- 1Department of Gastroenterology, Changzhou Hospital of Traditional Chinese Medicine Affiliated to Nanjing University of Chinese Medicine, Changzhou, China
- 2The First College of Clinical Medicine, Henan University of Chinese Medicine, Zhengzhou, China
- 3Heart Center, The First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou, China
- 4Department of Gastroenterology, The First Affiliated Hospital of Henan University of Chinese Medicine, Zhengzhou, China
Background: Previous research has demonstrated an association between gut microbiota and immune status with the development of several diseases. However, whether these factors contribute to polyps remains unclear. This study aims to use Mendelian randomization (MR) to investigate the causal relationship between gut microbiota and 4 types of polyps (nasal, gallbladder, colon, and gastric polyps), as well as to analyze the mediating role of immune traits.
Methods: This study utilized large-scale GWAS meta-analyses of gut microbiota (MiBioGen Consortium), 731 immune traits, and 4 types of polyps (one from the FinnGen Consortium and three from the NBDC Human Database). Univariate MR with the inverse variance weighted (IVW) estimation method was employed as the primary analytical approach. A two-step MR analysis was performed to identify potential mediating immune traits. Additionally, multivariable MR approach based on Bayesian model averaging (MR-BMA) was employed to further prioritize gut microbiota and immune traits associated with polyp development.
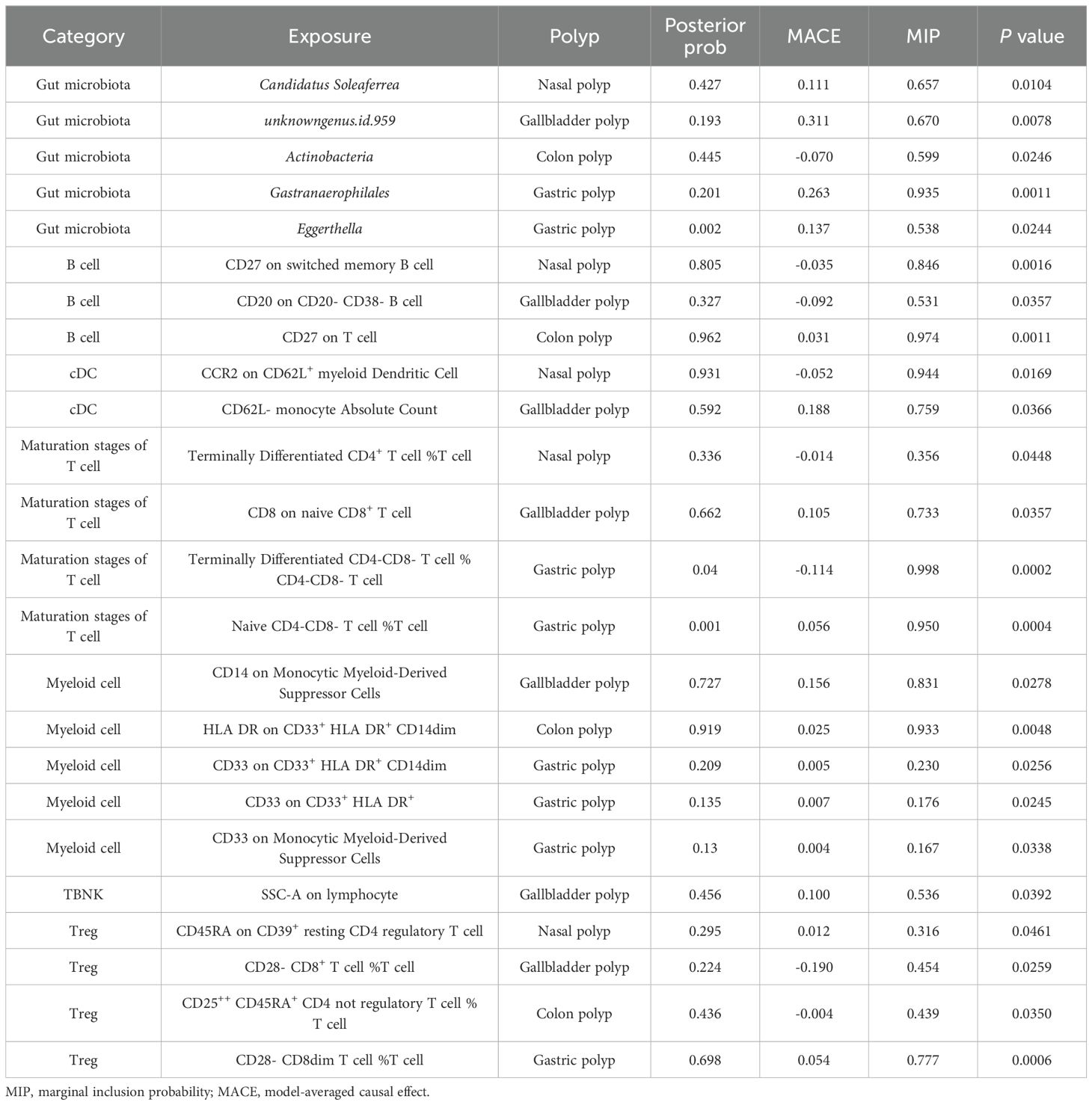
Results: Based on IVW method in univariate MR analysis, we identified 39 gut microbial taxa and 135 immune traits significantly causally associated with at least one type of polyp. For nasal polyps, 13 microbial taxa and 61 immune traits were causally associated. After false discovery rate (FDR) correction, CD3 on Central Memory CD8+ T cells and CD3 on CD4 regulatory T cells remained significant. MR-BMA identified 4 gut microbial taxa and 4 immune traits as high priority. For gallbladder polyps, 9 microbial taxa and 30 immune traits were causally associated. MR-BMA identified 8 microbial taxa and 6 immune traits as higher importance. For colon polyps, 6 microbial taxa and 21 immune traits were causally associated. MR-BMA identified 4 microbial taxa and 3 immune traits as higher importance. For gastric polyps, 12 microbial taxa and 33 immune traits were causally associated. Actinobacteria remained significant after FDR correction, and MR-BMA identified 7 gut microbial taxa and 6 immune traits as high priority. We identified 16 causal pathways with mediator directions consistent with the direction of gut microbiome-polyp association. Of these, 6 pathways were associated with the mechanism of nasal polyps, 1 with gallbladder polyps, 2 with colon polyps, and 7 with gastric polyps.
Conclusions: Our findings shed light on the causal relationships between gut microbiota, immune traits, and polyp development, underscoring the crucial roles of gut microbiota and immune status in polypogenesis. Furthermore, these findings suggest potential applications in polyp prevention, early screening, and the development of effective strategies to reduce polyp risk.
1 Introduction
Growths on the mucosal surfaces of the human body are collectively referred to as polyps, encompassing various pathological conditions such as hyperplasia, inflammation, and adenomas. While polyps are primarily classified according to their site of occurrence, including nasal polyps, gallbladder polyps, colon polyps, and gastric polyps, among others, this anatomical categorization is merely a starting point in the multidimensional process of polyp classification. In clinical practice and research, more refined and standardized classification systems are employed, considering not only the anatomical location but also morphological characteristics, size, surface features, histological type, and potential malignancy risk. These specialized systems, such as the NICE (NBI International Colorectal Endoscopic) classification for colon polyps, the Paris Classification for gastrointestinal polyps, and various methods for gallbladder and nasal polyps, provide crucial guidance for clinical decision-making and treatment strategies. While most polyps are benign, a small proportion can be malignant, underscoring the importance of accurate classification. Among individuals undergoing gastroscopy, the incidence of gastric polyps is 6.35%, with 77% being fundic gland polyps, 17% hyperplastic polyps, and malignant tumors being relatively uncommon (1). Polyps often develop insidiously, without specific clinical symptoms, and their pathogenesis remains unclear. Polyp development is closely associated with endocrine disorders, genetics, age, medications, and lifestyle habits (2). Notably, immune imbalance due to gut microbiota dysbiosis is considered a potential risk factor for polyp formation.
Microorganisms and the host body maintain a balanced relationship, with most microorganisms participating in host metabolism. These microorganisms aid digestion, promote vitamin synthesis, stimulate the development of the intestinal immune system, and together with epithelial cells, form an intrinsic barrier against pathogen invasion. Numerous studies have demonstrated that the fecal microbial composition of individuals with colon polyps significantly differs from that of healthy individuals (3, 4). Furthermore, early immunological changes associated with polyps may be linked to the microenvironment resulting from gut microbiota dysbiosis. Notably, bacterial drug delivery systems targeting the gut microbiota may emerge as an important therapeutic approach for both colon polyps and colorectal cancer (5). Since the late 20th century, Helicobacter pylori has been recognized as a crucial microorganism in the development of gastric polyps (6). Following numerous clinical studies validating its role, the eradication of H. pylori has become a routine treatment for gastric polyps (7). The gallbladder is directly connected to the intestine, with the bile acid cycle serving as an important link between these two organs. Hu et al. (8) demonstrated that intestinal microbiota, especially Desulfovibrionales, can induce gallbladder disease by increasing the secretion of secondary bile acids and enhancing bile acid hydrophobicity. For non-intestinal diseases, the intestinal microbiota and their metabolites can affect distant organs through the bloodstream or lymphatic circulation, either by active absorption or disruption of the epithelial barrier (9, 10). Therefore, the role of the intestinal microbiota should not be disregarded in the development of polyps, including nasal polyps, gallbladder polyps, colon polyps, and gastric polyps. Metabolic abnormalities resulting from gut microbial dysbiosis have been identified as a risk factor for chronic sinusitis (11). Chronic sinusitis frequently coexists with nasal polyps, characterized by eosinophil recruitment due to abnormal activation of immune cells, such as CD4+ T cells and Th2 cells (12). Notably, this localized immune remodeling, in turn, is considered an important contributor to nasal polyp development (13). Therefore, we focused on analyzing the intestinal microbiota and 731 immune profiles to investigate their potential associations with polyp development.
The clinical diagnosis of polyps relies primarily on endoscopy, including gastroenteroscopy and capsule endoscopy. These endoscopic examinations can be complemented by non-invasive imaging techniques (ultrasound and magnetic resonance imaging) to evaluate the size and type of polyps. Endoscopic resection techniques, including strangulation polypectomy, endoscopic mucosal resection, and endoscopic submucosal dissection, provide the most direct and effective means for diagnosing and treating polyps. However, these invasive procedures may not be suitable for all patients. For certain patients, treatment involves symptomatic and etiologic interventions, coupled with regular follow-up monitoring to assess polyp size progression. However, for the majority of patients, polyps tend to grow, necessitating eventual endoscopic surgery to halt disease progression and prevent malignant transformation. Therefore, identifying polyp-related biomarkers could provide new insights into the pathogenesis of polyps and contribute to polyp prevention, early screening, and the development of effective strategies to mitigate polyp risk.
Mendelian randomization (MR) is a method used to evaluate the causal relationship between an exposure and an outcome, utilizing single nucleotide polymorphisms (SNPs) strongly associated with the exposure as instrumental variables (IVs) (14). This approach is based on Mendelian laws of inheritance, treating genotypes as naturally randomized factors. MR helps mitigate the effects of reverse causation and confounding factors, providing more reliable causal inferences. By studying the human genome, genetic variants associated with certain diseases can be identified, shedding light on disease pathogenesis and providing a basis for diagnosis and treatment. In this study, we employed a two-sample bidirectional and mediated MR design to assess the causal relationship between gut microbiota and 4 types of polyps (nasal, gallbladder, colon, and gastric polyps), as well as to investigate the potential mediating role of immune profiles. Additionally, multivariable MR approach based on Bayesian model averaging (MR-BMA) was employed to identify the most likely gut microbiota and immune traits associated with polyp development.
2 Materials and methods
2.1 Study design
Univariate Mendelian randomization (MR) analysis was employed to investigate the causal relationship between gut microbiota, immune traits, and 4 types of polyps in human (nasal, gallbladder, colon, and gastric polyps). A two-step MR approach was performed to assess the potential mediating effects of immune traits. Additionally, multivariate MR analysis based on Bayesian model averaging (MR-BMA) was employed to rank the importance of gut microbiota and immune traits. Single nucleotide polymorphisms (SNPs) associated with these factors were utilized as instrumental variables (IVs). The MR study was based on the following three assumptions (15): (1) SNPs are strongly associated with the exposure factors; (2) SNPs are independent of potential confounding factors; (3) SNPs influence the outcome only through the exposure factors. See Figure 1 for the study flowchart.
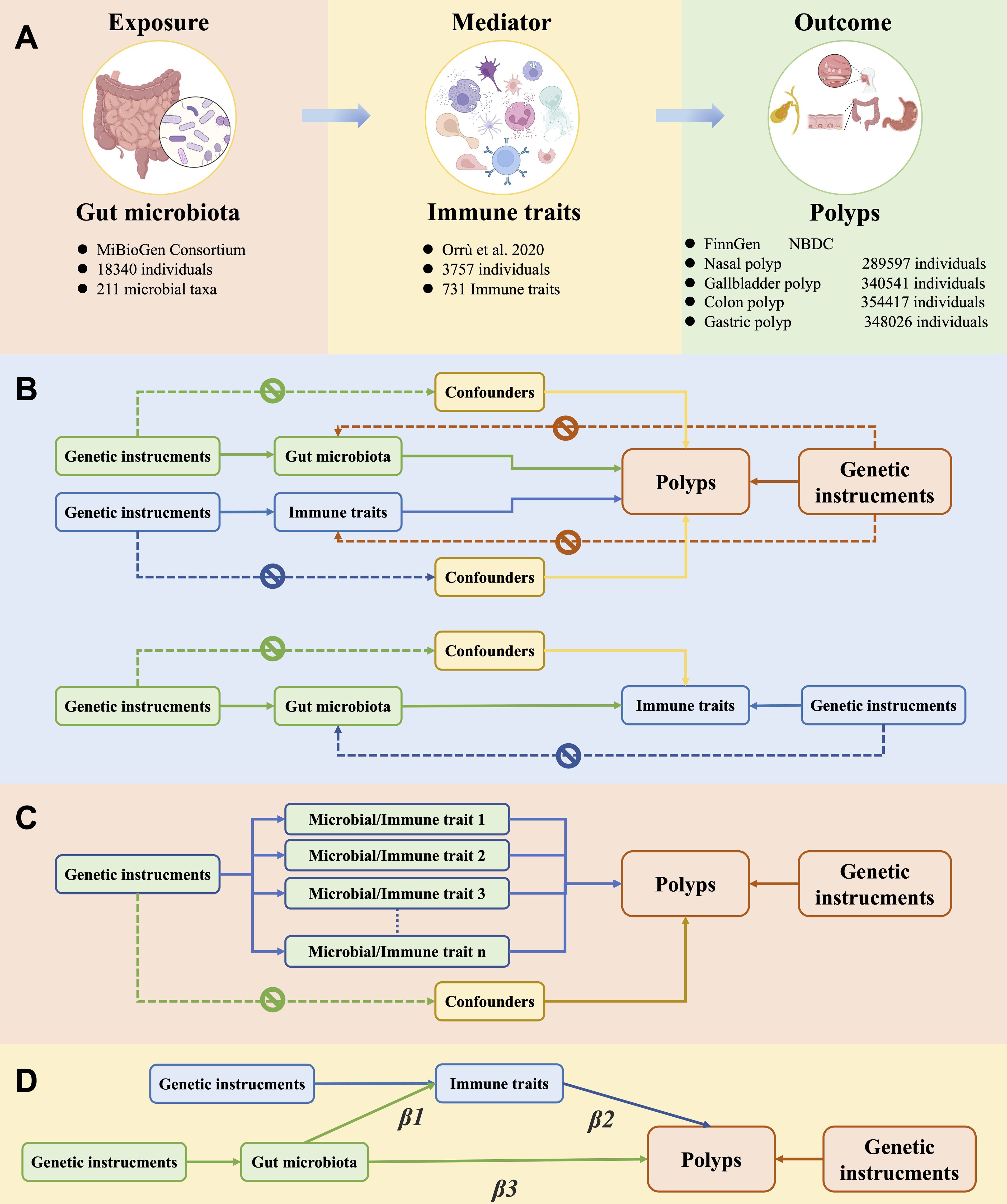
Figure 1. Overview of the study design and methods. (A) Details on exposure, mediator, and outcome data. (B) Univariate Mendelian randomization (MR) analyses with three components: 1) exposure to gut microbes and polyp as outcome, 2) exposure to immune traits and polyp as outcome, 3) exposure to gut microbiota and immune traits as outcome. Genetic instrumental variables were associated solely with exposure factors, did not directly influence polyp outcomes independently of these exposures, and were unaffected by confounding factors. (C) Multivariate MR analyses that ranked the importance of exposure factors using MR-BMA of positive exposure outcomes. (D) Mediating MR analysis to assess the mediating role of immune traits in the causal relationship between gut microbiota and polyps.
