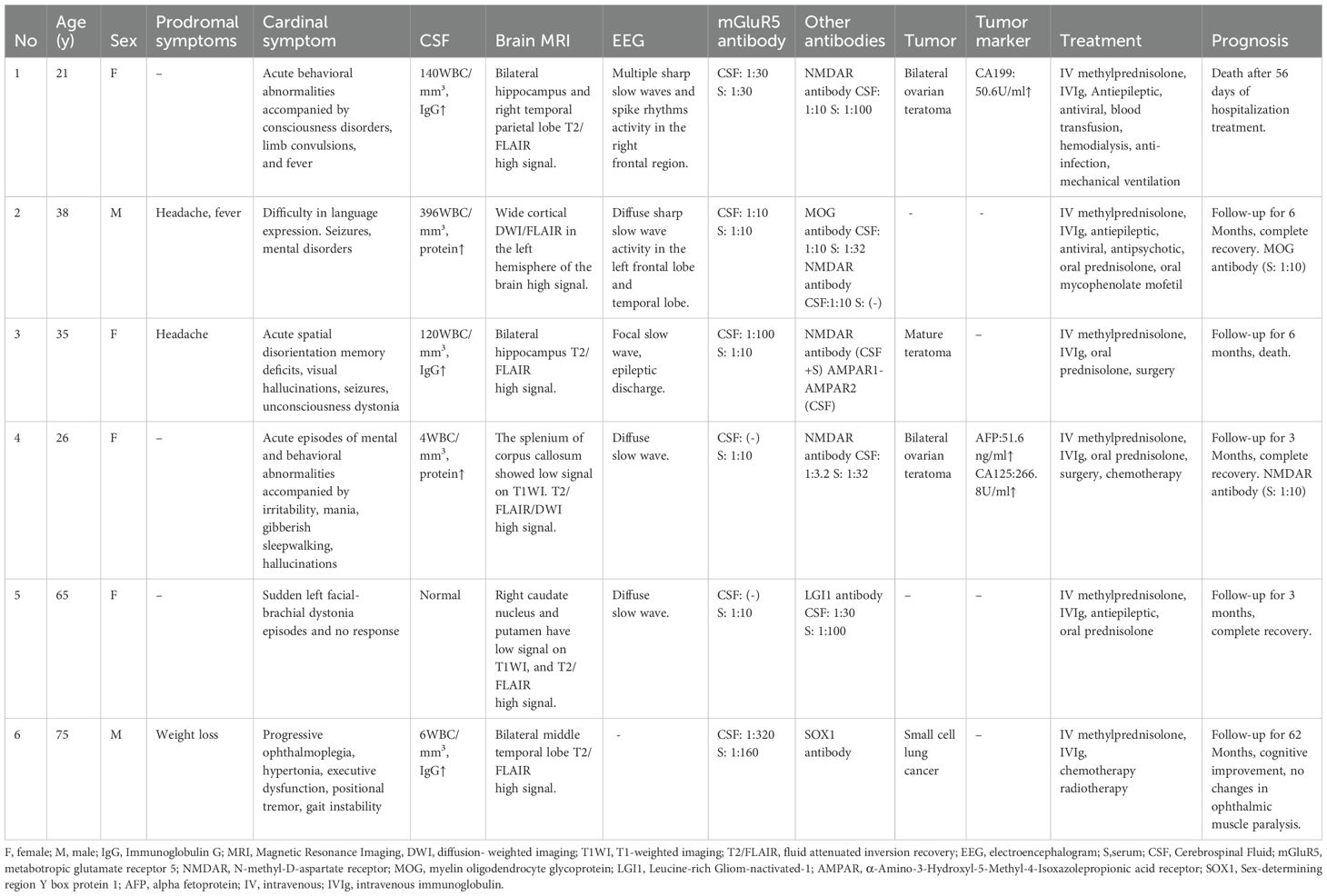
Table 1. Clinical characteristics, tumor status, treatment and prognosis of 6 patients with mGluR5 antibody overlapping encephalitis.