2.2 Source of GWAS summary statistics
GWAS data on gut microbiota were obtained from the MiBioGen consortium (https://mibiogen.gcc.rug.nl), comprising a total of 18,340 participants across 24 cohorts, including 13,266 participants from 16 European cohorts (16). The gut microbiota data encompassed a total of 211 taxa, including 131 genera, 35 family, 20 order, 16 class, and 9 phyla. GWAS data for 731 immune traits were derived from a cohort of 3,757 Sardinian individuals (17). The 731 immune traits could be categorized into 7 immune cell types: B cells (n=190), dendritic cells (n=64), T cell maturation stages (n=79), monocytes (n=43), myeloid cells (n=64), TBNK cells (n=124), and Treg cells (n=167). Data on polyps were obtained from two large databases. GWAS data (version R9) for nasal polyps, comprising 6,255 cases and 283,342 controls, were obtained from the FinnGen consortium (https://www.finngen.fi/fi) (18). The FinnGen research project is a nationwide Finnish genetic study that comprehensively collects genetic and electronic health record data, providing an important resource for studying disease-associated genetic variants. Data on gastric polyps (6,155 cases and 341,871 controls), colon polyps (22,049 cases and 332,368 controls), and gallbladder polyps (458 cases and 340,083 controls) were obtained from the NBDC Human Database (https://humandbs.biosciencedbc.jp/en/), a platform established by the NBDC Program Department of the Japan Science and Technology Agency (JST) for sharing various human-related data. The data for these three polyp types were derived from a European population study that included genome-wide data for 628,000 European individuals, with most of the data originating from the FinnGen consortium and the UK Biobank (19).
2.3 Selection of genetic variants
Independent SNPs form the basis of MR studies. Consistent with previous research (20), we selected SNPs with a p-value < 1 × 10−5 as instrumental variables. To exclude SNPs in linkage disequilibrium, we set k to 500 kb and r2 to 0.1 (21). To mitigate the risk of bias due to weak instrumental variables, we calculated the general F-statistic for each exposure factor. The F-statistics for SNPs associated with gut microbiota ranged from 14.59 to 88.43 (Supplementary File S1), while those for immune traits ranged from 19.55 to 2381.77 (Supplementary File S2), indicating strong correlations (F-statistic > 10) (22). To avoid the influence of confounding factors, we also removed SNPs strongly associated with alcohol consumption and smoking. Regarding potential sample overlap between exposure factors and outcomes, we consulted the relevant literature and found no sample overlap in the GWAS data used for exposures and outcomes.
2.4 Univariate Mendelian randomization
Analyses were conducted using the TwoSampleMR, MRPRESSO, and MendelianRandomization packages in R Studio (version 4.3.2) (23). In preliminary analyses, when the number of SNPs ≥2, we employed the random-effects inverse variance weighted (IVW) method, which is widely recognized as the most robust approach in MR analyses, to estimate the potential effect of exposure factors on polyp development. Additionally, we utilized the MR Egger, Weighted Median, Simple mode, and Weighted mode methods for the analysis (24–26). A heterogeneity test was employed to assess differences between IVs, and Cochran’s Q test was used to evaluate heterogeneity among IVs. The MR-Egger intercept and MR-PRESSO methods were utilized to detect horizontal pleiotropy (27, 28). Horizontal pleiotropy is when the factors we use to explore a link between two things might be affecting the outcome in more than one way, not just through the pathway we’re looking at. MR-Egger regression is a method that handles this by using a special kind of regression analysis that includes an extra term, the intercept. This intercept helps us understand how much the factors might be affecting the outcome in ways we didn’t expect. The slope of the regression line gives us a fair estimate of the actual effect we’re interested in. MR-PRESSO is a way to check if these unexpected effects, or pleiotropy, are happening with all the factors we’re using in our study. It has three main steps: First, it does a global test to see if there’s any sign of these extra effects. Second, it finds any outliers, or factors that seem to be behaving very differently, and removes them to get a corrected result. Third, it compares the results before and after removing these outliers to see if there’s a big difference. If the global test’s p-value is less than 0.05, it suggests that there are indeed some unexpected effects at play. When the number of SNPs <2, we employed the Wald ratio method for sensitivity analysis. To confirm the directionality of the MR analyses, we utilized the mr_steiger function to assess whether the results indicated a causal effect on the exposure factors. The mr_steiger function evaluates the putative direction of causality by quantifying the correlation coefficients of the SNPs’ effect sizes on both the exposure and outcome, while also accounting for sample size. It yields p-values that serve as a statistical measure to infer the likelihood of a directional causal relationship from exposure to outcome.
2.5 Multivariable MR approach based on Bayesian model averaging
Traditional multivariate Mendelian randomization (MVMR) analysis, which relies on standard linear regression, performs suboptimally when dealing with a large number of risk factors. Multivariable MR approach based on Bayesian model averaging (MR-BMA) is a Bayesian variable selection method for identifying potential causal determinants of disease from numerous candidate risk factors. As a novel multivariate MR approach, MR-BMA is well-suited for high-throughput sequencing studies with large data volumes, as it analyzes all possible combinations of risk factors and facilitates biomarker screening. We conducted MR-BMA analyses for significant gut microbiota and immune traits. The number of repeated iterations was set to 10,000, the prior probability was set to 0.1, and the prior variance was set to 0.5. The marginal inclusion probability (MIP), defined as the sum of the posterior probabilities (PP) for all specific models (i.e., a single biomarker or a combination of biomarkers), was calculated for each exposure factor. Additionally, the model-averaged causal estimate (MACE) was calculated, representing the direct causal effect of the biomarker averaged across all relevant models on polyp development. Exposures within the same category were ranked based on their MIP, with an MIP >0.1 considered indicative of high importance. All analyses were performed in R Studio (version 4.3.2).
2.6 Mediation analysis
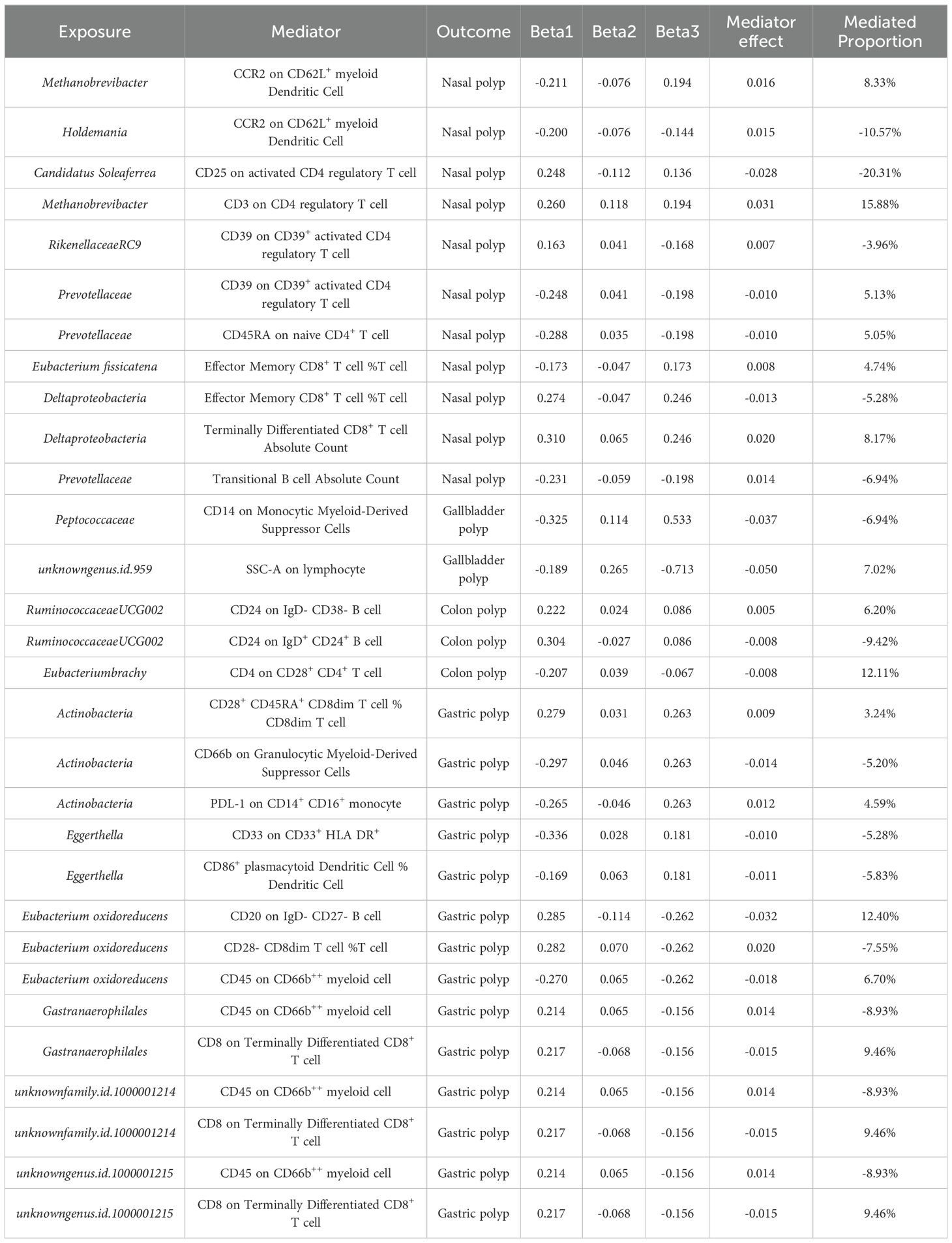
Our study employed a two-step MR approach to evaluate the potential mediating effect of immune traits on the relationship between gut microbiota and 4 types of polyps. Specifically, we sought to identify microbial taxa which displayed causal associations with the 4 types of polyps through their impacts on immunity. The causal effects of gut microbiota on polyps (Beta3), gut microbiota on immune traits (Beta1), and immune traits on polyps (Beta2) were analyzed separately using univariate MR. The proportion of the total effect mediated by the immune traits was estimated by dividing the indirect effect (Beta1×Beta2) by the total effect (Beta3). This analysis is grounded in the methodology of mediated effects analysis, aimed at elucidating influence of intestinal microbiota on the development of polyps via immune-mediated pathways. Here, β3 signifies the comprehensive effect of intestinal microbiota on polyps, encompassing both direct and indirect mechanisms; Beta1 quantifies the impact of intestinal microbiota on immune traits, whereas Beta2 captures the effect of these immune traits on polyps. By estimating the proportion of the indirect effect (Beta1×Beta2) within the total effect (Beta3), we quantitatively assess the magnitude of the mediating role played by immune traits in the intestinal microbiota-polyps relationship. This methodological approach facilitates a deeper understanding of intricate causal networks and enables the evaluation of the relative significance of various pathways of influence.
3 Result
3.1 Results of the weak instrumental variable test
The F-statistics for each SNP in gut microbiota ranged from 14.59 to 88.43 (Supplementary File S1), and the F-statistics for the immune traits ranged from 19.55 to 2381.77 (Supplementary File S2), all with high correlations (F-statistics >10).
3.2 Causal relationships between the gut microbiota, immune traits and nasal polyp
3.2.1 Univariate MR analysis and MR-BMA analysis of gut microbiota on nasal polyp
According to the IVW method, we identified a total of 39 gut microbiota taxa that were significantly causally associated with at least one type of polyp, of which 13 microbial taxa were potentially associated with nasal polyps (Figure 2). In assessing the causal effect of gut microbes on nasal polyps, we identified 1phylum, 1 family, and 4 genera as potential protective factors against the development of nasal polyps. The identified taxa were: Actinobacteria (OR=0.80, 95%CI:0.66-0.97, P=0.023), Prevotellaceae (OR=0.8, 95%CI:0.68-0.99, P=0.043), Bifidobacterium (OR=0.83, 95%CI:0.72-0.95, P=0.009), Actinomyces (OR=0.8, 95%CI:0.72-0.9, P=0.040), RikenellaceaeRC9 (OR=0.85, 95%CI:0.75-0.95, P=0.005), Holdemania (OR=0.87, 95%CI:0.76-0.98, P=0.02). 2 classes, 1 order, 1 family, and 3 genera of gut microbiota have been identified as risk factors for the occurrence of nasal polyps, including Methanobacteria (OR=1.13, 95%CI:1.00-1.27, P=0.046), Deltaproteobacteria (OR=1.28, 95%CI:1.05-1.56, P=0.014), Methanobacteriales (OR=1.14, 95%CI:1.01-1.30, P=0.042), Methanobacteriaceae (OR=1.13, 95%CI:1.00-1.27, P=0.046), Candidatus Soleaferrea (OR=1.15, 95%CI:1.01-1.30, P=0.036), Eubacterium fissicatena (OR=1.19, 95%CI:1.06-1.34, P=0.004), Methanobrevibacter (OR=1.21, 95%CI:1.01-1.45, P=0.035). Interestingly, Methanobacteria, Methanobacteriales, Methanobacteriaceae, and Methanobrevibacter all belong to the same class, suggesting that this group of gut microbiota may exert a more significant causal effect on nasal polyps. The mr_steiger algorithm indicated that the Mendelian randomization analysis was consistently directional. Additionally, we observed heterogeneity in the Actinobacteria results and horizontal pleiotropy in the Bifidobacterium results, which could potentially affect the reliability of the causal estimates. However, after correction for the false discovery rate (FDR), no taxa remained statistically significant (Figure 3A).
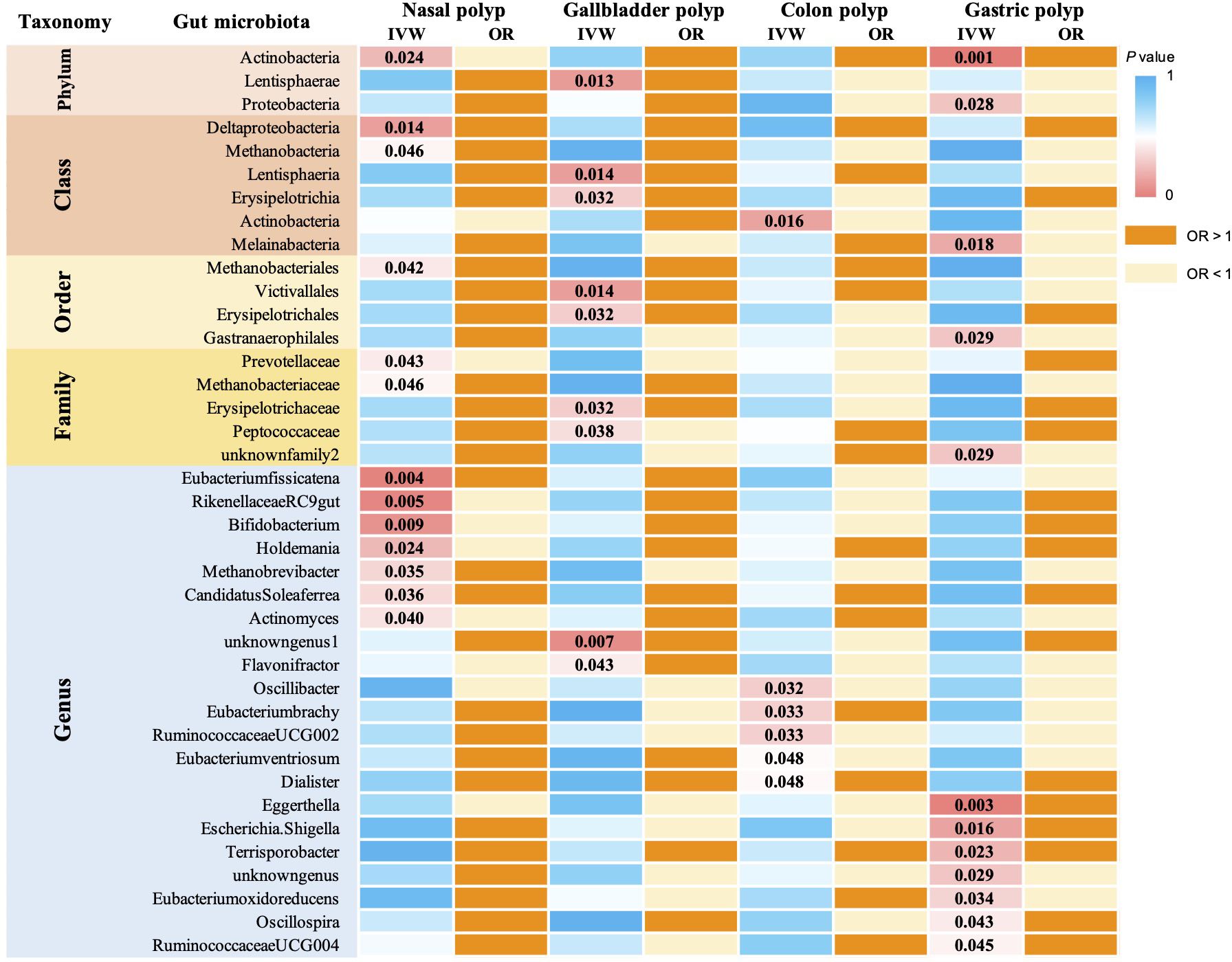
Figure 2. MR analysis examined the causal effect of gut microbiota on 4 types of polyps. The analysis identified 39 gut microorganisms that had a significant causal effect on at least 1 type of polyp, and their corresponding p-values are reported. Odds ratio (OR) greater than 1 indicates that the gut microorganism is a risk factor for polyps, while OR less than 1 suggests a protective effect.
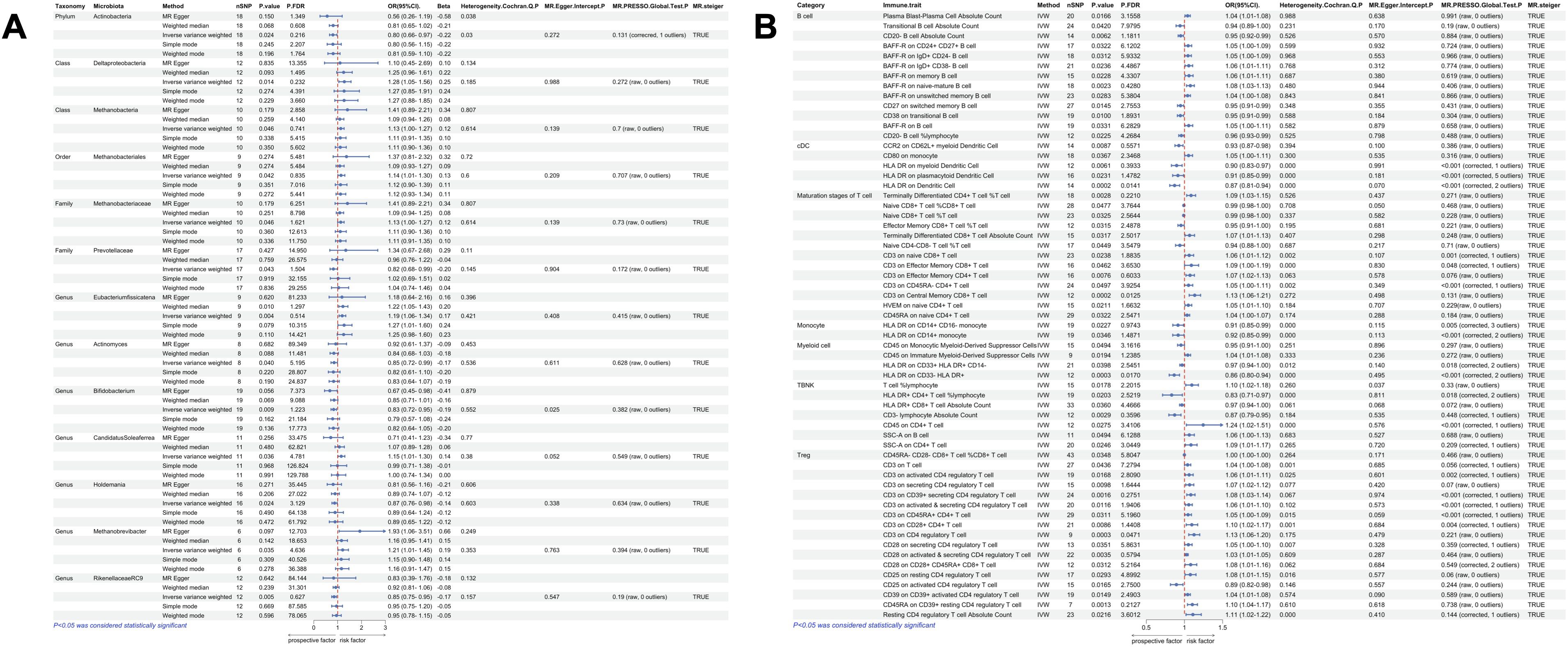
Figure 3. Univariate MR results of gut microbiota and immune traits on nasal polyps. (A) MR forest plot of the causal effect of gut microbiota on nasal polyps. (B) MR forest plot of the causal effect of immune traits on nasal polyps. OR>1 indicates an increased risk, whereas OR<1 indicates a decreased risk. The last 4 columns are analyses of horizontal pleiotropy, heterogeneity, and the directionality of MR, with P > 0.05 indicating no pleiotropy or heterogeneity and TRUE indicating no reverse causality.
MR-BMA analysis was conducted on gut microorganisms exhibiting a significant causal association with nasal polyps, enabling the ranking of their importance. Actinobacteria and Bifidobacterium were excluded from the analysis due to observed heterogeneity or horizontal pleiotropy. Important gut microbial taxa have potential as biomarkers. The results revealed 4 microorganisms with MIP values greater than 0.1: Candidatus Soleaferrea (MACE=0.112, MIP=0.427, P=0.0104), Methanobacteria (MACE=0.026, MIP=0.165, P=0.2329), Eubacterium fissicatena (MACE=0.010, MIP=0.111, P=0.1720), and RikenellaceaeRC9 (MACE=-0.010, MIP=0.118, P=0.1384). Candidatus Soleaferrea exhibited the highest posterior probability of 0.427, while the remaining 3 key microbes had posterior probabilities exceeding 0.03. See Supplementary File S3 for more details. Table 1 lists all gut microbiota and immune traits with significant P-values from the MR-BMA analysis.
3.2.2 Univariate MR analysis and MR-BMA analysis of immune traits on nasal polyp
According to IVW method, we identified a total of 135 immune traits that were significantly causally associated with at least one type of polyp, of which 61 immune traits were potentially associated with nasal polyps (Figure 4). Based on the immune cell types, they were specifically classified into 13 traits of B cells, 5 traits of cDC, 13 traits of Maturation stages of T cells, 2 traits of Monocyte, 4 traits of Myeloid cells, 7 traits of TBNK and 17 traits of Treg. Mr_steiger algorithm indicated that the MR analysis did not exhibit significant bidirectionality. Among these 61 immune traits, 35 did not exhibit heterogeneity or pleiotropy. After multiple test correction and sensitivity analysis, only two immune traits remained statistically significant (PFDR < 0.05). The significant immune traits were: CD3 on Central Memory CD8+ T cells (OR=1.13, 95%CI:1.06-1.21, PFDR=0.012), which belongs to the T cell maturation stage category, and CD3 on CD4 regulatory T cells (OR=1.13, 95%CI:1.06-1.20, PFDR=0.047), which belongs to Treg Figure 3B.
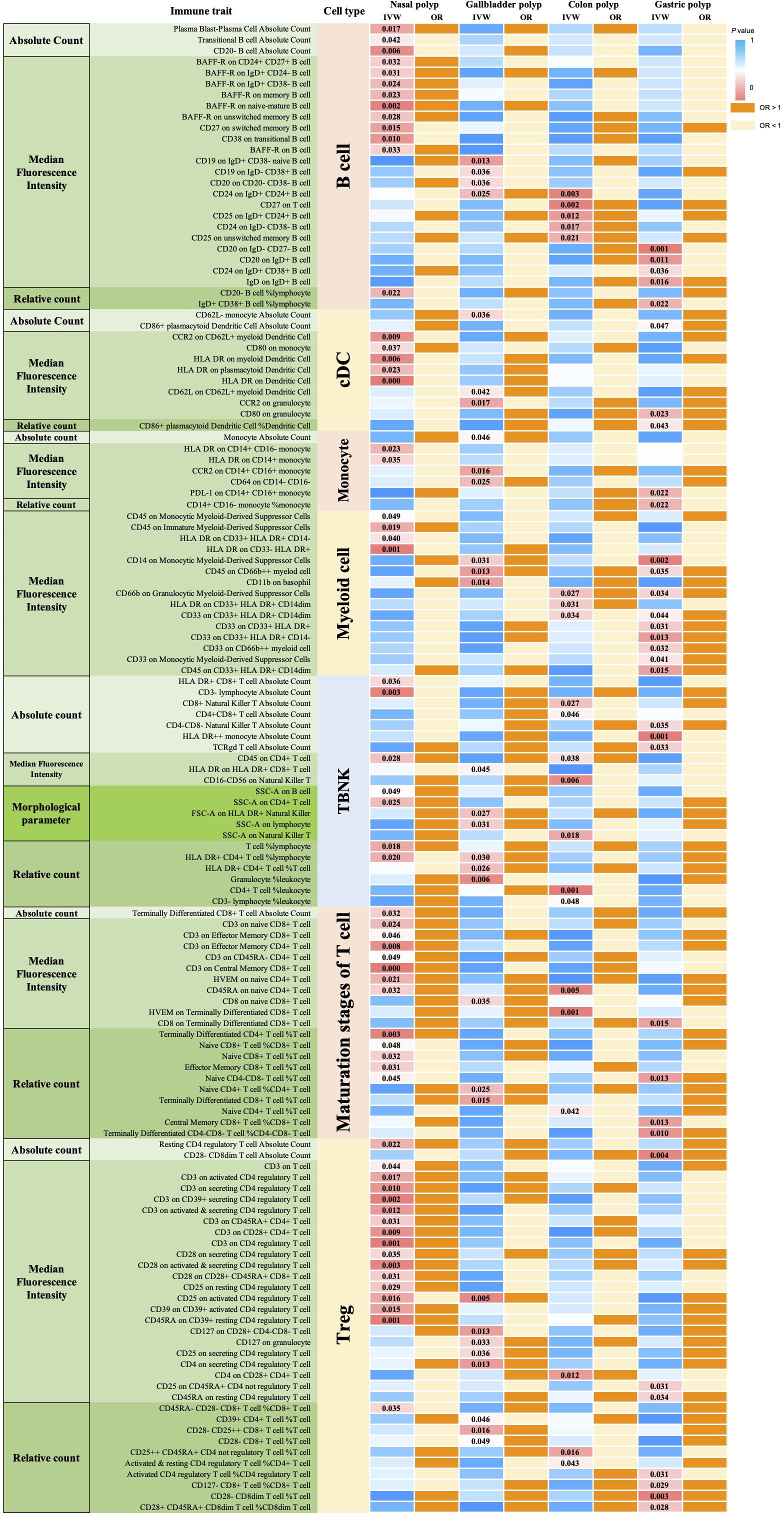
Figure 4. MR analysis of 731 immune traits for causal effects on 4 polyp types. 135 immune traits had significant causal effects on at least 1 polyp type. P-values for immune traits with significant causal effects associated with B cells (26) Dendritic cells (11), Monocyte (7), Myeloid cell (15), TBNK (21), Maturation stages of T cells (21), and Treg (34) are indicated. OR greater than 1 indicates that the immune trait is a risk factor for nasal polyps, while OR less than 1 indicates a protective effect.
We conducted MR-BMA analyses on immune traits exhibiting significant causal associations with nasal polyps, providing insights into the true importance of these immune traits for nasal polyp development. Among the 61 immune traits with significant causality, 26 exhibited heterogeneity or horizontal pleiotropy and were therefore excluded from the MR-BMA analysis. Consistent with the categorization in the univariate MR analysis, we classified the remaining immune traits into 7 categories for the MR-BMA analysis and ranked their significance based on MIP values. See Supplementary File S3 for details. 4 immune traits were identified as significant, with MIP and posterior probability greater than 0.2: CD27 on switched memory B cells (MACE=-0.035, MIP=0.846, P=0.0016), CCR2 on CD62L+ myeloid dendritic cells (MACE=-0.052, MIP=0.944, P=0.0169), Terminally Differentiated CD4+ T cell %T cell (MACE=-0.014, MIP=0.356, P=0.0448), with posterior probability models of 0.805, 0.931, and 0.336, respectively. Additionally, CD45RA on CD39+ resting CD4 regulatory T cells (MACE=0.012, MIP=0.316, P=0.0461) exhibited a posterior probability of 0.295 Table 1.