Anti-NMDAR encephalitis was first discovered in 2007 (3), which is the most common autoimmune encephalitis. Its primary clinical manifestations encompass mental and behavioral abnormalities, cognitive impairment, seizures, autonomic dysfunction, and disturbances in consciousness (19). The symptoms of anti-NMDAR encephalitis exhibit a wide range of manifestations. Some patients initially present with a solitary neurological or psychiatric symptom, while additional symptoms may progressively emerge over the course of several months. The occurrence of anti-mGluR5 antibody encephalitis was initially documented in 2011, when it was observed in two patients with limbic encephalitis and Hodgkin’s lymphoma. However, mGluR5 antibody encephalitis remains a rare condition in clinical practice. As the detection of antibodies becomes more widespread and our understanding of the disease improves, there has been a gradual increase in case reports. The association between mGluR5 antibody encephalitis and Hodgkin’s lymphoma has been reported in study (6, 19). However, our findings suggest that while this condition is indeed associated with tumors, there is no evidence of a specific link to Hodgkin’s lymphoma. This discrepancy may be attributed to the presence of overlapping antibodies or variations in genetic backgrounds. Previous research has indicated that anti-mGluR5 encephalitis be accompanied by peripheral neuropathy (20), suggesting that mGluRs antibodies can impact not only the central nervous system but also the peripheral nervous system (PNS). It is widely acknowledged that NMDAR antibody encephalitis exhibits a strong association with ovarian teratomas and demonstrates a higher prevalence among young female patients. A systematic review of studies conducted between 2007 and 2020 concluded that the incidence of ovarian teratoma among patients diagnosed with anti-NMDAR encephalitis is 37.4% (21). In this case, the clinical presentations of epileptic seizures and psychiatric abnormalities may be attributed to the presence of both mGluR5 and NMDAR antibodies; however, it is challenging to definitively determine which antibody exerts a predominant influence in this patient.
AE is a rare neurological disorder, and its diagnosis is on the rise due to the growing prevalence of autoantibody testing and enhanced awareness among clinicians. Nevertheless, it is undeniable that the differential diagnosis of AE occurs more frequently than the condition itself, encompassing a range of disorders such as central nervous system (CNS) infections, metabolic encephalopathy, primary central nervous system lymphoma, Creutzfeldt-Jakob disease (CJD), mitochondrial diseases (22).
Patients diagnosed with AE frequently exhibit prodromal symptoms, including headache and fever (notably in cases of anti-NMDAR encephalitis), which can complicate the differential diagnosis from infectious etiologies. Among infectious etiologies, viruses represent the most significant pathogens identified, with herpes simplex virus encephalitis being the predominant form of viral encephalitis. Kong (22) have developed and validated a novel diagnostic model that effectively differentiates viral encephalitis (VE) from autoimmune limbic encephalitis (ALE). Compared to patients whose conditions are driven by immunological factors, patients with infectious etiologies are more predisposed to experience headache, nausea/vomiting, and elevated white blood cell counts in the cerebrospinal fluid, while exhibiting a lower likelihood of status epilepticus. Furthermore, individuals diagnosed with viral encephalitis may present at an older age and demonstrate fewer neuropsychiatric symptoms than their counterparts with autoimmune encephalitis (23). Specific diagnosis is achieved through the detection of herpes simplex virus pathogens in both serum and CSF. MRI of the brain can be useful in the differential diagnosis of encephalitis. Most patients with AE have either bilateral or unilateral increased T2/FLAIR signals in the medial temporal lobes without contrast enhancement or abnormal DWI (24).