3.2.3 Mediated Mendelian randomization analysis
Through univariate MR analysis, we identified a total of 11 gut microbial taxa and 35 immune traits exhibiting significant causal effects on nasal polyps, without evidence of heterogeneity or horizontal pleiotropy. Subsequently, we conducted a two-sample MR analysis to investigate the relationships between these 11 gut microbes and 35 immune traits. See Supplementary File S4 for details. We identified a total of 11 causal pathways through which gut microbiota modulate immune function, thereby influencing the development of nasal polyps. The mediated proportion of these immune traits was also obtained. Among these, the mediating direction of 6 causal pathways (Beta1×Beta2) was consistent with the direction of the gut microbe-nasal polyp association (Beta3). Methanobrevibacter inhibited CCR2 on CD62L+ myeloid dendritic cells, a protective factor against nasal polyps, or mediated elevated levels of CD3 on CD4 regulatory T cells, which ultimately led to an increased risk of polyps. Prevotellaceae increased the risk of nasal polyps by decreasing CD39 on CD39+ activated CD4 regulatory T cells and CD45RA on naive CD4+ T cells, which are protective factors against nasal polyps. Additionally, Eubacterium fissicatena and Deltaproteobacteria affected Effector Memory CD8+ T cell %T cell and Terminally Differentiated CD8+ T cell Absolute Count, respectively, thereby increasing the risk of polyp. Notably, Eubacterium fissicatena was identified as a key microbial taxa in the MR-BMA analysis, suggesting that the mechanism by which Eubacterium fissicatena mediates Effector Memory CD8+ T cell %T cell levels may play an important role in the development of nasal polyps Table 2.
3.3 Causal relationships between the gut microbiota, immune traits and gallbladder polyp
3.3.1 Univariate MR analysis and MR-BMA analysis of gut microbiota on gallbladder polyp
According to the IVW method of univariate MR we identified a total of 9 gut microbial taxa that may be associated with gallbladder polyps (Figure 2). Among them, 1 family was a protective factor for the development of gallbladder polyps, Peptococcaceae (OR=0.49, 95%CI:0.25-0.96, P=0.038). 1 phylum, 2 orders, 2 orders, 1 family, and 2 genera were risk factors for the development of nasal polyps. Lentisphaerae (OR=1.74, 95%CI:1.12-2.70, P=0.013), Lentisphaeria (OR=1.78, 95%CI:1.12-2.82, P=0.014), Erysipelotrichia (OR=2.23, 95%CI:1.07-4.62, P=0.032), Erysipelotrichales (OR=2.23, 95%CI:1.07-4.62, P=0.032), Victivallales (OR=1.78, 95%CI:1.12-2.82, P=0.014), Erysipelotrichaceae (OR=2.23, 95%CI:1.07-4.62, P=0.032), Flavonifractor (OR=2.31, 95%CI:1.03-5.20, P=0.044), unknowngenus.id.959 (OR=1.70, 95%CI:1.15-2.52, P=0.007). No heterogeneity or pleiotropy was observed in any of the taxa. Lentisphaeria and Lentisphaerae belonged to one taxonomic group, while Erysipelotrichia, Erysipelotrichales, and Erysipelotrichaceae belonged to another group. These two groups might have a more significant causal effect. Mr_steiger algorithm showed consistent directionality in the MR analysis. After correcting for FDR, no microbiota reached statistical significance (Figure 5A).
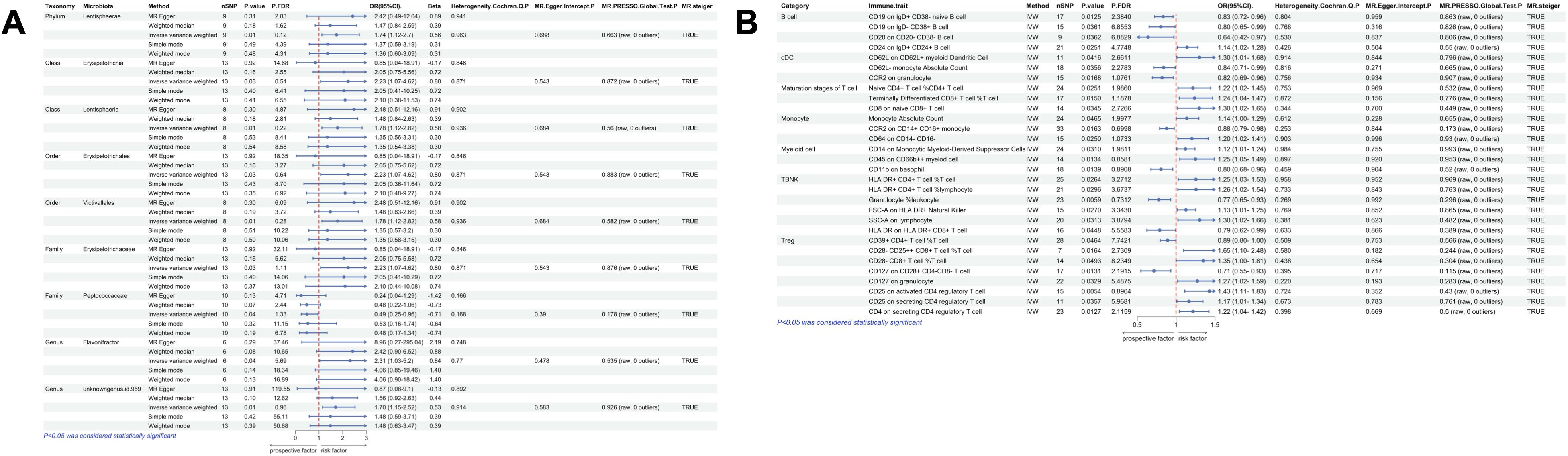
Figure 5. Univariate MR results of gut microbiota and immune traits on gallbladder polyps. (A) MR forest plot of the causal effect of gut microbiota on gallbladder polyps. (B) MR forest plot of the causal effect of immune traits on gallbladder polyps. OR>1 indicates an increased risk, whereas OR<1 indicates a decreased risk. The last 4 columns are analyses of horizontal pleiotropy, heterogeneity, and the directionality of MR, with P > 0.05 indicating no pleiotropy or heterogeneity and TRUE indicating no reverse causality.
To understand the true importance of gut microorganisms causally associated with gallbladder polyps, we performed MR-BMA analysis. 8 microorganisms had MIP values greater than 0.1: unknowngenus.id.959 (MACE=0.311, MIP=0.670, P=0.0078), Lentisphaerae (MACE=0.057, MIP=0.176, P=0.1440), Lentisphaeria (MACE=0.053, MIP=0.168, P=0.1638), Victivallales (MACE=0.053, MIP=0.168, P=0.1638), Erysipelotrichales (MACE=0.037, MIP=0.120, P=0.8759), Erysipelotrichia (MACE=0.037, MIP=0.120, P=0.8759), Erysipelotrichaceae (MACE=0.037, MIP=0.120, P=0.8759), and Peptococcaceae (MACE=-0.142, MIP=0.290, P=0.0847) (Supplementary File S3). Unknowngenus.id.959 had the highest posterior probability of 0.193. See Table 1 for detail.
3.3.2 Univariate MR analysis and MR-BMA analysis of immune traits on gallbladder polyp
Using IVW method of univariate MR, we identified a total of 30 immune traits potentially associated with gallbladder polyps (Figure 4). These included 4 traits of B cell, 3 traits of cDC, 3 traits of Maturation stages of T cell, 3 traits of Monocyte, 3 traits of Myeloid cell, 6 traits of TBNK and 8 traits of Treg. Mr_steiger algorithm indicated that the MR analysis did not show significant bidirectionality. None of the 30 immune traits exhibited heterogeneity or pleiotropy. After correcting the p-values for multiple testing using FDR, no immune traits remained statistically significant (P < 0.05 after FDR adjustment) (Figure 5B).
We also performed MR-BMA analysis on the 30 immune traits to gain further insight into their true significance for gallbladder polyps, as this method accounts for potential causal associations. Consistent with the univariate MR analysis, we categorized the immune traits into 7 classes for the MR-BMA analysis and ranked their importance based on MIP values (Supplementary File S3). The posterior probability models for CD20 on CD20- CD38- B cell (MACE=-0.092, MIP=0.531, P=0.0357), CD28- CD8+ T cell %T cell (MACE=-0.190, MIP=0.454, P=0.0259) reached 0.327, and 0.224, respectively. CD62L- monocyte Absolute Count (MACE=0.188, MIP=0.759, P=0.0366), CD8 on naive CD8+ T cell (MACE=0.105, MIP=0.733, P=0.0357), CD14 on Monocytic Myeloid-Derived Suppressor Cells (MACE=0.156, MIP=0.831, P=0.0278), and SSC-A on lymphocyte (MACE=0.100, MIP=0.536, P=0.0392) with posterior probabilities of 0.592, 0.662 0.727 and 0.456. Table 1. The posterior probability of each of these immune traits exceeds 0.2 and is a crucial player in the development of gallbladder polyps.
3.3.3 Mediated Mendelian randomization analysis
Using univariate MR, we identified 9 gut microbial taxa and 30 immune traits that had a significant causal effect on gallbladder polyps, without evidence of heterogeneity or horizontal pleiotropy. We then performed a two-sample MR analysis to investigate the relationship between these 9 gut microbial taxa and the 30 immune traits (Supplementary File S4). Two causal pathways were identified, through which gut microbiota modulate immune function and consequently influence the development of gallbladder polyps. The mediated proportion of these immune traits was also obtained. One of these causal pathways had a mediating direction (Beta1×Beta2) that was consistent with the direction of the effect of gut microbiota on gallbladder polyps (Beta3). The gut microbe unknowngenus.id.959 acts by inhibiting SSC-A (a marker) on lymphocytes, which is a risk factor for colon polyps. Notably, unknowngenus.id.959 was the most important gut microbe identified in the MR-BMA analysis. Therefore, the mechanism by which unknowngenus.id.959 mediates the level of SSC-A on lymphocytes may be an important protective mechanism against gallbladder polyps Table 2.
3.4 Causal relationships between the gut microbiota, immune traits and colon polyp
3.4.1 Univariate MR analysis and MR-BMA analysis of gut microbiota on colon polyp
Using IVW method of Mendelian randomization, we identified 6 gut microbial taxa potentially associated with colon polyps (Figure 2). Oscillibacter (OR=1.08, 95%CI:1.01-1.17, P=0.032) and RuminococcaceaeUCG002 (OR=1.09, 95%CI:1.01-1.18, P=0.033) were identified as having a potentially positive causative effect, increasing the risk of colon polyps. In contrast, Dialister (OR=0.88, 95%CI:0.78-1.00, P=0.048), Eubacteriumventriosum (OR=0.90, 95%CI:0.82-1.00, P=0.048), Actinobacteria (OR=0.91, 95%CI:0.85-0.98, P=0.016), and Eubacterium brachy (OR=0.94, 95%CI:0.88-0.99, P=0.033) were associated with a reduced risk of colon polyps. Mr_steiger algorithm did not reveal an inverse causal relationship between these 6 taxa and colon polyps. However, none of the results remained statistically significant after correcting for multiple testing using FDR method Figure 6A.
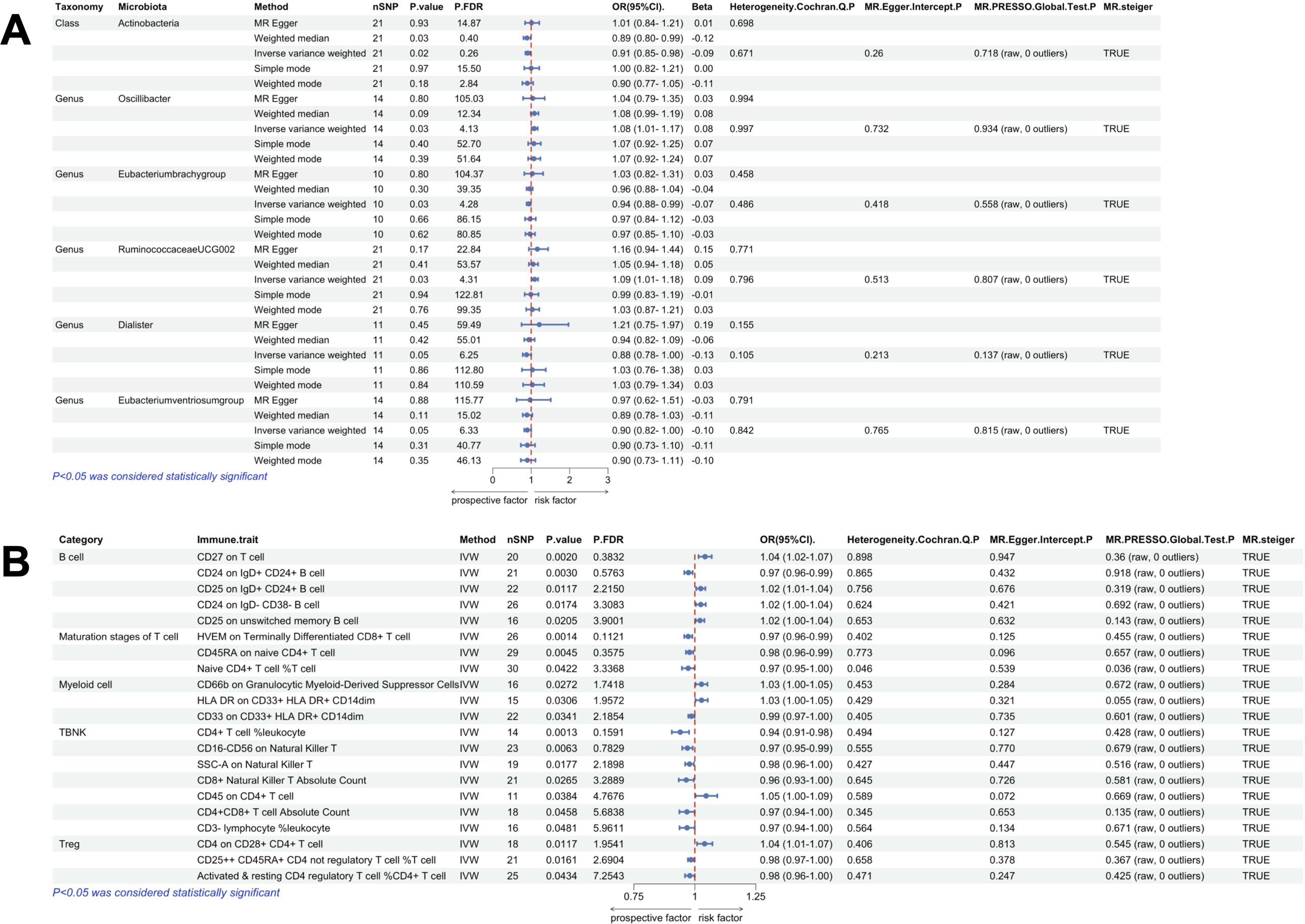
Figure 6. Univariate MR results of gut microbiota and immune traits on colon polyps. (A) MR forest plot of the causal effect of gut microbiota on colon polyps. (B) MR forest plot of the causal effect of immune traits on colon polyps. OR>1 indicates an increased risk, whereas OR<1 indicates a decreased risk. The last 4 columns are analyses of horizontal pleiotropy, heterogeneity, and the directionality of MR, with P > 0.05 indicating no pleiotropy or heterogeneity and TRUE indicating no reverse causality.
MR-BMA analysis of gut microorganisms with significant causative associations for colon polyps provides further insight into their significance. MIP values of 4 microorganisms greater than 0.1: Actinobacteria (MACE=-0.070, MIP=0.599, P=0.0246), Oscillibacter (MACE=0.012, MIP=0.145, P=0.3453), Eubacteriumventriosum (MACE=-0.029, MIP=0.255, P=0.2027), and Dialister (MACE=-0.010, MIP=0.105, P=0.8050) (Supplementary File S3). Actinobacteria had the highest posterior probability of 0.445. See Table 1 for detailed results.
3.4.2 Univariate MR analysis and MR-BMA analysis of immune traits on colon polyp
Using IVW method of univariate MR, we identified a total of 21 immune traits potentially associated with colon polyps (Figure 4). These included 5 traits of B cell, 3 traits of Maturation stages of T cell, 3 traits of Myeloid cell, 7 traits of TBNK and 3 traits of Treg. Mr_steiger algorithm indicated that MR analysis did not show significant bidirectionality. However, the Cochran’s Q test for heterogeneity and the MR-PRESSO test revealed some heterogeneity in the Naive CD4+ T cell %T cell, which affected the causality results. After correcting the p-values for FDR method, no immune trait remained statistically significant (P < 0.05 after FDR adjustment) (Figure 6B).
The 20 immune traits that had a significant causal effect on colon polyps were subjected to MR-BMA analysis to gain insight into their true importance for colon polyps. Consistent with the univariate MR analysis, we categorized these immune traits into 7 classes for the MR-BMA analysis and ranked their importance based on MIP values (Supplementary File S3). The immune traits CD27 on T cell (MACE=0.031, MIP=0.974, P=0.0011) and HLA DR on CD33+ HLA DR+ CD14dim (MACE=0.025, MIP=0.933, P=0.005) had posterior probabilities of 0.962 and 0.919, respectively. The immune trait cd25++ CD45RA+ CD4 not regulatory T cell %T cell (MACE=-0.004, MIP=0.439, P=0.0350) had a posterior probability of 0.436. These immune traits play an important role in the development of colon polyps.
3.4.3 Mediated Mendelian randomization analysis
Using univariate MR, we identified 6 gut microbial taxa and 20 immune traits that had a significant causal effect on colon polyps, without evidence of heterogeneity or horizontal pleiotropy. Two-sample MR analysis was performed to investigate the relationship between these 6 gut microbial taxa and the 20 immune traits (Supplementary File S4). We identified two causal pathways through which gut microbes modulate immune function and consequently influence the development of colon polyps, and obtained the mediated proportion for these immune traits. Among these, the mediating direction (Beta1×Beta2) of three causal pathways was consistent with the direction of the effect of gut microbes on colon polyps (Beta3). Increased abundance of RuminococcaceaeUCG002 can cause increased levels of CD24 on IgD- CD38- B cells, which in turn leads to an increased risk of colon polyps. Conversely, Eubacterium brachy may reduce the risk of colon polyps by decreasing the levels of CD4 on CD28+ CD4+ T cells (Table 2).
3.5 Causal relationships between the gut microbiome, immune traits and gastric polyp
3.5.1 Univariate MR analysis and MR-BMA analysis of gut microbiota on gastric polyp
We identified a total of 12 gut microbial taxa potentially associated with gastric polyps by IVW method Figure 2. There was evidence of horizontal pleiotropy and heterogeneity for the taxa Oscillospira and Proteobacteria. RuminococcaceaeUCG004 (OR=1.12, 95%CI:1.00-1.43, P=0.045), Eggerthella (OR=1.20, 95%CI:1.06-1.35, P=0.003), Terrisporobacter (OR=1.25, 95%CI:1.03-1.53, P=0.023), Escherichia.Shigella (OR=1.26, 95%CI:1.04-1.51, P=0.016), and Actinobacteria (OR=1.30, 95%CI:1.11-1.52, P=0.001) were identified as having a positive causal effect on gastric polyps, potentially increasing the risk. In contrast, Eubacterium oxidoreducens (OR=0.77, 95%CI:0.60-0.98, P=0.034), Gastranaerophilales (OR=0.86, 95%CI:0.74-0.98, P=0.029), unknownfamily.id.1000001214 (OR=0.86, 95%CI:0.74-0.98, P=0.029), unknowngenus.id.1000001215 (OR=0.86, 95%CI:0.74-0.98, P=0.029), and Melainabacteria (OR=0.86, 95%CI:0.76-0.97, P=0.018) were associated with a reduced risk of gastric polyps. Mr_steiger algorithm did not reveal any evidence of reverse causality between 10 microbial taxa and gastric polyps. Actinobacteria remained statistically significant after correcting for multiple testing using FDR method (FDR-adjusted P < 0.05) Figure 7A.
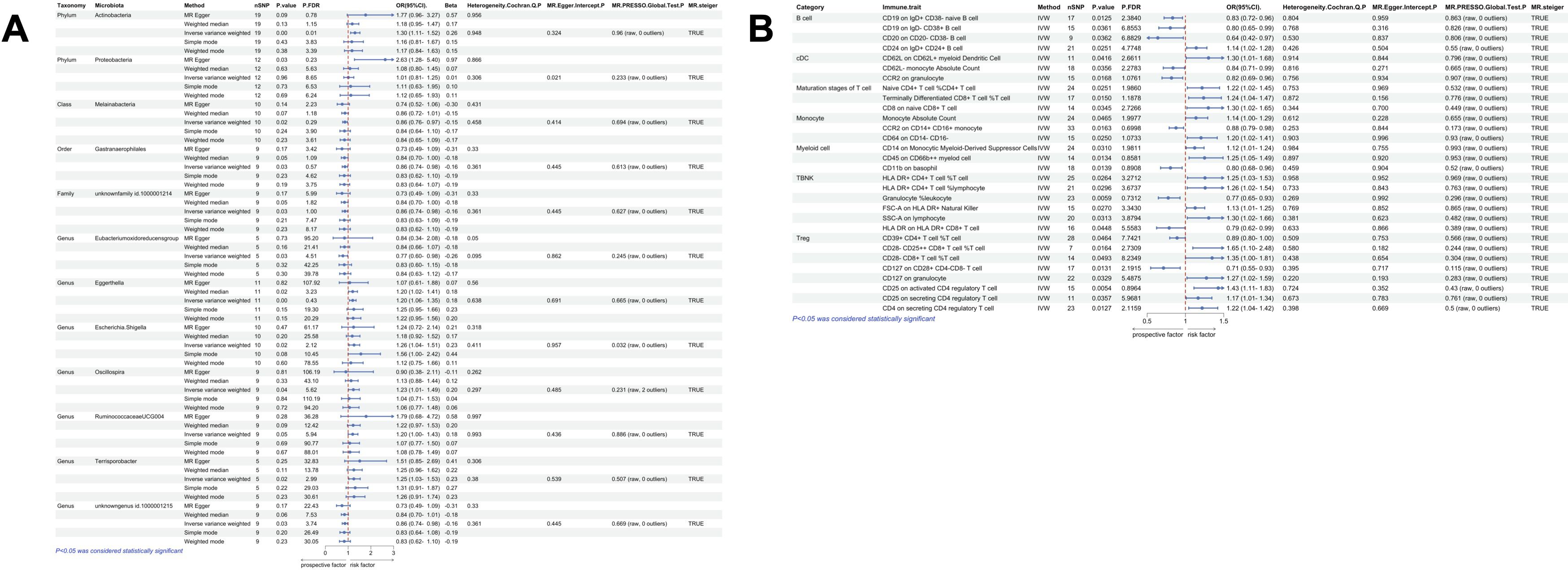
Figure 7. Univariate MR results of gut microbiota and immune traits on gastric polyps. (A) MR forest plot of the causal effect of gut microbiota on gastric polyps. (B) MR forest plot of the causal effect of immune traits on gastric polyps. OR>1 indicates an increased risk, whereas OR<1 indicates a decreased risk. The last 4 columns are analyses of horizontal pleiotropy, heterogeneity, and the directionality of MR, with P > 0.05 indicating no pleiotropy or heterogeneity and TRUE indicating no reverse causality.
We performed MR-BMA analysis on the gut microorganisms that were significantly causally associated with gastric polyps to gain further insight into their significance. 7 microorganisms had MIP values greater than 0.1: Gastranaerophilales (MACE=0.263, MIP=0.935, P=0.0011), Eggerthella (MACE=0.137, MIP=0.538, P=0.0244), Actinobacteria (MACE=-0.025, MIP=0.180, P=0.0729), Melainabacteria (MACE=-0.023, MIP=0.173, P=0.0822), Terrisporobacter (MACE=-0.023, MIP=0.173, P=0.0822), unknowngenus id.1000001215 (MACE=-0.023, MIP=0.173, P=0.0822), and unknownfamily id.1000001214 (MACE=0.022, MIP=0.172, P=0.1138). Supplementary File S3. 7 Gastranaerophilales had the highest posterior probability of 0.201. See Table 1 for detailed results.
3.5.2 Univariate MR analysis and MR-BMA analysis of immune traits on gastric polyp
According to the IVW method of univariate MR we identified a total of 33 immune traits that may has causal relationship with gastric polyps Figure 4. These included 5 traits of B cell, 3 traits of cDC, 4 traits of Maturation stages of T cell, 2 traits of Monocyte, 9 traits of Myeloid cell, 3 traits of TBNK and 7 traits of Treg. Mr_steiger algorithm indicated that the MR analysis did not show significant bidirectionality. Sensitivity analysis did not reveal heterogeneity and horizontal pleiotropy in these 33 immune traits. After correcting the p-values for multiple testing using FDR method, no immune trait remained statistically significant (P < 0.05 after FDR adjustment) Figure 7B.
Consistent with the univariate MR analysis, we categorized these immune traits into 7 classes for the MR-BMA analysis and ranked their importance based on MIP values Supplementary File S3. A total of 6 immune traits had MIP > 0.1 and p-value < 0.05, including Terminally Differentiated CD4-CD8- T cell %CD4-CD8- T cell (MACE=-0.114, MIP=0.998, P=0.0002), Naive CD4-CD8- T cell %T cell (MACE=0.056, MIP=0.950, P=0.0004), CD28- CD8dim T cell %T cell (MACE=0.054, MIP=0.777, P=0.0006), CD33 on CD33+ HLA DR+ (MACE=0.007, MIP=0.176, P=0.0245), CD33 on CD33+ HLA DR+ CD14dim (MACE=0.005, MIP=0.230, P=0.0256), and CD33 on Monocytic Myeloid-Derived Suppressor Cells (MACE=0.004, MIP=0.167, P=0.0338).
3.5.3 Mediated Mendelian randomization analysis
By univariate MR, we found a total of 10 gut microbial taxa as well as 33 immune traits to have significant causal effects on gallbladder polyps without heterogeneity or horizontal pleiotropy. Two-sample MR analysis was performed to investigate the relationship between these 10 gut microbial taxa and the 33 immune traits Supplementary File S4. We identified a total of 14 causal pathways through which gut microbes modulate immune function and consequently influence the development of gastric polyps, and obtained the mediated proportion for these immune traits. Among these, the mediating direction (Beta1×Beta2) of 7 causal pathways was consistent with the direction of the effect of gut microbes on gastric polyps (Beta3). Actinobacteria induced polyp development by inhibiting PDL-1 on CD14+ CD16+ monocytes or increasing the percentage of CD28+ CD45RA+ CD8dim T cells of CD8dim T cells. Eubacterium oxidoreducens induced polyp development by mediating the levels of CD45 on CD66b++ myeloid cells and CD20 on IgD- CD27- B cells, which exert a suppressive effect on polyp development. Gastranaerophilales may reduce the risk of gastric polyps by increasing the level of CD8 on terminally differentiated CD8+ T cells. Additionally, unknownfamily.id.1000001214 and unknowngenus.id.1000001215 may exert their protective effects through CD8 on terminally differentiated CD8+ T cells Table 2. Based on MR-BMA results, Gastranaerophilales, Actinobacteria, unknowngenus id.1000001215, and unknownfamily id.1000001214 are key gut microbes that play an important protective role against gastric polyps.
4 Discussion
In this large-scale, comprehensive MR analysis, we identified 11 microbial taxa with 35 immune traits causally associated with the risk of nasal polyps. 9 microbial taxa with 30 immune traits causally associated with the risk of gallbladder polyps.6 microbial taxa with 20 immune traits causally associated with the risk of colon polyps.10 microbial taxa with 33 immune traits causally associated with the risk of nasal polyps. 10 microbial taxa and 33 immune profiles were causally associated with the risk of nasal polyps. In addition, the MR-BMA results prioritized the importance of these factors. A total of 16 causal pathways were identified, in which the mediating direction (Beta1×Beta2) was consistent with the direction of the effect of gut microbes on polyps (Beta3), as shown in Figure 8. Among them, 6 causal pathways were found to be associated with the mechanism of nasal polyp development, including Eubacterium fissicatena- Effector Memory CD8+ T cell %T cell- Nasal polyp, mediated effect 4.74%; Prevotellaceae- CD45RA on naive CD4 + T cell- Nasal polyp, mediator effect 5.05%; Prevotellaceae- CD39 on CD39+ activated CD4 regulatory T cell- Nasal polyp, mediator effect 5.13%; Deltaproteobacteria- Terminally Differentiated CD8+ T cell Absolute Count- Nasal polyp, mediating effect 8.17%; Methanobrevibacter- CCR2 on CD62L+ myeloid Dendritic Cell- Nasal polyp with a mediator effect of 8.33%; Methanobrevibacter- CD3 on CD4 regulatory T cell- Nasal polyp with a mediator effect of 15.88%. 1 causal pathway was found to be associated with the mechanism of gallbladder polypogenesis was unknowngenus.id.959- SSC-A on lymphocyte- Gallbladder polyp with a mediating effect of 7.02%. 2 causal pathways were found to be associated with the mechanism of colon polyp development including RuminococcaceaeUCG002- CD24 on IgD- CD38- B cell- Colon polyp with a mediating effect of 6.20%; Eubacterium brachy- CD4 on CD28+ CD4+ T cell- Colon polyp, with a mediating effect of 12.11%. 7 causal pathways were found to be associated with the mechanism of gastric polyp development, including Actinobacteria- CD28+ CD45RA+ CD8dim T cell %CD8dim T cell- Gastric polyp with a mediating effect of 3.24%; Actinobacteria- PDL-1 on CD14+ CD16+ monocyte- Gastric polyp with a mediating effect of 4.59%; Eubacterium oxidoreducens- CD45 on CD66b++ myeloid cell- Gastric polyp, mediating effect 6.70%; Eubacterium oxidoreducens- CD20 on IgD- CD27- B cell- Gastric polyp, mediating effect 12.40%; Gastranaerophilales, unknownfamily.id.1000001214, and unknowngenus.id.1000001215 all of which inhibited gastric polyp development via CD8 on Terminally Differentiated CD8+ T cell, mediating effect 9.46%.