Temporal lobe gliomas that fail to exhibit a sustained response to immunotherapy, demonstrate a mass effect on MRI, and lack an inflammatory response in CSF can help differentiate them from autoimmune encephalitis (25). Primary central nervous system lymphoma (PCNSL) exhibits characteristics similar to AE, including unilateral involvement of the medial temporal lobe on MRI, elevated lymphocyte counts in CSF, and heightened sensitivity to steroid therapy. A definitive diagnosis necessitates histopathological examination (26). Mitochondrial encephalomyopathy with lactic acidosis and stroke-like episodes (MELAS) can initially manifest with altered consciousness and psychiatric symptoms, elevated cerebrospinal fluid cell counts, and focal lesions in the lateral temporal lobe on MRI. Negative CSF neuronal surface antibodies, MRS findings, and muscle biopsy results will necessitate a more detailed differential diagnosis (27).
Similar to other forms of antibody-associated AE, most patients with anti-mGluR5 encephalitis showed good responses to the immunotherapy (10). Anti-mGluR5 encephalitis should be treated with first-line immunotherapies, which include high-dose intravenous corticosteroids, intravenous immunoglobulin, and plasmapheresis (28). For severe cases, a combination of corticosteroids shock and intravenous immunoglobulin therapy may be employed (29). For patients who do not respond adequately to first-line immunotherapy, the consideration of second-line immunotherapy using agents such as rituximab, cyclophosphamide, or mycophenolate mofetil is warranted (30). Meanwhile, symptomatic and supportive treatments were administered based on clinical manifestations, encompassing the management of psychotic symptoms and antiepileptic therapy. Previous research has demonstrated that upon detection of an ovarian teratoma, prompt surgical intervention is imperative. Delaying the excision of a teratoma beyond one month is correlated with adverse clinical outcomes (31).
Previous research has indicated that the majority of patients with mGluR5 antibody encephalitis exhibit a favorable prognosis upon follow-up, with only a minority experiencing an unfavorable outcome (32). Compared to patients with good outcomes, patients with bad outcomes had higher frequency of hypoventilation and higher severity at peak of the disease, reflected by higher mRS score (10). The results of a large observational study on the prognosis of anti-NMDAR encephalitis indicate that (33) within the initial 24 months, 78.6% of the patients had a good prognosis, 12% relapsed, and 5.9% died. Titulaer et al. (33) demonstrated that factors associated with a favorable prognosis included non-admission to the ICU, early initiation of treatment, and lower disease severity within the first four weeks of onset. Schubert et al. (34) highlighted that the incidence of autonomic dysfunction and the utilization of mechanical ventilation during hospitalization were significantly correlated with adverse neurological outcomes at discharge, and these factors may potentially contribute to unfavorable long-term prognosis (35).
Unfortunately, the patient in this study exhibited a poor prognosis. Previous literature has indicated that surgical intervention can significantly enhance the prognosis. Due to concerns regarding the potential impact on the patient’s fertility, following initial hospitalization, the family declined surgical intervention and discontinued immunotherapy, subsequently choosing to leave the hospital of their own accord. Upon readmission, the optimal window for surgical treatment had elapsed, and the patient’s condition was critically severe. The primary cause of mortality for the patient was hemorrhagic shock caused by lower gastrointestinal hemorrhage. The pathogeny of lower gastrointestinal hemorrhage was evaluated as follows: Severe infection results in bone marrow suppression and abnormal coagulation function, with the patient’s coagulation function repeatedly reported as critical value during hospitalization. The patient had multiple systemic infections, including hematogenous infection, pulmonary infection, urinary tract infection, and abdominal infection, may experience intestinal flora imbalance due to the use of various antibiotics. Moreover, the patient has intermittent diarrhea, it is not excluded that antibiotic-related diarrhea leads to complications such as intestinal wall edema, inflammation, bleeding and even necrosis. Hemorrhage resulting from rupture of intestinal vascular malformation: during the course of the disease, the patient suddenly had a large amount of bloody stool, with a significant amount of bleeding and the possibility of intestinal vascular malformation was not excluded. Administration of high-dose hormone pulse therapy may also lead to gastrointestinal hemorrhage. The patient was admitted to the hospital in a critical condition, presenting numerous complications including septic shock and potential stress-induced gastrointestinal mucosal bleeding.