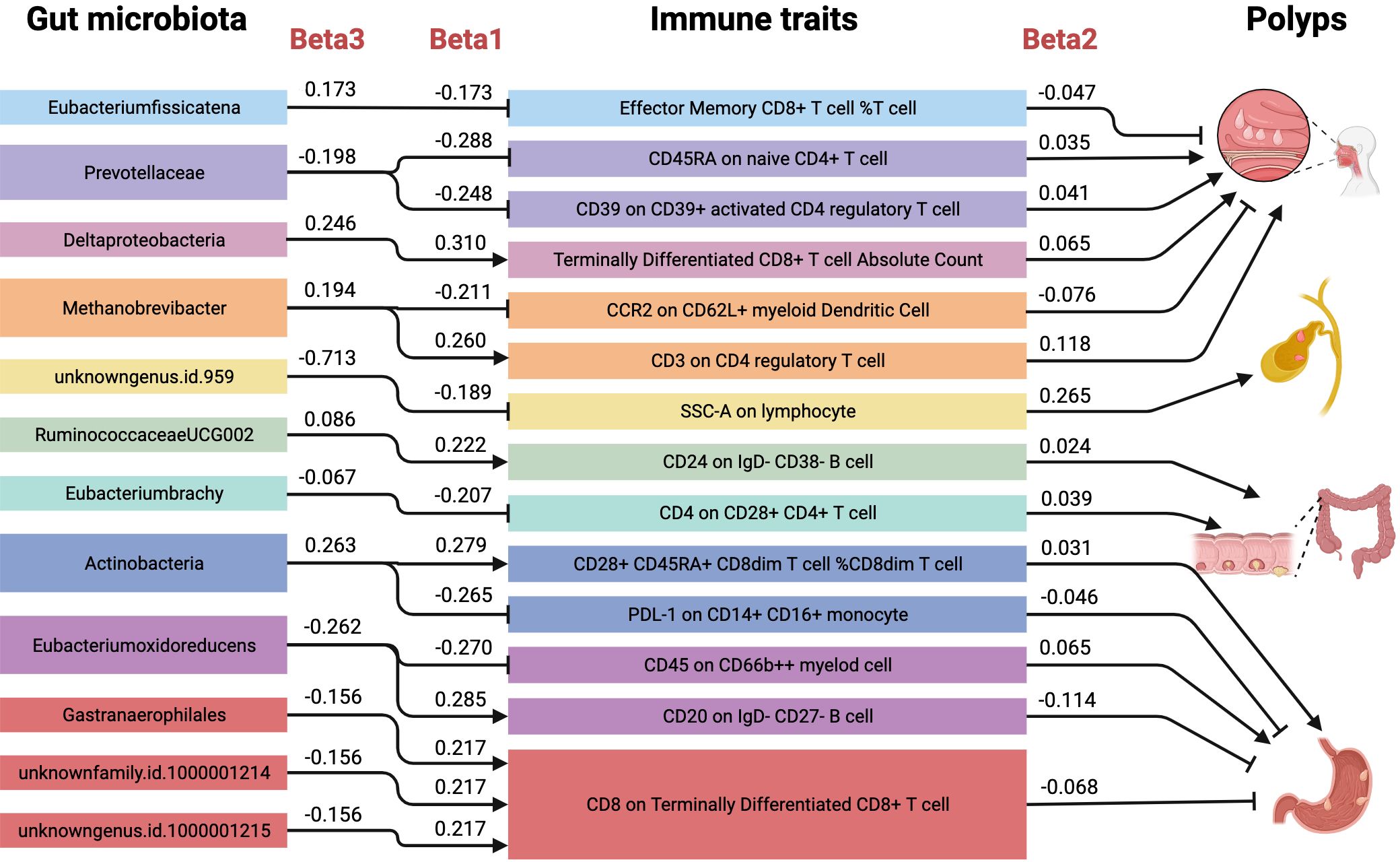
Figure 8. Mediating role of immune traits in the causal effect of gut microbiota on polyps. Causal effects of gut microbiota on polyps (Beta3), gut microbiota on immune traits (Beta1), and immune traits on polyps (Beta2). There were a total of 16 causal pathways where the mediating direction (Beta1*Beta2) was consistent with the direction of gut microbiota-polyps (Beta3).
With the advancement of human gut microbial sequencing technology (29), the gut microbial community, a complex and dynamic ecosystem, has emerged as a forefront and a key area of scientific inquiry, underscoring its pivotal role in human health and disease progression. Initially, research concentrated on exploring the potential relationship between gut microbes and digestive diseases, seeking to elucidate the microecosystem’s regulatory mechanisms in digestive physiology and pathological conditions (30–32). As research evolved, the gut microbiota’s broader immunomodulatory role became evident, particularly during immune-mediated and disease-induced immune injury phases, characterized by marked alterations in gut microbiota composition and function (33, 34). Paulos et al. (35) demonstrated that gut microbes regulate CD8+ T-cell function via the Toll-like receptor 4 signaling pathway, reinforcing this regulatory mechanism. This tumor-specific immune responses offered fresh insights into tumor immunotherapy. Subsequent studies (36, 37) have extended these findings, underscoring the gut microbiota’s pivotal role in modulating adaptive immune responses, particularly by modulating the function and activity of Th1 and Th17 cell subsets, as well as myeloid-derived cells, which collectively shape the immune response landscape within the tumor microenvironment.
In 1958, American surgeon Eiseman et al. (38) pioneered the use of healthy human fecal enemas to treat four patients with severe postoperative pseudomembranous enterocolitis, introducing gut microbiota regulation as a potential therapeutic approach. The gut microbiota is highly plastic, with its composition influenced by factors such as diet, prebiotics, probiotics, and antibiotics. Recent studies have revealed a strong link between gut microbiota composition and the efficacy of immune checkpoint inhibitors (ICIs). Oral administration of Bifidobacterium bifidum promotes dendritic cell maturation and CD8+ T-cell accumulation in tumors, enhancing the efficacy of PD-L1 blockade therapy (39). This observation was further corroborated, wherein transplantation of fecal microbiota from ICI-responsive patients to germ-free mice reduced tumor burden and improved therapeutic outcomes, while fecal microbiota transplantation (FMT) from non-responders showed no such effects (40, 41). Furthermore, microbial metabolism-derived circulating molecules, particularly secondary bile acids and other metabolites, have emerged as key regulators of various immune processes in humans (42). The intricate interplay between the gut microbiota and bile acids represents a pivotal axis in the modulation of immune responses and the pathogenesis of gastrointestinal diseases. The gut microbiota, through its metabolic activities, transforms bile acids, endowing them with the capacity to act as signaling molecules that engage with specific receptors, such as the farnesoid X receptor (FXR) and the G protein-coupled bile acid receptor 1 (TGR5) (43, 44). These receptors, in turn, are implicated in the regulation of inflammation, with activated FXR known to attenuate the nuclear factor kappa-B (NF-κB) signaling pathway, a key mediator of inflammatory processes (45).
Recent advances in engineered microbiomes and FMT have shown promise in modulating the immune environment and improving therapeutic outcomes, opening new avenues for personalized and precision medicine (46, 47). The modulation of the gut microbiota and bile acid profiles has emerged as a promising therapeutic strategy for gastrointestinal disorders, underscoring the significance of elucidating the complex dynamics within the bile acid–microbiota axis (48).
Previous studies (49, 50) have shown that Eubacterium fissicatena, a microorganism closely linked to inflammatory conditions, is strongly associated with obesity-related metabolic disorders. Its abundance level positively correlates with the severity of non-alcoholic fatty liver disease. Additionally, Misiak et al. (51) found that Eubacterium fissicatena may be involved in activating immunoinflammatory processes, such as altering intestinal barrier permeability, interfering with lipid metabolism, and disrupting glucose homeostasis. Therefore, Eubacterium fissicatena is considered a potential disease-associated bacterium that may play an unfavorable role in the development of various diseases (52). In our study, Eubacterium fissicatena was the key bacterial group identified by MR-BMA analysis as affecting the proportion of Effector Memory CD8+ T cells among total T cells, thereby inducing the development of nasal polyps.
Prevotellaceae are dominant commensal gut microbiota that play a crucial role in regulating host immunity and metabolism by producing short-chain fatty acids (e.g., butyrate) (53, 54). Ruiz-Saavedra et al. (55) found that the abundance of Prevotellaceae was significantly higher in healthy controls than in patients with intestinal polyps and adenomas. Notably, the abundance was even lower in patients with adenomas compared to those with intestinal polyps. This finding suggests that Prevotellaceae play a crucial role in maintaining intestinal homeostasis and preventing intestinal diseases. Our study suggests that Prevotellaceae protect against nasal polyps, potentially by reducing their risk through inhibiting CD39 levels on CD39+ activated CD4 regulatory T cells and CD45RA on naive CD4+ T cells.
Deltaproteobacteria, an important microbial taxon, play a key role in the material cycle of nature, with their metabolic activities being important for the transformation of elements such as carbon, sulfur, and heavy metals (56, 57). However, in humans, Deltaproteobacteria are considered pathogenic microorganisms that colonize both the oral cavity and the intestinal tract, and are associated with the development of periodontitis and other systemic diseases (58, 59). Terminally differentiated CD8+ T cells comprise a wide range of cell types, with cytotoxic T lymphocytes (CTLs) being the predominant and most typical type (60). Studies (61) have shown that the nasal cavity is a unique immune site where CTLs play an important role in the local immune defense mechanism and in the pathogenesis of atypical allergic rhinitis (NARES) and chronic nasal mucosal inflammation (62). It is noteworthy that there is a large CTL infiltration in the nasal mucosal tissues of NARES patients, and the cytokines and cytotoxic molecules secreted by CTLs can induce mucosal damage, thereby triggering or exacerbating the symptoms of rhinitis. Our study found that both Deltaproteobacteria and terminally differentiated CD8+ T cells are risk factors for the development of nasal polyps, and that Deltaproteobacteria and terminally differentiated CD8+ T cells are also causally associated with each other.
Previous studies (63) have found a significant association between the presence of Methanobrevibacter, a methanogenic bacteria taxon, and the incidence of colon polyps, with a higher percentage of these strains detected in patients with colon polyps. In recent years, methanogenic bacteria, besides being intestinal residents, have been found to potentially act as pathogens in diseased tissues, such as muscle and brain abscesses (64, 65). Notably, Sogodogo et al. (66) showed that Methanobrevibacter may be involved in the pathogenesis of refractory sinusitis by persistently colonizing the nasal cavity of patients and triggering a chronic inflammatory response. Ali et al. (67) integrated multi-omics data (peripheral blood, feces, and liver tissues), found a reduced abundance of Methanobrevibacter in patients with hepatitis C compared to healthy controls, which was negatively correlated with disease severity. However, a systematic review (68) suggested that Methanobrevibacter abundance is lower in the gut of obese individuals. Methanobrevibacter may play a beneficial role in the intestine by participating in physiological processes such as metabolism and immune regulation, but it may become a potential pathogen and cause disease upon colonizing extraintestinal tissues. This phenomenon highlights the importance of the ecological location of microbial communities in determining their roles. Our study suggests that Methanobrevibacter may contribute to an increased risk of nasal polyps by affecting the levels of CCR2 on CD62L+ myeloid dendritic cells and CD3 on CD4 regulatory T cells.
As one of the dominant gut genera, Ruminococcaceae plays a crucial role in maintaining the diversity and homeostasis of the gut microbial community. However, the abundance of this genus was found to be significantly reduced in the gut of patients with dark spot polyp syndrome, colorectal cancer, and its precancerous lesions (69, 70). Some members of the Ruminococcaceae are capable of producing bile acid-stimulating metabolites and are therefore often regarded as beneficial strains (71). However, it is noteworthy that Ruminococcaceae is a large taxon containing numerous species, and therefore, individual roles may vary significantly. Jiang et al. (72) showed that RuminococcaceaeUCG002 may be a harmful strain, with its elevated abundance being associated with a high risk of chronic insomnia and cardio-metabolic diseases. Radjabzadeh et al. (73) further found that RuminococcaceaeUCG002 was significantly associated with depressive symptoms through double-cohort validation and may play a key role in the pathogenesis of depression. A systematic review suggested that Eubacterium brachy may be a beneficial oral strain, with its higher abundance being associated with a lower risk of periodontal disease (74). Rezasoltani et al. (75) found that salivary Eubacterium brachy may serve as a novel biomarker for colorectal cancer screening. In our study, we found that RuminococcaceaeUCG002 is a risk factor for colon polyps, with its mechanism of action potentially related to the expression of CD24 on IgD-CD38- B cell subsets. Additionally, Eubacterium brachy was a protective factor against colon polyps, with its mechanism of action being related to CD4 on CD28+ CD4+ T cells.
Actinobacteria are usually considered beneficial gut microbial taxa and play an important role in colorectal tumorigenesis (76). This is consistent with our results that Actinobacteria are beneficial microbiota in colon polyps, which showed the highest posterior probability of 0.445 based on MR-BMA. However, for the development of gastric polyps, Actinobacteria may be present as pathogens. Alarcón et al. (77) suggest that Actinobacteria may be involved in H. pylori-associated intestinal metaplasia and gastric carcinogenesis. The reason for this is the interesting finding from previous studies that H. pylori-associated gastritis led to a reduction in the diversity of gastric microorganisms, but when gastric microorganisms were present only in H. pylori infection, it instead significantly prolonged the emergence of gastric malignant tumors (78). This result was verified by the study of Lertpiriyapong et al. (79), in which even a limited variety of intestinal microbiota could contribute to the H. pylori-induced gastric tumorigenesis. This reveals a potential mechanism for the role of gut microbiota in gastric carcinogenesis. We found that Actinobacteria could promote gastric polypogenesis by inhibiting the protective effect of PD-L1 on CD14+CD16+ monocytes. It can also induce gastric polypogenesis by elevating the ratio of CD28+ CD45RA+ CD8dim T cells to CD8dim T cells.
We identified CD45 on CD66b++ myeloid cells as a risk factor for gastric polyps, a class of myeloid-derived cells that highly express CD66b and CD45. Mao et al. (80) found that myeloid-derived suppressor cells (MDSCs) with the CD45+CD33lowCD11bdim phenotype depleted L-arginine in the microenvironment through the IL-6/IL-8-arginase I axis and inhibited tumor cell killing by CD8+ T cells, and it is noteworthy that this type of MDSCs exhibits concomitant high expression of CD66b. Similar results were obtained by later studies, in which higher numbers of CD66b+ neutrophils infiltrating within the tumor tissues of patients with gastric adenocarcinoma and esophageal adenocarcinoma were associated with poorer disease-free survival and overall survival, and this association of high infiltration with poor prognosis was more pronounced in female patients. In vitro experiments showed that CD66b+ neutrophils could promote the migration and invasive ability of gastric cancer cell lines by secreting inducible epithelial-mesenchymal transition-associated factors (e.g., TGF-β and IL-6). Additionally, we identified CD20 on IgD-CD27- B cells as a protective factor against gastric polyps. CD20 is a cell surface protein predominantly expressed on B cells and is widely used as a marker for identifying and characterizing B cells. IgD-CD27- B cells, also known as double-negative B cells, are a subpopulation of poorly differentiated or naïve B cells characterized by the lack of antigenic experience and memory cell markers. Previous studies (81) have shown that high levels of CD20+ B-cell follicles are highly correlated with the prognosis of gastric cancer, and that the level of tumor-infiltrating CD20+ IgD-CD27- B cells is positively correlated with the overall and disease-free survival of patients with gastric cancer, who have a better survival prognosis when exhibiting high levels of CD20+ IgD-CD27- B-cell infiltration.