This study is a single case report and has certain limitations in assessing the efficacy of immunotherapy and prognosis. To date, only case reports of mGluR5 antibody-overlapping patients with encephalitis have been documented, and the lack of large-scale studies has resulted in an inadequate evaluation of long-term outcomes and the effect of immunotherapy.
We reported a case of fatal mGluR5 overlapping NMDAR antibody-associated AE with bilateral ovarian teratoma, which widens the understanding of this disease. This case underscores the hazards of severe AE and emphasizes the need for clinicians to be vigilant in identifying severe encephalitis, particularly in instances where mGluR5 coexists with other encephalitis.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by Medical Research Ethics Review Committee, General Hospital of Ningxia Medical University. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article. Written informed consent was obtained from the participant/patient(s) for the publication of this case report.
YG: Writing – original draft, Data curation, Investigation. TX: Writing – original draft. PL: Writing – original draft. JZ: Writing – original draft. ZW: Writing – review & editing.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study was supported by National Natural Science Foundation of China (82371358 to ZW).
We would like to express our sincere gratitude to our patient and his family for their cooperation in the preparation of this report.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2025.1436246/full#supplementary-material
1. Dalmau J, Tüzün E, Wu HY, Masjuan J, Rossi JE, Voloschin A, et al. Paraneoplastic anti-N-methyl-D-aspartate receptor encephalitis associated with ovarian teratoma. Ann Neurol. (2007) 61:25–36. doi: 10.1002/ana.21050
2. Ren H, Fan S, Zhao Y, Guan H. The changing spectrum of antibody-mediated encephalitis in China. J Neuroimmunol. (2021) 361:577753. doi: 10.1016/j.jneuroim.2021.577753
3. Carr I. The Ophelia syndrome: memory loss in Hodgkin’s disease. Lancet. (1982) 1:844–5. doi: 10.1016/s0140-6736(82)91887-6
4. Mazzitelli M, Presto P, Antenucci N, Meltan S, Neugebauer V. Recent advances in the modulation of pain by the metabotropic glutamate receptors. Cells. (2022) 11:2608. doi: 10.3390/cells11162608
5. Dalmau J, Geis C, Graus F. Autoantibodies to synaptic receptors and neuronal cell surface proteins in autoimmune diseases of the central nervous system. Physiol Rev. (2017) 97:839–87. doi: 10.1152/physrev.00010.2016
6. Lancaster E, Martinez-Hernandez E, Titulaer MJ, Boulos M, Weaver S, Antoine JC, et al. Antibodies to metabotropic glutamate receptor 5 in the Ophelia syndrome. Neurology. (2011) 77:1698–701. doi: 10.1212/WNL.0b013e3182364a44
7. Spatola M, Sabater L, Planagumà J, Martínez-Hernandez E, Armangué T, Prüss H, et al. Encephalitis with mGluR5 antibodies: Symptoms and antibody effects. Neurology. (2018) 90:e1964–1964e1972. doi: 10.1212/WNL.0000000000005614
8. Fu J, Peng L, Yang Y, Xie Y, Li Z, Rong B. Case report: Overlapping syndrome mimicking infectious meningoencephalitis in a patient with coexistent MOG, NMDAR, mGluR5 antibody positivity. Front Immunol. (2022) 13:919125. doi: 10.3389/fimmu.2022.919125
9. Huo T, Luo X, Zhao J, Wang T, Chen J. A Chinese female patient with LGI1 and mGluR5 antibodies: A case report. Med (Baltimore). (2022) 101:e31063. doi: 10.1097/MD.0000000000031063
10. Guo K, Liu X, Gong X, Li A, Liu Y, Li X, et al. Autoimmune encephalitis with mGluR5 antibodies: A case series from China and review of the literature. Front Immunol. (2023) 14:1146536. doi: 10.3389/fimmu.2023.1146536
11. Li Y, Zhang M, Liu D, Wei M, Sheng J, Wang Z, et al. Case report: Autoimmune encephalitis with multiple auto-antibodies with reversible splenial lesion syndrome and bilateral ovarian teratoma. Front Immunol. (2022) 13:1029294. doi: 10.3389/fimmu.2022.1029294
12. Maudes E, Mannara F, García-Serra A, Radosevic M, Mellado A, Serafim AB, et al. Human metabotropic glutamate receptor 5 antibodies alter receptor levels and behavior in mice. Ann Neurol. (2022) 92:81–6. doi: 10.1002/ana.26362
13. Aloisi E, Le Corf K, Dupuis J, Zhang P, Ginger M, Labrousse V, et al. Altered surface mGluR5 dynamics provoke synaptic NMDAR dysfunction and cognitive defects in Fmr1 knockout mice. Nat Commun. (2017) 8:1103. doi: 10.1038/s41467-017-01191-2
14. Faas GC, Adwanikar H, Gereau RW4, Saggau P. Modulation of presynaptic calcium transients by metabotropic glutamate receptor activation: a differential role in acute depression of synaptic transmission and long-term depression. J Neurosci. (2002) 22:6885–90. doi: 10.1523/JNEUROSCI.22-16-06885.2002
15. Dalmau J, Gleichman AJ, Hughes EG, Rossi JE, Peng X, Lai M, et al. Anti-NMDA-receptor encephalitis: case series and analysis of the effects of antibodies. Lancet Neurol. (2008) 7:1091–8. doi: 10.1016/S1474-4422(08)70224-2
16. Lai M, Hughes EG, Peng X, Zhou L, Gleichman AJ, Shu H, et al. AMPA receptor antibodies in limbic encephalitis alter synaptic receptor location. Ann Neurol. (2009) 65:424–34. doi: 10.1002/ana.21589
17. Güre AO, Stockert E, Scanlan MJ, Keresztes RS, Jäger D, Altorki NK, et al. Serological identification of embryonic neural proteins as highly immunogenic tumor antigens in small cell lung cancer. Proc Natl Acad Sci U S A. (2000) 97:4198–203. doi: 10.1073/pnas.97.8.4198
18. Dalmau J, Lancaster E, Martinez-Hernandez E, Rosenfeld MR, Balice-Gordon R. Clinical experience and laboratory investigations in patients with anti-NMDAR encephalitis. Lancet Neurol. (2011) 10:63–74. doi: 10.1016/S1474-4422(10)70253-2
19. Mat A, Adler H, Merwick A, Chadwick G, Gullo G, Dalmau JO, et al. Ophelia syndrome with metabotropic glutamate receptor 5 antibodies in CSF. Neurology. (2013) 80:1349–50. doi: 10.1212/WNL.0b013e31828ab325
20. Yan W, Zhao C, Zhang H, Hu Z, Wang C. Case report: guillain-barré Syndrome characterized by severe headache associated with metabotropic glutamate receptor 5 antibody. Front Immunol. (2022) 13:808131. doi: 10.3389/fimmu.2022.808131
21. Wu CY, Wu JD, Chen CC. The association of ovarian teratoma and anti-N-methyl-D-aspartate receptor encephalitis: an updated integrative review. Int J Mol Sci. (2021) 22:10911. doi: 10.3390/ijms222010911
22. Graus F, Titulaer MJ, Balu R, Benseler S, Bien CG, Cellucci T, et al. A clinical approach to diagnosis of autoimmune encephalitis. Lancet Neurol. (2016) 15:391–404. doi: 10.1016/S1474-4422(15)00401-9
23. Kong X, Guo K, Liu X, Gong X, Li A, Cai L, et al. Differentiation between viral and autoimmune limbic encephalitis: a prospective cohort study with development and validation of a diagnostic model. J Neurol. (2024) 271:5301–11. doi: 10.1007/s00415-024-12468-0