Huang et al. (82) constructed a model mouse for chronic atrophic gastritis and analyzed it by high-throughput sequencing, finding that the abundance of Gastranaerophilales was significantly reduced in the intestines of the model mouse with chronic atrophic gastritis. By monitoring the dynamics of the gut microbiota in mice, Pessoa et al. (83) found that the abundance of Gastranaerophilales was decreased in the intestines of mice fed a high-fat diet. In most studies (84, 85), Gastranaerophilales have been recognized as a group of beneficial bacteria with important roles in lipid metabolism in vivo and immunity. Terminally differentiated CD8+ T cells are characterized by high cell surface expression of CD8. These cells usually undergo multiple rounds of cytokinesis and differentiate into effector cells capable of rapidly producing cytotoxic molecules, such as perforin and granzymes, to destroy target cells (86). During the early stages of H. pylori infection, CD8+ T cells of the tissue-resident memory phenotype are specific for CagA, the virulence factor of H. pylori, and are capable of recognizing the CagA antigen and producing the cytokine IFN-γ, which accelerates H. pylori clearance and attenuates gastric mucosal damage and inflammatory responses induced by CagA (87). In our study, both Gastranaerophilales and CD8 on terminally differentiated CD8+ T cells were protective factors against gastric polyps, and Gastranaerophilales also achieved this protective effect through CD8 on terminally differentiated CD8+ T cells, realizing this causal pathway.
This investigation presents several strengths. Mendelian randomization analyses were employed to elucidate potential causal relationships between gut microbiota, immune traits, and polyp development, while mitigating the influence of confounding factors. The genetic instruments for gut microbiota were derived from the largest genome-wide association study (GWAS) meta-analysis to date, enhancing the robustness of our MR analytical approach. The inclusion of 731 immune traits ensured comprehensive coverage of the immunophenotypes. To address potential violations of MR assumptions, horizontal pleiotropy and heterogeneity was assessed using MR-PRESSO method and MR-Egger regression intercept test. A two-sample MR design was implemented, utilizing non-overlapping exposure and outcome datasets at the aggregate level to minimize bias.
Notwithstanding these strengths, several limitations warrant consideration. Primarily, the study population was restricted to individuals of European ancestry, necessitating cautious extrapolation of findings to other ethnic groups due to potential genetic heterogeneity. Secondly, constraints in the available datasets precluded exploration of causal relationships between gut microbiota and polyps at the species level. Lastly, the utilization of summary statistics rather than individual-level data precluded subgroup analyses, such as differentiation between hyperplastic polyps and adenomas. These limitations underscore the need for future studies incorporating diverse populations, species-level microbial data, and individual-level phenotypic information to further elucidate the complex interplay between gut microbiota, immune function, and polyp formation.
5 Conclusion
Mendelian randomization analysis revealed new evidence for the pathogenesis of polyps, highlighting the crucial roles of gut microbiota and immune status in polyp development. We emphasize the key role that specific gut microbiota and immune traits play in influencing the risk of different types of polyps, including nasal polyps, gallbladder polyps, colon polyps, and gastric polyps. The identification of causal pathways through which gut microbes modulate immune function and thus influence polyp development contributes to a better understanding of the underlying mechanisms. MR-BMA approach prioritizes the importance of specific factors. These findings have potential implications for polyp prevention strategies, early screening efforts, and the reduction of polyp risk through the development of effective interventions targeting the identified etiologies. Overall, we emphasize the importance of the findings in elucidating the complex interactions between gut microbiota, immune traits, and polyp development, as well as the potential translational application of these insights in clinical practice and disease prevention.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by National Research Ethics Service. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants’ legal guardians/next of kin in accordance with the national legislation and institutional requirements.
Author contributions
CZ: Visualization, Validation, Supervision, Software, Resources, Project administration, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization, Writing – review & editing, Writing – original draft. XY: Writing – review & editing. ZL: Writing – original draft, Data curation. TL: Writing – original draft, Data curation. SL: Writing – review & editing, Software. JY: Writing – original draft, Validation. JW: Writing – original draft, Conceptualization. PY: Writing – original draft, Conceptualization. JR: Writing – review & editing, Writing – original draft. WZ: Writing – review & editing, Supervision.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This study has been supported by the National Natural Science Foundation of China (No.82030120), the Special Research Project on Traditional Chinese Medicine in Henan Province (No.2022JDZX006), National Famous TCM Practitioner Inheritance Workshop Construction Project (No: National TCM Office Human Education Letter (2022) No. 245).
Acknowledgments
The author would like to thank Chenlu Zhao, Dongfang Shang and Qing Zhao for reviewing this article and providing detailed suggestions.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2024.1431990/full#supplementary-material
Supplementary File S1 | Detailed information on instrumental variables for gut microbiota.
Supplementary File S2 | Detailed information on instrumental variables for immune traits.
Supplementary File S3 | MR-BMA analysis of gut Microbiota and immune traits.
Supplementary File S4 | Mediated MR analysis of gut Microbiota and immune traits.
Abbreviations
MR, Mendelian randomization; MR-BMA, MR approach based on Bayesian model averaging; IVW, Inverse variance weighted; FDR, False discovery rate; SNP, Single nucleotide polymorphism; IV, Instrumental variable; MVMR, Multivariate Mendelian randomization; MIP, Marginal inclusion probability; PP, posterior probability; MACE, Model-averaged causal estimate; CTL, Cytotoxic T lymphocyte; NARES, Atypical allergic rhinitis; ICI, Immune checkpoint inhibitor; FMT, Fecal microbiota transplantation; MDSC, Myeloid-derived suppressor cell.
References
1. Waldum H, Fossmark R. Gastritis, gastric polyps and gastric cancer. Int J Mol Sci. (2021) 22. doi: 10.3390/ijms22126548
2. Sung JJY, Chiu HM, Lieberman D, Kuipers EJ, Rutter MD, Macrae F, et al. Third Asia-Pacific consensus recommendations on colorectal cancer screening and postpolypectomy surveillance. Gut. (2022) 71:2152–66. doi: 10.1136/gutjnl-2022-327377
3. Tse BCY, Welham Z, Engel AF, Molloy MP. Genomic, microbial and immunological microenvironment of colorectal polyps. Cancers (Basel). (2021) 13:3382. doi: 10.3390/cancers13143382
4. Zhong X, Wang Y, Xu J, Cao H, Zhang F, Wang X. Gut microbiota signatures in tissues of the colorectal polyp and normal colorectal mucosa, and faeces. Front Cell Infect Microbiol. (2022) 12:1054808. doi: 10.3389/fcimb.2022.1054808
5. Chung Y, Ryu Y, An BC, Yoon YS, Choi O, Kim TY, et al. A synthetic probiotic engineered for colorectal cancer therapy modulates gut microbiota. Microbiome. (2021) 9:122. doi: 10.1186/s40168-021-01071-4
6. Nam SY, Lee SW, Jeon SW, Kwon YH, Lee HS. Helicobacter pylori eradication regressed gastric hyperplastic polyp: A randomized controlled trial. Dig Dis Sci. (2020) 65:3652–9. doi: 10.1007/s10620-020-06065-0
7. Nam SY, Park BJ, Ryu KH, Nam JH. Effect of Helicobacter pylori infection and its eradication on the fate of gastric polyps. Eur J Gastroenterol Hepatol. (2016) 28:449–54. doi: 10.1097/meg.0000000000000553
8. Hu H, Shao W, Liu Q, Liu N, Wang Q, Xu J, et al. Gut microbiota promotes cholesterol gallstone formation by modulating bile acid composition and biliary cholesterol secretion. Nat Commun. (2022) 13:252. doi: 10.1038/s41467-021-27758-8
9. Opazo MC, Ortega-Rocha EM, Coronado-Arrázola I, Bonifaz LC, Boudin H, Neunlist M, et al. Intestinal microbiota influences non-intestinal related autoimmune diseases. Front Microbiol. (2018) 9:432. doi: 10.3389/fmicb.2018.00432
10. Varesi A, Campagnoli LIM, Fahmideh F, Pierella E, Romeo M, Ricevuti G, et al. The interplay between gut microbiota and parkinson’s disease: implications on diagnosis and treatment. Int J Mol Sci. (2022) 23:12289. doi: 10.3390/ijms232012289
11. Michalik M, Podbielska-Kubera A, Basińska AM, Szewc M, Gałęcka M, Schwiertz A. Alteration of indicator gut microbiota in patients with chronic sinusitis. Immun Inflammation Dis. (2023) 11:e996. doi: 10.1002/iid3.996
12. Ma J, Tibbitt CA, Georén SK, Christian M, Murrell B, Cardell LO, et al. Single-cell analysis pinpoints distinct populations of cytotoxic CD4(+) T cells and an IL-10(+)CD109(+) T(H)2 cell population in nasal polyps. Sci Immunol. (2021) 6:6356. doi: 10.1126/sciimmunol.abg6356
13. Gevaert P, Han JK, Smith SG, Sousa AR, Howarth PH, Yancey SW, et al. The roles of eosinophils and interleukin-5 in the pathophysiology of chronic rhinosinusitis with nasal polyps. Int Forum Allergy Rhinol. (2022) 12:1413–23. doi: 10.1002/alr.22994
14. Burgess S, Timpson NJ, Ebrahim S, Davey Smith G. Mendelian randomization: where are we now and where are we going? Int J Epidemiol. (2015) 44:379–88. doi: 10.1093/ije/dyv108
15. Burgess S, Butterworth A, Malarstig A, Thompson SG. Use of Mendelian randomisation to assess potential benefit of clinical intervention. Bmj. (2012) 345:e7325. doi: 10.1136/bmj.e7325
16. Kurilshikov A, Medina-Gomez C, Bacigalupe R, Radjabzadeh D, Wang J, Demirkan A, et al. Large-scale association analyses identify host factors influencing human gut microbiome composition. Nat Genet. (2021) 53:156–65. doi: 10.1038/s41588-020-00763-1
17. Orrù V, Steri M, Sidore C, Marongiu M, Serra V, Olla S, et al. Complex genetic signatures in immune cells underlie autoimmunity and inform therapy. Nat Genet. (2020) 52:1036–45. doi: 10.1038/s41588-020-0684-4
18. Kurki MI, Karjalainen J, Palta P, Sipilä TP, Kristiansson K, Donner KM, et al. FinnGen provides genetic insights from a well-phenotyped isolated population. Nature. (2023) 613:508–18. doi: 10.1038/s41586-022-05473-8
19. Sakaue S, Kanai M, Tanigawa Y, Karjalainen J, Kurki M, Koshiba S, et al. A cross-population atlas of genetic associations for 220 human phenotypes. Nat Genet. (2021) 53:1415–24. doi: 10.1038/s41588-021-00931-x
20. Sanna S, van Zuydam NR, Mahajan A, Kurilshikov A, Vich Vila A, Võsa U, et al. Causal relationships among the gut microbiome, short-chain fatty acids and metabolic diseases. Nat Genet. (2019) 51:600–5. doi: 10.1038/s41588-019-0350-x
21. Zhou C, Wei J, Yu P, Yang J, Liu T, Jia R, et al. Convergent application of traditional Chinese medicine and gut microbiota in ameliorate of cirrhosis: a data mining and Mendelian randomization study. Front Cell Infect Microbiol. (2023) 13:1273031. doi: 10.3389/fcimb.2023.1273031
22. Pierce BL, Ahsan H, Vanderweele TJ. Power and instrument strength requirements for Mendelian randomization studies using multiple genetic variants. Int J Epidemiol. (2011) 40:740–52. doi: 10.1093/ije/dyq151
23. Hemani G, Zheng J, Elsworth B, Wade KH, Haberland V, Baird D, et al. The MR-Base platform supports systematic causal inference across the human phenome. Elife. (2018) 7:34408. doi: 10.7554/eLife.34408
24. Burgess S, Butterworth A, Thompson SG. Mendelian randomization analysis with multiple genetic variants using summarized data. Genet Epidemiol. (2013) 37:658–65. doi: 10.1002/gepi.21758
25. Bowden J, Davey Smith G, Haycock PC, Burgess S. Consistent estimation in mendelian randomization with some invalid instruments using a weighted median estimator. Genet Epidemiol. (2016) 40:304–14. doi: 10.1002/gepi.21965
26. Burgess S, Small DS, Thompson SG. A review of instrumental variable estimators for Mendelian randomization. Stat Methods Med Res. (2017) 26:2333–55. doi: 10.1177/0962280215597579
27. Burgess S, Thompson SG. Interpreting findings from Mendelian randomization using the MR-Egger method. Eur J Epidemiol. (2017) 32:377–89. doi: 10.1007/s10654-017-0255-x
28. Verbanck M, Chen CY, Neale B, Do R. Detection of widespread horizontal pleiotropy in causal relationships inferred from Mendelian randomization between complex traits and diseases. Nat Genet. (2018) 50:693–8. doi: 10.1038/s41588-018-0099-7
29. Wilson KH, Blitchington RB. Human colonic biota studied by ribosomal DNA sequence analysis. Appl Environ Microbiol. (1996) 62:2273–8. doi: 10.1128/aem.62.7.2273-2278.1996
30. Weingarden AR, Vaughn BP. Intestinal microbiota, fecal microbiota transplantation, and inflammatory bowel disease. Gut Microbes. (2017) 8:238–52. doi: 10.1080/19490976.2017.1290757
31. Aron-Wisnewsky J, Vigliotti C, Witjes J, Le P, Holleboom AG, Verheij J, et al. Gut microbiota and human NAFLD: disentangling microbial signatures from metabolic disorders. Nat Rev Gastroenterol Hepatol. (2020) 17:279–97. doi: 10.1038/s41575-020-0269-9
32. Pittayanon R, Lau JT, Leontiadis GI, Tse F, Yuan Y, Surette M, et al. Differences in gut microbiota in patients with vs without inflammatory bowel diseases: A systematic review. Gastroenterology. (2020) 158:930–946.e931. doi: 10.1053/j.gastro.2019.11.294
33. Matson V, Chervin CS, Gajewski TF. Cancer and the microbiome-influence of the commensal microbiota on cancer, immune responses, and immunotherapy. Gastroenterology. (2021) 160:600–13. doi: 10.1053/j.gastro.2020.11.041
34. Tomofuji Y, Maeda Y, Oguro-Igashira E, Kishikawa T, Yamamoto K, Sonehara K, et al. Metagenome-wide association study revealed disease-specific landscape of the gut microbiome of systemic lupus erythematosus in Japanese. Ann Rheum Dis. (2021) 80:1575–83. doi: 10.1136/annrheumdis-2021-220687
35. Paulos CM, Wrzesinski C, Kaiser A, Hinrichs CS, Chieppa M, Cassard L, et al. Microbial translocation augments the function of adoptively transferred self/tumor-specific CD8+ T cells via TLR4 signaling. J Clin Invest. (2007) 117:2197–204. doi: 10.1172/jci32205
36. Iida N, Dzutsev A, Stewart CA, Smith L, Bouladoux N, Weingarten RA, et al. Commensal bacteria control cancer response to therapy by modulating the tumor microenvironment. Science. (2013) 342:967–70. doi: 10.1126/science.1240527
37. Viaud S, Saccheri F, Mignot G, Yamazaki T, Daillère R, Hannani D, et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science. (2013) 342:971–6. doi: 10.1126/science.1240537
38. Eiseman B, Silen W, Bascom GS, Kauvar AJ. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery. (1958) 44:854–9.
39. Sivan A, Corrales L, Hubert N, Williams JB, Aquino-Michaels K, Earley ZM, et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science. (2015) 350:1084–9. doi: 10.1126/science.aac4255
40. Gopalakrishnan V, Spencer CN, Nezi L, Reuben A, Andrews MC, Karpinets TV, et al. Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science. (2018) 359:97–103. doi: 10.1126/science.aan4236
41. Routy B, Le Chatelier E, Derosa L, Duong CPM, Alou MT, Daillère R, et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. (2018) 359:91–7. doi: 10.1126/science.aan3706
42. Yang M, Gu Y, Li L, Liu T, Song X, Sun Y, et al. Bile acid-gut microbiota axis in inflammatory bowel disease: from bench to bedside. Nutrients. (2021) 13:3143. doi: 10.3390/nu13093143
43. Ananthanarayanan M, Balasubramanian N, Makishima M, Mangelsdorf DJ, Suchy FJ. Human bile salt export pump promoter is transactivated by the farnesoid X receptor/bile acid receptor. J Biol Chem. (2001) 276:28857–65. doi: 10.1074/jbc.M011610200
44. Chiang JYL, Ferrell JM. Discovery of farnesoid X receptor and its role in bile acid metabolism. Mol Cell Endocrinol. (2022) 548:111618. doi: 10.1016/j.mce.2022.111618
45. Jia W, Xie G, Jia W. Bile acid-microbiota crosstalk in gastrointestinal inflammation and carcinogenesis. Nat Rev Gastroenterol Hepatol. (2018) 15:111–28. doi: 10.1038/nrgastro.2017.119
46. Canale FP, Basso C, Antonini G, Perotti M, Li N, Sokolovska A, et al. Metabolic modulation of tumours with engineered bacteria for immunotherapy. Nature. (2021) 598:662–6. doi: 10.1038/s41586-021-04003-2
47. Yang R, Chen Z, Cai J. Fecal microbiota transplantation: Emerging applications in autoimmune diseases. J Autoimmun. (2023) 141:103038. doi: 10.1016/j.jaut.2023.103038
48. Wei M, Huang F, Zhao L, Zhang Y, Yang W, Wang S, et al. A dysregulated bile acid-gut microbiota axis contributes to obesity susceptibility. EBioMedicine. (2020) 55:102766. doi: 10.1016/j.ebiom.2020.102766
49. Song Y, Shen H, Liu T, Pan B, De Alwis S, Zhang W, et al. Effects of three different mannans on obesity and gut microbiota in high-fat diet-fed C57BL/6J mice. Food Funct. (2021) 12:4606–20. doi: 10.1039/d0fo03331f
50. Zhuge A, Li S, Lou P, Wu W, Wang K, Yuan Y, et al. Longitudinal 16S rRNA Sequencing Reveals Relationships among Alterations of Gut Microbiota and Nonalcoholic Fatty Liver Disease Progression in Mice. Microbiol Spectr. (2022) 10:e0004722. doi: 10.1128/spectrum.00047-22
51. Misiak B, Pawlak E, Rembacz K, Kotas M, Żebrowska-Różańska P, Kujawa D, et al. Associations of gut microbiota alterations with clinical, metabolic, and immune-inflammatory characteristics of chronic schizophrenia. J Psychiatr Res. (2024) 171:152–60. doi: 10.1016/j.jpsychires.2024.01.036
52. Liu Y, Huang W, Ji S, Wang J, Luo J, Lu B. Sophora japonica flowers and their main phytochemical, rutin, regulate chemically induced murine colitis in association with targeting the NF-κB signaling pathway and gut microbiota. Food Chem. (2022) 393:133395. doi: 10.1016/j.foodchem.2022.133395
53. Cignarella F, Cantoni C, Ghezzi L, Salter A, Dorsett Y, Chen L, et al. Intermittent fasting confers protection in CNS autoimmunity by altering the gut microbiota. Cell Metab. (2018) 27:1222–1235.e1226. doi: 10.1016/j.cmet.2018.05.006
54. Chen Y, Liu Y, Wang Y, Chen X, Wang C, Chen X, et al. Prevotellaceae produces butyrate to alleviate PD-1/PD-L1 inhibitor-related cardiotoxicity via PPARα-CYP4X1 axis in colonic macrophages. J Exp Clin Cancer Res. (2022) 41:1. doi: 10.1186/s13046-021-02201-4
55. Ruiz-Saavedra S, Arboleya S, Nogacka AM, González Del Rey C, Suárez A, Diaz Y, et al. Commensal fecal microbiota profiles associated with initial stages of intestinal mucosa damage: A pilot study. Cancers (Basel). (2023) 16:104. doi: 10.3390/cancers16010104
56. Jameson E, Stephenson J, Jones H, Millard A, Kaster AK, Purdy KJ, et al. Deltaproteobacteria (Pelobacter) and Methanococcoides are responsible for choline-dependent methanogenesis in a coastal saltmarsh sediment. Isme J. (2019) 13:277–89. doi: 10.1038/s41396-018-0269-8
57. Langwig MV, De Anda V, Dombrowski N, Seitz KW, Rambo IM, Greening C, et al. Large-scale protein level comparison of Deltaproteobacteria reveals cohesive metabolic groups. Isme J. (2022) 16:307–20. doi: 10.1038/s41396-021-01057-y
58. Campbell AG, Campbell JH, Schwientek P, Woyke T, Sczyrba A, Allman S, et al. Multiple single-cell genomes provide insight into functions of uncultured Deltaproteobacteria in the human oral cavity. PloS One. (2013) 8:e59361. doi: 10.1371/journal.pone.0059361
59. Leite GGS, Weitsman S, Parodi G, Celly S, Sedighi R, Sanchez M, et al. Mapping the segmental microbiomes in the human small bowel in comparison with stool: A REIMAGINE study. Dig Dis Sci. (2020) 65:2595–604. doi: 10.1007/s10620-020-06173-x
60. Barry M, Bleackley RC. Cytotoxic T lymphocytes: all roads lead to death. Nat Rev Immunol. (2002) 2:401–9. doi: 10.1038/nri819
61. Qiu S, Du Y, Duan X, Geng X, Xie J, Gao H, et al. Cytotoxic T lymphocytes mediate chronic inflammation of the nasal mucosa of patients with atypical allergic rhinitis. N Am J Med Sci. (2011) 3:378–83. doi: 10.4297/najms.2011.3378
62. Porzia A, Cavaliere C, Begvarfaj E, Masieri S, Mainiero F. Human nasal immune system: a special site for immune response establishment. J Biol Regul Homeost Agents. (2018) 32:3–8.
63. Weaver GA, Krause JA, Miller TL, Wolin MJ. Incidence of methanogenic bacteria in a sigmoidoscopy population: an association of methanogenic bacteria and diverticulosis. Gut. (1986) 27:698–704. doi: 10.1136/gut.27.6.698
64. Nkamga VD, Lotte R, Roger PM, Drancourt M, Ruimy R. Methanobrevibacter smithii and Bacteroides thetaiotaomicron cultivated from a chronic paravertebral muscle abscess. Clin Microbiol Infect. (2016) 22:1008–9. doi: 10.1016/j.cmi.2016.09.007
65. Drancourt M, Nkamga VD, Lakhe NA, Régis JM, Dufour H, Fournier PE, et al. Evidence of archaeal methanogens in brain abscess. Clin Infect Dis. (2017) 65:1–5. doi: 10.1093/cid/cix286
66. Sogodogo E, Fellag M, Loukil A, Nkamga VD, Michel J, Dessi P, et al. Nine cases of methanogenic archaea in refractory sinusitis, an emerging clinical entity. Front Public Health. (2019) 7:38. doi: 10.3389/fpubh.2019.00038
67. Ali RO, Quinn GM, Umarova R, Haddad JA, Zhang GY, Townsend EC, et al. Longitudinal multi-omics analyses of the gut-liver axis reveals metabolic dysregulation in hepatitis C infection and cirrhosis. Nat Microbiol. (2023) 8:12–27. doi: 10.1038/s41564-022-01273-y
68. Michels N, Zouiouich S, Vanderbauwhede B, Vanacker J, Indave Ruiz BI, Huybrechts I. Human microbiome and metabolic health: An overview of systematic reviews. Obes Rev. (2022) 23:e13409. doi: 10.1111/obr.13409
69. Peters BA, Dominianni C, Shapiro JA, Church TR, Wu J, Miller G, et al. The gut microbiota in conventional and serrated precursors of colorectal cancer. Microbiome. (2016) 4:69. doi: 10.1186/s40168-016-0218-6
70. Wang S, Huang G, Wang JX, Tian L, Zuo XL, Li YQ, et al. Altered gut microbiota in patients with peutz-jeghers syndrome. Front Microbiol. (2022) 13:881508. doi: 10.3389/fmicb.2022.881508
71. Sinha SR, Haileselassie Y, Nguyen LP, Tropini C, Wang M, Becker LS, et al. Dysbiosis-induced secondary bile acid deficiency promotes intestinal inflammation. Cell Host Microbe. (2020) 27:659–670.e655. doi: 10.1016/j.chom.2020.01.021
72. Jiang Z, Zhuo LB, He Y, Fu Y, Shen L, Xu F, et al. The gut microbiota-bile acid axis links the positive association between chronic insomnia and cardiometabolic diseases. Nat Commun. (2022) 13:3002. doi: 10.1038/s41467-022-30712-x
73. Radjabzadeh D, Bosch JA, Uitterlinden AG, Zwinderman AH, Ikram MA, van Meurs JBJ, et al. Gut microbiome-wide association study of depressive symptoms. Nat Commun. (2022) 13:7128. doi: 10.1038/s41467-022-34502-3
74. Antezack A, Etchecopar-Etchart D, La Scola B, Monnet-Corti V. New putative periodontopathogens and periodontal health-associated species: A systematic review and meta-analysis. J Periodontal Res. (2023) 58:893–906. doi: 10.1111/jre.13173
75. Rezasoltani S, Aghdaei HA, Jasemi S, Gazouli M, Dovrolis N, Sadeghi A, et al. Oral microbiota as novel biomarkers for colorectal cancer screening. Cancers (Basel). (2022) 15:192. doi: 10.3390/cancers15010192
76. Feng Q, Liang S, Jia H, Stadlmayr A, Tang L, Lan Z, et al. Gut microbiome development along the colorectal adenoma-carcinoma sequence. Nat Commun. (2015) 6:6528. doi: 10.1038/ncomms7528
77. Alarcón T, Llorca L, Perez-Perez G. Impact of the microbiota and gastric disease development by helicobacter pylori. Curr Top Microbiol Immunol. (2017) 400:253–75. doi: 10.1007/978-3-319-50520-6_11
78. Rolig AS, Cech C, Ahler E, Carter JE, Ottemann KM. The degree of Helicobacter pylori-triggered inflammation is manipulated by preinfection host microbiota. Infect Immun. (2013) 81:1382–9. doi: 10.1128/iai.00044-13
79. Lertpiriyapong K, Whary MT, Muthupalani S, Lofgren JL, Gamazon ER, Feng Y, et al. Gastric colonisation with a restricted commensal microbiota replicates the promotion of neoplastic lesions by diverse intestinal microbiota in the Helicobacter pylori INS-GAS mouse model of gastric carcinogenesis. Gut. (2014) 63:54–63. doi: 10.1136/gutjnl-2013-305178
80. Mao FY, Zhao YL, Lv YP, Teng YS, Kong H, Liu YG, et al. CD45(+)CD33(low)CD11b(dim) myeloid-derived suppressor cells suppress CD8(+) T cell activity via the IL-6/IL-8-arginase I axis in human gastric cancer. Cell Death Dis. (2018) 9:763. doi: 10.1038/s41419-018-0803-7
81. Hennequin A, Derangère V, Boidot R, Apetoh L, Vincent J, Orry D, et al. Tumor infiltration by Tbet+ effector T cells and CD20+ B cells is associated with survival in gastric cancer patients. Oncoimmunology. (2016) 5:e1054598. doi: 10.1080/2162402x.2015.1054598
82. Huang W, Yau Y, Zhu J, Wang Y, Dai Z, Gan H, et al. Effect of electroacupuncture at zusanli (ST36) on intestinal microbiota in rats with chronic atrophic gastritis. Front Genet. (2022) 13:824739. doi: 10.3389/fgene.2022.824739
83. Pessoa J, Belew GD, Barroso C, Egas C, Jones JG. The gut microbiome responds progressively to fat and/or sugar-rich diets and is differentially modified by dietary fat and sugar. Nutrients. (2023) 15:2097. doi: 10.3390/nu15092097
84. Du Y, Li DX, Lu DY, Zhang R, Zheng XX, Xu BJ, et al. Morus alba L. water extract changes gut microbiota and fecal metabolome in mice induced by high-fat and high-sucrose diet plus low-dose streptozotocin. Phytother Res. (2022) 36:1241–57. doi: 10.1002/ptr.7343
85. Liu J, Zong C, Yu X, Ding Y, Chang B, Wang R, et al. Alanyl-glutamine (Ala-gln) ameliorates dextran sulfate sodium (DSS)-induced acute colitis by regulating the gut microbiota, PI3K-Akt/NF-κB/STAT3 signaling, and associated pulmonary injury. ACS Infect Dis. (2023) 9:979–92. doi: 10.1021/acsinfecdis.3c00014
86. Milner JJ, Nguyen H, Omilusik K, Reina-Campos M, Tsai M, Toma C, et al. Delineation of a molecularly distinct terminally differentiated memory CD8 T cell population. Proc Natl Acad Sci U.S.A. (2020) 117:25667–78. doi: 10.1073/pnas.2008571117
Keywords: Mendelian randomization, gut microbiota, immune cell, nasal polyp, gastric polyp, colon polyp
Citation: Zhou C, Ye X, Liu Z, Liu T, Li S, Yang J, Wei J, Yu P, Jia R and Zhao W (2024) Dissecting the causal links between gut microbiome, immune traits and polyp using genetic evidence. Front. Immunol. 15:1431990. doi: 10.3389/fimmu.2024.1431990
Received: 13 May 2024; Accepted: 19 August 2024;
Published: 13 September 2024.
Edited by:
Juan Maza-Solano, University Hospital Virgen Macarena, SpainReviewed by:
Theocharis Konstantinidis, Democritus University of Thrace, GreeceRavi Verma, Jawaharlal Nehru University, India
Copyright © 2024 Zhou, Ye, Liu, Liu, Li, Yang, Wei, Yu, Jia and Zhao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wenxia Zhao, Wmhhby13ZW54aWFAMTYzLmNvbQ==
†These authors have contributed equally to this work
 Cheng Zhou
Cheng Zhou Xiaofeng Ye1†
Xiaofeng Ye1† Zhinuo Liu
Zhinuo Liu Tong Liu
Tong Liu Shanzheng Li
Shanzheng Li Jinqiu Yang
Jinqiu Yang Jingjing Wei
Jingjing Wei Peng Yu
Peng Yu Ran Jia
Ran Jia Wenxia Zhao
Wenxia Zhao