24. Armangue T, Leypoldt F, Dalmau J. Autoimmune encephalitis as differential diagnosis of infectious encephalitis. Curr Opin Neurol. (2014) 27:361–8. doi: 10.1097/WCO.0000000000000087
25. Vogrig A, Joubert B, Ducray F, Thomas L, Izquierdo C, Decaestecker K, et al. Glioblastoma as differential diagnosis of autoimmune encephalitis. J Neurol. (2018) 265:669–77. doi: 10.1007/s00415-018-8767-1
26. Thomas C, Lehrich C, Gross CC, Wiendl H, Meuth SG, Melzer N. Primary B cell lymphoma of the CNS mimicking anti-LGI1 limbic encephalitis. Front Neurol. (2018) 9:658. doi: 10.3389/fneur.2018.00658
27. Yokota Y, Hara M, Akimoto T, Mizoguchi T, Goto YI, Nishino I, et al. Late-onset MELAS syndrome with mtDNA 14453G→A mutation masquerading as an acute encephalitis: a case report. BMC Neurol. (2020) 20:247. doi: 10.1186/s12883-020-01818-w
28. Neuroinfectious Diseases and Cerebrospinal Fluid Cytology Group, Neurology Branch of Chinese Medical Association. Chinese expert consensus on the diagnosis and management of autoimmune encephalitis. Chin J Neurol. (2022) 55:931–49. doi: 10.3760/cma.j.cn113694-20220219-00118
29. Abboud H, Probasco JC, Irani S, Ances B, Benavides DR, Bradshaw M, et al. Autoimmune encephalitis: proposed best practice recommendations for diagnosis and acute management. J Neurol Neurosurg Psychiatry. (2021) 92:757–68. doi: 10.1136/jnnp-2020-325300
30. Lee WJ, Lee ST, Byun JI, Sunwoo JS, Kim TJ, Lim JA, et al. Rituximab treatment for autoimmune limbic encephalitis in an institutional cohort. Neurology. (2016) 86:1683–91. doi: 10.1212/WNL.0000000000002635
31. Lee WJ, Lee ST, Shin YW, Lee HS, Shin HR, Kim DY, et al. Teratoma removal, steroid, IVIG, rituximab and tocilizumab (T-SIRT) in anti-NMDAR encephalitis. Neurotherapeutics. (2021) 18:474–87. doi: 10.1007/s13311-020-00921-7
32. Sun Y, Tao JX, Han X, Wang X, Zhu Y, Lian Y, et al. Clinical features and brain MRI volumetric changes in anti-mGluR5 encephalitis. Ann Clin Transl Neurol. (2023) 10:1407–16. doi: 10.1002/acn3.51831
33. Titulaer MJ, McCracken L, Gabilondo I, Armangué T, Glaser C, Iizuka T, et al. Treatment and prognostic factors for long-term outcome in patients with anti-NMDA receptor encephalitis: an observational cohort study. Lancet Neurol. (2013) 12:157–65. doi: 10.1016/S1474-4422(12)70310-1
34. Schubert J, Brämer D, Huttner HB, Gerner ST, Fuhrer H, Melzer N, et al. Management and prognostic markers in patients with autoimmune encephalitis requiring ICU treatment. Neurol Neuroimmunol Neuroinflamm. (2019) 6:e514. doi: 10.1212/NXI.0000000000000514
Keywords: autoimmune encephalitis, metabotropic glutamate receptor 5, antibody overlap, ovarian teratoma, case report
Citation: Gu Y, Xuan T, Li P, Zhou J and Wang Z (2025) Anti-metabotropic glutamate receptor 5 coexistent anti-N-methyl-D-aspartate receptor encephalitis: a case report and literature review. Front. Immunol. 16:1436246. doi: 10.3389/fimmu.2025.1436246
Received: 02 June 2024; Accepted: 29 January 2025;
Published: 14 February 2025.
Edited by:
Karl Bechter, University of Ulm, GermanyReviewed by:
Jinzhou Feng, First Affiliated Hospital of Chongqing Medical University, ChinaCopyright © 2025 Gu, Xuan, Li, Zhou and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhenhai Wang, bnlmeXd6aEAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.